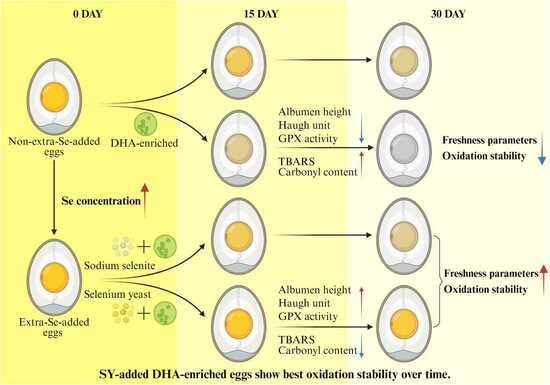
Comparative Effects of Selenium Yeast and Sodium Selenite on the Selenium Distribution, Interior Quality and Oxidative Stability of Docosahexaenoic Acid-Enriched Eggs During Storage
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental Design and Diet
2.3. Blood and Tissue Collection
2.4. Egg Sample Collection and Preparation
2.5. Se Concentration in Feed, Serum, Tissue, and Egg
2.6. Chemical Composition Analysis
2.7. Egg Freshness Quality Traits
2.8. Determination of GPX Activity, Lipid and Protein Oxidation
2.9. Statistical Analysis
3. Results and Discussion
3.1. Actual Se Content of the Experimental Diet
3.2. Selenium Contents in Serum and Tissues
3.3. Selenium Concentration and Total Selenium Amount in Eggs
3.4. Chemical Composition of Eggs
3.5. Oxidative Stability of Eggs During Storage
3.6. Egg Freshness During Storage
3.7. Pearson Correlation Analysis Between Selenium Content and Key Egg Quality Indicators
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Van Dael, P. Role Of N-3 Long-Chain Polyunsaturated Fatty Acids in Human Nutrition and Health: Review of Recent Studies and Recommendations. Nutr. Res. Pract. 2021, 15, 137–159. [Google Scholar] [CrossRef]
- Innes, J.K.; Calder, P.C. Marine Omega-3 (N-3) Fatty Acids for Cardiovascular Health: An Update for 2020. Int. J. Mol. Sci. 2020, 21, 1362. [Google Scholar] [CrossRef]
- Kralik, Z.; Kralik, G.; Grčević, M.; Hanžek, D.; Margeta, P. Microalgae Schizochytrium limacinum as an Alternative to Fish Oil in Enriching Table Eggs with N-3 Polyunsaturated Fatty Acids. J. Sci. Food Agric. 2020, 100, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Macit, M.; Karaoglu, M.; Celebi, S.; Esenbuga, N.; Yoruk, M.A.; Kaya, A. Effects of Supplementation of Dietary Humate, Probiotic, and Their Combination on Performance, Egg Quality, and Yolk Fatty Acid Composition of Laying Hens. Trop. Anim. Health Prod. 2021, 53, 63. [Google Scholar] [CrossRef] [PubMed]
- Panaite, T.D.; Cornescu, G.M.; Predescu, N.C.; Cismileanu, A.; Turcu, R.P.; Saracila, M.; Soica, C. Microalgae (Chlorella vulgaris and Spirulina platensis) as a Protein Alternative and Their Effects on Productive Performances, Blood Parameters, Protein Digestibility, and Nutritional Value of Laying Hens’ Egg. Appl. Sci. 2023, 13, 10451. [Google Scholar] [CrossRef]
- Wang, H.; He, W.; Mahukpégo Dansou, D.; Zhang, H.; Dwi Nugroho, R.; Tang, C.; Guo, X.; Yu, Y.; Zhao, Q.; Qin, Y.; et al. Astaxanthin Improved the Storage Stability of Docosahexaenoic Acid-Enriched Eggs by Inhibiting Oxidation of Non-Esterified Poly-Unsaturated Fatty Acids. Food Chem. 2022, 381, 132256. [Google Scholar] [CrossRef]
- Liu, B.; Zhou, Q.; Zhu, J.; Lin, G.; Yu, D.; Ao, T. Time Course of Nutritional and Functional Property Changes in Egg Yolk from Laying Hens Fed Docosahexaenoic Acid-Rich Microalgae. Poult. Sci. 2020, 99, 4616–4625. [Google Scholar] [CrossRef]
