Anti-Inflammatory, Anti-Apoptotic, and Antioxidant Roles of Honey, Royal Jelly, and Propolis in Suppressing Nephrotoxicity Induced by Doxorubicin in Male Albino Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Natural Products
2.2. Experimental Animals
2.3. Induction of Nephrotoxicity
2.4. Experimental Design
2.5. Biochemical Determinations
2.6. Cell Examination of Kidney Tissues
2.7. Immuno-Histochemical Study (Caspase-3)
2.8. Gene Expression Analysis
2.9. Statistical Analysis
3. Results
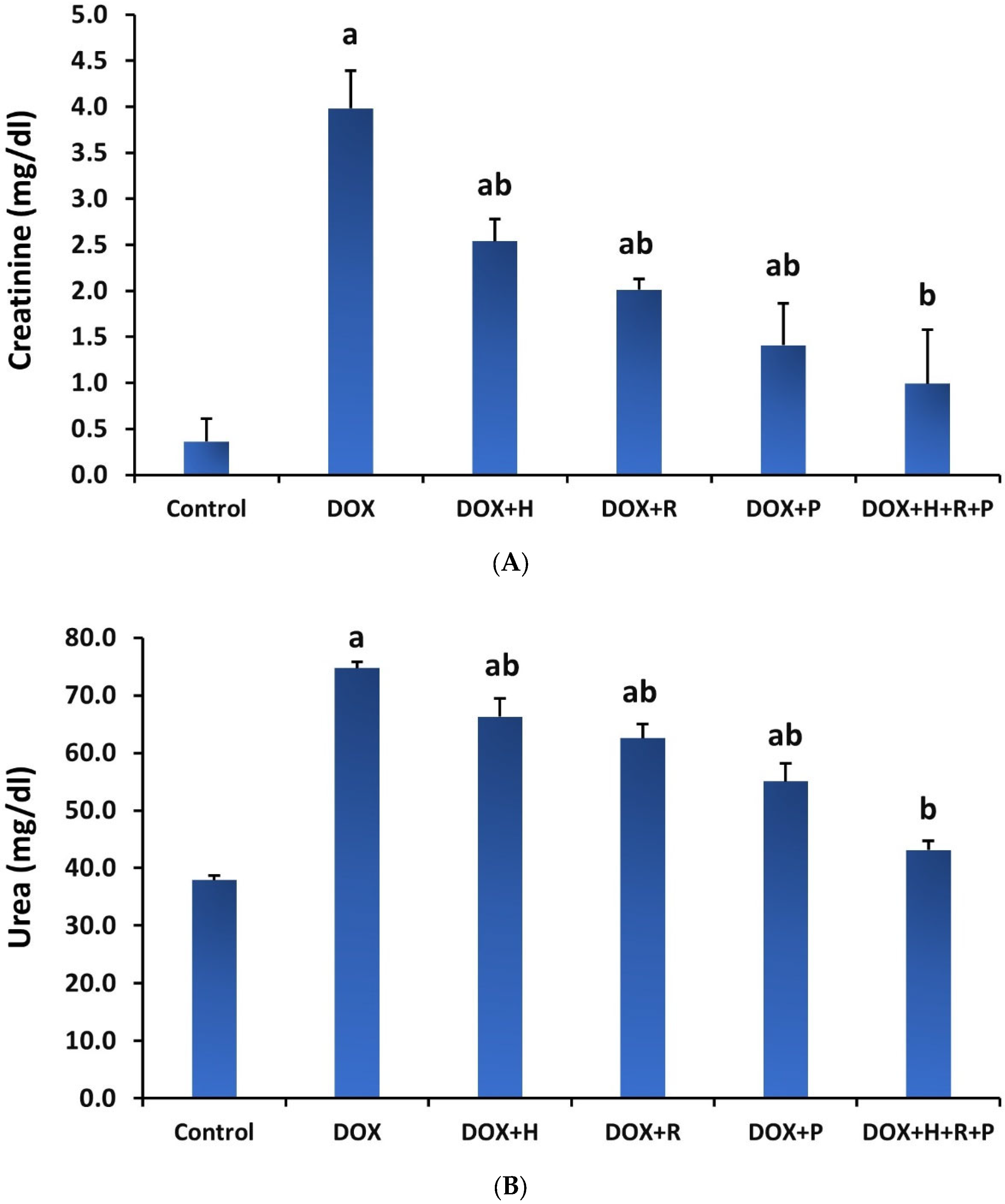
3.1. Renal Biomarkers
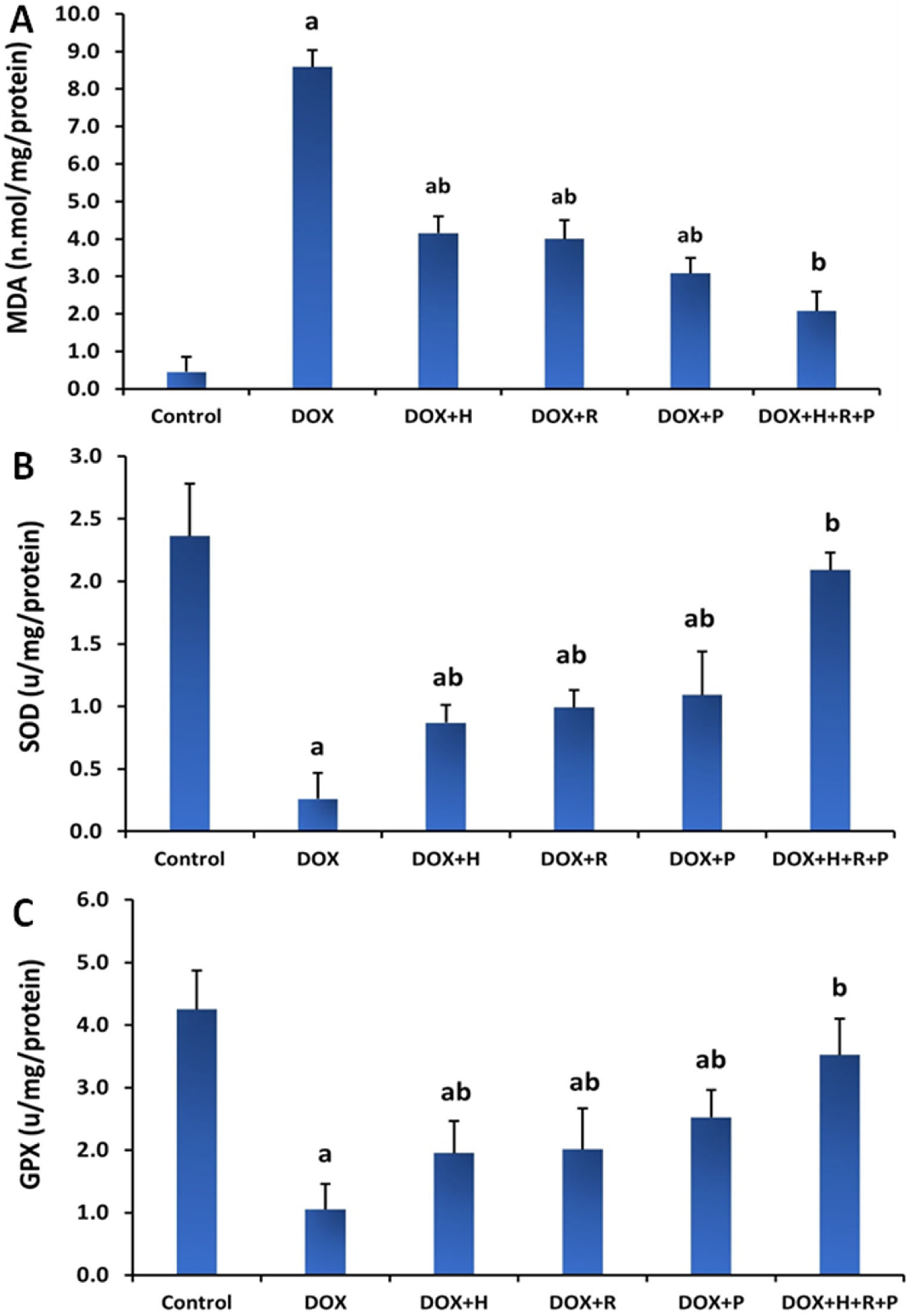
3.2. Antioxidants Status
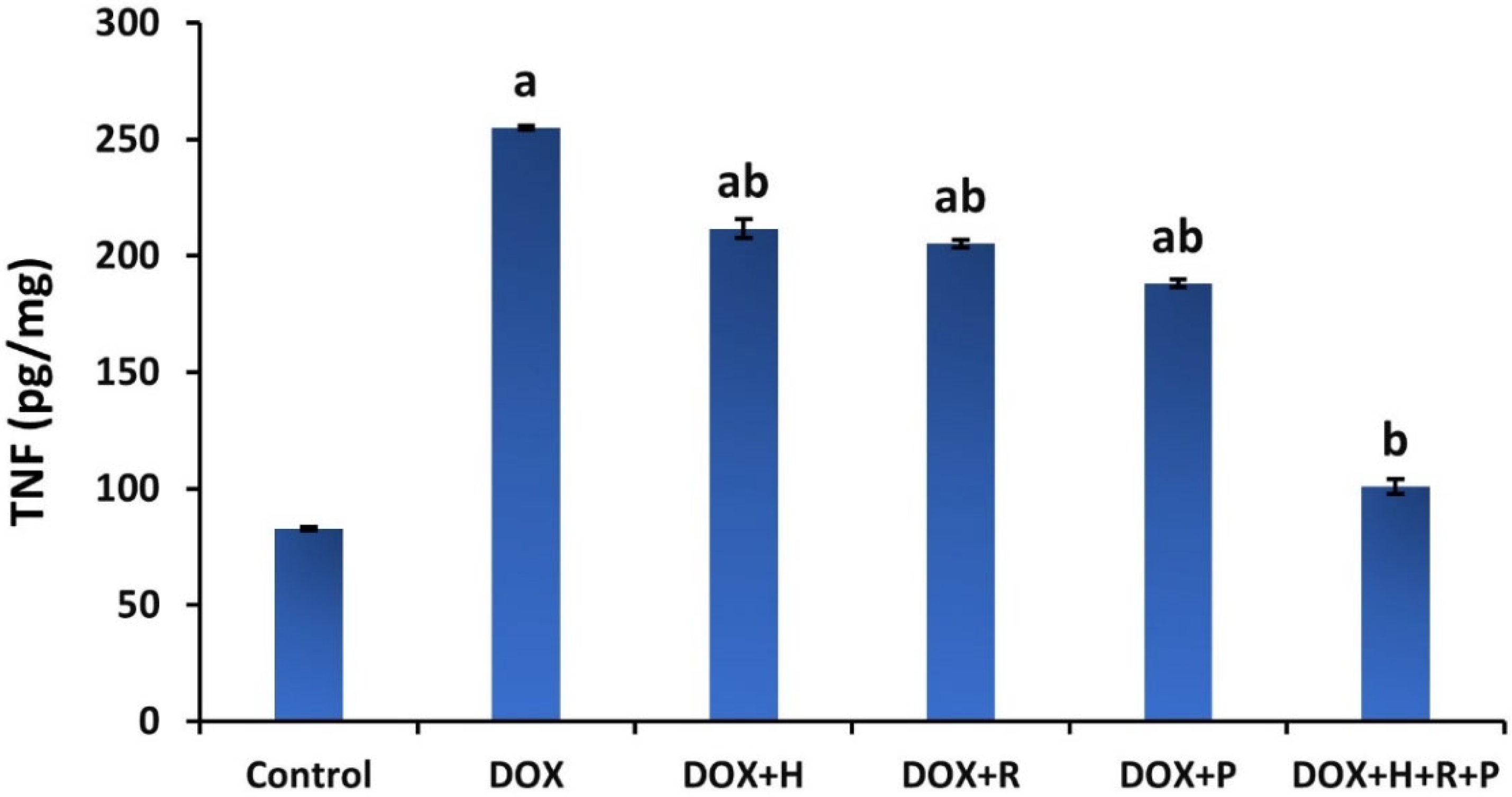
3.3. Inflammatory Markers
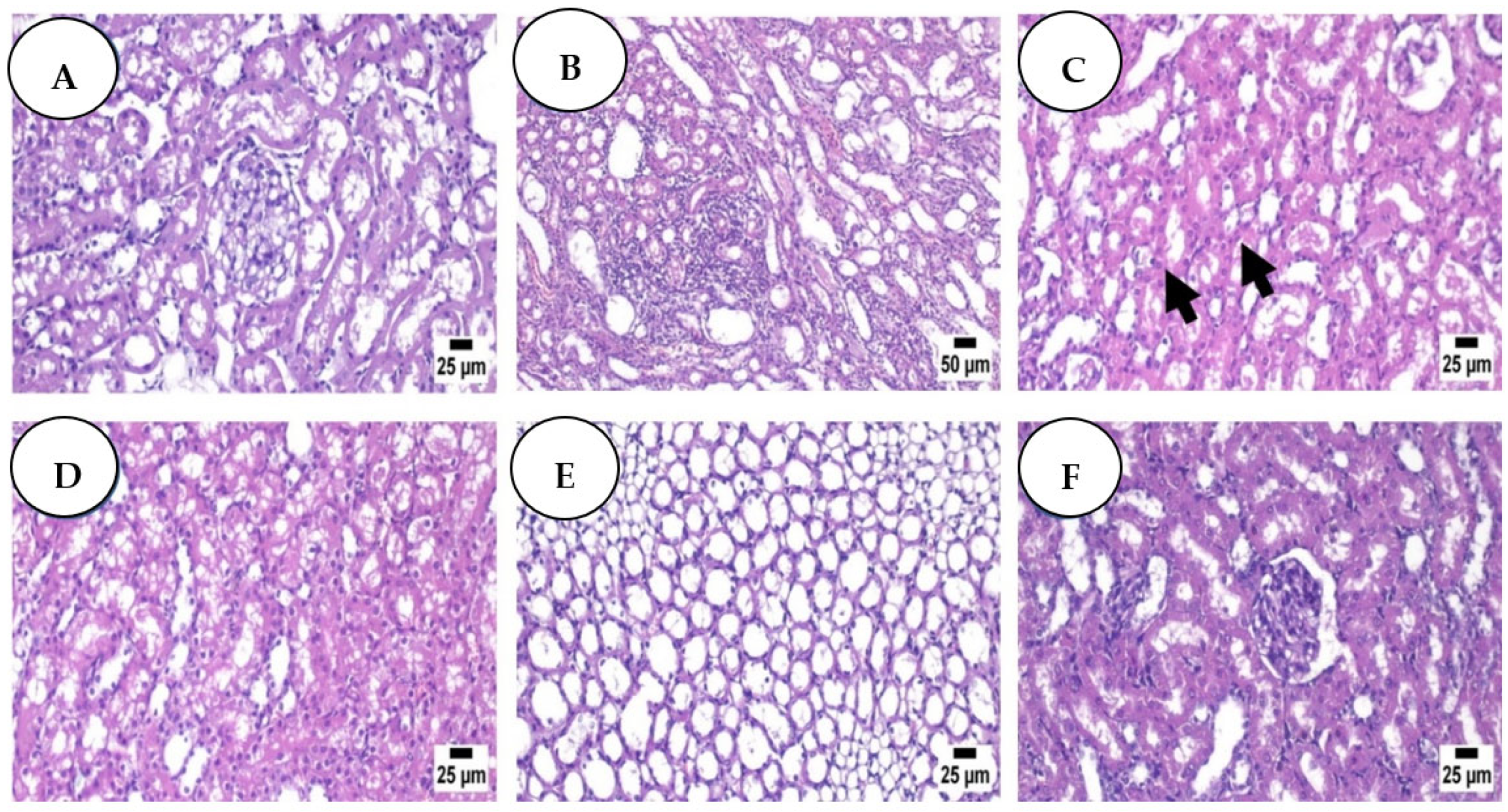
3.4. Histopathological Analysis: Haematoxylin and Eosin
3.5. Immuno-Histochemical Studies: Caspase 3
3.6. Gene Expression Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Refaie, M.M.; Amin, E.F.; El-Tahawy, N.F.; Abdelrahman, A.M. Possible protective effect of diacerein on doxorubicin-induced nephrotoxicity in rats. J. Toxicol. 2016, 2016, 9507563. [Google Scholar] [CrossRef] [PubMed]
- Hekmat, A.S.; Chenari, A.; Alipanah, H.; Javanmardi, K. Protective effect of alamandine on doxorubicin-induced nephrotoxicity in rats. BMC Pharm. Toxicol. 2021, 22, 31. [Google Scholar]
- Özgermen, B.; Bulut, G.; Alpaslan Pinarli, F.; Gültekin, S.; Özen, D.; Yavuz, O.; Haydardedeoglu, A.E. Investigation of the effects of fetal rat kidney-derived mesenchymal stem cells implementation on doxorubicin-induced nephropathy in male Sprague–Dawley rats. Ank. Univ. Vet. Fak. Derg. 2022, 69, 201–209. [Google Scholar] [CrossRef]
- Saad, S.Y.; Najjar, T.A.; Al-Rýkabý, A.C. The preventive role of deferoxamine against acute doxorubicin-induced cardiac, renal and hepatic toxicity in rats. Pharmacol. Res. 2001, 43, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Khodeer, D.M.; Mehanna, E.T.; Abushouk, A.I.; Abdel-Daim, M.M. Protective Effects of Evening Primrose Oil against Cyclophosphamide-Induced Biochemical, Histopathological, and Genotoxic Alterations in Mice. Pathogens 2020, 9, 98. [Google Scholar] [CrossRef]
- Rafiee, Z.; Moaiedi, M.Z.; Gorji, A.V.; Mansouri, E. p-Coumaric Acid Mitigates Doxorubicin-Induced Nephrotoxicity Through Suppression of Oxidative Stress, Inflammation and Apoptosis. Arch. Med. Res. 2020, 51, 32–40. [Google Scholar] [CrossRef]
- Saad, Z.A.; Khodeer, D.M.; Zaitone, S.A.; Ahmed, A.A.M.; Moustafa, Y.M. Exenatide Ameliorates Experimental Non-Alcoholic Fatty Liver in Rats via Suppression of Toll-like Receptor 4/NFκB Signaling: Comparison to Metformin. Life Sci. 2020, 253, 117725. [Google Scholar] [CrossRef]
