Coenzyme Q10 and Male Infertility: A Systematic Review
Abstract
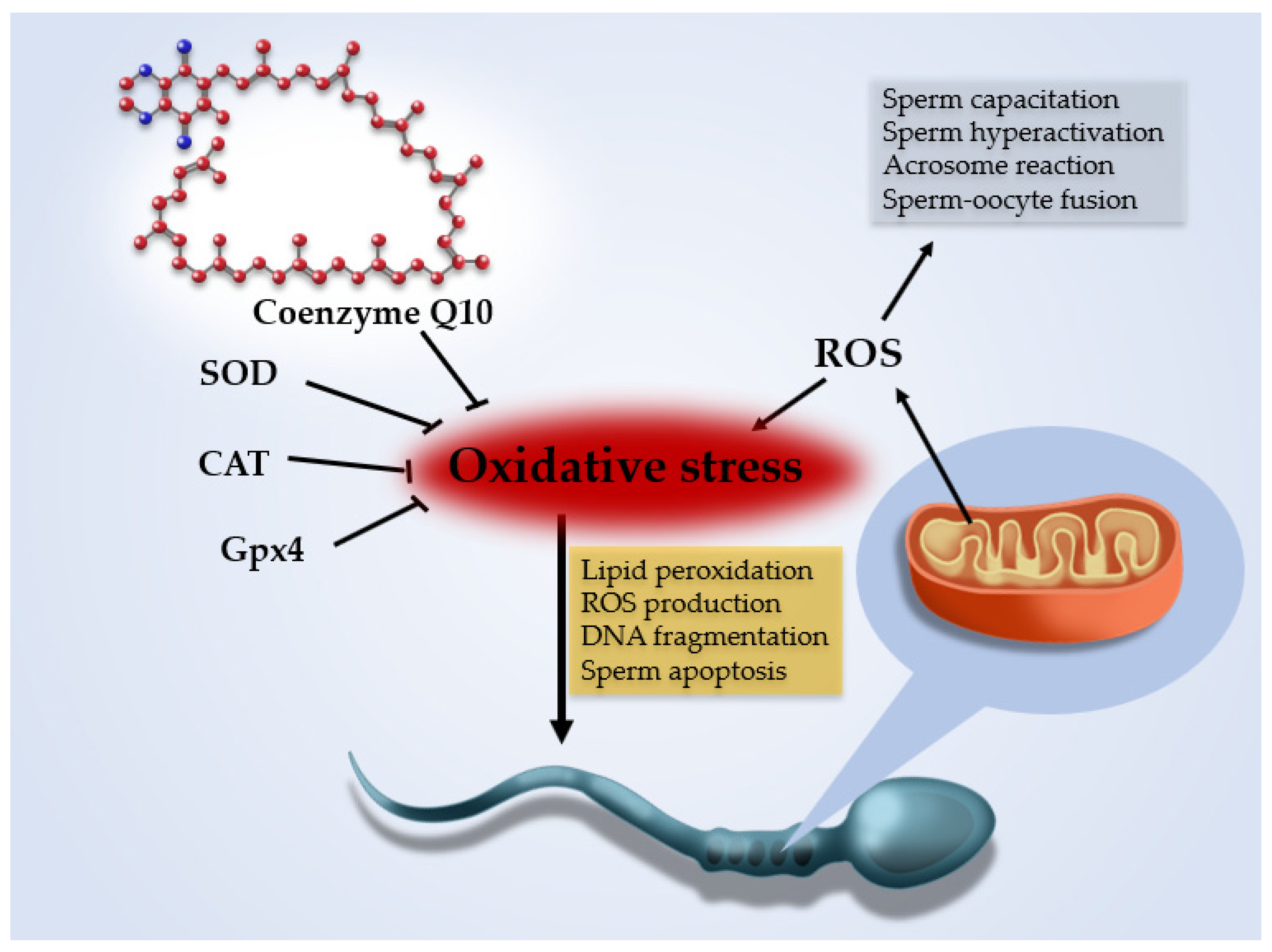
1. Introduction
2. Materials and Methods
2.1. Literature Search
2.2. Inclusion Criteria
- Studies evaluating the effect of CoQ10 or ubiquinol (a reduced form of CoQ10) supplementation (alone or with other antioxidant molecules) on male fertility
2.3. Exclusion Criteria
- Animal studies
- Scientific articles that were not published in English
- Review or conference abstracts or letters to the editor
- Studies published before 2000
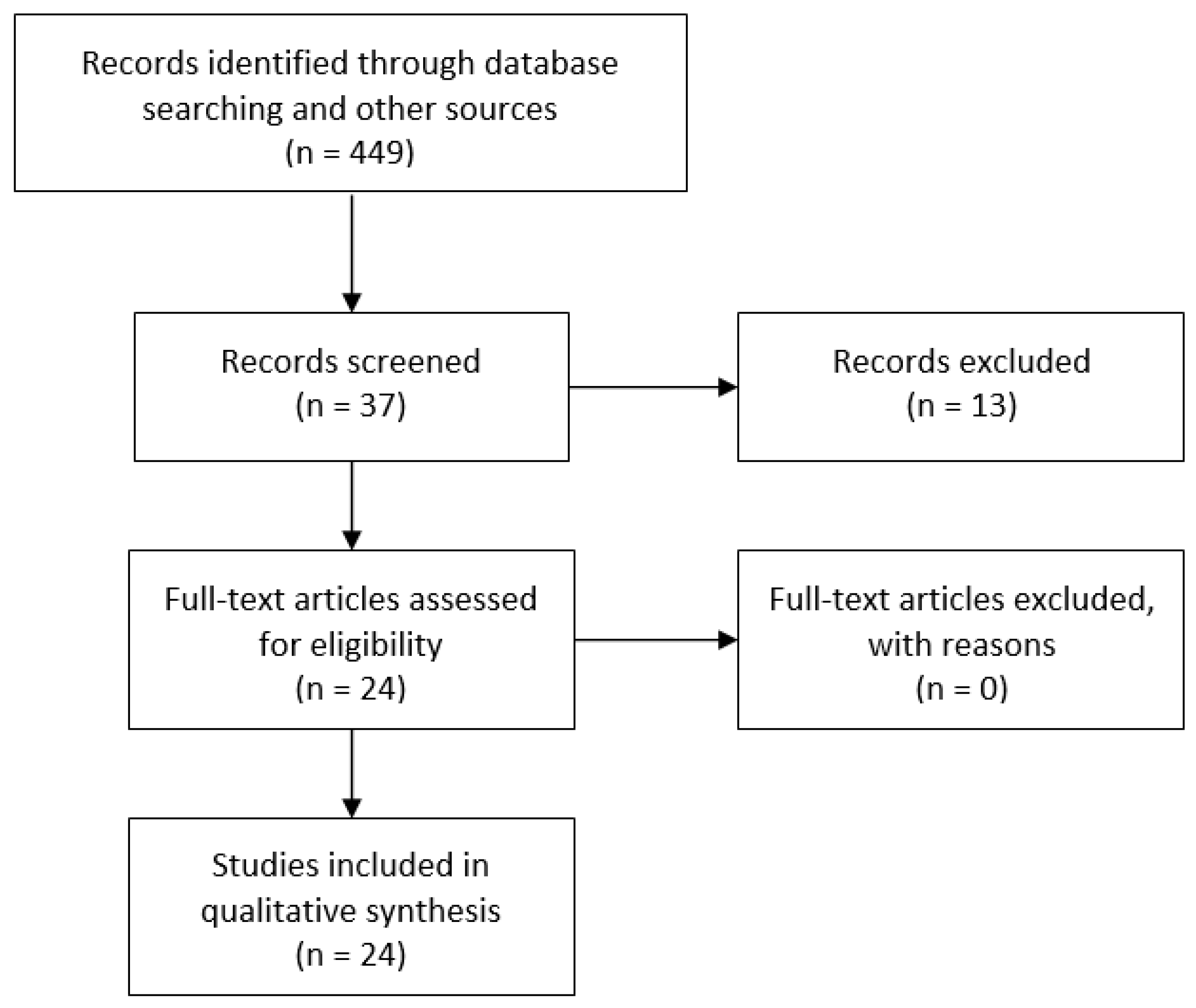
2.4. Selection of Studies
3. Results
3.1. CoQ10 and Male Fertility: Monotherapy
3.2. Compound Combinations and Male Infertility
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. WHO Laboratory Manual for the Examination and Processing of Human Semen; World Health Organization: Geneva, Switzerland, 2010; ISBN 9789241547789. [Google Scholar]
- Santi, D.; De Vincentis, S.; Alfano, P.; Balercia, G.; Calogero, A.E.; Cargnelutti, F.; Coccia, M.E.; Condorelli, R.A.; Dal Lago, A.; de Angelis, C.; et al. Use of follicle-stimulating hormone for the male partner of idiopathic infertile couples in Italy: Results from a multicentre, observational, clinical practice survey. Andrology 2020, 8, 637–644. [Google Scholar] [CrossRef]
- Agarwal, A.; Parekh, N.; Panner Selvam, M.K.; Henkel, R.; Shah, R.; Homa, S.T.; Ramasamy, R.; Ko, E.; Tremellen, K.; Esteves, S.; et al. Male Oxidative Stress Infertility (MOSI): Proposed Terminology and Clinical Practice Guidelines for Management of Idiopathic Male Infertility. World J. Mens. Health 2019, 37, 296–312. [Google Scholar] [CrossRef]
- Bui, A.D.; Sharma, R.; Henkel, R.; Agarwal, A. Reactive oxygen species impact on sperm DNA and its role in male infertility. Andrologia 2018, 50, 1–10. [Google Scholar] [CrossRef]
- Drevet, J.R. The antioxidant glutathione peroxidase family and spermatozoa: A complex story. Mol. Cell. Endocrinol. 2006, 16, 70–79. [Google Scholar] [CrossRef]
- Balercia, G.; Mancini, A.; Paggi, F.; Tiano, L.; Pontecorvi, A.; Boscaro, M.; Lenzi, A.; Littarru, G.P. Coenzyme Q10 and male infertility. J. Endocrinol. Investig. 2009, 32, 626–632. [Google Scholar] [CrossRef]
- Buhling, K.; Schumacher, A.; Eulenburg, C.; Zu Laakmann, E. Influence of oral vitamin and mineral supplementation on male infertility: A meta-analysis and systematic review. Reprod. Biomed. Online 2019, 39, 269–279. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. J. Clin. Epidemiol. 2009, 62, e1–e34. [Google Scholar] [CrossRef]
- Cakiroglu, B.; Eyyupoglu, S.E.; Gozukucuk, R.; Uyanik, B.S. Ubiquinol effect on sperm parameters in subfertile men who have astheno-teratozoospermia with normal sperm concentration. Nephrourol. Mon. 2014, 6, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Busetto, G.M.; Koverech, A.; Messano, M.; Antonini, G.; De Berardinis, E.; Gentile, V. Prospective open-label study on the efficacy and tolerability of a combination of nutritional supplements in primary infertile patients with idiopathic astenoteratozoospermia. Arch. Ital. Urol. E Androl. 2012, 84, 137–140. [Google Scholar]
