Novel Therapeutic Approaches Enhance PGC1-alpha to Reduce Oxidant Stress-Inflammatory Signaling and Improve Functional Recovery in Hibernating Myocardium
Abstract
1. Introduction
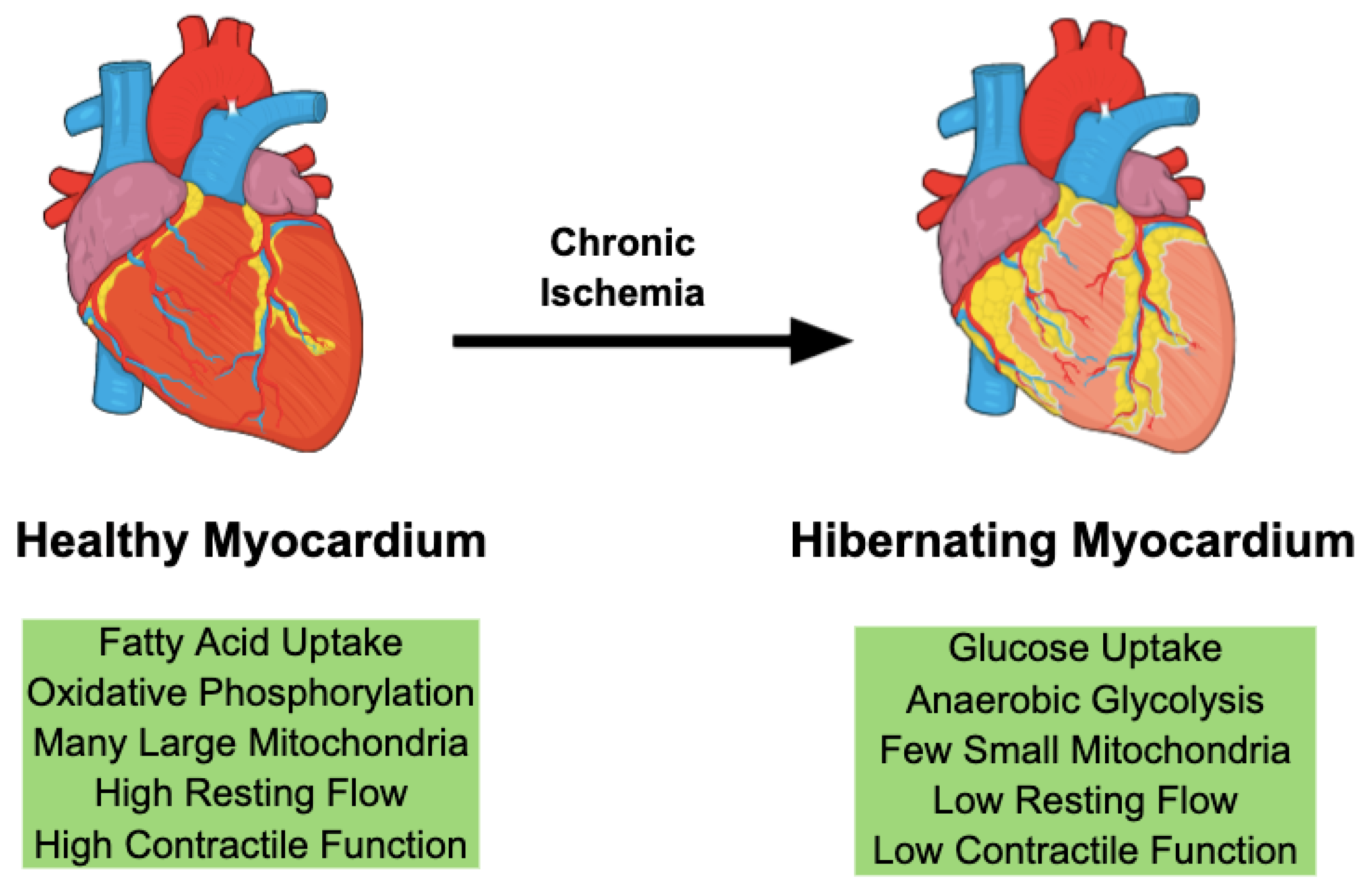
2. Hibernating Myocardium and Revascularization
3. PGC1-alpha Is a Key Mediator of Mitochondrial Energy Metabolism
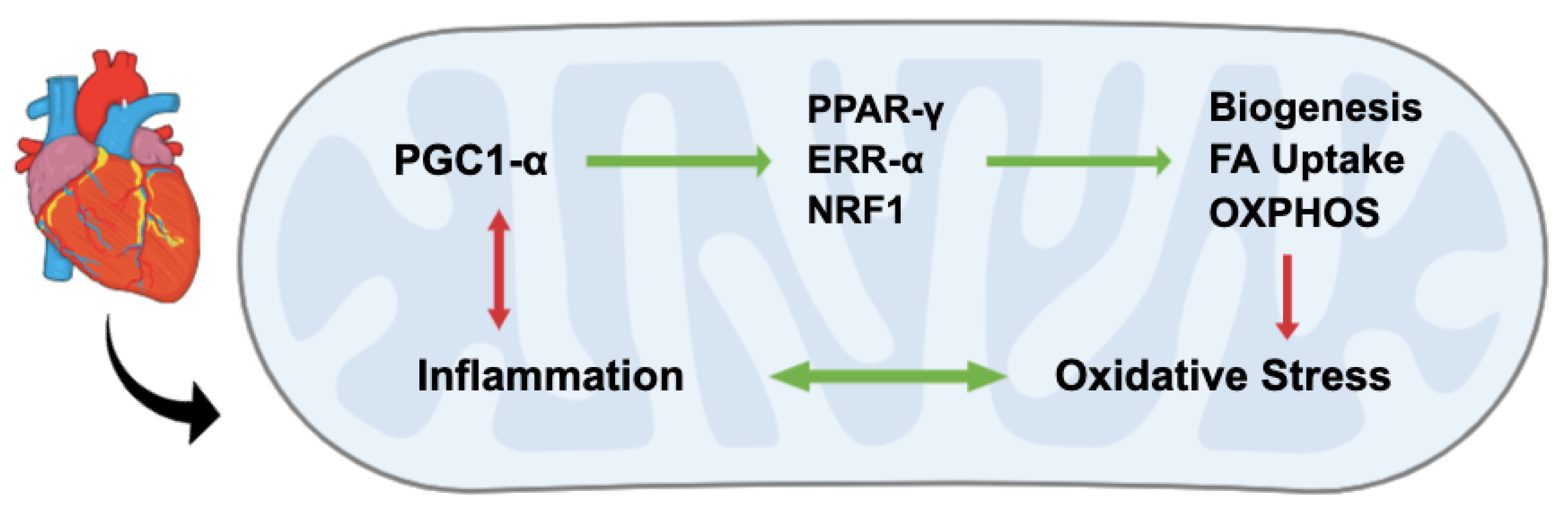
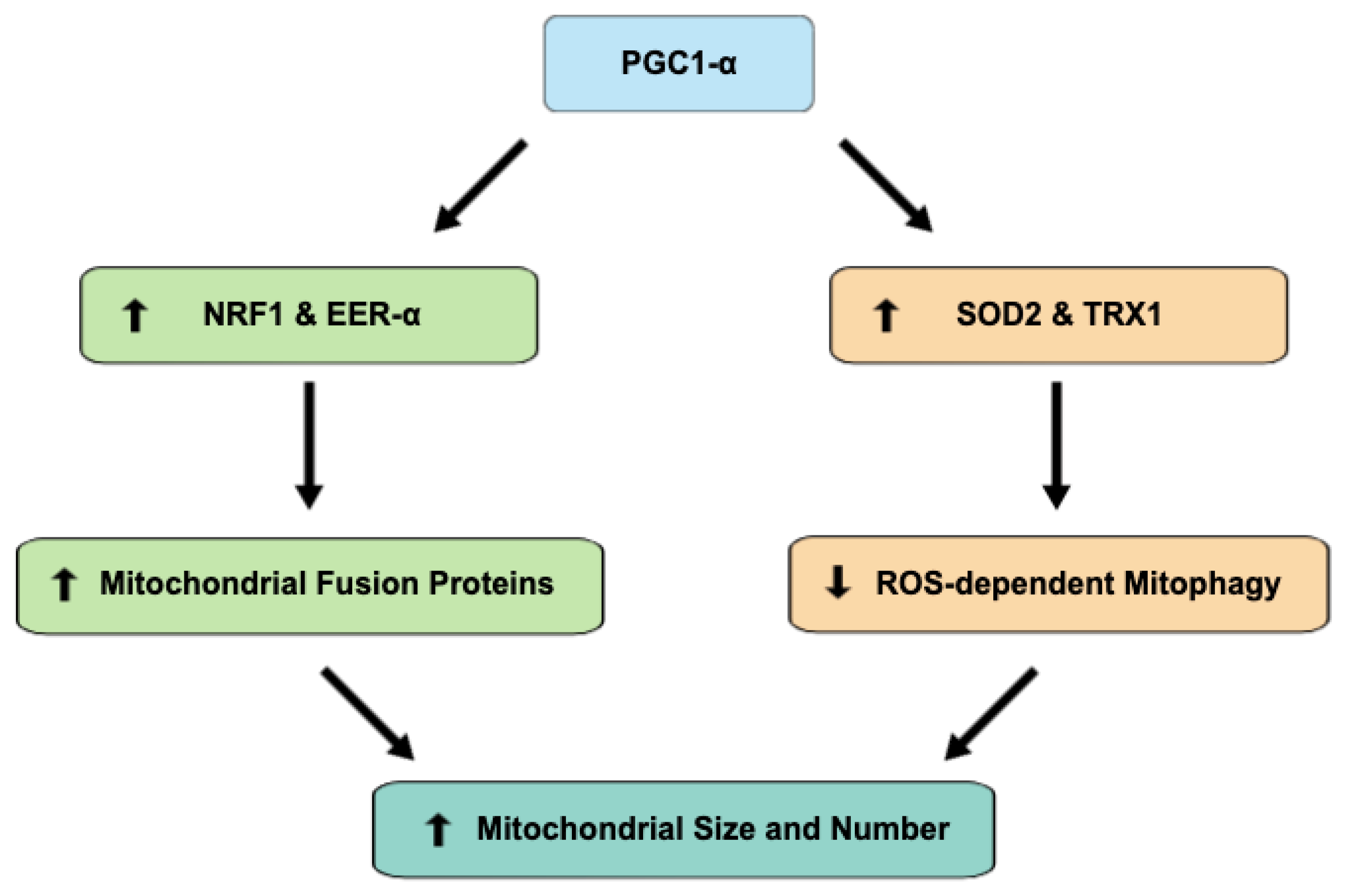
4. PGC1-alpha Regulates Oxidative Stress and Biogenesis
5. PGC1-alpha Links Cellular Metabolism and Inflammation in the Ischemic Heart
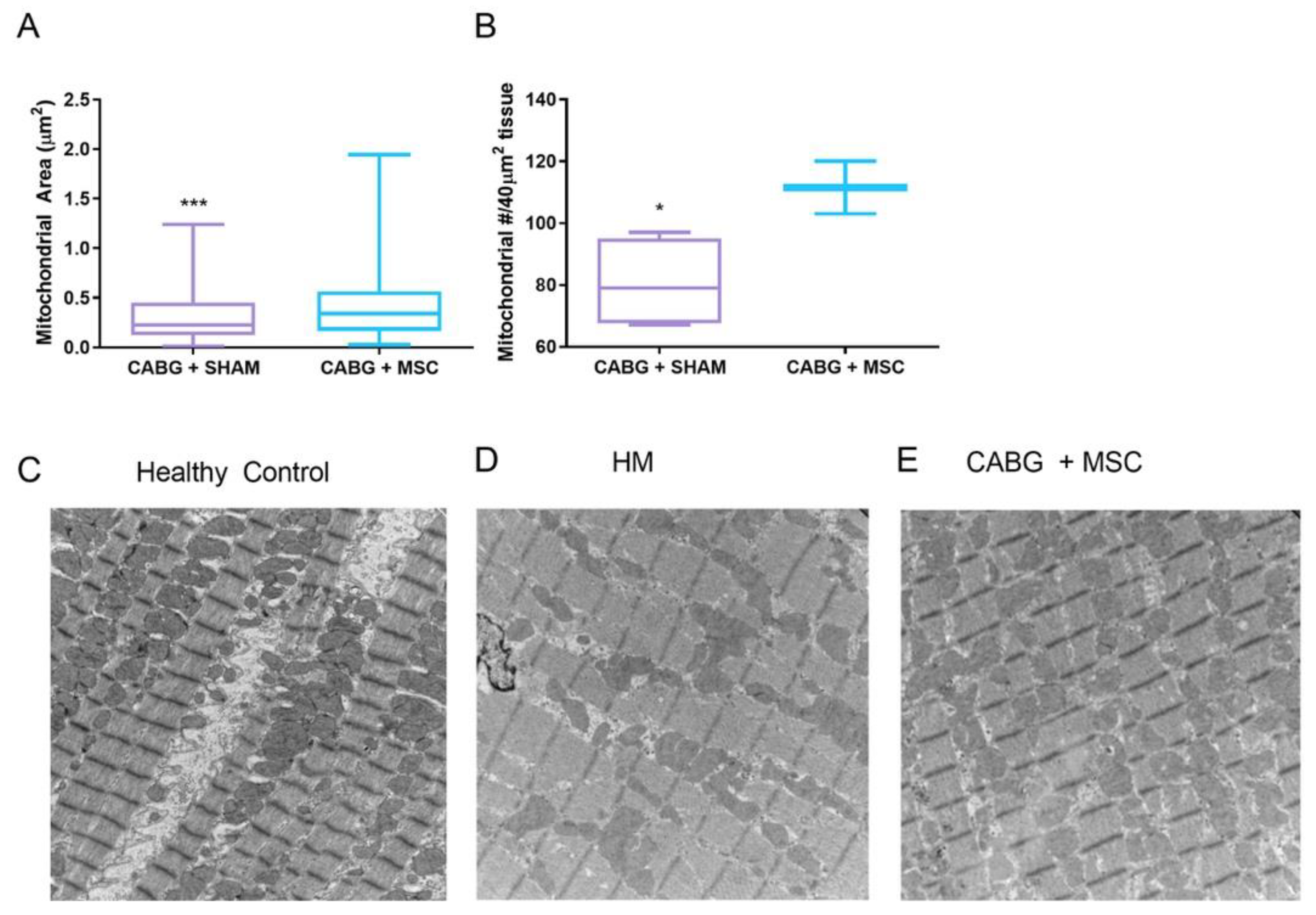
6. Enhanced PGC1-alpha Signaling Improves Recovery in Hibernating Myocardium
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Khan, M.A.; Hashim, M.J.; Mustafa, H.; Baniyas, M.Y.; Al Suwaidi SK, B.M.; AlKatheeri, R.; Alblooshi, F.M.K.; Almatrooshi, E.A.H.; Alzaabi, E.H.; Al Darmaki, R.S.; et al. Global Epidemiology of Ischemic Heart Disease: Results from the Global Burden of Disease Study. Cureus 2020, 12, e9349. [Google Scholar] [CrossRef] [PubMed]
- Diodato, M.; Chedrawy, E.G. Coronary Artery Bypass Graft Surgery: The Past, Present, and Future of Myocardial Revascularisation. Surg. Res. Pract. 2014, 2014, 726158. [Google Scholar] [CrossRef]
- Velazquez, E.J.; Lee, K.L.; Deja, M.A.; Jain, A.; Sopko, G.; Marchenko, A.; Ali, I.S.; Pohost, G.; Gradinac, S.; Abraham, W.T.; et al. Coronary-Artery Bypass Surgery in Patients with Left Ventricular Dysfunction. N. Engl. J. Med. 2011, 364, 1607–1616. [Google Scholar] [CrossRef] [PubMed]
- Shah, B.N.; Khattar, R.S.; Senior, R. The hibernating myocardium: Current concepts, diagnostic dilemmas, and clinical challenges in the post-STICH era. Eur. Heart J. 2013, 34, 1323–1336. [Google Scholar] [CrossRef] [PubMed]
- Finck, B.N.; Kelly, D.P. Peroxisome proliferator-activated receptor gamma coactivator-1 (PGC-1) regulatory cascade in cardiac physiology and disease. Circulation 2007, 115, 2540–2548. [Google Scholar] [CrossRef] [PubMed]
- Rahimtoola, S.H. The hibernating myocardium. Am. Heart J. 1989, 117, 211–221. [Google Scholar] [CrossRef]
- McFalls, E.O.; Kelly, R.F.; Hu, Q.; Mansoor, A.; Lee, J.; Kuskowski, M.; Sikora, J.; Ward, H.B.; Zhang, J. The energetic state within hibernating myocardium is normal during dobutamine despite inhibition of ATP-dependent potassium channel opening with glibenclamide. Am. J. Physiol. Circ. Physiol. 2007, 293, H2945–H2951. [Google Scholar] [CrossRef]
- Canty, J.M., Jr.; Fallavollita, J.A. Hibernating myocardium. J. Nucl. Cardiol. 2005, 12, 104–119. [Google Scholar] [CrossRef]
- Kelly, R.F.; Sluiter, W.; McFalls, E.O. Hibernating myocardium: Is the program to survive a pathway to failure? Circ. Res. 2008, 102, 3–5. [Google Scholar] [CrossRef]
- Depre, C.; Kim, S.-J.; John, A.S.; Huang, Y.; Rimoldi, O.E.; Pepper, J.R.; Dreyfus, G.D.; Gaussin, V.; Pennell, D.J.; Vatner, D.E.; et al. Program of Cell Survival Underlying Human and Experimental Hibernating Myocardium. Circ. Res. 2004, 95, 433–440. [Google Scholar] [CrossRef]
