The Role Played by Ferroptosis in Osteoarthritis: Evidence Based on Iron Dyshomeostasis and Lipid Peroxidation
Abstract
1. Introduction
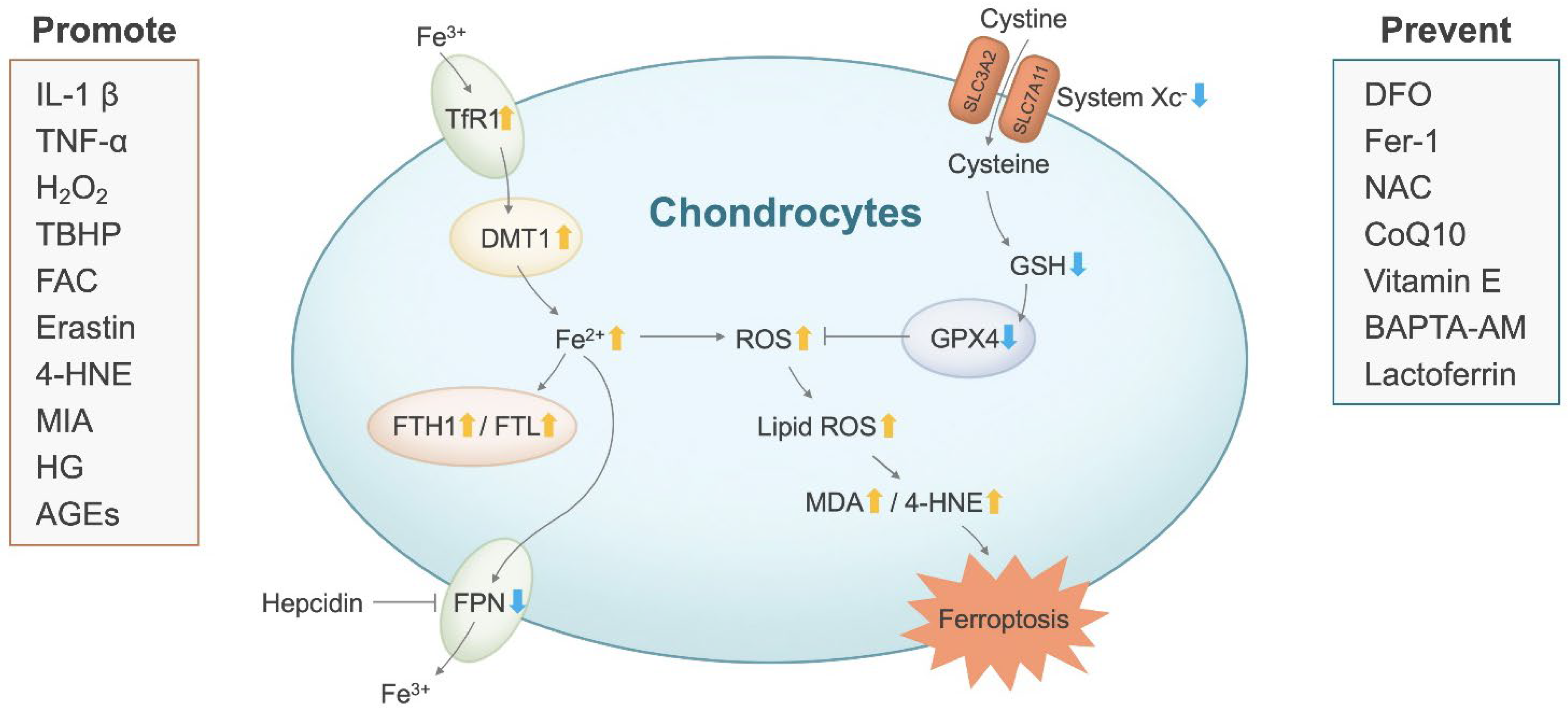
2. Main Characteristics of Ferroptosis
2.1. Iron Homeostasis and Ferroptosis
2.2. Lipid Peroxidation and Ferroptosis
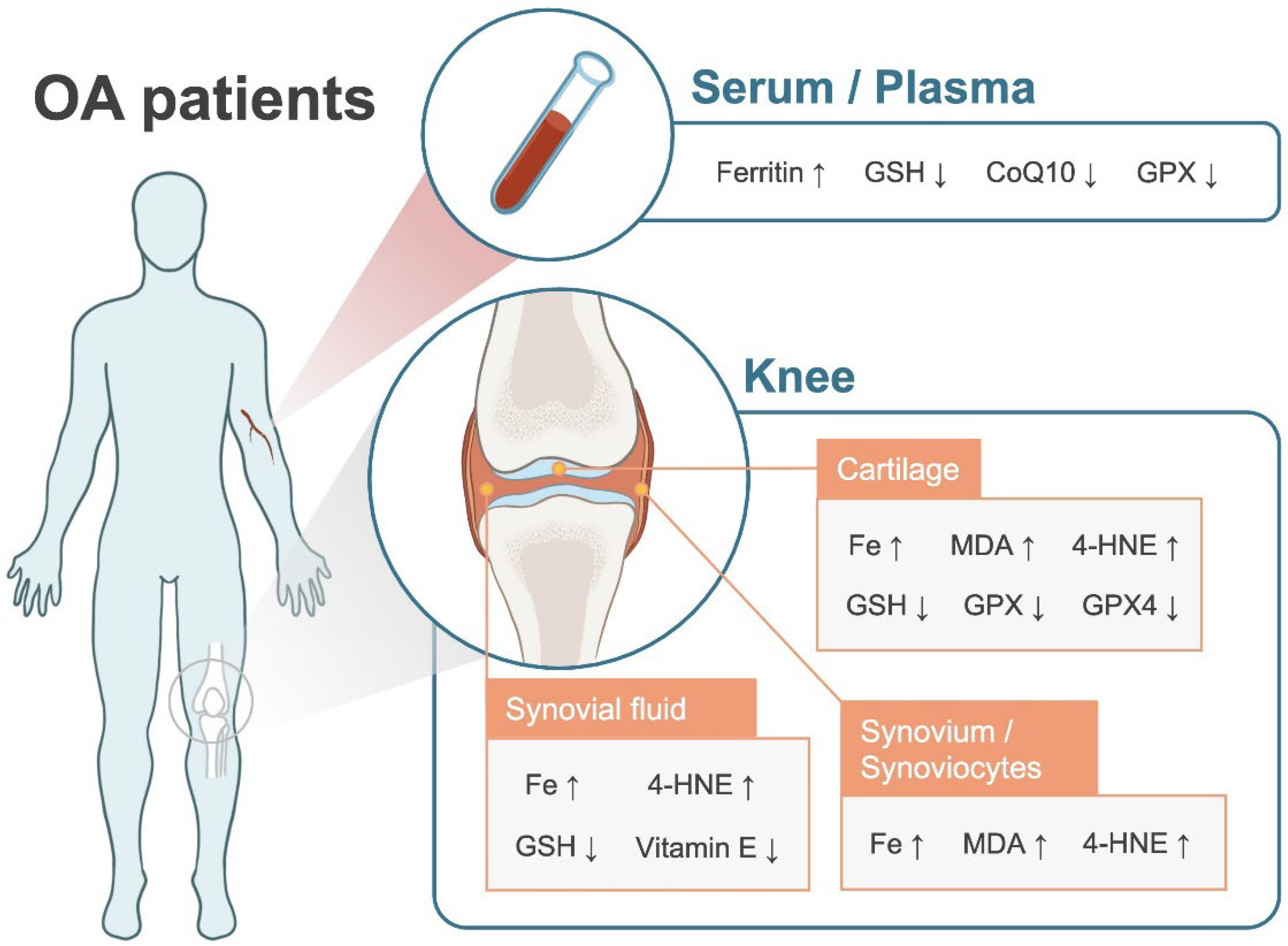
3. Potential Association between Ferroptosis and OA: Clinical Findings
3.1. Iron Dyshomeostasis
3.2. Lipid Peroxidation
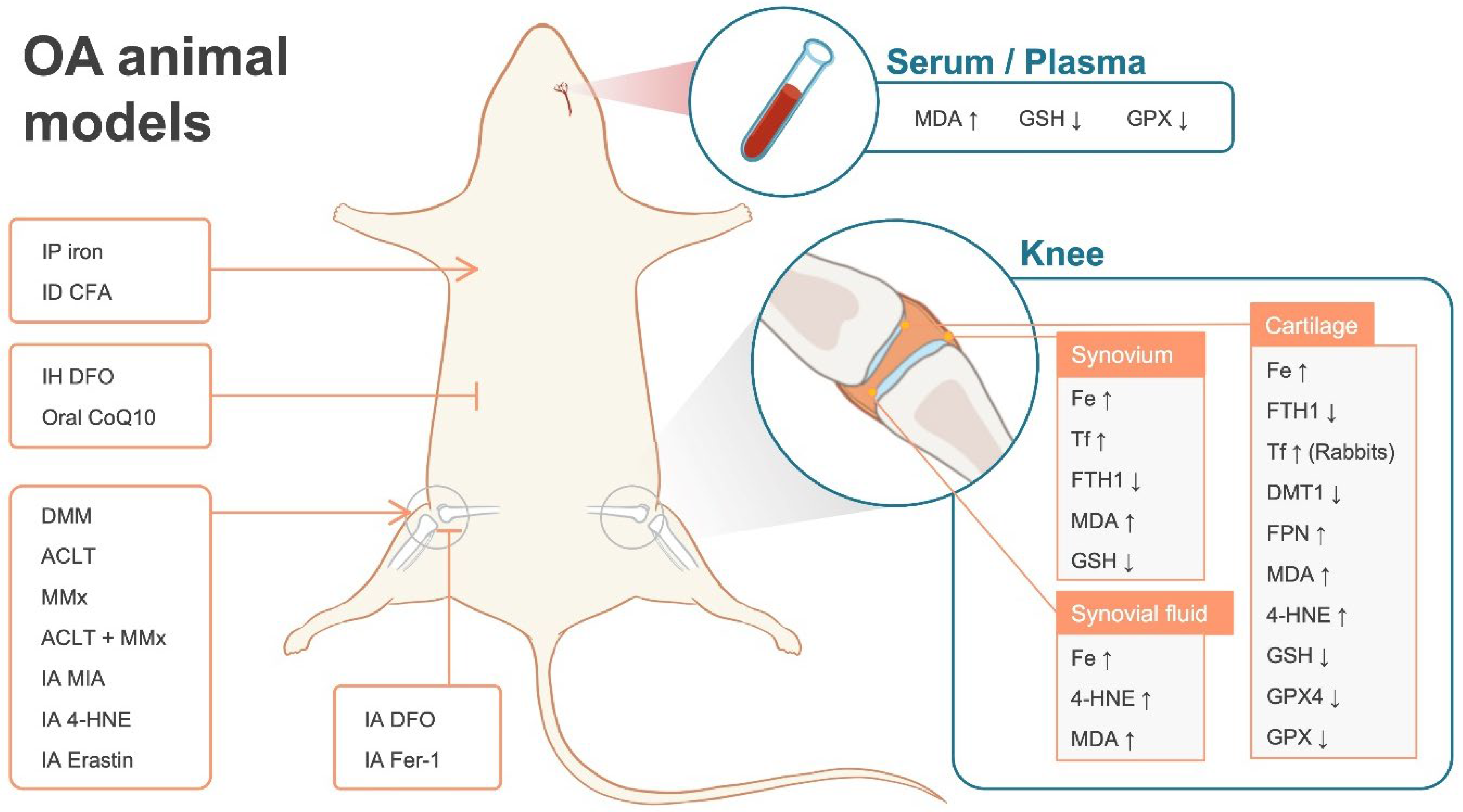
4. Potential Association between Ferroptosis and OA: Animal Models
4.1. Iron Dyshomeostasis
4.2. Lipid Peroxidation
4.3. Iron Dyshomeostasis and Lipid Peroxidation
5. Potential Association between Ferroptosis and OA: Cell Research
5.1. Iron Dyshomeostasis
5.2. Lipid Peroxidation
5.3. Iron Dyshomeostasis and Lipid Peroxidation
6. Discussion
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
Appendix A
| Index | Location | Number of Patients | Reference | |
|---|---|---|---|---|
| Iron dyshomeostasis | Fe ↑ | Cartilage | OA: undamaged = 4:4 | Miao et al. [9] |
| Synovial fluid | K–L stage 1–4 = 9:11:7:3 | Miao et al. [9] | ||
| Synovial fluid | OA: control = 25:25 | Yazar et al. [38] | ||
| Synovium | OA: control = 25:20 | Ogilvie-Harris et al. [39] | ||
| Ferritin ↑ | Serum | OB stage 1–4 = 10:10:10:10 | Nugzar et al. [43] | |
| Serum | OA: control = 129:20 | Kennish et al. [44] | ||
| Lipid peroxidation | MDA ↑ | Synoviocytes | OA: control = 14:10 | Grigolo et al. [46] |
| Cartilage | OA: control = 14:3 | Shah et al. [48] | ||
| Cartilage | OA: control = 11:11 | Gavriilidis et al. [49] | ||
| 4-HNE ↑ | Synoviocytes | OA: control = 14:10 | Grigolo et al. [46] | |
| Synovial fluid | OA: control = 18:5 | Morquette et al. [47] | ||
| Cartilage | OA: control = 14:3 | Shah et al. [48] | ||
| GSH ↓ | Cartilage | OA: undamaged = 7:7 | Miao et al. [9] | |
| Synovial fluid | OA: control = 27:12 | Regan et al. [50] | ||
| Plasma | OA: control = 15:15 | Maneesh et al. [51] | ||
| GPX ↓ | Cartilage | OA: undamaged = 4:4 | Miao et al. [9] | |
| Plasma | OA: control = 15:15 | Maneesh et al. [51] | ||
| GPX4 ↓ | Cartilage | OA: undamaged = 55:55 | Miao et al. [9] | |
| Vitamin E ↓ | Synovial fluid | OA: control = 32:10 | Sutipornpalangkul et al. [53] | |
| Synovial fluid | KSS > 46: ≤46 = 14:9 | Angthong et al. [54] | ||
| CoQ10 ↓ | Plasma | OA: control = 74:33 | Chang et al. [118] | |
| Index | Intervention | Control | Animal | Effects on Ferroptosis | Effects on OA | Reference |
|---|---|---|---|---|---|---|
| Iron dyshomeostasis | IP iron dextran | IP dextran | Strain 13 guinea pigs | Fe ↑, DMT1 ↓, FPN ↑, and FTH1 ↑ in cartilage and infrapatellar fat pads, and TfR ↓ in cartilage | Promote | Burton et al. [56] |
| IP iron dextran and DMM | IP iron dextran or DMM | C57BL/6 mice | Fe ↑ in cartilage and synovium | Promote | Jing et al. [57] | |
| DMM | Sham | SD rats | Fe ↑ in synovial fluid | Promote | Lv et al. [82] | |
| ACLT | Sham | C57BL/6 mice | FTH1 ↓ in cartilage | Promote | Miao et al. [9] | |
| ACLT | Sham | New Zealand rabbits | Tf ↑ and FTH1 ↓ in synovium | Promote | Luo et al. [59] | |
| Obese model | Calorie-restricted model | Dunkin-Hartley guinea pigs | TfR1 ↑ and ferritin ↑ in cartilage | Promote | Radakovich et al. [58] | |
| IH DFO | IH sodium lactate solution | Dunkin-Hartley guinea pigs | Fe ↑ in serum and Fe ↓ in cartilage | Prevent | Burton et al. [60] | |
| IA DFO or Fer-1 after ACLT | ACLT | C57BL/6 mice | FTH1 ↑ in cartilage | Prevent | Miao et al. [9] | |
| IA DFO after DMM | DMM | C57BL/6 mice | ― | Prevent | Guo et al. [83] | |
| IA DFO after IA erastin | IA erastin | C57BL/6 mice | ― | Prevent | Guo et al. [83] | |
| Lipid peroxidation | ACLT | Sham | C57BL/6 mice | GPX4 ↓ in cartilage | Promote | Miao et al. [9] |
| ACLT | Sham | Rabbits | MDA ↑ in serum | Promote | Karakurum et al. [61] | |
| ACLT | Sham | SD rats | MDA ↑, GSH ↓, and GPX ↓ in serum, and 4-HNE ↑ in cartilage | Promote | Yang et al. [62] | |
| ACLT | Sham | Rats | MDA ↑ in serum | Promote | Gladkova et al. [65], Zubavlenko et al. [66] | |
| ACLT | Sham | New Zealand rabbits | MDA ↑ and GSH ↓ in synovium and cartilage | Promote | Bai et al. [68] | |
| ACLT | Sham | Dogs | 4-HNE ↑ in synovial fluid and cartilage | Promote | Shi et al. [70] | |
| ACLT | Sham | C57BL/6 mice | MDA ↑ and GPX4 ↓ in cartilage | Promote | Zhou et al. [71] | |
| ACLT | Before ACLT | Dogs | MDA ↑ in serum | Promote | Goranov et al. [63] | |
| ACLT | Before ACLT | C57BL/6 mice | 4-HNE ↑ in cartilage | Promote | Aulin et al. [69] | |
| ACLT+MMx | Sham | Obese SD rats | MDA ↑ in serum | Promote | Chang et al. [64] | |
| MMx | Sham | SD rats | GSH ↓ and GPX ↓ in cartilage | Promote | Qiu et al. [72] | |
| DMM | Sham | C57BL/6 mice | GPX4 ↓ in cartilage | Promote | Yao et al. [8] | |
| DMM | Sham | SD rats | MDA ↑ and GSH ↓ in serum | Promote | Bai et al. [67] | |
| DMM | Sham | SD rats | MDA ↑ in synovial fluid, and GPX4 ↓ in cartilage | Promote | Lv et al. [82] | |
| IA MIA | IA NS | Wistar rats | MDA ↑ and GSH ↓ in plasma | Promote | Pathak et al. [73] Abdel Jaleel et al. [74] | |
| IA MIA | IA NS | Wistar rats | GSH ↓ in serum | Promote | Huang et al. [75] | |
| IA MIA | IA NS | SD rats | MDA ↑, GSH ↓, and GPX ↓ in serum | Promote | Fusco et al. [76] | |
| IA MIA | IA NS | Wistar rats | MDA ↑ and GPX ↓ in serum | Promote | Yabas et al. [77] | |
| IA MIA | IA NS | Wistar rats | GSH ↓ in serum | Promote | Ragab et al. [78]. | |
| IA MIA | Untreated | Wistar rats | GSH ↓ in cartilage | Promote | Ajeeshkumar et al. [79] | |
| ID CFA | Untreated | SD rats | MDA ↑ and GPX ↓ in serum | Promote | Ma et al. [80] | |
| IA 4-HNE | IA NS | Dogs | ― | Promote | Shi et al. [70] | |
| IA Fer-1 after DMM | DMM | C57BL/6 mice | GPX4 ↑ in cartilage | Prevent | Yao et al. [8] | |
| IA Fer-1 or DFO after ACLT | ACLT | C57BL/6 mice | GPX4 ↑ in cartilage | Prevent | Miao et al. [9] | |
| Oral CoQ10 after IA MIA | IA MIA | Wistar rats | ― | Prevent | Lee et al. [119] |