- Abad, C.; Amengual, M.J.; Gosálvez, J.; Coward, K.; Hannaoui, N.; Benet, J.; García-Peiró, A.; Prats, J. Effects of oral antioxidant treatment upon the dynamics of human sperm DNA fragmentation and subpopulations of sperm with highly degraded DNA. Andrologia 2013, 45, 211–216. [Google Scholar] [CrossRef]
- Stenqvist, A.; Oleszczuk, K.; Leijonhufvud, I.; Giwercman, A. Impact of antioxidant treatment on DNA fragmentation index: A double-blind placebo-controlled randomized trial. Andrology 2018, 6, 811–816. [Google Scholar] [CrossRef]
- Alahmar, A.T. The impact of two doses of coenzyme Q10 on semen parameters and antioxidant status in men with idiopathic oligoasthenoteratozoospermia. Clin. Exp. Reprod. Med. 2019, 46, 112–118. [Google Scholar] [CrossRef]
- Balercia, G.; Mosca, F.; Mantero, F.; Boscaro, M.; Mancini, A.; Ricciardo-Lamonica, G.; Littarru, G.P. Coenzyme Q10 supplementation in infertile men with idiopathic asthenozoospermia: An open, uncontrolled pilot study. Fertil. Steril. 2004, 81, 93–98. [Google Scholar] [CrossRef]
- Balercia, G.; Buldreghini, E.; Vignini, A.; Tiano, L.; Paggi, F.; Amoroso, S.; Ricciardo-Lamonica, G.; Boscaro, M.; Lenzi, A.; Littarru, G.P. Coenzyme Q10 treatment in infertile men with idiopathic asthenozoospermia: A placebo-controlled, double-blind randomized trial. Fertil. Steril. 2009, 91, 1785–1792. [Google Scholar] [CrossRef]
- Safarinejad, M.R. Efficacy of Coenzyme Q10 on Semen Parameters, Sperm Function and Reproductive Hormones in Infertile Men. J. Urol. 2009, 182, 237–248. [Google Scholar] [CrossRef]
- Nadjarzadeh, A.; Sadeghi, M.R.; Amirjannati, N.; Vafa, M.R.; Motevalian, S.A.; Gohari, M.R.; Akhondi, M.A.; Yavari, P.; Shidfar, F. Coenzyme Q 10 improves seminal oxidative defense but does not affect on semen parameters in idiopathic oligoasthenoteratozoospermia: A randomized double-blind, placebo controlled trial. J. Endocrinol. Investig. 2011, 34, 224–228. [Google Scholar]
- Safarinejad, M.R.; Safarinejad, S.; Shafiei, N.; Safarinejad, S. Effects of the reduced form of coenzyme Q10 (ubiquinol) on semen parameters in men with idiopathic infertility: A double-blind, placebo controlled, randomized study. J. Urol. 2012, 188, 526–531. [Google Scholar] [CrossRef]
- Safarinejad, M.R. The effect of coenzyme Q 10 supplementation on partner pregnancy rate in infertile men with idiopathic oligoasthenoteratozoospermia: An open-label prospective study. Int. Urol. Nephrol. 2012, 44, 689–700. [Google Scholar] [CrossRef]
