Extraction of Phenolic Compounds and Terpenes from Cannabis sativa L. By-Products: From Conventional to Intensified Processes
Abstract
1. Introduction
2. Extraction of Phenolic Compounds from Cannabis
2.1. Phenolic Compounds Present in Cannabis and Their Biological Activities
2.2. The Extraction Processes of PC from Cannabis
2.2.1. Conventional Solvent Extraction
2.2.2. Ultrasound Assisted Extraction (UAE)
| Plant Variety | Plant Organ | Solvent | Operating Conditions | Best PC Results | Best Biological Activities | Ref. |
|---|---|---|---|---|---|---|
| Kompoti, Tiborszallasi, Antal, Carmagnola Cs | Inflorescence | MeOH | 150 mL/g, 4 °C, 30 min |
|
| [14] |
| Bialobrzeskie, Felina 32, Tygra, Futura 75, Santhica 27, Fedora 17, Finola | Defatted seeds | MeOH 80% | 65 °C, 30 min |
|
| [21] |
| Ermes, Santhica 27, Ermo | Inflorescence | MeOH | 100 mL/g, 50 °C, 1 h |
| Nd | [61] |
| Fedora | Seeds | MeOH 80% | 10 mL/g, 4 °C, 30 min |
|
| [62] |
| Defatted seed flour |
|
| ||||
| Oil |
|
| ||||
| Alyssa, Anka, CanMa, CFX 1, CFX 2, CRS 1, Delores, Finola | Seeds | MeOH 90% | 9 mL/g, cold room, 30 min |
| Nd | [63] |
| Fedora | Seeds | MeOH 80% | 10 mL/g, 30 min |
|
| [64] |
| Defatted seed flour |
|
| ||||
| Oil |
|
| ||||
| Futura 75, Kc virtus, Carmagnola Cs, Villanova | Inflorescence | Water | 50 mL/g, 60 °C, 10 min |
|
| [65] |
| Helena | Seed meal | MeOH 80% | 80 mL/g, 10 min |
|
| [66] |
| Bama, Yunma | Defatted kernels | MeOH, EtOH and acetone at 50% 75% 100% | 10 mL/g, 30 min |
|
| [43] |
| Defatted hulls |
|
| ||||
| Hungary | Mix of leaves, flowers and seed husks | MeOH, 20, 50, 80% | 20 mL/g, 90, 120, 150 W, 5, 10, 15 min |
|
| [59] |
| Nd | Seed cake | MAW (35:35:30) | 5–20 mL/g, 40–70 °C, 20, 30, 40 min, 200 W |
|
| [67] |
| Nd | Hempseed cake | MeOH 70%, Acetone 80%, MeOH 70%:Acetone 70% (1:1 v/v) | 9 mL/g, 1 min |
| Nd | [60] |
| Futura 75 | Inflorescence | EtOH | 5 mL/g, 15 min |
|
| [54] |
| Skunk, Fourway, Kompolti, Fasamo | Fruits | MeOH 50%:CHCl3 (1:1) | 80 mL/g, 10 min |
| Nd | [20] |
| Seedling |
| |||||
| Bracts |
| |||||
| Leaves |
| |||||
| Flowers |
|
2.2.3. Other Extraction Techniques
3. Extraction of Terpenes from Cannabis
3.1. The Terpenes Present in Cannabis sativa L. and Their Biological Activities
3.2. The Extraction of Terpenes from Cannabis
3.2.1. Recovery by Steam and Hydrodistillation
3.2.2. Conventional Solvent Extraction
3.2.3. Supercritical Fluid Extraction
3.2.4. Other Extraction Techniques
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Small, E. Classification of Cannabis sativa L. In Relation to Agricultural, Biotechnological, Medical and Recreational Utilization. In Cannabis sativa L.—Botany and Biotechnology; Chandra, S., ElSohly, M.A., Lata, H., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 1–62. [Google Scholar]
- Pollastro, F.; Minassi, A.; Fresu, L.G. Cannabis Phenolics and their Bioactivities. Curr. Med. Chem. 2018, 25, 1160–1185. [Google Scholar] [CrossRef] [PubMed]
- Frassinetti, S.; Moccia, E.; Caltavuturo, L.; Gabriele, M.; Longo, V.; Bellani, L.; Giorgi, G.; Giorgetti, L. Nutraceutical potential of hemp (Cannabis sativa L.) seeds and sprouts. Food Chem. 2018, 262, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Valizadehderakhshan, M.; Shahbazi, A.; Kazem-Rostami, M.; Todd, M.S.; Bhowmik, A.; Wang, L. Extraction of Cannabinoids from Cannabis sativa L. (Hemp)—Review. Agriculture 2021, 11, 384. [Google Scholar] [CrossRef]
- Andre, C.M.; Hausman, J.-F.; Guerriero, G. Cannabis sativa: The Plant of the Thousand and One Molecules. Front. Plant Sci. 2016, 7, 19. [Google Scholar] [CrossRef]
- Ranalli, P.; Venturi, G. Hemp as a raw material for industrial applications. Euphytica 2004, 140, 1–6. [Google Scholar] [CrossRef]
- Flores-Sanchez, I.J.; Verpoorte, R. Secondary metabolism in cannabis. Phytochem. Rev. 2008, 7, 615–639. [Google Scholar] [CrossRef]
- ElSohly, M.A.; Slade, D. Chemical constituents of marijuana: The complex mixture of natural cannabinoids. Life Sci. 2005, 78, 539–548. [Google Scholar] [CrossRef]
