Insights on Dietary Polyphenols as Agents against Metabolic Disorders: Obesity as a Target Disease
Abstract
1. Introduction
2. Existing Treatment for Obesity and Their Safety Limitations
3. Diversity of Polyphenols in Diet and Their Biological Significance
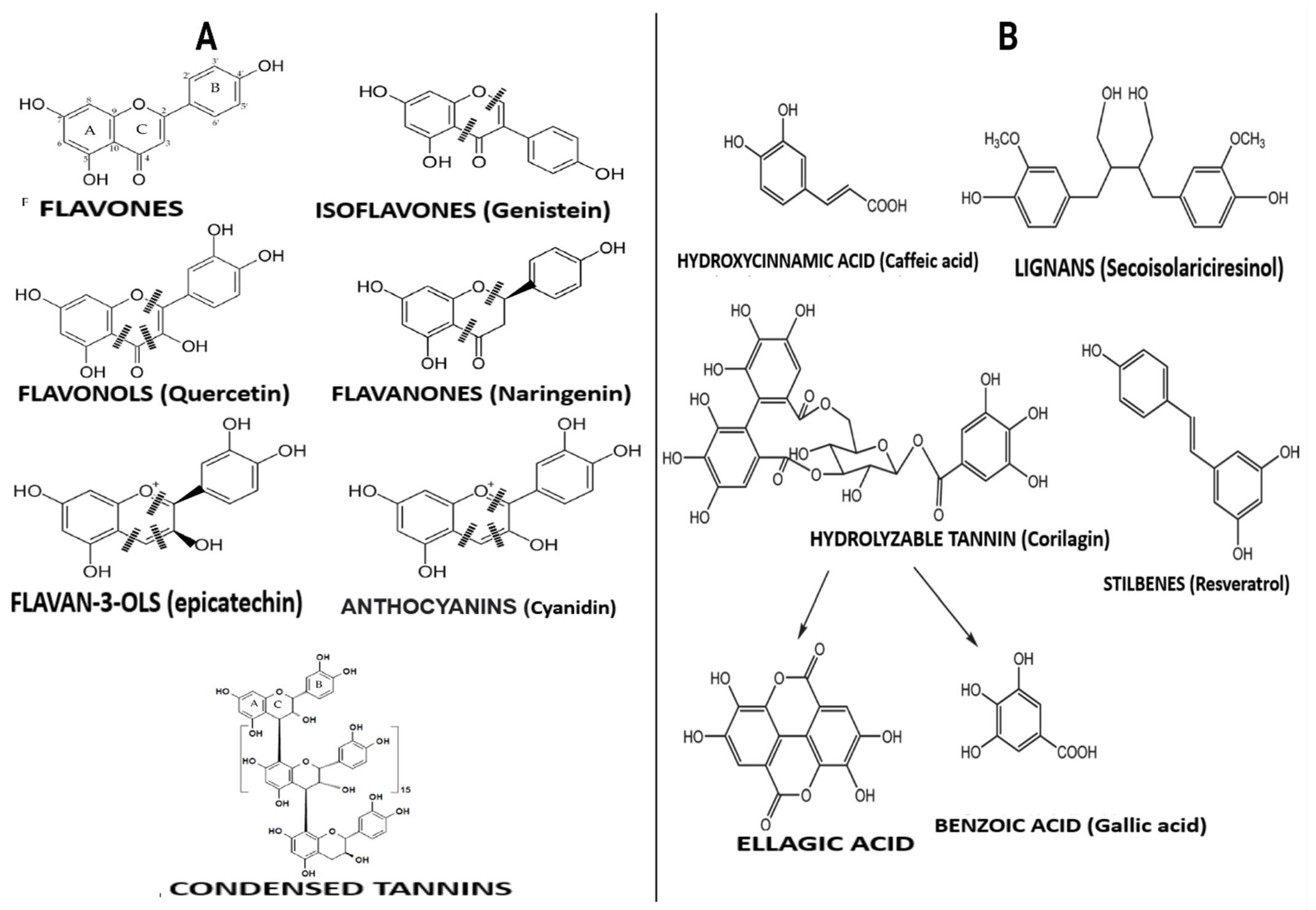
3.1. Flavonoid-Type Phenolic Compounds
3.2. Non-Flavonoid-Type Phenolic Compounds in Foods
4. Integrating Polyphenols as Bioactive Ingredients in Foods: Foods for Weight Loss
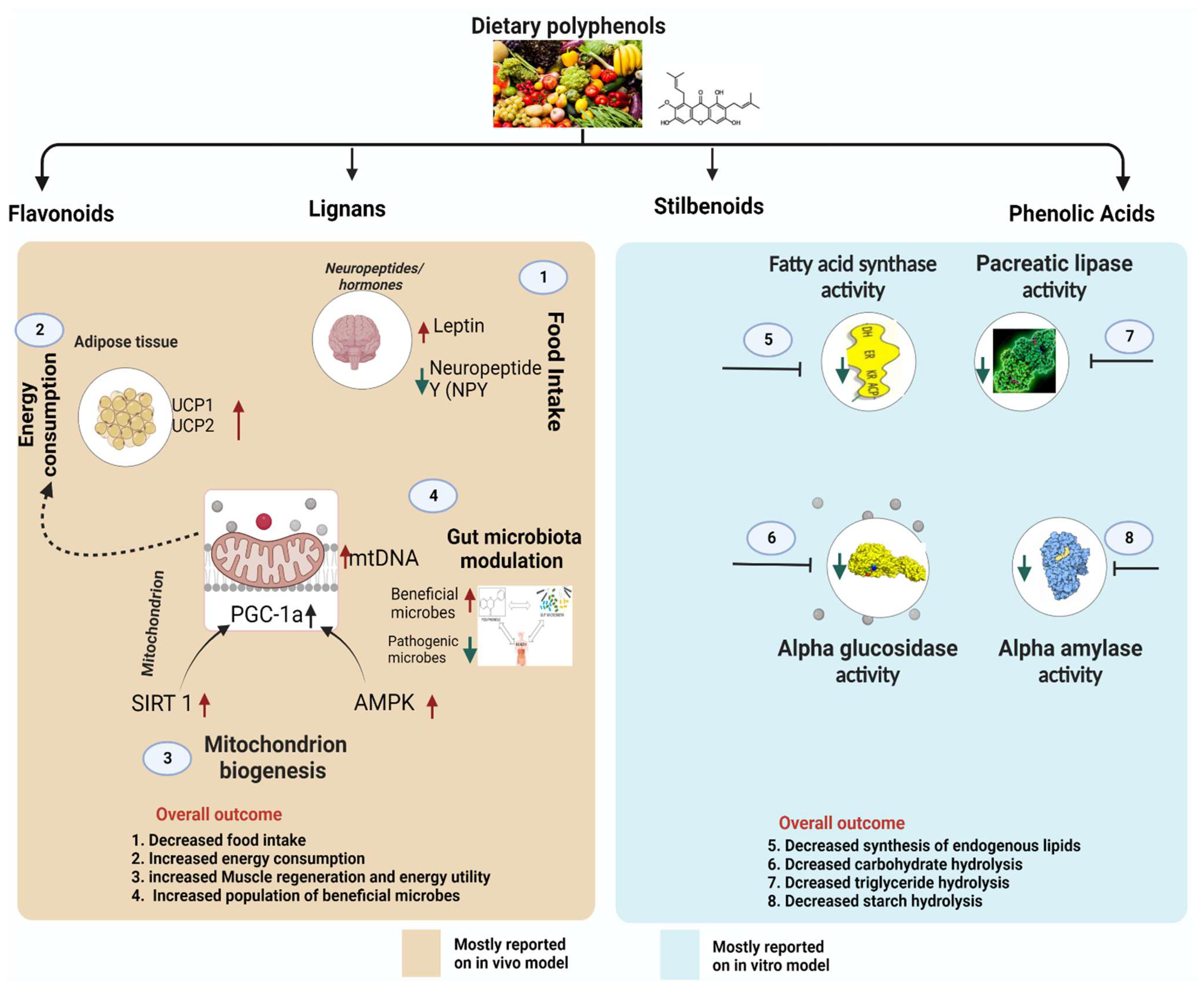
5. Molecular Target and Mode of Action for Antiobesity of Polyphenols
5.1. Polyphenols Influence Neuropeptides/Hormones Involved in Food Intake and Satiety
5.2. Polyphenols Inhibit Pro-Obesity Enzymes
5.3. Polyphenols Suppress Lipogenesis of the Adipose Tissue via Obesity-Related Transcription Factors
5.4. Polyphenols Modulate Thermogenesis and Mitochondrial Biogenesis
5.5. Gut Microbiota Modulation
6. A Comprehensive Review of Cellular, Animal, and Human Models Investigating the Potential of Polyphenol-Rich Extracts against Obesity
6.1. Studies on the Anti-Obesity Effects of Polyphenol-Rich Extracts Performed in Cell and Animal Models
6.2. Studies on the Anti-Obesity Effect of Polyphenol-Rich Plant Extracts Performed in Humans
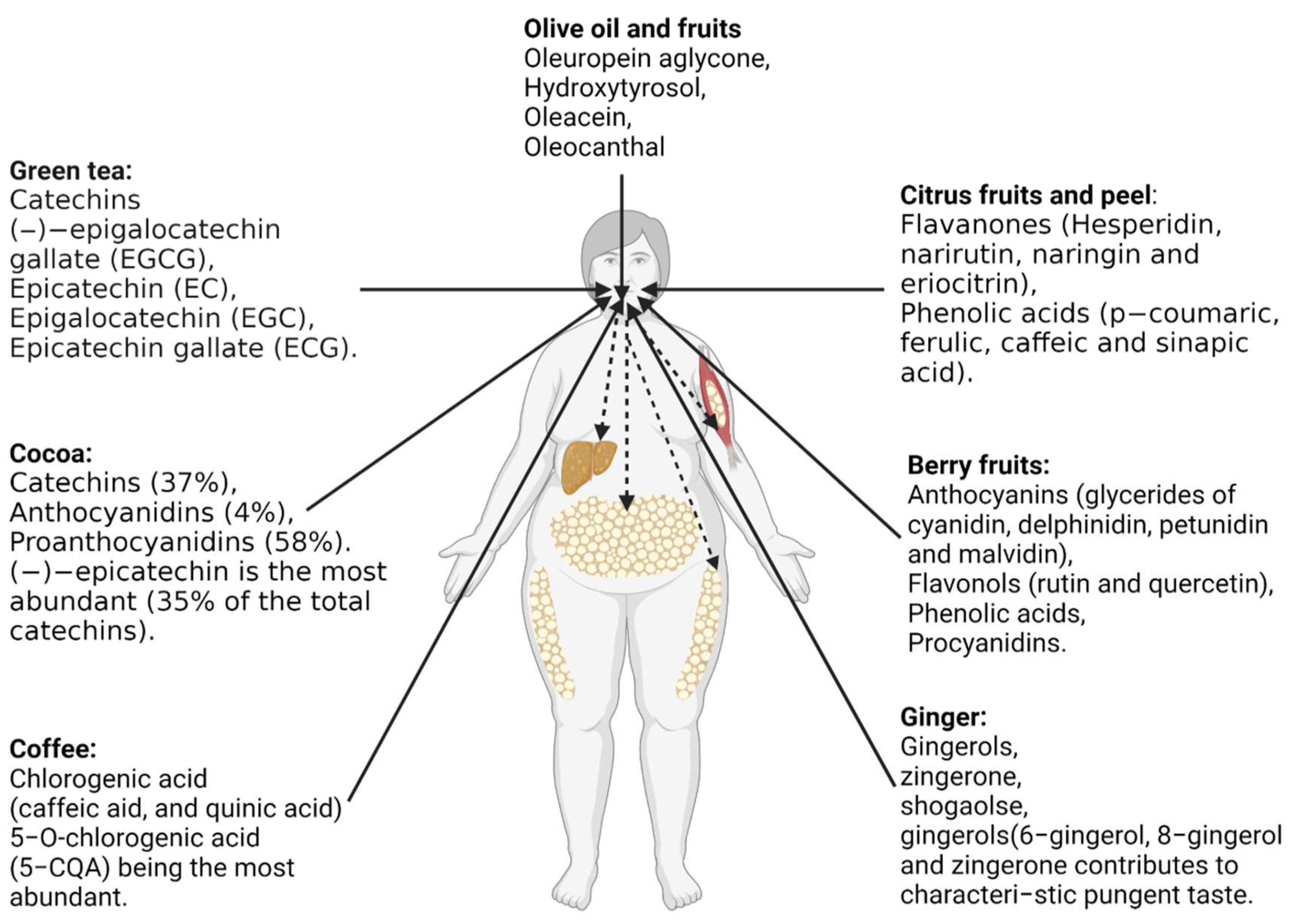
6.3. Anti-Obesity Effects of Commonly Studied Polyphenols-Rich Foods
7. Bioavailability of Polyphenols, Metabolism by Gut Microbes, Post Absorption Fate, and Eventual Effect on Obesity
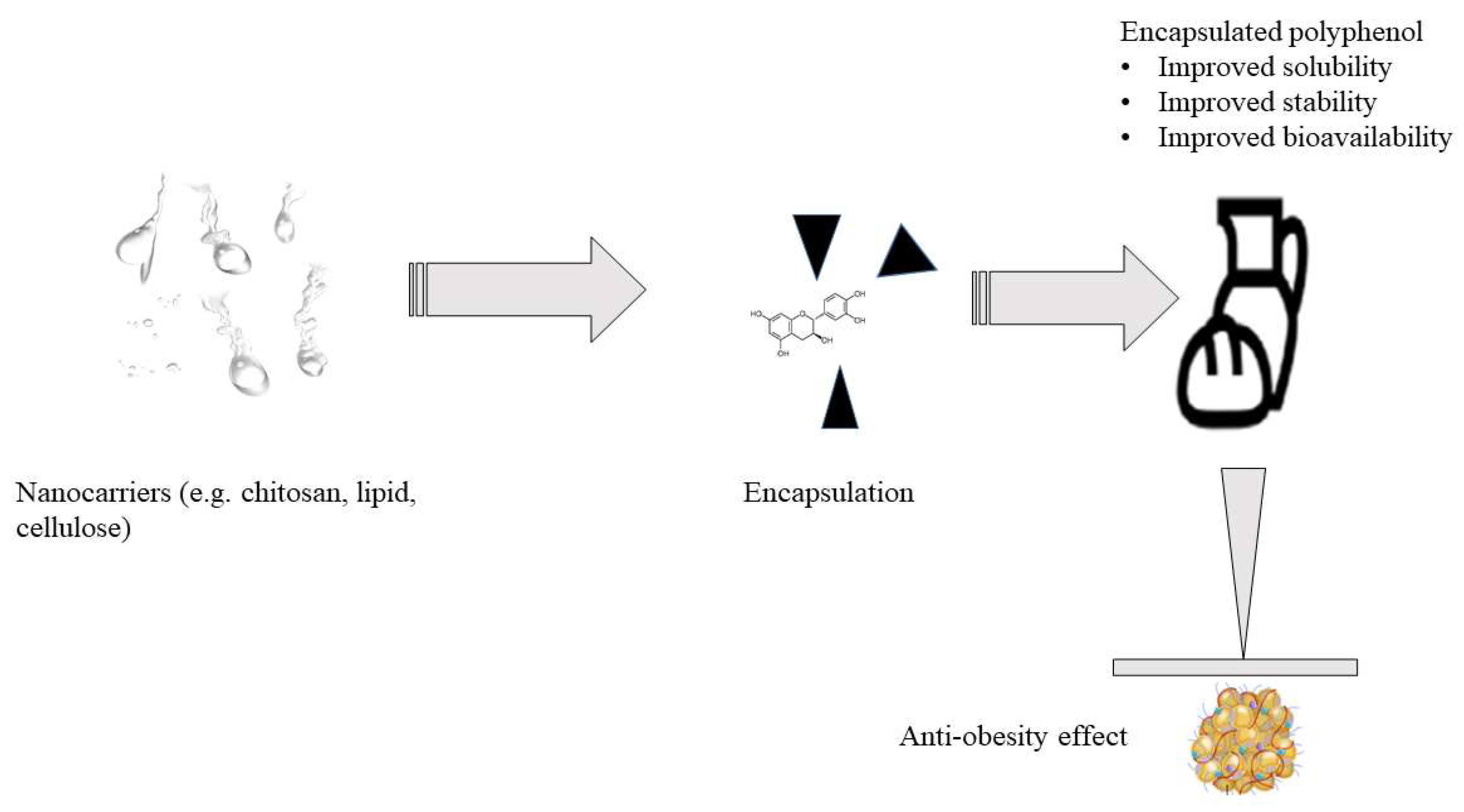
8. Improving the Bioavailability and Delivery of Polyphenols in the Body
9. Effects of Processing on Polyphenol Content of Foods and Ultimate Impact on Anti-Obesity Potential of the Final Product
10. Drawbacks of Polyphenols as Phytonutrients in Foods: Benefits versus Risks
11. Research Limitations
12. Perspectives
- In the study of plant phyto-ingredients, human and animal experiments are the most relevant in clinical nutrition but possess certain limitations. In the research on the health functions of plant metabolites, the two models have sometimes shown differences in the outcome. In most cases, the differences in the genetic composition between humans and mice are the causes of this variation [160]. Given these potential inconsistencies and to enable the public to make informed choices on polyphenol consumption, future research is needed to be more robust in clinical trials using human subjects to assess the anti-obesity effectiveness of polyphenols instead of relying on in vitro or animal findings for conclusions;
- The biological activity of polyphenols can be affected by various factors. The length of intake and mode of intake of polyphenols are key determinants of polyphenols’ biological activity [182]. However, most current studies have largely ignored the significance of the correlation between polyphenol intake and the length of intake. Furthermore, as already been mentioned, polyphenols doses administered during clinical trials are often higher than in the common diet [182]. However, only a few human studies have considered this factor in drawing a scientific substantiation for the relative claimed effects of polyphenols. In future research, it is essential to assess the relevance of these factors in the anti-obesity potential of polyphenol-rich foods to offer a verifiable claim on the health effects of polyphenols;
- Although trials have shown a correlation between polyphenol consumption and a reduction in risk factors for chronic diseases, discrepancies in explaining their positive effects have been found due to their low content in most daily diets as well as bioavailability. More studies are needed to find safe and effective methods to incorporate polyphenols in foods to improve their levels consumed in diets and to enhance the bioavailability to realize similar outcomes observed in in vitro, in vivo, and clinical trials studies. Nanocarriers are a potential technology for polyphenol encapsulation that could enhance their bioavailability, solubility and stability at their target sites in the body. In the current settings, most nanotechnology reports have addressed diseases such as cancer, but none of the studies have been reported for polyphenols targeting obesity. Future research needs to evaluate the effectiveness of encapsulated polyphenols against obesity development, both in animal and human trials, not just in vitro;
- The translation of food composition into intakes of a specific dietary compound is often achieved using food composition databases. However, for polyphenols, the approach needs to be revised since the currently available databases contain limited information regarding the diversity and concentration of phenolic compounds in plant foods. These limitations arise mainly because, unlike most nutrients, there has been a narrow systematic approach to comprehensively characterize and quantify the diverse polyphenols in plant foods using standardized analytical methods. Moreover, much of the available information has been assembled solely from heterogeneous sources, in which the original food sampling and description are still being investigated. Furthermore, the complexity arises due to the uneven distribution of polyphenolic compounds in different parts of plants and the loss of specific polyphenols during food processing. For example, in apples, quercetin is mainly found in the peel; however, peeled fruit contains no quercetin. Similarly, most polyphenols in wheat grain are located primarily in the outer layers and are usually lost during the refining process of the flour. Further research is required to expand our current knowledge regarding alternative dietary assessment methods that could help overcome these challenges;
- Finally, to establish firm evidence for the health effects of dietary polyphenol consumption, it is essential to have quantitative information regarding their dietary intake on food labels. This is particularly essential for enriched foods or foods that are known to contain high amounts of these compounds. The usefulness of such information is that consumers can have a planned dietary intake based on specific foods. Thus, similar to other known nutrients and drugs, in the future, food records and labels for polyphenols need to be developed to allow consumers to quickly assess total intake and required precautionary measures accurately.
13. Concluding Remarks
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Sergent, T.; Vanderstraeten, J.; Winand, J.; Beguin, P.; Schneider, Y.-J. Phenolic compounds and plant extracts as potential natural anti-obesity substances. Food Chem. 2012, 135, 68–73. [Google Scholar] [CrossRef]
- Boccellino, M.; D’Angelo, S. Anti-obesity effects of polyphenol intake: Current status and future possibilities. Int. J. Mol. Sci. 2020, 21, 5642. [Google Scholar] [CrossRef]
- WHO. Adolescent Obesity and Related Behaviours: Trends and Inequalities in the WHO European Region, 2002–2014; WHO: Geneva, Switzerland, 2017.
- Singh, M.; Thrimawithana, T.; Shukla, R.; Adhikari, B. Managing obesity through natural polyphenols: A review. Future Foods 2020, 1, 100002. [Google Scholar] [CrossRef]
- Dietrich, M.O.; Horvath, T.L. Limitations in anti-obesity drug development: The critical role of hunger-promoting neurons. Nat. Rev. Drug Discov. 2012, 11, 675–691. [Google Scholar]
- Mayer, M.A.; Hocht, C.; Puyó, A.; Taira, C.A. Recent advances in obesity pharmacotherapy. Curr. Clin. Pharmacol. 2009, 4, 53–61. [Google Scholar] [CrossRef]
- Jepsen, M.M.; Christensen, M.B. Emerging glucagon-like peptide 1 receptor agonists for the treatment of obesity. Expert Opin. Emerg. Drugs 2021, 26, 231–243. [Google Scholar]
- Cercato, C.; Roizenblatt, V.; Leança, C.; Segal, A.; Lopes Filho, A.; Mancini, M.; Halpern, A. A randomized double-blind placebo-controlled study of the long-term efficacy and safety of diethylpropion in the treatment of obese subjects. Int. J. Obes. 2009, 33, 857–865. [Google Scholar]
- Wigal, T.L.; Newcorn, J.H.; Handal, N.; Wigal, S.B.; Mulligan, I.; Schmith, V.; Konofal, E. A double-blind, placebo-controlled, phase II study to determine the efficacy, safety, tolerability and pharmacokinetics of a controlled release (CR) formulation of mazindol in adults with DSM-5 attention-deficit/hyperactivity disorder (ADHD). CNS Drugs 2018, 32, 289–301. [Google Scholar] [CrossRef]
- Stoops, W.W.; Strickland, J.C.; Hays, L.R.; Rayapati, A.O.; Lile, J.A.; Rush, C.R. Safety and tolerability of intranasal cocaine during phendimetrazine maintenance. Psychopharmacology 2016, 233, 2055–2063. [Google Scholar]
- Kang, J.; Park, C.Y.; Kang, J.; Park, Y.W.; Park, S. Randomized controlled trial to investigate the effects of a newly developed formulation of phentermine diffuse-controlled release for obesity. Diabetes Obes. Metab. 2010, 12, 876–882. [Google Scholar]
- Shirai, K.; Fujita, T.; Tanaka, M.; Fujii, Y.; Shimomasuda, M.; Sakai, S.; Samukawa, Y. Efficacy and safety of lipase inhibitor orlistat in Japanese with excessive visceral fat accumulation: 24-week, double-blind, randomized, placebo-controlled study. Adv. Ther. 2019, 36, 86–100. [Google Scholar] [CrossRef]
