Role of Redox Signaling and Inflammation in Skeletal Muscle Adaptations to Training
Abstract
:1. Introduction
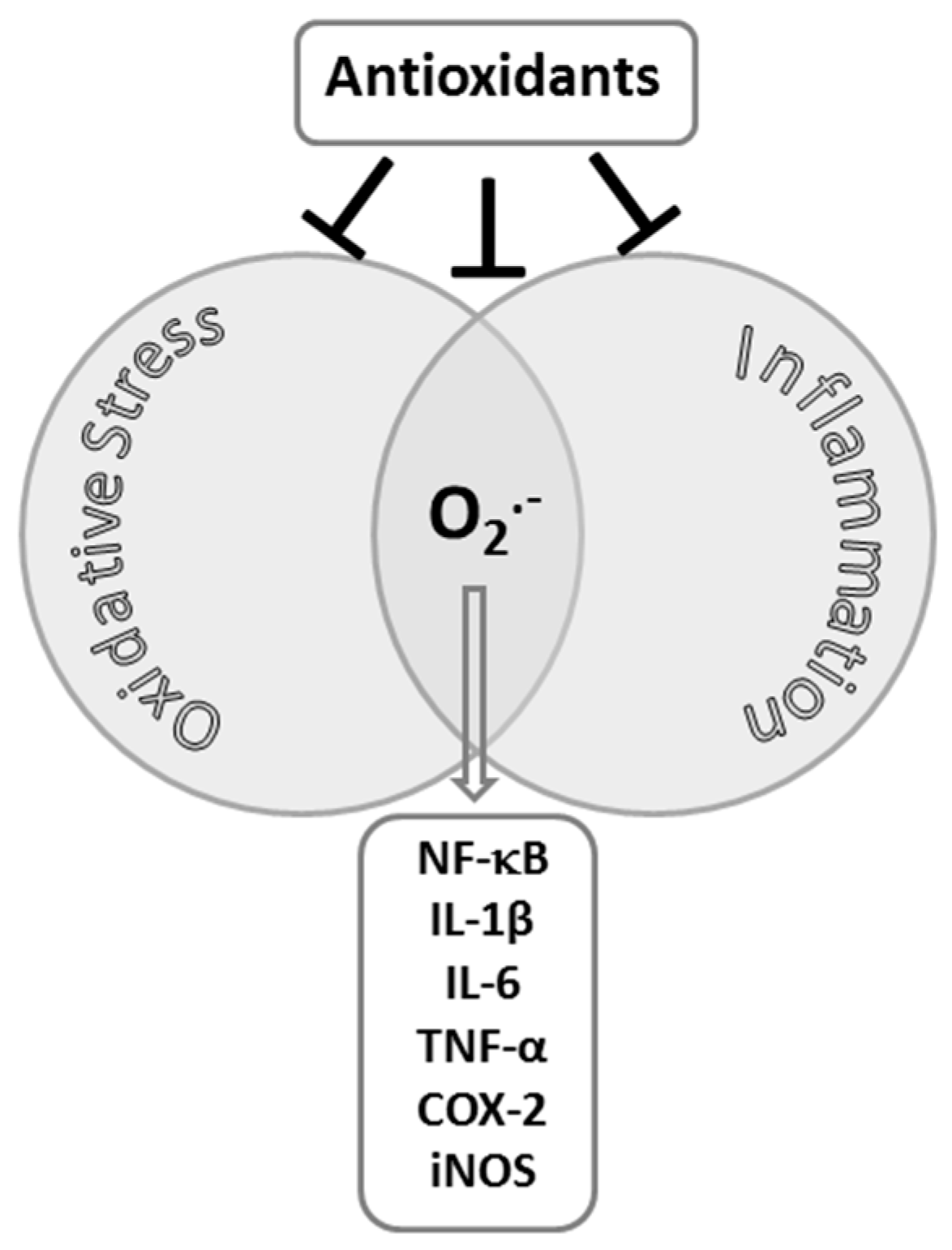
2. Exercise-Induced Inflammation and Oxidative Stress in Skeletal Muscle
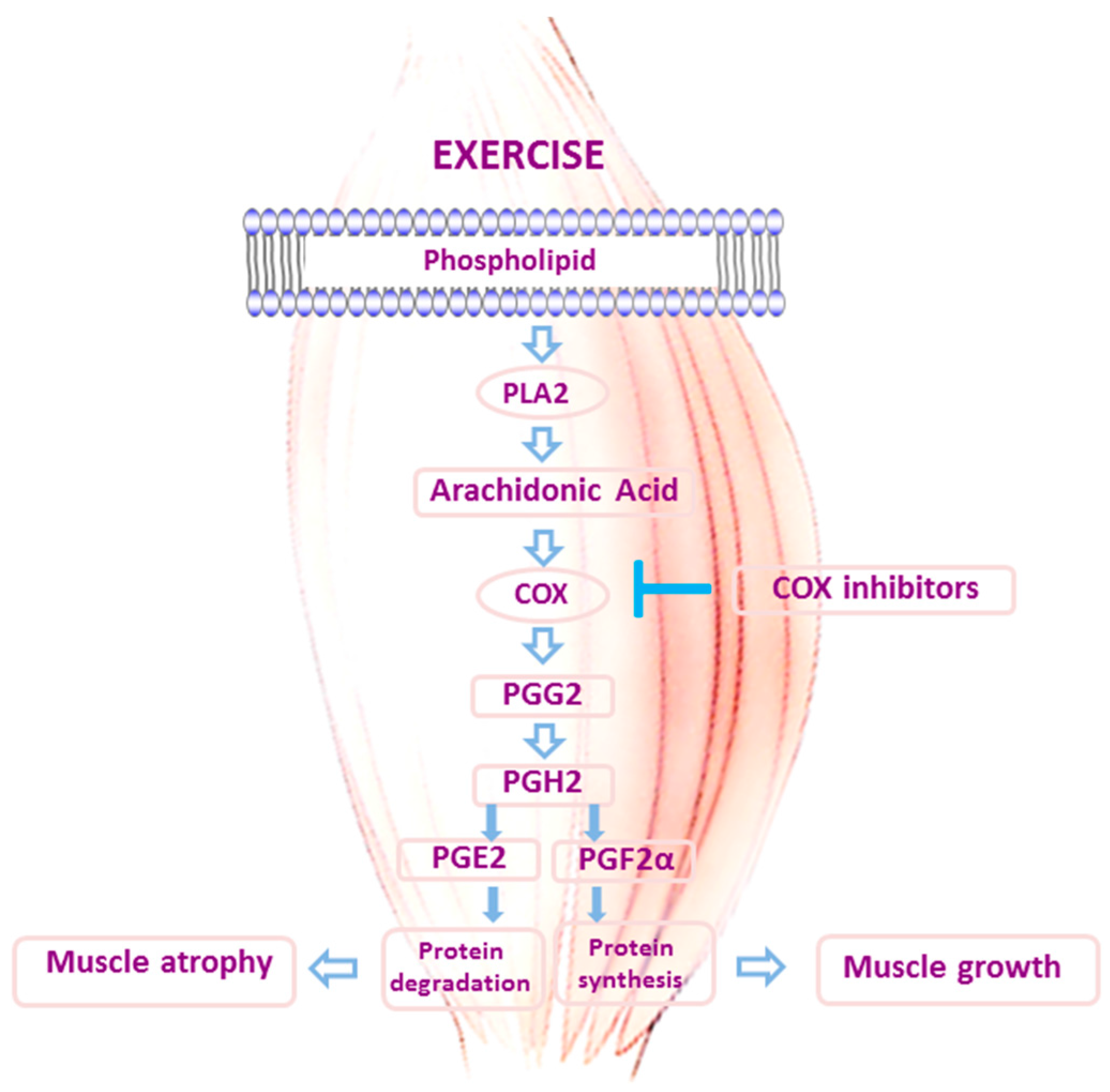
3. Role of Inflammation in Skeletal Muscle Adaptations to Exercise: Should We Block It with Anti-Inflammatory Drugs?
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mueller, K. Inflammation. Inflammation’s Yin-Yang. Introduction. Science 2013, 339, 155. [Google Scholar] [CrossRef] [PubMed]
- Young, H.A.; Ortaldo, J. Cytokines as critical co-stimulatory molecules in modulating the immune response of natural killer cells. Cell Res. 2006, 16, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Luster, A.D. Chemokines—Chemotactic cytokines that mediate inflammation. N. Engl. J. Med. 1998, 338, 436–445. [Google Scholar] [PubMed]
- Opal, S.M.; DePalo, V.A. Anti-inflammatory cytokines. Chest J. 2000, 117, 1162–1172. [Google Scholar] [CrossRef]
- Franceschi, C.; Capri, M.; Monti, D.; Giunta, S.; Olivieri, F.; Sevini, F.; Panourgia, M.P.; Invidia, L.; Celani, L.; Scurti, M.; et al. Inflammaging and anti-inflammaging: A systemic perspective on aging and longevity emerged from studies in humans. Mech. Ageing Dev. 2007, 128, 92–105. [Google Scholar] [CrossRef] [PubMed]
- Liao, P.; Zhou, J.; Ji, L.L.; Zhang, Y. Eccentric contraction induces inflammatory responses in rat skeletal muscle: Role of tumor necrosis factor-alpha. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 298, R599–R607. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K.; Hoffman-Goetz, L. Exercise and the immune system: Regulation, integration, and adaptation. Physiol. Rev. 2000, 80, 1055–1081. [Google Scholar] [PubMed]
- Cai, D.; Frantz, J.D.; Tawa, N.E., Jr.; Melendez, P.A.; Oh, B.C.; Lidov, H.G.; Hasselgren, P.O.; Frontera, W.R.; Lee, J.; Glass, D.J.; et al. IKKbeta/NF-kappaB activation causes severe muscle wasting in mice. Cell 2004, 119, 285–298. [Google Scholar] [CrossRef] [PubMed]
- Trappe, T.A.; White, F.; Lambert, C.P.; Cesar, D.; Hellerstein, M.; Evans, W.J. Effect of ibuprofen and acetaminophen on postexercise muscle protein synthesis. Am. J. Physiol. Endocrinol. Metab. 2002, 282, E551–E556. [Google Scholar] [CrossRef] [PubMed]
- Rodeman, H.P.; Goldberg, A.L. Arachidonic acid, prostaglandin E2 and F2 alpha influence rates of protein turnover in skeletal and cardiac muscle. J. Biol. Chem. 1982, 257, 1632–1638. [Google Scholar]
- Trappe, T.A.; Liu, S.Z. Effects of prostaglandins and cox-inhibiting drugs on skeletal muscle adaptations to exercise. J. Appl. Physiol. 2013, 115, 909–919. [Google Scholar] [CrossRef] [PubMed]
- Trappe, T.A.; Standley, R.A.; Jemiolo, B.; Carroll, C.C.; Trappe, S.W. Prostaglandin and myokine involvement in the cyclooxygenase-inhibiting drug enhancement of skeletal muscle adaptations to resistance exercise in older adults. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 304, R198–R205. [Google Scholar] [CrossRef] [PubMed]
