Evaluation of Antioxidant Activity, Total Flavonoids, Tannins and Phenolic Compounds in Psychotria Leaf Extracts
Abstract
:1. Introduction
2. Experimental Section
2.1. General Information
2.2. Plant Material
2.3. Preparation of Extracts
2.4. Measurement of Antioxidant Activity
2.4.1. β-Carotene/Linoleic Acid Method
2.4.2. Scavenging of 1,1-Diphenyl-2-Picrylhydrazyl (DPPH)
2.4.3. ABTS+ Scavenging Activity
2.5. Concentrations of Constituents
2.5.1. Total Phenol Concentration
2.5.2. Total Flavonoid Concentration
2.5.3. Condensed Tannin Concentration (CT)
2.5.4. Flavonol Concentration
2.6. HPLC/PDA Analysis
3. Results and Discussion
3.1. Antioxidant Activity
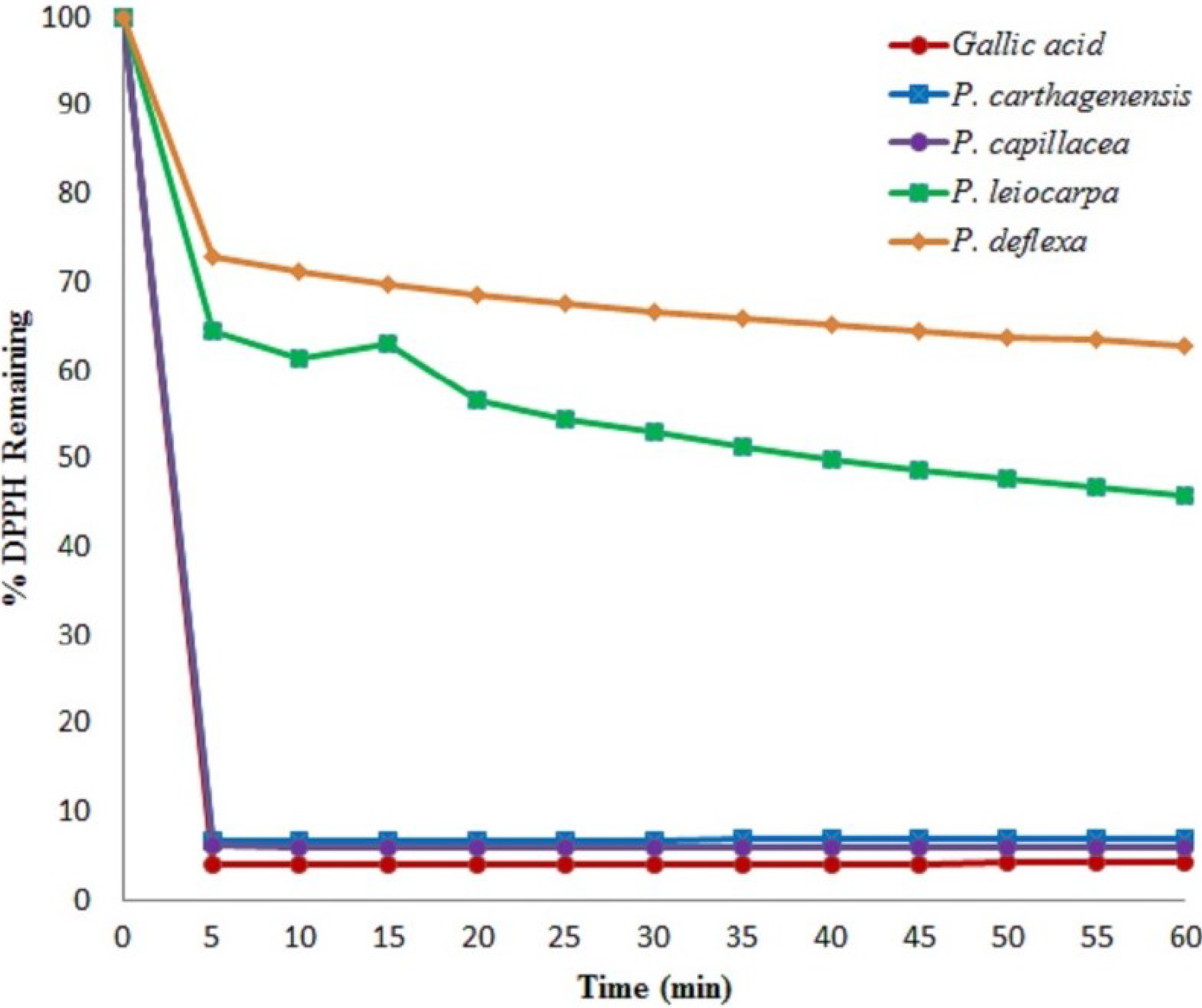
| Test | |||||
|---|---|---|---|---|---|
| DPPH | β-Carotene/Linoleic Acid | ABTS+ | |||
| IC50 (μg/mL) (Limit Confidence 95%) | %FRS * | (%AA) | (%) | ||
| Extracts | P. carthagenensis | 16.92 ± 4.58 | 93.52 ± 8.41 | 79.12 ± 3.70 | 92.5 ± 7.43 |
| (12.03–22.71) | |||||
| P. leiocarpa | 127.00 ± 10.55 | 54.13 ± 11.10 | 22.30 ± 7.14 | 12.20 ± 4.44 | |
| (123.50–135.94) | |||||
| P. capillacea | 30.05 ± 6.22 | 91.78 ± 4.23 | 33.40 ± 15.22 | 87.34 ± 8.32 | |
| (26.68–37.27) | |||||
| P. deflexa | 146.40 ± 12.47 | 66.37 ± 8.12 | 26.05 ± 10.60 | 15.58 ± 5.22 | |
| (141.81–148.42) | |||||
| Standards | BHT | 16.72 ± 1.34 | 92.19 ± 1.29 | 91.20 ± 4.54 | 96.4 ± 2.44 |
| (14.08–17.22) | |||||
| Ascorbic acid | 22.28 ± 0.53 | 96.40 ± 0.27 | 4.13 ± 1.42 | 80.9 ± 5.56 | |
| (20.20–23.43) | |||||
| Quercetin | n.d. | n.d. | 80.65 ± 1.25 | n.d. | |
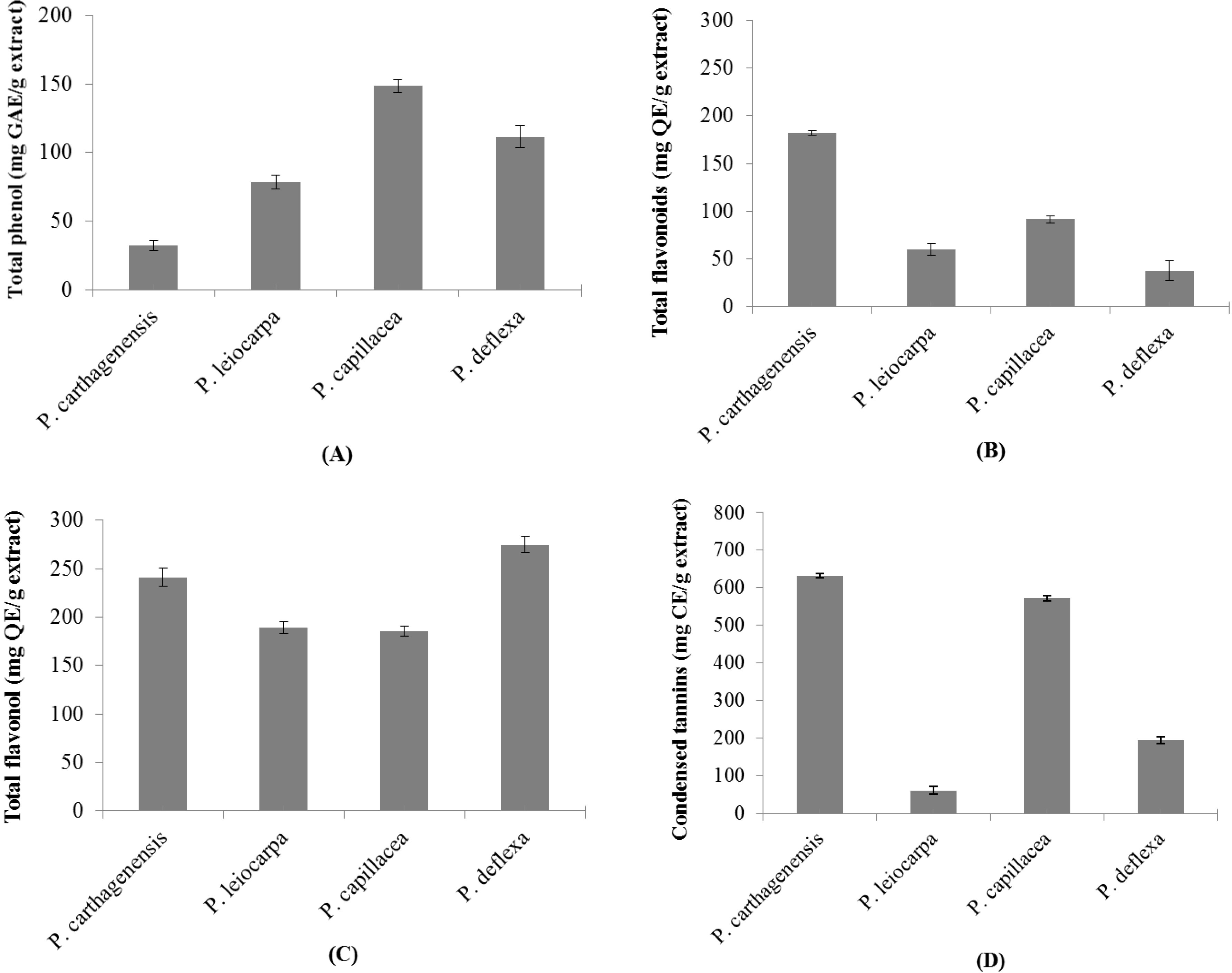
3.2. Levels of Constituents
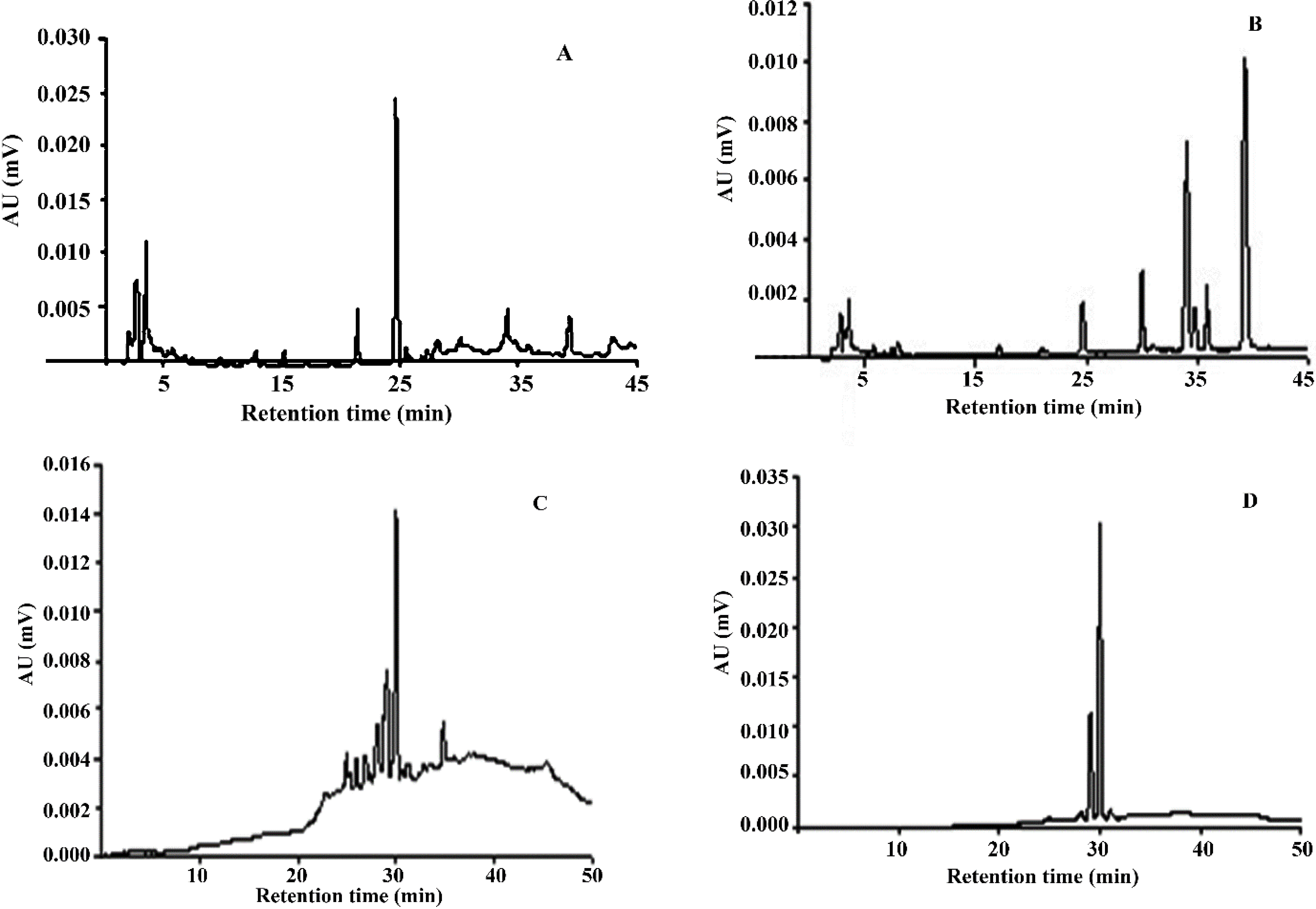
3.3. HPLC/PAD Analysis from Psychotria Extracts
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lindley, M. The impact of food processing on antioxidants in vegetable oils, fruits and vegetables. Trends Food Sci. Technol. 1998, 9, 336–340. [Google Scholar] [CrossRef]