- Festa, R.; Giacchi, E.; Raimondo, S.; Tiano, L.; Zuccarelli, P.; Silvestrini, A.; Meucci, E.; Littarru, G.P.; Mancini, A. Coenzyme Q10 supplementation in infertile men with low-grade varicocele: An open, uncontrolled pilot study. Andrologia 2014, 46, 805–807. [Google Scholar] [CrossRef]
- Nadjarzadeh, A.; Shidfar, F.; Amirjannati, N.; Vafa, M.R.; Motevalian, S.A.; Gohari, M.R.; Nazeri Kakhki, S.A.; Akhondi, M.M.; Sadeghi, M.R. Effect of Coenzyme Q10 supplementation on antioxidant enzymes activity and oxidative stress of seminal plasma: A double-blind randomised clinical trial. Andrologia 2014, 46, 177–183. [Google Scholar] [CrossRef]
- Thakur, A.S.; Littattarru, G.P.; Funahashi, I.; Painkara, U.S.; Dange, N.S.; Chauhan, P. Effect of ubiquinol therapy on sperm parameters and serum testosterone levels in oligoasthenozoospermic infertile men. J. Clin. Diagn. Res. 2015, 9, 11–13. [Google Scholar] [CrossRef]
- Alahmar, A.T.; Sengupta, P. Impact of Coenzyme Q10 and Selenium on Seminal Fluid Parameters and Antioxidant Status in Men with Idiopathic Infertility. Biol. Trace Elem. Res. 2021, 199, 1246–1252. [Google Scholar] [CrossRef]
- Kobori, Y.; Ota, S.; Sato, R.; Yagi, H.; Soh, S.; Arai, G.; Okada, H. Antioxidant cosupplementation therapy with vitamin C, vitamin E, and coenzyme Q10 in patients with oligoasthenozoospermia. Arch. Ital. Urol. E Androl. 2014, 86, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Tirabassi, G.; Vignini, A.; Tiano, L.; Buldreghini, E.; Brugè, F.; Silvestri, S.; Orlando, P.; D’Aniello, A.; Mazzanti, L.; Lenzi, A.; et al. Protective effects of coenzyme Q10 and aspartic acid on oxidative stress and DNA damage in subjects affected by idiopathic asthenozoospermia. Endocrine 2015, 49, 549–552. [Google Scholar] [CrossRef]
- Gvozdjáková, A.; Kucharská, J.; Dubravicky, J.; Mojto, V.; Singh, R.B. Coenzyme Q10, α-tocopherol, and oxidative stress could be important metabolic biomarkers of male infertility. Dis. Markers 2015, 2015. [Google Scholar] [CrossRef]
- Lipovac, M.; Bodner, F.; Imhof, M.; Chedraui, P. Comparison of the effect of a combination of eight micronutrients versus a standard mono preparation on sperm parameters. Reprod. Biol. Endocrinol. 2016, 14, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Terai, K.; Horie, S.; Fukuhara, S.; Miyagawa, Y.; Kobayashi, K.; Tsujimura, A. Combination therapy with antioxidants improves total motile sperm counts: A Preliminary Study. Reprod. Med. Biol. 2020, 19, 89–94. [Google Scholar] [CrossRef]
- Arafa, M.; Agarwal, A.; Majzoub, A.; Selvam, M.K.P.; Baskaran, S.; Henkel, R.; Elbardisi, H. Efficacy of antioxidant supplementation on conventional and advanced sperm function tests in patients with idiopathic male infertility. Antioxidants 2020, 9, 219. [Google Scholar] [CrossRef]
- Sadaghiani, S.; Fallahi, S.; Heshmati, H.; Hosseini Teshnizi, S.; Abedini Chaijan, H.; Farzad Amir Ebrahimi, F.; Khorrami, F.; Poorrezaeian, M.; Alizadeh, F. Effect of antioxidant supplements on sperm parameters in infertile male smokers: A single-blinded clinical trial. AIMS Public Health 2020, 7, 92–99. [Google Scholar] [CrossRef]
