New Unnatural Gallotannins: A Way toward Green Antioxidants, Antimicrobials and Antibiofilm Agents
Abstract
:1. Introduction
2. Materials and Methods
2.1. Physico-Chemical Methods
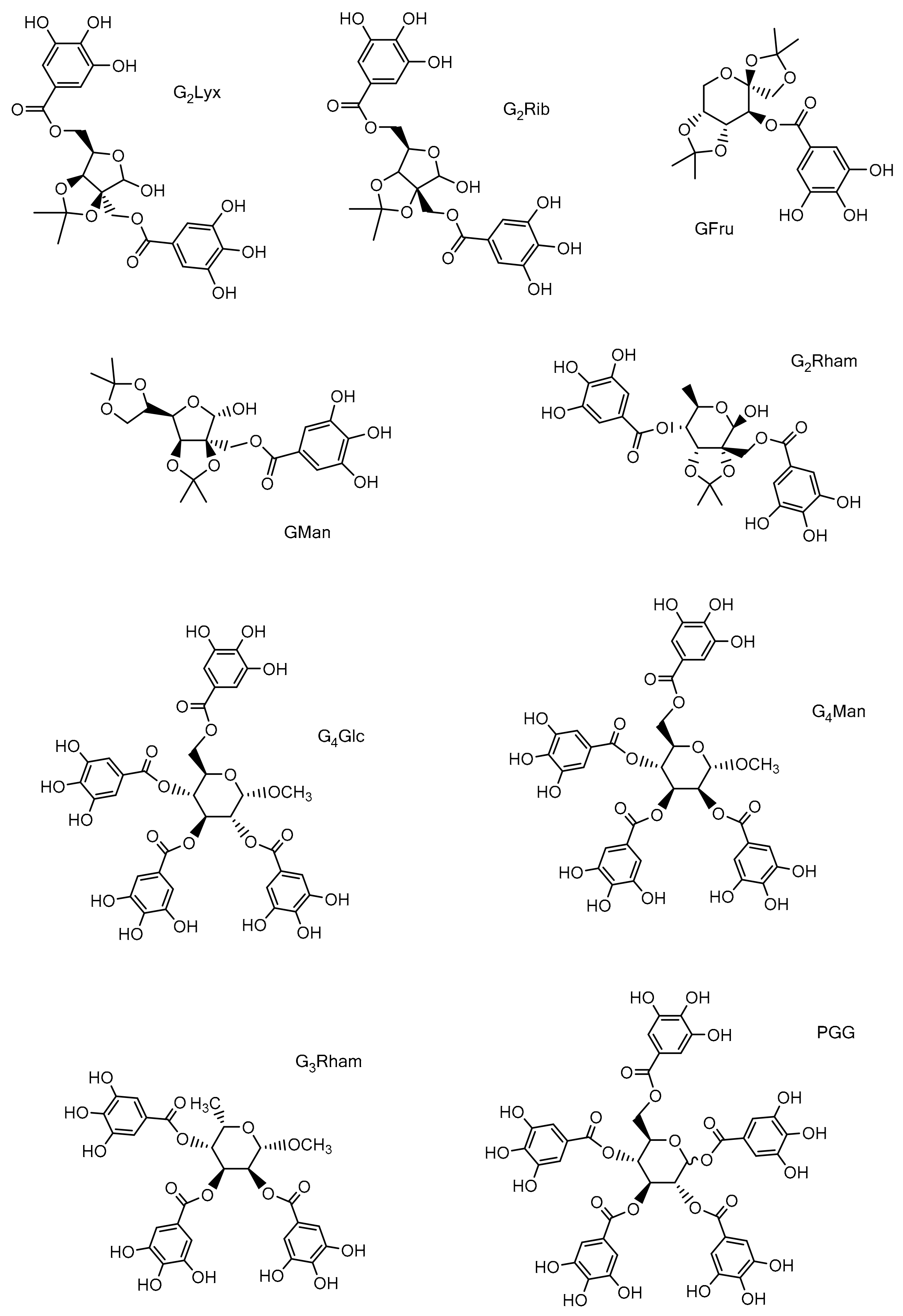
2.2. Studied Compounds
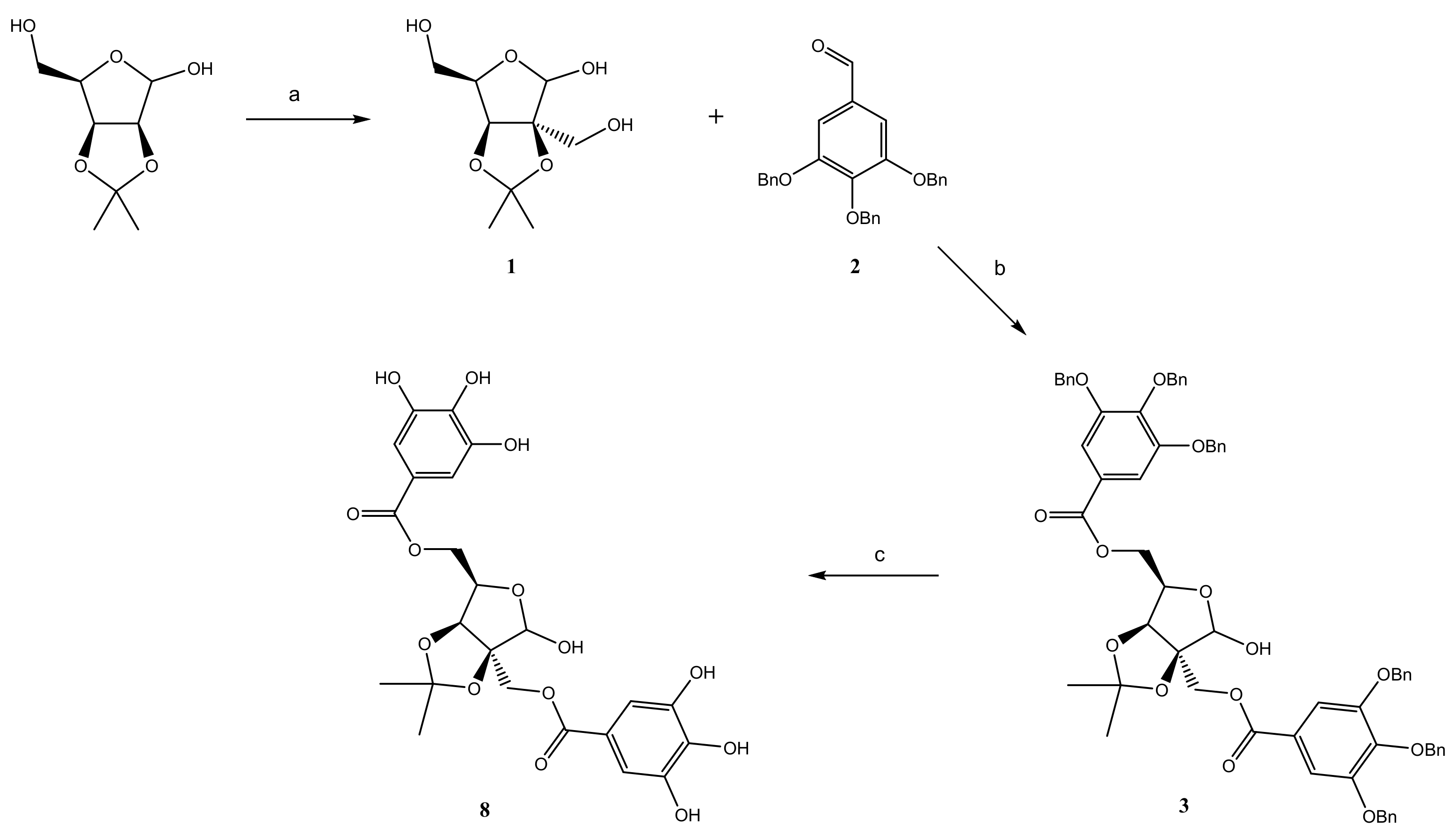
2.3. Synthesis of Galloyl Derivatives of 2-C-(Hydroxymethyl)-branched Saccharides
2.3.1. Preparation of 2-C-(Hydroxymethyl)-2,3-O-isopropylidene-α-d-lyxofuranose (1)
2.3.2. Preparation of 2-C-(Hydroxymethyl)-2,3-O-isopropylidene-α-d-lyxofuranose (2)
2.3.3. Preparation of Di-O-(3,4,5-tri-O-benzylgalloyl)-2-C-(hydroxymethyl)-2,3-O-isopro- pylidene Aldoses
2.3.4. Debenzylation Procedure
2.4. Determination of Antioxidant Activity
2.4.1. 1,1-Diphenyl-2-picrylhydrazyl Radical Scavenging Activity (DPPH Assay)
2.4.2. Iron(III)-Reducing Power Assay (FRAP Assay)
2.5. Biological Studies
2.5.1. Test Microorganisms
2.5.2. Determination of Antimicrobial Activity
2.5.3. Biofilm Inhibition
2.5.4. Biofilm Disruption
2.5.5. Interaction with Quorum Sensing of C. violaceum
2.5.6. Interaction with sortase A
2.6. Statistical Analysis
3. Results and Discussion
3.1. Chemistry
3.2. Antioxidant Activity
3.3. Antibacterial and Antimycobacterial Activity
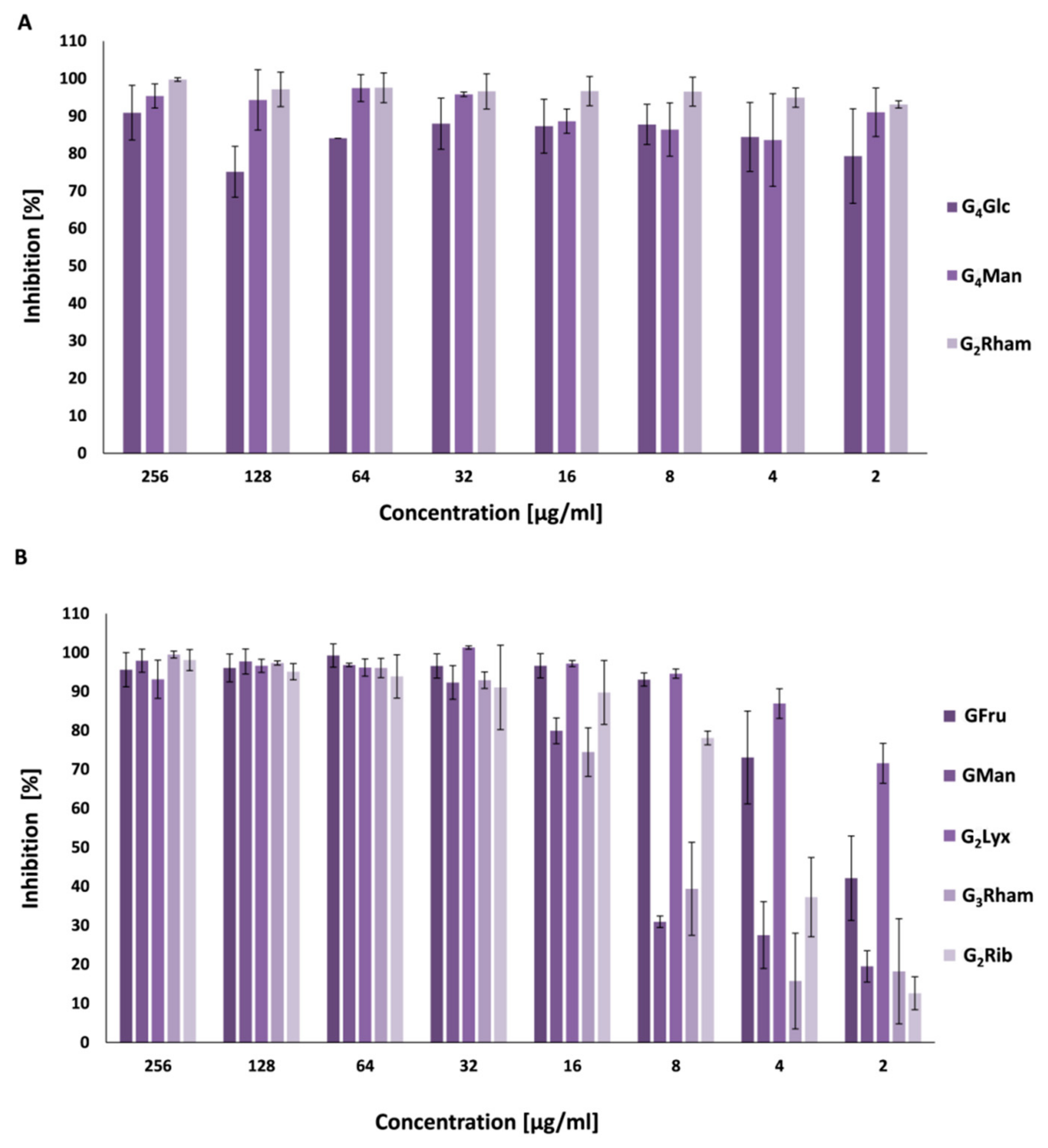
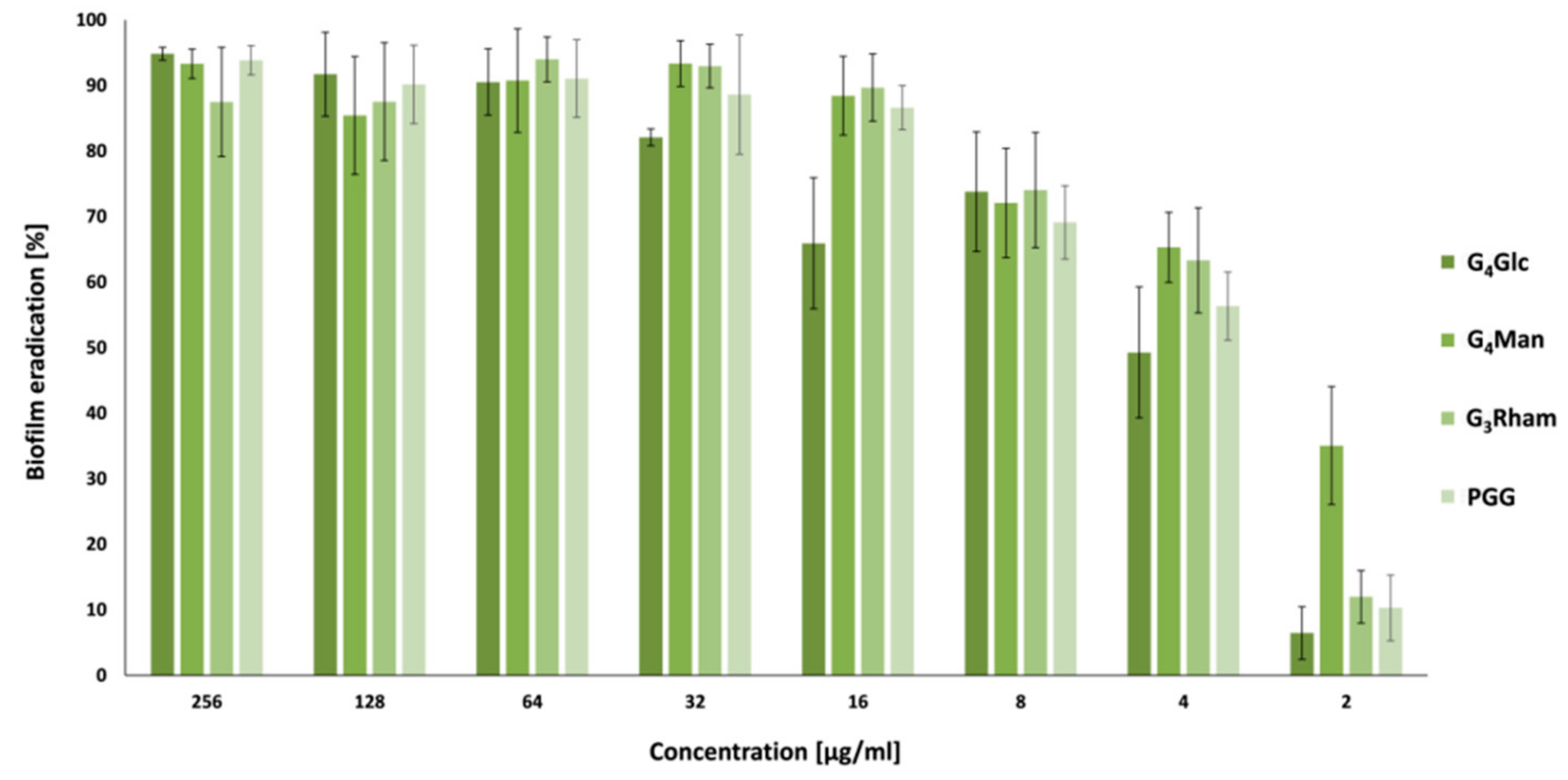
3.4. Antibiofilm Activity
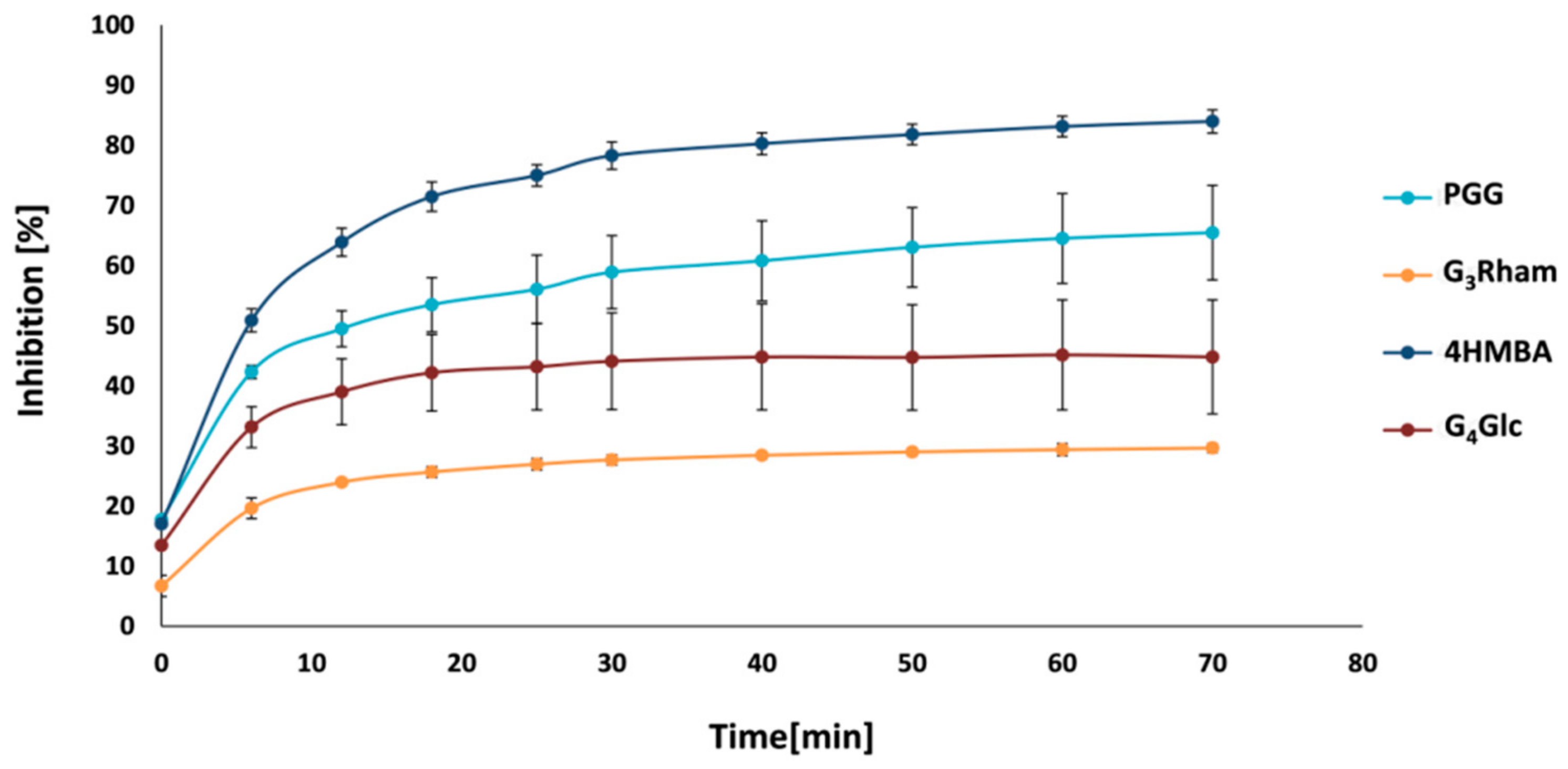
3.5. Quorum Sensing Inhibition
3.6. Inhibition of Sortase A
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Khanbabaee, K.; van Ree, T. Tannins: Classification and definition. Nat. Prod. Rep. 2001, 18, 641–649. [Google Scholar] [CrossRef]
- Quideau, S.; Deffieux, D.; Douat-Casassus, C.; Pouysegu, L. Plant polyphenols: Chemical properties, biological activities, and synthesis. Angew. Chem. Int. Ed. 2011, 50, 586–621. [Google Scholar] [CrossRef] [PubMed]
- Barbehenn, R.V.; Constabel, C.P. Tannins in plant–herbivore interactions. Phytochemistry 2011, 72, 1551–1565. [Google Scholar] [CrossRef] [PubMed]
- Chung, K.T.; Wong, T.Y.; Wei, C.I.; Huang, Y.W.; Lin, Y. Tannins and human health: A review. Crit. Rev. Food Sci. Nutr. 1998, 38, 421–464. [Google Scholar] [CrossRef]
- Kiss, A.K.; Piwowarski, J.P. Ellagitannins, gallotannins and their metabolites—The contribution to the anti-inflammatory effect of food products and medicinal plants. Curr. Med. Chem. 2018, 25, 4946–4967. [Google Scholar] [CrossRef]
