The Effects of Unconventional Feed Fermentation on Intestinal Oxidative Stress in Animals
Abstract
1. Introduction
2. Types and Applications of Unconventional Feed
2.1. By-Products of Grain and Oil Processing
2.2. By-Products of Livestock and Poultry Processing
2.3. By-Products of Aquatic Products Processing
2.4. Other Processing By-Products
3. The Effects and Mechanism of Fermentation on the Improvement of Unconventional Feed Quality
3.1. Reductions in Anti-Nutritional Factors and Toxins
3.2. Increases Feed Flavor and Improves Growth Performance
3.3. Improves Intestinal Health and Strengthens Immunity
3.4. Reduces Pollution from Livestock and Poultry Farming
4. Unconventional Fermented Feed Improves Oxidative Stress by Regulating Gut Health
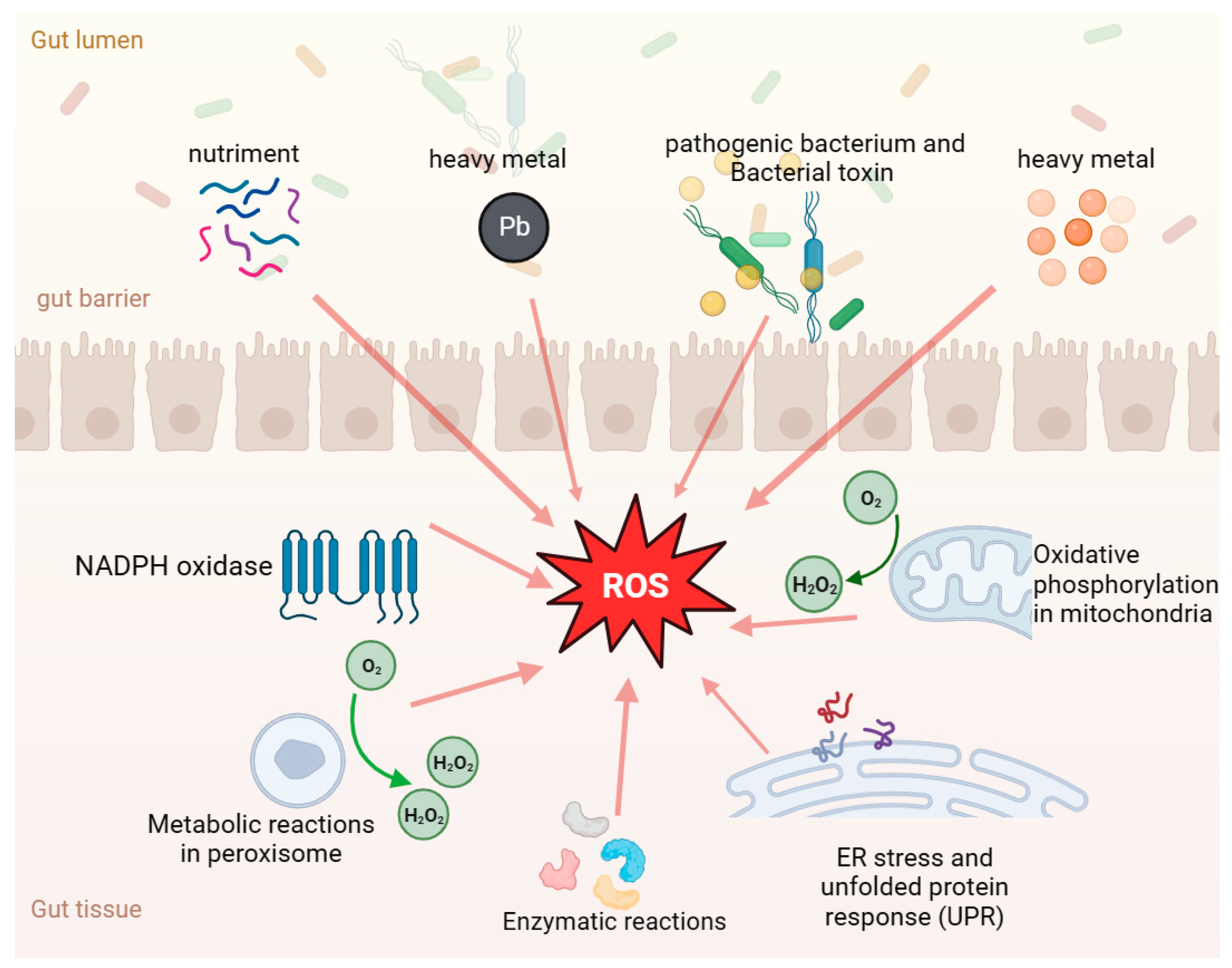
4.1. Gut Health and Oxidative Stress
4.2. Effects of Unconventional Fermented Feed on the Morphological Structure of Intestinal Mucosa
4.3. Effects of Unconventional Fermented Feed on Intestinal Barrier and Absorption Function
4.4. Effects of Unconventional Fermented Feed on Gut Microbiota
4.5. Effects of Unconventional Fermented Feed on Immune Function
4.6. Effects of Unconventional Fermented Feed on Antioxidant Capacity
5. Prospects
- (1)
- Diversified sources of raw materials: Exploring a greater variety of unconventional raw materials, such as agricultural by-products, aquatic by-products, and industrial by-products, can reduce supply instability through inter-regional and inter-seasonal diversification. The introduction of diversified sources of raw materials can not only reduce excessive dependence on a specific raw material, but can also help to improve the diversity and nutritional balance of feed.
- (2)
- Optimization of the fermentation process: By studying and optimizing the fermentation process, such as by controlling parameters such as temperature, humidity, and PH, the stability and efficiency of the fermentation process can be improved. The introduction of automation technology and intelligent monitoring systems can be used to help reduce human error, improve production efficiency, and improve product quality.
- (3)
- Anti-nutritional factor processing technology: The development of efficient treatment technologies, such as enzymatic hydrolysis, heat treatment, and acid–base treatment, can effectively reduce the content of antinutritional factors in raw materials. Further research into the mechanisms of different antinutritional factors in animal digestion and absorption is needed to optimize treatment techniques and ensure the nutritional value of feed.
- (4)
- Economic benefit evaluation: A comprehensive economic benefit evaluation study should be conducted to consider production costs, animal growth performance, feed utilization efficiency, and other factors to provide a more concrete and reliable economic decision-making basis for farmers and breeders. Tailored economic assessments should be conducted for different farming scales and regional characteristics to ensure the practical feasibility and sustainability of solutions.
- (5)
- An in-depth study of animal health and oxidative stress mechanisms: researchers should investigate the mechanism of fermented feed on the intestinal microecological balance, and explore its specific effects on animal immunity and disease resistance in order to further understand the regulatory effects of active ingredients in fermented feed on oxidative stress in animals, as well as the mechanism of their effects on the overall growth performance and meat quality.
