Immune Regulation of Heme Oxygenase-1 in Allergic Airway Inflammation
Abstract
1. Introduction
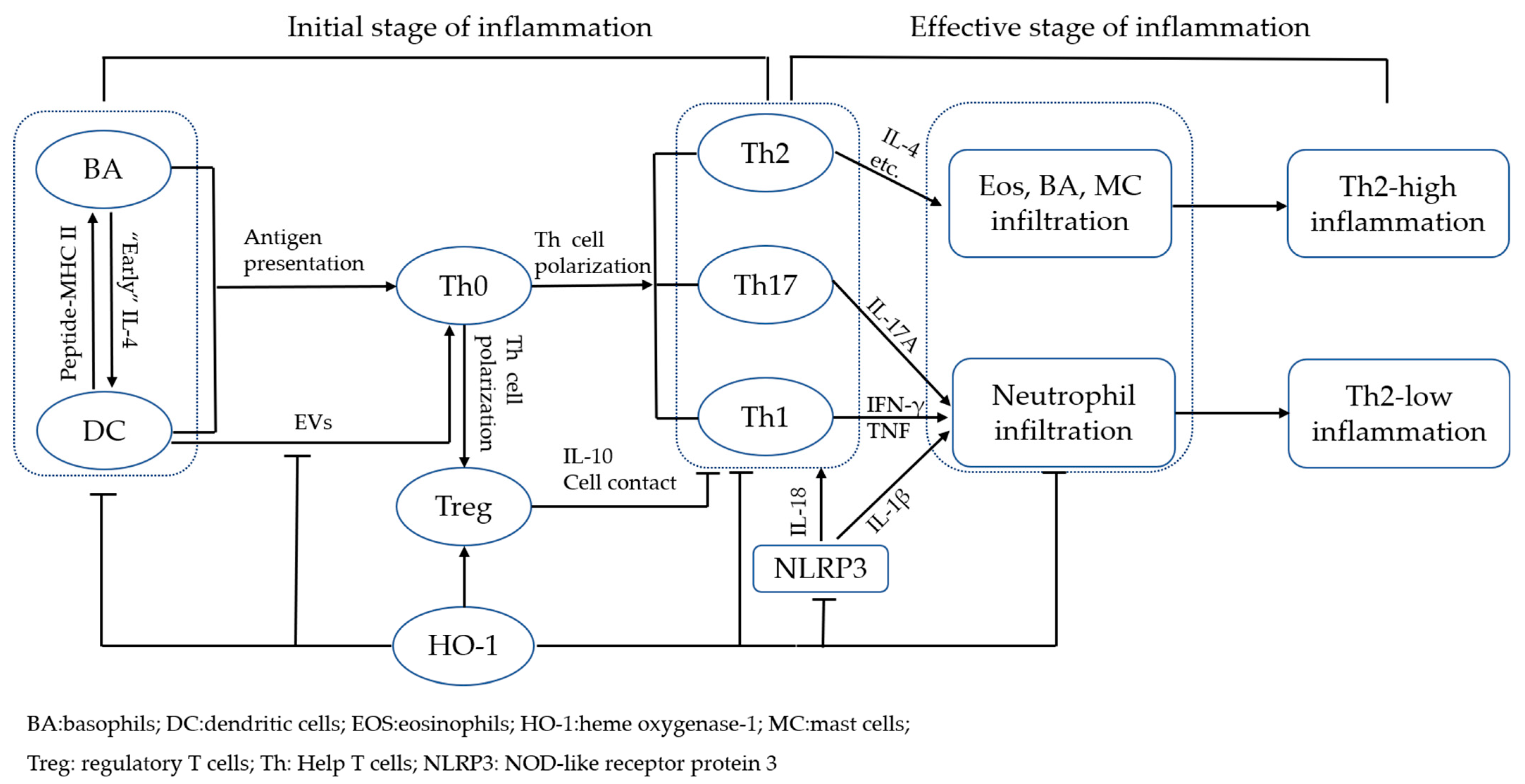
2. Mechanisms of Asthmatic Airway Inflammation and Role of HO-1
3. HO-1 Inhibits Inflammation during the Initial Stage
3.1. Inhibition of DC Function
3.2. Inhibition of BA Function
4. HO-1 Inhibits Inflammation during the Effective Stage
4.1. HO-1 Promotes Treg Cell Function and Inhibits Th2- and Th17 Cell-Mediated Inflammation
4.2. HO-1 Inhibits Th17 Cell-Mediated Inflammation
4.3. HO-1 Inhibits MC Function
4.4. HO-1 Regulated Inflammation by Inhibiting NLRP3 Inflammasomes
4.5. HO-1 Promotes Polarization of Macrophages to M2 Phenotype
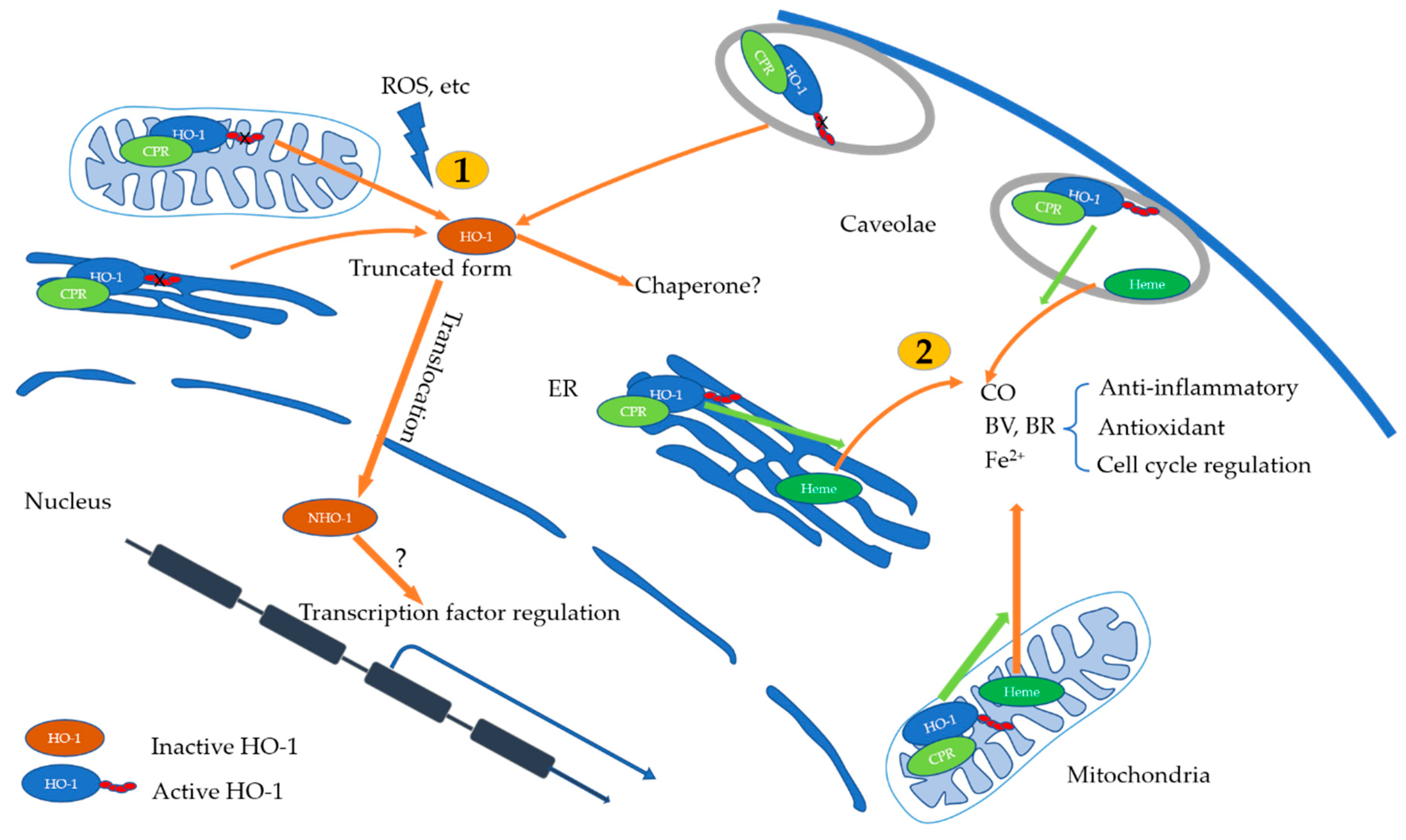
5. Subcellular Localization and Anti-Inflammatory Mechanism of HO-1
6. Potential Clinical Application of HO-1
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Consoli, V.; Sorrenti, V.; Grosso, S.; Vanella, L. Heme Oxygenase-1 Signaling and Redox Homeostasis in Physiopathological Conditions. Biomolecules 2021, 11, 589. [Google Scholar] [CrossRef]
- Ryter, S.W. Heme Oxgenase-1, a Cardinal Modulator of Regulated Cell Death and Inflammation. Cells 2021, 10, 515. [Google Scholar] [CrossRef]
- Ryter, S.W. Therapeutic Potential of Heme Oxygenase-1 and Carbon Monoxide in Acute Organ Injury, Critical Illness, and Inflammatory Disorders. Antioxidants 2020, 9, 1153. [Google Scholar] [CrossRef] [PubMed]
- Weis, N.; Weigert, A.; von Knethen, A.; Brune, B. Heme oxygenase-1 contributes to an alternative macrophage activation profile induced by apoptotic cell supernatants. Mol. Biol. Cell 2009, 20, 1280–1288. [Google Scholar] [CrossRef] [PubMed]
- Takamiya, R.; Murakami, M.; Kajimura, M.; Goda, N.; Makino, N.; Takamiya, Y.; Yamaguchi, T.; Ishimura, Y.; Hozumi, N.; Suematsu, M. Stabilization of mast cells by heme oxygenase-1: An anti-inflammatory role. Am. J. Physiol. Heart Circ. Physiol. 2002, 283, H861–H870. [Google Scholar] [CrossRef]
- Zhong, W.; Di, C.; Lv, J.; Zhang, Y.; Lin, X.; Yuan, Y.; Lv, J.; Xia, Z. Heme oxygenase-1 inhibits basophil maturation and activation but promotes its apoptosis in T helper type 2-mediated allergic airway inflammation. Immunology 2016, 147, 321–337. [Google Scholar] [CrossRef] [PubMed]
- Pae, H.O.; Oh, G.S.; Choi, B.M.; Chae, S.C.; Chung, H.T. Differential expressions of heme oxygenase-1 gene in CD25– and CD25+ subsets of human CD4+ T cells. Biochem. Biophys. Res. Commun. 2003, 306, 701–705. [Google Scholar] [CrossRef]
- Pae, H.O.; Lee, Y.C.; Chung, H.T. Heme oxygenase-1 and carbon monoxide: Emerging therapeutic targets in inflammation and allergy. Recent Pat. Inflamm. Allergy Drug Discov. 2008, 2, 159–165. [Google Scholar] [CrossRef]
- Kuribayashi, K.; Iida, S.; Nakajima, Y.; Funaguchi, N.; Tabata, C.; Fukuoka, K.; Fujimori, Y.; Ihaku, D.; Nakano, T. Suppression of heme oxygenase-1 activity reduces airway hyperresponsiveness and inflammation in a mouse model of asthma. J. Asthma 2015, 52, 662–668. [Google Scholar] [CrossRef]
- Xia, Z.W.; Zhong, W.W.; Xu, L.Q.; Sun, J.L.; Shen, Q.X.; Wang, J.G.; Shao, J.; Li, Y.Z.; Yu, S.C. Heme oxygenase-1-mediated CD4+CD25high regulatory T cells suppress allergic airway inflammation. J. Immunol. 2006, 177, 5936–5945. [Google Scholar] [CrossRef]
