Sex-Dependent Protective Effect of Combined Application of Solubilized Ubiquinol and Selenium on Monocrotaline-Induced Pulmonary Hypertension in Wistar Rats
Abstract
1. Introduction
2. Materials and Methods
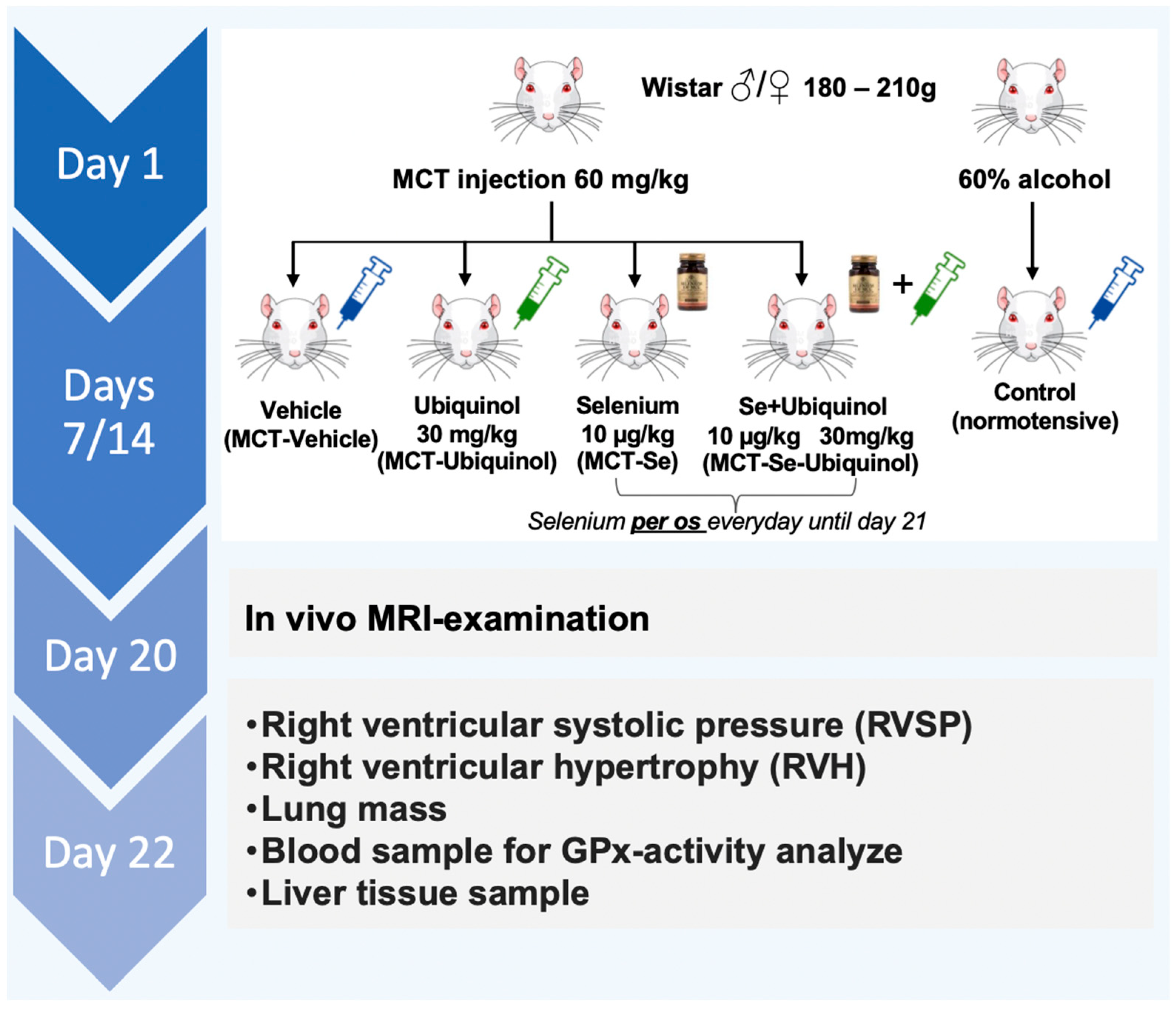
2.1. Animals and Experimental Design
2.2. Measurement of Systolic Pressure in the Tail Artery
2.3. Cycle Phase Determination
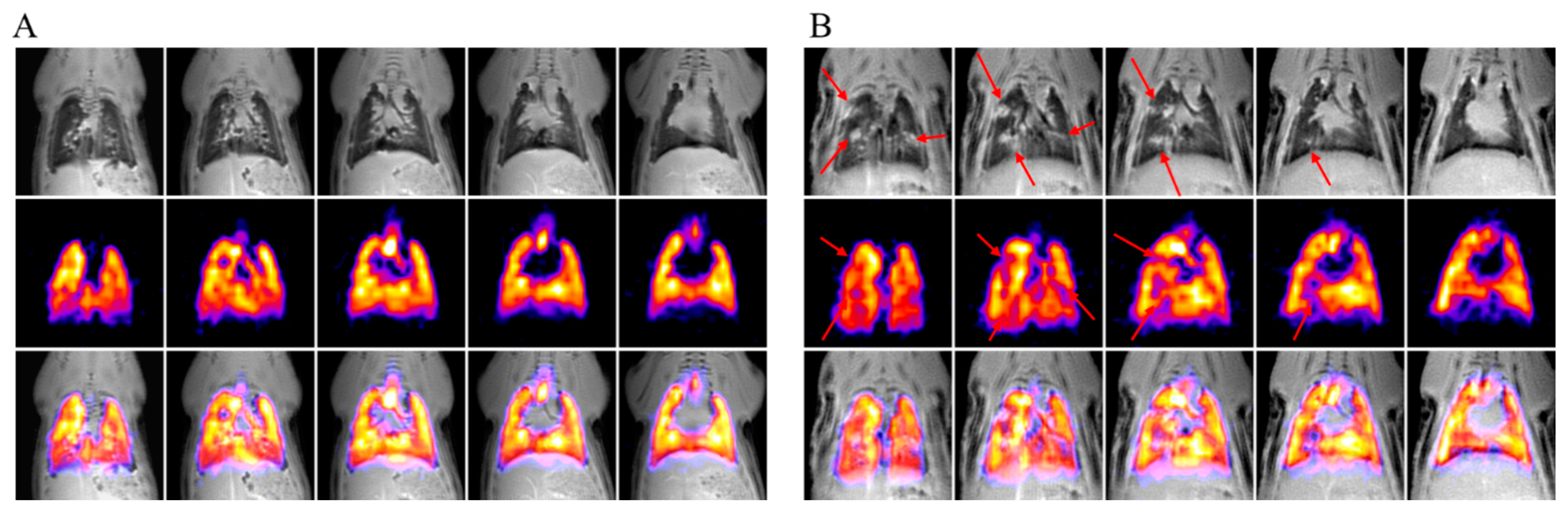
2.4. MRI Study
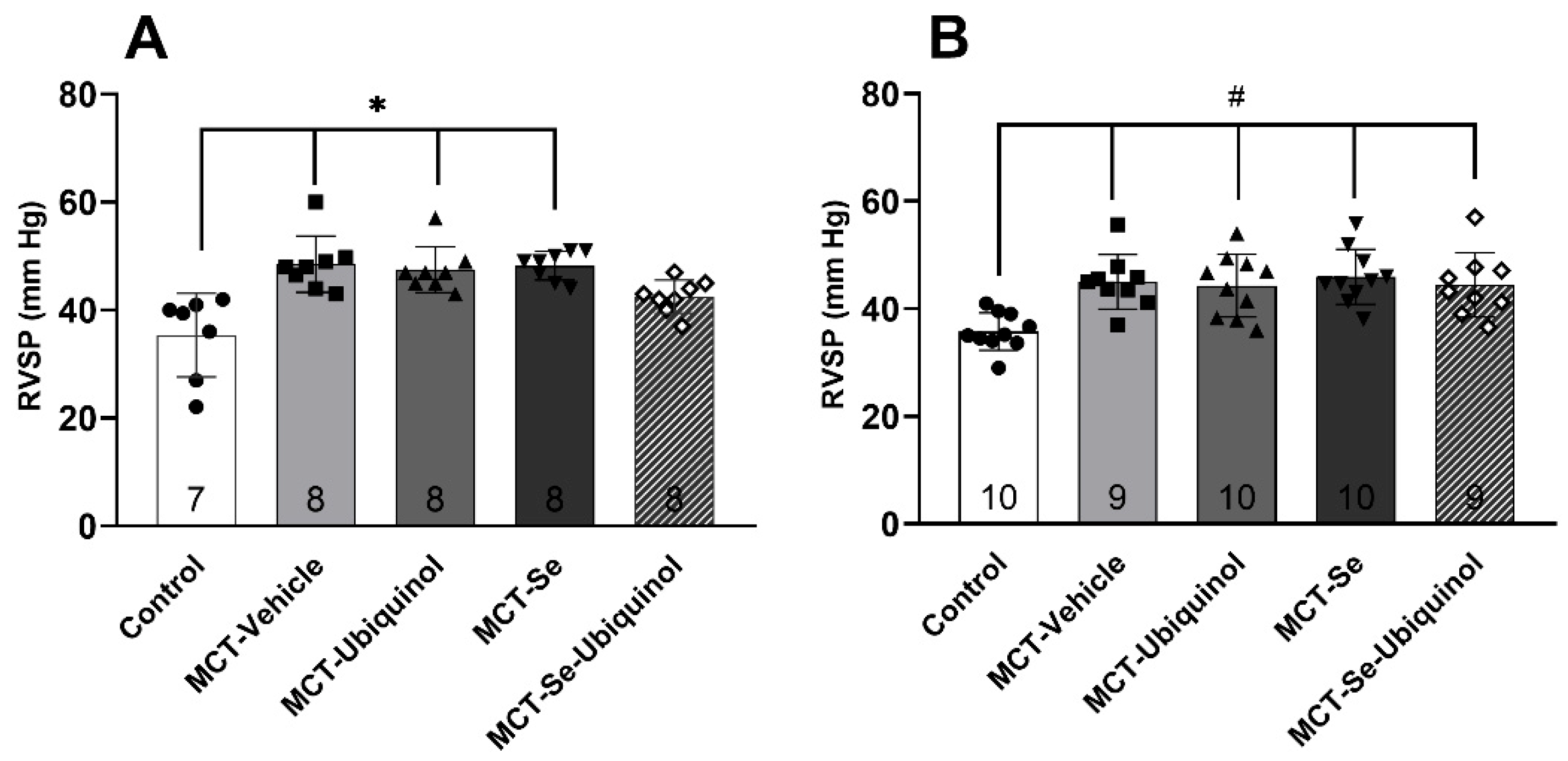
2.5. Registration of Hemodynamic Parameters In Vivo
2.6. Morphometric Measurement
2.7. Determination of Glutathione Peroxidase Activity in Venous Blood Hemolysates
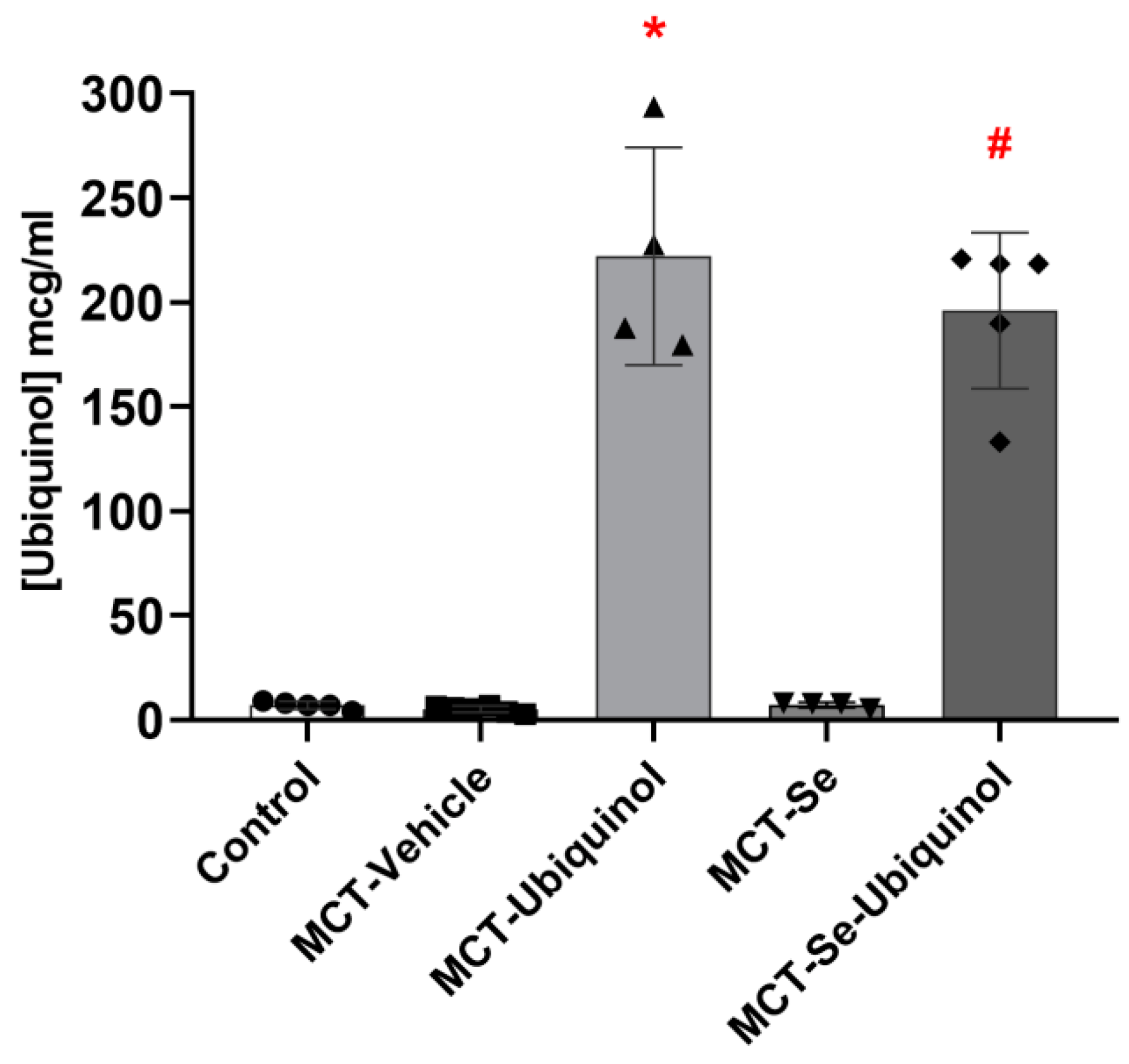
2.8. Method for Ubiquinol Determination in Plasma and Liver of Rats by High-Performance Gas-Liquid Chromatograph (HLPC)
2.9. Bioethical Approvement
2.10. Statistical Analysis
3. Results
4. Results and Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bhagavan, H.N.; Chopra, R.K. Coenzyme Q10: Absorption, tissue uptake, metabolism and pharmacokinetics. Free Radic. Res. 2006, 40, 445–453. [Google Scholar] [CrossRef]
- Zozina, V.I.; Covantev, S.; Goroshko, O.A.; Krasnykh, L.M.; Kukes, V.G. Coenzyme Q10 in Cardiovascular and Metabolic Diseases: Current State of the Problem. Curr. Cardiol. Rev. 2018, 14, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Cirilli, I.; Damiani, E.; Dludla, P.V.; Hargreaves, I.; Marcheggiani, F.; Millichap, L.E.; Orlando, P.; Silvestri, S.; Tiano, L. Role of Coenzyme Q10 in Health and Disease: An Update on the Last 10 Years (2010–2020). Antioxidants 2021, 10, 1325. [Google Scholar] [CrossRef] [PubMed]
- Nakagami, H.; Takemoto, M.; Liao, J. NADPH oxidase-derived superoxide anion mediates angiotensin II-induced cardiac hypertrophy. J. Mol. Cell. Cardiol. 2003, 35, 851–859. [Google Scholar] [CrossRef]
- Luo, K.; Yu, J.H.; Quan, Y.; Shin, Y.J.; Lee, K.E.; Kim, H.L.; Ko, E.J.; Chung, B.H.; Lim, S.W.; Yang, C.W. Therapeutic potential of coenzyme Q10 in mitochondrial dysfunction during tacrolimus-induced beta cell injury. Sci. Rep. 2019, 9, 7995. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.-K.; Wang, L.-P.; Chen, L.; Yao, X.-P.; Yang, K.-Q.; Gao, L.-G.; Zhou, X.