Comprehensive Analysis of Metabolites in Brews Prepared from Naturally and Technologically Treated Coffee Beans
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Sample Preparation
2.3. LC-MS/MS
2.4. Optimization of Green and Roasted Arabica and Robusta Coffee Brewing Conditions
2.5. Statistical Analysis
3. Results and Discussion
3.1. Determination of Chlorogenic Acids and Their Isomers
3.2. Determination of Methylxanthines and Trigonelline
3.3. Determination of Nicotinic Acid and Nicotinamide
3.4. Determination of Bioactive Amines: Theanine, Serotonin, and Melatonin
3.5. Determination of Quinic Acid and Other Phenolic Acids (Ferulic, Caffeic, and P-coumaric Acids)
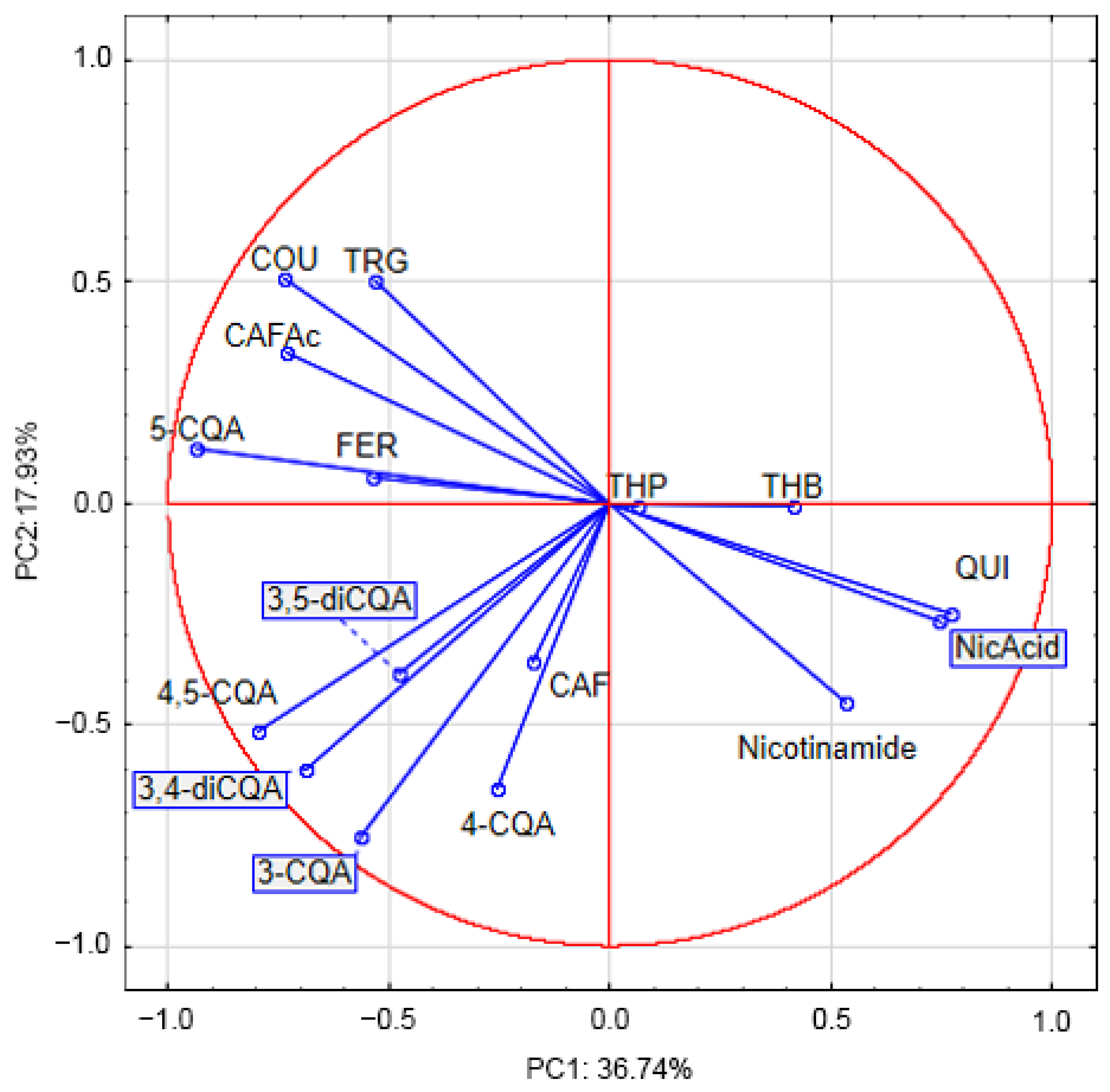
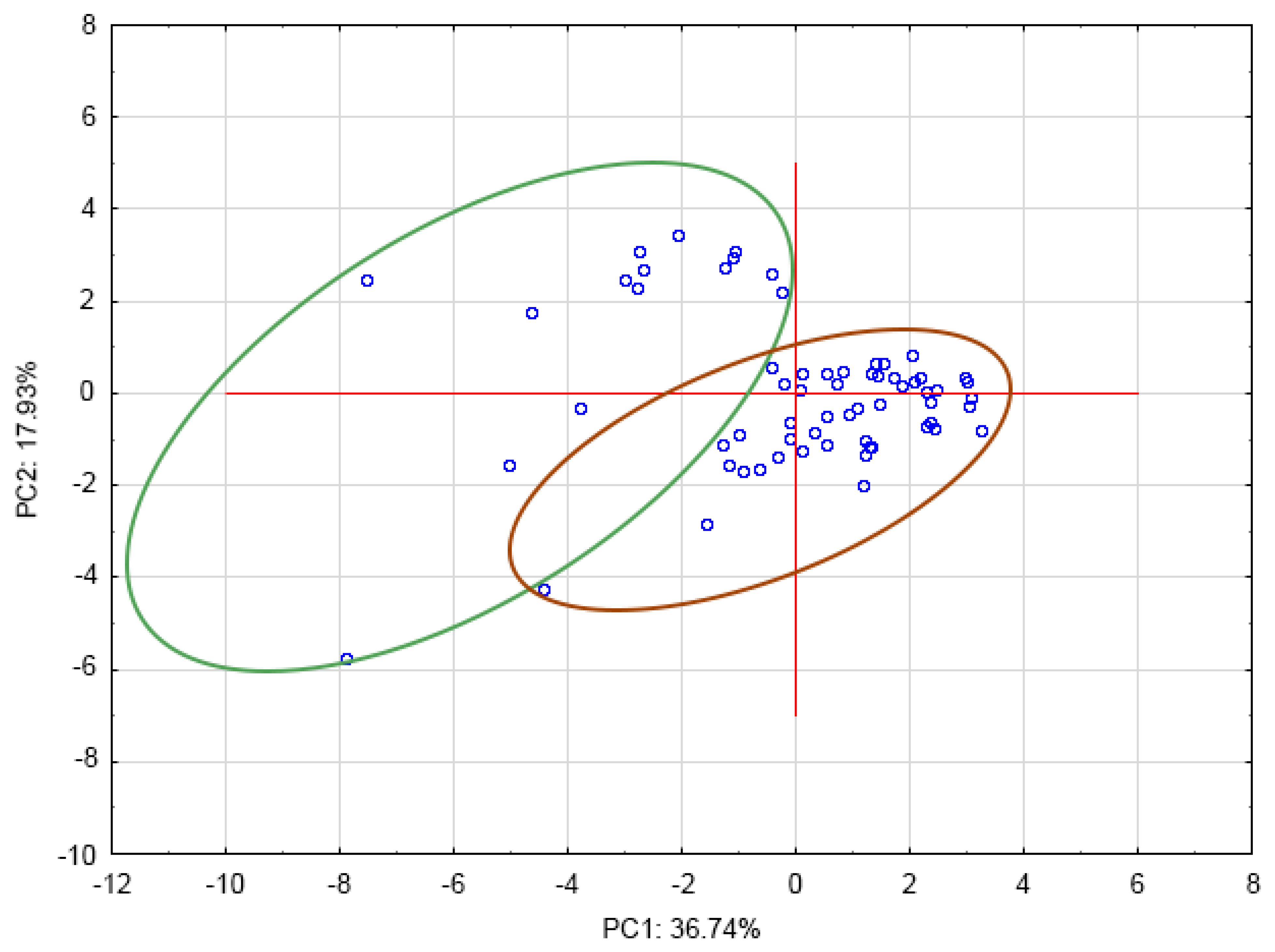
3.6. Principal Component Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shao, C.; Tang, H.; Wang, X.; He, J. Coffee consumption and stroke risk: Evidence from a systematic review and meta-analysis of more than 2.4 Million men and women. J. Stroke Cerebrovasc. Dis. 2021, 30, 105452. [Google Scholar] [CrossRef] [PubMed]
- Abe, S.K.; Saito, E.; Sawada, N.; Tsugane, S.; Ito, H.; Lin, Y.; Inoue, M. Coffee consumption and mortality in Japanese men and women: A pooled analysis of eight population-based cohort studies in Japan (Japan Cohort Consortium). Prev. Med. 2019, 123, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, T.; Bors, W.; Stettmaier, K. Studies on radical intermediates in the early stage of the nonenzymatic browning reaction of carbohydrates and amino acids. J. Agric. Food Chem. 1999, 47, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Clifford, M.N. Chlorogenic acids and other cinnamates—Nature, occurrence, dietary burden, absorption and metabolism. J. Sci. Food Agric. 2000, 80, 1033–1043. [Google Scholar] [CrossRef]
- Jaiswal, R.; Matei, M.F.; Golon, A.; Witt, M.; Kuhnert, N. Understanding the fate of chlorogenic acids in coffee roasting using mass spectrometry based targeted and non-targeted analytical strategies. Food Funct. 2012, 3, 976–984. [Google Scholar] [CrossRef]
- Farah, A.; de Paulis, T.; Moreira, D.P.; Trugo, L.C.; Martin, P.R. Chlorogenic acids and lactones in regular and water-decaffeinated Arabica coffees. J. Agric. Food Chem. 2006, 54, 374–381. [Google Scholar] [CrossRef]
