1. Introduction
Excessive generation of reactive oxygen species (ROS) during cellular stress or injury can trigger inflammation and damage to cells and tissues. Thus, controlling ROS and the associated inflammatory response is crucial for maintaining homeostasis, and recent research has focused on natural functional foods with antioxidant and anti-inflammatory capabilities [
1,
2,
3,
4].
Quercus Fructus (QF), the fruit of commonly encountered oak tree species in Korea, exhibits diverse varieties, such as
Quercus serrata,
Quercus dentata, and
Quercus acutissima. Although it has been used as a common culinary ingredient in Korea for centuries, it is one of the most promising natural resources due to its nutritional and medicinal properties [
5,
6,
7]. QF consists of an outer shell, an inner shell, and a fruit positioned between them. QF is known for its astringent taste, attributed to plant-derived polyphenolic compounds such as gallic acid, ellagic acid, and gallotannin [
8,
9,
10]. These compounds have demonstrated antioxidant and anti-inflammatory effects [
11,
12,
13].
One underexplored aspect of QF utilization is the role of the inner shell. Recent studies reported that QF powder retains higher total polyphenol and flavonoid contents when the inner shell is included versus when only the fruit is used [
7,
14]. This suggests that the often-discarded inner shell may significantly contribute to the bioactive compound profile [
5,
15]. However, it remains unclear how inner shell inclusion influences the functional antioxidant efficacy of QF extracts, especially under different extraction conditions. Limited research has addressed the combined impact of plant tissue composition and extraction method on antioxidant outcomes in biologically relevant systems. Additionally, the choice of extraction method is known to greatly affect the yield and activity of phytochemicals [
16,
17]. Conventional and innovative extraction techniques, including stirring, ultrasonication, boiling, and autoclave extraction, which represent high-temperature and high-pressure conditions, can differentially isolate antioxidant compounds. To date, few studies have systematically examined how varying extraction processes modulate the antioxidant capacity of QF extracts, particularly in conjunction with inner shell inclusion. Furthermore, separating the inner shell from the fruit is labor-intensive in practice; if its inclusion proves beneficial, it could streamline processing and improve functional outcomes.
In this study, we addressed these gaps by systematically evaluating four different extraction methods—stirring extraction and ultrasonication extraction with 80% ethanol, boiling extraction and autoclave extraction with water—applied to QF powder either with the inner shell (both fruit and inner shell) or without (fruit only) in terms of antioxidant activity and its biological effects in vitro and in vivo. This multifaceted approach enabled us to determine an optimal extraction strategy and, for the first time, demonstrate that specific extraction conditions translate into superior antioxidant efficacy in living systems.
2. Materials and Methods
2.1. Extraction Preparation and Reagents
QF samples were collected on 5 October 2022, from the university forest of Chungnam National University (Daejeon, Republic of Korea), and fully matured fruits were selected. QF was divided into two groups: one with all shells removed, called fruit (F), and the other one only the outer shell removed, called inner shell + fruit (I). They were dried in a 55 °C food dryer for 3 h, followed by pulverization using a high-speed grinder in 30 cycles of 5 s each.
For ethanol extraction, 5 g of QF powder and 50 mL of 80% ethanol were mixed and stirred at 200 rpm for 30 min using a plate stirrer (HS1-A, LABTron, Seoul, Republic of Korea) at room temperature. This process was repeated three times to obtain the stirred extract (S). The same procedure was performed using an ultrasonic bath at room temperature and 40 kHz ultrasonication (WUC.A03H, DAIHAN SCIENTIFIC, Wonju, Korea) for 15 min, repeated three times to obtain the ultrasonication extracts (U). The choice of 80% ethanol as the extraction solvent was based on its similar polarity to methanol, while avoiding the latter’s toxicological concerns in food-related studies (Plaskova and Mlcek [
18]). Gallic acid, a representative phenolic compound found in acorns, is known to have the highest solubility in methanol, followed by ethanol (Daneshfar et al. [
19]). Thus, 80% ethanol offered a suitable balance between extraction efficiency and safety [
8,
18,
19].
For water extraction, 5 g of crushed sample was added to 50 mL of distilled water. The boiling extract was prepared by boiling using a hot plate for 30 min (B). The autoclave extract was prepared at 121 °C and 0.135 MPa pressure for 15 min by autoclave (HB-506, Hanbaek Scientific Co., Bucheon, Republic of Korea) (A).
All eight extracts were then centrifuged at 27,000 rpm at 5 °C for 20 min using a centrifuge (Avanti JXN-30, Beckman Coulter, Brea, CA, USA), and the supernatant was collected. The concentration of all extracts was adjusted to 100 mg/mL [
20,
21,
22]. For the in vitro experiments, the supernatant was filtered using a 0.25 µm filter before use. The extraction conditions were chosen based on prior studies. Stirring and ultrasonication conditions were applied according to the method described by Cho et al. [
23], and the boiled water extraction was adopted from Hong et al. [
24]. Autoclave extraction, following a standard sterilization protocol, was used to investigate the effectiveness of high-pressure heat treatment.