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Liang, J.; Nie, Z.; Zhao, Y.; Qin, S.; Nian, F.; Tang, D. Effects of Jujube Powder on Growth Performance, Blood Biochemical Indices, and Intestinal Microbiota of Broiler. Animals 2023, 13, 3398. [Google Scholar] [CrossRef]
- Ren, Y.; Liu, L.; Zhou, S.; Li, Y.; Wang, Y.; Yang, K.; Chen, W.; Zhao, S. Effects of Different Proportions of Amaranthus hypochondriacus Stem and Leaf Powder Inclusions on Growth Performance, Carcass Traits, and Blood Biochemical Parameters of Broilers. Animals 2023, 13, 2818. [Google Scholar] [CrossRef]
- Abd El-Hack, M.E.; Samak, D.H.; Noreldin, A.E.; Arif, M.; Yaqoob, H.S.; Swelum, A.A. Towards saving freshwater: Halophytes as unconventional feedstuffs in livestock feed: A review. Environ. Sci. Pollut. Res. Int. 2018, 25, 14397–14406. [Google Scholar] [CrossRef] [PubMed]
- Sugiharto, S.; Ranjitkar, S. Recent advances in fermented feeds towards improved broiler chicken performance, gastrointestinal tract microecology and immune responses: A review. Anim. Nutr. 2019, 5, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, G.; Liu, J.; Hao, S.; Huang, S. The Influence of Converting Food Crops to Forage Crops Policy Implementation on Herbivorous Livestock Husbandry Development—Based on Policy Pilot Counties in Hebei, China. Agriculture 2022, 12, 1872. [Google Scholar] [CrossRef]
- Yin, J.; Li, Y.; Han, H.; Liu, Z.; Zeng, X.; Li, T.; Yin, Y. Long-term effects of lysine concentration on growth performance, intestinal microbiome, and metabolic profiles in a pig model. Food Funct. 2018, 9, 4153–4163. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhu, F.; Yang, H.; Li, J.; Li, Y.; Ding, X.; Xiong, X.; Yin, Y. Effects of dietary supplementation with epidermal growth factor on nutrient digestibility, intestinal development and expression of nutrient transporters in early-weaned piglets. J. Anim. Physiol. Anim. Nutr. (Berl.) 2019, 103, 618–625. [Google Scholar] [CrossRef] [PubMed]
- Sabour, S.; Tabeidian, S.A.; Sadeghi, G. Dietary organic acid and fiber sources affect performance, intestinal morphology, immune responses and gut microflora in broilers. Anim. Nutr. 2019, 5, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Shi, C.; Zhang, Y.; Song, D.; Lu, Z.; Wang, Y. Microbiota in fermented feed and swine gut. Appl. Microbiol. Biotechnol. 2018, 102, 2941–2948. [Google Scholar] [CrossRef] [PubMed]
- Kizir, D.; Karaman, M.; Ceylan, H. Tannic acid may ameliorate doxorubicin-induced changes in oxidative stress parameters in rat spleen. Naunyn Schmiedebergs Arch. Pharmacol. 2023, 396, 3605–3613. [Google Scholar] [CrossRef]
- Ponnampalam, E.N.; Kiani, A.; Santhiravel, S.; Holman, B.W.B.; Lauridsen, C.; Dunshea, F.R. The Importance of Dietary Antioxidants on Oxidative Stress, Meat and Milk Production, and Their Preservative Aspects in Farm Animals: Antioxidant Action, Animal Health, and Product Quality-Invited Review. Animals 2022, 12, 3279. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Zhang, H.; Li, S.; Jiang, Y.; Deng, J.; Yang, C.; Chen, X.; Jiang, L. Effects of different levels of Citri Sarcodactylis Fructus by-products fermented feed on growth performance, serum biochemical, and intestinal health of cyan-shank partridge birds. Sci. Rep. 2023, 13, 20130. [Google Scholar] [CrossRef]
- Fu, Z.; Ao, N.; Liang, X.; Chen, J.; Wang, Y.; Wang, Q.; Fu, J.; Liu, C.; Lu, L. Effects of fermented feed on growth performance, serum biochemical indexes, antioxidant capacity, and intestinal health of lion-head goslings. Front. Vet. Sci. 2023, 10, 1284523. [Google Scholar] [CrossRef]
- Jairath, G.; Verma, A.K.; Rani, D.; Marappan, G.; Bs, Y.; Singh, B.; Mal, G.; Gopinath, D.; Sharma, R.; Katoch, S.; et al. Self-fermented agro-wastes as antioxidant enriched maize grain replacer for sustainable animal feeding. J. Clean. Prod. 2023, 427, 139223. [Google Scholar] [CrossRef]
- Liu, S.; Tu, Y.; Sun, J.; Cai, P.; Zhou, Y.; Huang, Y.; Zhang, S.; Chen, W.; Wang, L.; Du, M.; et al. Fermented mixed feed regulates intestinal microbial community and metabolism and alters pork flavor and umami. Meat Sci. 2023, 201, 109177. [Google Scholar] [CrossRef]
- Hu, M.; Zhou, X.; Wang, Y.; Li, J.; Wu, Q.; Bao, S.; Jiang, L.; Liu, B. Use of fermented tea residues as a feed additive and effects on growth performance, body composition, intestinal enzyme activities, and inflammatory biomarkers in juvenile largemouth bass (Micropterus salmoides). Aquac. Rep. 2023, 31, 101671. [Google Scholar] [CrossRef]
- Feng, Y.; Zhang, W.; Xu, X.; Wang, W.; Xu, Y.; Wang, M.; Zhang, J.; Xu, H.; Fu, F. Protective effect of Luffa cylindrica fermentation liquid on cyclophosphamide-induced premature ovarian failure in female mice by attenuating oxidative stress, inflammation and apoptosis. J. Ovarian Res. 2024, 17, 24. [Google Scholar] [CrossRef]
- Bhavna, A.; Zindove, T.J.; Iji, P.A.; Bakare, A.G. Growth performance, carcass characteristics and meat sensory evaluation of broiler chickens fed diets with fermented cassava leaves. Anim. Biosci. 2024, 23, 362. [Google Scholar] [CrossRef]
- Martini, D.; Marino, M.; Venturi, S.; Tucci, M.; Klimis-Zacas, D.; Riso, P.; Porrini, M.; Del Bo, C. Blueberries and their bioactives in the modulation of oxidative stress, inflammation and cardio/vascular function markers: A systematic review of human intervention studies. J. Nutr. Biochem. 2023, 111, 109154. [Google Scholar] [CrossRef]
- Pan, Z.; Wang, W.; Chen, J.; Chen, Z.; Avellán-Llaguno, R.D.; Xu, W.; Duan, Y.; Liu, B.; Huang, Q. Temporal dynamics of microbial composition and antibiotic resistome in fermentation bed culture pig farms across various ages. Sci. Total Environ. 2024, 912, 168728. [Google Scholar] [CrossRef]
- Sun, H.; Kang, X.; Tan, H.; Cai, H.; Chen, D. Progress in Fermented Unconventional Feed Application in Monogastric Animal Production in China. Fermentation 2023, 9, 947. [Google Scholar] [CrossRef]
- Araiza Ponce, K.A.; Gurrola Reyes, J.N.; Martínez Estrada, S.C.; Salas Pacheco, J.M.; Palacios Torres, J.; Murillo Ortiz, M. Fermentation Patterns, Methane Production and Microbial Population under In Vitro Conditions from Two Unconventional Feed Resources Incorporated in Ruminant Diets. Animals 2023, 13, 2940. [Google Scholar] [CrossRef]
- Zhang, X.; Long, J.; Liu, J.; Hua, Y.; Zhang, C.; Li, X. Fermentation Characteristics, Antinutritional Factor Level and Flavor Compounds of Soybean Whey Yogurt. Foods 2024, 13, 330. [Google Scholar] [CrossRef]
- Li, J.; Wang, W.; Chen, S.; Shao, T.; Tao, X.; Yuan, X. Effect of Lactic Acid Bacteria on the Fermentation Quality and Mycotoxins Concentrations of Corn Silage Infested with Mycotoxigenic Fungi. Toxins 2021, 13, 699. [Google Scholar] [CrossRef]
- Kulabhusan, P.K.; Campbell, K. Physico-chemical treatments for the removal of cyanotoxins from drinking water: Current challenges and future trends. Sci. Total Environ. 2024, 917, 170078. [Google Scholar] [CrossRef]
- Qiu, Y.; Zhao, H.; He, X.; Zhu, F.; Zhang, F.; Liu, B.; Liu, Q. Effects of fermented feed of Pennisetum giganteum on growth performance, oxidative stress, immunity and gastrointestinal microflora of Boer goats under thermal stress. Front. Microbiol. 2022, 13, 1030262. [Google Scholar] [CrossRef]
- Guo, W.; Xu, L.N.; Guo, X.J.; Wang, W.; Hao, Q.H.; Wang, S.Y.; Zhu, B.C. The impacts of fermented feed on laying performance, egg quality, immune function, intestinal morphology and microbiota of laying hens in the late laying cycle. Animal 2022, 16, 100676. [Google Scholar] [CrossRef]
- Zhu, X.; Tao, L.; Liu, H.; Yang, G. Effects of fermented feed on growth performance, immune organ indices, serum biochemical parameters, cecal odorous compound production, and the microbiota community in broilers. Poult. Sci. 2023, 102, 102629. [Google Scholar] [CrossRef]
