Antioxidant Properties of Protein-Rich Plant Foods in Gastrointestinal Digestion—Peanuts as Our Antioxidant Friend or Foe in Allergies
Abstract
1. Introduction
2. Peanut as a Functional, Plant-Based Food
2.1. Peanut Proteins and Allergens as a Potential Reservoir of Antioxidant Power
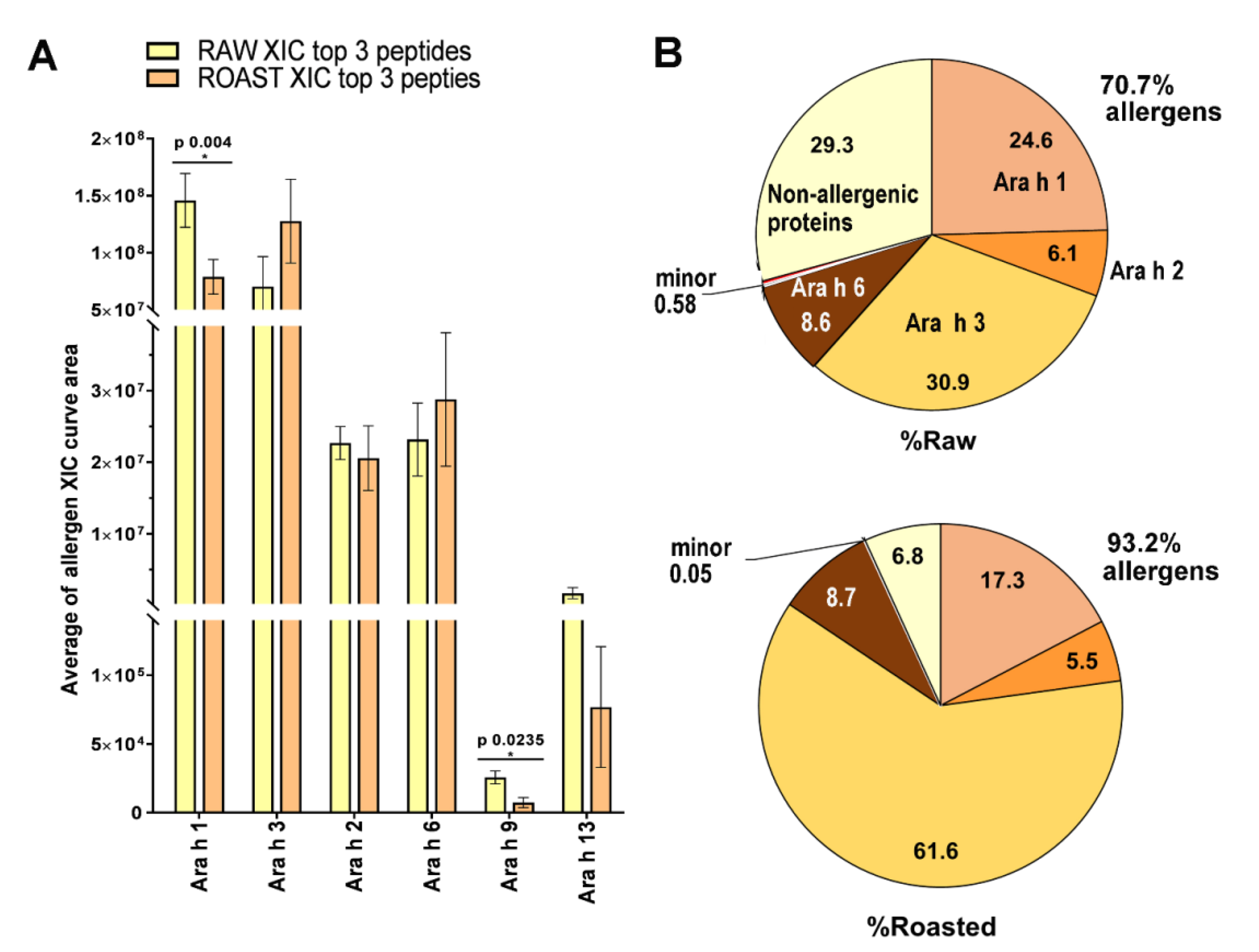
Peanut Allergy and Allergens
2.2. Effects of Thermal Processing on Bioaccessibility and Allergenic Properties of Peanut Proteins
2.3. The Effects of Thermal Processing on Bioaccessibility and Antioxidant Activity of Other Peanut Biomolecules
2.3.1. Peanut Lipids and Lipid-Soluble Vitamins
2.3.2. Peanut Carbohydrates
2.3.3. Peanut Phenolics and Water-Soluble Vitamins
3. Digestibility of Biomolecules from Complete Food Matrix and Their Antioxidant Potential
3.1. Perspectives on the Oral Phase of Simulated Digestion
3.2. Perspectives on the Gastric Phase of Simulated Digestion
3.3. Perspectives on the Intestinal Phase of Simulated Digestion
4. Food-Derived Bioactive Peptides with Antioxidant Activity
5. Conclusions and Future Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118–126. [Google Scholar] [CrossRef]
- Masilamani, M.; Commins, S.; Shreffler, W. Determinants of food allergy. Immunol. Allergy Clin. N. Am. 2012, 32, 11–33. [Google Scholar] [CrossRef] [PubMed]
- Ndhlala, A.R.; Moyo, M.; Van Staden, J. Natural antioxidants: Fascinating or mythical biomolecules? Molecules 2010, 15, 6905–6930. [Google Scholar] [CrossRef] [PubMed]
- Gorissen, S.H.M.; Crombag, J.J.R.; Senden, J.M.G.; Waterval, W.A.H.; Bierau, J.; Verdijk, L.B.; van Loon, L.J.C. Protein content and amino acid composition of commercially available plant-based protein isolates. Amino Acids 2018, 50, 1685–1695. [Google Scholar] [CrossRef] [PubMed]
- Messina, M. Soy and Health Update: Evaluation of the Clinical and Epidemiologic Literature. Nutrients 2016, 8, 754. [Google Scholar] [CrossRef]
- Bonku, R.; Yu, J. Health aspects of peanuts as an outcome of its chemical composition. Food Sci. Hum. Wellness 2020, 9, 21–30. [Google Scholar] [CrossRef]
- Cruz-Casas, D.E.; Aguilar, C.N.; Ascacio-Valdés, J.A.; Rodríguez-Herrera, R.; Chávez-González, M.L.; Flores-Gallegos, A.C. Enzymatic hydrolysis and microbial fermentation: The most favorable biotechnological methods for the release of bioactive peptides. Food Chem. Mol. Sci. 2021, 3, 100047. [Google Scholar] [CrossRef]
- Pellinen, T.; Päivärinta, E.; Isotalo, J.; Lehtovirta, M.; Itkonen, S.T.; Korkalo, L.; Erkkola, M.; Pajari, A.-M. Replacing dietary animal-source proteins with plant-source proteins changes dietary intake and status of vitamins and minerals in healthy adults: A 12-week randomized controlled trial. Eur. J. Nutr. 2022, 61, 1391–1404. [Google Scholar] [CrossRef]
- Zaky, A.A.; Simal-Gandara, J.; Eun, J.B.; Shim, J.H.; Abd El-Aty, A.M. Bioactivities, Applications, Safety, and Health Benefits of Bioactive Peptides From Food and By-Products: A Review. Front. Nutr. 2021, 8, 815640. [Google Scholar] [CrossRef]
- Bochnak-Niedźwiecka, J.; Szymanowska, U.; Kapusta, I.; Świeca, M. Antioxidant Content and Antioxidant Capacity of the Protein-Rich Powdered Beverages Enriched with Flax Seeds Gum. Antioxidants 2022, 11, 582. [Google Scholar] [CrossRef]
- Ayala, A.; Muñoz, M.F.; Argüelles, S. Lipid peroxidation: Production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid. Med. Cell. Longev. 2014, 2014, 360438. [Google Scholar] [CrossRef]
- Tylewicz, U.; Inchingolo, R.; Rodriguez-Estrada, M.T. Chapter 9—Food Aroma Compounds. In Nutraceutical and Functional Food Components; Galanakis, C.M., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 297–334. [Google Scholar]
- Hellwig, M. The Chemistry of Protein Oxidation in Food. Angew. Chem. Int. Ed. 2019, 58, 16742–16763. [Google Scholar] [CrossRef]
- Peña-Ramos, E.; Xiong, Y. Whey and soy protein, hydrolysates inhibit lipid oxidation in, cooked pork patties. Meat Sci. 2003, 64, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Verghese, M.; Willis, S.; Boateng, J.; Gomaa, A.; Kaur, R. Effect of Food Processing on Antioxidant Potential, Availability, and Bioavailability. Annu. Rev. Food Sci. Technol. 2021, 12, 307–329. [Google Scholar] [CrossRef]
- Domínguez, R.; Pateiro, M.; Munekata, P.E.S.; Zhang, W.; Garcia-Oliveira, P.; Carpena, M.; Prieto, M.A.; Bohrer, B.; Lorenzo, J.M. Protein Oxidation in Muscle Foods: A Comprehensive Review. Antioxidants 2021, 11, 60. [Google Scholar] [CrossRef]
- Perusko, M.; Ghnimi, S.; Simovic, A.; Stevanovic, N.; Radomirovic, M.; Gharsallaoui, A.; Smiljanic, K.; Van Haute, S.; Stanic-Vucinic, D.; Cirkovic Velickovic, T. Maillard reaction products formation and antioxidative power of spray dried camel milk powders increases with the inlet temperature of drying. LWT 2021, 143, 111091. [Google Scholar] [CrossRef]
- Đukić, T.; Smiljanić, K.; Mihailović, J.; Prodić, I.; Apostolović, D.; Liu, S.-H.; Epstein, M.M.; van Hage, M.; Stanić-Vučinić, D.; Ćirković Veličković, T. Proteomic Profiling of Major Peanut Allergens and Their Post-Translational Modifications Affected by Roasting. Foods 2022, 11, 3993. [Google Scholar] [CrossRef]
- Prodić, I.; Smiljanić, K.; Simović, A.; Radosavljević, J.; Ćirković Veličković, T. Thermal Processing of Peanut Grains Impairs Their Mimicked Gastrointestinal Digestion While Downstream Defatting Treatments Affect Digestomic Profiles. Foods 2019, 8, 463. [Google Scholar] [CrossRef]
- Cömert, E.D.; Gökmen, V. Effect of food combinations and their co-digestion on total antioxidant capacity under simulated gastrointestinal conditions. Curr. Res. Food Sci. 2022, 5, 414–422. [Google Scholar] [CrossRef]
- Coelho, M.C.; Ribeiro, T.B.; Oliveira, C.; Batista, P.; Castro, P.; Monforte, A.R.; Rodrigues, A.S.; Teixeira, J.; Pintado, M. In Vitro Gastrointestinal Digestion Impact on the Bioaccessibility and Antioxidant Capacity of Bioactive Compounds from Tomato Flours Obtained after Conventional and Ohmic Heating Extraction. Foods 2021, 10, 554. [Google Scholar] [CrossRef]
