Effect of the Extraction Process on the Biological Activity of Lyophilized Apricot Extracts Recovered from Apricot Pomace
Abstract
:1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Extraction Experiments
2.2.1. Infrared-Assisted Extraction
2.2.2. Heat-Assisted Extraction
2.3. Lyophilization
2.4. Analysis
2.4.1. Dry Matter Content
2.4.2. Quantification of Total Polyphenol Content by Folin-Ciocalteu Method
2.4.3. Determination of the Antiradical Activity
2.4.4. HPLC-DAD Analysis
2.4.5. Determination of the Antibacterial Activity
Bacterial Strains, Culture Media and Growth Conditions
Preparation of the Bacterial Inocula for Minimum Inhibitory Concentration (MIC)
Phenolic Extracts Preparation for MIC Assessment
2.4.6. Statistical Analysis
3. Results and Discussion
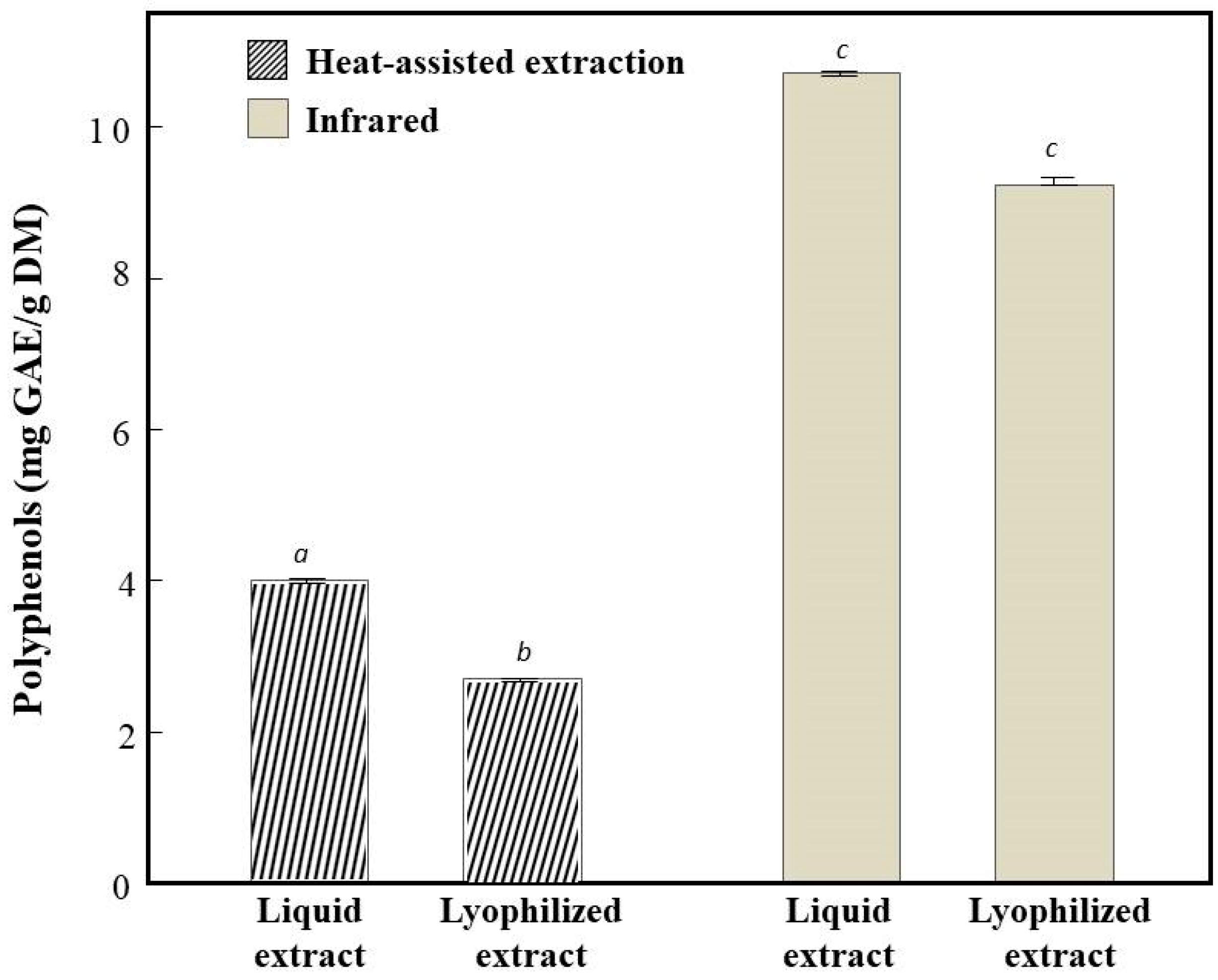
3.1. Total Polyphenols Content
3.2. Biological Activities
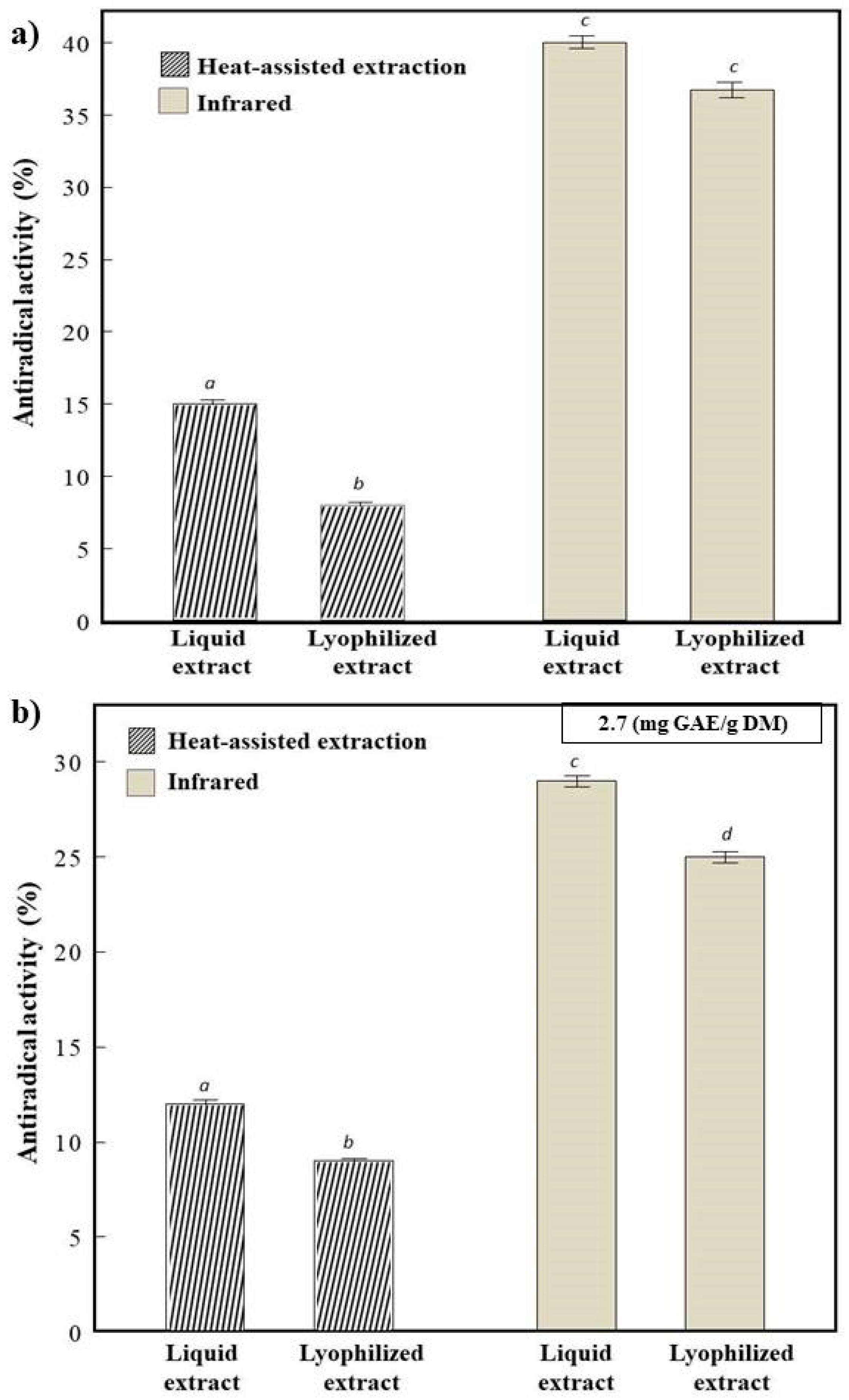
3.2.1. Antiradical Activity (AA)
3.2.2. Antibacterial Activity of the Apricot Pomace Liquid and Lyophilized Extracts
3.3. Quantification of Polyphenol Extracts by High-Performance Liquid Chromatography
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Balasundram, N.; Sundram, K.; Samman, S. Phenolic compounds in plants and agri-industrial by-products: Antioxidant activity, occurrence, and potential uses. Food Chem. 2006, 99, 191–203. [Google Scholar] [CrossRef]
- Rinaldo, D.; Mbéguié-A-Mbéguié, D.; Fils-Lycaon, B. Advances on polyphenols and their metabolism in sub-tropical and tropical fruits. Trends Food Sci. Technol. 2010, 21, 599–606. [Google Scholar] [CrossRef]
- Ames, B.N.; Shigenaga, M.K.; Hagen, T.M. Oxidants, antioxidants, and the degenerative diseases of aging. Proc. Natl. Acad. Sci. USA 1993, 90, 7915–7922. [Google Scholar] [CrossRef] [PubMed]
- Patthamakanokporn, O.; Puwastien, P.; Nitithamyong, A.; Sirichakwal, P.P. Changes of antioxidant activity and total phenolic compounds during storage of selected fruits. J. Food Compos. Anal. 2008, 21, 241–248. [Google Scholar] [CrossRef]