-L. Coenzyme Q10 treatment of cardiovascular disorders of ageing including heart failure, hypertension and endothelial dysfunction. Clin. Chim. Acta 2015, 450, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Senkevich, O.; Golubkina, N.; Kovalsky Yu Sirotina, Z.; Iskrenok, G.; Beldy, D. Selenium supply of the Khabarovsk Region residents. Far East Med. J. 2009, 1, 82–84. [Google Scholar]
- Shimada, B.K.; Alfulaij, N.; Seale, L.A. The Impact of Selenium Deficiency on Cardiovascular Function. Int. J. Mol. Sci. 2021, 22, 10713. [Google Scholar] [CrossRef] [PubMed]
- Benstoem, C.; Goetzenich, A.; Kraemer, S.; Borosch, S.; Manzanares, W.; Hardy, G.; Stoppe, C. Selenium and Its Supplementation in Cardiovascular Disease—What do We Know? Nutrients 2015, 7, 3094–3118. [Google Scholar] [CrossRef]
- Kuropatkina, T.A.; Medvedeva, N.A.; Medvedev, O.S. The role of selenium in cardiology. Kardiologiia 2021, 61, 96–104. [Google Scholar] [CrossRef]
- Bansal, M.P.; Kaur, P. Selenium, a versatile trace element: Current research implications. Indian J. Exp. Boil. 2005, 43, 1119–1129. [Google Scholar]
- Brown, K.M.; Arthur, J.R. Selenium, selenoproteins and human health: A review. Public Health Nutr. 2001, 4, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Qiu, F.; Zhou, H.; Peng, Y.; Hao, W.; Xu, J.; Yuan, J.; Wang, S.; Qiang, B.; Xu, C.; et al. Identification and characterization of selenoprotein K: An antioxidant in cardiomyocytes. FEBS Lett. 2006, 580, 5189–5197. [Google Scholar] [CrossRef] [PubMed]
- Alanne, M.; Kristiansson, K.; Auro, K.; Silander, K.; Kuulasmaa, K.; Peltonen, L.; Salomaa, V.; Perola, M. Variation in the selenoprotein S gene locus is associated with coronary heart disease and ischemic stroke in two independent Finnish cohorts. Qual. Life Res. 2007, 122, 355–365. [Google Scholar] [CrossRef] [PubMed]
- Nordman, T.; Xia, L.; Björkhem-Bergman, L.; Damdimopoulos, A.; Nalvarte, I.; Arnér, E.S.J.; Spyrou, G.; Eriksson, L.C.; Björnstedt, M.; Olsson, J.M. Regeneration of the antioxidant ubiquinol by lipoamide dehydrogenase, thioredoxin reductase and glutathione reductase. BioFactors 2003, 18, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Xia, L.; Nordman, T.; Olsson, J.M.; Damdimopoulos, A.; Björkhem-Bergman, L.; Nalvarte, I.; Eriksson, L.C.; Arnér, E.; Spyrou, G.; Björnstedt, M. The Mammalian Cytosolic Selenoenzyme Thioredoxin Reductase Reduces Ubiquinone. J. Biol. Chem. 2003, 278, 2141–2146. [Google Scholar] [CrossRef]
