Molecular Study on Twin Cohort with Discordant Birth Weight
Abstract
1. Introduction
2. Materials and Methods
2.1. Clinical Samples
2.2. Immunohistochemistry in the Umbilical Cord Sections
2.3. Immunocytochemistry and Fluorescence-Activated Cell Sorting (FACS) on the Fetal RBCs
2.4. Determination of Peroxynitrite (ONOO−) Level
2.5. Blood Smear Image Processing and Data Analysis
2.6. Statistical Analysis
3. Results
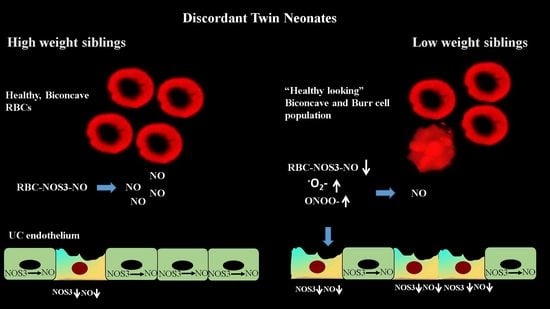
3.1. Expression of Endothelial and Inducible NOS in the Birth Weight–Discordant Populations
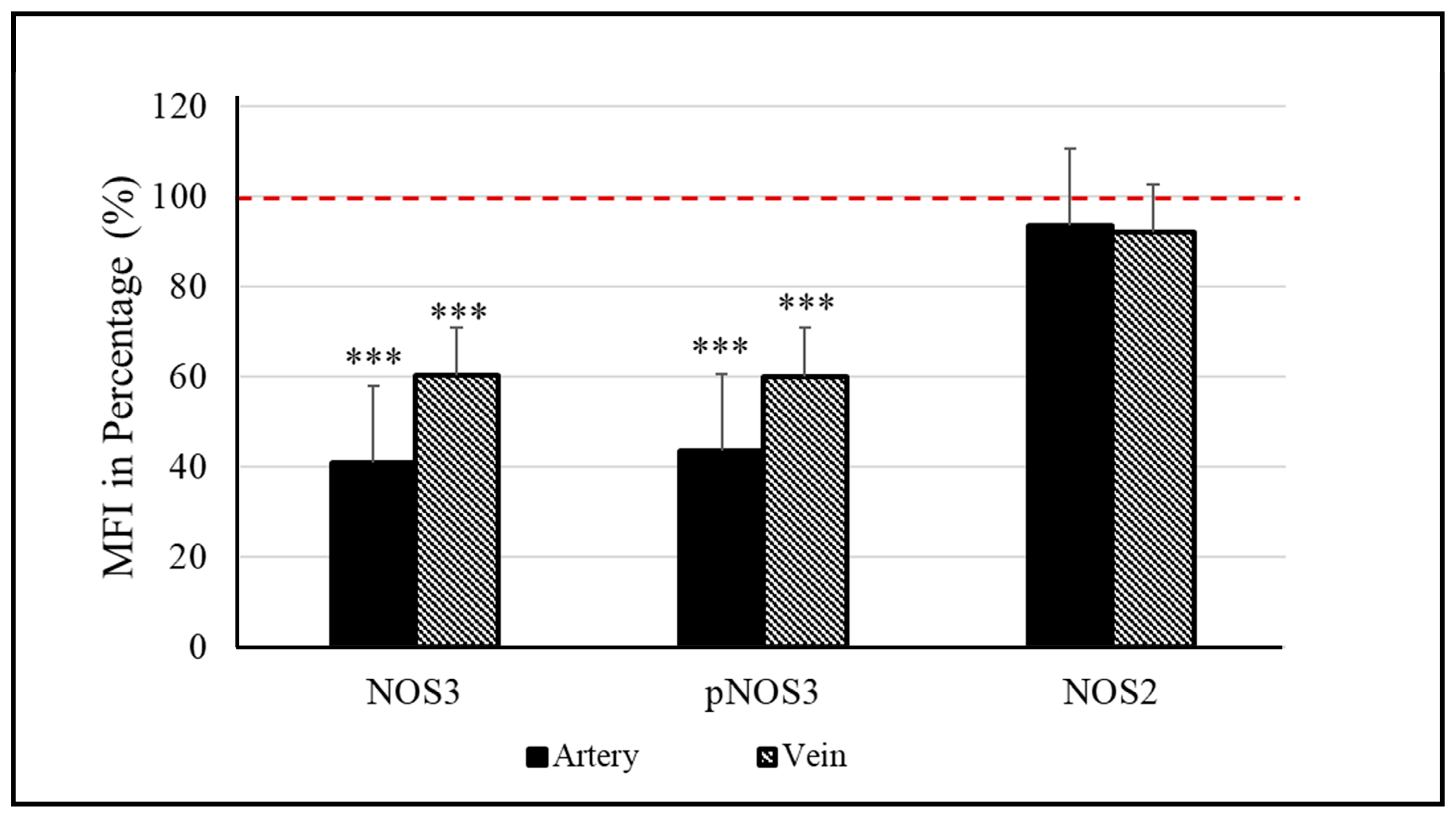
3.2. NOS3 Expression and Activation Level in the Red Blood Cell of the Birth Weight–Discordant Study Populations
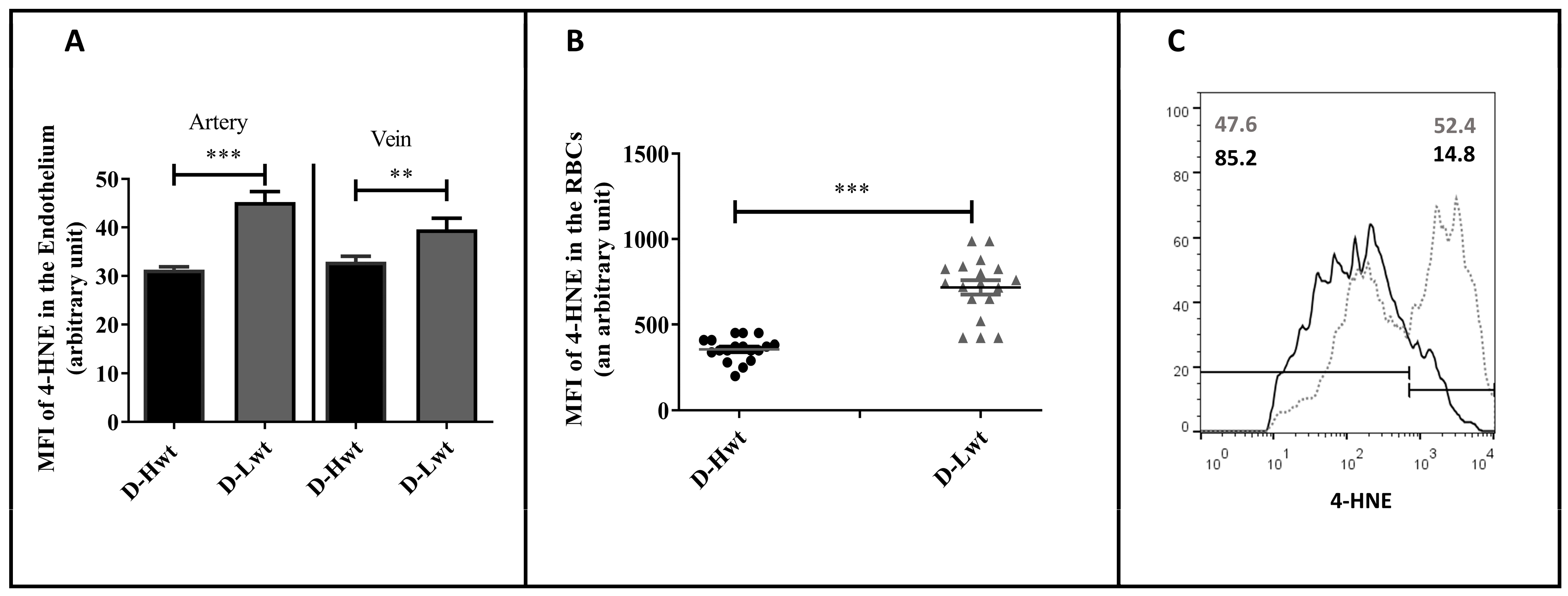
3.3. Membrane Damage Verification in the UC Vessels’ Endothelium and Red Blood Cells; Measurement of 4-Hydroxynonenal Level as a Lipid Peroxidation Marker
3.4. Peroxynitrite (ONOO−) Accumulation in Red Blood Cell Populations
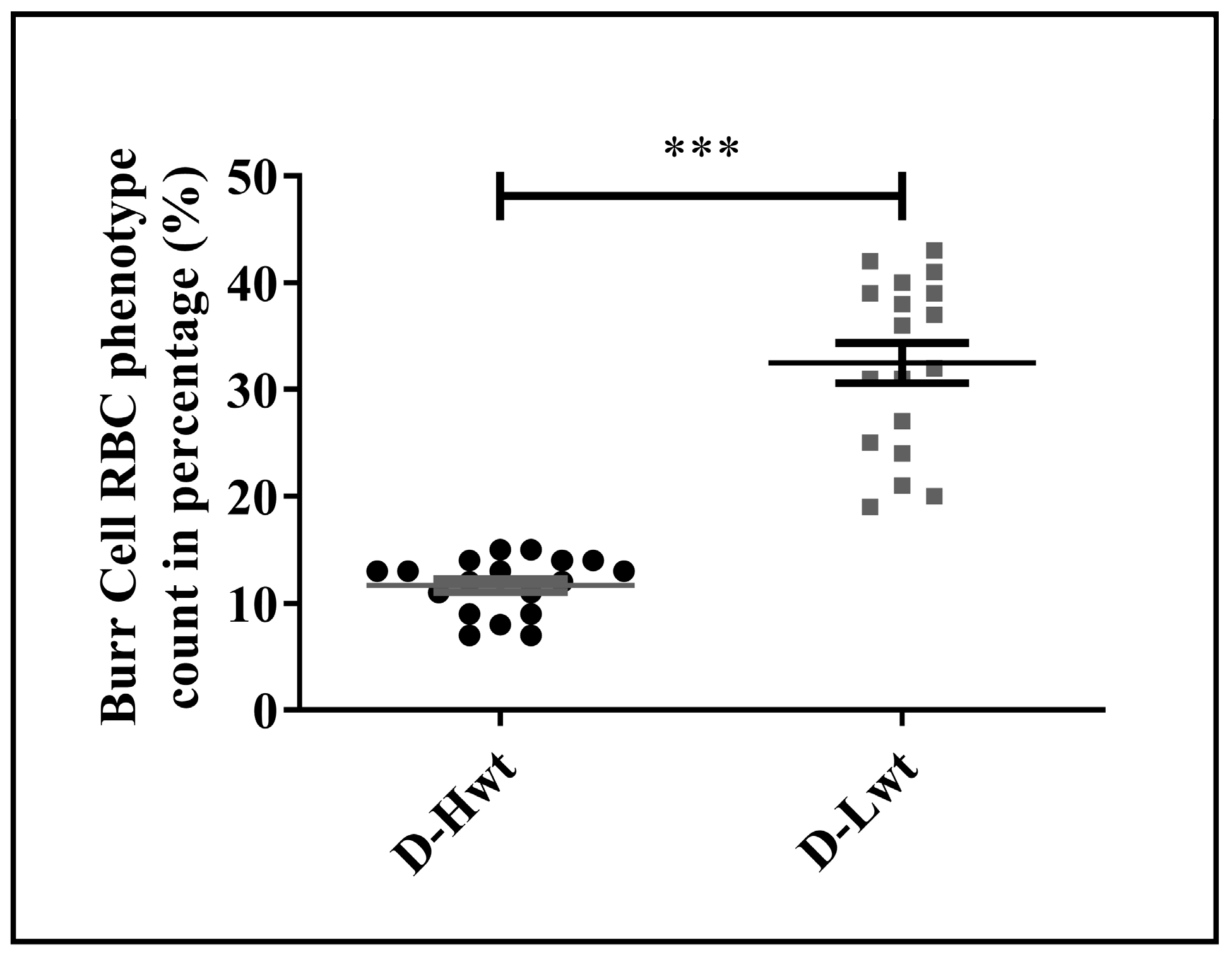
3.5. Accumulation of Morphological Variants in the Birth Weight–Discordant Red Blood Cell Populations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ochoa, J.J.; Ramirez-Tortosa, M.C.; Quiles, J.L.; Palomino, N.; Robles, R.; Mataix, J.; Huertas, J.R. Oxidative stress in erythrocytes from premature and full-term infants during their first 72 h of life. Free Radic. Res. 2003, 37, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Umranikar, A.; Parmar, D.; Davies, S.; Fountain, S. Multiple births following in vitro fertilization treatment: Redefining success. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 170, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Puccio, G.; Giuffré, M.; Piccione, M.; Piro, E.; Malerba, V.; Corsello, G. Intrauterine growth pattern and birthweight discordance in twin pregnancies: A retrospective study. Ital. J. Pediatr. 2014, 40, 43. [Google Scholar] [CrossRef] [PubMed]