- Afsar, T.; Razak, S.; Almajwal, A.; Al-Disi, D. Doxorubicin-induced alterations in kidney functioning, oxidative stress, DNA damage, and renal tissue morphology; Improvement by Acacia hydaspica tannin-rich ethyl acetate fraction. Saudi J. Biol. Sci. 2020, 27, 2251–2260. [Google Scholar] [CrossRef]
- Sami, M.M.; Ali, E.A.; Galhom, R.A.; Youssef, A.M.; Mohammad, H.M. Boswellic acids ameliorate doxorubicin-induced nephrotoxicity in mice: A focus on antioxidant and anti-apoptotic effects. Egypt. J. Basic Appl. Sci. 2019, 6, 10–24. [Google Scholar] [CrossRef]
- Deaval, D.G.; Martin, E.A.; Horner, J.M. Drug-induced oxidative stress and toxicity. J. Toxicol. 2012, 21, 645460. [Google Scholar] [CrossRef]
- Crook, M.A. Clinical Biochemistry and Metabolic Medicine, 8th ed.; Hodder and Stoughton Ltd.: London, UK, 2012. [Google Scholar]
- El-Rahman, G.I.A.; Behairy, A.; Elseddawy, N.M.; Batiha, G.E.-S.; Hozzein, W.N.; Khodeer, D.M.; Abd-Elhakim, Y.M. Saussurea Lappa Ethanolic Extract Attenuates Triamcinolone Acetonide-Induced Pulmonary and Splenic Tissue Damage in Rats via Modulation of Oxidative Stress, Inflammation, and Apoptosis. Antioxidants 2020, 9, 396. [Google Scholar] [CrossRef] [PubMed]
- Montaser, M.; Ebiya, R.A.; Afifi, M.; Saddick, S.; Allogmani, A.S.; Almaghrabi, O.A. Effect of natural and synthetic food colorants on spermatogenesis and the expression of its controlling genes. Slov. Vet. Res. 2018, 55, 187–199. [Google Scholar] [CrossRef]
- Madbouly, S.; Ebiya, R. Histopathological and molecular assessment of moringa and sesame oil as protective potential in gentamicin induced nephrotoxicity. Cienc. Tec. Vitivinic. 2018, 33, 248–267. [Google Scholar]
- Zhang, X.; Yeung, E.D.; Wang, J.; Panzhinskiy, E.E.; Tong, C.; Li, W.; Li, J. Isoliquiritigenin, a natural antioxidant, selectively inhibits the proliferation of prostate cancer cells. Clin. Exp. Pharmacol. Physiol. 2010, 4, 841–847. [Google Scholar] [CrossRef] [PubMed]
- Atrooz, O.M.; Al-Sabayleh, M.A.; Al-Abbadi, S.Y. Studies on physical and chemical analysis of various honey samples and their antioxidant activities. J. Biol. Sci. 2008, 8, 1338–1342. [Google Scholar] [CrossRef]
- Ahmed, S.; Othman, N.H. Honey as a potential natural anti-cancer agent: A review of its mechanisms. Evid.-Based Complement. Altern. Med. 2013, 2013, 829070. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, A.P.; John, A.A.; Vellayappan, M.V.; Balaji, A.; Jaganathan, S.K.; Mandal, M.; Supriyanto, E. Honey and its phytochemicals: Plausible agents in combating colon cancer through its diversified actions. J. Food Biochem. 2016, 40, 613–629. [Google Scholar] [CrossRef]
