Zebrafish Model Insights into Mediterranean Diet Liquids: Olive Oil and Wine
Abstract
:1. Review Outline
2. The Mediterranean Diet
2.1. Mediterranean Diet Definition
2.2. Mediterranean Diet Health Benefits
2.2.1. Mediterranean Diet and Longevity
2.2.2. Mediterranean Diet and Cardiovascular Diseases
3. Translational Research: From Observational to Animal Studies
3.1. One Type of Effect at a Time: Here, the Antioxidant
3.2. One Food at a Time: Here, the Liquid Foods
3.2.1. Olive Oil and the Animal Studies on Its Antioxidant Properties
3.2.2. Wine and the Animal Studies on Its Antioxidant Properties
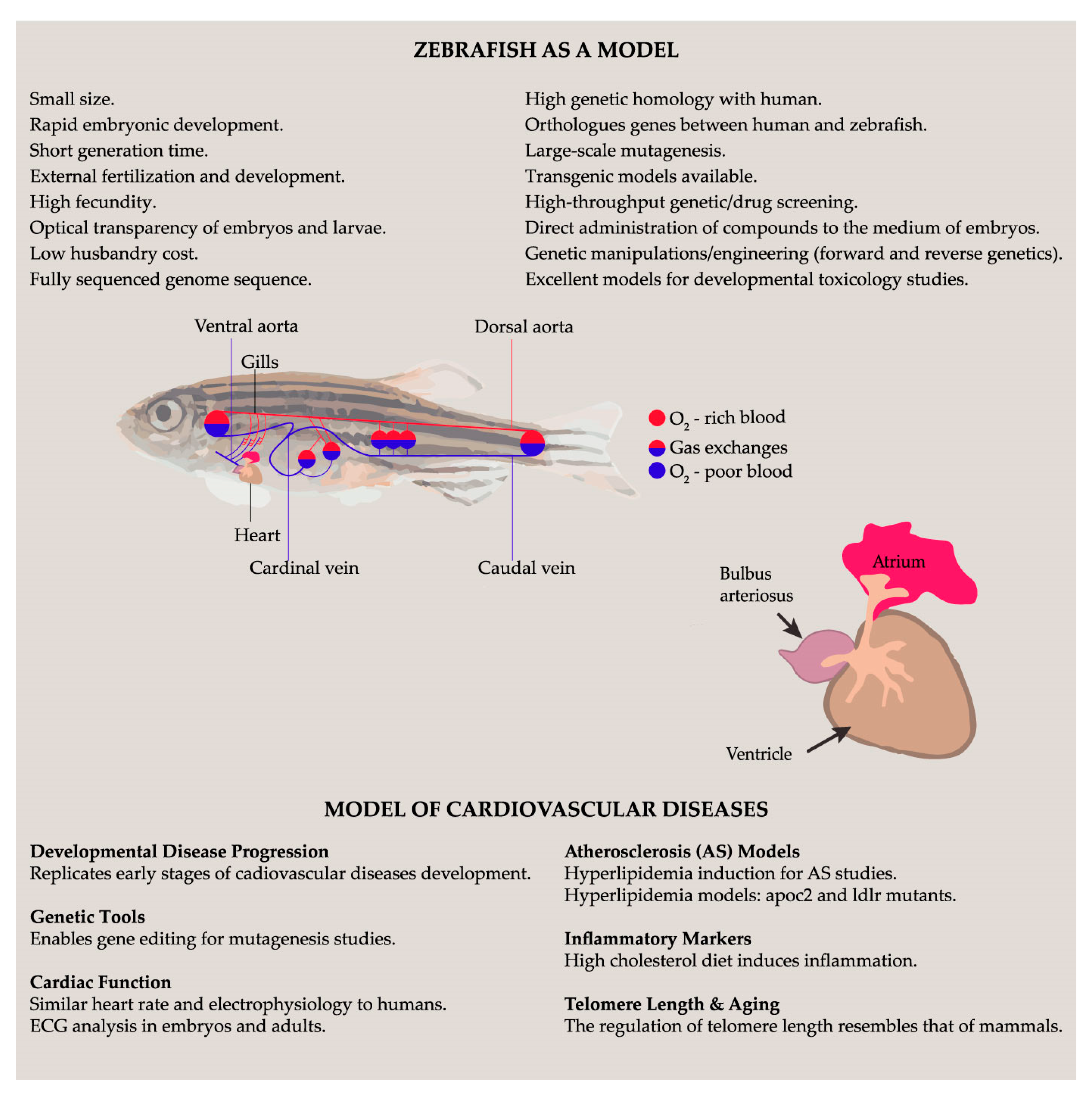
3.3. Zebrafish as a Potential Model to Illustrate Olive Oil and Wine Antioxidant Effects
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Keys, A. Eat Well & Stay Well; Doubleday & Company, Inc.: New York, NY, USA, 1959; p. 359. [Google Scholar]
- Berry, E.M.; Arnoni, Y.; Aviram, M. The Middle Eastern and biblical origins of the Mediterranean diet. Public Health Nutr. 2011, 14, 2288–2295. [Google Scholar] [CrossRef]
- Arnoni, Y.; Berry, E.M. Chapter 1—On the Origins and Evolution of the Mediterranean Diet. In the Mediterranean Diet; Preedy, V.R., Watson, R.R., Eds.; Academic Press: San Diego, CA, USA, 2015; pp. 3–11. [Google Scholar]
- Garnsey, P. Food and Society in Classical Antiquity; Cambridge University Press: Cambridge, UK, 1999. [Google Scholar]
- Grivetti, L.E.; Matalas, A.; Zampelas, V.; Wolinsky, I. Mediterranean food patterns: The view from antiquity, ancient Greeks and Romans. In The Mediterranean Diet: Constituents and Health Promotion; CRC Press Boca Raton: Boca Raton, FL, USA, 2001. [Google Scholar]
- Ferro-Luzzi, A.; Branca, F. Mediterranean diet, Italian-style: Prototype of a healthy diet. Am. J. Clin. Nutr. 1995, 61, 1338S–1345S. [Google Scholar] [CrossRef] [PubMed]
- UNESCO. Nomination File 00394. Available online: http://www.unesco.org/culture/ich/index.php (accessed on 8 July 2023).
