The Multiple Effects of Vitamin D against Chronic Diseases: From Reduction of Lipid Peroxidation to Updated Evidence from Clinical Studies
Abstract
1. Introduction
2. Materials and Methods
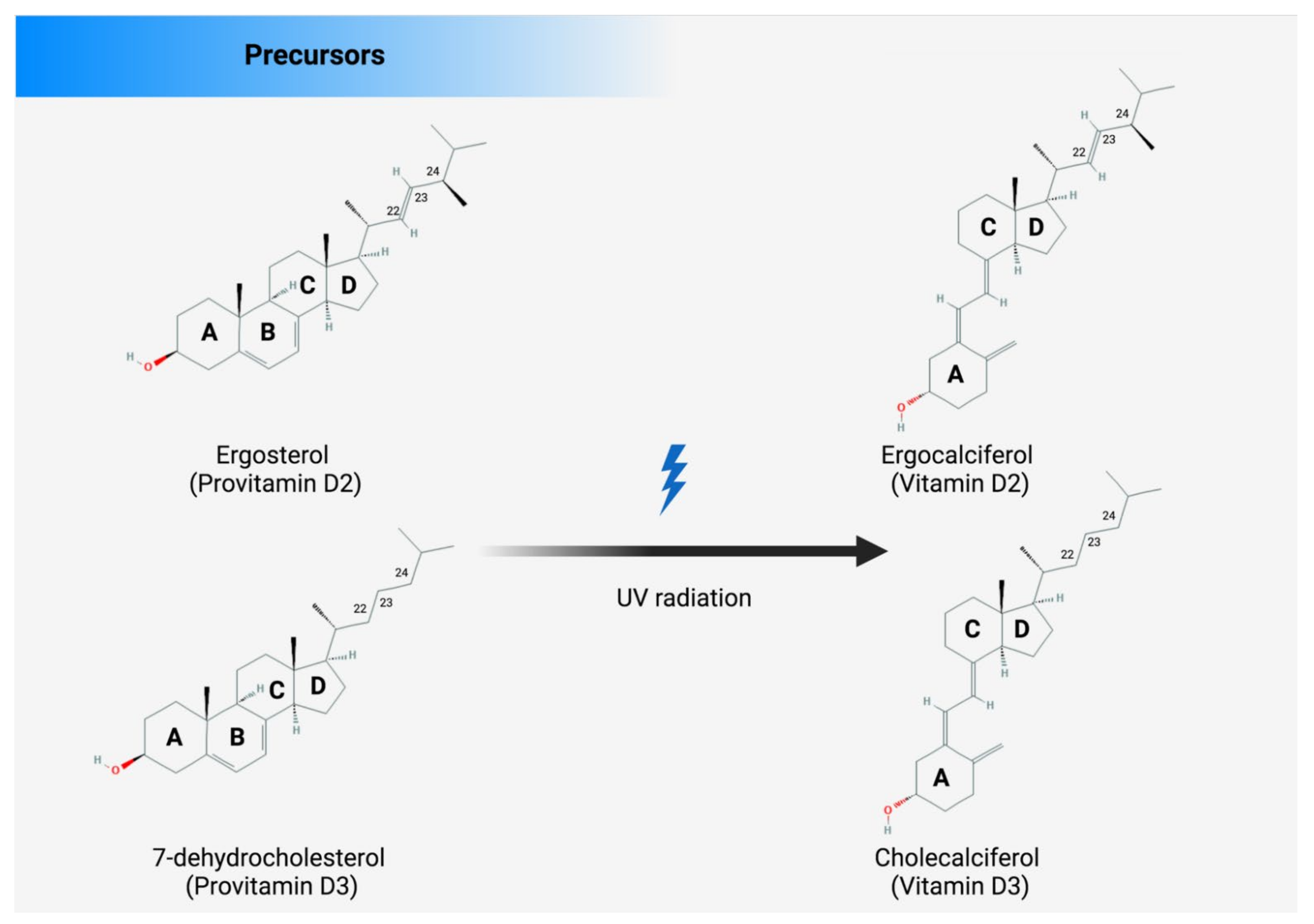
3. Endogenous and Exogenous Sources of Vitamin D
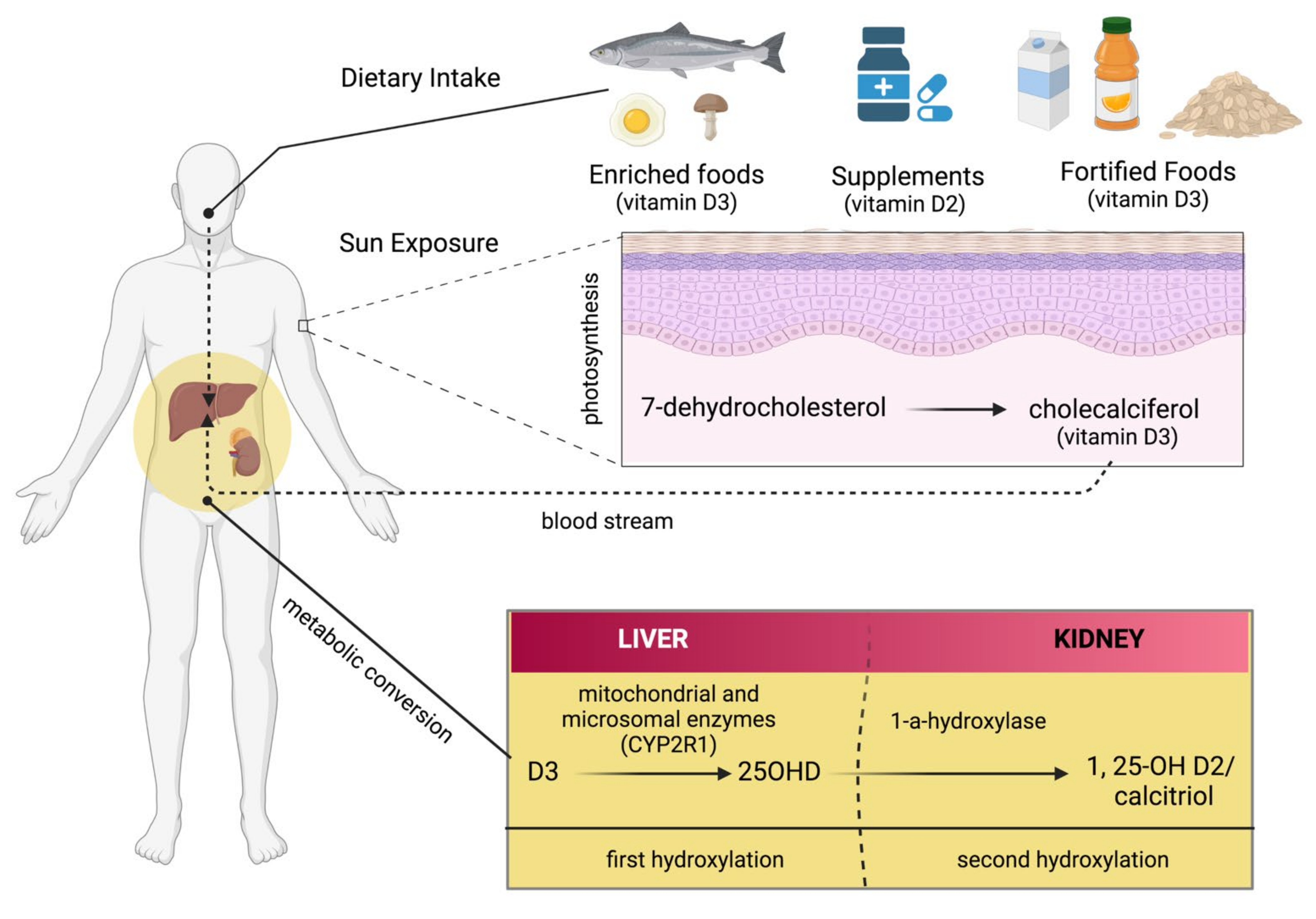
4. Metabolism of Vitamin D
5. Overall Effects of Vitamin D: From Antioxidant to Genomic Effects
6. Drug Interactions
7. Genetic Factors Influencing Vitamin D Homeostasis
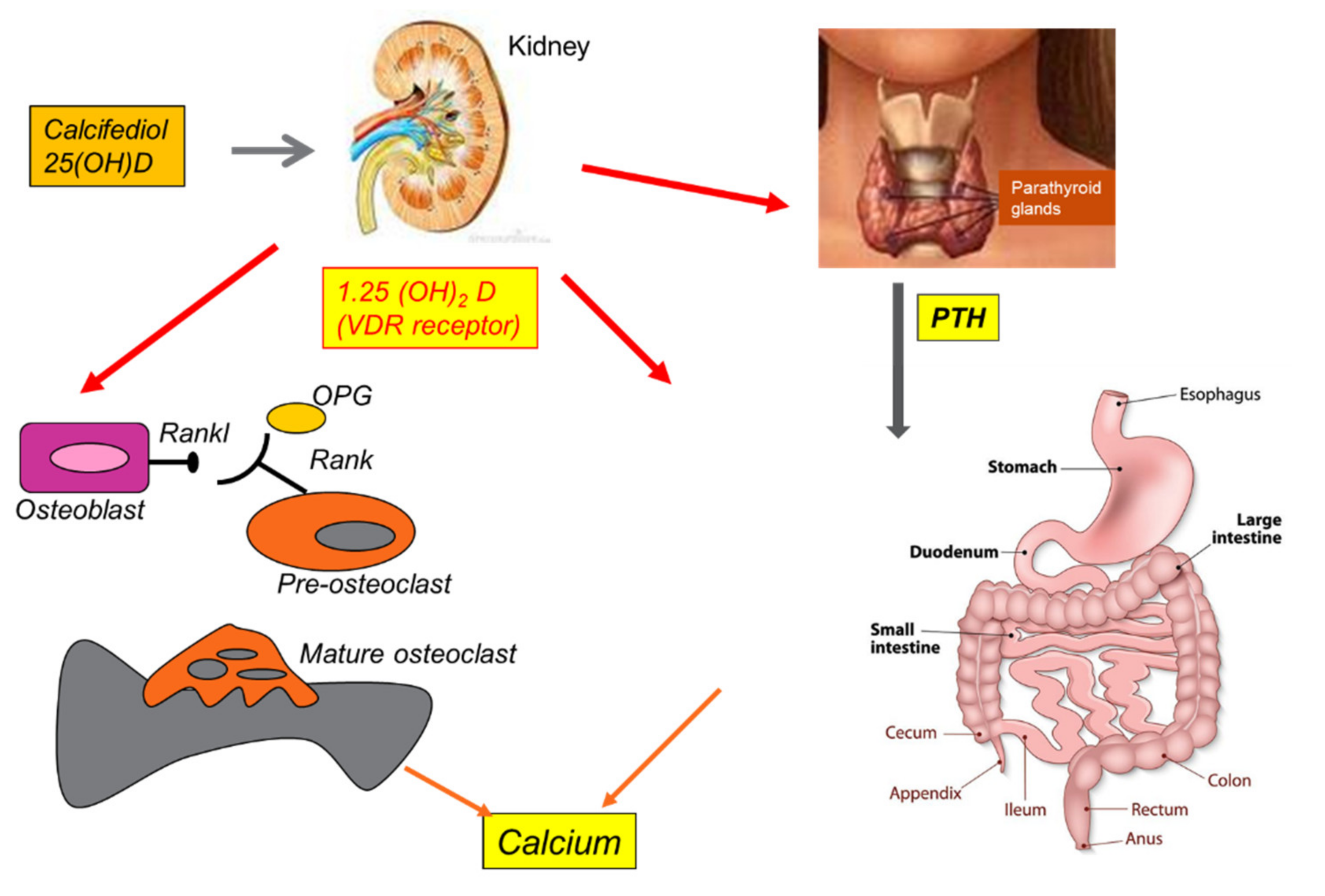
8. Vitamin D and Osteoporosis
9. Vitamin D and Muscle Homeostasis
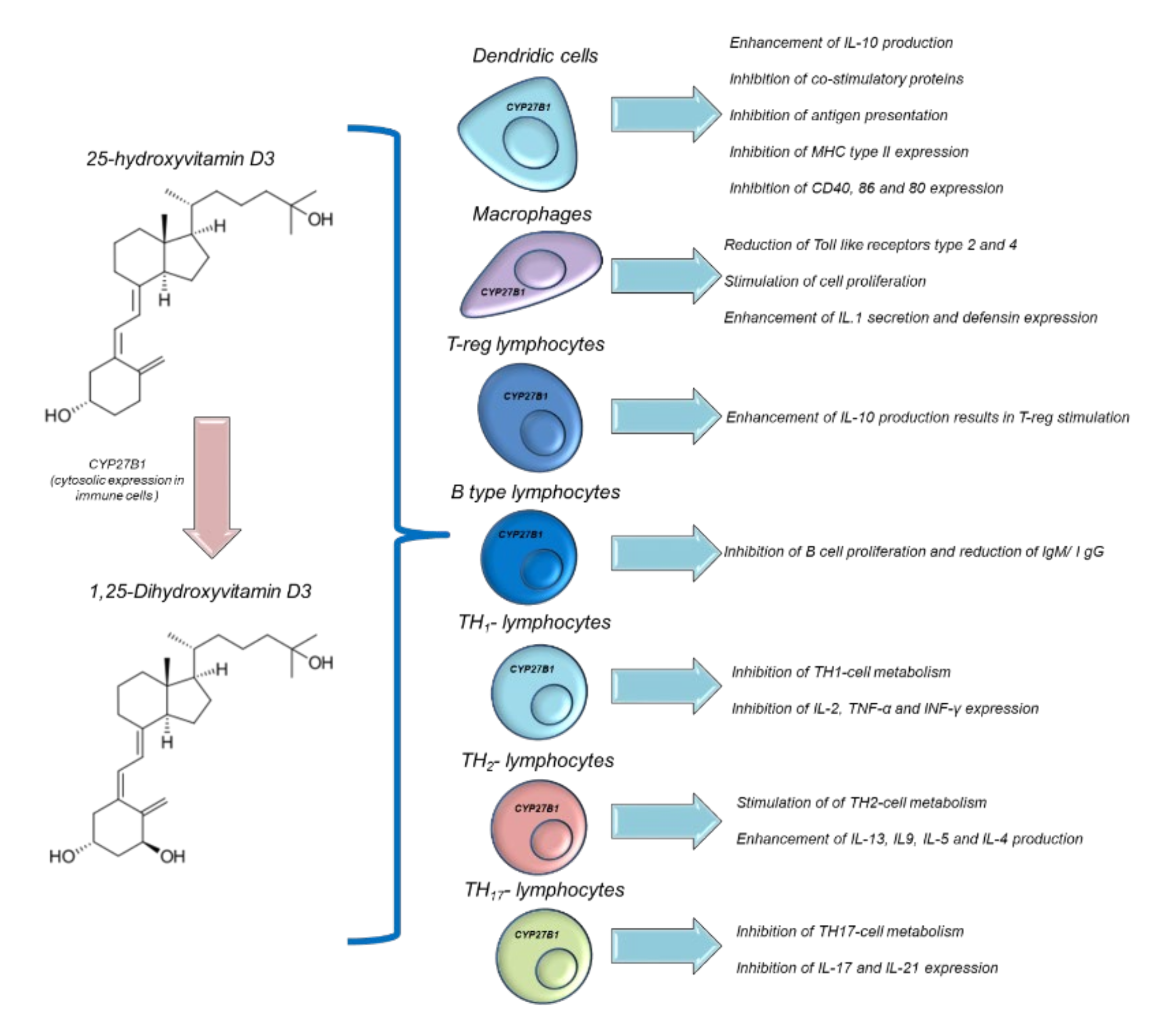
10. Vitamin D Supplementation and Modulation of Immune Functions: Putative Implications for Cancer Patients
11. Vitamin D and Cancer
11.1. Association between Vitamin D and Risk and Prevention of Cancer
11.2. Pathophysiology of Vitamin D in Cancer Prevention
11.3. Vitamin D levels and Breast Cancer
11.4. Vitamin D and Prostate Cancer
11.5. Vitamin D and Melanoma
11.6. Vitamin D and Colorectal Cancer
11.7. Vitamin D and Head-and-Neck Cancer
11.8. Vitamin D and Bladder Cancer
11.9. Vitamin D and Onco-Hematological Cancer
12. Vitamin D and Cardiovascular Diseases
13. Vitamin D and Human Microbiota
14. Vitamin D and Overall Risk of Infection
15. Conclusions
16. Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Reijven, P.L.M.; Soeters, P.B. Vitamin D: A Magic Bullet or a Myth? Clin. Nutr. 2020, 39, 2663–2674. [Google Scholar] [CrossRef] [PubMed]
- Carlberg, C. The Physiology of Vitamin D-Far More than Calcium and Bone. Front. Physiol. 2014, 5, 335. [Google Scholar] [CrossRef] [PubMed]
- Biancuzzo, R.M.; Clarke, N.; Reitz, R.E.; Travison, T.G.; Holick, M.F. Serum Concentrations of 1,25-Dihydroxyvitamin D2 and 1,25-Dihydroxyvitamin D3 in Response to Vitamin D2 and Vitamin D3 Supplementation. J. Clin. Endocrinol. Metab. 2013, 98, 973–979. [Google Scholar] [CrossRef] [PubMed]
- Medrano, M.; Carrillo-Cruz, E.; Montero, I.; Perez-Simon, J.A. Vitamin D: Effect on Haematopoiesis and Immune System and Clinical Applications. Int. J. Mol. Sci. 2018, 19, 2663. [Google Scholar] [CrossRef]
- Uchiyama, Y.; Higuchi, Y.; Takeda, S.; Masaki, T.; Shira-Ishi, A.; Sato, K.; Kubodera, N.; Ikeda, K.; Ogata, E. ED-71, a Vitamin D Analog, Is a More Potent Inhibitor of Bone Resorption than Alfacalcidol in an Estrogen-Deficient Rat Model of Osteoporosis. Bone 2002, 30, 582–588. [Google Scholar] [CrossRef]
- Bouillon, R.; Marcocci, C.; Carmeliet, G.; Bikle, D.; White, J.H.; Dawson-Hughes, B.; Lips, P.; Munns, C.F.; Lazaretti-Castro, M.; Giustina, A.; et al. Skeletal and Extraskeletal Actions of Vitamin D: Current Evidence and Outstanding Questions. Endocr. Rev. 2019, 40, 1109–1151. [Google Scholar] [CrossRef]
- Hossein-nezhad, A.; Holick, M.F. Vitamin D for Health: A Global Perspective. Mayo Clin. Proc. 2013, 88, 720–755. [Google Scholar] [CrossRef]
- Rusińska, A.; Płudowski, P.; Walczak, M.; Borszewska-Kornacka, M.K.; Bossowski, A.; Chlebna-Sokół, D.; Czech-Kowalska, J.; Dobrzańska, A.; Franek, E.; Helwich, E.; et al. Vitamin D Supplementation Guidelines for General Population and Groups at Risk of Vitamin D Deficiency in Poland-Recommendations of the Polish Society of Pediatric Endocrinology and Diabetes and the Expert Panel With Participation of National Specialist Consultants and Representatives of Scientific Societies-2018 Update. Front. Endocrinol. 2018, 9, 246. [Google Scholar] [CrossRef]
- Gandini, S.; Francesco, F.; Johanson, H.; Bonanni, B.; Testori, A. Why Vitamin D for Cancer Patients? Available online: https://pubmed.ncbi.nlm.nih.gov/22276021/ (accessed on 13 April 2022).
- PRISMA. Available online: http://prisma-statement.org/PRISMAStatement/FlowDiagram.aspx (accessed on 15 April 2022).
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; PRISMA-P Group. Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) 2015 Statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef]
- Lamberg-Allardt, C. Vitamin D in Foods and as Supplements. Prog. Biophys. Mol. Biol. 2006, 92, 33–38. [Google Scholar] [CrossRef]
- Holick, M.F.; Uskokovic, M.; Henley, J.W.; MacLaughlin, J.; Holick, S.A.; Potts, J.T. The Photoproduction of 1 Alpha,25-Dihydroxyvitamin D3 in Skin: An Approach to the Therapy of Vitamin-D-Resistant Syndromes. N. Engl. J. Med. 1980, 303, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Frommer, J.E.; McNeill, S.C.; Richtand, N.M.; Henley, J.W.; Potts, J.T. Photometabolism of 7-Dehydrocholesterol to Previtamin D3 in Skin. Biochem. Biophys. Res. Commun. 1977, 76, 107–114. [Google Scholar] [CrossRef]
- Dominguez, L.J.; Farruggia, M.; Veronese, N.; Barbagallo, M. Vitamin D Sources, Metabolism, and Deficiency: Available Compounds and Guidelines for Its Treatment. Metabolites 2021, 11, 255. [Google Scholar] [CrossRef] [PubMed]
- Klein, G.L.; Chen, T.C.; Holick, M.F.; Langman, C.B.; Price, H.; Celis, M.M.; Herndon, D.N. Synthesis of Vitamin D in Skin after Burns. Lancet 2004, 363, 291–292. [Google Scholar] [CrossRef]
- Tripkovic, L.; Lambert, H.; Hart, K.; Smith, C.P.; Bucca, G.; Penson, S.; Chope, G.; Hyppönen, E.; Berry, J.; Vieth, R.; et al. Comparison of Vitamin D2 and Vitamin D3 Supplementation in Raising Serum 25-Hydroxyvitamin D Status: A Systematic Review and Meta-Analysis. Am. J. Clin. Nutr. 2012, 95, 1357–1364. [Google Scholar] [CrossRef]
- Bouillon, R.; Schuit, F.; Antonio, L.; Rastinejad, F. Vitamin D Binding Protein: A Historic Overview. Front. Endocrinol. 2019, 10, 910. [Google Scholar] [CrossRef]
- Logan, V.F.; Gray, A.R.; Peddie, M.C.; Harper, M.J.; Houghton, L.A. Long-Term Vitamin D3 Supplementation Is More Effective than Vitamin D2 in Maintaining Serum 25-Hydroxyvitamin D Status over the Winter Months. Br. J. Nutr. 2013, 109, 1082–1088. [Google Scholar] [CrossRef]
- Houghton, L.A.; Vieth, R. The Case against Ergocalciferol (Vitamin D2) as a Vitamin Supplement. Am. J. Clin. Nutr. 2006, 84, 694–697. [Google Scholar] [CrossRef]
- Araya, Z.; Hosseinpour, F.; Bodin, K.; Wikvall, K. Metabolism of 25-Hydroxyvitamin D3 by Microsomal and Mitochondrial Vitamin D3 25-Hydroxylases (CYP2D25 and CYP27A1): A Novel Reaction by CYP27A1. Biochim. Biophys. Acta 2003, 1632, 40–47. [Google Scholar] [CrossRef]
