The Role of Isoflavones in the Prevention of Breast Cancer and Prostate Cancer
Abstract
1. Introduction
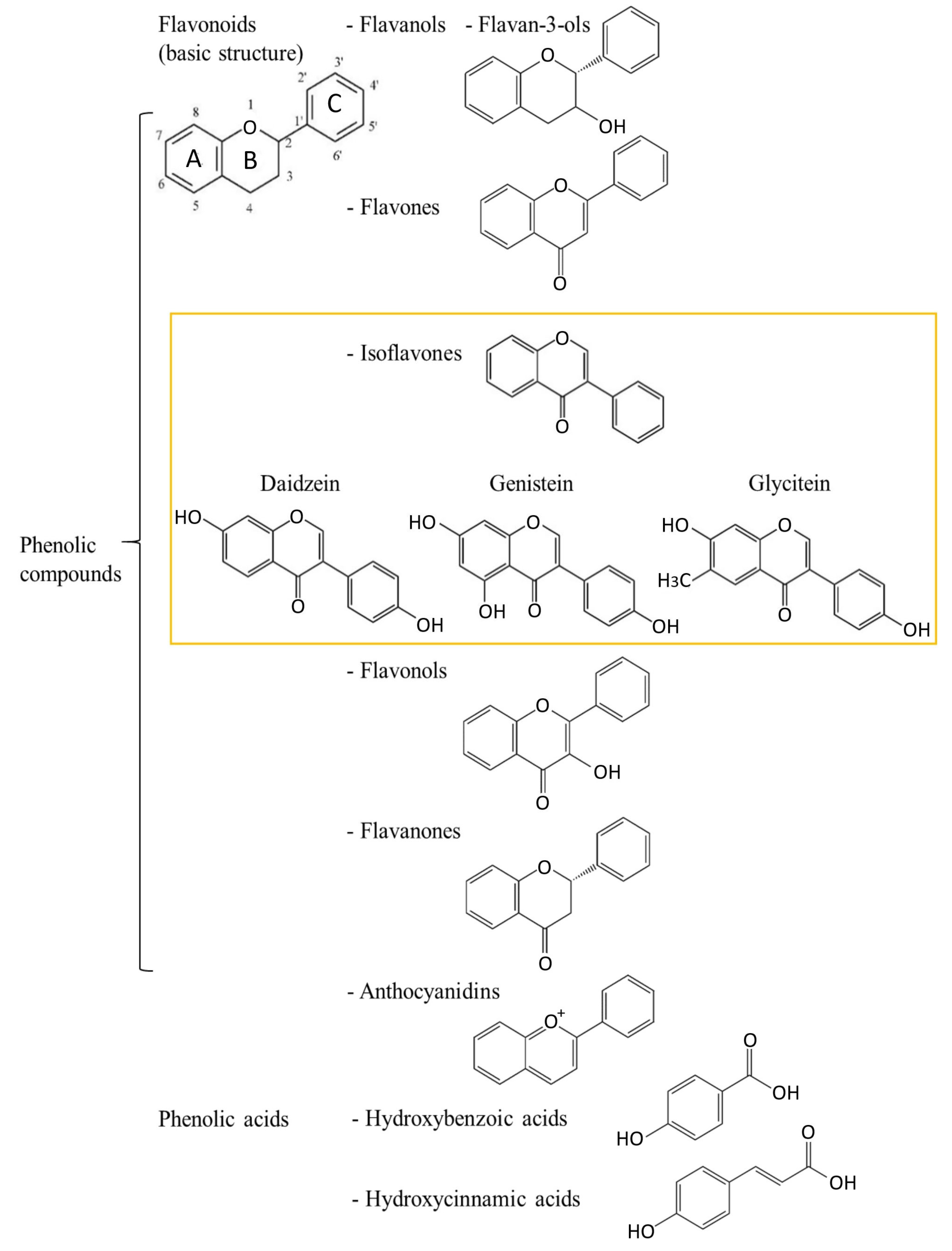
2. Polyphenols—Chemical Characteristics and Division
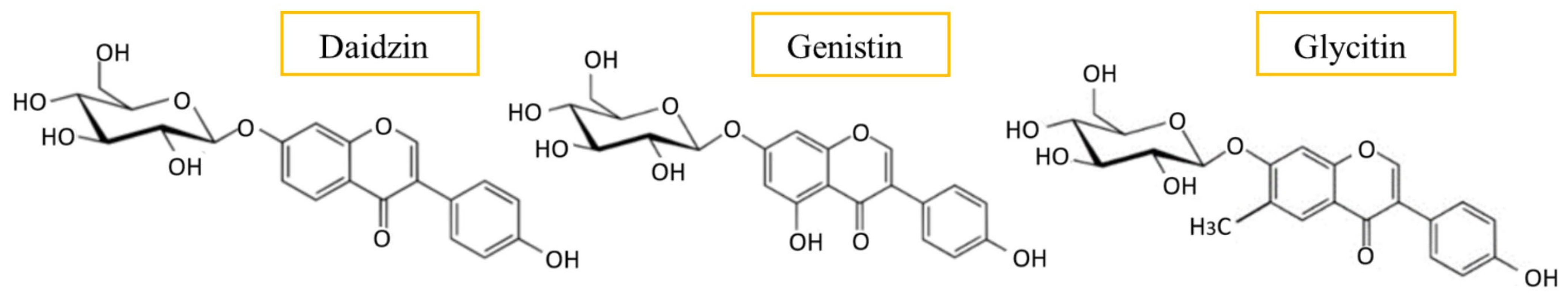
2.1. Isoflavones
Health-Related Properties of Isoflavones
3. Breast Cancer and Isoflavones
3.1. Breast Cancer—Epidemiology and Risk Factors
3.2. Breast Cancer Chemoprevention and Dietary Isoflavone Intake
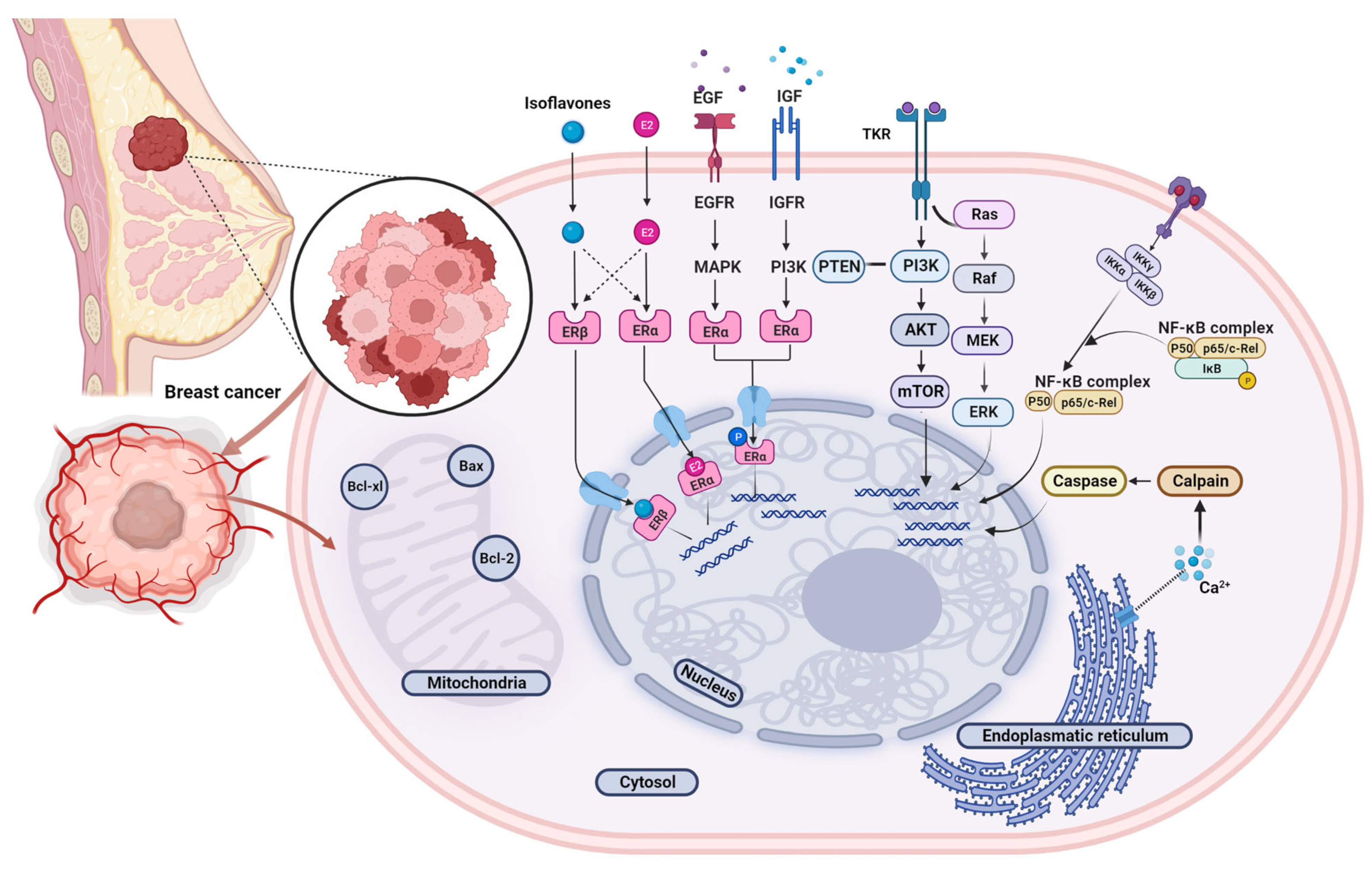
3.3. Isoflavone Mechanisms of Action and Impact on Breast Cancer
4. Prostate Cancer and Isoflavones
4.1. Prostate Cancer—Epidemiology and Risk Factors
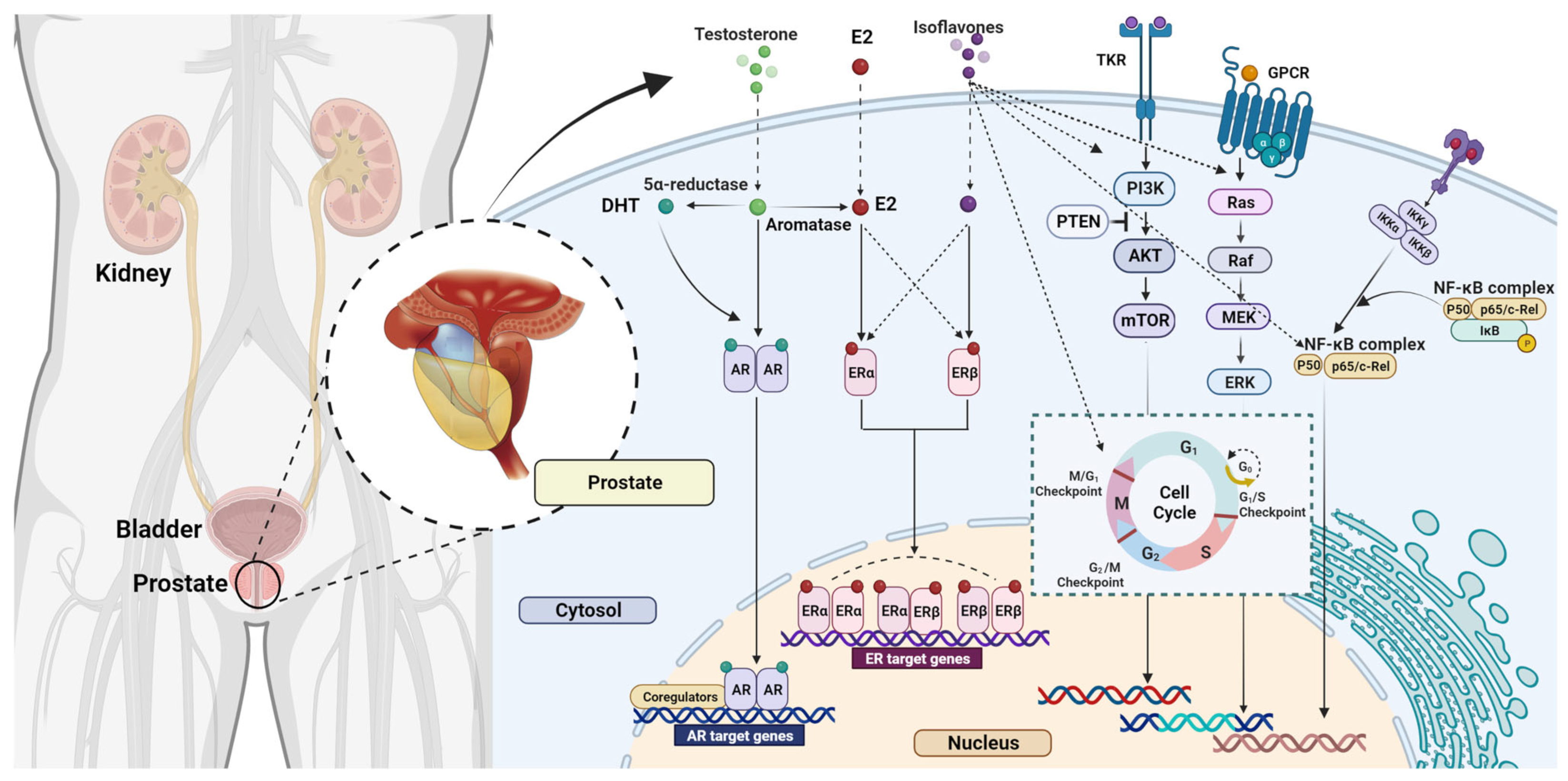
4.2. The Effects of Isoflavones on Prostate Cancer
4.2.1. Animal Studies
4.2.2. Human Studies
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Coffey, D.S. Similarities of prostate and breast cancer: Evolution, diet, and estrogens. Urology 2001, 57, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Teaford, M.F.; Ungar, P.S. Diet and the evolution of the earliest human ancestors. Proc. Natl. Acad. Sci. USA 2000, 97, 13506–13511. [Google Scholar] [CrossRef] [PubMed]
- Go, V.L.W.; Butrum, R.R.; Wong, D.A. Diet, Nutrition, and cancer prevention: The postgenomic era. J. Nutr. 2003, 133, 3830S–3836S. [Google Scholar] [CrossRef] [PubMed]
- Pejčić, T.; Tosti, T.; Džamić, Z.; Gašić, U.; Vuksanović, A.; Dolićanin, Z.; Tešić, Ž. The polyphenols as potential agents in prevention and therapy of prostate diseases. Molecules 2019, 24, 3982. [Google Scholar] [CrossRef]
- Fuentes, N.; Silveyra, P. Estrogen receptor signaling mechanisms. Adv. Protein Chem. Struct. Biol. 2019, 116, 135. [Google Scholar] [CrossRef]
- Hüser, S.; Guth, S.; Joost, H.G.; Soukup, S.T.; Köhrle, J.; Kreienbrock, L.; Diel, P.; Lachenmeier, D.W.; Eisenbrand, G.; Vollmer, G.; et al. Effects of isoflavones on breast tissue and the thyroid hormone system in humans: A comprehensive safety evaluation. Arch. Toxicol. 2018, 92, 2703. [Google Scholar] [CrossRef]
- Paterni, I.; Granchi, C.; Katzenellenbogen, J.A.; Minutolo, F. Estrogen receptors alpha (ERα) and beta (ERβ): Subtype-selective ligands and clinical potential. Steroids 2014, 90, 13–29. [Google Scholar] [CrossRef]
- Kim, I.S. Current perspectives on the beneficial effects of soybean isoflavones and their metabolites for humans. Antioxidants 2021, 10, 1064. [Google Scholar] [CrossRef]
- Szeja, W.; Grynkiewicz, G.; Rusin, A. Isoflavones, their glycosides and glycoconjugates. Synthesis and biological activity. Curr. Org. Chem. 2017, 21, 218–235. [Google Scholar] [CrossRef]
- Leclercq, G.; Jacquot, Y. Interactions of isoflavones and other plant derived estrogens with estrogen receptors for prevention and treatment of breast cancer—considerations concerning related efficacy and safety. J. Steroid Biochem. Mol. Biol. 2014, 139, 237–244. [Google Scholar] [CrossRef]
- Nešović, M.; Gašić, U.; Tosti, T.; Horvacki, N.; Nedić, N.; Sredojević, M.; Blagojević, S.; Ignjatović, L.; Tešić, Ž. Distribution of polyphenolic and sugar compounds in different buckwheat plant parts. RSC Adv. 2021, 11, 25816–25829. [Google Scholar] [CrossRef] [PubMed]