- Fox, S.B.; Khong, T.Y. Lack of innervation of human umbilical cord. An immunohistological and histochemical study. Placenta 1990, 11, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Vanhoutte, P.M.; Shimokawa, H.; Feletou, M.; Tang, E.H.C. Endothelial dysfunction and vascular disease—A 30th anniversary update. Acta Physiol. 2017, 219, 22–96. [Google Scholar] [CrossRef]
- Berk, B.C.; Haendeler, J.; Sottile, J. Angiotensin II, atherosclerosis, and aortic aneurysms. J. Clin. Investig. 2000, 105, 1525–1526. [Google Scholar] [CrossRef]
- Granger, J.P.; Alexander, B.T.; Llinas, M.T.; Bennett, W.A.; Khalil, R.A. Pathophysiology of hypertension during preeclampsia linking placental ischemia with endothelial dysfunction. Hypertension 2001, 38, 718–722. [Google Scholar] [CrossRef]
- Nedeljkovic, Z.S.; Gokce, N.; Loscalzo, J. Mechanisms of oxidative stress and vascular dysfunction. Postgrad. Med. J. 2003, 79, 195–200. [Google Scholar] [CrossRef]
- Selye, H. Stress and disease. Science 1955, 122, 625–631. [Google Scholar] [CrossRef]
- Geng, Y.J.; Wu, Q.; Muszynski, M.; Hansson, G.K.; Libby, P. Apoptosis of vascular smooth muscle cells induced by in vitro stimulation with interferon-γ, tumor necrosis factor-α, and interleukin-1β. Arter. Thromb. Vasc. Biol. 1996, 16, 19–27. [Google Scholar] [CrossRef]
- Farah, C.; Michel, L.Y.M.; Balligand, J.L. Nitric oxide signalling in cardiovascular health and disease. Nat. Rev. Cardiol. 2018, 15, 292–316. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.M.; Chapman, G.B.; Peyton, K.J.; Schafer, A.I.; Durante, W. Carbon monoxide inhibits apoptosis in vascular smooth muscle cells. Cardiovasc. Res. 2002, 55, 396–405. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, P.; Dugmonits, K.N.; Orvos, H.; Hermesz, E. Mature twin neonates exhibit oxidative stress via nitric oxide synthase dysfunctionality: A prognostic stress marker in the red blood cells and umbilical cord vessels. Antioxidants 2020, 9, 845. [Google Scholar] [CrossRef] [PubMed]
- Cortese-Krott, M.; Rodriguez-Mateos, A.; Sansone, R.; Kuhnle, G.; Thasian-Sivarajah, S.; Krenz, T.; Horn, P.; Krisp, C.; Wolters, D.; Heiss, C.; et al. Human red blood cells at work: Identification and visualization of erythrocytic eNOS activity in health and disease. Blood 2012, 120, 4229–4237. [Google Scholar] [CrossRef]