- Pasupuleti, V.; Sammugam, L.; Ramesh, N.; Gan, S.H. Honey, Propolis, and royal jelly: A comprehensive review of their biological actions and health benefits. Oxid. Med. Cell Longev. 2017, 2017, 21. [Google Scholar] [CrossRef]
- Abeshu, M.A.; Geleta, B. Medicinal uses of honey. Biol. Med. 2016, 8, 1–7. [Google Scholar]
- Alvarez-Suarez, J.M.; Giampieri, F.; Battino, M. Honey as a source of dietary antioxidants: Structures, bioavailability, and evidence of protective effects against human chronic diseases. Curr. Med. Chem. 2013, 20, 621–638. [Google Scholar] [CrossRef]
- Alhumaydhi, F.A. Biochemical studies on the protective effect of honey against doxorubicin-induced toxicity in BALB/C mice. Int. J. Health Sci. 2020, 14, 31. [Google Scholar]
- Ibrahim, R.S.; El-Banna, A.A. Royal jelly fatty acids bioprofiling using TLC-MS and digital image analysis coupled with chemometrics and non-parametric regression for discovering efficient biomarkers against melanoma. RSC Adv. 2021, 11, 18717. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y. Royal Jelly Reduces Cholesterol Levels, Ameliorates Abeta Pathology and Enhances Neuronal Metabolic Activities in a Rabbit Model of Alzheimer’s Disease. Front. Aging Neurosci. 2018, 10, 50. [Google Scholar] [CrossRef] [PubMed]
- Kunugi, H.; Ali, M.A. Royal Jelly and Its Components Promote Healthy Aging and Longevity: From Animal Models to Humans. J. Mol. Sci. 2019, 20, 4662. [Google Scholar] [CrossRef] [PubMed]
- Bhadauria, M. Propolis prevents hepatorenal injury induced by chronic exposure to carbon tetrachloride. Evid.-Based Complement. Altern. Med. 2012, 2012, 235358. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, E.K.; Osman, A.A.; Moghazy, A.M.; Rahman, A.; AS, A. Propolis Protective Effects Against Doxorubicin-Induced Multi-Organ Toxicity via Suppression of Oxidative Stress, Inflammation, Apoptosis, and Histopathological Alterations in Female Albino Rats. Biointerface Res. Appl. Chem. 2021, 12, 1762–1777. [Google Scholar]
- Zhao, X.; Zhang, J.; Tong, N.; Chen, Y.; Luo, Y. Protective effects of berberine on doxorubicin-induced hepatotoxicity in mice. Biol. Pharm. Bull. 2012, 35, 796–800. [Google Scholar] [CrossRef]
- Fawcett, J.K.; Scott, J.E. A rapid and precise method for the determination of urea. J. Clin. Pathol. 1960, 13, 156–159. [Google Scholar] [CrossRef]
- Schirmeister, J.; Willmann, H.; Kiefer, H. Plasma Creatinine as Rough Indicator of Renal Function. Dtsch. Med. Wochenschr. 1964, 89, 1940. [Google Scholar]
- Satoh, K. Serum lipid peroxide in cerebrovascular disorders determined by a new colorimetric method. Clin. Chim. Acta 1978, 90, 37. [Google Scholar]