- Wang, M.L.; Chen, C.Y.; Tonnis, B.; Pinnow, D.; Davis, J.; An, Y.C.; Dang, P. Changes of Seed Weight, Fatty Acid Composition, and Oil and Protein Contents from Different Peanut FAD2 Genotypes at Different Seed Developmental and Maturation Stages. J. Agric. Food Chem. 2018, 66, 3658–3665. [Google Scholar] [CrossRef] [PubMed]
- King, J.C.; Blumberg, J.; Ingwersen, L.; Jenab, M.; Tucker, K.L. Tree Nuts and Peanuts as Components of a Healthy Diet. J. Nutr. 2008, 138, 1736S–1740S. [Google Scholar] [CrossRef] [PubMed]
- Arya, S.S.; Salve, A.R.; Chauhan, S. Peanuts as functional food: A review. J. Food Sci. Technol. 2016, 53, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Hertzler, S.R.; Lieblein-Boff, J.C.; Weiler, M.; Allgeier, C. Plant Proteins: Assessing Their Nutritional Quality and Effects on Health and Physical Function. Nutrients 2020, 12, 3704. [Google Scholar] [CrossRef] [PubMed]
- Aderibigbe, A.S.; Park, C.S.; Adebiyi, A.; Olukosi, O.A.; Adeola, O. Digestibility of Amino Acids in Protein-Rich Feed Ingredients Originating from Animals, Peanut Flour, and Full-Fat Soybeans Fed to Pigs. Animals 2020, 10, 2062. [Google Scholar] [CrossRef]
- Salem, M.A.; Aborehab, N.M.; Al-Karmalawy, A.A.; Fernie, A.R.; Alseekh, S.; Ezzat, S.M. Potential Valorization of Edible Nuts By-Products: Exploring the Immune-Modulatory and Antioxidants Effects of Selected Nut Shells Extracts in Relation to Their Metabolic Profiles. Antioxidants 2022, 11, 462. [Google Scholar] [CrossRef]
- Joint WHO/FAO/UNU Expert Consultation. Protein and Amino Acid Requirements in Human Nutrition; World Health Organization Technical Report Series; WHO: Geneva, Switzerland, 2007; pp. 1–265.
- Wu, H.; Wang, Q.; Ma, T.; Ren, J. Comparative studies on the functional properties of various protein concentrate preparations of peanut protein. Food Res. Int. 2009, 42, 343–348. [Google Scholar] [CrossRef]
- Zhao, X.; Chen, J.; Du, F. Potential use of peanut by-products in food processing: A review. J. Food Sci. Technol. 2012, 49, 521–529. [Google Scholar] [CrossRef]
- Young, C. Amino acid composition of three peanut varieties. J. Food Sci. 1984, 45, 1086–1087. [Google Scholar] [CrossRef]
- Zhao, Y.; Vanhoutte, P.M.; Leung, S.W.S. Vascular nitric oxide: Beyond eNOS. J. Pharmacol. Sci. 2015, 129, 83–94. [Google Scholar] [CrossRef]
- Liu, H.C.; Chen, W.L.; Mao, S.J.T. Antioxidant Nature of Bovine Milk β-Lactoglobulin. J. Dairy Sci. 2007, 90, 547–555. [Google Scholar] [CrossRef]
- Sebei, K.; Gnouma, A.; Herchi, W.; Sakouhi, F.; Boukhchina, S. Lipids, proteins, phenolic composition, antioxidant and antibacterial activities of seeds of peanuts (Arachis hypogaea l) cultivated in Tunisia. Biol. Res. 2013, 46, 257–263. [Google Scholar] [CrossRef]
- Prodic, I.; Stanic-Vucinic, D.; Apostolovic, D.; Mihailovic, J.; Radibratovic, M.; Radosavljevic, J.; Burazer, L.; Milcic, M.; Smiljanic, K.; van Hage, M.; et al. Influence of peanut matrix on stability of allergens in gastric-simulated digesta: 2S albumins are main contributors to the IgE reactivity of short digestion-resistant peptides. Clin. Exp. Allergy 2018, 48, 731–740. [Google Scholar] [CrossRef]
- Hebling, C.M.; Ross, M.M.; Callahan, J.H.; McFarland, M.A. Size-Selective Fractionation and Visual Mapping of Allergen Protein Chemistry in Arachis hypogaea. J. Proteome Res. 2012, 11, 5384–5395. [Google Scholar] [CrossRef]
- Kottapalli, K.; Rakwal, R.; Agrawal, G.; Shibato, J.; Burow, M.; Puppala, N. Proteomics analysis of mature seed of four peanut cultivars using two-dimensional gel electrophoresis reveals distinct differential expression of storage, anti-nutritional, and allergenic proteins. Plant Sci. 2008, 175, 321–329. [Google Scholar] [CrossRef]
- Johnson, P.E.; Sayers, R.L.; Gethings, L.A.; Balasundaram, A.; Marsh, J.T.; Langridge, J.I.; Mills, E.N. Quantitative Proteomic Profiling of Peanut Allergens in Food Ingredients Used for Oral Food Challenges. Anal. Chem. 2016, 88, 5689–5695. [Google Scholar] [CrossRef]
- Schwager, C.; Kull, S.; Behrends, J.; Röckendorf, N.; Schocker, F.; Frey, A.; Homann, A.; Becker, W.M.; Jappe, U. Peanut oleosins associated with severe peanut allergy-importance of lipophilic allergens for comprehensive allergy diagnostics. J. Allergy Clin. Immunol. 2017, 140, 1331–1338.e8. [Google Scholar] [CrossRef]
- Petersen, A.; Kull, S.; Rennert, S.; Becker, W.M.; Krause, S.; Ernst, M.; Gutsmann, T.; Bauer, J.; Lindner, B.; Jappe, U. Peanut defensins: Novel allergens isolated from lipophilic peanut extract. J. Allergy Clin. Immunol. 2015, 136, 1295–1301.e5. [Google Scholar] [CrossRef]
- Turner, P.J.; Jerschow, E.; Umasunthar, T.; Lin, R.; Campbell, D.E.; Boyle, R.J. Fatal Anaphylaxis: Mortality Rate and Risk Factors. J. Allergy Clin. Immunol. Pract. 2017, 5, 1169–1178. [Google Scholar] [CrossRef]
- Lieberman, J.A.; Gupta, R.S.; Knibb, R.C.; Haselkorn, T.; Tilles, S.; Mack, D.P.; Pouessel, G. The global burden of illness of peanut allergy: A comprehensive literature review. Allergy 2021, 76, 1367–1384. [Google Scholar] [CrossRef]
- Krstić Ristivojević, M.; Apostolović, D.; Smiljanić, K. Enterocytes in Food Hypersensitivity Reactions. Animals 2021, 11, 2713. [Google Scholar] [CrossRef] [PubMed]
- Koid, A.E.; Chapman, M.D.; Hamilton, R.G.; van Ree, R.; Versteeg, S.A.; Dreskin, S.C.; Koppelman, S.J.; Wünschmann, S. Ara h 6 complements Ara h 2 as an important marker for IgE reactivity to peanut. J. Agric. Food Chem. 2014, 62, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Finkina, E.I.; Melnikova, D.N.; Bogdanov, I.V.; Ovchinnikova, T.V. Plant Pathogenesis-Related Proteins PR-10 and PR-14 as Components of Innate Immunity System and Ubiquitous Allergens. Curr. Med. Chem. 2017, 24, 1772–1787. [Google Scholar] [CrossRef] [PubMed]