- Toplak, H.; Hamann, A.; Moore, R.; Masson, E.; Gorska, M.; Vercruysse, F.; Sun, X.; Fitchet, M. Efficacy and safety of topiramate in combination with metformin in the treatment of obese subjects with type 2 diabetes: A randomized, double-blind, placebo-controlled study. Int. J. Obes. 2007, 31, 138–146. [Google Scholar] [CrossRef]
- O’neil, P.M.; Smith, S.R.; Weissman, N.J.; Fidler, M.C.; Sanchez, M.; Zhang, J.; Raether, B.; Anderson, C.M.; Shanahan, W.R. Randomized placebo-controlled clinical trial of lorcaserin for weight loss in type 2 diabetes mellitus: The BLOOM-DM study. Obesity 2012, 20, 1426–1436. [Google Scholar] [CrossRef]
- Sramek, J.; Leibowitz, M.; Weinstein, S.; Rowe, E.; Mendel, C.; Levy, B.; McMahon, F.; Mullican, W.; Toth, P.; Cutler, N. Efficacy and safety of sibutramine for weight loss in obese patients with hypertension well controlled by β-adrenergic blocking agents: A placebo-controlled, double-blind, randomised trial. J. Hum. Hypertens. 2002, 16, 13–19. [Google Scholar]
- Greenway, F.L.; Fujioka, K.; Plodkowski, R.A.; Mudaliar, S.; Guttadauria, M.; Erickson, J.; Kim, D.D.; Dunayevich, E.; Group, C.-I.S. Effect of naltrexone plus bupropion on weight loss in overweight and obese adults (COR-I): A multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2010, 376, 595–605. [Google Scholar] [CrossRef]
- Ji, L.; Onishi, Y.; Ahn, C.W.; Agarwal, P.; Chou, C.W.; Haber, H.; Guerrettaz, K.; Boardman, M.K. Efficacy and safety of exenatide once-weekly vs exenatide twice-daily in A sian patients with type 2 diabetes mellitus. J. Diabetes Investig. 2013, 4, 53–61. [Google Scholar]
- Pinget, M.; Goldenberg, R.; Niemoeller, E.; Muehlen-Bartmer, I.; Guo, H.; Aronson, R. Efficacy and safety of lixisenatide once daily versus placebo in type 2 diabetes insufficiently controlled on pioglitazone (GetGoal-P). Diabetes Obes. Metab. 2013, 15, 1000–1007. [Google Scholar] [CrossRef]
- O’Neil, P.M.; Birkenfeld, A.L.; McGowan, B.; Mosenzon, O.; Pedersen, S.D.; Wharton, S.; Carson, C.G.; Jepsen, C.H.; Kabisch, M.; Wilding, J.P. Efficacy and safety of semaglutide compared with liraglutide and placebo for weight loss in patients with obesity: A randomised, double-blind, placebo and active controlled, dose-ranging, phase 2 trial. Lancet 2018, 392, 637–649. [Google Scholar]
- Reusch, J.; Stewart, M.; Perkins, C.; Cirkel, D.; Ye, J.; Perry, C.; Reinhardt, R.; Bode, B. Efficacy and safety of once-weekly glucagon-like peptide 1 receptor agonist albiglutide (HARMONY 1 trial): 52-week primary endpoint results from a randomized, double-blind, placebo-controlled trial in patients with type 2 diabetes mellitus not controlled on pioglitazone, with or without metformin. Diabetes Obes. Metab. 2014, 16, 1257–1264. [Google Scholar]
- Umpierrez, G.; Povedano, S.T.; Manghi, F.P.; Shurzinske, L.; Pechtner, V. Efficacy and safety of dulaglutide monotherapy versus metformin in type 2 diabetes in a randomized controlled trial (AWARD-3). Diabetes Care 2014, 37, 2168–2176. [Google Scholar]
- Ozdal, T.; Sela, D.A.; Xiao, J.; Boyacioglu, D.; Chen, F.; Capanoglu, E. The reciprocal interactions between polyphenols and gut microbiota and effects on bioaccessibility. Nutrients 2016, 8, 78. [Google Scholar] [CrossRef]
- Akhlaghi, M.; Ghobadi, S.; Hosseini, M.M.; Gholami, Z.; Mohammadian, F. Flavanols are potential anti-obesity agents, a systematic review and meta-analysis of controlled clinical trials. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 675–690. [Google Scholar] [CrossRef]
- Wu, S.; Tian, L. A new flavone glucoside together with known ellagitannins and flavones with anti-diabetic and anti-obesity activities from the flowers of pomegranate (Punica granatum). Nat. Prod. Res. 2019, 33, 252–257. [Google Scholar] [CrossRef]
- Corrêa, T.A.F.; Rogero, M.M.; Hassimotto, N.M.A.; Lajolo, F.M. The two-way polyphenols-microbiota interactions and their effects on obesity and related metabolic diseases. Front. Nutr. 2019, 6, 188. [Google Scholar] [CrossRef]
- Otton, R.; Bolin, A.P.; Ferreira, L.T.; Marinovic, M.P.; Rocha, A.L.S.; Mori, M.A. Polyphenol-rich green tea extract improves adipose tissue metabolism by down-regulating miR-335 expression and mitigating insulin resistance and inflammation. J. Nutr. Biochem. 2018, 57, 170–179. [Google Scholar] [CrossRef]
- Yamakoshi, J.; Saito, M.; Kataoka, S.; Kikuchi, M. Safety evaluation of proanthocyanidin-rich extract from grape seeds. Food Chem. Toxicol. 2002, 40, 599–607. [Google Scholar] [CrossRef]
- Lee, H.; Jeong, J.H.; Ryu, J.-H. Lignan from Alnus japonica inhibits adipocyte differentiation via cell cycle and FOXO1 regulation. Molecules 2020, 25, 3346. [Google Scholar] [CrossRef]
- Mir, S.A.; Shah, M.A.; Ganai, S.A.; Ahmad, T.; Gani, M. Understanding the role of active components from plant sources in obesity management. J. Saudi Soc. Agric. Sci. 2019, 18, 168–176. [Google Scholar] [CrossRef]
- Adaim, A.; Zhang, J.; Lauren, D.; Stanley, R.; Skinner, M. Polyphenols and health: Using cell-based assays to aid in the development of new functional foods. Asia Pac. J. Clin. Nutr. 2005, 14, S106. [Google Scholar]
- Ravi, A.; Perera, C.; Quek, S.; Sun-Waterhouse, D. Physico-chemical properties of bread dough fortified with fruit fibers and phenolic antioxidants. In Proceedings of the Oral Presentation at the 2009 Institute of Food Technologists (IFT) Annual Meeting & Food Expo, Anaheim, CA, USA, 6–9 June 2009. [Google Scholar]
- Bora, A.F.M.; Ma, S.; Li, X.; Liu, L. Application of microencapsulation for the safe delivery of green tea polyphenols in food systems: Review and recent advances. Food Res. Int. 2018, 105, 241–249. [Google Scholar]
- Sun-Waterhouse, D. The development of fruit-based functional foods targeting the health and wellness market: A review. Int. J. Food Sci. Technol. 2011, 46, 899–920. [Google Scholar] [CrossRef]
- Kiss, A.; Takács, K.; Nagy, A.; Nagy-Gasztonyi, M.; Cserhalmi, Z.; Naár, Z.; Halasi, T.; Csáki, J.; Némedi, E. In vivo and in vitro model studies on noodles prepared with antioxidant-rich pseudocereals. J. Food Meas. Charact. 2019, 13, 2696–2704. [Google Scholar]
- Mayneris-Perxachs, J.; Alcaide-Hidalgo, J.M.; de la Hera, E.; del Bas, J.M.; Arola, L.; Caimari, A. Supplementation with biscuits enriched with hesperidin and naringenin is associated with an improvement of the Metabolic Syndrome induced by a cafeteria diet in rats. J. Funct. Foods 2019, 61, 103504. [Google Scholar] [CrossRef]
- Coe, S.; Ryan, L. White bread enriched with polyphenol extracts shows no effect on glycemic response or satiety, yet may increase postprandial insulin economy in healthy participants. Nutr. Res. 2016, 36, 193–200. [Google Scholar] [CrossRef]
- Serrano, A.; González-Sarrías, A.; Tomás-Barberán, F.A.; Avellaneda, A.; Gironés-Vilaplana, A.; Nieto, G.; Ros-Berruezo, G. Anti-inflammatory and antioxidant effects of regular consumption of cooked ham enriched with dietary phenolics in diet-induced obese mice. Antioxidants 2020, 9, 639. [Google Scholar]
- Colantuono, A.; Ferracane, R.; Vitaglione, P. In vitro bioaccessibility and functional properties of polyphenols from pomegranate peels and pomegranate peels-enriched cookies. Food Funct. 2016, 7, 4247–4258. [Google Scholar] [CrossRef]
- Bajerska, J.; Mildner-Szkudlarz, S.; Walkowiak, J. Effects of rye bread enriched with green tea extract on weight maintenance and the characteristics of metabolic syndrome following weight loss: A pilot study. J. Med. Food 2015, 18, 698–705. [Google Scholar]
- Bajerska, J.; Mildner-Szkudlarz, S.; Pruszynska-Oszmalek, E. May rye bread enriched with green tea extract be useful in the prevention of obesity in rats? Acta Aliment. 2013, 42, 69–78. [Google Scholar]
- Macho-González, A.; López-Oliva, M.E.; Merino, J.J.; García-Fernández, R.A.; Garcimartín, A.; Redondo-Castillejo, R.; Bastida, S.; Sánchez-Muniz, F.J.; Benedí, J. Carob fruit extract-enriched meat improves pancreatic beta-cell dysfunction, hepatic insulin signaling and lipogenesis in late-stage type 2 diabetes mellitus model. J. Nutr. Biochem. 2020, 84, 108461. [Google Scholar]
- Hsu, T.; Kusumoto, A.; Abe, K.; Hosoda, K.; Kiso, Y.; Wang, M.; Yamamoto, S. Polyphenol-enriched oolong tea increases fecal lipid excretion. Eur. J. Clin. Nutr. 2006, 60, 1330–1336. [Google Scholar] [CrossRef]
- Isganaitis, E.; Lustig, R.H. Fast food, central nervous system insulin resistance, and obesity. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 2451–2462. [Google Scholar]
- Sigal, R.J.; El-Hashimy, M.; Martin, B.C.; Soeldner, J.S.; Krolewski, A.S.; Warram, J.H. Acute postchallenge hyperinsulinemia predicts weight gain: A prospective study. Diabetes 1997, 46, 1025–1029. [Google Scholar] [CrossRef]
- Mayer, J. Regulation of energy intake and the body weight: The glucostatic theory and the lipostatic hypothesis. Ann. N. Y. Acad. Sci. 1955, 63, 15–43. [Google Scholar] [CrossRef]
- Lim, K.J.; Bisht, S.; Bar, E.E.; Maitra, A.; Eberhart, C.G. A polymeric nanoparticle formulation of curcumin inhibits growth, clonogenicity and stem-like fraction in malignant brain tumors. Cancer Biol. Ther. 2011, 11, 464–473. [Google Scholar]