- Jeszka-Skowron, M.; Frankowski, R.; Zgoła-Grześkowiak, A. Comparison of methylxantines, trigonelline, nicotinic acid and nicotinamide contents in brews of green and processed Arabica and Robusta coffee beans—Influence of steaming, decaffeination and roasting processes on coffee beans. LWT-Food Sci. Technol. 2020, 125, 109344. [Google Scholar] [CrossRef]
- Jumhawan, U.; Putri, S.P.; Yusianto; Bamba, T.; Fukusaki, E. Quantification of coffee blends for authentication of Asian palm civet coffee (Kopi Luwak) via metabolomics: A proof of concept. J. Biosci. Bioeng. 2016, 122, 79–84. [Google Scholar] [CrossRef]
- Angeloni, G.; Guerrini, L.; Masella, P.; Bellumori, M.; Daluiso, S.; Parenti, A.; Innocenti, M. What kind of coffee do you drink? An investigation on effects of eight different extraction methods. Food Res. Int. 2019, 116, 1327–1337. [Google Scholar] [CrossRef]
- Ludwig, I.A.; Mena, P.; Calani, L.; Cid, C.; Del Rio, D.; Leand, M.E.J.; Crozier, A. Variations in caffeine and chlorogenic acid contents of coffees: What are we drinking? Food Funct. 2014, 5, 1718–1726. [Google Scholar] [CrossRef]
- Farah, A.; de Paula Lima, J. Consumption of chlorogenic acids through coffee and health implications. Beverages 2019, 5, 11. [Google Scholar] [CrossRef]
- Jeszka-Skowron, M.; Frankowski, R.; Zgoła-Grześkowiak, A. Influence of coffee bean processing on the phenolic acid and flavonoid content in coffee brews (Coffea arabica and Coffea robusta). In Coffee Science: Biotechnological Advances, Economics and Health Benefits; Ramakrishna, A., Giridhar, P., Jeszka-Skowron, M., Eds.; CRC Press, Taylor Francis Group: Boca Raton, FL, USA, 2022; pp. 139–144. [Google Scholar]
- Król, K.; Gantner, M.; Tatarak, A.; Hallmann, E. The content of polyphenols in coffee beans as roasting, origin and storage effect. Eur. Food Res. Technol. 2020, 246, 33–39. [Google Scholar] [CrossRef]
- Angeloni, S.; Navarini, L.; Khamitova, G.; Sagratini, G.; Vittori, S.; Caprioli, G. Quantification of lignans in 30 ground coffee samples and evaluation of theirs extraction yield in espresso coffee by HPLC-MS/MS triple quadrupole. Int. J. Food Sci. Nutr. 2020, 71, 193–200. [Google Scholar] [CrossRef]