- Shi, H.T.; Wang, B.Y.; Bian, C.Z.; Han, Y.Q.; Qiao, H.X. Fermented Astragalus in diet improved laying performance, egg quality, antioxidant and immunological status and intestinal microbiota in laying hens. AMB Express 2020, 10, 159. [Google Scholar] [CrossRef]
- Wu, Z.; Yang, K.; Zhang, A.; Chang, W.; Zheng, A.; Chen, Z.; Cai, H.; Liu, G. Effects of Lactobacillus acidophilus on the growth performance, immune response, and intestinal barrier function of broiler chickens challenged with Escherichia coli O157. Poult. Sci. 2021, 100, 101323. [Google Scholar] [CrossRef]
- Yang, Y.; Yan, G.; Meng, X.; Wang, X.; Zhao, Z.; Zhou, S.; Li, G.; Zhang, Q.; Wei, X. Effects of Lactobacillus plantarum and Pediococcus acidilactici co-fermented feed on growth performance and gut microbiota of nursery pigs. Front. Vet. Sci. 2022, 9, 1076906. [Google Scholar] [CrossRef]
- Yu, D.Y.; Oh, S.H.; Kim, I.S.; Kim, G.I.; Kim, J.A.; Moon, Y.S.; Jang, J.C.; Lee, S.S.; Jung, J.H.; Park, J.; et al. Intestinal microbial composition changes induced by Lactobacillus plantarum GBL 16, 17 fermented feed and intestinal immune homeostasis regulation in pigs. J. Anim. Sci. Technol. 2022, 64, 1184–1198. [Google Scholar] [CrossRef]
- Zhang, Q.; Jing, X.; Zhao, Y.; Xia, D.; Liu, S.; Li, D.; Hao, Q.; Wang, M.; Yu, Z.; Li, S.; et al. Partial replacement of pelleted feed by moist fermented feed improved the feed conversion efficiency, liver and intestine health, and gut microbiota structure in common carp (Cyprinus carpio). Aquac. Rep. 2023, 32, 101690. [Google Scholar] [CrossRef]
- Oncul, F.O.; Aya, F.A.; Ali, H.; Seonghun, W.; Geon, L.; Han, K.R.; Bai, S.C. Effects of the dietary fermented tuna by-product meal on growth, blood parameters, nonspecific immune response, and disease resistance in juvenile olive flounder, Paralichthys olivaceus. J. World Aquac. Soc. 2018, 50, 65–77. [Google Scholar] [CrossRef]
- Selim, A.S.M.; Hasan, M.N.; Rahman, M.A.; Rahman, M.M.; Islam, M.R.; Bostami, A.; Islam, S.; Tedeschi, L.O. Nutrient content and in vitro degradation study of some unconventional feed resources of Bangladesh. Heliyon 2022, 8, e09496. [Google Scholar] [CrossRef]
- Wang, J.; Lu, R.; Li, Y.; Lu, J.; Liang, Q.; Zheng, Z.; Huang, H.; Deng, F.; Huang, H.; Jiang, H.; et al. Dietary supplementation with jasmine flower residue improves meat quality and flavor of goat. Front. Nutr. 2023, 10, 1145841. [Google Scholar] [CrossRef]
- Omar, A.E.; Al-Khalaifah, H.S.; Ismail, T.A.; Abd El-Aziz, R.M.; El-Mandrawy, S.A.M.; Shalaby, S.I.; Ibrahim, D. Performance, Serum Biochemical and Immunological Parameters, and Digestive Enzyme and Intestinal Barrier-Related Gene Expression of Broiler Chickens Fed Fermented Fava Bean By-Products as a Substitute for Conventional Feed. Front. Vet. Sci. 2021, 8, 696841. [Google Scholar] [CrossRef]
- Yuchao, H.; Ziqi, M.; Wenwen, W.; Xiran, H.; Yuan, W.; Jingwei, Q. Carcase traits, meat quality, and lipogenic gene expression in muscle of lambs fed wheat bran feruloyl oligosaccharides. Ital. J. Anim. Sci. 2023, 22, 369–378. [Google Scholar]
- Veluri, S.; Gonzalez-Ortiz, G.; Bedford, M.R.; Olukosi, O.A. Interactive effects of a stimbiotic supplementation and wheat bran inclusion in corn- or wheat-based diets on growth performance, ileal digestibility, and expression of nutrient transporters of broilers chickens. Poult. Sci. 2024, 103, 103178. [Google Scholar] [CrossRef]
- Phomkaivon, N.; Pan-utai, W.; Surojanametakul, V.; Varichanan, P.; Kaewtathip, T.; Kanyakam, K.; Klinsoda, J. Isoflavone aglycone-rich powder from soybean residue submerged fermentation using Lactobacillus fermentum 44197. NFS J. 2023, 33, 100157. [Google Scholar] [CrossRef]
- Fan, Z.; Chen, T.; Cai, G.; Huang, X.; Zhong, S.; Li, X.; Zhang, E. Effect of Aspergillus niger Fermentation on the Metabolites in Corn Stalks. Fermentation 2023, 9, 50. [Google Scholar] [CrossRef]
- Nunes, A.J.P.; Dalen, L.L.; Leonardi, G.; Burri, L. Developing sustainable, cost-effective and high-performance shrimp feed formulations containing low fish meal levels. Aquac. Rep. 2022, 27, 101422. [Google Scholar] [CrossRef]
- Xia, W.G.; Abouelezz, K.F.M.; Makled, M.N.; Wang, S.; Chen, W.; Zhang, Y.N.; Elokil, A.A.; Wang, S.L.; Huang, X.B.; Li, K.C.; et al. Effects of dietary substitution of peanut meal for soybean meal on egg production, egg quality, oxidative status, and yolk fatty acid profile in laying ducks. Animal 2022, 16, 100652. [Google Scholar] [CrossRef]
- Valena, R.D.L.; Sobrinho, A.G.d.S.; Romanzini, E.P.; Andrade, N.D.; Oliveira, V.D.S. Peanut meal and crude glycerin in lamb diets: Meat quality and fatty acid profile. Small Rumin. Res. 2020, 185, 106076. [Google Scholar] [CrossRef]
- Wang, T.; Xu, M.; Wang, J.; Wan, W.; Sun, H. A combination of rapeseed, cottonseed and peanut meal as a substitute of soybean meal in diets of Yellow River carp Cyprinus carpio var. Aquac. Nutr. 2020, 26, 1520–1532. [Google Scholar] [CrossRef]
- Azad, M.A.K.; Jiang, H.; Ni, H.; Liu, Y.; Huang, P.; Fang, J.; Kong, X. Diets Partially Replaced With Cassava Residue Modulate Antioxidant Capacity, Lipid Metabolism, and Gut Barrier Function of Huanjiang Mini-Pigs. Front. Vet. Sci. 2022, 9, 902328. [Google Scholar] [CrossRef]
- Santana, T.M.; Dantas, F.d.M.; Monteiro Dos Santos, D.K.; Kojima, J.T.; Pastrana, Y.M.; De Jesus, R.S.; Gonçalves, L.U. Fish Viscera Silage: Production, Characterization, and Digestibility of Nutrients and Energy for Tambaqui Juveniles. Fishes 2023, 8, 111. [Google Scholar] [CrossRef]
- Lasekan, A.; Abu Bakar, F.; Hashim, D. Potential of chicken by-products as sources of useful biological resources. Waste Manag. 2013, 33, 552–565. [Google Scholar] [CrossRef]
- Chen, H.; Gao, S.; Li, Y.; Xu, H.-J.; Li, W.; Wang, J.; Zhang, Y. Valorization of Livestock Keratin Waste: Application in Agricultural Fields. Int. J. Environ. Res. Public Health 2022, 19, 6681. [Google Scholar] [CrossRef]
- Flores Tapia, N.E.; Brito Moina, H. Exploring Tannery Solid Wastes as a Source of Animal Feed. Processes 2023, 11, 2965. [Google Scholar] [CrossRef]
- Suryadi, U.; Hertamawati, R.T.; Imam, A.S. Hydrolyzation of snail (Achatina fulica) meat with rice water as novel probiotic supplements for animal feed. Vet. World 2022, 15, 937–942. [Google Scholar] [CrossRef]
- Nag, M.; Lahiri, D.; Dey, A.; Sarkar, T.; Pati, S.; Joshi, S.; Bunawan, H.; Mohammed, A.; Edinur, H.A.; Ghosh, S.; et al. Seafood Discards: A Potent Source of Enzymes and Biomacromolecules With Nutritional and Nutraceutical Significance. Front. Nutr. 2022, 9, 879929. [Google Scholar] [CrossRef]
- Terzioğlu, P.; Öğüt, H.; Kalemtaş, A. Natural calcium phosphates from fish bones and their potential biomedical applications. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 91, 899–911. [Google Scholar] [CrossRef]
- Naghdi, S.; Rezaei, M.; Tabarsa, M.; Abdollahi, M. Fish Protein Hydrolysate from Sulfated Polysaccharides Extraction Residue of Tuna Processing By-Products with Bioactive and Functional Properties. Glob. Chall. 2023, 7, 2200214. [Google Scholar] [CrossRef]
- Zhu, K.; Yan, W.; Dai, Z.; Zhang, Y. Astaxanthin Extract from Shrimp (Trachypenaeus curvirostris) By-Products Improves Quality of Ready-to-Cook Shrimp Surimi Products during Frozen Storage at −18 °C. Foods 2022, 11, 2122. [Google Scholar] [CrossRef]
- Ge, Q.; Chen, L.; Tang, M.; Zhang, S.; Liu, L.; Gao, L.; Ma, S.; Kong, M.; Yao, Q.; Feng, F.; et al. Analysis of mulberry leaf components in the treatment of diabetes using network pharmacology. Eur. J. Pharmacol. 2018, 833, 50–62. [Google Scholar] [CrossRef]