- Rodrigues, A.S.; Pérez-Gregorio, M.R.; García-Falcón, M.S.; Simal-Gándara, J. Effect of curing and cooking on flavonols and anthocyanins in traditional varieties of onion bulbs. Food Res. Int. 2009, 42, 1331–1336. [Google Scholar] [CrossRef]
- Franks, F. Freeze-drying of bioproducts: Putting principles into practice. Eur. J. Pharm. Biopharm. 1998, 45, 221–229. [Google Scholar] [CrossRef]
- Abdelwahed, W.; Degobert, G.; Stainmesse, S.; Fessi, H. Freeze-drying of nanoparticles: Formulation, process and storage considerations. Adv. Drug Deliv. Rev. 2006, 58, 1688–1713. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Gregorio, M.R.; Regueiro, J.; González-Barreiro, C.; Rial-Otero, R.; Simal-Gándara, J. Changes in antioxidant flavonoids during freeze-drying of red onions and subsequent storage. Food Control 2011, 22, 1108–1113. [Google Scholar] [CrossRef]
- Chang, C.H.; Lin, H.Y.; Chang, C.Y.; Liu, Y.C. Comparisons on the antioxidant properties of fresh, freeze-dried and hot-air-dried tomatoes. J. Food Eng. 2006, 77, 478–485. [Google Scholar] [CrossRef]
- Shofian, N.M.; Hamid, A.A.; Osman, A.; Saari, N.; Anwar, F.; Pak Dek, M.S.; Hairuddin, M.R. Effect of freeze-drying on the antioxidant compounds and antioxidant activity of selected tropical fruits. Int. J. Mol. Sci. 2011, 12, 4678–4692. [Google Scholar] [CrossRef] [PubMed]
- Marques, L.G.; Silveira, A.M.; Freire, J.T. Freeze-drying characteristics of tropical fruits. Dry. Technol. 2006, 24, 457–463. [Google Scholar] [CrossRef]
- Cai, Y.Z.; Corke, H. Production and Properties of Spray-dried Amaranthus Betacyanin Pigments. J. Food Sci. 2000, 65, 1248–1252. [Google Scholar] [CrossRef]
- Murcia, M.A.; López-Ayerra, B.; Martinez-Tomé, M.; Vera, A.M.; García-Carmona, F. Evolution of ascorbic acid and peroxidase during industrial processing of broccoli. J. Sci. Food Agric. 2000, 80, 1882–1886. [Google Scholar] [CrossRef]