- Perrone, D.; Donangelo, C.M.; Farah, A. Fast simultaneous analysis of caffeine, trigonelline, nicotinic acid and sucrose in coffee by liquid chromatography–mass spectrometry. Food Chem. 2008, 110, 1030–1035. [Google Scholar] [CrossRef]
- Oliveira, S.D.; Franca, A.S.; Glória, M.B.A.; Borges, M.L.A. The effect of roasting on the presence of bioactive amines in coffees of different qualities. Food Chem. 2005, 90, 287–291. [Google Scholar] [CrossRef]
- Halász, A.; Baráth, Á.; Simon-Sarkadi, L.; Holzapfel, W. Biogenic amines and their production by microorganisms in food. Trends Food Sci. Technol. 1994, 5, 42–49. [Google Scholar] [CrossRef]
- Cheng, S.; Fu, X.; Wang, X.; Liao, Y.; Zeng, L.; Dong, F.; Yang, Z. Studies on the biochemical formation pathway of the amino acid l-theanine in tea (Camellia sinensis) and other plants. J. Agric. Food Chem. 2017, 65, 7210–7216. [Google Scholar] [CrossRef]
- Jeszka-Skowron, M.; Sentkowska, A.; Pyrzyńska, K.; De Peña, M.P. Chlorogenic acids, caffeine content and antioxidant properties of green coffee extracts: Influence of green coffee bean preparation. Eur. Food Res. Technol. 2016, 242, 1403–1409. [Google Scholar] [CrossRef]
- Jeszka-Skowron, M.; Stanisz, E.; De Peña, M.P. Relationship between antioxidant activity, chlorogenic acids and elemental composition of green coffee. LWT-Food Sci. Technol. 2016, 73, 243–250. [Google Scholar] [CrossRef]
- Lee, L.W.; Cheong, M.W.; Curran, P.; Yu, B.; Liu, S.Q. Coffee fermentation and flavor—An intricate and delicate relationship. Food Chem. 2015, 185, 182–191. [Google Scholar] [CrossRef]
- Perrone, D.; Farah, A.; Donangelo, C.M.; de Paulis, T.; Martin, P.R. Comprehensive analysis of major and minor chlorogenic acids and lactones in economically relevant Brazilian coffee cultivars. Food Chem. 2008, 106, 859–867. [Google Scholar] [CrossRef]
- Liang, N.; Xue, W.; Kennepohl, P.; Kitts, D.D. Interactions between major chlorogenic acid isomers and chemical changes in coffee brew that affect antioxidant activities. Food Chem. 2016, 213, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Farah, A.; de Paula Lima, J. Major chlorogenic acids’ contents and distribution in coffees. In Coffee: Production, Quality and Chemistry; Farah, A., Ed.; Royal Society of Chemistry: London, UK, 2019; pp. 584–610. [Google Scholar]
