Bioactive Compounds and Antioxidant Capacity of Rosa rugosa Depending on Degree of Ripeness
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Samples Description
2.3. Carotenoids and Vitamin E Determinations
2.3.1. Extraction Procedure
2.3.2. Carotenoids Identification and Quantification
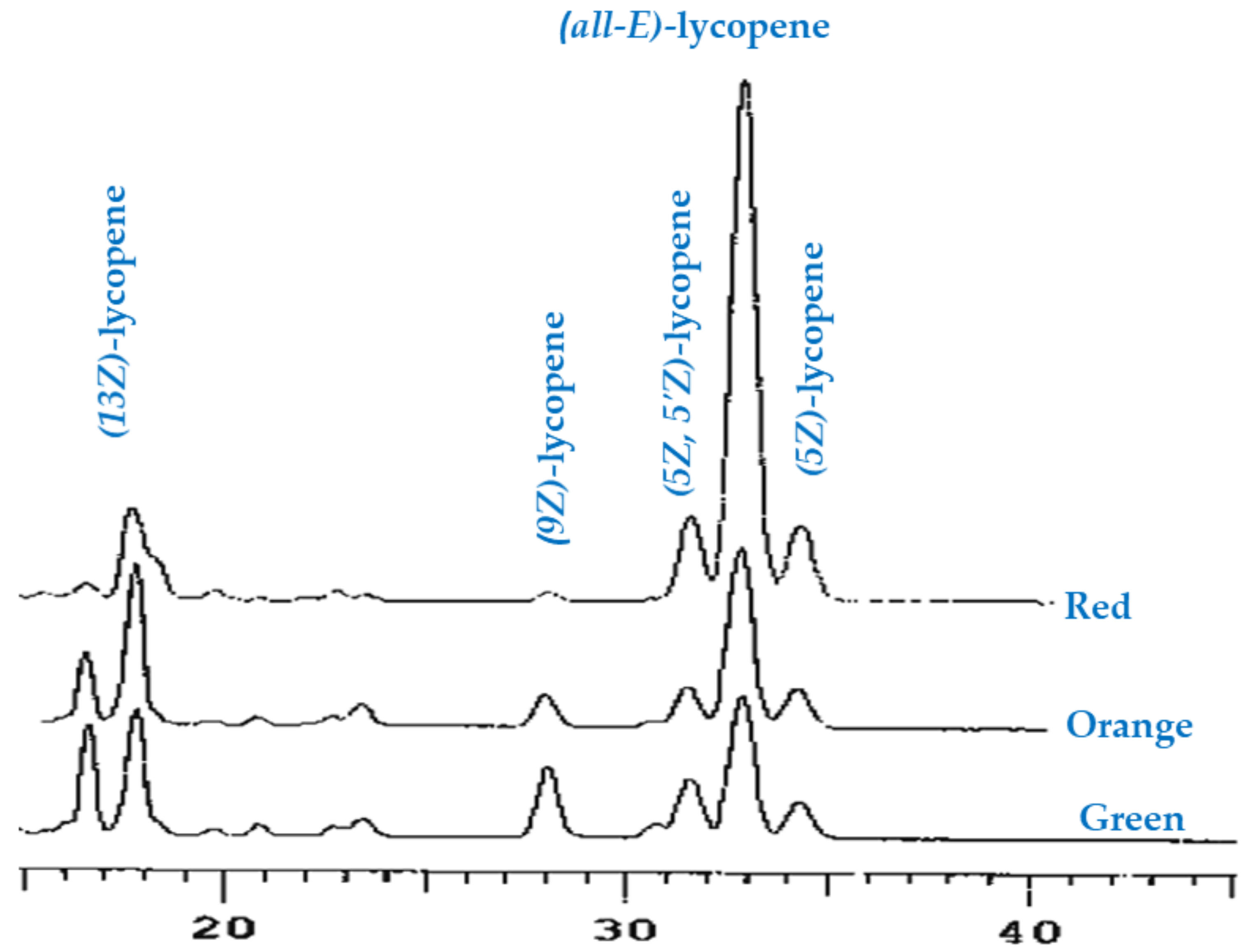
2.3.3. Analysis of Lycopene Composition
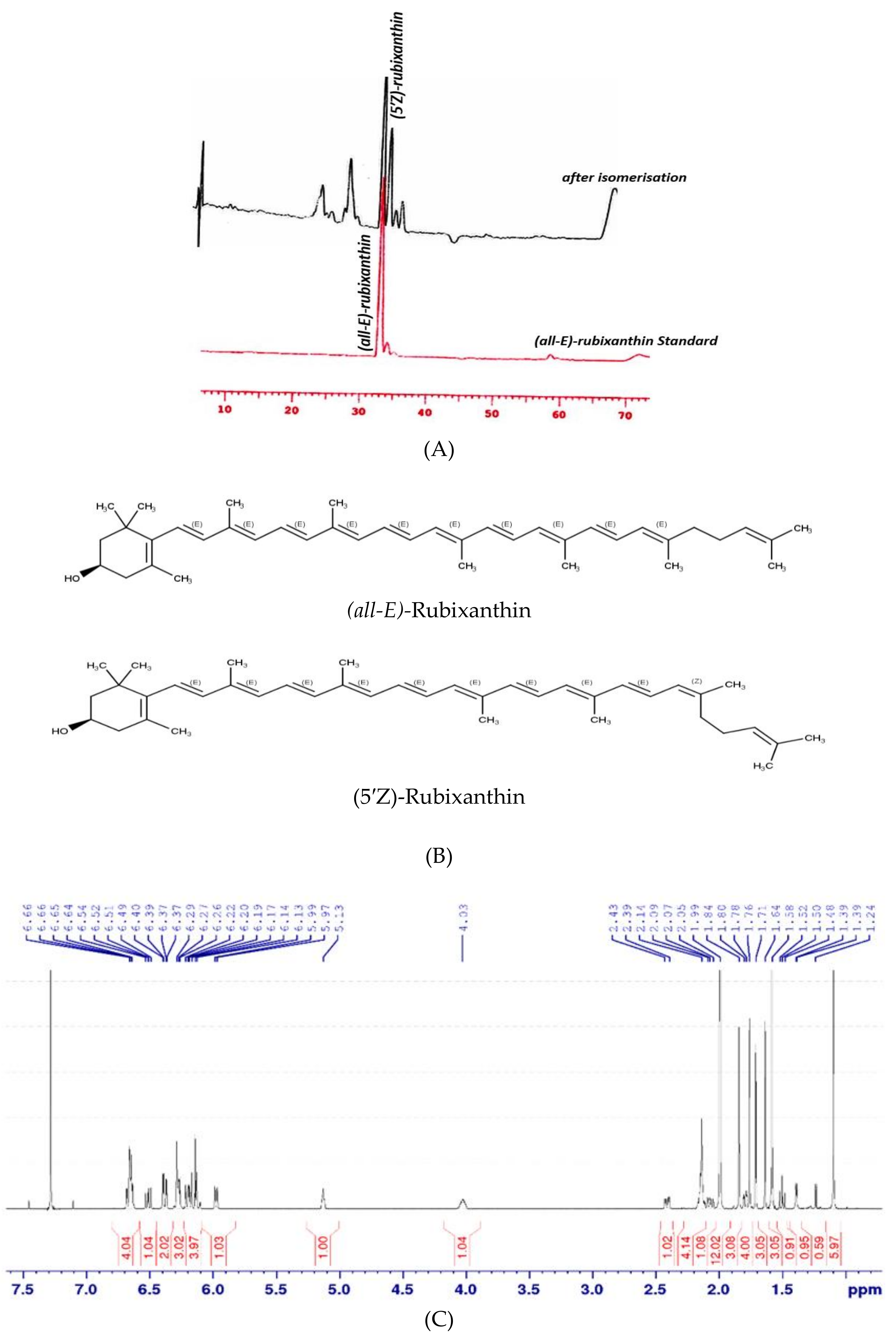
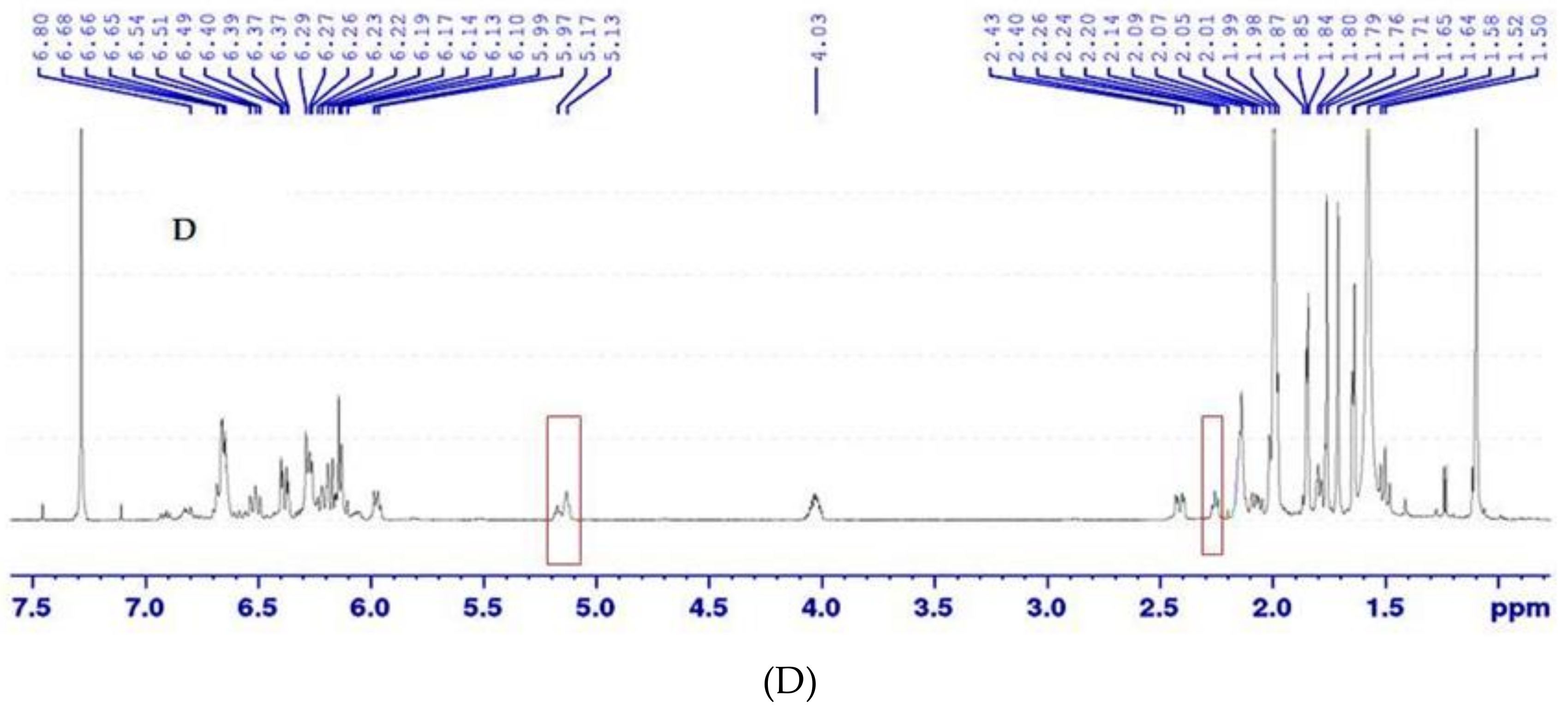
2.3.4. Isomerisation and NMR Analysis
2.3.5. Vitamin E Identification and Quantification
2.4. Vitamin C
2.4.1. Extraction Procedure
2.4.2. Quantification of Vitamin C
2.5. Antioxidants Capacity and Total Phenolic Contents
2.5.1. Extraction Procedures
2.5.2. Determination of Total Phenolic Contents (TP)
2.5.3. Hydrophilic Trolox Equivalent Antioxidant Capacity (H-TEAC) Assay
2.5.4. Hydrophilic Oxygen Radical Absorbance Capacity (H-ORAC) Assay
2.6. Statistical Analysis
3. Results and Discussion
3.1. Carotenoids Identification and Quantification
3.2. Isomerisation and NMR Analysis of (all-E)- and (5′Z)-Rubixanthin
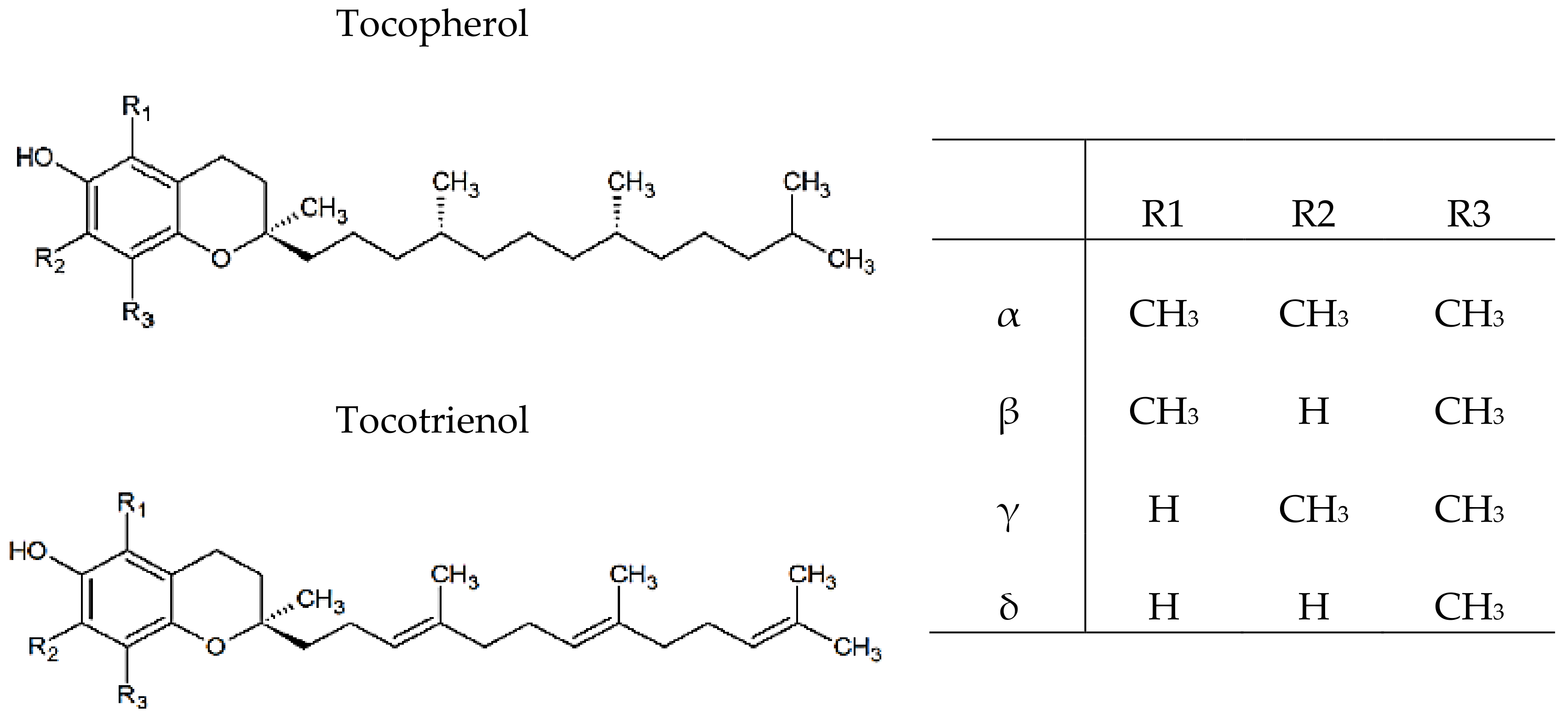
3.3. Vitamin E Identification and Quantification
3.4. Quantification of Vitamin C
3.5. Antioxidant Capacity
3.5.1. Total Phenolic Contents (TP)
3.5.2. Hydrophilic Antioxidant Capacity
3.5.3. Lipophilic Antioxidant Capacity
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Singh, P.; Goyal, G.K. Dietary lycopene: Its properties and anticarcinogenic effects. Compr. Rev. Food Sci. Food Saf. 2008, 7, 255–270. [Google Scholar] [CrossRef]
- Nilsson, O. Rosa. In Flora of Turkey and the East Aegean Islands, 4th ed.; Davis, P.H., Ed.; Edinburgh University Press: Edinburgh, UK, 1997; pp. 106–128. [Google Scholar]