- Mahlberg, P.G.; Kim, E.S. Accumulation of Cannabinoids in Glandular Trichomes of Cannabis (Cannabaceae). J. Ind. Hemp 2004, 9, 15–36. [Google Scholar] [CrossRef]
- De Petrocellis, L.; Ligresti, A.; Moriello, A.S.; Allarà, M.; Bisogno, T.; Petrosino, S.; Stott, C.G.; Di Marzo, V. Effects of cannabinoids and cannabinoid-enriched Cannabis extracts on TRP channels and endocannabinoid metabolic enzymes. Br. J. Pharmacol. 2011, 163, 1479–1494. [Google Scholar] [CrossRef]
- Burstein, S. Cannabidiol (CBD) and its analogs: A review of their effects on inflammation. Bioorganic Med. Chem. 2015, 23, 1377–1385. [Google Scholar] [CrossRef]
- Russo, E.B. Taming THC: Potential cannabis synergy and phytocannabinoid-terpenoid entourage effects: Phytocannabinoid-Terpenoid Entourage Effects. Br. J. Pharmacol. 2011, 163, 1344–1364. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, C.L.; Fanovich, M.A.; Churio, M.S. Cannabinoids: Extraction Methods, Analysis, and Physicochemical Characterization. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2019; Volume 61, pp. 143–173. ISBN 978-0-444-64183-0. [Google Scholar]
- Izzo, L.; Castaldo, L.; Narváez, A.; Graziani, G.; Gaspari, A.; Rodríguez-Carrasco, Y.; Ritieni, A. Analysis of Phenolic Compounds in Commercial Cannabis sativa L. Inflorescences Using UHPLC-Q-Orbitrap HRMS. Molecules 2020, 25, 631. [Google Scholar] [CrossRef]
- Gómez-Caravaca, A.M.; Verardo, V.; Segura-Carretero, A.; Fernández-Gutiérrez, A.; Caboni, M.F. Phenolic Compounds and Saponins in Plants Grown Under Different Irrigation Regimes. In Polyphenols in Plants; Elsevier: Amsterdam, The Netherlands, 2014; pp. 37–52. ISBN 978-0-12-397934-6. [Google Scholar]
- Gharaati Jahromi, S. Extraction Techniques of Phenolic Compounds from Plants. In Plant Physiological Aspects of Phenolic Compounds; Soto-Hernández, M., García-Mateos, R., Palma-Tenango, M., Eds.; IntechOpen: London, UK, 2019; pp. 1–18. ISBN 978-1-78984-033-9. [Google Scholar]
- Stojanov, N.; Zeremski, T.; Kiprovski, B.; Koren, A.; Sikora, V.; Miladinovic, J.; Acimovic, M. Possibility of Fiber Hemp Essential Oil Utilization as an Aroma and Fragrance Additive. In Proceedings of the 4th International Congress “Food Techlogy”, Quality and Safety, Novi Sad, Serbia, 23–25 October 2018. [Google Scholar]
- Nahler, G.; Jones, T.M.; Russo, E.B. Cannabidiol and Contributions of Major Hemp Phytocompounds to the “Entourage Effect”; Possible Mechanisms. J. Altern. Complement. Integr. Med. 2019, 5, 1–16. [Google Scholar] [CrossRef]
- Clark, M.N.; Bohm, B.A. Flavonoid variation in Cannabis L. Bot. J. Lannean Soc. 1979, 79, 249–257. [Google Scholar] [CrossRef]
- Flores-Sanchez, I.J.; Verpoorte, R. PKS Activities and Biosynthesis of Cannabinoids and Flavonoids in Cannabis sativa L. Plants. Plant Cell Physiol. 2008, 49, 1767–1782. [Google Scholar] [CrossRef]
- Irakli, M.; Tsaliki, E.; Kalivas, A.; Kleisiaris, F.; Sarrou, E.; Cook, C.M. Effect οf Genotype and Growing Year on the Nutritional, Phytochemical, and Antioxidant Properties of Industrial Hemp (Cannabis sativa L.) Seeds. Antioxidants 2019, 8, 491. [Google Scholar] [CrossRef]
- Ross, S.A.; ElSohly, M.A.; Sultana, G.N.N.; Mehmedic, Z.; Hossain, C.F.; Chandra, S. Flavonoid glycosides and cannabinoids from the pollen of Cannabis sativa L. Phytochem. Anal. 2005, 16, 45–48. [Google Scholar] [CrossRef]
- Vanhoenacker, G.; Van Rompaey, P.; De Keukeleire, D.; Sandra, P. Chemotaxonomic Features Associated with Flavonoids of Cannabinoid-Free Cannabis (Cannabis sativa subsp. sativa L.) in Relation to hops (Humulus Lupulus L.). Nat. Prod. Lett. 2002, 16, 57–63. [Google Scholar] [CrossRef]
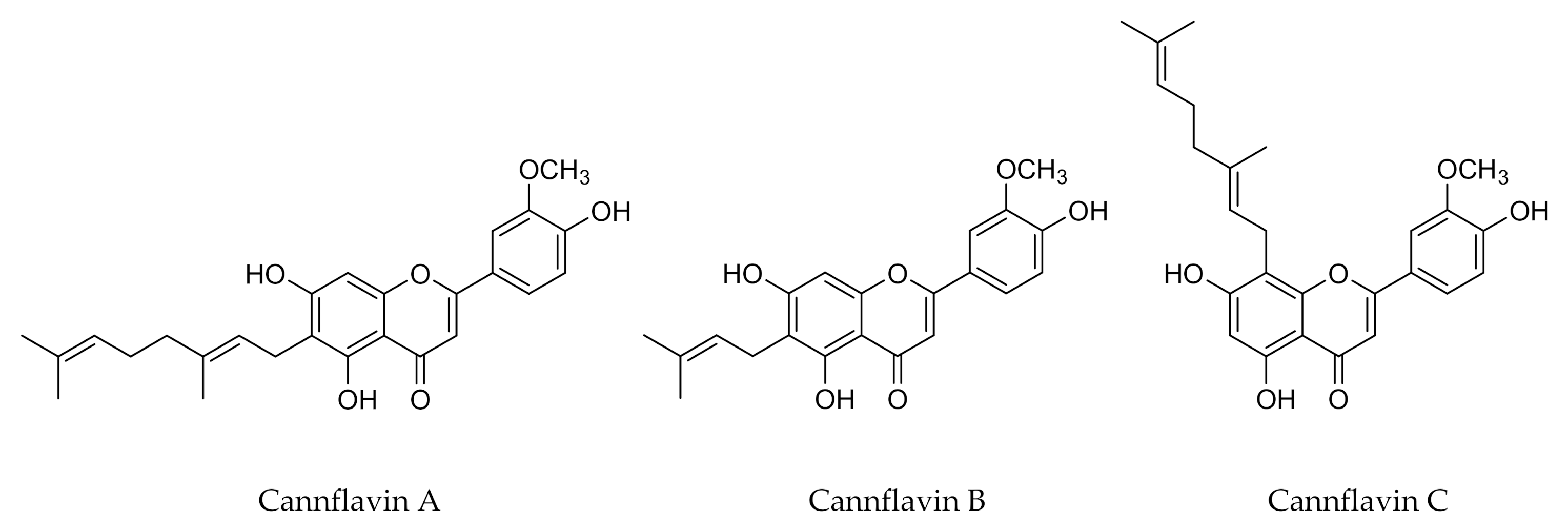
- Barrett, M.L.; Gordon, D.; Evans, F.J. Isolation from Cannabis sativa L. of cannflavin—a novel inhibitor of prostaglandin production. Biochem. Pharmacol. 1985, 34, 2019–2024. [Google Scholar] [CrossRef]
- Radwan, M.M.; Ross, S.A.; Slade, D.; Ahmed, S.A.; Zulfiqar, F.; ElSohly, M.A. Isolation and Characterization of New Cannabis Constituents from a High Potency Variety. Planta Med. 2008, 74, 267–272. [Google Scholar] [CrossRef]