- Pantelić, M.M.; Dabić Zagorac, D.; Davidović, S.M.; Todić, S.R.; Bešlić, Z.S.; Gašić, U.M.; Tešić, Ž.L.; Natić, M.M. Identification and quantification of phenolic compounds in berry skin, pulp, and seeds in 13 grapevine varieties grown in Serbia. Food Chem. 2016, 211, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Fotirić Akšić, M.; Nešović, M.; Ćirić, I.; Tešić, Ž.; Pezo, L.; Tosti, T.; Gašić, U.; Dojčinović, B.; Lončar, B.; Meland, M. Chemical fruit profiles of different raspberry cultivars grown in specific Norwegian agroclimatic conditions. Horticulturae 2022, 8, 765. [Google Scholar] [CrossRef]
- Fotirić Akšić, M.; Nešović, M.; Ćirić, I.; Tešić, Ž.; Pezo, L.; Tosti, T.; Gašić, U.; Dojčinović, B.; Lončar, B.; Meland, M. Polyphenolics and chemical profiles of domestic Norwegian apple (Malus × domestica Borkh.) cultivars. Front. Nutr. 2022, 9, 941487. [Google Scholar] [CrossRef] [PubMed]
- Pantelić, M.; Dabić Zagorac, D.; Natić, M.; Gašić, U.; Jović, S.; Vujović, D.; Popović Djordjević, J. Impact of clonal variability on phenolics and radical scavenging activity of grapes and wines: A study on the recently developed Merlot and Cabernet Franc clones (Vitis vinifera L.). PLoS ONE 2016, 11, e0163823. [Google Scholar] [CrossRef] [PubMed]
- Tešić, Ž.L.; Gašić, U.M.; Milojković-Opsenica, D.M. Chapter 3: Polyphenolic profile of the fruits grown in Serbia. In Advances in Plant Phenolics: From Chemistry to Human Health; Jayaprakasha, K.G., Patil, S.B., Gattuso, G., Eds.; American Chemical Society: Washington, DC, USA, 2018; Volume 1286, pp. 47–66. [Google Scholar] [CrossRef]
- Skrt, M.; Albreht, A.; Vovk, I.; Constantin, O.E.; Râpeanu, G.; Sežun, M.; Osojnik Črnivec, I.G.; Zalar, U.; Poklar Ulrih, N. Extraction of polyphenols and valorization of fibers from Istrian-grown pomegranate (Punica granatum L.). Foods 2022, 11, 2740. [Google Scholar] [CrossRef]
- Simonovska, B.; Vovk, I.; Andrenšek, S.; Valentová, K.; Ulrichová, J. Investigation of phenolic acids in yacon (Smallanthus sonchifolius) leaves and tubers. J. Chromatogr. A 2003, 1016, 89–98. [Google Scholar] [CrossRef]
- Ivanović, M.; Albreht, A.; Krajnc, P.; Vovk, I.; Islamčević Razboršek, M. Sustainable ultrasound-assisted extraction of valuable phenolics from inflorescences of Helichrysum arenarium L. using natural deep eutectic solvents. Ind. Crops Prod. 2021, 160, 113102. [Google Scholar] [CrossRef]
- Orsini, F.; Vovk, I.; Glavnik, V.; Jug, U.; Corradini, D. HPTLC, HPTLC-MS/MS and HPTLC-DPPH methods for analyses of flavonoids and their antioxidant activity in Cyclanthera pedata leaves, fruits and dietary supplement. J. Liq. Chromatogr. Relat. Technol. 2019, 42, 290–301. [Google Scholar] [CrossRef]
- Jug, U.; Glavnik, V.; Kranjc, E.; Vovk, I. High-performance thin-layer chromatography and high-performance thin-layer chromatography–mass spectrometry methods for the analysis of phenolic acids. J. Planar Chromatogr. 2018, 31, 13–22. [Google Scholar] [CrossRef]
- Guzelmeric, E.; Vovk, I.; Yesilada, E. Development and validation of an HPTLC method for apigenin 7-O-glucoside in chamomile flowers and its application for fingerprint discrimination of chamomile-like materials. J. Pharm. Biomed. Anal. 2015, 107, 108–118. [Google Scholar] [CrossRef]
- Nedić, N.; Nešović, M.; Radišić, P.; Gašić, U.; Baošić, R.; Joksimović, K.; Pezo, L.; Tešić, Ž.; Vovk, I. Polyphenolic and chemical profiles of honey from the Tara mountain in Serbia. Front. Nutr. 2022, 9, 941463. [Google Scholar] [CrossRef]