- Fujioka, K.; Shibamoto, T. Chlorogenic acid and caffeine contents in various commercial brewed coffees. Food Chem. 2008, 106, 217–221. [Google Scholar] [CrossRef]
- Deshpande, S.; Jaiswal, R.; Matei, M.F.; Kuhnert, N. Investigation of acyl migration in mono- and dicaffeoylquinic acids under aqueous basic, aqueous acidic, and dry roasting conditions. J. Agric. Food Chem. 2014, 62, 9160–9170. [Google Scholar] [CrossRef] [PubMed]
- Kulapichitr, F.; Borompichaichartkul, C.; Fang, M.; Suppavorasatit, I.; Cadwallader, K.R. Effect of post-harvest drying process on chlorogenic acids, antioxidant activities and CIE-Lab color of Thai Arabica green coffee beans. Food Chem. 2022, 366, 130504. [Google Scholar] [CrossRef] [PubMed]
- Lafay, S.; Gil-Izquierdo, A.; Manach, C.; Morand, C.; Besson, C.; Scalbert, A. Chlorogenic acid is absorbed in its intact form in the stomach of rats. J. Nutr. 2006, 136, 1192–1197. [Google Scholar] [CrossRef]
- Rodrigues, N.P.; Bragagnolo, N. Identification and quantification of bioactive compounds in coffee brews by HPLC-DAD-MSn. J. Food Compos. Anal. 2013, 32, 105–115. [Google Scholar] [CrossRef]
- Febrina, L.; Happyana, N.; Syah, Y.M. Metabolite profiles and antidiabetic activity of the green beans of Luwak (civet) coffees. Food Chem. 2021, 355, 129496. [Google Scholar] [CrossRef]
- Caprioli, G.; Cortese, M.; Maggi, F.; Minnetti, C.; Odello, L.; Sagratini, G.; Vittori, S. Quantification of caffeine, trigonelline and nicotinic acid in espresso coffee: The influence of espresso machines and coffee cultivars. Int. J. Food Sci. Nutr. 2014, 65, 465–469. [Google Scholar] [CrossRef]
- Kitzberger, C.S.G.; dos Santos Scholz, M.B.; de Toledo Benassi, M. Bioactive compounds content in roasted coffee from traditional and modern Coffea arabica cultivars grown under the same edapho-climatic conditions. Food Res. Int. 2014, 61, 61–66. [Google Scholar] [CrossRef]
- Kocadağlı, T.; Yılmaz, C.; Gökmen, V. Determination of melatonin and its isomer in foods by liquid chromatography tandem mass spectrometry. Food Chem. 2014, 153, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Dias, E.C.; Pereira, R.G.F.A.; Borém, F.M.; Mendes, E.; de Lima, R.R.; Fernandes, J.O.; Casal, S. Biogenic amine profile in unripe arabica coffee beans processed according to dry and wet methods. J. Agric. Food Chem. 2012, 60, 4120–4125. [Google Scholar] [CrossRef] [PubMed]
