The Role of Selenium in Pathologies: An Updated Review
Abstract
1. Introduction
2. Cancer
3. Inflammatory States
4. Cardiovascular Disease
5. Thyroid Disease
6. Fertility and Reproduction
7. Bone and Skeleton Health
8. HIV
9. COVID-19
10. Cognitive Decline and Alzheimer’s Disease
11. Parkinson’s Disease
12. Schizophrenia, Anxiety and Depression
13. Type-2 Diabetes
14. A Gender Medicine Approach for Selenium-Related Health?
15. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Roman, M.; Jitaru, P.; Barbante, C. Selenium biochemistry and its role for human health. Metallomics 2014, 6, 25–54. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. Selenium and human health. Lancet 2012, 379, 1256–1268. [Google Scholar] [CrossRef]
- Flohé, L.; Brigelius-Flohé, R. Selenoproteins of the Glutathione Peroxidase Family. In Selenium. Its Molecular Biology and Role in Human Health, 3rd ed.; Hatfield, D.L., Berry, M.J., Gladyshev, V.N., Eds.; Springer: London, UK, 2012; pp. 167–180. [Google Scholar]
- Brigelius-Flohé, R.; Flohé, L. Regulatory Phenomena in the Glutathione Peroxidase Superfamily. Antioxid. Redox Signal. 2020, 33, 498–516. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zeng, H.; Cheng, W.H. Beneficial and paradoxical roles of selenium at nutritional levels of intake in healthspan and longevity. Free Radic. Biol. Med. 2018, 127, 3–13. [Google Scholar] [CrossRef]
- Benhar, M. Roles of mammalian glutathione peroxidase and thioredoxin reductase enzymes in the cellular response to nitrosative stress. Free Radic. Biol. Med. 2018, 127, 160–164. [Google Scholar] [CrossRef]
- Pothion, H.; Jehan, C.; Tostivint, H.; Cartier, D.; Bucharles, C.; Falluel-Morel, A.; Boukhzar, L.; Anouar, Y.; Lihrmann, I. Selenoprotein T: An Essential Oxidoreductase Serving as a Guardian of Endoplasmic Reticulum Homeostasis. Antioxid. Redox Signal. 2020, 33, 1257–1275. [Google Scholar] [CrossRef]
- Anouar, Y.; Lihrmann, I.; Falluel-Morel, A.; Boukhzar, L. Selenoprotein T is a key player in ER proteostasis, endocrine homeostasis and neuroprotection. Free Radic. Biol. Med. 2018, 127, 145–152. [Google Scholar] [CrossRef]
- Gromer, S.; Johansson, L.; Bauer, H.; Arscott, L.D.; Rauch, S.; Ballou, D.P.; Williams, C.H., Jr.; Schirmer, R.H.; Arnér, E.S.J. Active sites of thioredoxin reductases: Why selenoproteins? Proc. Natl. Acad. Sci. USA 2003, 100, 12618–12623. [Google Scholar] [CrossRef]
- Flohé, L. Looking Back at the Early Stages of Redox Biology. Antioxidants 2020, 9, 1254. [Google Scholar] [CrossRef]
- Bayse, C.A.; Marsan, E.S.; Garcia, J.R.; Tran-Thompson, A.T. Thyroxine binding to type III iodothyronine deiodinase. Sci. Rep. 2020, 10, 15401. [Google Scholar] [CrossRef]
- Zhong, L.; Arnér, E.S.J.; Holmgren, A. Structure and mechanism of mammalian thioredoxin reductase: The active site is a redox-active selenolthiolyselenenylsulfide formed from the conserved cysteine-selenocysteine sequence. Proc. Natl. Acad. Sci. USA 2000, 97, 5854–5859. [Google Scholar] [CrossRef] [PubMed]
- Tanini, D.; Capperucci, A. Synthesis and applications of organic selenols. Adv. Synth. Catal. 2021, 363, 5360–5385. [Google Scholar] [CrossRef]
- Dalla Tiezza, M.; Bickelhaupt, F.M.; Flohe, L.; Orian, L. Proton Transfer and SN2 Reactions as Steps of Fast Selenol and Thiol Oxidation in Proteins: A Model Molecular Study Based on GPx. ChemPlusChem 2021, 86, 525–532. [Google Scholar] [CrossRef] [PubMed]
- Randi, E.B.; Casili, G.; Jacquemai, S.; Szabo, C. Selenium-Binding Protein 1 (SELENBP1) Supports Hydrogen Sulfide Biosynthesis and Adipogenesis. Antioxidants 2021, 10, 361. [Google Scholar] [CrossRef]
- Elhodaky, M.; Hong, L.K.; Kadkol, S.; Diamond, A.M. Selenium-binding protein 1 alters energy metabolism in prostate cancer cells. Prostate 2020, 80, 962–976. [Google Scholar] [CrossRef]
- Dóka, É.; Ida, T.; Dagnell, M.; Abiko, Y.; Luong, N.C.; Balog, N.; Takata, T.; Espinosa, B.; Nishimura, A.; Cheng, Q.; et al. Control of protein function through oxidation and reduction of persulfidated states. Sci. Adv. 2020, 6, eaax8358. [Google Scholar] [CrossRef] [PubMed]
- Wenzhong, W.; Tong, Z.; Hongjin, L.; Ying, C.; Jun, X. Role of Hydrogen Sulfide on Autophagy in Liver Injuries Induced by Selenium Deficiency in Chickens. Biol. Trace Elem. Res. 2017, 175, 194–203. [Google Scholar] [CrossRef]
- Zheng, S.F.; Bao, R.K.; Zhang, Q.J.; Wang, S.C.; Lin, H.J. Endogenous Hydrogen Sulfide Promotes Apoptosis via Mitochondrial Pathways in the Livers of Broilers with Selenium Deficiency Exudative Diathesis Disease. Biol. Trace Elem. Res. 2018, 186, 249–257. [Google Scholar] [CrossRef]
- Wu, C.; Xu, Z.; Huang, K. Effects of Dietary Selenium on Inflammation and Hydrogen Sulfide in the Gastrointestinal Tract in Chickens. Biol. Trace Elem. Res. 2016, 174, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Elhodaky, M.; Diamond, A.M. Selenium-Binding Protein 1 in Human Health and Disease. Int. J. Mol. Sci. 2018, 19, 3437. [Google Scholar] [CrossRef]
- Steinbrenner, H.; Micoogullari, M.; Hoang, N.A.; Bergheim, I.; Klotz, L.O.; Sies, H. Selenium-binding protein 1 (SELENBP1) is a marker of mature adipocytes. Redox Biol. 2019, 20, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Kuganesan, M.; Samra, K.; Evans, E.; Singer, M.; Dyson, A. Selenium and hydrogen selenide: Essential micronutrient and the fourth gasotransmitter? Intensive Care Med. Exp. 2019, 7, 71. [Google Scholar] [CrossRef] [PubMed]
- Samra, K.; Kuganesan, M.; Smith, W.; Kleyman, A.; Tidswell, R.; Arulkumaran, N.; Singer, M.; Dyson, A. The Pharmacology and Therapeutic Utility of Sodium Hydroselenide. Int. J. Mol. Sci. 2021, 22, 3258. [Google Scholar] [CrossRef] [PubMed]
- Greasley, A.; Zhang, Y.; Wu, B.; Pei, Y.; Belzile, N.; Yang, G. H2S Protects against Cardiac Cell Hypertrophy through Regulation of Selenoproteins. Oxid. Med. Cell. Long. 2019, 2019, 6494306. [Google Scholar] [CrossRef] [PubMed]
- Grman, M.; Misak, A.; Kurakova, L.; Brezova, V.; Cacanyiova, S.; Berenyiova, A.; Balis, P.; Tomasova, L.; Kharma, A.; Domínguez-Álvarez, E.; et al. Products of Sulfide/Selenite Interaction Possess Antioxidant Properties, Scavenge Superoxide-Derived Radicals, React with DNA, and Modulate Blood Pressure and Tension of Isolated Thoracic Aorta. Oxid. Med. Cell. Long. 2019, 2019, 9847650. [Google Scholar] [CrossRef]
- Schomburg, L. Variations in Selenium Metabolism in Males and Females. In Selenium. Its Molecular Biology and Role in Human Health, 3rd ed.; Hatfield, D.L., Berry, M.J., Gladyshev, V.N., Eds.; Springer: London, UK, 2012; pp. 419–432. [Google Scholar]
- Golubkina, N.; Moldovan, A.; Kekina, H.; Kharchenko, V.; Sekara, A.; Vasileva, V.; Skrypnik, L.; Tallarita, A.; Caruso, G. Joint Biofortification of Plants with Selenium and Iodine: New Field of Discoveries. Plants 2021, 10, 1352. [Google Scholar] [CrossRef]
- Hossain, A.; Skalicky, M.; Brestic, M.; Maitra, S.; Sarkar, S.; Ahmad, Z.; Vemuri, H.; Garai, S.; Mondal, M.; Bhatt, R.; et al. Selenium Biofortification: Roles, Mechanisms, Responses and Prospects. Molecules 2021, 26, 881. [Google Scholar] [CrossRef]
- Dong, Z.; Liu, Y.; Dong, G.; Wu, H. Effect of boiling and frying on the selenium content, speciation, and in vitro bioaccessibility of selenium-biofortified potato (Solanum tuberosum L.). Food Chem. 2021, 348, 129150. [Google Scholar] [CrossRef]
- D’Amato, R.M.; Regni, L.; Falcinelli, B.; Mattioli, S.; Benincasa, P.; Dal Bosco, A.; Pacheco, P.; Proietti, P.; Troni, E.; Santi, C.; et al. Current Knowledge on Selenium Biofortification to Improve the Nutraceutical Profile of Food: A Comprehensive Review. J. Agric. Food Chem. 2020, 68, 4075–4097. [Google Scholar] [CrossRef]
- Giacosa, A.; Faliva, M.A.; Perna, S.; Minoia, C.; Ronchi, A.; Rondanelli, M. Selenium Fortification of an Italian Rice Cultivar via Foliar Fertilization with Sodium Selenate and Its Effects on HumanSerum Selenium Levels and on Erythrocyte GlutathionePeroxidase Activity. Nutrients 2014, 6, 1251–1261. [Google Scholar] [CrossRef]
- Filippini, T.; Michalke, B.; Wise, L.A.; Malagoli, C.; Malavolti, M.; Vescovi, L.; Salvia, C.; Bargellini, A.; Sieri, S.; Krogh, V.; et al. Dietcomposition and serum levels of selenium species: A cross-sectional study. Food Chem. Toxicol. 2018, 115, 482–490. [Google Scholar] [CrossRef] [PubMed]
- Jablonska, E.; Vinceti, M. Selenium and Human Health: Witnessing a Copernican Revolution? J. Environ. Sci. Health C. Environ. Carcinog. Ecotoxicol. Rev. 2015, 33, 328–368. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, C.W.; Barbosa, N.V.; Rocha, J.B.T. Toxicology and pharmacology of synthetic organoselenium compounds: An update. Arch. Toxicol. 2021, 95, 1179–1226. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, C.W.; Zeni, G.; Rocha, J.B. Organoselenium and organotellurium compounds: Toxicology and pharmacology. Chem. Rev. 2004, 104, 6255–6285. [Google Scholar] [CrossRef] [PubMed]
- Human Vitamin and Mineral Requirements. Report of a Joint FAO/WHO Expert Consultation, Bangkok, Thailand, 2nd ed.; Food and Agriculture Organization of the United Nations—World Health Organization, Ed.; Food and Agriculture Organization (FAO): Geneva, Switzerland, 2004; Chapter 10; p. 194. [Google Scholar]
- Hu, W.; Zhao, C.; Hu, H.; Yin, S. Food Sources of Selenium and Its Relationship with Chronic Diseases. Nutrients 2021, 13, 1739. [Google Scholar] [CrossRef]
- Angeli, A.; Pinteala, M.; Maier, S.S.; Simionescu, B.C.; Milaneschi, A.; Abbas, G.; del Prete, S.; Capasso, C.; Capperucci, A.; Tanini, D.; et al. Evaluation of Thio- and Seleno-Acetamides Bearing Benzenesulfonamide as Inhibitor of Carbonic Anhydrases from Different Pathogenic Bacteria. Int. J. Mol. Sci. 2020, 21, 598. [Google Scholar] [CrossRef]
- Ren, X.; Zou, L.; Lu, J.; Holmgren, A. Selenocysteine in mammalian thioredoxin reductase and application of ebselen as a therapeutic. Free Radic. Biol. Med. 2018, 127, 238–247. [Google Scholar] [CrossRef]
- Capperucci, A.; Petrucci, A.; Faggi, C.; Tanini, D. Click Reaction of Selenols with Isocyanates: Rapid Access to Selenocarbamates as Peroxide-Switchable Reservoir of Thiol-peroxidase-Like Catalysts. Adv. Sinth. Catal. 2021, 363, 4256–4263. [Google Scholar] [CrossRef]
- Capperucci, A.; Coronnello, M.; Salvini, F.; Tanini, D.; Dei, S.; Teodori, E.; Giovannelli, L. Synthesis of functionalised organochalcogenides and in vitro evaluation of their antioxidant activity. Bioorg. Chem. 2021, 110, 104812. [Google Scholar] [CrossRef]
- Angeli, A.; Carta, F.; Donnini, S.; Capperucci, A.; Ferraroni, M.; Tanini, D.; Supuran, C.T. Selenolesterase enzyme activity of carbonic anhydrases. Chem. Commun. 2020, 56, 4444–4447. [Google Scholar] [CrossRef]
- Tanini, D.; Capperucci, A.; Ferraroni, M.; Carta, F.; Angeli, A.; Supuran, C.T. Direct and straightforward access to substituted alkyl selenols as novel carbonic anhydrase inhibitors. Eur. J. Med. Chem. 2020, 185, 111811. [Google Scholar] [CrossRef] [PubMed]
- Tanini, D.; Capperucci, A.; Supuran, C.T.; Angeli, A. Sulfur, selenium and tellurium containing amines act as effective carbonic anhydrase activators. Bioorganic Chem. 2019, 87, 516–522. [Google Scholar] [CrossRef] [PubMed]
- Tanini, D.; Lupori, B.; Malevolti, G.; Ambrosi, M.; Lo Nostro, P.; Capperucci, A. Direct biocatalysed synthesis of first sulfur-, selenium- and tellurium- containing L-ascorbyl hybrid derivatives with radical trapping and GPx-like properties. Chem. Commun. 2019, 55, 5705–5708. [Google Scholar] [CrossRef] [PubMed]