- Xiang, L.; Wu, Q.; Cheng, L.; Sun, K.; Li, J.; Yoshida, M.; Qi, J. Leptin and adiponectin signaling pathways are involved in the antiobesity effects of peanut skin extract. Oxidative Med. Cell. Longev. 2019, 2019, 2935315. [Google Scholar]
- Ardid-Ruiz, A.; Ibars, M.; Mena, P.; Del Rio, D.; Muguerza, B.; Bladé, C.; Arola, L.; Aragonès, G.; Suárez, M. Potential involvement of peripheral leptin/STAT3 signaling in the effects of resveratrol and its metabolites on reducing body fat accumulation. Nutrients 2018, 10, 1757. [Google Scholar]
- Badshah, H.; Ullah, I.; Kim, S.E.; Kim, T.-h.; Lee, H.Y.; Kim, M.O. Anthocyanins attenuate body weight gain via modulating neuropeptide Y and GABAB1 receptor in rats hypothalamus. Neuropeptides 2013, 47, 347–353. [Google Scholar]
- Buchholz, T.; Melzig, M.F. Polyphenolic compounds as pancreatic lipase inhibitors. Planta Med. 2015, 81, 771–783. [Google Scholar]
- McDougall, G.J.; Kulkarni, N.N.; Stewart, D. Berry polyphenols inhibit pancreatic lipase activity in vitro. Food Chem. 2009, 115, 193–199. [Google Scholar]
- Griffiths, D.W. The inhibition of digestive enzymes by polyphenolic compounds. In Nutritional and Toxicological Significance of Enzyme Inhibitors in Foods; Springer: Berlin/Heidelberg, Germany, 1986; pp. 509–516. [Google Scholar]
- Othman, Z.A.; Wan Ghazali, W.S.; Noordin, L.; Mohamed, M. Phenolic compounds and the anti-atherogenic effect of bee bread in high-fat diet-induced obese rats. Antioxidants 2020, 9, 33. [Google Scholar] [CrossRef]
- Brusselmans, K.; De Schrijver, E.; Heyns, W.; Verhoeven, G.; Swinnen, J.V. Epigallocatechin-3-gallate is a potent natural inhibitor of fatty acid synthase in intact cells and selectively induces apoptosis in prostate cancer cells. Int. J. Cancer 2003, 106, 856–862. [Google Scholar]
- Kim, N.-H.; Jegal, J.; Kim, Y.N.; Heo, J.-D.; Rho, J.-R.; Yang, M.H.; Jeong, E.J. Chokeberry extract and its active polyphenols suppress adipogenesis in 3T3-L1 adipocytes and modulates fat accumulation and insulin resistance in diet-induced obese mice. Nutrients 2018, 10, 1734. [Google Scholar] [CrossRef]
- Xie, H.; Sun, L.; Lodish, H.F. Targeting microRNAs in obesity. Expert Opin. Ther. Targets 2009, 13, 1227–1238. [Google Scholar] [CrossRef]
- Rocha, A.; Bolin, A.P.; Cardoso, C.A.L.; Otton, R. Green tea extract activates AMPK and ameliorates white adipose tissue metabolic dysfunction induced by obesity. Eur. J. Nutr. 2016, 55, 2231–2244. [Google Scholar] [CrossRef]
- Pollard, A.E.; Martins, L.; Muckett, P.J.; Khadayate, S.; Bornot, A.; Clausen, M.; Admyre, T.; Bjursell, M.; Fiadeiro, R.; Wilson, L. AMPK activation protects against diet-induced obesity through Ucp1-independent thermogenesis in subcutaneous white adipose tissue. Nat. Metab. 2019, 1, 340–349. [Google Scholar] [CrossRef]
- Fang, C.; Kim, H.; Noratto, G.; Sun, Y.; Talcott, S.T.; Mertens-Talcott, S.U. Gallotannin derivatives from mango (Mangifera indica L.) suppress adipogenesis and increase thermogenesis in 3T3-L1 adipocytes in part through the AMPK pathway. J. Funct. Foods 2018, 46, 101–109. [Google Scholar] [CrossRef]
- dos Santos, T.W.; Cristina Pereira, Q.; Teixeira, L.; Gambero, A.; Villena, J.A.; Lima Ribeiro, M. Effects of polyphenols on thermogenesis and mitochondrial biogenesis. Int. J. Mol. Sci. 2018, 19, 2757. [Google Scholar]
- de Mello, A.H.; Costa, A.B.; Engel, J.D.G.; Rezin, G.T. Mitochondrial dysfunction in obesity. Life Sci. 2018, 192, 26–32. [Google Scholar]
- Fernandez-Marcos, P.J.; Auwerx, J. Regulation of PGC-1α, a nodal regulator of mitochondrial biogenesis. Am. J. Clin. Nutr. 2011, 93, 884S–890S. [Google Scholar] [CrossRef]
- Lagouge, M.; Argmann, C.; Gerhart-Hines, Z.; Meziane, H.; Lerin, C.; Daussin, F.; Messadeq, N.; Milne, J.; Lambert, P.; Elliott, P. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1α. Cell 2006, 127, 1109–1122. [Google Scholar]
- Yoneshiro, T.; Aita, S.; Matsushita, M.; Kameya, T.; Nakada, K.; Kawai, Y.; Saito, M. Brown adipose tissue, whole-body energy expenditure, and thermogenesis in healthy adult men. Obesity 2011, 19, 13–16. [Google Scholar]
- Zhang, Z.; Di Yang, J.X.; Zhou, J.; Cao, H.; Che, Q.; Bai, Y.; Guo, J.; Su, Z. Non-shivering Thermogenesis Signalling Regulation and Potential Therapeutic Applications of Brown Adipose Tissue. Int. J. Biol. Sci. 2021, 17, 2853. [Google Scholar]
- Han, X.; Guo, J.; You, Y.; Yin, M.; Liang, J.; Ren, C.; Zhan, J.; Huang, W. Vanillic acid activates thermogenesis in brown and white adipose tissue. Food Funct. 2018, 9, 4366–4375. [Google Scholar]
- Lee, M.-S.; Shin, Y.; Jung, S.; Kim, Y. Effects of epigallocatechin-3-gallate on thermogenesis and mitochondrial biogenesis in brown adipose tissues of diet-induced obese mice. Food Nutr. Res. 2017, 61, 1325307. [Google Scholar]
- Carrera-Quintanar, L.; Lopez Roa, R.I.; Quintero-Fabián, S.; Sánchez-Sánchez, M.A.; Vizmanos, B.; Ortuño-Sahagún, D. Phytochemicals that influence gut microbiota as prophylactics and for the treatment of obesity and inflammatory diseases. Mediat. Inflamm. 2018, 2018, 9734845. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, M.; Ho, C.-T.; Guo, X.; Wu, Z.; Weng, P.; Yan, M.; Cao, J. Metagenomics analysis of gut microbiota modulatory effect of green tea polyphenols by high fat diet-induced obesity mice model. J. Funct. Foods 2018, 46, 268–277. [Google Scholar]
- Yen, G.-C.; Cheng, H.-L.; Lin, L.-Y.; Chen, S.-C.; Hsu, C.-L. The potential role of phenolic compounds on modulating gut microbiota in obesity. J. Food Drug Anal. 2020, 28, 195. [Google Scholar]
- Liu, Z.; Chen, Z.; Guo, H.; He, D.; Zhao, H.; Wang, Z.; Zhang, W.; Liao, L.; Zhang, C.; Ni, L. The modulatory effect of infusions of green tea, oolong tea, and black tea on gut microbiota in high-fat-induced obese mice. Food Funct. 2016, 7, 4869–4879. [Google Scholar] [CrossRef]
- Anhê, F.F.; Roy, D.; Pilon, G.; Dudonné, S.; Matamoros, S.; Varin, T.V.; Garofalo, C.; Moine, Q.; Desjardins, Y.; Levy, E. A polyphenol-rich cranberry extract protects from diet-induced obesity, insulin resistance and intestinal inflammation in association with increased Akkermansia spp. population in the gut microbiota of mice. Gut 2015, 64, 872–883. [Google Scholar] [CrossRef]
- Soeng, S.; Evacuasiany, E.; Widowati, W.; Fauziah, N.; Manik, V.T.; Maesaroh, M. Inhibitory potential of rambutan seeds extract and fractions on adipogenesis in 3T3-L1 cell line. J. Exp. Integr. Med. 2015, 5, 55–60. [Google Scholar] [CrossRef]
- Kim, H.-K.; Kim, J.N.; Han, S.N.; Nam, J.-H.; Na, H.-N.; Ha, T.J. Black soybean anthocyanins inhibit adipocyte differentiation in 3T3-L1 cells. Nutr. Res. 2012, 32, 770–777. [Google Scholar] [CrossRef]
- Pinent, M.; Blade, M.C.; Salvado, M.J.; Arola, L.; Hackl, H.; Quackenbush, J.; Trajanoski, Z.; Ardevol, A. Grape-seed derived procyanidins interfere with adipogenesis of 3T3-L1 cells at the onset of differentiation. Int. J. Obes. 2005, 29, 934–941. [Google Scholar]
- Jiao, X.; Wang, Y.; Lin, Y.; Lang, Y.; Li, E.; Zhang, X.; Zhang, Q.; Feng, Y.; Meng, X.; Li, B. Blueberry polyphenols extract as a potential prebiotic with anti-obesity effects on C57BL/6 J mice by modulating the gut microbiota. J. Nutr. Biochem. 2019, 64, 88–100. [Google Scholar] [CrossRef]
- Zhou, F.; Guo, J.; Han, X.; Gao, Y.; Chen, Q.; Huang, W.; Zhan, J.; Huang, D.; You, Y. Cranberry polyphenolic extract exhibits an antiobesity effect on high-fat diet–fed mice through increased thermogenesis. J. Nutr. 2020, 150, 2131–2138. [Google Scholar] [CrossRef]
- van der Zande, H.J.; Lambooij, J.M.; Chavanelle, V.; Zawistowska-Deniziak, A.; Otero, Y.; Otto, F.; Lantier, L.; McGuinness, O.P.; Le Joubioux, F.; Giera, M. Effects of a novel polyphenol-rich plant extract on body composition, inflammation, insulin sensitivity, and glucose homeostasis in obese mice. Int. J. Obes. 2021, 45, 2016–2027. [Google Scholar] [CrossRef]
- Marimoutou, M.; Le Sage, F.; Smadja, J.; Lefebvre d’Hellencourt, C.; Gonthier, M.-P.; Silva, R.-D. Antioxidant polyphenol-rich extracts from the medicinal plants Antirhea borbonica, Doratoxylon apetalum and Gouania mauritiana protect 3T3-L1 preadipocytes against H2O2, TNFα and LPS inflammatory mediators by regulating the expression of superoxide dismutase and NF-κB genes. J. Inflamm. 2015, 12, 1–15. [Google Scholar]
- Wu, T.; Jiang, Z.; Yin, J.; Long, H.; Zheng, X. Anti-obesity effects of artificial planting blueberry (Vaccinium ashei) anthocyanin in high-fat diet-treated mice. Int. J. Food Sci. Nutr. 2016, 67, 257–264. [Google Scholar]
- Kaume, L.; Gilbert, W.C.; Brownmiller, C.; Howard, L.R.; Devareddy, L. Cyanidin 3-O-β-D-glucoside-rich blackberries modulate hepatic gene expression, and anti-obesity effects in ovariectomized rats. J. Funct. Foods 2012, 4, 480–488. [Google Scholar]
- Ikarashi, N.; Toda, T.; Okaniwa, T.; Ito, K.; Ochiai, W.; Sugiyama, K. Anti-obesity and anti-diabetic effects of acacia polyphenol in obese diabetic KKAy mice fed high-fat diet. Evid.-Based Complement. Altern. Med. 2011, 2011, 952031. [Google Scholar] [CrossRef]
