Apples and Apple By-Products: Antioxidant Properties and Food Applications
Abstract
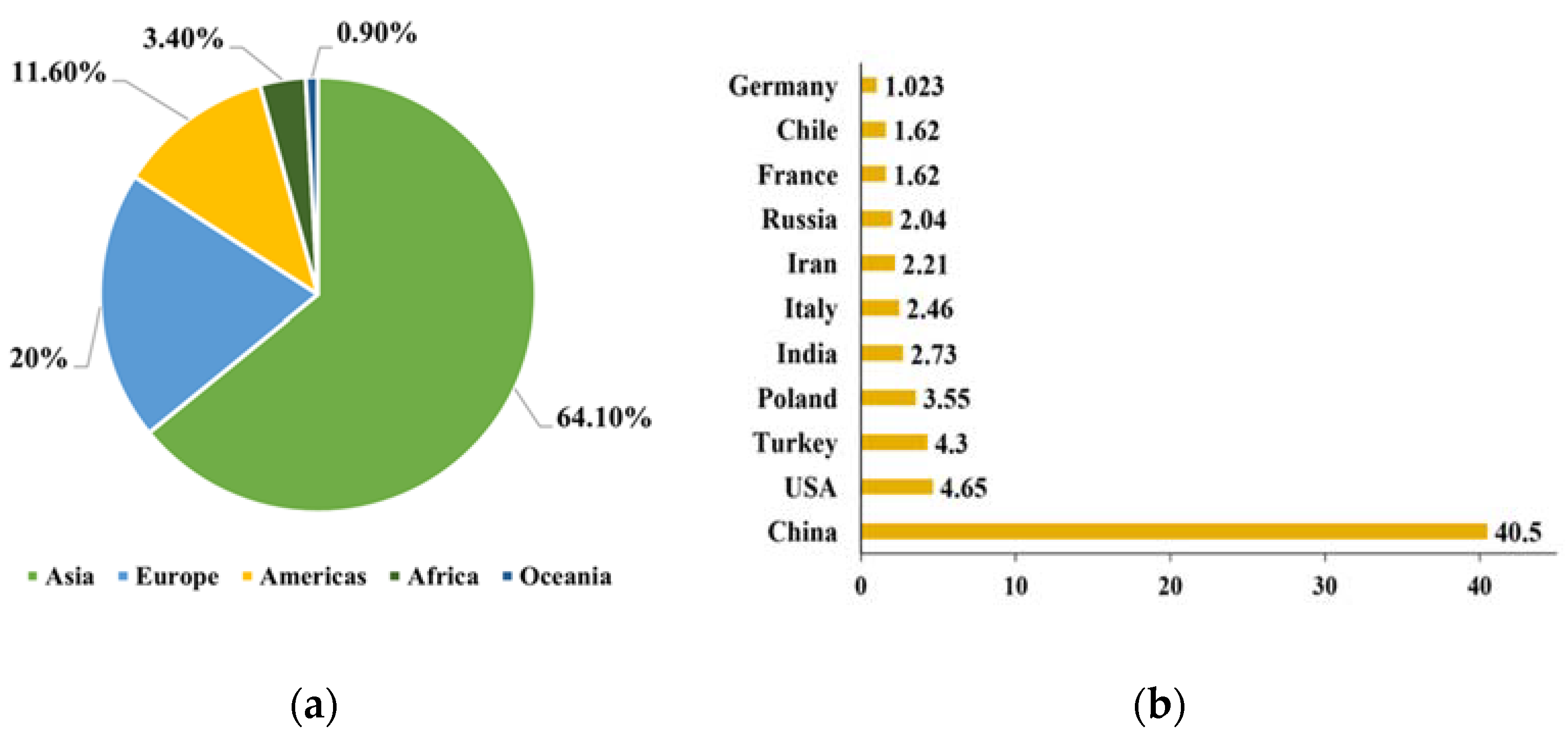
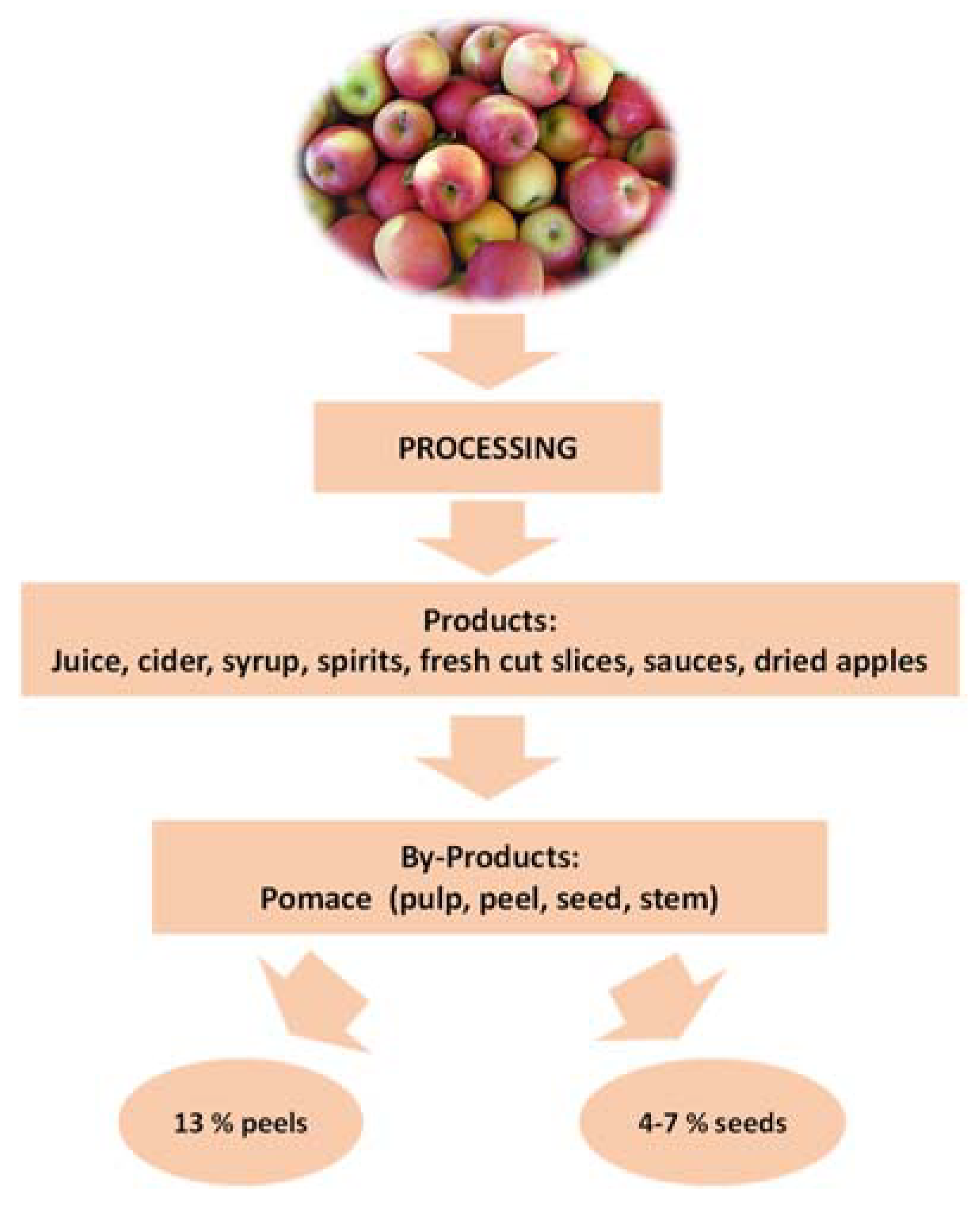
1. Introduction
2. Antioxidant Compounds Present in Apples and Apple By-Products
2.1. Vitamins
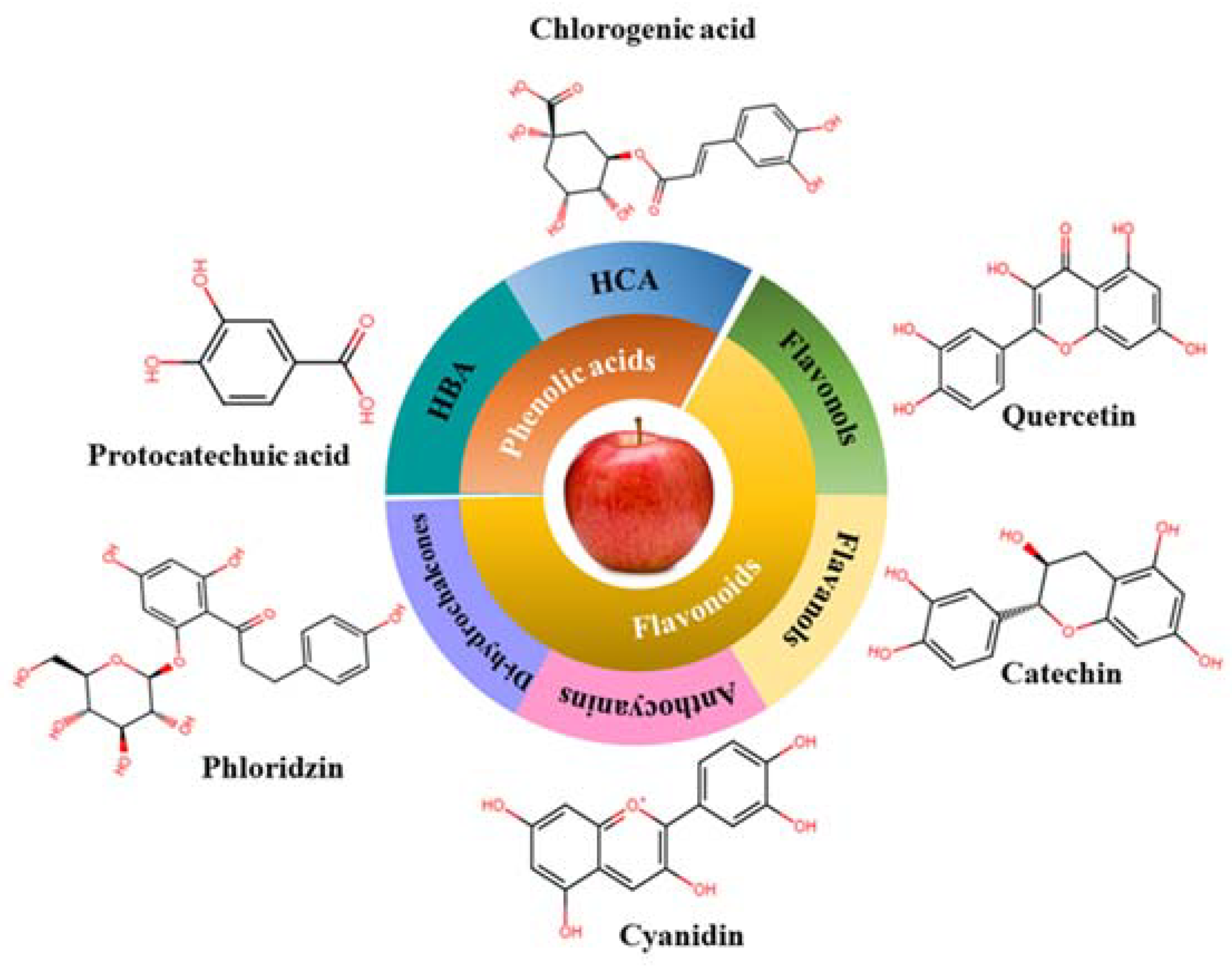
2.2. Phenolic Compounds
2.2.1. Phenolic Acids
2.2.2. Flavonoids
3. Extraction of Phenolic Compounds from Apples and Apple By-Products
3.1. Conventional Extraction
3.2. Innovative Extraction
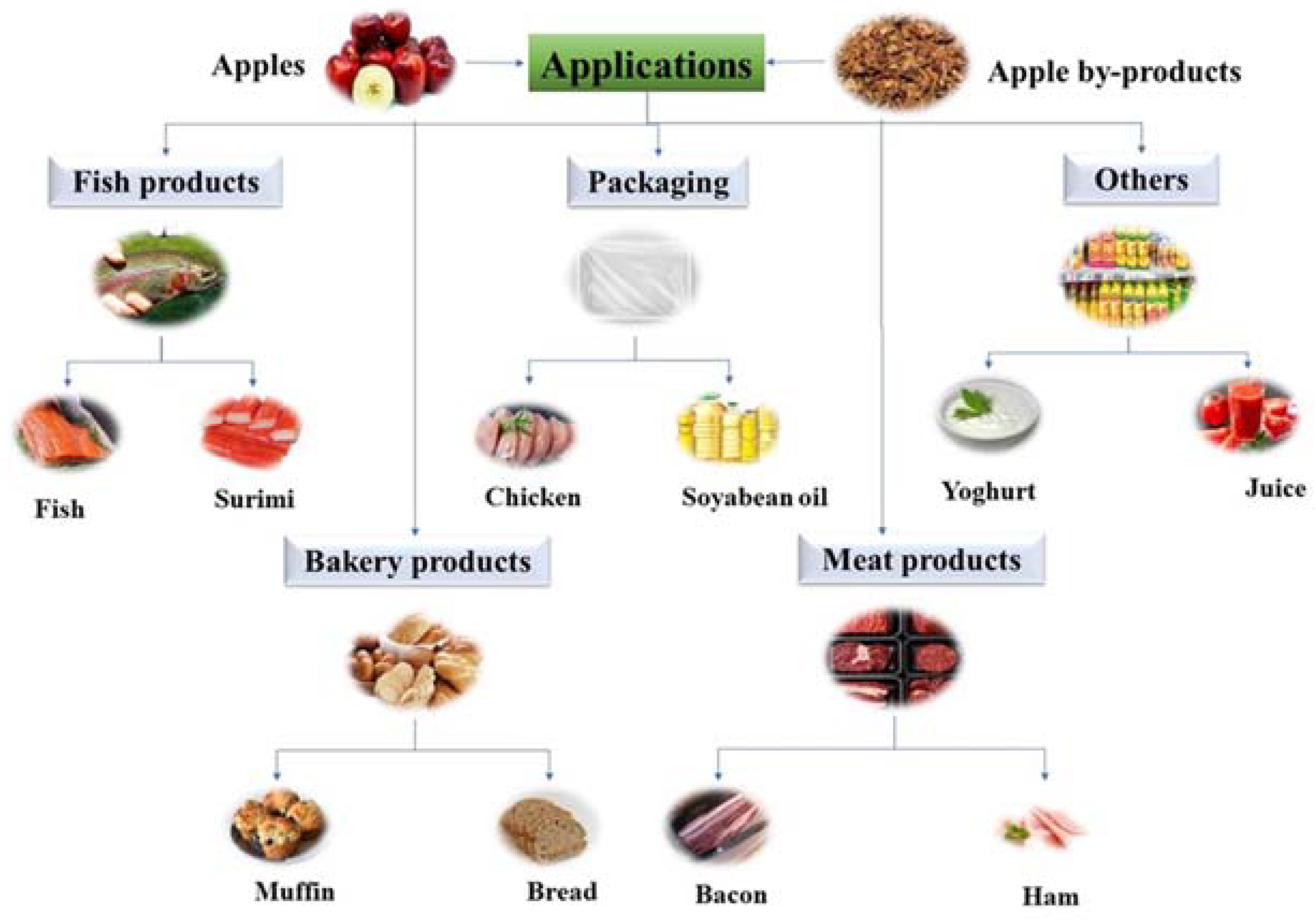
4. Applications of Apple Phenolic Compounds in Food Products
4.1. Bread and Bakery Products
4.2. Fish and Fish Products
4.3. Meat and Meat Products
4.4. Functional Packaging Materials
4.5. Other Products
| Food Product | Formulation | Apple Variety | Outcomes | References | |
|---|---|---|---|---|---|
| Bakery | Bread | Incorporation of defatted apple seed powder (5% and 20%) | Golden Delicious, Idared, and Šumatovka | Incorporation of 5% apple seed powder: TPC and TAC increased 1.7- and 1.1-fold Incorporation of 20% apple seed powder: TPC and TAC increased 2.9- and 2.1-fold | [58] |
| Gluten-free bread | Addition of apple pomace (up to 12.5%) | - | Satisfactory sensory quality TPC increased TAC increased 5.8-fold (ORAC) | [64] | |
| Muffins | Addition of apple peel powder (up to 32%) | Idared and Northern Spy | TAC increased 4.9-fold (FRAP) | [61] | |
| Cookies | Addition of apple pomace powder (5%, 10%, 15%, 20%, and 25%) | - | TPC increased | [62] | |
| Biscuits | Addition of apple pomace (10% and 20%) | Golden Delicious | TPC increase and reduced glycemic index (65 and 60, respectively) | [65] | |
| Fish and fish products | Rainbow trout fish | Apple peel extracts (from 10 to 100 mg/kg of fish) | - | TPC and TAC increased | [67] |
| Surimi | Young apple extracts | Qinguan | Lipid and protein oxidation retarded | [69] | |
| Fish oil | Apple peel extracts (400 µg/mL of fish oil) | Northern Spy | TPC and TAC increased (ORAC and FRAP); lipid oxidation retarded | [66] | |
| Meat and meat products | Ham | Apple extracts (300 mg/kg) | - | Lipid oxidation retarded | [70] |
| Beef jerky | Wet apple pomace (10 and 20%) | - | TPC and TAC increased | [63] | |
| Chicken sausages | Apple pomace (3%, 4%, and 9%) | - | TAC increased | [71] | |
| Bacon | Apple polyphenol powder (300 mg/kg) | - | Lipid and protein oxidation retarded | [72] | |
| Mutton | Apple pomace powder (1%, 3%, and 5%) | - | Lipid oxidation retarded | [73] | |
| Polyvinyl alcohol matrix | Apple pomace powder (1%, 5%, and 10%) | - | TPC and RSA increased; Lipid oxidation retarded | [74] | |
| Chitosan film | Young apple polyphenols (1% w/v) | - | RSA increased; Lipid and protein oxidation retarded | [77,78] | |
| Functional packaging materials | Edible coating material | Apple peel powder | Fuji | Lipid oxidation and microbial growth retarded | [80] |
| Edible film | Apple polyphenols powder (from 40 to 80 mg/mL) | - | RSA increased; Lipid oxidation retarded | [76] | |
| Others | Juices (Carrot and tomato) | Apple peel extracts (from 40 to 800 mg/L) | Bramley | TAC increased | [82] |