- Bozic, M.; Álvarez, Á.; de Pablo, C.; Sanchez-Niño, M.-D.; Ortiz, A.; Dolcet, X.; Encinas, M.; Fernandez, E.; Valdivielso, J.M. Impaired Vitamin D Signaling in Endothelial Cell Leads to an Enhanced Leukocyte-Endothelium Interplay: Implications for Atherosclerosis Development. PLoS ONE 2015, 10, e0136863. [Google Scholar] [CrossRef]
- Slominski, A.T.; Kim, T.-K.; Li, W.; Yi, A.-K.; Postlethwaite, A.; Tuckey, R.C. The Role of CYP11A1 in the Production of Vitamin D Metabolites and Their Role in the Regulation of Epidermal Functions. J. Steroid Biochem. Mol. Biol. 2014, 144 Pt A, 28–39. [Google Scholar] [CrossRef]
- Slominski, A.T.; Janjetovic, Z.; Fuller, B.E.; Zmijewski, M.A.; Tuckey, R.C.; Nguyen, M.N.; Sweatman, T.; Li, W.; Zjawiony, J.; Miller, D.; et al. Products of Vitamin D3 or 7-Dehydrocholesterol Metabolism by Cytochrome P450scc Show Anti-Leukemia Effects, Having Low or Absent Calcemic Activity. PLoS ONE 2010, 5, e9907. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.G.; Ochalek, J.T.; Kaufmann, M.; Jones, G.; Deluca, H.F. CYP2R1 Is a Major, but Not Exclusive, Contributor to 25-Hydroxyvitamin D Production in Vivo. Proc. Natl. Acad. Sci. USA 2013, 110, 15650–15655. [Google Scholar] [CrossRef] [PubMed]
- Guryev, O.; Carvalho, R.A.; Usanov, S.; Gilep, A.; Estabrook, R.W. A Pathway for the Metabolism of Vitamin D3: Unique Hydroxylated Metabolites Formed during Catalysis with Cytochrome P450scc (CYP11A1). Proc. Natl. Acad. Sci. USA 2003, 100, 14754–14759. [Google Scholar] [CrossRef] [PubMed]
- Hollis, B.W. Circulating 25-Hydroxyvitamin D Levels Indicative of Vitamin D Sufficiency: Implications for Establishing a New Effective Dietary Intake Recommendation for Vitamin D. J. Nutr. 2005, 135, 317–322. [Google Scholar] [CrossRef]
- Mazahery, H.; von Hurst, P.R. Factors Affecting 25-Hydroxyvitamin D Concentration in Response to Vitamin D Supplementation. Nutrients 2015, 7, 5111–5142. [Google Scholar] [CrossRef]
- Bischof, M.G.; Siu-Caldera, M.L.; Weiskopf, A.; Vouros, P.; Cross, H.S.; Peterlik, M.; Reddy, G.S. Differentiation-Related Pathways of 1 Alpha,25-Dihydroxycholecalciferol Metabolism in Human Colon Adenocarcinoma-Derived Caco-2 Cells: Production of 1 Alpha,25-Dihydroxy-3epi-Cholecalciferol. Exp. Cell Res. 1998, 241, 194–201. [Google Scholar] [CrossRef]
- Kamao, M.; Tatematsu, S.; Hatakeyama, S.; Sakaki, T.; Sawada, N.; Inouye, K.; Ozono, K.; Kubodera, N.; Reddy, G.S.; Okano, T. C-3 Epimerization of Vitamin D3 Metabolites and Further Metabolism of C-3 Epimers: 25-Hydroxyvitamin D3 Is Metabolized to 3-Epi-25-Hydroxyvitamin D3 and Subsequently Metabolized through C-1alpha or C-24 Hydroxylation. J. Biol. Chem. 2004, 279, 15897–15907. [Google Scholar] [CrossRef]
- Slominski, A.T.; Kim, T.-K.; Shehabi, H.Z.; Semak, I.; Tang, E.K.Y.; Nguyen, M.N.; Benson, H.A.E.; Korik, E.; Janjetovic, Z.; Chen, J.; et al. In Vivo Evidence for a Novel Pathway of Vitamin D₃ Metabolism Initiated by P450scc and Modified by CYP27B1. FASEB J. 2012, 26, 3901–3915. [Google Scholar] [CrossRef]
- Slominski, A.T.; Kim, T.-K.; Li, W.; Postlethwaite, A.; Tieu, E.W.; Tang, E.K.Y.; Tuckey, R.C. Detection of Novel CYP11A1-Derived Secosteroids in the Human Epidermis and Serum and Pig Adrenal Gland. Sci. Rep. 2015, 5, 14875. [Google Scholar] [CrossRef]
- Slominski, A.T.; Li, W.; Kim, T.-K.; Semak, I.; Wang, J.; Zjawiony, J.K.; Tuckey, R.C. Novel Activities of CYP11A1 and Their Potential Physiological Significance. J. Steroid Biochem. Mol. Biol. 2015, 151, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Moghadasian, M.H. Cerebrotendinous Xanthomatosis: Clinical Course, Genotypes and Metabolic Backgrounds. Clin. Investig. Med. 2004, 27, 42–50. [Google Scholar]
- Cheng, J.B.; Motola, D.L.; Mangelsdorf, D.J.; Russell, D.W. De-Orphanization of Cytochrome P450 2R1: A Microsomal Vitamin D 25-Hydroxilase. J. Biol. Chem. 2003, 278, 38084–38093. [Google Scholar] [CrossRef] [PubMed]
- Farhan, F.; Almarhoun, M.; Wong, A.; Findlay, A.S.; Bartholomew, C.; Williams, M.T.S.; Hurd, T.W.; Shu, X. Deletion of TSPO Causes Dysregulation of Cholesterol Metabolism in Mouse Retina. Cells 2021, 10, 3066. [Google Scholar] [CrossRef] [PubMed]
- Molin, A.; Wiedemann, A.; Demers, N.; Kaufmann, M.; Do Cao, J.; Mainard, L.; Dousset, B.; Journeau, P.; Abeguile, G.; Coudray, N.; et al. Vitamin D-Dependent Rickets Type 1B (25-Hydroxylase Deficiency): A Rare Condition or a Misdiagnosed Condition? J. Bone Miner. Res. 2017, 32, 1893–1899. [Google Scholar] [CrossRef]
- Liu, M.; Zhu, Q.; Wu, J.; Yu, X.; Hu, M.; Xie, X.; Yang, Z.; Yang, J.; Feng, Y.-Q.; Yue, J. Glutamate Affects the Production of Epoxyeicosanoids within the Brain: The up-Regulation of Brain CYP2J through the MAPK-CREB Signaling Pathway. Toxicology 2017, 381, 31–38. [Google Scholar] [CrossRef]
- Hosseinpour, F.; Wikvall, K. Porcine Microsomal Vitamin D(3) 25-Hydroxylase (CYP2D25). Catalytic Properties, Tissue Distribution, and Comparison with Human CYP2D6. J. Biol. Chem. 2000, 275, 34650–34655. [Google Scholar] [CrossRef]
- Rahmaniyan, M.; Patrick, K.; Bell, N.H. Characterization of Recombinant CYP2C11: A Vitamin D 25-Hydroxylase and 24-Hydroxylase. Am. J. Physiol. Endocrinol. Metab. 2005, 288, E753–E760. [Google Scholar] [CrossRef]
- Fu, G.K.; Lin, D.; Zhang, M.Y.; Bikle, D.D.; Shackleton, C.H.; Miller, W.L.; Portale, A.A. Cloning of Human 25-Hydroxyvitamin D-1 Alpha-Hydroxylase and Mutations Causing Vitamin D-Dependent Rickets Type 1. Mol. Endocrinol. 1997, 11, 1961–1970. [Google Scholar] [CrossRef][Green Version]
- Shinki, T.; Shimada, H.; Wakino, S.; Anazawa, H.; Hayashi, M.; Saruta, T.; DeLuca, H.F.; Suda, T. Cloning and Expression of Rat 25-Hydroxyvitamin D3-1alpha-Hydroxylase CDNA. Proc. Natl. Acad. Sci. USA 1997, 94, 12920–12925. [Google Scholar] [CrossRef]
- Bikle, D.D. Vitamin D: Production, Metabolism and Mechanisms of Action. In Endotext; Feingold, K.R., Anawalt, B., Boyce, A., Chrousos, G., de Herder, W.W., Dhatariya, K., Dungan, K., Hershman, J.M., Hofland, J., et al., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- St-Arnaud, R.; Arabian, A.; Travers, R.; Barletta, F.; Raval-Pandya, M.; Chapin, K.; Depovere, J.; Mathieu, C.; Christakos, S.; Demay, M.B.; et al. Deficient Mineralization of Intramembranous Bone in Vitamin D-24-Hydroxylase-Ablated Mice Is Due to Elevated 1,25-Dihydroxyvitamin D and Not to the Absence of 24,25-Dihydroxyvitamin D. Endocrinology 2000, 141, 2658–2666. [Google Scholar] [CrossRef] [PubMed]