- Al-Yafeai, A.; Malarski, A.; Böhm, V. Characterization of carotenoids and vitamin E in R. rugosa and R. canina: Comparative analysis. Food Chem. 2018, 242, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Andersson, S.C.; Rumpunen, K.; Johansson, E.; Olsson, M.E. Carotenoid content and composition in rose hips (Rosa spp.) during ripening, determination of suitable maturity marker and implications for health promoting food products. Food Chem. 2011, 128, 689–696. [Google Scholar] [CrossRef]
- Chrubasik, C.; Roufogalis, B.D.; Müller-Ladner, U.; Chrubasik, S. A systematic review on the Rosa canina effect and efficacy profiles. Phytother. Res. 2008, 22, 725–733. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Bjork, L.; Trajkovski, V.; Uggla, M. Evaluation of antioxidant activities of rosehip ethanol extracts in different test systems. J. Sci. Food Agric. 2000, 80, 2021–2027. [Google Scholar] [CrossRef]
- Böhm, V.; Fröhlich, K.; Bitsch, R. Rosehip-a “new” source of lycopene. Mol. Aspects. Med. 2003, 24, 385–389. [Google Scholar] [CrossRef]
- Daels-Rakotoarison, D.A.; Gressier, B.; Trotin, F.; Brunet, C.; Luyckx, M.; Dine, T.; Cazin, J.-C. Effects of Rosa canina fruit extract on neutrophil respiratory burst. Phytother. Res. 2002, 16, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.; Xu, L.; Cao, X.; Guo, Y.; Chan, C.-O.; Mok, D.K.W.; Yu, Z.; Chen, S. Anti-inflammatory activities and mechanisms of action of the petroleum ether fraction of Rosa multiflora Thunb. hips. J. Ethnopharmacol. 2011, 138, 717–722. [Google Scholar] [CrossRef] [PubMed]
- Barros, L.; Carvalho, A.M.; Ferreira, I.C.R. Exotic fruits as a source of important phytochemicals: Improving the traditional use of Rosa canina fruits in Portugal. Food Res. Int. 2011, 44, 2233–2236. [Google Scholar] [CrossRef]