- Bach-Faig, A.; Berry, E.M.; Lairon, D.; Reguant, J.; Trichopoulou, A.; Dernini, S.; Medina, F.X.; Battino, M.; Belahsen, R.; Miranda, G.; et al. Mediterranean diet pyramid today. Science and cultural updates. Public Health Nutr. 2011, 14, 2274–2284. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.; Bryan, J.; Hodgson, J.; Murphy, K. Definition of the mediterranean diet: A literature review. Nutrients 2015, 7, 9139–9153. [Google Scholar] [CrossRef] [PubMed]
- Keys, A. Seven Countries: A Multivariate Analysis of Death and Coronary Heart Disease; Harvard University Press: Cambridge, MA, USA, 1980. [Google Scholar]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. Hallmarks of aging: An expanding universe. Cell 2023, 186, 243–278. [Google Scholar] [CrossRef]
- Niccoli, T.; Partridge, L. Ageing as a risk factor for disease. Curr. Biol. 2012, 22, R741–R752. [Google Scholar] [CrossRef]
- Shannon, O.M.; Ashor, A.W.; Scialo, F.; Saretzki, G.; Martin-Ruiz, C.; Lara, J.; Matu, J.; Griffiths, A.; Robinson, N.; Lillà, L. Mediterranean diet and the hallmarks of ageing. Eur. J. Clin. Nutr. 2021, 75, 1176–1192. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Kouris-Blazos, A.; Wahlqvist, M.L.; Gnardellis, C.; Lagiou, P.; Polychronopoulos, E.; Vassilakou, T.; Lipworth, L.; Trichopoulos, D. Diet and overall survival in elderly people. BMJ 1995, 311, 1457–1460. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean diet and survival in a Greek population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef]
- De Lorgeril, M.; Salen, P.; Martin, J.-L.; Monjaud, I.; Delaye, J.; Mamelle, N. Mediterranean diet, traditional risk factors, and the rate of cardiovascular complications after myocardial infarction: Final report of the Lyon Diet Heart Study. Circulation 1999, 99, 779–785. [Google Scholar] [CrossRef]
- Lasheras, C.; Fernandez, S.; Patterson, A.M. Mediterranean diet and age with respect to overall survival in institutionalized, nonsmoking elderly people. Am. J. Clin. Nutr. 2000, 71, 987–992. [Google Scholar] [CrossRef] [PubMed]
- Knoops, K.T.; de Groot, L.C.; Kromhout, D.; Perrin, A.-E.; Moreiras-Varela, O.; Menotti, A.; Van Staveren, W.A. Mediterranean diet, lifestyle factors, and 10-year mortality in elderly European men and women: The HALE project. JAMA 2004, 292, 1433–1439. [Google Scholar] [CrossRef] [PubMed]
- Osler, M.; Schroll, M. Diet and mortality in a cohort of elderly people in a north European community. Int. J. Epidemiol. 1997, 26, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Orfanos, P.; Norat, T.; Bueno-de-Mesquita, B.; Ocké, M.C.; Peeters, P.H.; van der Schouw, Y.T.; Boeing, H.; Hoffmann, K.; Boffetta, P. Modified Mediterranean diet and survival: EPIC-elderly prospective cohort study. BMJ 2005, 330, 991. [Google Scholar] [CrossRef] [PubMed]
- Wahlqvist, M.; Darmadi-Blackberry, I.; Kouris-Blazos, A.; Jolley, D.; Steen, B.; Lukito, W.; Horie, Y. Does diet matter for survival in long-lived cultures? Asia Pac. J. Clin. Nutr. 2005, 14, 2. [Google Scholar]
- Mitrou, P.N.; Kipnis, V.; Thiébaut, A.C.; Reedy, J.; Subar, A.F.; Wirfält, E.; Flood, A.; Mouw, T.; Hollenbeck, A.R.; Leitzmann, M.F. Mediterranean dietary pattern and prediction of all-cause mortality in a US population: Results from the NIH-AARP Diet and Health Study. Arch. Intern. Med. 2007, 167, 2461–2468. [Google Scholar] [CrossRef]
- Reedy, J.; Krebs-Smith, S.M.; Miller, P.E.; Liese, A.D.; Kahle, L.L.; Park, Y.; Subar, A.F. Higher diet quality is associated with decreased risk of all-cause, cardiovascular disease, and cancer mortality among older adults. J. Nutr. 2014, 144, 881–889. [Google Scholar] [CrossRef]
- Lagiou, P.; Trichopoulos, D.; Sandin, S.; Lagiou, A.; Mucci, L.; Wolk, A.; Elisabete, E.; Adami, H.-O. Mediterranean dietary pattern and mortality among young women: A cohort study in Sweden. Br. J. Nutr. 2006, 96, 384–392. [Google Scholar] [CrossRef]
- Sofi, F.; Cesari, F.; Abbate, R.; Gensini, G.F.; Casini, A. Adherence to Mediterranean diet and health status: Meta-analysis. BMJ 2008, 337, a1344. [Google Scholar] [CrossRef]