- Dallas, C.; Gerbi, A.; Elbez, Y.; Caillard, P.; Zamaria, N.; Cloarec, M. Clinical study to assess the efficacy and safety of a citrus polyphenolic extract of red Orange, grapefruit, and Orange (Sinetrol-XPur) on weight management and metabolic parameters in healthy overweight individuals. Phytother. Res. 2014, 28, 212–218. [Google Scholar] [CrossRef]
- Hsu, C.-H.; Tsai, T.-H.; Kao, Y.-H.; Hwang, K.-C.; Tseng, T.-Y.; Chou, P. Effect of green tea extract on obese women: A randomized, double-blind, placebo-controlled clinical trial. Clin. Nutr. 2008, 27, 363–370. [Google Scholar] [CrossRef]
- Golomb, B.A.; Koperski, S.; White, H.L. Association between more frequent chocolate consumption and lower body mass index. Arch. Intern. Med. 2012, 172, 519–521. [Google Scholar]
- Kim, S.-Y.; Oh, M.-R.; Kim, M.-G.; Chae, H.-J.; Chae, S.-W. Anti-obesity effects of Yerba Mate (Ilex Paraguariensis): A randomized, double-blind, placebo-controlled clinical trial. BMC Complement. Altern. Med. 2015, 15, 1–8. [Google Scholar]
- Choquette, S.; Riesco, É.; Cormier, É.; Dion, T.; Aubertin-Leheudre, M.; Dionne, I.J. Effects of soya isoflavones and exercise on body composition and clinical risk factors of cardiovascular diseases in overweight postmenopausal women: A 6-month double-blind controlled trial. Br. J. Nutr. 2011, 105, 1199–1209. [Google Scholar] [CrossRef]
- Guo, X.; Tresserra-Rimbau, A.; Estruch, R.; Martínez-González, M.A.; Medina-Remón, A.; Fitó, M.; Corella, D.; Salas-Salvadó, J.; Portillo, M.P.; Moreno, J.J. Polyphenol levels are inversely correlated with body weight and obesity in an elderly population after 5 years of follow up (the randomised PREDIMED study). Nutrients 2017, 9, 452. [Google Scholar]
- Basu, A.; Sanchez, K.; Leyva, M.J.; Wu, M.; Betts, N.M.; Aston, C.E.; Lyons, T.J. Green tea supplementation affects body weight, lipids, and lipid peroxidation in obese subjects with metabolic syndrome. J. Am. Coll. Nutr. 2010, 29, 31–40. [Google Scholar] [CrossRef]
- Bell, Z.W.; Canale, R.E.; Bloomer, R.J. A dual investigation of the effect of dietary supplementation with licorice flavonoid oil on anthropometric and biochemical markers of health and adiposity. Lipids Health Dis. 2011, 10, 1–10. [Google Scholar] [CrossRef]
- Kim, J.; Park, J.; Lim, K. Nutrition supplements to stimulate lipolysis: A review in relation to endurance exercise capacity. J. Nutr. Sci. Vitaminol. 2016, 62, 141–161. [Google Scholar] [CrossRef]
- Rains, T.M.; Agarwal, S.; Maki, K.C. Antiobesity effects of green tea catechins: A mechanistic review. J. Nutr. Biochem. 2011, 22, 1–7. [Google Scholar] [CrossRef]
- Klaus, S.; Pültz, S.; Thöne-Reineke, C.; Wolfram, S. Epigallocatechin gallate attenuates diet-induced obesity in mice by decreasing energy absorption and increasing fat oxidation. Int. J. Obes. 2005, 29, 615–623. [Google Scholar]
- Murase, T.; Nagasawa, A.; Suzuki, J.; Hase, T.; Tokimitsu, I. Beneficial effects of tea catechins on diet-induced obesity: Stimulation of lipid catabolism in the liver. Int. J. Obes. 2002, 26, 1459–1464. [Google Scholar] [CrossRef]
- Nabi, B.N.; Sedighinejad, A.; Haghighi, M.; Farzi, F.; Rimaz, S.; Atrkarroushan, Z.; Biazar, G. The Anti-Obesity Effects of Green Tea: A Controlled, Randomized, Clinical Trial. Iran. Red Crescent Med. J. 2018, 20. [Google Scholar]
- Auvichayapat, P.; Prapochanung, M.; Tunkamnerdthai, O.; Sripanidkulchai, B.-O.; Auvichayapat, N.; Thinkhamrop, B.; Kunhasura, S.; Wongpratoom, S.; Sinawat, S.; Hongprapas, P. Effectiveness of green tea on weight reduction in obese Thais: A randomized, controlled trial. Physiol. Behav. 2008, 93, 486–491. [Google Scholar]
- Jiang, H.; Zhang, W.; Li, X.; Xu, Y.; Cao, J.; Jiang, W. The anti-obesogenic effects of dietary berry fruits: A review. Food Res. Int. 2021, 147, 110539. [Google Scholar]
- Stull, A.J.; Cash, K.C.; Johnson, W.D.; Champagne, C.M.; Cefalu, W.T. Bioactives in blueberries improve insulin sensitivity in obese, insulin-resistant men and women. J. Nutr. 2010, 140, 1764–1768. [Google Scholar]
- Paquette, M.; Larqué, A.S.M.; Weisnagel, S.; Desjardins, Y.; Marois, J.; Pilon, G.; Dudonné, S.; Marette, A.; Jacques, H. Strawberry and cranberry polyphenols improve insulin sensitivity in insulin-resistant, non-diabetic adults: A parallel, double-blind, controlled and randomised clinical trial. Br. J. Nutr. 2017, 117, 519–531. [Google Scholar]
- Sikora, J.; Broncel, M.; Markowicz, M.; Chałubiński, M.; Wojdan, K.; Mikiciuk-Olasik, E. Short-term supplementation with Aronia melanocarpa extract improves platelet aggregation, clotting, and fibrinolysis in patients with metabolic syndrome. Eur. J. Nutr. 2012, 51, 549–556. [Google Scholar] [CrossRef]
- Lavefve, L.; Howard, L.R.; Carbonero, F. Berry polyphenols metabolism and impact on human gut microbiota and health. Food Funct. 2020, 11, 45–65. [Google Scholar]
- Kim, G.-S.; Park, H.J.; Woo, J.-H.; Kim, M.-K.; Koh, P.-O.; Min, W.; Ko, Y.-G.; Kim, C.-H.; Won, C.-K.; Cho, J.-H. Citrus aurantium flavonoids inhibit adipogenesis through the Akt signaling pathway in 3T3-L1 cells. BMC Complement. Altern. Med. 2012, 12, 1–10. [Google Scholar] [CrossRef]
- Park, J.; Kim, H.-L.; Jung, Y.; Ahn, K.S.; Kwak, H.J.; Um, J.-Y. Bitter orange (Citrus aurantium Linné) improves obesity by regulating adipogenesis and thermogenesis through AMPK activation. Nutrients 2019, 11, 1988. [Google Scholar] [CrossRef]
- Dallas, C.; Gerbi, A.; Tenca, G.; Juchaux, F.; Bernard, F.-X. Lipolytic effect of a polyphenolic citrus dry extract of red orange, grapefruit, orange (SINETROL) in human body fat adipocytes. Mechanism of action by inhibition of cAMP-phosphodiesterase (PDE). Phytomedicine 2008, 15, 783–792. [Google Scholar] [CrossRef]
- Fukuchi, Y.; Hiramitsu, M.; Okada, M.; Hayashi, S.; Nabeno, Y.; Osawa, T.; Naito, M. Lemon polyphenols suppress diet-induced obesity by up-regulation of mRNA levels of the enzymes involved in β-oxidation in mouse white adipose tissue. J. Clin. Biochem. Nutr. 2008, 43, 201–209. [Google Scholar]
- Harton, A.; Myszkowska-Ryciak, J.; Gajewska, D.; Webb, M. The Role of Selected Bioactive Compounds in Teas, Spices, Cocoa and Coffee in Body Weight Control. Pol. J. Appl. Sci. 2017, 1, 56–66. [Google Scholar]
- Andújar, I.; Recio, M.C.; Giner, R.M.; Ríos, J. Cocoa polyphenols and their potential benefits for human health. Oxidative Med. Cell. Longev. 2012, 2012, 906252. [Google Scholar] [CrossRef]
- Ferrazzano, G.F.; Amato, I.; Ingenito, A.; De Natale, A.; Pollio, A. Anti-cariogenic effects of polyphenols from plant stimulant beverages (cocoa, coffee, tea). Fitoterapia 2009, 80, 255–262. [Google Scholar] [CrossRef]
- Shahmohammadi, H.A.; Hosseini, S.A.; Hajiani, E.; Malehi, A.S.; Alipour, M. Effects of green coffee bean extract supplementation on patients with non-alcoholic fatty liver disease: A randomized clinical trial. Hepat. Mon. 2017, 17. [Google Scholar]
- Nordestgaard, A.T.; Thomsen, M.; Nordestgaard, B.G. Coffee intake and risk of obesity, metabolic syndrome and type 2 diabetes: A Mendelian randomization study. Int. J. Epidemiol. 2015, 44, 551–565. [Google Scholar] [CrossRef]
- Kim, H.-J.; Kim, B.; Mun, E.-G.; Jeong, S.-Y.; Cha, Y.-S. The antioxidant activity of steamed ginger and its protective effects on obesity induced by high-fat diet in C57BL/6J mice. Nutr. Res. Pract. 2018, 12, 503–511. [Google Scholar]
- Suk, S.; Kwon, G.T.; Lee, E.; Jang, W.J.; Yang, H.; Kim, J.H.; Thimmegowda, N.; Chung, M.Y.; Kwon, J.Y.; Yang, S. Gingerenone A, a polyphenol present in ginger, suppresses obesity and adipose tissue inflammation in high-fat diet-fed mice. Mol. Nutr. Food Res. 2017, 61, 1700139. [Google Scholar]
- Ebrahimzadeh Attari, V.; Asghari Jafarabadi, M.; Zemestani, M.; Ostadrahimi, A. Effect of Zingiber officinale supplementation on obesity management with respect to the uncoupling protein 1-3826A>G and ß3-adrenergic receptor Trp64Arg polymorphism. Phytother. Res. 2015, 29, 1032–1039. [Google Scholar]
- Park, S.-H.; Jung, S.-J.; Choi, E.-K.; Ha, K.-C.; Baek, H.-I.; Park, Y.-K.; Han, K.-H.; Jeong, S.-Y.; Oh, J.-H.; Cha, Y.-S. The effects of steamed ginger ethanolic extract on weight and body fat loss: A randomized, double-blind, placebo-controlled clinical trial. Food Sci. Biotechnol. 2020, 29, 265–273. [Google Scholar]
- Bucciantini, M.; Leri, M.; Nardiello, P.; Casamenti, F.; Stefani, M. Olive Polyphenols: Antioxidant and Anti-Inflammatory Properties. Antioxidants 2021, 10, 1044, Diet. Polyphen. Neuroprotection2021, 71. [Google Scholar]
- Araki, R.; Fujie, K.; Yuine, N.; Watabe, Y.; Nakata, Y.; Suzuki, H.; Isoda, H.; Hashimoto, K. Olive leaf tea is beneficial for lipid metabolism in adults with prediabetes: An exploratory randomized controlled trial. Nutr. Res. 2019, 67, 60–66. [Google Scholar]
- Kandemir, K.; Tomas, M.; McClements, D.J.; Capanoglu, E. Recent advances on the improvement of quercetin bioavailability. Trends Food Sci. Technol. 2022, 119, 192–200. [Google Scholar] [CrossRef]
- Jakobek, L. Interactions of polyphenols with carbohydrates, lipids and proteins. Food Chem. 2015, 175, 556–567. [Google Scholar]
- Duarte, G.S.; Farah, A. Effect of simultaneous consumption of milk and coffee on chlorogenic acids’ bioavailability in humans. J. Agric. Food Chem. 2011, 59, 7925–7931. [Google Scholar]