- Xia, Z.W.; Xu, L.Q.; Zhong, W.W.; Wei, J.J.; Li, N.L.; Shao, J.; Li, Y.Z.; Yu, S.C.; Zhang, Z.L. Heme oxygenase-1 attenuates ovalbumin-induced airway inflammation by up-regulation of foxp3 T-regulatory cells, interleukin-10, and membrane-bound transforming growth factor-β1. Am. J. Pathol. 2007, 171, 1904–1914. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, L.; Wu, J.; Di, C.; Xia, Z. Heme oxygenase-1 exerts a protective role in ovalbumin-induced neutrophilic airway inflammation by inhibiting Th17 cell-mediated immune response. J. Biol. Chem. 2013, 288, 34612–34626. [Google Scholar] [CrossRef] [PubMed]
- Horvath, I.; Donnelly, L.E.; Kiss, A.; Paredi, P.; Kharitonov, S.A.; Barnes, P.J. Raised levels of exhaled carbon monoxide are associated with an increased expression of heme oxygenase-1 in airway macrophages in asthma: A new marker of oxidative stress. Thorax 1998, 53, 668–672. [Google Scholar] [CrossRef] [PubMed]
- Harju, T.; Soini, Y.; Paakko, R.; Kinnula, V.L. Up-regulation of heme oxygenase-I in alveolar macrophages of newly diagnosed asthmatics. Respir. Med. 2002, 96, 418–423. [Google Scholar] [CrossRef][Green Version]
- Mo, B.; Zhang, Z.; Xu, Y.; Xiong, W.; Liu, X.A.; Zhen, G. Expression of heme oxygenase-1 in the peripheral blood mononuclear cells from asthmatic patients. J. Huazhong Univ. Sci. Technol. Med. Sci. 2005, 25, 385–388. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, C.; Luo, J.; Hua, S.; Li, D.; Peng, L.; Liu, H.; Song, L. The protective role of Zingerone in a murine asthma model via activation of the AMPK/Nrf2/HO-1 pathway. Food Funct. 2021, 12, 3120–3131. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.M.; Ryu, H.W.; Kwon, O.K.; Hwang, D.; Kim, M.G.; Min, J.H.; Zhang, Z.; Kim, S.Y.; Paik, J.H.; Oh, S.R.; et al. Callicarpa japonica Thunb. ameliorates allergic airway inflammation by suppressing NF-kappaB activation and upregulating HO-1 expression. J. Ethnopharmacol. 2021, 267, 113523. [Google Scholar] [CrossRef]
- Kitada, O.; Kodama, T.; Kuribayashi, K.; Ihaku, D.; Fujita, M.; Matsuyama, T.; Sugita, M. Heme oxygenase-1 (HO-1) protein induction in a mouse model of asthma. Clin. Exp. Allergy 2001, 31, 1470–1477. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.X.; Sekizawa, K.; Okinaga, S.; Lie, R.; Sasaki, H. Role of heme oxygenase in pulmonary response to antigen challenge in sensitized rats in vivo. Int. Arch. Allergy Immunol. 1999, 120, 141–145. [Google Scholar] [CrossRef] [PubMed]
- Chapman, J.T.; Otterbein, L.E.; Elias, J.A.; Choi, A.M. Carbon monoxide attenuates aeroallergen-induced inflammation in mice. Am. J. Physiol. Lung Cell Mol. Physiol. 2001, 281, L209–L216. [Google Scholar] [CrossRef]
- Almolki, A.; Taille, C.; Martin, G.F.; Jose, P.J.; Zedda, C.; Conti, M.; Megret, J.; Henin, D.; Aubier, M.; Boczkowski, J. Heme oxygenase attenuates allergen-induced airway inflammation and hyperreactivity in guinea pigs. Am. J. Physiol. Lung Cell Mol. Physiol. 2004, 287, L26–L34. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Jiang, G.; Li, X.; Xiao, Q.; Chen, Y.; Xu, H.; Liu, G.; Lei, A.; Zhou, P.; Shi, K.; et al. Bilirubin represents a negative regulator of ILC2 in allergic airway inflammation. Mucosal. Immunol. 2022, 15, 314–326. [Google Scholar] [CrossRef]