Gallic acid, 3,4-dihydroxybenzoic acid (3,4-DHBA), p-coumaric acid, Chlorogenic acid, caffeic acid, syringic acid, quercetin-7-O-glucoside (Q7G), 1,3,5-tri-O-caffeoylquinic acid (1,3,5-TCQA), luteolin, quercetin, apigenin, and kaempferol were purchased from Chengdu Biopurify Phytochemicals Ltd. (Chengdu, China). Catechin, epigallocatechin gallate (EGCG), myricetin-3-O-glucoside, and quercetin-3-O-glucoside were obtained from Extrasynthese SAS (Genay Cedex, France). All standard compounds had a purity of ≥95%. HPLC-grade acetonitrile, methanol, formic acid, Ferulic acid, quercetin, 2,2-diphenyl-1-picrylhydrazyl (DPPH), 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS), potassium persulfate, Folin–Ciocalteu’s reagent, sodium carbonate, sodium phosphate buffer, potassium ferricyanide, trichloroacetic acid, ferric chloride, pyrogallol, HCl, Trolox and 2′,7′-dichlorofluorescein diacetate (DCFH-DA) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Diethylene glycol and NaOH were obtained from Daejung Chemicals & Metals Co. (Siheung, Republic of Korea). Lipopolysaccharide (LPS) was purchased from Invitrogen (San Diego, CA, USA), and hydrogen peroxide (H2O2) was purchased from Samchun Chemicals (Seoul, Republic of Korea).
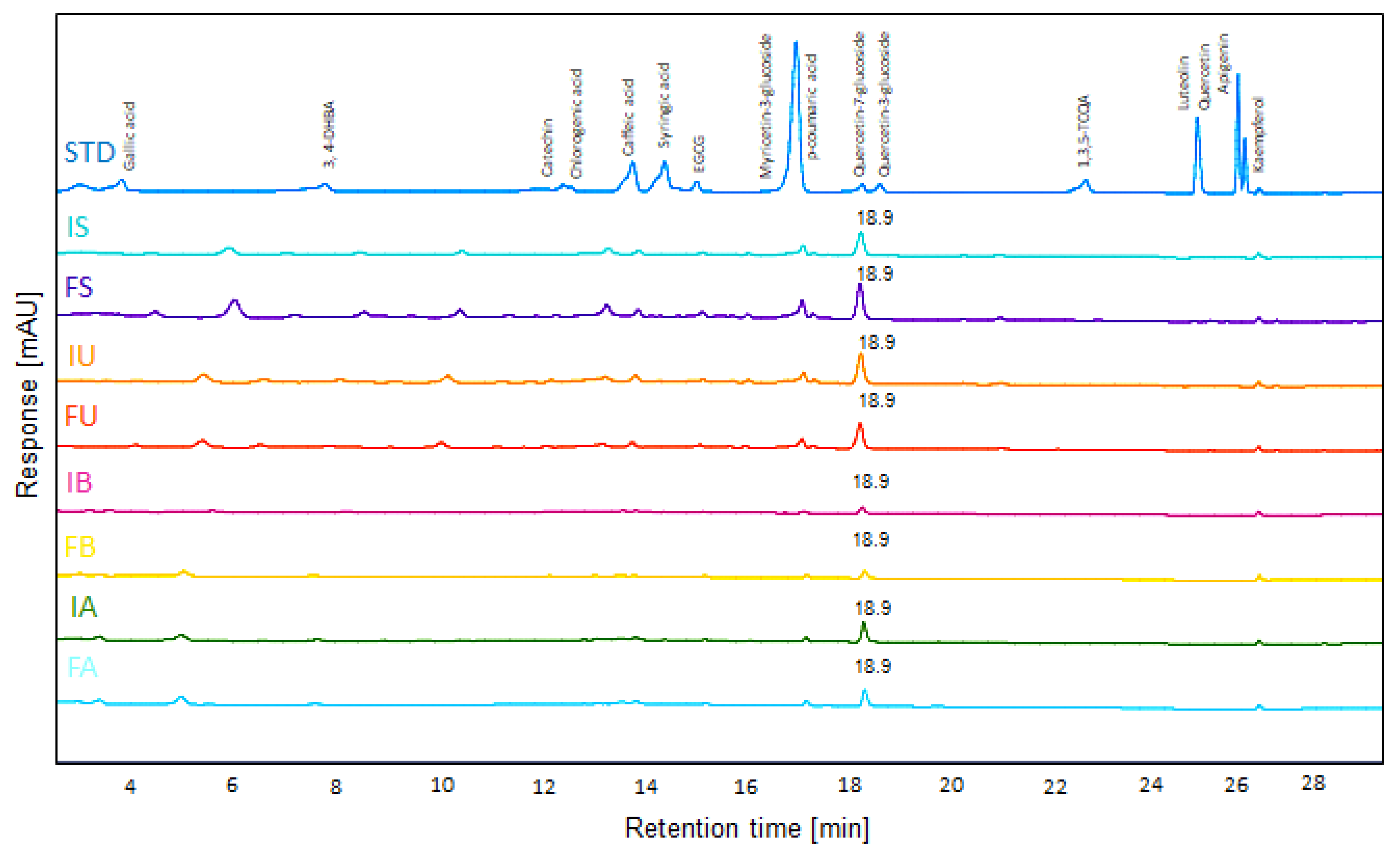
2.2. HPLC-UV Analysis
Detection of polyphenolic compounds was performed at 280 nm. Compound identification was conducted by comparing retention times and UV absorption spectra with those of authentic standards. Quantification was carried out using calibration curves constructed from serial dilutions of standard compounds. A total of 16 flavonoids and phenolic acids were quantified: gallic acid, 3,4-dihydroxybenzoic acid (3,4-DHBA), catechin, chlorogenic acid, caffeic acid, syringic acid, epigallocatechin gallate (EGCG), myricetin-3-glucoside, p-coumaric acid, quercetin-7-glucoside, quercetin-3-glucoside, 1,3,5-Tri-O-caffeoyl quinic acid (1,3,5-TCQA), luteolin, quercetin, apigenin, and kaempferol. Analysis was performed using HPLC coupled with a UV detector (1260 Agilent, Santa Clara, CA, USA), and the separation was performed using an Agilent Zorbax Eclipse Plus C18 column (4.6 × 150 mm, 5-micron) maintained at 35 °C. The mobile phase consisted of solvent A (0.1% (v/v) formic acid in distilled water) and solvent B (0.1% (v/v) formic acid in acetonitrile solution). The flow rate was 0.8 mL/min.