- Lee, S.J.; Lee, H.K.; Jung, M.K.; Mar, W. In vitro antiviral activity of 1,2,3,4,6-penta-O-galloyl-β-d-glucose against hepatitis B virus. Biol. Pharm. Bull. 2006, 29, 2131–2134. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Li, L.; Kim, S.H.; Hagerman, A.E.; Lü, J. Anti-cancer, anti-diabetic and other pharmacologic and biological activities of penta galloyl-glucose. Pharmaceut. Res. 2009, 26, 2066–2080. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yin, S.; Dong, Y.; Li, J.; Lü, J.; Hu, H. Penta-1,2,3,4,6-O-galloyl-β-d-glucose induces senescence-like terminal S-phase arrest in human hepatoma and breast cancer cells. Mol. Carcinog. 2011, 50, 592–600. [Google Scholar] [CrossRef] [PubMed]
- Jourdes, M.; Pouysegu, L.; Deffieux, D.; Teissedre, P.-L.; Quideau, S. Hydrolyzable tannins: Gallotannins and ellagitannins. In Natural Products: Phytochemistry, Botany and Metabolism of Alkaloids, Phenolics and Terpenes; Ramawat, K.G., Merillon, J.M., Eds.; Springer: Berlin, Germany, 2013; pp. 1975–2010. [Google Scholar]
- Wu, Y.P.; Liu, X.Y.; Bai, J.R.; Xie, H.C.; Ye, S.L.; Zhong, K.; Huang, Y.N.; Gao, H. Inhibitory effect of a natural phenolic compound, 3-p-trans-coumaroyl-2-hydroxyquinic acid against the attachment phase of biofilm formation of Staphylococcus aureus through targeting sortase A. RSC Adv. 2019, 9, 32453–32461. [Google Scholar] [CrossRef] [Green Version]
- Blando, F.; Russo, R.; Negro, C.; De Bellis, L.; Frassinetti, S. Antimicrobial and antibiofilm activity against Staphylococcus aureus of Opuntiaficus indica (L.) Mill. cladode polyphenolic extracts. Antioxidants 2019, 8, 117. [Google Scholar] [CrossRef] [Green Version]
- Gotz, F. Staphylococcus and biofilms. Molec. Microbiol. 2002, 43, 1367–1378. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; He, L.; Asiamah, T.K.; Otto, M. Colonization of medical devices by staphylococci. Environm. Microbiol. 2018, 20, 3141–3153. [Google Scholar] [CrossRef] [Green Version]
- Takó, M.; Kerekes, E.B.; Zambrano, C.; Kotogán, A.; Papp, T.; Krisch, J.; Vágvölgyi, C. Plant phenolics and phenolic-enriched extracts as antimicrobial agents against food contaminating microorganisms. Antioxidants 2020, 9, 165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amakura, Y.; Yoshimura, M.; Sugimoto, N.; Yamazaki, T.; Yoshida, T. Marker constituents of the natural antioxidant eucalyptus leaf extract for the evaluation of food additives. Biosci. Biotechnol. Biochem. 2009, 73, 1060–1065. [Google Scholar] [CrossRef] [Green Version]
- Abou-Zaid, M.M.; Nozzolillo, C. 1-O-galloyl-α-l-rhamnose from Acer rubrum. Phytochemistry 1999, 52, 1629–1631. [Google Scholar] [CrossRef]
- Xie, Y.; Zhao, Y. Synthesis of 7-O-galloyl-d-sedoheptulose. Carbohydr. Res. 2007, 342, 1510–1513. [Google Scholar] [CrossRef]
