Antioxidant Phytochemicals as Potential Therapy for Diabetic Complications
Abstract
1. Introduction
2. Diabetic Complications
2.1. Diabetic Retinopathy (DR)
2.2. Diabetic Peripheral Neuropathy (DPN)
2.3. Diabetic Nephropathy (DN)
2.4. Diabetes-Induced Cardiovascular and Cerebrovascular Diseases
3. Implications of Oxidative Stress in Diabetic Complications
Hyperglycemia and Oxidative Stress
4. Enzymatic Endogenous Antioxidants and Diabetic Complications
4.1. Enzymatic Endogenous Systems Implicated in Diabetic Retinopathy
4.2. Endogenous Antioxidants in Diabetic Nephropathy (DN)
4.3. Endogenous Antioxidants in Diabetic Cardiomyopathy
4.4. Endogenous Antioxidants in Cerebrovascular Diabetic Complications
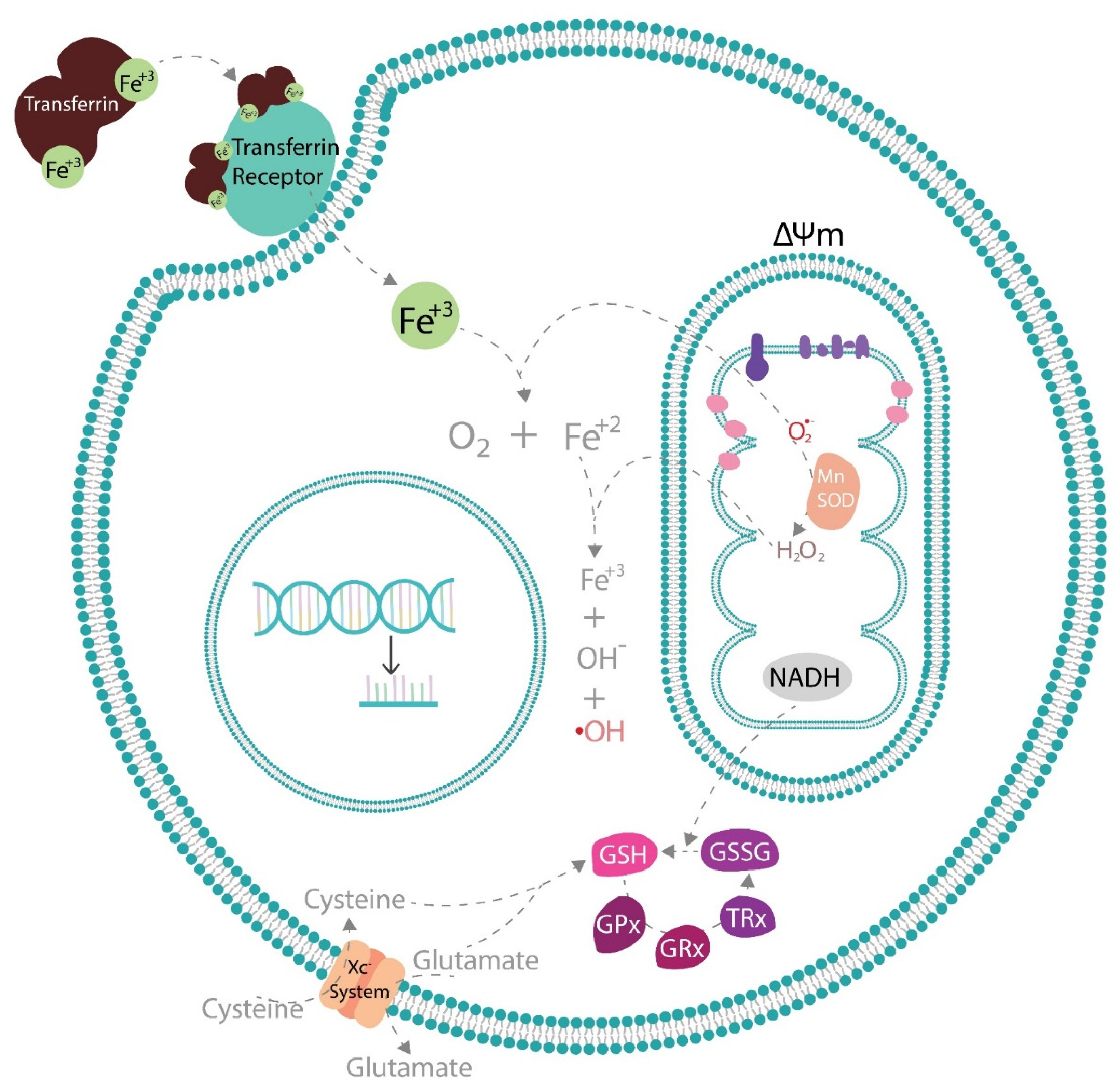
5. Effects of Antioxidant Phytochemicals on Endogenous Antioxidant Pathways
5.1. Effects of Antioxidant Phytochemicals on Mitochondrial Uncoupling Protein 2 (UCP2) and Mitochondrial Function in Diabetic Complications
5.2. Effects of Antioxidant Phytochemicals on Glutathione Peroxidase 4 (GPx4) and Coenzyme Q (CoQ) in Diabetic Complications and Ferroptosis
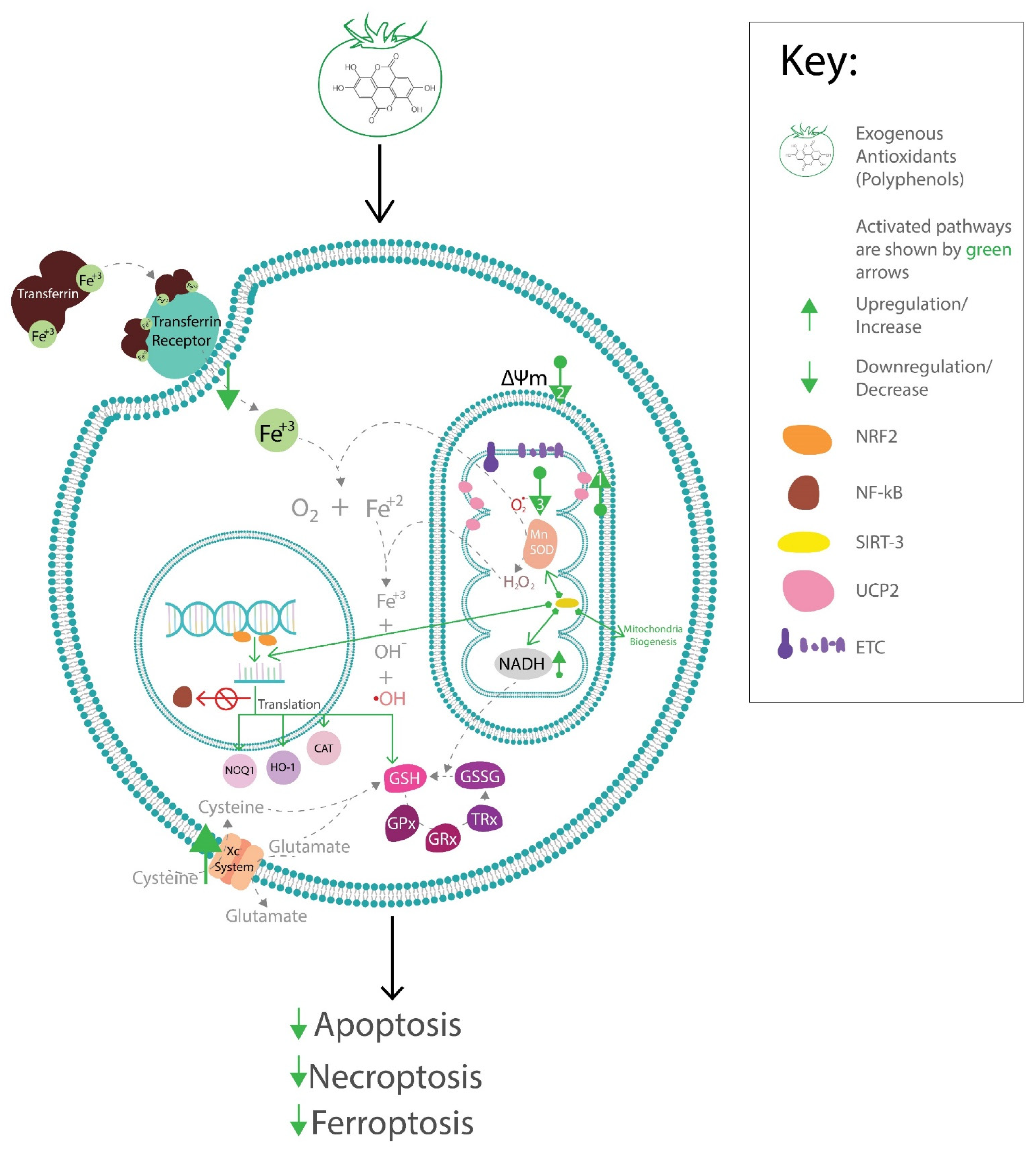
5.3. SIRTs and Polyphenols
5.4. Effects of Polyphenols on the Nuclear Factor (erythroid-derived 2)-like 2 (Nrf2) and Endogenous Antioxidant System
5.4.1. Nrf2 Regulation
5.4.2. Nrf2 in Diabetic Cardiomyopathy
5.4.3. Nrf2 in Diabetic Nephropathy
5.4.4. Nrf2 in Diabetic Retinopathy
5.4.5. Nrf2 in Diabetic Peripheral Neuropathy
5.4.6. Polyphenol and Endogenous Antioxidants Effect
6. Potential Therapeutic Strategies to Alleviate Diabetic Complications via the Activation of Endogenous Antioxidants by Exogenous Antioxidant Phytochemicals
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Misra, A.; Gopalan, H.; Jayawardena, R.; Hills, A.P.; Soares, M.; Reza-Albarrán, A.A.; Ramaiya, K.L. Diabetes in Developing Countries. J. Diabetes 2019, 11, 522–539. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.K.; Pearson-Stuttard, J.; Selvin, E.; Gregg, E.W. Interpreting Global Trends in Type 2 Diabetes Complications and Mortality. Diabetologia 2022, 65, 3–13. [Google Scholar] [CrossRef]
- Sun, H.; Saeedi, P.; Karuranga, S.; Pinkepank, M.; Ogurtsova, K.; Duncan, B.B.; Stein, C.; Basit, A.; Chan, J.C.N.; Mbanya, J.C. IDF Diabetes Atlas: Global, Regional and Country-Level Diabetes Prevalence Estimates for 2021 and Projections for 2045. Diabetes Res. Clin. Pract. 2021, 183, 109119. [Google Scholar] [CrossRef] [PubMed]
- Harding, J.L.; Pavkov, M.E.; Magliano, D.J.; Shaw, J.E.; Gregg, E.W. Global Trends in Diabetes Complications: A Review of Current Evidence. Diabetologia 2019, 62, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Jing, X.; Chen, J.; Dong, Y.; Han, D.; Zhao, H.; Wang, X.; Gao, F.; Li, C.; Cui, Z.; Liu, Y. Related Factors of Quality of Life of Type 2 Diabetes Patients: A Systematic Review and Meta-Analysis. Health Qual. Life Outcomes 2018, 16, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Hasler, C.M.; Blumberg, J.B. Phytochemicals: Biochemistry and Physiology. Introduction. J. Nutr. 1999, 129, 756S–757S. [Google Scholar] [CrossRef] [PubMed]
- Martinez, K.B.; Mackert, J.D.; McIntosh, M.K. Polyphenols and Intestinal Health. In Nutrition and Functional Foods for Healthy Aging; Elsevier: Amsterdam, The Netherlands, 2017; pp. 191–210. [Google Scholar]
- Koche, D.; Shirsat, R.; Kawale, M. An Overview of Major Classes of Phytochemicals: Their Types and Role in Disease Prevention. Hislopia J. 2016, 9, 976–2124. [Google Scholar]
- Ramawat, K.G.; Dass, S.; Mathur, M. The Chemical Diversity of Bioactive Molecules and Therapeutic Potential of Medicinal Plants. In Herbal Drugs: Ethnomedicine to Modern Medicine; Springer: Berlin, Germany, 2009; pp. 7–32. [Google Scholar]
- Oran, S.A.; Althaher, A.R.; Mubarak, M.S. Cinchona Officinalis (Cinchona Tree) and Corylus Avellana (Common Hazel), 1st ed.; Herbs, Shrubs, and Trees of Potential Medicinal Benefits; CRC Press: Boca Raton, FL, USA, 2022; ISBN 978-1-00-320506-7. [Google Scholar]
- Desborough, M.J.R.; Keeling, D.M. The Aspirin Story—From Willow to Wonder Drug. Br. J. Haematol. 2017, 177, 674–683. [Google Scholar] [CrossRef]
- Drummond, E.M.; Harbourne, N.; Marete, E.; Martyn, D.; Jacquier, J.; O’Riordan, D.; Gibney, E.R. Inhibition of Proinflammatory Biomarkers in THP1 Macrophages by Polyphenols Derived from Chamomile, Meadowsweet and Willow Bark. Phytother. Res. 2013, 27, 588–594. [Google Scholar] [CrossRef]
- Bharadwaj, K.C.; Gupta, T.; Singh, R.M. Chapter 9—Alkaloid Group of Cinchona Officinalis: Structural, Synthetic, and Medicinal Aspects. In Synthesis of Medicinal Agents from Plants; Tewari, A., Tiwari, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 205–227. ISBN 978-0-08-102071-5. [Google Scholar]
- Pietta, P.G. Flavonoids as Antioxidants. J. Nat. Prod. 2000, 63, 1035–1042. [Google Scholar] [CrossRef]
- Gu, R.; Wang, Y.; Long, B.; Kennelly, E.; Wu, S.; Liu, B.; Li, P.; Long, C. Prospecting for Bioactive Constituents from Traditional Medicinal Plants through Ethnobotanical Approaches. Biol. Pharm. Bull. 2014, 37, 903–915. [Google Scholar] [CrossRef] [PubMed]
- Hall, S.D.; Wang, Z.; Huang, S.-M.; Hamman, M.A.; Vasavada, N.; Adigun, A.Q.; Hilligoss, J.K.; Miller, M.; Gorski, J.C. The Interaction between St John’s Wort and an Oral Contraceptive. Clin. Pharmacol. Ther. 2003, 74, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Cade, W.T. Diabetes-Related Microvascular and Macrovascular Diseases in the Physical Therapy Setting. Phys. Ther. 2008, 88, 1322–1335. [Google Scholar] [CrossRef] [PubMed]
- Cheung, N.; Mitchell, P.; Wong, T.Y. Diabetic Retinopathy. Lancet 2010, 376, 124–136. [Google Scholar] [CrossRef] [PubMed]
- Biallosterski, C.; van Velthoven, M.E.J.; Michels, R.P.J.; Schlingemann, R.O.; DeVries, J.H.; Verbraak, F.D. Decreased Optical Coherence Tomography-Measured Pericentral Retinal Thickness in Patients with Diabetes Mellitus Type 1 with Minimal Diabetic Retinopathy. Br. J. Ophthalmol. 2007, 91, 1135–1138. [Google Scholar] [CrossRef]
- Lamoureux, E.L.; Tai, E.S.; Thumboo, J.; Kawasaki, R.; Saw, S.-M.; Mitchell, P.; Wong, T.Y. Impact of Diabetic Retinopathy on Vision-Specific Function. Ophthalmology 2010, 117, 757–765. [Google Scholar] [CrossRef]
- Fenwick, E.K.; Pesudovs, K.; Rees, G.; Dirani, M.; Kawasaki, R.; Wong, T.Y.; Lamoureux, E.L. The Impact of Diabetic Retinopathy: Understanding the Patient’s Perspective. Br. J. Ophthalmol. 2011, 95, 774–782. [Google Scholar] [CrossRef]
- Shi, R.; Chen, L.; Wang, W.; Deng, Y.; Liu, Y.; Zhou, H.; Lin, R. Plasma MiR-26a-5p Is a Biomarker for Retinal Neurodegeneration of Early Diabetic Retinopathy. Eye 2021, 35, 1587–1599. [Google Scholar] [CrossRef]
- Coyne, K.S.; Margolis, M.K.; Kennedy-Martin, T.; Baker, T.M.; Klein, R.; Paul, M.D.; Revicki, D.A. The Impact of Diabetic Retinopathy: Perspectives from Patient Focus Groups. Fam. Pract. 2004, 21, 447–453. [Google Scholar] [CrossRef]
- Watkinson, S.; Seewoodhary, R. Ocular Complications Associated with Diabetes Mellitus. Nurs. Stand. 2008, 22, 51–57, quiz 58, 60. [Google Scholar] [CrossRef]
- Stitt, A.W.; Lois, N.; Medina, R.J.; Adamson, P.; Curtis, T.M. Advances in Our Understanding of Diabetic Retinopathy. Clin. Sci. 2013, 125, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Duh, E.J.; Sun, J.K.; Stitt, A.W. Diabetic Retinopathy: Current Understanding, Mechanisms, and Treatment Strategies. JCI Insight 2017, 2, e93751. [Google Scholar] [CrossRef] [PubMed]
- Stitt, A.W.; Curtis, T.M.; Chen, M.; Medina, R.J.; McKay, G.J.; Jenkins, A.; Gardiner, T.A.; Lyons, T.J.; Hammes, H.-P.; Simó, R. The Progress in Understanding and Treatment of Diabetic Retinopathy. Prog. Retin. Eye Res. 2016, 51, 156–186. [Google Scholar] [CrossRef] [PubMed]
- Meyerle, C.B.; Chew, E.Y.; Ferris, F.L. Nonproliferative Diabetic Retinopathy. In Diabetic Retinopathy; Duh, E.J., Ed.; Humana Press: Totowa, NJ, USA, 2008; pp. 3–27. ISBN 978-1-934115-83-1. [Google Scholar]
- Isner, J.M.; Asahara, T. Angiogenesis and Vasculogenesis as Therapeutic Strategies for Postnatal Neovascularization. J. Clin. Investig. 1999, 103, 1231–1236. [Google Scholar] [CrossRef]