- Li, M.-N.; Yang, L.; Cui, H.-T.; Liao, J.-B.; Wang, N.; Miao, J.; Zhang, Y.; Wang, H.-D. Changes of Mitochondrial Dynamics During the Progression of Non-Alcoholic Fatty Liver Disease: A Review. Biomed. Eng. Commun. 2024, 3, 3. [Google Scholar] [CrossRef]
- Mazalli, M.R.; Bragagnolo, N. Increase of Cholesterol Oxidation and Decrease of PUFA as A Result of Thermal Processing and Storage in Eggs Enriched with N-3 Fatty Acids. J. Agric. Food Chem. 2009, 57, 5028–5034. [Google Scholar] [CrossRef] [PubMed]
- Xue, H.; Wang, J.; Xie, J.; Xiong, C.; Li, J.; Tu, Y.; Zhao, Y. Isolation, Purification, and Structure Identification of Antioxidant Peptides from Embryonated Eggs. Poult. Sci. 2019, 98, 2360–2370. [Google Scholar] [CrossRef]
- Lan, Y.; He, Y.; Chen, X.; Jiang, S.; Wang, Z.; Li, S.; Hui, T.; Li, S.; Fang, Z.; Chen, H. Thermal Processing and in Vitro Digestion of N-3 Pork: Effects on The Oxidative and Digestive Properties of Proteins and Lipids. Food Chem. 2025, 468, 142472. [Google Scholar] [CrossRef]
- Shahid, M.S.; Zhou, S.; Nie, W.; Wang, L.; Lv, H.; Yuan, J. Phytogenic Antioxidants Prolong n-3 Fatty Acid-Enriched Eggs’ Shelf Life by Activating the Nrf-2 Pathway through Phosphorylation of MAPK. Foods 2022, 11, 3158. [Google Scholar] [CrossRef]
- Gou, X.-B.; Xu, Y.-S.; Shao, C.-Y.; Zhao, X.-R.; Zhang, Z.; Shi, Y.-P.; Lei, Y.-J. The Anti-Tumor and Mechanism of the Benzoisoselenazolone Derivatives on the Human Lung Cancer Adenocarcinoma A549 Cells. Biomed. Eng. Commun. 2024, 3, 19. [Google Scholar] [CrossRef]
- Dos Reis, J.H.; Gebert, R.R.; Fortuoso, B.F.; Dos Santos, D.S.; Souza, C.F.; Baldissera, M.D.; Tavernari, F.C.; Boiago, M.M.; Paiano, D.; Da Silva, A.S. Selenomethionine as a Dietary Supplement for Laying Hens: Impacts on Lipid Peroxidation and Antioxidant Capacity in Fresh and Stored Eggs. J. Food Biochem. 2019, 43, e12957. [Google Scholar] [CrossRef]
- Li, H.; Liu, H.; Tang, X.; Deng, Z.; Li, H. From Soil to Table: A Comprehensive Review of Selenium-Fortified Foods. Compr. Rev. Food Sci. Food Saf. 2025, 24, e70250. [Google Scholar] [CrossRef]
- Chantiratikul, A.; Chinrasri, O.; Chantiratikul, P. Effect of Selenium from Selenium-Enriched Kale Sprout Versus Other Selenium Sources on Productivity and Selenium Concentrations in Egg and Tissue of Laying Hens. Biol. Trace Elem. Res. 2018, 182, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, A.I.; Mohamed, D.A.; Chwen, L.T.; Akit, H.; Samsudin, A.A. Effect of Selenium Sources on Laying Performance, Egg Quality Characteristics, Intestinal Morphology, Microbial Population and Digesta Volatile Fatty Acids in Laying Hens. Animals 2021, 11, 1681. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Mu, T.; Li, R.; Miao, S.; Jian, H.; Dong, X.; Zou, X. Effects of Different Selenium Sources and Levels on the Physiological State, Selenoprotein Expression, and Production and Preservation of Selenium-Enriched Eggs in Laying Hens. Poult. Sci. 2024, 103, 103347. [Google Scholar] [CrossRef]
- NY/T33-2004; Feeding Standard of Chicken (NY/T 33-2004). China Agricultural Industry Standards: Beijing, China, 2004.