- Bugeja Douglas, A.; Nešović, M.; Šikoparija, B.; Radišić, P.; Tosti, T.; Trifković, J.; Russi, L.; Attard, E.; Tešić, Ž.; Gašić, U. Melissopalynology analysis, determination of physicochemical parameters, sugars and phenolics in Maltese honey collected in different seasons. J. Serbian Chem. Soc. 2022, 87, 983–995. [Google Scholar] [CrossRef]
- Nešović, M.; Gašić, U.; Tosti, T.; Horvacki, N.; Šikoparija, B.; Nedić, N.; Blagojević, S.; Ignjatović, L.; Tešić, Ž. Polyphenol profile of buckwheat honey, nectar and pollen. R. Soc. Open Sci. 2020, 7, 1–15. [Google Scholar] [CrossRef]
- Fraga, C.G.; Croft, K.D.; Kennedy, D.O.; Tomás-Barberán, F.A. The effects of polyphenols and other bioactives on human health. Food Funct. 2019, 10, 514–528. [Google Scholar] [CrossRef]
- Halliwell, B.; Aeshbach, R.; Loliger, J.; Aruoma, O.I. The characterization of antioxidants. Food Chem. Toxicol. 1995, 33, 601–617. [Google Scholar] [CrossRef]
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radic. Biol. Med. 2010, 49, 1603–1616. [Google Scholar] [CrossRef]
- Król-Grzymała, A.; Amarowicz, R. Phenolic compounds of soybean seeds from two European countries and their antioxidant properties. Molecules 2020, 25, 2075. [Google Scholar] [CrossRef]
- Liu, J.; Chang, S.K.C.; Wiesenborn, D. Antioxidant properties of soybean isoflavone extract and tofu in vitro and in vivo. J. Agric. Food Chem. 2005, 53, 2333–2340. [Google Scholar] [CrossRef]
- Choi, E.J.; Kim, G.H. The antioxidant activity of daidzein metabolites, O-desmethylangolensin and equol, in HepG2 cells. Mol. Med. Rep. 2014, 9, 328–332. [Google Scholar] [CrossRef]
- Kalaiselvan, V.; Kalaivani, M.; Vijayakumar, A.; Sureshkumar, K.; Venkateskumar, K. Current knowledge and future direction of research on soy isoflavones as a therapeutic agents. Pharmacogn. Rev. 2010, 4, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, G. The antioxidant role of soy and soy foods in human health. Antioxidants 2020, 9, 635. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, R.K.; Tang, S.-N.; Zhu, W.; Meeker, D.; Shankar, S. Sulforaphane synergizes with quercetin to inhibit self-renewal capacity of pancreatic cancer stem cells. Front. Biosci. (Elite Ed.) 2011, 3, 515–528. [Google Scholar] [CrossRef] [PubMed]
- Izuegbuna, O.O. Polyphenols: Chemoprevention and therapeutic potentials in hematological malignancies. Front. Nutr. 2022, 9, 1008893. [Google Scholar] [CrossRef]
- Rudrapal, M.; Khairnar, S.J.; Khan, J.; Dukhyil, A.B.; Ansari, M.A.; Alomary, M.N.; Alshabrmi, F.M.; Palai, S.; Deb, P.K.; Devi, R. Dietary polyphenols and their role in oxidative stress-induced human diseases: Insights into protective effects, antioxidant potentials and mechanism(s) of action. Front. Pharmacol. 2022, 13, 806470. [Google Scholar] [CrossRef]
- Veitch, N.C. Isoflavonoids of the leguminosae. Nat. Prod. Rep. 2007, 24, 417–464. [Google Scholar] [CrossRef]
- U.S. Department of Agriculture. Agricultural Research Service: 2015. USDA Database for the Isoflavone Content of Selected Foods, Release 2.1; Nutrient Data Laboratory Home Page. Available online: https://data.nal.usda.gov/dataset/usda-database-isoflavone-content-selected-foods-release-21-november-2015/resource/1de757af (accessed on 22 December 2022).