- Mugesh, G.; Singh, H.B. Synthetic organoselenium compounds as antioxidants: Glutathione peroxidase activity. Chem. Soc. Rev. 2000, 29, 347–357. [Google Scholar] [CrossRef]
- Lenardão, E.J.; Santi, C.; Sancineto, L. New Frontiers in Organoselenium Compounds; Springer: New York, NY, USA, 2018. [Google Scholar]
- Organoselenium Compounds in Biology and Medicine: Synthesis, Biological and Therapeutic Treatments; Jain, V.K., Priyadarsini, K.I., Eds.; Royal Society of Chemistry: London, UK, 2018. [Google Scholar]
- Shamberger, R.J.; Rudolph, G. Protection against cocarcinogenesis by antioxidants. Experientia 1966, 22, 116. [Google Scholar] [CrossRef] [PubMed]
- Shamberger, R.J.; Frost, D.V. Possible protective effect of Selenium against human cancer. Can. Med. Assoc. J. 1969, 100, 682. [Google Scholar]
- Shamberger, R.J.; Willis, C.E. Selenium distribution and human cancer mortality. Crit. Rev. Clin. Lab. Sci. 1971, 2, 211–221. [Google Scholar] [CrossRef]
- Clark, L.C.; Combs, G.F., Jr.; Turnbull, B.W.; Slate, E.H.; Chalker, D.K.; Chow, J.; Davis, L.S.; Glover, R.A.; Graham, G.F.; Gross, E.G.; et al. Effects of Selenium supplementation for cancer prevention in patients with carcinoma of the skin. A randomized controlled trial. Nutritional Prevention of Cancer Study Group. JAMA 1996, 276, 1957–1963. [Google Scholar] [CrossRef]
- Combs, G.F., Jr.; Lü, J. Selenium as an Anticancer Agent. In Selenium. Its Molecular Biology and Role in Human Health, 2nd ed.; Hatfield, D.L., Berry, M.J., Gladyshev, V.N., Eds.; Kluwer Academic: New York, NY, USA, 2006; p. 205. [Google Scholar]
- Radomska, D.; Czarnomysy, R.; Radomski, D.; Bielawska, A.; Bielawski, K. Selenium as a Bioactive Micronutrient in the Human Diet and Its Cancer Chemopreventive Activity. Nutrients 2021, 13, 1649. [Google Scholar] [CrossRef]
- Jackson, M.I.; Combs, G.F., Jr. Selenium as a Cancer Preventive Agent. In Selenium. Its Molecular Biology and Role in Human Health, 3rd ed.; Hatfield, D.L., Berry, M.J., Gladyshev, V.N., Eds.; Springer: London, UK, 2012; pp. 313–323. [Google Scholar]
- Wright, M.E.; Diamond, A.M. Polymorphisms in Selenoprotein Genes and Cancer. In Selenium. Its Molecular Biology and Role in Human Health, 3rd ed.; Hatfield, D.L., Berry, M.J., Gladyshev, V.N., Eds.; Springer: London, UK, 2012; pp. 345–354. [Google Scholar]
- Petronek, M.S.; Stolwijk, J.M.; Murray, S.D.; Steinbach, E.J.; Zakharia, Y.; Buettner, G.R.; Spitz, D.R.; Allen, B.G. Utilization of redox modulating small molecules that selectively act as pro-oxidants in cancer cells to open a therapeutic window for improving cancer therapy. Redox Biol. 2021, 42, 101864. [Google Scholar] [CrossRef]
- Peters, K.M.; Carlson, B.A.; Gladyshev, V.N.; Tsuji, P.A. Selenoproteins in colon cancer. Free Radic. Biol. Med. 2018, 127, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Bertz, M.; Kühn, K.; Koeberle, S.C.; Müller, M.F.; Hoelzer, D.; Thies, K.; Deubel, S.; Thierbach, R.; Kipp, A.P. Selenoprotein H controls cell cycle progression and proliferation of human colorectal cancer cells. Free Radic. Biol. Med. 2018, 127, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Scalcon, V.; Bindoli, A.; Rigobello, M.P. Significance of the mitochondrial thioredoxin reductase in cancer cells: An update on role, targets and inhibitors. Free Radic. Biol. Med. 2018, 127, 62–79. [Google Scholar] [CrossRef] [PubMed]
- Reich, H.J.; Hondal, R.J. Why nature chose Selenium? ACS Chem. Biol. 2016, 11, 821–841. [Google Scholar] [CrossRef] [PubMed]
- Tanini, D.; Carradori, S.; Capperucci, A.; Lupori, L.; Zara, S.; Ferraroni, M.; Ghelardini, C.; Di Cesare Mannelli, L.; Micheli, L.; Lucarini, E.; et al. Chalcogenides-incorporating carbonic anhydrase inhibitors concomitantly reverted oxaliplatin-induced neuropathy and enhanced antiproliferative action. Eur. J. Med. Chem. 2021, 225, 113793. [Google Scholar] [CrossRef]
- Gandin, V.; Khalkar, P.; Braude, J.; Fernandes, A.P. Organic selenium compounds as potential chemotherapeutic agents for improved cancer treatment. Free Radic. Biol. Med. 2018, 127, 80–97. [Google Scholar] [CrossRef]
- Gopalakrishna, R.; Gundimeda, U.; Zhou, S.; Bui, H.; Holmgren, A. Redox regulation of protein kinase C by selenometabolites and selenoprotein thioredoxin reductase limits cancer prevention by selenium. Free Radic. Biol. Med. 2018, 127, 55–61. [Google Scholar] [CrossRef]
- Kadkol, S.; Diamond, A.M. The Interaction between Dietary Selenium Intake and Genetics in Determining Cancer Risk and Outcome. Nutrients 2020, 12, 2424. [Google Scholar] [CrossRef]
- Brigelius-Flohé, R.; Kipp, A.P. Glutathione Peroxidase 2, a Selenoprotein Exhibiting a Dual Personality in Preventing and Promoting Cancer. In Selenium. Its Molecular Biology and Role in Human Health, 4th ed.; Hatfield, D.L., Schweizer, U., Tsuji, P.A., Gladyshev, V.N., Eds.; Springer International Publishing: New York, NY, USA, 2016; Chapter 38; pp. 451–462. [Google Scholar]
- National Institute of Health, Office of Dietary Supplements. Selenium. Fact Sheet for Health Professionals. Updated: 26 March 2021. Available online: https://ods.od.nih.gov/factsheets/Selenium-HealthProfessional (accessed on 12 August 2021).
- Yu, S.Y.; Zhu, Y.J.; Li, W.G. Protective role of selenium against hepatitis B virus and primary liver cancer in Qidong. Biol. Trace Elem. Res. 1997, 56, 117–124. [Google Scholar] [CrossRef]
- Yu, S.Y.; Zhu, Y.J.; Li, W.G.; Huang, Q.S.; Huang, C.Z.; Zhang, Q.N.; Hou, C. A preliminary report on the intervention trials of primary liver cancer in high-risk populations with nutritional supplementation of Selenium in China. Biol. Trace Elem. Res. 1991, 29, 289–294. [Google Scholar] [CrossRef]
- Taylor, P.R.; Li, B.; Dawsey, S.M.; Li, J.Y.; Yang, C.S.; Guo, W.; Blot, W.J. Prevention of esophageal cancer: The nutrition intervention trials in Linxian, China. Linxian Nutrition Intervention Trials Study Group. Cancer Res. 1994, 54, 2029s–2031s. [Google Scholar] [PubMed]
- Qiao, Y.L.; Dawsey, S.M.; Kamangar, F.; Fan, J.H.; Abnet, C.C.; Sun, X.D.; Johnson, L.L.; Gail, M.H.; Dong, Z.W.; Yu, B.; et al. Total and cancer mortality after supplementation with vitamins and minerals: Follow-up of the Linxian General Population Nutrition Intervention Trial. J. Natl. Cancer Inst. 2009, 101, 507–518. [Google Scholar] [CrossRef] [PubMed]
- Krishnaswamy, K.; Prasad, M.P.; Krishna, T.P.; Annapurna, V.V.; Reddy, G.A. A case study of nutrient intervention of oral precancerous lesions in India. Eur. J. Cancer. B Oral Oncol. 1995, 31B, 41–48. [Google Scholar] [CrossRef]
- Prasad, M.P.; Mukundan, M.A.; Krishnaswamy, K. Micronuclei and carcinogen DNA adducts as intermediate end points in nutrient intervention trial of precancerous lesions in the oral cavity. Eur. J. Cancer. B Oral Oncol. 1995, 31B, 155–159. [Google Scholar] [CrossRef]
- Lippman, S.M.; Klein, E.A.; Goodman, P.J.; Lucia, M.S.; Thompson, I.M.; Ford, L.G.; Parnes, H.L.; Minasian, L.M.; Gaziano, J.M.; Hartline, J.A.; et al. Effect of Selenium and vitamin E on risk of prostate cancer and other cancers: The Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA 2009, 301, 39–51. [Google Scholar] [CrossRef]
- Meyer, F.; Galan, P.; Douville, P.; Bairati, I.; Kegle, P.; Bertrais, S.; Estaquio, C.; Hercberg, S. Antioxidant vitamin and mineral supplementation and prostate cancer prevention in the SU.VI.MAX trial. Int. J. Cancer 2005, 116, 182–186. [Google Scholar] [CrossRef]
- Jackson, M.I.; Combs, G.F., Jr. Selenium and anticarcinogenesis: Underlying mechanisms. Curr. Opin. Clin. Nutr. Metab. Care 2008, 11, 718–726. [Google Scholar] [CrossRef]
- Hsueh, Y.M.; Su, C.T.; Shiue, H.S.; Chen, W.J.; Pu, Y.S.; Lin, Y.C.; Tsai, C.S.; Huang, C.Y. Levels of plasma Selenium and urinary total arsenic interact to affect the risk for prostate cancer. Food Chem. Toxicol. 2017, 107, 167–175. [Google Scholar] [CrossRef]
- Dunn, B.D.; Taylor, P.R. Prostate Cancer Prevention and the Selenium and Vitamin E Cancer Prevention Trial (SELECT): A Selenium Perspective. In Selenium. Its Molecular Biology and Role in Human Health, 3rd ed.; Hatfield, D.L., Berry, M.J., Gladyshev, V.N., Eds.; Springer: London, UK, 2012; pp. 297–311. [Google Scholar]
- Strope, S.A.; Andriole, G.L. Update on chemoprevention for prostate cancer. Curr. Opin. Urol. 2010, 20, 194–197. [Google Scholar] [CrossRef]
- Grammatikopoulou, M.G.; Gkiouras, K.; Papageorgiou, S.Τ.; Myrogiannis, I.; Mykoniatis, I.; Papamitsou, T.; Bogdanos, D.P.; Goulis, D.G. Dietary Factors and Supplements Influencing Prostate Specific-Antigen (PSA) Concentrations in Men with Prostate Cancer and Increased Cancer Risk: An Evidence Analysis Review Based on Randomized Controlled Trials. Nutrients 2020, 12, 2985. [Google Scholar] [CrossRef]
- Kuria, A.; Fang, X.; Li, M.; Han, H.; He, J.; Aaseth, J.O.; Cao, Y. Does dietary intake of selenium protect against cancer? A systematic review and meta-analysis of population-based prospective studies. Crit. Rev. Food Sci. Nutr. 2020, 60, 684–694. [Google Scholar] [CrossRef] [PubMed]
- Karunasinghe, N.; Ng, L.; Wang, A.; Vaidyanathan, V.; Zhu, S.; Ferguson, L.R. Selenium Supplementation and Prostate Health in a New Zealand Cohort. Nutrients 2019, 12, 2. [Google Scholar] [CrossRef] [PubMed]
- Evans, S.O.; Jacobson, G.M.; Goodman, H.; Bird, S.; Jameson, M.B. Comparison of three oral selenium compounds in cancer patients: Evaluation of differential pharmacodynamic effects in normal and malignant cells. J. Trace Elem. Med. Biol. 2020, 58, 126446. [Google Scholar] [CrossRef] [PubMed]
- Evans, S.O.; Jacobson, G.M.; Goodman, H.; Bird, S.; Jameson, M.B. Comparative Safety and Pharmacokinetic Evaluation of Three Oral Selenium Compounds in Cancer Patients. Biol. Trace Elem. Res. 2019, 189, 395–404. [Google Scholar] [CrossRef]
- Kuršvietienė, L.; Mongirdienė, A.; Bernatonienė, J.; Šulinskienė, J.; Stanevičienė, I. Selenium Anticancer Properties and Impact on Cellular Redox Status. Antioxidants 2020, 9, 80. [Google Scholar] [CrossRef] [PubMed]
- Vinceti, M.; Filippini, T.; Cilloni, S.; Crespi, C.M. The Epidemiology of Selenium and Human Cancer. Adv. Cancer Res. 2017, 136, 1–48. [Google Scholar] [PubMed]
- Vernia, F.; Longo, S.; Stefanelli, G.; Viscido, A.; Latella, G. Dietary Factors Modulating Colorectal Carcinogenesis. Nutrients 2021, 13, 143. [Google Scholar] [CrossRef]
- Short, S.P.; Pilat, J.M.; Williams, C.S. Roles for selenium and selenoprotein P in the development, progression, and prevention of intestinal disease. Free Radic. Biol. Med. 2018, 127, 26–35. [Google Scholar] [CrossRef]
- Zhu, X.; Pan, D.; Wang, N.; Wang, S.; Sun, G. Relationship between Selenium in Human Tissues and Breast Cancer: A Meta-analysis Based on Case-Control Studies. Biol. Trace Elem Res. 2021, 199, 4439–4446. [Google Scholar] [CrossRef]
- Bevinakoppamath, S.; Saleh Ahmed, A.M.; Ramachandra, S.C.; Vishwanath, P.; Prashant, A. Chemopreventive and Anticancer Property of Selenoproteins in Obese Breast Cancer. Front. Pharmacol. 2021, 12, 618172. [Google Scholar] [CrossRef]