- National Research Council. Nutrient Requirements of Poultry; National Academies Press: Washington, DC, USA, 1994. [Google Scholar]
- Ge, C.; Luo, X.; Wu, L.; Lv, Y.; Hu, Z.; Yu, D.; Liu, B. Plant Essential Oils Improve Growth Performance by Increasing Antioxidative Capacity, Enhancing Intestinal Barrier Function, and Modulating Gut Microbiota in Muscovy Ducks. Poult. Sci. 2023, 102, 102813. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Wu, L.; Lv, Y.; Ge, C.; Luo, X.; Zhan, S.; Huang, W.; Shen, X.; Yu, D.; Liu, B. Integrated Analysis of Microbiome and Transcriptome Reveals the Mechanisms Underlying the Chlorogenic Acid-Mediated Attenuation tf Oxidative Stress and Systemic Inflammatory Responses Via Gut-Liver Axis in Post-Peaking Laying Hens. J. Anim. Sci. Biotechnol. 2025, 16, 82. [Google Scholar] [CrossRef]
- Liu, B.; Jiang, J.; Lin, G.; Yu, D.; Xiong, Y.L. Upregulation of Antioxidant Enzymes by Organic Mineral Co-Factors to Improve Oxidative Stability and Quality Attributes of Muscle from Laying Hens. Food Res. Int. 2019, 125, 108575. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of AOAC International; AOAC INTERNATIONAL: Washington, DC, USA, 2000. [Google Scholar]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A Simple Method for the Isolation and Purification of Total Lipides from Animal Tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef] [PubMed]
- Miao, S.; Mu, T.; Li, R.; Li, Y.; Zhao, W.; Li, J.; Dong, X.; Zou, X. Coated Sodium Butyrate Ameliorates High-Energy and Low-Protein Diet Induced Hepatic Dysfunction Via Modulating Mitochondrial Dynamics, Autophagy and Apoptosis in Laying Hens. J. Anim. Sci. Biotechnol. 2024, 15, 15. [Google Scholar] [CrossRef] [PubMed]
- Galobart, J.; Barroeta, A.C.; Baucells, M.D.; Guardiola, F. Lipid Oxidation in Fresh and Spray-Dried Eggs Enriched with Omega3 and Omega6 Polyunsaturated Fatty Acids During Storage as Affected by Dietary Vitamin E and Canthaxanthin Supplementation. Poult. Sci. 2001, 80, 327–337. [Google Scholar] [CrossRef]
- Bayarsaikhan, G.; Avan, A.N.; Çekiç, S.D.; Apak, R. Use of Modified CUPRAC and Dinitrophenylhydrazine Colorimetric Methods for Simultaneous Measurement of Oxidative Protein Damage and Antioxidant Defense Against Oxidation. Talanta 2019, 204, 613–625. [Google Scholar] [CrossRef]
- Lv, Y.; Ge, C.; Wu, L.; Hu, Z.; Luo, X.; Huang, W.; Zhan, S.; Shen, X.; Yu, D.; Liu, B. Hepatoprotective Effects of Magnolol in Fatty Liver Hemorrhagic Syndrome Hens Through Shaping Gut Microbiota and Tryptophan Metabolic Profile. J. Anim. Sci. Biotechnol. 2024, 15, 120. [Google Scholar] [CrossRef] [PubMed]