- Crawford, T.N.; Alfaro, D.V., 3rd; Kerrison, J.B.; Jablon, E.P. Diabetic Retinopathy and Angiogenesis. Curr. Diabetes Rev. 2009, 5, 8–13. [Google Scholar] [CrossRef]
- Capitão, M.; Soares, R. Angiogenesis and Inflammation Crosstalk in Diabetic Retinopathy. J. Cell. Biochem. 2016, 117, 2443–2453. [Google Scholar] [CrossRef]
- Gupta, N.; Mansoor, S.; Sharma, A.; Sapkal, A.; Sheth, J.; Falatoonzadeh, P.; Kuppermann, B.; Kenney, M. Diabetic Retinopathy and VEGF. Open Ophthalmol. J. 2013, 7, 4–10. [Google Scholar] [CrossRef]
- Campbell, M.; Doyle, S.L. Current Perspectives on Established and Novel Therapies for Pathological Neovascularization in Retinal Disease. Biochem. Pharmacol. 2019, 164, 321–325. [Google Scholar] [CrossRef]
- Praidou, A.; Androudi, S.; Brazitikos, P.; Karakiulakis, G.; Papakonstantinou, E.; Dimitrakos, S. Angiogenic Growth Factors and Their Inhibitors in Diabetic Retinopathy. Curr. Diabetes Rev. 2010, 6, 304–312. [Google Scholar] [CrossRef]
- Barber, A.J.; Lieth, E.; Khin, S.A.; Antonetti, D.A.; Buchanan, A.G.; Gardner, T.W. Neural Apoptosis in the Retina during Experimental and Human Diabetes. J. Clin. Investig. 1998, 102, 783–791. [Google Scholar] [CrossRef]
- Kern, T.S.; Barber, A.J. Retinal Ganglion Cells in Diabetes. J. Physiol. 2008, 586, 4401–4408. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, Q.; Gillies, M.C.; Wong, T.Y. Management of Diabetic Retinopathy: A Systematic Review. JAMA 2007, 298, 902–916. [Google Scholar] [CrossRef] [PubMed]
- Fong, D.S.; Girach, A.; Boney, A. Visual Side Effects of Successful Scatter Laser Photocoagulation Surgery for Proliferative Diabetic Retinopathy: A Literature Review. Retina 2007, 27, 816–824. [Google Scholar] [CrossRef] [PubMed]
- Gross, J.G.; Glassman, A.R.; Jampol, L.M.; Inusah, S.; Aiello, L.P.; Antoszyk, A.N.; Baker, C.W.; Berger, B.B.; Bressler, N.M.; Browning, D. Panretinal Photocoagulation vs Intravitreous Ranibizumab for Proliferative Diabetic Retinopathy: A Randomized Clinical Trial. JAMA 2015, 314, 2137–2146. [Google Scholar] [PubMed]
- Sivaprasad, S.; Prevost, A.T.; Vasconcelos, J.C.; Riddell, A.; Murphy, C.; Kelly, J.; Bainbridge, J.; Tudor-Edwards, R.; Hopkins, D.; Hykin, P. Clinical Efficacy of Intravitreal Aflibercept versus Panretinal Photocoagulation for Best Corrected Visual Acuity in Patients with Proliferative Diabetic Retinopathy at 52 Weeks (CLARITY): A Multicentre, Single-Blinded, Randomised, Controlled, Phase 2b, Non-Inferiority Trial. Lancet 2017, 389, 2193–2203. [Google Scholar]
- Preti, R.C.; Ramirez, L.M.; Monteiro, M.L.; Carra, M.K.; Pelayes, D.E.; Takahashi, W.Y. Contrast Sensitivity Evaluation in High Risk Proliferative Diabetic Retinopathy Treated with Panretinal Photocoagulation Associated or Not with Intravitreal Bevacizumab Injections: A Randomised Clinical Trial. Br. J. Ophthalmol. 2013, 97, 885–889. [Google Scholar] [CrossRef]
- McCannel, C.A. Meta-Analysis of Endophthalmitis after Intravitreal Injection of Anti-Vascular Endothelial Growth Factor Agents: Causative Organisms and Possible Prevention Strategies. Retina 2011, 31, 654–661. [Google Scholar] [CrossRef]
- Wubben, T.J.; Johnson, M.W. Anti-Vascular Endothelial Growth Factor Therapy for Diabetic Retinopathy: Consequences of Inadvertent Treatment Interruptions. Am. J. Ophthalmol. 2019, 204, 13–18. [Google Scholar] [CrossRef]
- Zehden, J.A.; Mortensen, X.M.; Reddy, A.; Zhang, A.Y. Systemic and Ocular Adverse Events with Intravitreal Anti-VEGF Therapy Used in the Treatment of Diabetic Retinopathy: A Review. Curr. Diabetes Rep. 2022, 22, 525–536. [Google Scholar] [CrossRef]
- Gonzalez, V.H.; Campbell, J.; Holekamp, N.M.; Kiss, S.; Loewenstein, A.; Augustin, A.J.; Ma, J.; Ho, A.C.; Patel, V.; Whitcup, S.M. Early and Long-Term Responses to Anti-Vascular Endothelial Growth Factor Therapy in Diabetic Macular Edema: Analysis of Protocol I Data. Am. J. Ophthalmol. 2016, 172, 72–79. [Google Scholar] [CrossRef]
- Faselis, C.; Katsimardou, A.; Imprialos, K.; Deligkaris, P.; Kallistratos, M.; Dimitriadis, K. Microvascular Complications of Type 2 Diabetes Mellitus. Curr. Vasc. Pharmacol. 2020, 18, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Tesfaye, S.; Selvarajah, D. Advances in the Epidemiology, Pathogenesis and Management of Diabetic Peripheral Neuropathy: Advances in Epidemiology, Pathogenesis and Management of DPN. Diabetes Metab. Res. Rev. 2012, 28, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, Z.; Azmi, S.; Yadav, R.; Ferdousi, M.; Kumar, M.; Cuthbertson, D.J.; Lim, J.; Malik, R.A.; Alam, U. Diabetic Peripheral Neuropathy: Epidemiology, Diagnosis, and Pharmacotherapy. Clin. Ther. 2018, 40, 828–849. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, M.; Fargo, E.; Prasad-Reddy, L.; Mahoney, K.M.; Isaacs, D. Diabetes: How to Manage Diabetic Peripheral Neuropathy. Drugs Context 2022, 11, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Nitta, K.; Masakane, I.; Hanafusa, N.; Taniguchi, M.; Hasegawa, T.; Nakai, S.; Goto, S.; Wada, A.; Hamano, T.; Hoshino, J.; et al. Annual Dialysis Data Report 2017, JSDT Renal Data Registry. Ren. Replace. Ther. 2019, 5, 53. [Google Scholar] [CrossRef]
- Rossing, K.; Christensen, P.K.; Hovind, P.; Tarnow, L.; Rossing, P.; Parving, H.-H. Progression of Nephropathy in Type 2 Diabetic Patients. Kidney Int. 2004, 66, 1596–1605. [Google Scholar] [CrossRef] [PubMed]
- Sagoo, M.K.; Gnudi, L. Diabetic Nephropathy: An Overview. In Diabetic Nephropathy; Gnudi, L., Long, D.A., Eds.; Methods in Molecular Biology; Springer: New York, NY, USA, 2020; Volume 2067, pp. 3–7. ISBN 978-1-4939-9840-1. [Google Scholar]
- Nakhoul, F.; Nakhoul, N.; Asleh, R.; Miller-Lotan, R.; Levy, A.P. Is the Hp 2-2 Diabetic Mouse Model a Good Model to Study Diabetic Nephropathy? Diabetes Res. Clin. Pract. 2013, 100, 289–297. [Google Scholar] [CrossRef]
- Gnudi, L.; Gentile, G.; Ruggenenti, P. The Patient with Diabetes Mellitus. In Oxford Textbook of Clinical Nephrology; Turner, N., Lameire, N., Goldsmith, D.J., Winearls, C.G., Himmelfarb, J., Remuzzi, G., Eds.; Oxford University Press: Oxford, UK, 2015; pp. 1199–1247. ISBN 978-0-19-959254-8. [Google Scholar]
- Jia, G.; Hill, M.A.; Sowers, J.R. Diabetic Cardiomyopathy: An Update of Mechanisms Contributing to This Clinical Entity. Circ. Res. 2018, 122, 624–638. [Google Scholar] [CrossRef]
- Wagenknecht, L.E.; Zaccaro, D.; Espeland, M.A.; Karter, A.J.; O’Leary, D.H.; Haffner, S.M. Diabetes and Progression of Carotid Atherosclerosis: The Insulin Resistance Atherosclerosis Study. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 1035–1041. [Google Scholar] [CrossRef]
- Folsom, A.R.; Rasmussen, M.L.; Chambless, L.E.; Howard, G.; Cooper, L.S.; Schmidt, M.I.; Heiss, G. Prospective Associations of Fasting Insulin, Body Fat Distribution, and Diabetes with Risk of Ischemic Stroke. The Atherosclerosis Risk in Communities (ARIC) Study Investigators. Diabetes Care 1999, 22, 1077–1083. [Google Scholar] [CrossRef]
- Ergul, A.; Kelly-Cobbs, A.; Abdalla, M.; Fagan, S.C. Cerebrovascular Complications of Diabetes: Focus on Stroke. Endocrine Metab. Immune Disord.-Drug Targets 2012, 12, 148–158. [Google Scholar] [CrossRef] [PubMed]
- Lusis, A.J. Atherosclerosis. Nature 2000, 407, 233–241. [Google Scholar] [CrossRef]
- Borghetti, G.; von Lewinski, D.; Eaton, D.M.; Sourij, H.; Houser, S.R.; Wallner, M. Diabetic Cardiomyopathy: Current and Future Therapies. Beyond Glycemic Control. Front. Physiol. 2018, 9, 1514. [Google Scholar] [CrossRef] [PubMed]
- Westermeier, F.; Riquelme, J.A.; Pavez, M.; Garrido, V.; Díaz, A.; Verdejo, H.E.; Castro, P.F.; García, L.; Lavandero, S. New Molecular Insights of Insulin in Diabetic Cardiomyopathy. Front. Physiol. 2016, 7, 125. [Google Scholar] [CrossRef]
- Dal Canto, E.; Ceriello, A.; Rydén, L.; Ferrini, M.; Hansen, T.B.; Schnell, O.; Standl, E.; Beulens, J.W. Diabetes as a Cardiovascular Risk Factor: An Overview of Global Trends of Macro and Micro Vascular Complications. Eur. J. Prev. Cardiol. 2019, 26, 25–32. [Google Scholar] [CrossRef]