- Sanchis-Gomar, F.; Pareja-Galeano, H.; Gomez-Cabrera, M.C.; Candel, J.; Lippi, G.; Salvagno, G.L.; Mann, G.E.; Vina, J. Allopurinol prevents cardiac and skeletal muscle damage in professional soccer players. Scand. J. Med. Sci. Sports 2014, 25, e110–e115. [Google Scholar] [CrossRef] [PubMed]
- Petrone, W.F.; English, D.K.; Wong, K.; McCord, J.M. Free radicals and inflammation: Superoxide-dependent activation of a neutrophil chemotactic factor in plasma. Proc. Natl. Acad. Sci. USA 1980, 77, 1159–1163. [Google Scholar] [CrossRef] [PubMed]
- McCord, J.M.; Wong, K.; Stokes, S.H.; Petrone, W.F.; English, D. Superoxide and inflammation: A mechanism for the anti-inflammatory activity of superoxide dismutase. Acta Physiol. Scand. Suppl. 1980, 492, 25–30. [Google Scholar] [PubMed]
- McCord, J.M. Free radicals and inflammation: Protection of synovial fluid by superoxide dismutase. Science 1974, 185, 5229–5531. [Google Scholar] [CrossRef]
- Chung, H.Y.; Sung, B.; Jung, K.J.; Zou, Y.; Yu, B.P. The molecular inflammatory process in aging. Antioxid. Redox Signal. 2006, 8, 572–581. [Google Scholar] [CrossRef] [PubMed]
- Meyer, M.; Pahl, H.L.; Baeuerle, P.A. Regulation of the transcription factors NF-κB and AP-1 by redox changes. Chem. Biol. Interact. 1994, 91, 91–100. [Google Scholar] [CrossRef]
- Ghosh, S.; Karin, M. Missing pieces in the NF-κB puzzle. Cell 2002, 109 (Suppl. 1), S81–S96. [Google Scholar] [CrossRef]
- Hollander, J.; Fiebig, R.; Gore, M.; Bejma, J.; Ookawara, T.; Ohno, H.; Ji, L.L. Superoxide dismutase gene expression in skeletal muscle: Fiber-specific adaptation to endurance training. Am. J. Physiol. 1999, 277, R856–R862. [Google Scholar] [PubMed]
- Ji, L.L.; Gomez-Cabrera, M.C.; Steinhafel, N.; Vina, J. Acute exercise activates nuclear factor (NF)-κB signaling pathway in rat skeletal muscle. FASEB J. 2004, 18, 1499–1506. [Google Scholar] [CrossRef] [PubMed]
- Hollander, J.; Fiebig, R.; Gore, M.; Ookawara, T.; Ohno, H.; Ji, L.L. Superoxide dismutase gene expression is activated by a single bout of exercise in rat skeletal muscle. Pflug. Arch. 2001, 442, 426–434. [Google Scholar] [CrossRef]
- Hansen, J.M.; Zhang, H.; Jones, D.P. Differential oxidation of thioredoxin-1, thioredoxin-2, and glutathione by metal ions. Free Radic. Biol. Med. 2006, 40, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Cabrera, M.C.; Vina, J.; Ji, L.L. Interplay of oxidants and antioxidants during exercise: Implications for muscle health. Phys. Sportsmed. 2009, 37, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K.; Ostrowski, K.; Rohde, T.; Bruunsgaard, H. The cytokine response to strenuous exercise. Can. J. Physiol. Pharmacol. 1998, 76, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Aoi, W.; Naito, Y.; Takanami, Y.; Kawai, Y.; Sakuma, K.; Ichikawa, H.; Yoshida, N.; Yoshikawa, T. Oxidative stress and delayed-onset muscle damage after exercise. Free Radic. Biol. Med. 2004, 37, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Powers, S.K.; Ji, L.L.; Kavazis, A.N.; Jackson, M.J. Reactive oxygen species: Impact on skeletal muscle. Compr. Physiol. 2011, 1, 941–969. [Google Scholar] [PubMed]