- Kopets, R.; Kuibida, I.; Chernyavska, I.; Cherepanyn, V.; Mazo, R.; Fedevych, V.; Gerasymov, S. Dietary supplementation with a novel l-carnitine multi-micronutrient in idiopathic male subfertility involving oligo-, astheno-, teratozoospermia: A randomized clinical study. Andrology 2020, 8, 1184–1193. [Google Scholar] [CrossRef]
- Nazari, L.; Salehpour, S.; Hosseini, S.; Allameh, F.; Jahanmardi, F.; Azizi, E.; Ghodssi-Ghassemabadi, R.; Hashemi, T. Effect of antioxidant supplementation containing l-carnitine on semen parameters: A prospective interventional study. J. Bras. Reprod. Assist. 2021, 25, 76–80. [Google Scholar] [CrossRef]
- Lei, L.; Liu, Y. Efficacy of coenzyme Q10 in patients with cardiac failure: A meta-analysis of clinical trials. BMC Cardiovasc. Disord. 2017, 17, 196. [Google Scholar] [CrossRef] [PubMed]
- Jorat, M.V.; Tabrizi, R.; Mirhosseini, N.; Lankarani, K.B.; Akbari, M.; Heydari, S.T.; Mottaghi, R.; Asemi, Z. The effects of coenzyme Q10 supplementation on lipid profiles among patients with coronary artery disease: A systematic review and meta-analysis of randomized controlled trials. Lipids Health Dis. 2018, 17, 230. [Google Scholar] [CrossRef]
- Zhang, S.Y.; Yang, K.L.; Zeng, L.T.; Wu, X.H.; Huang, H.Y. Effectiveness of coenzyme Q10 supplementation for Type 2 diabetes mellitus: A systematic review and meta-analysis. Int. J. Endocrinol. 2018, 2018, 6484839. [Google Scholar] [CrossRef] [PubMed]
- Zhai, J.; Bo, Y.; Lu, Y.; Liu, C.; Zhang, L. Effects of coenzyme Q10 on markers of inflammation: A systematic review and meta-analysis. PLoS ONE 2017, 12, e0170172. [Google Scholar] [CrossRef]
- Lafuente, R.; González-Comadrán, M.; Solà, I.; López, G.; Brassesco, M.; Carreras, R.; Checa, M.A. Coenzyme Q10 and male infertility: A meta-analysis. J. Assist. Reprod. Genet. 2013, 30, 1147–1156. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhu, L.; Jiang, H.; Chen, H.; Chen, Y.; Dai, Y. Sperm DNA fragmentation index and pregnancy outcome after IVF or ICSI: A meta-analysis. J. Assist. Reprod. Genet. 2015, 32, 17–26. [Google Scholar] [CrossRef]
- Meachem, S.J.; Nieschlag, E.; Simoni, M. Inhibin B in male reproduction: Pathophysiology and clinical relevance. Eur. J. Endocrinol. 2001, 145, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Ilacqua, A.; Izzo, G.; Emerenziani, G.P.; Baldari, C.; Aversa, A. Lifestyle and fertility: The influence of stress and quality of life on male fertility. Reprod. Biol. Endocrinol. 2018, 16, 115. [Google Scholar] [CrossRef]
| Author(s) | Design | Sample Size | Age | Target Population | Daily Dosage | Consumption | Length of Follow-up | Results |
|---|---|---|---|---|---|---|---|---|
