Green Tea and Other Tea Polyphenols: Effects on Sebum Production and Acne Vulgaris
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Selection of Studies
2.3. Data Extraction
3. Results
3.1. Sebum Production Studies
3.2. Acne Studies
3.2.1. Topical Tea Polyphenols
3.2.2. Systemic Tea Polyphenols
4. Discussion
4.1. Tea Polyphenol Bioavailability
4.2. Skin Penetration
4.3. Tolerability
4.4. Mechanisms of Action
4.5. Limitations
5. Conclusions
Author Contributions
Conflicts of Interest
Appendix A
| Tea | Polyphenol Subtypes and Content | Therapy (Topical or Systemic; Dosage) | Comparison (or Control/Placebo) | Subjects (n, Age) | Study Design, Duration | Major Outcome Measures | Major Results | Adverse Events | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Sebum Production Studies | |||||||||
| Green tea | GT formulation: 5% green tea; GT-L formulation: 2.5% green tea + 2.5% lotus extract. | Green tea (GT) Topical | Green tea + Lotus (GT-L) Topical; Placebo Topical | n = 22; all healthy men; Age 22–28 years-old. | Single-blinded, placebo-controlled, monocentric study; Pakistan (months December-February); 2 groups (G): G1: used GT topical on one cheek, placebo on the other; n = 11; G2: GT-L topical on one cheek, placebo on the other; n = 11. Intervention length: 60 days; Amount of tea polyphenol: information not provided; Frequency of application: once daily at bedtime; Follow up time: baseline, day 15, 30, 45, 60; Sebum excretion on both cheeks measured with Sebumeter at each visit. | Percent change in sebum secretion every 15 days (calculated by formula % change = ((Dx − D0)/D0) × 100; Dx = sebum value on day 15, 30 45, 60; D0 = baseline sebum value) | G1 (GT group): significant reduction in sebum secretion from baseline to 60 days of treatment compared to placebo control (p = 0.0060) (27% decrease from baseline); G2 (GT-L group): significant reduction in sebum secretion from baseline to 60 days of treatment compared to placebo control (p = 0.0002) (25% decrease from baseline); Placebo: no significant effect on reducing sebum secretion. | None reported | Mahmood et al., 2013 [15] |
| Green tea | 3% GT extract | Topical | No control group | n = 10; Age 24 to 40 years-old; all healthy men | Therapeutic study Intervention length: 60 days; Frequency of topical use: not stated; Amount of topical: not stated; Follow up: skin sebum production measured at week 1, 2, 3, 4, 6, and 8 via sebumeter. | Percent change in skin sebum production (calculated via formula % change = ((A − B)/B) × 100 A = individual value of any parameter (from 1st to 8th week); B = zero value for that parameter. | 3% GTE formulation decreased sebum production by nearly 10% at week 1, and 60% at week 8) (p < 0.05) | Mahmood et al., 2010 [34] | |
| Acne Studies | |||||||||