- Lin, X.L.; Lv, J.J.; Lv, J.; Di, C.X.; Zhang, Y.J.; Zhou, T.; Liu, J.L.; Xia, Z.W. Heme oxygenase-1 directly binds STAT3 to control the generation of pathogenic Th17 cells during neutrophilic airway inflammation. Allergy 2017, 72, 1972–1987. [Google Scholar] [CrossRef]
- Grayson, M.H.; Feldman, S.; Prince, B.T.; Patel, P.J.; Matsui, E.C.; Apter, A.J. Advances in asthma in 2017: Mechanisms, biologics, and genetics. J. Allergy Clin. Immunol. 2018, 142, 1423–1436. [Google Scholar] [CrossRef]
- Cevhertas, L.; Ogulur, I.; Maurer, D.J.; Burla, D.; Ding, M.; Jansen, K.; Koch, J.; Liu, C.; Ma, S.; Mitamura, Y.; et al. Advances and recent developments in asthma in 2020. Allergy 2020, 75, 3124–3146. [Google Scholar] [CrossRef]
- Boonpiyathad, T.; Sozener, Z.C.; Satitsuksanoa, P.; Akdis, C.A. Immunologic mechanisms in asthma. Semin. Immunol. 2019, 46, 101333. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.; Lee, H.K. The Role of CD4+ T Cells and Microbiota in the Pathogenesis of Asthma. Int. J. Mol. Sci. 2021, 22, 11822. [Google Scholar] [CrossRef] [PubMed]
- Holgate, S.T. Innate and adaptive immune responses in asthma. Nat. Med. 2012, 18, 673–683. [Google Scholar] [CrossRef]
- Riquelme, S.A.; Carreno, L.J.; Espinoza, J.A.; Mackern-Oberti, J.P.; Alvarez-Lobos, M.M.; Riedel, C.A.; Bueno, S.M.; Kalergis, A.M. Modulation of antigen processing by haem-oxygenase 1. Implications on inflammation and tolerance. Immunology 2016, 149, 1–12. [Google Scholar] [CrossRef]
- Peron, G.; de Lima Thomaz, L.; da Rosa, L.C.; Thome, R.; Verinaud, L.M.C. Modulation of dendritic cell by pathogen antigens: Where do we stand? Immunol. Lett. 2018, 196, 91–102. [Google Scholar] [CrossRef]
- Ferreira, I.; Liberal, J.; Martins, J.D.; Silva, A.; Neves, B.M.; Cruz, M.T. Inflammasome in Dendritic Cells Immunobiology: Implications to Diseases and Therapeutic Strategies. Curr. Drug Targets 2017, 18, 1003–1018. [Google Scholar] [CrossRef]
- Chauveau, C.; Remy, S.; Royer, P.J.; Hill, M.; Tanguy-Royer, S.; Hubert, F.X.; Tesson, L.; Brion, R.; Beriou, G.; Gregoire, M.; et al. Heme oxygenase-1 expression inhibits dendritic cell maturation and proinflammatory function but conserves IL-10 expression. Blood 2005, 106, 1694–1702. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.C.; Liu, X.; Kwon, M.Y.; Kang, Y.H.; Chung, S.W.; Perrella, M.A. Regulation of heme oxygenase-1 gene by peptidoglycan involves the interaction of Elk-1 and C/EBPalpha to increase expression. Am. J. Physiol. Lung Cell Mol. Physiol. 2010, 298, L870–L879. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wong, T.H.; Chen, H.A.; Gau, R.J.; Yen, J.H.; Suen, J.L. Heme Oxygenase-1-Expressing Dendritic Cells Promote Foxp3+ Regulatory T Cell Differentiation and Induce Less Severe Airway Inflammation in Murine Models. PLoS ONE 2016, 11, e0168919. [Google Scholar] [CrossRef] [PubMed]
- Moreau, A.; Hill, M.; Thebault, P.; Deschamps, J.Y.; Chiffoleau, E.; Chauveau, C.; Moullier, P.; Anegon, I.; Alliot-Licht, B.; Cuturi, M.C. Tolerogenic dendritic cells actively inhibit T cells through heme oxygenase-1 in rodents and in nonhuman primates. FASEB J. 2009, 23, 3070–3077. [Google Scholar] [CrossRef]