- Barrea, L.; Gallo, M.; Ruggeri, R.M.; Di Giacinto, P.; Sesti, F.; Prinzi, N.; Adinolfi, V.; Barucca, V.; Renzelli, V.; Muscogiuri, G.; et al. Nutritional status and follicular-derived thyroid cancer: An update. Crit. Rev. Food Sci. Nutr. 2020, 61, 25–59. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira Maia, M.; Batista, B.; Sousa, M.P.; de Souza, L.M.; Maia, C. Selenium and thyroid cancer: A systematic review. Nutr. Cancer 2020, 72, 1255–1263. [Google Scholar] [CrossRef] [PubMed]
- Kipp, A.P. Selenium in colorectal and differentiated thyroid cancer. Hormones 2020, 19, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Jablonska, E.; Li, Q.; Reszka, E.; Wieczorek, E.; Tarhonska, K.; Wang, T. Therapeutic Potential of Selenium and Selenium Compounds in Cervical Cancer. Cancer Control 2021, 28, 10732748211001808. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Kumaran, M.N.; Gounder, M.; Gibbon, D.G.; Nieves-Neira, W.; Vaidya, A.; Hellmann, M.; Kane, M.P.; Buckley, B.; Shih, W.; et al. Phase I trial of selenium plus chemotherapy in gynecologic cancers. Gynecol. Oncol. 2018, 150, 478–486. [Google Scholar] [CrossRef]
- Jamier, V.; Ba, L.A.; Jacob, C. Selenium- and Tellurium-Containing Multifunctional Redox Agents as Biochemical Redox Modulators with Selective Cytotoxicity. Chem. Eur. J. 2010, 16, 10920–10928. [Google Scholar] [CrossRef]
- Lee, K.H.; Jeong, D. Bimodal actions of selenium essential for antioxidant and toxic pro-oxidant activities: The selenium paradox (Review). Mol. Med. Rep. 2012, 5, 299–304. [Google Scholar]
- Ganther, H.E. Selenium metabolism, selenoproteins and mechanisms of cancer prevention: Complexities with thioredoxin reductase. Carcinogenesis 1999, 20, 1657–1666. [Google Scholar] [CrossRef]
- Jeong, D.W.; Yoo, M.H.; Kim, T.S.; Kim, J.H.; Kim, I.Y. Protection of mice from allergen-induced asthma by selenite: Prevention of eosinophil infiltration by inhibition of NF-kappa B activation. J. Biol. Chem. 2002, 277, 17871–17876. [Google Scholar] [CrossRef]
- Kim, J.Y.; Park, H.S.; Kang, S.I.; Choi, E.J.; Kim, I.Y. Redox regulation of cytosolic glycerol-3-phosphate dehydrogenase: Cys (102) is the target of the redox control and essential for the catalytic activity. Biochim. Biophys. Acta 2002, 1569, 67–74. [Google Scholar] [CrossRef]
- Ghosh, J. Rapid induction of apoptosis in prostate cancer cells by selenium: Reversal by metabolites of arachidonate 5-lipoxygenase. Biochem. Biophys. Res. Commun. 2004, 315, 624–635. [Google Scholar] [CrossRef] [PubMed]
- Park, H.S.; Huh, S.H.; Kim, Y.; Shim, J.; Lee, S.H.; Parki, I.S.; Jung, Y.K.; Kim, I.Y.; Choi, E.J. Selenite negatively regulates caspase-3 through a redox mechanism. J. Biol. Chem. 2000, 275, 8487–8491. [Google Scholar] [CrossRef] [PubMed]
- Park, H.S.; Park, E.; Kim, M.S.; Ahn, K.; Kim, I.Y.; Choi, E.J. Selenite inhibits the c-Jun N-terminal kinase/stress-activated protein kinase (JNK/SAPK) through a thiol redox mechanism. J. Biol. Chem. 2000, 275, 2527–2531. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.S.; Yun, B.Y.; Kim, I.Y. Induction of the mitochondrial permeability transition by selenium compounds mediated by oxidation of the protein thiol groups and generation of the superoxide. Biochem. Pharmacol. 2003, 66, 2301–2311. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Alarcon, M.; Cabrera-Vique, C. Selenium in food and the human body: A review. Sci. Total Environ. 2008, 400, 115–141. [Google Scholar] [CrossRef]
- Wallenberg, M.; Misra, S.; Björnstedt, M. Selenium cytotoxicity in cancer. Basic Clin. Pharmacol. Toxicol. 2014, 114, 377–386. [Google Scholar] [CrossRef]
- Jiang, Z.; Chi, J.; Li, H.; Wang, Y.; Liu, W.; Han, B. Effect of chitosanoligosaccharide-conjugated Selenium on improving immune function and blocking gastric cancer growth. Eur. J. Pharmacol. 2020, 891, 173673. [Google Scholar] [CrossRef]
- Yakubov, E.; Eibl, T.; Hammer, A.; Holtmannspötter, M.; Savaskan, N.; Steiner, H.H. Therapeutic Potential of Selenium in Glioblastoma. Front. Neurosci. 2021, 15, 666679. [Google Scholar] [CrossRef]
- Kaushal, N.; Gandhi, U.H.; Nelson, S.M.; Narayan, V.; Sandeep Prabhu, K. Selenium and Inflammation. In Selenium. Its Molecular Biology and Role in Human Health, 3rd ed.; Hatfield, D.L., Berry, M.J., Gladyshev, V.N., Eds.; Springer: London, UK, 2012; pp. 443–456. [Google Scholar]
- Maehira, F.; Luyo, G.A.; Miyagi, I.; Oshiro, M.; Yamane, N.; Kuba, M.H.; Nakazato, Y. Alterations of serum Selenium concentrations in the acute phase of pathological conditions. Clin. Chim. Acta 2002, 316, 137–146. [Google Scholar] [CrossRef]
- Sakr, Y.; Reinhart, K.; Bloos, F.; Marx, G.; Russwurm, S.; Bauer, M.; Brunkhorst, F. Time course and relationship between plasma Selenium concentrations, systemic inflammatory response, sepsis, and multiorganfailure. Br. J. Anaesth. 2007, 98, 775–784. [Google Scholar] [CrossRef]
- Angstwurm, M.W.; Engelmann, L.; Zimmermann, T.; Lehmann, C.; Spes, C.H.; Abel, P.; Strauss, R.; Meier-Hellmann, A.; Insel, R.; Radke, J.; et al. Selenium in Intensive Care (SIC): Results of a prospective randomized, placebo-controlled, multiple-center study in patients with severe systemic inflammatory response syndrome, sepsis, and septic shock. Crit. Care Med. 2007, 35, 118. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.; Lv, Z.; Zhang, Y.; Wang, Y.; Qiao, X.; Sun, C.; Chen, Y.; Guo, M.; Han, W.; Ye, A.; et al. Precision Redox: The Key for Antioxidant Pharmacology. Antioxid. Redox Signal. 2021, 34, 1069–1082. [Google Scholar] [CrossRef] [PubMed]
- Frank, D.H.; Roe, D.J.; Chow, H.H.; Guillen, J.M.; Choquette, K.; Gracie, D.; Francis, J.; Fish, A.; Alberts, D.S. Effects of a high-Selenium yeast supplement on celecoxib plasma levels: A randomized phase II trial. Cancer Epidemiol. Biomark. Prev. 2004, 13, 299–303. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Desai, D.; Kaushal, N.; Gandhi, U.H.; Arner, R.J.; D’Souza, C.; Chen, G.; Vunta, H.; El-Bayoumy, K.; Amin, S.; Prabhu, K.S. Synthesis and evaluation of the anti-inflammatory properties of Selenium-derivatives of celecoxib. Chem. Biol. Interact. 2010, 188, 446–456. [Google Scholar] [CrossRef]
- Vaghari-Tabari, M.; Jafari-Gharabaghlou, D.; Sadeghsoltani, F.; Hassanpour, P.; Qujeq, D.; Rashtchizadeh, N.; Ghorbanihaghjo, A. Zinc and Selenium in Inflammatory Bowel Disease: Trace Elements with Key Roles? Biol. Trace Elem. Res. 2021, 199, 3190–3204. [Google Scholar] [CrossRef]
- Turrubiates-Hernández, F.J.; Márquez-Sandoval, Y.F.; González-Estevez, G.; Reyes-Castillo, Z.; Muñoz-Valle, J.F. The Relevance of Selenium Status in Rheumatoid Arthritis. Nutrients 2020, 12, 3007. [Google Scholar] [CrossRef]
- Qin, J.; Huang, X.; Wang, N.; Zhou, P.; Zhang, H.; Chen, Z.; Liang, K.; Gong, D.; Zeng, Q.; Niu, P.; et al. Supranutritional selenium suppresses ROS-induced generation of RANKL-expressing osteoclastogenic CD4+ T cells and ameliorates rheumatoid arthritis. Clin. Transl. Immunol. 2021, 10, e1338. [Google Scholar] [CrossRef]
- Zhao, S.; Cheng, C.K.; Zhang, C.L.; Huang, Y. Interplay between Oxidative Stress, Cyclooxygenases, and Prostanoids in Cardiovascular Diseases. Antioxid. Redox Signal. 2021, 34, 784–799. [Google Scholar] [CrossRef]
- Brigelius-Flohé, R.; Banning, A.; Schnurr, K. Selenium-dependent enzymes in endothelial cell function. Antioxid. Redox Signal. 2003, 5, 205–215. [Google Scholar] [CrossRef]
- Zachariah, M.; Maamoun, H.; Milano, L.; Rayman, M.P.; Meira, L.B.; Agouni, A. Endoplasmic reticulum stress and oxidative stress drive endothelial dysfunction induced by high selenium. J. Cell. Physiol. 2021, 236, 4348–4359. [Google Scholar] [CrossRef]
- Prabhu, K.S.; Zamamiri-Davis, F.; Stewart, J.B.; Thompson, J.T.; Sordillo, L.M.; Reddy, C.C. Selenium deficiency increases the expression of inducible nitric oxide synthase in RAW 264.7 macrophages: Role of nuclear factor-kappaB in up-regulation. Biochem. J. 2002, 366, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Cui, J.; Lu, Y.; Huang, C.; Liu, H.; Xu, S. Selenium Deficiency Induces Inflammation via the iNOS/NF-κB Pathway in the Brain of Pigs. Biol. Trace Elem. Res. 2020, 196, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Casaril, A.M.; Domingues, M.; Bampi, S.R.; de Andrade Lourenço, D.; Padilha, N.B.; Lenardão, E.J.; Sonego, M.; Seixas, F.K.; Collares, T.; Nogueira, C.W.; et al. The selenium-containing compound 3-((4-chlorophenyl)selanyl)-1-methyl-1H-indole reverses depressive-like behavior induced by acute restraint stress in mice: Modulation of oxido-nitrosative stress and inflammatory pathway. Psychopharmacology 2019, 236, 2867–2880. [Google Scholar] [CrossRef] [PubMed]
- Ge, J.; Guo, K.; Zhang, C.; Talukder, M.; Lv, M.W.; Li, J.Y.; Li, J.L. Comparison of nanoparticle-selenium, selenium-enriched yeast and sodium selenite on the alleviation of cadmium-induced inflammation via NF-κB/IκB pathway in heart. Sci. Total Environ. 2021, 773, 145442. [Google Scholar] [CrossRef] [PubMed]
- Fan, R.F.; Liu, J.X.; Yan, Y.X.; Wang, L.; Wang, Z.Y. Selenium relieves oxidative stress, inflammation, and apoptosis within spleen of chicken exposed to mercuric chloride. Poult. Sci. 2020, 99, 5430–5439. [Google Scholar] [CrossRef]
- Chomchan, R.; Puttarak, P.; Brantner, A.; Siripongvutikorn, S. Selenium-Rich Ricegrass Juice Improves Antioxidant Properties and Nitric Oxide Inhibition in Macrophage Cells. Antioxidants 2018, 7, 57. [Google Scholar] [CrossRef]
- Benstoem, C.; Goetzenich, A.; Kraemer, S.; Borosch, S.; Manzanares, W.; Hardy, G.; Stoppe, C. Selenium and its supplementation in cardiovascular disease—What do we know? Nutrients 2015, 7, 3094–3118. [Google Scholar] [CrossRef]
- Li, J.; Lo, K.; Shen, G.; Feng, Y.Q.; Huang, Y.Q. Gender difference in the association of serum selenium with all-cause and cardiovascular mortality. Postgrad. Med. 2020, 132, 148–155. [Google Scholar] [CrossRef]
- Bomer, N.; Grote Beverborg, N.; Hoes, M.F.; Streng, K.W.; Vermeer, M.; Dokter, M.M.; IJmker, J.; Anker, S.D.; Cleland, J.; Hillege, H.L.; et al. Selenium and outcome in heart failure. Eur. J. Heart Fail. 2020, 22, 1415–1423. [Google Scholar] [CrossRef]
- Bleys, J.; Navas-Acien, A.; Guallar, E. Serum Selenium levels and all-cause, cancer, and cardiovascular mortality among US adults. Arch. Intern. Med. 2008, 168, 404–410. [Google Scholar] [CrossRef]
- Mohammadifard, N.; Humphries, K.H.; Gotay, C.; Mena-Sánchez, G.; Salas-Salvadó, J.; Esmaillzadeh, A.; Ignaszewski, A.; Sarrafzadegan, N. Trace minerals intake: Risks and benefits for cardiovascular health. Crit. Rev. Food Sci. Nutr. 2019, 59, 1334–1346. [Google Scholar] [CrossRef] [PubMed]
- Handy, D.E.; Loscalzo, J. Selenoproteins in Cardiovascular Redox Pathology. In Selenium. Its Molecular Biology and Role in Human Health, 4th ed.; Hatfield, D.L., Schweizer, U., Tsuji, P.A., Gladyshev, V.N., Eds.; Springer International Publishing: New York, NY, USA, 2016; Chapter 39; pp. 463–474. [Google Scholar]
- Tanguy, S.; Rakotovao, A.; Jouan, M.G.; Ghezzi, C.; de Leiris, J.; Boucher, F. Dietary selenium intake influences Cx43 dephosphorylation, TNF-α expression and cardiac remodeling after reperfused infarction. Mol. Nutr. Food Res. 2011, 55, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Alehagen, U.; Aaseth, J. Selenium and coenzyme Q10 interrelationship in cardiovascular diseases—A clinician’s point of view. J. Trace Elem. Med. Biol. 2015, 31, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Alehagen, U.; Aaseth, J.; Johansson, P. Reduced Cardiovascular Mortality 10 Years after Supplementation with Selenium and Coenzyme Q10 for Four Years: Follow-Up Results of a Prospective Randomized Double-Blind Placebo-Controlled Trial in Elderly Citizens. PLoS ONE 2015, 10, e0141641. [Google Scholar]