- Bakhshalinejad, R.; Hassanabadi, A.; Swick, R.A. Dietary Sources and Levels of Selenium Supplements Affect Growth Performance, Carcass Yield, Meat Quality and Tissue Selenium Deposition in Broilers. Anim. Nutr. 2019, 5, 256–263. [Google Scholar] [CrossRef]
- Liu, Z.; Cao, Y.; Ai, Y.; Lin, G.; Yin, X.; Wang, L.; Wang, M.; Zhang, B.; Wu, K.; Guo, Y.; et al. Effects of Selenium Yeast on Egg Quality, Plasma Antioxidants, Selenium Deposition and Eggshell Formation in Aged Laying Hens. Animals 2023, 13, 902. [Google Scholar] [CrossRef]
- Payne, R.L.; Southern, L.L. Comparison of Inorganic and Organic Selenium Sources for Broilers. Poult. Sci. 2005, 84, 898–902. [Google Scholar] [CrossRef]
- Ghazi Harsini, S.; Habibiyan, M.; Moeini, M.M.; Abdolmohammadi, A.R. Effects of Dietary Selenium, Vitamin E, And Their Combination on Growth, Serum Metabolites, and Antioxidant Defense System in Skeletal Muscle of Broilers Under Heat Stress. Biol. Trace Elem. Res. 2012, 148, 322–330. [Google Scholar] [CrossRef]
- Meng, T.; Liu, Y.L.; Xie, C.Y.; Zhang, B.; Huang, Y.Q.; Zhang, Y.W.; Yao, Y.; Huang, R.; Wu, X. Effects of Different Selenium Sources on Laying Performance, Egg Selenium Concentration, and Antioxidant Capacity in Laying Hens. Biol. Trace Elem. Res. 2019, 189, 548–555. [Google Scholar] [CrossRef]
- Lazard, M. Selenium Metabolism and Toxicity in the Yeast Saccharomyces Cerevisiae. Med. Res. Arch. 2021, 9. [Google Scholar] [CrossRef]
- Schrauzer, G.N. Selenomethionine: A Review of Its Nutritional Significance, Metabolism and Toxicity. J. Nutr. 2000, 130, 1653–1656. [Google Scholar] [CrossRef]
- Shulhai, A.M.; Rotondo, R.; Petraroli, M.; Patianna, V.; Predieri, B.; Iughetti, L.; Esposito, S.; Street, M.E. The Role of Nutrition on Thyroid Function. Nutrients 2024, 16, 2496. [Google Scholar] [CrossRef] [PubMed]
- Turcu, R.P.; Sărăcilă, M.; Alexandru, P.; Vișinescu, P.; Criste, R.D. Obtaining Eggs Enriched in Polyunsaturated Fatty Acids (PUFA) 2. Clinical Studies, Consumer Perception and Market of the PUFA-Enriched Eggs: A review. Arch. Zootech. 2019, 21, 86–119. [Google Scholar]
- Boukhatem, A.S.; Chahed, A. Experimental Approach to Preserving Eggs Using the Century Egg Method; Ecole Nationale Supérieure Vétérinaire (ENSV) Alger: Bab Ezzouar, Algeria, 2024. [Google Scholar]