- Restuccia, D.; Spizzirri, U.G.; Parisi, O.I.; Cirillo, G.; Picci, N. Brewing effect on levels of biogenic amines in different coffee samples as determined by LC-UV. Food Chem. 2015, 175, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Casal, S.; Oliveira, M.B.P.P.; Ferreira, M.A. Determination of biogenic amines in coffee by an optimized liquid chromatographic method. J. Liq. Chromatogr. Relat. Technol. 2002, 25, 2535–2549. [Google Scholar] [CrossRef]
- Casal, S.; Mendes, E.; Alves, M.R.; Alves, R.C.; Beatriz, M.; Oliveira, P.P.; Ferreira, M.A. Free and conjugated biogenic amines in green and roasted coffee beans. J. Agric. Food Chem. 2004, 52, 6188–6192. [Google Scholar] [CrossRef]
- Özdestan, Ö. Evaluation of bioactive amine and mineral levels in Turkish coffee. Food Res. Int. 2014, 61, 167–175. [Google Scholar] [CrossRef]
- Servillo, L.; Giovane, A.; Casale, R.; Cautela, D.; D’Onofrio, N.; Balestrieri, M.L.; Castaldo, D. Glucosylated forms of serotonin and tryptophan in green coffee beans. LWT-Food Sci. Technol. 2016, 73, 117–122. [Google Scholar] [CrossRef]
- Górnaś, P.; Dwiecki, K.; Siger, A.; Tomaszewska-Gras, J.; Michala, M.; Polewski, K. Contribution of phenolic acids isolated from green and roasted boiled-type coffee brews to total coffee antioxidant capacity. Eur. Food Res. Technol. 2016, 242, 641–653. [Google Scholar] [CrossRef]
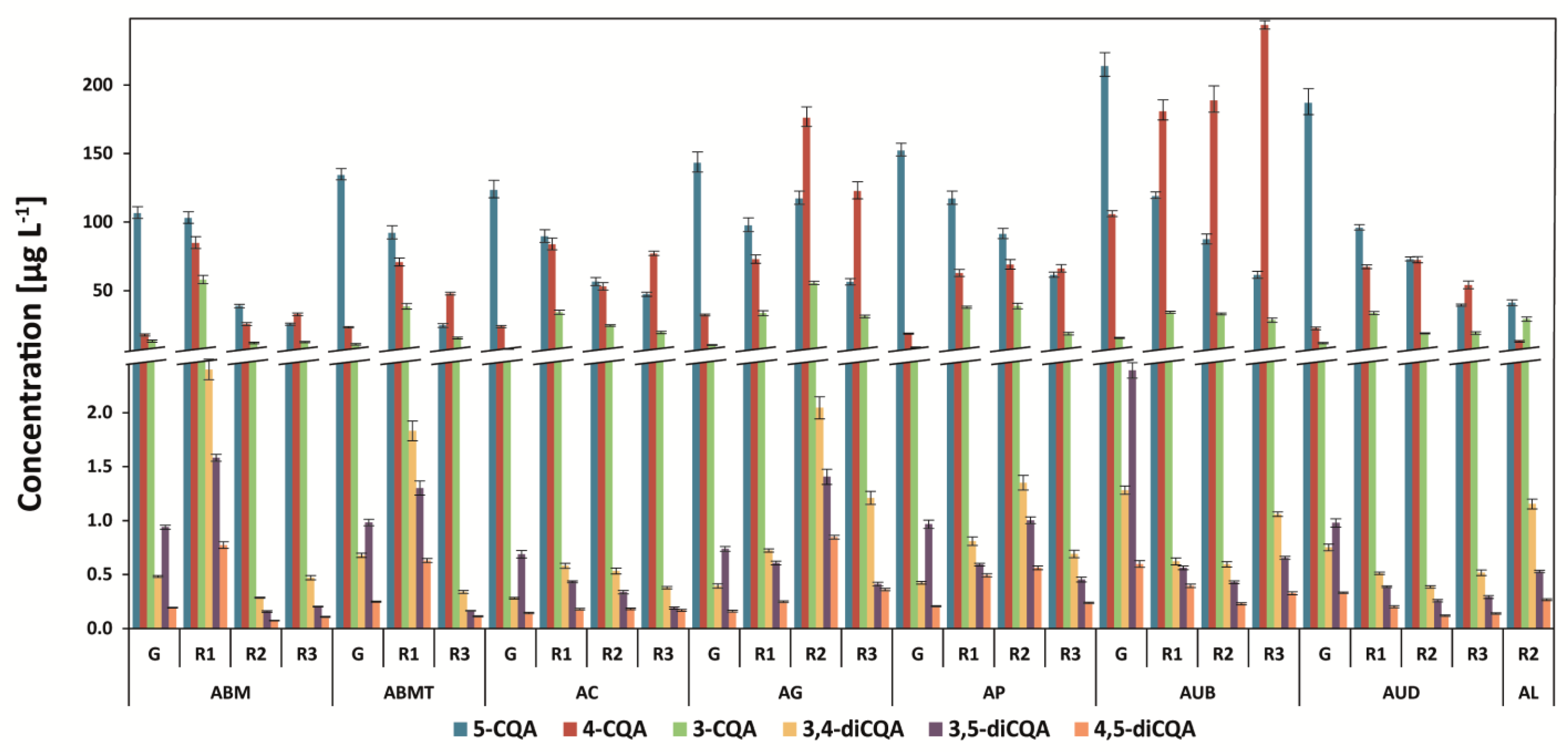
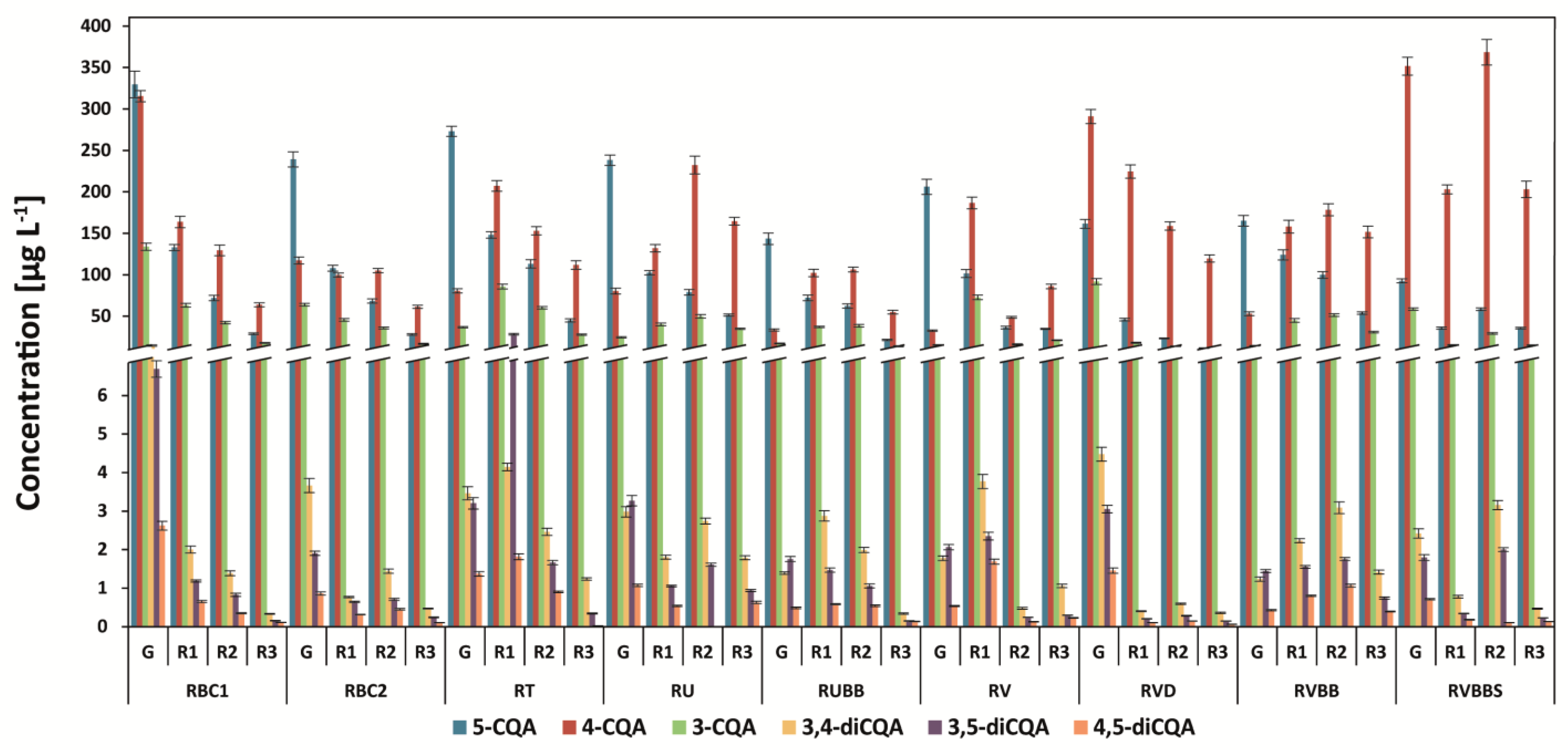
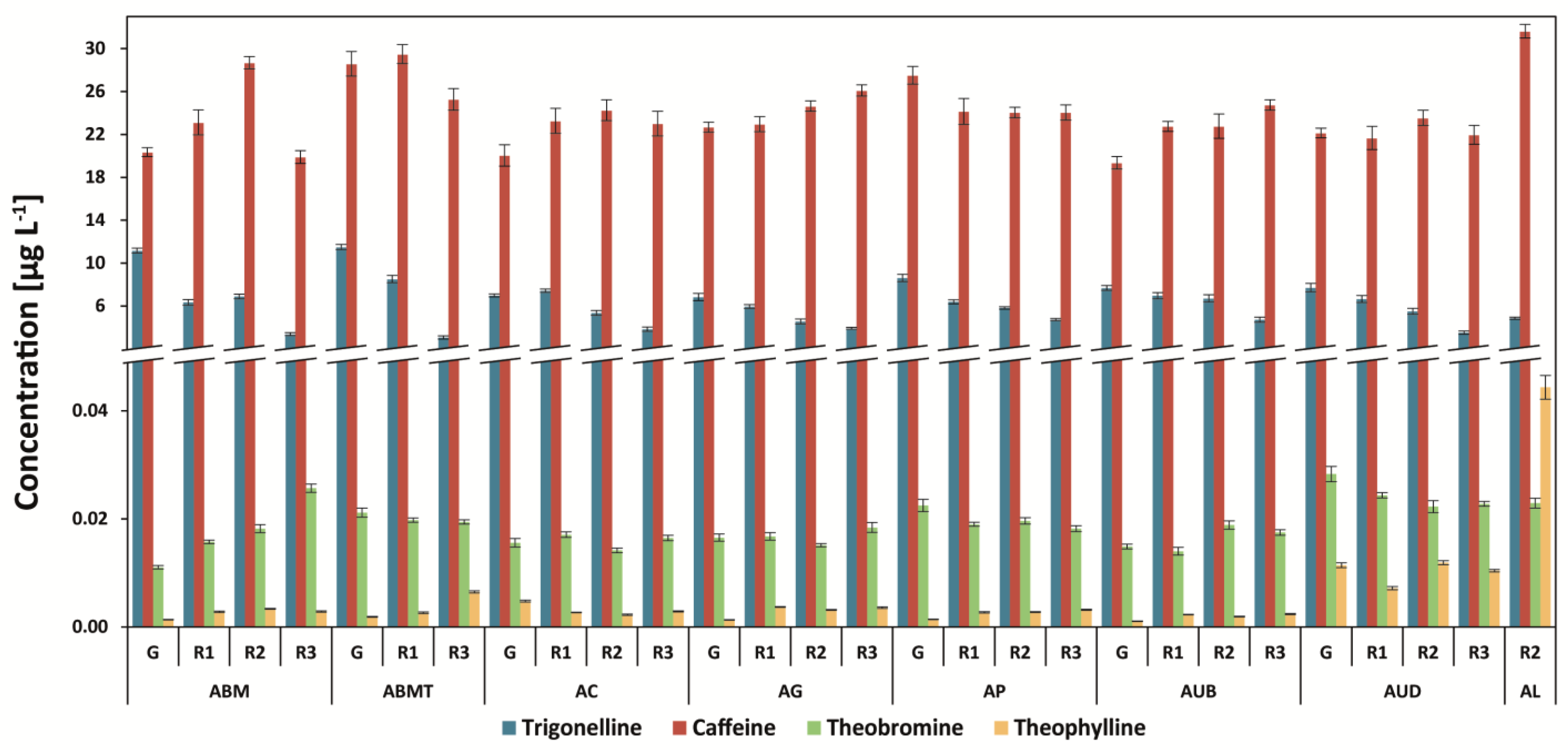
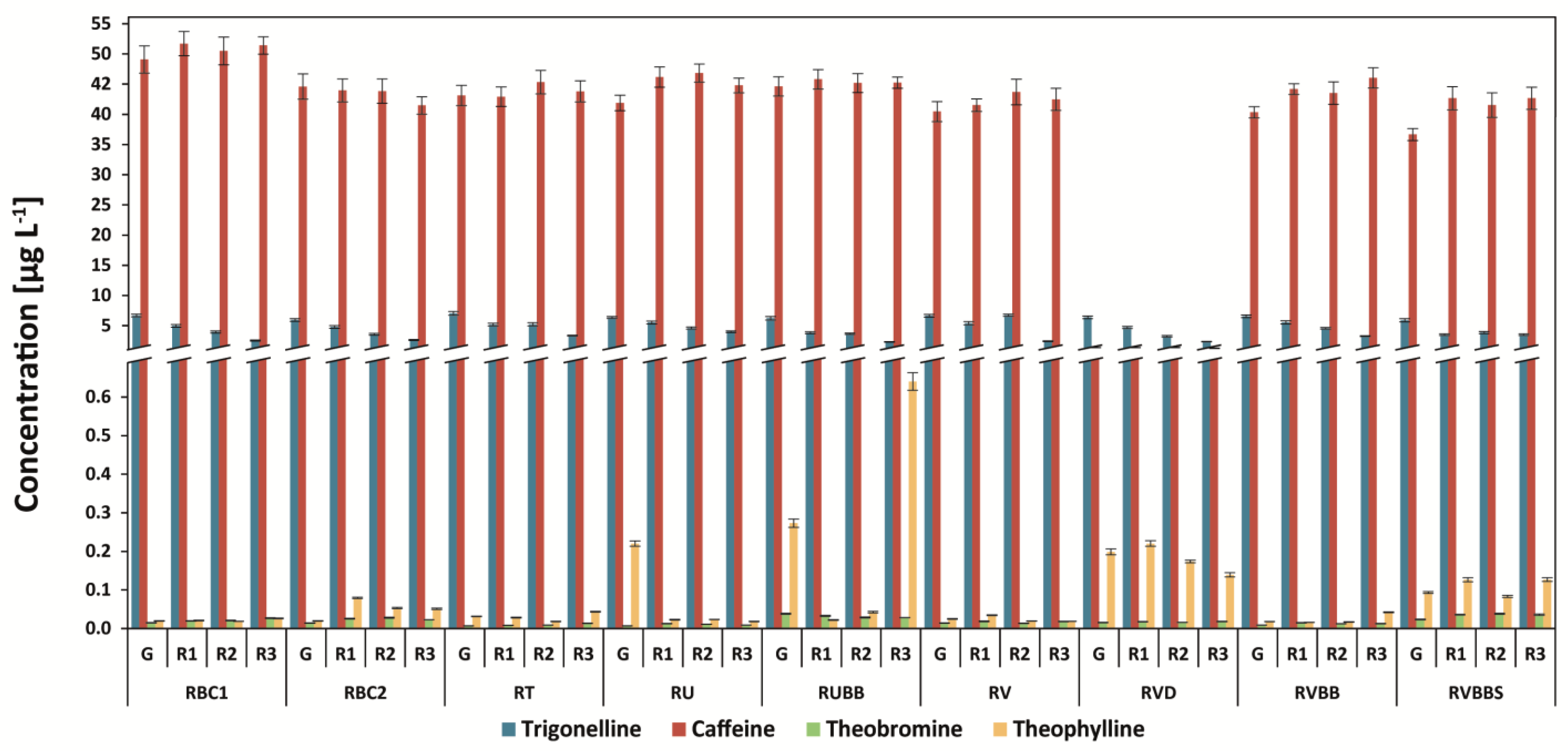
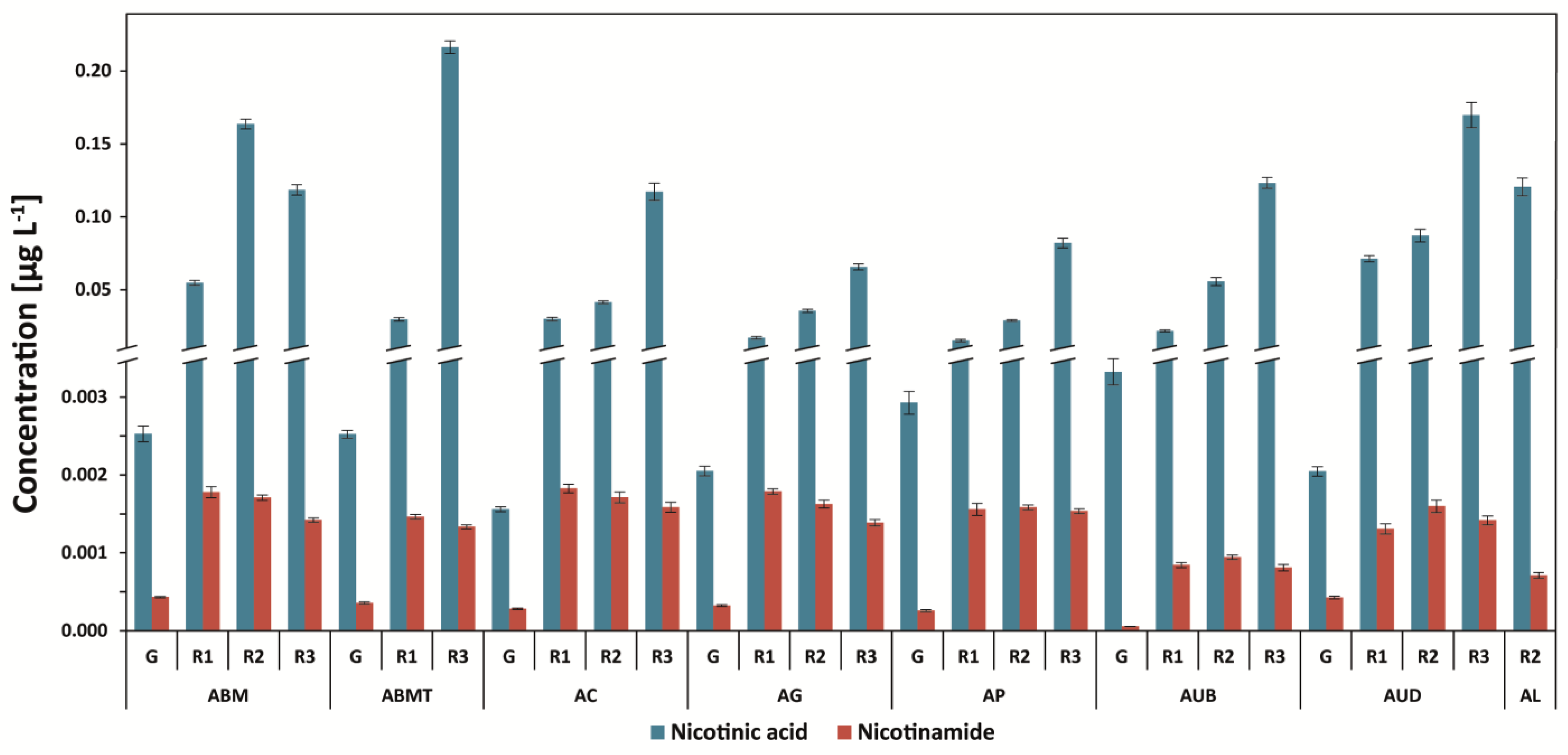
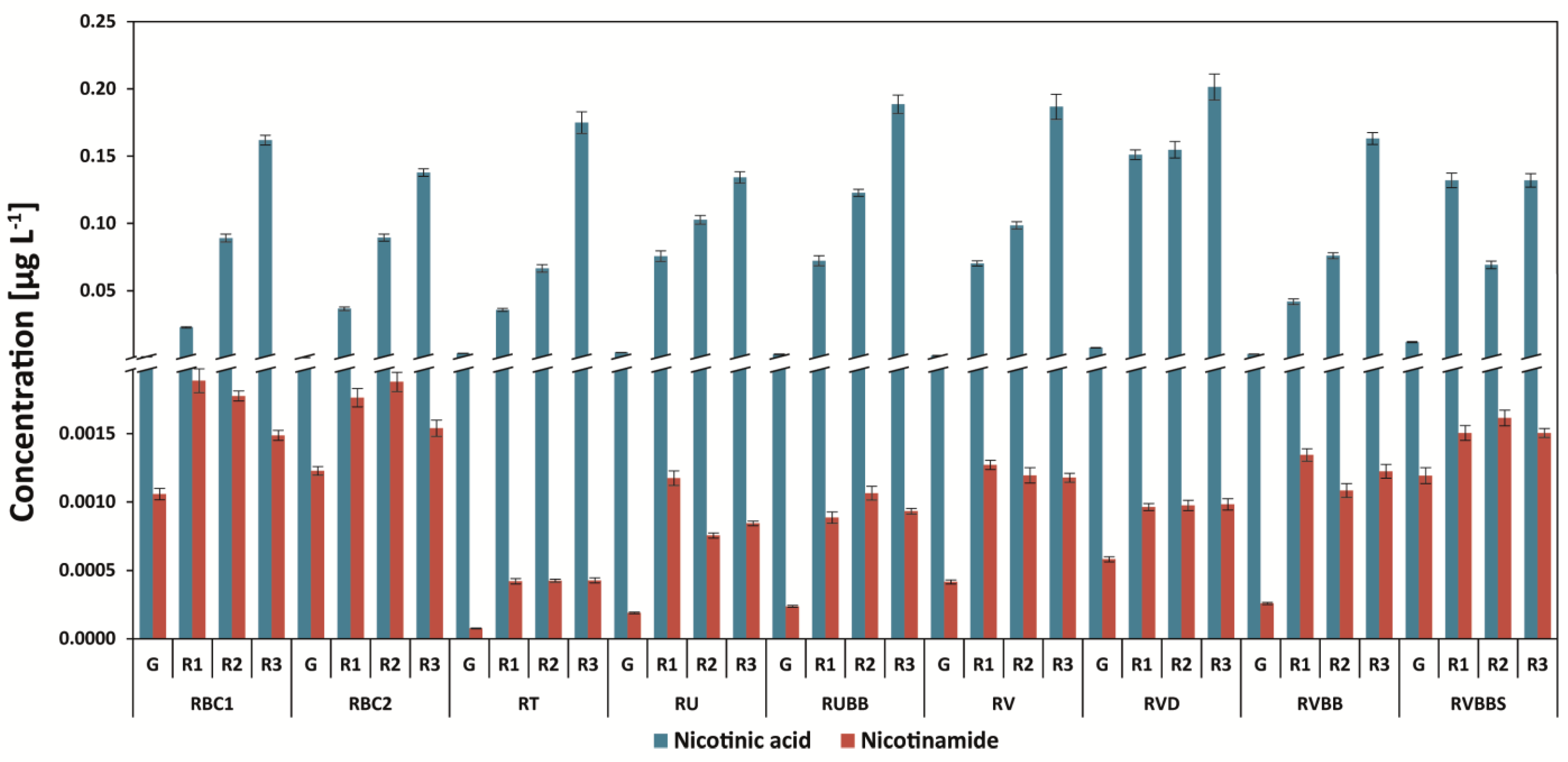
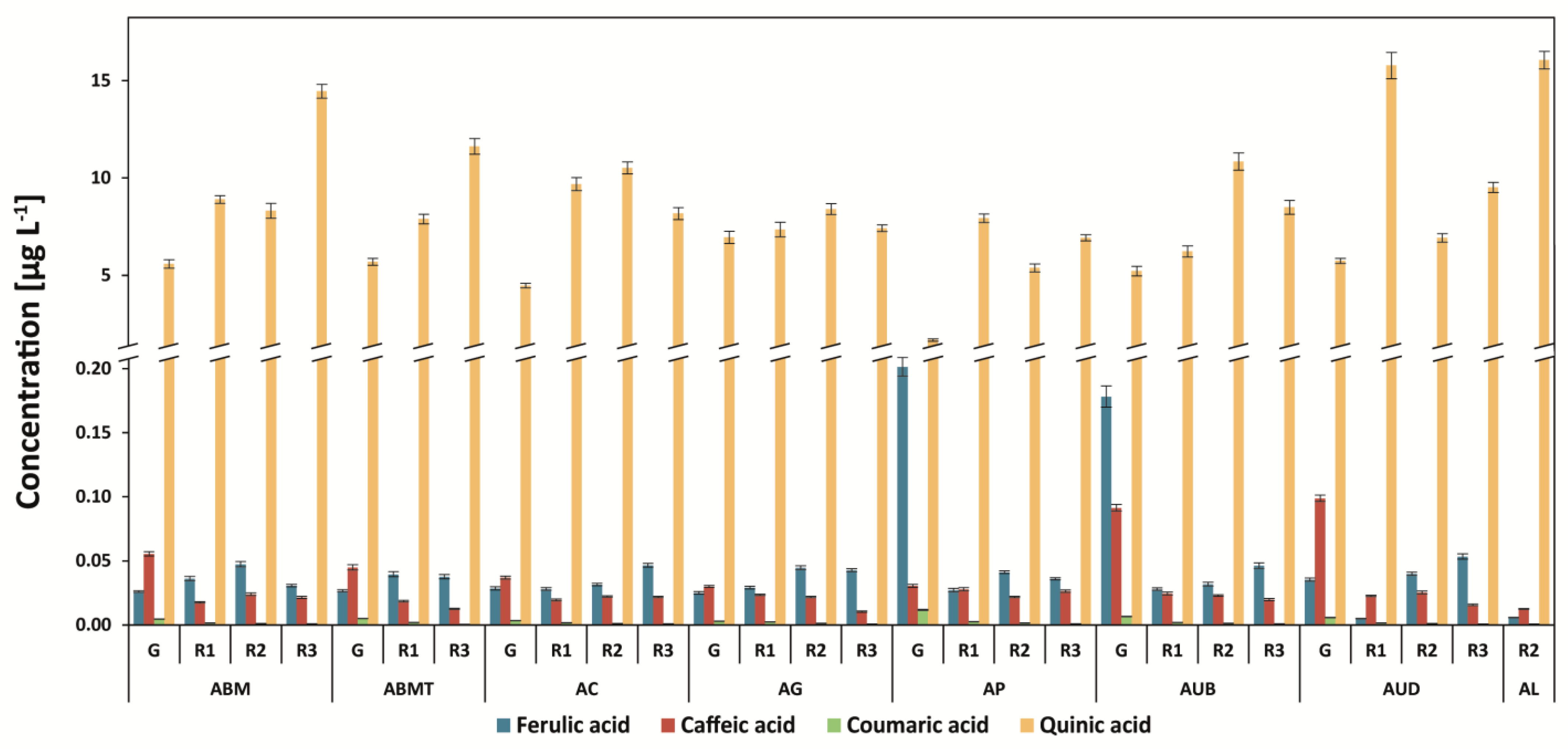
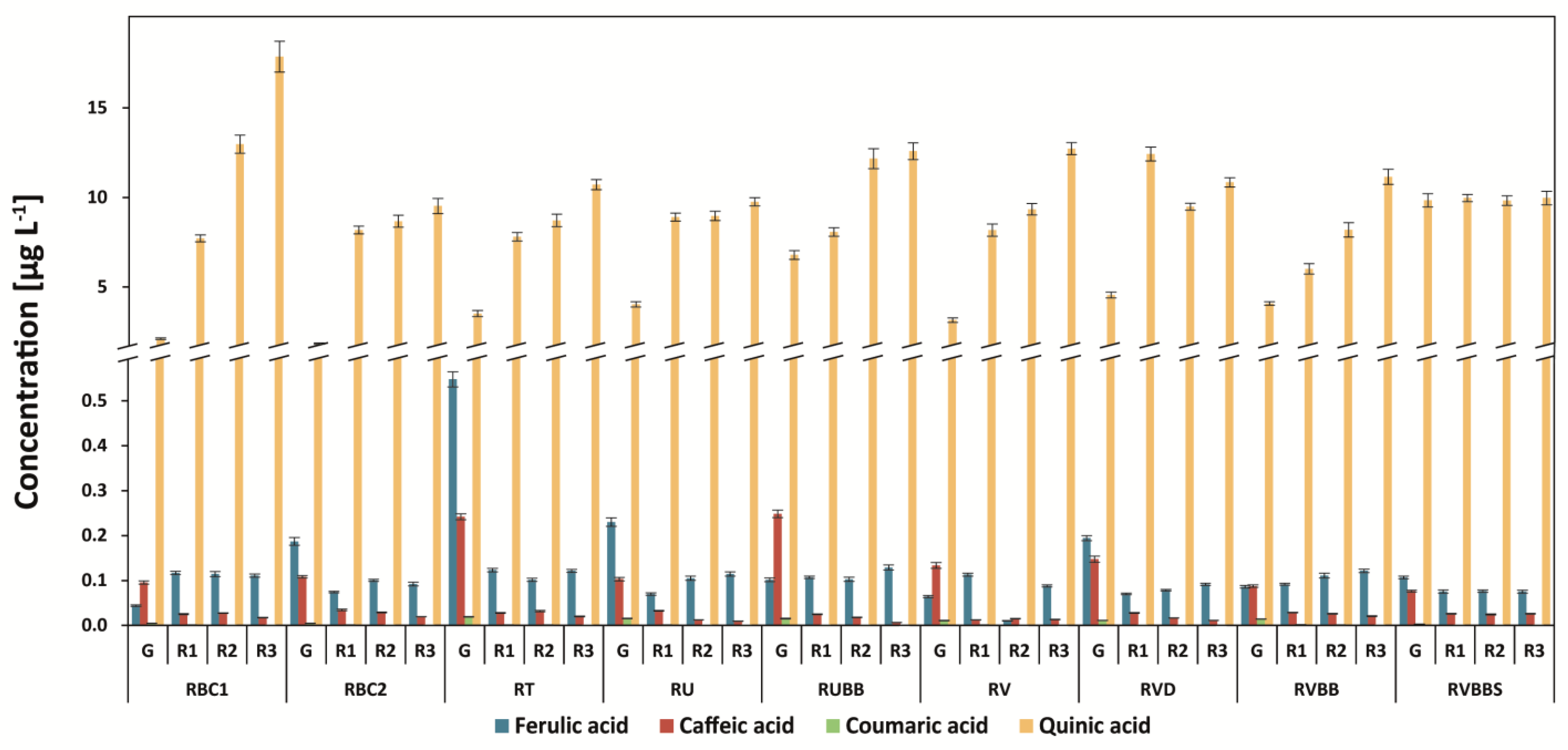
| Abbr. | Origin of Coffee | Washed/Unwashed | Detailed Information |
|---|---|---|---|
| Coffee arabica | |||
| ABM | Brazil São Paulo, region: Mogiana | Unwashed coffee | Mogiana TF-–type New York 2/3 Fancy–no black beans allowed |
| ABMT | Brazil MTGB | Unwashed coffee | MTGB—medium to good beans; Flat beans-screen 15/16 |
| AC | Colombia | Fully washed coffee | Excelso, screen 15 |
| AG | Guatemala | Washed coffee | SHB—strictly hard beans 1600/1700 m The “Strictly Hard Bean” coffees—the best in the world: complete, full-bodied taste, acid and fragrant cup. |