- Thompson, D.; Williams, C.; Garcia-Roves, P.; McGregor, S.J.; McArdle, F.; Jackson, M.J. Post-exercise vitamin C supplementation and recovery from demanding exercise. Eur. J. Appl. Physiol. 2003, 89, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Howatson, G.; McHugh, M.P.; Hill, J.A.; Brouner, J.; Jewell, A.P.; van Someren, K.A.; Shave, R.E.; Howatson, S.A. Influence of tart cherry juice on indices of recovery following marathon running. Scand. J. Med. Sci. Sports 2009, 20, 843–852. [Google Scholar] [CrossRef] [PubMed]
- Seeram, N.P.; Momin, R.A.; Nair, M.G.; Bourquin, L.D. Cyclooxygenase inhibitory and antioxidant cyanidin glycosides in cherries and berries. Phytomedicine 2001, 8, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Bell, P.G.; Walshe, I.H.; Davison, G.W.; Stevenson, E.; Howatson, G. Montmorency cherries reduce the oxidative stress and inflammatory responses to repeated days high-intensity stochastic cycling. Nutrients 2014, 6, 829–843. [Google Scholar] [CrossRef] [PubMed]
- Kevil, C.G.; Pruitt, H.; Kavanagh, T.J.; Wilkerson, J.; Farin, F.; Moellering, D.; Darley-Usmar, V.M.; Bullard, D.C.; Patel, R.P. Regulation of endothelial glutathione by ICAM-1: Implications for inflammation. FASEB J. 2004, 18, 1321–1323. [Google Scholar] [CrossRef] [PubMed]
- Best, T.M.; Fiebig, R.; Corr, D.T.; Brickson, S.; Ji, L. Free radical activity, antioxidant enzyme, and glutathione changes with muscle stretch injury in rabbits. J. Appl. Physiol. 1999, 87, 74–82. [Google Scholar] [PubMed]
- Viña, J.; Gimeno, A.; Sastre, J.; Desco, C.; Asensi, M.; Pallardo, F.V.; Cuesta, A.; Ferrero, J.A.; Terada, L.S.; Repine, J.E. Mechanism of free radical production in exhaustive exercise in humans and rats; role of xanthine oxidase and protection by allopurinol. IUBMB Life 2000, 49, 539–544. [Google Scholar] [PubMed]
- Gomez-Cabrera, M.C.; Pallardo, F.V.; Sastre, J.; Vina, J.; Garcia-del-Moral, L. Allopurinol and markers of muscle damage among participants in the Tour de France. JAMA 2003, 289, 2503–2504. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Cabrera, M.C.; Martinez, A.; Santangelo, G.; Pallardo, F.V.; Sastre, J.; Vina, J. Oxidative stress in marathon runners: Interest of antioxidant supplementation. Br. J. Nutr. 2006, 96 (Suppl. S1), S31–S33. [Google Scholar] [CrossRef] [PubMed]
- Peake, J.; Suzuki, K. Neutrophil activation, antioxidant supplements and exercise-induced oxidative stress. Exerc. Immunol. Rev. 2004, 10, 129–141. [Google Scholar] [PubMed]