- Ma, J.; Wang, J.; Jin, X.; Liu, S.; Tang, S.; Zhang, Z.; Long, S.; Piao, X. Effect of Dietary Supplemented with Mulberry Leaf Powder on Growth Performance, Serum Metabolites, Antioxidant Property and Intestinal Health of Weaned Piglets. Antioxidants 2023, 12, 307. [Google Scholar] [CrossRef]
- Herosimczyk, A.; Lepczyński, A.; Ożgo, M.; Tuśnio, A.; Taciak, M.; Barszcz, M. Effect of dietary inclusion of 1% or 3% of native chicory inulin on the large intestinal mucosa proteome of growing pigs. Animal 2020, 14, 1647–1658. [Google Scholar] [CrossRef]
- Li, X.; Jensen, B.B.; Canibe, N. The Mode of Action of Chicory Roots on Skatole Production in Entire Male Pigs Is neither via Reducing the Population of Skatole-Producing Bacteria nor via Increased Butyrate Production in the Hindgut. Appl. Environ. Microbiol. 2019, 85, e02327-18. [Google Scholar] [CrossRef]
- Chen, Y.; Xie, Y.; Zhong, R.; Han, H.; Liu, L.; Chen, L.; Zhang, H.; Beckers, Y.; Everaert, N. Effects of graded levels of xylo-oligosaccharides on growth performance, serum parameters, intestinal morphology, and intestinal barrier function in weaned piglets. J. Anim. Sci. 2021, 99, skab183. [Google Scholar] [CrossRef]
- Zou, S.; Sun, C.; Li, F.; Xie, Y.; Liang, T.; Yang, Y.; Shi, B.; Ma, Q.; Shi, Z.; Chai, S.; et al. Effect of Gardenia Pomace Supplementation on Growth Performance, Blood Metabolites, Immune and Antioxidant Indices, and Meat Quality in Xiangcun Pigs. Animals 2022, 12, 2280. [Google Scholar] [CrossRef]
- Chen, Q.; Wang, C.; Sun, Y.; Chen, Y.; Chen, S.; Han, T.; Wang, J. Excessive Substitution of Fish Meal with Fermented Soybean Meal Induces Oxidative Stress by Impairing Glutathione Metabolism in Largemouth Bass (Micropterus salmoides). Antioxidants 2023, 12, 2096. [Google Scholar] [CrossRef]
- Wu, H.; Barrow, C.; Dunshea, F.R.; Suleria, H.A.R. Phenolic bioaccessibility, antioxidant, and antidiabetic effects of indigenous fermented coffee beans after simulated gastrointestinal digestion and colonic fermentation. Food Biosci. 2023, 54, 102920. [Google Scholar] [CrossRef]
- Zhou, R.; Wang, L.; Li, Y.; Wu, H.; Lu, L.; Zang, R.; Xu, H. Effects of Tail Vegetable Fermented Feed on the Growth and Rumen Microbiota of Lambs. Animals 2024, 14, 303. [Google Scholar] [CrossRef]
- Chen, X.; Zhou, X.; Li, S.; Zhang, H.; Liu, Z. Effects of tea residues-fermented feed on production performance, egg quality, antioxidant capacity, caecal microbiota, and ammonia emissions of laying hens. Front. Vet. Sci. 2023, 10, 1195074. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhao, F.; Shi, T.; Xiong, Z.; Gao, R.; Yuan, L. Suanyu fermentation strains screening, process optimization and the effect of thermal processing methods on its flavor. Food Res. Int. 2023, 173, 113296. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Jin, Z.; Hu, D.; Yang, W.; Yan, Y.; Nie, X.; Lin, J.; Zhang, Q.; Gai, D.; Ji, Y.; et al. Effect of solid-state fermentation with Lactobacillus casei on the nutritional value, isoflavones, phenolic acids and antioxidant activity of whole soybean flour. LWT 2020, 125, 109264. [Google Scholar] [CrossRef]
- Yiannikouris, A.; Apajalahti, J.; Siikanen, O.; Dillon, G.P.; Moran, C.A. Saccharomyces cerevisiae Cell Wall-Based Adsorbent Reduces Aflatoxin B1 Absorption in Rats. Toxins 2021, 13, 209. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, H.; Luo, J.; Chen, T.; Xi, Q.; Sun, J.; Wei, L.; Zhang, Y. Synergism of fermented feed and ginseng polysaccharide on growth performance, intestinal development, and immunity of Xuefeng black-bone chickens. BMC Vet. Res. 2024, 20, 13. [Google Scholar] [CrossRef]
- Wang, G.; Meng, Z.; Chen, L.; Jiang, J.; Feng, Y.; Zhang, B. Effects of kelp residues fermented with probiotics on the culture of sea cucumber, Apostichopus japonicus. Aquac. Res. 2020, 51, 1133–1142. [Google Scholar] [CrossRef]
- Olukomaiya, O.O.; Pan, L.; Zhang, D.; Mereddy, R.; Sultanbawa, Y.; Li, X. Performance and ileal amino acid digestibility in broilers fed diets containing solid-state fermented and enzyme-supplemented canola meals. Anim. Feed Sci. Technol. 2021, 275, 114876. [Google Scholar] [CrossRef]
- Adisa, A.M.; Badejo, A.A.; Ifesan, B.O.T.; Enujiugha, V.N. Phenotypic and molecular differentiation of lactic acid bacteria in fonio millet ogi fermentation and their potential as starter cultures. Food Humanit. 2024, 2, 100230. [Google Scholar] [CrossRef]
- Gao, Y.; Hu, M.; Meng, W.; Wen, W.; Zhang, P.; Fan, B.; Wang, F.; Li, S. Study on the quality of soybean proteins fermented by Bacillus subtilis BSNK-5: Insights into nutritional, functional, safety, and flavor properties. Food Chem. 2024, 443, 138523. [Google Scholar] [CrossRef] [PubMed]
- Shangguan, L.; Liu, Z.; Zhang, H.; Yang, Q.; Zhang, X.; Yao, L.; Li, P.; Chen, X.; Dai, J. Improved umami taste of the enzymatic hydrolysate of soybean protein isolate by Corynebacterium glutamicum P-45 fermentation. Food Biosci. 2024, 58, 103565. [Google Scholar] [CrossRef]
- Guo, S.; Zhang, Y.; Cheng, Q.; Xv, J.; Hou, Y.; Wu, X.; Du, E.; Ding, B. Partial Substitution of Fermented Soybean Meal for Soybean Meal Influences the Carcass Traits and Meat Quality of Broiler Chickens. Animals 2020, 10, 225. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.H.; Horng, Y.B.; Chen, W.J.; Hua, K.F.; Dybus, A.; Yu, Y.H. Effect of Fermented Products Produced by Bacillus licheniformis on the Growth Performance and Cecal Microbial Community of Broilers under Coccidial Challenge. Animals 2021, 11, 1245. [Google Scholar] [CrossRef]
- Qiu, Y.; Li, K.; Zhao, X.; Liu, S.; Wang, L.; Yang, X.; Jiang, Z. Fermented Feed Modulates Meat Quality and Promotes the Growth of Longissimus Thoracis of Late-Finishing Pigs. Animals 2020, 10, 1682. [Google Scholar] [CrossRef]
- Lu, J.; Han, Q.; Wang, S.; Wang, Z.; Li, X.; Hu, J.; Yang, G.; Wang, L.; Shi, X. Effect of fermented corn-soybean meal on carcass and meat quality of grower-finisher pigs. J. Anim. Physiol. Anim. Nutr. (Berl.) 2021, 105, 693–698. [Google Scholar] [CrossRef]
- Zhu, J.J.; Gao, M.X.; Song, X.J.; Zhao, L.; Li, Y.W.; Hao, Z.H. Changes in bacterial diversity and composition in the faeces and colon of weaned piglets after feeding fermented soybean meal. J. Med. Microbiol. 2018, 67, 1181–1190. [Google Scholar] [CrossRef]
- Liu, S.; Du, M.; Tu, Y.; You, W.; Chen, W.; Liu, G.; Li, J.; Wang, Y.; Lu, Z.; Wang, T.; et al. Fermented mixed feed alters growth performance, carcass traits, meat quality and muscle fatty acid and amino acid profiles in finishing pigs. Anim. Nutr. 2023, 12, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Neves, N.; De Dea Lindner, J.; Stockhausen, L.; Delziovo, F.R.; Bender, M.; Serzedello, L.; Cipriani, L.A.; Ha, N.; Skoronski, E.; Gisbert, E.; et al. Fermentation of Plant-Based Feeds with Lactobacillus acidophilus Improves the Survival and Intestinal Health of Juvenile Nile Tilapia (Oreochromis niloticus) Reared in a Biofloc System. Animals 2024, 14, 332. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, M.; Song, F.; Zhu, X.; Lu, Q.; Liu, R. Fermentation enhances the amelioration effect of bee pollen on Caco-2 monolayer epithelial barrier dysfunction based on NF-κB-mediated MLCK-MLC signaling pathway. Food Res. Int. 2024, 178, 113938. [Google Scholar] [CrossRef]
- Mirsalami, S.M.; Mirsalami, M. Impact of solid-state fermentation utilizing Saccharomyces boulardii on the chemical composition and bioactive constituents of rice husk. J. Agric. Food Res. 2024, 15, 100957. [Google Scholar] [CrossRef]
- Wang, C.; Shi, C.; Su, W.; Jin, M.; Xu, B.; Hao, L.; Zhang, Y.; Lu, Z.; Wang, F.; Wang, Y.; et al. Dynamics of the Physicochemical Characteristics, Microbiota, and Metabolic Functions of Soybean Meal and Corn Mixed Substrates during Two-Stage Solid-State Fermentation. mSystems 2020, 5, e00501-19. [Google Scholar] [CrossRef]