| Yoghurt | Apple pomace extracts (3.3%) | Royal Gala | TPC and TAC increased | [83] | |
| Probiotic yoghurt | Apple peel extracts (1–5%) | Red Delicious Golden Royal Gala | TPC and antioxidant capacity increased | [84] |
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Arias, A.; Feijoo, G.; Moreira, M.T. Exploring the Potential of Antioxidants from Fruits and Vegetables and Strategies for Their Recovery. Innov. Food Sci. Emerg. Technol. 2022, 77, 102974. [Google Scholar] [CrossRef]
- Fraga, C.G.; Croft, K.D.; Kennedy, D.O.; Tomás-Barberán, F.A. The Effects of Polyphenols and Other Bioactives on Human Health. Food Funct. 2019, 10, 514–528. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, J.M.; Pateiro, M.; Domínguez, R.; Barba, F.J.; Putnik, P.; Kovačević, D.B.; Shpigelman, A.; Granato, D.; Franco, D. Berries Extracts as Natural Antioxidants in Meat Products: A Review. Food Res. Int. 2018, 106, 1095–1104. [Google Scholar] [CrossRef] [PubMed]
- Lyu, F.; Luiz, S.F.; Azeredo, D.R.P.; Cruz, A.G.; Ajlouni, S.; Ranadheera, C.S. Apple Pomace as a Functional and Healthy Ingredient in Food Products: A Review. Processes 2020, 8, 319. [Google Scholar] [CrossRef]
- Domínguez, R.; Gullon, P.; Pateiro, M.; Munekata, P.E.S.; Zhang, W.; Lorenzo, J.M. Tomato as Potential Source of Natural Additives for Meat Industry. A Review. Antioxidant 2020, 9, 73. [Google Scholar] [CrossRef]
- Domínguez, R.; Pateiro, M.; Gagaoua, M.; Barba, F.J.; Zhang, W.; Lorenzo, J.M. A Comprehensive Review on Lipid Oxidation in Meat and Meat Products. Antioxidants 2019, 8, 429. [Google Scholar] [CrossRef]
- Ribeiro, J.S.; Santos, M.J.M.C.; Silva, L.K.R.; Pereira, L.C.L.; Santos, I.A.; da Silva Lannes, S.C.; da Silva, M.V. Natural Antioxidants Used in Meat Products: A Brief Review. Meat Sci. 2019, 148, 181–188. [Google Scholar] [CrossRef]
- Halliwell, B. Letters to the Editors. Defin. Meas. Antioxid. Free Radic. Biol. Med. 1995, 18, 125–126. [Google Scholar] [CrossRef]
- Ozsoy, N.; Candoken, E.; Akev, N. Implications for Degenerative Disorders. Oxid. Med. Cell. Longev. 2009, 2, 99–106. [Google Scholar] [CrossRef]
- Agati, G.; Matteini, P.; Goti, A.; Tattini, M. Chloroplast-Located Flavonoids Can Scavenge Singlet Oxygen. New Phytol. 2007, 174, 77–89. [Google Scholar] [CrossRef]
- Ćetković, G.; Čanadanović-Brunet, J.; Djilas, S.; Savatović, S.; Mandić, A.; Tumbas, V. Assessment of Polyphenolic Content and in Vitro Antiradical Characteristics of Apple Pomace. Food Chem. 2008, 109, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Raudone, L.; Raudonis, R.; Liaudanskas, M.; Janulis, V.; Viskelis, P. Phenolic Antioxidant Profiles in the Whole Fruit, Flesh and Peel of Apple Cultivars Grown in Lithuania. Sci. Hortic. 2017, 216, 186–192. [Google Scholar] [CrossRef]
- Kalinowska, M.; Gryko, K.; Wróblewska, A.M.; Jabłońska-Trypuć, A.; Karpowicz, D. Phenolic Content, Chemical Composition and Anti-/pro-Oxidant Activity of Gold Milenium and Papierowka Apple Peel Extracts. Sci. Rep. 2020, 10, 14951. [Google Scholar] [CrossRef]