| Balercia et al., 2004 [14] | Open, uncontrolled | 22 | 31 (25–39) | Idiopathic asthenozoospermia | CoQ10 200 mg/day | Twice daily orally | 6 months | Imrprovement in the forward (a + b) motility of sperm cells (p < 0.05), improvement in computer-assisted determination of kinetic parameters (p < 0.05) and increase in seminal coQ10 levels (p < 0.05) after treatment. |
| Balercia et al., 2009 [15] | Randomized, double-blind, placebo-controlled | 60 | 32 (27–39) | Idiopathic asthenozoospermia | CoQ10 200 mg/day | Twice daily orally | 6 months | Improvement in the forward (p < 0.0001) and total (p < 0.0003) motility of sperm cells, improvement in computer-assisted determination of kinetic parameters and increase in seminal coQ10 and ubiquinol levels (p < 0.0001) after treatment. Lower baseline value of motility and levels of coQ10 had higher probability to be responders to the treatment. |
| Safarinejad, 2009 [16] | Randomized, double-blind, placebo-controlled | 212 | 28 (21–42) | iOAT | CoQ10 300 mg/day | Once daily orally | 26 weeks | Improvement in sperm density and motility (p = 0.01) after coQ10 treatment. Positive correlation between treatment duration and sperm count (p = 0.01), sperm motility (p = 0.01) and sperm morphology (p = 0.02). Decrease in FSH and LH levels (p = 0.03) and increase in inhibin levels and acrosome reaction (p = 0.03) after coQ10 treatment. |
| Nadjarzadeh et al., 2011 [17] | Randomized, double-blind, placebo-controlled | 60 | 34 (25–46) | iOAT | CoQ10 200 mg/day | - | 3 months | Non-significant changes in semen parameters of CoQ10 group. Increase in total antioxidant capacity of seminal plasma (p = 0.017) and significant reduction in concentration of thiobarbituric acid-reactive substances (p = 0.013) in the CoQ10 group. |
| Safarinejad et al., 2012 [18] | Randomized, double-blind, placebo-controlled | 228 | 25–44 | iOAT | Ubiquinol 200 mg/day | Once daily orally | 26 weeks | Improvement in sperm density (p = 0.005), motility (p = 0.008) and morphology (p = 0.01) after ubiquinol treatment. Positive correlation between treatment duration, seminal plasma antioxidant capacity and semen parameters. Decrease in FSH (p = 0.02) and LH levels (p = 0.03) and increase in inhibin levels (p = 0.01) after ubiquinol treatment. |
| Safarinejad et al., 2012 [19] | Open, uncontrolled | 287 | 32 (26–43) | iOAT | CoQ10 300 mg/day | Twice daily orally | 12 months | Improvement in sperm density, motility and morphology after CoQ10 treatment (p < 0.05). Decrease in FSH and LH levels and increase in inhibin levels after CoQ10 treatment (p < 0.05). Improvements remained significant 12 months after CoQ10 discontinuation. Beneficial effect on pregnancy rate. |
| Festa et al., 2014 [20] | Open, uncontrolled | 38 | 19–40 | Varicocele-related infertility | CoQ10 100 mg/day | Twice daily orally | 3 months | Increase in sperm density (p = 0.03), forward motility (p = 0.03) and seminal plasma total antioxidant capacity (p < 0.01). |