- Murota, K.; Nakamura, Y.; Uehara, M. Flavonoid metabolism: The interaction of metabolites and gut microbiota. Biosci. Biotechnol. Biochem. 2018, 82, 600–610. [Google Scholar]
- Nagar, E.E.; Okun, Z.; Shpigelman, A. Digestive fate of polyphenols: Updated view of the influence of chemical structure and the presence of cell wall material. Curr. Opin. Food Sci. 2020, 31, 38–46. [Google Scholar] [CrossRef]
- Lu, Y.; Fan, C.; Li, P.; Lu, Y.; Chang, X.; Qi, K. Short chain fatty acids prevent high-fat-diet-induced obesity in mice by regulating G protein-coupled receptors and gut microbiota. Sci. Rep. 2016, 6, 1–13. [Google Scholar]
- van der Beek, C.M.; Canfora, E.E.; Kip, A.M.; Gorissen, S.H.; Damink, S.W.O.; van Eijk, H.M.; Holst, J.J.; Blaak, E.E.; Dejong, C.H.; Lenaerts, K. The prebiotic inulin improves substrate metabolism and promotes short-chain fatty acid production in overweight to obese men. Metabolism 2018, 87, 25–35. [Google Scholar]
- Den Besten, G.; Bleeker, A.; Gerding, A.; van Eunen, K.; Havinga, R.; van Dijk, T.H.; Oosterveer, M.H.; Jonker, J.W.; Groen, A.K.; Reijngoud, D.-J. Short-chain fatty acids protect against high-fat diet–induced obesity via a PPARγ-dependent switch from lipogenesis to fat oxidation. Diabetes 2015, 64, 2398–2408. [Google Scholar] [CrossRef]
- Weitkunat, K.; Stuhlmann, C.; Postel, A.; Rumberger, S.; Fankhänel, M.; Woting, A.; Petzke, K.J.; Gohlke, S.; Schulz, T.J.; Blaut, M. Short-chain fatty acids and inulin, but not guar gum, prevent diet-induced obesity and insulin resistance through differential mechanisms in mice. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef]
- De la Cuesta-Zuluaga, J.; Mueller, N.T.; Álvarez-Quintero, R.; Velásquez-Mejía, E.P.; Sierra, J.A.; Corrales-Agudelo, V.; Carmona, J.A.; Abad, J.M.; Escobar, J.S. Higher fecal short-chain fatty acid levels are associated with gut microbiome dysbiosis, obesity, hypertension and cardiometabolic disease risk factors. Nutrients 2018, 11, 51. [Google Scholar] [CrossRef]
- Hackman, R.M.; Polagruto, J.A.; Zhu, Q.Y.; Sun, B.; Fujii, H.; Keen, C.L. Flavanols: Digestion, absorption and bioactivity. Phytochem. Rev. 2008, 7, 195–208. [Google Scholar] [CrossRef]
- Filosa, S.; Di Meo, F.; Crispi, S. Polyphenols-gut microbiota interplay and brain neuromodulation. Neural Regen. Res. 2018, 13, 2055. [Google Scholar]
- Roowi, S.; Stalmach, A.; Mullen, W.; Lean, M.E.; Edwards, C.A.; Crozier, A. Green tea flavan-3-ols: Colonic degradation and urinary excretion of catabolites by humans. J. Agric. Food Chem. 2010, 58, 1296–1304. [Google Scholar]
- Loke, W.M.; Jenner, A.M.; Proudfoot, J.M.; McKinley, A.J.; Hodgson, J.M.; Halliwell, B.; Croft, K.D. A metabolite profiling approach to identify biomarkers of flavonoid intake in humans. J. Nutr. 2009, 139, 2309–2314. [Google Scholar]
- Park, S.; Oh, S.; Kim, T.; Ham, J.; Lee, S. Synthesis of 3,4,5-Trimethoxybenzene Derivatives with Substituted Triazole Group by Huisgen 1,3-Cycloaddition and Their Inhibitory Effects on 3T3-L1 Adipocyte Differentiation. ChemInform 2016, 47, 1364–1367. [Google Scholar] [CrossRef]
- Aura, A.-M.; O’leary, K.; Williamson, G.; Ojala, M.; Bailey, M.; Puupponen-Pimiä, R.; Nuutila, A.-M.; Oksman-Caldentey, K.-M.; Poutanen, K. Quercetin derivatives are deconjugated and converted to hydroxyphenylacetic acids but not methylated by human fecal flora in vitro. J. Agric. Food Chem. 2002, 50, 1725–1730. [Google Scholar]
- Appeldoorn, M.M.; Vincken, J.-P.; Aura, A.-M.; Hollman, P.C.; Gruppen, H. Procyanidin dimers are metabolized by human microbiota with 2-(3,4-dihydroxyphenyl) acetic acid and 5-(3,4-dihydroxyphenyl)-γ-valerolactone as the major metabolites. J. Agric. Food Chem. 2009, 57, 1084–1092. [Google Scholar]
- Coldham, N.; Darby, C.; Hows, M.; King, L.; Zhang, A.-Q.; Sauer, M. Comparative metabolism of genistin by human and rat gut microflora: Detection and identification of the end-products of metabolism. Xenobiotica 2002, 32, 45–62. [Google Scholar] [CrossRef]
- Hidalgo, M.; Oruna-Concha, M.J.; Kolida, S.; Walton, G.E.; Kallithraka, S.; Spencer, J.P.; de Pascual-Teresa, S. Metabolism of anthocyanins by human gut microflora and their influence on gut bacterial growth. J. Agric. Food Chem. 2012, 60, 3882–3890. [Google Scholar] [CrossRef]
- Hsu, C.-L.; Huang, S.-L.; Yen, G.-C. Inhibitory effect of phenolic acids on the proliferation of 3T3-L1 preadipocytes in relation to their antioxidant activity. J. Agric. Food Chem. 2006, 54, 4191–4197. [Google Scholar]
- Cerdá, B.; Periago, P.; Espín, J.C.; Tomás-Barberán, F.A. Identification of urolithin A as a metabolite produced by human colon microflora from ellagic acid and related compounds. J. Agric. Food Chem. 2005, 53, 5571–5576. [Google Scholar]
- Xia, B.; Shi, X.C.; Xie, B.C.; Zhu, M.Q.; Chen, Y.; Chu, X.Y.; Cai, G.H.; Liu, M.; Yang, S.Z.; Mitchell, G.A. Urolithin A exerts antiobesity effects through enhancing adipose tissue thermogenesis in mice. PLoS Biol. 2020, 18, e3000688. [Google Scholar]
- Selma, M.V.; Beltrán, D.; García-Villalba, R.; Espín, J.C.; Tomás-Barberán, F.A. Description of urolithin production capacity from ellagic acid of two human intestinal Gordonibacter species. Food Funct. 2014, 5, 1779–1784. [Google Scholar]
- Serra, A.; Macià, A.; Romero, M.-P.; Reguant, J.; Ortega, N.; Motilva, M.-J. Metabolic pathways of the colonic metabolism of flavonoids (flavonols, flavones and flavanones) and phenolic acids. Food Chem. 2012, 130, 383–393. [Google Scholar]
- Rechner, A.R.; Smith, M.A.; Kuhnle, G.; Gibson, G.R.; Debnam, E.S.; Srai, S.K.S.; Moore, K.P.; Rice-Evans, C.A. Colonic metabolism of dietary polyphenols: Influence of structure on microbial fermentation products. Free. Radic. Biol. Med. 2004, 36, 212–225. [Google Scholar]
- Bode, L.M.; Bunzel, D.; Huch, M.; Cho, G.-S.; Ruhland, D.; Bunzel, M.; Bub, A.; Franz, C.M.; Kulling, S.E. In vivo and in vitro metabolism of trans-resveratrol by human gut microbiota. Am. J. Clin. Nutr. 2013, 97, 295–309. [Google Scholar] [CrossRef]
- Trinh, H.-T.; Joh, E.-H.; Kwak, H.-Y.; Baek, N.-I.; Kim, D.-H. Anti-pruritic effect of baicalin and its metabolites, baicalein and oroxylin A, in mice. Acta Pharmacol. Sin. 2010, 31, 718–724. [Google Scholar]
- Min, W.; Wu, M.; Fang, P.; Yu, M.; Shi, M.; Zhang, Z.; Bo, P. Effect of baicalein on GLUT4 translocation in adipocytes of diet-induced obese mice. Cell. Physiol. Biochem. 2018, 50, 426–436. [Google Scholar] [CrossRef]
- Griffiths, L.; Smith, G. Metabolism of apigenin and related compounds in the rat. Metabolite formation in vivo and by the intestinal microflora in vitro. Biochem. J. 1972, 128, 901–911. [Google Scholar]
- Hsu, C.-L.; Yen, G.-C. Effects of flavonoids and phenolic acids on the inhibition of adipogenesis in 3T3-L1 adipocytes. J. Agric. Food Chem. 2007, 55, 8404–8410. [Google Scholar] [CrossRef]
- Kumar Singh, A.; Cabral, C.; Kumar, R.; Ganguly, R.; Kumar Rana, H.; Gupta, A.; Rosaria Lauro, M.; Carbone, C.; Reis, F.; Pandey, A.K. Beneficial effects of dietary polyphenols on gut microbiota and strategies to improve delivery efficiency. Nutrients 2019, 11, 2216. [Google Scholar] [CrossRef]
- Gonçalves, R.F.; Martins, J.T.; Duarte, C.M.; Vicente, A.A.; Pinheiro, A.C. Advances in nutraceutical delivery systems: From formulation design for bioavailability enhancement to efficacy and safety evaluation. Trends Food Sci. Technol. 2018, 78, 270–291. [Google Scholar] [CrossRef]
- Sansone, F.; Picerno, P.; Mencherini, T.; Russo, P.; Gasparri, F.; Giannini, V.; Lauro, M.R.; Puglisi, G.; Aquino, R.P. Enhanced technological and permeation properties of a microencapsulated soy isoflavones extract. J. Food Eng. 2013, 115, 298–305. [Google Scholar]
- Papillo, V.A.; Locatelli, M.; Travaglia, F.; Bordiga, M.; Garino, C.; Coïsson, J.D.; Arlorio, M. Cocoa hulls polyphenols stabilized by microencapsulation as functional ingredient for bakery applications. Food Res. Int. 2019, 115, 511–518. [Google Scholar]
- Zhou, H.; Zheng, B.; McClements, D.J. In Vitro Gastrointestinal Stability of Lipophilic Polyphenols is Dependent on their Oil–Water Partitioning in Emulsions: Studies on Curcumin, Resveratrol, and Quercetin. J. Agric. Food Chem. 2021, 69, 3340–3350. [Google Scholar] [CrossRef]
- Ning, P.; Lü, S.; Bai, X.; Wu, X.; Gao, C.; Wen, N.; Liu, M. High encapsulation and localized delivery of curcumin from an injectable hydrogel. Mater. Sci. Eng. C 2018, 83, 121–129. [Google Scholar]
- Wang, L.-C.; Pan, T.-M.; Tsai, T.-Y. Lactic acid bacteria-fermented product of green tea and Houttuynia cordata leaves exerts anti-adipogenic and anti-obesity effects. J. Food Drug Anal. 2018, 26, 973–984. [Google Scholar] [CrossRef]
- Khan, M.K.; Ahmad, K.; Hassan, S.; Imran, M.; Ahmad, N.; Xu, C. Effect of novel technologies on polyphenols during food processing. Innov. Food Sci. Emerg. Technol. 2018, 45, 361–381. [Google Scholar]
- Ofosu, F.K.; Elahi, F.; Daliri, E.B.-M.; Han, S.-I.; Oh, D.-H. Impact of thermal treatment and fermentation by lactic acid bacteria on sorghum metabolite changes, their antioxidant and antidiabetic activities. Food Biosci. 2022, 45, 101502. [Google Scholar]