- Cömert, E.D.; Mogol, B.A.; Gökmen, V. Relationship between Color and Antioxidant Capacity of Fruits and Vegetables. Curr. Res. Food Sci. 2020, 2, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Al Daccache, M.; Koubaa, M.; Maroun, R.G.; Salameh, D.; Louka, N.; Vorobiev, E. Impact of the Physicochemical Composition and Microbial Diversity in Apple Juice Fermentation Process: A Review. Molecules 2020, 25, 3698. [Google Scholar] [CrossRef] [PubMed]
- Cruz, M.G.; Bastos, R.; Pinto, M.; Ferreira, J.M.; Santos, J.F.; Wessel, D.F.; Coelho, E.; Coimbra, M.A. Waste Mitigation: From an Effluent of Apple Juice Concentrate Industry to a Valuable Ingredient for Food and Feed Applications. J. Clean. Prod. 2018, 193, 652–660. [Google Scholar] [CrossRef]
- Dugmore, T.I.J.; Clark, J.H.; Bustamante, J.; Houghton, J.A.; Matharu, A.S. Valorisation of Biowastes for the Production of Green Materials Using Chemical Methods. Top. Curr. Chem. 2017, 375, 46. [Google Scholar] [CrossRef]
- Mirzaei-Aghsaghali, A.; Maheri-Sis, N. Nutritive Value of Some Agro-Industrial by-Products for Ruminants—A Review. World J. Zool. 2008, 3, 40–46. [Google Scholar]
- Parfitt, J.; Barthel, M.; MacNaughton, S. Food waste within food supply chains: Quantification and potential for change to 2050. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 3065–3081. [Google Scholar] [CrossRef]
- de la Rosa, L.A.; Moreno-Escamilla, J.O.; Rodrigo-García, J.; Alvarez-Parrilla, E. Phenolic Compounds; Elsevier Inc.: Amsterdam, The Netherlands, 2019; ISBN 9780128132784. [Google Scholar]
- Skinner, R.C.; Gigliotti, J.C.; Ku, K.M.; Tou, J.C. A Comprehensive Analysis of the Composition, Health Benefits, and Safety of Apple Pomace. Nutr. Rev. 2018, 76, 893–909. [Google Scholar] [CrossRef]
- Campeanu, G.; Neata, G.; Darjanschi, G. Chemical Composition of the Fruits of Several Apple Cultivars Growth as Biological Crop. Not. Bot. Horti Agrobot. Cluj-Napoca 2009, 37, 161–164. [Google Scholar]
- da Silva, L.C.; Viganó, J.; de Souza Mesquita, L.M.; Dias, A.L.B.; de Souza, M.C.; Sanches, V.L.; Chaves, J.O.; Pizani, R.S.; Contieri, L.S.; Rostagno, M.A. Recent Advances and Trends in Extraction Techniques to Recover Polyphenols Compounds from Apple By-Products. Food Chem. 2021, 12, 100133. [Google Scholar] [CrossRef] [PubMed]
- Trigo, J.P.; Alexandre, E.M.C.; Saraiva, J.A.; Pintado, M.E. High Value-Added Compounds from Fruit and Vegetable by-Products–Characterization, Bioactivities, and Application in the Development of Novel Food Products. Crit. Rev. Food Sci. Nutr. 2020, 60, 1388–1416. [Google Scholar] [CrossRef] [PubMed]
- Barros, L.; Dueñas, M.; Ferreira, I.C.F.R.; Baptista, P.; Santos-Buelga, C. Phenolic Acids Determination by HPLC-DAD-ESI/MS in Sixteen Different Portuguese Wild Mushrooms Species. Food Chem. Toxicol. 2009, 47, 1076–1079. [Google Scholar] [CrossRef]
- Kschonsek, J.; Wolfram, T.; Stöckl, A.; Böhm, V. Polyphenolic Compounds Analysis of Old and New Apple Cultivars and Contribution of Polyphenolic Profile to the in Vitro Antioxidant Capacity. Antioxidants 2018, 7, 20. [Google Scholar] [CrossRef]