- Campbell, N.K.; Fitzgerald, H.K.; Malara, A.; Hambly, R.; Sweeney, C.M.; Kirby, B.; Fletcher, J.M.; Dunne, A. Naturally derived Heme-Oxygenase 1 inducers attenuate inflammatory responses in human dendritic cells and T cells: Relevance for psoriasis treatment. Sci. Rep. 2018, 8, 10287. [Google Scholar] [CrossRef]
- Zhao, Y.; Jia, Y.; Wang, L.; Chen, S.; Huang, X.; Xu, B.; Zhao, G.; Xiang, Y.; Yang, J.; Chen, G. Upregulation of Heme Oxygenase-1 Endues Immature Dendritic Cells With More Potent and Durable Immunoregulatory Properties and Promotes Engraftment in a Stringent Mouse Cardiac Allotransplant Model. Front. Immunol. 2018, 9, 1515. [Google Scholar] [CrossRef]
- Al-Huseini, L.M.; Yeang, H.X.A.; Hamdam, J.M.; Sethu, S.; Alhumeed, N.; Wong, W.; Sathish, J.G. Heme oxygenase-1 regulates dendritic cell function through modulation of p38 MAPK-CREB/ATF1 signaling. J. Biol. Chem. 2014, 289, 16442–16451. [Google Scholar] [CrossRef]
- Riquelme, S.A.; Pogu, J.; Anegon, I.; Bueno, S.M.; Kalergis, A.M. Carbon monoxide impairs mitochondria-dependent endosomal maturation and antigen presentation in dendritic cells. Eur. J. Immunol. 2015, 45, 3269–3288. [Google Scholar] [CrossRef]
- Tardif, V.; Riquelme, S.A.; Remy, S.; Carreno, L.J.; Cortes, C.M.; Simon, T.; Hill, M.; Louvet, C.; Riedel, C.A.; Blancou, P.; et al. Carbon monoxide decreases endosome-lysosome fusion and inhibits soluble antigen presentation by dendritic cells to T cells. Eur. J. Immunol. 2013, 43, 2832–2844. [Google Scholar] [CrossRef]
- Jung, I.D.; Lee, J.S.; Lee, C.M.; Noh, K.T.; Jeong, Y.I.; Park, W.S.; Chun, S.H.; Jeong, S.K.; Park, J.W.; Son, K.H.; et al. Induction of indoleamine 2,3-dioxygenase expression via heme oxygenase-1-dependant pathway during murine dendritic cell maturation. Biochem. Pharmacol. 2010, 80, 491–505. [Google Scholar] [CrossRef]
- Riquelme, S.A.; Bueno, S.M.; Kalergis, A.M. Carbon monoxide down-modulates Toll-like receptor 4/MD2 expression on innate immune cells and reduces endotoxic shock susceptibility. Immunology 2015, 144, 321–332. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Habtezion, A. Carbon monoxide-based therapy ameliorates acute pancreatitis via TLR4 inhibition. J. Clin. Investig. 2014, 124, 437–447. [Google Scholar] [CrossRef] [PubMed]
- George, J.F.; Braun, A.; Brusko, T.M.; Joseph, R.; Bolisetty, S.; Wasserfall, C.H.; Atkinson, M.A.; Agarwal, A.; Kapturczak, M.H. Suppression by CD4+CD25+ regulatory T cells is dependent on expression of heme oxygenase-1 in antigen-presenting cells. Am. J. Pathol. 2008, 173, 154–160. [Google Scholar] [CrossRef]
- Wu, Y.; Yu, Q.; Zhang, M.; Zhou, Y.; Su, X.; Wu, M.; Lv, J.; Xia, Z. Hemin-primed dendritic cells suppress allergic airway inflammation through releasing extracellular vesicles. J. Leukoc. Biol. 2021, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Poddighe, D.; Mathias, C.B.; Freyschmidt, E.J.; Kombe, D.; Caplan, B.; Marseglia, G.L.; Oettgen, H.C. Basophils are rapidly mobilized following initial aeroallergen encounter in naive mice and provide a priming source of IL-4 in adaptive immune responses. J. Biol. Regul. Homeost. Agents 2014, 28, 91–103. [Google Scholar]
- Miyake, K.; Karasuyama, H. Emerging roles of basophils in allergic inflammation. Allergol. Int. 2017, 66, 382–391. [Google Scholar] [CrossRef]