- Rahman, M.M.; Das, R.; Hoque, M.M.; Zzaman, W. Effect of freeze drying on antioxidant activity and phenolic contents of mango (Mangifera indica). Int. Food Res. J. 2015, 22, 613–617. [Google Scholar]
- Cai, Y.; Yu, Y.; Duan, G.; Li, Y. Study on infrared-assisted extraction coupled with high performance liquid chromatography (HPLC) for determination of catechin, epicatechin, and procyanidin B2 in grape seeds. Food Chem. 2011, 127, 1872–1877. [Google Scholar] [CrossRef]
- Qu, Y.; Li, C.; Zhang, C.; Zeng, R.; Fu, C. Optimization of infrared-assisted extraction of Bletilla striata polysaccharides based on response surface methodology and their antioxidant activities. Carbohydr. Polym. 2016, 148, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Duan, G.; Xie, M.; Chen, B.; Li, Y. Infrared-assisted extraction coupled with high-performance liquid chromatography for simultaneous determination of eight active compounds in Radix Salviae miltiorrhizae. J. Sep. Sci. 2010, 33, 2888–2897. [Google Scholar] [CrossRef] [PubMed]
- Madrau, M.A.; Piscopo, A.; Sanguinetti, A.M.; Del Caro, A.; Poiana, M.; Romeo, F.V.; Piga, A. Effect of drying temperature on polyphenolic content and antioxidant activity of apricots. Eur. Food Res. Technol. 2009, 228, 441–448. [Google Scholar] [CrossRef]
- Slinkard, K.; Singleton, V. Total phenol analysis: Automation and comparison with manual methods. Am. J. Enol. Vitic. 1977, 28, 49–55. [Google Scholar]
- Zhang, D.; Hamauzu, Y. Phenolic compounds, ascorbic acid, carotenoids and antioxidant properties of green, red and yellow bell peppers. J. Food Agric. Environ. 2003, 1, 22–27. [Google Scholar]
- Vizzotto, M.; Cisneros-zevallos, L.; Byrne, D.H. Large Variation Found in the Phytochemical and Antioxidant Activity of Peach and Plum Germplasm. J. Am. Soc. Hortic. Sci. 2007, 132, 334–340. [Google Scholar]