| AP | Papua New Guinea | Washed coffee | Grows at altitudes of 1000 m, |
| AUB | Uganda, region: Bugishu | Fully washed coffee | 1300–2300 m above sea Grows mostly on the high slopes of Mount Elgon—to the East (the Sipi region), along the Kenyan border |
| AUD | Uganda, region: Drugar | Unwashed coffee | West—alongside the mountain border with Congo |
| AL | Bali, Indonesia | Natural fermentation in animal gut | Kopi Luwak Degree of roasting: city–full city |
| Coffee robusta | |||
| RBC1 | Brazil Conilon 5/6 | Unwashed coffee | Type 5/6—screen 13 small beans; max 100 defects |
| RBC2 | Brazil Conilon 6/7 | Unwashed coffee | Type 6/7; max 200 defects |
| RT | Tanzania sc. 14 | Unwashed coffee | Screen 14—maximum 5% below screen 14 Dried in cherry in the sun and later milled to remove the outer hard skin and the husks |
| RU | Uganda sc. 12 | Unwashed coffee | 900–1500 meters in a radius around Lake Victoria as well as in the western regions. Screen 12—dry-processed beans—no more than 20% of defective beans and not more than one-tenth of 1% by the weight of the extraneous matter shall have a maximum moisture content of 12.5% |
| RUBB | Uganda BB | Unwashed coffee | 900–1500 meters in a radius around Lake Victoria as well as in the western regions; BB—Black Beans—black and discolored coffee beans, separated from clean coffee electronically or by hand |
| RV | Vietnam sc. 16 clean | Unwashed coffee | Grade 1: > 7 mm bean size |
| RVD | Vietnam sc. 16 clean decaf | Unwashed coffee | Grade 1: > 7 mm bean size, decaffeinated coffee beans |
| RVBB | Vietnam gr. 2, 5% BB | Unwashed coffee | Grade 2: Black beans and broken 2–3%, bean size > 6–7 mm |
| RVBBS | Vietnam gr. 2, 5% BB, Steamed | Unwashed coffee | Grade 2: Black beans and broken 2–3%, bean size > 6–7 mm Steamed beans |
| Coffee 1 | Unroasted–Green (G) /Roasting Degree (R1–R3; Light, Medium, Dark) | Theanine | Melatonin | Serotonin |
|---|---|---|---|---|
| Coffea arabica | ||||
| ABM | G | 5.27 ± 0.21 | 0.0006 ± 0.0001 | 15.48 ± 0.13 |
| R1 | 0.09 ± 0.01 | 0.0030 ± 0.0003 | 0.13 ± 0.02 | |
| R2 | 0.16 ± 0.02 | 0.0051 ± 0.0004 | 0.05 ± 0.01 | |
| R3 | 0.12 ± 0.02 | 0.0027 ± 0.0002 | 0.09 ± 0.02 | |