- Sokol, C.L.; Chu, N.Q.; Yu, S.; Nish, S.A.; Laufer, T.M.; Medzhitov, R. Basophils function as antigen-presenting cells for an allergen-induced T helper type 2 response. Nat. Immunol. 2009, 10, 713–720. [Google Scholar] [CrossRef]
- Sarfati, M.; Wakahara, K.; Chapuy, L.; Delespesse, G. Mutual Interaction of Basophils and T Cells in Chronic Inflammatory Diseases. Front. Immunol. 2015, 6, 399. [Google Scholar] [CrossRef]
- Chirumbolo, S.; Bjorklund, G.; Sboarina, A.; Vella, A. The role of basophils as innate immune regulatory cells in allergy and immunotherapy. Hum. Vaccin. Immunother. 2018, 14, 815–831. [Google Scholar] [CrossRef]
- Pae, H.O.; Oh, G.S.; Choi, B.M.; Chae, S.C.; Kim, Y.M.; Chung, K.R.; Chung, H.T. Carbon monoxide produced by heme oxygenase-1 suppresses T cell proliferation via inhibition of IL-2 production. J. Immunol. 2004, 172, 4744–4751. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, P.; Lu, J.; Xiong, W.; Oger, J.; Tetzlaff, W.; Cynader, M. Bilirubin possesses powerful immunomodulatory activity and suppresses experimental autoimmune encephalomyelitis. J. Immunol. 2008, 181, 1887–1897. [Google Scholar] [CrossRef] [PubMed]
- Grover, P.; Goel, P.N.; Greene, M.I. Regulatory T Cells: Regulation of Identity and Function. Front. Immunol. 2021, 12, 750542. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A. Regulatory T cells mediated immunomodulation during asthma: A therapeutic standpoint. J. Transl. Med. 2020, 18, 456. [Google Scholar] [CrossRef] [PubMed]
- Lan, F.; Zhang, N.; Bachert, C.; Zhang, L. Stability of regulatory T cells in T helper 2-biased allergic airway diseases. Allergy 2020, 75, 1918–1926. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, S.; Mikami, N.; Wing, J.B.; Tanaka, A.; Ichiyama, K.; Ohkura, N. Regulatory T Cells and Human Disease. Annu. Rev. Immunol. 2020, 38, 541–566. [Google Scholar] [CrossRef]
- Choi, B.M.; Pae, H.O.; Jeong, Y.R.; Kim, Y.M.; Chung, H.T. Critical role of heme oxygenase-1 in Foxp3-mediated immune suppression. Biochem. Biophys. Res. Commun. 2005, 327, 1066–1071. [Google Scholar] [CrossRef]
- Hudey, S.N.; Ledford, D.K.; Cardet, J.C. Mechanisms of non-type 2 asthma. Curr. Opin. Immunol. 2020, 66, 123–128. [Google Scholar] [CrossRef]
- Sze, E.; Bhalla, A.; Nair, P. Mechanisms and therapeutic strategies for non-T2 asthma. Allergy 2020, 75, 311–325. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, Y.; Zhong, W.; Di, C.; Lin, X.; Xia, Z. Heme oxygenase-1 ameliorates dextran sulfate sodium-induced acute murine colitis by regulating Th17/Treg cell balance. J. Biol. Chem. 2014, 289, 26847–26858. [Google Scholar] [CrossRef]
- Da Silva, E.Z.; Jamur, M.C.; Oliver, C. Mast cell function: A new vision of an old cell. J. Histochem. Cytochem. 2014, 62, 698–738. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, M.; Cervantes-Garcia, D.; Cordova-Davalos, L.E.; Perez-Rodriguez, M.J.; Gonzalez-Espinosa, C.; Salinas, E. Responses of Mast Cells to Pathogens: Beneficial and Detrimental Roles. Front. Immunol. 2021, 12, 685865. [Google Scholar] [CrossRef] [PubMed]
- Skokos, D.; Le Panse, S.; Villa, I.; Rousselle, J.C.; Peronet, R.; David, B.; Namane, A.; Mecheri, S. Mast cell-dependent B and T lymphocyte activation is mediated by the secretion of immunologically active exosomes. J. Immunol. 2001, 166, 868–876. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Chen, G. Mast cell and autoimmune diseases. Mediat. Inflamm. 2015, 2015, 246126. [Google Scholar] [CrossRef]