- Alehagen, U.; Johansson, P.; Björnstedt, M.; Rosén, A.; Dahlström, U. Cardiovascular mortality and N-terminal-proBNP reduced after combined selenium and coenzyme Q10 supplementation: A 5-year prospective randomized double-blind placebo-controlled trial among elderly Swedish citizens. Int. J. Cardiol. 2013, 167, 1860–1866. [Google Scholar] [CrossRef]
- Alehagen, U.; Aaseth, J.; Johansson, P. Less increase of copeptin and MR-proADM due to intervention with selenium and coenzyme Q10 combined: Results from a 4-year prospective randomized double-blind placebo-controlled trial among elderly Swedish citizens. BioFactors 2015, 41, 443–452. [Google Scholar] [CrossRef]
- Johansson, P.; Dahlström, Ö.; Dahlström, U.; Alehagen, U. Improved health-related quality of life, and more days out of hospital with supplementation with selenium and coenzyme Q10 combined. Results from a double blind, placebo-controlled prospective study. J. Nutr. Health Aging 2015, 19, 870–877. [Google Scholar] [CrossRef]
- Gelzinis, T.A. Pulmonary Hypertension in 2021: Part I—Definition, Classification, Pathophysiology, and Presentation. J. Cardiothorac. Vasc. Anesth. 2021. [Google Scholar] [CrossRef]
- Thenappan, T.; Ormiston, M.; Ryan, J.J.; Archer, S.L. Pulmonary arterial hypertension: Pathogenesis and clinical management. BMJ 2018, 360, j5492. [Google Scholar] [CrossRef] [PubMed]
- Schermuly, R.T.; Ghofrani, A.; Wilkins, M.R.; Grimminger, F. Mechanisms of disease: Pulmonary arterial hypertension. Nat. Rev. Cardiol. 2011, 8, 443–455. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, F.W.; Sorge, I.; Vogel-Claussen, J.; Roth, C.; Gräfe, D.; Päts, A.; Voskrebenzev, A.; Anders, R.M. The current status and further prospects for lung magnetic resonance imaging in pediatric radiology. Pediatr. Radiol. 2020, 50, 734–749. [Google Scholar] [CrossRef] [PubMed]