- Middleton, E.; Kandaswamy, C.; Theoharides, T.C. The effects of plant flavonoids on mammalian cells: Implications for inflammation, heart disease, and cancer. Pharmacol. Rev. 2000, 52, 673–751. [Google Scholar] [PubMed]
- Salah, N.; Miller, N.J.; Paganga, G.; Tijburg, L.; Rice-Evans, C.A. Polyphenolic flavonols as scavengers of aqueous phase radicals and as chain-breaking antioxidants. Arch. Biochem. Biophys. 1995, 322, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, T.P.; Robinson, D.; Allaway, S.; Syms, J.; Hale, A. Effect of red wine ingestion on the antioxidant capacity of serum. Clin. Chem. 1995, 41, 32–35. [Google Scholar] [PubMed]
- Perry, L.M.; Metzger, J. Medicinal Plants of East and Southeast; MIT Press: Cambridge, UK, 1980. [Google Scholar]
- Lajis, N.H.; Mahmud, Z.; Toia, R.F. The alkaloids of Psychotria rostrata. Planta Med. 1993, 59, 383–384. [Google Scholar] [CrossRef] [PubMed]
- Hart, N.K.; Johns, S.R.; Lamberton, J.A.; Summons, R.E. Psychotridine, a C55H62N10 alkaloid from Psychotria beccarioides (Rubiaceae). Austrian J. Chem. 1974, 27, 639–646. [Google Scholar] [CrossRef]
- Roth, A.; Kuballa, B.; Bounthanh, C.; Cabalion, P.; Sévenet, T.; Beck, J.P.; Anton, R. Cytotoxic activity of polyindoline alkaloids of Psychotria fosteriana (Rubiaceae). Planta Med. 1986, 5, 450–453. [Google Scholar]
- Libot, F.; Miet, C.; Kunesch, N.; Poisson, J.E.; Pusset, J.; Sévenet, T. Rubiacées d’océanie: Alcaloïdes de Psychotria oleoides de Nouvelle-Calédonie et de Calycodendron milnei du Vanuatu (Nouvelle-Hébrides). J. Nat. Prod. 1987, 50, 468–473. [Google Scholar] [CrossRef]
- Adjibade, Y.; Weniger, B.; Quirion, J.C.; Kuballa, B.; Cabalion, P.; Anton, R. Dimeric alkaloids from Psychotria fosteriana. Phytochemistry 1992, 31, 317–319. [Google Scholar] [CrossRef]