- Peake, J.; Nosaka, K.; Suzuki, K. Characterization of inflammatory responses to eccentric exercise in humans. Exerc. Immunol. Rev. 2005, 11, 64–85. [Google Scholar] [PubMed]
- Bourgeois, J.; MacDougall, D.; MacDonald, J.; Tarnopolsky, M. Naproxen does not alter indices of muscle damage in resistance-exercise trained men. Med. Sci. Sports Exerc. 1999, 31, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Hasson, S.M.; Daniels, J.C.; Divine, J.G.; Niebuhr, B.R.; Richmond, S.; Stein, P.G.; Williams, J.H. Effect of ibuprofen use on muscle soreness, damage, and performance: A preliminary investigation. Med. Sci. Sports Exerc. 1993, 25, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Pizza, F.X.; Cavender, D.; Stockard, A.; Baylies, H.; Beighle, A. Anti-inflammatory doses of ibuprofen: Effect on neutrophils and exercise-induced muscle injury. Int. J. Sports Med. 1999, 20, 98–102. [Google Scholar] [CrossRef] [PubMed]
- O’Grady, M.; Hackney, A.C.; Schneider, K.; Bossen, E.; Steinberg, K.; Douglas, J.M., Jr.; Murray, W.J.; Watkins, W.D. Diclofenac sodium (Voltaren) reduced exercise-induced injury in human skeletal muscle. Med. Sci. Sports Exerc. 2000, 32, 1191–1196. [Google Scholar] [CrossRef] [PubMed]
- Sayers, S.P.; Knight, C.A.; Clarkson, P.M.; van Wegen, E.H.; Kamen, G. Effect of ketoprofen on muscle function and sEMG activity after eccentric exercise. Med. Sci. Sports Exerc. 2001, 33, 702–710. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Cabrera, M.C.; Salvador-Pascual, A.; Cabo, H.; Ferrando, B.; Vina, J. Redox modulation of mitochondriogenesis in exercise. Does antioxidant supplementation blunt the benefits of exercise training? Free Radic. Biol. Med. 2015, 86, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Powers, S.K.; Radak, Z.; Ji, L.L. Exercise-induced oxidative stress: Past, present and future. J. Physiol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.J.; Jones, D.A.; Edwards, R.H. Vitamin E and skeletal muscle. Ciba Found. Symp. 1983, 101, 224–239. [Google Scholar] [PubMed]
- Gomez-Cabrera, M.C.; Borras, C.; Pallardo, F.V.; Sastre, J.; Ji, L.L.; Vina, J. Decreasing xanthine oxidase mediated oxidative stress prevents useful cellular adaptations to exercise in rats. J. Physiol. 2005, 567, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Sen, C.K.; Atalay, M.; Hänninen, O. Exercise-induced oxidative stress: Glutahione supplementation and deficiency. J. Appl. Physiol. 1994, 79, 675–686. [Google Scholar]
- Sastre, J.; Asensi, M.; Gasco, E.; Pallardo, F.V.; Ferrero, J.A.; Furukawa, T.; Vina, J. Exhaustive physical exercise causes oxidation of glutathione status in blood: Prevention by antioxidant administration. Am. J. Physiol. 1992, 263, R992–R995. [Google Scholar] [PubMed]