- Ren, Y.; Himmeldirk, K.; Chen, X. Synthesis and structure-activity relationship study of antidiabetic penta-O-galloyl-d-glucopyranose and its analogues. J. Med. Chem. 2006, 49, 2829–2837. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Himmeldirk, K.; Qian, Y.; Ren, Y.; Malki, A.; Chen, X. Biological and biomedical functions of penta-O-galloyl-d-glucose and its derivatives. J. Nat. Med. 2014, 68, 465–472. [Google Scholar] [CrossRef] [PubMed]
- González-Sarrías, A.; Yuan, T.; Seeram, N.P. Cytotoxicity and structure activity relationship studies of maplexins A–I, gallotannins from red maple (Acer rubrum). Food Chem. Toxicol. 2012, 50, 1369–1376. [Google Scholar] [CrossRef]
- Torres-León, C.; Ventura-Sobrevilla, J.; Serna-Cock, L.; Ascacio-Valdés, J.A.; Contreras-Esquivel, J.; Aguilar, C.N. Pentagalloylglucose (PGG): A valuable phenolic compound with functional properties. J. Funct. Foods 2017, 37, 176–189. [Google Scholar] [CrossRef]
- Jiamboonsri, P.; Pithayanukul, P.; Bavovada, R.; Chomnawang, M.T. The inhibitory potential of Thai mango seed kernel extract against methicillin-resistant Staphylococcus aureus. Molecules 2011, 16, 6255–6270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, M.H.; Chang, F.R.; Hua, M.Y.; Wu, Y.C.; Liu, S.T. Inhibitory effects of 1,2,3,4,6-penta-O-galloyl-β-d-glucopyranose on biofilm formation by Staphylococcus aureus. Antimicrob. Agents Chemother. 2011, 55, 1021–1027. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.; Wang, B.; Zhang, S.; Yang, S.; Wang, H.; Ren, A.; Yi, E. Isolation of antifungal compound from Paeonia suffruticosa and its antifungal mechanism. Chin. J. Integrat. Med. 2014, 21, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Shafizadeh, F. Branched-chain sugars of natural occurrence. Adv. Carbohydr. Chem. 1956, 48, 263–283. [Google Scholar] [PubMed]
- Grisebach, H.; Schmid, R. Chemistry and biochemistry of branched-chain sugars. Angew. Chem. Internat. Edit. 1972, 1, 159–248. [Google Scholar] [CrossRef]
- Beck, E.; Hopf, H. Branched-chain sugars and sugar alcohols. Carbohydrates 1990, 235–289. [Google Scholar] [CrossRef]
- Masaki, H.; Atsumi, T.; Sakurai, H. Hamamelitannin as a new potent active oxygen scavenger. Phytochemistry 1994, 37, 337–343. [Google Scholar] [CrossRef]
- Masaki, H.; Atsumi, T.; Sakurai, H. Peroxyl radical scavenging activities of hamamelitannin in chemical and biological systems. Free. Rad. Res. 1995, 22, 419–430. [Google Scholar] [CrossRef]
- Lizárraga, D.; Touriño, S.; Reyes-Zurita, F.J.; de Kok, T.M.; van Delft, J.H.; Maas, L.M.; Briedé, J.J.; Centelles, J.J.; Torres, J.L.; Cascante, M. Witch hazel (Hamamelis virginiana) fractions and the importance of gallate moieties-electron transfer capacities in their antitumoral properties. J. Agric. Food Chem. 2008, 56, 11675–11682. [Google Scholar] [CrossRef]