- Cortese-Krott, M.M.; Kelm, M. Endothelial nitric oxide synthase in red blood cells: Key to a new erythrocrine function? Redox Biol. 2014, 2, 251–258. [Google Scholar] [CrossRef]
- Dugmonits, K.N.; Chakraborty, P.; Hollandi, R.; Zahorán, S.; Pankotai-Bodó, G.; Horváth, P.; Orvos, H.; Hermesz, E. Maternal Smoking Highly Affects the Function, Membrane Integrity, and Rheological Properties in Fetal Red Blood Cells. Oxid. Med. Cell. Longev. 2019, 2019, 1509798. [Google Scholar] [CrossRef]
- Chakraborty, P.; Khamit, A.; Hermesz, E. Fetal oxygen supply can be improved by an effective cross-talk between fetal erythrocytes and vascular endothelium. Biochim. Biophys. Acta-Mol. Basis Dis. 2021, 1867, 166243. [Google Scholar] [CrossRef]
- Ding, H.; Triggle, C.R. Endothelial cell dysfunction and the vascular complications associated with type 2 diabetes: Assessing the health of the endothelium. Vasc. Health Risk Manag. 2005, 1, 55–71. [Google Scholar] [CrossRef]
- Barthelmes, J.; Nägele, M.P.; Ludovici, V.; Ruschitzka, F.; Sudano, I.; Flammer, A.J. Endothelial dysfunction in cardiovascular disease and Flammer syndrome-similarities and differences. EPMA J. 2017, 8, 99–109. [Google Scholar] [CrossRef]
- Nishikawa, E.; Matsumoto, T.; Isige, M.; Tsuji, T.; Mugisima, H.; Takahashi, S. Comparison of capacities to maintain hematopoietic stem cells among different types of stem cells derived from the placenta and umbilical cord. Regen. Ther. 2016, 4, 48–61. [Google Scholar] [CrossRef]
- Chakraborty, P.; Dugmonits, K.N.; Végh, A.G.; Hollandi, R.; Horváth, P.; Maléth, J.; Hegyi, P.; Németh, G.; Hermesz, E. Failure in the compensatory mechanism in red blood cells due to sustained smoking during pregnancy. Chem. Biol. Interact. 2019, 313, 108821. [Google Scholar] [CrossRef] [PubMed]