- Alehagen, U.; Alexander, J.; Aaseth, J. Supplementation with Selenium and Coenzyme Q10 Reduces Cardiovascular Mortality in Elderly with Low Selenium Status. A Secondary Analysis of a Randomised Clinical Trial. PLoS ONE 2016, 11, e0157541. [Google Scholar] [CrossRef] [PubMed]
- Alehagen, U.; Aaseth, J.; Alexander, J.; Svensson, E.; Johansson, P.; Larsson, A. Less fibrosis in elderly subjects supplemented with selenium and coenzyme Q10-A mechanism behind reduced cardiovascular mortality? Biofactors 2018, 44, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Alehagen, U.; Johansson, P.; Aaseth, J.; Alexander, J.; Brismar, K. Increase in insulin-like growth factor 1 (IGF-1) and insulin-like growth factor binding protein 1 after supplementation with selenium and coenzyme Q10. A prospective randomized double-blind placebo-controlled trial among elderly Swedish citizens. PLoS ONE 2017, 12, e0178614. [Google Scholar] [CrossRef]
- Alehagen, U.; Aaseth, J.; Alexander, J.; Johansson, P. Still reduced cardiovascular mortality 12 years after supplementation with selenium and coenzyme Q10 for four years: A validation of previous 10-year follow-up results of a prospective randomized double-blind placebo-controlled trial in elderly. PLoS ONE 2018, 13, e0193120. [Google Scholar] [CrossRef]
- Alehagen, U.; Aaseth, J.; Alexander, J.; Johansson, P.; Larsson, A. Supplemental selenium and coenzyme Q10 reduce glycation along with cardiovascular mortality in an elderly population with low selenium status—A four-year, prospective, randomised, double-blind placebo-controlled trial. J. Trace Elem. Med. Biol. 2020, 61, 126541. [Google Scholar] [CrossRef]
- Alehagen, U.; Aaseth, J.; Lindahl, T.L.; Larsson, A.; Alexander, J. Dietary Supplementation with Selenium and Coenzyme Q10 Prevents Increase in Plasma D-Dimer While Lowering Cardiovascular Mortality in an Elderly Swedish Population. Nutrients 2021, 13, 1344. [Google Scholar] [CrossRef]
- Flores-Mateo, G.; Navas-Acien, A.; Pastor-Barriuso, R.; Guallar, E. Selenium and coronary heart disease: A meta-analysis. Am. J. Clin. Nutr. 2006, 84, 762–773. [Google Scholar] [CrossRef] [PubMed]
- Ju, W.; Li, X.; Li, Z.; Wu, G.R.; Fu, X.F.; Yang, X.M.; Zhang, X.Q.; Gao, X.B. The effect of selenium supplementation on coronary heart disease: A systematic review and meta-analysis of randomized controlled trials. J. Trace Elem. Med. Biol. 2017, 44, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Navas-Acien, A.; Bleys, J.; Guallar, E. Selenium intake and cardiovascular risk: What is new? Curr. Opin. Lipidol. 2008, 19, 43–49. [Google Scholar] [CrossRef]
- Hercberg, S.; Galan, P.; Preziosi, P.; Bertrais, S.; Mennen, L.; Malvy, D.; Roussel, A.M.; Favier, A.; Briancon, S. The SU.VI.MAX Study: A randomized, placebo-controlled trial of the health effects of antioxidant vitamins and minerals. Arch. Intern. Med. 2004, 164, 2335–2342. [Google Scholar] [CrossRef] [PubMed]
- Hercberg, S.; Bertrais, S.; Czernichow, S.; Noisette, N.; Galan, P.; Jaouen, A.; Tichet, J.; Briancon, S.; Favier, A.; Mennen, L.; et al. Alterations of the lipid profile after 7.5 years of low-dose antioxidant supplementation in the SU.VI.MAX Study. Lipids 2005, 40, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Hercberg, S.; Kesse-Guyot, E.; Druesne-Pecollo, N.; Touvier, M.; Favier, A.; Latino-Martel, P.; Briançon, S.; Galan, P. Incidence of cancers, ischemic cardiovascular diseases and mortality during 5-year follow-up after stopping antioxidant vitamins and minerals supplements: A postintervention follow-up in the SU.VI.MAX Study. Int. J. Cancer 2010, 127, 1875–1881. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P.; Stranges, S.; Griffin, B.A.; Pastor-Barriuso, R.; Guallar, E. Effect of supplementation with high-Selenium yeast on plasma lipids: A randomized trial. Ann. Intern. Med. 2011, 154, 656–665. [Google Scholar] [CrossRef]
- Salimian, M.; Soleimani, A.; Bahmani, F.; Tabatabaei, S.M.H.; Asemi, Z.; Talari, H.R. The effects of selenium administration on carotid intima-media thickness and metabolic status in diabetic hemodialysis patients: A randomized, double-blind, placebo-controlled trial. Clin. Nutr. ESPEN 2021, in press. [Google Scholar] [CrossRef]
- Maugeri, A.; Hruskova, J.; Jakubik, J.; Kunzova, S.; Sochor, O.; Barchitta, M.; Agodi, A.; Bauerova, H.; Medina-Inojosa, J.R.; Vinciguerra, M. Dietary antioxidant intake decreases carotid intima media thickness in women but not in men: A cross-sectional assessment in the Kardiovize study. Free Radic. Biol. Med. 2019, 131, 274–281. [Google Scholar] [CrossRef]
- Mirdamadi, A.; Rafiei, R.; Kahazaipour, G.; Fouladi, L. Selenium Level in Patients with Heart Failure versus Normal Individuals. Int. J. Prev. Med. 2019, 10, 210. [Google Scholar]
- Hu, H.; Bi, C.; Lin, T.; Liu, L.; Song, Y.; Wang, B.; Wang, P.; Zhou, Z.; Fang, C.; Ma, H.; et al. Sex difference in the association between plasma selenium and first stroke: A community-based nested case-control study. Biol. Sex Differ. 2021, 12, 39. [Google Scholar] [CrossRef] [PubMed]
- Al-Taesh, H.; Çelekli, A.; Sucu, M.; Taysi, S. Trace elements in patients with aortic valve sclerosis. Ther. Adv. Cardiovasc. Dis. 2021, 15, 1753944720985985. [Google Scholar] [CrossRef]
- Bleys, J.; Navas-Acien, A.; Laclaustra, M.; Pastor-Barriuso, R.; Menke, A.; Ordovas, J. Serum Selenium and peripheral arterial disease: Results from the national health and nutrition examination survey, 2003–2004. Am. J. Epidemiol. 2009, 169, 996–1003. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Liu, C.; Guo, J.; Song, Y. Selenium status and cardiovascular diseases: Meta-analysis of prospective observational studies and randomized controlled trials. Eur. J. Clin. Nutr. 2016, 70, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Dumitrescu, A.M.; Liao, X.H.; Abdullah, M.S.; Lado-Abeal, J.; Majed, F.A.; Moeller, L.C.; Boran, G.; Schomburg, L.; Weiss, R.E.; Refetoff, S. Mutations in SECISBP2 result in abnormal thyroid hormone metabolism. Nat. Genet. 2005, 37, 1247–1252. [Google Scholar] [CrossRef] [PubMed]
- Schomburg, L.; Dumitrescu, A.M.; Liao, X.H.; Bin-Abbas, B.; Hoeflich, J.; Köhrle, J.; Refetoff, S. Selenium supplementation fails to correct the selenoprotein synthesis defect in subjects with SBP2 gene mutations. Thyroid 2009, 19, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Duntas, L.H. Selenium and the thyroid: A close-knit connection. J. Clin. Endocrinol. Metab. 2010, 95, 5180–5188. [Google Scholar] [CrossRef] [PubMed]
- Winther, K.H.; Rayman, M.P.; Bonnema, S.J.; Hegedüs, L. Selenium in thyroid disorders—Essential knowledge for clinicians. Nat. Rev. Endocrinol. 2020, 16, 165–176. [Google Scholar] [CrossRef]
- Zuo, Y.; Li, Y.; Gu, X.; Lei, Z. The correlation between selenium levels and autoimmune thyroid disease: A systematic review and meta-analysis. Ann. Palliat. Med. 2021, 10, 4398–4408. [Google Scholar] [CrossRef]
- Kyrgios, I.; Giza, S.; Kotanidou, E.P.; Kleisarchaki, A.; Tsinopoulou, V.R.; Papadopoulou, A.; Markantonatou, A.M.; Kanellidou, E.; Giannakou, A.; Galli-Tsinopoulou, A. L-selenomethionine supplementation in children and adolescents with autoimmune thyroiditis: A randomized double-blind placebo-controlled clinical trial. J. Clin. Pharm.Ther. 2019, 44, 102–108. [Google Scholar] [CrossRef]
- Karimi, F.; Omrani, G.R. Effects of selenium and vitamin C on the serum level of antithyroid peroxidase antibody in patients with autoimmune thyroiditis. J. Endocrinol. Investig. 2019, 42, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Krysiak, R.; Kowalcze, K.; Okopień, B. Selenomethionine potentiates the impact of vitamin D on thyroid autoimmunity in euthyroid women with Hashimoto’s thyroiditis and low vitamin D status. Pharmacol. Rep. 2019, 71, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, L.; Mobasseri, M.; Ostadrahimi, A.; Pourmoradian, S.; Soleimanzadeh, H.; Kafili, B. Effect of selenium-enriched yeast supplementation on serum thyroid-stimulating hormone and anti-thyroid peroxidase antibody levels in subclinical hypothyroidism: Randomized controlled trial. Adv. Biomed. Res. 2021, 10, 33. [Google Scholar] [PubMed]
- Almanza-Monterrubio, M.; Garnica-Hayashi, L.; Dávila-Camargo, A.; Nava-Castañeda, Á. Oral selenium improved the disease activity in patients with mild Graves’ orbitopathy. J. Fr. Ophtalmol. 2021, 44, 643–651. [Google Scholar] [CrossRef] [PubMed]
- Reid, S.M.; Middleton, P.; Cossich, M.C.; Crowther, C.A.; Bain, E. Interventions for clinical and subclinical hypothyroidism pre-pregnancy and during pregnancy. Cochrane Database Syst. Rev. 2013, 5, CD007752. [Google Scholar] [CrossRef]
- Guo, X.; Zhou, L.; Xu, J.; Liu, Z.; Liu, J.; Yan, C. Prenatal Maternal Low Selenium, High Thyrotropin, and Low Birth Weights. Biol. Trace Elem. Res. 2021, 199, 18–25. [Google Scholar] [CrossRef]
- Maiorino, M.; Flohé, L.; Roveri, A.; Steinert, P.; Wissing, J.B.; Ursini, F. Selenium and reproduction. Biofactors 1999, 10, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Flohé, L. Selenium in mammalian spermiogenesis. Biol. Chem. 2007, 388, 987–995. [Google Scholar] [CrossRef] [PubMed]
- Turanov, A.A.; Malinouski, M.; Gladyshev, V.N. Selenium and Male Reproduction. In Selenium. Its Molecular Biology and Role in Human Health, 3rd ed.; Hatfield, D.L., Berry, M.J., Gladyshev, V.N., Eds.; Springer: London, UK, 2012; pp. 409–417. [Google Scholar]
- Jacobsson, S.O.; Hansson, E. Distribution of Selenium in mice studied by whole-body autoradiography after injection off SE-75-sodium selenite. Acta Vet. Scand. 1965, 6, 287–298. [Google Scholar] [CrossRef]
- Hansson, E.; Jacobsson, S.O. Uptake of [75Se] selenomethionine in the tissues of the mouse studied by whole-body autoradiography. Biochim. Biophys. Acta 1966, 115, 285–293. [Google Scholar] [CrossRef]
- Maiorino, M.; Wissing, J.B.; Brigelius-Flohé, R.; Calabrese, F.; Roveri, A.; Steinert, P.; Ursini, F.; Flohé, L. Testosterone mediates expression of the selenoproteins PHGPx by induction of spermatogenesis and not by direct transcriptional gene activation. FASEB J. 1998, 12, 1359–1370. [Google Scholar] [CrossRef] [PubMed]
- Scarlata, E.; O’Flaherty, C. Antioxidant Enzymes and Male Fertility: Lessons from Knockout Models. Antioxid. Redox Signal. 2020, 32, 569–580. [Google Scholar] [CrossRef] [PubMed]
- Dias, T.R.; Martin-Hidalgo, D.; Silva, B.M.; Oliveira, P.F.; Alves, M.G. Endogenous and Exogenous Antioxidants As a Tool to Ameliorate Male Infertility Induced by Reactive Oxygen Species. Antioxid. Redox Signal. 2020, 33, 767–785. [Google Scholar] [CrossRef]
- Roveri, A.; Casasco, A.; Maiorino, M.; Dalan, P.; Calligaro, A.; Ursini, F. Phospholipid hydroperoxide glutathione peroxidase of rat testis. Gonadotropin dependence and immunocytochemicalidentification. J. Biol. Chem. 1992, 267, 6142–6146. [Google Scholar] [CrossRef]
- Pfeifer, H.; Conrad, M.; Roethlein, D.; Kyriakopoulos, A.; Brielmeier, M.; Bornkamm, G.W.; Behne, D. Identification of a specific sperm nuclei selenoenzyme necessary for protamine thiol cross-linking during sperm maturation. FASEB J. 2001, 15, 1236–1238. [Google Scholar] [CrossRef] [PubMed]