- Neal, M.; Pippard, B.J.; Hollingsworth, K.G.; Maunder, A.; Dutta, P.; Simpson, A.J.; Blamire, A.M.; Wild, J.M.; Thelwall, P.E. Optimized and accelerated 19 F-MRI of inhaled perfluoropropane to assess regional pulmonary ventilation. Magn. Reson. Med. 2019, 82, 1301–1311. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Arroyo, J.G.; Farkas, L.; Alhussaini, A.A.; Farkas, D.; Kraskauskas, D.; Voelkel, N.F.; Bogaard, H.J. The monocrotaline model of pulmonary hypertension in perspective. Am. J. Physiol. Cell. Mol. Physiol. 2012, 302, L363–L369. [Google Scholar] [CrossRef]
- Desowitz, R. Thorn in the Starfish: The Immune System and How It Works; WW Norton & Company: New York, NY, USA, 1988; p. 256. [Google Scholar]
- Swathy, S.S.; Panicker, S.; Indira, M. Effect of exogenous selenium on the testicular toxicity induced by ethanol in rats. Indian J. Physiol. Pharmacol. 2006, 50, 215–224. [Google Scholar]
- Pavlova, O.S.; Anisimov, N.V.; Gervits, L.L.; Gulyaev, M.V.; Semenova, V.N.; Pirogov, Y.A.; Panchenko, V.Y. 19F MRI of human lungs at 0.5 Tesla using octafluorocyclobutane. Magn. Reson. Med. 2020, 84, 2117–2123. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 Years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Flohé, L.; Günzler, W.A. Assays of glutathione peroxidase. Methods Enzymol. 1984, 105, 114–120. [Google Scholar] [CrossRef]
- Littarru, G.; Battino, M.; Tomasetti, M.; Mordente, A.; Santini, S.; Oradei, A.; Manto, A.; Ghirlanda, G. Metabolic implications of Coenzyme Q10 in red blood cells and plasma lipoproteins. Mol. Asp. Med. 1994, 15, s67–s72. [Google Scholar] [CrossRef]
- Forsmark-Andrée, P.; Ernster, L. Evidence for a protective effect of endogenous ubiquinol against oxidative damage to mitochondrial protein and DNA during lipid peroxidation. Mol. Asp. Med. 1994, 15, s73–s81. [Google Scholar] [CrossRef]
- Abdullaev, S.H.; Kuropatkina, T.; Pankova Artemieva, M.; Makarov, V.; Medvedeva, N.; Medvedev, O. The effect of intravenous ubiquinol administration on MCT PH development in experiments on rats. Exp. Clin. Pharmacol. 2019, 85, 20–23. [Google Scholar]
- Kuropatkina, T.A.; Pankova, N.V.; Medvedeva, N.A.; Medvedev, O.S. Ubiquinol ameliorates endothelial dysfunction and increases expression of miRNA-34a in a rat model of pulmonary hypertension. Res. Results Pharmacol. 2021, 7, 23–31. [Google Scholar] [CrossRef]