- Ohkawa, H.; Ohishi, W.; Yagi, K. Anal Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351. [Google Scholar] [CrossRef]
- Nishikimi, M.; Roa, N.A.; Yogi, K. The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochem. Biophys. Res. Commun. 1972, 46, 849–854. [Google Scholar] [CrossRef]
- Paglia, D.E.; Valentine, W.N. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 1967, 70, 158–169. [Google Scholar] [PubMed]
- Taylor, P.C. Anti-TNF therapy for rheumatoid arthritis and other inflammatory diseases. Mol. Biotechnol. 2001, 19, 153–168. [Google Scholar] [CrossRef]
- Bancroft, J.; Gamble, M. Theory and Practice of Histological Techniques; Churchill Livingstone Elsevier: London, UK, 2002. [Google Scholar]
- Wang, L.; Liu, X.; Chen, H.; Chen, Z.; Weng, X.; Qiu, T.; Liu, L. Effect of picroside II on apoptosis induced by renal ischemia/reperfusion injury in rats. Exp. Ther. Med. 2015, 9, 817–822. [Google Scholar] [CrossRef]
- Ebiya, R.A.; Montaser, M.M.; Darwish, S.M. Downregulated StAR gene and male reproductive dysfunction caused by nifedipine and ethosuximide. J. Basic Appl. Zool. 2016, 76, 42–51. [Google Scholar] [CrossRef]
- Montaser, M.; Mahmoud, S.; Mahdi, E.; El-Hallous, E.; Ebiya, R.A. Molecular and histopathological assessment of Arabic Coffee effect on fertility in Male Albino Rats. Res. J. Pharm. Biol. Chem. Sci. 2017, 8, 666–677. [Google Scholar]
- Ruggiero, A.; Ferrara, P.; Attinà, G.; Rizzo, D.; Riccardi, R. Renal toxicity and chemotherapy in children with cancer. Brit. J. Clin. Pharmacol. 2017, 83, 2605–2614. [Google Scholar] [CrossRef]
- Abdelmeguid, N.E.; Chmaisse, H.N.; Zeinab, N.A. Protective effect of silymarin on cisplatin-induced nephrotoxicity in rats. Pak. J. Nutr. 2010, 9, 624–636. [Google Scholar] [CrossRef][Green Version]
- Khan, N.; Sultana, S. Abrogation of potassium bromate induced renal oxidative stress and subsequent cell proliferation response by soy isoflavones in Wistar rats. Toxicology 2004, 201, 173–184. [Google Scholar] [CrossRef]
- Razaa, Z.; Naureen, Z. Melatonin ameliorates the drug induced nephrotoxicity: Molecular insights. Nefrologia 2020, 40, 12–25. [Google Scholar] [CrossRef] [PubMed]
- Hamad, R.; Jayakumar, C.; Ranganathan, P.; Mohamed, R.; El-Hamamy, M.M.; Dessouki, A.A.; Ibrahim, A.; Ramesh, G. Honey feeding protects kidney against cisplatin nephrotoxicity through suppression of inflammation. Clin. Exp. Pharm. Physiol. 2015, 42, 843–848. [Google Scholar] [CrossRef] [PubMed]
- Oktem, G.; Ayla, S.; Seckin, I.; Tanriverdi, G.; Cengiz, M.; Eser, M. Doxorubicin induced nephrotoxicity: Protective effect of nicotinamide. Int. J. Cell Biol. 2011, 2011, 390238. [Google Scholar]