- Setchell, K.D.R.; Maynard Brown, N.; Desai, P.B.; Zimmer-Nechimias, L.; Wolfe, B.; Jakate, A.S.; Creutzinger, V.; Heubi, J.E. Bioavailability, disposition, and dose-response effects of soy isoflavones when consumed by healthy women at physiologically typical dietary intakes. J. Nutr. 2003, 133, 1027–1035. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Rios, E.; Castro, L.; Liu, J.; Yan, Y.; Dixon, D. Genistein: Dual role in women’s health. Nutrients 2021, 13, 3048. [Google Scholar] [CrossRef]
- Selvakumar, P.; Badgeley, A.; Murphy, P.; Anwar, H.; Sharma, U.; Lawrence, K.; Lakshmikuttyamma, A. Flavonoids and other polyphenols act as epigenetic modifiers in breast cancer. Nutrients 2020, 12, 761. [Google Scholar] [CrossRef]
- Frankenfeld, C.L. Cardiometabolic risk and gut microbial phytoestrogen metabolite phenotypes. Mol. Nutr. Food Res. 2017, 61, 1500900. [Google Scholar] [CrossRef]
- Jackson, R.L.; Greiwe, J.S.; Schwen, R.J. Emerging evidence of the health benefits of S-equol, an estrogen receptor β agonist. Nutr. Rev. 2011, 69, 432–448. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, Y.H.; Ho, C.T.; Pan, M.H. Bioavailability and health benefits of major isoflavone aglycones and their metabolites. J. Funct. Foods 2020, 74, 104164. [Google Scholar] [CrossRef]
- Richelle, M.; Pridmore-Merten, S.; Bodenstab, S.; Enslen, M.; Offord, E.A. Hydrolysis of isoflavone glycosides to aglycones by beta-glycosidase does not alter plasma and urine isoflavone pharmacokinetics in postmenopausal women. J. Nutr. 2002, 132, 2587–2592. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Harbeck, N.; Penault-Llorca, F.; Cortes, J.; Gnant, M.; Houssami, N.; Poortmans, P.; Ruddy, K.; Tsang, J.; Cardoso, F. Breast cancer. Nat. Rev. Dis. Prim. 2019, 5, 66. [Google Scholar] [CrossRef]
- Heer, E.; Harper, A.; Escandor, N.; Sung, H.; McCormack, V.; Fidler-Benaoudia, M.M. Global burden and trends in premenopausal and postmenopausal breast cancer: A population-based study. Lancet Glob. Health 2020, 8, e1027–e1037. [Google Scholar] [CrossRef]
- Porter, P. “Westernizing” women’s risk? Breast cancer in lower-income countries. N. Engl. J. Med. 2008, 358, 213–216. [Google Scholar] [CrossRef]
- Ilic, L.; Haidinger, G.; Simon, J.; Hackl, M.; Schernhammer, E.; Papantoniou, K. Trends in female breast cancer incidence, mortality, and survival in Austria, with focus on age, stage, and birth cohorts (1983–2017). Sci. Rep. 2022, 12, 1–10. [Google Scholar] [CrossRef]
- Becker, S. A historic and scientific review of breast cancer: The next global healthcare challenge. Int. J. Gynecol. Obstet. 2015, 131, S36–S39. [Google Scholar] [CrossRef]
- Rositch, A.F.; Unger-Saldaña, K.; DeBoer, R.J.; Ng’ang’a, A.; Weiner, B.J. The role of dissemination and implementation science in global breast cancer control programs: Frameworks, methods, and examples. Cancer 2020, 126 (Suppl. 10), 2394–2404. [Google Scholar] [CrossRef]
- Duggan, C.; Dvaladze, A.; Rositch, A.F.; Ginsburg, O.; Yip, C.H.; Horton, S.; Camacho Rodriguez, R.; Eniu, A.; Mutebi, M.; Bourque, J.M.; et al. The Breast Health Global Initiative 2018 Global Summit on Improving Breast Healthcare Through Resource-Stratified Phased Implementation: Methods and overview. Cancer 2020, 126, 2339–2352. [Google Scholar] [CrossRef]
- Prat, A.; Perou, C.M. Deconstructing the molecular portraits of breast cancer. Mol. Oncol. 2011, 5, 5–23. [Google Scholar] [CrossRef]
- Polyak, K. Heterogeneity in breast cancer. J. Clin. Investig. 2011, 121, 3786. [Google Scholar] [CrossRef]
- Loibl, S.; Poortmans, P.; Morrow, M.; Denkert, C.; Curigliano, G. Breast cancer. Lancet 2021, 397, 1750–1769. [Google Scholar] [CrossRef]
- Bianchini, G.; De Angelis, C.; Licata, L.; Gianni, L. Treatment landscape of triple-negative breast cancer—Expanded options, evolving needs. Nat. Rev. Clin. Oncol. 2022, 19, 91–113. [Google Scholar] [CrossRef]
- Łukasiewicz, S.; Czeczelewski, M.; Forma, A.; Baj, J.; Sitarz, R.; Stanisławek, A. Breast cancer—Epidemiology, risk factors, classification, prognostic markers, and current treatment strategies—An updated review. Cancers 2021, 13, 4287. [Google Scholar] [CrossRef]
- Giordano, S.H. Breast cancer in men. N. Engl. J. Med. 2018, 378, 2311–2320. [Google Scholar] [CrossRef]
- McGuire, A.; Brown, J.A.L.; Malone, C.; McLaughlin, R.; Kerin, M.J. Effects of age on the detection and management of breast cancer. Cancers 2015, 7, 908–929. [Google Scholar] [CrossRef]
- Shiyanbola, O.O.; Arao, R.F.; Miglioretti, D.L.; Sprague, B.L.; Hampton, J.M.; Stout, N.K.; Kerlikowske, K.; Braithwaite, D.; Buist, D.S.M.; Egan, K.M.; et al. Emerging trends in family history of breast cancer and associated risk. Cancer Epidemiol. Biomarkers Prev. 2017, 26, 1753–1760. [Google Scholar] [CrossRef]
- Wendt, C.; Margolin, S. Identifying breast cancer susceptibility genes—A review of the genetic background in familial breast cancer. Acta Oncol. 2019, 58, 135–146. [Google Scholar] [CrossRef]
- Sun, Y.S.; Zhao, Z.; Yang, Z.N.; Xu, F.; Lu, H.J.; Zhu, Z.Y.; Shi, W.; Jiang, J.; Yao, P.P.; Zhu, H.P. Risk factors and preventions of breast cancer. Int. J. Biol. Sci. 2017, 13, 1387. [Google Scholar] [CrossRef] [PubMed]
- Daly, A.A.; Rolph, R.; Cutress, R.I.; Copson, E.R. A Review of modifiable risk factors in young women for the prevention of breast cancer. Breast Cancer 2021, 13, 241. [Google Scholar] [CrossRef]
- Steward, W.P.; Brown, K. Cancer chemoprevention: A rapidly evolving field. Br. J. Cancer 2013, 109, 1–7. [Google Scholar] [CrossRef] [PubMed]
- George, B.P.; Chandran, R.; Abrahamse, H. Role of phytochemicals in cancer chemoprevention: Insights. Antioxidants 2021, 10, 1455. [Google Scholar] [CrossRef]
- Mohan Shankar, G.; Swetha, M.; Keerthana, C.K.; Rayginia, T.P.; Anto, R.J. Cancer chemoprevention: A strategic approach using phytochemicals. Front. Pharmacol. 2022, 12, 4044. [Google Scholar] [CrossRef]
- Chen, C.; Kong, A.-N.T. Dietary cancer-chemopreventive compounds: From signaling and gene expression to pharmacological effects. Trends Pharmacol. Sci. 2005, 26, 318–326. [Google Scholar] [CrossRef]
- Pabich, M.; Materska, M. Biological effect of soy isoflavones in the prevention of civilization diseases. Nutrients 2019, 11, 1660. [Google Scholar] [CrossRef]
- Finkeldey, L.; Schmitz, E.; Ellinger, S. Effect of the intake of isoflavones on risk factors of breast cancer—A systematic review of randomized controlled intervention studies. Nutrients 2021, 13, 2309. [Google Scholar] [CrossRef]
- Rizzo, G.; Baroni, L. Soy, soy foods and their role in vegetarian diets. Nutrients 2018, 10, 43. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Knaze, V.; Luján-Barroso, L.; Kuhnle, G.G.C.; Mulligan, A.A.; Touillaud, M.; Slimani, N.; Romieu, I.; Powell, N.; Tumino, R.; et al. Dietary intakes and food sources of phytoestrogens in the European Prospective Investigation into Cancer and Nutrition (EPIC) 24-hour dietary recall cohort. Eur. J. Clin. Nutr. 2012, 66, 932–941. [Google Scholar] [CrossRef]
- Mulligan, A.A.; Welch, A.A.; McTaggart, A.A.; Bhaniani, A.; Bingham, S.A. Intakes and sources of soya foods and isoflavones in a UK population cohort study (EPIC-Norfolk). Eur. J. Clin. Nutr. 2007, 61, 248–254. [Google Scholar] [CrossRef]
- Mortensen, A.; Kulling, S.E.; Schwartz, H.; Rowland, I.; Ruefer, C.E.; Rimbach, G.; Cassidy, A.; Magee, P.; Millar, J.; Hall, W.L.; et al. Analytical and compositional aspects of isoflavones in food and their biological effects. Mol. Nutr. Food Res. 2009, 53 (Suppl. 2), S266–S309. [Google Scholar] [CrossRef]