- Mittag, D.; Akkerdaas, J.; Ballmer-Weber, B.K.; Vogel, L.; Wensing, M.; Becker, W.M.; Koppelman, S.J.; Knulst, A.C.; Helbling, A.; Hefle, S.L.; et al. Ara h 8, a Bet v 1-homologous allergen from peanut, is a major allergen in patients with combined birch pollen and peanut allergy. J. Allergy Clin. Immunol. 2004, 114, 1410–1417. [Google Scholar] [CrossRef] [PubMed]
- Florsheim, E.B.; Sullivan, Z.A.; Khoury-Hanold, W.; Medzhitov, R. Food allergy as a biological food quality control system. Cell 2021, 184, 1440–1454. [Google Scholar] [CrossRef]
- Zhao, X.; Fu, Y.; Song, C. Proteins. In Essentials of Food Chemistry; Kan, J., Chen, K., Eds.; Springer: Singapore, 2021; pp. 49–121. [Google Scholar]
- Sathe, S.K.; Venkatachalam, M.; Sharma, G.M.; Kshirsagar, H.H.; Teuber, S.S.; Roux, K.H. Solubilization and electrophoretic characterization of select edible nut seed proteins. J. Agric. Food Chem. 2009, 57, 7846–7856. [Google Scholar] [CrossRef]
- Guo, C.; Xie, Y.-J.; Zhu, M.-T.; Xiong, Q.; Chen, Y.; Yu, Q.; Xie, J.-H. Influence of different cooking methods on the nutritional and potentially harmful components of peanuts. Food Chem. 2020, 316, 126269. [Google Scholar] [CrossRef]
- Koppelman, S.; Apostolovic, D.; Warmenhoven, H.; Verbart, D.; Taylor, S.; Isleib, T.; Maleki, S. The content of allergens Ara h1, Ara h2, Ara h3, and Ara h6 in different peanut cultivars commonly consumed in Europe and the USA. Allergy 2012, 96, 452–586. [Google Scholar]
- Perkins, D.; Toledo, R. Effect of Heat Treatment for Trypsin Inhibitor Inactivation on Physical and Functional Properties of Peanut Protein. J. Food Sci. 1982, 47, 917–923. [Google Scholar] [CrossRef]
- Viquez, O.M.; Konan, K.N.; Dodo, H.W. Structure and organization of the genomic clone of a major peanut allergen gene, Ara h 1. Mol. Immunol. 2003, 40, 565–571. [Google Scholar] [CrossRef]
- Maleki, S.J.; Viquez, O.; Jacks, T.; Dodo, H.; Champagne, E.T.; Chung, S.Y.; Landry, S.J. The major peanut allergen, Ara h 2, functions as a trypsin inhibitor, and roasting enhances this function. J. Allergy Clin. Immunol. 2003, 112, 190–195. [Google Scholar] [CrossRef]
- Dodo, H.W.; Viquez, O.M.; Maleki, S.J.; Konan, K.N. cDNA clone of a putative peanut (Arachis hypogaea L.) trypsin inhibitor has homology with peanut allergens Ara h 3 and Ara h 4. J. Agric. Food Chem. 2004, 52, 1404–1409. [Google Scholar] [CrossRef]
- Schmitt, D.A.; Nesbit, J.B.; Hurlburt, B.K.; Cheng, H.; Maleki, S.J. Processing Can Alter the Properties of Peanut Extract Preparations. J. Agric. Food Chem. 2010, 58, 1138–1143. [Google Scholar] [CrossRef]
- Fu, T.-J.; Maks, N. Impact of Thermal Processing on ELISA Detection of Peanut Allergens. J. Agric. Food Chem. 2013, 61, 5649–5658. [Google Scholar] [CrossRef]
- Poms, R.E.; Capelletti, C.; Anklam, E. Effect of roasting history and buffer composition on peanut protein extraction efficiency. Mol. Nutr. Food Res. 2004, 48, 459–464. [Google Scholar] [CrossRef]
- Koppelman, S.J.; Smits, M.; Tomassen, M.; de Jong, G.A.H.; Baumert, J.; Taylor, S.L.; Witkamp, R.; Veldman, R.J.; Pieters, R.; Wichers, H. Release of Major Peanut Allergens from Their Matrix under Various pH and Simulated Saliva Conditions-Ara h2 and Ara h6 Are Readily Bio-Accessible. Nutrients 2018, 10, 1281. [Google Scholar] [CrossRef]
- Tian, Y.; Rao, H.; Tao, S.; Xue, W.-T. Effect of boiling on the structure and immunoreactivity of recombinant peanut protein Ara h 1. Food Agric. Immunol. 2018, 29, 845–858. [Google Scholar] [CrossRef]
- Webb, K.E., Jr. Amino acid and peptide absorption from the gastrointestinal tract. Fed. Proc. 1986, 45, 2268–2271. [Google Scholar]
- Kopper, R.; Odum, N.; Helm, R.; Stanley, J.S.; Burks, A. Peanut Protein Allergens: The Effect of Roasting on Solubility and Allergenicity. Int. Arch. Allergy Immunol. 2005, 136, 16–22. [Google Scholar] [CrossRef]
- Zhang, T.; Shi, Y.; Zhao, Y.; Wang, J.; Wang, M.; Niu, B.; Chen, Q. Different thermal processing effects on peanut allergenicity. J. Sci. Food Agric. 2019, 99, 2321–2328. [Google Scholar] [CrossRef]
- Cabanillas, B.; Novak, N. Effects of daily food processing on allergenicity. Crit. Rev. Food Sci. Nutr. 2019, 59, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Rao, H.; Wenhui, F.; Xue, W. In vitro digestibility and Immunoreactivity of thermally processed peanut. Food Agric. Immunol. 2018, 29, 989–1001. [Google Scholar] [CrossRef]
- Beyer, K.; Morrowa, E.; Li, X.-M.; Bardina, L.; Bannon, G.A.; Burks, A.W.; Sampson, H.A. Effects of cooking methods on peanut allergenicity. J. Allergy Clin. Immunol. 2001, 107, 1077–1081. [Google Scholar] [CrossRef] [PubMed]
- Maleki, S.J.; Chung, S.-Y.; Champagne, E.T.; Raufman, J.-P. The effects of roasting on the allergenic properties of peanut proteins. J. Allergy Clin. Immunol. 2000, 106, 763–768. [Google Scholar] [CrossRef]
- Vissers, Y.M.; Iwan, M.; Adel-Patient, K.; Stahl Skov, P.; Rigby, N.M.; Johnson, P.E.; Mandrup Müller, P.; Przybylski-Nicaise, L.; Schaap, M.; Ruinemans-Koerts, J.; et al. Effect of roasting on the allergenicity of major peanut allergens Ara h 1 and Ara h 2/6: The necessity of degranulation assays. Clin. Exp. Allergy 2011, 41, 1631–1642. [Google Scholar] [CrossRef]
- Zhou, H.; Wu, Z.; Chang, X.; Tang, Y.; Yuan, J.; Li, X.; Yang, A.; Tong, P.; Chen, H. The effect of roasting on peanut allergens’ digestibility, allergenicity, and structure. Food Biosci. 2021, 44, 101454. [Google Scholar] [CrossRef]
- Zhang, D.; Guo, X.; Wang, Q.; Zhao, L.; Sun, Q.; Duan, X.; Cao, Y.; Sun, H. Investigation on lipid profile of peanut oil and changes during roasting by lipidomic approach. LWT 2022, 154, 112594. [Google Scholar] [CrossRef]
- Bilal, M.; Shabbir, M.A.; Xiaobo, Z.; Arslan, M.; Usman, M.; Azam, M.; Aadil, R.M.; Ahmad, N. Characterization of peanut seed oil of selected varieties and its application in the cereal-based product. J. Food Sci. Technol. 2020, 57, 4044–4053. [Google Scholar] [CrossRef]