| Green tea | Amount of tea polyphenols: Each GTE capsule contained: 285.6 mg EGCG, 78.7 mg ECG, 38.5 mg EGC, 24 mg EC, 21.25 mg GCG, <0.3 mg GC, <0.3 mg caffeine, 51.65 mg cellulose | Systemic Decaffeinated GTE; each capsule 500 mg decaffeinated GTE or cellulose | Placebo (cellulose) | n = 80; All women; Age 25–45 years-old with moderate to severe acne (determined by Investigator’s Global Assessment (score of 3 or 4 on a scale 0 to 5)); None of the women received systemic retinoid or hormone treatment during previous 3 months. | Randomized, double blind, placebo controlled trial; May 2012 to October 2013, Taiwan; Intervention length: 4 weeks; Follow up: baseline and at 4 weeks; G1: received decaffeinated GTE for 4 weeks n = 40; G2: placebo (pure cellulose) for 4 weeks n = 40; Products packaged into identical capsules for blinding purposes; Frequency: Subjects took 1 capsule 30 min after meals 3 times daily for 4 weeks. | Acne lesion counts (inflammatory and non-inflammatory) recorded for entire face by blinded dermatologist at baseline and at 4 weeks | Between group comparison: G1 vs. G2: significant reduction in inflammatory lesion counts on nose (p = 0.03), perioral area (p = 0.04) and chin (p = 0.03); no difference in total lesion count; G1 did not have significant reduction on forehead, cheek and total lesion count compared to placebo; Within group comparison: G1: Compared with baseline, G1 (decaffeinated GTE) had significantly lower number of inflammatory lesions on forehead (p = 0.04), cheeks (p = 0.04) and entire face (total lesion count) (p = 0.003); G2: in placebo group, there was significant reduction in inflammatory lesion count on cheeks (p = 0.01), chin (p = 0.01) and entire face (p = 0.02). | G1: 1 subject developed mild constipation, 2 subjects abdominal discomfort after GTE treatment; G2: 1 subject thirsty, 1 difficulty falling asleep; No major adverse effects noted. | Lu et al., 2016 [39] |
| Tea | Apple brand tea leaves; Amount of tea polyphenol: not stated | Topical 2% tea lotion; | 5% zinc sulphate solution | n = 47; 33 females; 14 males; Age 13–27 years-old with acne vulgaris (severe acne excluded); Excluded comedonal, severe acne and nodulo-cystic acne and if on acne treatment in past 2 months; also systemic disease therapy with ie corticosteroids, hormones, medications that induce acniform rash. | Single-blind randomly comparative therapeutic clinical trial; Iraq, June 2006 to December 2007; Intervention length 8 weeks; Frequency of topical: twice daily; G1: 2% tea lotion n = 24 (17 females, 7 males); G2: 5% zinc sulphate solution n = 23 (16 females, 7 males); Follow up: Inflammatory (papules and pustules) lesions counted every 2 weeks for a total of 8 weeks; Photograph taken at baseline and subsequent visits. | Clinical improvement of acne vulgaris—scored by inflammatory lesion counting before and after treatment: Mild acne: <20 pustules and <10 papules; Moderate acne: 20–40 pustules, >30 papules). Good response: inflammatory (papules and pustules) lesion count decrease by >50%; Moderate response: lesion count decrease between 10% and 50%; No response: reduction in lesion count <10%. | n = 40 completed the study; n = 20 in each group; G1: 2% tea lotion statistically significant decrease in number of inflammatory lesions; Response