- Anderson, P.H.; Sawyer, R.K.; May, B.K.; O’Loughlin, P.D.; Morris, H.A. 25-Hydroxyvitamin D Requirement for Maintaining Skeletal Health Utilising a Sprague-Dawley Rat Model. J. Steroid Biochem. Mol. Biol. 2007, 103, 592–595. [Google Scholar] [CrossRef] [PubMed]
- Sheng, L.; Turner, A.G.; Barratt, K.; Kremer, R.; Morris, H.A.; Callen, D.F.; Anderson, P.H.; Tarulli, G.A. Mammary-Specific Ablation of Cyp24a1 Inhibits Development, Reduces Proliferation and Increases Sensitivity to Vitamin D. J. Steroid Biochem. Mol. Biol. 2019, 189, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Reddy, G.S.; Muralidharan, K.R.; Okamura, W.H.; Tserng, K.Y.; McLane, J.A. Metabolism of 1alpha,25-Dihydroxyvitamin D(3) and Its C-3 Epimer 1alpha,25-Dihydroxy-3-Epi-Vitamin D(3) in Neonatal Human Keratinocytes. Steroids 2001, 66, 441–450. [Google Scholar] [CrossRef]
- Pike, J.W.; Meyer, M.B. The Vitamin D Receptor: New Paradigms for the Regulation of Gene Expression by 1,25-Dihydroxyvitamin D(3). Endocrinol. Metab. Clin. N. Am. 2010, 39, 255–269. [Google Scholar] [CrossRef]
- Haussler, M.R.; Jurutka, P.W.; Mizwicki, M.; Norman, A.W. Vitamin D Receptor (VDR)-Mediated Actions of 1α,25(OH)₂vitamin D₃: Genomic and Non-Genomic Mechanisms. Best Pract. Res. Clin. Endocrinol. Metab. 2011, 25, 543–559. [Google Scholar] [CrossRef]
- Rochel, N.; Wurtz, J.M.; Mitschler, A.; Klaholz, B.; Moras, D. The crystal structure of the nuclear receptor for vitamin D bound to its natural ligand. Mol Cell. 2000, 5, 173–179. [Google Scholar] [CrossRef]
- Zella, L.A.; Meyer, M.B.; Nerenz, R.D.; Lee, S.M.; Martowicz, M.L.; Pike, J.W. Multifunctional Enhancers Regulate Mouse and Human Vitamin D Receptor Gene Transcription. Mol. Endocrinol. 2010, 24, 128–147. [Google Scholar] [CrossRef]
- Meyer, M.B.; Benkusky, N.A.; Sen, B.; Rubin, J.; Pike, J.W. Epigenetic Plasticity Drives Adipogenic and Osteogenic Differentiation of Marrow-Derived Mesenchymal Stem Cells. J. Biol. Chem. 2016, 291, 17829–17847. [Google Scholar] [CrossRef]
- Norman, A.W.; Okamura, W.H.; Hammond, M.W.; Bishop, J.E.; Dormanen, M.C.; Bouillon, R.; van Baelen, H.; Ridall, A.L.; Daane, E.; Khoury, R.; et al. Comparison of 6-s-Cis- and 6-s-Trans-Locked Analogs of 1alpha,25-Dihydroxyvitamin D3 Indicates That the 6-s-Cis Conformation Is Preferred for Rapid Nongenomic Biological Responses and That Neither 6-s-Cis- nor 6-s-Trans-Locked Analogs Are Preferred for Genomic Biological Responses. Mol. Endocrinol. 1997, 11, 1518–1531. [Google Scholar] [CrossRef]
- Dede, S.; Taşpinar, M.; Yüksek, V.; Çetin, S.; Usta, A. The Effects of Vitamin D Application on NaF-Induced Cytotoxicity in Osteoblast Cells (HFOB 1.19). Biol. Trace Elem. Res. 2022. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Wang, W.; Shi, L.; Yang, X.; Chen, Y.; Liu, X.; Li, J.; Ye, F.; An, X.; Zhang, X. Severe Vitamin D Deficiency Is Strongly Associated with Liver Dysfunction and Disease Severity in Hepatitis B Virus Related Cirrhosis and Liver Failure Patients. J. Nutr. Sci. Vitaminol. 2022, 68, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Russo, C.; Valle, M.S.; Casabona, A.; Spicuzza, L.; Sambataro, G.; Malaguarnera, L. Vitamin D Impacts on Skeletal Muscle Dysfunction in Patients with COPD Promoting Mitochondrial Health. Biomedicines 2022, 10, 898. [Google Scholar] [CrossRef] [PubMed]
- Śledzińska, K.; Landowski, P.; Żmijewski, M.A.; Kamińska, B.; Kowalski, K.; Liberek, A. Diet, Sun, Physical Activity and Vitamin D Status in Children with Inflammatory Bowel Disease. Nutrients 2022, 14, 1029. [Google Scholar] [CrossRef] [PubMed]
- Trasciatti, S.; Piras, F.; Bonaretti, S.; Marini, S.; Nencioni, S.; Biasci, E.; Egan, C.G.; Nannipieri, F. Effect of Oral Cholecalciferol in a Murine Model of Celiac Disease: A Dose Ranging Study. J. Steroid Biochem. Mol. Biol. 2022, 220, 106083. [Google Scholar] [CrossRef]
- Balion, C.; Griffith, L.E.; Strifler, L.; Henderson, M.; Patterson, C.; Heckman, G.; Llewellyn, D.J.; Raina, P. Vitamin D, Cognition, and Dementia: A Systematic Review and Meta-Analysis. Neurology 2012, 79, 1397–1405. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Willett, W.C.; Wong, J.B.; Giovannucci, E.; Dietrich, T.; Dawson-Hughes, B. Fracture Prevention with Vitamin D Supplementation: A Meta-Analysis of Randomized Controlled Trials. JAMA 2005, 293, 2257–2264. [Google Scholar] [CrossRef]
- Peterson, A.L.; Murchison, C.; Zabetian, C.; Leverenz, J.B.; Watson, G.S.; Montine, T.; Carney, N.; Bowman, G.L.; Edwards, K.; Quinn, J.F. Memory, Mood, and Vitamin D in Persons with Parkinson’s Disease. J. Parkinsons Dis. 2013, 3, 547–555. [Google Scholar] [CrossRef]
- Koduah, P.; Paul, F.; Dörr, J.-M. Vitamin D in the Prevention, Prediction and Treatment of Neurodegenerative and Neuroinflammatory Diseases. EPMA J. 2017, 8, 313–325. [Google Scholar] [CrossRef]
- Lee, S.S.; Ling, K.H.; Tusimin, M.; Subramaniam, R.; Rahim, K.F.; Loh, S.P. Interplay between Maternal and Neonatal Vitamin D Deficiency and Vitamin-D-Related Gene Polymorphism with Neonatal Birth Anthropometry. Nutrients 2022, 14, 564. [Google Scholar] [CrossRef]
- Manousaki, D.; Mitchell, R.; Dudding, T.; Haworth, S.; Harroud, A.; Forgetta, V.; Shah, R.L.; Luan, J.; Langenberg, C.; Timpson, N.J.; et al. Genome-Wide Association Study for Vitamin D Levels Reveals 69 Independent Loci. Am. J. Hum. Genet. 2020, 106, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Lin, Y.S.; Zheng, X.E.; Senn, T.; Hashizume, T.; Scian, M.; Dickmann, L.J.; Nelson, S.D.; Baillie, T.A.; Hebert, M.F.; et al. An Inducible Cytochrome P450 3A4-Dependent Vitamin D Catabolic Pathway. Mol. Pharmacol. 2012, 81, 498–509. [Google Scholar] [CrossRef] [PubMed]
- Nicolaidou, P.; Georgouli, H.; Kotsalis, H.; Matsinos, Y.; Papadopoulou, A.; Fretzayas, A.; Syriopoulou, V.; Krikos, X.; Karantana, A.; Karpathios, T. Effects of Anticonvulsant Therapy on Vitamin D Status in Children: Prospective Monitoring Study. J. Child. Neurol. 2006, 21, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Christiansen, C.; Rodbro, P.; Lund, M. Incidence of Anticonvulsant Osteomalacia and Effect of Vitamin D: Controlled Therapeutic Trial. Br. Med. J. 1973, 4, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Vitamin D3 Interactions Checker. Available online: https://www.drugs.com/drug-interactions/cholecalciferol,vitamin-d3.html (accessed on 13 April 2022).