- Bondonno, N.P.; Bondonno, C.P.; Ward, N.C.; Hodgson, J.M.; Croft, K.D. The Cardiovascular Health Benefits of Apples: Whole Fruit vs. Isolated Compounds. Trends Food Sci. Technol. 2017, 69, 243–256. [Google Scholar] [CrossRef]
- Nkuimi Wandjou, J.G.; Lancioni, L.; Barbalace, M.C.; Hrelia, S.; Papa, F.; Sagratini, G.; Vittori, S.; Dall’Acqua, S.; Caprioli, G.; Beghelli, D. Comprehensive Characterization of Phytochemicals and Biological Activities of the Italian Ancient Apple ‘Mela Rosa Dei Monti Sibillini’. Food Res. Int. 2020, 137, 109422. [Google Scholar] [CrossRef]
- Hyson, D.A. A Comprehensive Review of Apples and Apple Components and Their Relationship to Human Health. Adv. Nutr. 2011, 2, 408–420. [Google Scholar] [CrossRef]
- da Silva, L.C.; Souza, M.C.; Sumere, B.R.; Silva, L.G.S.; da Cunha, D.T.; Barbero, G.F.; Bezerra, R.M.N.; Rostagno, M.A. Simultaneous Extraction and Separation of Bioactive Compounds from Apple Pomace Using Pressurized Liquids Coupled On-Line with Solid-Phase Extraction. Food Chem. 2020, 318, 126450. [Google Scholar] [CrossRef]
- Jakobek, L.; Barron, A.R. Ancient Apple Varieties from Croatia as a Source of Bioactive Polyphenolic Compounds. J. Food Compos. Anal. 2016, 45, 9–15. [Google Scholar] [CrossRef]
- Górnaś, P.; Mišina, I.; Olšteine, A.; Krasnova, I.; Pugajeva, I.; Lacis, G.; Siger, A.; Michalak, M.; Soliven, A.; Segliņa, D. Phenolic Compounds in Different Fruit Parts of Crab Apple: Dihydrochalcones as Promising Quality Markers of Industrial Apple Pomace by-Products. Ind. Crops Prod. 2015, 74, 607–612. [Google Scholar] [CrossRef]
- Casazza, A.A.; Pettinato, M.; Perego, P. Polyphenols from Apple Skins: A Study on Microwave-Assisted Extraction Optimization and Exhausted Solid Characterization. Sep. Purif. Technol. 2020, 240, 116640. [Google Scholar] [CrossRef]
- Bekele, E.A.; Annaratone, C.E.P.; Hertog, M.L.A.T.M.; Nicolai, B.M.; Geeraerd, A.H. Multi-Response Optimization of the Extraction and Derivatization Protocol of Selected Polar Metabolites from Apple Fruit Tissue for GC-MS Analysis. Anal. Chim. Acta 2014, 824, 42–56. [Google Scholar] [CrossRef] [PubMed]
- López-fernández, O.; Domínguez, R.; Pateiro, M.; Munekata, P.E.S.; Rocchetti, G.; Lorenzo, J.M. Determination of Polyphenols Using Liquid Chromatography–Tandem Mass Spectrometry Technique (LC–MS/MS): A Review. Antioxidants 2020, 9, 479. [Google Scholar] [CrossRef] [PubMed]
- Perussello, C.A.; Zhang, Z.; Marzocchella, A.; Tiwari, B.K. Valorization of Apple Pomace by Extraction of Valuable Compounds. Compr. Rev. Food Sci. Food Saf. 2017, 16, 776–796. [Google Scholar] [CrossRef]
- Mehinagic, E.; Prost, C.; Demaimay, M. Representativeness of Apple Aroma Extract Obtained by Vacuum Hydrodistillation: Comparison of Two Concentration Techniques. J. Food Sci. 2003, 68, 2411–2415. [Google Scholar] [CrossRef]
- Rabetafika, H.N.; Bchir, B.; Blecker, C.; Richel, A. Fractionation of Apple By-Products as Source of New Ingredients: Current Situation and Perspectives. Trends Food Sci. Technol. 2014, 40, 99–114. [Google Scholar] [CrossRef]