to treatment: 15%—no response; 25%—moderate; 60%—good. Papule count before treatment 23.3 ± 10.9; After treatment 12 ± 8.7, statistically significant difference (p = 0.0001). Pustule count before treatment 29.5 ± 5.8; After treatment 14.5 ± 10.7, statistically significant reduction (p < 0.001). G2: zinc sulphate beneficial but result not statistically significant Response to treatment: 35%—no response; 50%—moderate; 15%—good. Papules before treatment 19.5 ± 9.7 and after treatment 14.4 ± 8.6 (reduction not statistically significant; p = 0.08); Pustules before treatment 19.4 ± 9.3 and after treatment 15.2 ± 9.5 (reduction not significant; p = 0.16). | G1: 5 subjects (25%): mild itching only in early course of treatment which then disappeared; G2: burning sensation in 5 subjects (25%), itching in 2 (10%) in early course of treatment which was subsequently reduced; No major adverse effects. | Sharquie et al., 2008 [35] |
| Green tea | EGCG | 1% EGCG and 5% EGCG Topical | Vehicle with 3% ethanol | n = 35; 17 men; 18 women; mean age: 22.1 (range not stated). | Randomly assigned split body trial; Intervention length: 8 weeks; Follow up: baseline, week 1, 2, 4, 6, and 8; Frequency of application: twice daily; Amount per application: not stated; G1: used 1% EGCG, n = 17; G2: used 5% EGCG, n = 18; Both groups applied lotion to one side of face, vehicle containing 3% ethanol to the other. | Dose response relationship between 1% and 5% EGCG in improving acne lesion count; Leeds grading and acne lesion count to calculate changes in non inflammatory lesions (whiteheads, blackheads, comedones) and inflammatory lesions (papules, pustules, nodules) during EGCG application period. | Baseline Leeds score: 5.1 ± 0.4; Week 8 Leeds score: 1.2 ± 0.4 (1% EGCG); 1.7 ± 0.6 (5% EGCG); 1% EGCG resulted in: 79% reduction of non-inflammatory acne (53.8 ± 19.8 mean lesions at baseline, 15.6 ± 6.2 at week 8, p < 0.05); 89% reduction in inflammatory acne (10 ± 3.1 mean lesions at baseline, 1.1±0.5 at week 8, p < 0.05); 5% EGCG significantly improved inflammatory and non-inflammatory lesion counts (p < 0.05), but details not provided. | Yoon et al., 2013 [31] | |
| Tea | Swan brand tea | Topical 2% tea lotion: (prepared by adding 75 mL tea extract + 25 mL ethanol (95% purity)) | Control lotion: (prepared by adding 75 mL distilled water + 25 mL ethanol (95% purity)) | n = 60; 25 males; 35 females; Age 14–22 years-old. | Iraq, October 2002 to October 2004; Intervention length: 2 months; Follow up: at 1 and 2 months; Frequency of topical application: twice daily; Amount applied: not stated; 2 groups: G1: n = 30, mean age 17.5 ± 3.9 years, treated with 2% tea lotion twice per day for 2 months, clinical evaluation monthly (inflammatory lesion count—papules and pustules). G2: n = 30, mean age 18.1 ± 3.7 years treated in the same manner but with control lotion. | Inflammatory lesion count (papules and pustules) assessed as follows: Mild acne: < 20 pustules, papules < 10; Moderate acne: 20–40 pustules, 10–30 papules; Severe acne: pustules > 40, papules > 30. Response to treatment classified as: Good response: >50% reduction in inflammatory lesion count; Moderate response: 10%–50% reduction in inflammatory lesion count; No response: <10% reduction in inflammatory lesion count. Patient satisfaction to treatment assessed as: - full satisfaction - partial satisfaction - no satisfaction | n = 49 completed treatment; G1: n = 25 Significant reduction in lesion count of papules (baseline 12 ± 3.3, week eight 8.1 ± 0.19, p < 0.001) and pustules (baseline 20.7 ±5.8, week eight 8.9 ± 2.3, p < 0.001); G2: n = 24. No significant reduction of papules or pustules at 8 weeks compared to baseline. Patient satisfaction after 8 weeks: G1: 64% fully satisfied, 6 patients (24%) partially satisfied, 12% not satisfied; G2: 83.3% not satisfied, 12.5% partially satisfied, 4.2% fully satisfied. | None | Sharquie et al., 2006 [36] |
| Green tea | Topical 2% green tea lotion (prepared by adding 75 mL tea extract + 25 mL ethanol (95% purity) | n = 20; 6 males; 14 females; Age 15–36 years old. | Egypt, May 2007 to February 2008; Intervention length: 6 weeks; Follow up: every 2 weeks; Frequency of application: twice daily; Amount of topical applied: not stated; Acne severity assessed, baseline photographs taken, green tea lotion given to be applied on face twice daily for 6 weeks, follow up every 2 weeks; TLC considered 100% at baseline and any decrease was considered improvement. | Change in TLC and SI scores at the end of treatment compared to baseline; Acne severity assessed every 2 weeks via total lesion count (TLC) and severity index (SI); TLC = papules + pustules; SI was determined as follows: SI = 1 if TLC < 10 SI = 2 if TLC = 10–20 SI = 3 if TLC > 20 | Mean TLC decreased from 24 (baseline) to 10 after 6 weeks of treatment (58.33% reduction, p < 0.0001, 95% CI = 8.58–19.42); Mean SI decreased from 2.05 (baseline) to 1.25 after 6 weeks of treatment (39.02% reduction, p < 0.0001, 95% CI = 0.54–1.26). | Stinging sensation in 2 subjects (10%) on first day of lotion use, disappeared in 48 h; Minimal pruritus in 3 of 20 subjects (15%) on first day of lotion use and lasted 3 days. | Elsaie et al., 2009 [37] | ||
| Green tea | Polyphenon-60 (green tea extract compound) | Topical 20 mg/mL polyphenon-60 applied to face twice daily for 8 weeks | None | n = 30, with mild to moderate acne; Age: not stated; Proportion of sex: not stated. | Therapeutic study; Intervention length: 8 weeks; Follow up time: baseline and at 8 weeks; Frequency of application: twice daily; Amount applied: not stated; Korea Topical polyphenon-60 (20 mg/mL) from green tea lotion applied onto facial acne twice daily for 8 weeks; Acne lesion count at baseline and at 8 weeks done by dermatologists. | Acne lesion count: open comedones, closed comedones, and pustules at baseline vs. 8 weeks after treatment | 8 weeks post-treatment compared to baseline: Number of open comedones decreased by 61% (p < 0.05); Number of pustules decreased by 28% (p < 0.05); Number of closed comedones was not significantly decreased. | Jung et al., 2012 [38] | |