- O’Donnell, M.J.; Xavier, D.; Liu, L.; Zhang, H.; Chin, S.L.; Rao-Melacini, P.; Rangarajan, S.; Islam, S.; Pais, P.; McQueen, M.J.; et al. Risk Factors for Ischaemic and Intracerebral Haemorrhagic Stroke in 22 Countries (the INTERSTROKE Study): A Case-Control Study. Lancet 2010, 376, 112–123. [Google Scholar] [CrossRef] [PubMed]
- Lau, L.; Lew, J.; Borschmann, K.; Thijs, V.; Ekinci, E.I. Prevalence of Diabetes and Its Effects on Stroke Outcomes: A Meta-analysis and Literature Review. J. Diabetes Investig. 2019, 10, 780–792. [Google Scholar] [CrossRef]
- Mankovsky, B.N.; Ziegler, D. Stroke in Patients with Diabetes Mellitus. Diabetes Metab. Res. Rev. 2004, 20, 268–287. [Google Scholar] [CrossRef]
- Tun, N.N.; Arunagirinathan, G.; Munshi, S.K.; Pappachan, J.M. Diabetes Mellitus and Stroke: A Clinical Update. World J. Diabetes 2017, 8, 235. [Google Scholar] [CrossRef]
- Zinman, B.; Wanner, C.; Lachin, J.M.; Fitchett, D.; Bluhmki, E.; Hantel, S.; Mattheus, M.; Devins, T.; Johansen, O.E.; Woerle, H.J.; et al. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N. Engl. J. Med. 2015, 373, 2117–2128. [Google Scholar] [CrossRef]
- Zhou, Y.; Huang, Y.; Ji, X.; Wang, X.; Shen, L.; Wang, Y. Pioglitazone for the Primary and Secondary Prevention of Cardiovascular and Renal Outcomes in Patients with or at High Risk of Type 2 Diabetes Mellitus: A Meta-Analysis. J. Clin. Endocrinol. Metab. 2020, 105, 1670–1681. [Google Scholar] [CrossRef] [PubMed]
- Committee, C.S. A Randomised, Blinded, Trial of Clopidogrel versus Aspirin in Patients at Risk of Ischaemic Events (CAPRIE). Lancet 1996, 348, 1329–1339. [Google Scholar] [CrossRef]
- Dormandy, J.A.; Charbonnel, B.; Eckland, D.J.; Erdmann, E.; Massi-Benedetti, M.; Moules, I.K.; Skene, A.M.; Tan, M.H.; Lefèbvre, P.J.; Murray, G.D.; et al. Secondary Prevention of Macrovascular Events in Patients with Type 2 Diabetes in the PROactive Study (PROspective PioglitAzone Clinical Trial in MacroVascular Events): A Randomised Controlled Trial. Lancet 2005, 366, 1279–1289. [Google Scholar] [CrossRef] [PubMed]
- Asmat, U.; Abad, K.; Ismail, K. Diabetes Mellitus and Oxidative Stress—A Concise Review. Saudi Pharm. J. 2016, 24, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Bhatti, J.S.; Sehrawat, A.; Mishra, J.; Sidhu, I.S.; Navik, U.; Khullar, N.; Kumar, S.; Bhatti, G.K.; Reddy, P.H. Oxidative Stress in the Pathophysiology of Type 2 Diabetes and Related Complications: Current Therapeutics Strategies and Future Perspectives. Free Radic. Biol. Med. 2022, 184, 114–134. [Google Scholar] [CrossRef] [PubMed]
- Brownlee, M. Biochemistry and Molecular Cell Biology of Diabetic Complications. Nature 2001, 414, 813–820. [Google Scholar] [CrossRef]
- Giacco, F.; Brownlee, M. Oxidative Stress and Diabetic Complications. Circ. Res. 2010, 107, 1058–1070. [Google Scholar] [CrossRef]
- Brownlee, M. The Pathobiology of Diabetic Complications: A Unifying Mechanism. Diabetes 2005, 54, 1615–1625. [Google Scholar] [CrossRef]
- Paul, S.; Ali, A.; Katare, R. Molecular Complexities Underlying the Vascular Complications of Diabetes Mellitus—A Comprehensive Review. J. Diabetes Its Complicat. 2020, 34, 107613. [Google Scholar] [CrossRef]
- Díaz-Flores, M.; Baiza-Gutman, L.A. Biochemical Mechanisms of Vascular Complications in Diabetes. In The Diabetes Textbook; Rodriguez-Saldana, J., Ed.; Springer International Publishing: Cham, Switzerland, 2019; pp. 695–707. ISBN 978-3-030-11814-3. [Google Scholar]
- Wu, M.Y.; Yiang, G.T.; Lai, T.T.; Li, C.J. The Oxidative Stress and Mitochondrial Dysfunction during the Pathogenesis of Diabetic Retinopathy. Oxidative Med. Cell. Longev. 2018, 2018, 3420187. [Google Scholar] [CrossRef]
- Xu, J.; Chen, L.J.; Yu, J.; Wang, H.J.; Zhang, F.; Liu, Q.; Wu, J. Involvement of Advanced Glycation End Products in the Pathogenesis of Diabetic Retinopathy. Cell. Physiol. Biochem. 2018, 48, 705–717. [Google Scholar] [CrossRef] [PubMed]
- Hayden, M.R.; Sowers, J.R.; Tyagi, S.C. The Central Role of Vascular Extracellular Matrix and Basement Membrane Remodeling in Metabolic Syndrome and Type 2 Diabetes: The Matrix Preloaded. Cardiovasc. Diabetol. 2005, 4, 9. [Google Scholar] [CrossRef] [PubMed]
- Koya, D.; King, G.L. Protein Kinase C Activation and the Development of Diabetic Complications. Diabetes 1998, 47, 859–866. [Google Scholar] [CrossRef]
- Mima, A.; Kitada, M.; Geraldes, P.; Li, Q.; Matsumoto, M.; Mizutani, K.; Qi, W.; Li, C.; Leitges, M.; Rask-Madsen, C. Glomerular VEGF Resistance Induced by PKCδ/SHP-1 Activation and Contribution to Diabetic Nephropathy. FASEB J. 2012, 26, 2963–2974. [Google Scholar] [CrossRef] [PubMed]
- Buse, M.G. Hexosamines, Insulin Resistance, and the Complications of Diabetes: Current Status. Am. J. Physiol. Endocrinol. Metab. 2006, 290, E1–E8. [Google Scholar] [CrossRef]
- Chatham, J.C.; Young, M.E.; Zhang, J. Role of O-Linked N-Acetylglucosamine (O-GlcNAc) Modification of Proteins in Diabetic Cardiovascular Complications. Curr. Opin. Pharmacol. 2020, 57, 1–12. [Google Scholar] [CrossRef]
- Liu, C.; Dong, W.; Li, J.; Kong, Y.; Ren, X. O-GlcNAc Modification and Its Role in Diabetic Retinopathy. Metabolites 2022, 12, 725. [Google Scholar] [CrossRef]
- Frey, T.; Antonetti, D.A. Alterations to the Blood-Retinal Barrier in Diabetes: Cytokines and Reactive Oxygen Species. Antioxid. Redox Signal. 2011, 15, 1271–1284. [Google Scholar] [CrossRef]
- Asnaghi, V.; Gerhardinger, C.; Hoehn, T.; Adeboje, A.; Lorenzi, M. A Role for the Polyol Pathway in the Early Neuroretinal Apoptosis and Glial Changes Induced by Diabetes in the Rat. Diabetes 2003, 52, 506–511. [Google Scholar] [CrossRef]
- Li, Q.R.; Wang, Z.; Zhou, W.; Fan, S.R.; Ma, R.; Xue, L.; Yang, L.; Li, Y.S.; Tan, H.L.; Shao, Q.H. Epalrestat Protects against Diabetic Peripheral Neuropathy by Alleviating Oxidative Stress and Inhibiting Polyol Pathway. Neural Regen. Res. 2016, 11, 345–351. [Google Scholar]
- Hotta, N.; Kawamori, R.; Fukuda, M.; Shigeta, Y. Long-Term Clinical Effects of Epalrestat, an Aldose Reductase Inhibitor, on Progression of Diabetic Neuropathy and Other Microvascular Complications: Multivariate Epidemiological Analysis Based on Patient Background Factors and Severity of Diabetic Neuropathy. Diabet. Med. A J. Br. Diabet. Assoc. 2012, 29, 1529–1533. [Google Scholar]
- Fridlyand, L.E.; Philipson, L.H. Does the Glucose-Dependent Insulin Secretion Mechanism Itself Cause Oxidative Stress in Pancreatic β-Cells? Diabetes 2004, 53, 1942–1948. [Google Scholar] [CrossRef] [PubMed]
- Munteanu, C.; Rotariu, M.; Turnea, M.-A.; Anghelescu, A.; Albadi, I.; Dogaru, G.; Silișteanu, S.C.; Ionescu, E.V.; Firan, F.C.; Ionescu, A.M. Topical Reappraisal of Molecular Pharmacological Approaches to Endothelial Dysfunction in Diabetes Mellitus Angiopathy. Curr. Issues Mol. Biol. 2022, 44, 3378–3397. [Google Scholar] [CrossRef] [PubMed]
- Elumalai, S.; Karunakaran, U.; Moon, J.-S.; Won, K.-C. NADPH Oxidase (NOX) Targeting in Diabetes: A Special Emphasis on Pancreatic β-Cell Dysfunction. Cells 2021, 10, 1573. [Google Scholar] [CrossRef]
- Förstermann, U.; Xia, N.; Li, H. Roles of Vascular Oxidative Stress and Nitric Oxide in the Pathogenesis of Atherosclerosis. Circ. Res. 2017, 120, 713–735. [Google Scholar] [CrossRef]
- Kim, W.-H.; Lee, J.W.; Suh, Y.H.; Lee, H.J.; Lee, S.H.; Oh, Y.K.; Gao, B.; Jung, M.H. AICAR Potentiates ROS Production Induced by Chronic High Glucose: Roles of AMPK in Pancreatic β-Cell Apoptosis. Cell. Signal. 2007, 19, 791–805. [Google Scholar] [CrossRef]
- Chau, G.C.; Im, D.U.; Kang, T.M.; Bae, J.M.; Kim, W.; Pyo, S.; Moon, E.-Y.; Um, S.H. MTOR Controls ChREBP Transcriptional Activity and Pancreatic β Cell Survival under Diabetic Stress. J. Cell Biol. 2017, 216, 2091–2105. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, C. Targeting β-Cell Dedifferentiation and Transdifferentiation: Opportunities and Challenges. Endocr. Connect. 2021, 10, R213–R228. [Google Scholar] [CrossRef]
- Wang, F.; Wang, L.; Qu, C.; Chen, L.; Geng, Y.; Cheng, C.; Yu, S.; Wang, D.; Yang, L.; Meng, Z. Kaempferol Induces ROS-Dependent Apoptosis in Pancreatic Cancer Cells via TGM2-Mediated Akt/MTOR Signaling. BMC Cancer 2021, 21, 396. [Google Scholar] [CrossRef]
- Zhao, D.; Yang, J.; Yang, L. Insights for Oxidative Stress and MTOR Signaling in Myocardial Ischemia/Reperfusion Injury under Diabetes. Oxidative Med. Cell. Longev. 2017, 2017, 6437467. [Google Scholar] [CrossRef]