- Sheppard, A.J.; Rudolf, T.S. Analysis of Peanuts and Peanut Products for Total Lipids, Fatty Acids and Proximates. Peanut Sci. 1991, 18, 51–54. [Google Scholar] [CrossRef]
- Sanders, T.H. Peanuts. In Encyclopedia of Food Sciences and Nutrition, 2nd ed.; Caballero, B., Ed.; Academic Press: Oxford, UK, 2003; pp. 4420–4427. [Google Scholar]
- Huang, Y.; Ma, R.; Xu, Y.; Zhong, K.; Bu, Q.; Gao, H. A Comparison of Lipid Contents in Different Types of Peanut Cultivars Using UPLC-Q-TOF-MS-Based Lipidomic Study. Foods 2021, 11, 4. [Google Scholar] [CrossRef]
- Zhang, D.; Li, X.; Cao, Y.; Wang, C.; Xue, Y. Effect of roasting on the chemical components of peanut oil. LWT 2020, 125, 109249. [Google Scholar] [CrossRef]
- Dun, Q.; Yao, L.; Deng, Z.; Li, H.; Li, J.; Fan, Y.; Zhang, B. Effects of hot and cold-pressed processes on volatile compounds of peanut oil and corresponding analysis of characteristic flavor components. LWT 2019, 112, 107648. [Google Scholar] [CrossRef]
- Juan-Polo, A.; Sanahuja, A.B.; Prats Moya, M.S.; Monedero Prieto, M.; Sanchez Reig, C.; Maestre Pérez, S.E. Optimization and validation of a simplified methodology for simultaneous extraction of fatty acids and tocopherol homologues in peanuts. J. Food Compos. Anal. 2022, 106, 104287. [Google Scholar] [CrossRef]
- Silva, M.; Martinez, M.J.; Casini, C.; Grosso, N. Tocopherol content, peroxide value and sensory attributes in roasted peanuts during storage. Int. J. Food Sci. Technol. 2010, 45, 1499–1504. [Google Scholar] [CrossRef]
- Lee, S.; Choi, Y.; Jeong, H.S.; Lee, J.; Sung, J. Effect of different cooking methods on the content of vitamins and true retention in selected vegetables. Food Sci. Biotechnol. 2018, 27, 333–342. [Google Scholar] [CrossRef]
- Matros, A.; Peshev, D.; Peukert, M.; Mock, H.P.; Van den Ende, W. Sugars as hydroxyl radical scavengers: Proof-of-concept by studying the fate of sucralose in Arabidopsis. Plant J. Cell Mol. Biol. 2015, 82, 822–839. [Google Scholar] [CrossRef]
- Kopjar, M.; Loncaric, A.; Mikulinjak, M.; Srajbek, Z.; Srajbek, M.; Pichler, A. Evaluation of Antioxidant Interactions of Combined Model Systems of Phenolics in the Presence of Sugars. Nat. Prod. Commun. 2016, 11, 1445–1448. [Google Scholar] [CrossRef]
- Bolouri-Moghaddam, M.R.; Le Roy, K.; Xiang, L.; Rolland, F.; Van den Ende, W. Sugar signalling and antioxidant network connections in plant cells. FEBS J. 2010, 277, 2022–2037. [Google Scholar] [CrossRef]
- Capuano, E. The behavior of dietary fiber in the gastrointestinal tract determines its physiological effect. Crit. Rev. Food Sci. Nutr. 2017, 57, 3543–3564. [Google Scholar] [CrossRef]
- Zhang, T.; Yang, Y.; Liang, Y.; Jiao, X.; Zhao, C. Beneficial Effect of Intestinal Fermentation of Natural Polysaccharides. Nutrients 2018, 10, 1055. [Google Scholar] [CrossRef]
- Zhao, L.; Qin, Y.; Guan, R.; Zheng, W.; Liu, J.; Zhao, J. Digestibility of fucosylated glycosaminoglycan from sea cucumber and its effects on digestive enzymes under simulated salivary and gastrointestinal conditions. Carbohydr. Polym. 2018, 186, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Mohanta, B.; Sen, D.J.; Mahanti, B.; Nayak, A.K. Antioxidant potential of herbal polysaccharides: An overview on recent researches. Sens. Int. 2022, 3, 100158. [Google Scholar] [CrossRef]
- Vibhakar, C.; Sheena, A.; Rohan, V.P.; Jigna, G.T. Physiological Function of Phenolic Compounds in Plant Defense System. In Phenolic Compounds; Farid, A.B., Ed.; IntechOpen: Rijeka, Croatia, 2021; pp. 185–205, Ch. 10. [Google Scholar]
- Tiemi Abe, L.; Maria Lajolo, F.; Genovese, M.I. Comparison of phenol content and antioxidant capacity of nuts. Ciência Tecnol. Aliment. 2010, 30, 254–259. [Google Scholar]
- Craft, B.D.; Kosińska, A.; Amarowicz, R.; Pegg, R.B. Antioxidant properties of extracts obtained from raw, dry-roasted, and oil-roasted US peanuts of commercial importance. Plant Foods Hum. Nutr. 2010, 65, 311–318. [Google Scholar] [CrossRef]
- Attree, R.; Du, B.; Xu, B. Distribution of phenolic compounds in seed coat and cotyledon, and their contribution to antioxidant capacities of red and black seed coat peanuts (Arachis hypogaea L.). Ind. Crops Prod. 2015, 67, 448–456. [Google Scholar] [CrossRef]
- Dean, L.L. Extracts of Peanut Skins as a Source of Bioactive Compounds: Methodology and Applications. Appl. Sci. 2020, 10, 8546. [Google Scholar] [CrossRef]
- Resurreccion, A.V.A.; Sales, J.; Potrebko, I.; Francisco, L.; Hitchcock, H. Peanuts bioactive food in a shell. Food Technol. 2009, 63, 30–36. [Google Scholar]
- Stuetz, W.; Schlörmann, W.; Glei, M. B-vitamins, carotenoids and α-/γ-tocopherol in raw and roasted nuts. Food Chem. 2017, 221, 222–227. [Google Scholar] [CrossRef]
- Embaby, H.E.-S. Effect of heat treatments on certain antinutrients and in vitro protein digestibility of peanut and sesame seeds. Food Sci. Technol. Res. 2011, 17, 31–38. [Google Scholar] [CrossRef]
- Soleas, G.J.; Diamandis, E.P.; Goldberg, D.M. Resveratrol: A molecule whose time has come? And gone? Clin. Biochem. 1997, 30, 91–113. [Google Scholar] [CrossRef]
- Olas, B.; Wachowicz, B. Resveratrol, a phenolic antioxidant with effects on blood platelet functions. Platelets 2005, 16, 251–260. [Google Scholar] [CrossRef]
- Wojtunik-Kulesza, K.; Oniszczuk, A.; Oniszczuk, T.; Combrzyński, M.; Nowakowska, D.; Matwijczuk, A. Influence of In Vitro Digestion on Composition, Bioaccessibility and Antioxidant Activity of Food Polyphenols-A Non-Systematic Review. Nutrients 2020, 12, 1401. [Google Scholar] [CrossRef]
- Woźniak, D.; Cichy, W.; Przysławski, J.; Drzymała-Czyż, S. The role of microbiota and enteroendocrine cells in maintaining homeostasis in the human digestive tract. Adv. Med. Sci. 2021, 66, 284–292. [Google Scholar] [CrossRef]