- Barclay, J.K.; Hansel, M. Free radicals may contribute to oxidative skeletal muscle fatigue. Can. J. Physiol. Pharmacol. 1991, 69, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Shindoh, A.; Dimarco, A.; Thomas, A. Effect of N-acetylcysteine on diaphragm fatigue. J. Appl. Physiol. 1990, 68, 2107–2113. [Google Scholar] [PubMed]
- Reid, M.B.; Stoik, D.S.; Koch, S.M.; Khawli, F.A.; Lois, A.A. N-acetylcysteine inhibits muscle fatigue in humans. J. Clin. Investig. 1994, 94, 2468–2474. [Google Scholar] [CrossRef] [PubMed]
- Medved, I.; Brown, M.J.; Bjorksten, A.R.; Murphy, K.T.; Petersen, A.C.; Sostaric, S.; Gong, X.; McKenna, M.J. N-acetylcysteine enhances muscle cysteine and glutathione availability and attenuates fatigue during prolonged exercise in endurance-trained individuals. J. Appl. Physiol. 2004, 97, 1477–1485. [Google Scholar] [CrossRef] [PubMed]
- McKenna, M.J.; Medved, I.; Goodman, C.A.; Brown, M.J.; Bjorksten, A.R.; Murphy, K.T.; Petersen, A.C.; Sostaric, S.; Gong, X. N-acetylcysteine attenuates the decline in muscle Na+, K+-pump activity and delays fatigue during prolonged exercise in humans. J. Physiol. 2006, 576, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Leeuwenburgh, C.; Ji, L.L. Glutathione and glutathione ethyl ester supplementation of mice alter glutathione homeostasis during exercise. J. Nutr. 1998, 128, 2420–2426. [Google Scholar] [PubMed]
- Ristow, M.; Zarse, K.; Oberbach, A.; Kloting, N.; Birringer, M.; Kiehntopf, M.; Stumvoll, M.; Kahn, C.R.; Bluher, M. Antioxidants prevent health-promoting effects of physical exercise in humans. Proc. Natl. Acad. Sci. USA 2009, 106, 8665–8670. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Cabrera, M.C.; Domenech, E.; Romagnoli, M.; Arduini, A.; Borras, C.; Pallardo, F.V.; Sastre, J.; Vina, J. Oral administration of vitamin C decreases muscle mitochondrial biogenesis and hampers training-induced adaptations in endurance performance. Am. J. Clin. Nutr. 2008, 87, 142–149. [Google Scholar] [PubMed]
- Paulsen, G.; Cumming, K.T.; Holden, G.; Hallen, J.; Ronnestad, B.R.; Sveen, O.; Skaug, A.; Paur, I.; Bastani, N.E.; Ostgaard, H.N.; et al. Vitamin C and E supplementation hampers cellular adaptation to endurance training in humans: A double-blind, randomised, controlled trial. J. Physiol. 2014, 592, 1887–1901. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Cabrera, M.C.; Domenech, E.; Vina, J. Moderate exercise is an antioxidant: Upregulation of antioxidant genes by training. Free Radic. Biol. Med. 2008, 44, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Kometiani, P.; Liu, J.; Li, J.; Shapiro, J.I.; Askari, A. Intracellular reactive oxygen species mediate the linkage of Na+/K+-atpase to hypertrophy and its marker genes in cardiac myocytes. J. Biol. Chem. 1999, 274, 19323–19328. [Google Scholar] [CrossRef] [PubMed]
- McGinley, C.; Shafat, A.; Donnelly, A.E. Does antioxidant vitamin supplementation protect against muscle damage? Sports Med. 2009, 39, 1011–1032. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, R.R. The underappreciated role of muscle in health and disease. Am. J. Clin. Nutr. 2006, 84, 475–482. [Google Scholar] [PubMed]