| Index | Intervention | Control | Cells | Effects on Ferroptosis | Effects on OA | Reference |
|---|---|---|---|---|---|---|
| Iron dyshomeostasis | FAC | Control | C-20/A4 human chondrocytes | Intracellular iron ↑, FTH1 ↑, hepcidin ↓, FPN ↓, TfR1 ↓, and TfR2 ↓ | Promote | Karim et al. [85] |
| DFO | Control | Human OA cartilage | ― | Prevent | Tchetina et al. [88] | |
| Lactoferrin + IL-1β | IL-1β | Human chondrocytes | ― | Prevent | Rasheed et al. [89] | |
| Lipid peroxidation | 4-HNE | Control | Human OA cartilage or chondrocytes | ― | Promote | Morquette et al. [47] |
| 4-HNE | Control | Human OA chondrocytes | ― | Promote | Vaillancourt et al. [90] | |
| 4-HNE or IL-1β | Control | Human OA chondrocytes | GSH ↓ | Promote | Benabdoune et al. [92] | |
| IL-1β | Control | C28/I2 human chondrocytes | Intracellular ROS ↑, MDA ↑, GPX1 ↓, and GPX4 ↓ | Promote | Hosseinzadeh et al. [95,96] | |
| IL-1β | Control | HC-A human chondrocytes | Intracellular ROS ↑ and GPX ↓ | Promote | Zuo et al. [97] | |
| IL-1β | Control | C28/I2 human chondrocytes | Intracellular ROS ↑ and GPX ↓ | Promote | Yin et al. [98] | |
| IL-1β | Control | CHON-001 human chondrocytes | Intracellular ROS ↑, MDA ↑, and GSH ↓ | Promote | Zhu et al. [99] | |
| TNF-α | Control | CHON-001 human chondrocytes | Intracellular ROS ↑ and GSH ↓ | Promote | Wang et al. [100] | |
| HG | Control | C28/I2 human chondrocytes | Intracellular ROS ↑, MDA ↑, GPX1 ↓, GPX3 ↓, and GPX4 ↓ | Promote | Hosseinzadeh et al. [106] | |
| AGEs | Control | Human chondrocytes | Intracellular ROS ↑ and GSH ↓ | Promote | Hu et al. [108] | |
| SAC or colchicine | Control | Human OA chondrocytes | Intracellular ROS ↓, LPO ↓, 4-HNE ↓, and GPX ↑ | Prevent | Elmazoglu et al. [93] | |
| Nifedipine | Control | Human OA chondrocytes | Intracellular ROS ↓, and GPX ↑ | Prevent | Yao et al. [94] |
| Index | Intervention | Control | Cells | Effects on Ferroptosis | Effects on OA | Reference |
|---|---|---|---|---|---|---|
| Iron dyshomeostasis | Fe3+, or Fe2+, or ferritin | Control | Rabbit chondrocytes | ― | Promote | Kirkpatrick et al. [84] |
| FAC | Control | Mouse chondrocytes | ― | Promote | Jing et al. [57] | |
| FAC | Control | Mouse chondrocytes | Intracellular iron ↑ and ROS ↑ | Promote | Jing et al. [86] | |
| FAC | Control | ATDC5 mouse chondrocytes | Intracellular iron ↑, FTH1 ↑, and FTL ↑ | Promote | Ohno et al. [87] | |
| IL-1β | Control | Mouse chondrocytes | Intracellular iron ↑, TfR1 ↑, DMT1 ↑, and FPN ↓ | Promote | Jing et al. [57] | |
| IL-1β | Control | Mouse chondrocytes | Intracellular iron ↑ | Promote | Lv et al. [82] | |
| IL-1β | Control | ATDC5 mouse chondrocytes | Intracellular iron ↑ and TfR1↑ | Promote | Mo et al. [117] | |
| IL-1β or TNF-α | Control | Mouse chondrocytes | TfR1 ↑ and FPN ↓ | Promote | Jing et al. [116] | |
| IL-1β or erastin | Control | Mouse chondrocytes | Intracellular iron ↑ | Promote | Guo et al. [83] | |
| TBHP | Control | Mouse chondrocytes | Intracellular iron ↑ | Promote | Miao et al. [9] | |
| FAC + IL-1β | IL-1β | Mouse chondrocytes | ― | Promote | Jing et al. [116] | |
| DFO + FAC | FAC | Mouse chondrocytes | ― | Prevent | Jing et al. [116] | |
| DFO + IL-1β | IL-1β | Mouse chondrocytes | ― | Prevent | Jing et al. [57] | |
| DFO + TBHP | TBHP | Mouse chondrocytes | Intracellular iron ↓ | Prevent | Miao et al. [9] | |
| DFO + IL-1β or erastin | IL-1β or erastin | Mouse chondrocytes | Intracellular iron ↓ | Prevent | Guo et al. [83] | |
| Fer-1 + IL-1β | IL-1β | Mouse chondrocytes | Intracellular iron↓ | Prevent | Lv et al. [82] | |
| Fer-1 + TBHP | TBHP | Mouse chondrocytes | Intracellular iron ↓ | Prevent | Miao et al. [9] | |
| Fer-1 + IL-1β or erastin | IL-1β or erastin | Mouse chondrocytes | Intracellular iron ↓ | Prevent | Guo et al. [83] | |
| BAPTA-AM + FAC | FAC | Mouse chondrocytes | Intracellular iron ↓ and ROS ↓ | Prevent | Jing et al. [86] | |
| Lipid peroxidation | IL-1β | Control | Mouse chondrocytes | Intracellular MDA ↑, lipid-ROS ↑, GSH ↓, GPX4 ↓, and SLC7A11 ↓ | Promote | Zhou et al. [71] |
| IL-1β | Control | Mouse chondrocytes | Intracellular ROS ↑, MDA ↑, and GPX4 ↓ | Promote | Lv et al. [82] | |
| IL-1β | Control | ATDC5 mouse chondrocytes | Intracellular MDA ↑, GSH ↓, GPX4 ↓, and SLC7A11 ↓ | Promote | Mo et al. [117] | |
| IL-1β or FAC | Control | Mouse chondrocytes | Intracellular ROS ↑ and lipid-ROS ↑, GPX4 ↓ and SLC7A11 ↓ | Promote | Yao et al. [8] | |
| IL-1β or erastin | Control | Mouse chondrocytes | Intracellular ROS ↑, MDA ↑, lipid-ROS ↑, GPX4 ↓, and SLC7A11 ↓ | Promote | Guo et al. [83] | |
| Erastin | Control | Mouse chondrocytes | ― | Promote | Yao et al. [8] | |
| H2O2 | Control | Canine chondrocytes | GSH ↓ | Promote | Dycus et al. [101] | |
| H2O2 | Control | Rat chondrocytes | Intracellular ROS ↑, GSH ↓, and GPX ↓ | Promote | Guo et al. [102] | |
| H2O2 | Control | Mouse chondrocytes | Intracellular ROS ↑, LPO ↑, and GSH/GSSG ↓ | Promote | Zhang et al. [103] | |
| MIA | Control | Rat chondrocytes | Intracellular ROS ↑ and MDA ↑ | Promote | Qiao et al. [105] | |
| TBHP | Control | Mouse chondrocytes | Intracellular ROS ↑, MDA ↑, lipid-ROS ↑, GSH ↓, and GPX ↓ | Promote | Miao et al. [9] | |
| Fe2+ + TBHP or H2O2 | TBHP or H2O2 | Bovine chondrocytes | LPO ↑ | Promote | Dombrecht et al. [115] | |
| DFO + TBHP | TBHP | Mouse chondrocytes | Intracellular ROS ↓, MDA ↓, lipid-ROS ↓, GSH ↑, and GPX ↑ | Prevent | Miao et al. [9] | |
| DFO + IL-1β or erastin | IL-1β or erastin | Mouse chondrocytes | Intracellular ROS ↓, MDA ↓, lipid-ROS ↓, GPX4 ↑, and SLC7A11 ↑ | Prevent | Guo et al. [83] | |
| Fer-1 + IL-1β | IL-1β | Mouse chondrocytes | Intracellular ROS ↓, MDA ↓, and GPX4 ↑ | Prevent | Lv et al. [82] | |
| Fer-1 + IL-1β | IL-1β | ATDC5 mouse chondrocytes | Intracellular MDA ↓ and GSH ↑ | Prevent | Mo et al. [117] | |
| Fer-1 + IL-1β or FAC | IL-1β or FAC | Mouse chondrocytes | Intracellular ROS ↓ and lipid-ROS ↓, GPX4 ↑ and SLC7A11 ↑ | Prevent | Yao et al. [8] | |