- Page, B.J.; Banas, M.D.; Suzuki, G.; Weil, B.R.; Young, R.F.; Fallavollita, J.A.; Palka, B.A.; Canty, J.M. Revascularization of Chronic Hibernating Myocardium Stimulates Myocyte Proliferation and Partially Reverses Chronic Adaptations to Ischemia. J. Am. Coll. Cardiol. 2015, 65, 684–697. [Google Scholar] [CrossRef] [PubMed]
- Fedele, F.A.; Gewirtz, H.; Capone, R.J.; Sharaf, B.; Most, A.S. Metabolic response to prolonged reduction of myocardial blood flow distal to a severe coronary artery stenosis. Circulation 1988, 78, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Page, B.; Young, R.; Iyer, V.; Suzuki, G.; Lis, M.; Korotchkina, L.; Patel, M.S.; Blumenthal, K.M.; Fallavollita, J.A.; Canty, J.J.M. Persistent Regional Downregulation in Mitochondrial Enzymes and Upregulation of Stress Proteins in Swine with Chronic Hibernating Myocardium. Circ. Res. 2008, 102, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Qu, J.; Young, R.; Page, B.J.; Shen, X.; Tata, N.; Li, J.; Duan, X.; Fallavollita, J.A.; Canty, J.M. Reproducible Ion-Current-Based Approach for 24-Plex Comparison of the Tissue Proteomes of Hibernating versus Normal Myocardium in Swine Models. J. Proteome Res. 2014, 13, 2571–2584. [Google Scholar] [CrossRef]
- Kloner, R.A. Stunned and Hibernating Myocardium: Where Are We Nearly 4 Decades Later? J. Am. Heart Assoc. 2020, 9, e015502. [Google Scholar] [CrossRef]
- Frangogiannis, N.G.; Shimoni, S.; Chang, S.M.; Ren, G.; Shan, K.; Aggeli, C.; Reardon, M.J.; Letsou, G.V.; Espada, R.; Ramchandani, M.; et al. Evidence for an Active Inflammatory Process in the Hibernating Human Myocardium. Am. J. Pathol. 2002, 160, 1425–1433. [Google Scholar] [CrossRef]
- Nian, M.; Lee, P.; Khaper, N.; Liu, P. Inflammatory Cytokines and Postmyocardial Infarction Remodeling. Circ. Res. 2004, 94, 1543–1553. [Google Scholar] [CrossRef]
- Akhmedov, A.T.; Rybin, V.; Marín-García, J. Mitochondrial oxidative metabolism and uncoupling proteins in the failing heart. Heart Fail. Rev. 2015, 20, 227–249. [Google Scholar] [CrossRef]
- Stone, L.L.H.; Swingen, C.; Holley, C.; Wright, C.; Chappuis, E.; Ward, H.B.; McFalls, E.O.; Kelly, R.F. Magnetic resonance imaging assessment of cardiac function in a swine model of hibernating myocardium 3 months following bypass surgery. J. Thorac. Cardiovasc. Surg. 2017, 153, 582–590. [Google Scholar] [CrossRef] [PubMed]
- Kelly, R.F.; Cabrera, J.A.; Ziemba, E.A.; Crampton, M.; Anderson, L.B.; McFalls, E.O.; Ward, H.B. Continued depression of maximal oxygen consumption and mitochondrial proteomic expression despite successful coronary artery bypass grafting in a swine model of hibernation. J. Thorac. Cardiovasc. Surg. 2011, 141, 261–268. [Google Scholar] [CrossRef][Green Version]