- Kocetkovs, V.; Radenkovs, V.; Juhnevica-Radenkova, K.; Jakovlevs, D.; Muizniece-Brasava, S. The Impact of Eggshell Thickness on the Qualitative Characteristics of Stored Eggs Produced by Three Breeds of Laying Hens of the Cage and Cage-Free Housed Systems. Appl. Sci. 2022, 12, 11539. [Google Scholar] [CrossRef]
- Wang, N.; Wang, J.; Zhang, Y.; Wu, Z.; Wang, D.; Xiao, H.; Lyu, X.; Chen, H.; Wei, F. Comprehensive Lipidomics and Volatile Compounds Profiling Reveals Correlation of Lipids and Flavors in DHA-Enriched Egg Yolk. Oil Crop Sci. 2023, 8, 27–34. [Google Scholar] [CrossRef]
- Edens, F.W.; Sefton, A.E. Sel-PlexTM, A Source of Organic Selenium in Selenised Yeast Protein, as a Factor That Influences Meat Quality. J. Appl. Anim. Nutr. 2016, 4, e10. [Google Scholar] [CrossRef]
- Surai, P.F.; Kochish, I.I. Nutritional Modulation of the Antioxidant Capacities in Poultry: The Case of Selenium. Poult. Sci. 2019, 98, 4231–4239. [Google Scholar] [CrossRef]
- Adeniran, S.O.; Zheng, P.; Feng, R.; Adegoke, E.O.; Huang, F.; Ma, M.; Wang, Z.; Ifarajimi, O.O.; Li, X.; Zhang, G. The Antioxidant Role of Selenium via GPx1 and GPx4 in LPS-Induced Oxidative Stress in Bovine Endometrial Cells. Biol. Trace Elem. Res. 2022, 200, 1140–1155. [Google Scholar] [CrossRef]
- Chen, Y.; He, H.; Bi, X.; Zhang, Y.; Yin, H. Effects of Various Selenium-Enriched Yeasts, Selenomethionine, and Nanoselenium on Production Performance, Quality, and Antioxidant Capacity in Laying Hens. Poult. Sci. 2024, 103, 103387. [Google Scholar] [CrossRef]
- Jing, C.L.; Dong, X.F.; Wang, Z.M.; Liu, S.; Tong, J.M. Comparative Study of DL-Selenomethionine Vs Sodium Selenite and Seleno-Yeast on Antioxidant Activity and Selenium Status in Laying Hens. Poult. Sci. 2015, 94, 965–975. [Google Scholar] [CrossRef]
- Obianwuna, U.E.; Oleforuh-Okoleh, V.U.; Wang, J.; Zhang, H.J.; Qi, G.H.; Qiu, K.; Wu, S.G. Potential Implications of Natural Antioxidants of Plant Origin on Oxidative Stability of Chicken Albumen during Storage: A Review. Antioxidants 2022, 11, 630. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.R.; Ward, G.E.; Regmi, P.; Karcher, D.M. Impact of Egg Handling and Conditions During Extended Storage on Egg Quality. Poult. Sci. 2018, 97, 716–723. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.H.; Lee, K.T.; Lee, W.I.; Han, Y.K. Effects of Storage Temperature and Time on the Quality of Eggs from Laying Hens at Peak Production. Asian-Australas. J. Anim. Sci. 2010, 24, 279–284. [Google Scholar] [CrossRef]