- Ferrentino, G.; Morozova, K.; Mosibo, O.K.; Ramezani, M.; Scampicchio, M. Biorecovery of Antioxidants from Apple Pomace by Supercritical Fluid Extraction. J. Clean. Prod. 2018, 186, 253–261. [Google Scholar] [CrossRef]
- Hernández-Carranza, P.; Ávila-Sosa, R.; Guerrero-Beltrán, J.A.; Navarro-Cruz, A.R.; Corona-Jiménez, E.; Ochoa-Velasco, C.E. Optimization of Antioxidant Compounds Extraction from Fruit By-Products: Apple Pomace, Orange and Banana Peel. J. Food Process. Preserv. 2016, 40, 103–115. [Google Scholar] [CrossRef]
- Reis, S.F.; Rai, D.K.; Abu-Ghannam, N. Water at Room Temperature as a Solvent for the Extraction of Apple Pomace Phenolic Compounds. Food Chem. 2012, 135, 1991–1998. [Google Scholar] [CrossRef]
- Alberti, A.; Zielinski, A.A.F.; Zardo, D.M.; Demiate, I.M.; Nogueira, A.; Mafra, L.I. Optimisation of the Extraction of Phenolic Compounds from Apples Using Response Surface Methodology. Food Chem. 2014, 149, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Fromm, M.; Loos, H.M.; Bayha, S.; Carle, R.; Kammerer, D.R. Recovery and Characterisation of Coloured Phenolic Preparations from Apple Seeds. Food Chem. 2013, 136, 1277–1287. [Google Scholar] [CrossRef] [PubMed]
- Azmir, J.; Zaidul, I.S.M.; Rahman, M.M.; Sharif, K.M.; Mohamed, A.; Sahena, F.; Jahurul, M.H.A.; Ghafoor, K.; Norulaini, N.A.N.; Omar, A.K.M. Techniques for Extraction of Bioactive Compounds from Plant Materials: A Review. J. Food Eng. 2013, 117, 426–436. [Google Scholar] [CrossRef]
- Putnik, P.; Lorenzo, J.M.; Barba, F.J.; Roohinejad, S.; Jambrak, A.R.; Granato, D.; Montesano, D.; Kovačević, D.B. Novel Food Processing and Extraction Technologies of High-Added Value Compounds from Plant Materials. Foods 2018, 7, 106. [Google Scholar] [CrossRef]
- Soquetta, M.B.; Terra, L.d.M.; Bastos, C.P. Green Technologies for the Extraction of Bioactive Compounds in Fruits and Vegetables. CYTA J. Food 2018, 16, 400–412. [Google Scholar] [CrossRef]
- Alirezalu, K.; Pateiro, M.; Yaghoubi, M.; Alirezalu, A.; Peighambardoust, S.H.; Lorenzo, J.M. Phytochemical Constituents, Advanced Extraction Technologies and Techno-Functional Properties of Selected Mediterranean Plants for Use in Meat Products. A Comprehensive Review. Trends Food Sci. Technol. 2020, 100, 292–306. [Google Scholar] [CrossRef]
- Bai, X.L.; Yue, T.L.; Yuan, Y.H.; Zhang, H.W. Optimization of Microwave-Assisted Extraction of Polyphenols from Apple Pomace Using Response Surface Methodology and HPLC Analysis. J. Sep. Sci. 2010, 33, 3751–3758. [Google Scholar] [CrossRef]
- Khawli, F.; Al Pateiro, M.; Domínguez, R.; Lorenzo, J.M.; Gullón, P.; Kousoulaki, K.; Ferrer, E.; Berrada, H.; Barba, F.J. Innovative Green Technologies of Intensification for Valorization of Seafood and Their By-Products. Mar. Drugs 2019, 17, 689. [Google Scholar] [CrossRef]
- Medina-Torres, N.; Ayora-Talavera, T.; Espinosa-Andrews, H.; Sánchez-Contreras, A.; Pacheco, N. Ultrasound Assisted Extraction for the Recovery of Phenolic Compounds from Vegetable Sources. Agronomy 2017, 7, 47. [Google Scholar] [CrossRef]