The gradient elution program for flavonoid and phenolic acid separation was set as follows: 0–3 min, 0–5% B; 3–8 min, 5–10% B; 8–22 min, 10–30% B; 22–25 min, 30–70% B; and 25–27 min, 70–100% B. Subsequently, column washing was performed at 100% solvent B for 10 min, followed by re-equilibration with 5% solvent B to prepare for the next sequence.
2.3. Evaluation of Bioactive Compounds and Antioxidant Activity
2.3.1. Determination of Total Polyphenol Content
Total polyphenol content was determined using the Folin–Ciocalteu method [
25,
26]. A 0.1 mL sample was mixed with 10 mL of 2% sodium carbonate solution. After 2 min, 0.1 mL of 50% Folin–Ciocalteu reagent was added. The mixture was allowed to react at room temperature for 30 min. Absorbance was measured at 750 nm using a UV-Vis spectrophotometer. Ferulic acid in 80% ethanol was used as the standard. Results were expressed as µg ferulic acid equivalents (FAE) per mL of extract (µg FAE/mL) [
27,
28]. All measurements were performed in triplicate, and the results are presented as mean ± standard deviation (SD). A sample blank was used to exclude the absorbance originating from the sample itself.
2.3.2. Determination of Total Flavonoid Content
Total flavonoid content was determined by mixing 0.2 mL of sample with 2 mL of diethylene glycol and 0.2 mL of 1 N NaOH [
29]. The mixture was incubated at 37 °C for 1 h. Absorbance was measured at 420 nm. Quercetin in 80% ethanol was used as the standard. Results were expressed as µg quercetin equivalents (QE) per mL of extract (µg QE/mL). All measurements were performed in triplicate, and the results are presented as mean ± standard deviation (SD). A sample blank was used to exclude the absorbance originating from the sample itself.
2.3.3. 2,2-Diphenyl-1-picrylhydrazyl (DPPH) Radical Scavenging Activity
The antioxidant activity was evaluated using 100 µM DPPH in methanol [
30]. A 0.5 mL sample was mixed with 5 mL of DPPH solution and incubated in the dark for 30 min. Absorbance was measured at 517 nm. Trolox in 80% ethanol was used as the standard. A sample blank was used to exclude the absorbance originating from the sample itself.
2.3.4. 2,2′-Azino-bis (3-Ethylbenzothiazoline-6-Sulfonic Acid) (ABTS) Radical Scavenging Activity
An ABTS radical cation was generated by reacting 7 mM ABTS with 140 mM potassium persulfate in distilled water and allowing the mixture to stand in the dark for 12–16 h [
31]. The solution was diluted with ethanol to obtain an absorbance of 0.7 ± 0.002 at 734 nm. For the assay, 150 µL of the sample was mixed with 3 mL of ABTS solution and allowed to react for 2.5 min [
31,
32]. Absorbance was measured at 734 nm. Trolox in 80% ethanol was used as the standard. A sample blank was used to exclude the absorbance originating from the sample itself.
2.3.5. Reducing Power Assay
The reducing power was determined by mixing 100 µL of sample with 500 µL of 0.2 M sodium phosphate buffer (pH 6.6) and 50 µL of 1% potassium ferricyanide [
33]. The mixture was incubated at 50 °C for 20 min, followed by the addition of 2.5 mL of 10% trichloroacetic acid. After centrifugation at 1000×
g for 10 min, 500 µL of the supernatant was mixed with 500 µL of distilled water and 100 µL of 1% ferric chloride. Absorbance was measured at 700 nm. Trolox in 80% ethanol was used as the standard. A sample blank was used to exclude the absorbance originating from the sample itself.
2.3.6. Superoxide Dismutase (SOD)-like Activity
To assess SOD-like activity, 100 µL of sample was mixed with 1.5 mL of 50 mM Tris-HCl buffer (containing 10 mM EDTA, pH 8.0) and 0.1 mL of 7.2 mM pyrogallol [
34]. The mixture was incubated at 25 °C for 10 min. The reaction was stopped by adding 0.5 mL of 0.1 N HCl, and the absorbance was measured at 420 nm. A sample blank was used to exclude the absorbance originating from the sample itself.
2.4. Cell Culture, Viability Assay, and Imaging Assays
Mouse macrophage cells (Raw 264.7) were purchased from the American Type Culture Collection (Manassas, VA, USA). The cells were cultured in Dulbecco’s Modified Eagle’s medium (DMEM; Thermo Fisher Scientific, Waltham, MA, USA) with 1% Penicillin-Streptomycin (Sigma-Aldrich Co., Saint louis, MO, USA) and 10% heat-inactivated fetal bovine serum (FBS) (Thermo Fisher Scientific) at 37 °C under an atmosphere containing 5% CO2.
For the cell viability assay, Raw 264.7 cells were seeded in 96-well plates and treated with various concentrations of QF extracts (12.5, 25, 50, 100, 200, 400, 800 µg/mL) for 48 h. Viability was examined using an EZ-Cytox WST assay kit according to the manufacturer’s instructions (Daeil Lab Service Co., Ltd., Seoul, Republic of Korea). Absorbance was measured at 450 nm using a Microplate Spectrophotometer (xMark™, Bio-Rad, Hercules, CA, USA). The cell viability rate was calculated based on the untreated control group.