- Verotta, L.; Pilati, T.; Tato, M.; Elisabetsky, E.; Amador, T.A.; Nunes, D.S. Pyrrolidinoindoline alkaloids from Psychotria colorata. J. Nat. Prod. 1998, 61, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Jannic, V.; Gueritte, F.; Laprevote, O.; Serani, L.; Martin, M.T.; Sevenet, T.; Potier, P. Pyrrolidinoindoline alkaloids from Psychotria oleoides and Psychotria lyciiflora. J. Nat. Prod. 1999, 62, 838–843. [Google Scholar] [CrossRef] [PubMed]
- Takayama, M.; Hamano, H.; Ochi, Y.; Seaegusa, H.; Komatsu, K.; Murati, T.; Arakura, N.; Imai, Y.; Hasebe, O.; Kawa, S.; et al. Recurrent attacks of autoimune pancreatites results in pancreatic stone formation. Am. J. Gastrienterol. 2004, 99, 932–937. [Google Scholar] [CrossRef]
- Itoh, A.; Tanahashi, T.; Nagakura, N. Six tetrahydroisoquinoline-monoterpene glucosides from Cephaelis ipecacuanha. Phytochemistry 1991, 30, 3117–3123. [Google Scholar] [CrossRef]
- Itoh, A.; Tanahashi, T.; Nagakura, N.; Nayeshiro, H. Tetrahydroisoquinoline-monoterpene glucosides from Alangium lamarckii and Cephaelis ipecacuanha. Phytochemistry 1994, 36, 383–387. [Google Scholar] [CrossRef]
- Itoh, A.; Ikuta, Y.; Baba, Y.; Tanahashi, T.; Nagakura, N. Ipecac alkaloids from Cephaelis acuminata. Phytochemistry 1999, 52, 1169–1176. [Google Scholar] [CrossRef] [PubMed]
- Nagakura, N.; Itoh, A.; Tanahashi, T. Four tetrahydroisoquinoline-monoterpene glucosides from Cephaelis ipecacuanha. Phytochemistry 1993, 32, 761–765. [Google Scholar] [CrossRef]
- Solis, P.; Ravelo, A.G.; Palenzuela, J.A.; Gupta, M.P.; Gonzalez, A.; Phillipson, J.D. Quinoline alkaloids from Psychotria glomerata. Phytochemistry 1997, 44, 963–969. [Google Scholar] [CrossRef]
- Muhammad, I.; Dunbar, C.D.; Shabana, I.K.; Tekwani, B.L.; Bedir, E.; Takamatsu, S.; Ferreira, D.; Walker, L.A. Antiparasitic alkaloids from Psychotria klugii. J. Nat. Prod. 2003, 66, 962–967. [Google Scholar] [CrossRef] [PubMed]