- Ziaei, S.; Halaby, R.; Vinjamury, S.; Sommers, E. Dietary isoflavones and breast cancer risk. Medicines 2017, 4, 18. [Google Scholar] [CrossRef]
- Xie, Q.; Chen, M.L.; Qin, Y.; Zhang, Q.Y.; Xu, H.X.; Zhou, Y.; Mi, M.T.; Zhu, J.D. Isoflavone consumption and risk of breast cancer: A dose-response meta-analysis of observational studies. Asia Pac. J. Clin. Nutr. 2013, 22, 118–127. [Google Scholar] [CrossRef]
- Qin, L.Q.; Xu, J.Y.; Wang, P.Y.; Hoshi, K. Soyfood intake in the prevention of breast cancer risk in women: A meta-analysis of observational epidemiological studies. J. Nutr. Sci. Vitaminol. 2006, 52, 428–436. [Google Scholar] [CrossRef]
- Chen, M.; Rao, Y.; Zheng, Y.; Wei, S.; Li, Y.; Guo, T.; Yin, P. Association between soy isoflavone intake and breast cancer risk for pre- and post-menopausal women: A meta-analysis of epidemiological studies. PLoS ONE 2014, 9, e89288. [Google Scholar] [CrossRef]
- Wu, A.H.; Yu, M.C.; Tseng, C.C.; Pike, M.C. Epidemiology of soy exposures and breast cancer risk. Br. J. Cancer 2008, 98, 9. [Google Scholar] [CrossRef]
- Wei, Y.; Lv, J.; Guo, Y.; Bian, Z.; Gao, M.; Du, H.; Yang, L.; Chen, Y.; Zhang, X.; Wang, T.; et al. Soy intake and breast cancer risk: A prospective study of 300,000 Chinese women and a dose–response meta-analysis. Eur. J. Epidemiol. 2020, 35, 567–578. [Google Scholar] [CrossRef]
- Dong, J.Y.; Qin, L.Q. Soy isoflavones consumption and risk of breast cancer incidence or recurrence: A meta-analysis of prospective studies. Breast Cancer Res. Treat. 2011, 125, 315–323. [Google Scholar] [CrossRef]
- Boutas, I.; Kontogeorgi, A.; Dimitrakakis, C.; Kalantaridou, S.N. Soy isoflavones and breast cancer risk: A meta-analysis. In Vivo 2022, 36, 556–562. [Google Scholar] [CrossRef]
- Qiu, S.; Jiang, C. Soy and isoflavones consumption and breast cancer survival and recurrence: A systematic review and meta-analysis. Eur. J. Nutr. 2019, 58, 3079–3090. [Google Scholar] [CrossRef] [PubMed]
- Dai, Q.; Shu, X.O.; Jin, F.; Potter, J.D.; Kushi, L.H.; Teas, J.; Gao, Y.T.; Zheng, W. Population-based case-control study of soyfood intake and breast cancer risk in Shanghai. Br. J. Cancer 2001, 85, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Matsuo, K.; Tsunoda, N.; Hirose, K.; Hiraki, A.; Kawase, T.; Yamashita, T.; Iwata, H.; Tanaka, H.; Tajima, K. Effect of soybean on breast cancer according to receptor status: A case-control study in Japan. Int. J. Cancer 2008, 123, 1674–1680. [Google Scholar] [CrossRef]
- Baglia, M.L.; Zheng, W.; Li, H.; Yang, G.; Gao, J.; Gao, Y.T.; Shu, X.O. The association of soy food consumption with the risk of subtype of breast cancers defined by hormone receptor and HER2 status. Int. J. Cancer 2016, 139, 742–748. [Google Scholar] [CrossRef]
- Sim, E.J.; Ko, K.P.; Ahn, C.; Park, S.M.; Surh, Y.J.; An, S.; Kim, S.W.; Lee, M.H.; Lee, J.W.; Lee, J.E.; et al. Isoflavone intake on the risk of overall breast cancer and molecular subtypes in women at high risk for hereditary breast cancer. Breast Cancer Res. Treat. 2020, 184, 615–626. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.V.; Agatonovic-Kustrin, S.; Glass, B.D. Molecular aspects of phytoestrogen selective binding at estrogen receptors. J. Pharm. Sci. 2007, 96, 1879–1885. [Google Scholar] [CrossRef]
- Mukherjee, S.; Mukherjee, A.; Saha, A. QSAR modeling on binding affinity of diverse estrogenic flavonoids: Electronic, topological and spatial functions in quantitative approximation. J. Mol. Struct. Theochem. 2005, 715, 85–90. [Google Scholar] [CrossRef]
- Stubert, J.; Gerber, B. Isoflavones—Mechanism of action and impact on breast cancer risk. Breast Care 2009, 4, 22–29. [Google Scholar] [CrossRef]
- Russo, M.; Russo, G.L.; Daglia, M.; Kasi, P.D.; Ravi, S.; Nabavi, S.F.; Nabavi, S.M. Understanding genistein in cancer: The “good” and the “bad” effects: A review. Food Chem. 2016, 196, 589–600. [Google Scholar] [CrossRef]
- Van Duursen, M.B.M. Modulation of estrogen synthesis and metabolism by phytoestrogens in vitro and the implications for women’s health. Toxicol. Res. (Camb.) 2017, 6, 772–794. [Google Scholar] [CrossRef]
- Liu, R.; Yu, X.; Chen, X.; Zhong, H.; Liang, C.; Xu, X.; Xu, W.; Cheng, Y.; Wang, W.; Yu, L.; et al. Individual factors define the overall effects of dietary genistein exposure on breast cancer patients. Nutr. Res. 2019, 67, 1–16. [Google Scholar] [CrossRef]
- Hsieh, C.-J.; Hsu, Y.-L.; Huang, Y.-F.; Tsai, E.-M. Molecular mechanisms of anticancer effects of phytoestrogens in breast cancer. Curr. Protein Pept. Sci. 2018, 19, 323–332. [Google Scholar] [CrossRef]
- Allred, C.D.; Allred, K.F.; Ju, Y.H.; Virant, S.M.; Helferich, W.G. Soy diets containing varying amounts of genistein stimulate growth of estrogen-dependent (MCF-7) tumors in a dose-dependent manner. Cancer Res. 2001, 61, 5045–5050. [Google Scholar]
- Ahn, S.Y.; Jo, M.S.; Lee, D.; Baek, S.E.; Baek, J.; Yu, J.S.; Jo, J.; Yun, H.; Kang, K.S.; Yoo, J.E.; et al. Dual effects of isoflavonoids from Pueraria lobata roots on estrogenic activity and anti-proliferation of MCF-7 human breast carcinoma cells. Bioorg. Chem. 2019, 83, 135–144. [Google Scholar] [CrossRef]
- Li, Z.; Li, J.; Mo, B.; Hu, C.; Liu, H.; Qi, H.; Wang, X.; Xu, J. Genistein induces cell apoptosis in MDA-MB-231 breast cancer cells via the mitogen-activated protein kinase pathway. Toxicol. Vitro 2008, 22, 1749–1753. [Google Scholar] [CrossRef]
- Yu, X.; Zhu, J.; Mi, M.; Chen, W.; Pan, Q.; Wei, M. Anti-angiogenic genistein inhibits VEGF-induced endothelial cell activation by decreasing PTK activity and MAPK activation. Med. Oncol. 2012, 29, 349–357. [Google Scholar] [CrossRef]
- Zhang, X.; Cook, K.L.; Warri, A.; Cruz, I.M.; Rosim, M.; Riskin, J.; Helferich, W.; Doerge, D.; Clarke, R.; Hilakivi-Clarke, L. Lifetime genistein intake increases the response of mammary tumors to tamoxifen in rats. Clin. Cancer Res. 2017, 23, 814–824. [Google Scholar] [CrossRef]
- Li, Y.; Meeran, S.M.; Patel, S.N.; Chen, H.; Hardy, T.M.; Tollefsbol, T.O. Epigenetic reactivation of estrogen receptor-α (ERα) by genistein enhances hormonal therapy sensitivity in ERα-negative breast cancer. Mol. Cancer 2013, 12, 9. [Google Scholar] [CrossRef]
- Lee, K.L.; Peehl, D.M. Molecular and cellular pathogenesis of benign prostatic hyperplasia. J. Urol. 2004, 172, 1784–1791. [Google Scholar] [CrossRef]
- Pejčić, T.; Tosti, T.; Tešić, Ž.; Milković, B.; Dragičević, D.; Kozomara, M.; Čekerevac, M.; Džamić, Z. Testosterone and dihydrotestosterone levels in the transition zone correlate with prostate volume. Prostate 2017, 77, 1082–1092. [Google Scholar] [CrossRef]
- Sandhu, S.; Moore, C.M.; Chiong, E.; Beltran, H.; Bristow, R.G.; Williams, S.G. Prostate cancer. Lancet 2021, 398, 1075–1090. [Google Scholar] [CrossRef]
- De Marzo, A.M.; DeWeese, T.L.; Platz, E.A.; Meeker, A.K.; Nakayama, M.; Epstein, J.I.; Isaacs, W.B.; Nelson, W.G. Pathological and molecular mechanisms of prostate carcinogenesis: Implications for diagnosis, detection, prevention, and treatment. J. Cell. Biochem. 2004, 91, 459–477. [Google Scholar] [CrossRef]
- Ali, A.; Du Feu, A.; Oliveira, P.; Choudhury, A.; Bristow, R.G.; Baena, E. Prostate zones and cancer: Lost in transition? Nat. Rev. Urol. 2022, 19, 101–115. [Google Scholar] [CrossRef]
- Stephenson, A.; Klein, A. Epidemiology, etiology, and prevention of prostate cancer. In Campbell- Walsh Urology, 12th ed.; Wein, A., Kavouss, L., Partin, A., Peters, C., Eds.; Elsevier: Philadelphia, PA, USA, 2016; pp. 2543–2564. [Google Scholar]