- Erdelmeier, C.; Cinatl, J.; Rabenau, H.; Doerr, H.; Biber, A.; Koch, E. Antiviral and antiphlogistic activities of Hamamelis virginiana Bark. Planta Med. 1996, 62, 241–245. [Google Scholar] [CrossRef]
- Sánchez-Tena, S.; Fernández-Cachón, M.F.; Carreras, A.A.; Mateos-Martín, M.L.; Costoya, N.; Moyer, M.P.; Nuñez, M.J.; Torres, J.L.; Cascante, M. Hamamelitannin from Witch hazel (Hamamelis virginiana) displays specific cytotoxic activity against colon cancer cells. J. Nat. Prod. 2012, 75, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Vermote, A.; Brackman, G.; Risseeuw, M.D.P.; Cappoen, D.; Cos, P.; Coenye, T.; Van Calenbergh, S. Novel potentiators for vancomycin in the treatment of biofilm-related MRSA infections via a mix and match approach. ACS Med. Chem. Lett. 2016, 8, 38–42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brackman, G.; Breyne, K.; De Rycke, R.; Vermote, A.; Van Nieuwerburgh, F.; Meyer, E.; Van Calenbergh, S.; Coenye, T. The quorum sensing inhibitor hamamelitannin increases antibiotic susceptibility of Staphylococcus aureus bioflms by affecting peptidoglycan biosynthesis and eDNA release. Sci. Rep. 2016, 6, 20321. [Google Scholar] [CrossRef]
- Hricovíniová, J.; Ševčovičová, A.; Hricovíniová, Z. Evaluation of the genotoxic, DNA-protective and antioxidant profile of synthetic alkyl gallates and gallotannins using in vitro assays. Toxicol. Vitro 2020, 65, 104789. [Google Scholar] [CrossRef]
- Barbat, J.; Gelas, J.; Horton, D. Reactions of D-lyxose and D-xylose with 2-methoxypropene under kinetic conditions. Carbohydr. Res. 1991, 219, 115–121. [Google Scholar] [CrossRef]
- Clinton, R.O.; Geissman, T.A. Gallaldehyde tribenzyl ether. J. Am. Chem. Soc. 1943, 65, 85–87. [Google Scholar] [CrossRef]
- Ho, P.-T. Branched-chain sugars. Reaction of furanoses with formaldehyde: A stereospecific synthesis of l-dendroketose. Can. J. Chem. 1979, 57, 384–386. [Google Scholar] [CrossRef]
- Hricovíniová, Z. Isomerization as a route to rare ketoses. The beneficial effect of microwave irradiation on Mo(VI)-catalyzed stereospecific rearrangement. Tetrahedron Asymmetry 2008, 19, 204–208. [Google Scholar] [CrossRef]
- Ho, P.-T. Branched-chain sugars. Reaction of furanoses with formaldehyde: A simple synthesis of d- and l-apiose. Can. J. Chem. 1979, 57, 381–383. [Google Scholar] [CrossRef] [Green Version]
- Locatelli, M.; Gindro, R.; Travaglia, F.; Coïsson, J.D.; Rinaldi, M.; Arlorio, M. Study of the DPPH-scavenging activity: Development of a free software for the correct interpretation of data. Food Chem. 2009, 114, 889–897. [Google Scholar] [CrossRef]
- Gupta, D. Methods for determination of antioxidant capacity: A review. Int. J. Pharm. Sci. Res. 2015, 6, 546–566. [Google Scholar] [CrossRef]