- Aloo, S.-O.; Ofosu, F.-K.; Daliri, E.-B.-M.; Oh, D.-H. UHPLC-ESI-QTOF-MS/MS metabolite profiling of the antioxidant and antidiabetic activities of red cabbage and broccoli seeds and sprouts. Antioxidants 2021, 10, 852. [Google Scholar] [CrossRef]
- Lambert, J.D.; Sang, S.; Yang, C.S. Possible controversy over dietary polyphenols: Benefits vs risks. Chem. Res. Toxicol. 2007, 20, 583–585. [Google Scholar] [CrossRef]
- Bentivegna, S.; Whitney, K. Subchronic 3-month oral toxicity study of grape seed and grape skin extracts. Food Chem. Toxicol. 2002, 40, 1731–1743. [Google Scholar] [CrossRef]
- Mennen, L.I.; Walker, R.; Bennetau-Pelissero, C.; Scalbert, A. Risks and safety of polyphenol consumption. Am. J. Clin. Nutr. 2005, 81, 326S–329S. [Google Scholar] [CrossRef]
- Boocock, D.J.; Faust, G.E.; Patel, K.R.; Schinas, A.M.; Brown, V.A.; Ducharme, M.P.; Booth, T.D.; Crowell, J.A.; Perloff, M.; Gescher, A.J. Phase I dose escalation pharmacokinetic study in healthy volunteers of resveratrol, a potential cancer chemopreventive agent. Cancer Epidemiol. Prev. Biomark. 2007, 16, 1246–1252. [Google Scholar]
- Popat, R.; Plesner, T.; Davies, F.; Cook, G.; Cook, M.; Elliott, P.; Jacobson, E.; Gumbleton, T.; Oakervee, H.; Cavenagh, J. A phase 2 study of SRT501 (resveratrol) with bortezomib for patients with relapsed and or refractory multiple myeloma. Br. J. Haematol. 2012, 160, 714–717. [Google Scholar] [CrossRef]
- Bhatt, J.K.; Thomas, S.; Nanjan, M.J. Resveratrol supplementation improves glycemic control in type 2 diabetes mellitus. Nutr. Res. 2012, 32, 537–541. [Google Scholar] [CrossRef]
- Ottaviani, J.I.; Borges, G.; Momma, T.Y.; Spencer, J.P.; Keen, C.L.; Crozier, A.; Schroeter, H. The metabolome of [2-14C](−)-epicatechin in humans: Implications for the assessment of efficacy, safety and mechanisms of action of polyphenolic bioactives. Sci. Rep. 2016, 6, 29034. [Google Scholar]
- Heber, D.; Seeram, N.P.; Wyatt, H.; Henning, S.M.; Zhang, Y.; Ogden, L.G.; Dreher, M.; Hill, J.O. Safety and antioxidant activity of a pomegranate ellagitannin-enriched polyphenol dietary supplement in overweight individuals with increased waist size. J. Agric. Food Chem. 2007, 55, 10050–10054. [Google Scholar] [CrossRef]
- Chow, H.S.; Cai, Y.; Hakim, I.A.; Crowell, J.A.; Shahi, F.; Brooks, C.A.; Dorr, R.T.; Hara, Y.; Alberts, D.S. Pharmacokinetics and safety of green tea polyphenols after multiple-dose administration of epigallocatechin gallate and polyphenon E in healthy individuals. Clin. Cancer Res. 2003, 9, 3312–3319. [Google Scholar]
- Frank, J.; George, T.W.; Lodge, J.K.; Rodriguez-Mateos, A.M.; Spencer, J.P.; Minihane, A.M.; Rimbach, G. Daily consumption of an aqueous green tea extract supplement does not impair liver function or alter cardiovascular disease risk biomarkers in healthy men. J. Nutr. 2009, 139, 58–62. [Google Scholar]
- Shoba, G.; Joy, D.; Joseph, T.; Majeed, M.; Rajendran, R.; Srinivas, P. Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med. 1998, 64, 353–356. [Google Scholar] [CrossRef]
- Yoshitomi, R.; Yamamoto, M.; Kumazoe, M.; Fujimura, Y.; Yonekura, M.; Shimamoto, Y.; Nakasone, A.; Kondo, S.; Hattori, H.; Haseda, A. The combined effect of green tea and α-glucosyl hesperidin in preventing obesity: A randomized placebo-controlled clinical trial. Sci. Rep. 2021, 11, 19067. [Google Scholar] [CrossRef]
- Jamar, G.; Santamarina, A.B.; Flygare, A.C.; Gagliardi, A.; de Rosso, V.V.; Dourado, V.Z.; Pisani, L.P. Effects of the jucara fruit supplementation on metabolic parameters in individuals with obesity: A double-blind randomized controlled trial. J. Nutr. Biochem. 2020, 83, 108430. [Google Scholar] [CrossRef]
- Boix-Castejón, M.; Herranz-López, M.; Gago, A.P.; Olivares-Vicente, M.; Caturla, N.; Roche, E.; Micol, V. Hibiscus and lemon verbena polyphenols modulate appetite-related biomarkers in overweight subjects: A randomized controlled trial. Food Funct. 2018, 9, 3173–3184. [Google Scholar]
- Serna, A.; Marhuenda, J.; Arcusa, R.; Pérez-Piñeiro, S.; Sánchez-Macarro, M.; Victoria-Montesinos, D.; Cánovas, F.; Jones, J.; Caturla, N.; López-Román, J. Effectiveness of a Polyphenolic Extract (Lippia Citriodora and Hibiscus Sabdariffa) on Appetite Regulation in Overweight and Obese Grade I Population: A 8 weeks Randomized, Double-Blind, Cross-Over, Placebo-Controlled Trial. Randomized Control. Trial 2020, 61, 825–841. [Google Scholar] [CrossRef]
- Poulsen, M.M.; Vestergaard, P.F.; Clasen, B.F.; Radko, Y.; Christensen, L.P.; Stødkilde-Jørgensen, H.; Møller, N.; Jessen, N.; Pedersen, S.B.; Jørgensen, J.O.L. High-dose resveratrol supplementation in obese men: An investigator-initiated, randomized, placebo-controlled clinical trial of substrate metabolism, insulin sensitivity, and body composition. Diabetes 2013, 62, 1186–1195. [Google Scholar]
- Zhang, Y.; Yu, Y.; Li, X.; Meguro, S.; Hayashi, S.; Katashima, M.; Yasumasu, T.; Wang, J.; Li, K. Effects of catechin-enriched green tea beverage on visceral fat loss in adults with a high proportion of visceral fat: A double-blind, placebo-controlled, randomized trial. J. Funct. Foods 2012, 4, 315–322. [Google Scholar]
- Baba, A.; Hoshino, T.; Ogawa, S.; Takara, T. Improvement of glucose metabolism and safety of acacia bark-derived proanthocyanidins in healthy Japanese adults: A Randomized, Double-blind, Placebo-controlled, Parallel-group Trial. Funct. Foods Health Dis. 2021, 11, 431–455. [Google Scholar]
- Roberts, A.T.; Martin, C.K.; Liu, Z.; Amen, R.J.; Woltering, E.A.; Rood, J.C.; Caruso, M.K.; Yu, Y.; Xie, H.; Greenway, F.L. The safety and efficacy of a dietary herbal supplement and gallic acid for weight loss. J. Med. Food 2007, 10, 184–188. [Google Scholar] [CrossRef]
- Ifie, I.; Marshall, L.J. Food processing and its impact on phenolic constituents in food. Cogent Food Agric. 2018, 4, 1507782. [Google Scholar] [CrossRef]
- Dao, L.; Friedman, M. Chlorogenic acid content of fresh and processed potatoes determined by ultraviolet spectrophotometry. J. Agric. Food Chem. 1992, 40, 2152–2156. [Google Scholar]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxidative Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef]
- Ou, J.; Wang, M.; Zheng, J.; Ou, S. Positive and negative effects of polyphenol incorporation in baked foods. Food Chem. 2019, 284, 90–99. [Google Scholar] [CrossRef]
- Aloo, S.O.; Mwiti, G.; Ngugi, L.W.; Oh, D.-H. Uncovering the secrets of industrial hemp in food and nutrition: The trends, challenges, and new-age perspectives. Crit. Rev. Food Sci. Nutr. 2022, 1–20. [Google Scholar] [CrossRef]
- Shen, P.; Gao, Z.; Fang, B.; Rao, J.; Chen, B. Ferreting out the secrets of industrial hemp protein as emerging functional food ingredients. Trends Food Sci. Technol. 2021, 112, 1–15. [Google Scholar] [CrossRef]
- Spencer, J.P.; Abd El Mohsen, M.M.; Minihane, A.-M.; Mathers, J.C. Biomarkers of the intake of dietary polyphenols: Strengths, limitations and application in nutrition research. Br. J. Nutr. 2008, 99, 12–22. [Google Scholar]
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The role of polyphenols in human health and food systems: A mini-review. Front. Nutr. 2018, 5, 87. [Google Scholar]
- Hursel, R.; Viechtbauer, W.; Westerterp-Plantenga, M.S. The effects of green tea on weight loss and weight maintenance: A meta-analysis. Int. J. Obes. 2009, 33, 956–961. [Google Scholar]
- D’Archivio, M.; Filesi, C.; Varì, R.; Scazzocchio, B.; Masella, R. Bioavailability of the polyphenols: Status and controversies. Int. J. Mol. Sci. 2010, 11, 1321–1342. [Google Scholar]



| Drug | Country | Participants | Dose | Reported Adverse Effect | References |
|---|---|---|---|---|---|
| Common Drugs | |||||
| For short-term use | |||||
| Diethylpropion | Brazil | N = 69 | 50 mg per day for 6 months | Dry mouth and insomnia | [8] |
| Mazindol | USA | N = 85 | 1–3 mg per day for 6 weeks. | Dry mouth, nausea, decreased appetite, fatigue, heart rate, and constipation | [9] |
| Phendimetrazine | USA | N = 13 | 140–210 mg per day for 7-days | No observable adverse effects | [10] |
| Phentermine | Korea | N = 74 | 30 mg per day for 12 weeks | Dry mouth and insomnia | [11] |
| For long-term use | |||||
| Orlistat | Japan | N = 100 | 60 mg three times daily for 24 weeks | Oily spotting and flatus with discharge | [12] |
| Topiramate ER | Canada, Europe and South Africa | N = 646 | 192 mg per day for 24 weeks. | Paresthesia and central nervous system-related effects. | [13] |
| Lorcaserin | USA | N = 604 | 10 mg per day for 12 months | Headache, nasopharyngitis, nausea, and back pain | [14] |
| Sibutramine | USA | N = 61 | 20 mg once daily for 12 weeks | No adverse effect | [15] |
| Naltrexone/Bupropion ER | USA | N = 1650 | Naltrexone 32 mg per day plus bupropion 360 mg per day 56 weeks | Headache, constipation, nausea, dizziness, vomiting, and dry mouth | [16] |
| GLP-1 receptor agonists | |||||
| For short-term use | |||||
| Exenatide | China | N = 681 | 2 mg per day for 10 weeks | Nausea, dyslipidemia, and vomiting | [17] |
| Lixisenatide | France | N = 484 | 20 µg per day for 24 weeks | Acute pancreatitis | [18] |
| For long-term use | |||||