- Stanley, W.C.; Chandler, M.P. Energy Metabolism in the Normal and Failing Heart: Potential for Therapeutic Interventions. Heart Fail. Rev. 2002, 7, 115–130. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Qin, Y.; Liu, B.; Gao, M.; Li, A.; Li, X.; Gong, G. PGC-1α-Mediated Mitochondrial Quality Control: Molecular Mechanisms and Implications for Heart Failure. Front. Cell Dev. Biol. 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Huss, J.M.; Kelly, D.P. Mitochondrial energy metabolism in heart failure: A question of balance. J. Clin. Investig. 2005, 115, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Gottlieb, R.A.; Piplani, H.; Sin, J.; Sawaged, S.; Hamid, S.M.; Taylor, D.J.; Germano, J.D.F. At the heart of mitochondrial quality control: Many roads to the top. Cell. Mol. Life Sci. 2021, 78, 3791–3801. [Google Scholar] [CrossRef]
- Finck, B.N.; Kelly, D.P. PGC-1 coactivators: Inducible regulators of energy metabolism in health and disease. J. Clin. Investig. 2006, 116, 615–622. [Google Scholar] [CrossRef]
- Li, Y.; Jiao, Y.; Liu, Y.; Fu, J.; Sun, L.; Su, J. PGC-1α protects from myocardial ischaemia-reperfusion injury by regulating mitonuclear communication. J. Cell. Mol. Med. 2022, 26, 593–600. [Google Scholar] [CrossRef]
- Duncan, J.G.; Finck, B.N. The PPARalpha-PGC-1alpha Axis Controls Cardiac Energy Metabolism in Healthy and Diseased Myocardium. PPAR Res. 2008, 2008, 253817. [Google Scholar] [CrossRef]
- Lehman, J.J.; Barger, P.M.; Kovacs, A.; Saffitz, J.E.; Medeiros, D.M.; Kelly, D.P. Peroxisome proliferator-activated receptor gamma coactivator-1 promotes cardiac mitochondrial biogenesis. J. Clin. Investig. 2000, 106, 847–856. [Google Scholar] [CrossRef]
- Schreiber, S.N.; Knutti, D.; Brogli, K.; Uhlmann, T.; Kralli, A. The transcriptional coactivator PGC-1 regulates the expression and activity of the orphan nuclear receptor estrogen-related receptor alpha (ERRalpha). J. Biol. Chem. 2003, 278, 9013–9018. [Google Scholar] [CrossRef]
- Wu, Z.; Puigserver, P.; Andersson, U.; Zhang, C.; Adelmant, G.; Mootha, V.; Troy, A.; Cinti, S.; Lowell, B.; Scarpulla, R.C.; et al. Mechanisms Controlling Mitochondrial Biogenesis and Respiration through the Thermogenic Coactivator PGC-1. Cell 1999, 98, 115–124. [Google Scholar] [CrossRef]
- Huss, J.M.; Kopp, R.P.; Kelly, D.P. Peroxisome proliferator-activated receptor coactivator-1alpha (PGC-1alpha) coactivates the cardiac-enriched nuclear receptors estrogen-related receptor-alpha and -gamma. Identification of novel leucine-rich interaction motif within PGC-1alpha. J. Biol. Chem. 2002, 277, 40265–40274. [Google Scholar] [CrossRef] [PubMed]
- Wenz, T. Regulation of mitochondrial biogenesis and PGC-1α under cellular stress. Mitochondrion 2013, 13, 134–142. [Google Scholar] [CrossRef]