- Imai, H.; Hakkaku, N.; Iwamoto, R.; Suzuki, J.; Suzuki, T.; Tajima, Y.; Konishi, K.; Minami, S.; Ichinose, S.; Ishizaka, K.; et al. Depletion of selenoprotein GPx4 in spermatocytes causes male infertility in mice. J. Biol. Chem. 2009, 284, 32522–32532. [Google Scholar] [CrossRef]
- Schneider, M.; Förster, H.; Boersma, A.; Seiler, A.; Wehnes, H.; Sinowatz, F.; Neumüller, C.; Deutsch, M.J.; Walch, A.; Hrabé de Angelis, M.; et al. Mitochondrial glutathione peroxidase 4 disruption causes male infertility. FASEB J. 2009, 23, 3233–3242. [Google Scholar] [CrossRef]
- Imai, H.; Suzuki, K.; Ishizaka, K.; Ichinose, S.; Oshima, H.; Okayasu, I.; Emoto, K.; Umeda, M.; Nakagawa, Y. Failure of the expression of phospholipid hydroperoxide glutathione peroxidase in the spermatozoa of human infertile males. Biol. Reprod. 2001, 64, 674–683. [Google Scholar] [CrossRef]
- Foresta, C.; Flohé, L.; Garolla, A.; Roveri, A.; Ursini, F.; Maiorino, M. Male fertility is linked to the selenoprotein phospholipid hydroperoxide glutathione peroxidase. Biol. Reprod. 2002, 67, 967–971. [Google Scholar] [CrossRef]
- Renko, K.; Werner, M.; Renner-Müller, I.; Cooper, T.G.; Yeung, C.H.; Hollenbach, B.; Scharpf, M.; Köhrle, J.; Schomburg, L.; Schweizer, U. Hepatic selenoprotein P (SePP) expression restores Selenium transport and prevents infertility and motor-incoordination in Sepp-knockout mice. Biochem. J. 2008, 409, 741–749. [Google Scholar] [CrossRef]
- Su, D.; Novoselov, S.V.; Sun, Q.A.; Moustafa, M.E.; Zhou, Y.; Oko, R.; Hatfield, D.L.; Gladyshev, V.N. Mammalian selenoproteinthioredoxin-glutathione reductase. Roles in disulfide bond formation and sperm maturation. J. Biol. Chem. 2005, 280, 26491–26498. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Xiong, W.; Chen, L.L.; Huang, J.Q.; Lei, X.G. Selenoprotein V protects against endoplasmic reticulum stress and oxidative injury induced by pro-oxidants. Free Radic. Biol. Med. 2020, 160, 670–679. [Google Scholar] [CrossRef] [PubMed]
- Salas-Huetos, A.; Rosique-Esteban, N.; Becerra-Tomás, N.; Vizmanos, B.; Bulló, M.; Salas-Salvadó, J. The Effect of Nutrients and Dietary Supplements on Sperm Quality Parameters: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. Adv. Nutr. 2018, 9, 833–848. [Google Scholar] [CrossRef]
- Liu, Y.; Peterson, K.E.; Sánchez, B.N.; Jones, A.D.; Cantoral, A.; Mercado-García, A.; Solano-González, M.; Ettinger, A.S.; Téllez-Rojo, M.M. Dietary Intake of Selenium in Relation to Pubertal Development in Mexican Children. Nutrients 2019, 11, 1595. [Google Scholar] [CrossRef] [PubMed]
- Hawkes, W.C.; Alkan, Z.; Wong, K. Selenium supplementation does not affect testicular selenium status or semen quality in North American men. J. Androl. 2009, 30, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Steiner, A.Z.; Hansen, K.R.; Barnhart, K.T.; Cedars, M.I.; Legro, R.S.; Diamond, M.P.; Krawetz, S.A.; Usadi, R.; Baker, V.L.; Coward, R.M.; et al. Reproductive Medicine Network. The effect of antioxidants on male factor infertility: The Males, Antioxidants, and Infertility (MOXI) randomized clinical trial. Fertil. Steril. 2020, 113, 552–560.e3. [Google Scholar] [CrossRef]
- Cannarella, R.; Condorelli, R.A.; Calogero, A.E.; Bagnara, V.; Aversa, A.; Greco, E.A.; Brunetti, A.; La Vignera, S. Effects of Selenium Supplementation on Sperm Parameters and DNA-Fragmentation Rate in Patients with Chronic Autoimmune Thyroiditis. J. Clin. Med. 2021, 10, 3755. [Google Scholar] [CrossRef]
- Qazi, I.H.; Yang, H.; Wei, S.; Angel, C.; Pan, B.; Zhou, G.; Han, H. Dietary selenium deficiency and supplementation differentially modulate the expression of two ER-resident selenoproteins (selenoprotein K and selenoprotein M) in the ovaries of aged mice: Preliminary data. Reprod. Biol. 2020, 20, 441–446. [Google Scholar] [CrossRef]
- Lin, J.; Wang, L. Oxidative Stress in Oocytes and Embryo Development: Implications for In Vitro Systems. Antioxid. Redox Signal. 2021, 34, 1394–1406. [Google Scholar] [CrossRef]
- Safiyeh, F.D.; Mojgan, M.; Parviz, S.; Sakineh, M.A.; Behnaz, S.O. The effect of selenium and vitamin E supplementation on anti-Mullerian hormone and antral follicle count in infertile women with occult premature ovarian insufficiency: A randomized controlled clinical trial. Complement. Ther. Med. 2021, 56, 102533. [Google Scholar] [CrossRef]
- Vescini, F.; Chiodini, I.; Palermo, A.; Cesareo, R.; De Geronimo, V.; Scillitani, A.; Gennari, L.; Falchetti, A. Selenium: A Trace Element for a Healthy Skeleton—A Narrative Review. Endocr. Metab. Immune Disord. Drug Targets 2021, 21, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xie, D.; Li, J.; Long, H.; Wu, J.; Wu, Z.; He, H.; Wang, H.; Yang, T.; Wang, Y. Association between dietary Selenium intake and the prevalence of osteoporosis: A cross-sectional study. BMC Musculoskelet. Disord. 2019, 20, 585. [Google Scholar] [CrossRef] [PubMed]
- Park, K.C.; Kwon, Y.; Lee, Y.; Kim, D.K.; Jang, Y.; Lee, S. Low Selenium levels are associated with decreased bone mineral densities. J. Trace Elem. Med. Biol. 2020, 61, 126534. [Google Scholar] [CrossRef] [PubMed]
- Arikan, D.C.; Coskun, A.; Ozer, A.; Kilinc, M.; Atalay, F.; Arikan, T. Plasmaselenium, zinc, copper and lipid levels in postmenopausal Turkish women and their relationwithosteoporosis. Biol. Trace Elem. Res. 2011, 144, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Al, E.A.A.; Parsian, H.; Fathi, M.; Faghihzadeh, S.; Hosseini, S.R.; Nooreddini, H.G.; Mosapour, A. ALOX12 gene polymorphisms and serum Selenium status in elderly osteoporotic patients. Adv. Clin. Exp. Med. 2018, 27, 1717–1722. [Google Scholar]
- Beukhof, C.M.; Medici, M.; van den Beld, A.W.; Hollenbach, B.; Hoeg, A.; Visser, W.E.; de Herder, W.W.; Visser, T.J.; Schomburg, L.; Peeters, R.P. Selenium Status isPositivelyAssociated with Bone Mineral Density in Healthy Aging European Men. PLoS ONE 2016, 11, e0152748. [Google Scholar] [CrossRef]
- Pedrera-Zamorano, J.D.; Calderon-Garcia, J.F.; Roncero-Martin, R.; Manas-Nunez, P.; Moran, J.M.; Lavado-Garcia, J.M. The protective effect of calcium on bonemass in postmenopausal women with high Selenium intake. J. Nutr. Health Aging. 2012, 16, 743–748. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.S.; Jacques, R.M.; Schomburg, L.; Hill, T.R.; Mathers, J.C.; Williams, G.R.; Eastell, R. Effect of selenium supplementation on musculoskeletal health in older women: A randomised, double-blind, placebo-controlled trial. Lancet Healthy Longev. 2021, 2, e212–e221. [Google Scholar] [CrossRef]
- Labunskyy, V.M.; Hatfield, D.L.; Gladyshev, V.N. Selenoproteins: Molecular Pathways and Physiological Roles. Physiol. Rev. 2014, 94, 739–777. [Google Scholar] [CrossRef]
- Campa, A.; Sales Martinez, S.; Baum, M.K. Selenium in HIV/AIDS. In Selenium. Its Molecular Biology and Role in Human Health, 4th ed.; Hatfield, D.L., Schweizer, U., Tsuji, P.A., Gladyshev, V.N., Eds.; Springer International Publishing: New York, NY, USA, 2016; Chapter 28; pp. 333–342. [Google Scholar]
- Campa, A.; Baum, M.K. Role of Selenium in HIV/AIDS. In Selenium. Its Molecular Biology and Role in Human Health, 3rd ed.; Hatfield, D.L., Berry, M.J., Gladyshev, V.N., Eds.; Springer: London, UK, 2012; pp. 383–397. [Google Scholar]
- Ogunro, P.S.; Ogungbamigbe, T.O.; Elemie, P.O.; Egbewale, B.E.; Adewole, T.A. Plasma Selenium concentration and glutathione peroxidase activity in HIV-1/AIDS infected patients: A correlation with the disease progression. Niger. Postgrad. Med. J. 2006, 13, 1–5. [Google Scholar]
- Allard, J.P.; Aghdassi, E.; Chau, J.; Salit, I.; Walmsley, S. Oxidative stress and plasma antioxidant micronutrients in humans with HIV infection. Am. J. Clin. Nutr. 1998, 67, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Cirelli, A.; Ciardi, M.; de Simone, C.; Sorice, F.; Giordano, R.; Ciaralli, L.; Costantini, S. Serum Selenium concentration and disease progress in patients with HIV infection. Clin. Biochem. 1991, 24, 211–214. [Google Scholar] [CrossRef]
- Hulgan, T.; Morrow, J.; D’Aquila, R.T.; Raffanti, S.; Morgan, M.; Rebeiro, P.; Haas, D.W. Oxidant stress is increased during treatment of human immunodeficiency virus infection. Clin. Infect. Dis. 2003, 37, 1711–1717. [Google Scholar] [CrossRef]
- Wang, X.; Chai, H.; Yao, Q.; Chen, C. Molecular mechanisms of HIV protease inhibitor-induced endothelial dysfunction. J. Acquir. Immune Defic. Syndr. 2007, 44, 493–499. [Google Scholar] [CrossRef]
- Mondal, D.; Pradhan, L.; Ali, M.; Agrawal, K.C. HAART drugs induce oxidative stress in human endothelial cells and increase endothelial recruitment of mononuclear cells: Exacerbation by inflammatory cytokines and amelioration by antioxidants. Cardiovasc. Toxicol. 2004, 4, 287–302. [Google Scholar] [CrossRef]
- Campa, A.; Shor-Posner, G.; Indacochea, F.; Zhang, G.; Lai, H.; Asthana, D.; Scott, G.B.; Baum, M.K. Mortality risk in Selenium-deficient HIV-positive children. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 1999, 20, 508–513. [Google Scholar] [CrossRef]
- Baum, M.K.; Shor-Posner, G.; Lai, S.; Zhang, G.; Lai, H.; Fletcher, M.A.; Sauberlich, H.; Page, J.B. High risk of HIV-related mortality is associated with Seleniumdeficiency. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 1997, 15, 370–374. [Google Scholar] [CrossRef]
- Kupka, R.; Msamanga, G.I.; Spiegelman, D.; Morris, S.; Mugusi, F.; Hunter, D.J.; Fawzi, W.W. Selenium status is associated with accelerated HIV disease progression among HIV-1-infected pregnant women in Tanzania. J. Nutr. 2004, 134, 2556–2560. [Google Scholar] [CrossRef]
- Burbano, X.; Miguez-Burbano, M.J.; McCollister, K.; Zhang, G.; Rodriguez, A.; Ruiz, P.; Lecusay, R.; Shor-Posner, G. Impact of a Selenium chemoprevention clinical trial on hospital admissions of HIV-infected participants. HIV Clin. Trials 2002, 3, 483–491. [Google Scholar]
- Hurwitz, B.E.; Klaus, J.R.; Llabre, M.M.; Gonzalez, A.; Lawrence, P.J.; Maher, K.J.; Greeson, J.M.; Baum, M.K.; Shor-Posner, G.; Skyler, J.S.; et al. Suppression of human immunodeficiency virus type 1 viral load with Selenium supplementation: A randomized controlled trial. Arch. Intern. Med. 2007, 167, 148–154. [Google Scholar] [CrossRef]
- Kupka, R.; Mugusi, F.; Aboud, S.; Msamanga, G.I.; Finkelstein, J.L.; Spiegelman, D.; Fawzi, W.W. Randomized, double-blind, placebo-controlled trial of Selenium supplements among HIV-infected pregnant women in Tanzania: Effects on maternal and child outcomes. Am. J. Clin. Nutr. 2008, 87, 1802–1808. [Google Scholar] [CrossRef] [PubMed]
- Spallholz, J.E.; Boylan, L.M.; Larsen, H.S. Advances in understanding Selenium’s role in the immune system. Ann. N. Y. Acad. Sci. 1990, 587, 123–139. [Google Scholar] [CrossRef] [PubMed]
- Sappey, C.; Legrand-Poels, S.; Best-Belpomme, M.; Favier, A.; Rentier, B.; Piette, J. Stimulation of glutathione peroxidase activity decreases HIV type 1 activation after oxidative stress. AIDS Res. Hum. Retrovir. 1994, 10, 1451–1461. [Google Scholar] [CrossRef] [PubMed]