- Mandalari, G.; Vardakou, M.; Faulks, R.; Bisignano, C.; Martorana, M.; Smeriglio, A.; Trombetta, D. Food Matrix Effects of Polyphenol Bioaccessibility from Almond Skin during Simulated Human Digestion. Nutrients 2016, 8, 568. [Google Scholar] [CrossRef]
- Sousa, R.; Portmann, R.; Dubois, S.; Recio, I.; Egger, L. Protein digestion of different protein sources using the INFOGEST static digestion model. Food Res. Int. 2020, 130, 108996. [Google Scholar] [CrossRef]
- Brodkorb, A.; Egger, L.; Alminger, M.; Alvito, P.; Assunção, R.; Ballance, S.; Bohn, T.; Bourlieu-Lacanal, C.; Boutrou, R.; Carrière, F.; et al. INFOGEST static in vitro simulation of gastrointestinal food digestion. Nat. Protoc. 2019, 14, 991–1014. [Google Scholar] [CrossRef]
- Ribeiro, T.B.; Oliveira, A.; Campos, D.; Nunes, J.; Vicente, A.A.; Pintado, M. Simulated digestion of an olive pomace water-soluble ingredient: Relationship between the bioaccessibility of compounds and their potential health benefits. Food Funct. 2020, 11, 2238–2254. [Google Scholar] [CrossRef]
- Tomé-Sánchez, I.; Martín-Diana, A.B.; Peñas, E.; Frias, J.; Rico, D.; Jiménez-Pulido, I.; Martínez-Villaluenga, C. Bioprocessed Wheat Ingredients: Characterization, Bioaccessibility of Phenolic Compounds, and Bioactivity During in vitro Digestion. Front. Plant Sci. 2021, 12, 790898. [Google Scholar] [CrossRef]
- Tonolo, F.; Moretto, L.; Grinzato, A.; Fiorese, F.; Folda, A.; Scalcon, V.; Ferro, S.; Arrigoni, G.; Bellamio, M.; Feller, E.; et al. Fermented Soy-Derived Bioactive Peptides Selected by a Molecular Docking Approach Show Antioxidant Properties Involving the Keap1/Nrf2 Pathway. Antioxidants 2020, 9, 1306. [Google Scholar] [CrossRef]
- Calvo-Lerma, J.; Paz-Yépez, C.; Asensio-Grau, A.; Heredia, A.; Andrés, A. Impact of Processing and Intestinal Conditions on in Vitro Digestion of Chia (Salvia hispanica) Seeds and Derivatives. Foods 2020, 9, 290. [Google Scholar] [CrossRef]
- Darewicz, M.; Pliszka, M.; Borawska-Dziadkiewicz, J.; Minkiewicz, P.; Iwaniak, A. Multi-Bioactivity of Protein Digests and Peptides from Oat (Avena sativa L.) Kernels in the Prevention of the Cardiometabolic Syndrome. Molecules 2022, 27, 7907. [Google Scholar] [CrossRef]
- Sánchez-Velázquez, O.A.; Cuevas-Rodríguez, E.O.; Mondor, M.; Ribéreau, S.; Arcand, Y.; Mackie, A.; Hernández-Álvarez, A.J. Impact of in vitro gastrointestinal digestion on peptide profile and bioactivity of cooked and non-cooked oat protein concentrates. Curr. Res. Food Sci. 2021, 4, 93–104. [Google Scholar] [CrossRef]
- Walle, T.; Browning, A.M.; Steed, L.L.; Reed, S.G.; Walle, U.K. Flavonoid Glucosides Are Hydrolyzed and Thus Activated in the Oral Cavity in Humans. J. Nutr. 2005, 135, 48–52. [Google Scholar] [CrossRef]
- Lu, Y.; Bennick, A. Interaction of tannin with human salivary proline-rich proteins. Arch. Oral Biol. 1998, 43, 717–728. [Google Scholar] [CrossRef]
- Zhu, K.; Yao, S.; Zhang, Y.; Liu, Q.; Xu, F.; Wu, G.; Dong, W.; Tan, L. Effects of in vitro saliva, gastric and intestinal digestion on the chemical properties, antioxidant activity of polysaccharide from Artocarpus heterophyllus Lam. (Jackfruit) Pulp. Food Hydrocoll. 2019, 87, 952–959. [Google Scholar] [CrossRef]
- Feng, X.; Bie, N.; Li, J.; Zhang, M.; Feng, Y.; Ding, T.; Zhao, Y.; Wang, C. Effect of in vitro simulated gastrointestinal digestion on the antioxidant activity, molecular weight, and microstructure of polysaccharides from Chinese yam. Int. J. Biol. Macromol. 2022, 207, 873–882. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on the evaluation of allergenic foods and food ingredients for labelling purposes. EFSA J. 2014, 12, 3894. [Google Scholar] [CrossRef]
- Prodic, I.; Smiljanic, K.; Nagl, C.; Ballmer-Weber, B.; Hoffmann-Sommergruber, K.; Velickovic, T.C. INFOGEST Digestion Assay of Raw and Roasted Hazelnuts and Its Impact on Allergens and Their IgE Binding Activity. Foods 2022, 11, 2914. [Google Scholar] [CrossRef]
- Di Stasio, L.; Picariello, G.; Mongiello, M.; Nocerino, R.; Berni Canani, R.; Bavaro, S.; Monaci, L.; Ferranti, P.; Mamone, G. Peanut digestome: Identification of digestion resistant IgE binding peptides. Food Chem. Toxicol. 2017, 107, 88–98. [Google Scholar] [CrossRef]
- Smiljanic, K.; Prodic, I.; Apostolovic, D.; Cvetkovic, A.; Veljovic, D.; Mutic, J.; van Hage, M.; Burazer, L.; Cirkovic Velickovic, T. In-depth quantitative profiling of post-translational modifications of Timothy grass pollen allergome in relation to environmental oxidative stress. Environ. Int. 2019, 126, 644–658. [Google Scholar] [CrossRef]
- Koppelman, S.J.; Hefle, S.L.; Taylor, S.L.; de Jong, G.A.H. Digestion of peanut allergens Ara h 1, Ara h 2, Ara h 3, and Ara h 6: A comparative in vitro study and partial characterization of digestion-resistant peptides. Mol. Nutr. Food Res. 2010, 54, 1711–1721. [Google Scholar] [CrossRef]
- Apostolovic, D.; Stanic-Vucinic, D.; de Jongh, H.H.; de Jong, G.A.; Mihailovic, J.; Radosavljevic, J.; Radibratovic, M.; Nordlee, J.A.; Baumert, J.L.; Milcic, M.; et al. Conformational stability of digestion-resistant peptides of peanut conglutins reveals the molecular basis of their allergenicity. Sci. Rep. 2016, 6, 29249. [Google Scholar] [CrossRef]
- Mouécoucou, J.; Frémont, S.; Sanchez, C.; Villaume, C.; Méjean, L. In vitro allergenicity of peanut after hydrolysis in the presence of polysaccharides. Clin. Exp. Allergy 2004, 34, 1429–1437. [Google Scholar] [CrossRef]
- Cao, H.; Saroglu, O.; Karadag, A.; Diaconeasa, Z.; Zoccatelli, G.; Conte-Junior, C.A.; Gonzalez-Aguilar, G.A.; Ou, J.; Bai, W.; Zamarioli, C.M.; et al. Available technologies on improving the stability of polyphenols in food processing. Food Front. 2021, 2, 109–139. [Google Scholar] [CrossRef]
- Martinez-Gonzalez, A.I.; Díaz-Sánchez, Á.G.; Rosa, L.A.d.l.; Vargas-Requena, C.L.; Bustos-Jaimes, I.; Alvarez-Parrilla, A.E. Polyphenolic Compounds and Digestive Enzymes: In Vitro Non-Covalent Interactions. Molecules 2017, 22, 669. [Google Scholar] [CrossRef]