- Achenbach, H.; Lottes, M.; Waibel, R.; Karias, G.A.; Correa, M.D.; Gupta, M.P. Alkaloids and other compounds from Psychotria correae. Phytochemistry 1995, 38, 1537–1545. [Google Scholar] [CrossRef]
- Kerber, V.A.; Gregianni, T.S.; Paranhos, J.S.; Schwanbach, J.; Farias, F.; Fett, J.P.; Fett-Neto, A.G.; Zuanazzi, J.A.; Quirion, J.C.; Elisabetsky, E.; et al. Brachycerine, a novel monoterpene indole alkaloid from Psychotria brachyceras. J. Nat. Prod. 2001, 64, 677–679. [Google Scholar] [CrossRef] [PubMed]
- Santos, L.V.; Fett-Neto, A.G.; Kerber, V.A.; Elisabetsky, E.; Quirion, J.C.; Henriques, A.T. Indole monoterpene alkaloids from leaves of Psychotria suterella Müll. Arg. (Rubiaceae). Biochem. Syst. Ecol. 2001, 29, 1185–1187. [Google Scholar] [CrossRef]
- Paul, J.H.A.; Maxwell, A.R.; Reynolds, W.F. Novel bis(monoterpenoid) indole alkaloids from Psychotria bahiensis. J. Nat. Prod. 2003, 66, 752–754. [Google Scholar] [CrossRef] [PubMed]
- Henriques, A.T.; Lopes, S.O.; Paranhos, J.T.; Gregianini, T.S.; von Poser, G.L.; Fett-Neto, A.G.; Schripsema, J. N,β-d-Glucopyranosyl vincosamide, a light regulated indole alkaloid from the shoots of Psychotria leiocarpa. Phytochemistry 2004, 65, 449–454. [Google Scholar] [CrossRef] [PubMed]
- Simoes-Pires, C.A.; Farias, F.N.; Marston, A.; Queiroz, E.F.; Chaves, C.G.; Henriques, A.T.; Hostettman, K. Indole monoterpenes with antichemotactic activity from Psychotria myriantha: Chemotaxonomic significance. Nat. Prod. Commun. 2006, 1, 1101–1106. [Google Scholar]
- Nomura, T.; Quesada, A.L.; Kutchan, T.M. The New β-d-glucosidade in terpenoide isoquinoline alkaloid biosynthesis in Psychotria ipecacuanha. J. Biol. Chem. 2008, 283, 34650–34659. [Google Scholar] [CrossRef] [PubMed]