References
- Watson, R.R.; Preedy, V.R.; Zibadi, S. Polyphenols in Human Health and Disease; Academic Press: New York, NY, USA, 2013. [Google Scholar]
- Habauzit, V.; Morand, C. Evidence for a protective effect of polyphenols-containing foods on cardiovascular health: An update for clinicians. Ther. Adv. Chronic Dis. 2012, 3, 87–106. [Google Scholar] [CrossRef] [PubMed]
- Zern, T.L.; Fernandez, M.L. Cardioprotective effects of dietary polyphenols. J. Nutr. 2005, 135, 2291–2294. [Google Scholar] [PubMed]
- Johnston, C. Functional foods as modifiers of cardiovascular disease. Am. J. Lifestyle Med. 2009, 3, 39S–43S. [Google Scholar] [CrossRef] [PubMed]
- Curin, Y.; Andriantsitohaina, R. Polyphenols as potential therapeutical agents against cardiovascular diseases. Pharmacol. Rep. 2005, 57, 97–107. [Google Scholar] [PubMed]
- Tsao, R. Chemistry and biochemistry of dietary polyphenols. Nutrients 2010, 2, 1231–1246. [Google Scholar] [CrossRef] [PubMed]
- Sies, H.; Stahl, W. Nutritional protection against skin damage from sunlight. Annu. Rev. Nutr. 2004, 24, 173–200. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.D. Does tea prevent cancer? Evidence from laboratory and human intervention studies. Am. J. Clin. Nutr. 2013, 98, 1667s–1675s. [Google Scholar] [CrossRef] [PubMed]
- Afaq, F.; Syed, D.N.; Malik, A.; Hadi, N.; Sarfaraz, S.; Kweon, M.H.; Khan, N.; Zaid, M.A.; Mukhtar, H. Delphinidin, an anthocyanidin in pigmented fruits and vegetables, protects human HaCaT keratinocytes and mouse skin against UVB-mediated oxidative stress and apoptosis. J. Investig. Dermatol. 2007, 127, 222–232. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.P.; Oh, K.N.; Yun, H.J.; Jeong, H.G. The flavonoids apigenin and luteolin suppress ultraviolet A-induced matrix metalloproteinase-1 expression via MAPKs and AP-1-dependent signaling in HaCaT cells. J. Dermatol. Sci. 2011, 61, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, U.; Moore, C.E.; de Spirt, S.; Tronnier, H.; Stahl, W. Green tea polyphenols provide photoprotection, increase microcirculation, and modulate skin properties of women. J. Nutr. 2011, 141, 1202–1208. [Google Scholar] [CrossRef] [PubMed]
- OyetakinWhite, P.; Tribout, H.; Baron, E. Protective mechanisms of green tea polyphenols in skin. Oxid. Med. Cell. Longev. 2012, 2012, 560682. [Google Scholar] [CrossRef] [PubMed]
- Graham, H.N. Green tea composition, consumption, and polyphenol chemistry. Prev. Med. 1992, 21, 334–350. [Google Scholar] [CrossRef]
- Smith, K.R.; Thiboutot, D.M. Thematic review series: Skin lipids. Sebaceous gland lipids: Friend or foe? J. Lipid Res. 2008, 49, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, T.; Akhtar, N.; Moldovan, C. A comparison of the effects of topical green tea and lotus on facial sebum control in healthy humans. Hippokratia 2013, 17, 64–67. [Google Scholar] [PubMed]