- Medvedeva, N.; Pankova, N.; Artemyeva, M. Gender differences PH manifestation. 2017 The female sex hormone estradiol in-fluence the Arterial hypertension. Promotion of arterial hypertension prevention and treatment “Antihypertensive League”. Arter. Hypertens. 2017, 23, 421–433. [Google Scholar]
- Kalenikova, E.; Gorodetskaya, E.; Obolenskaya, O.; Shapavo, N.S.; Makarov, V.G.; Medvedev, O.S. Pharmacokinetics and tissue distribution of ubiquinol and its intravenous administration. Exp. Clin. Pharmacol. 2018, 81, 103–104. [Google Scholar]
- Kalenikova, E.I.; Gorodetskaya, E.; Obolenskaya, O.N.; Shapoval, N.S.; Makarov, V.G.; Medvedev, O. Tissue-Level Dynamics and Redox State of Coenzyme Q10 in Rats After Intravenous Injection of Ubiquinol. Pharm. Chem. J. 2018, 52, 690–693. [Google Scholar] [CrossRef]
- Zhang, Y.; Roh, Y.J.; Han, S.-J.; Park, I.; Lee, H.M.; Ok, Y.S.; Lee, B.C.; Lee, S.-R. Role of Selenoproteins in Redox Regulation of Signaling and the Antioxidant System: A Review. Antioxidants 2020, 9, 383. [Google Scholar] [CrossRef] [PubMed]
- Rock, C.; Moos, P.J. Selenoprotein P protects cells from lipid hydroperoxides generated by 15-LOX-1. Prostaglandins Leukot. Essent. Fat. Acids 2010, 83, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Pinto, R.E.; Bartley, W. The effect of age and sex on glutathione reductase and glutathione peroxidase activities and on aerobic glutathione oxidation in rat liver homogenates. Biochem. J. 1969, 112, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Borras, C.; Sastre, J.; García-Sala, D.; Lloret, A.; Pallardó, F.V.; Viña, J. Mitochondria from females exhibit higher antioxidant gene expression and lower oxidative damage than males. Free Radic. Biol. Med. 2003, 34, 546–552. [Google Scholar] [CrossRef]
- Wang, L.; Ahn, Y.J.; Asmis, R. Sexual dimorphism in glutathione metabolism and glutathione-dependent responses. Redox Biol. 2019, 31, 101410. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Mao, Q.; Cao, J.; Wang, Y.; Zhou, X.; Fan, L. Effects of coenzyme Q10 on vascular endothelial function in humans: A meta-analysis of randomized controlled trials. Atherosclerosis 2011, 221, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Kao, C.; Carsten, M. Cellular Aspects of Smooth Muscle Function; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2005. [Google Scholar]