- Gandaglia, G.; Leni, R.; Bray, F.; Fleshner, N.; Freedland, S.J.; Kibel, A.; Stattin, P.; Van Poppel, H.; La Vecchia, C. Epidemiology and prevention of prostate cancer. Eur. Urol. Oncol. 2021, 4, 877–892. [Google Scholar] [CrossRef]
- Patel, A.R.; Klein, E.A. Risk factors for prostate cancer. Nat. Clin. Pract. Urol. 2009, 6, 87–95. [Google Scholar] [CrossRef]
- Bostwick, D.G.; Burke, H.B.; Djakiew, D.; Euling, S.; Ho, S.M.; Landolph, J.; Morrison, H.; Sonawane, B.; Shifflett, T.; Waters, D.J.; et al. Human prostate cancer risk factors. Cancer 2004, 101, 2371–2490. [Google Scholar] [CrossRef]
- Gann, P.H. Risk factors for prostate cancer. Rev. Urol. 2002, 4 (Suppl. 5), S3. [Google Scholar]
- Carter, B.S.; Beaty, T.H.; Steinberg, G.D.; Childs, B.; Walsh, P.C. Mendelian inheritance of familial prostate cancer. Proc. Natl. Acad. Sci. USA 1992, 89, 3367–3371. [Google Scholar] [CrossRef]
- Giri, V.N.; Beebe-Dimmer, J.L. Familial prostate cancer. Semin. Oncol. 2016, 43, 560–565. [Google Scholar] [CrossRef]
- Zeegers, M.P.A.; Jellema, A.; Ostrer, H. Empiric risk of prostate carcinoma for relatives of patients with prostate carcinoma: A meta-analysis. Cancer 2003, 97, 1894–1903. [Google Scholar] [CrossRef]
- Yu, H.; Harris, R.E.; Gao, Y.T.; Gao, R.; Wynder, E.L. Comparative epidemiology of cancers of the colon, rectum, prostate and breast in Shanghai, China versus the United States. Int. J. Epidemiol. 1991, 20, 76–81. [Google Scholar] [CrossRef]
- Kimura, T. East meets West: Ethnic differences in prostate cancer epidemiology between East Asians and Caucasians. Chin. J. Cancer 2012, 31, 421–429. [Google Scholar] [CrossRef]
- Shimizu, H.; Ross, R.K.; Bernstein, L.; Henderson, B.E.; Mack, T.M.; Yatani, R. Cancers of the prostate and breast among Japanese and white immigrants in Los Angeles county. Br. J. Cancer 1991, 63, 963–966. [Google Scholar] [CrossRef]
- Zhang, J.; Dhakal, I.B.; Zhao, Z.; Li, L. Trends in mortality from cancers of the breast, colon, prostate, esophagus, and stomach in East Asia: Role of nutrition transition. Eur. J. Cancer Prev. 2012, 21, 480–489. [Google Scholar] [CrossRef]
- De Martel, C.; Ferlay, J.; Franceschi, S.; Vignat, J.; Bray, F.; Forman, D.; Plummer, M. Global burden of cancers attributable to infections in 2008: A review and synthetic analysis. Lancet. Oncol. 2012, 13, 607–615. [Google Scholar] [CrossRef]
- Giovannucci, E. The epidemiology of vitamin D and cancer incidence and mortality: A review (United States). Cancer Causes Control 2005, 16, 83–95. [Google Scholar] [CrossRef]
- Robles, L.A.; Harrison, S.; Tan, V.Y.; Beynon, R.; McAleenan, A.; Higgins, J.P.; Martin, R.M.; Lewis, S.J. Does testosterone mediate the relationship between vitamin D and prostate cancer progression? A systematic review and meta-analysis. Cancer Causes Control 2022, 33, 1025–1038. [Google Scholar] [CrossRef]
- Krill, D.; DeFlavia, P.; Dhir, R.; Luo, J.; Becich, M.J.; Lehman, E.; Getzenberg, R.H. Expression patterns of vitamin D receptor in human prostate. J. Cell. Biochem. 2001, 82, 566–572. [Google Scholar] [CrossRef]
- Schwartz, G.G. Vitamin D and the epidemiology of prostate cancer. Semin. Dial. 2005, 18, 276–289. [Google Scholar] [CrossRef]
- Galor, O.; Moav, O.; The Neolithic Revolution and Contemporary Variations in Life Expectancy, Working Paper, No. 2007-14; Brown University, Department of Economics, 2007, Providence, RI. Available online: https://www.econstor.eu/handle/10419/80105 (accessed on 22 December 2022).
- Bell, K.J.L.; Del Mar, C.; Wright, G.; Dickinson, J.; Glasziou, P. Prevalence of incidental prostate cancer: A systematic review of autopsy studies. Int. J. Cancer 2015, 137, 1749–1757. [Google Scholar] [CrossRef]
- Tzenios, N.; Tazanios, M.E.; Chahine, M. The impact of body mass index on prostate cancer: An updated systematic review and meta-analysis. Medicine 2022, 101, e30191. [Google Scholar] [CrossRef] [PubMed]
- Wright, M.E.; Chang, S.C.; Schatzkin, A.; Albanes, D.; Kipnis, V.; Mouw, T.; Hurwitz, P.; Hollenbeck, A.; Leitzmann, M.F. Prospective study of adiposity and weight change in relation to prostate cancer incidence and mortality. Cancer 2007, 109, 675–684. [Google Scholar] [CrossRef]
- Zhong, S.; Yan, X.; Wu, Y.; Zhang, X.; Chen, L.; Tang, J.; Zhao, J. Body mass index and mortality in prostate cancer patients: A dose-response meta-analysis. Prostate Cancer Prostatic Dis. 2016, 19, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Bonn, S.E.; Wiklund, F.; Sjölander, A.; Szulkin, R.; Stattin, P.; Holmberg, E.; Grönberg, H.; Bälter, K. Body mass index and weight change in men with prostate cancer: Progression and mortality. Cancer Causes Control 2014, 25, 933–943. [Google Scholar] [CrossRef] [PubMed]
- Bassett, J.K.; Severi, G.; Baglietto, L.; MacInnis, R.J.; Hoang, H.N.; Hopper, J.L.; English, D.R.; Giles, G.G. Weight change and prostate cancer incidence and mortality. Int. J. Cancer 2012, 131, 1711–1719. [Google Scholar] [CrossRef]
- Buschemeyer, W.C.; Freedland, S.J. Obesity and prostate cancer: Epidemiology and clinical implications. Eur. Urol. 2007, 52, 331–343. [Google Scholar] [CrossRef]
- Peterson, G.; Barnes, S. Genistein and biochanin A inhibit the growth of human prostate cancer cells but not epidermal growth factor receptor tyrosine autophosphorylation. Prostate 1993, 22, 335–345. [Google Scholar] [CrossRef]
- Davis, J.N.; Singh, B.; Bhuiyan, M.; Sarkar, F.H. Genistein-induced upregulation of p21WAF1, downregulation of cyclin B, and induction of apoptosis in prostate cancer cells. Nutr. Cancer 1998, 32, 123–131. [Google Scholar] [CrossRef]
- Geller, J.; Sionit, L.; Partido, C.; Li, L.; Tan, X.; Youngkin, T.; Nachtsheim, D.; Hoffman, R.M. Genistein inhibits the growth of human-patient bph and prostate cancer in histoculture. Prostate 1998, 34, 75–79. [Google Scholar] [CrossRef]
- Ji, X.; Liu, K.; Li, Q.; Shen, Q.; Han, F.; Ye, Q.; Zheng, C. A mini-review of flavone isomers apigenin and genistein in prostate cancer treatment. Front. Pharmacol. 2022, 13, 851589. [Google Scholar] [CrossRef]
- Kyle, E.; Neckers, L.; Takimoto, C.; Curt, G.; Bergan, R. Genistein-induced apoptosis of prostate cancer cells is preceded by a specific decrease in focal adhesion kinase activity. Mol. Pharmacol. 1997, 51, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Santibáñez, J.F.; Navarro, A.; Martínez, J. Genistein inhibits proliferation and in vitro invasive potential of human prostatic cancer cell lines. Anticancer Res. 1997, 17, 1199–1204. [Google Scholar] [PubMed]
- Chae, H.S.; Xu, R.; Won, J.Y.; Chin, Y.W.; Yim, H. Molecular targets of genistein and its related flavonoids to exert anticancer effects. Int. J. Mol. Sci. 2019, 20, 2420. [Google Scholar] [CrossRef]
- Zhang, L.L.; Li, L.; Wu, D.P.; Fan, J.H.; Li, X.; Wu, K.J.; Wang, X.Y.; He, D.L. A novel anti-cancer effect of genistein: Reversal of epithelial mesenchymal transition in prostate cancer cells. Acta Pharmacol. Sin. 2008, 29, 1060–1068. [Google Scholar] [CrossRef]
- Rao, A.; Woodruff, R.D.; Wade, W.N.; Kute, T.E.; Cramer, S.D. Genistein and vitamin D synergistically inhibit human prostatic epithelial cell growth. J. Nutr. 2002, 132, 3191–3194. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Blackburn, G.L.; Zhou, J.-R. Genistein and daidzein downregulate prostate androgen-regulated transcript-1 (PART-1) gene expression induced by dihydrotestosterone in human prostate LNCaP cancer cells. J. Nutr. 2003, 133, 389–392. [Google Scholar] [CrossRef] [PubMed]