- Dyer, S.; Nesbit, J.B.; Cabanillas, B.; Cheng, H.; Hurlburt, B.K.; Maleki, S.J. Contribution of Chemical Modifications and Conformational Epitopes to IgE Binding by Ara h 3. Foods 2018, 7, 189. [Google Scholar] [CrossRef]
- Kolarich, D.; Altmann, F. N-Glycan analysis by matrix-assisted laser desorption/ionization mass spectrometry of electrophoretically separated nonmammalian proteins: Application to peanut allergen Ara h 1 and olive pollen allergen Ole e 1. Anal. Biochem. 2000, 285, 64–75. [Google Scholar] [CrossRef]
- Li, J.; Shefcheck, K.; Callahan, J.; Fenselau, C. Primary sequence and site-selective hydroxylation of prolines in isoforms of a major peanut allergen protein Ara h 2. Protein Sci. 2010, 19, 174–182. [Google Scholar] [CrossRef]
- Marsh, J.T.; Palmer, L.K.; Koppelman, S.J.; Johnson, P.E. Determination of Allergen Levels, Isoforms, and Their Hydroxyproline Modifications Among Peanut Genotypes by Mass Spectrometry. Front. Allergy 2022, 3, 872714. [Google Scholar] [CrossRef]
- Teodorowicz, M.; van Neerven, J.; Savelkoul, H. Food Processing: The Influence of the Maillard Reaction on Immunogenicity and Allergenicity of Food Proteins. Nutrients 2017, 9, 835. [Google Scholar] [CrossRef]
- Deng, Y.; Wierenga, P.A.; Schols, H.A.; Sforza, S.; Gruppen, H. Effect of Maillard induced glycation on protein hydrolysis by lysine/arginine and non-lysine/arginine specific proteases. Food Hydrocoll. 2017, 69, 210–219. [Google Scholar] [CrossRef]
- Zhu, Y.; Lao, F.; Pan, X.; Wu, J. Food Protein-Derived Antioxidant Peptides: Molecular Mechanism, Stability and Bioavailability. Biomolecules 2022, 12, 1622. [Google Scholar] [CrossRef] [PubMed]
- Galvez, A.F.; de Lumen, B.O. A soybean cDNA encoding a chromatin-binding peptide inhibits mitosis of mammalian cells. Nat. Biotechnol. 1999, 17, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Akbarian, M.; Khani, A.; Eghbalpour, S.; Uversky, V.N. Bioactive Peptides: Synthesis, Sources, Applications, and Proposed Mechanisms of Action. Int. J. Mol. Sci. 2022, 23, 1445. [Google Scholar] [CrossRef]
- Tadesse, S.A.; Emire, S.A. Production and processing of antioxidant bioactive peptides: A driving force for the functional food market. Heliyon 2020, 6, e04765. [Google Scholar] [CrossRef]
- Elias, R.J.; Kellerby, S.S.; Decker, E.A. Antioxidant activity of proteins and peptides. Crit. Rev. Food Sci. Nutr. 2008, 48, 430–441. [Google Scholar] [CrossRef]
- Abd El-Fattah, A.M.; Sakr, S.S.; El-Dieb, S.M.; Elkashef, H.A.S. Bioactive peptides with ACE-I and antioxidant activity produced from milk proteolysis. Int. J. Food Prop. 2017, 20, 3033–3042. [Google Scholar] [CrossRef]
- He, R.; Ju, X.; Yuan, J.; Wang, L.; Girgih, A.T.; Aluko, R.E. Antioxidant activities of rapeseed peptides produced by solid state fermentation. Food Res. Int. 2012, 49, 432–438. [Google Scholar] [CrossRef]
- Zou, T.B.; He, T.P.; Li, H.B.; Tang, H.W.; Xia, E.Q. The Structure-Activity Relationship of the Antioxidant Peptides from Natural Proteins. Molecules 2016, 21, 72. [Google Scholar] [CrossRef]
- Shi, C.; Liu, M.; Zhao, H.; Lv, Z.; Liang, L.; Zhang, B. A Novel Insight into Screening for Antioxidant Peptides from Hazelnut Protein: Based on the Properties of Amino Acid Residues. Antioxidants 2022, 11, 127. [Google Scholar] [CrossRef]
- Girgih, A.T.; He, R.; Malomo, S.; Offengenden, M.; Wu, J.; Aluko, R.E. Structural and functional characterization of hemp seed (Cannabis sativa L.) protein-derived antioxidant and antihypertensive peptides. J. Funct. Foods 2014, 6, 384–394. [Google Scholar] [CrossRef]
- Chen, G.-T.; Zhao, L.; Zhao, L.-Y.; Cong, T.; Bao, S.-F. In vitro study on antioxidant activities of peanut protein hydrolysate. J. Sci. Food Agric. 2007, 87, 357–362. [Google Scholar] [CrossRef]
- Hwang, J.-Y.; Wang, Y.T.; Shyu, Y.-S.; Wu, J.S.-B. Antimutagenic and antiproliferative effects of roasted and defatted peanut dregs on human leukemic U937 and HL-60 cells. Phytother. Res. 2008, 22, 286–290. [Google Scholar] [CrossRef]
- Liu, M.-C.; Yang, S.-J.; Hong, D.; Yang, J.-P.; Liu, M.; Lin, Y.; Huang, C.-H.; Wang, C.-J. A simple and convenient method for the preparation of antioxidant peptides from walnut (Juglans regia L.) protein hydrolysates. Chem. Cent. J. 2016, 10, 39. [Google Scholar] [CrossRef]
- Zhao, F.; Liu, C.; Bordoni, L.; Petracci, I.; Wu, D.; Fang, L.; Wang, J.; Wang, X.; Gabbianelli, R.; Min, W. Advances on the Antioxidant Peptides from Nuts: A Narrow Review. Antioxidants 2022, 11, 2020. [Google Scholar] [CrossRef]
- Nyo, M.K.; Nguyen, L.T. Value-Addition of Defatted Peanut Cake by Proteolysis: Effects of Proteases and Degree of Hydrolysis on Functional Properties and Antioxidant Capacity of Peptides. Waste Biomass Valorization 2019, 10, 1251–1259. [Google Scholar] [CrossRef]

| Protein Family (Pfam Database) | Allergome/Enzyme | UniProt Access. | Protein Name from UniProt Database | Relative Abundance % (XIC Area) | |||
|---|---|---|---|---|---|---|---|
| Avrg. Mass (kDa) | Gastric Ctrl [19] | Gastric Digest [19] | PBS pH7,4 [18] | ||||
| 1. Cupin (Vicillin-type, 7S globulin) | Ara h 1 | Q6PSU4 | Conarachin (Fragment) | 48.1 | 0.1 | 0.3 | / |
| P43238 = N1NG13 | Seed storage protein Ara h1 | 71.3 | 0.4 | 10.4 | / | ||
| Q6PSU3 | Conarachin (Fragment) | 66.6 | 0.3 | 2.4 | / | ||
| P43237 = B3IXL2 | Allergen Ara h 1 clone P17 | 70.3 | 0 | 4.9 | 20.3 | ||
| E9LFE7 | 7S conarachin (Fragment) | 15.9 | 0 | 0 | / | ||
| Q6PSU5 | Conarachin (Fragment) | 33.6 | 0 | 0.1 | / | ||
| Q6PSU6 | Conarachin (Fragment) | 34.1 | 0 | 0.1 | / | ||
| E5G076 | Ara h 1 allergen | 70.8 | 0.3 | 0 | 4.3 | ||
| 2. Conglutin (2S albumin) | Ara h 2 | Q6PSU2 | Conglutin-7 | 20.1 | / | / | 6.1 |
| Ara h 6 | Q647G9 | Conglutin | 16.9 | / | / | 8.6 | |