| Fer-1 + IL-1β or erastin | IL-1β or erastin | Mouse chondrocytes | Intracellular ROS ↓, MDA ↓, lipid-ROS ↓, GPX4 ↑, and SLC7A11 ↑ | Prevent | Guo et al. [83] | |
| Fer-1 + TBHP | TBHP | Mouse chondrocytes | Intracellular ROS ↓, MDA ↓, lipid-ROS ↓, GSH ↑, and GPX ↑ | Prevent | Miao et al. [9] | |
| NAC + H2O2 | H2O2 | Canine chondrocytes | GSH ↑ | Prevent | Dycus et al. [101] | |
| NAC + FAC | FAC | Mouse chondrocytes | ― | Prevent | Jing et al. [116] | |
| CoQ10 + IL-1β | IL-1β | Rat chondrocytes | ― | Prevent | Li et al. [120] | |
| Vitamin E + calcium ionophore | Calcium ionophore | Rabbit chondrocytes | Intracellular MDA ↓ and 4-HNE ↓ | Prevent | Tiku et al. [109] | |
| IL-1 β or LPS | Control | Rabbit synoviocytes | Intracellular MDA ↑ | Promote | Ingale et al. [128] |
References
- Hunter, D.J.; March, L.; Chew, M. Osteoarthritis in 2020 and beyond: A Lancet Commission. Lancet 2020, 396, 1711–1712. [Google Scholar] [CrossRef]
- Hunter, D.J.; Bierma-Zeinstra, S. Osteoarthritis. Lancet 2019, 393, 1745–1759. [Google Scholar] [CrossRef]
- Jiang, Y. Osteoarthritis year in review 2021: Biology. Osteoarthr. Cartil. 2022, 30, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Zhang, Z.; Sheng, P.; Mobasheri, A. The role of metabolism in chondrocyte dysfunction and the progression of osteoarthritis. Ageing Res. Rev. 2021, 66, 101249. [Google Scholar] [CrossRef] [PubMed]
- Komori, T. Cell Death in Chondrocytes, Osteoblasts, and Osteocytes. Int. J. Mol. Sci. 2016, 17, 2045. [Google Scholar] [CrossRef]
- Abusarah, J.; Bentz, M.; Benabdoune, H.; Rondon, P.E.; Shi, Q.; Fernandes, J.C.; Fahmi, H.; Benderdour, M. An overview of the role of lipid peroxidation-derived 4-hydroxynonenal in osteoarthritis. Inflamm. Res. 2017, 66, 637–651. [Google Scholar] [CrossRef]
- Dixon, S.J.; Lemberg, K.M.; Lamprecht, M.R.; Skouta, R.; Zaitsev, E.M.; Gleason, C.E.; Patel, D.N.; Bauer, A.J.; Cantley, A.M.; Yang, W.S.; et al. Ferroptosis: An Iron-Dependent Form of Nonapoptotic Cell Death. Cell 2012, 149, 1060–1072. [Google Scholar] [CrossRef]
- Yao, X.; Sun, K.; Yu, S.; Luo, J.; Guo, J.; Lin, J.; Wang, G.; Guo, Z.; Ye, Y.; Guo, F. Chondrocyte ferroptosis contribute to the progression of osteoarthritis. J. Orthop. Transl. 2021, 27, 33–43. [Google Scholar] [CrossRef]
- Miao, Y.; Chen, Y.; Xue, F.; Liu, K.; Zhu, B.; Gao, J.; Yin, J.; Zhang, C.; Li, G. Contribution of ferroptosis and GPX4’s dual functions to osteoarthritis progression. EBioMedicine 2022, 76, 103847. [Google Scholar] [CrossRef]
- Yagoda, N.; von Rechenberg, M.; Zaganjor, E.; Bauer, A.J.; Yang, W.S.; Fridman, D.J.; Wolpaw, A.J.; Smukste, I.; Peltier, J.M.; Boniface, J.J.; et al. RAS-RAF-MEK-dependent oxidative cell death involving voltage-dependent anion channels. Nature 2007, 447, 864–868. [Google Scholar] [CrossRef]
- Dixon, S.J.; Stockwell, B.R. The role of iron and reactive oxygen species in cell death. Nat. Chem. Biol. 2014, 10, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.Y.; Babitt, J.L. Liver iron sensing and body iron homeostasis. Blood 2019, 133, 18–29. [Google Scholar] [CrossRef]
- Gunshin, H.; Fujiwara, Y.; Custodio, A.O.; Direnzo, C.; Robine, S.; Andrews, N.C. Slc11a2 is required for intestinal iron absorption and erythropoiesis but dispensable in placenta and liver. J. Clin. Investig. 2005, 115, 1258–1266. [Google Scholar] [CrossRef] [PubMed]
- Donovan, A.; Brownlie, A.; Zhou, Y.; Shepard, J.; Pratt, S.J.; Moynihan, J.; Paw, B.H.; Drejer, A.; Barut, B.; Zapata, A.; et al. Positional cloning of zebrafish ferroportin1 identifies a conserved vertebrate iron exporter. Nature 2000, 403, 776–781. [Google Scholar] [CrossRef] [PubMed]
- Vulpe, C.D.; Kuo, Y.M.; Murphy, T.L.; Cowley, L.; Askwith, C.; Libina, N.; Gitschier, J.; Anderson, G.J. Hephaestin, a ceruloplasmin homologue implicated in intestinal iron transport, is defective in the sla mouse. Nat. Genet. 1999, 21, 195–199. [Google Scholar] [CrossRef]
- Nemeth, E.; Ganz, T. Hepcidin-Ferroportin Interaction Controls Systemic Iron Homeostasis. Int. J. Mol. Sci. 2021, 22, 6493. [Google Scholar] [CrossRef]
- Anderson, G.J.; Frazer, D.M. Current understanding of iron homeostasis. Am. J. Clin. Nutr. 2017, 106 (Suppl. S6), 1559s–1566s. [Google Scholar] [CrossRef]
- Ohgami, R.S.; Campagna, D.R.; Greer, E.L.; Antiochos, B.; McDonald, A.; Chen, J.; Sharp, J.J.; Fujiwara, Y.; Barker, J.E.; Fleming, M.D. Identification of a ferrireductase required for efficient transferrin-dependent iron uptake in erythroid cells. Nat. Genet. 2005, 37, 1264–1269. [Google Scholar] [CrossRef]
- Torti, F.M.; Torti, S.V. Regulation of ferritin genes and protein. Blood 2002, 99, 3505–3516. [Google Scholar] [CrossRef]
- Andrews, N.C. Probing the iron pool. Focus on “Detection of intracellular iron by its regulatory effect”. Am. J. Physiol. Cell Physiol. 2004, 287, C1537–C1538. [Google Scholar] [CrossRef]
- Xie, Y.; Hou, W.; Song, X.; Yu, Y.; Huang, J.; Sun, X.; Kang, R.; Tang, D. Ferroptosis: Process and function. Cell Death Differ 2016, 23, 369–379. [Google Scholar] [CrossRef]
- Feng, H.; Schorpp, K.; Jin, J.; Yozwiak, C.E.; Hoffstrom, B.G.; Decker, A.M.; Rajbhandari, P.; Stokes, M.E.; Bender, H.G.; Csuka, J.M.; et al. Transferrin Receptor Is a Specific Ferroptosis Marker. Cell Rep. 2020, 30, 3411–3423.e7. [Google Scholar] [CrossRef] [PubMed]
- Kong, N.; Chen, X.; Feng, J.; Duan, T.; Liu, S.; Sun, X.; Chen, P.; Pan, T.; Yan, L.; Jin, T.; et al. Baicalin induces ferroptosis in bladder cancer cells by downregulating FTH1. Acta Pharm. Sin. B 2021, 11, 4045–4054. [Google Scholar] [CrossRef] [PubMed]
- Bao, W.D.; Pang, P.; Zhou, X.T.; Hu, F.; Xiong, W.; Chen, K.; Wang, J.; Wang, F.; Xie, D.; Hu, Y.Z.; et al. Loss of ferroportin induces memory impairment by promoting ferroptosis in Alzheimer’s disease. Cell Death Differ. 2021, 28, 1548–1562. [Google Scholar] [CrossRef] [PubMed]
- Ayala, A.; Muñoz, M.F.; Argüelles, S. Lipid peroxidation: Production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid. Med. Cell Longev. 2014, 2014, 360438. [Google Scholar] [CrossRef]