- Guinebretière, M.; Puterflam, J.; Keïta, A.; Réhault-Godbert, S.; Thomas, R.; Chartrin, P.; Cailleau-Audouin, E.; Coudert, E.; Collin, A. Storage Temperature or Thermal Treatments During Long Egg Storage Duration Influences Hatching Performance and Chick Quality. Front. Physiol. 2022, 13, 852733. [Google Scholar] [CrossRef]

| Items | Control (CON) | Microalgae (MA) |
|---|---|---|
| Ingredients | ||
| Corn | 61.50 | 61.50 |
| Soybean meal, 46% CP | 25.00 | 24.50 |
| Soybean oil | 1.50 | 0.50 |
| Microalgae | 0 | 1.50 |
| Limestone | 8.50 | 8.50 |
| CaHPO4 | 1.00 | 1.00 |
| Salts | 0.30 | 0.30 |
| DL-Methionine | 0.12 | 0.12 |
| Lysine-HCl | 0.08 | 0.08 |
| V + M Premix 1 | 2.00 | 2.00 |
| Total | 100.00 | 100.00 |
| Nutrient levels 2 | ||
| ME, Mcal/kg | 2.73 | 2.72 |
| CP, % | 16.63 (16.57) | 16.61 (16.58) |
| Lys, % | 0.89 | 0.89 |
| Met, % | 0.38 | 0.38 |
| Cys + Met, % | 0.75 | 0.75 |
| Ca, % | 3.66 (3.70) | 3.65 (3.69) |
| Total P, % | 0.55 | 0.55 |
| Av. P, % | 0.35 | 0.35 |
| Se, mg/kg | 0.11 (0.10) | 0.12 (0.11) |
| Treatment | Se Sources | Supplemental Se (mg/kg) | Calculated Total Se (mg/kg) | Analyzed Total Se (mg/kg) |
|---|---|---|---|---|
| CON | - | 0 | 0.11 | 0.100 ± 0.008 |
| MA | - | 0 | 0.12 | 0.110 ± 0.008 |
| MA + SS | sodium selenite | 0.25 | 0.36 | 0.360 ± 0.007 |
| MA + SY | selenium yeast | 0.25 | 0.37 | 0.350 ± 0.004 |
| Items | CON | MA | MA + SS | MA + SY | p-Value |
|---|---|---|---|---|---|
| Serum | 0.074 ± 0.009 c | 0.068 ± 0.008 c | 0.176 ± 0.013 b | 0.196 ± 0.009 a | <0.001 |
| Liver | 0.258 ± 0.015 c | 0.219 ± 0.012 d | 0.458 ± 0.023 b | 0.496 ± 0.016 a | <0.001 |
| Kidney | 0.408 ± 0.025 b | 0.420 ± 0.026 b | 0.627 ± 0.032 a | 0.603 ± 0.033 a | <0.001 |
| Spleen | 0.403 ± 0.013 b | 0.406 ± 0.018 b | 0.575 ± 0.015 a | 0.583 ± 0.017 a | <0.001 |
| Heart | 0.302 ± 0.021 b | 0.311 ± 0.019 b | 0.474 ± 0.021 a | 0.492 ± 0.011 a | <0.001 |
| Item | CON | MA | MA + SS | MA + SY | p-Value |
|---|---|---|---|---|---|
| Se concentration | |||||
| Albumen | 0.061 ± 0.002 c | 0.059 ± 0.004 c | 0.110 ± 0.014 b | 0.239 ± 0.010 a | <0.001 |
| Yolk | 0.228 ± 0.017 b | 0.216 ± 0.016 b | 0.603± 0.035 a | 0.598± 0.038 a | <0.001 |
| Total Se amount | |||||
| Albumen | 2.44 ± 0.09 c | 2.40 ± 0.20 c | 4.33 ± 0.62 b | 9.61 ± 0.36 a | <0.001 |
| Yolk | 3.46 ± 0.29 b | 3.33 ± 0.25 b | 9.15 ± 0.60 a | 9.21 ± 0.39 a | <0.001 |
| Whole egg | 5.90 ± 0.23 c | 5.73 ± 0.24 c | 13.48 ± 0.91 b | 18.82 ± 0.31 a | <0.001 |
| Item | CON | MA | MA + SS | MA + SY | p-Value |