- Georgé, S.; Tourniaire, F.; Gautier, H.; Goupy, P.; Rock, E.; Caris-Veyrat, C. Changes in the contents of carotenoids, phenolic compounds and vitamin C during technical processing and lyophilisation of red and yellow tomatoes. Food Chem. 2011, 124, 1603–1611. [Google Scholar] [CrossRef]
- Liliana, S.-C.; Diana, P.V.-M.; Alfredo, A.A. Structural, physical, functional and nutraceutical changes of freeze-dried fruit. Afr. J. Biotechnol. 2015, 14, 442–450. [Google Scholar] [CrossRef]
- Alfaro, S.; Mutis, A.; Quiroz, A.; Seguel, I.; Scheuermann, E. Effects of Drying Techniques on Murtilla Fruit Polyphenols and Antioxidant Activity. J. Food Res. 2014, 3, 73. [Google Scholar] [CrossRef]
- Deladino, L.; Teixeira, A.S.; Reta, M.; García, A.D.M.; Navarro, A.S.; Martino, M.N. Major phenolics in yerba mate extracts (Ilex paraguariensis) and their contribution to the total antioxidant capacity. Food Nutr. Sci. 2013, 4, 154–162. [Google Scholar] [CrossRef]
- Abascal, K.; Ganora, L.; Yarnell, E. The effect of freeze-drying and its implications for botanical medicine: A review. Phytother. Res. 2005, 19, 655–660. [Google Scholar] [CrossRef] [PubMed]
- Hasmida, M.N.; Nur Syukriah, A.R.; Liza, M.S.; Mohd Azizi, C.Y. Effect of different extraction techniques on total phenolic content and antioxidant activity of quercus infectoria galls. Int. Food Res. J. 2014, 21, 1039–1043. [Google Scholar]
- Delcour, A.H. Outer Membrane Permeability and Antibiotic Resistance. Biochim. Biophys. Acta 2009, 1794, 808–816. [Google Scholar] [CrossRef] [PubMed]
- Russell, A.D. Similarities and differences in the responses of microorganisms to biocides 1. J. Antimicrob. Chemother. 2003, 52, 750–763. [Google Scholar] [CrossRef] [PubMed]
- Veberic, R.; Stampar, F. Selected polyphenols in fruits of different cultivars of genus Prunus. Phyton—Ann. Rei Bot. 2005, 45, 375–383. [Google Scholar]