- Tuomainen, T.-P. Randomized Controlled Trial of Vitamin D Supplementation on Glucose Metabolism in Subjects With Components of the Metabolic Syndrome; 2013. Available online: https://www.medifind.com/articles/clinical-trial/5822311 (accessed on 26 April 2022).
- Carlberg, C.; Haq, A. The Concept of the Personal Vitamin D Response Index. J. Steroid Biochem. Mol. Biol. 2018, 175, 12–17. [Google Scholar] [CrossRef]
- Smith, L.M.; Gallagher, J.C.; Suiter, C. Medium Doses of Daily Vitamin D Decrease Falls and Higher Doses of Daily Vitamin D3 Increase Falls: A Randomized Clinical Trial. J. Steroid Biochem. Mol. Biol. 2017, 173, 317–322. [Google Scholar] [CrossRef]
- Seuter, S.; Virtanen, J.K.; Nurmi, T.; Pihlajamäki, J.; Mursu, J.; Voutilainen, S.; Tuomainen, T.-P.; Neme, A.; Carlberg, C. Molecular Evaluation of Vitamin D Responsiveness of Healthy Young Adults. J. Steroid Biochem. Mol. Biol. 2017, 174, 314–321. [Google Scholar] [CrossRef]
- Kim, Y.A.; Yoon, J.W.; Lee, Y.; Choi, H.J.; Yun, J.W.; Bae, E.; Kwon, S.-H.; Ahn, S.E.; Do, A.-R.; Jin, H.; et al. Unveiling Genetic Variants Underlying Vitamin D Deficiency in Multiple Korean Cohorts by a Genome-Wide Association Study. Endocrinol. Metab. 2021, 36, 1189–1200. [Google Scholar] [CrossRef]
- Thacher, T.D.; Fischer, P.R.; Singh, R.J.; Roizen, J.; Levine, M.A. CYP2R1 Mutations Impair Generation of 25-Hydroxyvitamin D and Cause an Atypical Form of Vitamin D Deficiency. J. Clin. Endocrinol. Metab. 2015, 100, E1005–E1013. [Google Scholar] [CrossRef]
- Cheng, J.B.; Levine, M.A.; Bell, N.H.; Mangelsdorf, D.J.; Russell, D.W. Genetic Evidence That the Human CYP2R1 Enzyme Is a Key Vitamin D 25-Hydroxylase. Proc. Natl. Acad. Sci. USA 2004, 101, 7711–7715. [Google Scholar] [CrossRef]
- Ahn, J.; Yu, K.; Stolzenberg-Solomon, R.; Simon, K.C.; McCullough, M.L.; Gallicchio, L.; Jacobs, E.J.; Ascherio, A.; Helzlsouer, K.; Jacobs, K.B.; et al. Genome-Wide Association Study of Circulating Vitamin D Levels. Hum. Mol. Genet. 2010, 19, 2739–2745. [Google Scholar] [CrossRef] [PubMed]
- Bu, F.-X.; Armas, L.; Lappe, J.; Zhou, Y.; Gao, G.; Wang, H.-W.; Recker, R.; Zhao, L.-J. Comprehensive Association Analysis of Nine Candidate Genes with Serum 25-Hydroxy Vitamin D Levels among Healthy Caucasian Subjects. Hum. Genet. 2010, 128, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Simon, K.C.; Munger, K.L.; Kraft, P.; Hunter, D.J.; De Jager, P.L.; Ascherio, A. Genetic Predictors of 25-Hydroxyvitamin D Levels and Risk of Multiple Sclerosis. J. Neurol. 2011, 258, 1676–1682. [Google Scholar] [CrossRef] [PubMed]
- Cooper, J.D.; Smyth, D.J.; Walker, N.M.; Stevens, H.; Burren, O.S.; Wallace, C.; Greissl, C.; Ramos-Lopez, E.; Hyppönen, E.; Dunger, D.B.; et al. Inherited Variation in Vitamin D Genes Is Associated with Predisposition to Autoimmune Disease Type 1 Diabetes. Diabetes 2011, 60, 1624–1631. [Google Scholar] [CrossRef] [PubMed]
- Dorjgochoo, T.; Shi, J.; Gao, Y.T.; Long, J.; Delahanty, R.; Xiang, Y.B.; Cai, Q.; Shu, X.O. Genetic Variants in Vitamin D Metabolism-Related Genes and Body Mass Index: Analysis of Genome-Wide Scan Data of Approximately 7000 Chinese Women. Int. J. Obes. 2012, 36, 1252–1255. [Google Scholar] [CrossRef]
- Di Spigna, G.; Del Puente, A.; Covelli, B.; Abete, E.; Varriale, E.; Salzano, S.; Postiglione, L. Vitamin D Receptor Polymorphisms as Tool for Early Screening of Severe Bone Loss in Women Patients with Rheumatoid Arthritis. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 4664–4669. [Google Scholar]
- Divanoglou, N.; Komninou, D.; Stea, E.A.; Argiriou, A.; Papatzikas, G.; Tsakalof, A.; Pazaitou-Panayiotou, K.; Georgakis, M.K.; Petridou, E. Association of Vitamin D Receptor Gene Polymorphisms with Serum Vitamin D Levels in a Greek Rural Population (Velestino Study). Lifestyle Genom. 2021, 14, 81–90. [Google Scholar] [CrossRef]
- Usategui-Martín, R.; De Luis-Román, D.-A.; Fernández-Gómez, J.M.; Ruiz-Mambrilla, M.; Pérez-Castrillón, J.-L. Vitamin D Receptor (VDR) Gene Polymorphisms Modify the Response to Vitamin D Supplementation: A Systematic Review and Meta-Analysis. Nutrients 2022, 14, 360. [Google Scholar] [CrossRef]
- Zhao, X.-Q.; Chen, K.; Wan, H.-Y.; He, S.-Y.; Qin, H.-J.; Yu, B.; Jiang, N. Vitamin D Receptor Genetic Variations May Associate with the Risk of Developing Late Fracture-Related Infection in the Chinese Han Population. J. Immunol. Res. 2022, 2022, 9025354. [Google Scholar] [CrossRef]
- Jurutka, P.W.; Remus, L.S.; Whitfield, G.K.; Thompson, P.D.; Hsieh, J.C.; Zitzer, H.; Tavakkoli, P.; Galligan, M.A.; Dang, H.T.; Haussler, C.A.; et al. The Polymorphic N Terminus in Human Vitamin D Receptor Isoforms Influences Transcriptional Activity by Modulating Interaction with Transcription Factor IIB. Mol. Endocrinol. 2000, 14, 401–420. [Google Scholar] [CrossRef]
- Di Francia, R.; Amitrano, F.; De Lucia, D. Evaluation of Genotyping Methods and Costs for VDR Polymorphisms. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 1–3. [Google Scholar] [PubMed]
- D’Alésio, A.; Garabédian, M.; Sabatier, J.P.; Guaydier-Souquières, G.; Marcelli, C.; Lemaçon, A.; Walrant-Debray, O.; Jehan, F. Two Single-Nucleotide Polymorphisms in the Human Vitamin D Receptor Promoter Change Protein-DNA Complex Formation and Are Associated with Height and Vitamin D Status in Adolescent Girls. Hum. Mol. Genet. 2005, 14, 3539–3548. [Google Scholar] [CrossRef] [PubMed]
- Arai, H.; Miyamoto, K.I.; Yoshida, M.; Yamamoto, H.; Taketani, Y.; Morita, K.; Kubota, M.; Yoshida, S.; Ikeda, M.; Watabe, F.; et al. The Polymorphism in the Caudal-Related Homeodomain Protein Cdx-2 Binding Element in the Human Vitamin D Receptor Gene. J. Bone Miner. Res. 2001, 16, 1256–1264. [Google Scholar] [CrossRef] [PubMed]
- Rowland, G.W.; Schwartz, G.G.; John, E.M.; Ingles, S.A. Calcium Intake and Prostate Cancer among African Americans: Effect Modification by Vitamin D Receptor Calcium Absorption Genotype. J. Bone Miner. Res. 2012, 27, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Goltzman, D. Functions of Vitamin D in Bone. Histochem. Cell Biol. 2018, 149, 305–312. [Google Scholar] [CrossRef]
- Khundmiri, S.J.; Murray, R.D.; Lederer, E. PTH and Vitamin D. Compr. Physiol. 2016, 6, 561–601. [Google Scholar] [CrossRef]
- Goltzman, D.; Mannstadt, M.; Marcocci, C. Physiology of the Calcium-Parathyroid Hormone-Vitamin D Axis. Front. Horm. Res. 2018, 50, 1–13. [Google Scholar] [CrossRef]
- Marks, K.H.; Kilav, R.; Naveh-Many, T.; Silver, J. Calcium, Phosphate, Vitamin D, and the Parathyroid. Pediatr. Nephrol. 1996, 10, 364–367. [Google Scholar] [CrossRef]
- DeLuca, H.F. The Metabolism and Functions of Vitamin D. Adv. Exp. Med. Biol. 1986, 196, 361–375. [Google Scholar] [CrossRef]
- Epstein, O.; Kato, Y.; Dick, R.; Sherlock, S. Vitamin D, Hydroxyapatite, and Calcium Gluconate in Treatment of Cortical Bone Thinning in Postmenopausal Women with Primary Biliary Cirrhosis. Am. J. Clin. Nutr. 1982, 36, 426–430. [Google Scholar] [CrossRef]
- Ubesie, A.C.; Heubi, J.E.; Kocoshis, S.A.; Henderson, C.J.; Mezoff, A.G.; Rao, M.B.; Cole, C.R. Vitamin D Deficiency and Low Bone Mineral Density in Pediatric and Young Adult Intestinal Failure. J. Pediatr. Gastroenterol. Nutr. 2013, 57, 372–376. [Google Scholar] [CrossRef] [PubMed]
- Salovaara, K.; Tuppurainen, M.; Kärkkäinen, M.; Rikkonen, T.; Sandini, L.; Sirola, J.; Honkanen, R.; Alhava, E.; Kröger, H. Effect of Vitamin D(3) and Calcium on Fracture Risk in 65- to 71-Year-Old Women: A Population-Based 3-Year Randomized, Controlled Trial—The OSTPRE-FPS. J. Bone Miner. Res. 2010, 25, 1487–1495. [Google Scholar] [CrossRef] [PubMed]
- Seeman, E. Bone Modeling and Remodeling. Crit. Rev. Eukaryot. Gene Expr. 2009, 19, 219–233. [Google Scholar] [CrossRef] [PubMed]
- Langdahl, B.; Ferrari, S.; Dempster, D.W. Bone Modeling and Remodeling: Potential as Therapeutic Targets for the Treatment of Osteoporosis. Ther. Adv. Musculoskelet. Dis. 2016, 8, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Boyce, B.F.; Yao, Z.; Xing, L. Osteoclasts Have Multiple Roles in Bone in Addition to Bone Resorption. Crit. Rev. Eukaryot. Gene Expr. 2009, 19, 171–180. [Google Scholar] [CrossRef]
- Raggatt, L.J.; Partridge, N.C. Cellular and Molecular Mechanisms of Bone Remodeling. J. Biol. Chem. 2010, 285, 25103–25108. [Google Scholar] [CrossRef]
- Natsag, J.; Kendall, M.A.; Sellmeyer, D.E.; McComsey, G.A.; Brown, T.T. Vitamin D, Osteoprotegerin/Receptor Activator of Nuclear Factor-KappaB Ligand (OPG/RANKL) and Inflammation with Alendronate Treatment in HIV-Infected Patients with Reduced Bone Mineral Density. HIV Med. 2016, 17, 196–205. [Google Scholar] [CrossRef]
- Anderson, P.H.; Sawyer, R.K.; Moore, A.J.; May, B.K.; O’Loughlin, P.D.; Morris, H.A. Vitamin D Depletion Induces RANKL-Mediated Osteoclastogenesis and Bone Loss in a Rodent Model. J. Bone Miner. Res. 2008, 23, 1789–1797. [Google Scholar] [CrossRef]
- Chu, Y.-R.; Xu, S.-Q.; Wang, J.-X.; Zong, H.-X.; Chen, K.-M.; Wang, C.; Tong, W.-Q.; Wang, X. Synergy of Sarcopenia and Vitamin D Deficiency in Vertebral Osteoporotic Fractures in Rheumatoid Arthritis. Clin. Rheumatol. 2022. [Google Scholar] [CrossRef]
- Kulenović, I.; Rasić, S.; Kulenović, E. Osteoporosis: Current Trends in Diagnosis and Management. Bosn. J. Basic Med. Sci. 2006, 6, 24–28. [Google Scholar] [CrossRef]