- Barrett, M.L.; Scutt, A.M.; Evans, F.J. Cannflavin A and B, prenylated flavones from Cannabis sativa L. Experientia 1986, 42, 452–453. [Google Scholar] [CrossRef] [PubMed]
- Rea, K.A.; Casaretto, J.A.; Al-Abdul-Wahid, M.S.; Sukumaran, A.; Geddes-McAlister, J.; Rothstein, S.J.; Akhtar, T.A. Biosynthesis of cannflavins A and B from Cannabis sativa L. Phytochemistry 2019, 164, 162–171. [Google Scholar] [CrossRef] [PubMed]
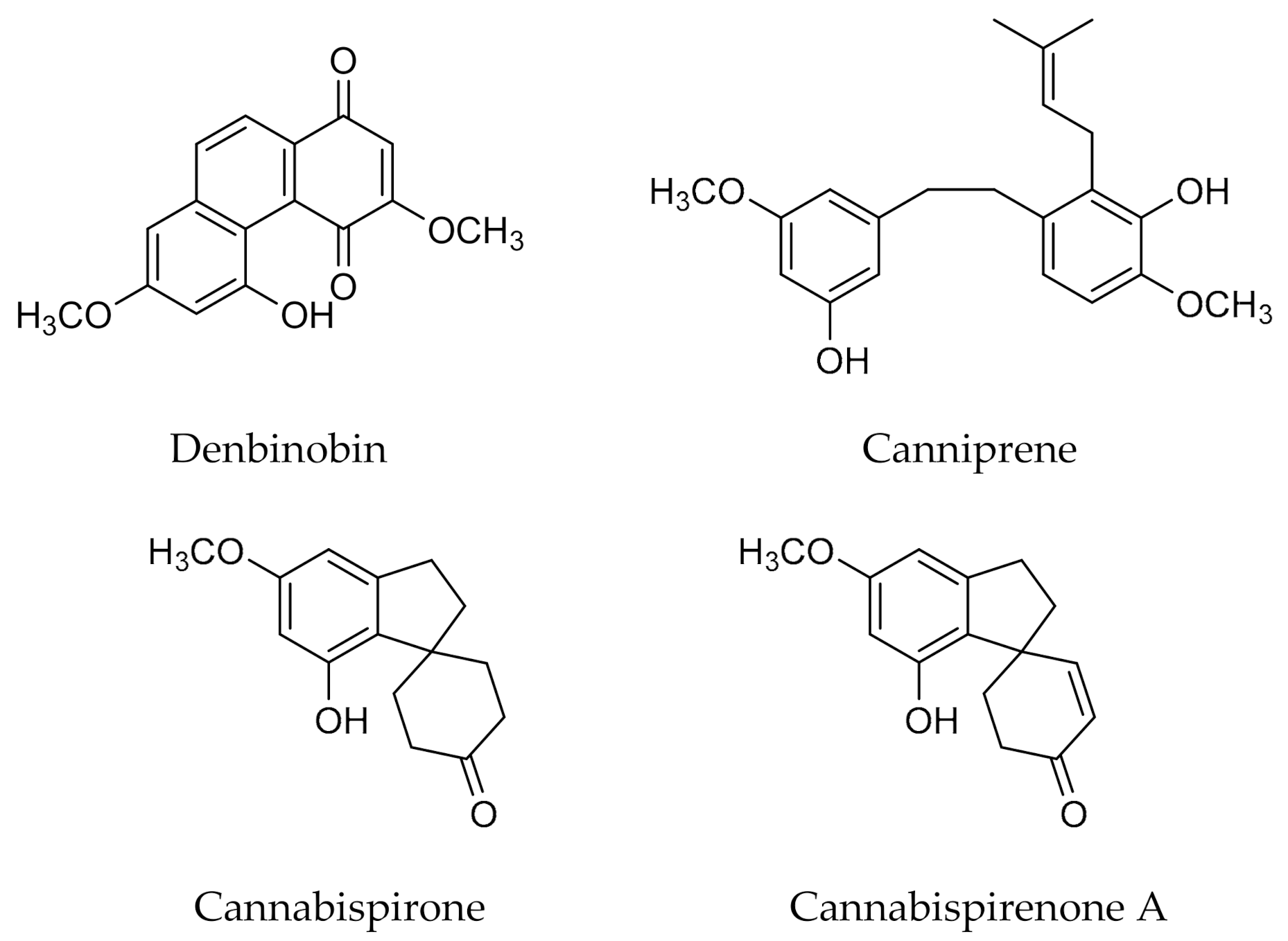
- Radwan, M.M.; ElSohly, M.A.; Slade, D.; Ahmed, S.A.; Wilson, L.; El-Alfy, A.T.; Khan, I.A.; Ross, S.A. Non-cannabinoid constituents from a high potency Cannabis sativa variety. Phytochemistry 2008, 69, 2627–2633. [Google Scholar] [CrossRef] [PubMed]
- Guo, T.-T.; Zhang, J.-C.; Zhang, H.; Liu, Q.-C.; Zhao, Y.; Hou, Y.-F.; Bai, L.; Zhang, L.; Liu, X.-Q.; Liu, X.-Y.; et al. Bioactive spirans and other constituents from the leaves of Cannabis sativa f. sativa. J. Asian Nat. Prod. Res. 2017, 19, 793–802. [Google Scholar] [CrossRef]
- Smeriglio, A.; Galati, E.M.; Monforte, M.T.; Lanuzza, F.; D’Angelo, V.; Circosta, C. Polyphenolic Compounds and Antioxidant Activity of Cold-Pressed Seed Oil from Finola Cultivar of Cannabis sativa L.: Polyphenolic Compounds and Antioxidant Activity of FHSO. Phytother. Res. 2016, 30, 1298–1307. [Google Scholar] [CrossRef]
- Pagani, A.; Scala, F.; Chianese, G.; Grassi, G.; Appendino, G.; Taglialatela-Scafati, O. Cannabioxepane, a novel tetracyclic cannabinoid from hemp, Cannabis sativa L. Tetrahedron 2011, 67, 3369–3373. [Google Scholar] [CrossRef]
- Crombie, L.; Crombie, W.M.L. Natural Products of Thailand High Hl-THC-Strain Cannabis. The Bi- Benzyl-Spiran-Dihydrophenanthrene Group: Relations with Cannabinoids and Canniflavones. J. Chem. Soc. Perkin Trans. 1982, 1, 1455–1466. [Google Scholar] [CrossRef]
- El-Feraly, F.S.; ElSohly, M.A.; Boeren, E.G.; Turner, C.E.; Ottersen, T.; Aasen, A. Crystal and molecular structure of cannabispiran and its correlation to dehydrocannabispiran. Tetrahedron 1977, 33, 2373–2378. [Google Scholar] [CrossRef]
- Allegrone, G.; Pollastro, F.; Magagnini, G.; Taglialatela-Scafati, O.; Seegers, J.; Koeberle, A.; Werz, O.; Appendino, G. The Bibenzyl Canniprene Inhibits the Production of Pro-Inflammatory Eicosanoids and Selectively Accumulates in Some Cannabis sativa Strains. J. Nat. Prod. 2017, 80, 731–734. [Google Scholar] [CrossRef]
- Guo, T.; Liu, Q.; Hou, P.; Li, F.; Guo, S.; Song, W.; Zhang, H.; Liu, X.; Zhang, S.; Zhang, J.; et al. Stilbenoids and cannabinoids from the leaves of Cannabis sativa f. sativa with potential reverse cholesterol transport activity. Food Funct. 2018, 9, 6608–6617. [Google Scholar] [CrossRef]
- Tomassini, L.; Frezza, C.; Ventrone, A.; Serafini, M.; Nicoletti, M.; Piovano, M. Dihydrostilbene derivatives plus kinsenoside from the roots of the rare species Bipinnula fimbriata (Poepp.) I.M.Johnst. Biochem. Syst. Ecol. 2020, 91, 104073. [Google Scholar] [CrossRef]
- Chong, J.; Poutaraud, A.; Hugueney, P. Metabolism and roles of stilbenes in plants. Plant Sci. 2009, 177, 143–155. [Google Scholar] [CrossRef]
- Sánchez-Duffhues, G.; Calzado, M.A.; de Vinuesa, A.G.; Appendino, G.; Fiebich, B.L.; Loock, U.; Lefarth-Risse, A.; Krohn, K.; Muñoz, E. Denbinobin inhibits nuclear factor-κB and induces apoptosis via reactive oxygen species generation in human leukemic cells. Biochem. Pharmacol. 2009, 77, 1401–1409. [Google Scholar] [CrossRef]
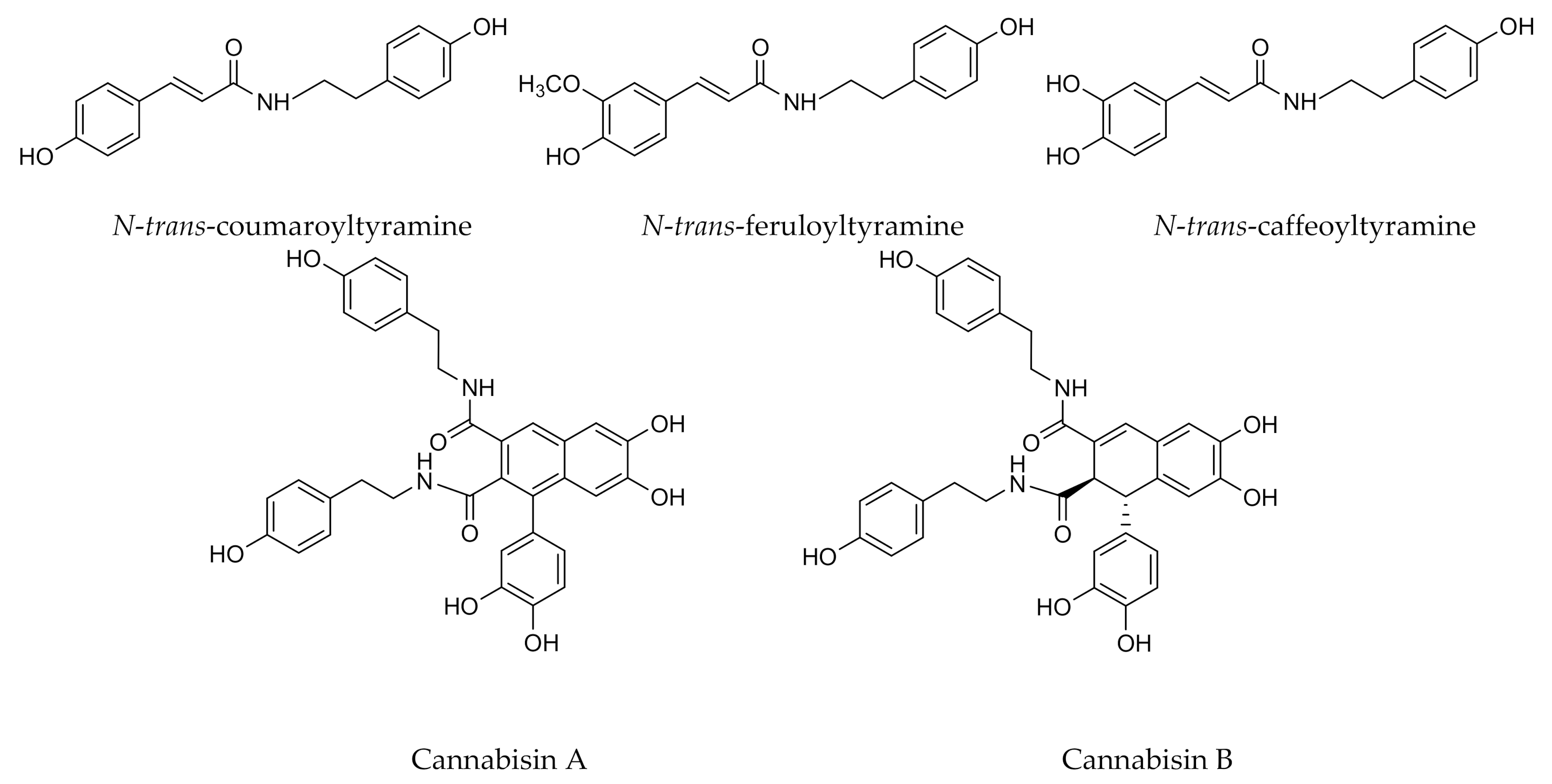
- Yan, X.; Tang, J.; dos Santos Passos, C.; Nurisso, A.; Avello Simões-Pires, C.; Ji, M.; Lou, H.; Fan, P. Characterization of Lignanamides from Hemp (Cannabis sativa L.) Seed and Their Antioxidant and Acetylcholinesterase Inhibitory Activities. J. Agric. Food Chem. 2015, 63, 10611–10619. [Google Scholar] [CrossRef]