- Bhalchandra, W.; Alqadhi, Y.A. Administration of Honey and Royal Jelly Ameliorate Cisplatin-Induced Changes in Liver and Kidney Function in Rat. Biomed. Pharmacol. J. 2018, 11, 2191–2199. [Google Scholar]
- Al-Yahya, M.; Mothana, R.; Al-Said, M.; Al-Dosari, M.; Al-Musayeib, N.; Al-Sohaibani, M.; Parvez, M.K.; Rafatullah, S. Attenuation of CCl4-Induced Oxidative Stress and Hepatonephrotoxicity by Saudi Sidr Honeyin Rats. Evid.-Based Complement. Altern. Med. 2013, 10, 569037. [Google Scholar]
- Baykara, M.; Silici, S.; Özçelik, M.; Güler, O.; Erdoğan, N.; Bilgen, M. In vivo nephroprotective efficacy of Propolis against contrast induced nephropathy. Diagn. Interv. Radiol. 2015, 21, 317. [Google Scholar] [CrossRef]
- Promsan, S.; Jaikumkao, K.; Pongchaidecha, A.; Chattipakorn, N.; Chatsudthipong, V.; Arjinajarn, P.; Lungkaphin, A. Pinocembrin attenuates gentamicin-induced nephrotoxicity in rats. Can. J. Physiol. Pharmacol. 2016, 94, 808–818. [Google Scholar] [CrossRef]
- Cauich-Kumul, R.; Campos, M.R. Bee Propolis: Properties, Chemical Composition, Applications, and Potential Health Effects. In Bioactive Compounds; Woodhead Publishing: Sawston, UK, 2019; pp. 227–243. [Google Scholar]
- El-Sheikh, A.A.; Morsy, M.A.; Mahmoud, M.M.; Rifaai, R.A.; Abdelrahman, A.M. Effect of coenzyme-Q10 on doxorubicin-induced nephrotoxicity in rats. Adv. Pharm. Sci. 2012, 2012, 981461. [Google Scholar] [CrossRef]
- Balli, E.; Mete, U.O.; Tuli, A. Effect of melatonin on the cardiotoxicity of Doxorubicin. Histol. Histopathol. 2004, 19, 1101–1108. [Google Scholar]
- Al-Waili, N.S. Identification of nitric oxide metabolites in various honeys: Effects of intravenous honey on plasma and urinary nitric oxide metabolites concentrations. J. Med. Food 2003, 6, 359–364. [Google Scholar] [CrossRef]
- Al-Seeni, M.N.; El Rabey, H.A.; Al-Solamy, S.M. The protective role of bee honey against the toxic effect of melamine in the male rat kidney. Toxicol. Ind. Health 2015, 31, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.P.; Zaidi, A.I.A.; Raisuddin, S.; Saxena, A.K.; Murthy, R.C.; Ray, P.K. Effect of zink on immune functions and host resistance against infectionand tumor challence. Immunopharmacol. Immunotoxicol. 1992, 14, 813–840. [Google Scholar] [CrossRef] [PubMed]
- Geiger, S.; Lange, V.; Suhl, P.; Heinemann, V.; Stemmler, H.J. Anti-cancer therapy induced cardiotoxicity: Review of thl literature. Anti-Cancer Drugs 2010, 21, 578–590. [Google Scholar] [CrossRef] [PubMed]
- Erejuwa, O.O.; Sulaiman, S.A.; Ab Wahab, M.S. Honey: A novel antioxidant. Molecules 2012, 17, 4400–4423. [Google Scholar] [CrossRef]