- Mendez-Enriquez, E.; Hallgren, J. Mast Cells and Their Progenitors in Allergic Asthma. Front. Immunol. 2019, 10, 821. [Google Scholar] [CrossRef]
- Galli, S.J.; Tsai, M. IgE and mast cells in allergic disease. Nat. Med. 2012, 18, 693–704. [Google Scholar] [CrossRef]
- Tsai, M.; Grimbaldeston, M.; Galli, S.J. Mast cells and immunoregulation/immunomodulation. Adv. Exp. Med. Biol. 2011, 716, 186–211. [Google Scholar] [CrossRef]
- Yasui, Y.; Sasao, E.; Sakata, M.; Matsui, N.; Fukuishi, N.; Akagi, R.; Akagi, M. Upregulation of heme oxygenase-1 by degranulation in rat basophilic leukemia cells. Biol. Pharm. Bull. 2007, 30, 443–446. [Google Scholar] [CrossRef]
- Yasui, Y.; Nakamura, M.; Onda, T.; Uehara, T.; Murata, S.; Matsui, N.; Fukuishi, N.; Akagi, R.; Suematsu, M.; Akagi, M. Heme oxygenase-1 inhibits cytokine production by activated mast cells. Biochem. Biophys. Res. Commun. 2007, 354, 485–490. [Google Scholar] [CrossRef]
- Ma, Y.Y.; Yang, M.Q.; Wang, C.F.; Ding, J.; Li, J.Y. Inhibiting mast cell degranulation by HO-1 affects dendritic cell maturation in vitro. Inflamm. Res. 2014, 63, 527–537. [Google Scholar] [CrossRef]
- Lee, T.H.; Song, H.J.; Park, C.S. Role of inflammasome activation in development and exacerbation of asthma. Asia Pac. Allergy 2014, 4, 187–196. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pinkerton, J.W.; Kim, R.Y.; Robertson, A.A.B.; Hirota, J.A.; Wood, L.G.; Knight, D.A.; Cooper, M.A.; O’Neill, L.A.J.; Horvat, J.C.; Hansbro, P.M. Inflammasomes in the lung. Mol. Immunol. 2017, 86, 44–55. [Google Scholar] [CrossRef] [PubMed]
- Im, H.; Ammit, A.J. The NLRP3 inflammasome: Role in airway inflammation. Clin. Exp. Allergy 2014, 44, 160–172. [Google Scholar] [CrossRef]
- Li, H.; Zhou, X.; Zhang, J. Induction of heme oxygenase-1 attenuates lipopolysaccharide-induced inflammasome activation in human gingival epithelial cells. Int. J. Mol. Med. 2014, 34, 1039–1044. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.P.; Jiang, L.; Kang, K.; Fei, D.S.; Meng, X.L.; Nan, C.C.; Pan, S.H.; Zhao, M.R.; Zhao, M.Y. Hemin inhibits NLRP3 inflammasome activation in sepsis-induced acute lung injury, involving heme oxygenase-1. Int. Immunopharmacol. 2014, 20, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Joe, Y.; Jeong, S.O.; Zheng, M.; Back, S.H.; Park, S.W.; Ryter, S.W.; Chung, H.T. Endoplasmic reticulum stress is sufficient for the induction of IL-1beta production via activation of the NF-kappaB and inflammasome pathways. Innate Immun. 2014, 20, 799–815. [Google Scholar] [CrossRef]
- Jung, S.S.; Moon, J.S.; Xu, J.F.; Ifedigbo, E.; Ryter, S.W.; Choi, A.M.; Nakahira, K. Carbon monoxide negatively regulates NLRP3 inflammasome activation in macrophages. Am. J. Physiol. Lung Cell Mol. Physiol. 2015, 308, L1058–L1067. [Google Scholar] [CrossRef]
- Jiang, L.; Fei, D.; Gong, R.; Yang, W.; Yu, W.; Pan, S.; Zhao, M.; Zhao, M. CORM-2 inhibits TXNIP/NLRP3 inflammasome pathway in LPS-induced acute lung injury. Inflamm. Res. 2016, 65, 905–915. [Google Scholar] [CrossRef]