- Crescente, G.; Piccolella, S.; Esposito, A.; Scognamiglio, M.; Fiorentino, A.; Pacifico, S. Chemical composition and nutraceutical properties of hempseed: An ancient food with actual functional value. Phytochem. Rev. 2018, 17, 733–749. [Google Scholar] [CrossRef]
- Slatkin, D.J.; Doorenbos, N.J.; Harris, L.S.; Masoud, A.N.; Quimby, M.W.; Schiff, P.L. Chemical Constituents of Cannabis sativa L. Root. J. Pharm. Sci. 1971, 60, 1891–1892. [Google Scholar] [CrossRef]
- Leonard, W.; Zhang, P.; Ying, D.; Fang, Z. Lignanamides: Sources, biosynthesis and potential health benefits—A minireview. Crit. Rev. Food Sci. Nutr. 2021, 61, 1404–1414. [Google Scholar] [CrossRef]
- Chen, T.; He, J.; Zhang, J.; Li, X.; Zhang, H.; Hao, J.; Li, L. The isolation and identification of two compounds with predominant radical scavenging activity in hempseed (seed of Cannabis sativa L.). Food Chem. 2012, 134, 1030–1037. [Google Scholar] [CrossRef]
- Zengin, G.; Menghini, L.; Di Sotto, A.; Mancinelli, R.; Sisto, F.; Carradori, S.; Cesa, S.; Fraschetti, C.; Filippi, A.; Angiolella, L.; et al. Chromatographic Analyses, In Vitro Biological Activities, and Cytotoxicity of Cannabis sativa L. Essential Oil: A Multidisciplinary Study. Molecules 2018, 23, 3266. [Google Scholar] [CrossRef]
- Gutiérrez, A.; Rodríguez, I.M.; Del Río, C.D. Chemical Characterization of Lignin and Lipid Fractions in Industrial Hemp Bast Fibers Used for Manufacturing High-Quality Paper Pulps. J. Agric. Food Chem. 2006, 54, 2138–2144. [Google Scholar] [CrossRef]
- Bassil, S. Etude de La Répartition Structurale Des Acides Férulique et P-Coumarique Dans La Chènevotte et La Poudre Organique de Chanvre (Cannabis Sativa) Exploration Des Voies de Fractionnement Pour L’obtention D’extraits à Valeur Ajoutée. Ph.D. Thesis, Université Paul Sabatier, Toulouse, France, 2015. [Google Scholar]
- Candy, L.; Bassil, S.; Rigal, L.; Simon, V.; Raynaud, C. Thermo-mechano-chemical extraction of hydroxycinnamic acids from industrial hemp by-products using a twin-screw extruder. Ind. Crops Prod. 2017, 109, 335–345. [Google Scholar] [CrossRef]
- Lesma, G.; Consonni, R.; Gambaro, V.; Remuzzi, C.; Roda, G.; Silvani, A.; Vece, V.; Visconti, G.L. Cannabinoid-free Cannabis sativa L. grown in the Po valley: Evaluation of fatty acid profile, antioxidant capacity and metabolic content. Nat. Prod. Res. 2014, 28, 1801–1807. [Google Scholar] [CrossRef]
- Drinić, Z.; Vidović, S.; Vladić, J.; Koren, A.; Kiprovski, B.; Sikora, V. Effect of Extraction Solvent on Total Polyphenols Content and Antioxidant Activity of Industrial Hemp (Cannabis Sativa L.). Lek. Sirovine 2018, 17–21. [Google Scholar] [CrossRef]
- Teh, S.-S.; Bekhit, A.E.-D.; Birch, J. Antioxidative Polyphenols from Defatted Oilseed Cakes: Effect of Solvents. Antioxidants 2014, 3, 67–80. [Google Scholar] [CrossRef]
- Mkpenie, V.N.; Essien, E.E.; Udoh, I.I. Effect of Extraction Conditions on Total Polyphenol Contents, Antioxidant and Antimicrobial Activities of Cannabis sativa L. EJEAFChe 2012, 11, 300–307. [Google Scholar]
- Mourtzinos, I.; Menexis, N.; Iakovidis, D.; Makris, D.P.; Goula, A. A Green Extraction Process to Recover Polyphenols from Byproducts of Hemp Oil Processing. Recycling 2018, 3, 15. [Google Scholar] [CrossRef]
- Ahmed, M.; Ji, M.; Qin, P.; Gu, Z.; Liu, Y.; Sikandar, A.; Iqbal, M.F.; Javeed, A. Phytochemical Screening, Total Phenolic and Flavonoids Contents and Antioxidant Activities of Citrullus Colocynthis L. and Cannabis Sativa L. Appl. Ecol. Environ. Res. 2019, 17, 6961–6979. [Google Scholar] [CrossRef]
- Palmieri, S.; Pellegrini, M.; Ricci, A.; Compagnone, D.; Lo Sterzo, C. Chemical Composition and Antioxidant Activity of Thyme, Hemp and Coriander Extracts: A Comparison Study of Maceration, Soxhlet, UAE and RSLDE Techniques. Foods 2020, 9, 1221. [Google Scholar] [CrossRef]
- Dhapte, V.; Mehta, P. Advances in hydrotropic solutions: An updated review. St. Petersburg Polytech. Univ. J. Phys. Math. 2015, 1, 424–435. [Google Scholar] [CrossRef]
- Rajha, H.N.; Chacar, S.; Afif, C.; Vorobiev, E.; Louka, N.; Maroun, R.G. β-Cyclodextrin-Assisted Extraction of Polyphenols from Vine Shoot Cultivars. J. Agric. Food Chem. 2015, 63, 3387–3393. [Google Scholar] [CrossRef]