- Müller, L.; Goupy, P.; Fröhlich, K.; Dangles, O.; Caris-Veyrat, C.; Böhm, V. Comparative study on antioxidant activity of lycopene (Z)-isomers in different assays. J. Agric. Food Chem. 2011, 59, 4504–4511. [Google Scholar] [CrossRef] [PubMed]
- Arpin, N.; Liaaen -Jensen, S. Carotenoids of higher plants-11 Rubixanthin and Gazaniaxanthin. Phytochemistry 1969, 8, 185–193. [Google Scholar] [CrossRef]
- Kishimoto, S.; Maoka, T.; Sumitomo, K.; Ohmiya, A. Analysis of carotenoid composition in petals of calendula (Calendula officinalis L.). Biosci. Biotechnol. Biochem. 2005, 69, 2122–2128. [Google Scholar] [CrossRef] [PubMed]
- Halgren, T.A. Merck molecular force field. I. Basis, form, scope, parameterization, and performance of MMFF94. J. Comput. Chem. 1996, 17, 490–519. [Google Scholar] [CrossRef]
- Al-Duais, M.; Hohbein, J.; Werner, S.; Böhm, V.; Jetschke, G. Contents of vitamin C, carotenoids, tocopherols, and tocotrienols in the subtropical plant species Cyphostemma digitatum as affected by processing. J. Agric. Food Chem. 2009, 57, 5420–5427. [Google Scholar] [CrossRef] [PubMed]
- Müller, L.; Gnoyke, S.; Popken, A.; Böhm, V. Antioxidant capacity and related parameters of different fruit formulations. LWT—Food Sci. Technol. 2010, 43, 992–999. [Google Scholar] [CrossRef]
- Müller, L.; Theile, K.; Böhm, V. In vitro antioxidant activity of tocopherols and tocotrienols and comparison of vitamin E concentration and lipophilic antioxidant capacity in human plasma. Mol. Nutr. Food Rer. 2010, 54, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Fraser, P.D.; Truesdale, M.R.; Bird, C.R.; Schuch, W.; Bramley, P.M. Carotenoid biosynthesis during tomato fruit development. Plant Physiol. 1994, 105, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Staffan, C.A.; Kimmo, R.; Eva, J.; Marie, E.O. Changes in tocopherols and carotenoids during ripening in rose hips and sea buckthorn berries. J. Plan. Food Medic. 2008, 765, 255–262. [Google Scholar] [CrossRef]