- Chen, Q.M. Nrf2 for cardiac protection: Pharmacological options against oxidative stress. Trends Pharmacol. Sci. 2021, 42, 729–744. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Hu, Z.; Tang, X.; Hu, K. Overexpression of PGC-1α reduces inflammation and protects against focal cerebral ischaemia/reperfusion injury. Arch. Med. Sci. 2020, 16. [Google Scholar] [CrossRef]
- Irrcher, I.; Adhihetty, P.J.; Sheehan, T.; Joseph, A.M.; Hood, D.A. PPARgamma coactivator-1alpha expression during thyroid hormone- and contractile activity-induced mitochondrial adaptations. Am. J. Physiol. Cell Physiol. 2003, 284, C1669–C1677. [Google Scholar] [CrossRef] [PubMed]
- Riehle, C.; Abel, E.D. PGC-1 Proteins and Heart Failure. Trends Cardiovasc. Med. 2012, 22, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Handschin, C.; Spiegelman, B.M. The role of exercise and PGC1α in inflammation and chronic disease. Nature 2008, 454, 463–469. [Google Scholar] [CrossRef]
- Adhihetty, P.J.; O’Leary, M.F.N.; Chabi, B.; Wicks, K.L.; Hood, D.A. Effect of denervation on mitochondrially mediated apoptosis in skeletal muscle. J. Appl. Physiol. 2007, 102, 1143–1151. [Google Scholar] [CrossRef]
- Hu, X.; Xu, X.; Huang, Y.; Fassett, J.; Flagg, T.P.; Zhang, Y.; Nichols, C.G.; Bache, R.J.; Chen, Y. Disruption of Sarcolemmal ATP-Sensitive Potassium Channel Activity Impairs the Cardiac Response to Systolic Overload. Circ. Res. 2008, 103, 1009–1017. [Google Scholar] [CrossRef]
- Bhat, S.; Chin, A.; Shirakabe, A.; Ikeda, Y.; Ikeda, S.; Zhai, P.; Hsu, C.; Sayed, D.; Abdellatif, M.; Byun, J.; et al. Recruitment of RNA Polymerase II to Metabolic Gene Promoters Is Inhibited in the Failing Heart Possibly Through PGC-1alpha (Peroxisome Proliferator-Activated Receptor-gamma Coactivator-1alpha) Dysregulation. Circ. Heart Fail. 2019, 12, e005529. [Google Scholar] [CrossRef]
- Dorn, G.W., 2nd; Vega, R.B.; Kelly, D.P. Mitochondrial biogenesis and dynamics in the developing and diseased heart. Genes Dev. 2015, 29, 1981–1991. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Shen, W.; Yao, K.; Wang, H.; Liu, B.; Li, T.; Song, L.; Diao, D.; Mao, G.; Huang, P.; et al. Fine-Tuning of PGC1α Expression Regulates Cardiac Function and Longevity. Circ. Res. 2019, 125, 707–719. [Google Scholar] [CrossRef]
- Russell, L.K.; Mansfield, C.M.; Lehman, J.J.; Kovacs, A.; Courtois, M.; Saffitz, J.E.; Medeiros, D.M.; Valencik, M.L.; McDonald, J.A.; Kelly, D.P. Cardiac-specific induction of the transcriptional coactivator peroxisome proliferator-activated receptor gamma coactivator-1alpha promotes mitochondrial biogenesis and reversible cardiomyopathy in a developmental stage-dependent manner. Circ. Res. 2004, 94, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Summermatter, S.; Shui, G.; Maag, D.; Santos, G.; Wenk, M.R.; Handschin, C. PGC-1α Improves Glucose Homeostasis in Skeletal Muscle in an Activity-Dependent Manner. Diabetes 2013, 62, 85–95. [Google Scholar] [CrossRef]