- Layton, A.M. Optimal management of acne to prevent scarring and psychological sequelae. Am. J. Clin. Dermatol. 2001, 2, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Zouboulis, C.C.; Eady, A.; Philpott, M.; Goldsmith, L.A.; Orfanos, C.; Cunliffe, W.C.; Rosenfield, R. What is the pathogenesis of acne? Exp. Dermatol. 2005, 14, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Melnik, B.C.; Zouboulis, C.C. Potential role of FoxO1 and mTORC1 in the pathogenesis of western diet-induced acne. Exp. Dermatol. 2013, 22, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Monfrecola, G.; Lembo, S.; Caiazzo, G.; de Vita, V.; di Caprio, R.; Balato, A.; Fabbrocini, G. Mechanistic target of rapamycin (mTOR) expression is increased in acne patients’ skin. Exp. Dermatol. 2016, 25, 153–155. [Google Scholar] [CrossRef] [PubMed]
- Agamia, N.F.; Abdallah, D.M.; Sorour, O.; Mourad, B.; Younan, D.N. Skin expression of mammalian target of rapamycin and forkhead box transcription factor O1, and serum insulin-like growth factor-1 in patients with acne vulgaris and their relationship with diet. Br. J. Dermatol. 2016, 174, 1299–1307. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.M.; Gilliland, K.; Clawson, G.A.; Thiboutot, D. IGF-1 induces SREBP-1 expression and lipogenesis in SEB-1 sebocytes via activation of the phosphoinositide 3-kinase/Akt pathway. J. Investig. Dermatol. 2008, 128, 1286–1293. [Google Scholar] [CrossRef] [PubMed]
- Melnik, B.C. Western diet-mediated mTORC1-signaling in acne, psoriasis, atopic dermatitis, and related diseases of civilization: Therapeutic role of plant-derived natural mTORC1 inhibitors. In Bioactive Dietary Factors and Plant Extracts in Dermatology; Humana Press: Totowa, NJ, USA, 2013; pp. 397–419. [Google Scholar]
- Im, M.; Kim, S.Y.; Sohn, K.C.; Choi, D.K.; Lee, Y.; Seo, Y.J.; Kim, C.D.; Hwang, Y.L.; Zouboulis, C.C.; Lee, J.H. Epigallocatechin-3-gallate suppresses IGF-I-induced lipogenesis and cytokine expression in SZ95 sebocytes. J. Investig. Dermatol. 2012, 132, 2700–2708. [Google Scholar] [CrossRef] [PubMed]
- Van Aller, G.S.; Carson, J.D.; Tang, W.; Peng, H.; Zhao, L.; Copeland, R.A.; Tummino, P.J.; Luo, L. Epigallocatechin gallate (EGCG), a major component of green tea, is a dual phosphoinositide-3-kinase/mTOR inhibitor. Biochem. Biophys. Res. Commun. 2011, 406, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Melnik, B.C. Linking diet to acne metabolomics, inflammation, and comedogenesis: An update. Clin. Cosmet. Investig. Dermatol. 2015, 8, 371–388. [Google Scholar] [CrossRef] [PubMed]
- Melnik, B.C.; Schmitz, G. Are therapeutic effects of antiacne agents mediated by activation of FoxO1 and inhibition of mTORC1? Exp. Dermatol. 2013, 22, 502–504. [Google Scholar] [CrossRef] [PubMed]
- Kuo, K.L.; Weng, M.S.; Chiang, C.T.; Tsai, Y.J.; Lin-Shiau, S.Y.; Lin, J.K. Comparative Studies on the Hypolipidemic and Growth Suppressive Effects of Oolong, Black, Pu-Erh, and Green Tea Leaves in Rats. J. Agric. Food Chem. 2005, 53, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Malahlela, P.; Motswaledi, M.H. Management of mild to moderate acne vulgaris. S. Afr. Fam. Pract. 2013, 55, 241–244. [Google Scholar] [CrossRef]