- Thelander, M.; Narita, J.O.; Gruissem, W. Plastid differentiation and pigment biosynthesis during tomato fruit ripening. J. Plant Biochem. Physiol. 1986, 5, 128–141. [Google Scholar]
- Li, L.; Yuna, H. Chromoplast biogenesis and carotenoid accumulation. J. Arch. Biochem. Biophys. 2013, 539, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Kotıkova, Z.; Lachman, J.; Hejtmankova, A.; Hejtmankov, K. Determination of antioxidant activity and antioxidant content in tomato varieties and evaluation of mutual interactions between antioxidants. LWT—Food Sci. Technol. 2011, 44, 1703–1710. [Google Scholar] [CrossRef]
- Cazzaniga, S.; Bressan, M.; Carbonera, D.; Agostini, A.; Dall’Osto, L. Differential roles of carotenes and xanthophylls in photosystem I photoprotection. J. Biochem. 2016, 55, 3636–3649. [Google Scholar] [CrossRef] [PubMed]
- Zhong, L.; Gustavsson, K.E.; Oredsson, S.; Gła, B.; Yilmaz, J.L.; Olsson, M.E. Determination of free and esterified carotenoid composition in rosehip fruit by HPLC DAD-APCI+-MS. J. Food Chem. 2016, 210, 541–550. [Google Scholar] [CrossRef] [PubMed]
- Honda, M.; Takahashi, N.; Kuwa, T.; Takehara, M.; Inoue, Y.; Kumagai, T. Spectral characterisation of Z-isomers of lycopene formed during heat treatment and solvent effects on the E/Z isomerisation. J. Food Chem. 2015, 171, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Huawei, Z.; Xiaowen, W.; Elshareif, O.; Hong, L.; Qingrui, S.; Lianfu, Z. Isomerisation and degradation of lycopene during heat processing in simulated food system. Food Res. Int. J. 2014, 21, 45–50. [Google Scholar]