- James, D.E.; Stöckli, J.; Birnbaum, M.J. The Aetiology and Molecular Landscape of Insulin Resistance. Nat. Rev. Mol. Cell Biol. 2021, 22, 751–771. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Pan, J.; Liu, D.; Zhang, M.; Li, X.; Tian, J.; Liu, M.; Jin, T.; An, F. Nicorandil Alleviates Apoptosis in Diabetic Cardiomyopathy through PI3K/Akt Pathway. J. Cell. Mol. Med. 2019, 23, 5349–5359. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.-G.; Li, P.-L.; Dai, Y.; Deng, J.-L.; Shan, M.-Y.; Chen, B.; Zhang, K.-B.; Guo, S.-D.; Xu, Z.-H. Mibefradil Alleviates High-Glucose–Induced Cardiac Hypertrophy by Inhibiting PI3K/Akt/MTOR-Mediated Autophagy. J. Cardiovasc. Pharmacol. 2020, 76, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Paneni, F.; Costantino, S.; Cosentino, F. Role of Oxidative Stress in Endothelial Insulin Resistance. World J. Diabetes 2015, 6, 326. [Google Scholar] [CrossRef] [PubMed]
- Shamshoum, H.; Vlavcheski, F.; MacPherson, R.E.; Tsiani, E. Rosemary Extract Activates AMPK, Inhibits MTOR and Attenuates the High Glucose and High Insulin-Induced Muscle Cell Insulin Resistance. Appl. Physiol. Nutr. Metab. 2021, 46, 819–827. [Google Scholar] [CrossRef]
- Tang, Z.; Wang, P.; Dong, C.; Zhang, J.; Wang, X.; Pei, H. Oxidative Stress Signaling Mediated Pathogenesis of Diabetic Cardiomyopathy. Oxidative Med. Cell. Longev. 2022, 2022, 5913374. [Google Scholar] [CrossRef]
- Sies, H.; Jones, D.P. Reactive Oxygen Species (ROS) as Pleiotropic Physiological Signalling Agents. Nat. Rev. Mol. Cell Biol. 2020, 21, 363–383. [Google Scholar] [CrossRef]
- Volpe, C.M.O.; Villar-Delfino, P.H.; Dos Anjos, P.M.F.; Nogueira-Machado, J.A. Cellular Death, Reactive Oxygen Species (ROS) and Diabetic Complications. Cell Death Dis. 2018, 9, 1–9. [Google Scholar] [CrossRef]
- Ahsan, M.K.; Lekli, I.; Ray, D.; Yodoi, J.; Das, D.K. Redox Regulation of Cell Survival by the Thioredoxin Superfamily: An Implication of Redox Gene Therapy in the Heart. Available online: https://www.liebertpub.com/doi/10.1089/ars.2009.2683 (accessed on 12 November 2022).
- Santilli, F.; D’Ardes, D.; Davì, G. Oxidative Stress in Chronic Vascular Disease: From Prediction to Prevention. Vasc. Pharmacol. 2015, 74, 23–37. [Google Scholar] [CrossRef]
- Mirończuk-Chodakowska, I.; Witkowska, A.M.; Zujko, M.E. Endogenous Non-Enzymatic Antioxidants in the Human Body. Adv. Med. Sci. 2018, 63, 68–78. [Google Scholar] [CrossRef]
- Jung, K.-A.; Kwak, M.-K. The Nrf2 System as a Potential Target for the Development of Indirect Antioxidants. Molecules 2010, 15, 7266–7291. [Google Scholar] [CrossRef] [PubMed]
- Seco-Cervera, M.; González-Cabo, P.; Pallardó, F.V.; Romá-Mateo, C.; García-Giménez, J.L. Thioredoxin and Glutaredoxin Systems as Potential Targets for the Development of New Treatments in Friedreich’s Ataxia. Antioxidants 2020, 9, 1257. [Google Scholar] [CrossRef] [PubMed]
- Kowluru, R.A.; Kern, T.S.; Engerman, R.L. Abnormalities of Retinal Metabolism in Diabetes or Experimental Galactosemia. IV. Antioxidant Defense System. Free Radic. Biol. Med. 1997, 22, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Kowluru, R.A.; Mishra, M. Oxidative Stress, Mitochondrial Damage and Diabetic Retinopathy. Biochim. Et Biophys. Acta (BBA) Mol. Basis Dis. 2015, 1852, 2474–2483. [Google Scholar] [CrossRef]
- Lee, J.-Y.; Kim, M.; Oh, S.B.; Kim, H.-Y.; Kim, C.; Kim, T.-Y.; Park, Y.-H. Superoxide Dismutase 3 Prevents Early Stage Diabetic Retinopathy in Streptozotocin-Induced Diabetic Rat Model. PLoS ONE 2022, 17, e0262396. [Google Scholar] [CrossRef]
- Kwong-Han, K.; Zunaina, E.; Hanizasurana, H.; Che-Badariah, A.A.; Che-Maraina, C.H. Comparison of Catalase, Glutathione Peroxidase and Malondialdehyde Levels in Tears among Diabetic Patients with and without Diabetic Retinopathy. J. Diabetes Metab. Disord. 2022, 21, 681–688. [Google Scholar] [CrossRef]
- Zhao, J.-S.; Jin, H.-X.; Gao, J.-L.; Pu, C.; Zhang, P.; Huang, J.-J.; Cheng, L.; Feng, G. Serum Extracellular Superoxide Dismutase Is Associated with Diabetic Retinopathy Stage in Chinese Patients with Type 2 Diabetes Mellitus. Dis. Mrk. 2018, 2018, 8721379. [Google Scholar] [CrossRef]
- Lu, J.; Holmgren, A. The Thioredoxin Antioxidant System. Free Radic. Biol. Med. 2014, 66, 75–87. [Google Scholar] [CrossRef]
- Singh, L.P. Thioredoxin Interacting Protein (TXNIP) and Pathogenesis of Diabetic Retinopathy. J. Clin. Exp. Ophthalmol. 2013, 4. [Google Scholar] [CrossRef]
- Cheng, Y.; Zhang, X.; Ma, F.; Sun, W.; Wang, W.; Yu, J.; Shi, Y.; Cai, L.; Xu, Z. The Role of Akt2 in the Protective Effect of Fenofibrate against Diabetic Nephropathy. Int. J. Biol. Sci. 2020, 16, 553–567. [Google Scholar] [CrossRef]
- Wei, P.Z.; Szeto, C.C. Mitochondrial Dysfunction in Diabetic Kidney Disease. Clin. Chim. Acta 2019, 496, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Sakashita, M.; Tanaka, T.; Inagi, R. Metabolic Changes and Oxidative Stress in Diabetic Kidney Disease. Antioxidants 2021, 10, 1143. [Google Scholar] [CrossRef] [PubMed]
- Nezu, M.; Suzuki, N. Roles of Nrf2 in Protecting the Kidney from Oxidative Damage. Int. J. Mol. Sci. 2020, 21, 2951. [Google Scholar] [CrossRef] [PubMed]
- Duni, A.; Liakopoulos, V.; Roumeliotis, S.; Peschos, D.; Dounousi, E. Oxidative Stress in the Pathogenesis and Evolution of Chronic Kidney Disease: Untangling Ariadne’s Thread. Int. J. Mol. Sci. 2019, 20, 3711. [Google Scholar] [CrossRef]
- Hong, Y.A.; Park, C.W. Catalytic Antioxidants in the Kidney. Antioxidants 2021, 10, 130. [Google Scholar] [CrossRef]
- Tucker, P.S.; Dalbo, V.J.; Han, T.; Kingsley, M.I. Clinical and Research Markers of Oxidative Stress in Chronic Kidney Disease. Biomarkers 2013, 18, 103–115. [Google Scholar] [CrossRef]
- Modlinger, P.S.; Wilcox, C.S.; Aslam, S. Nitric Oxide, Oxidative Stress, and Progression of Chronic Renal Failure. Semin. Nephrol. 2004, 24, 354–365. [Google Scholar] [CrossRef]
- Shurtz-Swirski, R.; Mashiach, E.; Kristal, B.; Shkolnik, T.; Shasha, S.M. Antioxidant Enzymes Activity in Polymorphonuclear Leukocytes in Chronic Renal Failure. Nephron 1995, 71, 176–179. [Google Scholar] [CrossRef]
- DeRubertis, F.R.; Craven, P.A.; Melhem, M.F.; Salah, E.M. Attenuation of Renal Injury in Db / Db Mice Overexpressing Superoxide Dismutase. Diabetes 2004, 53, 762–768. [Google Scholar] [CrossRef]
- Kitada, M.; Xu, J.; Ogura, Y.; Monno, I.; Koya, D. Manganese Superoxide Dismutase Dysfunction and the Pathogenesis of Kidney Disease. Front. Physiol. 2020, 11, 755. [Google Scholar] [CrossRef]
- Kuo, C.-W.; Shen, C.-J.; Tung, Y.-T.; Chen, H.-L.; Chen, Y.-H.; Chang, W.-H.; Cheng, K.-C.; Yang, S.-H.; Chen, C.-M. Extracellular Superoxide Dismutase Ameliorates Streptozotocin-Induced Rat Diabetic Nephropathy via Inhibiting the ROS/ERK1/2 Signaling. Life Sci. 2015, 135, 77–86. [Google Scholar] [CrossRef]
- Hong, Y.A.; Lim, J.H.; Kim, M.Y.; Kim, Y.; Park, H.S.; Kim, H.W.; Choi, B.S.; Chang, Y.S.; Kim, H.W.; Kim, T.-Y.; et al. Extracellular Superoxide Dismutase Attenuates Renal Oxidative Stress Through the Activation of Adenosine Monophosphate-Activated Protein Kinase in Diabetic Nephropathy. Antioxid. Redox Signal. 2018, 28, 1543–1561. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Tran, S.; Sachetelli, S.; Filep, J.G.; Ingelfinger, J.R.; Chan, J.S.D. Catalase Overexpression Attenuates Angiotensinogen Expression and Apoptosis in Diabetic Mice. Kidney Int. 2007, 71, 912–923. [Google Scholar] [CrossRef]
- Brezniceanu, M.-L.; Liu, F.; Wei, C.-C.; Chénier, I.; Godin, N.; Zhang, S.-L.; Filep, J.G.; Ingelfinger, J.R.; Chan, J.S.D. Attenuation of Interstitial Fibrosis and Tubular Apoptosis in Db/Db Transgenic Mice Overexpressing Catalase in Renal Proximal Tubular Cells. Diabetes 2008, 57, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Chiu, Y.I.-W.; Kuo, M.-C.; Kuo, H.-T.; Chang, J.-M.; Guh, J.-Y.; Lai, Y.-H.; Chen, H.-C. Alterations of Glomerular and Extracellular Levels of Glutathione Peroxidase in Patients and Experimental Rats with Diabetic Nephropathy. J. Lab. Clin. Med. 2005, 145, 181–186. [Google Scholar] [CrossRef]