| ABMT | G | 3.64 ± 0.09 | 0.0009 ± 0.0001 | 9.96 ± 0.11 |
| R1 | 0.06 ± 0.01 | 0.0036 ± 0.0003 | 0.13 ± 0.03 | |
| R2 | 0.08 ± 0.01 | 0.0038 ± 0.0003 | 0.03 ± 0.01 | |
| R3 | 0.15 ± 0.02 | 0.0072 ± 0.0005 | 0.09 ± 0.01 | |
| AC | G | 3.57 ± 0.09 | 0.0014 ± 0.0001 | 20.67 ± 1.01 |
| R1 | 0.05 ± 0.01 | 0.0028 ± 0.0003 | 0.20 ± 0.02 | |
| R2 | 0.03 ± 0.01 | 0.0019 ± 0.0002 | 0.11 ± 0.01 | |
| R3 | 0.29 ± 0.03 | 0.0022 ± 0.0002 | 0.10 ± 0.01 | |
| AG | G | 3.87 ± 0.09 | 0.0006 ± 0.0001 | 22.41 ± 1.21 |
| R1 | 0.09 ± 0.01 | 0.0015 ± 0.0002 | 0.25 ± 0.03 | |
| R2 | 0.03 ± 0.01 | 0.0064 ± 0.0005 | 0.16 ± 0.02 | |
| R3 | 0.11 ± 0.01 | 0.0053 ± 0.0005 | 0.10 ± 0.01 | |
| AP | G | 4.68 ± 0.12 | 0.0009 ± 0.0001 | 33.26 ± 1.13 |
| R1 | 0.04 ± 0.01 | 0.0019 ± 0.0002 | 0.23 ± 0.02 | |
| R2 | 0.06 ± 0.01 | 0.0019 ± 0.0002 | 0.13 ± 0.01 | |
| R3 | 0.10 ± 0.01 | 0.0023 ± 0.0002 | 0.10 ± 0.01 | |
| AUB | G | 2.08 ± 0.08 | 0.0016 ± 0.0002 | 13.67 ± 0.74 |
| R1 | 0.04 ± 0.01 | 0.0018 ± 0.0002 | 0.16 ± 0.01 | |
| R2 | 0.03 ± 0.01 | 0.0014 ± 0.0001 | 0.10 ± 0.01 | |
| R3 | 0.03 ± 0.01 | 0.0020 ± 0.0002 | 0.11 ± 0.01 | |
| AUD | G | 6.34 ± 0.13 | 0.0024 ± 0.0002 | 31.15 ± 1.23 |
| R1 | 0.06 ± 0.01 | 0.0034 ± 0.0002 | 0.15 ± 0.03 | |
| R2 | 0.11 ± 0.01 | 0.0018 ± 0.0001 | 0.13 ± 0.01 | |
| R3 | 0.18 ± 0.02 | 0.0016 ± 0.0001 | 0.09 ± 0.01 | |
| AL | R2 | 0.06 ± 0.01 | 0.0038 ± 0.0002 | 0.06 ± 0.01 |
| Coffea robusta | ||||
| RBC1 | G | 6.60 ± 0.17 | 0.0014 ± 0.0002 | 16.22 ± 0.97 |
| R1 | 0.15 ± 0.01 | 0.0038 ± 0.0003 | 0.13 ± 0.01 | |
| R2 | 0.13 ± 0.01 | 0.0120 ± 0.0009 | 0.07 ± 0.01 | |
| R3 | 0.18 ± 0.02 | 0.0039 ± 0.0002 | 0.07 ± 0.01 | |
| RBC2 | G | 8.73 ± 0.41 | 0.0039 ± 0.0002 | 28.11 ± 1.21 |
| R1 | 0.09 ± 0.01 | 0.0040 ± 0.0002 | 0.11 ± 0.01 | |
| R2 | 0.40 ± 0.08 | 0.0044 ± 0.0003 | 0.12 ± 0.01 | |
| R3 | 0.08 ± 0.01 | 0.0026 ± 0.0002 | 0.10 ± 0.01 | |
| RT | G | 2.75± 0.11 | 0.0072 ± 0.0003 | 18.52 ± 0.87 |
| R1 | 0.20 ± 0.01 | 0.0025 ± 0.0002 | 0.21 ± 0.02 | |
| R2 | 0.12 ± 0.01 | 0.0039 ± 0.0002 | 0.13 ± 0.01 | |
| R3 | 0.11 ± 0.01 | 0.0052 ± 0.0003 | 0.11 ± 0.01 | |
| RU | G | 7.39 ± 0.18 | 0.0053 ± 0.0002 | 19.52 ± 0.99 |
| R1 | 0.11 ± 0.01 | 0.0084 ± 0.0004 | 0.22 ± 0.01 | |
| R2 | 0.08 ± 0.01 | 0.0060 ± 0.0003 | 0.16 ± 0.02 | |
| R3 | 0.63 ± 0.06 | 0.0035 ± 0.0002 | 0.11 ± 0.01 | |