- Berezhnov, A.V.; Fedotova, E.I.; Nenov, M.N.; Kasymov, V.A.; Pimenov, O.Y.; Dynnik, V.V. Dissecting Cellular Mechanisms of Long-Chain Acylcarnitines-Driven Cardiotoxicity: Disturbance of Calcium Homeostasis, Activation of Ca2+-Dependent Phospholipases, and Mitochondrial Energetics Collapse. Int. J. Mol. Sci. 2020, 21, 7461. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.H.; Hoppel, C.L.; Lok, K.H.; Zhao, L.; Wong, S.W.; Minkler, P.E.; Hwang, D.H.; Newman, J.W.; Garvey, W.T. Plasma acylcarnitine profiles suggest incomplete long-chain fatty acid beta-oxidation and altered tricarboxylic acid cycle activity in type 2 diabetic African-American women. J. Nutr. 2009, 139, 1073–1081. [Google Scholar] [CrossRef]
- Supruniuk, E.; Mikłosz, A.; Chabowski, A. The Implication of PGC-1α on Fatty Acid Transport across Plasma and Mitochondrial Membranes in the Insulin Sensitive Tissues. Front. Physiol. 2017, 8, 923. [Google Scholar] [CrossRef]
- Adhihetty, P.J.; Uguccioni, G.; Leick, L.; Hidalgo, J.; Pilegaard, H.; Hood, D.A. The role of PGC-1α on mitochondrial function and apoptotic susceptibility in muscle. Am. J. Physiol.-Cell Physiol. 2009, 297, C217–C225. [Google Scholar] [CrossRef]
- Leone, T.C.; Lehman, J.J.; Finck, B.N.; Schaeffer, P.J.; Wende, A.R.; Boudina, S.; Courtois, M.; Wozniak, D.F.; Sambandam, N.; Bernal-Mizrachi, C.; et al. PGC-1alpha deficiency causes multi-system energy metabolic derangements: Muscle dysfunction, abnormal weight control and hepatic steatosis. PLoS Biol. 2005, 3, e101. [Google Scholar] [CrossRef]
- Arany, Z.; He, H.; Lin, J.; Hoyer, K.; Handschin, C.; Toka, O.; Ahmad, F.; Matsui, T.; Chin, S.; Wu, P.-H.; et al. Transcriptional coactivator PGC-1α controls the energy state and contractile function of cardiac muscle. Cell Metab. 2005, 1, 259–271. [Google Scholar] [CrossRef]
- Arany, Z.; Novikov, M.; Chin, S.; Ma, Y.; Rosenzweig, A.; Spiegelman, B.M. Transverse aortic constriction leads to accelerated heart failure in mice lacking PPAR-gamma coactivator 1alpha. Proc. Natl. Acad. Sci. USA 2006, 103, 10086–10091. [Google Scholar] [CrossRef] [PubMed]
- Aquilano, K.; Baldelli, S.; Pagliei, B.; Cannata, S.M.; Rotilio, G.; Ciriolo, M.R. p53 Orchestrates the PGC-1α-Mediated Antioxidant Response Upon Mild Redox and Metabolic Imbalance. Antioxid. Redox Signal. 2013, 18, 386–399. [Google Scholar] [CrossRef] [PubMed]
- St-Pierre, J.; Drori, S.; Uldry, M.; Silvaggi, J.M.; Rhee, J.; Jäger, S.; Handschin, C.; Zheng, K.; Lin, J.; Yang, W.; et al. Suppression of Reactive Oxygen Species and Neurodegeneration by the PGC-1 Transcriptional Coactivators. Cell 2006, 127, 397–408. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Xu, X.; Hu, X.; Fassett, J.; Zhu, G.; Tao, Y.; Li, J.; Huang, Y.; Zhang, P.; Zhao, B.; et al. PGC-1α Regulates Expression of Myocardial Mitochondrial Antioxidants and Myocardial Oxidative Stress After Chronic Systolic Overload. Antioxid. Redox Signal. 2010, 13, 1011–1022. [Google Scholar] [CrossRef]