- Wang, Y.; Chang, D.; Zhao, M.; Chen, M. Glutathione Peroxidase 4 Is a Predictor of Diabetic Kidney Disease Progression in Type 2 Diabetes Mellitus. Oxidative Med. Cell. Longev. 2022, 2022, 2948248. [Google Scholar] [CrossRef]
- Wang, X.; Fang, H.; Xu, G.; Yang, Y.; Xu, R.; Liu, Q.; Xue, X.; Liu, J.; Wang, H. Resveratrol Prevents Cognitive Impairment in Type 2 Diabetic Mice by Upregulating Nrf2 Expression and Transcriptional Level. Diabetes Metab. Syndr. Obes. Targets Ther. 2020, 13, 1061–1075. [Google Scholar] [CrossRef]
- Sharma, A.; Tate, M.; Mathew, G.; Vince, J.E.; Ritchie, R.H.; de Haan, J.B. Oxidative Stress and NLRP3-Inflammasome Activity as Significant Drivers of Diabetic Cardiovascular Complications: Therapeutic Implications. Front. Physiol. 2018, 9, 114. [Google Scholar] [CrossRef]
- Tan, Y.; Zhang, Z.; Zheng, C.; Wintergerst, K.A.; Keller, B.B.; Cai, L. Mechanisms of Diabetic Cardiomyopathy and Potential Therapeutic Strategies: Preclinical and Clinical Evidence. Nat. Rev. Cardiol. 2020, 17, 585–607. [Google Scholar] [CrossRef]
- Bigagli, E.; Lodovici, M. Circulating Oxidative Stress Biomarkers in Clinical Studies on Type 2 Diabetes and Its Complications. Oxidative Med. Cell. Longev. 2019, 2019, 5953685. [Google Scholar] [CrossRef]
- Saisho, Y. Glycemic Variability and Oxidative Stress: A Link between Diabetes and Cardiovascular Disease? Int. J. Mol. Sci. 2014, 15, 18381–18406. [Google Scholar] [CrossRef] [PubMed]
- Frustaci, A.; Kajstura, J.; Chimenti, C.; Jakoniuk, I.; Leri, A.; Maseri, A.; Nadal-Ginard, B.; Anversa, P. Myocardial Cell Death in Human Diabetes. Circ. Res. 2000, 87, 1123–1132. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Zheng, S.; Metreveli, N.S.; Epstein, P.N. Protection of Cardiac Mitochondria by Overexpression of MnSOD Reduces Diabetic Cardiomyopathy. Diabetes 2006, 55, 798–805. [Google Scholar] [CrossRef]
- Matsushima, S.; Kinugawa, S.; Ide, T.; Matsusaka, H.; Inoue, N.; Ohta, Y.; Yokota, T.; Sunagawa, K.; Tsutsui, H. Overexpression of Glutathione Peroxidase Attenuates Myocardial Remodeling and Preserves Diastolic Function in Diabetic Heart. Am. J. Physiol.-Heart Circ. Physiol. 2006, 291, H2237–H2245. [Google Scholar] [CrossRef] [PubMed]
- Ge, Z.-D.; Lian, Q.; Mao, X.; Xia, Z. Current Status and Challenges of NRF2 as a Potential Therapeutic Target for Diabetic Cardiomyopathy. Int. Heart J. 2019, 60, 512–520. [Google Scholar] [CrossRef]
- Jiang, T.; Sun, Q.; Chen, S. Oxidative Stress: A Major Pathogenesis and Potential Therapeutic Target of Antioxidative Agents in Parkinson’s Disease and Alzheimer’s Disease. Prog. Neurobiol. 2016, 147, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Niedzielska, E.; Smaga, I.; Gawlik, M.; Moniczewski, A.; Stankowicz, P.; Pera, J.; Filip, M. Oxidative Stress in Neurodegenerative Diseases. Mol. Neurobiol. 2016, 53, 4094–4125. [Google Scholar] [CrossRef]
- Allen, C.L.; Bayraktutan, U. Oxidative Stress and Its Role in the Pathogenesis of Ischaemic Stroke. Int. J. Stroke 2009, 4, 461–470. [Google Scholar] [CrossRef]
- Cherubini, A.; Polidori, M.C.; Bregnocchi, M.; Pezzuto, S.; Cecchetti, R.; Ingegni, T.; di Iorio, A.; Senin, U.; Mecocci, P. Antioxidant Profile and Early Outcome in Stroke Patients. Stroke 2000, 31, 2295–2300. [Google Scholar] [CrossRef]
- Domínguez, C.; Delgado, P.; Vilches, A.; Martín-Gallán, P.; Ribó, M.; Santamarina, E.; Molina, C.; Corbeto, N.; Rodríguez-Sureda, V.; Rosell, A.; et al. Oxidative Stress after Thrombolysis-Induced Reperfusion in Human Stroke. Stroke 2010, 41, 653–660. [Google Scholar] [CrossRef]
- Cojocaru, I.M.; Cojocaru, M.; Sapira, V.; Ionescu, A. Evaluation of Oxidative Stress in Patients with Acute Ischemic Stroke. Rom. J. Intern. Med. 2013, 51, 97–106. [Google Scholar] [PubMed]
- Chekalina, N.I.; Kazakov, Y.M.; Mamontova, T.V.; Vesnina, L.E.; Kaidashev, I.P. Resveratrol More Effectively than Quercetin Reduces Endothelium Degeneration and Level of Necrosis Factor α in Patients with Coronary Artery Disease. Wiad. Lek. 2016, 69, 475–479. [Google Scholar] [PubMed]
- Csiszar, A.; Labinskyy, N.; Pinto, J.T.; Ballabh, P.; Zhang, H.; Losonczy, G.; Pearson, K.; de Cabo, R.; Pacher, P.; Zhang, C.; et al. Resveratrol Induces Mitochondrial Biogenesis in Endothelial Cells. Am. J. Physiol.-Heart Circ. Physiol. 2009, 297, H13–H20. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Xia, N.; Förstermann, U. Cardiovascular Effects and Molecular Targets of Resveratrol. Nitric Oxide 2012, 26, 102–110. [Google Scholar] [CrossRef]
- Della-Morte, D.; Dave, K.R.; DeFazio, R.A.; Bao, Y.C.; Raval, A.P.; Perez-Pinzon, M.A. Resveratrol Pretreatment Protects Rat Brain from Cerebral Ischemic Damage via a Sirtuin 1–Uncoupling Protein 2 Pathway. Neuroscience 2009, 159, 993–1002. [Google Scholar] [CrossRef]
- Han, J.; Wang, M.; Jing, X.; Shi, H.; Ren, M.; Lou, H. (−)-Epigallocatechin Gallate Protects Against Cerebral Ischemia-Induced Oxidative Stress via Nrf2/ARE Signaling. Neurochem. Res. 2014, 39, 1292–1299. [Google Scholar] [CrossRef]
- Alfieri, A.; Srivastava, S.; Siow, R.C.M.; Modo, M.; Fraser, P.A.; Mann, G.E. Targeting the Nrf2-Keap1 Antioxidant Defence Pathway for Neurovascular Protection in Stroke: Nrf2-Mediated Neurovascular Protection. J. Physiol. 2011, 589, 4125–4136. [Google Scholar] [CrossRef]
- Biessels, G.J.; Koffeman, A.; Scheltens, P. Diabetes and Cognitive Impairment: Clinical Diagnosis and Brain Imaging in Patients Attending a Memory Clinic. J. Neurol. 2006, 253, 477–482. [Google Scholar] [CrossRef]
- Han, J.; Liu, X.; Li, Y.; Zhang, J.; Yu, H. Sirt1/Nrf2 Signalling Pathway Prevents Cognitive Impairment in Diabetic Rats through Anti-oxidative Stress Induced by MiRNA-23b-3p Expression. Mol. Med. Rep. 2018, 17, 8414–8422. [Google Scholar] [CrossRef]
- Donadelli, M.; Dando, I.; Fiorini, C.; Palmieri, M. UCP2, a Mitochondrial Protein Regulated at Multiple Levels. Cell. Mol. Life Sci. 2014, 71, 1171–1190. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, L.; Wen, D.; Ren, C.; Chen, S.; Zhang, Z.; Hu, L.; Yu, Z.; Tombran-Tink, J.; Zhang, X.; et al. Neuroprotection of Retinal Cells by Caffeic Acid Phenylethyl Ester (CAPE) Is Mediated by Mitochondrial Uncoupling Protein UCP2. Neurochem. Int. 2021, 151, 105214. [Google Scholar] [CrossRef]
- Bhagani, H.; Nasser, S.A.; Dakroub, A.; El-Yazbi, A.F.; Eid, A.A.; Kobeissy, F.; Pintus, G.; Eid, A.H. The Mitochondria: A Target of Polyphenols in the Treatment of Diabetic Cardiomyopathy. Int. J. Mol. Sci. 2020, 21, 4962. [Google Scholar] [CrossRef]
- Fang, W.; Wang, C.; He, Y.; Zhou, Y.; Peng, X.; Liu, S. Resveratrol Alleviates Diabetic Cardiomyopathy in Rats by Improving Mitochondrial Function through PGC-1α Deacetylation. Acta Pharm. Sin. 2018, 39, 59–73. [Google Scholar] [CrossRef]
- Naoi, M.; Wu, Y.; Shamoto-Nagai, M.; Maruyama, W. Mitochondria in Neuroprotection by Phytochemicals: Bioactive Polyphenols Modulate Mitochondrial Apoptosis System, Function and Structure. Int. J. Mol. Sci. 2019, 20, 2451. [Google Scholar] [CrossRef]
- Zhu, H.; Toan, S.; Mui, D.; Zhou, H. Mitochondrial Quality Surveillance as a Therapeutic Target in Myocardial Infarction. Acta Physiol. 2021, 231, e13590. [Google Scholar] [CrossRef]
- Skemiene, K.; Liobikas, J.; Borutaite, V. Anthocyanins as Substrates for Mitochondrial Complex I—Protective Effect against Heart Ischemic Injury. FEBS J. 2015, 282, 963–971. [Google Scholar] [CrossRef]
- Huang, P.-C.; Wang, G.-J.; Fan, M.-J.; Asokan Shibu, M.; Liu, Y.-T.; Padma Viswanadha, V.; Lin, Y.-L.; Lai, C.-H.; Chen, Y.-F.; Liao, H.-E.; et al. Cellular Apoptosis and Cardiac Dysfunction in STZ-Induced Diabetic Rats Attenuated by Anthocyanins via Activation of IGFI-R/PI3K/Akt Survival Signaling. Environ. Toxicol. 2017, 32, 2471–2480. [Google Scholar] [CrossRef]
- Diao, J.; Zhao, H.; You, P.; You, H.; Wu, H.; Shou, X.; Cheng, G. Rosmarinic Acid Ameliorated Cardiac Dysfunction and Mitochondrial Injury in Diabetic Cardiomyopathy Mice via Activation of the SIRT1/PGC-1α Pathway. Biochem. Biophys. Res. Commun. 2021, 546, 29–34. [Google Scholar] [CrossRef]
- Diao, J.; Wei, J.; Yan, R.; Fan, G.; Lin, L.; Chen, M. Effects of Resveratrol on Regulation on UCP2 and Cardiac Function in Diabetic Rats. J. Physiol. Biochem. 2019, 75, 39–51. [Google Scholar] [CrossRef]