- Koo, B.; Kim, J.W.; Nyachoti, C.M. Nutrient and energy digestibility, and microbial metabolites in weaned pigs fed diets containing Lactobacillus–fermented wheat. Anim. Feed Sci. Technol. 2018, 241, 27–37. [Google Scholar] [CrossRef]
- Kulprachakarn, K.; Chaipoot, S.; Phongphisutthinant, R.; Paradee, N.; Prommaban, A.; Ounjaijean, S.; Rerkasem, K.; Parklak, W.; Prakit, K.; Saengsitthisak, B.; et al. Antioxidant Potential and Cytotoxic Effect of Isoflavones Extract from Thai Fermented Soybean (Thua-Nao). Molecules 2021, 26, 7432. [Google Scholar] [CrossRef]
- de Oliveira, N.S.; Ha, N.; da Cunha, L.; Cipriani, L.A.; Neto, A.T.; Skoronski, E.; Gisbert, E.; Perez Fabregat, T.E.H. Fermentation of Soybean Meal with Lactobacillus acidophilus Allows Greater Inclusion of Vegetable Protein in the Diet and Can Reduce Vibrionacea in the Intestine of the South American Catfish (Rhamdia quelen). Animals 2022, 12, 690. [Google Scholar] [CrossRef]
- Wang, W.; Tan, Z.; Gu, L.; Ma, H.; Wang, Z.; Wang, L.; Wu, G.; Qin, G.; Wang, Y.; Pang, H. Dynamics Changes of Microorganisms Community and Fermentation Quality in Soybean Meal Prepared with Lactic Acid Bacteria and Artemisia argyi through Fermentation and Aerobic Exposure Processes. Foods 2022, 11, 795. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Zeng, N.; Jiang, S.; Wang, X.; Yan, H.; Gao, C. Dietary replacement of soybean meal by fermented feedstuffs for aged laying hens: Effects on laying performance, egg quality, nutrient digestibility, intestinal health, follicle development, and biological parameters in a long-term feeding period. Poult. Sci. 2023, 102, 102478. [Google Scholar] [CrossRef]
- Sun, J.; Li, M.; Tang, Z.; Zhang, X.; Chen, J.; Sun, Z. Effects of Rhodotorula mucilaginosa fermentation product on the laying performance, egg quality, jejunal mucosal morphology and intestinal microbiota of hens. J. Appl. Microbiol. 2020, 128, 54–64. [Google Scholar] [CrossRef]
- Tokach, M.D.; Menegat, M.B.; Gourley, K.M.; Goodband, R.D. Review: Nutrient requirements of the modern high-producing lactating sow, with an emphasis on amino acid requirements. Animal 2019, 13, 2967–2977. [Google Scholar] [CrossRef]
- Wang, C.; Lin, C.; Su, W.; Zhang, Y.; Wang, F.; Wang, Y.; Shi, C.; Lu, Z. Effects of supplementing sow diets with fermented corn and soybean meal mixed feed during lactation on the performance of sows and progeny. J. Anim. Sci. 2018, 96, 206–214. [Google Scholar] [CrossRef]
- Grela, E.R.; Czech, A.; Kiesz, M.; Wlazło, Ł.; Nowakowicz-Dębek, B. A fermented rapeseed meal additive: Effects on production performance, nutrient digestibility, colostrum immunoglobulin content and microbial flora in sows. Anim. Nutr. 2019, 5, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Qiang, X.; Xia, T.; Geng, B.; Zhao, M.; Li, X.; Zheng, Y.; Wang, M. Bioactive Components of Lycium barbarum and Deep-Processing Fermentation Products. Molecules 2023, 28, 8044. [Google Scholar] [CrossRef]
- Krueger, L.A.; Koester, L.R.; Jones, D.F.; Spangler, D.A. Carbon dioxide equivalent emissions from corn silage fermentation. Front. Microbiol. 2022, 13, 1092315. [Google Scholar] [CrossRef]
- Delahoy, M.J.; Wodnik, B.; McAliley, L.; Penakalapati, G.; Swarthout, J.; Freeman, M.C.; Levy, K. Pathogens transmitted in animal feces in low- and middle-income countries. Int. J. Hyg. Environ. Health 2018, 221, 661–676. [Google Scholar] [CrossRef] [PubMed]
- Mollenkopf, D.F.; Mathys, D.A.; Feicht, S.M.; Stull, J.W.; Bowman, A.S.; Daniels, J.B.; Wittum, T.E. Maintenance of Carbapenemase-Producing Enterobacteriaceae in a Farrow-to-Finish Swine Production System. Foodborne Pathog. Dis. 2018, 15, 372–376. [Google Scholar] [CrossRef]
- Hăbeanu, M.; Lefter, N.A.; Gheorghe, A.; Untea, A.; Ropotă, M.; Grigore, D.M.; Varzaru, I.; Toma, S.M. Evaluation of Performance, Nitrogen Metabolism and Tissue Composition in Barrows Fed an n-3 PUFA-Rich Diet. Animals 2019, 9, 234. [Google Scholar] [CrossRef]
- Liao, S.; Liao, L.; Huang, P.; Wang, Y.; Zhu, S.; Wang, X.; Lv, T.; Li, Y.; Fan, Z.; Liu, T.; et al. Effects of Different Levels of Garlic Straw Powder on Growth Performance, Meat Quality, Antioxidant and Intestinal Mucosal Morphology of Yellow-Feathered Broilers. Front. Physiol. 2022, 13, 902995. [Google Scholar] [CrossRef]
- Custódio, L.; Prados, L.F.; Oliveira, I.M.; Resende, F.D.; Pettigrew, J.E.; Yiannikouris, A.; Siqueira, G.R. Do mycotoxin contaminated diets and yeast cell wall adsorbent affect meat quality of Nellore bulls finished in feedlot?-A short communication. Meat Sci. 2019, 158, 107865. [Google Scholar] [CrossRef]
- Lobionda, S.; Sittipo, P.; Kwon, H.Y.; Lee, Y.K. The Role of Gut Microbiota in Intestinal Inflammation with Respect to Diet and Extrinsic Stressors. Microorganisms 2019, 7, 271. [Google Scholar] [CrossRef]
- Gentile, C.L.; Weir, T.L. The gut microbiota at the intersection of diet and human health. Science 2018, 362, 776–780. [Google Scholar] [CrossRef]
- Patnode, M.L.; Beller, Z.W.; Han, N.D.; Cheng, J.; Peters, S.L.; Terrapon, N.; Henrissat, B.; Gall, S.L.; Saulnier, L.; Hayashi, D.K. Interspecies Competition Impacts Targeted Manipulation of Human Gut Bacteria by Fiber-Derived Glycans. Cell 2019, 179, 59–73.e13. [Google Scholar] [CrossRef]
- Liping, Z.; Feng, Z.; Xiaoying, D.; Guojun, W.; Lam, Y.Y.; Xuejiao, W.; Huaqing, F.; Xinhe, X.; Chunhua, L.; Jilin, M. Gut bacteria selectively promoted by dietary fibers alleviate type 2 diabetes. Science 2018, 359, 1151–1156. [Google Scholar]
- Leprun, P.M.B.; Clarke, G. The gut microbiome and pharmacology: A prescription for therapeutic targeting of the gut-brain axis. Curr. Opin. Pharmacol. 2019, 49, 17–23. [Google Scholar] [CrossRef]
- Xu, F.; Wu, H.; Xie, J.; Zeng, T.; Hao, L.; Xu, W.; Lu, L. The Effects of Fermented Feed on the Growth Performance, Antioxidant Activity, Immune Function, Intestinal Digestive Enzyme Activity, Morphology, and Microflora of Yellow-Feather Chickens. Animals 2023, 13, 3545. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.A.; Liu, Z.; Qian, C.; Wu, J.; Sultana, N.; Zhong, X. Potential effect of the microbial fermented feed utilization on physicochemical traits, antioxidant enzyme and trace mineral analysis in rabbit meat. J. Anim. Physiol. Anim. Nutr. (Berl.) 2020, 104, 767–775. [Google Scholar] [CrossRef]
- Zeng, X.; Zheng, Y.; He, Y.; Zhang, J.; Peng, W.; Su, W. Microbial Metabolism of Naringin and the Impact on Antioxidant Capacity. Nutrients 2022, 14, 3765. [Google Scholar] [CrossRef]
- Lee, S.; Ryu, C.H.; Back, Y.C.; Lee, S.D.; Kim, H. Effect of Fermented Concentrate on Ruminal Fermentation, Ruminal and Fecal Microbiome, and Growth Performance of Beef Cattle. Animals 2023, 13, 3622. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Tao, L.; Zhang, R.; Yang, G. Effects of fermented feed on growth performance, nutrient metabolism and cecal microflora of broilers. Anim. Biosci. 2022, 35, 596–604. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Hack, M.E.; Samak, D.H.; Noreldin, A.E.; El-Naggar, K.; Abdo, M. Probiotics and plant-derived compounds as eco-friendly agents to inhibit microbial toxins in poultry feed: A comprehensive review. Environ. Sci. Pollut. Res. Int. 2018, 25, 31971–31986. [Google Scholar] [CrossRef]