- Oravcová, V.; Zurek, L.; Townsend, A.; Clark, A.B.; Ellis, J.C.; Čížek, A.; Literák, I. American crows as carriers of vancomycin-resistant enterococci with vanA gene. Environ. Microbiol. 2014, 16, 939–949. [Google Scholar] [CrossRef]
- CLSI. Clinical and Laboratory Standards Institute Performance Standards for Antimicrobial Susceptibility Testing; The 8th Informational Supplement Document; CLSI: Wayne, PA, USA, 2012; p. M100-S22. [Google Scholar]
- Schwalbe, R.; Steele-Moore, L.; Goodwin, A.C. Antimicrobial Susceptibility Testing Protocols; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar] [CrossRef]
- Pospíšilová, Š.; Malík, I.; Bezoušková, K.; Kauerová, T.; Kollár, P.; Csöllei, J.; Oravec, M.; Čížek, A.; Jampílek, J. Dibasic derivatives of phenylcarbamic acid as prospective antibacterial agents interacting with cytoplasmic membrane. Antibiotics 2020, 9, 64. [Google Scholar] [CrossRef] [Green Version]
- Pospíšilová, Š.; Malík, I.; Čurillová, J.; Michnová, H.; Černá, L.; Padrtová, T.; Hošek, J.; Pecher, D.; Čížek, A.; Jampílek, J. Insight into antimicrobial activity of substituted phenylcarbamoyloxypiperazinyl propanols. Bioorg. Chem. 2020, 102, 104060. [Google Scholar] [CrossRef]
- O’Toole, G.A. Microtiter dish biofilm formation assay. J. Vis. Exp. 2011, 47, 2437. [Google Scholar] [CrossRef]
- Merghni, A.; Marzouki, H.; Hentati, H.; Aouni, M.; Mastouri, M. Antibacterial and antibiofilm activities of Laurus nobilis, L. essential oil against Staphylococcus aureus strains associated with oral infections. Curr. Res. Translat. Med. 2016, 64, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Szabó, M.Á.; Varga, G.Z.; Hohmann, J.; Schelz, Z.; Szegedi, E.; Amaral, L.; Molnár, J. Inhibition of quorum-sensing signals by essential oils. Phytother. Res. 2010, 24, 782–786. [Google Scholar] [CrossRef]
- Fraga-Corral, M.; García-Oliveira, P.; Pereira, A.G.; Lourenço-Lopes, C.; Jimenez-Lopez, C.; Prieto, M.A.; Simal-Gandara, J. Technological application of tannin-based extracts. Molecules 2020, 25, 614. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siquet, C.; Paiva-Martins, F.; Lima, J.L.F.C.; Reis, S.; Borges, F. Antioxidant profile of dihydroxy- and trihydroxyphenolic acids—A structure-activity relationship study. Free Rad. Res. 2006, 40, 433–442. [Google Scholar] [CrossRef] [PubMed]
- Jing, P.; Zhao, S.J.; Jian, W.J.; Qian, B.J.; Dong, Y.; Pang, J. Quantitative studies on structure-DPPH·scavenging activity relationships of food phenolic acids. Molecules 2012, 17, 12910–12924. [Google Scholar] [CrossRef] [PubMed]