- Zheng, J.; Conrad, M. The Metabolic Underpinnings of Ferroptosis. Cell Metab. 2020, 32, 920–937. [Google Scholar] [CrossRef]
- Liu, J.; Li, Q.; Yang, Y.; Ma, L. Iron Metabolism and Type 2 Diabetes Mellitus: A Meta-Analysis and Systematic Review. J. Diabetes Investig. 2020, 11, 946–955. [Google Scholar] [CrossRef]
- Chaudhary, K.; Promsote, W.; Ananth, S.; Veeranan-Karmegam, R.; Tawfik, A.; Arjunan, P.; Martin, P.; Smith, S.B.; Thangaraju, M.; Kisselev, O.; et al. Iron Overload Accelerates the Progression of Diabetic Retinopathy in Association with Increased Retinal Renin Expression. Sci. Rep. 2018, 8, 3025. [Google Scholar] [CrossRef]
- Zhao, L.; Bartnikas, T.; Chu, X.; Klein, J.; Yun, C.; Srinivasan, S.; He, P. Hyperglycemia Promotes Microvillus Membrane Expression of DMT1 in Intestinal Epithelial Cells in a PKCα-dependent Manner. FASEB J. 2019, 33, 3549–3561. [Google Scholar] [CrossRef]
- Sha, W.; Hu, F.; Xi, Y.; Chu, Y.; Bu, S. Mechanism of Ferroptosis and Its Role in Type 2 Diabetes Mellitus. J. Diabetes Res. 2021, 2021, 9999612. [Google Scholar] [CrossRef]
- Jakaria, M.; Belaidi, A.A.; Bush, A.I.; Ayton, S. Ferroptosis as a Mechanism of Neurodegeneration in Alzheimer’s Disease. J. Neurochem. 2021, 159, 804–825. [Google Scholar] [CrossRef]
- Chandra, K.; Jain, V.; Jabin, A.; Dwivedi, S.; Joshi, S.; Ahmad, S.; Jain, S.K. Effect of Cichorium Intybus Seeds Supplementation on the Markers of Glycemic Control, Oxidative Stress, Inflammation, and Lipid Profile in Type 2 Diabetes Mellitus: A Randomized, Double-Blind Placebo Study. Phytother. Res. 2020, 34, 1609–1618. [Google Scholar] [CrossRef]
- Yang, X.-D.; Yang, Y.-Y. Ferroptosis as a Novel Therapeutic Target for Diabetes and Its Complications. Front. Endocrinol. 2022, 13, 853822. [Google Scholar] [CrossRef]
- Fernández-del-Río, L.; Nag, A.; Gutiérrez Casado, E.; Ariza, J.; Awad, A.M.; Joseph, A.I.; Kwon, O.; Verdin, E.; de Cabo, R.; Schneider, C.; et al. Kaempferol Increases Levels of Coenzyme Q in Kidney Cells and Serves as a Biosynthetic Ring Precursor. Free Radic. Biol. Med. 2017, 110, 176–187. [Google Scholar] [CrossRef]
- Xie, L.X.; Williams, K.J.; He, C.H.; Weng, E.; Khong, S.; Rose, T.E.; Kwon, O.; Bensinger, S.J.; Marbois, B.N.; Clarke, C.F. Resveratrol and Para-Coumarate Serve as Ring Precursors for Coenzyme Q Biosynthesis [S]. J. Lipid Res. 2015, 56, 909–919. [Google Scholar] [CrossRef]
- Meza-Torres, C.; Hernández-Camacho, J.D.; Cortés-Rodríguez, A.B.; Fang, L.; Bui Thanh, T.; Rodríguez-Bies, E.; Navas, P.; López-Lluch, G. Resveratrol Regulates the Expression of Genes Involved in CoQ Synthesis in Liver in Mice Fed with High Fat Diet. Antioxidants 2020, 9, 431. [Google Scholar] [CrossRef]
- Al-Numair, K.S.; Chandramohan, G.; Veeramani, C.; Alsaif, M.A. Ameliorative Effect of Kaempferol, a Flavonoid, on Oxidative Stress in Streptozotocin-Induced Diabetic Rats. Redox Rep. 2015, 20, 198–209. [Google Scholar] [CrossRef]
- Yu, Q.; Lee, C.F.; Wang, W.; Karamanlidis, G.; Kuroda, J.; Matsushima, S.; Sadoshima, J.; Tian, R. Elimination of NADPH Oxidase Activity Promotes Reductive Stress and Sensitizes the Heart to Ischemic Injury. J. Am. Heart Assoc. 2014, 3, e000555. [Google Scholar] [CrossRef]
- Zhang, Y.; Murugesan, P.; Huang, K.; Cai, H. NADPH Oxidases and Oxidase Crosstalk in Cardiovascular Diseases: Novel Therapeutic Targets. Nat. Rev. Cardiol. 2020, 17, 170–194. [Google Scholar] [CrossRef]
- Tan, H.; Chen, J.; Li, Y.; Li, Y.; Zhong, Y.; Li, G.; Liu, L.; Li, Y. Glabridin, a Bioactive Component of Licorice, Ameliorates Diabetic Nephropathy by Regulating Ferroptosis and the VEGF/Akt/ERK Pathways. Mol. Med. 2022, 28, 58. [Google Scholar] [CrossRef]
- Huang, D.; Shen, P.; Wang, C.; Gao, J.; Ye, C.; Wu, F. Calycosin Plays a Protective Role in Diabetic Kidney Disease through the Regulation of Ferroptosis. Pharm. Biol. 2022, 60, 990–996. [Google Scholar] [CrossRef]
- He, Y.; Wang, S.; Sun, H.; Li, Y.; Feng, J. Naringenin Ameliorates Myocardial Injury in STZ-Induced Diabetic Mice by Reducing Oxidative Stress, Inflammation and Apoptosis via Regulating the Nrf2 and NF-ΚB Signaling Pathways. Front. Cardiovasc. Med. 2022, 9, 946766. [Google Scholar] [CrossRef]
- Tang, X.; Li, X.; Zhang, D.; Han, W. Astragaloside-IV Alleviates High Glucose-Induced Ferroptosis in Retinal Pigment Epithelial Cells by Disrupting the Expression of MiR-138-5p/Sirt1/Nrf2. Bioengineered 2022, 13, 8238–8253. [Google Scholar] [CrossRef]
- Zhou, Y. The Protective Effects of Cryptochlorogenic Acid on β-Cells Function in Diabetes in Vivo and Vitro via Inhibition of Ferroptosis. Diabetes Metab. Syndr. Obes. 2020, 13, 1921–1931. [Google Scholar] [CrossRef]
- Li, D.; Jiang, C.; Mei, G.; Zhao, Y.; Chen, L.; Liu, J.; Tang, Y.; Gao, C.; Yao, P. Quercetin Alleviates Ferroptosis of Pancreatic β Cells in Type 2 Diabetes. Nutrients 2020, 12, 2954. [Google Scholar] [CrossRef]
- Singh, C.K.; Chhabra, G.; Ndiaye, M.A.; Garcia-Peterson, L.M.; Mack, N.J.; Ahmad, N. The Role of Sirtuins in Antioxidant and Redox Signaling. Antioxid. Redox Signal. 2018, 28, 643–661. [Google Scholar] [CrossRef]
- Liu, J.; Chen, S.; Biswas, S.; Nagrani, N.; Chu, Y.; Chakrabarti, S.; Feng, B. Glucose-Induced Oxidative Stress and Accelerated Aging in Endothelial Cells Are Mediated by the Depletion of Mitochondrial SIRTs. Physiol. Rep. 2020, 8, e14331. [Google Scholar] [CrossRef]
- Wang, X.; Meng, L.; Zhao, L.; Wang, Z.; Liu, H.; Liu, G.; Guan, G. Resveratrol Ameliorates Hyperglycemia-Induced Renal Tubular Oxidative Stress Damage via Modulating the SIRT1/FOXO3a Pathway. Diabetes Res. Clin. Pract. 2017, 126, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Chi, Y.; Kang, Y.; Lu, H.; Niu, H.; Liu, W.; Li, Y. Resveratrol Ameliorates Podocyte Damage in Diabetic Mice via SIRT1/PGC-1α Mediated Attenuation of Mitochondrial Oxidative Stress. J. Cell. Physiol. 2019, 234, 5033–5043. [Google Scholar] [CrossRef] [PubMed]
- Bernal-Ramirez, J.; Silva-Platas, C.; Jerjes-Sánchez, C.; Chapoy-Villanueva, H.; Ramos, M.; Garcia, N.; García-Rivas, G. Resveratrol Preserves Mitochondrial Function By Sirt3 Activation in a Right Ventricle Dysfunction Model. J. Card. Fail. 2022, 28, S82. [Google Scholar] [CrossRef]
- Kim, A.; Lee, W.; Yun, J.-M. Luteolin and Fisetin Suppress Oxidative Stress by Modulating Sirtuins and Forkhead Box O3a Expression under in vitro Diabetic Conditions. Nutr. Res. Pract. 2017, 11, 430–434. [Google Scholar] [CrossRef]
- Hu, H.-C.; Lei, Y.-H.; Zhang, W.-H.; Luo, X.-Q. Antioxidant and Anti-Inflammatory Properties of Resveratrol in Diabetic Nephropathy: A Systematic Review and Meta-Analysis of Animal Studies. Front. Pharm. 2022, 13, 841818. [Google Scholar] [CrossRef] [PubMed]
- Xian, Y.; Gao, Y.; Lv, W.; Ma, X.; Hu, J.; Chi, J.; Wang, W.; Wang, Y. Resveratrol Prevents Diabetic Nephropathy by Reducing Chronic Inflammation and Improving the Blood Glucose Memory Effect in Non-Obese Diabetic Mice. Naunyn-Schmiedeberg’s Arch. Pharm. 2020, 393, 2009–2017. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; Dinkova-Kostova, A.T. The Nrf2 Regulatory Network Provides an Interface between Redox and Intermediary Metabolism. Trends Biochem. Sci. 2014, 39, 199–218. [Google Scholar] [CrossRef]
- Nguyen, P.M.; Park, M.S.; Chow, M.; Chang, J.H.; Wrischnik, L.; Chan, W.K. Benzo[a]Pyrene Increases the Nrf2 Content by Downregulating the Keap1 Message. Toxicol. Sci. 2010, 116, 549–561. [Google Scholar] [CrossRef]
- Rada, P.; Rojo, A.I.; Evrard-Todeschi, N.; Innamorato, N.G.; Cotte, A.; Jaworski, T.; Tobón-Velasco, J.C.; Devijver, H.; García-Mayoral, M.F.; Van Leuven, F.; et al. Structural and Functional Characterization of Nrf2 Degradation by the Glycogen Synthase Kinase 3/β-TrCP Axis. Mol. Cell. Biol. 2012, 32, 3486–3499. [Google Scholar] [CrossRef]
- Wang, H.; Liu, K.; Geng, M.; Gao, P.; Wu, X.; Hai, Y.; Li, Y.; Li, Y.; Luo, L.; Hayes, J.D.; et al. RXRα Inhibits the NRF2-ARE Signaling Pathway through a Direct Interaction with the Neh7 Domain of NRF2. Cancer Res. 2013, 73, 3097–3108. [Google Scholar] [CrossRef] [PubMed]