- Yassin, S.A.; Abdel-Moneim, A.; Abdel-Reheim, E.S. Protective Effect of Propolis on Doxorubicin Induced Cardio- and Nephrotoxicity. Ann. Cardiol. Vasc. Med. 2020, 3, 1028. [Google Scholar]
- Neale, T.J.; Rüger, B.; Macaulay, H.; Dunbar, P.R.; Hasan, Q.; Bourke, A. Tumor necrosis factor-alpha is expressed by glomerular visceral epithelial cells in human membranous nephropathy. Am. J. Pathol. 1995, 146, 1444–1454. [Google Scholar]
- Hosseinian, S.; Khajavi, R.A.; Ebrahimzadeh, A.; Soukhtanloo, M.; Sadeghnia, H.R.; Shafei, M.N. Thymoquinone ameliorates renal damage in unilateral ureteral obstruction in rats. Pharmacol. Rep. 2017, 69, 648–657. [Google Scholar] [CrossRef]
- Al-Saedi, H.F.; Al-Zubaidy, A.A.; Khattab, Y.I. Thepossible effects of montelukast against doxorubicin-induced nephrotoxicity in rabbits. Int. J. Adv. Res. 2014, 2, 723–729. [Google Scholar]
- Chien, S.; Wu, Y.; Chen, Z.; Yang, W. Naturally occurring anthraquinones: Chemistry and therapeutic potential in autoimmune diabetes. Evid.-Based Complement. Altern. Med. 2015, 2015, 357357. [Google Scholar] [CrossRef]
- Thi Lan Nguyen, H.; Panyoyai, N.; Kasapis, S.; Pang, E.; Mantri, N. Honey and Its Role in Relieving Multiple Facets of Atherosclerosis. Nutrients 2019, 11, 167. [Google Scholar] [CrossRef]
- Ahmad, A.; Khan, R.A.; Mesaik, M.A. Anti-inflammatory effect of natural honey on bovine thrombin-induced oxidative burst in phagocytes. Phytother. Res. 2009, 23, 801–808. [Google Scholar] [CrossRef] [PubMed]
- Kassim, M.; Yusoff, K.M.; Ong, G.; Sekaran, S.; Yusof, M.Y.B.M.; Mansor, M. Gelam honey inhibits lipopolysaccharide-induced endotoxemia in rats through the induction of heme oxygenase-1 and the inhibition of cytokines, nitric oxide, and high-mobility group protein B1. Fitoterapia 2012, 83, 1054–1059. [Google Scholar] [CrossRef] [PubMed]
- Rizk, S.M.; Zaki, H.F.; Mina, M.A.M. Propolis Attenuates Doxorubicin-Induced Testicular Toxicity in Rats. Food Chem. Toxicology 2014, 67, 176–186. [Google Scholar] [CrossRef] [PubMed]
- Köse, E.; Oğuz, F.; Vardi, N.; Sarihan, M.E.; Beytur, A.; Yücel, A.; Polat, A.; Ekïncï, N. Therapeutic and protective effects of montelukast against doxorubicininduced acute kidney damage in rats. Iran. J. Basic Med. Sci. 2019, 22, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Weng, M.S.; Ho, Y.S.; Lin, J.K. Chrysin induces G1 phase cell cycle arrest in C6 glioma cells through inducing p21Waf1/Cip1 expression: Involvement of p38 mitogen-activated protein kinase. Biochem. Pharmacol. 2005, 69, 1815–1827. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Katsuda, S.; Takahashi, H.; Koh, E. Fas-mediated apoptosis in adriamycin-induced cardiomyopathy in rats: In vivo study. Circulation 2000, 102, 572–578. [Google Scholar] [CrossRef]