- Sadat-Ali, M.; Al Elq, A.H.; Al-Turki, H.A.; Al-Mulhim, F.A.; Al-Ali, A.K. Influence of Vitamin D Levels on Bone Mineral Density and Osteoporosis. Ann. Saudi Med. 2011, 31, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Mokta, J.; Balraj; Mokta, K.; Ranjan, A.; Joshi, I.; Garg, M. High Prevalence of Hypovitaminosis D in Patients Presenting with Proximal Muscle Weakness: A Sub-Himalayan Study. J. Assoc. Phys. India 2017, 65, 55–58. [Google Scholar]
- Vieth, R. Why the Minimum Desirable Serum 25-Hydroxyvitamin D Level Should Be 75 Nmol/L (30 Ng/Ml). Best Pract. Res. Clin. Endocrinol. Metab. 2011, 25, 681–691. [Google Scholar] [CrossRef] [PubMed]
- Bischoff-Ferrari, H.A. Optimal Serum 25-Hydroxyvitamin D Levels for Multiple Health Outcomes. Adv. Exp. Med. Biol. 2014, 810, 500–525. [Google Scholar] [CrossRef] [PubMed]
- Van Leeuwen, J.P.; van Driel, M.; van den Bemd, G.J.; Pols, H.A. Vitamin D Control of Osteoblast Function and Bone Extracellular Matrix Mineralization. Crit. Rev. Eukaryot. Gene Expr. 2001, 11, 199–226. [Google Scholar] [CrossRef] [PubMed]
- Chiang, C.-M.; Ismaeel, A.; Griffis, R.B.; Weems, S. Effects of Vitamin D Supplementation on Muscle Strength in Athletes: A Systematic Review. J. Strength Cond. Res. 2017, 31, 566–574. [Google Scholar] [CrossRef]
- Bislev, L.S.; Grove-Laugesen, D.; Rejnmark, L. Vitamin D and Muscle Health: A Systematic Review and Meta-Analysis of Randomized Placebo-Controlled Trials. J. Bone Miner. Res. 2021, 36, 1651–1660. [Google Scholar] [CrossRef]
- Liu, P.; Hao, Q.; Hai, S.; Wang, H.; Cao, L.; Dong, B. Sarcopenia as a Predictor of All-Cause Mortality among Community-Dwelling Older People: A Systematic Review and Meta-Analysis. Maturitas 2017, 103, 16–22. [Google Scholar] [CrossRef]
- Girgis, C.M.; Mokbel, N.; Cha, K.M.; Houweling, P.J.; Abboud, M.; Fraser, D.R.; Mason, R.S.; Clifton-Bligh, R.J.; Gunton, J.E. The Vitamin D Receptor (VDR) Is Expressed in Skeletal Muscle of Male Mice and Modulates 25-Hydroxyvitamin D (25OHD) Uptake in Myofibers. Endocrinology 2014, 155, 3227–3237. [Google Scholar] [CrossRef]
- Garcia, L.A.; King, K.K.; Ferrini, M.G.; Norris, K.C.; Artaza, J.N. 1,25(OH)2vitamin D3 Stimulates Myogenic Differentiation by Inhibiting Cell Proliferation and Modulating the Expression of Promyogenic Growth Factors and Myostatin in C2C12 Skeletal Muscle Cells. Endocrinology 2011, 152, 2976–2986. [Google Scholar] [CrossRef]
- Lee, Y.M.; Kim, S.A.; Lee, D.H. Can Current Recommendations on Sun Exposure Sufficiently Increase Serum Vitamin D Level?: One-Month Randomized Clinical Trial. J. Korean Med. Sci. 2020, 35, e50. [Google Scholar] [CrossRef] [PubMed]
- Tepper, S.; Shahar, D.R.; Geva, D.; Ish-Shalom, S. Predictors of Serum 25(Oh)D Increase Following Bimonthly Supplementation with 100,000IU Vitamin D in Healthy, Men Aged 25-65 Years. J. Steroid Biochem. Mol. Biol. 2014, 144 Pt A, 163–166. [Google Scholar] [CrossRef]
- Bassatne, A.; Chakhtoura, M.; Saad, R.; Fuleihan, G.E.-H. Vitamin D Supplementation in Obesity and during Weight Loss: A Review of Randomized Controlled Trials. Metabolism 2019, 92, 193–205. [Google Scholar] [CrossRef] [PubMed]
- Delle Monache, S.; Di Fulvio, P.; Iannetti, E.; Valerii, L.; Capone, L.; Nespoli, M.G.; Bologna, M.; Angelucci, A. Body Mass Index Represents a Good Predictor of Vitamin D Status in Women Independently from Age. Clin. Nutr. 2019, 38, 829–834. [Google Scholar] [CrossRef] [PubMed]
- Arora, J.; Wang, J.; Weaver, V.; Zhang, Y.; Cantorna, M.T. Novel Insight into the Role of the Vitamin D Receptor in the Development and Function of the Immune System. J. Steroid Biochem. Mol. Biol. 2022, 219, 106084. [Google Scholar] [CrossRef]
- Charoenngam, N.; Holick, M.F. Immunologic Effects of Vitamin D on Human Health and Disease. Nutrients 2020, 12, 2097. [Google Scholar] [CrossRef]
- Wu, Y.; Lin, X.; Song, F.; Xue, D.; Wang, Y. Vitamin D3 Promotes Autophagy in THP-1 Cells Infected with Mycobacterium Tuberculosis. Exp. Ther. Med. 2022, 23, 240. [Google Scholar] [CrossRef]
- Matos, C.; Renner, K.; Peuker, A.; Schoenhammer, G.; Schreiber, L.; Bruss, C.; Eder, R.; Bruns, H.; Flamann, C.; Hoffmann, P.; et al. Physiological Levels of 25-Hydroxyvitamin D3 Induce a Suppressive CD4+ T Cell Phenotype Not Reflected in the Epigenetic Landscape. J Immunol. 2022, 95, e13146. [Google Scholar] [CrossRef]
- Bikle, D.D. Vitamin D Regulation of Immune Function during Covid-19. Rev. Endocr. Metab. Disord. 2022, 23, 279–285. [Google Scholar] [CrossRef]
- Schlingmann, K.P. Vitamin D-Dependent Hypercalcemia. Endocrino. Metab. Clin. N. Am. 2021, 50, 729–742. [Google Scholar] [CrossRef]
- Pérez-Ferro, M.; Romero-Bueno, F.I.; Serrano Del Castillo, C.; Mahillo, I.; Alvear, A.; Largo, R.; Herrero-Beaumont, G.; Sánchez-Pernaute, O. A Subgroup of Lupus Patients with Nephritis, Innate T Cell Activation and Low Vitamin D Is Identified by the Enhancement of Circulating MHC Class I-Related Chain A. Clin. Exp. Immunol. 2019, 196, 336–344. [Google Scholar] [CrossRef] [PubMed]
- Komisarenko, Y.I.; Bobryk, M.I. Vitamin D Deficiency and Immune Disorders in Combined Endocrine Pathology. Front. Endocrinol. 2018, 9, 600. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.; Chun, S.; Hwang, S.; Kim, J.; Cho, Y.; Lee, J.; Kwack, K.; Choi, S.-W. Vitamin D and Exercise Are Major Determinants of Natural Killer Cell Activity, Which Is Age- and Gender-Specific. Front. Immunol. 2021, 12, 594356. [Google Scholar] [CrossRef] [PubMed]
- Abrahamsson, H.; Porojnicu, A.C.; Lindstrøm, J.C.; Dueland, S.; Flatmark, K.; Hole, K.H.; Seierstad, T.; Moan, J.; Redalen, K.R.; Meltzer, S.; et al. High Level of Circulating Vitamin D during Neoadjuvant Therapy May Lower Risk of Metastatic Progression in High-Risk Rectal Cancer. BMC Cancer 2019, 19, 488. [Google Scholar] [CrossRef]
- Buttigliero, C.; Monagheddu, C.; Petroni, P.; Saini, A.; Dogliotti, L.; Ciccone, G.; Berruti, A. Prognostic Role of Vitamin d Status and Efficacy of Vitamin D Supplementation in Cancer Patients: A Systematic Review. Oncologist 2011, 16, 1215–1227. [Google Scholar] [CrossRef]
- Karata, F.; Adahan, D. Is Low Serum Vitamin D Level Associated with Cancer? VITAMIN D AND CANCER 7. WCRJ 2020, 7, e1683. [Google Scholar]
- Shah, S.I.A. Beneficial Role of Vitamin D in Common Cancers:Is the Evidence Compelling Enough? WCRJ 2020, 7, e1574. [Google Scholar]
- Feldman, D.; Krishnan, A.V.; Swami, S.; Giovannucci, E.; Feldman, B.J. The Role of Vitamin D in Reducing Cancer Risk and Progression. Nat. Rev. Cancer 2014, 14, 342–357. [Google Scholar] [CrossRef]
- Dalle Carbonare, L.; Valenti, M.T.; Del Forno, F.; Caneva, E.; Pietrobelli, A. Vitamin D: Daily vs. Monthly Use in Children and Elderly-What Is Going On? Nutrients 2017, 9, 652. [Google Scholar] [CrossRef]
- Garland, C.F.; Garland, F.C.; Gorham, E.D.; Lipkin, M.; Newmark, H.; Mohr, S.B.; Holick, M.F. The Role of Vitamin D in Cancer Prevention. Am. J. Public Health 2006, 96, 252–261. [Google Scholar] [CrossRef]
- Sluyter, J.D.; Manson, J.E.; Scragg, R. Vitamin D and Clinical Cancer Outcomes: A Review of Meta-Analyses. JBMR Plus 2021, 5, e10420. [Google Scholar] [CrossRef] [PubMed]
- Manson, J.E.; Bassuk, S.S.; Lee, I.-M.; Cook, N.R.; Albert, M.A.; Gordon, D.; Zaharris, E.; Macfadyen, J.G.; Danielson, E.; Lin, J.; et al. The VITamin D and OmegA-3 TriaL (VITAL): Rationale and Design of a Large Randomized Controlled Trial of Vitamin D and Marine Omega-3 Fatty Acid Supplements for the Primary Prevention of Cancer and Cardiovascular Disease. Contemp. Clin. Trials 2012, 33, 159–171. [Google Scholar] [CrossRef] [PubMed]
- Chandler, P.D.; Chen, W.Y.; Ajala, O.N.; Hazra, A.; Cook, N.; Bubes, V.; Lee, I.-M.; Giovannucci, E.L.; Willett, W.; Buring, J.E.; et al. Effect of Vitamin D3 Supplements on Development of Advanced Cancer: A Secondary Analysis of the VITAL Randomized Clinical Trial. JAMA Netw. Open 2020, 3, e2025850. [Google Scholar] [CrossRef] [PubMed]
- Shao, T.; Klein, P.; Grossbard, M.L. Vitamin D and Breast Cancer. Oncologist 2012, 17, 36–45. [Google Scholar] [CrossRef]
- Hossain, S.; Beydoun, M.A.; Beydoun, H.A.; Chen, X.; Zonderman, A.B.; Wood, R.J. Vitamin D and Breast Cancer: A Systematic Review and Meta-Analysis of Observational Studies. Clin. Nutr. ESPEN 2019, 30, 170–184. [Google Scholar] [CrossRef]
- Townsend, K.; Banwell, C.M.; Guy, M.; Colston, K.W.; Mansi, J.L.; Stewart, P.M.; Campbell, M.J.; Hewison, M. Autocrine Metabolism of Vitamin D in Normal and Malignant Breast Tissue. Clin. Cancer Res. 2005, 11, 3579–3586. [Google Scholar] [CrossRef]
- Bauer, S.R.; Hankinson, S.E.; Bertone-Johnson, E.R.; Ding, E.L. Plasma Vitamin D Levels, Menopause, and Risk of Breast Cancer: Dose-Response Meta-Analysis of Prospective Studies. Medicine 2013, 92, 123–131. [Google Scholar] [CrossRef]
- Krishnan, A.V.; Swami, S.; Feldman, D. The Potential Therapeutic Benefits of Vitamin D in the Treatment of Estrogen Receptor Positive Breast Cancer. Steroids 2012, 77, 1107–1112. [Google Scholar] [CrossRef]
- Swami, S.; Krishnan, A.V.; Peng, L.; Lundqvist, J.; Feldman, D. Transrepression of the Estrogen Receptor Promoter by Calcitriol in Human Breast Cancer Cells via Two Negative Vitamin D Response Elements. Endocr. Relat. Cancer 2013, 20, 565–577. [Google Scholar] [CrossRef]
- Chen, T.C.; Holick, M.F. Vitamin D and Prostate Cancer Prevention and Treatment. Trends Endocrinol. Metab. 2003, 14, 423–430. [Google Scholar] [CrossRef]