- Motohashi, H.; Katsuoka, F.; Engel, J.D.; Yamamoto, M. Small Maf Proteins Serve as Transcriptional Cofactors for Keratinocyte Differentiation in the Keap1–Nrf2 Regulatory Pathway. Proc. Natl. Acad. Sci. USA 2004, 101, 6379–6384. [Google Scholar] [CrossRef] [PubMed]
- Kopacz, A.; Kloska, D.; Forman, H.J.; Jozkowicz, A.; Grochot-Przeczek, A. Beyond Repression of Nrf2: An Update on Keap1. Free Radic. Biol. Med. 2020, 157, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.Y.; Wang, C.C.; Lai, T.-I.; Tsu, H.N.; Wang, C.H.; Liang, H.Y.; Kuo, W.W. Antioxidant Effects of Diallyl Trisulfide on High Glucose-Induced Apoptosis Are Mediated by the PI3K/Akt-Dependent Activation of Nrf2 in Cardiomyocytes. Int. J. Cardiol. 2013, 168, 1286–1297. [Google Scholar] [CrossRef] [PubMed]
- Mohan, T.; Narasimhan, K.K.S.; Ravi, D.B.; Velusamy, P.; Chandrasekar, N.; Chakrapani, L.N.; Srinivasan, A.; Karthikeyan, P.; Kannan, P.; Tamilarasan, B.; et al. Role of Nrf2 Dysfunction in the Pathogenesis of Diabetic Nephropathy: Therapeutic Prospect of Epigallocatechin-3-Gallate. Free Radic. Biol. Med. 2020, 160, 227–238. [Google Scholar] [CrossRef]
- Nezu, M.; Souma, T.; Yu, L.; Suzuki, T.; Saigusa, D.; Ito, S.; Suzuki, N.; Yamamoto, M. Transcription Factor Nrf2 Hyperactivation in Early-Phase Renal Ischemia-Reperfusion Injury Prevents Tubular Damage Progression. Kidney Int. 2017, 91, 387–401. [Google Scholar] [CrossRef]
- Sun, W.; Liu, X.; Zhang, H.; Song, Y.; Li, T.; Liu, X.; Liu, Y.; Guo, L.; Wang, F.; Yang, T.; et al. Epigallocatechin Gallate Upregulates NRF2 to Prevent Diabetic Nephropathy via Disabling KEAP1. Free Radic. Biol. Med. 2017, 108, 840–857. [Google Scholar] [CrossRef]
- ALTamimi, J.Z.; AlFaris, N.A.; Alshammari, G.M.; Alagal, R.I.; Aljabryn, D.H.; Aldera, H.; Alrfaei, B.M.; Alkhateeb, M.A.; Yahya, M.A. Ellagic Acid Protects against Diabetic Nephropathy in Rats by Regulating the Transcription and Activity of Nrf2. J. Funct. Foods 2021, 79, 104397. [Google Scholar] [CrossRef]
- Alaofi, A.L. Sinapic Acid Ameliorates the Progression of Streptozotocin (STZ)-Induced Diabetic Nephropathy in Rats via NRF2/HO-1 Mediated Pathways. Front. Pharmacol. 2020, 11, 1119. [Google Scholar] [CrossRef]
- Zhu, X.; Chen, Y.; Chen, Q.; Yang, H.; Xie, X. Astaxanthin Promotes Nrf2/ARE Signaling to Alleviate Renal Fibronectin and Collagen IV Accumulation in Diabetic Rats. J. Diabetes Res. 2018, 2018, e6730315. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, Z.; Gong, W.; Zou, Y.; Xu, F.; Chen, L.; Huang, H. Paeonol Ameliorates Diabetic Renal Fibrosis Through Promoting the Activation of the Nrf2/ARE Pathway via Up-Regulating Sirt1. Front. Pharmacol. 2018, 9, 512. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-J.; Kong, L.; Tang, Z.-Z.; Zhang, Y.-M.; Liu, Y.; Wang, T.-Y.; Liu, Y.-W. Hesperetin Ameliorates Diabetic Nephropathy in Rats by Activating Nrf2/ARE/Glyoxalase 1 Pathway. Biomed. Pharmacother. 2019, 111, 1166–1175. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.; Cheng, Y.; Dong, X.; Zhang, M.; Pei, C.; Zhang, M. Effects of Rhaponticin on Retinal Oxidative Stress and Inflammation in Diabetes through NRF2/HO-1/NF-κB Signalling. J. Biochem. Mol. Toxicol. 2020, 34, e22568. [Google Scholar] [CrossRef] [PubMed]
- Xie, T.; Chen, X.; Chen, W.; Huang, S.; Peng, X.; Tian, L.; Wu, X.; Huang, Y. Curcumin Is a Potential Adjuvant to Alleviates Diabetic Retinal Injury via Reducing Oxidative Stress and Maintaining Nrf2 Pathway Homeostasis. Front. Pharmacol. 2021, 12, 796565. [Google Scholar] [CrossRef]
- Bucolo, C.; Drago, F.; Maisto, R.; Romano, G.L.; D’Agata, V.; Maugeri, G.; Giunta, S. Curcumin Prevents High Glucose Damage in Retinal Pigment Epithelial Cells through ERK1/2-mediated Activation of the Nrf2/HO-1 Pathway. J. Cell. Physiol. 2019, 234, 17295–17304. [Google Scholar] [CrossRef]
- Song, Y.; Huang, L.; Yu, J. Effects of Blueberry Anthocyanins on Retinal Oxidative Stress and Inflammation in Diabetes through Nrf2/HO-1 Signaling. J. Neuroimmunol. 2016, 301, 1–6. [Google Scholar] [CrossRef]
- Huang, W.; Yan, Z.; Li, D.; Ma, Y.; Zhou, J.; Sui, Z. Antioxidant and Anti-Inflammatory Effects of Blueberry Anthocyanins on High Glucose-Induced Human Retinal Capillary Endothelial Cells. Oxidative Med. Cell. Longev. 2018, 2018, e1862462. [Google Scholar] [CrossRef]
- Herrera-Bravo, J.; Beltrán, J.F.; Huard, N.; Saavedra, K.; Saavedra, N.; Alvear, M.; Lanas, F.; Salazar, L.A. Anthocyanins Found in Pinot Noir Waste Induce Target Genes Related to the Nrf2 Signalling in Endothelial Cells. Antioxidants 2022, 11, 1239. [Google Scholar] [CrossRef]
- Millán, I.; Desco, M.d.C.; Torres-Cuevas, I.; Pérez, S.; Pulido, I.; Mena-Mollá, S.; Mataix, J.; Asensi, M.; Ortega, Á.L. Pterostilbene Prevents Early Diabetic Retinopathy Alterations in a Rabbit Experimental Model. Nutrients 2019, 12, 82. [Google Scholar] [CrossRef]
- Matos, A.L.; Bruno, D.F.; Ambrósio, A.F.; Santos, P.F. The Benefits of Flavonoids in Diabetic Retinopathy. Nutrients 2020, 12, 3169. [Google Scholar] [CrossRef]
- Liang, F.; Wang, J.; Zhu, X.; Wang, Z.; Zheng, J.; Sun, Z.; Xu, S.; Zhang, J.; Zhou, J.; Shi, L. Melatonin Alleviates Neuronal Damage After Intracerebral Hemorrhage in Hyperglycemic Rats. Drug Des. Dev. Ther. 2020, 14, 2573–2584. [Google Scholar] [CrossRef] [PubMed]
- Sandireddy, R.; Yerra, V.G.; Komirishetti, P.; Areti, A.; Kumar, A. Fisetin Imparts Neuroprotection in Experimental Diabetic Neuropathy by Modulating Nrf2 and NF-ΚB Pathways. Cell. Mol. Neurobiol. 2016, 36, 883–892. [Google Scholar] [CrossRef] [PubMed]
- Bachewal, P.; Gundu, C.; Yerra, V.G.; Kalvala, A.K.; Areti, A.; Kumar, A. Morin Exerts Neuroprotection via Attenuation of ROS Induced Oxidative Damage and Neuroinflammation in Experimental Diabetic Neuropathy: Pharmacological Effects of Morin in Diabetic Neuropathy. BioFactors 2018, 44, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Mittal, R.; Kumar, A.; Singh, D.P.; Bishnoi, M.; Nag, T.C. Ameliorative Potential of Rutin in Combination with Nimesulide in STZ Model of Diabetic Neuropathy: Targeting Nrf2/HO-1/NF-KB and COX Signalling Pathway. Inflammopharmacol 2018, 26, 755–768. [Google Scholar] [CrossRef]
- Malakoti, F.; Mohammadi, E.; Akbari Oryani, M.; Shanebandi, D.; Yousefi, B.; Salehi, A.; Asemi, Z. Polyphenols Target MiRNAs as a Therapeutic Strategy for Diabetic Complications. Crit. Rev. Food Sci. Nutr. 2022, 1–17. [Google Scholar] [CrossRef]
- Yehuda, I.; Madar, Z.; Leikin-Frenkel, A.; Szuchman-Sapir, A.; Magzal, F.; Markman, G.; Tamir, S. Glabridin, an Isoflavan from Licorice Root, Upregulates Paraoxonase 2 Expression under Hyperglycemia and Protects It from Oxidation. Mol. Nutr. Food Res. 2016, 60, 287–299. [Google Scholar] [CrossRef]
- Roxo, D.F.; Arcaro, C.A.; Gutierres, V.O.; Costa, M.C.; Oliveira, J.O.; Lima, T.F.O.; Assis, R.P.; Brunetti, I.L.; Baviera, A.M. Curcumin Combined with Metformin Decreases Glycemia and Dyslipidemia, and Increases Paraoxonase Activity in Diabetic Rats. Diabetol. Metab. Syndr. 2019, 11, 33. [Google Scholar] [CrossRef]
- Yehuda, I.; Madar, Z.; Leikin-Frenkel, A.; Tamir, S. Glabridin, an Isoflavan from Licorice Root, Downregulates INOS Expression and Activity under High-Glucose Stress and Inflammation. Mol. Nutr. Food Res. 2015, 59, 1041–1052. [Google Scholar] [CrossRef]
- An, X.; Zhang, Y.; Cao, Y.; Chen, J.; Qin, H.; Yang, L. Punicalagin Protects Diabetic Nephropathy by Inhibiting Pyroptosis Based on TXNIP/NLRP3 Pathway. Nutrients 2020, 12, 1516. [Google Scholar] [CrossRef]
- Kotewicz, M.; Krauze-Baranowska, M.; Daca, A.; Płoska, A.; Godlewska, S.; Kalinowski, L.; Lewko, B. Urolithins Modulate the Viability, Autophagy, Apoptosis, and Nephrin Turnover in Podocytes Exposed to High Glucose. Cells 2022, 11, 2471. [Google Scholar] [CrossRef]
- Iatcu, C.O.; Steen, A.; Covasa, M. Gut Microbiota and Complications of Type-2 Diabetes. Nutrients 2022, 14, 166. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Jian, S.; Guo, D.; Wen, C.; Xin, Z.; Zhang, L.; Kuang, T.; Wen, J.; Yin, Y.; Deng, B. Fecal Microbiota and Metabolomics Revealed the Effect of Long-Term Consumption of Gallic Acid on Canine Lipid Metabolism and Gut Health. Food Chem. X 2022, 15, 100377. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Zhuo, L.; He, Y.; Fu, Y.; Shen, L.; Xu, F.; Gou, W.; Miao, Z.; Shuai, M.; Liang, Y.; et al. The Gut Microbiota-Bile Acid Axis Links the Positive Association between Chronic Insomnia and Cardiometabolic Diseases. Nat. Commun. 2022, 13, 3002. [Google Scholar] [CrossRef]