- Beretz, A.; Roth-Georger, A.; Corre, G.; Kuballa, B.; Anton, R.; Cazenale, J.P. Polyindolinic alkaloids from Psychotria fosteriana. Potent inhibitors of the aggregation of human platelets. Planta Med. 1985, 4, 300–303. [Google Scholar] [PubMed]
- Leal, M.B.; Elisabetsky, E. Opioid-like activity of Psychotria brachypoda. Int. J. Pharmacogn. 1996, 34, 267–272. [Google Scholar] [CrossRef]
- Lopes, S.O. Análise Química e Cultivo in Vitro de Psychotria leiocarpa Cham. et Schlecht. e Psychotria carthagenensis Jacq. (Rubiaceae). Master’s Thesis, Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil, 1998. [Google Scholar]
- Lopes, S.; Poser, G.L.; Kerber, V.A.; Farias, F.M.; Konrath, E.L.; Moreno, P.; Sobral, M.E.; Zuanazzi, J.A.S.; Henriques, A.T. Taxonomic significance of alkaloids and iridoid glucosides in the tribe Psychotrieae (Rubiaceae). Biochem. Syst. Ecol. 2004, 32, 1187–1195. [Google Scholar] [CrossRef]
- Dillenburg, C.R.; Porto, M.L. Rubiaceae-tribo Psychotriae. Boletim Instituto de Biociências—UFRGS 1985, 39, 1–76. [Google Scholar]
- Rivier, L.; Lindgren, J.E. “Ayahuasca”, the South American hallucinogenic drink: An ethnobotanical and chemical investigation. Econ. Bot. 1972, 26, 101–129. [Google Scholar] [CrossRef]
- McKenna, D.J.; Towers, G.N.T.; Abbot, F. Monoamine oxidase inhibitors in South American hallucinogenic plants: Trypatamine and β-carboline constituents of Ayahusca. J. Ethnopharmacol. 1984, 10, 195–223. [Google Scholar] [CrossRef] [PubMed]
- Adedapo, A.A.; Jimoh, F.O.; Koduru, S.; Afolayan, A.J.; Masika, P.J. Antibacterial and antioxidant properties of the methanol extracts of the leaves and stems of Calpurnia aurea. BMC Complement. Altern. Med. 2008, 53, 1–8. [Google Scholar]
- Blois, M.S. Antioxidant determinations by the use of a stable free radical. Nature 1958, 181, 1199–1200. [Google Scholar] [CrossRef]
- Kaur, C.; Kapoor, H.C. Antioxidant activity and total phenolic content of some Asian vegetables. Int. J. Food Sci. Technol. 2002, 37, 153–161. [Google Scholar] [CrossRef]