- Petrou, S.; Mamais, I.; Lavranos, G.; Tzanetakou, I.P.; Chrysostomou, S. Effect of Vitamin D Supplementation in Prostate Cancer: A Systematic Review of Randomized Control Trials. Int. J. Vitam Nutr. Res. 2018, 88, 100–112. [Google Scholar] [CrossRef] [PubMed]
- Flaig, T.W.; Barqawi, A.; Miller, G.; Kane, M.; Zeng, C.; Crawford, E.D.; Glodé, L.M. A Phase II Trial of Dexamethasone, Vitamin D, and Carboplatin in Patients with Hormone-Refractory Prostate Cancer. Cancer 2006, 107, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Trump, D.L.; Potter, D.M.; Muindi, J.; Brufsky, A.; Johnson, C.S. Phase II Trial of High-Dose, Intermittent Calcitriol (1,25 Dihydroxyvitamin D3) and Dexamethasone in Androgen-Independent Prostate Cancer. Cancer 2006, 106, 2136–2142. [Google Scholar] [CrossRef]
- Beer, T.M. ASCENT: The Androgen-Independent Prostate Cancer Study of Calcitriol Enhancing Taxotere. BJU Int. 2005, 96, 508–513. [Google Scholar] [CrossRef]
- Fink, M. Febrile Neutropenia and Infection in the ASCENT Studies. J. Clin. Oncol. 2011, 29, 4337, author reply 4337–4338. [Google Scholar] [CrossRef]
- Podgorska, E.; Kim, T.-K.; Janjetovic, Z.; Urbanska, K.; Tuckey, R.C.; Bae, S.; Slominski, A.T. Knocking out the Vitamin D Receptor Enhances Malignancy and Decreases Responsiveness to Vitamin D3 Hydroxyderivatives in Human Melanoma Cells. Cancers 2021, 13, 3111. [Google Scholar] [CrossRef]
- Skobowiat, C.; Oak, A.S.W.; Kim, T.-K.; Yang, C.H.; Pfeffer, L.M.; Tuckey, R.C.; Slominski, A.T. Noncalcemic 20-Hydroxyvitamin D3 Inhibits Human Melanoma Growth in in Vitro and in Vivo Models. Oncotarget 2017, 8, 9823–9834. [Google Scholar] [CrossRef]
- Newton-Bishop, J.A.; Chang, Y.-M.; Elliott, F.; Chan, M.; Leake, S.; Karpavicius, B.; Haynes, S.; Fitzgibbon, E.; Kukalizch, K.; Randerson-Moor, J.; et al. Relationship between Sun Exposure and Melanoma Risk for Tumours in Different Body Sites in a Large Case-Control Study in a Temperate Climate. Eur. J. Cancer 2011, 47, 732–741. [Google Scholar] [CrossRef]
- Johansson, H.; Spadola, G.; Tosti, G.; Mandalà, M.; Minisini, A.M.; Queirolo, P.; Aristarco, V.; Baldini, F.; Cocorocchio, E.; Albertazzi, E.; et al. Vitamin D Supplementation and Disease-Free Survival in Stage II Melanoma: A Randomized Placebo Controlled Trial. Nutrients 2021, 13, 1931. [Google Scholar] [CrossRef]
- Universitaire Ziekenhuizen Leuven. Vitamin D Supplementation in Cutaneous Malignant Melanoma Outcome; Universitaire Ziekenhuizen Leuven, 2020; KU Leuven. Available online: https://clinicaltrials.gov/ct2/show/NCT01748448 (accessed on 1 April 2022).
- De Smedt, J.; Van Kelst, S.; Boecxstaens, V.; Stas, M.; Bogaerts, K.; Vanderschueren, D.; Aura, C.; Vandenberghe, K.; Lambrechts, D.; Wolter, P.; et al. Vitamin D Supplementation in Cutaneous Malignant Melanoma Outcome (ViDMe): A Randomized Controlled Trial. BMC Cancer 2017, 17, 562. [Google Scholar] [CrossRef]
- Lopez-Caleya, J.F.; Ortega-Valín, L.; Fernández-Villa, T.; Delgado-Rodríguez, M.; Martín-Sánchez, V.; Molina, A.J. The Role of Calcium and Vitamin D Dietary Intake on Risk of Colorectal Cancer: Systematic Review and Meta-Analysis of Case-Control Studies. Cancer Causes Control. 2022, 33, 167–182. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.E.; Li, H.; Chan, A.T.; Hollis, B.W.; Lee, I.-M.; Stampfer, M.J.; Wu, K.; Giovannucci, E.; Ma, J. Circulating Levels of Vitamin D and Colon and Rectal Cancer: The Physicians’ Health Study and a Meta-Analysis of Prospective Studies. Cancer Prev. Res. 2011, 4, 735–743. [Google Scholar] [CrossRef] [PubMed]
- Gandini, S.; Boniol, M.; Haukka, J.; Byrnes, G.; Cox, B.; Sneyd, M.J.; Mullie, P.; Autier, P. Meta-Analysis of Observational Studies of Serum 25-Hydroxyvitamin D Levels and Colorectal, Breast and Prostate Cancer and Colorectal Adenoma. Int. J. Cancer 2011, 128, 1414–1424. [Google Scholar] [CrossRef]
- Maalmi, H.; Walter, V.; Jansen, L.; Boakye, D.; Schöttker, B.; Hoffmeister, M.; Brenner, H. Association between Blood 25-Hydroxyvitamin D Levels and Survival in Colorectal Cancer Patients: An Updated Systematic Review and Meta-Analysis. Nutrients 2018, 10, 896. [Google Scholar] [CrossRef]
- Touvier, M.; Chan, D.S.M.; Lau, R.; Aune, D.; Vieira, R.; Greenwood, D.C.; Kampman, E.; Riboli, E.; Hercberg, S.; Norat, T. Meta-Analyses of Vitamin D Intake, 25-Hydroxyvitamin D Status, Vitamin D Receptor Polymorphisms, and Colorectal Cancer Risk. Cancer Epidemiol. Biomark. Prev. 2011, 20, 1003–1016. [Google Scholar] [CrossRef]
- Ng, K.; Nimeiri, H.S.; McCleary, N.J.; Abrams, T.A.; Yurgelun, M.B.; Cleary, J.M.; Rubinson, D.A.; Schrag, D.; Miksad, R.; Bullock, A.J.; et al. Effect of High-Dose vs Standard-Dose Vitamin D3 Supplementation on Progression-Free Survival Among Patients With Advanced or Metastatic Colorectal Cancer: The SUNSHINE Randomized Clinical Trial. JAMA 2019, 321, 1370–1379. [Google Scholar] [CrossRef]
- Bochen, F.; Balensiefer, B.; Körner, S.; Bittenbring, J.T.; Neumann, F.; Koch, A.; Bumm, K.; Marx, A.; Wemmert, S.; Papaspyrou, G.; et al. Vitamin D Deficiency in Head and Neck Cancer Patients—Prevalence, Prognostic Value and Impact on Immune Function. Oncoimmunology 2018, 7, e1476817. [Google Scholar] [CrossRef]
- Pu, Y.; Zhu, G.; Xu, Y.; Zheng, S.; Tang, B.; Huang, H.; Wu, I.X.Y.; Hunag, D.; Liu, Y.; Zhng, X. Association Between Vitamin D Exposure and Head and Neck Cancer: A Systematic Review With Meta-Analysis. Available online: https://pubmed.ncbi.nlm.nih.gov/33732250/ (accessed on 13 April 2022).
- Izreig, S.; Hajek, M.; Edwards, H.A.; Mehra, S.; Sasaki, C.; Judson, B.L.; Rahmati, R.W. The Role of Vitamin D in Head and Neck Cancer. Laryngoscope Investig. Otolaryngol. 2020, 5, 1079–1088. [Google Scholar] [CrossRef]
- Baykan, O.; Akgul, M.; Uren, N.; Yaman, A.; Tinay, I.; Ergul, E.; Sazci, A.; Turkeri, L.; Haklar, G. The Relationship Between Urothelial Type Bladder Cancer, Plasma 25-Hydroxyvitamin D Levels, and Vitamin D Receptor ApaI BsmI FokI, and TaqI Polymorphisms. Clin. Lab. 2019, 65. [Google Scholar] [CrossRef]
- Duan, G.-Q.; Zheng, X.; Li, W.-K.; Zhang, W.; Li, Z.; Tan, W. The Association Between VDR and GC Polymorphisms and Lung Cancer Risk: A Systematic Review and Meta-Analysis. Genet. Test. Mol. Biomark. 2020, 24, 285–295. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, H.; Wen, X.; Zhang, Y.; Wei, X.; Liu, T. Vitamin D Deficiency and Increased Risk of Bladder Carcinoma: A Meta-Analysis. Cell Physiol. Biochem. 2015, 37, 1686–1692. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Chen, C.; Pan, W.; Gao, M.; He, W.; Mao, R.; Lin, T.; Huang, J. Comparative Efficacy of Vitamin D Status in Reducing the Risk of Bladder Cancer: A Systematic Review and Network Meta-Analysis. Nutrition 2016, 32, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Thandra, K.C.; Barsouk, A.; Saginala, K.; Padala, S.A.; Barsouk, A.; Rawla, P. Epidemiology of Non-Hodgkin’s Lymphoma. Med. Sci. 2021, 9, 5. [Google Scholar] [CrossRef] [PubMed]
- Park, H.Y.; Hong, Y.-C.; Lee, K.; Koh, J. Vitamin D Status and Risk of Non-Hodgkin Lymphoma: An Updated Meta-Analysis. PLoS ONE 2019, 14, e0216284. [Google Scholar] [CrossRef]
- Friedberg, J. ILyAD (Indolent Lymphoma And Vitamin D) A Phase III Double Blind., Randomized Trial to Evaluate the Supplemental Effect of Vitamin D on Progression-Free Survival in Patients With Low Tumor-Burden Indolent Non-Hodgkin Lymphoma Treated With Rituximab; 2022. Available online: https://ascopubs.org/doi/abs/10.1200/JCO.2018.36.15_suppl.TPS7587 (accessed on 26 April 2022).
- Drake, M.T.; Maurer, M.J.; Link, B.K.; Habermann, T.M.; Ansell, S.M.; Micallef, I.N.; Kelly, J.L.; Macon, W.R.; Nowakowski, G.S.; Inwards, D.J.; et al. Vitamin D Insufficiency and Prognosis in Non-Hodgkin’s Lymphoma. J. Clin. Oncol 2010, 28, 4191–4198. [Google Scholar] [CrossRef] [PubMed]
- Warwick, T.; Schulz, M.H.; Gilsbach, R.; Brandes, R.P.; Seuter, S. Nuclear Receptor Activation Shapes Spatial Genome Organization Essential for Gene Expression Control: Lessons Learned from the Vitamin D Receptor. Nucleic Acids Res. 2022, 50, 745–3763. [Google Scholar] [CrossRef]
- Gouni-Berthold, I.; Berthold, H.K. Vitamin D and Vascular Disease. Curr. Vasc. Pharmacol. 2021, 19, 250–268. [Google Scholar] [CrossRef]
- Latic, N.; Erben, R.G. Vitamin D and Cardiovascular Disease, with Emphasis on Hypertension, Atherosclerosis, and Heart Failure. Int. J. Mol. Sci. 2020, 21, 6483. [Google Scholar] [CrossRef]
- Grandi, N.C.; Breitling, L.P.; Brenner, H. Vitamin D and Cardiovascular Disease: Systematic Review and Meta-Analysis of Prospective Studies. Prev. Med. 2010, 51, 228–233. [Google Scholar] [CrossRef]
- Zhang, W.; Yi, J.; Liu, D.; Wang, Y.; Jamilian, P.; Gaman, M.-A.; Prabahar, K.; Fan, J. The Effect of Vitamin D on the Lipid Profile as a Risk Factor for Coronary Heart Disease in Postmenopausal Women: A Meta-Analysis and Systematic Review of Randomized Controlled Trials. Exp. Gerontol. 2022, 161, 111709. [Google Scholar] [CrossRef]
- Scragg, R.; Stewart, A.W.; Waayer, D.; Lawes, C.M.M.; Toop, L.; Sluyter, J.; Murphy, J.; Khaw, K.T.; Camargo, C.A., Jr. Effect of Monthly High-Dose Vitamin D Supplementation on Cardiovascular Disease in the Vitamin D Assessment Study: A Randomized Clinical Trial. Available online: https://pubmed.ncbi.nlm.nih.gov/28384800/ (accessed on 19 April 2022).