- Chan, E.W.L.; Gray, A.I.; Igoli, J.O.; Lee, S.M.; Goh, J.K. Galloylated flavonol rhamnosides from the leaves of Calliandra tergemina with antibacterial activity against methicillin-resistant Staphylococcus aureus (MRSA). Phytochemistry 2014, 107, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Bag, A.; Bhattacharyya, S.K.; Chattopadhyay, R.R. Isolation and identification of a gallotannin 1,2,6-tri-O-galloyl- β-d-glucopyranose from hydroalcoholic extract of Terminalia chebula fruits effective against multidrug-resistant uropathogens. J. Appl. Microbiol. 2013, 115, 390–397. [Google Scholar] [CrossRef]
- Aguilar-Galvez, A.; Noratto, G.; Chambi, F.; Debaste, F.; Campos, D. Potential of tara (Caesalpinia spinosa) gallotannins and hydrolysates as natural antibacterial compounds. Food Chem. 2014, 156, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Maisetta, G.; Batoni, G.; Caboni, P.; Esin, S.; Rinaldi, A.C.; Zucca, P. Tannin profile, antioxidant properties, and antimicrobial activity of extracts from two Mediterranean species of parasitic plant Cytinus. BMC Complement. Alternat. Med. 2019, 19, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bag, A.; Chattopadhyay, R.R. Synergistic antibiofilm efficacy of a gallotannin 1,2,6-tri-O-galloyl-β-d-glucopyranose from Terminalia chebula fruit in combination with gentamicin and trimethoprim against multidrug resistant uropathogenic Escherichia coli biofilms. PLoS ONE 2017, 12, e0178712. [Google Scholar] [CrossRef] [Green Version]
- Ta, C.; Arnason, J. Mini review of phytochemicals and plant taxa with activity as microbial biofilm and quorum sensing inhibitors. Molecules 2016, 21, 29. [Google Scholar] [CrossRef]
- Narla, A.V.; Borenstein, D.B.; Wingreen, N.S. A biophysical limit for quorum sensing in biofilms. Proc. Natl. Acad. Sci. USA 2021, 118, e2022818118. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Gao, Y.; Wang, H.; Niu, X.; Wang, J. Baicalin weakens Staphylococcus aureus pathogenicity by targeting sortase B. Front. Cell. Infect. Microbiol. 2018, 8, 418. [Google Scholar] [CrossRef] [Green Version]
- Oh, I.; Yang, W.-Y.; Chung, S.-C.; Kim, T.-Y.; Oh, K.-B.; Shin, J. In vitro sortase A inhibitory and antimicrobial activity of flavonoids isolated from the roots of Sophora flavescens. Arch. Pharm. Res. 2011, 34, 217–222. [Google Scholar] [CrossRef]
- Mu, D.; Xiang, H.; Dong, H.; Wang, D.; Wang, T. Isovitexin, a potential candidate inhibitor of sortase A of Staphylococcus aureus USA 300. J. Microbiol. Biotechnol. 2018, 28, 1426–1432. [Google Scholar] [CrossRef]