- Marmolino, D.; Manto, M.; Acquaviva, F.; Vergara, P.; Ravella, A.; Monticelli, A.; Pandolfo, M. PGC-1alpha down-regulation affects the antioxidant response in Friedreich’s ataxia. PLoS ONE 2010, 5, e10025. [Google Scholar] [CrossRef]
- Baldelli, S.; Aquilano, K.; Ciriolo, M.R. PGC-1α buffers ROS-mediated removal of mitochondria during myogenesis. Cell Death Dis. 2014, 5, e1515. [Google Scholar] [CrossRef]
- Holley, C.T.; Long, E.K.; Butterick, T.A.; Duffy, C.M.; Lindsey, M.E.; Stone, L.H.; McFalls, E.O.; Kelly, R.F. Mitochondrial fusion proteins in revascularized hibernating hearts. J. Surg. Res. 2015, 195, 29–36. [Google Scholar] [CrossRef]
- Cartoni, R.; Léger, B.; Hock, M.B.; Praz, M.; Crettenand, A.; Pich, S.; Ziltener, J.-C.; Luthi, F.; Dériaz, O.; Zorzano, A.; et al. Mitofusins 1/2 and ERRalpha expression are increased in human skeletal muscle after physical exercise. J. Physiol. 2005, 567 Pt 1, 349–358. [Google Scholar] [CrossRef]
- Valle, I.; Álvarez-Barrientos, A.; Arza, E.; Lamas, S.; Monsalve, M. PGC-1α regulates the mitochondrial antioxidant defense system in vascular endothelial cells. Cardiovasc. Res. 2005, 66, 562–573. [Google Scholar] [CrossRef]
- Ronnebaum, S.M.; Patterson, C. The FoxO Family in Cardiac Function and Dysfunction. Annu. Rev. Physiol. 2010, 72, 81–94. [Google Scholar] [CrossRef]
- Olmos, Y.; Valle, I.; Borniquel, S.; Tierrez, A.; Soria, E.; Lamas, S.; Monsalve, M. Mutual Dependence of Foxo3a and PGC-1α in the Induction of Oxidative Stress Genes. J. Biol. Chem. 2009, 284, 14476–14484. [Google Scholar] [CrossRef] [PubMed]
- Yamada, S.; Kane, G.C.; Behfar, A.; Liu, X.K.; Dyer, R.B.; Faustino, R.S.; Miki, T.; Seino, S.; Terzic, A. Protection conferred by myocardial ATP-sensitive K+ channels in pressure overload-induced congestive heart failure revealed in KCNJ11 Kir6.2-null mutant. J. Physiol. 2006, 577 Pt 3, 1053–1065. [Google Scholar] [CrossRef] [PubMed]
- Rabinovich-Nikitin, I.; Blant, A.; Dhingra, R.; Kirshenbaum, L.A.; Czubryt, M.P. NF-kappaB p65 Attenuates Cardiomyocyte PGC-1alpha Expression in Hypoxia. Cells 2022, 11, 2193. [Google Scholar] [CrossRef]
- Fiordelisi, A.; Iaccarino, G.; Morisco, C.; Coscioni, E.; Sorriento, D. NFkappaB is a Key Player in the Crosstalk between Inflammation and Cardiovascular Diseases. Int. J. Mol. Sci. 2019, 20, 1599. [Google Scholar] [CrossRef]
- Zhao, T.C.; Taher, M.M.; Valerie, K.C.; Kukreja, R.C. p38 Triggers late preconditioning elicited by anisomycin in heart: Involvement of NF-kappaB and iNOS. Circ. Res. 2001, 89, 915–922. [Google Scholar] [CrossRef] [PubMed]
- McFalls, E.O.; Hou, M.; Bache, R.J.; Best, A.; Marx, D.; Sikora, J.; Ward, H.B. Activation of p38 MAPK and increased glucose transport in chronic hibernating swine myocardium. Am. J. Physiol. Circ. Physiol. 2004, 287, H1328–H1334. [Google Scholar] [CrossRef] [PubMed]