- Soltani, S.; Jayedi, A.; Shab-Bidar, S.; Becerra-Tomás, N.; Salas-Salvadó, J. Adherence to the Mediterranean diet in relation to all-cause mortality: A systematic review and dose-response meta-analysis of prospective cohort studies. Adv. Nutr. 2019, 10, 1029–1039. [Google Scholar] [CrossRef]
- Canudas, S.; Becerra-Tomás, N.; Hernández-Alonso, P.; Galié, S.; Leung, C.; Crous-Bou, M.; De Vivo, I.; Gao, Y.; Gu, Y.; Meinilä, J. Mediterranean diet and telomere length: A systematic review and meta-analysis. Adv. Nutr. 2020, 11, 1544–1554. [Google Scholar] [CrossRef]
- Crous-Bou, M.; Fung, T.T.; Prescott, J.; Julin, B.; Du, M.; Sun, Q.; Rexrode, K.M.; Hu, F.B.; De Vivo, I. Mediterranean diet and telomere length in Nurses’ Health Study: Population based cohort study. BMJ 2014, 349, g6674. [Google Scholar] [CrossRef] [PubMed]
- Guasch-Ferré, M.; Willett, W.C. The Mediterranean diet and health: A comprehensive overview. J. Intern. Med. 2021, 290, 549–566. [Google Scholar] [CrossRef] [PubMed]
- Laslett, L.J.; Alagona, P.; Clark, B.A.; Drozda, J.P.; Saldivar, F.; Wilson, S.R.; Poe, C.; Hart, M. The worldwide environment of cardiovascular disease: Prevalence, diagnosis, therapy, and policy issues: A report from the American College of Cardiology. J. Am. Coll. Cardiol. 2012, 60, S1–S49. [Google Scholar] [CrossRef] [PubMed]
- Virani, S.S.; Alonso, A.; Aparicio, H.J.; Benjamin, E.J.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Cheng, S.; Delling, F.N. Heart disease and stroke statistics-2021 update: A report from the American Heart Association. Circulation 2021, 143, e139–e596. [Google Scholar] [CrossRef]
- Stampfer, M.J.; Hu, F.B.; Manson, J.E.; Rimm, E.B.; Willett, W.C. Primary prevention of coronary heart disease in women through diet and lifestyle. N. Engl. J. Med. 2000, 343, 16–22. [Google Scholar] [CrossRef]
- Dilis, V.; Katsoulis, M.; Lagiou, P.; Trichopoulos, D.; Naska, A.; Trichopoulou, A. Mediterranean diet and CHD: The Greek European Prospective Investigation into Cancer and Nutrition cohort. Br. J. Nutr. 2012, 108, 699–709. [Google Scholar] [CrossRef]
- Buckland, G.; González, C.A.; Agudo, A.; Vilardell, M.; Berenguer, A.; Amiano, P.; Ardanaz, E.; Arriola, L.; Barricarte, A.; Basterretxea, M. Adherence to the Mediterranean diet and risk of coronary heart disease in the Spanish EPIC Cohort Study. Am. J. Epidemiol. 2009, 170, 1518–1529. [Google Scholar] [CrossRef]
- Hoevenaar-Blom, M.P.; Nooyens, A.C.; Kromhout, D.; Spijkerman, A.M.; Beulens, J.W.; Van Der Schouw, Y.T.; Bueno-de-Mesquita, B.; Verschuren, W.M. Mediterranean style diet and 12-year incidence of cardiovascular diseases: The EPIC-NL cohort study. PLoS ONE 2012, 7, e45458. [Google Scholar] [CrossRef]
- Tognon, G.; Lissner, L.; Sæbye, D.; Walker, K.Z.; Heitmann, B.L. The Mediterranean diet in relation to mortality and CVD: A Danish cohort study. Br. J. Nutr. 2014, 111, 151–159. [Google Scholar] [CrossRef]
- Gardener, H.; Wright, C.B.; Gu, Y.; Demmer, R.T.; Boden-Albala, B.; Elkind, M.S.; Sacco, R.L.; Scarmeas, N. Mediterranean-style diet and risk of ischemic stroke, myocardial infarction, and vascular death: The Northern Manhattan Study. Am. J. Clin. Nutr. 2011, 94, 1458–1464. [Google Scholar] [CrossRef] [PubMed]
- Panagiotakos, D.B.; Pitsavos, C.; Chrysohoou, C.; Skoumas, I.; Stefanadis, C. Five-year incidence of cardiovascular disease and its predictors in Greece: The ATTICA study. Vasc. Med. 2008, 13, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Martínez-González, M.A.; García-López, M.; Bes-Rastrollo, M.; Toledo, E.; Martínez-Lapiscina, E.H.; Delgado-Rodriguez, M.; Vazquez, Z.; Benito, S.; Beunza, J.J. Mediterranean diet and the incidence of cardiovascular disease: A Spanish cohort. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 237–244. [Google Scholar] [CrossRef] [PubMed]
- George, S.M.; Ballard-Barbash, R.; Manson, J.E.; Reedy, J.; Shikany, J.M.; Subar, A.F.; Tinker, L.F.; Vitolins, M.; Neuhouser, M.L. Comparing indices of diet quality with chronic disease mortality risk in postmenopausal women in the Women’s Health Initiative Observational Study: Evidence to inform national dietary guidance. Am. J. Epidemiol. 2014, 180, 616–625. [Google Scholar] [CrossRef]