- Huie, R.E.; Padmaja, S. The reaction of no with superoxide. Free Radic. Res. 1993, 18, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, A.E.; Jones, T.R.; Lamprecht, M.R.; Clarke, C.; Kang, I.H.; Friman, O.; Guertin, D.A.; Chang, J.H.; Lindquist, R.A.; Moffat, J.; et al. CellProfiler: Image analysis software for identifying and quantifying cell phenotypes. Genome. Biol. 2006, 7, R100. [Google Scholar] [CrossRef] [PubMed]
- Michel, T.; Feron, O. Nitric oxide synthases: Which, where, how, and why? J. Clin. Investig. 1997, 100, 2146–2152. [Google Scholar] [CrossRef] [PubMed]
- Förstermann, U.; Li, H. Therapeutic effect of enhancing endothelial nitric oxide synthase (eNOS) expression and preventing eNOS uncoupling. Br. J. Pharmacol. 2011, 164, 213–223. [Google Scholar] [CrossRef]
- Förstermann, U.; Sessa, W.C. Nitric oxide synthases: Regulation and function. Eur. Heart J. 2012, 33, 829–837. [Google Scholar] [CrossRef] [PubMed]
- Cau, S.B.A.; Carneiro, F.S.; Tostes, R.C. Differential modulation of nitric oxide synthases in aging: Therapeutic opportunities. Front. Physiol. 2012, 3, 218. [Google Scholar] [CrossRef]
- Cosentino, F.; Lüscher, T.F. Tetrahydrobiopterin and endothelial nitric oxide synthase activity. Cardiovasc. Res. 1999, 43, 274–278. [Google Scholar] [CrossRef]
- Pritchard, K.A., Jr.; Groszek, L.; Smalley, D.M.; Sessa, W.C.; Wu, M.; Villalon, P.; Wolin, M.S.; Stemerman, M.B. Native low-density lipoprotein increases endothelial cell nitric oxide synthase generation of superoxide anion. Circ. Res. 1995, 77, 510–518. [Google Scholar] [CrossRef]
- Landmesser, U.; Dikalov, S.; Price, S.R.; McCann, L.; Fukai, T.; Holland, S.M.; Mitch, W.E.; Harrison, D.G. Oxidation of tetrahydrobiopterin leads to uncoupling of endothelial cell nitric oxide synthase in hypertension. J. Clin. Investig. 2003, 111, 1201–1209. [Google Scholar] [CrossRef]
- Laursen, J.B.; Somers, M.; Kurz, S.; McCann, L.; Warnholtz, A.; Freeman, B.A.; Tarpey, M.; Fukai, T.; Harrison, D.G. Endothelial regulation of vasomotion in apoE-deficient mice: Implications for interactions between peroxynitrite and tetrahydrobiopterin. Circulation 2001, 103, 1282–1288. [Google Scholar] [CrossRef] [PubMed]