- Martín-Peláez, S.; Mosele, J.I.; Pizarro, N.; Farràs, M.; de la Torre, R.; Subirana, I.; Pérez-Cano, F.J.; Castañer, O.; Solà, R.; Fernandez-Castillejo, S.; et al. Effect of Virgin Olive Oil and Thyme Phenolic Compounds on Blood Lipid Profile: Implications of Human Gut Microbiota. Eur. J. Nutr. 2017, 56, 119–131. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, M.; Zhang, Y.; Tian, M.; Chen, P.; Lan, Y.; Zhou, B. Urolithin A Alleviates Acute Kidney Injury Induced by Renal Ischemia Reperfusion through the P62-Keap1-Nrf2 Signaling Pathway. Phytother. Res. 2022, 36, 984–995. [Google Scholar] [CrossRef] [PubMed]
- Gerasimova, E.L.; Gazizullina, E.R.; Borisova, M.V.; Igdisanova, D.I.; Nikiforov, E.A.; Moseev, T.D.; Varaksin, M.V.; Chupakhin, O.N.; Charushin, V.N.; Ivanova, A.V. Design and Antioxidant Properties of Bifunctional 2H-Imidazole-Derived Phenolic Compounds—A New Family of Effective Inhibitors for Oxidative Stress-Associated Destructive Processes. Molecules 2021, 26, 6534. [Google Scholar] [CrossRef] [PubMed]
- Yates, M.S.; Tauchi, M.; Katsuoka, F.; Flanders, K.C.; Liby, K.T.; Honda, T.; Gribble, G.W.; Johnson, D.A.; Johnson, J.A.; Burton, N.C.; et al. Pharmacodynamic Characterization of Chemopreventive Triterpenoids as Exceptionally Potent Inducers of Nrf2-Regulated Genes. Mol. Cancer Ther. 2007, 6, 154–162. [Google Scholar] [CrossRef]
- Rahnasto-Rilla, M.; Tyni, J.; Huovinen, M.; Jarho, E.; Kulikowicz, T.; Ravichandran, S.; Bohr, V.A.; Ferrucci, L.; Lahtela-Kakkonen, M.; Moaddel, R. Natural Polyphenols as Sirtuin 6 Modulators. Sci. Rep. 2018, 8, 4163. [Google Scholar] [CrossRef]
- Kancheva, V.D.; Dettori, M.A.; Fabbri, D.; Alov, P.; Angelova, S.E.; Slavova-Kazakova, A.K.; Carta, P.; Menshov, V.A.; Yablonskaya, O.I.; Trofimov, A.V.; et al. Natural Chain-Breaking Antioxidants and Their Synthetic Analogs as Modulators of Oxidative Stress. Antioxidants 2021, 10, 624. [Google Scholar] [CrossRef]
- Rodriguez-Mateos, A.; Pino-García, R.D.; George, T.W.; Vidal-Diez, A.; Heiss, C.; Spencer, J.P.E. Impact of Processing on the Bioavailability and Vascular Effects of Blueberry (Poly)Phenols. Mol. Nutr. Food Res. 2014, 58, 1952–1961. [Google Scholar] [CrossRef]
- Abukhabta, S.; Khalil Ghawi, S.; Karatzas, K.A.; Charalampopoulos, D.; McDougall, G.; Allwood, J.W.; Verrall, S.; Lavery, S.; Latimer, C.; Pourshahidi, L.K.; et al. Sulforaphane-Enriched Extracts from Glucoraphanin-Rich Broccoli Exert Antimicrobial Activity against Gut Pathogens in Vitro and Innovative Cooking Methods Increase in Vivo Intestinal Delivery of Sulforaphane. Eur. J. Nutr. 2021, 60, 1263–1276. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo-Liberona, N.; González-Domínguez, R.; Vegas, E.; Riso, P.; Del Bo’, C.; Bernardi, S.; Peron, G.; Guglielmetti, S.; Gargari, G.; Kroon, P.A.; et al. Increased Intestinal Permeability in Older Subjects Impacts the Beneficial Effects of Dietary Polyphenols by Modulating Their Bioavailability. J. Agric. Food Chem. 2020, 68, 12476–12484. [Google Scholar] [CrossRef] [PubMed]
- Williamson, G.; Kay, C.D.; Crozier, A. The Bioavailability, Transport, and Bioactivity of Dietary Flavonoids: A Review from a Historical Perspective. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1054–1112. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Milenkovic, D.; Van de Wiele, T.; Rodriguez-Mateos, A.; de Roos, B.; Garcia-Conesa, M.T.; Landberg, R.; Gibney, E.R.; Heinonen, M.; Tomás-Barberán, F.; et al. Addressing the Inter-Individual Variation in Response to Consumption of Plant Food Bioactives: Towards a Better Understanding of Their Role in Healthy Aging and Cardiometabolic Risk Reduction. Mol. Nutr. Food Res. 2017, 61, 1600557. [Google Scholar] [CrossRef] [PubMed]
- Polia, F.; Pastor-Belda, M.; Martínez-Blázquez, A.; Horcajada, M.-N.; Tomás-Barberán, F.A.; García-Villalba, R. Technological and Biotechnological Processes To Enhance the Bioavailability of Dietary (Poly)Phenols in Humans. J. Agric. Food Chem. 2022, 70, 2092–2107. [Google Scholar] [CrossRef]
- Iglesias-Aguirre, C.E.; Vallejo, F.; Beltrán, D.; Aguilar-Aguilar, E.; Puigcerver, J.; Alajarín, M.; Berná, J.; Selma, M.V.; Espín, J.C. Lunularin Producers versus Non-Producers: Novel Human Metabotypes Associated with the Metabolism of Resveratrol by the Gut Microbiota. J. Agric. Food Chem. 2022, 70, 10521–10531. [Google Scholar] [CrossRef]
- Cortés-Martín, A.; Selma, M.V.; Tomás-Barberán, F.A.; González-Sarrías, A.; Espín, J.C. Where to Look into the Puzzle of Polyphenols and Health? The Postbiotics and Gut Microbiota Associated with Human Metabotypes. Mol. Nutr. Food Res. 2020, 64, 1900952. [Google Scholar] [CrossRef]
- Frankenfeld, C.L. Cardiometabolic Risk and Gut Microbial Phytoestrogen Metabolite Phenotypes. Mol. Nutr. Food Res. 2017, 61, 1500900. [Google Scholar] [CrossRef]
- Cortés-Martín, A.; Iglesias-Aguirre, C.E.; Meoro, A.; Selma, M.V.; Espín, J.C. Pharmacological Therapy Determines the Gut Microbiota Modulation by a Pomegranate Extract Nutraceutical in Metabolic Syndrome: A Randomized Clinical Trial. Mol. Nutr. Food Res. 2021, 65, 2001048. [Google Scholar] [CrossRef]
- Islam, F.; Khadija, J.F.; Islam, M.R.; Shohag, S.; Mitra, S.; Alghamdi, S.; Babalghith, A.O.; Theyab, A.; Rahman, M.T.; Akter, A.; et al. Investigating Polyphenol Nanoformulations for Therapeutic Targets against Diabetes Mellitus. Evid.-Based Complement. Altern. Med. 2022, 2022, e5649156. [Google Scholar] [CrossRef]
- de Oliveira, W.Q.; Neri-Numa, I.A.; Arruda, H.S.; McClements, D.J.; Pastore, G.M. Encapsulation of Flavonoids in Foods for Diabetics: The Emerging Paradigm for an Effective Therapy. Trends Food Sci. Technol. 2022, 127, 198–206. [Google Scholar] [CrossRef]
- Haider, M.; Abdin, S.M.; Kamal, L.; Orive, G. Nanostructured Lipid Carriers for Delivery of Chemotherapeutics: A Review. Pharmaceutics 2020, 12, 288. [Google Scholar] [CrossRef] [PubMed]
- Medici, S.; Peana, M.; Pelucelli, A.; Zoroddu, M.A. An Updated Overview on Metal Nanoparticles Toxicity. Semin. Cancer Biol. 2021, 76, 17–26. [Google Scholar] [CrossRef] [PubMed]
| Antioxidant System | Examples of Polyphenol | Effects |
|---|---|---|
| UCP2/mitochondrial Complex 1 | Caffeic acid Anthocyanins | In vitro protection of retinal cells, improving mitochondrial function [160]. Improve cardiac function by preserving mitochondrial function in diabetic rat model [166]. |
| SIRTs | Resveratrol | Improve GSH, CAT SOD activity and mitochondria function via activation of SIRT1/PGC-1α/FOXO3a. |
| GSH/GPx4/Xc− system (Inhibition of ferroptosis) | Glabridin and calycosin | Reno-protective effect in DN rat model [183,184]. |
| Naringenin | Cardio protective effect [185]. | |
| Astragaloside-IV | In vitro protection of retinal cells [186]. | |
| Quercetin and cryptochlorogenic acid | Preserving β-cell function [87,188]. | |
| Nrf-2 | Epigallocatechin-3-gallate Curcumin | Reno-protective effect in DN [206]. Protective effective against retinal injury in diabetes [213]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akpoveso, O.-O.P.; Ubah, E.E.; Obasanmi, G. Antioxidant Phytochemicals as Potential Therapy for Diabetic Complications. Antioxidants 2023, 12, 123. https://doi.org/10.3390/antiox12010123
Akpoveso O-OP, Ubah EE, Obasanmi G. Antioxidant Phytochemicals as Potential Therapy for Diabetic Complications. Antioxidants. 2023; 12(1):123. https://doi.org/10.3390/antiox12010123
Chicago/Turabian StyleAkpoveso, Oke-Oghene Philomena, Emeka Emmanuel Ubah, and Gideon Obasanmi. 2023. "Antioxidant Phytochemicals as Potential Therapy for Diabetic Complications" Antioxidants 12, no. 1: 123. https://doi.org/10.3390/antiox12010123
APA StyleAkpoveso, O.-O. P., Ubah, E. E., & Obasanmi, G. (2023). Antioxidant Phytochemicals as Potential Therapy for Diabetic Complications. Antioxidants, 12(1), 123. https://doi.org/10.3390/antiox12010123