- Shenouda, N.S.; Zhou, C.; Browning, J.D.; Ansell, P.J.; Sakla, M.S.; Lubahn, D.B.; MacDonald, R.S. Phytoestrogens in common herbs regulate prostate cancer cell growth in vitro. Nutr. Cancer 2004, 49, 200–208. [Google Scholar] [CrossRef]
- Shen, J.C.; Klein, R.D.; Wei, Q.; Guan, Y.; Contois, J.H.; Wang, T.T.Y.; Chang, S.; Hursting, S.D. Low-dose genistein induces cyclin-dependent kinase inhibitors and G1 cell-cycle arrest in human prostate cancer cells. Mol. Carcinog. 2000, 29, 92–102. [Google Scholar] [CrossRef]
- Ouchi, H.; Ishiguro, H.; Ikeda, N.; Hori, M.; Kubota, Y.; Uemura, H. Genistein induces cell growth inhibition in prostate cancer through the suppression of telomerase activity. Int. J. Urol. 2005, 12, 73–80. [Google Scholar] [CrossRef]
- Li, Y.; Ahmed, F.; Ali, S.; Philip, P.A.; Kucuk, O.; Sarkar, F.H. Inactivation of nuclear factor kappaB by soy isoflavone genistein contributes to increased apoptosis induced by chemotherapeutic agents in human cancer cells. Cancer Res. 2005, 65, 6934–6942. [Google Scholar] [CrossRef]
- Kousidou, O.C.; Mitropoulou, T.N.; Roussidis, A.E.; Kletsas, D.; Theocharis, A.D.; Karamanos, N.K. Genistein suppresses the invasive potential of human breast cancer cells through transcriptional regulation of metalloproteinases and their tissue inhibitors. Int. J. Oncol. 2005, 26, 1101–1109. [Google Scholar] [CrossRef] [PubMed]
- Bektic, J.; Berger, A.P.; Pfeil, K.; Dobler, G.; Bartsch, G.; Klocker, H. Androgen receptor regulation by physiological concentrations of the isoflavonoid genistein in androgen-dependent LNCaP cells is mediated by estrogen receptor β. Eur. Urol. 2004, 45, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Chiyomaru, T.; Yamamura, S.; Fukuhara, S.; Yoshino, H.; Kinoshita, T.; Majid, S.; Saini, S.; Chang, I.; Tanaka, Y.; Enokida, H.; et al. Genistein inhibits prostate cancer cell growth by targeting miR-34a and oncogenic HOTAIR. PLoS ONE 2013, 8, e70372. [Google Scholar] [CrossRef]
- Chiyomaru, T.; Yamamura, S.; Fukuhara, S.; Hidaka, H.; Majid, S.; Saini, S.; Arora, S.; Deng, G.; Shahryari, V.; Chang, I.; et al. Genistein up-regulates tumor suppressor microRNA-574-3p in prostate cancer. PLoS ONE 2013, 8, e58929. [Google Scholar] [CrossRef] [PubMed]
- Thelen, P.; Wuttke, W.; Seidlová-Wuttke, D. Phytoestrogens selective for the estrogen receptor beta exert anti-androgenic effects in castration resistant prostate cancer. J. Steroid Biochem. Mol. Biol. 2014, 139, 290–293. [Google Scholar] [CrossRef] [PubMed]
- Bonkhoff, H.; Berges, R. The evolving role of oestrogens and their receptors in the development and progression of prostate cancer. Eur. Urol. 2009, 55, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Pollak, M. The insulin and insulin-like growth factor receptor family in neoplasia: An update. Nat. Rev. Cancer 2012, 12, 159–169. [Google Scholar] [CrossRef]
- Spagnuolo, C.; Russo, G.L.; Orhan, I.E.; Habtemariam, S.; Daglia, M.; Sureda, A.; Nabavi, S.F.; Devi, K.P.; Loizzo, M.R.; Tundis, R.; et al. Genistein and cancer: Current status, challenges, and future directions. Adv. Nutr. 2015, 6, 408–419. [Google Scholar] [CrossRef] [PubMed]
- Dalu, A.; Haskell, J.F.; Coward, L.; Lamartiniere, C.A. Genistein, a component of soy, inhibits the expression of the EGF and ErbB2/Neu receptors in the rat dorsolateral prostate. Prostate 1998, 37, 36–43. [Google Scholar] [CrossRef]
- Kim, H.; Peterson, T.G.; Barnes, S. Mechanisms of action of the soy isoflavone genistein: Emerging role for its effects via transforming growth factor beta signaling pathways. Am. J. Clin. Nutr. 1998, 68 (Suppl. 6), 1418S–1425S. [Google Scholar] [CrossRef]
- Cai, L.Q.; Cai, J.; Wu, W.; Zhu, Y.S. 17α-Estradiol and genistein inhibit high fat diet induced prostate gene expression and prostate growth in the rat. J. Urol. 2011, 186, 1489–1496. [Google Scholar] [CrossRef]
- Pollard, M.; Wolter, W. Prevention of spontaneous prostate-related cancer in Lobund-Wistar rats by a soy protein isolate/isoflavone diet. Prostate 2000, 45, 101–105. [Google Scholar] [CrossRef]
- Mentor-Marcel, R.; Lamartiniere, C.A.; Eltoum, I.E.; Greenberg, N.M.; Elgavish, A. Genistein in the diet reduces the incidence of poorly differentiated prostatic adenocarcinoma in transgenic mice (TRAMP). Cancer Res. 2001, 61, 6777–6782. [Google Scholar]
- Wang, J.; Eltoum, I.E.; Lamartiniere, C.A. Genistein chemoprevention of prostate cancer in TRAMP mice. J. Carcinog. 2007, 6, 3. [Google Scholar] [CrossRef]
- Schleicher, R.L.; Lamartiniere, C.A.; Zheng, M.; Zhang, M. The inhibitory effect of genistein on the growth and metastasis of a transplantable rat accessory sex gland carcinoma. Cancer Lett. 1999, 136, 195–201. [Google Scholar] [CrossRef]
- Pavese, J.M.; Farmer, R.L.; Bergan, R.C. Inhibition of cancer cell invasion and metastasis by genistein. Cancer Metastasis Rev. 2010, 29, 465–482. [Google Scholar] [CrossRef]
- Pavese, J.M.; Krishna, S.N.; Bergan, R.C. Genistein inhibits human prostate cancer cell detachment, invasion, and metastasis. Am. J. Clin. Nutr. 2014, 100, 431S–436S. [Google Scholar] [CrossRef]
- Fritz, W.A.; Wang, J.; Eltoum, I.E.; Lamartiniere, C.A. Dietary genistein down-regulates androgen and estrogen receptor expression in the rat prostate. Mol. Cell. Endocrinol. 2002, 186, 89–99. [Google Scholar] [CrossRef]
- Sĺusarz, A.; Jackson, G.A.; Day, J.K.; Shenouda, N.S.; Bogener, J.L.; Browning, J.D.; Fritsche, K.L.; MacDonald, R.S.; Besch-Williford, C.L.; Lubahn, D.B. Aggressive prostate cancer is prevented in ERαKO mice and stimulated in ERβKO TRAMP mice. Endocrinology 2012, 153, 4160–4170. [Google Scholar] [CrossRef]
- Lu, L.-J.; Anderson, K.E.; Grady, J.J.; Nagamani, M. Effects of soya consumption for one month on steroid hormones in premenopausal women: Implications for breast cancer risk reduction. Cancer Epidemiol. Biomarkers Prev. 1996, 5, 63–70. [Google Scholar]
- Busby, M.G.; Jeffcoat, A.R.; Bloedon, L.A.T.; Koch, M.A.; Black, T.; Dix, K.J.; Heizer, W.D.; Thomas, B.F.; Hill, J.M.; Crowell, J.A.; et al. Clinical characteristics and pharmacokinetics of purified soy isoflavones: Single-dose administration to healthy men. Am. J. Clin. Nutr. 2002, 75, 126–136. [Google Scholar] [CrossRef]
- Hong, S.J.; Kim, S.I.; Kwon, S.M.; Lee, J.R.; Chung, B.C. Comparative study of concentration of isoflavones and lignans in plasma and prostatic tissues of normal control and benign prostatic hyperplasia. Yonsei Med. J. 2002, 43, 236–241. [Google Scholar] [CrossRef]
- Adams, K.F.; Chen, C.; Newton, K.M.; Potter, J.D.; Lampe, J.W. Soy isoflavones do not modulate prostate-specific antigen concentrations in older men in a randomized controlled trial. Cancer Epidemiol. Biomarkers Prev. 2004, 13, 644–648. [Google Scholar] [CrossRef]
- Goetzl, M.A.; VanVeldhuizen, P.J.; Thrasher, J.B. Effects of soy phytoestrogens on the prostate. Prostate Cancer Prostatic Dis. 2007, 10, 216–223. [Google Scholar] [CrossRef]
- Yan, L.; Spitznagel, E.L. Soy consumption and prostate cancer risk in men: A revisit of a meta-analysis. Am. J. Clin. Nutr. 2009, 89, 1155–1163. [Google Scholar] [CrossRef]
- Andres, S.; Abraham, K.; Appel, K.E.; Lampen, A. Risks and benefits of dietary isoflavones for cancer. Crit. Rev. Toxicol. 2011, 41, 463–506. [Google Scholar] [CrossRef]
- Hebert, J.R.; Hurley, T.G.; Olendzki, B.C.; Teas, J.; Ma, Y.; Hampl, J.S. Nutritional and socioeconomic factors in relation to prostate cancer mortality: A cross-national study. J. Natl. Cancer Inst. 1998, 90, 1637–1647. [Google Scholar] [CrossRef]