| Nadjarzadeh et al., 2014 [21] | Randomized, double-blind, placebo-controlled | 60 | 34 (25–40) | iOAT | CoQ10 200 mg/day | Twice daily orally | 3 months | Increase in forward and total motility (p < 0.05) in the treatment group. Increase in seminal CoQ10 (p = 0.0001) and positive correlation with normal sperm morphology (p = 0.037) and CAT (p < 0.05) and SOD activity (p < 0.05) after treatment. Higher catalase and SOD activity and lower seminal 8-isoprostane concentration (p < 0.05) in the CoQ10 group after treatment. |
| Cakiroglu et al., 2014 [9] | Retrospective | 62 | 32 (23–50) | Normo- or mild oligospermic patients with asthenoteratozoospermia | Ubiquinol 200 mg/day | Twice daily orally | 6 months | Increase in normal morphology and sperm motility (a + b) (p < 0.001) after ubiquinol supplementation. |
| Thakur et al., 2015 [22] | Open, uncontrolled | 60 | 20-40 | Oligospermia | Ubiquinol 150 mg/day | Once daily orally | 6 months | Increase in total sperm count and total sperm motility (p < 0.05) and reduction of number of immobile and sluggish motile sperm (p < 0.05). |
| Alahmar, 2019 [13] | Prospective, randomized | 65 | 27 | iOAT | CoQ10 200 mg/day versus CoQ10 400 mg/day | Once daily orally | 3 months | Increase in seminal parameters (sperm concentration, sperm motility and sperm morphology) and seminal antioxidant activity in both groups after treatment (p < 0.05). Higher increase in kinetic parameters in subjects treated with 400 mg CoQ10. Correlation between sperm parameters and seminal antioxidant activity after treatment (p < 0.05). |
| Alahmar et al., 2021 [23] | Prospective, randomized | 70 | 25 | iOAT | CoQ10 200 mg/day Versus Selenium 200 mcg/day | Once daily orally | 3 months | Increase in sperm density, total sperm motility and progressive sperm motility (p < 0.01) and improvement of antioxidant capacity of seminal fluid (p < 0.05) after CoQ10 administration. |
| Author(s) | Design | Sample Size | Age | Target Population | Antioxidant | Consumption | Length of Follow-up | Results |
|---|---|---|---|---|---|---|---|---|
| Busetto et al., 2012 [10] | Open, uncontrolled | 114 | 31 (21–46) | iOAT | CoQ10 20 mg L-carnitine 145 mg Acetyl-L-carnitine 64 mg Fructose 250 mg Citric acid 50 mg Selenium 50 mcg Zinc 10 mg Ascorbic acid 90 mg Cyanocobalamin 1.5 mcg Folic acid 200 mcg | Once daily orally | 4 months | Increase in progressive sperm motility (p < 0.05). |
| Abad et al., 2013 [11] | Open, uncontrolled | 20 | - | Asthenoteratozoospermia | CoQ10 20 mg L-carinitine 1500 mg vitamin C 60 mg vitamin E 10 mg vitamin B9 200 mcg vitamin B12 1 mcg Zinc 10 mg selenium 50 mcg | - | 3 months | Slight increase in sperm density (p = 0.042) and normal morphology (p = 0.04), pronounced increase in A motility, A + B motility and vitality (p < 0.05) after antioxidant treatment. Improvement in DNA integrity and reduction in proportion of highly DNA degraded sperm (p = 0.04). |