| Liraglutide, | Australia, Belgium, the UK, the USA, Russia, Israel, and Germany | - | 30 mg per day for 52 weeks | Gastrointestinal symptoms, primarily nausea | [19] |
| Albiglutide | USA | N = 155 | 30 mg per week for 52 weeks | Diarrhea, nausea, and vomiting | [20] |
| Dulaglutide | Not reported | N = 807 | 1.5 mg or 0.75 mg per day for 52 weeks | Nausea, diarrhea, and vomiting | [21] |
| Polyphenol-Rich Extract | Enriched Product | Experimental Model | Main Observation | References |
|---|---|---|---|---|
| A mixture of polyphenol-rich extracts from green tea, grape seed, and baobab fruit | White bread | Clinical trial | Improved insulin sensitivity | [36] |
| Catechins, chlorogenic acids, and hydroxytyrosol | Cooked ham | In vivo | Decreased fat accumulation (23.08% reduction) | [37] |
| Extracts of pomegranate peels and pomegranate peels (rich in ellagitannins, gallic acid, and ellagic acid) | Cookies | In-vitro | Inhibited α-glucosidase, α-amylase, and lipase activities | [38] |
| Green tea extract (containing catechins) | Bread | Clinical trial | Improved body weight, waist circumference | [39] |
| Green tea polyphenols | Bread | In vivo | Significantly suppressed body weight gain | [40] |
| Proanthocyanidins from carob fruit extract | Meat | In vivo | Decreased accumulation of liver fats | [41] |
| Polymerized-polyphenol extract from oolong tea | Oolong tea | Clinical trial | Improved body lipid accumulation | [42] |
| Polyphenol | Model | Treatment Dose | Key Observation | References |
|---|---|---|---|---|
| Polyphenols-rich blueberry extract | Mice | Mice were provided with 15.6 mg/kg BW per day for 12 days | Inhibited body weight gain and reverted lipid metabolism to normal. | [76] |
| Rambutan seeds extract (containing alkaloid, terpenoid, triterpenoid, and flavonoids) | 3T3-L1 cell line | Cells were treated with varying concentrations of the extracts (10 and 50 µg/mL) | Decreased triglyceride levels. Inhibited glucose-6-phosphate dehydrogenase (G6PDH) which promote adipogenesis | [73] |
| Procyanidin-rich grape seed extract (GSPE) | 3T3-L1 | Cells were treated with 140 mg/L GSPE (dissolved in water) for 24 hr on days 0, 2, or 4. | Reduced by 32% triglyceride content in cells treated at day zero. Downregulated genes responsible for preadipocyte differentiation but elevated preadipocyte factor-1 (Pref-1). | [75] |
| Polyphenol-rich cranberry extracts | Mice | Mice were provided with 0.75% (w/w) of polyphenol-cranberry-rich extract per day for 16 weeks. | Elevated energy expenditure and brown adipose tissue thermogenesis. | [77] |
| Polyphenol-rich totum-63 extract | Mice | Mice were fed with a high-fat diet for 12 weeks, followed by supplementation with Totum-63 (2.7% w/w) for 4 weeks. | Decreased body weight and fat mass. Increased expression of insulin receptor β and insulin-induced phosphorylation of PKB in skeletal muscle, white adipose tissue (WAT), and brown adipose tissue (BAT), thereby inducing thermogenesis. | [78] |
| Polyphenol-rich extracts from Antirhea borbonica, Doratoxylon apetalum and Gouania mauritiana | 3T3-L1 | Cells were treated with polyphenol-rich from the respective extract (0–200 μM GAE) for 24, 48, and 72 h | Inhibited obesity-induced inflammation and oxidative stress | [79] |
| Anthocyanin-rich blueberry extract | Mice. | Mice were fed with 50–200 mg/kg per day for 8 weeks | High doses of 200 mg/kg reduced body weight by 19.4%, while 50 and 100 mg/kg doses did not significantly affect body weight. | [80] |
| Cyanidin 3-O-β-d-glucoside (C3G)-rich blackberries | Rats | Rats were given 10% (w/w) of C3G-rich blackberry | Reduced overall body weight | [81] |
| Acacia-rich polyphenols (containing catechin-like flavan-3-ols, such as robinetinidol and fisetinidol) | Mice | Mice were provided with a high-fat diet supplemented with 2.5–5.0% (w/w) of acacia polyphenol extract for 7 weeks. | Decreased body weight and also elevated the mRNA expression of energy expenditure-related genes | [82] |
| Anthocyanins-rich black soybean extract | 3T3-L1 cells | Cells were treated with anthocyanins-rich black soybean extract (at a concentration of 12.5–50 μg/mL) | Inhibited the proliferation of both preconfluent preadipocytes and mature postconfluent adipocytes | [74] |
| Polyphenol | Subject | Country | Study Objective | Intervention | Key Observations | References |
|---|---|---|---|---|---|---|
| Polyphenol-rich green tea extract | N = 100, Women, age: 16–60 years BMI: >27 kg/m2 | Taiwan | To examine the effect of green tea extract on obesity. | The subjects consumed green tea containing (491 mg of catechins containing 302 mg EGCG per day) for 12 weeks. | 0.3% reduction in body weight (Equiv. 0.15 kg) after 12 weeks of treatment. Significantly reduced triglyceride levels. | [84] |
| Polyphenol-rich chocolate (contained mainly epicatechin) | N = 1017, Men and women aged: 20 to 85 years | USA | To evaluate the effect of chocolate rich in phenolic compounds on body mass index. | Subjects ate chocolate, with a mean intake of 2.0 (2.5) times/week and exercised 3.6 (3.0) times/week. | Improved BMI. | [85] |
| Yerba Mate (Ilex Paraguariensis) (rich in (quercetin rutin, chlorogenic and caffeic acids) | N = 15 BMI < 35 and ≥ 25 kg/m2 and waist-hip ratio (WHR) ≥ 0.90 for men or ≥ 0.85 for women | Korea | To investigate the efficacy of Yerba Mate supplementation against obesity. | The subjects were given 13 g/day of Yerba Mate capsules for 12 weeks. | Decreased BMI (p = 0.036), body mass fat (p = 0.030), and waist-hip ratio (p = 0.004). | [86] |
| Soya isoflavones | N = 100, postmenopausal women age: 50–70 years BMI 28–40 kg/m2 | Canada | To assess the combined effect of exercise and soy isoflavones on obesity. | Subjects consumed a 70 mg/day dose of isoflavones for 12 months. | Decreased trunk fat mass and increased lean body mass. | [87] |
| Mixture of polyphenols | N = 573, 277 men, 296 women, age: 66.2–68.3 years BMI > 30 kg/m2 | Spain | To assess the associations between total polyphenol and obesity parameters among the elderly after a long period of polyphenol intake (measured by overall urinary polyphenol level). | Participants known to consume foods rich in polyphenols were recruited and followed up for 5 years. Spot urine samples were collected and analyzed for total polyphenols, and obesity indicators were measured. | Increased consumption of dietary polyphenols was associated with improved BMI after 5 years of consumption. | [88] |
| Citrus polyphenolic extract of red-orange, grapefruit, and orange (Sinetrol-XPur) | N = 95, 55 women and 40 men, age: 22–45 years BMI 26–29.9 kg/m2 | France | To investigate appropriate polyphenolic-rich combinations that would help reduce body fat, inflammation, and oxidative stress in overweight subjects. | Subjects consumed two capsules of citrus polyphenolic extract containing orange, grapefruit, sweet orange, and guarana daily for 12 weeks. | Reduced abdominal fat and overall body weight. | [83] |
| Polyphenol-rich green tea extract | N = 35, Men and women, mean, age 42.5 ± 1.7 years BMI: 36.1 ± 1.3 kg/m2 | USA | To compare the effects of green tea polyphenols with controls on body weight and safety parameters in obese subjects. | Subjects took either four cups of decaffeinated green tea beverage or two capsules of green tea extract containing either 28 mg or 870 mg of catechins (GC, GCG, EC, ECG, EGC, and EGCG) daily for 8 weeks. | Decreased body weight. Improved LDL-cholesterol level | [89] |
| Licorice flavonoid oil | N = 22, men and women, age: 20–53 years BMI: 25.0–36.0 kg/m2 | USA | To investigate the effect of licorice flavonoid oil supplementation on obesity-related health markers. | Subjects consumed three capsules of licorice flavonoid oil, 300 mg per day for 8 weeks. | Inhibited the total cholesterol level. Decreased total triglycerides. | [90] |
| Model | Experimental Procedure | Observation | References |
|---|---|---|---|
| Mice | Mice were fed with a high-fat diet supplemented with sodium acetate, sodium propionate, sodium butyrate or their mixture (ratio at 3:1:1) | Caused changes in the bacterial community: reduced the proportion of Firmicutes and increased Bacteroidetes. | [122] |
| Human | Participants received a daily dietary supplement of 24 g inulin (source of SCFA) for two investigation days, with at least 5 days of washout | Improved β-cell function with increased insulin secretion. No effects on plasma triglycerides, or free glycerol. | [123] |
| Mice | The mice were fed diets containing sodium acetate, sodium propionate or sodium butyrate at 5% (w/w). | Induced reduction in body weight and stimulated insulin sensitivity. | [124] |
| Mice | Animals were fed a high-fat diet supplemented with 5% acetate or propionate (in the presence of 5% cellulose). | SCFA lowered hepatic triglycerides and improved insulin sensitivity. | [125] |
| Human | Participants (n = 441) were recruited and examined for their fecal SCFA, and related markers of obesity were analyzed. | Higher SCFA in fecal excretion was associated with gut modulation effects. | [126] |
| Polyphenol | Gut Bacteria Involved | Model | Main Metabolite | Major Observation | References |