- Bertoia, M.L.; Triche, E.W.; Michaud, D.S.; Baylin, A.; Hogan, J.W.; Neuhouser, M.L.; Tinker, L.F.; Van Horn, L.; Waring, M.E.; Li, W. Mediterranean and dietary approaches to stop hypertension dietary patterns and risk of sudden cardiac death in postmenopausal women. Am. J. Clin. Nutr. 2014, 99, 344–351. [Google Scholar] [CrossRef]
- Mohseni, M.; Mohammadifard, N.; Hassannejad, R.; Aghabozorgi, M.; Shirani, F.; Sadeghi, M.; Roohafza, H.; Sarrafzadegan, N. Longitudinal association of dietary habits and the risk of cardiovascular disease among Iranian population between 2001 and 2013: The Isfahan Cohort Study. Sci. Rep. 2023, 13, 5364. [Google Scholar] [CrossRef]
- Sotos-Prieto, M.; Bhupathiraju, S.N.; Mattei, J.; Fung, T.T.; Li, Y.; Pan, A.; Willett, W.C.; Rimm, E.B.; Hu, F.B. Changes in diet quality scores and risk of cardiovascular disease among US men and women. Circulation 2015, 132, 2212–2219. [Google Scholar] [CrossRef]
- Pitsavos, C.; Panagiotakos, D.; Chrysohoou, C.; Stefanadis, C.; Toutouzas, P. Τhe benefits from Mediterranean diet on the risk of developing acute coronary syndromes, in hypercholesterolemic subjects: A case-control study (CARDIO2000). Coron. Artery Dis. 2002, 13, 295–300. [Google Scholar] [CrossRef]
- Pitsavos, C.; Panagiotakos, D.; Chrysohoou, C.; Kokkinos, P.; Skoumas, J.; Papaioannou, I.; Stefanadis, C.; Toutouzas, P. The effect of the combination of Mediterranean diet and leisure time physical activity on the risk of developing acute coronary syndromes, in hypertensive subjects. J. Hum. Hypertens. 2002, 16, 517–524. [Google Scholar] [CrossRef]
- Pitsavos, C.; Panagiotakos, D.B.; Chrysohoou, C.; Papaioannou, I.; Papadimitriou, L.; Tousoulis, D.; Stefanadis, C.; Toutouzas, P. The adoption of Mediterranean diet attenuates the development of acute coronary syndromes in people with the metabolic syndrome. Nutr. J. 2003, 2, 1. [Google Scholar] [CrossRef]
- Pitsavos, C.; Chrysohoou, C.; Panagiotakos, D.B.; Lentzas, Y.; Stefanadis, C. Abdominal obesity and inflammation predicts hypertension among prehypertensive men and women: The ATTICA Study. Heart Vessel. 2008, 23, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Panagiotakos, D.B.; Georgousopoulou, E.N.; Pitsavos, C.; Chrysohoou, C.; Skoumas, I.; Pitaraki, E.; Georgiopoulos, G.A.; Ntertimani, M.; Christou, A.; Stefanadis, C. Exploring the path of Mediterranean diet on 10-year incidence of cardiovascular disease: The ATTICA study (2002–2012). Nutr. Metab. Cardiovasc. Dis. 2015, 25, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Panagiotakos, D.B.; Pitsavos, C.; Leda Matalas, A.; Chrysohoou, C.; Stefanadis, C. Geographical influences on the association between adherence to the Mediterranean diet and the prevalence of acute coronary syndromes, in Greece: The CARDIO2000 study. Int. J. Cardiol. 2005, 100, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Martínez-González, M.A.; Fernández-Jarne, E.; Serrano-Martínez, M.; Marti, A.; Martinez, J.A.; Martín-Moreno, J.M. Mediterranean diet and reduction in the risk of a first acute myocardial infarction: An operational healthy dietary score. Eur. J. Nutr. 2002, 41, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Turati, F.; Pelucchi, C.; Galeone, C.; Praud, D.; Tavani, A.; La Vecchia, C. Mediterranean diet and non-fatal acute myocardial infarction: A case–control study from Italy. Public Health Nutr. 2015, 18, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Georgousopoulou, E.N.; Kastorini, C.-M.; Milionis, H.J.; Ntziou, E.; Kostapanos, M.S.; Nikolaou, V.; Vemmos, K.N.; Goudevenos, J.A.; Panagiotakos, D.B. Association between mediterranean diet and non-fatal cardiovascular events, in the context of anxiety and depression disorders: A case/case-control study. Hell. J. Cardiol. 2014, 55, 24–31. [Google Scholar]
- Tsivgoulis, G.; Psaltopoulou, T.; Wadley, V.G.; Alexandrov, A.V.; Howard, G.; Unverzagt, F.W.; Moy, C.; Howard, V.J.; Kissela, B.; Judd, S.E. Adherence to a Mediterranean Diet and prediction of incident stroke. Stroke 2015, 46, 780–785. [Google Scholar] [CrossRef]