| Minimum Inhibitory Concentration (µg/mL) | ||||
|---|---|---|---|---|
| Bacterica/POMs | Heat-Assisted Extraction | Infrared | ||
| Liquid Extract | Lyophilized Extract | Liquid Extract | Lyophilized Extract | |
| Methicillin Resistant Staphylococcus aureus (MRSA3) (gram+) | - | - | 6.75 | 13.5 |
| Staphylococcus aureus 1966 (gram+) | 6.75 | 13.5 | 3.3 | 6.75 |
| Coagulase-negative Staphylococci 1530 (gram+) | - | - | 3.3 | 6.75 |
| Staphylococcus epidermidis 2080 (gram+) | 6.75 | 13.5 | 3.3 | 6.75 |
| Klebsiella 118 metallo beta lactamase + (gram−) | - | - | - | - |
| Klebsiella (gram−) | - | - | - | - |
| E. coli ESBL 2238 (gram−) | - | - | - | - |
| Escherichia coli (1250) (gram−) | - | - | 6.75 | 13.5 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheaib, D.; El Darra, N.; Rajha, H.N.; El-Ghazzawi, I.; Maroun, R.G.; Louka, N. Effect of the Extraction Process on the Biological Activity of Lyophilized Apricot Extracts Recovered from Apricot Pomace. Antioxidants 2018, 7, 11. https://doi.org/10.3390/antiox7010011
Cheaib D, El Darra N, Rajha HN, El-Ghazzawi I, Maroun RG, Louka N. Effect of the Extraction Process on the Biological Activity of Lyophilized Apricot Extracts Recovered from Apricot Pomace. Antioxidants. 2018; 7(1):11. https://doi.org/10.3390/antiox7010011
Chicago/Turabian StyleCheaib, Dina, Nada El Darra, Hiba N. Rajha, Iman El-Ghazzawi, Richard G. Maroun, and Nicolas Louka. 2018. "Effect of the Extraction Process on the Biological Activity of Lyophilized Apricot Extracts Recovered from Apricot Pomace" Antioxidants 7, no. 1: 11. https://doi.org/10.3390/antiox7010011
APA StyleCheaib, D., El Darra, N., Rajha, H. N., El-Ghazzawi, I., Maroun, R. G., & Louka, N. (2018). Effect of the Extraction Process on the Biological Activity of Lyophilized Apricot Extracts Recovered from Apricot Pomace. Antioxidants, 7(1), 11. https://doi.org/10.3390/antiox7010011