- Yin, H.; Xu, L.; Porter, N.A. Free radical lipid peroxidation: Mechanisms and analysis. Chem. Rev. 2011, 111, 5944–5972. [Google Scholar] [CrossRef]
- Giera, M.; Lingeman, H.; Niessen, W.M. Recent Advancements in the LC- and GC-Based Analysis of Malondialdehyde (MDA): A Brief Overview. Chromatographia 2012, 75, 433–440. [Google Scholar] [CrossRef]
- Yang, H.; Hu, Y.; Weng, M.; Liu, X.; Wan, P.; Hu, Y.; Ma, M.; Zhang, Y.; Xia, H.; Lv, K. Hypoxia inducible lncRNA-CBSLR modulates ferroptosis through m6A-YTHDF2-dependent modulation of CBS in gastric cancer. J. Adv. Res. 2022, 37, 91–106. [Google Scholar] [CrossRef]
- Park, M.W.; Cha, H.W.; Kim, J.; Kim, J.H.; Yang, H.; Yoon, S.; Boonpraman, N.; Yi, S.S.; Yoo, I.D.; Moon, J.S. NOX4 promotes ferroptosis of astrocytes by oxidative stress-induced lipid peroxidation via the impairment of mitochondrial metabolism in Alzheimer’s diseases. Redox. Biol. 2021, 41, 101947. [Google Scholar] [CrossRef]
- Liu, P.; Feng, Y.; Li, H.; Chen, X.; Wang, G.; Xu, S.; Li, Y.; Zhao, L. Ferrostatin-1 alleviates lipopolysaccharide-induced acute lung injury via inhibiting ferroptosis. Cell Mol. Biol. Lett. 2020, 25, 10. [Google Scholar] [CrossRef]
- Zheng, J.; Conrad, M. The Metabolic Underpinnings of Ferroptosis. Cell Metab. 2020, 32, 920–937. [Google Scholar] [CrossRef] [PubMed]
- Bridges, R.J.; Natale, N.R.; Patel, S.A. System xc− cystine/glutamate antiporter: An update on molecular pharmacology and roles within the CNS. Br. J. Pharm. 2012, 165, 20–34. [Google Scholar] [CrossRef] [PubMed]
- Doll, S.; Freitas, F.P.; Shah, R.; Aldrovandi, M.; da Silva, M.C.; Ingold, I.; Goya Grocin, A.; Xavier da Silva, T.N.; Panzilius, E.; Scheel, C.H.; et al. FSP1 is a glutathione-independent ferroptosis suppressor. Nature 2019, 575, 693–698. [Google Scholar] [CrossRef] [PubMed]
- Badgley, M.A.; Kremer, D.M.; Maurer, H.C.; DelGiorno, K.E.; Lee, H.J.; Purohit, V.; Sagalovskiy, I.R.; Ma, A.; Kapilian, J.; Firl, C.E.M.; et al. Cysteine depletion induces pancreatic tumor ferroptosis in mice. Science 2020, 368, 85–89. [Google Scholar] [CrossRef]
- Yang, W.S.; SriRamaratnam, R.; Welsch, M.E.; Shimada, K.; Skouta, R.; Viswanathan, V.S.; Cheah, J.H.; Clemons, P.A.; Shamji, A.F.; Clish, C.B.; et al. Regulation of ferroptotic cancer cell death by GPX4. Cell 2014, 156, 317–331. [Google Scholar] [CrossRef]
- Bersuker, K.; Hendricks, J.M.; Li, Z.; Magtanong, L.; Ford, B.; Tang, P.H.; Roberts, M.A.; Tong, B.; Maimone, T.J.; Zoncu, R.; et al. The CoQ oxidoreductase FSP1 acts parallel to GPX4 to inhibit ferroptosis. Nature 2019, 575, 688–692. [Google Scholar] [CrossRef]
- Sun, K.; Guo, Z.; Hou, L.; Xu, J.; Du, T.; Xu, T.; Guo, F. Iron Homeostasis in Arthropathies: From Pathogenesis to Therapeutic Potential. Ageing Res. Rev. 2021, 72, 101481. [Google Scholar] [CrossRef]
- Yazar, M.; Sarban, S.; Kocyigit, A.; Isikan, U.E. Synovial fluid and plasma selenium, copper, zinc, and iron concentrations in patients with rheumatoid arthritis and osteoarthritis. Biol. Trace Elem. Res. 2005, 106, 123–132. [Google Scholar] [CrossRef]
- Ogilvie-Harris, D.J.; Fornaiser, V.L. Synovial iron deposition in osteoarthritis and rheumatoid arthritis. J. Rheumatol. 1980, 7, 30–36. [Google Scholar]
- Zhou, J.; Liu, C.; Sun, Y.; Francis, M.; Ryu, M.S.; Grider, A.; Ye, K. Genetically predicted circulating levels of copper and zinc are associated with osteoarthritis but not with rheumatoid arthritis. Osteoarthr. Cartil. 2021, 29, 1029–1035. [Google Scholar] [CrossRef]
- Qu, Z.; Yang, F.; Hong, J.; Wang, W.; Li, S.; Jiang, G.; Yan, S. Causal relationship of serum nutritional factors with osteoarthritis: A Mendelian randomization study. Rheumatology 2021, 60, 2383–2390. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yau, M.S.; Yerges-Armstrong, L.M.; Duggan, D.J.; Renner, J.B.; Hochberg, M.C.; Mitchell, B.D.; Jackson, R.D.; Jordan, J.M. Genetic Determinants of Radiographic Knee Osteoarthritis in African Americans. J. Rheumatol. 2017, 44, 1652–1658. [Google Scholar] [CrossRef] [PubMed]
- Nugzar, O.; Zandman-Goddard, G.; Oz, H.; Lakstein, D.; Feldbrin, Z.; Shargorodsky, M. The role of ferritin and adiponectin as predictors of cartilage damage assessed by arthroscopy in patients with symptomatic knee osteoarthritis. Best Pract. Res Clin. Rheumatol. 2018, 32, 662–668. [Google Scholar] [CrossRef] [PubMed]
- Kennish, L.; Attur, M.; Huang, X.; Lai, Y.; Liu, C.; Krasnokutsky, S.; Samuels, J.; Abramson, S.B. Iron Overload and Hemochromatosis (HFE) Mutation Correlate with Clinical Outcomes in an Osteoarthritis Cohort. Osteoarthr. Cartil. 2011, 19, S143–S144. [Google Scholar] [CrossRef]
- Wu, L.; Si, H.; Zeng, Y.; Wu, Y.; Li, M.; Liu, Y.; Shen, B. Association between Iron Intake and Progression of Knee Osteoarthritis. Nutrients 2022, 14, 1674. [Google Scholar] [CrossRef]
- Grigolo, B.; Roseti, L.; Fiorini, M.; Facchini, A. Enhanced lipid peroxidation in synoviocytes from patients with osteoarthritis. J. Rheumatol. 2003, 30, 345–347. [Google Scholar]
- Morquette, B.; Shi, Q.; Lavigne, P.; Ranger, P.; Fernandes, J.C.; Benderdour, M. Production of lipid peroxidation products in osteoarthritic tissues: New evidence linking 4-hydroxynonenal to cartilage degradation. Arthritis. Rheum. 2006, 54, 271–281. [Google Scholar] [CrossRef]
- 48. Shah, R.; Raska, K., Jr.; Tiku, M.L. The presence of molecular markers of in vivo lipid peroxidation in osteoarthritic cartilage: A pathogenic role in osteoarthritis. Arthritis. Rheum. 2005, 52, 2799–2807. [Google Scholar] [CrossRef]
- Gavriilidis, C.; Miwa, S.; von Zglinicki, T.; Taylor, R.W.; Young, D.A. Mitochondrial dysfunction in osteoarthritis is associated with down-regulation of superoxide dismutase 2. Arthritis. Rheum. 2013, 65, 378–387. [Google Scholar] [CrossRef]
- Regan, E.A.; Bowler, R.P.; Crapo, J.D. Joint fluid antioxidants are decreased in osteoarthritic joints compared to joints with macroscopically intact cartilage and subacute injury. Osteoarthr. Cartil. 2008, 16, 515–521. [Google Scholar] [CrossRef]