- Rashid, R.; Mohd Wani, S.; Manzoor, S.; Masoodi, F.A.; Masarat Dar, M. Green Extraction of Bioactive Compounds from Apple Pomace by Ultrasound Assisted Natural Deep Eutectic Solvent Extraction: Optimisation, Comparison and Bioactivity. Food Chem. 2022, 398, 133871. [Google Scholar] [CrossRef]
- He, Y.; Lu, Q.; Liviu, G. Effects of Extraction Processes on the Antioxidant Activity of Apple Polyphenols. CYTA J. Food 2015, 13, 603–606. [Google Scholar] [CrossRef]
- Pingret, D.; Fabiano-Tixier, A.S.; Bourvellec, C.; Le Renard, C.M.G.C.; Chemat, F. Lab and Pilot-Scale Ultrasound-Assisted Water Extraction of Polyphenols from Apple Pomace. J. Food Eng. 2012, 111, 73–81. [Google Scholar] [CrossRef]
- Virot, M.; Tomao, V.; Le Bourvellec, C.; Renard, C.M.C.G.; Chemat, F. Towards the Industrial Production of Antioxidants from Food Processing By-Products with Ultrasound-Assisted Extraction. Ultrason. Sonochem. 2010, 17, 1066–1074. [Google Scholar] [CrossRef]
- Barba, F.J.; Zhu, Z.; Koubaa, M.; Sant’Ana, A.S.; Orlien, V. Green Alternative Methods for the Extraction of Antioxidant Bioactive Compounds from Winery Wastes and By-Products: A Review. Trends Food Sci. Technol. 2016, 49, 96–109. [Google Scholar] [CrossRef]
- Filipčev, B.; Lević, L.; Bodroža-Solarov, M.; Mišljenovic, N.; Koprivica, G. Quality Characteristics and Antioxidant Properties of Breads Supplemented with Sugar Beet Molasses-Based Ingredients. Int. J. Food Prop. 2010, 13, 1035–1053. [Google Scholar] [CrossRef]
- Li, W.; Yang, R.; Ying, D.; Yu, J.; Sanguansri, L.; Augustin, M.A. Analysis of Polyphenols in Apple Pomace: A Comparative Study of Different Extraction and Hydrolysis Procedures. Ind. Crops Prod. 2020, 147, 112250. [Google Scholar] [CrossRef]
- Purić, M.; Rabrenović, B.; Rac, V.; Pezo, L.; Tomašević, I.; Demin, M. Application of Defatted Apple Seed Cakes as a By-Product for the Enrichment of Wheat Bread. LWT 2020, 130, 109391. [Google Scholar] [CrossRef]
- Sudha, M.L.; Dharmesh, S.M.; Pynam, H.; Bhimangouder, S.V.; Eipson, S.W.; Somasundaram, R.; Nanjarajurs, S.M. Antioxidant and Cyto/DNA Protective Properties of Apple Pomace Enriched Bakery Products. J. Food Sci. Technol. 2016, 53, 1909–1918. [Google Scholar] [CrossRef]
- Sudha, M.L.; Baskaran, V.; Leelavathi, K. Apple Pomace as a Source of Dietary Fiber and Polyphenols and Its Effect on the Rheological Characteristics and Cake Making. Food Chem. 2007, 104, 686–692. [Google Scholar] [CrossRef]
- Rupasinghe, H.P.V.; Wang, L.; Huber, G.M.; Pitts, N.L. Effect of Baking on Dietary Fibre and Phenolics of Muffins Incorporated with Apple Skin Powder. Food Chem. 2008, 107, 1217–1224. [Google Scholar] [CrossRef]
- Usman, M.; Ahmed, S.; Mehmood, A.; Bilal, M.; Patil, P.J.; Akram, K.; Farooq, U. Effect of Apple Pomace on Nutrition, Rheology of Dough and Cookies Quality. J. Food Sci. Technol. 2020, 57, 3244–3251. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.; Cavender, G.; Zhao, Y. Impingement Drying for Preparing Dried Apple Pomace Flour and Its Fortification in Bakery and Meat Products. J. Food Sci. Technol. 2015, 52, 5568–5578. [Google Scholar] [CrossRef] [PubMed]