- Perabo, F.G.E.; Von Löw, E.C.; Ellinger, J.; von Rücker, A.; Müller, S.C.; Bastian, P.J. Soy isoflavone genistein in prevention and treatment of prostate cancer. Prostate Cancer Prostatic Dis. 2008, 11, 6–12. [Google Scholar] [CrossRef]
- Van Die, M.D.; Bone, K.M.; Williams, S.G.; Pirotta, M.V. Soy and soy isoflavones in prostate cancer: A systematic review and meta-analysis of randomized controlled trials. BJU Int. 2014, 113, E119–E130. [Google Scholar] [CrossRef]
- He, J.; Wang, S.; Zhou, M.; Yu, W.; Zhang, Y.; He, X. Phytoestrogens and risk of prostate cancer: A meta-analysis of observational studies. World J. Surg. Oncol. 2015, 13, 231. [Google Scholar] [CrossRef]
- Russo, G.I.; Di Mauro, M.; Regis, F.; Reale, G.; Campisi, D.; Marranzano, M.; Lo Giudice, A.; Solinas, T.; Madonia, M.; Cimino, S.; et al. Association between dietary phytoestrogens intakes and prostate cancer risk in Sicily. Aging Male 2018, 21, 48–54. [Google Scholar] [CrossRef]
- Jarred, R.A.; Keikha, M.; Dowling, C.; McPherson, S.J.; Clare, A.M.; Husband, A.J.; Pedersen, J.S.; Frydenberg, M.; Risbridger, G.P. Induction of apoptosis in low to moderate-grade human prostate carcinoma by red clover-derived dietary isoflavones. Cancer Epidemiol. Biomarkers Prev. 2002, 12, 1689–1696. [Google Scholar]
- Fischer, L.; Mahoney, C.; Jeffcoat, A.R.; Koch, M.A.; Thomas, B.F.; Valentine, J.L.; Stinchcombe, T.; Boan, J.; Crowell, J.A.; Zeisel, S.H. Clinical characteristics and pharmacokinetics of purified soy isoflavones: Multiple-dose administration to men with prostate neoplasia. Nutr. Cancer 2004, 48, 160–170. [Google Scholar] [CrossRef]
- DeVere White, R.W.; Hackman, R.M.; Soares, S.E.; Beckett, L.A.; Li, Y.; Sun, B. Effects of a genistein-rich extract on PSA levels in men with a history of prostate cancer. Urology 2004, 63, 259–263. [Google Scholar] [CrossRef]
| Ref. | Ref. Period | Study Type/Design | Main Findings 1 |
|---|---|---|---|
| [76] | No explicit indication in the search protocol; includes studies published from 1992 to 2010 | Meta-analysis (22 studies, 7 cohorts, and 15 case-control studies) | For the high-dose category intake of isoflavones reduced the BCa risk in Asian populations (combined RR/OR = 0.68, 95% CI: 0.52–0.89) but not in Western populations (combined RR/OR = 0.98, 95% CI: 0.8–1.11) |
| [77] | 1966–2006 | Meta-analysis (21 studies: 14 case-control and 7 cohort studies) | The estimated pooled RR of BCa for soy food intake was 0.75, 95% CI: 0.59–0.95. Isoflavone intake resulted in a 20% decrease in risk (RR = 0.81, 95% CI: 0.67–0.99) |
| [78] | Up to January 2013 | Meta-analysis (35 studies: 30 on premenopausal women, including 10 cohort/nested case-control and 20 case-control studies; 31 on postmenopausal women, including 12 cohort/nested case-control and 19 case-control studies) | In Asian countries protective effect of isoflavones intake related to BCa was confirmed in both pre- and post-menopausal women (OR = 0.59, 95% CI: 0.48–0.69 and 0.44–0.74 for pre- and post-menopausal women, respectively); Among postmenopausal women living in Western countries soy isoflavone intake had a marginally significant protective effect (OR = 0.92, 95% CI: 0.83~1.00) |
| [79] | No explicit indication in the search protocol; includes studies published from 1991 to 2007 | Meta-analysis (8 studies for high-soy consuming Asians: 1 cohort and 7 case–control studies; 11 studies in low-soy consuming Western populations: 4 cohort/nested case-control and 7 case-control studies) | Among Asian women results implied a dose-dependent, statistically significant association between BCa RR and habitual soy food intake (approximately 16% risk decrease per 10 mg of isoflavones intake/day). No association was found in low-soy consuming Western populations. |
| [80] | Prospective study: 2004–2014; meta-analysis: up to 2019 | Prospective study and a dose-response meta-analysis (9 studies) | No association was found between soy intake and BCa incidence in the prospective study. Meta-analysis of prospective studies estimated that each 10 mg increment in daily soy isoflavones intake was associated with a 3% RR of BCa. |
| [81] | Up to September 2010 | Meta-analysis (18 prospective studies) | The protective effect of soy isoflavones against BCa risk was only observed among studies conducted in Asian populations (RR = 0.76, 95% CI: 0.65–0.86) but not in Western populations (RR = 0.97, 95% CI: 0.87–1.06). Isoflavone intake was also inversely associated with BCa recurrence (RR = 0.84, 95% CI: 0.70–0.99) |
| [82] | Up to April 2021 | Meta-analysis (8 prospective studies) | Isoflavone consumption of >15 mg/day is favored when compared to 0-15 mg/day (OR = 7.01 (95% CI: 6.58–7.47) with reference to BCa risk. |
| [83] | Up to February 2018 | Meta-analysis (12 studies: 10 prospective cohort studies, 1 case–control study, 1 pooled analysis) | Pre-diagnosis isoflavones intake was significantly associated with reduced risk of total mortality and recurrence. RR was predominantly observed among post-menopausal patients. |
| [84] | August 1996–March 1998 | Population-based case-control study | A 30% BCa RR was observed for women in the highest decile group based on isoflavones intake (OR = 0.66, 95% Cl: 0.46–0.95); superior risk reduction was reported for ER+/PR+ tumors than for other subtypes (OR = 0.44, 95% Cl: 0.25–0.78) |
| [85] | 2003–2005 | Case-control study | High intake of soybean products was associated with a decreased risk of ER+ (top tertile OR = 5 0.74; 95% CI, 0.58–0.94), HER2- (top tertile OR = 0.78; 95% CI, 0.61–0.99) and ER+/PR+/HER- BCa (top tertile OR = 0.73; 95% CI: 0.54–0.97) |
| [86] | 1996–2000 | Population-based prospective cohort study | Soy/isoflavones intake was associated with a significantly decreased risk of ER+/PR+ BCa in postmenopausal women (HR = 0.72; 95% CI: 0.53–0.96) and decreased risk of ER−/PR− BCa in premenopausal women (HR = 0.46; 95% CI: 0.22–0.97). The risk association did not vary by HER2 status. |
| [87] | 2007–2014 | Nationwide multicenter cohort study | Isoflavones intake above 15.50 mg/day was inversely associated with luminal A BCa risk in BRCA2 mutation carriers (HR = 0.14, 95% CI: 0.04–0.50) and triple-negative BCa in BRCA1 carriers (HR = 0.09, 95% CI: 0.02–0.40) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pejčić, T.; Zeković, M.; Bumbaširević, U.; Kalaba, M.; Vovk, I.; Bensa, M.; Popović, L.; Tešić, Ž. The Role of Isoflavones in the Prevention of Breast Cancer and Prostate Cancer. Antioxidants 2023, 12, 368. https://doi.org/10.3390/antiox12020368
Pejčić T, Zeković M, Bumbaširević U, Kalaba M, Vovk I, Bensa M, Popović L, Tešić Ž. The Role of Isoflavones in the Prevention of Breast Cancer and Prostate Cancer. Antioxidants. 2023; 12(2):368. https://doi.org/10.3390/antiox12020368
Chicago/Turabian StylePejčić, Tomislav, Milica Zeković, Uroš Bumbaširević, Milica Kalaba, Irena Vovk, Maja Bensa, Lazar Popović, and Živoslav Tešić. 2023. "The Role of Isoflavones in the Prevention of Breast Cancer and Prostate Cancer" Antioxidants 12, no. 2: 368. https://doi.org/10.3390/antiox12020368
APA StylePejčić, T., Zeković, M., Bumbaširević, U., Kalaba, M., Vovk, I., Bensa, M., Popović, L., & Tešić, Ž. (2023). The Role of Isoflavones in the Prevention of Breast Cancer and Prostate Cancer. Antioxidants, 12(2), 368. https://doi.org/10.3390/antiox12020368