| Kobori et al., 2014 [24] | Open, uncontrolled | 169 | 36 (25–58) | iOAT | CoQ10 120 mg Vitamin C 80 mg Vitamin E 40 mg | Twice daily orally | 6 months | Increase in sperm concentration and sperm motility at 3 and 6 months of treatment (p < 0.05). |
| Tirabassi et al., 2015 [25] | Open, uncontrolled | 20 | 32 | Idiopathic asthenozoospermia | CoQ10 200 mg Aspartic acid 2660 mg | Once daily orally | 3 months | Q10 and aspartic acid administration improved sperm kinetics, antioxidant defenses (SOD activity) and reduced nitric oxide-related oxidant species and oxidative DNA damage (p < 0.05). |
| Gvozdjáková et al., 2015 [26] | Open, uncontrolled | 40 | 28–40 | Oligoasthenoteratozoospermia | Ubiquinol 30 mg L-carnitine 440 mg Vitamin E 75 IU Vitamin C 12 mg | Twice daily during the first 3 months; once daily during the next 3 months | 6 months | Increase in sperm density (p < 0.001). Increase in seminal CoQ10 and α-tocopherol levels and decrease in oxidative stress markers (p < 0.05). |
| Lipovac et al., 2016 [27] | Open, uncontrolled | 299 | 20–60 | Infertile men with al least one pathologic sperm analysis | Carnitine 1000 mg/day versus CoQ10 15 mg Carnitine 440 mg Arginine 250 mg Zinc 40 mg Vitamin E 120 mg Glutathione 80 mg Selenium 60 mcg | Twice daily (mono-substance) versus once daily (combination) | 3 months | Improvement of all sperm parameters in both groups (p < 0.05), but higher relative changes in sperm density and progressive motility for the combined micronutrient treatment group. |
| Stenqvist et al., 2018 [12] | Randomized, double-blind, placebo-controlled | 77 | 38 | Infertile men with elevated DNA fragmentation index | CoQ10 10 mg Folic acid 100 mcg Vitamin C 30 mg Vitamin E 5 mg Vitamin B12 0.5 mcg Carnitine 750 mg Zinc 5 mg Selenium 25 mcg | Twice daily orally | 6 months | Higher sperm density compared to baseline in the treatment group after antioxidant supplementation (p = 0.028). No differences in DNA fragmentation index in any group and between groups after treatment. |
| Terai et al. 2020 [28] | Prospective, randomized | 31 | 38 | Oligozoospermia and/or asthenozoospermia | CoQ10 90 mg L-Carnitine 750 mg Zinc 30 mg Astaxanthin16 mg Vitamin C 1000 mg | Three times per day | 3 months | Increase in total sperm count in the supplement group after treatment (p = 0.04). |
| Arafa et al., 2020 [29] | Open, uncontrolled | 148 | 36 (31–41) | Idiopathic male infertility and unexplained male infertility | Coq10 200 mg Vitamin A 5000 IU Vitamin C 120 mg Vitamin D3: 1200 IU Vitamin E 200 IU Vitamin K 80 µg Thiamin 3 mg Riboflavin 3.4 mg Niacin 20 mg Vitamin B6 25 mg Folate 800 µg Vitamin B12 1000 µg Biotin 600 µg Zinc 30 mg Selenium 140 µg Copper 1 mg Manganese 2 mg Chromium 120 µg L-carnitine tartrate 2000 mg L-arginine 350 mg N-acetyl l-cysteine 200 mg Lycopene 10 mg Benfotiamine 1 mg | Three capsules