- Belardinelli, R.; Muçaj, A.; Lacalaprice, F.; Solenghi, M.; Seddaiu, G.; Principi, F.; Tiano, L.; Littarru, G.P. Coenzyme Q10 and exercise training in chronic heart failure. Eur. Heart J. 2006, 27, 2675–2681. [Google Scholar] [CrossRef]
- Tiano, L.; Belardinelli, R.; Carnevali, P.; Principi, F.; Seddaiu, G.; Littarru, G.P. Effect of coenzyme Q10 administration on endothelial function and extracellular superoxide dismutase in patients with ischaemic heart disease: A double-blind, randomized controlled study. Eur. Heart J. 2007, 28, 2249–2255. [Google Scholar] [CrossRef] [PubMed]
- Kozaeva, L.; Gorodetskaya, E.; Ruuge, E.; Kalenikova, E.; Medvedev, O. Beneficial effect of coenzyme Q10 injection on nitric oxide -related dilation of the rat aorta. Eur. J. Pharmacol. 2017, 794, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-B.; Han, J.-Y.; Jiang, W.; Wang, J. Selenium inhibits high glucose-induced cyclooxygenase-2 and P-selectin expression in vascular endothelial cells. Mol. Biol. Rep. 2010, 38, 2301–2306. [Google Scholar] [CrossRef]
- Zhu, M.; Gao, Z.; Fu, Y.; Qiu, Y.; Huang, K.; Zhu, C.; Wu, Y.; Zhu, T.; Wang, Q.; Yang, L.; et al. Amorphous Selenium Nanoparticles Improve Vascular Function in Rats with Chronic Isocarbophos Poisoning via Inhibiting the Apoptosis of Vascular Endothelial Cells. Front. Bioeng. Biotechnol. 2021, 9, 673327. [Google Scholar] [CrossRef]
- Smith, A.M.; Cha, C.; Kimura, R.E. Plasma selenium and glutathione peroxidase activity fluctuate during the rat estrous cycle. Nutr. Res. 1995, 15, 267–277. [Google Scholar] [CrossRef]
- Ha, E.; Smith, A. Plasma selenium and glutathione peroxidase activity increase during the pre-ovulatory phase of the menstrual cycle. J. Am. Coll. Nutr. 2003, 22, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Smith, A.M.; Failla, M.L.; Hill, K.E.; Yu, Z. Estrogen status alters tissue distribution and metabolism of selenium in female rats. J. Nutr. Biochem. 2012, 23, 532–538. [Google Scholar] [CrossRef]
- Mohamed, H.A.; Said, R.S. Coenzyme Q10 attenuates inflammation and fibrosis implicated in radiation enteropathy through suppression of NF-kB/TGF-β/MMP-9 pathways. Int. Immunopharmacol. 2021, 92, 107347. [Google Scholar] [CrossRef] [PubMed]