- Rosenberg, I. Summary comments: Epidemiological and methodological problems in determining nutritional status of older persons. Am. J. Clin. Nutr. 1989, 50, 1231–1233. [Google Scholar]
- Reid, M.B.; Judge, A.R.; Bodine, S.C. Crosstalk opposing view: The dominant mechanism causing disuse muscle atrophy is proteolysis. J. Physiol. 2014, 592, 5345–5347. [Google Scholar] [CrossRef] [PubMed]
- Von Euler, U.S. On the specific vaso-dilating and plain muscle stimulating substances from accessory genital glands in man and certain animals (prostaglandin and vesiglandin). J. Physiol. 1936, 88, 213–234. [Google Scholar] [CrossRef] [PubMed]
- Smith, W.L.; Urade, Y.; Jakobsson, P.J. Enzymes of the cyclooxygenase pathways of prostanoid biosynthesis. Chem. Rev. 2011, 111, 5821–5865. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, D.W.; Kelly, J.P.; Rosenberg, L.; Anderson, T.E.; Mitchell, A.A. Recent patterns of medication use in the ambulatory adult population of the United States: The Slone survey. JAMA 2002, 287, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Simmons, D.L.; Botting, R.M.; Hla, T. Cyclooxygenase isozymes: The biology of prostaglandin synthesis and inhibition. Pharmacol. Rev. 2004, 56, 387–437. [Google Scholar] [CrossRef] [PubMed]
- Carroll, C.C.; O’Connor, D.T.; Steinmeyer, R.; Del Mundo, J.D.; McMullan, D.R.; Whitt, J.A.; Ramos, J.E.; Gonzales, R.J. The influence of acute resistance exercise on cyclooxygenase-1 and -2 activity and protein levels in human skeletal muscle. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 305, R24–R30. [Google Scholar] [CrossRef] [PubMed]
- Herbaczynska-Cedro, K.; Staszewska-Barczak, J. Proceedings: Muscular work and prostaglandin release. Br. J. Pharmacol. 1974, 52, 454P–455P. [Google Scholar] [PubMed]
- Berlin, T.; Cronestrand, R.; Nowak, J.; Sonnenfeld, T.; Wennmalm, A. Conversion of arachidonic acid to prostaglandins in homogenates of human skeletal muscle and kidney. Acta. Physiol. Scand. 1979, 106, 441–445. [Google Scholar] [CrossRef] [PubMed]
- Markworth, J.F.; Cameron-Smith, D. Prostaglandin F2α stimulates PI3K/ERK/mTOR signaling and skeletal myotube hypertrophy. Am. J. Physiol. Cell Physiol. 2011, 300, C671–C682. [Google Scholar] [CrossRef] [PubMed]
- Markworth, J.F.; Cameron-Smith, D. Arachidonic acid supplementation enhances in vitro skeletal muscle cell growth via a COX-2-dependent pathway. Am. J. Physiol. Cell Physiol. 2013, 304, C56–C67. [Google Scholar] [CrossRef] [PubMed]
- Karamouzis, M.; Langberg, H.; Skovgaard, D.; Bulow, J.; Kjaer, M.; Saltin, B. In situ microdialysis of intramuscular prostaglandin and thromboxane in contracting skeletal muscle in humans. Acta Physiol. Scand. 2001, 171, 71–76. [Google Scholar] [PubMed]