- Maneesh, M.; Jayalekshmi, H.; Suma, T.; Chatterjee, S.; Chakrabarti, A.; Singh, T.A. Evidence for oxidative stress in osteoarthritis. Indian J. Clin. Biochem. 2005, 20, 129–130. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hu, Q.; Zhang, Y.; Lou, H.; Ou, Z.; Liu, J.; Duan, W.; Wang, H.; Ge, Y.; Min, J.; Wang, F.; et al. GPX4 and vitamin E cooperatively protect hematopoietic stem and progenitor cells from lipid peroxidation and ferroptosis. Cell Death Dis. 2021, 12, 706. [Google Scholar] [CrossRef] [PubMed]
- Sutipornpalangkul, W.; Morales, N.P.; Charoencholvanich, K.; Harnroongroj, T. Lipid peroxidation, glutathione, vitamin E, and antioxidant enzymes in synovial fluid from patients with osteoarthritis. Int. J. Rheum. Dis. 2009, 12, 324–328. [Google Scholar] [CrossRef] [PubMed]
- Angthong, C.; Morales, N.P.; Sutipornpalangkul, W.; Khadsongkram, A.; Pinsornsak, P.; Pongcharoen, B. Can levels of antioxidants in synovial fluid predict the severity of primary knee osteoarthritis: A preliminary study. Springerplus 2013, 2, 652. [Google Scholar] [CrossRef][Green Version]
- Bhattacharya, I.; Saxena, R.; Gupta, V. Efficacy of vitamin E in knee osteoarthritis management of North Indian geriatric population. Adv. Musculoskelet. Dis. 2012, 4, 11–19. [Google Scholar] [CrossRef]
- Burton, L.H.; Radakovich, L.B.; Marolf, A.J.; Santangelo, K.S. Systemic iron overload exacerbates osteoarthritis in the strain 13 guinea pig. Osteoarthr. Cartil. 2020, 28, 1265–1275. [Google Scholar] [CrossRef]
- Jing, X.; Lin, J.; Du, T.; Jiang, Z.; Li, T.; Wang, G.; Liu, X.; Cui, X.; Sun, K. Iron Overload Is Associated With Accelerated Progression of Osteoarthritis: The Role of DMT1 Mediated Iron Homeostasis. Front. Cell Dev. Biol. 2020, 8, 594509. [Google Scholar] [CrossRef]
- Radakovich, L.B.; Marolf, A.J.; Santangelo, K.S. ‘Iron Accumulation’ Gene Expression Profile in Obese Hartley Guinea Pig Knee Joints Is Associated with More Severe Osteoarthritis. Osteoarthr. Cartil. 2017, 25, S169. [Google Scholar] [CrossRef]
- Luo, Q.; Qin, X.; Qiu, Y.; Hou, L.; Yang, N. The change of synovial fluid proteome in rabbit surgery-induced model of knee osteoarthritis. Am. J. Transl. Res. 2018, 10, 2087–2101. [Google Scholar]
- Burton, L.H.; Afzali, M.F.; Radakovich, L.B.; Campbell, M.A.; Culver, L.A.; Olver, C.S.; Santangelo, K.S. Systemic administration of a pharmacologic iron chelator reduces cartilage lesion development in the Dunkin-Hartley model of primary osteoarthritis. Free Radic. Biol. Med. 2022, 179, 47–58. [Google Scholar] [CrossRef]
- Karakurum, G.; Karakok, M.; Tarakcioglu, M.; Kocer, N.E.; Kocabas, R.; Bagci, C. Comparative effect of intra-articular administration of hyaluronan and/or cortisone with evaluation of malondialdehyde on degenerative osteoarthritis of the rabbit’s knee. Tohoku. J. Exp. Med. 2003, 199, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Sun, S.; Wang, J.; Li, W.; Wang, X.; Yuan, L.; Li, S. S-Allylmercaptocysteine Targets Nrf2 in Osteoarthritis Treatment Through NOX4/NF-κB Pathway. Drug. Des. Devel. Ther. 2020, 14, 4533–4546. [Google Scholar] [CrossRef] [PubMed]
- Goranov, N.V. Serum markers of lipid peroxidation, antioxidant enzymatic defense, and collagen degradation in an experimental (Pond-Nuki) canine model of osteoarthritis. Vet. Clin. Pathol. 2007, 36, 192–195. [Google Scholar] [CrossRef]
- Chang, H.W.; Sudirman, S.; Yen, Y.W.; Mao, C.F.; Ong, A.D.; Kong, Z.L. Blue Mussel (Mytilus edulis) Water Extract Ameliorates Inflammatory Responses and Oxidative Stress on Osteoarthritis in Obese Rats. J. Pain. Res. 2020, 13, 1109–1119. [Google Scholar] [CrossRef] [PubMed]
- Gladkova, E.V. Role of Imbalance of Lipid Peroxidation and Articular Cartilage Remodeling in the Pathogenesis of Early Primary and Post-Traumatic Gonarthrosis in Rats. Bull. Exp. Biol. Med. 2022, 172, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Zubavlenko, R.; Belova, S.V.; Gladkova, E.V.; Matveeva, O.V.; Ulyanov, V.Y. Morphological Changes in Articular Cartilage and Free-Radical Lipid Peroxidation in Rats with Posttraumatic Osteoarthrosis. Bull. Exp. Biol. Med. 2021, 172, 214–217. [Google Scholar] [CrossRef] [PubMed]
- Bai, H.; Yuan, R.; Zhang, Z.; Liu, L.; Wang, X.; Song, X.; Ma, T.; Tang, J.; Liu, C.; Gao, L. Intra-articular Injection of Baicalein Inhibits Cartilage Catabolism and NLRP3 Inflammasome Signaling in a Posttraumatic OA Model. Oxid. Med. Cell Longev. 2021, 2021, 6116890. [Google Scholar] [CrossRef]
- Bai, B.; Li, Y. Danshen prevents articular cartilage degeneration via antioxidation in rabbits with osteoarthritis. Osteoarthr. Cartil. 2016, 24, 514–520. [Google Scholar] [CrossRef]
- Aulin, C.; Lundbäck, P.; Palmblad, K.; Klareskog, L.; Erlandsson Harris, H. An in vivo cross-linkable hyaluronan gel with inherent anti-inflammatory properties reduces OA cartilage destruction in female mice subjected to cruciate ligament transection. Osteoarthr. Cartil. 2017, 25, 157–165. [Google Scholar] [CrossRef]
- Shi, Q.; Abusarah, J.; Zaouter, C.; Moldovan, F.; Fernandes, J.C.; Fahmi, H.; Benderdour, M. New evidence implicating 4-hydroxynonenal in the pathogenesis of osteoarthritis in vivo. Arthritis. Rheumatol. 2014, 66, 2461–2471. [Google Scholar] [CrossRef]
- Zhou, X.; Zheng, Y.; Sun, W.; Zhang, Z.; Liu, J.; Yang, W.; Yuan, W.; Yi, Y.; Wang, J.; Liu, J. D-mannose alleviates osteoarthritis progression by inhibiting chondrocyte ferroptosis in a HIF-2α-dependent manner. Cell Prolif. 2021, 54, e13134. [Google Scholar] [CrossRef] [PubMed]
- Qiu, L.; Luo, Y.; Chen, X. Quercetin attenuates mitochondrial dysfunction and biogenesis via upregulated AMPK/SIRT1 signaling pathway in OA rats. Biomed. Pharm. 2018, 103, 1585–1591. [Google Scholar] [CrossRef] [PubMed]
- Pathak, N.N.; Balaganur, V.; Lingaraju, M.C.; Kant, V.; Kumar, D.; Kumar, D.; Sharma, A.K.; Tandan, S.K. Effect of atorvastatin, a HMG-CoA reductase inhibitor in monosodium iodoacetate-induced osteoarthritic pain: Implication for osteoarthritis therapy. Pharm. Rep. 2015, 67, 513–519. [Google Scholar] [CrossRef]