- Meldrum, D.R.; Dinarello, C.A.; Cleveland, J.C.; Cain, B.S.; Shames, B.D.; Meng, X.; Harken, A.H. Hydrogen peroxide induces tumor necrosis factor alpha-mediated cardiac injury by a P38 mitogen-activated protein kinase-dependent mechanism. Surgery 1998, 124, 291–296. [Google Scholar] [CrossRef]
- Sawyer, D.B.; Loscalzo, J. Myocardial hibernation: Restorative or preterminal sleep? Circulation 2002, 105, 1517–1519. [Google Scholar] [CrossRef]
- Hanna, A.; Frangogiannis, N.G. Inflammatory Cytokines and Chemokines as Therapeutic Targets in Heart Failure. Cardiovasc. Drugs Ther. 2020, 34, 849–863. [Google Scholar] [CrossRef]
- Eisele, P.S.; Salatino, S.; Sobek, J.; Hottiger, M.O.; Handschin, C. The peroxisome proliferator-activated receptor gamma coactivator 1alpha/beta (PGC-1) coactivators repress the transcriptional activity of NF-kappaB in skeletal muscle cells. J. Biol. Chem. 2013, 288, 2246–2260. [Google Scholar] [CrossRef]
- Palomer, X.; Álvarez-Guardia, D.; Rodríguez-Calvo, R.; Coll, T.; Laguna, J.C.; Davidson, M.M.; Chan, T.O.; Feldman, A.M.; Vázquez-Carrera, M. TNF-alpha reduces PGC-1alpha expression through NF-kappaB and p38 MAPK leading to increased glucose oxidation in a human cardiac cell model. Cardiovasc. Res. 2009, 81, 703–712. [Google Scholar] [CrossRef] [PubMed]
- Handschin, C.; Furrer, R. Körperliche Aktivität und PGC-1alpha bei Entzündung und chronischen Krankheiten. Dtsch. Z. Für Sportmed. 2015, 2015, 317–320. [Google Scholar] [CrossRef][Green Version]
- Álvarez-Guardia, D.; Palomer, X.; Coll, T.; Davidson, M.M.; Chan, T.O.; Feldman, A.M.; Laguna, J.C.; Vázquez-Carrera, M. The p65 subunit of NF-κB binds to PGC-1α, linking inflammation and metabolic disturbances in cardiac cells. Cardiovasc. Res. 2010, 87, 449–458. [Google Scholar] [CrossRef]
- Butterick, T.A.; Stone, L.H.; Duffy, C.; Holley, C.; Cabrera, J.A.; Crampton, M.; Ward, H.B.; Kelly, R.F.; McFalls, E.O. Pioglitazone increases PGC1-α signaling within chronically ischemic myocardium. Basic Res. Cardiol. 2016, 111, 37. [Google Scholar] [CrossRef] [PubMed]
- Stone, L.H.; Chappuis, E.; Wright, C.; Kelly, R.F.; McFalls, E.O. CoQ10 enhances PGC1α and increases expression of mitochondrial antioxidant proteins in chronically ischemic swine myocardium. Nutr. Metab. 2019, 16, 92. [Google Scholar] [CrossRef]
- Khan, A.; Johnson, D.K.; Carlson, S.; Hocum-Stone, L.; Kelly, R.F.; Gravely, A.A.; Mbai, M.; Green, D.L.; Santilli, S.; Garcia, S.; et al. NT-Pro BNP Predicts Myocardial Injury Post-vascular Surgery and is Reduced with CoQ10: A Randomized Double-Blind Trial. Ann. Vasc. Surg. 2020, 64, 292–302. [Google Scholar] [CrossRef]
- Chen, Q.; Qi, S.; Hocum-Stone, L.; Lesnefsky, E.; Kelly, R.; McFalls, E. Preventing Myocardial Injury Following Non-Cardiac Surgery: A Potential Role for Preoperative Antioxidant Therapy with Ubiquinone. Antioxidants 2021, 10, 276. [Google Scholar] [CrossRef]