|---|---|---|---|---|---|
| Yolk composition | |||||
| Dry matter, % | 52.09 ± 1.00 | 52.55 ± 0.83 | 53.13 ± 0.43 | 52.92 ± 0.97 | 0.181 |
| Ash, % | 1.98 ± 0.20 | 2.01 ± 0.18 | 2.08 ± 0.13 | 2.11 ± 0.13 | 0.502 |
| Total lipid, % | 35.86 ± 2.64 | 34.56 ± 1.56 | 34.89 ± 2.16 | 34.64 ± 1.45 | 0.673 |
| Proteins, % | 16.28 ± 1.10 | 16.61 ± 1.27 | 15.94 ± 1.08 | 16.57 ± 1.41 | 0.764 |
| Total cholesterol, mg/g egg | 13.24 ± 1.59 | 13.49 ± 1.52 | 13.07± 2.00 | 12.55 ± 1.63 | 0.804 |
| DHA, mg/100 g yolk | 65.17 ± 2.42 c | 219.89 ± 8.90 b | 222.56 ±7.41 ab | 233.78 ± 8.14 a | <0.001 |
| Albumen composition | |||||
| Dry matter, % | 14.66 ± 0.50 | 14.54 ± 0.40 | 14.50 ± 0.41 | 14.61 ± 0.60 | 0.658 |
| Ash, % | 0.74 ± 0.05 | 0.69 ± 0.05 | 0.72 ± 0.04 | 0.71 ± 0.03 | 0.460 |
| Proteins, % | 13.80 ± 0.38 | 13.69 ± 0.56 | 13.47 ± 0.32 | 13.62 ± 0.25 | 0.453 |
| Item | CON | MA | MA + SS | MA + SY |
|---|---|---|---|---|
| Albumen GPX activity, U/g protein | ||||
| 0 days | 69.42 ± 7.67 b,x | 65.70 ± 4.34 b,x | 88.30 ± 5.25 a,x | 91.84 ± 3.72 a,x |
| 15 days | 57.49 ± 4.47 b,y | 57.75 ± 6.24 b,y | 64.82 ± 6.11 ab,y | 73.26 ± 7.34 a,y |
| 30 days | 51.59 ± 3.35 bc,y | 47.12 ± 2.82 c,z | 57.78 ± 4.23 ab,y | 64.54 ± 7.36 a,y |
| Albumen carbonyl content, nm/mg protein | ||||
| 0 days | 4.44 ± 0.74 ab,z | 5.38 ± 0.49 a,z | 4.51 ± 0.41 ab,z | 4.40 ± 0.62 b,z |
| 15 days | 6.87 ± 0.71 ab,y | 7.73 ± 0.77 a,y | 6.20 ± 0.40 bc,y | 5.61 ± 0.46 c,y |
| 30 days | 8.40 ± 0.63 a,x | 9.19 ± 0.50 a,x | 7.31 ± 0.31 b,x | 6.61 ± 0.88 b,x |
| Yolk GPX activity, U/g protein | ||||
| 0 days | 53.59 ± 4.99 b,x | 50.75 ± 5.69 b,x | 64.63 ± 6.06 a,x | 71.71 ± 6.53 a,x |
| 15 days | 49.93 ± 4.48 b,xy | 47.07 ± 5.22 b,x | 59.62 ± 4.60 a,x | 66.33 ± 4.58 a,x |
| 30 days | 41.25 ± 6.21 b,y | 38.85 ± 2.97 b,y | 51.80 ± 3.33 a,y | 58.10 ± 2.38 a,y |
| TBARS in yolk, μg MDA/kg | ||||
| 0 days | 166.6 ± 13.3 ab,z | 182.1 ± 8.7 a,y | 153.5 ± 12.4 bc,y | 145.7 ± 9.5 c,y |
| 15 days | 353.0 ± 24.8 b,y | 438.0 ± 20.9 a,x | 337.2 ± 27.5 b,x | 317.2 ± 31.1 b,x |
| 30 days | 403.3 ± 12.8 bc,x | 495.8 ± 26.0 a,x | 408.7 ± 42.9 b,x | 361.2 ± 20.3 c,x |
| Item | CON | MA | MA + SS | MA + SY |
|---|---|---|---|---|
| Weight loss rate, % | ||||
| 15 days | 2.37 ± 0.25 b,y | 3.12 ± 0.19 a,y | 2.77 ± 0.42 ab,y | 2.36 ± 0.38 b,y |
| 30 days | 4.95 ± 0.35 b,x | 5.97 ± 0.63 a,x | 5.09 ± 0.44 b,x | 4.37 ± 0.41 b,x |
| Albumen height, mm | ||||
| 0 days | 7.20 ± 0.31 x | 7.10 ± 0.47 x | 7.13 ± 0.31 x | 7.13 ± 0.26 x |