- Delmas-Beauvieux, M.C.; Peuchant, E.; Couchouron, A.; Constans, J.; Sergeant, C.; Simonoff, M.; Pellegrin, J.L.; Leng, B.; Conri, C.; Clerc, M. The enzymatic antioxidant system in blood and glutathione status in human immunodeficiency virus (HIV)-infected patients: Effects of supplementation with Selenium or beta-carotene. Am. J. Clin. Nutr. 1996, 64, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Kalantari, P.; Narayan, V.; Natarajan, S.K.; Muralidhar, K.; Gandhi, U.H.; Vunta, H.; Henderson, A.J.; Prabhu, K.S. Thioredoxin reductase-1 negatively regulates HIV-1 transactivating protein Tat-dependent transcription in human macrophages. J. Biol. Chem. 2008, 283, 33183–33190. [Google Scholar] [CrossRef]
- Kamwesiga, J.; Mutabazi, V.; Kayumba, J.; Tayari, J.C.; Smyth, R.; Fay, H.; Umurerwa, A.; Baziruwiha, M.; Ntizimira, C.; Murebwayire, A.; et al. Effect of selenium supplementation on CD4 T-cell recovery, viral suppression, morbidity and quality of life of HIV-infected patients in Rwanda: Study protocol for a randomized controlled trial. Trials 2011, 12, 192. [Google Scholar] [CrossRef]
- Baum, M.K.; Campa, A.; Lai, S.; Sales Martinez, S.; Tsalaile, L.; Burns, P.; Farahani, M.; Li, Y.; van Widenfelt, E.; Page, J.B.; et al. Effect of micronutrient supplementation on disease progression in asymptomatic, antiretroviral-naive, HIV-infected adults in Botswana: A randomized clinical trial. JAMA 2013, 310, 2154–2163. [Google Scholar] [CrossRef]
- Okunade, K.S.; Olowoselu, O.F.; John-Olabode, S.; Hassan, B.O.; Akinsola, O.J.; Nwogu, C.M.; Ugwu, A.O.; Moses, O.E.; Rabiu, K.A.; Ajepe, A.; et al. Effects of selenium supplementation on pregnancy outcomes and disease progression in HIV-infected pregnant women in Lagos: A randomized controlled trial. Int. J. Gynaecol. Obstet. 2021, 153, 533–541. [Google Scholar] [CrossRef]
- Alexander, J.; Tinkov, A.; Strand, T.A.; Alehagen, U.; Skalny, A.; Aaseth, J. Early Nutritional Interventions with Zinc, Selenium and Vitamin D for Raising Anti-Viral Resistance Against Progressive COVID-19. Nutrients 2020, 12, 2358. [Google Scholar] [CrossRef]
- Fulzele, S.; Sahay, B.; Yusufu, I.; Lee, T.J.; Sharma, A.; Kolhe, R.; Isales, C.M. COVID-19 Virulence in Aged Patients Might Be Impacted by the Host Cellular MicroRNAs Abundance/Profile. Aging Dis. 2020, 11, 509–522. [Google Scholar] [CrossRef]
- World Health Organization Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int/ (accessed on 5 January 2022).
- Zhang, J.; Saad, R.; Taylor, E.W.; Rayman, M.P. Selenium and selenoproteins in viral infection with potential relevance to COVID-19. Redox Biol. 2020, 37, 101715. [Google Scholar] [CrossRef] [PubMed]
- Hiffler, L.; Rakotoambinina, B. Selenium and RNA Virus Interactions: Potential Implications for SARS-CoV-2 Infection (COVID-19). Front. Nutr. 2020, 7, 164. [Google Scholar] [CrossRef] [PubMed]
- Khatiwada, S.; Subedi, A. A Mechanistic Link between Selenium and Coronavirus Disease 2019 (COVID-19). Curr. Nutr. Rep. 2021, 10, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Moghaddam, A.; Heller, R.A.; Sun, Q.; Seelig, J.; Cherkezov, A.; Seibert, L.; Hackler, J.; Seemann, P.; Diegmann, J.; Pilz, M.; et al. Selenium Deficiency Is Associated with Mortality Risk from COVID-19. Nutrients 2020, 12, 2098. [Google Scholar] [CrossRef]
- Heller, R.A.; Sun, Q.; Hackler, J.; Seelig, J.; Seibert, L.; Cherkezov, A.; Minich, W.B.; Seemann, P.; Diegmann, J.; Pilz, M.; et al. Prediction of survival odds in COVID-19 by zinc, age and selenoprotein P as composite biomarker. Redox Boil. 2021, 38, 101764. [Google Scholar] [CrossRef]
- Sahin, E.; Orhan, C.; Uckun, F.M.; Sahin, K. Clinical Impact Potential of Supplemental Nutrients as Adjuncts of Therapy in High-Risk COVID-19 for Obese Patients. Front. Nutr. 2020, 7, 580504. [Google Scholar] [CrossRef]
- Diwaker, D.; Mishra, K.P.; Ganju, L. Potential roles of protein disulphide isomerase in viral infections. Acta Virol. 2013, 57, 293–304. [Google Scholar]
- Kieliszek, M.; Lipinski, B. Selenium supplementation in the prevention of coronavirus infections (COVID-19). Med. Hypotheses 2020, 143, 109878. [Google Scholar] [CrossRef]
- Loef, M.; Schrauzer, G.N.; Walach, H. Selenium and Alzheimer’s disease: A systematic review. J. Alzheimer’s Dis. 2011, 26, 81–104. [Google Scholar] [CrossRef]
- Berr, C.; Balansard, B.; Arnaud, J.; Roussel, A.M.; Alpérovitch, A. Cognitive decline is associated with systemic oxidative stress: The EVA study. Etude du Vieillissement Artériel. J. Am. Geriatr. Soc. 2000, 48, 1285–1291. [Google Scholar] [CrossRef]
- Gao, S.; Jin, Y.; Hall, K.S.; Liang, C.; Unverzagt, F.W.; Ji, R.; Murrell, J.R.; Cao, J.; Shen, J.; Ma, F.; et al. Selenium level and cognitive function in rural elderly Chinese. Am. J. Epidemiol. 2007, 165, 955–965. [Google Scholar] [CrossRef] [PubMed]
- Kesse-Guyot, E.; Fezeu, L.; Jeandel, C.; Ferry, M.; Andreeva, V.; Amieva, H.; Hercberg, S.; Galan, P. French adults’ cognitive performance after daily supplementation with antioxidant vitamins and minerals at nutritional doses: A post hoc analysis of the Supplementation in Vitamins and Mineral Antioxidants (SU.VI.MAX) trial. Am. J. Clin. Nutr. 2011, 94, 892–899. [Google Scholar] [CrossRef]
- Shahar, A.; Patel, K.V.; Semba, R.D.; Bandinelli, S.; Shahar, D.R.; Ferrucci, L.; Guralnik, J.M. Plasma selenium is positively related to performance in neurological tasks assessing coordination and motor speed. J. Mov. Disord. 2010, 25, 1909–1915. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, B.R.; Apolinário, D.; da Silva Bandeira, V.; Busse, A.L.; Magaldi, R.M.; Jacob-Filho, W.; Cozzolino, S. Effects of Brazil nut consumption on selenium status and cognitive performance in older adults with mild cognitive impairment: A randomized controlled pilot trial. Eur. J. Nutr. 2016, 55, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, B.R.; Hare, D.J.; Macpherson, H. Sex-dependent association between selenium status and cognitive performance in older adults. Eur. J. Nutr. 2021, 60, 1153–1159. [Google Scholar] [CrossRef] [PubMed]
- Bellinger, F.P.; Weeber, E.J. Selenium in Alzheimer’s Disease. In Selenium. Its Molecular Biology and Role in Human Health, 3rd ed.; Hatfield, D.L., Berry, M.J., Gladyshev, V.N., Eds.; Springer: London, UK, 2012; pp. 433–442. [Google Scholar]
- Burk, R.F.; Hill, K.E. Selenoprotein P-expression, functions, and roles in mammals. Biochim. Biophys. Acta 2009, 1790, 1441–1447. [Google Scholar] [CrossRef] [PubMed]
- Bellinger, F.P.; Raman, A.V.; Reeves, M.A.; Berry, M.J. Regulation and function of selenoproteins in human disease. Biochem. J. 2009, 422, 11–22. [Google Scholar] [CrossRef]
- Corcoran, N.M.; Martin, D.; Hutter-Paier, B.; Windisch, M.; Nguyen, T.; Nheu, L.; Sundstrom, L.E.; Costello, A.J.; Hovens, C.M. Sodium selenate specifically activates PP2A phosphatase, dephosphorylates tau and reverses memory deficits in an Alzheimer’s disease model. J. Clin. Neurosci. 2010, 17, 1025–1033. [Google Scholar] [CrossRef]
- Van Eersel, J.; Ke, Y.D.; Liu, X.; Delerue, F.; Kril, J.J.; Götz, J.; Ittner, L.M. Sodium selenate mitigates tau pathology, neurodegeneration, and functional deficits in Alzheimer’s disease models. Proc. Natl. Acad. Sci. USA 2010, 107, 13888–13893. [Google Scholar] [CrossRef]
- Butterfield, D.A.; Drake, J.; Pocernich, C.; Castegna, A. Evidence of oxidative damage in Alzheimer’s disease brain: Central role for amyloid beta-peptide. Trends. Mol. Med. 2001, 7, 548–554. [Google Scholar] [CrossRef]
- Sharma, G.; Shin, E.J.; Sharma, N.; Nah, S.Y.; Mai, H.N.; Nguyen, B.T.; Jeong, J.H.; Lei, X.G.; Kim, H.C. Glutathione peroxidase-1 and neuromodulation: Novel potentials of an old enzyme. Food Chem. Toxicol. 2021, 148, 111945. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.H.; Song, G.L. Roles of Selenoproteins in Brain Function and the Potential Mechanism of Selenium in Alzheimer’s Disease. Front. Neurosci. 2021, 15, 646518. [Google Scholar] [CrossRef] [PubMed]
- Solovyev, N. Selenoprotein P and its potential role in Alzheimer’s disease. Hormones 2020, 19, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Takemoto, A.S.; Berry, M.J.; Bellinger, F.P. Role of selenoprotein P in Alzheimer’s disease. Ethn. Dis. 2010, 20, S1–95. [Google Scholar] [PubMed]
- Bellinger, F.P.; He, Q.P.; Bellinger, M.T.; Lin, Y.; Raman, A.V.; White, L.R.; Berry, M.J. Association of selenoprotein P with Alzheimer’s pathology in human cortex. J. Alzheimer’s Dis. 2008, 15, 465–472. [Google Scholar] [CrossRef]
- Rueli, R.H.; Parubrub, A.C.; Dewing, A.S.; Hashimoto, A.C.; Bellinger, M.T.; Weeber, E.J.; Uyehara-Lock, J.H.; White, L.R.; Berry, M.J.; Bellinger, F.P. Increased selenoprotein P in choroid plexus and cerebrospinal fluid in Alzheimer’s disease brain. J. Alzheimer’s Dis. 2015, 44, 379–383. [Google Scholar] [CrossRef]
- Hatfield, D.L.; Gladyshev, V.N.; Park, J.M.; Park, S.I.; Chittum, H.S.; Huh, J.R.; Carlson, B.A.; Kim, M.; Moustafa, M.E.; Lee, B.J. Comprehensive Natural Products Chemistry; Kelly, J.F., Ed.; Elsevier: New York, NY, USA, 1999; pp. 353–380. [Google Scholar]
- Saito, Y.; Takahashi, K. Characterization of selenoprotein P as a selenium supply protein. Eur. J. Biochem. 2002, 269, 5746–5751. [Google Scholar] [CrossRef]
- Schomburg, L.; Schweizer, U.; Holtmann, B.; Flohé, L.; Sendtner, M.; Köhrle, J. Gene disruption discloses role of selenoprotein P in selenium delivery to target tissues. Biochem. J. 2003, 370, 397–402. [Google Scholar] [CrossRef]
- Vicente-Zurdo, D.; Romero-Sánchez, I.; Rosales-Conrado, N.; León-González, M.E.; Madrid, Y. Ability of selenium species to inhibit metal-induced Aβ aggregation involved in the development of Alzheimer’s disease. Anal. Bioanal. Chem. 2020, 412, 6485–6497. [Google Scholar] [CrossRef]
- Hoozemans, J.J.; van Haastert, E.S.; Nijholt, D.A.; Rozemuller, A.J.; Eikelenboom, P.; Scheper, W. The unfolded protein response is activated in pretangle neurons in Alzheimer’s disease hippocampus. Am. J. Pathol. 2009, 174, 1241–1251. [Google Scholar] [CrossRef]
- Resende, R.; Ferreiro, E.; Pereira, C.; Oliveira, C.R. ER stress is involved in Abeta-induced GSK-3beta activation and tau phosphorylation. J. Neurosci. Res. 2008, 86, 2091–2099. [Google Scholar] [CrossRef] [PubMed]
- Shchedrina, V.A.; Zhang, Y.; Labunskyy, V.M.; Hatfield, D.L.; Gladyshev, V.N. Structure-function relations, physiological roles, and evolution of mammalian ER-resident selenoproteins. Antioxid. Redox Signal. 2010, 12, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Kawahara, M.; Negishi-Kato, M.; Sadakane, Y. Calcium dyshomeostasis and neurotoxicity of Alzheimer’s beta-amyloid protein. Expert Rev. Neurother. 2009, 9, 681–693. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.T.; Chang, R.C.; Tan, L. Calcium dysregulation in Alzheimer’s disease: From mechanisms to therapeutic opportunities. Prog. Neurobiol. 2009, 89, 240–255. [Google Scholar] [CrossRef] [PubMed]
- Reeves, M.A.; Bellinger, F.P.; Berry, M.J. The neuroprotective functions of selenoprotein M and its role in cytosolic calcium regulation. Antioxid. Redox Signal. 2010, 12, 809–818. [Google Scholar] [CrossRef]
- Grumolato, L.; Ghzili, H.; Montero-Hadjadje, M.; Gasman, S.; Lesage, J.; Tanguy, Y.; Galas, L.; Ait-Ali, D.; Leprince, J.; Guérineau, N.C.; et al. Selenoprotein T is a PACAP-regulated gene involved in intracellular Ca2+ mobilization and neuroendocrine secretion. FASEB J. 2008, 22, 1756–1768. [Google Scholar] [CrossRef]