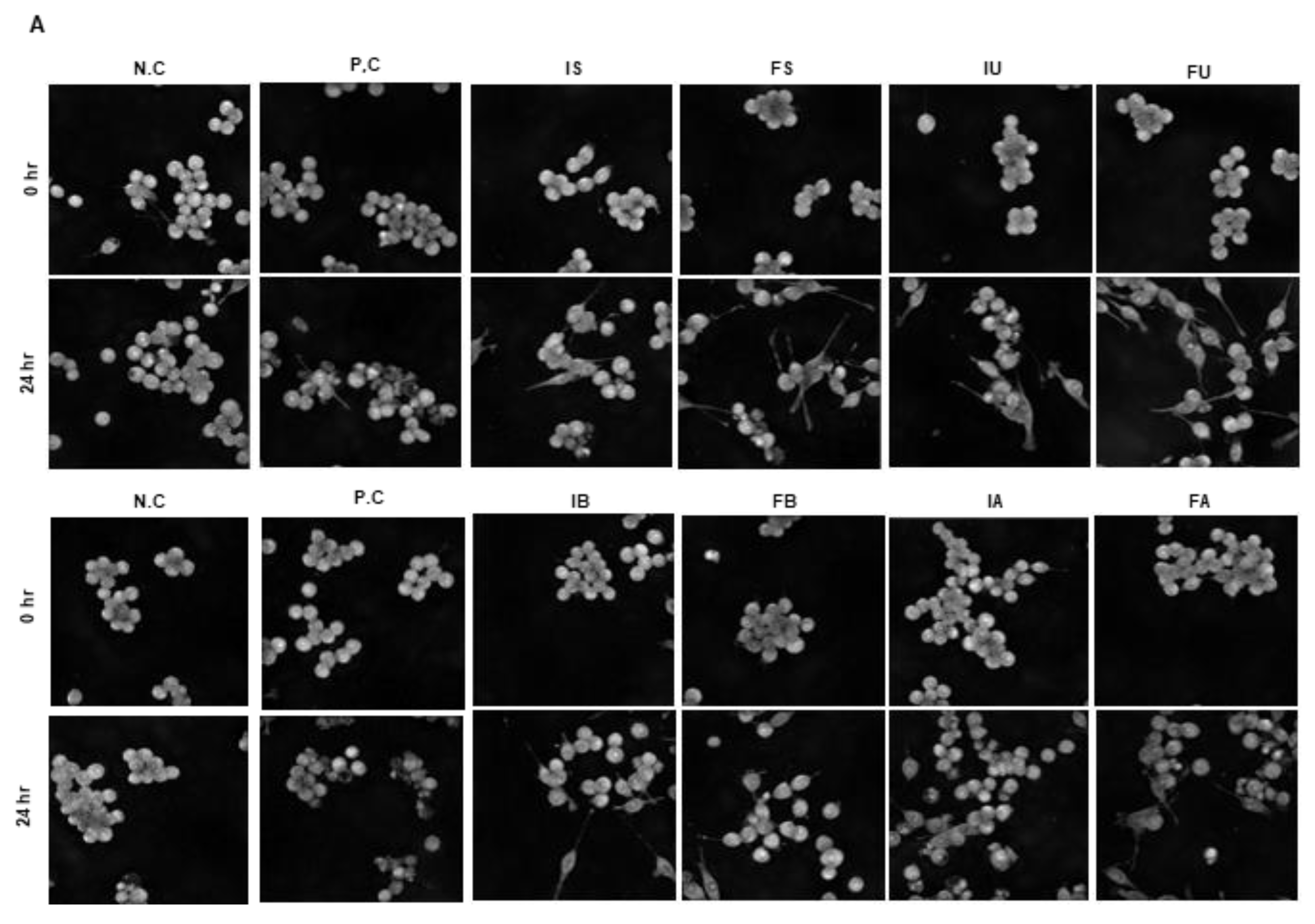
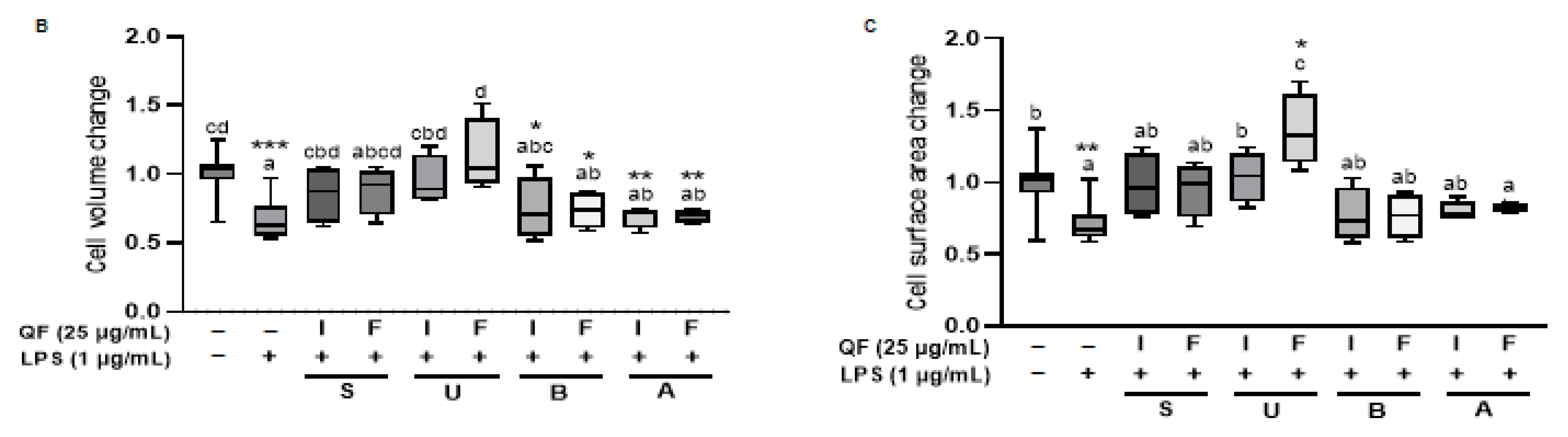
For live cell imaging, Raw 264.7 cells were seeded in glass-bottom 6-well plates (P06-1.5H-N) and treated with QF extracts (25 μg/mL) for 48 h. After 24 h, LPS (1 μg/mL) was added, followed by an additional 24-h incubation. Time-lapse holotomography (HT) imaging was performed for 24 h using the HT-X1™ system (Tomocube Inc., Daejeon, Republic of Korea), generating four-dimensional (3D + time) datasets. The acquired 4D HT data were analyzed using the ‘(3D) single cell analysis’ pipeline in Tomoanalysis™ 2.0 software (Tomocube Inc.), which quantifies cell volume (μm3) and surface area (μm2). The analysis procedure was performed with reference to the TomoAnalysis™ 2.1 User Manual provided by the manufacturer. Measurements from the 24 h timepoint were compared to baseline (0 h) to calculate fold changes. Data were normalized to the mean of the negative control group, which was set to 1.