- Kim, S.K.; Joe, Y.; Chen, Y.; Ryu, J.; Lee, J.H.; Cho, G.J.; Ryter, S.W.; Chung, H.T. Carbon monoxide decreases interleukin-1beta levels in the lung through the induction of pyrin. Cell Mol. Immunol. 2017, 14, 349–359. [Google Scholar] [CrossRef]
- Lv, J.; Su, W.; Yu, Q.; Zhang, M.; Di, C.; Lin, X.; Wu, M.; Xia, Z. Heme oxygenase-1 protects airway epithelium against apoptosis by targeting the proinflammatory NLRP3-RXR axis in asthma. J. Biol. Chem. 2018, 293, 18454–18465. [Google Scholar] [CrossRef]
- Alobaidi, A.H.; Alsamarai, A.M.; Alsamarai, M.A. Inflammation in Asthma Pathogenesis: Role of T Cells, Macrophages, Epithelial Cells and Type 2 Inflammation. Antiinflamm. Antiallergy Agents Med. Chem. 2021, 20, 317–332. [Google Scholar] [CrossRef] [PubMed]
- Saradna, A.; Do, D.C.; Kumar, S.; Fu, Q.L.; Gao, P. Macrophage polarization and allergic asthma. Transl. Res. 2018, 191, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Moreira, A.P.; Hogaboam, C.M. Macrophages in allergic asthma: Fine-tuning their pro- and anti-inflammatory actions for disease resolution. J. Interferon Cytokine Res. 2011, 31, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.; Akkoyunlu, M.; Rabin, R.L. Macrophages-common culprit in obesity and asthma. Allergy 2018, 73, 1196–1205. [Google Scholar] [CrossRef]
- Naito, Y.; Takagi, T.; Higashimura, Y. Heme oxygenase-1 and anti-inflammatory M2 macrophages. Arch. Biochem. Biophys. 2014, 564, 83–88. [Google Scholar] [CrossRef]
- Gwak, S.Y.; Kim, S.J.; Park, J.; Kim, S.H.; Joe, Y.; Lee, H.N.; Kim, W.; Muna, I.A.; Na, H.K.; Chung, H.T.; et al. Potential Role of Heme Oxygenase-1 in the Resolution of Experimentally Induced Colitis through Regulation of Macrophage Polarization. Gut Liver 2021, 16, 1–13. [Google Scholar] [CrossRef]
- Vijayan, V.; Wagener, F.; Immenschuh, S. The macrophage heme-heme oxygenase-1 system and its role in inflammation. Biochem. Pharmacol. 2018, 153, 159–167. [Google Scholar] [CrossRef]
- Zhang, M.; Nakamura, K.; Kageyama, S.; Lawal, A.O.; Gong, K.W.; Bhetraratana, M.; Fujii, T.; Sulaiman, D.; Hirao, H.; Bolisetty, S.; et al. Myeloid HO-1 modulates macrophage polarization and protects against ischemia-reperfusion injury. JCI Insight 2018, 3, e120596. [Google Scholar] [CrossRef]
- Dunn, L.L.; Midwinter, R.G.; Ni, J.; Hamid, H.A.; Parish, C.R.; Stocker, R. New insights into intracellular locations and functions of heme oxygenase-1. Antioxid. Redox Signal. 2014, 20, 1723–1742. [Google Scholar] [CrossRef]
- Tenhunen, R.; Marver, H.S.; Schmid, R. The enzymatic conversion of heme to bilirubin by microsomal heme oxygenase. Proc. Natl. Acad. Sci. USA 1968, 61, 748–755. [Google Scholar] [CrossRef]
- Linnenbaum, M.; Busker, M.; Kraehling, J.R.; Behrends, S. Heme oxygenase isoforms differ in their subcellular trafficking during hypoxia and are differentially modulated by cytochrome P450 reductase. PLoS ONE 2012, 7, e35483. [Google Scholar] [CrossRef] [PubMed]
- Wegiel, B.; Nemeth, Z.; Correa-Costa, M.; Bulmer, A.C.; Otterbein, L.E. Heme oxygenase-1: A metabolic nike. Antioxid. Redox Signal. 2014, 20, 1709–1722. [Google Scholar] [CrossRef]
- Kim, H.P.; Wang, X.; Galbiati, F.; Ryter, S.W.; Choi, A.M. Caveolae compartmentalization of heme oxygenase-1 in endothelial cells. FASEB J. 2004, 18, 1080–1089. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Li, S.; Li, C.; Cui, L.; Ma, J.; Hui, Y. The non-canonical effects of heme oxygenase-1, a classical fighter against oxidative stress. Redox Biol. 2021, 47, 102170. [Google Scholar] [CrossRef] [PubMed]