- Abdel Jaleel, G.A.; Saleh, D.O.; Al-Awdan, S.W.; Hassan, A.; Asaad, G.F. Impact of type III collagen on monosodium iodoacetate-induced osteoarthritis in rats. Heliyon 2020, 6, e04083. [Google Scholar] [CrossRef]
- Huang, T.C.; Chang, W.T.; Hu, Y.C.; Hsieh, B.S.; Cheng, H.L.; Yen, J.H.; Chiu, P.R.; Chang, K.L. Zinc Protects Articular Chondrocytes through Changes in Nrf2-Mediated Antioxidants, Cytokines and Matrix Metalloproteinases. Nutrients 2018, 10, 471. [Google Scholar] [CrossRef] [PubMed]
- Fusco, R.; Siracusa, R.; Peritore, A.F.; Gugliandolo, E.; Genovese, T.; D’Amico, R.; Cordaro, M.; Crupi, R.; Mandalari, G.; Impellizzeri, D.; et al. The Role of Cashew (Anacardium occidentale L.) Nuts on an Experimental Model of Painful Degenerative Joint Disease. Antioxidants 2020, 9, 511. [Google Scholar] [CrossRef]
- Yabas, M.; Orhan, C.; Er, B.; Tuzcu, M.; Durmus, A.S.; Ozercan, I.H.; Sahin, N.; Bhanuse, P.; Morde, A.A.; Padigaru, M.; et al. A Next Generation Formulation of Curcumin Ameliorates Experimentally Induced Osteoarthritis in Rats via Regulation of Inflammatory Mediators. Front. Immunol. 2021, 12, 609629. [Google Scholar] [CrossRef]
- Ragab, G.H.; Halfaya, F.M.; Ahmed, O.M.; Abou El-Kheir, W.; Mahdi, E.A.; Ali, T.M.; Almehmadi, M.M.; Hagag, U. Platelet-Rich Plasma Ameliorates Monosodium Iodoacetate-Induced Ankle Osteoarthritis in the Rat Model via Suppression of Inflammation and Oxidative Stress. Evid. Based Complement Altern. Med. 2021, 2021, 6692432. [Google Scholar] [CrossRef]
- Ajeeshkumar, K.K.; Vishnu, K.V.; Navaneethan, R.; Raj, K.; Remyakumari, K.R.; Swaminathan, T.R.; Suseela, M.; Asha, K.K.; Sreekanth, G.P. Proteoglycans isolated from the bramble shark cartilage show potential anti-osteoarthritic properties. Inflammopharmacology 2019, 27, 175–187. [Google Scholar] [CrossRef]
- Ma, D.; He, J.; He, D. Chamazulene reverses osteoarthritic inflammation through regulation of matrix metalloproteinases (MMPs) and NF-kβ pathway in in-vitro and in-vivo models. Biosci. Biotechnol. Biochem. 2020, 84, 402–410. [Google Scholar] [CrossRef]
- Kurz, B.; Jost, B.; Schünke, M. Dietary vitamins and selenium diminish the development of mechanically induced osteoarthritis and increase the expression of antioxidative enzymes in the knee joint of STR/1N mice. Osteoarthritis Cartilage 2002, 10, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Lv, M.; Cai, Y.; Hou, W.; Peng, K.; Xu, K.; Lu, C.; Yu, W.; Zhang, W.; Liu, L. The RNA-binding protein SND1 promotes the degradation of GPX4 by destabilizing the HSPA5 mRNA and suppressing HSPA5 expression, promoting ferroptosis in osteoarthritis chondrocytes. Inflamm. Res. 2022, 71, 461–472. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Lin, J.; Sun, K.; Guo, J.; Yao, X.; Wang, G.; Hou, L.; Xu, J.; Guo, J.; Guo, F. Deferoxamine Alleviates Osteoarthritis by Inhibiting Chondrocyte Ferroptosis and Activating the Nrf2 Pathway. Front. Pharm. 2022, 13, 791376. [Google Scholar] [CrossRef] [PubMed]
- Kirkpatrick, C.J.; Mohr, W.; Haferkamp, O. Alterations in chondrocyte morphology, proliferation and binding of 35SO4 due to Fe(III), Fe(II), ferritin and haemoglobin in vitro. Virchows. Arch. B Cell Pathol. Incl. Mol. Pathol. 1982, 38, 297–306. [Google Scholar] [CrossRef]
- Karim, A.; Bajbouj, K.; Shafarin, J.; Qaisar, R.; Hall, A.C.; Hamad, M. Iron Overload Induces Oxidative Stress, Cell Cycle Arrest and Apoptosis in Chondrocytes. Front. Cell Dev. Biol. 2022, 10, 821014. [Google Scholar] [CrossRef]
- Jing, X.; Wang, Q.; Du, T.; Zhang, W.; Liu, X.; Liu, Q.; Li, T.; Wang, G.; Chen, F.; Cui, X. Calcium chelator BAPTA-AM protects against iron overload-induced chondrocyte mitochondrial dysfunction and cartilage degeneration. Int. J. Mol. Med. 2021, 48, 196. [Google Scholar] [CrossRef]
- Ohno, T.; Hashimoto, N.; Mitsui, K.; Nishimura, H.; Hagiwara, H. Iron overload inhibits calcification and differentiation of ATDC5 cells. J. Biochem. 2012, 151, 109–114. [Google Scholar] [CrossRef]
- Tchetina, E.V.; Markova, G.A.; Poole, A.R.; Zukor, D.J.; Antoniou, J.; Makarov, S.A.; Kuzin, A.N. Deferoxamine Suppresses Collagen Cleavage and Protease, Cytokine, and COL10A1 Expression and Upregulates AMPK and Krebs Cycle Genes in Human Osteoarthritic Cartilage. Int. J. Rheumatol. 2016, 2016, 6432867. [Google Scholar] [CrossRef]
- Rasheed, N.; Alghasham, A.; Rasheed, Z. Lactoferrin from Camelus dromedarius Inhibits Nuclear Transcription Factor-kappa B Activation, Cyclooxygenase-2 Expression and Prostaglandin E2 Production in Stimulated Human Chondrocytes. Pharmacogn. Res. 2016, 8, 135–141. [Google Scholar] [CrossRef]
- Vaillancourt, F.; Morquette, B.; Shi, Q.; Fahmi, H.; Lavigne, P.; Di Battista, J.A.; Fernandes, J.C.; Benderdour, M. Differential regulation of cyclooxygenase-2 and inducible nitric oxide synthase by 4-hydroxynonenal in human osteoarthritic chondrocytes through ATF-2/CREB-1 transactivation and concomitant inhibition of NF-kappaB signaling cascade. J. Cell Biochem. 2007, 100, 1217–1231. [Google Scholar] [CrossRef]
- Bentz, M.; Zaouter, C.; Shi, Q.; Fahmi, H.; Moldovan, F.; Fernandes, J.C.; Benderdour, M. Inhibition of inducible nitric oxide synthase prevents lipid peroxidation in osteoarthritic chondrocytes. J. Cell Biochem. 2012, 113, 2256–2267. [Google Scholar] [CrossRef] [PubMed]
- Benabdoune, H.; Rondon, E.P.; Shi, Q.; Fernandes, J.; Ranger, P.; Fahmi, H.; Benderdour, M. The role of resolvin D1 in the regulation of inflammatory and catabolic mediators in osteoarthritis. Inflamm. Res. 2016, 65, 635–645. [Google Scholar] [CrossRef] [PubMed]
- Elmazoglu, Z.; Aydın Bek, Z.; Sarıbaş, S.G.; Özoğul, C.; Goker, B.; Bitik, B.; Aktekin, C.N.; Karasu, Ç. S-allylcysteine inhibits chondrocyte inflammation to reduce human osteoarthritis via targeting RAGE, TLR4, JNK, and Nrf2 signaling: Comparison with colchicine. Biochem. Cell Biol. 2021, 99, 645–654. [Google Scholar] [CrossRef]