- Amrein, K.; Parekh, D.; Westphal, S.; Preiser, J.-C.; Berghold, A.; Riedl, R.; Eller, P.; Schellongowski, P.; Thickett, D.; Meybohm, P.; et al. Effect of High-Dose Vitamin D3 on 28-Day Mortality in Adult Critically Ill Patients with Severe Vitamin D Deficiency: A Study Protocol of a Multicentre, Placebo-Controlled Double-Blind Phase III RCT (the VITDALIZE Study). BMJ Open 2019, 9, e031083. [Google Scholar] [CrossRef] [PubMed]
- Beveridge, L.A.; Khan, F.; Struthers, A.D.; Armitage, J.; Barchetta, I.; Bressendorff, I.; Cavallo, M.G.; Clarke, R.; Dalan, R.; Dreyer, G.; et al. Effect of Vitamin D Supplementation on Markers of Vascular Function: A Systematic Review and Individual Participant Meta-Analysis. J. Am. Heart Assoc. 2018, 7, e008273. [Google Scholar] [CrossRef]
- Abderhalden, L.A.; Meyer, S.; Dawson-Hughes, B.; Orav, E.J.; Meyer, U.; de Godoi Rezende Costa Molino, C.; Theiler, R.; Stähelin, H.B.; Ruschitzka, F.; Egli, A.; et al. Effect of Daily 2000 IU versus 800 IU Vitamin D on Blood Pressure among Adults Age 60 Years and Older: A Randomized Clinical Trial. Am. J. Clin. Nutr. 2020, 112, 527–537. [Google Scholar] [CrossRef] [PubMed]
- Rea, D.; Coppola, G.; Palma, G.; Barbieri, A.; Luciano, A.; Del Prete, P.; Rossetti, S.; Berretta, M.; Facchini, G.; Perdonà, S.; et al. Microbiota Effects on Cancer: From Risks to Therapies. Oncotarget 2018, 9, 17915–17927. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Hamady, M.; Fraser-Liggett, C.M.; Knight, R.; Gordon, J.I. The Human Microbiome Project. Nature 2007, 449, 804–810. [Google Scholar] [CrossRef]
- Sorboni, S.G.; Moghaddam, H.S.; Jafarzadeh-Esfehani, R.; Soleimanpour, S. A Comprehensive Review on the Role of the Gut Microbiome in Human Neurological Disorders. Clin. Microbiol Rev. 2022, 35, e0033820. [Google Scholar] [CrossRef]
- Baquero, F.; Nombela, C. The Microbiome as a Human Organ. Clin. Microbiol. Infect. 2012, 18 (Suppl. S4), 2–4. [Google Scholar] [CrossRef]
- Gominak, S.C. Vitamin D Deficiency Changes the Intestinal Microbiome Reducing B Vitamin Production in the Gut. The Resulting Lack of Pantothenic Acid Adversely Affects the Immune System, Producing a “pro-Inflammatory” State Associated with Atherosclerosis and Autoimmunity. Med. Hypotheses 2016, 94, 103–107. [Google Scholar] [CrossRef]
- Del Chierico, F.; Vernocchi, P.; Dallapiccola, B.; Putignani, L. Mediterranean Diet and Health: Food Effects on Gut Microbiota and Disease Control. Int. J. Mol. Sci. 2014, 15, 11678–11699. [Google Scholar] [CrossRef]
- Su, D.; Nie, Y.; Zhu, A.; Chen, Z.; Wu, P.; Zhang, L.; Luo, M.; Sun, Q.; Cai, L.; Lai, Y.; et al. Vitamin D Signaling through Induction of Paneth Cell Defensins Maintains Gut Microbiota and Improves Metabolic Disorders and Hepatic Steatosis in Animal Models. Front. Physiol. 2016, 7, 498. [Google Scholar] [CrossRef]
- Jin, D.; Wu, S.; Zhang, Y.-G.; Lu, R.; Xia, Y.; Dong, H.; Sun, J. Lack of Vitamin D Receptor Causes Dysbiosis and Changes the Functions of the Murine Intestinal Microbiome. Clin. Ther. 2015, 37, 996–1009.e7. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Yoon, S.; Zhang, Y.-G.; Lu, R.; Xia, Y.; Wan, J.; Petrof, E.O.; Claud, E.C.; Chen, D.; Sun, J. Vitamin D Receptor Pathway Is Required for Probiotic Protection in Colitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2015, 309, G341–G349. [Google Scholar] [CrossRef] [PubMed]
- Ooi, J.H.; Li, Y.; Rogers, C.J.; Cantorna, M.T. Vitamin D Regulates the Gut Microbiome and Protects Mice from Dextran Sodium Sulfate-Induced Colitis. J. Nutr. 2013, 143, 1679–1686. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.L.; Martoni, C.J.; Prakash, S. Oral Supplementation with Probiotic L. Reuteri NCIMB 30242 Increases Mean Circulating 25-Hydroxyvitamin D: A Post Hoc Analysis of a Randomized Controlled Trial. J. Clin. Endocrinol. Metab. 2013, 98, 2944–2951. [Google Scholar] [CrossRef]
- Wang, T.J.; Zhang, F.; Richards, J.B.; Kestenbaum, B.; van Meurs, J.B.; Berry, D.; Kiel, D.P.; Streeten, E.A.; Ohlsson, C.; Koller, D.L.; et al. Common Genetic Determinants of Vitamin D Insufficiency: A Genome-Wide Association Study. Lancet 2010, 376, 180–188. [Google Scholar] [CrossRef]
- Zhang, X.; Shang, X.; Jin, S.; Ma, Z.; Wang, H.; Ao, N.; Yang, J.; Du, J. Vitamin D Ameliorates High-Fat-Diet-Induced Hepatic Injury via Inhibiting Pyroptosis and Alters Gut Microbiota in Rats. Arch. Biochem. Biophys. 2021, 705, 108894. [Google Scholar] [CrossRef]
- Owen, J.L.; Mohamadzadeh, M. Microbial Activation of Gut Dendritic Cells and the Control of Mucosal Immunity. J. Interferon Cytokine Res. 2013, 33, 619–631. [Google Scholar] [CrossRef]
- Thomas, R.L.; Jiang, L.; Adams, J.S.; Xu, Z.Z.; Shen, J.; Janssen, S.; Ackermann, G.; Vanderschueren, D.; Pauwels, S.; Knight, R.; et al. Vitamin D Metabolites and the Gut Microbiome in Older Men. Nat. Commun. 2020, 11, 5997. [Google Scholar] [CrossRef]
- Gaschott, T.; Werz, O.; Steinmeyer, A.; Steinhilber, D.; Stein, J. Butyrate-Induced Differentiation of Caco-2 Cells Is Mediated by Vitamin D Receptor. Biochem. Biophys. Res. Commun. 2001, 288, 690–696. [Google Scholar] [CrossRef]
- Quagliariello, V.; Masarone, M.; Armenia, E.; Giudice, A.; Barbarisi, M.; Caraglia, M.; Barbarisi, A.; Persico, M. Chitosan-Coated Liposomes Loaded with Butyric Acid Demonstrate Anticancer and Anti-Inflammatory Activity in Human Hepatoma HepG2 Cells. Oncol. Rep. 2019, 41, 1476–1486. [Google Scholar] [CrossRef]
- Sokol, H.; Pigneur, B.; Watterlot, L.; Lakhdari, O.; Bermúdez-Humarán, L.G.; Gratadoux, J.-J.; Blugeon, S.; Bridonneau, C.; Furet, J.-P.; Corthier, G.; et al. Faecalibacterium Prausnitzii Is an Anti-Inflammatory Commensal Bacterium Identified by Gut Microbiota Analysis of Crohn Disease Patients. Proc. Natl. Acad. Sci. USA 2008, 105, 16731–16736. [Google Scholar] [CrossRef] [PubMed]
- Schäffler, H.; Herlemann, D.P.; Klinitzke, P.; Berlin, P.; Kreikemeyer, B.; Jaster, R.; Lamprecht, G. Vitamin D Administration Leads to a Shift of the Intestinal Bacterial Composition in Crohn’s Disease Patients, but Not in Healthy Controls. J. Dig. Dis. 2018, 19, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Garg, M.; Hendy, P.; Ding, J.N.; Shaw, S.; Hold, G.; Hart, A. The Effect of Vitamin D on Intestinal Inflammation and Faecal Microbiota in Patients with Ulcerative Colitis. J. Crohns Colitis 2018, 12, 963–972. [Google Scholar] [CrossRef] [PubMed]
- Berni Canani, R.; Sangwan, N.; Stefka, A.T.; Nocerino, R.; Paparo, L.; Aitoro, R.; Calignano, A.; Khan, A.A.; Gilbert, J.A.; Nagler, C.R. Lactobacillus Rhamnosus GG-Supplemented Formula Expands Butyrate-Producing Bacterial Strains in Food Allergic Infants. ISME J. 2016, 10, 742–750. [Google Scholar] [CrossRef]
- Wang, J.; Thingholm, L.B.; Skiecevičienė, J.; Rausch, P.; Kummen, M.; Hov, J.R.; Degenhardt, F.; Heinsen, F.-A.; Rühlemann, M.C.; Szymczak, S.; et al. Genome-Wide Association Analysis Identifies Variation in Vitamin D Receptor and Other Host Factors Influencing the Gut Microbiota. Nat. Genet. 2016, 48, 1396–1406. [Google Scholar] [CrossRef]
- Kanhere, M.; He, J.; Chassaing, B.; Ziegler, T.R.; Alvarez, J.A.; Ivie, E.A.; Hao, L.; Hanfelt, J.; Gewirtz, A.T.; Tangpricha, V. Bolus Weekly Vitamin D3 Supplementation Impacts Gut and Airway Microbiota in Adults With Cystic Fibrosis: A Double-Blind, Randomized, Placebo-Controlled Clinical Trial. J. Clin. Endocrinol. Metab. 2018, 103, 564–574. [Google Scholar] [CrossRef]
- Bashir, M.; Prietl, B.; Tauschmann, M.; Mautner, S.I.; Kump, P.K.; Treiber, G.; Wurm, P.; Gorkiewicz, G.; Högenauer, C.; Pieber, T.R. Effects of High Doses of Vitamin D3 on Mucosa-Associated Gut Microbiome Vary between Regions of the Human Gastrointestinal Tract. Eur. J. Nutr. 2016, 55, 1479–1489. [Google Scholar] [CrossRef]
- Yamamoto, E.A.; Jørgensen, T.N. Relationships Between Vitamin D, Gut Microbiome, and Systemic Autoimmunity. Front. Immunol. 2019, 10, 3141. [Google Scholar] [CrossRef]
- Mailhot, G.; White, J.H. Vitamin D and Immunity in Infants and Children. Nutrients 2020, 12, 1233. [Google Scholar] [CrossRef]
- Paiz, N.; Alonso, P.; Portillo, A.L. Vitamin D Status: Can It Affect the Risk of Infection and the Severity of COVID-19 Symptoms? Curr. Trop. Med. Rep 2021, 8, 204–211. [Google Scholar] [CrossRef]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D Supplementation to Prevent Acute Respiratory Tract Infections: Systematic Review and Meta-Analysis of Individual Participant Data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef] [PubMed]
- Radujkovic, A.; Hippchen, T.; Tiwari-Heckler, S.; Dreher, S.; Boxberger, M.; Merle, U. Vitamin D Deficiency and Outcome of COVID-19 Patients. Nutrients 2020, 12, 2757. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Liao, Q.; Shen, Y.; Li, H.; Cheng, L. Vitamin D Deficiency Is Associated with COVID-19 Incidence and Disease Severity in Chinese People [Corrected]. J. Nutr. 2021, 151, 98–103. [Google Scholar] [CrossRef]
- Cereda, E.; Bogliolo, L.; Lobascio, F.; Barichella, M.; Zecchinelli, A.L.; Pezzoli, G.; Caccialanza, R. Vitamin D Supplementation and Outcomes in Coronavirus Disease 2019 (COVID-19) Patients from the Outbreak Area of Lombardy, Italy. Nutrition 2021, 82, 111055. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D.; Ritz, C.; Kiely, M.; Odin Collaborators. Improved Dietary Guidelines for Vitamin D: Application of Individual Participant Data (IPD)-Level Meta-Regression Analyses. Nutrients 2017, 9, 469. [Google Scholar] [CrossRef]
- Yao, P.; Bennett, D.; Mafham, M.; Lin, X.; Chen, Z.; Armitage, J.; Clarke, R. Vitamin D and Calcium for the Prevention of Fracture: A Systematic Review and Meta-Analysis. JAMA Netw. Open 2019, 2, e1917789. [Google Scholar] [CrossRef]
- Burt, L.A.; Billington, E.O.; Rose, M.S.; Raymond, D.A.; Hanley, D.A.; Boyd, S.K. Effect of High-Dose Vitamin D Supplementation on Volumetric Bone Density and Bone Strength: A Randomized Clinical Trial. JAMA 2019, 322, 736–745. [Google Scholar] [CrossRef]
- Chakhtoura, M.; Chamoun, N.; Rahme, M.; Fuleihan, G.E.-H. Impact of Vitamin D Supplementation on Falls and Fractures-A Critical Appraisal of the Quality of the Evidence and an Overview of the Available Guidelines. Bone 2020, 131, 115112. [Google Scholar] [CrossRef]