| Ara h 7 | B4XID4 | Ara h 7 allergen | 19.3 | / | / | 0 | |
| Q647G8 | 2S protein 2 (Ara h 7.0301) | 18.5 | / | / | 0.0 | ||
| 3. Cupin (Legumin-type, 11S globulin, Glycinin) | Ara h 3 | A1DZF0 | Arachin 6 | 60.4 | 1.7 | 7.7 | 0 |
| Q9FZ11 | Gly1 | 60.4 | 1.3 | 0.2 | 0.3 | ||
| B5TYU1 | Arachin Arah3 isoform | 60.6 | 3.1 | 6.6 | / | ||
| Q5I6T2 | Arachin Ahy-4 | 60.7 | 0.8 | 0.1 | 0 | ||
| Q647H4 | Arachin Ahy-1 | 61.5 | 0.9 | 0.1 | 0.3 | ||
| Q647H3 | Arachin Ahy-2 | 61.5 | 17.6 | 3.3 | 4.4 | ||
| Q8LKN1 | Allergen Arah3/Arah4 | 61.7 | 1.6 | 4.1 | / | ||
| Q9SQH7 | Glycinin | 61.0 | 0.8 | 5.5 | 0 | ||
| Q8LL03 | Trypsin inhibitor (Fragment) | 25.5 | 0.8 | 0 | / | ||
| Q6T2T4 | Storage protein | 61.5 | 0 | 0.1 | / | ||
| Q6IWG5 = Q0GM57 | Glycinin (Fragment) | 58.1 | 62.9 | 49.3 | 24.9 | ||
| E5G077 | Ara h 3 allergen | 58.3 | 0.9 | 4.3 | / | ||
| Q647H2 | Arachin Ahy-3 | 54.6 | 2.2 | 0 | 1.0 | ||
/ | E9LFE9 | 11S arachin | 28.3 | 0 | 0 | 1.4 | |
| E9LFE8 | 11S arachin (Fragment) | 28.3 | 0 | 0 | 8.8 | ||
| A1E2B0 | 11S seed storage globulin B1 | 33.5 | 0 | 0 | 4.4 | ||
| A1E2B1 | 11S seed storage globulin B2 | 31.3 | / | / | 1.6 | ||
| A7LIS5 | Germin-like protein | 23.5 | / | / | 0 | ||
| D4NXQ0 | Germin-like protein subfamily 3 member 3 | 23.4 | / | / | 0 | ||
| Q647H1 | Conarachin | 75.9 | / | / | 0.4 | ||
| 4. Profilin | Ara h 5 | D3K177 | Profilin | 14.2 | Johnson et al., 2016 [38] | ||
| 5. Pathogenesis-related protein, PR-10, Bet v 1 family member | Ara h 8 | Q6VT83 | Ara h 8 allergen | 17.0 | / | / | 0 |
| B1PYZ4 | Ara h 8 allergen isoform 3 | 16.9 | / | / | 0 | ||
| B0YIU5 | Ara h 8 allergen isoform | 16.4 | / | / | 0 | ||
| Q2YHR1 | Pathogenesis-related protein 10 (Fragment) | 14.4 | / | / | 0.1 | ||
| B2ZGS2 | Pathogenesis-related class 10 protein | 16.9 | / | / | 0.1 | ||
| 6. Plant LTP family 1 | Ara h 9 | B6CEX8 | Non-specific lipid-transfer protein | 11.7 | / | / | 0.2 |
| B6CG41 | Non-specific lipid-transfer protein (Fragment) | 9.1 | / | / | 0.2 | ||
| Ara h 17 | A0A510A9S3 | Non-specific lipid-transfer protein 1 | 9.4 | http://allergen.org/viewallergen.php?aid=831, accessed on 1 February 2023 | |||
| 7. Plant LTP family 2 | Ara h 16 | A0A509ZX51 | Non-specific lipid transfer protein 2 | 7.0 | http://allergen.org/viewallergen.php?aid=830, accessed on 1 February 2023 | ||
| 8. Oleosin family | Ara h 10 | Q647G5 | Oleosin Ara h 10.0101 | 17.7 | / | / | 0.1 |
| Q647G4 | Oleosin 17.8 (Ara h 10.0102) | 15.5 | / | / | 0.1 | ||
| Ara h 11 | Q45W86 | Oleosin Ara h 11.0102 | 14.3 | ||||
| Q45W87 | Oleosin Ara h 11.0101 | 14.3 | |||||
| Ara h 14 | Q6J1J8 | Oleosin Ara h 14.0103 | 18.4 | ||||
| Q9AXI0 | Oleosin Ara h 14.0102 | 18.5 | |||||
| Q9AXI1 | Oleosin Ara h 14.0101 | 18.4 | |||||
| Ara h 15 | Q647G3 | Oleosin Ara h 15.0101 | 16.9 | [39] | |||
| 9. Defensin family | Ara h 12 | B3EWP3 | Defensin 1 | 7.9 | [40] | ||
| Ara h 13 | C0HJZ1 | Defensin 3 | 8.3 | / | / | 0.2 | |
| 10. Cyclophilin family | Ara h 18 | - peptidyl-prolyl cis-trans isomerase | |||||
| 11. Leguminous lectin family | Ara h agglutinin | P02872 | Galactose-Binding Lectin | 29.3 | |||
| Q38711 | Galactose-binding lectin (Fragment) | 29.1 | 0 | 0 | 0.3 | ||
| A0A089ZXL7 | Peanut agglutinin variant | 29.4 | 0 | 0 | 0.1 | ||
| P02872 | Galactose-binding lectin | 29.3 | 2.5 | 0 | / | ||
| Q43373 | Galactose-binding lectin | 29.6 | 0 | 0 | / | ||
| 12. Chitinase class I | Hydrolase | Q42515 | Chitinase (Class II) | 28.9 | / | / | 0.1 |
| 13. Proteasome subunit | Q1PCR5 | 20S proteasome beta subunit Endopeptidase | 24.0 | / | / | 0 | |
| N1NJN8 | Proteasome subunit Peptidase T1B | 23.5 | / | / | 0 | ||
| B4UWD5 | Proteasome subunit alpha type Peptidase T1A | 15.0 | / | / | 0.1 | ||
| 14. Phospholipase D family | Q2HWT8 | Phospholipase D | 92.3 | ||||
| 15. RuBisCO large chain family | Lyase | O20356 | Ribulose bisphosphate carboxylase | 51.9 | |||
| A0A191UJ50 | Ribulose bisphosphate carboxylase large chain | 52.6 | / | / | 0.1 | ||
| 16. Glyoxalase/ /Dioxygenase superfamily | B4UWB4 | Lactoylglutathione lyase | 23.1 | ||||
| 17. Fructose-bisphosphate aldolase class-I | T2B9M0 | Fructose-bisphosphate aldolase | 38.4 | 0.1 | 0.3 | 2.7 | |
| 18. PEPCase type 1 family | C9W981 | Phosphoenolpyruvate carboxylase | 116.4 | 0 | 0 | / | |
| 19. Short chain dehydrogenase | Oxidoreductase | A7LB60 | Steroleosin-A | 38.8 | |||
| 20. Glutathione peroxidase family | B4UW79 | Glutathione peroxidase | 24.4 | ||||
| 21. Redoxin | Q06H32 | Glutaredoxin-dependent peroxiredoxin | 17.5 | ||||
| 22. Glyceraldehyde 3-phosphate dehydrogenase C | A0A0A6ZDP1 | Glyceraldehyde-3-phosphate dehydrogenase C2 | 20.1 | 0 | 0 | 4.8 | |
| A0A0A6ZDT0 | Glyceraldehyde-3-phosphate dehydrogenase C2 | 20.1 | 0.3 | 0 | 0.2 | ||
| 23. Lipoxygenase family | Q4JME6 | Lipoxygenase | 97.5 | 0 | 0 | / | |
| Q4JME7 | Lipoxygenase | 97.6 | 0 | 0 | / | ||
| Q9M5D3 | Lipoxygenase | 97.6 | 0.5 | 0 | / | ||
| 24. SDR family | E6Y9A9 | Enoyl-ACP reductase | 41.5 | / | / | 0 | |
| D8KXZ8 | Enoyl-ACP reductase 1–3 | 41.5 | / | / | 0 | ||
| 25. Alcohol dehydrogenase | H6U596 | Alcohol dehydrogenase (Fragment) | 19.4 | / | / | 1.0 | |
| 26. NAD dependent epimerase/dehydratase family | B4UW57 | Putative dihydroflavonol reductase (Fragment) | 24.3 | / | / | 0 | |
| 27. Cu-Zn superoxide dismutase family | Q1HDS7 | Superoxide dismutase [Cu-Zn] | 15.2 | / | / | 0 | |
| Q45W82 | Cu-Zn superoxide dismutase | 15.1 | / | / | 0 | ||
| 28. Glutathione S-transferase. N | Transferase | B4UW81 | Glutathione S-Transferase 2 | 20.3 | |||
| 29. Beta-ketoacyl-ACP synthases family | E6Y9A7 | Beta-ketoacyl-ACP synthetase I | 49.9 | [38] | |||
| 30. UDPGP type 1 family | Q06H19 | UDP-Glucose Pyrophosphorylase | 16.9 | ||||
| 31. Leucine Rich Repeat family | A0A290G0J9 | Resistance protein (Fragment) | 57.8 | 0.2 | 0 | / | |
| A0A290FZZ3 | Resistance protein (Fragment) | 41.5 | / | / | 0 | ||