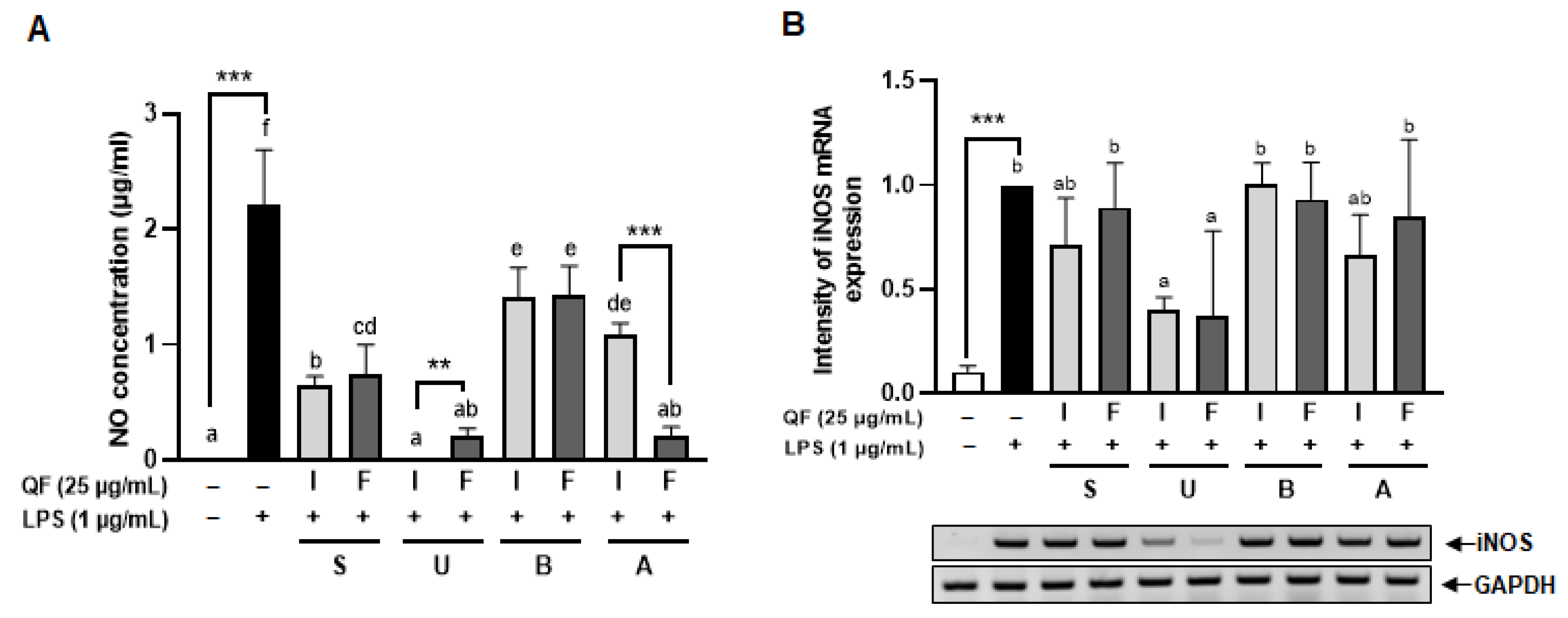
2.5. Nitric Oxide (NO) Assay
The amount of nitrite produced by cytokines in the culture medium was measured using Griess reagent (Promega Co., Madison, WI, USA) according to the manufacturer’s instructions using an xMark™ Microplate Spectrophotometer. The amounts of NO produced were calculated using a NO reference standard curve.
2.6. RNA Extraction & Reverse Transcription Polymerase Chain Reaction (RT-PCR)
Raw 264.7 cells were seeded in 6-well plates and treated with various concentrations of QF extracts (12.5, 25, 50 µg/mL) for 48 h. After 24 h, 1 µg/mL of LPS was added and the mixture was incubated for an additional 24 h. Cells were then homogenized using TRI reagent (MRC Inc., Cincinnati, OH, USA) for RNA extraction. Chloroform (Junsei Co., Tokyo, Japan) was added, and the homogenate was centrifuged at 12,000× g for 15 min at 4 °C. The supernatant was collected, isopropanol (Duksan Co., Ansan-si, Republic of Korea) was added, and the mixture was centrifuged at 12,000× g for 8 min at 20 °C. The supernatant was removed, and the RNA concentration from the pellet was quantified using a NanoDrop spectrophotometer (Thermo Scientific Inc., Waltham, MA, USA). The cDNA was synthesized using an RT Kit (Biofact Co., Daejeon, Republic of Korea). To quantify the mRNA expression of iNOS, RT-PCR was performed using 2 × Taq Basic PCR Master Mix according to the manufacturer’s instructions (Biofact Co., Daejeon, Republic of Korea) with the following gene-specific primer: iNOS (FW: 5′-AAT GGC AAC ATC AGG TCG GCC ATC ACT-3′, RV: 5′-GCT GTG TGT CAC AGA AGT CTC GAA CTC-3′). The amplification products were loaded onto 1.5% or 2% agarose gel for electrophoresis and observed using a gel documentation system (AE-9000 E-Graph, ATTO Co., Tokyo, Japan) under UV light.