- Deng, Y.; Zhao, Y.; Padilla-Zakour, O.; Yang, G. Polyphenols, antioxidant and antimicrobial activities of leaf and bark extracts of Solidago canadensis L. Ind. Crops Prod. 2015, 75, 803–809. [Google Scholar] [CrossRef]
- Wang, L.; Weller, C.L. Recent advances in extraction of nutraceuticals from plants. Trends Food Sci. Technol. 2006, 17, 300–312. [Google Scholar] [CrossRef]
- Agarwal, C.; Máthé, K.; Hofmann, T.; Csóka, L. Ultrasound-Assisted Extraction of Cannabinoids from Cannabis sativa L. Optimized by Response Surface Methodology. J. Food Sci. 2018, 83, 700–710. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Zago, E.; Nandasiri, R.; Khattab, R.; Eskin, N.A.M.; Eck, P.; Thiyam-Holländer, U. Effect of Solvent, Preheating Temperature, and Time on the Ultrasonic Extraction of Phenolic Compounds from Cold-Pressed Hempseed Cake. J. Am. Oil Chem. Soc. 2018, 95, 1319–1327. [Google Scholar] [CrossRef]
- Calzolari, D.; Magagnini, G.; Lucini, L.; Grassi, G.B.; Appendino, G.; Amaducci, S. High added-value compounds from Cannabis threshing residues. Ind. Crops Prod. 2017, 108, 558–563. [Google Scholar] [CrossRef]
- Siano, F.; Moccia, S.; Picariello, G.; Russo, G.L.; Sorrentino, G.; Di Stasio, M.; La Cara, F.; Volpe, M.G. Comparative Study of Chemical, Biochemical Characteristic and ATR-FTIR Analysis of Seeds, Oil and Flour of the Edible Fedora Cultivar Hemp (Cannabis sativa L.). Molecules 2019, 24, 83. [Google Scholar] [CrossRef]
- Vonapartis, E.; Aubin, M.-P.; Seguin, P.; Mustafa, A.F.; Charron, J.-B. Seed composition of ten industrial hemp cultivars approved for production in Canada. J. Food Compos. Anal. 2015, 39, 8–12. [Google Scholar] [CrossRef]
- Moccia, S.; Siano, F.; Russo, G.L.; Volpe, M.G.; La Cara, F.; Pacifico, S.; Piccolella, S.; Picariello, G. Antiproliferative and antioxidant effect of polar hemp extracts (Cannabis sativa L., Fedora cv.) in human colorectal cell lines. Int. J. Food Sci. Nutr. 2020, 71, 410–423. [Google Scholar] [CrossRef]
- Ferrante, C.; Recinella, L.; Ronci, M.; Menghini, L.; Brunetti, L.; Chiavaroli, A.; Leone, S.; Di Iorio, L.; Carradori, S.; Tirillini, B.; et al. Multiple pharmacognostic characterization on hemp commercial cultivars: Focus on inflorescence water extract activity. Food Chem. Toxicol. 2019, 125, 452–461. [Google Scholar] [CrossRef]
- Pojić, M.; Mišan, A.; Sakač, M.; Dapčević Hadnađev, T.; Šarić, B.; Milovanović, I.; Hadnađev, M. Characterization of Byproducts Originating from Hemp Oil Processing. J. Agric. Food Chem. 2014, 62, 12436–12442. [Google Scholar] [CrossRef]
- Teh, S.-S.; Birch, E.J. Effect of ultrasonic treatment on the polyphenol content and antioxidant capacity of extract from defatted hemp, flax and canola seed cakes. Ultrason. Sonochem. 2014, 21, 346–353. [Google Scholar] [CrossRef]
- Schmidt, S.; Pokorný, J. Potential application of oilseeds as sources of antioxidants for food lipids—A review. Czech J. Food Sci. 2005, 23, 93–102. [Google Scholar] [CrossRef]
- Li, Y.; Fabiano-Tixier, A.-S.; Abert-Vian, M.; Chemat, F. Microwave-Assisted Extraction of Antioxidants and Food Colors. In Microwave-Assisted Extraction for Bioactive Compounds; Chemat, F., Cravotto, G., Eds.; Food Engineering Series; Springer: Boston, MA, USA, 2012; pp. 103–125. ISBN 978-1-4614-4829-7. [Google Scholar]
- Drinić, Z.; Vladić, J.; Koren, A.; Zeremski, T.; Stojanov, N.; Kiprovski, B.; Vidović, S. Microwave-assisted extraction of cannabinoids and antioxidants from Cannabis sativa aerial parts and process modeling. J. Chem. Technol. Biotechnol. 2020, 95, 831–839. [Google Scholar] [CrossRef]
- Matešić, N.; Jurina, T.; Benković, M.; Panić, M.; Valinger, D.; Gajdoš Kljusurić, J.; Jurinjak Tušek, A. Microwave-assisted extraction of phenolic compounds from Cannabis sativa L.: Optimization and kinetics study. Sep. Sci. Technol. 2020, 1–14. [Google Scholar] [CrossRef]