- Wang, J.; Shi, Y.; Jing, S.; Dong, H.; Wang, D.; Wang, T. Astilbin inhibits the activity of sortase A from Streptococcus mutans. Molecules 2019, 24, 465. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dong, J.; Zhang, L.; Xu, N.; Zhou, S.; Song, Y.; Yang, Q.; Liu, Y.; Yang, Y.; Ai, X. Rutin reduces the pathogenicity of Streptococcus agalactiae to tilapia by inhibiting the activity of sortase A. Aquaculture 2021, 530, 735743. [Google Scholar] [CrossRef]
| Comp. | Concentration (mM) | DPPH Scavenging Activity (%) | Reducing Power (Absorbance) |
|---|---|---|---|
| G2Lyx | 0.1 | 49.97 ± 1.14 | 0.675 ± 0.041 |
| 0.25 | 63.98 ± 1.05 | 0.886 ± 0.015 | |
| 0.5 | 75.35 ± 1.61 | 1.263 ± 0.053 | |
| 1 | 87.36 ± 1.35 | 1.699 ± 0.082 | |
| G2Rib | 0.1 | 52.67 ± 1.27 * | 0.698 ± 0.028 |
| 0.25 | 67.55 ± 1.08 * | 0.979 ± 0.015 | |
| 0.5 | 78.39 ± 1.11 | 1.185 ± 0.031 | |
| 1 | 87.95 ± 1.39 | 1.761 ± 0.102 | |
| G2Rham | 0.1 | 49.68 ± 2.13 | 0.622 ± 0.036 |
| 0.25 | 67.04 ± 1.72 | 0.904 ± 0.045 | |
| 0.5 | 79.63 ± 1.21 | 1.325 ± 0.018 | |
| 1 | 85.18 ± 1.03 | 1.711 ± 0.072 | |
| GMan | 0.1 | 33.57 ± 1.66 *** | 0.534 ± 0.081 * |
| 0.25 | 48.19 ± 2.09 ** | 0.871 ± 0.016 * | |
| 0.5 | 68.42 ± 1.74 * | 1.054 ± 0.037 * | |
| 1 | 71.28 ± 1.16 ** | 1.128 ± 0.057 ** | |
| GFru | 0.1 | 27.67± 1.25 *** | 0.485 ± 0.044 * |
| 0.25 | 39.29 ± 1.08 *** | 0.621 ± 0.073 ** | |
| 0.5 | 62.18 ± 1.13 *** | 0.823 ± 0.051 ** | |
| 1 | 73.36± 2.19 *** | 1.105 ± 0.069 ** | |
| G4Glc | 0.1 | 69.72 ± 1.59 *** | 0.826 ± 0.017 * |
| 0.25 | 75.35 ± 1.98 ** | 1.297 ± 0.044 ** | |
| 0.5 | 84.49 ± 2.27 * | 1.999 ± 0.081 ** | |
| 1 | 95.96 ± 1.77 ** | 2.267 ± 0.086 ** | |
| G4Man | 0.1 | 68.19 ± 1.54 *** | 0.779 ± 0.051 |
| 0.25 | 77.02 ± 2.11 ** | 1.307 ± 0.079 *** | |
| 0.5 | 83.76 ± 1.99 * | 1.878 ± 0.068 ** | |
| 1 | 95.01 ± 1.63 ** | 2.238 ± 0.039 ** | |
| G3Rham | 0.1 | 71.38 ± 1.74 *** | 0.801 ± 0.073 * |
| 0.25 | 78.14 ± 1.49 ** | 1.464 ± 0.057 ** | |
| 0.5 | 86.63 ± 2.08 * | 1.935 ± 0.108 *** | |
| 1 | 94.13 ± 1.27 ** | 2.197 ± 0.096 ** | |
| PGG | 0.1 | 75.32 ± 2.01 *** | 0.918 ± 0.025 * |
| 0.25 | 80.27 ± 1.14 *** | 1.506 ± 0.037 ** | |
| 0.5 | 92.35 ± 1.58 ** | 2.104 ± 0.093 *** | |
| 1 | 97.88 ± 1.22 ** | 2.307 ± 0.034 ** | |
| GA | 0.1 | 47.56 ± 1.45 | 0.688 ± 0.015 |
| 0.25 | 63.03 ± 2.06 | 1.002 ± 0.071 | |
| 0.5 | 76.89 ± 1.73 | 1.289 ± 0.059 | |
| 1 | 88.14 ± 1.16 | 1.736 ± 0.084 |
| Comp. | MIC ([µg/mL]/[mM]) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| SA | MRSA-1 | MRSA-2 | MRSA-3 | EF | VRE-1 | VRE-2 | VRE-3 | MT | MK | MS | |
| G4Glc | 256/ 318 | 256/ 318 | 64/ 79.7 | 64/ 79.7 | 32/ 39.8 | 64/ 79.7 | 256/ 318 | 256/ 318 | >128/ >159 | 256 318 | 256/ 318 |
| G4Man | 64/ 79.7 | 256/ 318 | 64/ 79.7 | 64/ 79.7 | 16/ 19.9 | 32/ 39.8 | 256/ 318 | 256/ 318 | >128/ >159 | 128/ 159 | 256/ 318 |