2.7. Zebrafish Embryo Collection & ROS Measurement
Zebrafish (Danio rerio) embryos were supplied by the Zebrafish Center for Disease Modeling (ZCDM, Korea) and maintained in a temperature-controlled room at 28 °C with a 14:10 h day/night cycle. Zebrafish were fed brine shrimp 4 times per day. Experiments were performed following the Animal Research Guidelines at Chungnam National University (202410A-CNU-203).
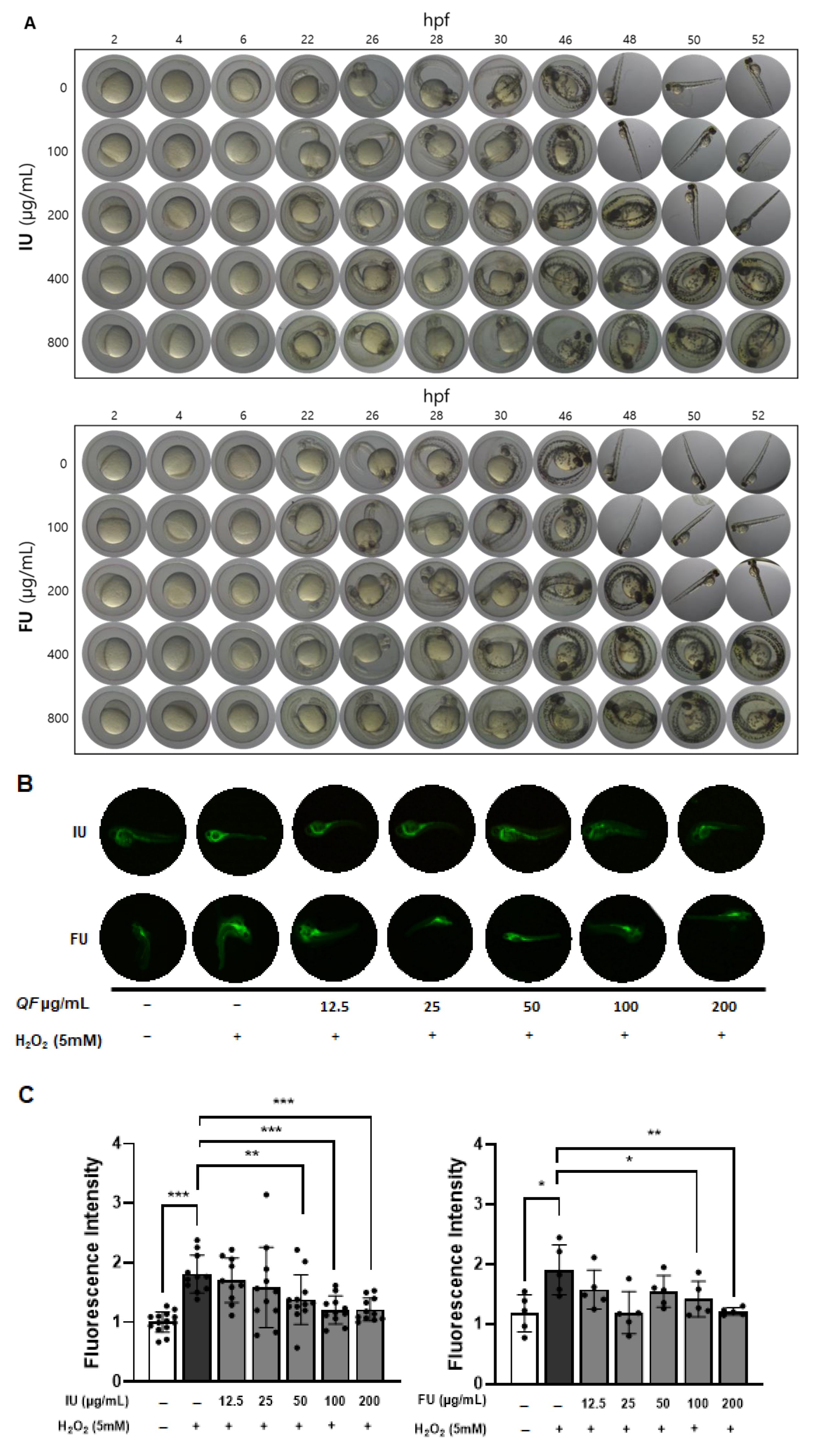
The embryo toxicity test was conducted as a preliminary screening experiment for the subsequent ROS assay [
35,
36]. At 2 to 52 h post fertilization (hpf), zebrafish embryos were isolated in 96-well cell culture plates (Hyundai Micro Co., Seoul, Republic of Korea) at 1 embryo/well in egg water supplemented with 0.1% methylene blue and maintained in a temperature-controlled room at 28 °C. The embryos were then treated with QF (0, 100, 200, 400, 800 µg/mL) for various times as indicated. Embryos (
n = 9 per group) were examined using an optical microscope (TM-10S, Taeshin Bioscience, Gyeonggi-do, Republic of Korea).