- Lucky, A.W.; Barber, B.L.; Girman, C.J.; Williams, J.; Ratterman, J.; Waldstreicher, J. A multirater validation study to assess the reliability of acne lesion counting. J. Am. Acad. Dermatol. 1996, 35, 559–565. [Google Scholar] [CrossRef]
- Doshi, A.; Zaheer, A.; Stiller, M.J. A comparison of current acne grading systems and proposal of a novel system. Int. J. Dermatol. 1997, 36, 416–418. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.Y.; Kwon, H.H.; Min, S.U.; Thiboutot, D.M.; Suh, D.H. Epigallocatechin-3-gallate improves acne in humans by modulating intracellular molecular targets and inhibiting P. Acnes. J. Investig. Dermatol. 2013, 133, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Leyden, J.J. Therapy for acne vulgaris. N. Engl. J. Med. 1997, 336, 1156–1162. [Google Scholar] [PubMed]
- Muthu, M.; Gopal, J.; Min, S.X.; Chun, S. Green tea versus traditional Korean teas: Antibacterial/antifungal or both? Appl. Biochem. Biotechnol. 2016, 180, 780–790. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, T.; Akhtar, N.; Khan, B.A.; Khan, H.M.; Saeed, T. Outcomes of 3% green tea emulsion on skin sebum production in male volunteers. Bosn. J. Basic Med. Sci. 2010, 10, 260–264. [Google Scholar] [PubMed]
- Sharquie, K.E.; Noaimi, A.A.; Al-Salih, M.M. Topical therapy of acne vulgaris using 2% tea lotion in comparison with 5% zinc sulphate solution. Saudi Med. J. 2008, 29, 1757–1761. [Google Scholar] [PubMed]
- Sharquie, K.E.; Al-Turfi, I.A.; Al-Shimary, W.M. Treatment of acne vulgaris with 2% topical tea lotion. Saudi Med. J. 2006, 27, 83–85. [Google Scholar] [PubMed]
- Elsaie, M.L.; Abdelhamid, M.F.; Elsaaiee, L.T.; Emam, H.M. The efficacy of topical 2% green tea lotion in mild-to-moderate acne vulgaris. J. Drugs Dermatol. 2009, 8, 358–364. [Google Scholar] [PubMed]
- Jung, M.K.; Ha, S.; Son, J.A.; Song, J.H.; Houh, Y.; Cho, E.; Chun, J.H.; Yoon, S.R.; Yang, Y.; Bang, S.I.; et al. Polyphenon-60 displays a therapeutic effect on acne by suppression of TLR2 and IL-8 expression via down-regulating the ERK1/2 pathway. Arch. Dermatol. Res. 2012, 304, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.H.; Hsu, C.H. Does supplementation with green tea extract improve acne in post-adolescent women? A randomized, double-blind, and placebo-controlled clinical trial. Complement. Ther. Med. 2016, 25, 159–163. [Google Scholar] [CrossRef] [PubMed]
- Levin, J.; Momin, S.B. How much do we really know about our favorite cosmeceutical ingredients? J. Clin. Aesthet. Dermatol. 2010, 3, 22–41. [Google Scholar] [PubMed]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Remesy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2005, 81, 230S–242S. [Google Scholar] [PubMed]
- Mereles, D.; Hunstein, W. Epigallocatechin-3-gallate (EGCG) for clinical trials: More pitfalls than promises? Int. J. Mol. Sci. 2011, 12, 5592–5603. [Google Scholar] [CrossRef] [PubMed]