- Jiang, X.; Liu, H.; You, Y.; Zhong, G.; Ruan, Z.; Liao, J.; Zhang, H.; Pan, J.; Tang, Z.; Hu, L. Multi-omics reveals the protective effects of curcumin against AFB1-induced oxidative stress and inflammatory damage in duckling intestines. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2024, 276, 109815. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, J.; Ding, J.; Zhang, Y.; Hou, C.; Shen, W.; Wu, X.; Zhu, J. Effects of hypoxia stress on oxidative stress, apoptosis and microorganisms in the intestine of large yellow croaker (Larimichthys crocea). Aquaculture 2024, 581, 740444. [Google Scholar] [CrossRef]
- Wang, H.; Liu, P.P.; Wei, Z.; Wang, X.W. PcEiger links the Imd/Relish pathway to ROS production in the intestine of the red swamp crayfish. EMBO Rep. 2023, 24, e55903. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Rommelaere, S.; Kondo, S.; Lemaitre, B. Renal Purge of Hemolymphatic Lipids Prevents the Accumulation of ROS-Induced Inflammatory Oxidized Lipids and Protects Drosophila from Tissue Damage. Immunity 2020, 52, 374–387.e376. [Google Scholar] [CrossRef]
- Luo, Q.; Wang, J.; Wang, P.; Liang, X.; Li, J.; Wu, C.; Fang, H.; Ding, S.; Shao, S.; Shi, K. Transcriptomic and genetic approaches reveal that low-light-induced disease susceptibility is related to cellular oxidative stress in tomato. Hortic. Res. 2023, 10, uhad173. [Google Scholar] [CrossRef]
- Zhu, H.; Chen, C.; Zeng, J.; Yun, Z.; Liu, Y.; Qu, H.; Jiang, Y.; Duan, X.; Xia, R. MicroRNA528, a hub regulator modulating ROS homeostasis via targeting of a diverse set of genes encoding copper-containing proteins in monocots. New Phytol. 2020, 225, 385–399. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Jiang, D.; Wang, X.; Jiang, Y.; Sun, Q.; Ling, W.; An, X.; Ji, C.; Li, S.; Qi, Y.; et al. Spermidine improves the antioxidant capacity and morphology of intestinal tissues and regulates intestinal microorganisms in Sichuan white geese. Front. Microbiol. 2023, 14, 1292984. [Google Scholar] [CrossRef]
- Liu, J.X.; Zhu, K.C.; Guo, H.Y.; Liu, B.S.; Zhang, N.; Zhang, D.C. Effects of cysteine addition to low-fishmeal diets on the growth, anti-oxidative stress, intestine immunity, and Streptococcus agalactiae resistance in juvenile golden pompano (Trachinotus ovatus). Front. Immunol. 2022, 13, 1066936. [Google Scholar] [CrossRef]
- Li, W.; Zhang, Y.; Chen, M.; Guo, X.; Ding, Z. The antioxidant strain Lactiplantibacillus plantarum AS21 and Clostridium butyricum ameliorate DSS-induced colitis in mice by remodeling the assembly of intestinal microbiota and improving gut functions. Food Funct. 2024, 5, 2022–2037. [Google Scholar] [CrossRef]
- Han, F.; Fan, H.; Yao, M.; Yang, S.; Han, J. Oral administration of yeast β-glucan ameliorates inflammation and intestinal barrier in dextran sodium sulfate-induced acute colitis. J. Funct. Foods 2017, 35, 115–126. [Google Scholar] [CrossRef]
- Zhao, J.; Zhang, Z.; Zhang, S.; Page, G.; Jaworski, N.W. The role of lactose in weanling pig nutrition: A literature and meta-analysis review. J. Anim. Sci. Biotechnol. 2021, 12, 10. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.; Zhao, S.; Jiang, L.; Lu, L.; Yang, Q.; Yu, Q. Lactobacillus reuteri Stimulates Intestinal Epithelial Proliferation and Induces Differentiation into Goblet Cells in Young Chickens. J. Agric. Food Chem. 2019, 67, 13758–13766. [Google Scholar] [CrossRef]
- Chuang, W.Y.; Lin, L.J.; Hsieh, Y.C.; Chang, S.C.; Lee, T.T. Effects of Saccharomyces cerevisiae and phytase co-fermentation of wheat bran on growth, antioxidation, immunity and intestinal morphology in broilers. Anim. Biosci. 2021, 34, 1157–1168. [Google Scholar] [CrossRef]
- Jacquier, V.; Nelson, A.; Jlali, M.; Rhayat, L.; Brinch, K.S.; Devillard, E. Bacillus subtilis 29784 induces a shift in broiler gut microbiome toward butyrate-producing bacteria and improves intestinal histomorphology and animal performance. Poult. Sci. 2019, 98, 2548–2554. [Google Scholar] [CrossRef]
- Sugiharto, S.; Yudiarti, T.; Isroli, I.; Widiastuti, E.; Wahyuni, H.I.; Sartono, T.A. Growth performance, haematological responses, intestinal microbiology and carcass traits of broiler chickens fed finisher diets containing two-stage fermented banana peel meal. Trop. Anim. Health Prod. 2020, 52, 1425–1433. [Google Scholar] [CrossRef]
- Liu, Y.; Feng, J.; Wang, Y.; Lv, J.; Li, J.; Guo, L.; Min, Y. Fermented Corn-Soybean Meal Mixed Feed Modulates Intestinal Morphology, Barrier Functions and Cecal Microbiota in Laying Hens. Animals 2021, 11, 3059. [Google Scholar] [CrossRef]
- Wu, P.; Golly, M.K.; Guo, Y.; Ma, H.; He, R.; Luo, X.; Luo, S.; Zhang, C.; Zhang, L.; Zhu, J. Effect of partial replacement of soybean meal with high-temperature fermented soybean meal in antibiotic-growth-promoter-free diets on growth performance, organ weights, serum indexes, intestinal flora and histomorphology of broiler chickens. Anim. Feed Sci. Technol. 2020, 269, 114616. [Google Scholar] [CrossRef]
- Yu, J.; Wang, Z.Y.; Yang, H.M.; Xu, L.; Wan, X.L. Effects of cottonseed meal on growth performance, small intestinal morphology, digestive enzyme activities, and serum biochemical parameters of geese. Poult. Sci. 2019, 98, 2066–2071. [Google Scholar] [CrossRef]
- Won, S.; Hamidoghli, A.; Choi, W.; Park, Y.; Jang, W.J.; Kong, I.S.; Bai, S.C. Effects of Bacillus subtilis WB60 and Lactococcus lactis on Growth, Immune Responses, Histology and Gene Expression in Nile tilapia, Oreochromis niloticus. Microorganisms 2020, 8, 67. [Google Scholar] [CrossRef]
- Kuebutornye, F.K.A.; Tang, J.; Cai, J.; Yu, H.; Wang, Z.; Abarike, E.D.; Lu, Y.; Li, Y.; Afriyie, G. In vivo assessment of the probiotic potentials of three host-associated Bacillus species on growth performance, health status and disease resistance of Oreochromis niloticus against Streptococcus agalactiae. Aquaculture 2020, 527, 735440. [Google Scholar] [CrossRef]
- Chen, S.; Mei, H.; Xu, L.; Zhan, L.; Yang, Y.; Zhao, D.; Bao, G.; Li, X.; Cao, Z. Impact of fermented feed of soybean hulls and rapeseed cake on immunity, antioxidant capacity, and gut microbiota in Chahua chicken. Poult. Sci. 2024, 103, 103451. [Google Scholar] [CrossRef]
- Yaman, M.A.; Yunita, T.; Daud, M.; Jeksi, S. Effect of wet fermented diet containing a combination of maggot flour (Hermetia ilucen) and active digestive enzymes on growth and protein retention of hybrid chickens in the early phase of growth. In Proceedings of the IOP Conference Series: Earth and Environmental Science, Banda Aceh, Indonesia, 1 May 2023; p. 012086. [Google Scholar]
- Koo, B.; Bustamante-García, D.; Nyachoti, C.M. Energy content and nutrient digestibility of diets containing Lactobacillus-fermented barley or wheat fed to weaned pigs. J. Anim. Sci. 2018, 96, 4802–4811. [Google Scholar] [CrossRef]
- Zhang, A.R.; Wei, M.; Yan, L.; Zhou, G.L.; Li, Y.; Wang, H.M.; Yang, Y.Y.; Yin, W.; Guo, J.Q.; Cai, X.H.; et al. Effects of feeding solid-state fermented wheat bran on growth performance and nutrient digestibility in broiler chickens. Poult. Sci. 2022, 101, 101402. [Google Scholar] [CrossRef]
- Kongphitee, K.; Sommart, K.; Phonbumrung, T.; Gunha, T.; Suzuki, T. Feed intake, digestibility and energy partitioning in beef cattle fed diets with cassava pulp instead of rice straw. Asian-Australas. J. Anim. Sci. 2018, 31, 1431–1441. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhang, C.; Du, M.; Wang, Y.; Zhang, G.; Lee, Y. Effects of dietary supplementation with different fermented feeds on performance, nutrient digestibility, and serum biochemical indexes of fattening lambs. Anim. Biosci. 2021, 34, 633–641. [Google Scholar] [CrossRef]
- Cao, S.; Hou, L.; Sun, L.; Gao, J.; Gao, K.; Yang, X.; Jiang, Z.; Wang, L. Intestinal morphology and immune profiles are altered in piglets by early-weaning. Int. Immunopharmacol. 2022, 105, 108520. [Google Scholar] [CrossRef] [PubMed]