- Schön, K. Studies on Carotenoids V. Gazaniaxanthin. Biochem. J. 1938, 32, 1566–1570. [Google Scholar] [CrossRef] [PubMed]
- Muné-Bosch, S.; Alegre, L. The function of tocopherols and tocotrienols in plants. CRC Crit. Rev. Plant Sci. 2002, 21, 31–57. [Google Scholar] [CrossRef]
- Andersson, S.; Olsson, M.; Gustavsson, K.; Johanssonc, E.; Rumpunenb, P. Tocopherols in rose hips (Rosa spp.) during ripening. J. Sci. Food Agric. 2012, 92, 2116–2121. [Google Scholar] [CrossRef] [PubMed]
- Zadernowski, R.; Naczk, M.; Amarowicz, R. Tocopherols in Sea Buckthorn (Hippophae rhamnoides L.) Berry Oil. JAOCS 2003, 80, 55–58. [Google Scholar] [CrossRef]
- Groff, J.L.; Gropper, S.S.; Hunt, S.M. The water soluble vitamins. In Advanced Nutrition and Human Metabolism; West Publishing Company: Minneapolis, MN, USA, 1995; pp. 222–231. [Google Scholar]
- Franck, C.; Baetens, M.; Lammertyn, J.; Verboven, P.; Davey, M.W.; Nicolai, B.M. Ascorbic acid concentration in cv. conference pears during fruit development and postharvest storage. J. Agric. Food Chem. 2003, 51, 4757–4763. [Google Scholar] [CrossRef] [PubMed]
- Davey, M.W.; Auwerkerken, A.; Keulemans, J. Relationship of apple vitamin C and antioxidant contents to harvest date and postharvest pathogen infection. J. Sci. Food Agric. 2007, 87, 802–813. [Google Scholar] [CrossRef]
- Uggla, M.; Gao, X.; Werlemark, G. Variation among and within dog rose taxa (Rosa sect. Caninae) in fruit weight, percentages of fruit flesh and dry matter, and vitamin C content. Acta Agric. Scand. B-Soil Plant Sci. 2003, 53, 147–155. [Google Scholar]
- Ercisli, S. Chemical composition of fruits in some rose (Rosa spp.) species. J. Food Chem. 2007, 104, 1379–1384. [Google Scholar] [CrossRef]