| 32. Protein kinase family | A0A290GKJ7 | Resistance protein (Fragment) | 54.6 | 0.1 | 0 | / | |
| 33. RNA polymerase Rpb1 | A0A191UJ63 | DNA-directed RNA polymerase subunit beta | 157.0 | / | / | 0 | |
| 34. SHMT family | A0A0A6ZDR9 | Serine hydroxymethyltransferase | 52.0 | / | / | ||
| A0A0A6ZDT3 | Serine hydroxymethyltransferase | 52.0 | / | / | |||
| 35. Thiolase family | A0A0R4UXQ1 | 3-ketoacyl-CoA thiolase | 48.3 | / | / | 0.1 | |
| 36. NDK family | Q45W80 | Nucleoside diphosphate kinase | 16.4 | / | / | ||
| 37. Glutathione S-transferase. N-terminal domain | B4UW81 | Glutathione S-transferase 2 | 20.3 | / | / | 0.6 | |
| 38. AMP-binding enzyme | Ligase | A0A109QJM5 | Long chain acyl-CoA synthetase 4 | 74.3 | 0 | 0 | / |
| 39. Complex1_ 49kDa 1 | Translocase | A0A191UJC0 | NAD(P)H-quinone oxidoreductase subunit H. chloroplastic | 145.5 | 0 | 0 | / |
| 40. Chalcone isomerase family | Isomerase | J9QGM3 | Chalcone-flavonone isomerase family protein | 27.6 | 0 | 0 | / |
| 41. LEA type 1 family | / | Q2PXN4 | Seed maturation protein LEA 4 | 5.8 | |||
| 42. LEA type 2 family | / | Q2PXN9 | Dessication Protectant Protein LEA 14 | 16.2 | |||
| 43. LEAP-3 | / | E5FHY6 | Late embryogenesis abundant protein group 3 protein (Fragment) | 18.0 | / | / | 0 |
| 44. LEA type seed maturation protein family | / | E5FHZ2 | Late embryogenesis abundant protein group 5 | 27.4 | / | / | 0.1 |
| / | N1NKG9 | Late embryogenesis abundant protein | 26.0 | / | / | 0.2 | |
| 45. Small hydrophilic plant seed protein family | / | Q4U4M1 | LEA protein | 10.4 | |||
| E5FHY2 | Late embryogenesis abundant protein group 1 protein | 10.7 | / | / | 0 | ||
| E5FHY1 | Late embryogenesis abundant protein group 1 protein | 10.1 | / | / | 0 | ||
| 46. EF-hand domain pair | / | Q6PWX0 | Calmodulin | 16.7 | |||
| 47. Glutathione S-transferase, C terminus | / | Q1PCR4 | Putative IN2-1 Protein | 16.1 | |||
| 48. Cystatin family | / | B3GR01 | Cysteine proteinase inhibitor | 10.7 | |||
| 49. Bowman-Birk serine protease inhibitor family | / | Q0PKR5 | Proteinase inhibitor | 7.9 | |||
| / | Q2VMU0 | Serine protease inhibitor | 12.2 | ||||
| / | Q7X973 | Bowman-Birk trypsin inhibitor | 9.5 | ||||
| 50. Plant PEC family metalothionein | / | Q0Q0Q8 | Type 4 metallothionein | 9.1 | |||
| 51. NB-ARC family | / | Q2KQ45 | Resistance protein PLTR (Fragment) | 19.6 | 0.5 | 0 | / |
| / | A0A1B3TNS1 | NBS-LRR type disease resistance protein | 108.6 | 0 | 0 | / | |
| 52. DnaJ family | / | A0A126DIH0 | DnaJ | 39.1 | 0 | 0 | / |
| 53. Annexin family | / | A0A0F7GF62 | Annexin | 36.1 | / | / | 0.1 |
| A0A0F6VX63 | Annexin | 36.1 | / | / | 2.3 | ||
| B4UW70 | Fiber annexin | 18.8 | |||||
| 54. Phosphatidylethanolamine-binding protein family | / | K7PQ65 | Mother of FT and TFL1 | 19.2 | / | / | 0 |
| 55. TCTP family | / | Q06H31 | Translationally controlled tumour-like protein | 19.1 | / | / | 0.1 |
| 56. Actin-binding proteins ADF family | / | Q2PK12 | Actin depolymerising factor-like protein | 16.1 | / | / | 0 |
| 57. Trypsin and protease inhibitor | / | B4UWB2 | Kunitz trypsin inhibitor 4 | 22.1 | |||
| 58. Protein of unknown function | / | B4UWA3 | Putative Uncharacterised Protein | 20.2 | |||
| 59. Endoribonuclease L-PSP | / | B4UWE2 | Perchloric Acid Soluble Translation Inhibitor Protein | 20.3 | |||
| 60. Major intrinsic protein | / | Q06H34 | Tonoplast intrinsic protein alpha TIP | 19.2 | |||
| 61. Ubiquitin family | / | Q06H40 | Ubiquitin-Ribosomal Protein S27a | 21.4 | |||
| 62. Actin family | / | A0A0A1EUV7 | Actin (fragment) | 35.9 | [38] | ||
| Reference | Food/Biomolecules as the Contributor of Antioxidant Capacity | Simulated Digestion Protocol | Antioxidative Test Employed | Outcome |
|---|---|---|---|---|
| Coelho et al., 2021 [21] | Tomato pomace/phenolics | Modified INFOGEST 2.0 [102] | ABTS·+ (TEAC) | Significantly increased in GP (more) and IP (less) |
| Comert and Gokmen, 2022 [20] | Hazelnuts/phenolics Flaxseed/phenolics | INFOGEST 1.0 | ABTS·+ (TEAC) | Significantly increased in GP (more) and IP (more) |
| Tome-Sachez et al., 2021 [103] | Germinated whole-wheat grain/phenolics | INFOGEST 2.0 | DPPH, ORAC, ABTS·+, FRAP | Increased in GP (more) and IP (significantly compared to GP) |
| Tonolo et al., 2020 [104] | Soy/peptides | INFOGEST 1.0 | ABTS·+, DPPH | Decreased after GP and IP |
| Calvo-Lerma et al., 2020 [105] | Chia seed/phenolics | INFOGEST 1.0 | DPPH | Decreased after GP and IP |
| Darewicz et al., 2022 [106] | Oat/peptides | INFOGEST 1.0 | DPPH | Increased in GP (more) and IP (significantly compared to GP) |
| Sánchez-Velázquez et al., 2021 [107] | Oat/peptides | INFOGEST 2.0 | ABTS·+, DPPH | Increased in GP (more) and IP (significantly compared to GP) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prodić, I.; Krstić Ristivojević, M.; Smiljanić, K. Antioxidant Properties of Protein-Rich Plant Foods in Gastrointestinal Digestion—Peanuts as Our Antioxidant Friend or Foe in Allergies. Antioxidants 2023, 12, 886. https://doi.org/10.3390/antiox12040886
Prodić I, Krstić Ristivojević M, Smiljanić K. Antioxidant Properties of Protein-Rich Plant Foods in Gastrointestinal Digestion—Peanuts as Our Antioxidant Friend or Foe in Allergies. Antioxidants. 2023; 12(4):886. https://doi.org/10.3390/antiox12040886
Chicago/Turabian StyleProdić, Ivana, Maja Krstić Ristivojević, and Katarina Smiljanić. 2023. "Antioxidant Properties of Protein-Rich Plant Foods in Gastrointestinal Digestion—Peanuts as Our Antioxidant Friend or Foe in Allergies" Antioxidants 12, no. 4: 886. https://doi.org/10.3390/antiox12040886
APA StyleProdić, I., Krstić Ristivojević, M., & Smiljanić, K. (2023). Antioxidant Properties of Protein-Rich Plant Foods in Gastrointestinal Digestion—Peanuts as Our Antioxidant Friend or Foe in Allergies. Antioxidants, 12(4), 886. https://doi.org/10.3390/antiox12040886