- Xin, H.; Wang, M.; Xia, Z.; Yu, B.; He, J.; Yu, J.; Mao, X.; Huang, Z.; Luo, Y.; Luo, J.; et al. Fermented Diet Liquid Feeding Improves Growth Performance and Intestinal Function of Pigs. Animals 2021, 11, 1452. [Google Scholar] [CrossRef]
- Wang, H.; Kim, I.H. Evaluation of Dietary Probiotic (Lactobacillus plantarum BG0001) Supplementation on the Growth Performance, Nutrient Digestibility, Blood Profile, Fecal Gas Emission, and Fecal Microbiota in Weaning Pigs. Animals 2021, 11, 2232. [Google Scholar] [CrossRef]
- Wang, W.; Wang, Y.; Hao, X.; Duan, Y.; Meng, Z.; An, X.; Qi, J. Dietary fermented soybean meal replacement alleviates diarrhea in weaned piglets challenged with enterotoxigenic Escherichia coli K88 by modulating inflammatory cytokine levels and cecal microbiota composition. BMC Vet. Res. 2020, 16, 245. [Google Scholar] [CrossRef]
- Guo, L.; Lv, J.; Liu, Y.; Ma, H.; Chen, B.; Hao, K.; Feng, J.; Min, Y. Effects of Different Fermented Feeds on Production Performance, Cecal Microorganisms, and Intestinal Immunity of Laying Hens. Animals 2021, 11, 2799. [Google Scholar] [CrossRef]
- Wang, J.; Ji, H.; Wang, S.; Liu, H.; Zhang, W.; Zhang, D.; Wang, Y. Probiotic Lactobacillus plantarum Promotes Intestinal Barrier Function by Strengthening the Epithelium and Modulating Gut Microbiota. Front. Microbiol. 2018, 9, 1953. [Google Scholar] [CrossRef]
- Yang, Z.; Liu, P.; Kong, Q.; Deng, Y.; Zhang, W.; Xu, G.; Tang, H. Effects of Co-Fermented Feed Using Lactobacillus acidophilus, Limosilactobacillus reuteri and Lactiplantibacillus plantarum on Growth, Antioxidant Capacity, Fatty Acids and Gut Microbiota of Largemouth Bass (Micropterus salmoides). Fishes 2023, 8, 433. [Google Scholar] [CrossRef]
- Tian, Y.; Zhang, R.; Li, G.; Zeng, T.; Chen, L.; Xu, W.; Gu, T.; Tao, Z.; Du, X.; Lu, L. Microbial fermented feed affects flavor amino acids and yolk trimethylamine of duck eggs via cecal microbiota-yolk metabolites crosstalk. Food Chem. 2024, 430, 137008. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Liu, X.; Zhang, K. Effects of Microbial Fermented Feed on Serum Biochemical Profile, Carcass Traits, Meat Amino Acid and Fatty Acid Profile, and Gut Microbiome Composition of Finishing Pigs. Front. Vet. Sci. 2021, 8, 744630. [Google Scholar] [CrossRef]
- Cook, L.; Rees, W.D.; Wong, M.Q.; Peters, H.; Levings, M.K.; Steiner, T.S. Fecal Microbiota Transplantation for Recurrent Clostridioides difficile Infection Enhances Adaptive Immunity to C difficile Toxin B. Gastroenterology 2021, 160, 2155–2158.e2154. [Google Scholar] [CrossRef]
- Nzabarushimana, E.; Tang, H. Functional profile of host microbiome indicates Clostridioides difficile infection. Gut Microbes 2022, 14, 2135963. [Google Scholar] [CrossRef]
- Hao, X.; Ding, N.; Zhang, Y.; Yang, Y.; Zhao, Y.; Zhao, J.; Li, Y.; Li, Z. Benign regulation of the gut microbiota: The possible mechanism through which the beneficial effects of manual acupuncture on cognitive ability and intestinal mucosal barrier function occur in APP/PS1 mice. Front. Neurosci. 2022, 16, 960026. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.W.; Li, R.Q.; An, J.X.; Xie, T.Q.; Han, Z.Y.; Xu, R.; Fang, Y.; Zhang, X.Z. Prebiotics-Encapsulated Probiotic Spores Regulate Gut Microbiota and Suppress Colon Cancer. Adv. Mater. 2020, 32, e2004529. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Jiang, Z.; Zhang, Z.; Liu, T.; Fan, Y.; Liu, T.; Peng, N. Bacillus coagulans in Combination with Chitooligosaccharides Regulates Gut Microbiota and Ameliorates the DSS-Induced Colitis in Mice. Microbiol. Spectr. 2022, 10, e0064122. [Google Scholar] [CrossRef]
- Oh, N.S.; Lee, J.Y.; Kim, Y.T.; Kim, S.H.; Lee, J.H. Cancer-protective effect of a synbiotic combination between Lactobacillus gasseri 505 and a Cudrania tricuspidata leaf extract on colitis-associated colorectal cancer. Gut Microbes 2020, 12, 1785803. [Google Scholar] [CrossRef]
- Huang, Y.; Zhang, K.; Guo, W.; Zhang, C.; Chen, H.; Xu, T.; Lu, Y.; Wu, Q.; Li, Y.; Chen, Y. Aspergillus niger fermented Tartary buckwheat ameliorates obesity and gut microbiota dysbiosis through the NLRP3/Caspase-1 signaling pathway in high-fat diet mice. J. Funct. Foods 2022, 95, 105171. [Google Scholar] [CrossRef]
- O’ Meara, F.M.; Gardiner, G.E.; O’Doherty, J.V.; Clarke, D.; Cummins, W.; Lawlor, P.G. Effect of wet/dry, fresh liquid, fermented whole diet liquid, and fermented cereal liquid feeding on feed microbial quality and growth in grow-finisher pigs. J. Anim. Sci. 2020, 98, skaa166. [Google Scholar] [CrossRef]
- Zamojska, D.; Nowak, A.; Nowak, I.; Macierzyńska-Piotrowska, E. Probiotics and Postbiotics as Substitutes of Antibiotics in Farm Animals: A Review. Animals 2021, 11, 3431. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Liu, J.; Jin, X.; Liu, C.; Fan, C.; Guo, L.; Liang, Y.; Zheng, J.; Peng, N. Developmental, Dietary, and Geographical Impacts on Gut Microbiota of Red Swamp Crayfish (Procambarus clarkii). Microorganisms 2020, 8, 1376. [Google Scholar] [CrossRef]
- Hajimohammadi, A.; Mottaghitalab, M.; Hashemi, M. Effects of microbial fermented sesame meal and enzyme supplementation on the intestinal morphology, microbiota, pH, tibia bone and blood parameters of broiler chicks. Ital. J. Anim. Sci. 2020, 19, 457–467. [Google Scholar] [CrossRef]
- Al-Khalaifah, H.S.; Shahin, S.E.; Omar, A.E.; Mohammed, H.A.; Mahmoud, H.I.; Ibrahim, D. Effects of graded levels of microbial fermented or enzymatically treated dried brewer’s grains on growth, digestive and nutrient transporter genes expression and cost effectiveness in broiler chickens. BMC Vet. Res. 2020, 16, 424. [Google Scholar] [CrossRef] [PubMed]
- Taranu, I.; Nguyen, T.T.; Pham, K.D.; Gras, M.A.; Pistol, G.C.; Marin, D.E.; Rotar, C.; Habeanu, M.; Ho, P.H.; Le, T.M. Rice and Cassava Distillers Dried Grains in Vietnam: Nutritional Values and Effects of Their Dietary Inclusion on Blood Chemical Parameters and Immune Responses of Growing Pigs. Waste Biomass Valorization 2019, 10, 3373–3382. [Google Scholar] [CrossRef]
- Zhu, F.; Zhang, B.; Li, J.; Zhu, L. Effects of fermented feed on growth performance, immune response, and antioxidant capacity in laying hen chicks and the underlying molecular mechanism involving nuclear factor-κB. Poult. Sci. 2020, 99, 2573–2580. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Jiang, Z.; Qiu, Y.; Wang, L. 447 Effects of probiotic solid-state fermented complete feed on growth performance and pork quality of finishing pigs. J. Anim. Sci. 2018, 96, 232. [Google Scholar] [CrossRef]
- Fan, L.; Dou, M.; Wang, X.; Han, Q.; Zhao, B.; Hu, J.; Yang, G.; Shi, X.; Li, X. Fermented corn-soybean meal elevated IGF1 levels in grower-finisher pigs. J. Anim. Sci. 2018, 96, 5144–5151. [Google Scholar] [CrossRef]
- Cheng, Y.; Wu, T.; Chu, X.; Tang, S.; Cao, W.; Liang, F.; Fang, Y.; Pan, S.; Xu, X. Fermented blueberry pomace with antioxidant properties improves fecal microbiota community structure and short chain fatty acids production in an in vitro mode. LWT 2020, 125, 109260. [Google Scholar] [CrossRef]
- Wang, X.; Hu, K.; Chen, Y.; Lai, J.; Zhang, M.; Li, J.; Li, Q.; Zhao, N.; Liu, S. Effect of Lactiplantibacillus plantarum fermentation on the physicochemical, antioxidant activity and immunomodulatory ability of polysaccharides from Lvjian okra. Int. J. Biol. Macromol. 2024, 257, 128649. [Google Scholar] [CrossRef]
- Duan, W.; Zhou, L.; Ren, Y.; Liu, F.; Xue, Y.; Wang, F.Z.; Lu, R.; Zhang, X.J.; Shi, J.S.; Xu, Z.H.; et al. Lactic acid fermentation of goji berries (Lycium barbarum) prevents acute alcohol liver injury and modulates gut microbiota and metabolites in mice. Food Funct. 2024, 15, 1612–1626. [Google Scholar] [CrossRef] [PubMed]