- Pilegaard, H.; Saltin, B.; Neufer, P.D. Exercise induces transient transcriptional activation of the PGC-1α gene in human skeletal muscle. J. Physiol. 2003, 546 Pt 3, 851–858. [Google Scholar] [CrossRef]
- Stone, L.L.H.; Swingen, C.; Wright, C.; Qi, S.S.; Rassette, M.; McFalls, E.O.; Kelly, R.F. Recovery of hibernating myocardium using stem cell patch with coronary bypass surgery. J. Thorac. Cardiovasc. Surg. 2021, 162, e3–e16. [Google Scholar] [CrossRef]
- Stone, L.L.H.; Chappuis, E.; Marquez, M.; McFalls, E.O.; Kelly, R.F.; Butterick, T. Mitochondrial Respiratory Capacity is Restored in Hibernating Cardiomyocytes Following Co-Culture with Mesenchymal Stem Cells. Cell Med. 2019, 11. [Google Scholar] [CrossRef]
- Hughey, C.C.; James, F.D.; Ma, L.; Bracy, D.P.; Wang, Z.; Wasserman, D.H.; Rottman, J.N.; Shearer, J. Diminishing impairments in glucose uptake, mitochondrial content, and ADP-stimulated oxygen flux by mesenchymal stem cell therapy in the infarcted heart. Am. J. Physiol.-Cell Physiol. 2014, 306, C19–C27. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, N.; Gill, J.F.; Brink, M.; Handschin, C. Moderate Modulation of Cardiac PGC-1α Expression Partially Affects Age-Associated Transcriptional Remodeling of the Heart. Front. Physiol. 2018, 9, 242. [Google Scholar] [CrossRef] [PubMed]
| Type | Intervention | Model | Sample | Biopsied Tissue | Reference |
|---|---|---|---|---|---|
| Lifestyle-based | Exercise | Human | n = 7 | Skeletal Muscle | [78] |
| Dietary | CoQ10 | Swine | n = 12 | Myocardium | [75] |
| Pharmacological | Pioglitazone | Swine | n = 18 | Myocardium | [74] |
| Cell-therapeutic | Mesenchymal Stem Cells | Swine | n = 12 | Myocardium | [79] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aggarwal, R.; Potel, K.N.; McFalls, E.O.; Butterick, T.A.; Kelly, R.F. Novel Therapeutic Approaches Enhance PGC1-alpha to Reduce Oxidant Stress-Inflammatory Signaling and Improve Functional Recovery in Hibernating Myocardium. Antioxidants 2022, 11, 2155. https://doi.org/10.3390/antiox11112155
Aggarwal R, Potel KN, McFalls EO, Butterick TA, Kelly RF. Novel Therapeutic Approaches Enhance PGC1-alpha to Reduce Oxidant Stress-Inflammatory Signaling and Improve Functional Recovery in Hibernating Myocardium. Antioxidants. 2022; 11(11):2155. https://doi.org/10.3390/antiox11112155
Chicago/Turabian StyleAggarwal, Rishav, Koray N. Potel, Edward O. McFalls, Tammy A. Butterick, and Rosemary F. Kelly. 2022. "Novel Therapeutic Approaches Enhance PGC1-alpha to Reduce Oxidant Stress-Inflammatory Signaling and Improve Functional Recovery in Hibernating Myocardium" Antioxidants 11, no. 11: 2155. https://doi.org/10.3390/antiox11112155
APA StyleAggarwal, R., Potel, K. N., McFalls, E. O., Butterick, T. A., & Kelly, R. F. (2022). Novel Therapeutic Approaches Enhance PGC1-alpha to Reduce Oxidant Stress-Inflammatory Signaling and Improve Functional Recovery in Hibernating Myocardium. Antioxidants, 11(11), 2155. https://doi.org/10.3390/antiox11112155