- Zhang, J.G.; Luo, H.M.; Huang, Q.J.Y.; Shan, W.C.Y. A comparative study on berry characteristics of large berry cultivars of sea buckthorn. For. Res. Beijing 2006, 18, 643–650. [Google Scholar]
- Rousi, A.; Hannele, A. Ascorbic acid contents in relation to ripeness in fruit of six Hippophae rhamnoides clones form pyharanta, SW. Finland. J. Ann. Agric. Fenn. 1977, 16, 80–87. [Google Scholar]
- Seung, K.L.; Adel, A. Kade Preharvest and postharvest factors influencing vitamin C content of horticultural. Postharvest Biol. Technol. 2000, 20, 207–222. [Google Scholar]
- Prior, R.L.; Hoang, H.; Gu, L.; Wu, X.; Bacchiocca, M.; Howard, L.; Hampsch-Woodill, M.; Huang, D.; Ou, B.; Jacob, R. Assays for Hydrophilic and Lipophilic Antioxidant Capacity (oxygen radical absorbance capacity (ORACFL)) of Plasma and Other Biological and Food Samples. J. Agric. Food Chem. 2003, 51, 3273–3279. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Ou, B.; Prior, R.L. The Chemistry behind Antioxidant Capacity Assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef] [PubMed]
- Balasundram, N.; Sundram, K.; Sammar, S. Phenolic compounds in plants and agro-industrial by-products: Antioxidant Activity. Occurrence and Potential Uses. J. Food Chem. 2006, 99, 191–203. [Google Scholar] [CrossRef]
- Montazeri, N.; Baher, E.; Mirzajani, F.; Barami, Z.; Yousefian, S. Phytochemical contents and biological activities of Rosa canina fruit from Iran. J. Med. Plant Res. 2011, 5, 4584–4589. [Google Scholar]
- Denev, P.; Lojek, A.; Ciz, M.; Kratchanova, M. Antioxidant activity and polyphenol content of Bulgarian fruits. J. Agric. Sci. 2013, 19, 22–27. [Google Scholar]
- Parr, A.J.; Bolwell, G.P. Phenols in the plant and in man. The potential for possible nutritional enhancement of the diet by modifying the phenols content or profile. J. Sci. Food Agric. 2009, 80, 985–1012. [Google Scholar] [CrossRef]