- Serebruany, V.L.; Gurbel, P.A.; Ordoñez, J.; Herzog, W.R.; Rohde, M.; Mortensen, S.A.; Folkers, K. Could coenzyme Q10 affect hemostasis by inhibiting platelet vitronectin (CD51CD61) receptor? Mol. Asp. Med. 1997, 18, 189–194. [Google Scholar] [CrossRef]
- Serebruany, V.L.; Ordonez, J.V.; Herzog, W.R.; Rohde, M.; Mortensen, S.A.; Folkers, K.; Gurbel, P.A. Dietary Coenzyme Q10 Supplementation Alters Platelet Size and Inhibits Human Vitronectin (CD51/CD61) Receptor Expression. J. Cardiovasc. Pharmacol. 1997, 29, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Moghaddam, A.K.Z.; Hamzekolaei, M.H.M.; Khajali, F.; Hassanpour, H. Role of Selenium from Different Sources in Prevention of Pulmonary Arterial Hypertension Syndrome in Broiler Chickens. Biol. Trace Elem. Res. 2017, 180, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Xiao, R.; Zhu, L.; Su, Y.; Zhang, J.; Lu, Y.; Li, J.; Wang, T.; Fang, J.; Jing, Z.-C.; Dupuis, J.; et al. Monocrotaline pyrrole induces pulmonary endothelial damage through binding to and release from erythrocytes in lung during venous blood reoxygenation. Am. J. Physiol. Cell. Mol. Physiol. 2019, 316, L798–L809. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.; Shih, H.; Huang, I.; Tsai, P.; Chen, K.; Huang, C. Magnesium Sulfate Mitigates the Progression of Monocrotaline Pulmonary Hypertension in Rats. Int. J. Mol. Sci. 2019, 20, 4622. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.-M.; Zhou, R.; Zhang, M.; Tan, H.-R.; Yu, J.-Q. Betaine Attenuates Monocrotaline-Induced Pulmonary Arterial Hypertension in Rats via Inhibiting Inflammatory Response. Molecules 2018, 23, 1274. [Google Scholar] [CrossRef]
- Lee, Y.-S.; Byun, J.; Kim, J.-A.; Lee, J.-S.; Kim, K.L.; Suh, Y.-L.; Kim, J.-M.; Jang, H.-S.; Lee, J.-Y.; Shin, I.-S.; et al. Monocrotaline-induced pulmonary hypertension correlates with upregulation of connective tissue growth factor expression in the lung. Exp. Mol. Med. 2005, 37, 27–35. [Google Scholar] [CrossRef]
- Shortness of Breath (Dyspnea) in Pulmonary Fibrosis URL. Available online: https://pulmonaryfibrosisnews.com/shortness-of-breath-dyspnea (accessed on 17 February 2022).