- Yao, J.; Long, H.; Zhao, J.; Zhong, G.; Li, J. Nifedipine inhibits oxidative stress and ameliorates osteoarthritis by activating the nuclear factor erythroid-2-related factor 2 pathway. Life Sci. 2020, 253, 117292. [Google Scholar] [CrossRef] [PubMed]
- Hosseinzadeh, A.; Bahrampour Juybari, K.; Fatemi, M.J.; Kamarul, T.; Bagheri, A.; Tekiyehmaroof, N.; Sharifi, A.M. Protective Effect of Ginger (Zingiber officinale Roscoe) Extract against Oxidative Stress and Mitochondrial Apoptosis Induced by Interleukin-1β in Cultured Chondrocytes. Cells Tissues Organs 2017, 204, 241–250. [Google Scholar] [CrossRef]
- Hosseinzadeh, A.; Jafari, D.; Kamarul, T.; Bagheri, A.; Sharifi, A.M. Evaluating the Protective Effects and Mechanisms of Diallyl Disulfide on Interlukin-1β-Induced Oxidative Stress and Mitochondrial Apoptotic Signaling Pathways in Cultured Chondrocytes. J. Cell Biochem. 2017, 118, 1879–1888. [Google Scholar] [CrossRef]
- Zuo, S.; Zou, W.; Wu, R.M.; Yang, J.; Fan, J.N.; Zhao, X.K.; Li, H.Y. Icariin Alleviates IL-1β-Induced Matrix Degradation By Activating The Nrf2/ARE Pathway In Human Chondrocytes. Drug. Des. Devel. Ther. 2019, 13, 3949–3961. [Google Scholar] [CrossRef] [PubMed]
- Yin, M.; Xu, Y. The protective effects of etomidate against interleukin-1β (IL-1β)-induced oxidative stress, extracellular matrix alteration and cellular senescence in chondrocytes. Bioengineered 2022, 13, 985–994. [Google Scholar] [CrossRef]
- Zhu, H.; Zhu, S.; Shang, X.; Meng, X.; Jing, S.; Yu, L.; Deng, Y. Exhausting circ_0136474 and Restoring miR-766-3p Attenuate Chondrocyte Oxidative Injury in IL-1β-Induced Osteoarthritis Progression Through Regulating DNMT3A. Front. Genet. 2021, 12, 648709. [Google Scholar] [CrossRef]
- Wang, C.; Qu, L. The anti-fibrotic agent nintedanib protects chondrocytes against tumor necrosis factor-α (TNF-α)-induced extracellular matrix degradation. Bioengineered 2022, 13, 5318–5329. [Google Scholar] [CrossRef]
- Dycus, D.L.; Au, A.Y.; Grzanna, M.W.; Wardlaw, J.L.; Frondoza, C.G. Modulation of inflammation and oxidative stress in canine chondrocytes. Am. J. Vet. Res. 2013, 74, 983–989. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Guo, Y.X.; Liu, L.; Yan, D.Z.; Guo, J.P. Plumbagin prevents osteoarthritis in human chondrocytes through Nrf-2 activation. Mol. Med. Rep. 2017, 15, 2333–2338. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Wang, X.; Peng, Q.; Jin, Y.; Shi, G.; Fan, Z.; Zhou, Z. Four-Octyl Itaconate Protects Chondrocytes against H(2)O(2)-Induced Oxidative Injury and Attenuates Osteoarthritis Progression by Activating Nrf2 Signaling. Oxid. Med. Cell Longev. 2022, 2022, 2206167. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Makosa, D.; Miller, B.; Griffin, T.M. Glutathione as a mediator of cartilage oxidative stress resistance and resilience during aging and osteoarthritis. Connect. Tissue Res. 2020, 61, 34–47. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Y.Q.; Jiang, P.F.; Gao, Y.Z. Lutein prevents osteoarthritis through Nrf2 activation and downregulation of inflammation. Arch. Med. Sci. 2018, 14, 617–624. [Google Scholar] [CrossRef]
- Hosseinzadeh, A.; Bahrampour Juybari, K.; Kamarul, T.; Sharifi, A.M. Protective effects of atorvastatin on high glucose-induced oxidative stress and mitochondrial apoptotic signaling pathways in cultured chondrocytes. J. Physiol. Biochem. 2019, 75, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, A.; Yabu, A.; Nakamura, H. Advanced glycation end products in musculoskeletal system and disorders. Methods 2020, 203, 179–186. [Google Scholar] [CrossRef]
- Hu, N.; Gong, X.; Yin, S.; Li, Q.; Chen, H.; Li, Y.; Li, F.; Qing, L.; Yang, J.; Zhu, S.; et al. Saxagliptin suppresses degradation of type II collagen and aggrecan in primary human chondrocytes: A therapeutic implication in osteoarthritis. Artif. Cells Nanomed. Biotechnol. 2019, 4, 3239–3245. [Google Scholar] [CrossRef] [PubMed]
- Tiku, M.L.; Shah, R.; Allison, G.T. Evidence linking chondrocyte lipid peroxidation to cartilage matrix protein degradation. Possible role in cartilage aging and the pathogenesis of osteoarthritis. J. Biol. Chem. 2000, 275, 20069–20076. [Google Scholar] [CrossRef] [PubMed]
- Tiku, M.L.; Allison, G.T.; Naik, K.; Karry, S.K. Malondialdehyde oxidation of cartilage collagen by chondrocytes. Osteoarthr. Cartil. 2003, 11, 159–166. [Google Scholar] [CrossRef]
- Tiku, M.L.; Narla, H.; Jain, M.; Yalamanchili, P. Glucosamine prevents in vitro collagen degradation in chondrocytes by inhibiting advanced lipoxidation reactions and protein oxidation. Arthritis. Res. Ther. 2007, 9, R76. [Google Scholar] [CrossRef] [PubMed]
- Mendis, E.; Kim, M.M.; Rajapakse, N.; Kim, S.K. Sulfated glucosamine inhibits oxidation of biomolecules in cells via a mechanism involving intracellular free radical scavenging. Eur. J. Pharm. 2008, 579, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, S.; Akagi, M.; Yoshida, K.; Hayakawa, S.; Sawamura, T.; Munakata, H.; Hamanishi, C. Oxidized low-density lipoprotein (ox-LDL) binding to lectin-like ox-LDL receptor-1 (LOX-1) in cultured bovine articular chondrocytes increases production of intracellular reactive oxygen species (ROS) resulting in the activation of NF-kappaB. Osteoarthr. Cartil. 2004, 12, 568–576. [Google Scholar] [CrossRef]
- Cheng, Y.H.; Chavez, E.; Tsai, K.L.; Yang, K.C.; Kuo, W.T.; Yang, Y.P.; Chiou, S.H.; Lin, F.H. Effects of thermosensitive chitosan-gelatin based hydrogel containing glutathione on Cisd2-deficient chondrocytes under oxidative stress. Carbohydr. Polym. 2017, 173, 17–27. [Google Scholar] [CrossRef]
- Dombrecht, E.J.; De Tollenaere, C.B.; Aerts, K.; Cos, P.; Schuerwegh, A.J.; Bridts, C.H.; Van Offel, J.F.; Ebo, D.G.; Stevens, W.J.; De Clerck, L.S. Antioxidant effect of bisphosphonates and simvastatin on chondrocyte lipid peroxidation. Biochem. Biophys. Res. Commun. 2006, 348, 459–464. [Google Scholar] [CrossRef]