- Ros, E.; Martínez-González, M.A.; Estruch, R.; Salas-Salvadó, J.; Fitó, M.; Martínez, J.A.; Corella, D. Mediterranean Diet and cardiovascular health: Teachings of the PREDIMED study. Adv. Nutr. 2014, 5, 330S–336S. [Google Scholar] [CrossRef]
- Kelleher, A.H.; Sikalidis, A.K. The effects of Mediterranean Diet on the human gut microbiota; a brief discussion of evidence in humans. OBM Hepatol. Gastroenterol. 2021, 5, 2101056. [Google Scholar] [CrossRef]
- Hajam, Y.A.; Rani, R.; Ganie, S.Y.; Sheikh, T.A.; Javaid, D.; Qadri, S.S.; Pramodh, S.; Alsulimani, A.; Alkhanani, M.F.; Harakeh, S.; et al. Oxidative stress in human pathology and aging: Molecular mechanisms and perspectives. Cells 2022, 11, 552. [Google Scholar] [CrossRef]
- Yang, C.S.; Ho, C.-T.; Zhang, J.; Wan, X.; Zhang, K.; Lim, J. Antioxidants: Differing meanings in food science and health science. J. Agric. Food Chem. 2018, 66, 3063–3068. [Google Scholar] [CrossRef] [PubMed]
- Zeb, A. Concept, mechanism, and applications of phenolic antioxidants in foods. J. Food Biochem. 2020, 44, e13394. [Google Scholar] [CrossRef] [PubMed]
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative stress, aging, and diseases. Clin. Interv. Aging 2018, 13, 757–772. [Google Scholar] [CrossRef] [PubMed]
- Fehér, J.; Lengyel, G.; Lugasi, A. The cultural history of wine-theoretical background to wine therapy. Cent. Eur. J. Med. 2007, 2, 379–391. [Google Scholar] [CrossRef]
- Mazzocchi, A.; Leone, L.; Agostoni, C.; Pali-Schöll, I. The secrets of the Mediterranean Diet. Does [Only] olive oil matter? Nutrients 2019, 11, 2941. [Google Scholar] [CrossRef]
- Nocella, C.; Cammisotto, V.; Fianchini, L.; D’Amico, A.; Novo, M.; Castellani, V.; Stefanini, L.; Violi, F.; Carnevale, R. Extra virgin olive oil and cardiovascular diseases: Benefits for human health. Endocr. Metab. Immune Disord. Drug Targets 2017, 18, 4–13. [Google Scholar] [CrossRef]
- Fernández Del Río, L.; Gutiérrez-Casado, E.; Varela-López, A.; Villalba, J. Olive oil and the hallmarks of aging. Molecules 2016, 21, 163. [Google Scholar] [CrossRef]
- Fytili, C.; Nikou, T.; Tentolouris, N.; Tseti, I.K.; Dimosthenopoulos, C.; Sfikakis, P.P.; Simos, D.; Kokkinos, A.; Skaltsounis, A.L.; Katsilambros, N.; et al. Effect of long-term hydroxytyrosol administration on body weight, fat mass and urine metabolomics: A randomized double-blind prospective human study. Nutrients 2022, 14, 1525. [Google Scholar] [CrossRef]
- Silva, P.; Rodríguez-Pérez, M.; Gómez-Torres, Ó.; Burgos-Ramos, E. Olive oil and wine as source of multi-target agents in the prevention of Alzheimer disease. Nutr. Res. Rev. 2023, 36, 140–154. [Google Scholar] [CrossRef]
- Chowanadisai, W.; Hart, M.D.; Strong, M.D.; Graham, D.M.; Rucker, R.B.; Smith, B.J.; Keen, C.L.; Messerli, M.A. Genetic and genomic advances in developmental models: Applications for nutrition research. Adv. Nutr. 2020, 11, 971–978. [Google Scholar] [CrossRef]
- Silva, P.; Fernandes, E.; Carvalho, F. Dual effect of red wine on liver redox status: A concise and mechanistic review. Arch. Toxicol. 2015, 89, 1681–1693. [Google Scholar] [CrossRef] [PubMed]
- Silva, P.; Vauzour, D. Wine polyphenols and neurodegenerative diseases: An update on the molecular mechanisms underpinning their protective effects. Beverages 2018, 4, 96. [Google Scholar] [CrossRef]
- Maykish, A.; Rex, R.; Sikalidis, A.K. Organic winemaking and its subsets; biodynamic, natural, and clean wine in California. Foods 2021, 10, 127. [Google Scholar] [CrossRef] [PubMed]
- Di Castelnuovo, A.; Costanzo, S.; Bonaccio, M.; Rago, L.; De Curtis, A.; Persichillo, M.; Bracone, F.; Olivieri, M.; Cerletti, C.; Donati, M.B.; et al. Moderate Alcohol consumption is associated with lower risk for heart failure but not atrial fibrillation. JACC Heart Fail. 2017, 5, 837–844. [Google Scholar] [CrossRef]
- Levantesi, G.; Marfisi, R.; Mozaffarian, D.; Franzosi, M.G.; Maggioni, A.; Nicolosi, G.L.; Schweiger, C.; Silletta, M.; Tavazzi, L.; Tognoni, G.; et al. Wine consumption and risk of cardiovascular events after myocardial infarction: Results from the GISSI-Prevenzione trial. Int. J. Cardiol. 2013, 163, 282–287. [Google Scholar] [CrossRef]