| 15 days | 5.15 ± 0.40 a,y | 4.40 ± 0.24 b,y | 5.00 ± 0.48 a,y | 5.20 ± 0.36 a,y |
| 30 days | 4.20 ± 0.24 a,z | 3.72 ± 0.22 b,z | 4.30 ± 0.48 a,y | 4.52 ± 0.63 a,y |
| Haugh unit | ||||
| 0 days | 84.56 ± 2.02 x | 83.96 ± 2.79 x | 84.36 ± 1.81 x | 84.25 ± 1.69 x |
| 15 days | 70.04 ± 3.40 a,y | 63.54 ± 2.65 b,y | 72.19 ± 4.10 a,y | 70.60 ± 2.99 a,y |
| 30 days | 61.48 ± 2.23 a,z | 54.93 ± 2.58 b,z | 59.62 ± 5.79 a,z | 62.97 ± 5.51 a,y |
| Yolk pH | ||||
| 0 days | 6.06 ± 0.06 z | 6.03 ± 0.10 z | 6.06 ± 0.12 z | 6.00 ± 0.12 y |
| 15 days | 6.48 ± 0.11 y | 6.54 ± 0.07 y | 6.44 ± 0.07 y | 6.40 ± 0.10 x |
| 30 days | 6.70 ± 0.10 ab,x | 6.82 ± 0.13 a,x | 6.53 ± 0.14 bc,x | 6.49 ± 0.07 c,x |
| Yolk index | ||||
| 0 days | 0.50 ± 0.01 x | 0.51 ± 0.02 x | 0.50 ± 0.01 x | 0.51 ± 0.02 x |
| 15 days | 0.45 ± 0.02 bc,y | 0.44 ± 0.01 c,y | 0.46 ± 0.02 ab,y | 0.47 ± 0.02 a,y |
| 30 days | 0.41 ± 0.01 bc,z | 0.39 ± 0.02 c,z | 0.42 ± 0.02 b,z | 0.44 ± 0.01 a,z |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zou, C.; Ge, C.; Lv, Y.; Huang, W.; Zhan, S.; Shen, X.; Yuan, H.; Wang, X.; Wu, L.; Yu, D.; et al. Comparative Effects of Selenium Yeast and Sodium Selenite on the Selenium Distribution, Interior Quality and Oxidative Stability of Docosahexaenoic Acid-Enriched Eggs During Storage. Antioxidants 2025, 14, 1333. https://doi.org/10.3390/antiox14111333
Zou C, Ge C, Lv Y, Huang W, Zhan S, Shen X, Yuan H, Wang X, Wu L, Yu D, et al. Comparative Effects of Selenium Yeast and Sodium Selenite on the Selenium Distribution, Interior Quality and Oxidative Stability of Docosahexaenoic Acid-Enriched Eggs During Storage. Antioxidants. 2025; 14(11):1333. https://doi.org/10.3390/antiox14111333
Chicago/Turabian StyleZou, Chenhao, Chaoyue Ge, Yujie Lv, Weichen Huang, Shenao Zhan, Xinyu Shen, Hongmeng Yuan, Xiaoxu Wang, Lianchi Wu, Dongyou Yu, and et al. 2025. "Comparative Effects of Selenium Yeast and Sodium Selenite on the Selenium Distribution, Interior Quality and Oxidative Stability of Docosahexaenoic Acid-Enriched Eggs During Storage" Antioxidants 14, no. 11: 1333. https://doi.org/10.3390/antiox14111333
APA StyleZou, C., Ge, C., Lv, Y., Huang, W., Zhan, S., Shen, X., Yuan, H., Wang, X., Wu, L., Yu, D., & Liu, B. (2025). Comparative Effects of Selenium Yeast and Sodium Selenite on the Selenium Distribution, Interior Quality and Oxidative Stability of Docosahexaenoic Acid-Enriched Eggs During Storage. Antioxidants, 14(11), 1333. https://doi.org/10.3390/antiox14111333