- Teh, S.-S.; Niven, B.E.; Bekhit, A.E.-D.A.; Carne, A.; Birch, E.J. The Use of Microwave and Pulsed Electric Field as a Pretreatment Step in Ultrasonic Extraction of Polyphenols from Defatted Hemp Seed Cake (Cannabis sativa) Using Response Surface Methodology. Food Bioprocess Technol. 2014, 7, 3064–3076. [Google Scholar] [CrossRef]
- Wibisono, R.; Zhang, J.; Saleh, Z.; Stevenson, D.E.; Joyce, N.I. Optimisation of accelerated solvent extraction for screening of the health benefits of plant food materials. Health 2009, 1, 220–230. [Google Scholar] [CrossRef]
- Kitrytė, V.; Bagdonaitė, D.; Rimantas Venskutonis, P. Biorefining of industrial hemp (Cannabis sativa L.) threshing residues into cannabinoid and antioxidant fractions by supercritical carbon dioxide, pressurized liquid and enzyme-assisted extractions. Food Chem. 2018, 267, 420–429. [Google Scholar] [CrossRef]
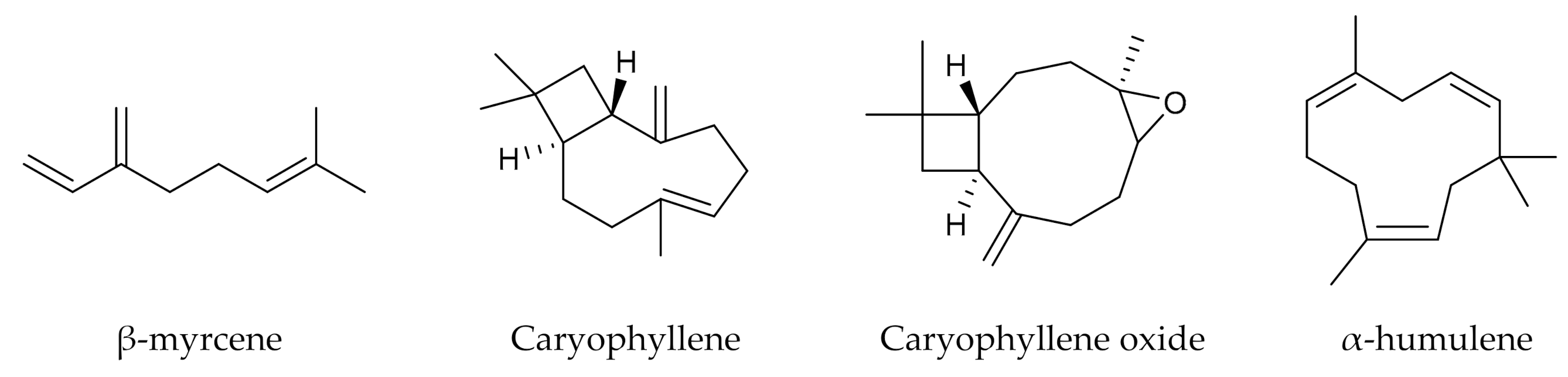
- Booth, J.K.; Page, J.E.; Bohlmann, J. Terpene synthases from Cannabis sativa. PLoS ONE 2017, 12, e0173911. [Google Scholar] [CrossRef]
- Ibrahim, E.A.; Wang, M.; Radwan, M.M.; Wanas, A.S.; Majumdar, C.G.; Avula, B.; Wang, Y.-H.; Khan, I.A.; Chandra, S.; Lata, H.; et al. Analysis of Terpenes in Cannabis sativa L. Using GC/MS: Method Development, Validation, and Application. Planta Med. 2019, 85, 431–438. [Google Scholar] [CrossRef]
- Hillig, K.W. A chemotaxonomic analysis of terpenoid variation in Cannabis. Biochem. Syst. Ecol. 2004, 32, 875–891. [Google Scholar] [CrossRef]
- Fischedick, J.T.; Hazekamp, A.; Erkelens, T.; Choi, Y.H.; Verpoorte, R. Metabolic fingerprinting of Cannabis sativa L., cannabinoids and terpenoids for chemotaxonomic and drug standardization purposes. Phytochemistry 2010, 71, 2058–2073. [Google Scholar] [CrossRef] [PubMed]
- Namdar, D.; Mazuz, M.; Ion, A.; Koltai, H. Variation in the compositions of cannabinoid and terpenoids in Cannabis sativa derived from inflorescence position along the stem and extraction methods. Ind. Crops Prod. 2018, 113, 376–382. [Google Scholar] [CrossRef]
- Naz, S.; Hanif, M.A.; Bhatti, H.N.; Ansari, T.M. Impact of Supercritical Fluid Extraction and Traditional Distillation on the Isolation of Aromatic Compounds fromCannabis indicaand Cannabis sativa. J. Essent. Oil Bear. Plants 2017, 20, 175–184. [Google Scholar] [CrossRef]
- Vuerich, M.; Ferfuia, C.; Zuliani, F.; Piani, B.; Sepulcri, A.; Baldini, M. Yield and Quality of Essential Oils in Hemp Varieties in Different Environments. Agronomy 2019, 9, 356. [Google Scholar] [CrossRef]
- Smeriglio, A.; Trombetta, D.; Alloisio, S.; Cornara, L.; Denaro, M.; Garbati, P.; Grassi, G.; Circosta, C. Promising in vitro antioxidant, anti-acetylcholinesterase and neuroactive effects of essential oil from two non-psychotropic Cannabis sativa L. biotypes. Phytotherapy Res. 2020, 1–16. [Google Scholar] [CrossRef]
- Wanas, A.S.; Radwan, M.M.; Chandra, S.; Lata, H.; Mehmedic, Z.; Ali, A.; Baser, K.; Demirci, B.; ElSohly, M.A. Chemical Composition of Volatile Oils of Fresh and Air-Dried Buds of Cannabis chemovars, Their Insecticidal and Repellent Activities. Nat. Prod. Commun. 2020, 15, 1–7. [Google Scholar] [CrossRef]
- Benelli, G.; Pavela, R.; Petrelli, R.; Cappellacci, L.; Santini, G.; Fiorini, D.; Sut, S.; Dall’Acqua, S.; Canale, A.; Maggi, F. The essential oil from industrial hemp (Cannabis sativa L.) by-products as an effective tool for insect pest management in organic crops. Ind. Crops Prod. 2018, 122, 308–315. [Google Scholar] [CrossRef]
- Zheljazkov, V.D.; Sikora, V.; Dincheva, I.; Kačániová, M.; Astatkie, T.; Semerdjieva, I.B.; Latkovic, D. Industrial, CBD, and Wild Hemp: How Different Are Their Essential Oil Profile and Antimicrobial Activity? Molecules 2020, 25, 4631. [Google Scholar] [CrossRef]
- Ascrizzi, R.; Ceccarini, L.; Tavarini, S.; Flamini, G.; Angelini, L.G. Valorisation of hemp inflorescence after seed harvest: Cultivation site and harvest time influence agronomic characteristics and essential oil yield and composition. Ind. Crops Prod. 2019, 139, 111541. [Google Scholar] [CrossRef]