| RUBB | G | 10.11 ± 0.19 | 0.0043 ± 0.0002 | 16.00 ± 0.87 |
| R1 | 0.91 ± 0.02 | 0.0036 ± 0.0002 | 0.08 ± 0.01 | |
| R2 | 0.14 ± 0.01 | 0.0017 ± 0.0002 | 0.10 ± 0.02 | |
| R3 | 1.34 ± 0.01 | 0.0039 ± 0.0002 | 0.09 ± 0.02 | |
| RV | G | 9.61 ± 0.21 | 0.0051 ± 0.0003 | 19.33 ± 0.98 |
| R1 | 0.65 ± 0.03 | 0.0092 ± 0.0004 | 0.09 ± 0.01 | |
| R2 | 0.39 ± 0.03 | 0.0021 ± 0.0002 | 0.23 ± 0.01 | |
| R3 | 0.19 ± 0.02 | 0.0065 ± 0.0004 | 0.12 ± 0.01 | |
| RVD | G | 7.79 ± 0.20 | 0.0025 ± 0.0002 | 0.90 ± 0.08 |
| R1 | 0.26 ± 0.03 | 0.0044 ± 0.0003 | 0.04 ± 0.01 | |
| R2 | 0.16 ± 0.02 | 0.0031± 0.0002 | 0.04 ± 0.01 | |
| R3 | 0.15 ± 0.02 | 0.0024 ± 0.0002 | 0.04 ± 0.01 | |
| RVBB | G | 8.63 ± 0.24 | 0.0061 ± 0.0005 | 19.67 ± 1.12 |
| R1 | 0.12 ± 0.02 | 0.0028 ± 0.0002 | 0.25 ± 0.03 | |
| R2 | 0.08 ± 0.01 | 0.0054 ± 0.0004 | 0.22 ± 0.03 | |
| R3 | 0.40 ± 0.01 | 0.0038 ± 0.0003 | 0.17 ± 0.02 | |
| RVBBS | G | 0.27 ± 0.03 | 0.0018 ± 0.0002 | 0.20 ± 0.02 |
| R1 | 0.10 ± 0.01 | 0.0015 ± 0.0002 | 0.05 ± 0.01 | |
| R2 | 0.14 ± 0.01 | 0.0035 ± 0.0003 | 0.08 ± 0.01 | |
| R3 | 0.19 ± 0.02 | 0.0024 ± 0.0002 | 0.05 ± 0.01 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jeszka-Skowron, M.; Frankowski, R.; Zgoła-Grześkowiak, A.; Płatkiewicz, J. Comprehensive Analysis of Metabolites in Brews Prepared from Naturally and Technologically Treated Coffee Beans. Antioxidants 2023, 12, 95. https://doi.org/10.3390/antiox12010095
Jeszka-Skowron M, Frankowski R, Zgoła-Grześkowiak A, Płatkiewicz J. Comprehensive Analysis of Metabolites in Brews Prepared from Naturally and Technologically Treated Coffee Beans. Antioxidants. 2023; 12(1):95. https://doi.org/10.3390/antiox12010095
Chicago/Turabian StyleJeszka-Skowron, Magdalena, Robert Frankowski, Agnieszka Zgoła-Grześkowiak, and Julia Płatkiewicz. 2023. "Comprehensive Analysis of Metabolites in Brews Prepared from Naturally and Technologically Treated Coffee Beans" Antioxidants 12, no. 1: 95. https://doi.org/10.3390/antiox12010095
APA StyleJeszka-Skowron, M., Frankowski, R., Zgoła-Grześkowiak, A., & Płatkiewicz, J. (2023). Comprehensive Analysis of Metabolites in Brews Prepared from Naturally and Technologically Treated Coffee Beans. Antioxidants, 12(1), 95. https://doi.org/10.3390/antiox12010095