For ROS measurement, we revised the present protocol for our experiment setting [
36]. The number of embryos per group for ROS measurement was determined based on statistical analyses from a prior experiment using the IU extract. In this study, 10–12 embryos per group were used, and the antioxidant effect of QF against H
2O
2-induced ROS generation was found to be large to very large (Cohen’s d = 1.252 to 2.867). G*Power 3.1.9.7 analysis indicated that such effect sizes would require a minimum of 5 embryos per group (Cohen’s d = 2.867) to achieve a statistical power of 0.8 (α = 0.05, two-tailed). To minimize unnecessary animal use, the IU experiment was terminated early once sufficient statistical significance was achieved. Based on these results, the subsequent FU experiment was conducted using only 5 embryos per group. All experimental designs were guided by the principles of the 3Rs (Replacement, Reduction, Refinement) to minimize animal use while maintaining statistical validity
Zebrafish embryos at 2 hpf were isolated in 96-well plates at 1 embryo/well in egg water supplemented with 0.1% methylene blue and treated with QF (0, 12.5, 25, 50, 100, 200 µg/mL) for 24 h. Then, 5 mM H2O2 was added, and the mixture was incubated for an additional 24 h to induce ROS. After incubation, the embryos were washed with egg water (n = 5–12 per group). At 48 hpf, the eggs were treated with egg water containing DCFH-DA (20 µg/mL), incubated for 1 h at 28 °C in the dark, and then washed with egg water. Images of stained embryos were observed using a digital microscope (Dino-Lite Digital Microscope, ANMO Electronics Co., New Taipei City, Taiwan) and analyzed by ImageJ 1.54k software.
2.8. Statistical Analysis
Data were analyzed using the SPSS 24.0 (SPSS Inc., Chicago, IL, USA) software. Results are displayed as mean ± standard deviation. Statistical analysis comparing two groups was performed using an independent t-test, and one-way analysis of variance (ANOVA), followed by Duncan’s multiple range tests, which were used to analyze two or more groups. Statistical significance was set at p < 0.05.
4. Discussion
While previous studies have investigated QF’s antioxidant properties [
39,
40,
41], our research provides the first comprehensive comparison of inner shell inclusion across multiple extraction methods, revealing previously unreported functional benefits. The combination of ultrasonication extraction with inner shell inclusion (IU) emerged as the optimal approach, demonstrating superior antioxidant activity through enhanced polyphenol extraction and preservation of bioactive compounds.
Previous research has established that ethanol extraction generally yields higher antioxidant activity than water-based methods for QF [
6,
42,
43], which is consistent with our findings where ethanol extracts (IS: 254.27 μg FAE/mL, IU: 239.66 μg FAE/mL) showed superior total phenolic content and NO inhibition compared to water extracts. Our identification of Q7G as the primary detectable flavonoid, known for its enhanced bioavailability and iNOS inhibitory properties [
44], provides mechanistic insight into the observed antioxidant effects, although the superior biological activity of ultrasonication extracts suggests involvement of additional co-extracted compounds.
Our findings suggest that Q7G may play a crucial role in reducing NO levels and enhancing antioxidant defense, reinforcing its potential as a key bioactive compound in QF extracts. This study underscores the significance of Q7G as a more effective functional ingredient compared to traditional phenolic acids, paving the way for further research into its therapeutic applications.
To ascertain the antioxidant effects of QF, oxidative stress induction was performed using LPS. We observed differences in antioxidant effects depending on the presence of the inner shell and the extraction method, as indicated by NO inhibition. The 25 μg/mL extracts were effective in decreasing iNOS expression. This is in agreement with Youn et al. (2016), whose study showed higher antioxidant activity at 50 μg/mL in
Quercus acutissima Carruth, although the extraction methods and methods of assessing antioxidant activities were different from those of our study [
12]. There might have been variations in the concentration of extracts causing a decrease in iNOS expression due to different extraction methods. The antioxidant activity of plant materials generally originates from their polyphenolic compounds. Several studies have shown a direct relationship between phenolic content and antioxidant activity of
Quercus species [
10]. As expected, the high antioxidant activity in this paper can be attributed to the phenolic content. These results indicate that different extraction methods may yield different mechanisms of action in antioxidant function.
The differential performance across four antioxidant evaluation methods reflects their distinct measurement mechanisms and target radical species. DPPH assays, utilizing nitrogen-centered radicals in organic solvents [
30], showed ethanol extracts (S, U) consistently outperforming other methods, with water boiling (B) showing slightly lower activity and autoclave (A) extracts demonstrating the poorest performance, particularly IA < FA. ABTS assays with water-soluble cationic radicals [
32] revealed that stirring, ultrasonication, and boiling methods (S, U, B) performed similarly well, while autoclave extracts showed lower overall activity but with a reversed pattern (IA > FA) compared to DPPH results. This discrepancy suggests that while autoclave processing generally reduces antioxidant capacity, it generates different antioxidant components in inner shell versus fruit extracts, with the former being more effective against ABTS radicals. The reducing power assay evaluates the electron-donating ability of antioxidants, independent of radical scavenging mechanisms [
45], consistently favoring inner shell-containing extracts across all methods, indicating that tannins and other multi-hydroxyl compounds in the inner shell contribute significantly to the total reducing capacity. SOD-like activity, which assesses the ability of antioxidants to mimic superoxide dismutase by scavenging superoxide anion radicals [
34], showed the most dramatic differences, with ethanol extracts (S, U) substantially outperforming water extracts (B, A), and demonstrating over 3-fold higher activity compared to FA extracts, highlighting the critical role of extraction solvent in preserving superoxide-scavenging compounds.
The superior performance of ethanol extracts (S, U) compared to water extracts (B, A) can be attributed to fundamental differences in solvent polarity and extraction conditions [
46,
47,
48,
49]. Organic solvents such as 80% ethanol possess higher solubility for polyphenols and flavonoid compounds than water, enabling the extraction of a broader spectrum of antioxidant components while maintaining relatively low temperatures that preserve thermolabile compounds. Conversely, water-based extraction methods, particularly autoclave processing under high temperature and pressure, can enhance release of bound phenolic acids through cell wall destruction but simultaneously cause structural degradation of heat-sensitive flavonoid glycosides [
46]. The superiority of ultrasonication over stirring with identical solvent conditions demonstrates the additional benefit of acoustic cavitation in disrupting cellular microstructures and promoting rapid compound release without thermal degradation. These findings demonstrate that comprehensive antioxidant evaluation requires multiple assays to capture the full spectrum of protective mechanisms, while extraction optimization must balance compound release with preservation.