- Panagiotakos, D.B.; Kouli, G.-M.; Magriplis, E.; Kyrou, I.; Georgousopoulou, E.N.; Chrysohoou, C.; Tsigos, C.; Tousoulis, D.; Pitsavos, C. Beer, wine consumption, and 10-year CVD incidence: The ATTICA study. Eur. J. Clin. Nutr. 2019, 73, 1015–1023. [Google Scholar] [CrossRef]
- Di Castelnuovo, A.; Costanzo, S.; Bonaccio, M.; McElduff, P.; Linneberg, A.; Salomaa, V.; Männistö, S.; Moitry, M.; Ferrières, J.; Dallongeville, J.; et al. Alcohol intake and total mortality in 142,960 individuals from the MORGAM Project: A population-based study. Addiction 2022, 117, 312–325. [Google Scholar] [CrossRef]
- Román, G.C.; Jackson, R.E.; Reis, J.; Román, A.N.; Toledo, J.B.; Toledo, E. Extra-virgin olive oil for potential prevention of Alzheimer disease. Rev. Neurol. 2019, 175, 705–723. [Google Scholar] [CrossRef]
- Drehmer, E.; Navarro-Moreno, M.Á.; Carrera-Juliá, S.; Moreno, M.L. A comparative study between olive oil and corn oil on oxidative metabolism. Food Funct. 2022, 13, 7157–7167. [Google Scholar] [CrossRef]
- Elgebaly, H.A.; Mosa, N.M.; Allach, M.; El-massry, K.F.; El-Ghorab, A.H.; Al Hroob, A.M.; Mahmoud, A.M. Olive oil and leaf extract prevent fluoxetine-induced hepatotoxicity by attenuating oxidative stress, inflammation and apoptosis. Biomed. Pharmacother. 2018, 98, 446–453. [Google Scholar] [CrossRef]
- Ghorbel, I.; Elwej, A.; Fendri, N.; Mnif, H.; Jamoussi, K.; Boudawara, T.; Grati Kamoun, N.; Zeghal, N. Olive oil abrogates acrylamide induced nephrotoxicity by modulating biochemical and histological changes in rats. Ren. Fail. 2017, 39, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Saleh, S.R.; Abdelhady, S.A.; Khattab, A.R.; El-Hadidy, W.F. Dual prophylactic/therapeutic potential of date seed, and nigella and olive oils-based nutraceutical formulation in rats with experimentally-induced Alzheimer’s disease: A mechanistic insight. J. Chem. Neuroanat. 2020, 110, 101878. [Google Scholar] [CrossRef]
- Vazquez, A.; Sanchez-Rodriguez, E.; Vargas, F.; Montoro-Molina, S.; Romero, M.; Espejo-Calvo, J.A.; Vilchez, P.; Jaramillo, S.; Olmo-García, L.; Carrasco-Pancorbo, A.; et al. Cardioprotective effect of a virgin olive oil enriched with bioactive compounds in spontaneously hypertensive rats. Nutrients 2019, 11, 1728. [Google Scholar] [CrossRef] [PubMed]
- Lafraxo, H.; Bakour, M.; Laaroussi, H.; El Ghouizi, A.; Ousaaid, D.; Aboulghazi, A.; Lyoussi, B. The synergistic beneficial effect of thyme honey and olive oil against diabetes and its complications induced by alloxan in wistar rats. Evid. Based Complement. Altern. Med. 2021, 2021, 9949056. [Google Scholar] [CrossRef] [PubMed]
- Pase, C.S.; Teixeira, A.M.; Roversi, K.; Dias, V.T.; Calabrese, F.; Molteni, R.; Franchi, S.; Panerai, A.E.; Riva, M.A.; Burger, M.E. Olive oil-enriched diet reduces brain oxidative damages and ameliorates neurotrophic factor gene expression in different life stages of rats. J. Nutr. Biochem. 2015, 26, 1200–1207. [Google Scholar] [CrossRef]
- Boss, A.; Bishop, K.; Marlow, G.; Barnett, M.; Ferguson, L. Evidence to support the anti-cancer effect of olive leaf extract and future directions. Nutrients 2016, 8, 513. [Google Scholar] [CrossRef]
- Nani, A.; Murtaza, B.; Sayed Khan, A.; Khan, N.A.; Hichami, A. Antioxidant and anti-inflammatory potential of polyphenols contained in mediterranean diet in obesity: Molecular mechanisms. Molecules 2021, 26, 985. [Google Scholar] [CrossRef]
- Roig, R.; Cascón, E.; Arola, L.; Bladé, C.; Josepa Salvadó, M. Moderate red wine consumption protects the rat against oxidation in vivo. Life Sci. 1999, 64, 1517–1524. [Google Scholar] [CrossRef]
- Simonetti, P.; Cervato, G.; Brusamolino, A.; Gatti, P.; Pellegrini, N.; Cestaro, B. Effect of red wine consumption on rat liver peroxidation. Alcohol 1996, 13, 41–45. [Google Scholar] [CrossRef]
- MacEdo, L.F.L.; Rogero, M.M.; Guimarães, J.P.; Granato, D.; Lobato, L.P.; Castro, I.A. Effect of red wines with different in vitro antioxidant activity on oxidative stress of high-fat diet rats. Food Chem. 2013, 137, 122–129. [Google Scholar] [CrossRef]