|---|---|---|---|---|---|
| Epicatechin | Eubacterium sp., Bifidobacterium sp., Lactobacillus, anaerobic cocci, and, Fusobacterium spp. | In vitro study (human feces) | 5-(3,4-dihydroxyphenyl)-γ-valeric acid,3-(3-hydroxyphenyl)propionic acid,4-hydroxyphenyl acetic acid | - | [129] |
| Quercetin | Eubacterium ramulus, Eggerthella sp. | Animal study (urine) | 4-ethylphenol, Benzoic acid,4-ethylbenzoic acid | Inhibitory effects on α-amylase enzyme | [130] |
| Epicatechin | - | Animal study (urine) | 1,3,5-Trimethoxybenzene | Inhibit adipocyte differentiation | [130,131] |
| Quercetin | Bacteroides | In vitro study (humans feces) | Hydroxyphenylacetic acid derivatives | Gut microbiota modulation | [132] |
| Rutin | Bacteroides | In vitro study (humans feces) | 3,4-dihydroxyphenylacetic acid | Gut microbiota modulation | [132] |
| Isoflavone | Streptococcus intermedius, Bifidobacterium spp., Bacteroides ovatus, Streptococcus intermedius, Escherichia coli | In vitro study (human urine) | Dihydrodaidzein (DHD), tetrahydrodaidzein (THD), equol, and O-DMA | - | [25] |
| Flavan-3-ols | Clostridium coccoides, Bifidobacterium spp. | In vitro study (human feces) | Dihydroxyphenylpropionic (dihydrocaffeic) acid and 3,4-dihydroxybenzoic (protocatechuic) acid (PCA) | Inhibit pancreatic lipase activities | [25,133] |
| Genistin, | No specific bacteria reported | In vitro study (human and animal feces) | 4-hydroxyphenyl-2-propionic acid and 1,3,5-trihydroxybenzene | - | [134] |
| Anthocyanin | Lactobacillus spp. | In vitro study (human feces) | Gallic acid, syringic acid and p-coumaric acid | Inhibition of preadipocytes growth | [135,136] |
| Ellagic acid | Gordonibacter urolithinfaciens | In vitro study (human feces) | Urolithins | Inducing thermogenesis in brown adipose tissue (BAT) and inducing browning of white adipose tissue (WAT). | [137,138] |
| Ellagitannins | Gordonibacter urolithinfaciens | In vitro study (human feces) | Urolithins | Inducing thermogenesis in brown adipose tissue (BAT) and inducing browning of white adipose tissue (WAT | [139] |
| Naringenin | No specific bacteria reported | In vitro study (rat feces) | Phenylacetic acid, protocatechuic acid | - | [140] |
| Chlorogenic acid | No specific bacteria reported | In vitro study (human feces) | 3-(3-hydroxyphenyl)-propionic acid | - | [141] |
| Resveratrol | Slackia equolifaciens, Adlercreutzia equolifaciens | In vivo and in vitro (human feces) | Dihydroresveratrol, and lunularin | - | [25,142] |
| Baicalin | No specific bacteria reported | In vitro study (human feces) | Baicalein and oroxylin A | Enhances pAKT, PGC-1α and UCP1 | [143,144] |
| Apigenin | No specific bacteria reported | Animal study (urine) | P-hydroxyphenyl acetic acid, P-hydroxycinnamic acid, P-hydroxybenzoic acid | Inhibition of adipogenesis | [145,146] |
| Polyphenol | Subject Description | Dosage | Side Effect | References |
|---|---|---|---|---|
| Resveratrol (RV) | 40 healthy volunteers aged 18–80 years | Participants took oral resveratrol (single doses of 0.5, 1, 2.5, or 5 g) per day for 1 week. | An intake of up to one dose of 5 g of resveratrol was safe, with only minor adverse events in some cases. | [160] |
| Sinetrol-XPur (polyphenolic citrus dry extract) | 95 healthy overweight volunteers (55 women and 40 men), age: 22–45 years, BMI: 26–29.9 kg/m2 | Subjects consumed two capsules of citrus polyphenolic extract containing orange, grapefruit, sweet orange, and guarana for 12 weeks | Mild effect, such as a slight increase in cardiac rate, was observed | [83] |
| Resveratrol | 24 overweight patients, median age: 66.5 years | Participants took 5.0 g resveratrol per day for 4 months | Serious adverse effects, including nausea, diarrhea, vomiting, and fatigue | [161] |
| Resveratrol | 62 participants (men and women) | Participants consumed 250 mg daily for 3 months | Improved glycemic control with no observable side effects | [162] |
| Epicatechin | age:18–50 years BMI: 19–30 kg/m2 | Subjects consumed 250 mg of cocoa flavanols; 40 mg of epicatechin for 14 days | Improved body weight parameters with no adverse events observed | [163] |
| Ellagitannin | 64 overweight individuals, age: 40–70 years, BMI: 25–32 kg/m2 | Subjects consumed 710 mg per day of a pomegranate ellagitannin-enriched polyphenol extract. | No serious adverse events on the subject upon the intake | [164] |
| Polyphenon E | 40 healthy participants with Fitzpatrick skin types II or III, age: ≥18 year | Participants took 800 mg polyphenol E once per day for 2 weeks | Adverse events, including stomach upset, nausea, heartburn, stomach pain, dizziness, headache, and muscle pain, were observed during the 4-week treatment period | [165] |
| Polyphenols-rich green tea | 17 healthy volunteers, age: 41 ± 9 years BMI 26.7 ± 3.3 kg/m2, | Participants consumed tea containing 119 mg polyphenols (epicatechin, 5 mg; epigallocatechin, 47 mg; epigallocatechingallate, 25 mg; epicatechingallate, 14 mg; gallocatechingallate, 8 mg; catechingallate, 3 mg; catechin, 1 mg; gallocatechin, 9 mg; and ellagic acid, 7 mg) and 19 mg caffeine per day for 3 weeks | The daily consumption of green tea polyphenols, even at high dose levels, was safe. No effects on cardiovascular risk biomarkers screened | [166] |
| Curcumin | 10 healthy male volunteers aged 20–26 years. Weight: 50–75 kg | Participants consumed beverages formulated with 2 g of curcumin | Improved serum concentration without any toxic effect observed | [167] |
| EGCG and α-glucosyl hesperidin (gH) | 60 healthy males and females aged: 30–75 years | Subjects were given green tea, 178 mg gH and 146 mg EGCG per day for 12 weeks | The amount of EGCG and gH consumed effectively reduced body weight with no adverse effects | [168] |
| Juçara fruit polyphenols | 35 adults known to be obese (men and women) aged 31–59 years. BMI: 30–39.9 kg/m2 | Subjects consumed 5 g of pulp powder per day for 6 weeks | No toxic effects were observed. Authors’ conclusion: safe for consumption | [169] |
| Hibiscus and lemon verbena polyphenols | 54 overweight subjects, age: 30–75 years BMI: 25–34.9 kg m−2 | 500 mg of Lippia citriodora (35%) and Hibiscus sabdariffa (65%) per day for 2 months was taken by the studied subjects | No toxic effects were reported | [170] |
| Green tea (containing majorly, catechins) | 35 subjects with obesity and metabolic syndrome | Subjects consumed green tea (four cups/day) or green tea extract (two capsules/day) for 8 weeks. | Improved obesity-related parameters without any observable side effect | [89] |
| Lippia Citriodora and Hibiscus Sabdariffa extract (Lc-Hs) | 33 Volunteers (male and female), age: 18–65 years, BMI: 25–34.9 kg/m2 | Oral administration of two capsules/day, each capsule containing 250 mg for 60 days | The supplementation with the Lc-Hs extract decreased appetite sensation; no toxicity was observed upon consumption | [171] |
| High dose of resveratrol | 24 obese but healthy volunteers (men), age: 18–70 years, BMI >30 kg/m2 | Subjects consumed 500 mg/day of resveratrol for 4 weeks | Improved key obesity indicators (total and lean body mass, total body fat mass, or visceral and abdominal subcutaneous fat). No adverse effects were observed. | [172] |
| Catechin-enriched green tea | 33 obese subjects (18 men, 15 women), age: 20–65 years, waist circumference: ≥80 cm (women) or 90 cm (men) | Subjects drank a 350-mL bottle of beverage after lunchtime within 30 min daily for 12 weeks. | Adverse events such as changes in stools, abdominal discomfort, and appetite were associated with catechin-enriched green tea consumption | [173] |
| Acacia bark-derived proanthocyanidins | Participants took proanthocyanidins derived from acacia bark (245 mg/day for 12 weeks) | No side effects or adverse events were observed upon the consumption | [174] | |
| Gallic acid | 105 healthy subjects age: 18–60 years, BMI: 25–35 kg/m2 | Subjects took a 300 mg/1.2 g NT-GA combination or 600 mg/2.4 g/day NT-GA for 24 weeks. | NT-GA consumption was well tolerated but was ineffective in causing weight loss or limiting food intake | [175] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aloo, S.-O.; Ofosu, F.K.; Kim, N.-H.; Kilonzi, S.M.; Oh, D.-H. Insights on Dietary Polyphenols as Agents against Metabolic Disorders: Obesity as a Target Disease. Antioxidants 2023, 12, 416. https://doi.org/10.3390/antiox12020416
Aloo S-O, Ofosu FK, Kim N-H, Kilonzi SM, Oh D-H. Insights on Dietary Polyphenols as Agents against Metabolic Disorders: Obesity as a Target Disease. Antioxidants. 2023; 12(2):416. https://doi.org/10.3390/antiox12020416
Chicago/Turabian StyleAloo, Simon-Okomo, Fred Kwame Ofosu, Nam-Hyeon Kim, Sheila M. Kilonzi, and Deog-Hwan Oh. 2023. "Insights on Dietary Polyphenols as Agents against Metabolic Disorders: Obesity as a Target Disease" Antioxidants 12, no. 2: 416. https://doi.org/10.3390/antiox12020416
APA StyleAloo, S.-O., Ofosu, F. K., Kim, N.-H., Kilonzi, S. M., & Oh, D.-H. (2023). Insights on Dietary Polyphenols as Agents against Metabolic Disorders: Obesity as a Target Disease. Antioxidants, 12(2), 416. https://doi.org/10.3390/antiox12020416