In the antioxidant component analysis using HPLC, only Q7G was clearly detected among the 16 tested phenolic standards, while colorimetric measurements showed significant differences in total phenolic and flavonoid contents between samples. This discrepancy indicates that the complex phytochemical diversity in QF extracts, including numerous unidentified compounds, contributes to the total antioxidant capacity. Q7G was detected at high concentrations in ethanol extracts containing only fruit (FS: 93.6 μg/mL) and IU (79.9 μg/mL). However, the lower Q7G concentration in the IU extract, which exhibited superior biological activity, suggests the possibility of synergistic effects among multiple components. Particularly intriguing was the FA extract’s high total flavonoid content (comparable to FS and IU) despite lower Q7G levels, suggesting that autoclave processing induces glycoside hydrolysis or structural modifications that reduce HPLC-detectable Q7G while creating degradation products reactive with colorimetric reagents. The inner shell’s primary contribution appears to be non-flavonoid polyphenols such as tannins, which explains the consistent enhancement of total phenolic content and reducing power in I-containing extracts. This compositional complexity underscores why extraction optimization cannot rely solely on individual compound quantification but must consider total bioactive profiles and their functional interactions.
The integration of in vitro cell culture and in vivo zebrafish studies provided robust validation of our biochemical findings. Raw 264.7 macrophage experiments revealed that ethanol extracts, particularly IU, effectively inhibited LPS-induced NO production and iNOS expression at 25 μg/mL, while water extracts showed limited or adverse effects. The holotomography analysis offered unique insights into cellular protection mechanisms, showing that extract treatment prevented LPS-induced cell volume reduction and death rather than typical inflammatory activation. The high LPS concentration (1 μg/mL) likely induced rapid cytotoxicity before classical morphological changes could manifest, making cell survival the primary measurable outcome. Zebrafish embryo studies confirmed these protective effects in a complex biological system, with IU extract demonstrating superior ROS suppression at lower concentrations (50 μg/mL) compared to the FU extract (100 μg/mL). This dose-dependent efficacy directly correlated with total phenolic content and Q7G concentrations, validating our biochemical measurements in a physiologically relevant context. The consistency between in vitro and in vivo results strengthens the evidence for bioactive compound stability and functional preservation across biological systems.
This study investigated the effects of the combination of inner shell inclusion and extraction methods on the antioxidant activity of QF from various perspectives, but the following limitations exist. First, in this study, HPLC-UV analysis was performed on a total of 16 standard compounds, and the detection wavelength was set to 280 nm. However, this condition may not be suitable for detecting hydrolysable tannins such as ellagitannins. In fact, several unresolved peaks were observed in the chromatogram of this study, suggesting the possible presence of polar polymeric compounds such as ellagitannins. However, since this study did not include standard materials for these compounds and did not perform LC-MS/MS analysis for structural confirmation, the exact identity of these compounds could not be determined. Therefore, future studies should combine LC-MS/MS-based structural analysis and multi-wavelength detection to clarify the presence of phenolic compounds not identified in the current analysis and their role in the antioxidant mechanism.
Second, although this study confirmed strong antioxidant activity through in vitro and in vivo models, there were limitations in quantitatively separating and analyzing the contributions of individual components such as Q7G and tannins due to the nature of the complex extract. Interactions between various bioactive substances (synergistic effects) and complex physiological activities may occur, which can hinder the clear distinction of the effects of specific individual components. Future studies should clarify the contribution of each component through fractionation extraction or component separation and recombination tests.
Third, in vivo experiments using zebrafish are useful models for confirming systemic biological effects, but they have limitations in terms of physiological similarity to humans. Therefore, follow-up studies using mammalian models such as mice are needed to validate and generalize these results. In addition, as this study adhered to the 3Rs principle (Replacement, Reduction, Refinement) to minimize animal use, the relatively small sample size per group (n) represents a limitation that should be considered when interpreting the data.
The greatest strength of this study is that it did not merely compare solvents but comprehensively analyzed the combination of inner shell inclusion and extraction method as key factors determining the antioxidant efficacy of QF extract. QF are raw materials that exist in a state where the shell and inner shell are mixed, and the process of removing the inner shell in actual industrial settings or processing can be cumbersome and inefficient. Therefore, one of the main objectives of this study was to experimentally clarify the validity of whether it is necessary to remove the inner shell and whether including the inner shell could be advantageous in terms of antioxidant activity.
To achieve this objective, this study applied four different extraction methods and systematically compared antioxidant activity based on whether the inner shell was included or removed. Notably, the highest antioxidant activity was observed under the ultrasonic extraction + inner shell (IU) condition. This finding is significant not only because it goes beyond simple chemical metrics but also because it is supported by biological activity through in vitro and in vivo analyses using cell and zebrafish models.
Additionally, HPLC analysis detected Q7G as one of the potential active components, and the high antioxidant activity observed in the ultrasonic extract, including the peel, suggests the potential contribution of non-flavonoid phenolic compounds with high-molecular-weight polyhydroxy structures, such as tannins, in addition to Q7G.
Although the limitations of the analytical method made it difficult to accurately quantify some high-molecular-weight antioxidant substances such as ellagitannins, this study pointed out their possible existence and suggested future analytical directions, thereby laying the foundation for future research. Furthermore, IU showed consistent efficacy, such as improved cell survival, NO production inhibition, and ROS reduction in zebrafish embryos, supporting actual biological functionality beyond simple antioxidant measurements.