- Rashid, S.; Ali, N.; Nafees, S.; Ahmad, S.T.; Arjumand, W.; Hasan, S.K.; Sultana, S. Alleviation of doxorubicin-induced nephrotoxicity and hepatotoxicity by chrysin in Wistar rats. Toxicol. Mech. Methods 2013, 23, 337–345. [Google Scholar] [CrossRef]
- Szliszka, E.; Krol, W. Polyphenols isolated from Propolis augment TRAILinduced apoptosis in cancer cells. Evid.-Based Complement. Altern. Med. 2013, 2013, 731940. [Google Scholar] [CrossRef]
- Krishnakumar, R.; Kraus, W.L. The PARP side of the nucleus: Molecular actions, physiological outcomes, and clinical targets. Mol. Cell 2010, 39, 8–24. [Google Scholar] [CrossRef]
- Ha, H.C.; Snyder, S.H. Poly (ADP-ribose) polymerase is a mediator of necrotic cell death by ATP depletion. Proc. Natl. Acad. Sci. USA 1999, 96, 13978–13982. [Google Scholar] [CrossRef]
- Szabó, C.; Dawson, V.L. Role of poly (ADP-ribose) synthetase in inflammation and ischaemia-reperfusion. Trends Pharm. Sci. 1998, 19, 287–298. [Google Scholar] [CrossRef]
- Jagtap, P.; Szabó, C. Poly(ADP-ribose) polymerase and the therapeutic effects of its inhibitors. Nat. Rev. Drug Discov. 2005, 4, 421–440. [Google Scholar] [CrossRef] [PubMed]
- Mamdouh, A.M.; Khodeer, D.M.; Tantawy, M.A.; Moustafa, Y.M. In-Vitro and in-Vivo Investigation of Amygdalin, Metformin, and Combination of Both against Doxorubicin on Hepatocellular Carcinoma. Life Sci. 2021, 285, 119961. [Google Scholar] [CrossRef] [PubMed]
- Hassen, M.T.; Mohamed, H.K.; Montaser, M.M.; El-Sharnouby, M.E.; Awad, N.; Ebiya, R.A. Molecular, Immunomodulatory, and Histopathological Role of Mesenchymal Stem Cells and Beetroot Extract on Cisplatin Induced Testicular Damage in Albino Rats. Animals 2021, 11, 1142. [Google Scholar] [CrossRef]
- Elkazzaz, S.K.; Khodeer, D.M.; El Fayoumi, H.M.; Moustafa, Y.M. Role of Sodium Glucose Cotransporter Type 2 Inhibitors Dapagliflozin on Diabetic Nephropathy in Rats; Inflammation, Angiogenesis and Apoptosis. Life Sci. 2021, 280, 119018. [Google Scholar] [CrossRef]
- Erejuwa, O.O.; Sulaiman, S.A.; Wahab, M.S.; Sirajudeen, K.N.S.; Salleh, M.M.; Gurtu, S. Antioxidant Protection of Malaysian Tualang Honey in Pancreas of Normal and Streptozotocininduced Diabetic Rats. In Annales d’Endocrinologie; Elsevier: Amsterdam, The Netherlands, 2010; pp. 291–296. [Google Scholar]
- Kocot, J.; Kiełczykowska, M.; Luchowska-Kocot, D.; Kurzepa, J.; Musik, I. Antioxidant Potential of Propolis, Bee Pollen, and Royal Jelly: Possible Medical Application. Oxid. Med. Cell. Longev. 2018, 2, 7074209. [Google Scholar] [CrossRef]
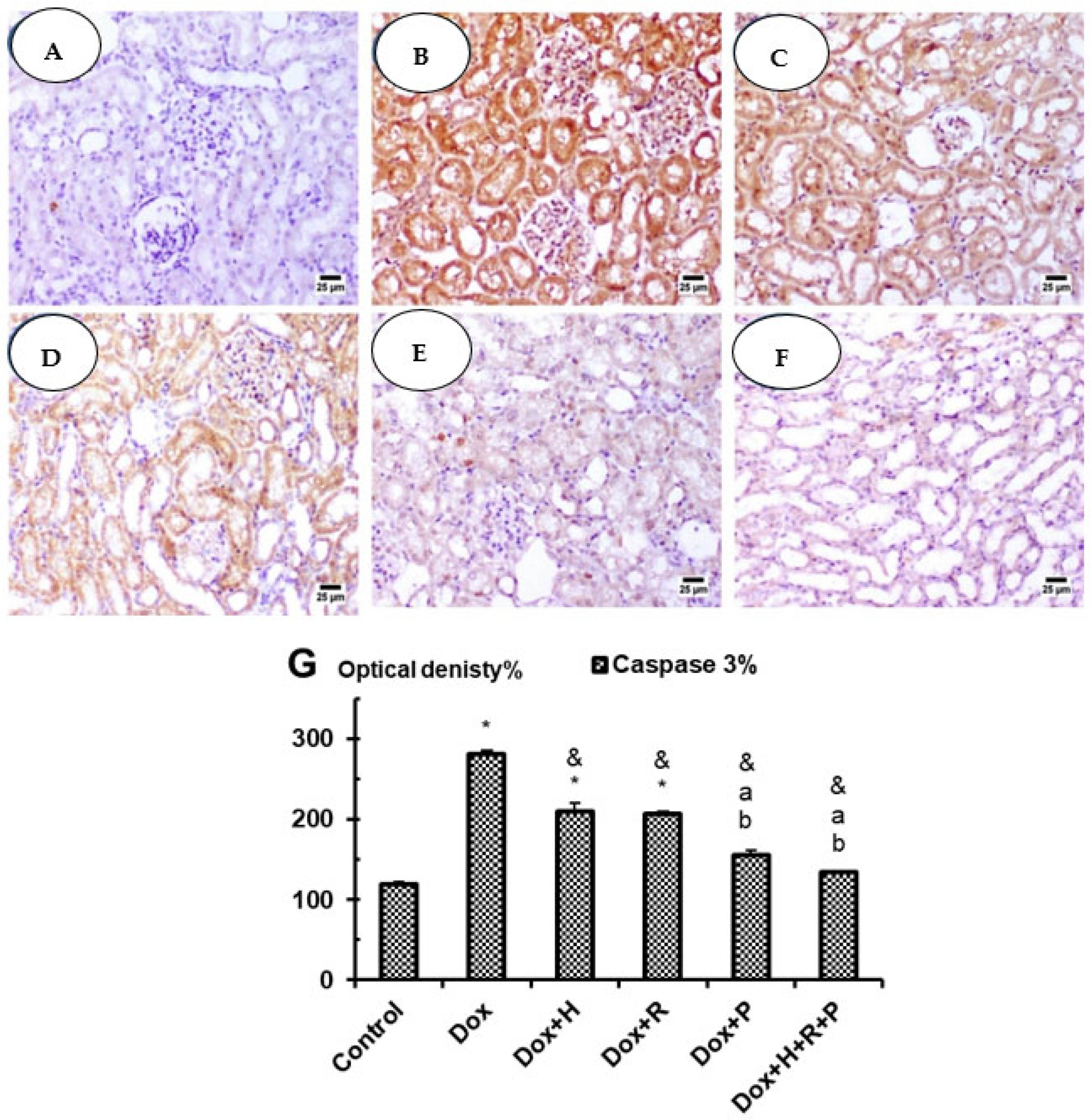
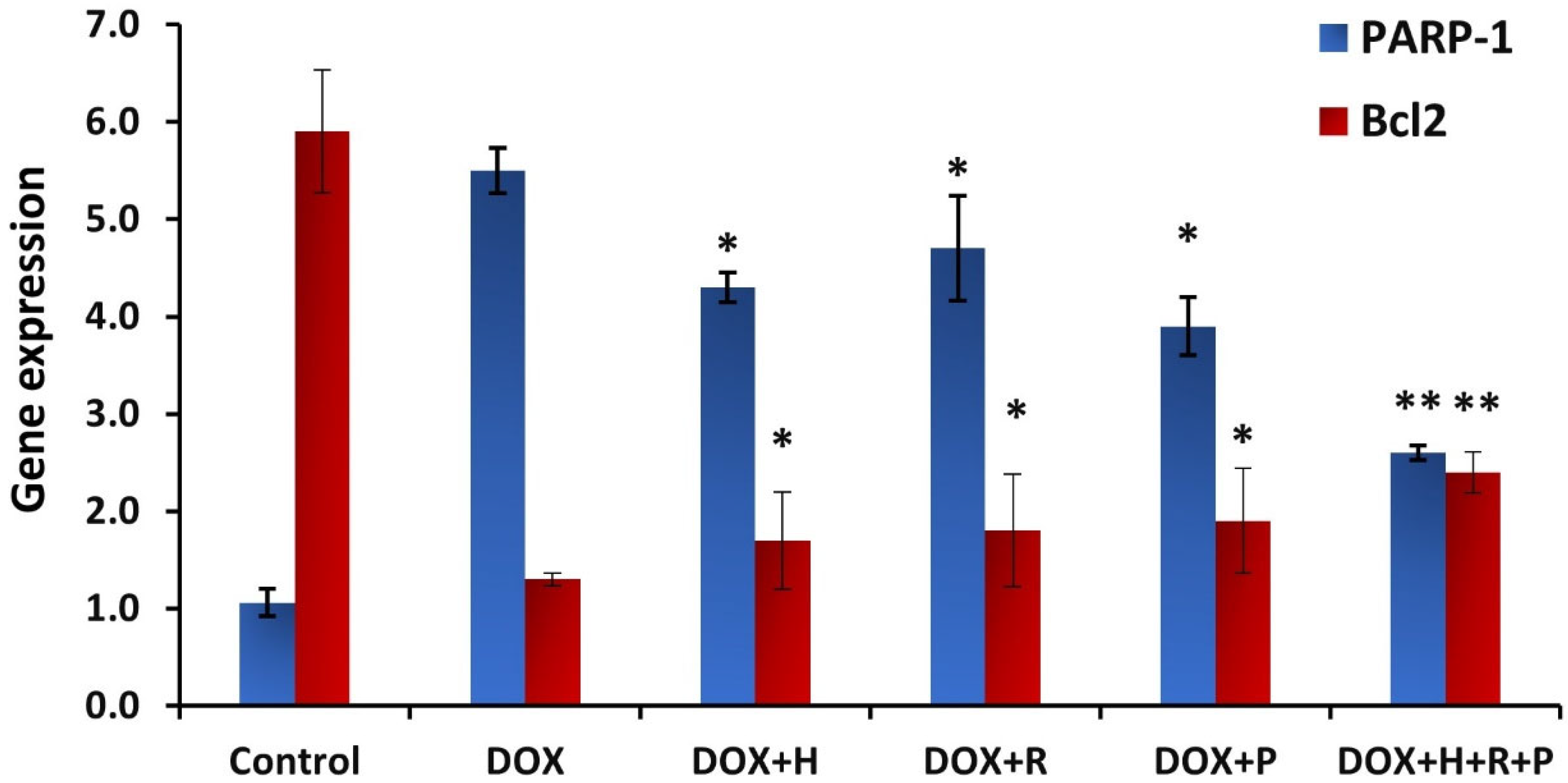
 Upregulation of the parameter.