- Assunção, M.; Santos-Marques, M.J.; Monteiro, R.; Azevedo, I.; Andrade, J.P.; Carvalho, F.; Martins, M.J. Red wine protects against ethanol-induced oxidative stress in rat liver. J. Agric. Food Chem. 2009, 57, 6066–6073. [Google Scholar] [CrossRef] [PubMed]
- Orellana, M.; Varela, N.; Guajardo, V.; Araya, J.; Rodrigo, R. Modulation of rat liver cytochrome P450 activity by prolonged red wine consumption. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2002, 131, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, K.; Sugamoto, K.; Arakawa, T.; Nishiyama, K.; Yamasaki, M. Chronic intake of high-dose of blueberry leaf extract does not augment the harmful effects of ethanol in rats. PeerJ 2019, 7, e6989. [Google Scholar] [CrossRef] [PubMed]
- Orellana, M.; Araya, J.; Guajardo, V.; Rodrigo, R. Modulation of cytochrome P450 activity in the kidney of rats following long-term red wine exposure. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2002, 132, 399–405. [Google Scholar] [CrossRef]
- Rodrigo, R.; Castillo, R.; Carrasco, R.; Huerta, P.; Moreno, M. Diminution of tissue lipid peroxidation in rats is related to the in vitro antioxidant capacity of wine. Life Sci. 2005, 76, 889–900. [Google Scholar] [CrossRef]
- Sabadashka, M.; Hertsyk, D.; Strugała-Danak, P.; Dudek, A.; Kanyuka, O.; Kucharska, A.Z.; Kaprelyants, L.; Sybirna, N. Anti-diabetic and antioxidant activities of red wine concentrate enriched with polyphenol compounds under experimental diabetes in rats. Antioxidants 2021, 10, 1399. [Google Scholar] [CrossRef]
- Landrault, N.; Poucheret, P.; Azay, J.; Krosniak, M.; Gasc, F.; Jenin, C.; Cros, G.; Teissedre, P.L. Effect of a polyphenols-enriched chardonnay white wine in diabetic rats. J. Agric. Food Chem. 2003, 51, 311–318. [Google Scholar] [CrossRef]
- Souza Bomfim, G.H.; Musial, D.C.; Rocha, K.; Jurkiewicz, A.; Jurkiewicz, N.H. Red wine but not alcohol consumption improves cardiovascular function and oxidative stress of the hypertensive-SHR and diabetic-STZ rats. Clin. Exp. Hypertens. 2022, 44, 573–584. [Google Scholar] [CrossRef]
- Banc, R.; Popa, D.-S.; Cozma-Petruţ, A.; Filip, L.; Kiss, B.; Fărcaş, A.; Nagy, A.; Miere, D.; Loghin, F. Protective effects of wine polyphenols on oxidative stress and hepatotoxicity induced by acrylamide in rats. Antioxidants 2022, 11, 1347. [Google Scholar] [CrossRef]
- Assunção, M.; Santos-Marques, M.J.; de Freitas, V.; Carvalho, F.; Andrade, J.P.; Lukoyanov, N.V.; Paula-Barbosa, M.M. Red wine antioxidants protect hippocampal neurons against ethanol-induced damage: A biochemical, morphological and behavioral study. Neuroscience 2007, 146, 1581–1592. [Google Scholar] [CrossRef]
- Assunção, M.; Santos-Marques, M.J.; De Freitas, V.; Paula-Barbosa, M.M.; Carvalho, F. Modulation of rat cerebellum oxidative status by prolonged red wine consumption. Addict. Biol. 2008, 13, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Liu, X.; Qin, Y.; Hou, Y.; Hou, J.; Wu, Q.; Xu, W. Zebrafish as model organisms for toxicological evaluations in the field of food science. Compr. Rev. Food Sci. Food Saf. 2023, 22, 3481–3505. [Google Scholar] [CrossRef] [PubMed]
- Borzì, A.; Biondi, A.; Basile, F.; Luca, S.; Vicari, E.; Vacante, M. Olive oil effects on colorectal cancer. Nutrients 2018, 11, 32. [Google Scholar] [CrossRef] [PubMed]
- Teranikar, T.; Nguyen, P.; Lee, J. Biomechanics of cardiac development in zebrafish model. Curr. Opin. Biomed. Eng. 2023, 27, 100459. [Google Scholar] [CrossRef]
- Bowley, G.; Kugler, E.; Wilkinson, R.; Lawrie, A.; Van Eeden, F.; Chico, T.J.A.; Evans, P.C.; Noël, E.S.; Serbanovic-Canic, J. Zebrafish as a tractable model of human cardiovascular disease. Br. J. Pharmacol. 2022, 179, 900–917. [Google Scholar] [CrossRef]
- Sukhorukov, V.N.; Khotina, V.A.; Chegodaev, Y.S.; Ivanova, E.; Soben, I.A.; Orekhov, A.N. Lipid metabolism in macrophages: Focus on Atherosclerosis. Biomedicines 2020, 8, 262. [Google Scholar] [CrossRef]
- Liu, C.; Gates, K.P.; Fang, L.; Amar, M.J.; Schneider, D.A.; Geng, H.; Huang, W.; Kim, J.; Pattison, J.; Zhang, J.; et al. Apoc2 loss-of-function zebrafish mutant as a genetic model of hyperlipidemia. Dis. Models Mech. 2015, 8, 989–998. [Google Scholar] [CrossRef]