- Peter, B.; Bosze, S.; Horvath, R. Biophysical characteristics of proteins and living cells exposed to the green tea polyphenol epigallocatechin-3-gallate (EGCG): Review of recent advances from molecular mechanisms to nanomedicine and clinical trials. Eur. Biophys. J. 2016. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Chen, L.; Lee, M.J.; Balentine, D.; Kuo, M.C.; Schantz, S.P. Blood and urine levels of tea catechins after ingestion of different amounts of green tea by human volunteers. Cancer Epidemiol. Biomarkers Prev. 1998, 7, 351–354. [Google Scholar] [PubMed]
- Yanagida, A.; Shoji, A.; Shibusawa, Y.; Shindo, H.; Tagashira, M.; Ikeda, M.; Ito, Y. Analytical separation of tea catechins and food-related polyphenols by high-speed counter-current chromatography. J. Chromatogr. A 2006, 1112, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Dal Belo, S.E.; Gaspar, L.R.; Maia Campos, P.M.; Marty, J.P. Skin penetration of epigallocatechin-3-gallate and quercetin from green tea and Ginkgo biloba extracts vehiculated in cosmetic formulations. Skin Pharmacol. Physiol. 2009, 22, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Zillich, O.V.; Schweiggert-Weisz, U.; Hasenkopf, K.; Eisner, P.; Kerscher, M. Release and in vitro skin permeation of polyphenols from cosmetic emulsions. Int. J. Cosmet. Sci. 2013, 35, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Bojar, R.A.; Holland, K.T. Acne and propionibacterium acnes. Clin. Dermatol. 2004, 22, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Bek-Thomsen, M.; Lomholt, H.B.; Kilian, M. Acne is not associated with yet-uncultured bacteria. J. Clin. Microbiol. 2008, 46, 3355–3360. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Summanen, P.H.; Downes, J.; Corbett, K.; Komoriya, T.; Henning, S.M.; Kim, J.; Finegold, S.M. Antimicrobial activity of pomegranate and green tea extract on propionibacterium acnes, propionibacterium granulosum, staphylococcus aureus and staphylococcus epidermidis. J. Drugs Dermatol. 2015, 14, 574–578. [Google Scholar] [PubMed]
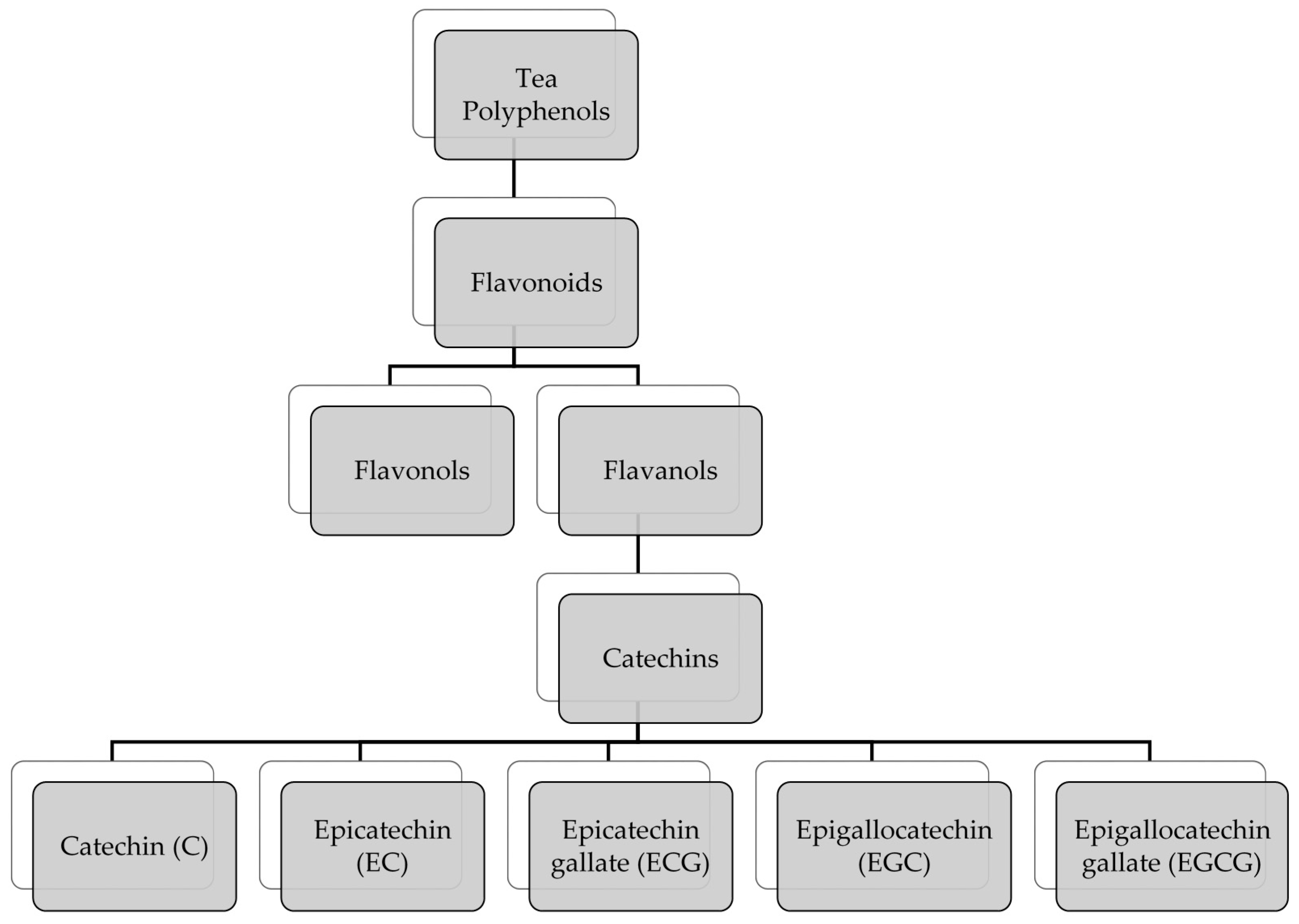
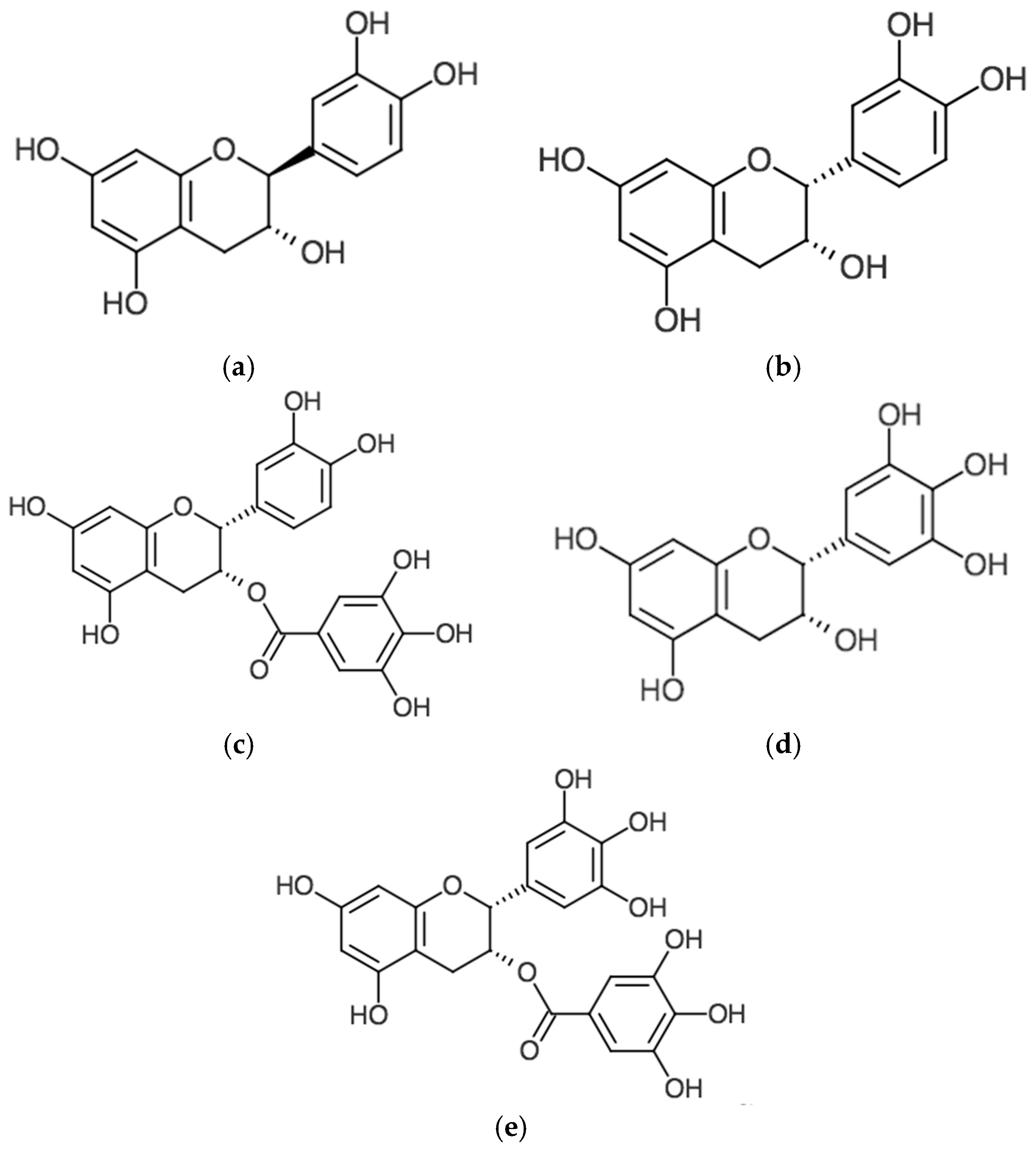
| Polyphenol | K(non-polar/polar) |
|---|---|
| Epicatechin gallate (ECG) | 6.25 |
| Epigallocatechin gallate (EGCG) | 2.94 |
| Epicatechin (EC) | 2.38 |
| Catechin (C) | 2.33 |
| Epigallocatechin (EGC) | 0.93 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saric, S.; Notay, M.; Sivamani, R.K. Green Tea and Other Tea Polyphenols: Effects on Sebum Production and Acne Vulgaris. Antioxidants 2017, 6, 2. https://doi.org/10.3390/antiox6010002
Saric S, Notay M, Sivamani RK. Green Tea and Other Tea Polyphenols: Effects on Sebum Production and Acne Vulgaris. Antioxidants. 2017; 6(1):2. https://doi.org/10.3390/antiox6010002
Chicago/Turabian StyleSaric, Suzana, Manisha Notay, and Raja K. Sivamani. 2017. "Green Tea and Other Tea Polyphenols: Effects on Sebum Production and Acne Vulgaris" Antioxidants 6, no. 1: 2. https://doi.org/10.3390/antiox6010002
APA StyleSaric, S., Notay, M., & Sivamani, R. K. (2017). Green Tea and Other Tea Polyphenols: Effects on Sebum Production and Acne Vulgaris. Antioxidants, 6(1), 2. https://doi.org/10.3390/antiox6010002