Upregulation of the parameter.  Downlegulation of the parameter.
Downlegulation of the parameter.
 Upregulation of the parameter.
Upregulation of the parameter.  Downlegulation of the parameter.
Downlegulation of the parameter.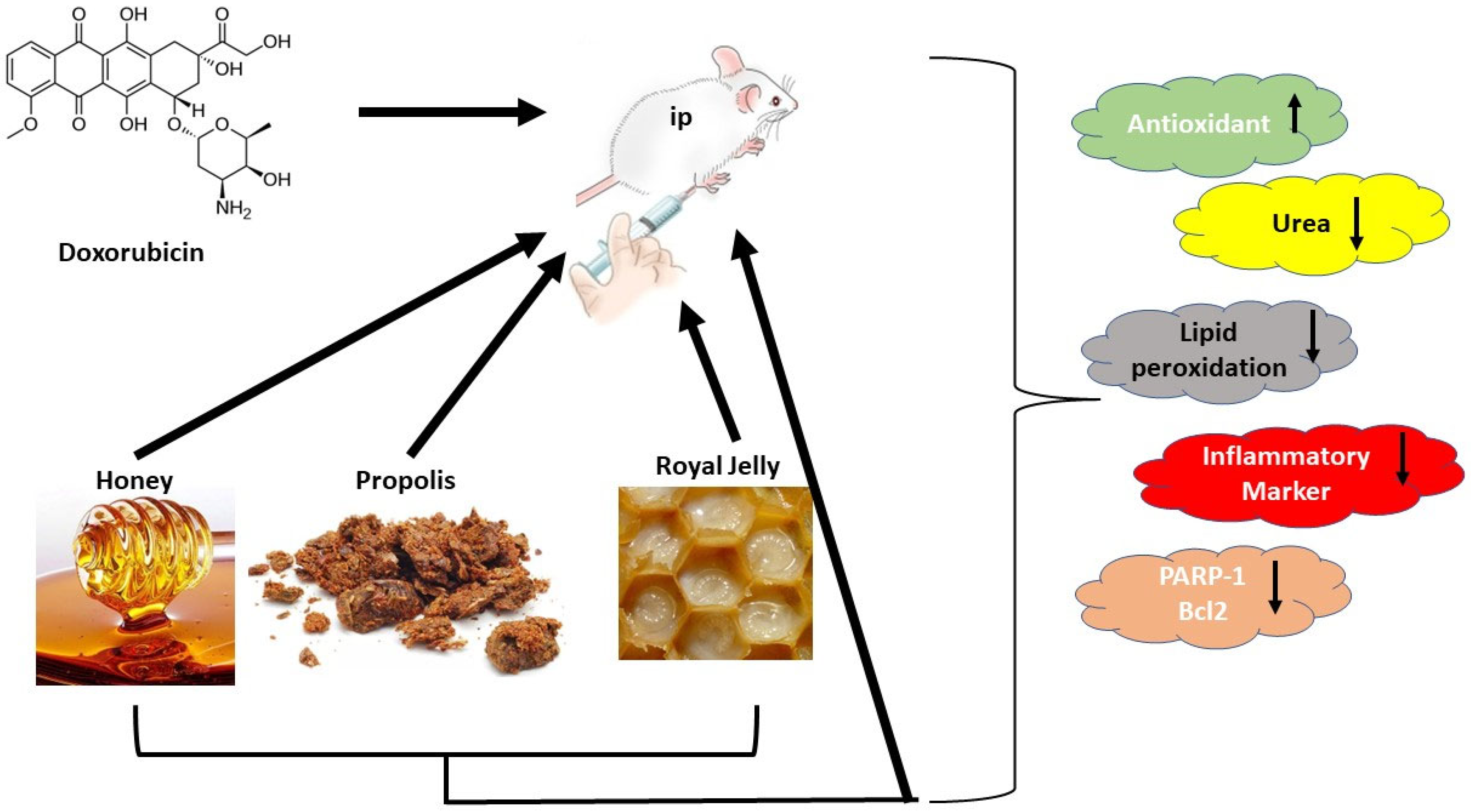
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohamed, H.K.; Mobasher, M.A.; Ebiya, R.A.; Hassen, M.T.; Hagag, H.M.; El-Sayed, R.; Abdel-Ghany, S.; Said, M.M.; Awad, N.S. Anti-Inflammatory, Anti-Apoptotic, and Antioxidant Roles of Honey, Royal Jelly, and Propolis in Suppressing Nephrotoxicity Induced by Doxorubicin in Male Albino Rats. Antioxidants 2022, 11, 1029. https://doi.org/10.3390/antiox11051029
Mohamed HK, Mobasher MA, Ebiya RA, Hassen MT, Hagag HM, El-Sayed R, Abdel-Ghany S, Said MM, Awad NS. Anti-Inflammatory, Anti-Apoptotic, and Antioxidant Roles of Honey, Royal Jelly, and Propolis in Suppressing Nephrotoxicity Induced by Doxorubicin in Male Albino Rats. Antioxidants. 2022; 11(5):1029. https://doi.org/10.3390/antiox11051029
Chicago/Turabian StyleMohamed, Hanaa K., Maysa A. Mobasher, Rasha A. Ebiya, Marwa T. Hassen, Howaida M. Hagag, Radwa El-Sayed, Shaimaa Abdel-Ghany, Manal M. Said, and Nabil S. Awad. 2022. "Anti-Inflammatory, Anti-Apoptotic, and Antioxidant Roles of Honey, Royal Jelly, and Propolis in Suppressing Nephrotoxicity Induced by Doxorubicin in Male Albino Rats" Antioxidants 11, no. 5: 1029. https://doi.org/10.3390/antiox11051029
APA StyleMohamed, H. K., Mobasher, M. A., Ebiya, R. A., Hassen, M. T., Hagag, H. M., El-Sayed, R., Abdel-Ghany, S., Said, M. M., & Awad, N. S. (2022). Anti-Inflammatory, Anti-Apoptotic, and Antioxidant Roles of Honey, Royal Jelly, and Propolis in Suppressing Nephrotoxicity Induced by Doxorubicin in Male Albino Rats. Antioxidants, 11(5), 1029. https://doi.org/10.3390/antiox11051029