- Koseoglu, E.; Kutuk, B.; Nalbantoglu, O.U.; Koseoglu, R.; Kendirci, M. Arsenic and selenium measurements in nail and hair show important relationships to Alzheimer’s disease in the elderly. J. Trace Elem. Med. Biol. 2021, 64, 126684. [Google Scholar] [CrossRef]
- Varikasuvu, S.R.; Prasad, V.S.; Kothapalli, J.; Manne, M. Brain Selenium in Alzheimer’s Disease (BRAIN SEAD Study): A Systematic Review and Meta-Analysis. Biol. Trace Elem. Res. 2019, 189, 361–369. [Google Scholar] [CrossRef]
- Martini, F.; Rosa, S.G.; Klann, I.P.; Fulco, B.; Carvalho, F.B.; Rahmeier, F.L.; Fernandes, M.C.; Nogueira, C.W. A multifunctional compound ebselen reverses memory impairment, apoptosis and oxidative stress in a mouse model of sporadic Alzheimer’s disease. J. Psychiatr. Res. 2019, 109, 107–117. [Google Scholar] [CrossRef]
- Zhang, Z.H.; Chen, C.; Jia, S.Z.; Cao, X.C.; Liu, M.; Tian, J.; Hoffmann, P.R.; Xu, H.X.; Ni, J.Z.; Song, G.L. Selenium Restores Synaptic Deficits by Modulating NMDA Receptors and Selenoprotein K in an Alzheimer’s Disease Model. Antioxid. Redox Signal. 2021, 35, 863–884. [Google Scholar] [CrossRef]
- Plascencia-Villa, G.; Perry, G. Preventive and Therapeutic Strategies in Alzheimer’s Disease: Focus on Oxidative Stress, Redox Metals, and Ferroptosis. Antioxid. Redox Signal. 2021, 34, 591–610. [Google Scholar] [CrossRef] [PubMed]
- Socha, K.; Klimiuk, K.; Naliwajko, S.K.; Soroczyńska, J.; Puścion-Jakubik, A.; Markiewicz-Żukowska, R.; Kochanowicz, J. Dietary Habits, Selenium, Copper, Zinc and Total Antioxidant Status in Serum in Relation to Cognitive Functions of Patients with Alzheimer’s Disease. Nutrients 2021, 13, 287. [Google Scholar] [CrossRef] [PubMed]
- Vinceti, M.; Michalke, B.; Malagoli, C.; Eichmüller, M.; Filippini, T.; Tondelli, M.; Bargellini, A.; Vinceti, G.; Zamboni, G.; Chiari, A. Selenium and selenium species in the etiology of Alzheimer’s dementia: The potential for bias of the case-control study design. J. Trace Elem. Med. Biol. 2019, 53, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, B.R.; Hare, D.J.; Bush, A.I.; Li, Q.X.; Fowler, C.J.; Masters, C.L.; Martins, R.N.; Ganio, K.; Lothian, A.; Mukherjee, S.; et al. AIBL research group. Selenium Levels in Serum, Red Blood Cells, and Cerebrospinal Fluid of Alzheimer’s Disease Patients: A Report from the Australian Imaging, Biomarker & Lifestyle Flagship Study of Ageing (AIBL). J. Alzheimer’s Dis. 2017, 57, 183–193. [Google Scholar]
- Meseguer, I.; Molina, J.A.; Jiménez-Jiménez, F.J.; Aguilar, M.V.; Mateos-Vega, C.J.; González-Muñoz, M.J.; de Bustos, F.; Ortí-Pareja, M.; Zurdo, M.; Berbel, A.; et al. Cerebrospinal fluid levels of selenium in patients with Alzheimer’s disease. J. Neural Transm. 1999, 106, 309–315. [Google Scholar] [CrossRef]
- Gerhardsson, L.; Blennow, K.; Lundh, T.; Londos, E.; Minthon, L. Concentrations of metals, beta-amyloid and tau-markers in cerebrospinal fluid in patients with Alzheimer’s disease. Dement. Geriatr. Cogn. Disord. 2009, 28, 88–94. [Google Scholar] [CrossRef]
- Cardoso, B.R.; Roberts, B.R.; Malpas, C.B.; Vivash, L.; Genc, S.; Saling, M.M.; Desmond, P.; Steward, C.; Hicks, R.J.; Callahan, J.; et al. Supranutritional Sodium Selenate Supplementation Delivers Selenium to the Central Nervous System: Results from a Randomized Controlled Pilot Trial in Alzheimer’s Disease. Neurotherapeutics 2019, 16, 192–202. [Google Scholar] [CrossRef]
- Tamtaji, O.R.; Heidari-Soureshjani, R.; Mirhosseini, N.; Kouchaki, E.; Bahmani, F.; Aghadavod, E.; Tajabadi-Ebrahimi, M.; Asemi, Z. Probiotic and selenium co-supplementation, and the effects on clinical, metabolic and genetic status in Alzheimer’s disease: A randomized, double-blind, controlled trial. Clin. Nutr. 2019, 38, 2569–2575. [Google Scholar] [CrossRef]
- Zecca, L.; Stroppolo, A.; Gatti, A.; Tampellini, D.; Toscani, M.; Gallorini, M.; Giaveri, G.; Arosio, P.; Santambrogio, P.; Fariello, R.G.; et al. The role of iron and copper molecules in the neuronal vulnerability of locus coeruleus and substantia nigra during aging. Proc. Natl. Acad. Sci. USA 2004, 101, 9843–9848. [Google Scholar] [CrossRef]
- Venkateshappa, C.; Harish, G.; Mythri, R.B.; Mahadevan, A.; Bharath, M.M.; Shankar, S.K. Increased oxidative damage and decreased antioxidant function in aging human substantia nigra compared to striatum: Implications for Parkinson’s disease. Neurochem. Res. 2012, 37, 358–369. [Google Scholar] [CrossRef]
- Gaki, G.S.; Papavassiliou, A.G. Oxidative stress-induced signaling pathways implicated in the pathogenesis of Parkinson’s disease. Neuromol. Med. 2014, 16, 217–230. [Google Scholar] [CrossRef] [PubMed]
- Recasens, A.; Dehay, B. Alpha-synuclein spreading in Parkinson’s disease. Front. Neuroanat. 2014, 8, 159. [Google Scholar] [CrossRef] [PubMed]
- Park, H.A.; Ellis, A.C. Dietary Antioxidants and Parkinson’s Disease. Antioxidants 2020, 9, 570. [Google Scholar] [CrossRef] [PubMed]
- Hemmati-Dinarvand, M.; Saedi, S.; Valilo, M.; Kalantary-Charvadeh, A.; Alizadeh Sani, M.; Kargar, R.; Safari, H.; Samadi, N. Oxidative stress and Parkinson’s disease: Conflict of oxidant-antioxidant systems. Neurosci. Lett. 2019, 709, 134296. [Google Scholar] [CrossRef] [PubMed]
- Ble, A.; Cherubini, A.; Volpato, S.; Bartali, B.; Walston, J.D.; Windham, B.G.; Bandinelli, S.; Lauretani, F.; Guralnik, J.M.; Ferrucci, L. Lower plasma vitamin E levels are associated with the frailty syndrome: The InCHIANTI study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2006, 61, 278–283. [Google Scholar] [CrossRef]
- Ellwanger, J.H.; Molz, P.; Dallemole, D.R.; Pereira dos Santos, A.; Müller, T.E.; Cappelletti, L.; Gonçalves da Silva, M.; Franke, S.I.; Prá, D.; Pêgas Henriques, J.A. Selenium reduces bradykinesia and DNA damage in a rat model of Parkinson’s disease. Nutrition 2015, 31, 359–365. [Google Scholar] [CrossRef]
- Adani, G.; Filippini, T.; Michalke, B.; Vinceti, M. Selenium and Other Trace Elements in the Etiology of Parkinson’s Disease: A Systematic Review and Meta-Analysis of Case-Control Studies. Neuroepidemiology 2020, 54, 1–23. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Sun, H.L.; Wang, T.; Liu, X.X.; Liu, C.; Li, W.J.; Li, X. Selenium level does not differ in blood but increased in cerebrospinal fluid in Parkinson’s disease: A meta-analysis. Int. J. Neurosci. 2021, 131, 95–101. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, R.P.; Cheng, W.H.; Zhu, J.H. Prioritized brain selenium retention and selenoprotein expression: Nutritional insights into Parkinson’s disease. Mech. Ageing Dev. 2019, 180, 89–96. [Google Scholar] [CrossRef]
- Maass, F.; Michalke, B.; Willkommen, D.; Schulte, C.; Tönges, L.; Boerger, M.; Zerr, I.; Bähr, M.; Lingor, P. Selenium speciation analysis in the cerebrospinal fluid of patients with Parkinson’s disease. J. Trace Elem. Med. Biol. 2020, 57, 126412. [Google Scholar] [CrossRef]
- Sampson, T.R.; Debelius, J.W.; Thron, T.; Janssen, S.; Shastri, G.G.; Ilhan, Z.E.; Challis, C.; Schretter, C.E.; Rocha, S.; Gradinaru, V.; et al. Gut Microbiota Regulate Motor Deficits and Neuroinflammation in a Model of Parkinson’s Disease. Cell 2016, 167, 1469–1480.e12. [Google Scholar] [CrossRef]
- Ma, J.; Yan, L.; Guo, T.; Yang, S.; Liu, Y.; Xie, Q.; Ni, D.; Wang, J. Association between Serum Essential Metal Elements and the Risk of Schizophrenia in China. Sci. Rep. 2020, 10, 10875. [Google Scholar] [CrossRef]
- Pitts, M.W.; Raman, A.V.; Berry, M.J. Schizophrenia, Oxidative Stress and Selenium. In Selenium. Its Molecular Biology and Role in Human Health, 3rd ed.; Hatfield, D.L., Berry, M.J., Gladyshev, V.N., Eds.; Springer: London, UK, 2012; pp. 355–367. [Google Scholar]
- Looney, J.M.; Childs, H.M. The Lactic Acid and Glutathione Content of the Blood of Schizophrenic Patients. J. Clin. Investig. 1934, 13, 963–968. [Google Scholar] [CrossRef]
- Do, K.Q.; Trabesinger, A.H.; Kirsten-Krüger, M.; Lauer, C.J.; Dydak, U.; Hell, D.; Holsboer, F.; Boesiger, P.; Cuénod, M. Schizophrenia: Glutathione deficit in cerebrospinalfluid and prefrontalcortex in vivo. Eur. J. Neurosci. 2000, 12, 3721–3728. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.K.; Leonard, S.; Reddy, R. Altered glutathione redox state in schizophrenia. Dis. Markers 2006, 22, 83–93. [Google Scholar] [CrossRef]
- Gysin, R.; Kraftsik, R.; Sandell, J.; Bovet, P.; Chappuis, C.; Conus, P.; Deppen, P.; Preisig, M.; Ruiz, V.; Steullet, P.; et al. Impaired glutathione synthesis in schizophrenia: Convergent genetic and functional evidence. Proc. Natl. Acad. Sci. USA 2007, 104, 16621–16626. [Google Scholar] [CrossRef]
- Padurariu, M.; Ciobica, A.; Dobrin, I.; Stefanescu, C. Evaluation of antioxidant enzymes activities and lipid peroxidation in schizophrenic patients treated with typical and atypical antipsychotics. Neurosci. Lett. 2010, 479, 317–320. [Google Scholar] [CrossRef]
- Romero-Ramos, M.; Venero, J.L.; Cano, J.; Machado, A. Low Selenium diet induces tyrosine hydroxylase enzyme in nigrostriatal system of the rat. Brain Res. Mol. Brain Res. 2000, 84, 7–16. [Google Scholar] [CrossRef]
- Tang, Y.L.; Wang, S.W.; Lin, S.M. Both inorganic and organic Selenium supplements can decrease brain monoamine oxidase B enzyme activity in adult rats. Br. J. Nutr. 2008, 100, 660–665. [Google Scholar] [CrossRef]
- Li, Z.; Liu, Y.; Li, X.; Ju, W.; Wu, G.; Yang, X.; Fu, X.; Gao, X. Association of Elements with Schizophrenia and Intervention of Selenium Supplements. Biol. Trace Elem. Res. 2018, 183, 16–21. [Google Scholar] [CrossRef]
- Saghazadeh, A.; Mahmoudi, M.; Shahrokhi, S.; Mojarrad, M.; Dastmardi, M.; Mirbeyk, M.; Rezaei, N. Trace elements in schizophrenia: A systematic review and meta-analysis of 39 studies (n = 5151 participants). Nutr. Rev. 2020, 78, 278–303. [Google Scholar] [CrossRef]
- Czaderny, K. Risk factors for depression. New evidence on selenium deficiency and depressive disorders. Psychiatr. Pol. 2020, 54, 1109–1121. [Google Scholar] [CrossRef]
- Li, Z.; Wang, W.; Xin, X.; Song, X.; Zhang, D. Association of total zinc, iron, copper and selenium intakes with depression in the US adults. J. Affect. Disord. 2018, 228, 68–74. [Google Scholar] [CrossRef]
- Wang, J.; Um, P.; Dickerman, B.A.; Liu, J. Zinc, Magnesium, Selenium and Depression: A Review of the Evidence, Potential Mechanisms and Implications. Nutrients 2018, 10, 584. [Google Scholar] [CrossRef]
- Ferreira de Almeida, T.L.; Petarli, G.B.; Cattafesta, M.; Zandonade, E.; Bezerra, O.; Tristão, K.G.; Salaroli, L.B. Association of Selenium Intake and Development of Depression in Brazilian Farmers. Front. Nutr. 2021, 8, 671377. [Google Scholar] [CrossRef]
- Turan, E.; Karaaslan, O. The Relationship between Iodine and Selenium Levels with Anxiety and Depression in Patients with Euthyroid Nodular Goiter. Oman Med. J. 2020, 35, e161. [Google Scholar] [CrossRef]
- Salehi-Abargouei, A.; Esmaillzadeh, A.; Azadbakht, L.; Keshteli, A.H.; Afshar, H.; Feizi, A.; Feinle-Bisset, C.; Adibi, P. Do patterns of nutrient intake predict self-reported anxiety, depression and psychological distress in adults? SEPAHAN study. Clin. Nut. 2019, 38, 940–947. [Google Scholar] [CrossRef]
- Jacobs, E.T.; Lance, P.; Mandarino, L.J.; Ellis, N.A.; Chow, H.S.; Foote, J.; Martinez, J.A.; Hsu, C.P.; Batai, K.; Saboda, K.; et al. Selenium supplementation and insulin resistance in a randomized, clinical trial. BMJ Open Diabetes Res. Care 2019, 7, e000613. [Google Scholar] [CrossRef]