- Mascaro, M.; Alonso, E.N.; Alonso, E.G.; Lacunza, E.; Curino, A.C.; Facchinetti, M.M. Nuclear Localization of Heme Oxygenase-1 in Pathophysiological Conditions: Does It Explain the Dual Role in Cancer? Antioxidants 2021, 10, 87. [Google Scholar] [CrossRef] [PubMed]
- Dennery, P.A. Signaling function of heme oxygenase proteins. Antioxid. Redox Signal. 2014, 20, 1743–1753. [Google Scholar] [CrossRef]
- Elguero, B.; Gueron, G.; Giudice, J.; Toscani, M.A.; De Luca, P.; Zalazar, F.; Coluccio-Leskow, F.; Meiss, R.; Navone, N.; De Siervi, A.; et al. Unveiling the association of STAT3 and HO-1 in prostate cancer: Role beyond heme degradation. Neoplasia 2012, 14, 1043–1056. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Lv, J.; Ge, D.; Bai, H.; Yang, Y.; Wu, J. Heme oxygenase-1 alleviates eosinophilic inflammation by inhibiting STAT3-SOCS3 signaling. Pediatr. Pulmonol. 2020, 55, 1440–1447. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Yao, X.; Yu, R.; Bai, J.; Sun, Y.; Huang, M.; Adcock, I.M.; Barnes, P.J. Exhaled carbon monoxide in asthmatics: A meta-analysis. Respir. Res. 2010, 11, 50. [Google Scholar] [CrossRef]
- Ueno, T.; Kataoka, M.; Hirano, A.; Iio, K.; Tanimoto, Y.; Kanehiro, A.; Okada, C.; Soda, R.; Takahashi, K.; Tanimoto, M. Inflammatory markers in exhaled breath condensate from patients with asthma. Respirology 2008, 13, 654–663. [Google Scholar] [CrossRef]
- Jesenak, M.; Banovcin, P.; Havlicekova, Z.; Dobrota, D.; Babusikova, E. Factors influencing the levels of exhaled carbon monoxide in asthmatic children. J. Asthma 2014, 51, 900–906. [Google Scholar] [CrossRef] [PubMed]
- Salerno, L.; Floresta, G.; Ciaffaglione, V.; Gentile, D.; Margani, F.; Turnaturi, R.; Rescifina, A.; Pittala, V. Progress in the development of selective heme oxygenase-1 inhibitors and their potential therapeutic application. Eur. J. Med. Chem. 2019, 167, 439–453. [Google Scholar] [CrossRef] [PubMed]
- Goebel, U.; Wollborn, J. Carbon monoxide in intensive care medicine-time to start the therapeutic application?! Intensive Care Med. Exp. 2020, 8, 2. [Google Scholar] [CrossRef] [PubMed]
- Ling, K.; Men, F.; Wang, W.C.; Zhou, Y.Q.; Zhang, H.W.; Ye, D.W. Carbon Monoxide and Its Controlled Release: Therapeutic Application, Detection, and Development of Carbon Monoxide Releasing Molecules (CORMs). J. Med. Chem. 2018, 61, 2611–2635. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xia, Z.; Zhong, W. Immune Regulation of Heme Oxygenase-1 in Allergic Airway Inflammation. Antioxidants 2022, 11, 465. https://doi.org/10.3390/antiox11030465
Xia Z, Zhong W. Immune Regulation of Heme Oxygenase-1 in Allergic Airway Inflammation. Antioxidants. 2022; 11(3):465. https://doi.org/10.3390/antiox11030465
Chicago/Turabian StyleXia, Zhenwei, and Wenwei Zhong. 2022. "Immune Regulation of Heme Oxygenase-1 in Allergic Airway Inflammation" Antioxidants 11, no. 3: 465. https://doi.org/10.3390/antiox11030465
APA StyleXia, Z., & Zhong, W. (2022). Immune Regulation of Heme Oxygenase-1 in Allergic Airway Inflammation. Antioxidants, 11(3), 465. https://doi.org/10.3390/antiox11030465