- Jing, X.; Du, T.; Li, T.; Yang, X.; Wang, G.; Liu, X.; Jiang, Z.; Cui, X. The detrimental effect of iron on OA chondrocytes: Importance of pro-inflammatory cytokines induced iron influx and oxidative stress. J. Cell Mol. Med. 2021, 25, 5671–5680. [Google Scholar] [CrossRef] [PubMed]
- Mo, Z.; Xu, P.; Li, H. Stigmasterol alleviates interleukin-1beta-induced chondrocyte injury by down-regulatingsterol regulatory element binding transcription factor 2 to regulateferroptosis. Bioengineered 2021, 12, 9332–9340. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.S.; Yen, C.H.; Huang, Y.Y.; Chiu, C.J.; Lin, P.T. Associations between Coenzyme Q10 Status, Oxidative Stress, and Muscle Strength and Endurance in Patients with Osteoarthritis. Antioxidants 2020, 9, 1275. [Google Scholar] [CrossRef]
- Lee, J.; Hong, Y.S.; Jeong, J.H.; Yang, E.J.; Jhun, J.Y.; Park, M.K.; Jung, Y.O.; Min, J.K.; Kim, H.Y.; Park, S.H.; et al. Coenzyme Q10 ameliorates pain and cartilage degradation in a rat model of osteoarthritis by regulating nitric oxide and inflammatory cytokines. PLoS ONE 2013, 8, e69362. [Google Scholar] [CrossRef]
- Li, X.; Guo, Y.; Huang, S.; He, M.; Liu, Q.; Chen, W.; Liu, M.; Xu, D.; He, P. Coenzyme Q10 Prevents the Interleukin-1 Beta Induced Inflammatory Response via Inhibition of MAPK Signaling Pathways in Rat Articular Chondrocytes. Drug. Dev. Res. 2017, 78, 403–410. [Google Scholar] [CrossRef]
- Ostalowska, A.; Birkner, E.; Wiecha, M.; Kasperczyk, S.; Kasperczyk, A.; Kapolka, D.; Zon-Giebel, A. Lipid peroxidation and antioxidant enzymes in synovial fluid of patients with primary and secondary osteoarthritis of the knee joint. Osteoarthr. Cartil. 2006, 14, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Mathy-Hartert, M.; Hogge, L.; Sanchez, C.; Deby-Dupont, G.; Crielaard, J.M.; Henrotin, Y. Interleukin-1beta and interleukin-6 disturb the antioxidant enzyme system in bovine chondrocytes: A possible explanation for oxidative stress generation. Osteoarthr. Cartil. 2008, 16, 756–763. [Google Scholar] [CrossRef]
- Tsai, W.Y.; Tsai, R.Y.; Liu, C.C.; Wu, J.L.; Wong, C.S. Sulfasalazine attenuates ACL transection and medial menisectomy-induced cartilage destruction by inhibition of cystine/glutamate antiporter. J. Orthop. Res. 2016, 34, 650–657. [Google Scholar] [CrossRef]
- Ansari, M.Y.; Ahmad, N.; Haqqi, T.M. Oxidative stress and inflammation in osteoarthritis pathogenesis: Role of polyphenols. Biomed. Pharm. 2020, 129, 110452. [Google Scholar] [CrossRef] [PubMed]
- Brodziak-Dopierała, B.; Roczniak, W.; Jakóbik-Kolon, A.; Kluczka, J.; Koczy, B.; Kwapuliński, J.; Babuśka-Roczniak, M. Correlations between iron content in knee joint tissues and chosen indices of peripheral blood morphology. Adv. Clin. Exp. Med. 2017, 26, 1077–1083. [Google Scholar] [CrossRef] [PubMed]
- Carlo, M.D., Jr.; Loeser, R.F. Increased oxidative stress with aging reduces chondrocyte survival: Correlation with intracellular glutathione levels. Arthritis. Rheum. 2003, 48, 3419–3430. [Google Scholar] [CrossRef]
- Ishibashi, A.; Maeda, N.; Kojima, C.; Goto, K. Iron Metabolism following Twice a Day Endurance Exercise in Female Long-Distance Runners. Nutrients 2022, 14, 1907. [Google Scholar] [CrossRef]
- Ingale, D.R.; Kulkarni, P.G.; Koppikar, S.J.; Harsulkar, A.M.; Moghe, A.S.; Jagtap, S.D. Reduced synovial inflammation and inhibition of matrix metalloproteinases explicates anti-osteoarthritis activity of polyherbal formulations. Indian J. Pharm. 2018, 50, 22–29. [Google Scholar] [CrossRef]
- Yang, K.C.; Wu, C.C.; Chen, W.Y.; Sumi, S.; Huang, T.L. l-Glutathione enhances antioxidant capacity of hyaluronic acid and modulates expression of pro-inflammatory cytokines in human fibroblast-like synoviocytes. J. Biomed. Mater. Res A 2016, 104, 2071–2079. [Google Scholar] [CrossRef]
- Kurz, B.; Schunke, M. Articular chondrocytes and synoviocytes in culture: Influence of antioxidants on lipid peroxidation and proliferation. Ann. Anat. 1997, 179, 439–446. [Google Scholar] [CrossRef]
- Li, G.; Cheng, T.; Yu, X. The Impact of Trace Elements on Osteoarthritis. Front Med. 2021, 8, 771297. [Google Scholar] [CrossRef] [PubMed]
- Konieczynski, P.; Szreder, G.; Tamowska, E.; Wesolowski, M. Essential elements in synovial fluid samples obtained from patients living in Northern Poland. J. Trace Elem. Med. Biol. 2018, 48, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Huang, Z.; Duan, J.; Nice, E.C.; Lin, J.; Huang, C. Elesclomol induces copper-dependent ferroptosis in colorectal cancer cells via degradation of ATP7A. Mol. Oncol. 2021, 15, 3527–3544. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.H.; Wu, J.; Xu, Y.; Ding, C.C.; Mestre, A.A.; Lin, C.C.; Yang, W.H.; Chi, J.T. Zinc transporter ZIP7 is a novel determinant of ferroptosis. Cell Death Dis. 2021, 12, 198. [Google Scholar] [CrossRef] [PubMed]
- Saeidnia, S.; Manayi, A.; Abdollahi, M. From in vitro Experiments to in vivo and Clinical Studies; Pros and Cons. Curr Drug. Discov. Technol. 2015, 12, 218–224. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, S.; Xu, J.; Si, H.; Wu, Y.; Zhou, S.; Shen, B. The Role Played by Ferroptosis in Osteoarthritis: Evidence Based on Iron Dyshomeostasis and Lipid Peroxidation. Antioxidants 2022, 11, 1668. https://doi.org/10.3390/antiox11091668
Zhang S, Xu J, Si H, Wu Y, Zhou S, Shen B. The Role Played by Ferroptosis in Osteoarthritis: Evidence Based on Iron Dyshomeostasis and Lipid Peroxidation. Antioxidants. 2022; 11(9):1668. https://doi.org/10.3390/antiox11091668
Chicago/Turabian StyleZhang, Shaoyun, Jiawen Xu, Haibo Si, Yuangang Wu, Shengliang Zhou, and Bin Shen. 2022. "The Role Played by Ferroptosis in Osteoarthritis: Evidence Based on Iron Dyshomeostasis and Lipid Peroxidation" Antioxidants 11, no. 9: 1668. https://doi.org/10.3390/antiox11091668
APA StyleZhang, S., Xu, J., Si, H., Wu, Y., Zhou, S., & Shen, B. (2022). The Role Played by Ferroptosis in Osteoarthritis: Evidence Based on Iron Dyshomeostasis and Lipid Peroxidation. Antioxidants, 11(9), 1668. https://doi.org/10.3390/antiox11091668