- Fiorini, D.; Molle, A.; Nabissi, M.; Santini, G.; Benelli, G. Valorizing industrial hemp (Cannabis sativa L.) by-products: Cannabidiol enrichment in the inflorescence essential oil optimizing sample pre-treatment prior to distillation. Ind. Crops Prod. 2019, 128, 581–589. [Google Scholar] [CrossRef]
- Bertoli, A.; Tozzi, S.; Pistelli, L.; Angelini, L.G. Fibre hemp inflorescences: From crop-residues to essential oil production. Ind. Crops Prod. 2010, 32, 329–337. [Google Scholar] [CrossRef]
- Casano, S.; Grassi, G.; Martini, V.; Michelozzi, M. Variations in Terpene Profiles of Different Strains of Cannabis Sativa L. Acta Hortic. 2011, 925, 115–121. [Google Scholar] [CrossRef]
- Abdollahi, M.; Sefidkon, F.; Calagari, M.; Mousavi, A.; Mahomoodally, M.F. Impact of four hemp (Cannabis sativa L.) varieties and stage of plant growth on yield and composition of essential oils. Ind. Crops Prod. 2020, 155, 112793. [Google Scholar] [CrossRef]
- Ross, S.A.; ElSohly, M.A. The Volatile Oil Composition of Fresh and Air-Dried Buds of Cannabis sativa. J. Nat. Prod. 1996, 59, 49–51. [Google Scholar] [CrossRef]
- Sethi, V.K.; Jain, M.P.; Thakur, R.S. Chemical Investigation of Wild Cannabis Sativa L. Roots. Planta Med. 1977, 32, 378–379. [Google Scholar] [CrossRef]
- Jin, D.; Dai, K.; Xie, Z.; Chen, J. Secondary Metabolites Profiled in Cannabis Inflorescences, Leaves, Stem Barks, and Roots for Medicinal Purposes. Sci. Rep. 2020, 10, 3309. [Google Scholar] [CrossRef]
- Sunil, C.; Irudayaraj, S.S.; Duraipandiyan, V.; Alrashood, S.T.; Alharbi, S.A.; Ignacimuthu, S. Friedelin exhibits antidiabetic effect in diabetic rats via modulation of glucose metabolism in liver and muscle. J. Ethnopharmacol. 2021, 268, 113659. [Google Scholar] [CrossRef]
- Gupta, M.; Kapoor, B.; Gupta, R.; Singh, N. Plants and phytochemicals for treatment of peptic ulcer: An overview. S. Afr. J. Bot. 2021, 138, 105–114. [Google Scholar] [CrossRef]
- Gonçalves Pereira, R.C.; Gontijo Evangelista, F.C.; dos Santos Júnior, V.S.; de Paula Sabino, A.; Gonçalves Maltarollo, V.; Pereira de Freitas, R.; Pains Duarte, L. Cytotoxic Activity of Triterpenoids from Cheiloclinium cognatum Branches against Chronic and Acute Leukemia Cell Lines. Chem. Biodivers. 2020, 17, e2000773. [Google Scholar] [CrossRef]
- Hernández Vázquez, L.; Palazon, J.; Navarro-Ocaña, A. The Pentacyclic Triterpenes α, β-amyrins: A Review of Sources and Biological Activities. In Phytochemicals—A Global Perspective of Their Role in Nutrition and Health; Rao, V., Ed.; IntechOpen: London, UK, 2012; pp. 487–502. ISBN 978-953-51-0296-0. [Google Scholar]
- Kwaśnica, A.; Pachura, N.; Masztalerz, K.; Figiel, A.; Zimmer, A.; Kupczyński, R.; Wujcikowska, K.; Carbonell-Barrachina, A.A.; Szumny, A.; Różański, H. Volatile Composition and Sensory Properties as Quality Attributes of Fresh and Dried Hemp Flowers (Cannabis sativa L.). Foods 2020, 9, 1118. [Google Scholar] [CrossRef]
- Nitsche, M.; Gbadamosi, R. Steam Distillation. In Practical Column Design Guide; Nitsche, M., Gbadamosi, R., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 187–213. ISBN 978-3-319-51687-5. [Google Scholar]
- Chemat, F.; Boutekedjiret, C. Extraction//Steam Distillation. In Reference Module in Chemistry, Molecular Sciences and Chemical Engineering; Elsevier: Amsterdam, The Netherlands, 2015; ISBN 978-0-12-409547-2. [Google Scholar]
- Zheljazkov, V.D.; Sikora, V.; Semerdjieva, I.B.; Kačániová, M.; Astatkie, T.; Dincheva, I. Grinding and Fractionation during Distillation Alter Hemp Essential Oil Profile and Its Antimicrobial Activity. Molecules 2020, 25, 3943. [Google Scholar] [CrossRef]
- Darani, K.K.; Mozafari, M.R. Supercritical Fluids Technology in Bioprocess Industries: A Review. J. Biochem. Tech. 2009, 2, 144–152. [Google Scholar]
- Wells, S.L.; DeSimone, J. CO2 Technology Platform: An Important Tool for Environmental Problem Solving. Angew. Chem. Int. Ed. 2001, 40, 518–527. [Google Scholar] [CrossRef]