- Nair, J.; De Flora, S.; Izzotti, A.; Bartsch, H. Lipid peroxidation-derived etheno-DNA adducts in human atherosclerotic lesions. Mutat. Res. 2007, 621, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Doorn, J.A.; Petersen, D.R. Covalent adduction of nucleophilic amino acids by 4-hydroxynonenal and 4-oxononenal. Chem. Biol. Interact. 2003, 143–144, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, A.; Comporti, M.; Esterbauer, H. Identification of 4-hydroxynonenal as a cytotoxic product originating from the peroxidation of liver microsomal lipids. Biochim. Biophys. Acta 1980, 620, 281–296. [Google Scholar] [CrossRef] [PubMed]
- Esterbauer, H.; Schaur, R.J.; Zollner, H. Chemistry and biochemistry of 4-hydroxynonenal, malonaldehyde and related aldehydes. Free Radic. Biol. Med. 1991, 11, 81–128. [Google Scholar] [CrossRef]
- Chapple, S.J.; Cheng, X.; Mann, G.E. Effects of 4-hydroxynonenal on vascular endothelial and smooth muscle cell redox signaling and function in health and disease. Redox Biol. 2013, 1, 319–331. [Google Scholar] [CrossRef]
- Mahdi, A.; Tengbom, J.; Alvarsson, M.; Wernly, B.; Zhou, Z.; Pernow, J. Red Blood Cell Peroxynitrite Causes Endothelial Dysfunction in Type 2 Diabetes Mellitus via Arginase. Cells 2020, 9, 1712. [Google Scholar] [CrossRef]
- Zipursky, A.; Brown, E.; Palko, J.; Brown, E.J. The erythrocyte differential count in newborn infants. Am. J. Pediatr. Hematol. Oncol. 1983, 5, 45–51. [Google Scholar]
- Elgsaeter, A.; Mikkelsen, A. Shapes and shape changes in vitro in normal red blood cells. Biochim. Biophys. Acta 1991, 1071, 273–290. [Google Scholar] [CrossRef]
- Chowdhury, A.; Dasgupta, R.; Majumder, S.K. Changes in hemoglobin-oxygen affinity with shape variations of red blood cells. J. Biomed. Opt. 2017, 22, 105006. [Google Scholar] [CrossRef]