- Ning, Y.; Wang, S.; Sun, Y.; Zhang, S.; Wen, Y.; Zou, D.; Zhou, D. Deciphering survival strategies: Oxidative stress and microbial interplay in Eisenia fetida under tetracycline contamination. Sci. Total Environ. 2024, 909, 168647. [Google Scholar] [CrossRef]
- Collart, L.; Jiang, D.; Halsey, K.H. The volatilome reveals microcystin concentration, microbial composition, and oxidative stress in a critical Oregon freshwater lake. mSystems 2023, 8, e0037923. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Lin, Y.; Miao, L.; Pan, W.; Ge, X. Ferulic acid alleviates lipopolysaccharide-induced acute liver injury in Megalobrama amblycephala. Aquaculture 2021, 532, 735972. [Google Scholar] [CrossRef]
- Dai, X.; Xing, C.; Cao, H.; Luo, J.; Wang, T.; Liu, P.; Guo, X.; Hu, G.; Zhang, C. Alterations of mitochondrial antioxidant indexes and apoptosis in duck livers caused by Molybdenum or/and cadmium. Chemosphere 2018, 193, 574. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Yang, Q.; Liang, H.; Ren, M.; Ge, X.; Ji, K. Effects of stocking density and dietary phosphorus levels on the growth performance, antioxidant capacity, and nitrogen and phosphorus emissions of juvenile blunt snout bream (Megalobrama amblycephala). Aquac. Nutr. 2021, 27, 581–591. [Google Scholar] [CrossRef]
- Dong, J.; Ping, L.; Xie, Q.; Liu, D.; Zhao, L.; Evivie, S.E.; Wang, Z.; Li, B.; Huo, G. Lactobacillus plantarum KLDS1.0386 with antioxidant capacity ameliorates the lipopolysaccharide-induced acute liver injury in mice by NF-κB and Nrf2 pathway. Food Biosci. 2022, 47, 101589. [Google Scholar] [CrossRef]
- Hao, L.; Su, W.; Zhang, Y.; Wang, C.; Lu, Z. Effects of Supplementing with Fermented Mixed Feed on the Performance and Meat Quality in Finishing Pigs. Anim. Feed Sci. Technol. 2020, 266, 114501. [Google Scholar] [CrossRef]
- Chen, Q.; Wang, R.; Wang, Y.; An, X.; Qi, J. Characterization and antioxidant activity of wheat bran polysaccharides modified by Saccharomyces cerevisiae and Bacillus subtilis fermentation. J. Cereal Sci. 2020, 97, 103157. [Google Scholar] [CrossRef]
- Czech, A.; Sembratowicz, I.; Kiesz, M. The Effects of a Fermented Rapeseed or/and Soybean Meal Additive on Antioxidant Parameters in the Blood and Tissues of Piglets. Animals 2021, 11, 1646. [Google Scholar] [CrossRef] [PubMed]
- Kalinowski, C.T.; Betancor, M.B.; Torrecillas, S.; Sprague, M.; Larroquet, L.; Véron, V.; Panserat, S.; Izquierdo, M.S.; Kaushik, S.J.; Fontagné-Dicharry, S. More Than an Antioxidant: Role of Dietary Astaxanthin on Lipid and Glucose Metabolism in the Liver of Rainbow Trout (Oncorhynchus mykiss). Antioxidants 2023, 12, 136. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.; Jin, M.; Yuan, Y.; Luo, J.-X.; Lu, Y.; Zhou, Q.-C.; Tan, Z.-L. Dietary nucleotide-rich yeast supplementation improves growth, innate immunity and intestinal morphology of Pacific white shrimp (Litopenaeus vannamei). Aquac. Nutr. 2018, 24, 1425–1435. [Google Scholar] [CrossRef]
- Long, S.; He, T.; Kim, S.W.; Shang, Q.; Kiros, T.; Mahfuz, S.U.; Wang, C.; Piao, X. Live Yeast or Live Yeast Combined with Zinc Oxide Enhanced Growth Performance, Antioxidative Capacity, Immunoglobulins and Gut Health in Nursery Pigs. Animals 2021, 11, 1626. [Google Scholar] [CrossRef] [PubMed]
- Gu, T.; Duan, M.; Zhang, R.; Zeng, T.; Xu, W.; Feng, W.; Jiang, C.; Tian, Y.; Chen, L.; Lu, L. Probiotic Fermented Feed Alleviates Liver Fat Deposition in Shaoxing Ducks via Modulating Gut Microbiota. Front. Microbiol. 2022, 13, 928670. [Google Scholar] [CrossRef]
- Peng, W.; Talpur, M.Z.; Zeng, Y.; Xie, P.; Li, J.; Wang, S.; Wang, L.; Zhu, X.; Gao, P.; Jiang, Q.; et al. Influence of fermented feed additive on gut morphology, immune status, and microbiota in broilers. BMC Vet. Res. 2022, 18, 218. [Google Scholar] [CrossRef]
- Hanum, F.; Atsuta, Y.; Daimon, H. Methane Production Characteristics of an Anaerobic Co-Digestion of Pig Manure and Fermented Liquid Feed. Molecules 2022, 27, 6509. [Google Scholar] [CrossRef]
- Lu, Y.; Zhang, R.; Lei, H.; Hang, Y.; Xue, H.; Cai, X.; Lu, Y. Supplementation with Fermented Feedstuff Enhances Orexin Expression and Secretion Associated with Increased Feed Intake and Weight Gain in Weaned Pigs. Animals 2022, 12, 1329. [Google Scholar] [CrossRef]




| Unconventional Fermented Feed Feeding | |||||
|---|---|---|---|---|---|
| Animal | Raw Materials | Probiotics | Regulated Items | Antioxidant Substance | References |
| Boer goats | Pennisetum giganteum | Bacillus coagulans preparation | Abundance of Lactobacillus and unidentified Clostridiales ↑ Anaerovibrio and Methanobrevibacter ↓ | CAT, GSH-Px activities and glutathione ↑ | Qiu et al. [26] |
| Laying hens | Corn–soybean meal wheat bran | Bacillus subtilis and Saccharomyces cerevisiae | In relative Lactobacillus, Megasphaera, and Peptococcus abundance ↑ Campylobacter abundance ↓ | Immunoglobulin A, immunoglobulin M, and immunoglobulin G ↑ | Guo et al. [27] |
| Broilers | Corn, soybean meal, corn–gluten meal, and corn dried distillers’ grains | Lactobacillus plantarum, Bacillus subtilis, and Saccharomyces cerevisiae | Abundance of Ruminococcaceae, Lactobacillaceae, and unclassified Clostridiales ↑ Abundance of Rikenellaceae, Lachnospiraceae, and Bacteroidaceae ↓ | Acetic acid, propionic acid, butyric acid, and lactic acid ↑ | Zhu et al. [28] |
| Laying hens | Astragalus | Lactobacillus plantarum | CAT, GSH-Px, superoxide dismutase and total antioxidant capacity in serum ↑ | CAT ↑ | Hong et al. [29] |
| Cobb male broilers | Corn–soybean meal | Lactobacillus acidophilus | Body weight, ADG, average daily feed intake, and jejunum and ileum V:C ratio at 14 d and 21 d ↑ | The mRNA expression of inducible nitric oxide synthase, interleukin-8, and interleukin-1β in the jejunum ↓ | Wu et al. [30] |
| Nursery pig | Corn–soybean meal | Lactobacillus plantarum and Pediococcus acidilactici | ADG and significantly increased fecal acetate, butyrate, and total short-chain fatty acid concentrations ↑ | Short-chain fatty acid ↑ | Yang et al. [31] |
| Berkshire pigs | Rubus coreanus | Lactobacillus plantarum | The mRNA expression of transcription factors and cytokines in Th1 and Treg cells ↑ The mRNA expression of T helper cell 2 and Th17 transcription factors and cytokines ↓ | The mRNA expression of transcription factors and cytokines in Th1 and Treg cells ↑ | Yu et al. [32] |
| Cyprinus carpio | Wheat, soybean meal, corn–gluten meal, chicken meal | C. somerae XMX-1, S. cerevisiae GCC−1, L. rhamnosus GCC-3, and B. subtilis HGcc-1 | Health and production ↑ | Liver anti-inflammatory factors transforming growth Factor-β↑ | Zhang et al. [33] |
| Juvenile olive flounder | Garlic husks, Tuna | Bacillus licheniformis and Bacillus subtilis | Weight gain, specific growth rate, and feed efficiency ↑ | Sucrose reductase↑ | Fatma et al. [34] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lian, X.; Shi, M.; Liang, Y.; Lin, Q.; Zhang, L. The Effects of Unconventional Feed Fermentation on Intestinal Oxidative Stress in Animals. Antioxidants 2024, 13, 305. https://doi.org/10.3390/antiox13030305
Lian X, Shi M, Liang Y, Lin Q, Zhang L. The Effects of Unconventional Feed Fermentation on Intestinal Oxidative Stress in Animals. Antioxidants. 2024; 13(3):305. https://doi.org/10.3390/antiox13030305
Chicago/Turabian StyleLian, Xiao, Mingyu Shi, Ying Liang, Qinlu Lin, and Lingyu Zhang. 2024. "The Effects of Unconventional Feed Fermentation on Intestinal Oxidative Stress in Animals" Antioxidants 13, no. 3: 305. https://doi.org/10.3390/antiox13030305
APA StyleLian, X., Shi, M., Liang, Y., Lin, Q., & Zhang, L. (2024). The Effects of Unconventional Feed Fermentation on Intestinal Oxidative Stress in Animals. Antioxidants, 13(3), 305. https://doi.org/10.3390/antiox13030305