- DA Porto, C.; Decorti, D.; Natolino, A. Separation of aroma compounds from industrial hemp inflorescences (Cannabis sativa L.) by supercritical CO2 extraction and on-line fractionation. Ind. Crops Prod. 2014, 58, 99–103. [Google Scholar] [CrossRef]
- Vági, E.; Balázs, M.; Komóczi, A.; Kiss, I.; Mihalovits, M.; Székely, E. Cannabinoids Enriched Extracts from Industrial Hemp Residues. Period. Polytech. Chem. Eng. 2019, 63, 357–363. [Google Scholar] [CrossRef]
- Omar, J.; Olivares, M.; Alzaga, M.; Etxebarria, N. Optimisation and characterisation of marihuana extracts obtained by supercritical fluid extraction and focused ultrasound extraction and retention time locking GC-MS. J. Sep. Sci. 2013, 36, 1397–1404. [Google Scholar] [CrossRef]
- Grijó, D.R.; Vieitez Osorio, I.A.; Cardozo-Filho, L. Supercritical extraction strategies using CO2 and ethanol to obtain cannabinoid compounds from Cannabis hybrid flowers. J. CO2 Util. 2018, 28, 174–180. [Google Scholar] [CrossRef]
- Aizpurua-Olaizola, O.; Soydaner, U.; Öztürk, E.; Schibano, D.; Simsir, Y.; Navarro, P.; Etxebarria, N.; Usobiaga, A. Evolution of the Cannabinoid and Terpene Content during the Growth of Cannabis sativa Plants from Different Chemotypes. J. Nat. Prod. 2016, 79, 324–331. [Google Scholar] [CrossRef]
- Fiorini, D.; Scortichini, S.; Bonacucina, G.; Greco, N.G.; Mazzara, E.; Petrelli, R.; Torresi, J.; Maggi, F.; Cespi, M. Cannabidiol-enriched hemp essential oil obtained by an optimized microwave-assisted extraction using a central composite design. Ind. Crops Prod. 2020, 154, 112688. [Google Scholar] [CrossRef]
- Ternelli, M.; Brighenti, V.; Anceschi, L.; Poto, M.; Bertelli, D.; Licata, M.; Pellati, F. Innovative methods for the preparation of medical Cannabis oils with a high content of both cannabinoids and terpenes. J. Pharm. Biomed. Anal. 2020, 186, 113296. [Google Scholar] [CrossRef]
- Jovanovic, A.; Petrovic, P.; Ðordjevic, V.; Zdunic, G.; Savikin, K.; Bugarski, B. Polyphenols extraction from plant sources. Lek. Sirovine 2017, 37, 45–49. [Google Scholar] [CrossRef]
| Plant Variety | Plant Organs | Solvent | Operating Conditions | Best PC Results | Best Biological Activities | Ref. |
|---|---|---|---|---|---|---|
| Futura 75 | Seeds | EtOH 80% | 10 mL/g, 4 °C, 3 h |
|
| [3] |
| Futura 75 | Seeds | MeOH 80% | 5 mL/g, 3 h |
|
| [48] |
| Carmagnola | Seeds | MeOH 80% | 5 mL/g, 3 h |
|
| |
| Felina 32 | Seeds | MeOH 80% | 5 mL/g, 3 h |
|
| |
| Helena | Aerial parts | Water EtOH 30, 50, 70, 90% | 20 mL/g, RT, 24 h |
|
| [49] |
| Nd | Seed meal | MeOH 80, 100%; EtOH 100%; Acetone 80, 100% MAW (35:35:30 v/v/v) | 16.6 mL/g, 25 °C, 1 h |
|
| [50] |
| Nd | Leaves | Acetone 50, 100% MeOH 50, 100% | 10 mL/g, RT, 2, 8 or 18 h |
|
| [51] |
| Helena | Seed meal | CD/water mixtures (1–40%) | 5–15.2 mL/g, 20–60 °C, 3 h |
|
| [52] |
| Nd | Leaves and flowers | MeOH | 30 mL/g, 40 min |
|
| [34] |
| Nd | Leaves | EtOH, MeOH, ethyl acetate, CHCl3, acetone, hexane, water | 4 mL/g, RT, 72 h |
|
| [53] |
| Futura 75 | Inflorescence | EtOH | 5 mL/g, RT, 30 days |
|
| [54] |
| 5 mL/g, 100 °C, 2–6 h |
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Isidore, E.; Karim, H.; Ioannou, I. Extraction of Phenolic Compounds and Terpenes from Cannabis sativa L. By-Products: From Conventional to Intensified Processes. Antioxidants 2021, 10, 942. https://doi.org/10.3390/antiox10060942
Isidore E, Karim H, Ioannou I. Extraction of Phenolic Compounds and Terpenes from Cannabis sativa L. By-Products: From Conventional to Intensified Processes. Antioxidants. 2021; 10(6):942. https://doi.org/10.3390/antiox10060942
Chicago/Turabian StyleIsidore, Emilie, Hamza Karim, and Irina Ioannou. 2021. "Extraction of Phenolic Compounds and Terpenes from Cannabis sativa L. By-Products: From Conventional to Intensified Processes" Antioxidants 10, no. 6: 942. https://doi.org/10.3390/antiox10060942
APA StyleIsidore, E., Karim, H., & Ioannou, I. (2021). Extraction of Phenolic Compounds and Terpenes from Cannabis sativa L. By-Products: From Conventional to Intensified Processes. Antioxidants, 10(6), 942. https://doi.org/10.3390/antiox10060942