- Karamouzis, M.; Karamouzis, I.; Vamvakoudis, E.; Ampatzidis, G.; Christoulas, K.; Angelopoulou, N.; Mandroukas, K. The response of muscle interstitial prostaglandin E2(PGE2), prostacyclin I2(PGI2) and thromboxane A2(TXA2) levels during incremental dynamic exercise in humans determined by in vivo microdialysis. Prostaglandins Leukot. Essent. Fat. Acids 2001, 64, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Mikkelsen, U.R.; Helmark, I.C.; Kjaer, M.; Langberg, H. Prostaglandin synthesis can be inhibited locally by infusion of nsaids through microdialysis catheters in human skeletal muscle. J. Appl. Physiol. 2008, 104, 534–537. [Google Scholar] [CrossRef] [PubMed]
- Boushel, R.; Langberg, H.; Gemmer, C.; Olesen, J.; Crameri, R.; Scheede, C.; Sander, M.; Kjaer, M. Combined inhibition of nitric oxide and prostaglandins reduces human skeletal muscle blood flow during exercise. J. Physiol. 2002, 543, 691–698. [Google Scholar] [CrossRef] [PubMed]
- Markworth, J.F.; Vella, L.D.; Figueiredo, V.C.; Cameron-Smith, D. Ibuprofen treatment blunts early translational signaling responses in human skeletal muscle following resistance exercise. J. Appl. Physiol. 2014, 117, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Krentz, J.R.; Quest, B.; Farthing, J.P.; Quest, D.W.; Chilibeck, P.D. The effects of ibuprofen on muscle hypertrophy, strength, and soreness during resistance training. Appl. Physiol. Nutr. Metab. 2008, 33, 470–475. [Google Scholar] [CrossRef] [PubMed]
- Petersen, S.G.; Beyer, N.; Hansen, M.; Holm, L.; Aagaard, P.; Mackey, A.L.; Kjaer, M. Nonsteroidal anti-inflammatory drug or glucosamine reduced pain and improved muscle strength with resistance training in a randomized controlled trial of knee osteoarthritis patients. Arch. Phys. Med. Rehabil. 2011, 92, 1185–1193. [Google Scholar] [CrossRef] [PubMed]
- Trappe, T.A.; Carroll, C.C.; Dickinson, J.M.; LeMoine, J.K.; Haus, J.M.; Sullivan, B.E.; Lee, J.D.; Jemiolo, B.; Weinheimer, E.M.; Hollon, C.J. Influence of acetaminophen and ibuprofen on skeletal muscle adaptations to resistance exercise in older adults. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 300, R655–R662. [Google Scholar] [CrossRef] [PubMed]
- Trappe, T.A.; Ratchford, S.M.; Brower, B.E.; Liu, S.Z.; Lavin, K.M.; Carroll, C.C.; Jemiolo, B.; Trappe, S.W. COX inhibitor influence on skeletal muscle fiber size and metabolic adaptations to resistance exercise in older adults. J. Gerontol. A Biol. Sci. Med. Sci. 2016, 71, 1289–1294. [Google Scholar] [CrossRef] [PubMed]
- Trappe, T.A.; Fluckey, J.D.; White, F.; Lambert, C.P.; Evans, W.J. Skeletal muscle PGF2α and PG2 in response to eccentric resistance exercise: Influence of ibuprofen acetaminophen. J. Clin. Endocrinol. Metab. 2001, 86, 5067–5070. [Google Scholar] [PubMed]