- Dawson-Hughes, B.; Nelson, J.; Pittas, A.G. Intratrial Exposure to Vitamin D and New-Onset Diabetes Among Adults With Prediabetes: A Secondary Analysis From the Vitamin D and Type 2 Diabetes (D2d) Study. Diabetes Care 2021, 44, e106. [Google Scholar] [CrossRef]
- Barbáchano, A.; Fernández-Barral, A.; Ferrer-Mayorga, G.; Costales-Carrera, A.; Larriba, M.J.; Muñoz, A. The Endocrine Vitamin D System in the Gut. Mol. Cell Endocrinol. 2017, 453, 79–87. [Google Scholar] [CrossRef]
- Murai, I.H.; Fernandes, A.L.; Antonangelo, L.; Gualano, B.; Pereira, R.M.R. Effect of a Single High-Dose Vitamin D3 on the Length of Hospital Stay of Severely 25-Hydroxyvitamin D-Deficient Patients with COVID-19. Clinics 2021, 76, e3549. [Google Scholar] [CrossRef] [PubMed]
- Boucher, B.J. No Evidence That Vitamin D Is Able to Prevent or Affect the Severity of COVID-19 in Individuals with European Ancestry: A Mendelian Randomisation Study of Open Data, by Amin et Al. BMJ Nutr. Prev. Health 2021, 4, 352–353. [Google Scholar] [CrossRef] [PubMed]
- Manson, J.E.; Cook, N.R.; Lee, I.-M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Gordon, D.; Copeland, T.; D’Agostino, D.; et al. Vitamin D Supplements and Prevention of Cancer and Cardiovascular Disease. N. Engl. J. Med. 2019, 380, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Manson, J.E.; Bassuk, S.S.; Buring, J.E.; VITAL Research Group. Principal Results of the VITamin D and OmegA-3 TriaL (VITAL) and Updated Meta-Analyses of Relevant Vitamin D Trials. J. Steroid Biochem. Mol. Biol. 2020, 198, 105522. [Google Scholar] [CrossRef]
- Di Benedetto, F.; Di Sandro, S.; De Ruvo, N.; Berretta, M.; Masetti, M.; Montalti, R.; Ballarin, R.; Cocchi, S.; Potenza, L.; Luppi, M.; et al. Kaposi’s Sarcoma after Liver Transplantation. J. Cancer Res. Clin. Oncol. 2008, 134, 653–658. [Google Scholar] [CrossRef] [PubMed]
- Berretta, M.; Lleshi, A.; Cappellani, A.; Bearz, A.; Spina, M.; Talamini, R.; Cacopardo, B.; Nunnari, G.; Montesarchio, V.; Izzi, I.; et al. Oxaliplatin Based Chemotherapy and Concomitant Highly Active Antiretroviral Therapy in the Treatment of 24 Patients with Colorectal Cancer and HIV Infection. Curr. HIV Res. 2010, 8, 218–222. [Google Scholar] [CrossRef]
- Zanet, E.; Berretta, M.; Martellotta, F.; Cacopardo, B.; Fisichella, R.; Tavio, M.; Berretta, S.; Tirelli, U. Anal Cancer: Focus on HIV-Positive Patients in the HAART-Era. Curr. HIV Res. 2011, 9, 70–81. [Google Scholar] [CrossRef]
- Nunnari, G.; Coco, C.; Pinzone, M.R.; Pavone, P.; Berretta, M.; Di Rosa, M.; Schnell, M.; Calabrese, G.; Cacopardo, B. The Role of Micronutrients in the Diet of HIV-1-Infected Individuals. Front. Biosci. 2012, 4, 2442–2456. [Google Scholar] [CrossRef][Green Version]
- D’Arminio Monforte, A.; Diaz-Cuervo, H.; De Luca, A.; Maggiolo, F.; Cingolani, A.; Bonora, S.; Castagna, A.; Girardi, E.; Antinori, A.; Lo Caputo, S.; et al. Evolution of Major Non-HIV-Related Comorbidities in HIV-Infected Patients in the Italian Cohort of Individuals, Naïve for Antiretrovirals (ICONA) Foundation Study Cohort in the Period 2004–2014. HIV Med. 2019, 20, 99–109. [Google Scholar] [CrossRef]
| 25-Hydroxylase Activity | |||
|---|---|---|---|
| CYP Enzyme | Hydroxylate Function | Location/Expression | Note |
| CYP27A1 | Does not 25-hydroxylate Vitamin D2 | Liver, mitochondrial 25-hydroxylase, widely distributed throughout the body | Mouse mutation: increase in 25OHD blood level [21]. Human: inactivating mutations cause cerebrotendinous xanthomatosis, abnormal bile and cholesterol metabolism, but not rickets [34]. |
| CYP2R1 | 25-hydroxylates both vitamin D2 and D3 | Microsomal fraction of mouse liver [35], expression in liver and testes | CYP27A1 null mouse: increase CYP2R1 expression, hence increased blood levels of 25OHD. CYP2R1 null mouse: 50% decrease in 25OHD blood level [36]. Human: leu99pro mutation caused severe bone disease, rickets, and low 25OHD [37]. |
| CYP3A4 | Prefers 1αOHD to 25OHD as substrate | Liver and intestine | Is a major drug-metabolizing enzyme |
| CYP2J3 | More 25-hydroxylase activity than CYP2J2 | Rat liver | More 25-hydroxylase activity than its human homolog (CYP2J2) |
| CYP2J2 | Less 25-hydroxylase activity than CYP2J3 | Primarily in the heart | It is the human homolog of CYP2J3, functioning mainly as an arachidonic acid epooxygenase [38]. |
| CYP2D25 | Does not have substantial 25-hydroxylase activity [34] | Its human homolog was initially isolated from pig livers and kidneys [39] | - |
| CYP2C11 | 25-hydroxylase activity for vitamin D3 and D2 and 1OHD analogs | Expression in livers of male rats | Known for hydroxylation of testosterone [40]. |
| 1α-hydroxylase | |||
| CYP27B1 | The only enzyme with 25OHD 1α-hydroxylase ativity | Kidney (mainly), other tissues such as epithelial cells in the lungs, breast, skin, intestine, prostate; parathyroid gland), pancreatic islets, thyroid, testes, ovary, and placenta; macrophages, and T and B lymphocytes and dendritic cells (DCs); osteoblasts and chondrocytes [37] | Gene mutation leads to pseudovitamin D deficiency caused by inadequate 1,25(OH)2D production [41,42]. Parathyroid hormone, fibroblast growth factor 23, and 1,25(OH)2D play important roles in 1α-hydroxylase regulation:
|
| 24-hydroxylase | |||
| CYP24A1 | The only enzyme with 24-hydroxylase ativity | Kidneys (mainly) | With both 24-hydroxylase and 23-hydroxylase activity, CYP24A1 mutation leads to removal of all 24-hydroxylated metabolites of vitamin D; therefore, it can cause defective mineralization of intramembranous (not endochondral) bone [44]. It is a marker of 1,25(OH)2D response in cells that express it. Main role of CYP24A1 is to regulate levels of 1,25(OH)2D in tissues. Moreover, a number of malignancies have elevated CYP24A1 expression [45]. In tumours, its inhibition can increase 1,25(OH)2D levels, leading to antiproliferative/prodifferentiating [46]. |
| 3-epimerase (3-epi) | |||
| - | It produces the 3- epi form of 1,25(OH)2D in keratinocyte [47] | Keratinocyte, hepatocyte-derived cells, parathyroid cells, colon cancer cells, osteo-blasts, but not in the kidneys | The enzyme has not been purified and sequenced yet. It therefore remains unclear whether one gene product is involved. |
| 20-hydroxylation | |||
| CYP11A1 | 20-hydroxylation of vitamin D | Keratinocyte | The product, 20-hydroxylation, or its metabolite, 20,23(OH)2D, appear to have similar activity to 1,25(OH)2D, although not for all functions [23]. |
| Drug/Susbance | Action | Clinical Action | Interaction |
|---|---|---|---|
| Erdafitinib | The mechanism appears to be related to the pharmacodynamic effects of fibroblast growth factor receptor (FGFR/aKlotho) inhibition by erdafitinib. | Minimizes risk of hyperphosphatemia; it is recommended to restrict phosphate intake to 600–800 mg daily | Major |
| Ergocalciferol and Vit D3 derivates | Additional effects result in toxicity, manifesting as hypercalcemia, hypercalciuria, and hyperphosphatemia. | In hypercalcemia, vitamin D and any calcium supplements should be immediately stopped | Major |
| Oxcarbazepin | It induces CYP450 inducers. It may decrease the pharmacologic effects of vitamin D analogs, inducing the hepatic conversion of vitamin D to inactive metabolites. | Patients who metabolize CYP450 poorly must be supplemented with double doses of Vit D when receiving oxicarbazepin. | Moderate |
| Magnesium salts and products | Possibly increases plasma hypermagnesemia, particularly in chronic renal dialysis patients, due to potentially additive pharmacologic effects. Chronic hypermagnesemia may have a role in the pathogenesis of adynamic bone disease in dialysis patients. | Patients on chronic renal dialysis treated with a vitamin D analog should avoid magnesium-containing products | Moderate |
| Indapamide and other thiazide diuretics | Thiazide diuretics inhibit the renal excretion of calcium and may also enhance the responsiveness of bone and renal tubule to parathyroid hormone.Thus, the concurrent use of large amounts of calcium or vitamin D can lead to excessively high plasma levels of calcium. | Serum calcium should be monitored if patients experience signs of hypercalcemia, such as dizziness, weakness, lethargy, headache, myalgia, anorexia, nausea, vomiting, and seizures | Moderate |
| Restriction Sites | PCR Amplicon | Allelic Variants | Fragments Post-Digestion (bp) |
|---|---|---|---|
| ApaI | 745 bp | A/A | 745 |
| A/a | 745 + 528 + 218 | ||
| a/a | 528 + 218 | ||
| TaqI | 745 bp | T/T | 745 |
| T/t | 745 + 293 + 251 + 201 | ||
| t/t | 293 + 251 + 201 | ||
| BmsI | 822 bp | B/B | 822 |
| B/b | 822 + 646 + 176 | ||
| b/b | 646 + 176 | ||
| FokI | 267 bp | f/f | 267 |
| F/f | 267 + 197 + 70 | ||
| F/F | 197 + 70 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berretta, M.; Quagliariello, V.; Bignucolo, A.; Facchini, S.; Maurea, N.; Di Francia, R.; Fiorica, F.; Sharifi, S.; Bressan, S.; Richter, S.N.; et al. The Multiple Effects of Vitamin D against Chronic Diseases: From Reduction of Lipid Peroxidation to Updated Evidence from Clinical Studies. Antioxidants 2022, 11, 1090. https://doi.org/10.3390/antiox11061090
Berretta M, Quagliariello V, Bignucolo A, Facchini S, Maurea N, Di Francia R, Fiorica F, Sharifi S, Bressan S, Richter SN, et al. The Multiple Effects of Vitamin D against Chronic Diseases: From Reduction of Lipid Peroxidation to Updated Evidence from Clinical Studies. Antioxidants. 2022; 11(6):1090. https://doi.org/10.3390/antiox11061090
Chicago/Turabian StyleBerretta, Massimiliano, Vincenzo Quagliariello, Alessia Bignucolo, Sergio Facchini, Nicola Maurea, Raffaele Di Francia, Francesco Fiorica, Saman Sharifi, Silvia Bressan, Sara N. Richter, and et al. 2022. "The Multiple Effects of Vitamin D against Chronic Diseases: From Reduction of Lipid Peroxidation to Updated Evidence from Clinical Studies" Antioxidants 11, no. 6: 1090. https://doi.org/10.3390/antiox11061090
APA StyleBerretta, M., Quagliariello, V., Bignucolo, A., Facchini, S., Maurea, N., Di Francia, R., Fiorica, F., Sharifi, S., Bressan, S., Richter, S. N., Camozzi, V., Rinaldi, L., Scaroni, C., & Montopoli, M. (2022). The Multiple Effects of Vitamin D against Chronic Diseases: From Reduction of Lipid Peroxidation to Updated Evidence from Clinical Studies. Antioxidants, 11(6), 1090. https://doi.org/10.3390/antiox11061090