- Vinceti, M.; Filippini, T.; Rothman, K.J. Selenium exposure and the risk of type 2 diabetes: A systematic review and meta-analysis. Eur. J. Epidemiol. 2018, 33, 789–810. [Google Scholar] [CrossRef]
- Kim, J.; Chung, H.S.; Choi, M.K.; Roh, Y.K.; Yoo, H.J.; Park, J.H.; Kim, D.S.; Yu, J.M.; Moon, S. Association between Serum Selenium Level and the Presence of Diabetes Mellitus: A Meta-Analysis of Observational Studies. Diabetes Metab. J. 2019, 43, 447–460. [Google Scholar] [CrossRef]
- Kohler, L.N.; Foote, J.; Kelley, C.P.; Florea, A.; Shelly, C.; Chow, H.S.; Hsu, P.; Batai, K.; Ellis, N.; Saboda, K.; et al. Selenium and Type 2 Diabetes: Systematic Review. Nutrients 2018, 10, 1924. [Google Scholar] [CrossRef]
- Dubey, P.; Thakur, V.; Chattopadhyay, M. Role of Minerals and Trace Elements in Diabetes and Insulin Resistance. Nutrients 2020, 12, 1864. [Google Scholar] [CrossRef]
- Siddiqi, S.M.; Sun, C.; Wu, X.; Shah, I.; Mehmood, A. The Correlation between Dietary Selenium Intake and Type 2 Diabetes: A Cross-Sectional Population-Based Study on North Chinese Adults. BioMed Res. Int. 2020, 2020, 8058463. [Google Scholar] [CrossRef]
- Wongdokmai, R.; Shantavasinkul, P.C.; Chanprasertyothin, S.; Panpunuan, P.; Matchariyakul, D.; Sritara, P.; Sirivarasai, J. The Involvement of Selenium in Type 2 Diabetes Development Related to Obesity and Low Grade Inflammation. Diabetes Metab. Synd. Obe. 2021, 14, 1669–1680. [Google Scholar] [CrossRef]
- Liao, X.L.; Wang, Z.H.; Liang, X.N.; Liang, J.; Wei, X.B.; Wang, S.H.; Guo, W.X. The Association of Circulating Selenium Concentrations with Diabetes Mellitus. Diabetes Metab. Synd. Obes. 2020, 13, 4755–4761. [Google Scholar] [CrossRef]
- Vinceti, M.; Bonaccio, M.; Filippini, T.; Costanzo, S.; Wise, L.A.; Di Castelnuovo, A.; Ruggiero, E.; Persichillo, M.; Cerletti, C.; Donati, M.B.; et al. Moli-sani Study Investigators. Dietary selenium intake and risk of hospitalization for type 2 diabetes in the Moli-sani study cohort. Nutr. Metab. Cardiovasc. Dis. 2021, 31, 1738–1746. [Google Scholar] [CrossRef]
- Stranges, S.; Rayman, M.P.; Winther, K.H.; Guallar, E.; Cold, S.; Pastor-Barriuso, R. Effect of selenium supplementation on changes in HbA1c: Results from a multiple-dose, randomized controlled trial. Diabetes Obes. Metab. 2019, 21, 541–549. [Google Scholar] [CrossRef]
- Huang, J.Q.; Zhou, J.C.; Wu, Y.Y.; Ren, F.Z.; Lei, X.G. Role of glutathione peroxidase 1 in glucose and lipid metabolism-related diseases. Free Radic. Biol. Med. 2018, 127, 108–115. [Google Scholar] [CrossRef]
- May, J.M.; de Haën, C. Insulin-stimulated intracellular hydrogen peroxide production in rat epididymal fat cells. J. Biol. Chem. 1979, 254, 2214–2220. [Google Scholar] [CrossRef]
- Wang, X.D.; Vatamaniuk, M.Z.; Wang, S.K.; Roneker, C.A.; Simmons, R.A.; Lei, X.G. Molecular mechanisms for hyperinsulinaemia induced by overproduction of selenium-dependent glutathione peroxidase-1 in mice. Diabetologia 2008, 51, 1515–1524. [Google Scholar] [CrossRef]
- Loh, K.; Deng, H.; Fukushima, A.; Cai, X.; Boivin, B.; Galic, S.; Bruce, C.; Shields, B.J.; Skiba, B.; Ooms, L.M.; et al. Reactive oxygen species enhance insulin sensitivity. Cell Metab. 2009, 10, 260–272. [Google Scholar] [CrossRef]
- Schoenmakers, E.; Agostini, M.; Mitchell, C.; Schoenmakers, N.; Papp, L.; Rajanayagam, O.; Padidela, R.; Ceron-Gutierrez, L.; Doffinger, R.; Prevosto, C.; et al. Mutations in the selenocysteine insertion sequence-binding protein 2 gene lead to a multisystem selenoprotein deficiency disorder in humans. J. Clin. Investig. 2010, 120, 4220–4235. [Google Scholar] [CrossRef]
- Yang, S.J.; Hwang, S.Y.; Choi, H.Y.; Yoo, H.J.; Seo, J.A.; Kim, S.G.; Kim, N.H.; Baik, S.H.; Choi, D.S.; Choi, K.M. Serum selenoprotein P levels in patients with type 2 diabetes and prediabetes: Implications for insulin resistance, inflammation, and atherosclerosis. J. Clin. Endocrinol. Metab. 2011, 96, E1325–E1329. [Google Scholar] [CrossRef]
- Misu, H.; Takamura, T.; Takayama, H.; Hayashi, H.; Matsuzawa-Nagata, N.; Kurita, S.; Ishikura, K.; Ando, H.; Takeshita, Y.; Ota, T.; et al. A liver-derived secretory protein, selenoprotein P, causes insulin resistance. Cell Metab. 2010, 12, 483–495. [Google Scholar] [CrossRef]
- Schomburg, L. Sex-Specific Differences in Biological Effects and Metabolism of Selenium. In Selenium. Its Molecular Biology and Role in Human Health, 4th ed.; Hatfield, D.L., Schweizer, U., Tsuji, P.A., Gladyshev, V.N., Eds.; Springer International Publishing: New York, NY, USA, 2016; pp. 377–388. [Google Scholar]
- Waters, D.J.; Chiang, E.C.; Cooley, D.M.; Morris, J.S. Making sense of sex and supplements: Differences in the anticarcinogenic effects of Selenium in men and women. Mutat. Res. 2004, 551, 91–107. [Google Scholar] [CrossRef]
- Galan, P.; Briançon, S.; Favier, A.; Bertrais, S.; Preziosi, P.; Faure, H.; Arnaud, J.; Arnault, N.; Czernichow, S.; Mennen, L.; et al. Antioxidant status and risk of cancer in the SU.VI.MAX study: Is the effect of supplementation dependent on baseline levels? Br. J. Nutr. 2005, 94, 125–132. [Google Scholar] [CrossRef]
- Kok, F.J.; de Bruijn, A.M.; Hofman, A.; Vermeeren, R.; Valkenburg, H.A. Is serum Selenium a risk factor for cancer in men only? Am. J. Epidemiol. 1987, 125, 12–16. [Google Scholar] [CrossRef]
- Knekt, P.; Aromaa, A.; Maatela, J.; Alfthan, G.; Aaran, R.K.; Hakama, M.; Hakulinen, T.; Peto, R.; Teppo, L. Serum Selenium and subsequent risk of cancer among Finnish men and women. J. Natl. Cancer Inst. 1990, 82, 864–868. [Google Scholar] [CrossRef]
- Amaral, A.F.; Cantor, K.P.; Silverman, D.T.; Malats, N. Selenium and bladder cancer risk: A meta-analysis. Cancer Epidemiol. Biomark. Prev. 2010, 19, 2407–2415. [Google Scholar] [CrossRef]
- Michaud, D.S.; De Vivo, I.; Morris, J.S.; Giovannucci, E. Toenail Selenium concentrations and bladder cancer risk in women and men. Br. J. Cancer 2005, 93, 804–806. [Google Scholar] [CrossRef][Green Version]
- Hughes, D.J.; Fedirko, V.; Jenab, M.; Schomburg, L.; Méplan, C.; Freisling, H.; Bueno-de-Mesquita, H.B.; Hybsier, S.; Becker, N.P.; Czuban, M.; et al. Selenium status is associated with colorectal cancer risk in the European prospective investigation of cancer and nutrition cohort. Int. J. Cancer Res. 2015, 136, 1149–1161. [Google Scholar] [CrossRef] [PubMed]
- Takata, Y.; Kristal, A.R.; King, I.B.; Song, X.; Diamond, A.M.; Foster, C.B.; Hutter, C.M.; Hsu, L.; Duggan, D.J.; Langer, R.D.; et al. Serum selenium, genetic variation in selenoenzymes, and risk of colorectal cancer: Primary analysis from the Women’s Health Initiative Observational Study and meta-analysis. Cancer Epidemiol. Biomark. Prev. 2011, 20, 1822–1830. [Google Scholar] [CrossRef] [PubMed]
- Toulis, K.A.; Anastasilakis, A.D.; Tzellos, T.G.; Goulis, D.G.; Kouvelas, D. Selenium supplementation in the treatment of Hashimoto’s thyroiditis: A systematic review and a meta-analysis. Thyroid 2010, 20, 1163–1173. [Google Scholar] [CrossRef] [PubMed]
- Duntas, L.H.; Mantzou, E.; Koutras, D.A. Effects of a six month treatment with selenomethionine in patients with autoimmune thyroiditis. Eur. J. Endocrinol. 2003, 148, 389–393. [Google Scholar] [CrossRef]
- Rasmussen, L.B.; Schomburg, L.; Köhrle, J.; Pedersen, I.B.; Hollenbach, B.; Hög, A.; Ovesen, L.; Perrild, H.; Laurberg, P. Selenium status, thyroid volume, and multiple nodule formation in an area with mild iodine deficiency. Eur. J. Endocrinol. 2011, 164, 585–590. [Google Scholar] [CrossRef]
- Derumeaux, H.; Valeix, P.; Castetbon, K.; Bensimon, M.; Boutron-Ruault, M.C.; Arnaud, J.; Hercberg, S. Association of selenium with thyroid volume and echostructure in 35- to 60-year-old French adults. Eur. J. Endocrinol. 2003, 148, 309–315. [Google Scholar] [CrossRef]
- Nawrot, T.S.; Staessen, J.A.; Roels, H.A.; Den Hond, E.; Thijs, L.; Fagard, R.H.; Dominiczak, A.F.; Struijker-Boudier, H.A. Blood pressure and blood Selenium: A cross-sectional and longitudinal population study. Eur. Heart J. 2007, 28, 628–633. [Google Scholar] [CrossRef]
- Riese, C.; Michaelis, M.; Mentrup, B.; Götz, F.; Köhrle, J.; Schweizer, U.; Schomburg, L. Selenium-dependent pre- and posttranscriptional mechanisms are responsible for sexual dimorphic expression of selenoproteins in murine tissues. Endocrinology 2006, 147, 5883–5892. [Google Scholar] [CrossRef]
- Schomburg, L.; Riese, C.; Renko, K.; Schweizer, U. Effect of age on sexually dimorphic selenoprotein expression in mice. Biol. Chem. 2007, 388, 1035–1041. [Google Scholar] [CrossRef]
- Seale, L.A.; Ogawa-Wong, A.N.; Berry, M.J. Sexual dimorphism in selenium metabolism and selenoproteins. Free Radic. Biol. Med. 2018, 127, 198–205. [Google Scholar] [CrossRef]
- Mauvais-Jarvis, F.; BaireyMerz, N.; Barnes, P.J.; Brinton, R.D.; Carrero, J.J.; DeMeo, D.L.; De Vries, G.J.; Epperson, C.N.; Govindan, R.; Klein, S.L.; et al. Sex and gender: Modifiers of health, disease, and medicine. Lancet 2020, 396, 565–582. [Google Scholar] [CrossRef]
- Bartz, D.; Chitnis, T.; Kaiser, U.B.; Rich-Edwards, J.W.; Rexrode, K.M.; Pennell, P.B.; Goldstein, J.M.; O’Neal, M.A.; LeBoff, M.; Behn, M.; et al. Clinical Advances in Sex- and Gender-Informed Medicine to Improve the Health of All: A Review. JAMA Intern. Med. 2020, 180, 574–583. [Google Scholar] [CrossRef]
- McClung, J.P.; Roneker, C.A.; Mu, W.; Lisk, D.J.; Langlais, P.; Liu, F.; Lei, X.G. Development of insulin resistance and obesity in mice overexpressing cellular glutathione peroxidase. Proc. Natl. Acad. Sci. USA 2004, 101, 8852–8857. [Google Scholar] [CrossRef] [PubMed]
- Steinbrenner, H.; Speckmann, B.; Pinto, A.; Sies, H. High selenium intake and increased diabetes risk: Experimental evidence for interplay between selenium and carbohydrate metabolism. J. Clin. Biochem. Nutr. 2011, 48, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Mahadev, K.; Motoshima, H.; Wu, X.; Ruddy, J.M.; Arnold, R.S.; Cheng, G.; Lambeth, J.D.; & Goldstein, B.J. The NAD(P)H oxidase homolog Nox4 modulates insulin-stimulated generation of H2O2 and plays an integral role in insulin signal transduction. Mol. Cell. Biol. 2004, 24, 1844–1854. [Google Scholar] [CrossRef]
- Sies, H.; Jones, D.P. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat. Rev. Mol. 2020, 21, 363–383. [Google Scholar] [CrossRef]
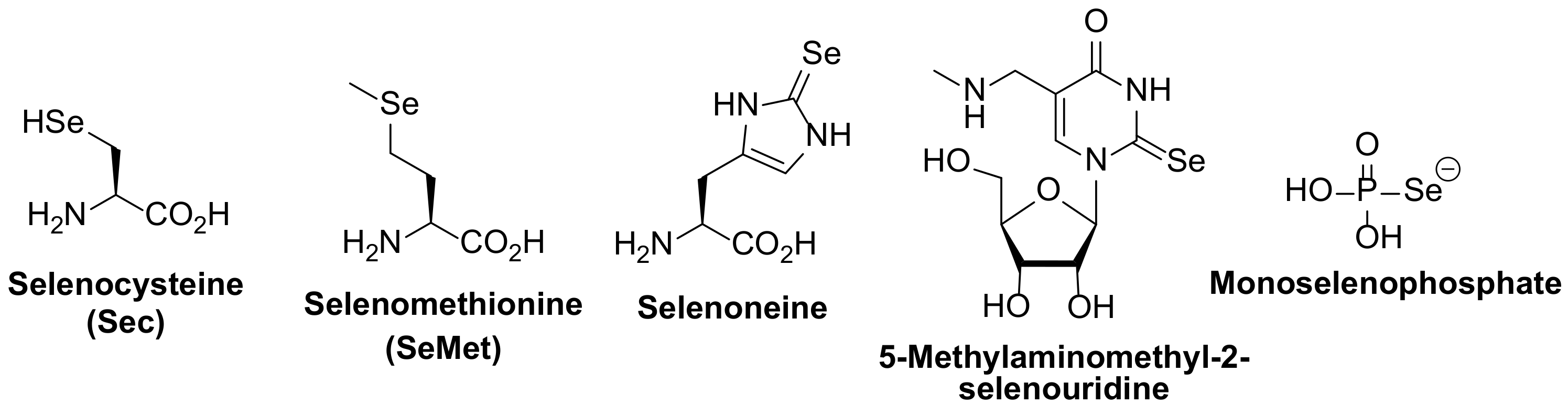

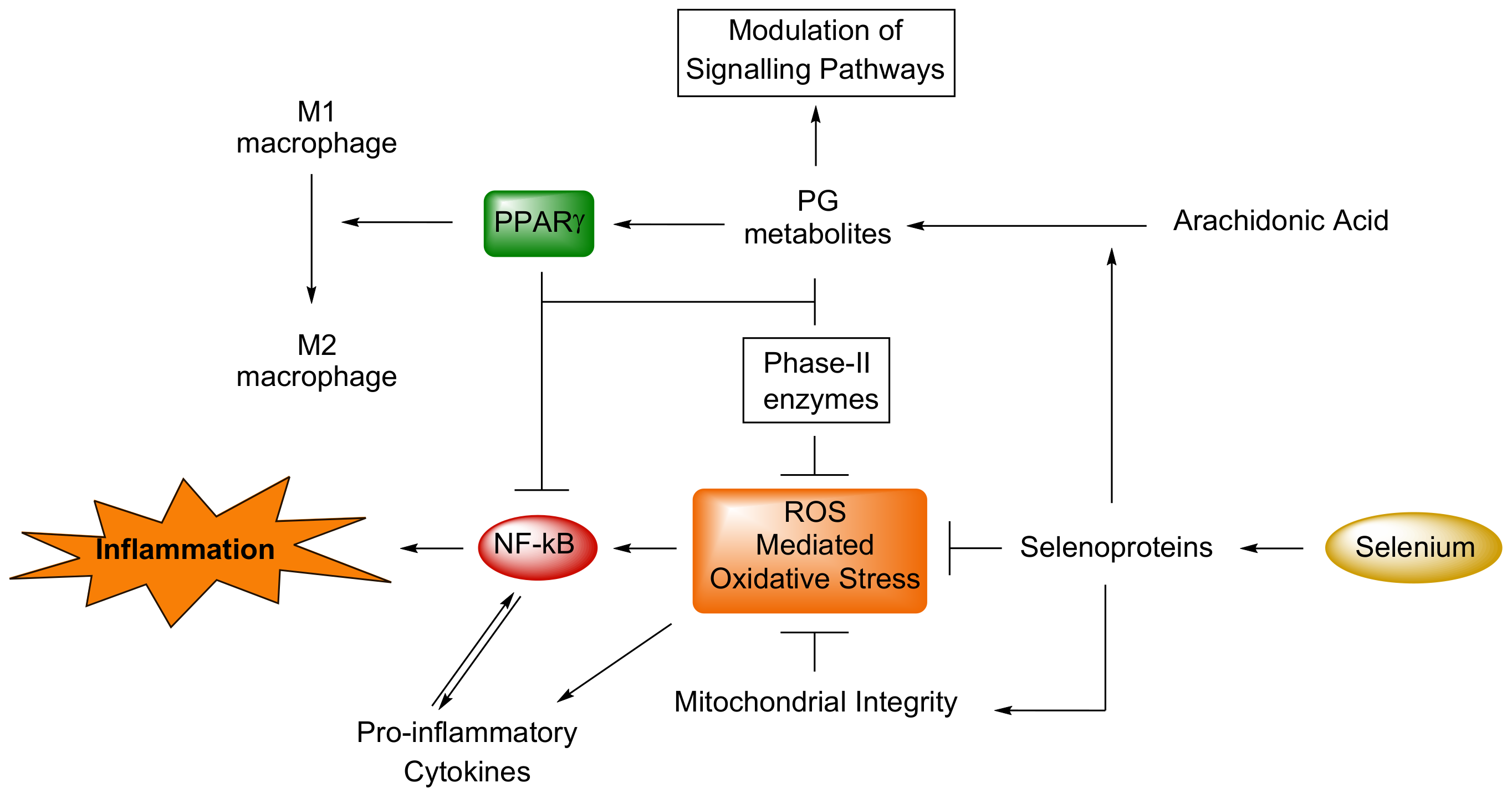
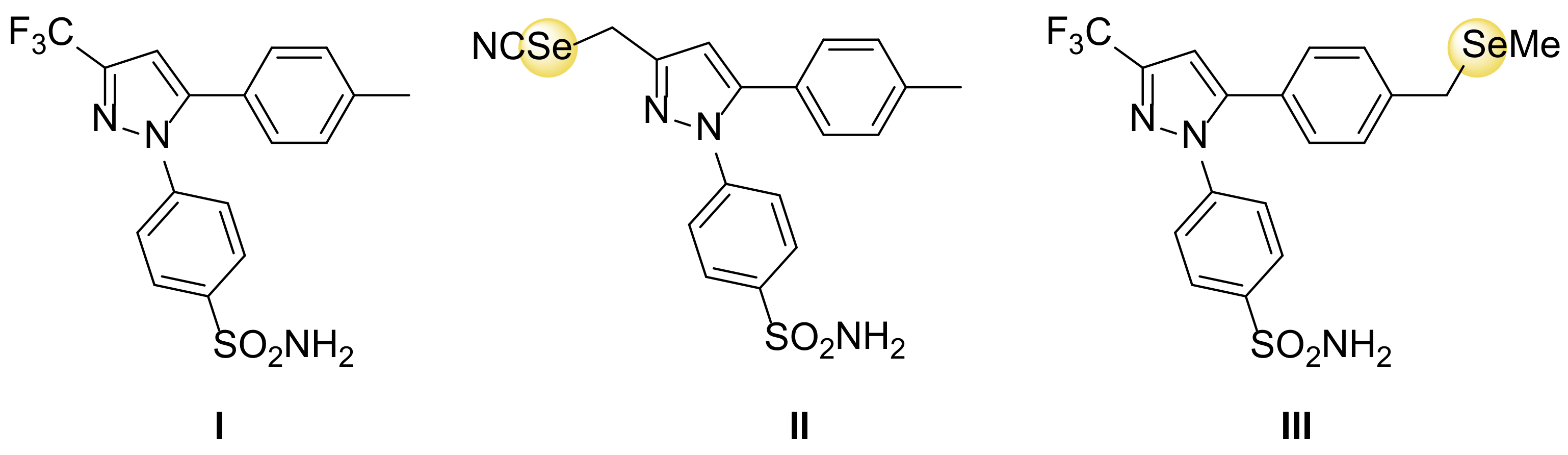
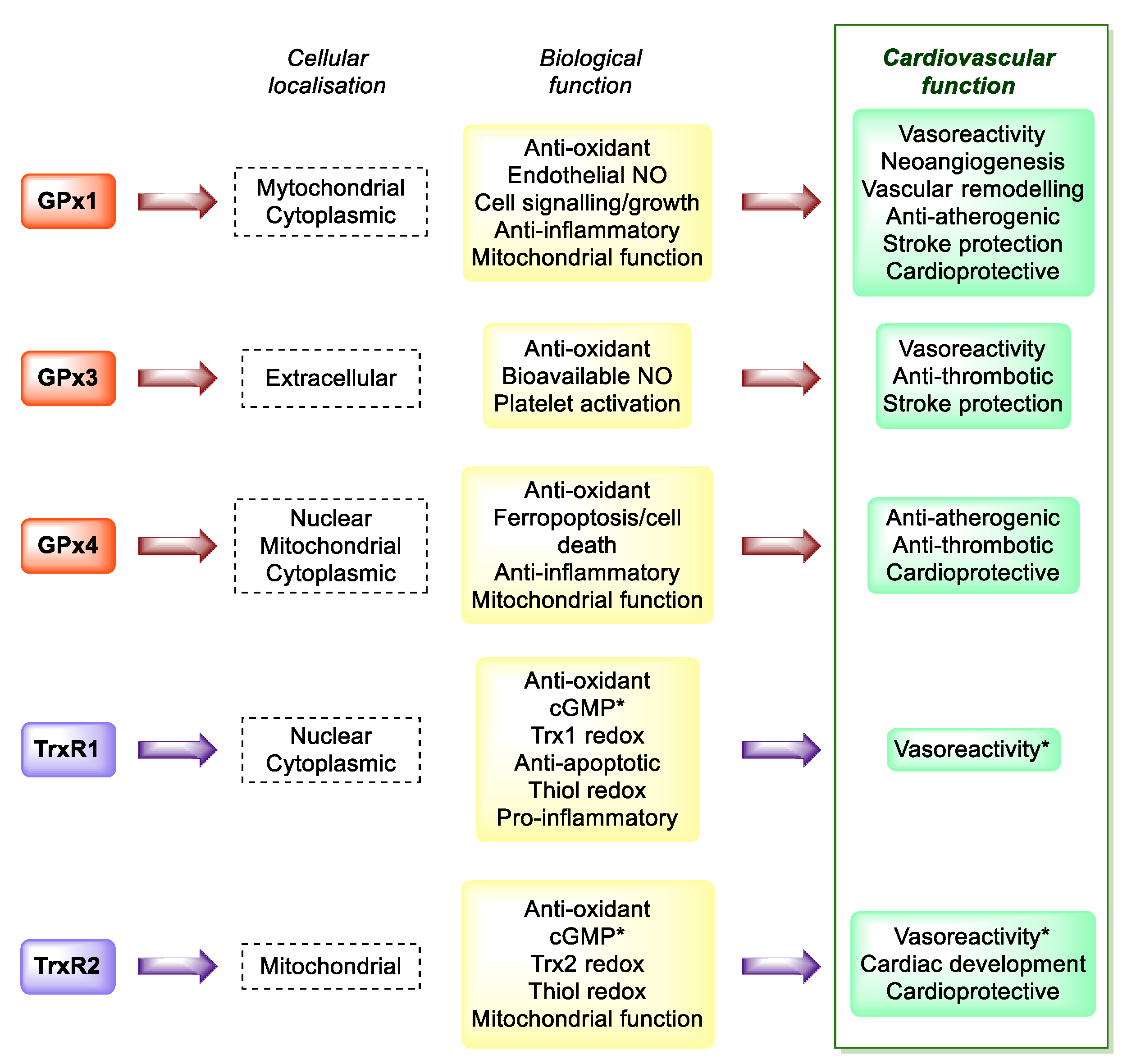

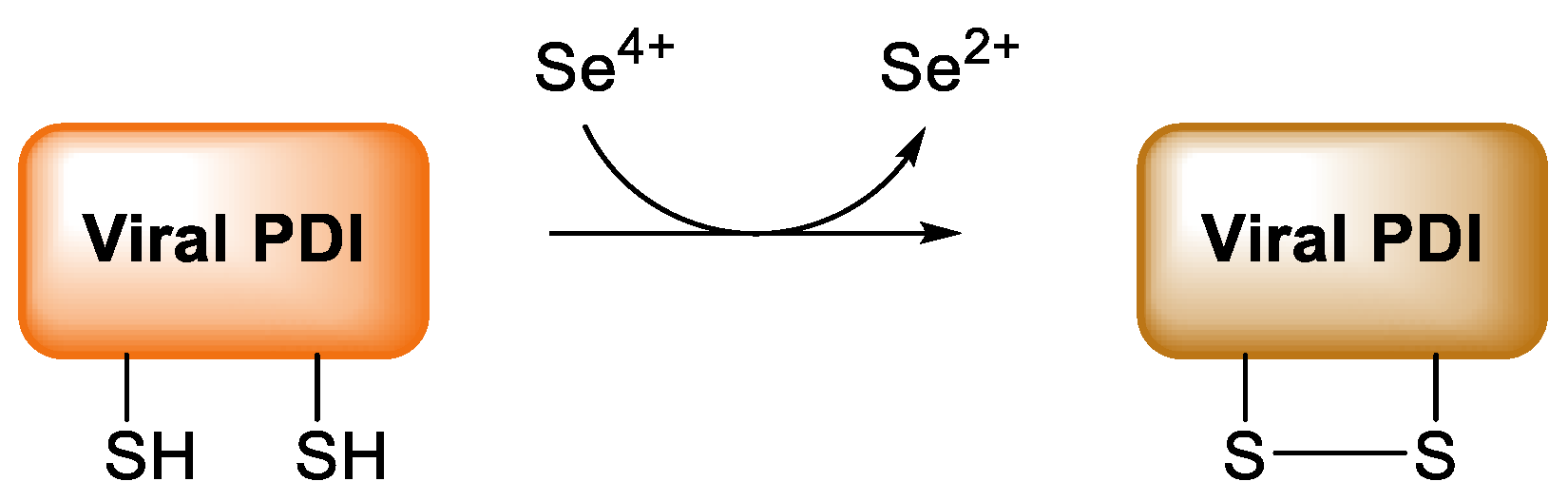
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barchielli, G.; Capperucci, A.; Tanini, D. The Role of Selenium in Pathologies: An Updated Review. Antioxidants 2022, 11, 251. https://doi.org/10.3390/antiox11020251
Barchielli G, Capperucci A, Tanini D. The Role of Selenium in Pathologies: An Updated Review. Antioxidants. 2022; 11(2):251. https://doi.org/10.3390/antiox11020251
Chicago/Turabian StyleBarchielli, Giulia, Antonella Capperucci, and Damiano Tanini. 2022. "The Role of Selenium in Pathologies: An Updated Review" Antioxidants 11, no. 2: 251. https://doi.org/10.3390/antiox11020251
APA StyleBarchielli, G., Capperucci, A., & Tanini, D. (2022). The Role of Selenium in Pathologies: An Updated Review. Antioxidants, 11(2), 251. https://doi.org/10.3390/antiox11020251