- Rocha Parra, A.F.; Ribotta, P.D.; Ferrero, C. Apple Pomace in Gluten-Free Formulations: Effect on Rheology and Product Quality. Int. J. Food Sci. Technol. 2015, 50, 682–690. [Google Scholar] [CrossRef]
- Alongi, M.; Melchior, S.; Anese, M. Reducing the Glycemic Index of Short Dough Biscuits by Using Apple Pomace as a Functional Ingredient. LWT 2019, 100, 300–305. [Google Scholar] [CrossRef]
- Rupasinghe, H.P.V.; Erkan, N.; Yasmin, A. Antioxidant Protection of Eicosapentaenoic Acid and Fish Oil Oxidation by Polyphenoljc-Enriched Apple Skin Extract. J. Agric. Food Chem. 2010, 58, 1233–1239. [Google Scholar] [CrossRef]
- Bitalebi, S.; Nikoo, M.; Rahmanifarah, K.; Noori, F.; Ahmadi Gavlighi, H. Effect of Apple Peel Extract as Natural Antioxidant on Lipid and Protein Oxidation of Rainbow Trout (Oncorhynchus Mykiss) Mince. Int. Aquat. Res. 2019, 11, 135–146. [Google Scholar] [CrossRef]
- Hellwig, M. The Chemistry of Protein Oxidation in Food. Angew. Chem. Int. Ed. 2019, 58, 16742–16763. [Google Scholar] [CrossRef]
- Sun, L.; Sun, J.; Thavaraj, P.; Yang, X.; Guo, Y. Effects of Thinned Young Apple Polyphenols on the Quality of Grass Carp (Ctenopharyngodon Idellus) Surimi during Cold Storage. Food Chem. 2017, 224, 372–381. [Google Scholar] [CrossRef]
- Sun, W.Q.; Zhang, Y.J.; Zhou, G.H.; Xu, X.L.; Peng, Z.Q. Effect of Apple Polyphenol on Oxidative Stability of Sliced Cooked Cured Beef and Pork Hams during Chilled Storage. J. Muscle Foods 2010, 21, 722–737. [Google Scholar] [CrossRef]
- Yadav, S.; Malik, A.; Pathera, A.; Islam, R.U.; Sharma, D. Development of Dietary Fibre Enriched Chicken Sausages by Incorporating Corn Bran, Dried Apple Pomace and Dried Tomato Pomace. Nutr. Food Sci. 2016, 46, 16–29. [Google Scholar] [CrossRef]
- Deng, S.; Shi, S.; Xia, X. Effect of Plant Polyphenols on the Physicochemical Properties, Residual Nitrites, and N-Nitrosamine Formation in Dry-Fried Bacon. Meat Sci. 2022, 191, 108872. [Google Scholar] [CrossRef] [PubMed]
- Rather, S.A.; Akhter, R.; Masoodi, F.A.; Gani, A.; Wani, S.M. Utilization of Apple Pomace Powder as a Fat Replacer in Goshtaba: A Traditional Meat Product of Jammu and Kashmir, India. J. Food Meas. Charact. 2015, 9, 389–399. [Google Scholar] [CrossRef]
- Gaikwad, K.K.; Lee, J.Y.; Lee, Y.S. Development of Polyvinyl Alcohol and Apple Pomace Bio-Composite Film with Antioxidant Properties for Active Food Packaging Application. J. Food Sci. Technol. 2016, 53, 1608–1619. [Google Scholar] [CrossRef]
- Lan, W.; Wang, S.; Zhang, Z.; Liang, X.; Liu, X.; Zhang, J. Development of Red Apple Pomace Extract/Chitosan-Based Films Reinforced by TiO2 Nanoparticles as a Multifunctional Packaging Material. Int. J. Biol. Macromol. 2021, 168, 105–115. [Google Scholar] [CrossRef]