- Shahidi, F.; Chavan, U.D.; Naczk, M.; Amarowicz, R. Nutrient distribution and phenolic antioxidants in air-classified fractions of beach pea (Lathyrus maritimus L.). J. Agric. Food Chem. 2001, 49, 926–933. [Google Scholar] [CrossRef] [PubMed]
- Rufino, M.S.M.; Alves, R.E.; Brito, E.S.; Morais, S.M.; Sampaio, C.G.; Pérez-Jiménez, J.; Saura-Calixto, F.D. Determinação da atividade antioxidante total em frutas pela captura do radical livre ABTS. Embrapa Agroindústria Tropical/Comunicado Técnico 2007, 128, 1–4. [Google Scholar]
- Harborne, J.B.; Williams, C.A. Advances in flavonoid research since 1992. Phytochemistry 2000, 55, 481–504. [Google Scholar] [CrossRef] [PubMed]
- Djeridane, A.; Yousfi, M.; Nadjemi, B.; Boutassouna, D.; Stocker, P.; Vidal, N. Antioxidant activity of some algerian medicinal plants extracts containing phenolic compounds. Food Chem. 2006, 97, 654–660. [Google Scholar] [CrossRef]
- Maxson, E.D.; Rooney, L.W. Evaluation of methods for tannin analysis in sorghum grain. Cereal Chem. 1972, 49, 719–729. [Google Scholar]
- Lin, J.Y.; Tang, C.Y. Determination of total phenolic and flavonoid contents in selected fruits and vegetables, as well as their stimulatory effects on mouse splenocyte proliferation. Food Chem. 2007, 101, 140–147. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Benset, C. Use of free radical method to evaluate antioxidant activity. Lebensm. Wiss. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Naczk, M.; Shahidi, F. Extraction and analysis of phenolics in food. J. Chromatogr. 2004, 1054, 95–111. [Google Scholar] [CrossRef]
- Hall, C.A.; Cuppett, S.L. Structure-activities of natural antioxidants. In Antioxidant Methodology: in Vivo and in Vitro Concepts; Aruoma, O.I., Cuppett, S.L., Eds.; AOCS Press: Champaign, IL, USA, 1997; pp. 141–172. [Google Scholar]