- Ruef, P.; Linderkamp, O. Deformability and Geometry of Neonatal Erythrocytes with Irregular Shapes. Pediatr. Res. 1999, 45, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Mandal, A.K.; Taylor, C.A.; Bell, R.D.; Hillman, N.M.; Jarnot, M.D.; Cunningham, J.D.; Phillips, L.G. Erythrocyte deformation in ischemic acute tubular necrosis and amelioration by splenectomy in the dog. Lab. Investig. 1991, 65, 566–576. [Google Scholar] [PubMed]
- Turchetti, V.; De Matteis, C.; Leoncini, F.; Trabalzini, L.; Guerrini, M.; Forconi, S. Variations of erythrocyte morphology in different pathologies. Clin. Hemorheol. Microcirc. 1997, 17, 209–215. [Google Scholar] [PubMed]
- Suljević, D.; Mitrašinović-Brulić, M.; Fočak, M. L-cysteine protective effects against platelet disaggregation and echinocyte occurrence in gentamicin-induced kidney injury. Mol. Cell. Biochem. 2023, 478, 13–22. [Google Scholar] [CrossRef]
- Dodd, J.M.; Crowther, C.A.; Haslam, R.R.; Robinson, J.S. Elective birth at 37 weeks of gestation versus standard care for women with an uncomplicated twin pregnancy at term: The Twins Timing of Birth Randomised Trial. BJOG 2012, 119, 964–973. [Google Scholar] [CrossRef] [PubMed]
- Cheong-See, F.; Schuit, E.; Arroyo-Manzano, D.; Khalil, A.; Barrett, J.; Joseph, K.S.; Asztalos, E.; Hack, K.; Lewi, L.; Lim, A.; et al. Prospective risk of stillbirth and neonatal complications in twin pregnancies: Systematic review and meta-analysis. BMJ 2016, 354, i4353. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chakraborty, P.; Orvos, H.; Hermesz, E. Molecular Study on Twin Cohort with Discordant Birth Weight. Antioxidants 2023, 12, 1370. https://doi.org/10.3390/antiox12071370
Chakraborty P, Orvos H, Hermesz E. Molecular Study on Twin Cohort with Discordant Birth Weight. Antioxidants. 2023; 12(7):1370. https://doi.org/10.3390/antiox12071370
Chicago/Turabian StyleChakraborty, Payal, Hajnalka Orvos, and Edit Hermesz. 2023. "Molecular Study on Twin Cohort with Discordant Birth Weight" Antioxidants 12, no. 7: 1370. https://doi.org/10.3390/antiox12071370
APA StyleChakraborty, P., Orvos, H., & Hermesz, E. (2023). Molecular Study on Twin Cohort with Discordant Birth Weight. Antioxidants, 12(7), 1370. https://doi.org/10.3390/antiox12071370