- Ilahy, R.; Hdider, C.; Lenucci, M.S.; Tlili, I.; Dalessandro, G. Antioxidant activity and bioactive compound changes during fruit ripening of high-lycopene tomato cultivars. J. Food Compost. Anal. 2001, 24, 588–595. [Google Scholar] [CrossRef]
- Rodríguez, J.C.; Gómez, D.; Pacetti, D.; Núñez, O.; Gagliardi, R.; Frega, N.; Ojeda, M.; Loizzo, M.; Tundis, R.; Lucci, P. Effects of the fruit ripening stage on antioxidant capacity, total phenolics, and polyphenolic composition of crude palm oil from interspecific hybrid Elaeis oleifera × Elaeis guineensis. J. Agric. Food Chem. 2016, 64, 852–859. [Google Scholar] [CrossRef] [PubMed]
- Young, A.J.; Lowe, G.M. Antioxidant and prooxidant properties of carotenoids. Arch. Biochem. Biophys. 2001, 385, 20–27. [Google Scholar] [CrossRef] [PubMed]
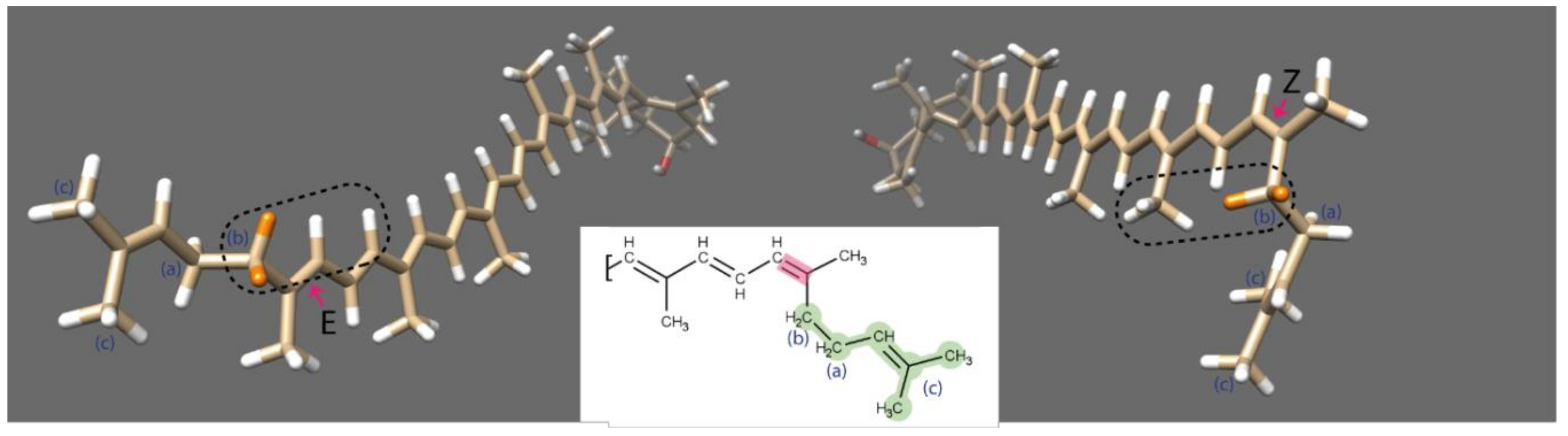
| Carotenoid \ Sample | Green | Orange | Red |
|---|---|---|---|
| (all-E)-β-carotene | 1.8 ± 0.1 a | 7.0 ± 0.4 b | 7.3 ± 0.2 b |
| (9Z)-β-carotene | 0.01± 0.0 a | n.d. | 0.1 ± 0.01 b |
| (13Z)-β-carotene | n.d. | n.d. | 0.3 ± 0.3 |
| (15Z)-β-carotene | n.d. | n.d. | 0.1 ± 0.01 |
| (all-E)-α-cryptoxanthin | 0.2 ± 0.01 | 0.2 ± 0.02 | 0.2 ± 0.01 |
| (all-E)-β-cryptoxanthin | 0.2 ± 0.02 a | 0.4 ± 0.04 b | 1.0 ± 0.03 c |
| (all-E)-lutein | 0.5 ± 0.03 | 0.5 ± 0.0 | 0.5 ± 0.01 |
| (all-E)-lycopene | 1.6 ± 0.2 a | 2.2 ± 0.4 b | 6.0 ± 0.6 c |
| (5Z)-lycopene | 0.4 ± 0.06 a | 0.6 ± 0.08 b | 1.4 ± 0.1 c |
| (9Z)-lycopene | 1.2 ± 0.1 b | 1.4 ± 0.2 b | 0.5 ± 0.05 a |
| (13Z)-lycopene | 4.2 ± 0.6 a | 6.0 ± 0.9 b | 6.5 ± 0.4 b |
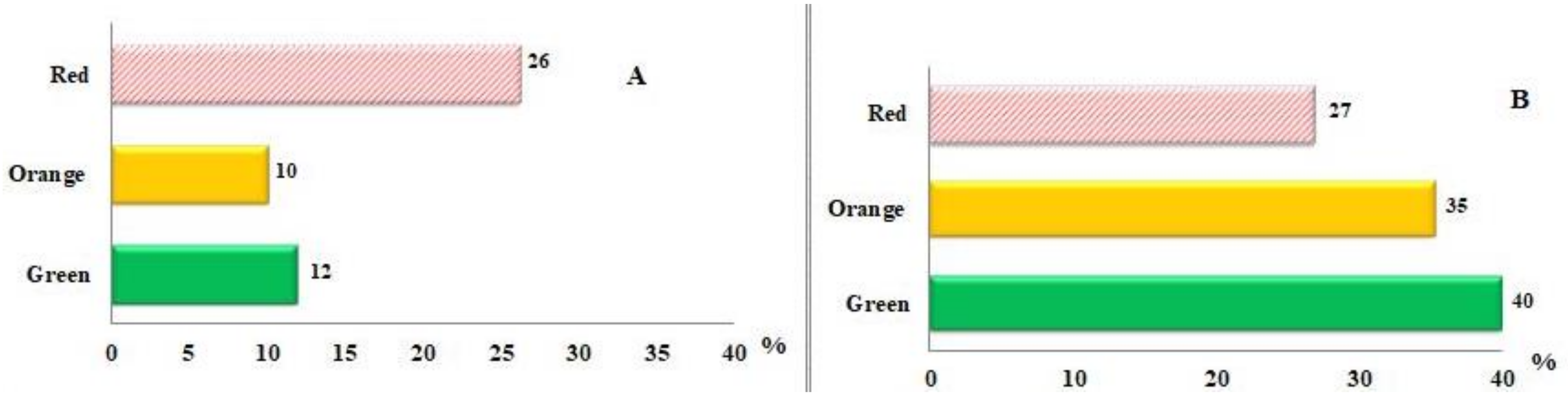
| (all-E)-rubixanthin | 0.9 ± 0.1 a | 2.1 ± 0.2 b | 3.0 ± 0.1 c |
| (5′Z)-rubixanthin | 1.8 ± 0.04 a | n.d. | 3.6 ± 0.3 b |