- Robbins, R.J. Phenolic acids in foods: An overview of analytical methodology. J. Agric. Food Chem. 2003, 51, 2866–2887. [Google Scholar] [CrossRef] [PubMed]
- Torres, J.L.; Rosazza, J.P.N. Microbial transformations of p-coumaric acid by Bacillus megaterium and Curvularia lunata. J. Nat. Prod. 2001, 64, 1408–1414. [Google Scholar] [CrossRef] [PubMed]
- Jayaprakasha, G.K.; Singh, R.P.; Sakariah, K.K. Antioxidant activity of grape seed (Vitis vinifera) extracts on peroxidation models in vitro. Food Chem. 2001, 73, 285–290. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Formagio, A.S.N.; Volobuff, C.R.F.; Santiago, M.; Cardoso, C.A.L.; Vieira, M.D.C.; Valdevina Pereira, Z. Evaluation of Antioxidant Activity, Total Flavonoids, Tannins and Phenolic Compounds in Psychotria Leaf Extracts. Antioxidants 2014, 3, 745-757. https://doi.org/10.3390/antiox3040745
Formagio ASN, Volobuff CRF, Santiago M, Cardoso CAL, Vieira MDC, Valdevina Pereira Z. Evaluation of Antioxidant Activity, Total Flavonoids, Tannins and Phenolic Compounds in Psychotria Leaf Extracts. Antioxidants. 2014; 3(4):745-757. https://doi.org/10.3390/antiox3040745
Chicago/Turabian StyleFormagio, Anelise Samara Nazari, Carla Roberta Ferreira Volobuff, Matheus Santiago, Claudia Andrea Lima Cardoso, Maria Do Carmo Vieira, and Zefa Valdevina Pereira. 2014. "Evaluation of Antioxidant Activity, Total Flavonoids, Tannins and Phenolic Compounds in Psychotria Leaf Extracts" Antioxidants 3, no. 4: 745-757. https://doi.org/10.3390/antiox3040745
APA StyleFormagio, A. S. N., Volobuff, C. R. F., Santiago, M., Cardoso, C. A. L., Vieira, M. D. C., & Valdevina Pereira, Z. (2014). Evaluation of Antioxidant Activity, Total Flavonoids, Tannins and Phenolic Compounds in Psychotria Leaf Extracts. Antioxidants, 3(4), 745-757. https://doi.org/10.3390/antiox3040745