twice a day orally | 3 months | In the idiopathic infertility group, supplementation showed a significant decrease in seminal ORP and SDF levels (p < 0.05). Sperm analysis revealed an improvement in all parameters investigated (p < 0.05), except for semen volume and sperm viability. Changes were also more evident in both the sub-categories of high ORP and high SDF. ORP levels significantly decreased in all the subjects with idiopathic infertility and in both the sub-categories of high ORP and high SDF levels (p < 0.05). In the unexplained male infertility group, only progressive motility significantly improved after treatment (p < 0.05). ORP levels decreased after treatment (p < 0.05). |
| Sadaghiani et al., 2020 [30] | Prospective, single-blinded | 50 | 32 | Astheno/oligozoospermia and cigarette smoking | CoQ10 30 mg * Zinc 8 mg * Vitamin C 100 mg * Vitamin E 12 mg * Folic acid 400 μg * Selenium 200 mg ** | * Once daily ** Every other day | 3 months | Increase in seminal volume (p = 0.032), sperm count (p = 0.001), sperm motility (total and progressive) (p = 0.001) and normal morphology (p = 0.003) after treatment. |
| Kopets et al., 2020 [31] | Randomized, double-blind, placebo-controlled | 83 | 33 | Idiopathic male infertility | Co Q10 40 Mg L-Carnitine/L-Acetyl-Carnitine 1990 Mg, L-Arginine 250 Mg Glutathione 100 Mg Zinc 7,5 Mg Vitamin B9 234 Mcg Vitamin B12 2 Mcg Selenium 50 Mcg | Once daily orally | 6 months | Improvement in sperm parameters (sperm density, sperm motility and sperm morphology) (p < 0.001) and increase in pregnancy rate at 4 months (p = 0.017) in the treatment group. |
| Nazari et al., 2021 [32] | Open, uncontrolled | 180 | 36 (26–40) | iOAT | Coq10 20 Mg L-Carnitine 1500 Mg Vitamin C 60 Mg Vitamin E 10 Mg Zinc 10 Mg Vitamin B9 200 µg Selenium 50 µg Vitamin B12 1 µg | Twice daily orally | 3 months | Increase in sperm density (p = 0.004) and morphology (p = 0.01), but not in sperm motility |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salvio, G.; Cutini, M.; Ciarloni, A.; Giovannini, L.; Perrone, M.; Balercia, G. Coenzyme Q10 and Male Infertility: A Systematic Review. Antioxidants 2021, 10, 874. https://doi.org/10.3390/antiox10060874
Salvio G, Cutini M, Ciarloni A, Giovannini L, Perrone M, Balercia G. Coenzyme Q10 and Male Infertility: A Systematic Review. Antioxidants. 2021; 10(6):874. https://doi.org/10.3390/antiox10060874
Chicago/Turabian StyleSalvio, Gianmaria, Melissa Cutini, Alessandro Ciarloni, Lara Giovannini, Michele Perrone, and Giancarlo Balercia. 2021. "Coenzyme Q10 and Male Infertility: A Systematic Review" Antioxidants 10, no. 6: 874. https://doi.org/10.3390/antiox10060874
APA StyleSalvio, G., Cutini, M., Ciarloni, A., Giovannini, L., Perrone, M., & Balercia, G. (2021). Coenzyme Q10 and Male Infertility: A Systematic Review. Antioxidants, 10(6), 874. https://doi.org/10.3390/antiox10060874