| (all-E)-violaxanthin | 1.0 ± 0.3 a | 1.2 ± 0.4 b | 0.9 ± 0.4 a |
| (all-E)-zeaxanthin | 0.5 ± 0.1 a | 2.0 ± 0.1 b | 1.7 ± 0.0 b |
| phytoene | 0.8 ± 0.1 c | 0.6 ± 0.03 b | 0.4 ± 0.1 a |
| phytofluene | n.d. | n.d. | n.d. |
| Parameter \ Sample | Green | Orange | Red | |
|---|---|---|---|---|
| α-Tocopherol | µmol/100 g | 15 ± 3 a | 17 ± 2 b | 14 ± 1 a |
| ɣ-Tocopherol | n.d. | n.d. | n.d. | |
| ∑ Tocotrienol | n.d. | n.d. | n.d. | |
| ∑ Vitamin E | 15 ± 3 a | 17 ± 2 b | 14 ± 1 a | |
| Parameter \ Sample | Green | Orange | Red |
|---|---|---|---|
| Ascorbic acid (mg/100 g) | 955 ± 71 b | 1090 ± 51 c | 798 ± 37 a |
| Total phenolics (mg GAE/100 g) | 1097 ± 123 a | 1009 ± 124 a | 1327 ± 43 b |
| H-TEAC (mmol TE/100 g) | 9.0 ± 1.0 a | 9.1 ± 1.0 a | 15 ± 1.0 b |
| H-ORAC (mmol TE/100 g) | 32 ± 4 b | 30 ± 4 b | 23 ± 0.3 a |
| L-TEAC (mmol α-TE/100 g) | 0.8 ± 0.2 a | 1.1 ± 0.1 a | 4.4 ± 0.3 b |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Yafeai, A.; Bellstedt, P.; Böhm, V. Bioactive Compounds and Antioxidant Capacity of Rosa rugosa Depending on Degree of Ripeness. Antioxidants 2018, 7, 134. https://doi.org/10.3390/antiox7100134
Al-Yafeai A, Bellstedt P, Böhm V. Bioactive Compounds and Antioxidant Capacity of Rosa rugosa Depending on Degree of Ripeness. Antioxidants. 2018; 7(10):134. https://doi.org/10.3390/antiox7100134
Chicago/Turabian StyleAl-Yafeai, Ahlam, Peter Bellstedt, and Volker Böhm. 2018. "Bioactive Compounds and Antioxidant Capacity of Rosa rugosa Depending on Degree of Ripeness" Antioxidants 7, no. 10: 134. https://doi.org/10.3390/antiox7100134
APA StyleAl-Yafeai, A., Bellstedt, P., & Böhm, V. (2018). Bioactive Compounds and Antioxidant Capacity of Rosa rugosa Depending on Degree of Ripeness. Antioxidants, 7(10), 134. https://doi.org/10.3390/antiox7100134