- Liu, C.; Kim, Y.S.; Kim, J.; Pattison, J.; Kamaid, A.; Miller, Y.I. Modeling hypercholesterolemia and vascular lipid accumulation in LDL receptor mutant zebrafish. J. Lipid Res. 2018, 59, 391–399. [Google Scholar] [CrossRef]
- Stoletov, K.; Fang, L.; Choi, S.-H.; Hartvigsen, K.; Hansen, L.F.; Hall, C.; Pattison, J.; Juliano, J.; Miller, E.R.; Almazan, F.; et al. Vascular lipid accumulation, lipoprotein oxidation, and macrophage lipid uptake in hypercholesterolemic zebrafish. Circ. Res. 2009, 104, 952–960. [Google Scholar] [CrossRef]
- Luo, H.; Li, Q.-Q.; Wu, N.; Shen, Y.-G.; Liao, W.-T.; Yang, Y.; Dong, E.; Zhang, G.-M.; Liu, B.-R.; Yue, X.-Z.; et al. Chronological in vivo imaging reveals endothelial inflammation prior to neutrophils accumulation and lipid deposition in HCD-fed zebrafish. Atherosclerosis 2019, 290, 125–135. [Google Scholar] [CrossRef]
- Herrmann, W.; Herrmann, M. The Importance of telomere shortening for atherosclerosis and mortality. J. Cardiovasc. Dev. Dis. 2020, 7, 29. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, M.C.; De Castro, I.P.; Ferreira, M.G. Telomeres in aging and disease: Lessons from zebrafish. DMM Dis. Models Mech. 2016, 9, 737–748. [Google Scholar] [CrossRef] [PubMed]
- Giordo, R.; Nasrallah, G.K.; Al-Jamal, O.; Paliogiannis, P.; Pintus, G. Resveratrol inhibits oxidative stress and prevents mitochondrial damage induced by zinc oxide nanoparticles in zebrafish (Danio rerio). Int. J. Mol. Sci. 2020, 21, 3838. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, C.; Zhang, J.; Xie, J.; Yang, L.; Xing, Y.; Li, Z. The effects of quercetin on immunity, antioxidant indices, and disease resistance in zebrafish (Danio rerio). Fish Physiol. Biochem. 2020, 46, 759–770. [Google Scholar] [CrossRef]
- Dong, Y.; Yu, M.; Wu, Y.; Xia, T.; Wang, L.; Song, K.; Zhang, C.; Lu, K.; Rahimnejad, S. Hydroxytyrosol promotes the mitochondrial function through activating mitophagy. Antioxidants 2022, 11, 893. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Sahu, K.; Kapil, L.; Singh, C.; Singh, A. Quercetin ameliorates lipopolysaccharide-induced neuroinflammation and oxidative stress in adult zebrafish. Mol. Biol. Rep. 2022, 49, 3247–3258. [Google Scholar] [CrossRef]
- Silva, N.-V.; Carregosa, D.; Gonçalves, C.; Vieira, O.V.; Nunes dos Santos, C.; Jacinto, A.; Crespo, C.L. A dietary cholesterol-based intestinal inflammation assay for improving drug-discovery on inflammatory bowel diseases. Front. Cell Dev. Biol. 2021, 9, 674749. [Google Scholar] [CrossRef]
- Arteaga, C.; Boix, N.; Teixido, E.; Marizande, F.; Cadena, S.; Bustillos, A. The zebrafish embryo as a model to test protective effects of food antioxidant compounds. Molecules 2021, 26, 5786. [Google Scholar] [CrossRef]
- Caro, M.; Sansone, A.; Amézaga, J.; Navarro, V.; Ferreri, C.; Tueros, I. Wine lees modulate lipid metabolism and induce fatty acid remodelling in Zebrafish. Food Funct. 2017, 8, 1652–1659. [Google Scholar] [CrossRef]
- Hoseinifar, S.H.; Fazelan, Z.; El-Haroun, E.; Yousefi, M.; Yazici, M.; Van Doan, H.; Paolucci, M. The effects of grapevine (Vitis vinifera L.) leaf extract on growth performance, antioxidant status, and immunity of zebrafish (Danio rerio). Fishes 2023, 8, 326. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, P.; Rodríguez-Pérez, M.; Burgos-Ramos, E. Zebrafish Model Insights into Mediterranean Diet Liquids: Olive Oil and Wine. Antioxidants 2023, 12, 1843. https://doi.org/10.3390/antiox12101843
Silva P, Rodríguez-Pérez M, Burgos-Ramos E. Zebrafish Model Insights into Mediterranean Diet Liquids: Olive Oil and Wine. Antioxidants. 2023; 12(10):1843. https://doi.org/10.3390/antiox12101843
Chicago/Turabian StyleSilva, Paula, María Rodríguez-Pérez, and Emma Burgos-Ramos. 2023. "Zebrafish Model Insights into Mediterranean Diet Liquids: Olive Oil and Wine" Antioxidants 12, no. 10: 1843. https://doi.org/10.3390/antiox12101843
APA StyleSilva, P., Rodríguez-Pérez, M., & Burgos-Ramos, E. (2023). Zebrafish Model Insights into Mediterranean Diet Liquids: Olive Oil and Wine. Antioxidants, 12(10), 1843. https://doi.org/10.3390/antiox12101843