- Gutierrez-Mariscal, F.M.; Larriva, A.P.A.-D.; Limia-Perez, L.; Romero-Cabrera, J.L.; Yubero-Serrano, E.M.; López-Miranda, J. Coenzyme Q10 Supplementation for the Reduction of Oxidative Stress: Clinical Implications in the Treatment of Chronic Diseases. Int. J. Mol. Sci. 2020, 21, 7870. [Google Scholar] [CrossRef]
- De Blasio, M.; Huynh, K.; Qin, C.; Rosli, S.; Kiriazis, H.; Ayer, A.; Cemerlang, N.; Stocker, R.; Du, X.-J.; McMullen, J.R.; et al. Therapeutic targeting of oxidative stress with coenzyme Q10 counteracts exaggerated diabetic cardiomyopathy in a mouse model of diabetes with diminished PI3K(p110α) signaling. Free Radic. Biol. Med. 2015, 87, 137–147. [Google Scholar] [CrossRef]
- Kikuchi, N.; Satoh, K.; Kurosawa, R.; Yaoita, N.; Mamun, E.A.; Siddique, M.A.H.; Omura, J.; Satoh, T.; Nogi, M.; Sunamura, S.; et al. Selenoprotein P Promotes the Development of Pulmonary Arterial Hypertension. Circulation 2018, 138, 600–623. [Google Scholar] [CrossRef] [PubMed]

| Group | Control (n = 6) | MCT-Vehicle (n = 6) | MCT-Ubiquinol (n = 6) | MCT-Se (n = 6) | MCT-Se-Ubiquinol (n = 6) |
|---|---|---|---|---|---|
| μmol of NADPH/min × V sample | |||||
| Males | 7.021 ± 0.826 | 8.079 ± 0.266 *,# | 6.611 ± 0.593 | 6.348 ± 0.569 | 7.172 ± 0.364 |
| Females | 10.880 ± 1.792 $ | 9.365 ± 1.346 | 9.420 ± 0.441 $ | 9.764 ± 1.418 $ | 9.637 ± 0.411 $ |
| Group | Control | MCT-Vehicle | MCT-Ubiquinol | MCT-Se | MCT-Se-Ubiquinol |
|---|---|---|---|---|---|
| Males | 0.611 ± 0.057 (n = 8) | 0.877 ± 0.328 * (n = 9) | 0.740 ± 0.115 (n = 8) | 0.796 ± 0.171 & (n = 9) | 0.751 ± 0.094 (n = 8) |
| Females | 0.636 ± 0.083 (n = 12) | 0.828 ± 0.248 # (n = 11) | 0.754 ± 0.083 (n = 11) | 0.792 ± 0.148 $ (n = 11) | 0.733 ± 0.113 (n = 10) |
| Group | 1H MRI Signs of Fibrotic Changes | 19F MRI Disruption in Ventilation |
|---|---|---|
| Control (n = 4) | 0 | 0 |
| MCT-Vehicle (n = 4) | 4 * | 4 $ |
| MCT-Ubiquinol (n = 4) | 2 | 3 & |
| MCT-Se (n = 4) | 4 * | 4 $ |
| MCT-Se-Ubiquinol (n = 4) | 3 # | 4 $ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuropatkina, T.; Pavlova, O.; Gulyaev, M.; Pirogov, Y.; Khutorova, A.; Stvolinsky, S.; Medvedeva, N.; Medvedev, O. Sex-Dependent Protective Effect of Combined Application of Solubilized Ubiquinol and Selenium on Monocrotaline-Induced Pulmonary Hypertension in Wistar Rats. Antioxidants 2022, 11, 549. https://doi.org/10.3390/antiox11030549
Kuropatkina T, Pavlova O, Gulyaev M, Pirogov Y, Khutorova A, Stvolinsky S, Medvedeva N, Medvedev O. Sex-Dependent Protective Effect of Combined Application of Solubilized Ubiquinol and Selenium on Monocrotaline-Induced Pulmonary Hypertension in Wistar Rats. Antioxidants. 2022; 11(3):549. https://doi.org/10.3390/antiox11030549
Chicago/Turabian StyleKuropatkina, Tatyana, Olga Pavlova, Mikhail Gulyaev, Yury Pirogov, Anastasiya Khutorova, Sergey Stvolinsky, Natalia Medvedeva, and Oleg Medvedev. 2022. "Sex-Dependent Protective Effect of Combined Application of Solubilized Ubiquinol and Selenium on Monocrotaline-Induced Pulmonary Hypertension in Wistar Rats" Antioxidants 11, no. 3: 549. https://doi.org/10.3390/antiox11030549
APA StyleKuropatkina, T., Pavlova, O., Gulyaev, M., Pirogov, Y., Khutorova, A., Stvolinsky, S., Medvedeva, N., & Medvedev, O. (2022). Sex-Dependent Protective Effect of Combined Application of Solubilized Ubiquinol and Selenium on Monocrotaline-Induced Pulmonary Hypertension in Wistar Rats. Antioxidants, 11(3), 549. https://doi.org/10.3390/antiox11030549