| G3Rham | 128/ 201 | 256/ 403 | 128/ 201 | 128/ 201 | 32/ 50.4 | 128/ 201 | 256/ 403 | 256/ 403 | >128/ >201 | 128/ 201 | 256/ 403 |
| G2Lyx | >256/ >504 | >256/ >504 | >256/ >504 | >256/ >504 | >256/ >504 | >256/ >504 | >256/ >504 | >256/ >504 | >128/ >252 | >256/ 504 | >256/ 504 |
| G2Rham | 256/ 456 | 256/ 456 | 256/ 456 | 256/ 456 | >256/ >456 | >256/ >456 | >256/ >456 | >256/ >456 | >256/ >456 | >256/ >456 | >256/ >456 |
| G2Rib | 128/ 244 | 256/ 488.2 | 256/ 488 | 128/ 244 | >256/ >488 | >256/ >488 | >256/ >488 | >256/ >488 | >128/ >244 | >256/ >488 | >256/ >488 |
| GMan | 128/ 289 | 256/ 578 | 256/ 578 | 128/ 289 | >256/ >578 | >256/ >578 | >256/ >578 | >256/ >578 | >128/ >289 | >256/ >578 | >256/ >578 |
| GFru | 128/ 310 | 128/ 310 | 256/ 620 | 128/ 310 | >256/ >620 | >256/ >620 | >256/ >620 | >256/ >620 | >128/ >310 | >256/ >620 | >256/ >620 |
| PGG | 32/ 34.0 | 128/ 136 | 32/ 34.0 | 16/ 17.0 | 16/ 17.0 | 64/ 68.0 | 128/ 136 | 256/ 272 | >128/ >136 | 256/ 272 | >256/ >272 |
| GA | 256/ 1487 | 64/ 371 | 64/ 371 | 32/ 185 | >256/ >1487 | >256/ >1487 | >256/ >1487 | >256/ >1487 | >128/ >743 | 256/ 1487 | >256/ >1487 |
| AMP | 2/ 5.72 | 16/ 45.8 | >16/ >45.8 | >16/ >45.8 | 1/ 2.86 | 4/ 11.5 | 4/ 11.5 | 4/ 11.5 | – | – | – |
| INH | – | – | – | – | – | – | – | – | 8 58.0 | 4 29.1 | 16 117 |
| Comp. (conc. 10 mg/mL) | Diameter of Inhibition Zone on Agar [mm] |
|---|---|
| G4Glc | 5.00 ± 0.01 |
| G4Man | 8.50 ± 2.90 |
| G3Rham | 7.00 ± 1.40 |
| G2Rham | – |
| G2Rib | 8.30 ± 0.62 |
| G2Lyx | 5.00 ± 1.01 |
| GMan | 3.00 ± 0.02 |
| GFru | 6.50 ± 1.31 |
| PGG | 6.00 ± 0.01 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hricovíniová, Z.; Mascaretti, Š.; Hricovíniová, J.; Čížek, A.; Jampílek, J. New Unnatural Gallotannins: A Way toward Green Antioxidants, Antimicrobials and Antibiofilm Agents. Antioxidants 2021, 10, 1288. https://doi.org/10.3390/antiox10081288
Hricovíniová Z, Mascaretti Š, Hricovíniová J, Čížek A, Jampílek J. New Unnatural Gallotannins: A Way toward Green Antioxidants, Antimicrobials and Antibiofilm Agents. Antioxidants. 2021; 10(8):1288. https://doi.org/10.3390/antiox10081288
Chicago/Turabian StyleHricovíniová, Zuzana, Šárka Mascaretti, Jana Hricovíniová, Alois Čížek, and Josef Jampílek. 2021. "New Unnatural Gallotannins: A Way toward Green Antioxidants, Antimicrobials and Antibiofilm Agents" Antioxidants 10, no. 8: 1288. https://doi.org/10.3390/antiox10081288
APA StyleHricovíniová, Z., Mascaretti, Š., Hricovíniová, J., Čížek, A., & Jampílek, J. (2021). New Unnatural Gallotannins: A Way toward Green Antioxidants, Antimicrobials and Antibiofilm Agents. Antioxidants, 10(8), 1288. https://doi.org/10.3390/antiox10081288