- Lin, L.; Peng, S.; Shi, C.; Li, C.; Hua, Z.; Cui, H. Preparation and Characterization of Cassava Starch/Sodium Carboxymethyl Cellulose Edible Film Incorporating Apple Polyphenols. Int. J. Biol. Macromol. 2022, 212, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Sun, J.; Liu, D.; Fu, M.; Yang, X.; Guo, Y. The Preservative Effects of Chitosan Film Incorporated with Thinned Young Apple Polyphenols on the Quality of Grass Carp (Ctenopharyngodon Idellus) Fillets during Cold Storage: Correlation between the Preservative Effects and the Active Properties. Food Packag. Shelf Life 2018, 17, 1–10. [Google Scholar] [CrossRef]
- Sun, L.; Sun, J.; Chen, L.; Niu, P.; Yang, X.; Guo, Y. Preparation and Characterization of Chitosan Film Incorporated with Thinned Young Apple Polyphenols as an Active Packaging Material. Carbohydr. Polym. 2017, 163, 81–91. [Google Scholar] [CrossRef]
- Riaz, A.; Lei, S.; Akhtar, H.M.S.; Wan, P.; Chen, D.; Jabbar, S.; Abid, M.; Hashim, M.M.; Zeng, X. Preparation and Characterization of Chitosan-Based Antimicrobial Active Food Packaging Film Incorporated with Apple Peel Polyphenols. Int. J. Biol. Macromol. 2018, 114, 547–555. [Google Scholar] [CrossRef]
- Shin, S.H.; Chang, Y.; Lacroix, M.; Han, J. Control of Microbial Growth and Lipid Oxidation on Beef Product Using an Apple Peel-Based Edible Coating Treatment. LWT 2017, 84, 183–188. [Google Scholar] [CrossRef]
- Gupta, R.K.; Yadav, S. Formulation of Noodles Using Apple Pomace and Evaluation of Its Phytochemicals and Antioxidant Activity. J. Pharmacogn. Phytochem. 2015, 4, 99–106. [Google Scholar]
- Massini, L.; Rico, D.; Martin-Diana, A.B.; Barry-Ryan, C. Apple Peel Flavonoids as Natural Antioxidants for Vegetable Juice Applications. Eur. Food Res. Technol. 2016, 242, 1459–1469. [Google Scholar] [CrossRef]
- Fernandes, P.A.R.; Ferreira, S.S.; Bastos, R.; Ferreira, I.; Cruz, M.T.; Pinto, A.; Coelho, E.; Passos, C.P.; Coimbra, M.A.; Cardoso, S.M. Apple Pomace Extract as a Sustainable Food Ingredient. Antioxidants 2019, 8, 189. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, I.; Khalique, A.; Shahid, M.Q.; Rashid, A.A.; Faiz, F.; Ikram, M.A.; Ahmed, S.; Imran, M.; Khan, M.A.; Nadeem, M. Studying the Influence of Apple Peel Polyphenol Extract Fortification on the Characteristics of Probiotic Yoghurt. Plants 2020, 9, 77. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Asma, U.; Morozova, K.; Ferrentino, G.; Scampicchio, M. Apples and Apple By-Products: Antioxidant Properties and Food Applications. Antioxidants 2023, 12, 1456. https://doi.org/10.3390/antiox12071456
Asma U, Morozova K, Ferrentino G, Scampicchio M. Apples and Apple By-Products: Antioxidant Properties and Food Applications. Antioxidants. 2023; 12(7):1456. https://doi.org/10.3390/antiox12071456
Chicago/Turabian StyleAsma, Umme, Ksenia Morozova, Giovanna Ferrentino, and Matteo Scampicchio. 2023. "Apples and Apple By-Products: Antioxidant Properties and Food Applications" Antioxidants 12, no. 7: 1456. https://doi.org/10.3390/antiox12071456
APA StyleAsma, U., Morozova, K., Ferrentino, G., & Scampicchio, M. (2023). Apples and Apple By-Products: Antioxidant Properties and Food Applications. Antioxidants, 12(7), 1456. https://doi.org/10.3390/antiox12071456