- Rieu, I.; Magne, H.; Savary-Auzeloux, I.; Averous, J.; Bos, C.; Peyron, M.A.; Combaret, L.; Dardevet, D. Reduction of low grade inflammation restores blunting of postprandial muscle anabolism and limits sarcopenia in old rats. J. Physiol. 2009, 587, 5483–5492. [Google Scholar] [CrossRef] [PubMed]
| Author | Sample | Training Protocol | Dose of the Anti-Inflammatory | Main Findings |
|---|---|---|---|---|
| Trappe and co-workers [9] | Humans (Young) | 10–14 sets of 10 eccentric repetitions at 120% of concentric one repetition maximum with the knee extensors | Ibuprofen (1200 mg/day), acetaminophen (4000 mg/day) after the exercise | Anti-inflammatory attenuated the increased rate of muscle protein synthesis 24 h after exercise. No effect on whole body protein breakdown, on serum CK, or on muscle soreness |
| Krentz and co-workers [79] | Humans (Young) | 6 sets of biceps curls (3 sets of 8 to 10 concentric repetitions at 70% 1 RM and 3 sets of 4 to 6 eccentric repetitions at 100% 1 RM), 2–3 days/week for 6 week | Ibuprofen (400 mg) taken after exercise | No effect of ibuprofen on muscle growth or strength adaptations |
| Petersen and co-workers [80] | Human (Old with osteoarthritis) | 12 week of progressive resistance training, 3 days/week (4 to 5 sets of 8 to 15 repetitions at 70%–80% of 1 RM) | Ibuprofen (1200 mg/day) | No effect on muscle mass gains, but muscle strength was enhanced in those individuals consuming the COX inhibitor (maybe explained by the pain relief). |
| Trappe and co-workers [11,12,81] | Human (Old healthy) | Resistance-exercise training 3 days/week (3 sets of 10 repetitions at 75% of 1 RM/day) for 12 week | Acetaminophen (4000 mg/day) or ibuprofen (1200 mg/day) | COX inhibitors enhance muscle mass and strength gains of 25%–50% over placebo: Reduced the PGE2-IL-6-MuRF-1 levels in skeletal muscle. Upregulated PGF2α receptor and its protein synthesis effect |
| Trappe and co-workers [82] | Human (Old healthy) | 12 weeks of knee-extensor resistance exercise (3 days/week) | Acetaminophen (4000 mg/day) | COX inhibitor enhances myocellular growth, and this effect is more pronounced in Type I muscle fibers |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gomez-Cabrera, M.C.; Viña, J.; Ji, L.L. Role of Redox Signaling and Inflammation in Skeletal Muscle Adaptations to Training. Antioxidants 2016, 5, 48. https://doi.org/10.3390/antiox5040048
Gomez-Cabrera MC, Viña J, Ji LL. Role of Redox Signaling and Inflammation in Skeletal Muscle Adaptations to Training. Antioxidants. 2016; 5(4):48. https://doi.org/10.3390/antiox5040048
Chicago/Turabian StyleGomez-Cabrera, Maria Carmen, Jose Viña, and Li Li Ji. 2016. "Role of Redox Signaling and Inflammation in Skeletal Muscle Adaptations to Training" Antioxidants 5, no. 4: 48. https://doi.org/10.3390/antiox5040048
APA StyleGomez-Cabrera, M. C., Viña, J., & Ji, L. L. (2016). Role of Redox Signaling and Inflammation in Skeletal Muscle Adaptations to Training. Antioxidants, 5(4), 48. https://doi.org/10.3390/antiox5040048







