Re-Evaluating the Oxidative Phenotype: Can Endurance Exercise Save the Western World?
Abstract
1. Introduction
2. Rate of ATP Hydrolysis at Myofilaments Creates Demand
3. Oxidative Phosphorylation Is Supported by Reducing Agents Made in the TCA Cycle
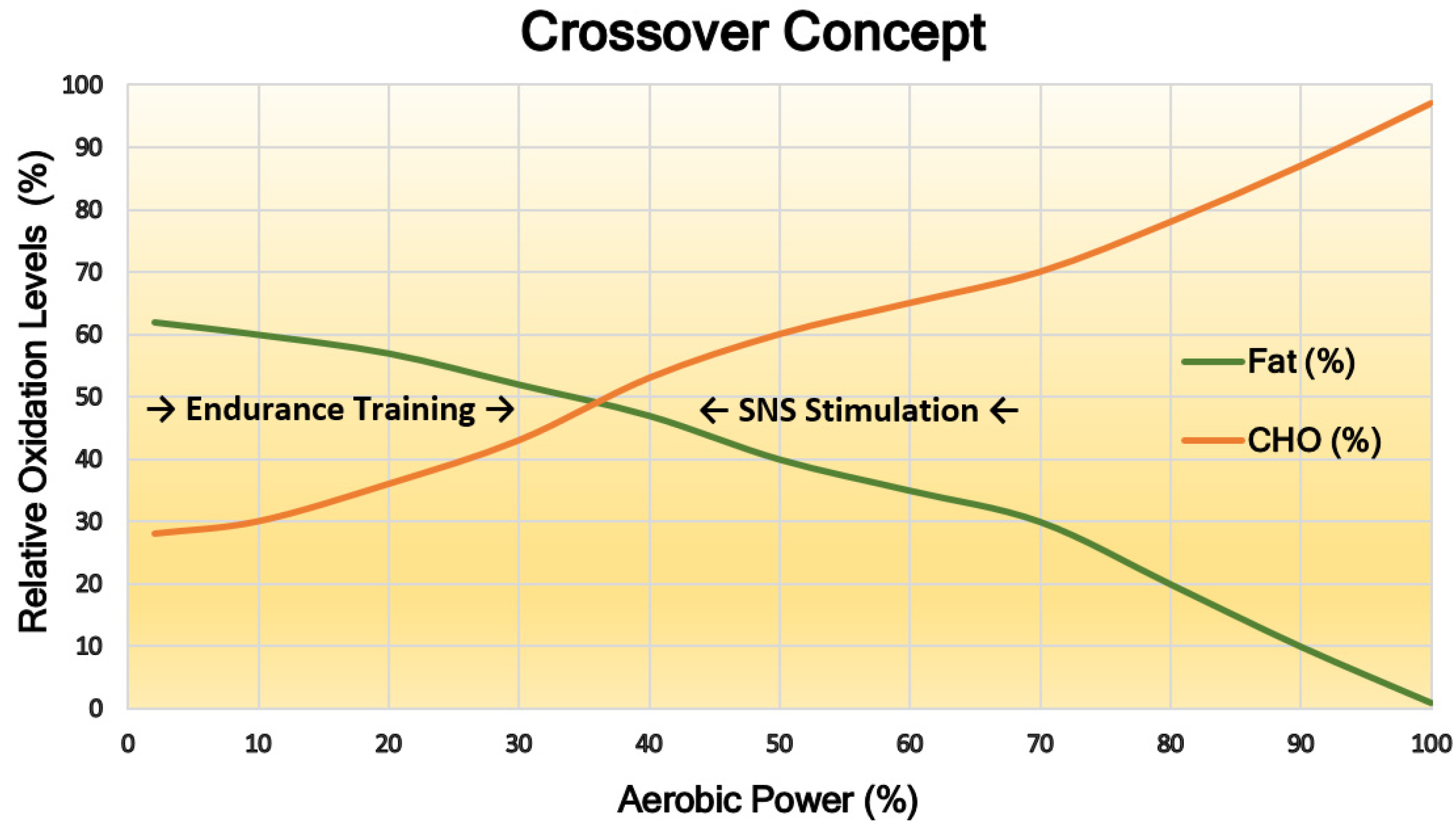
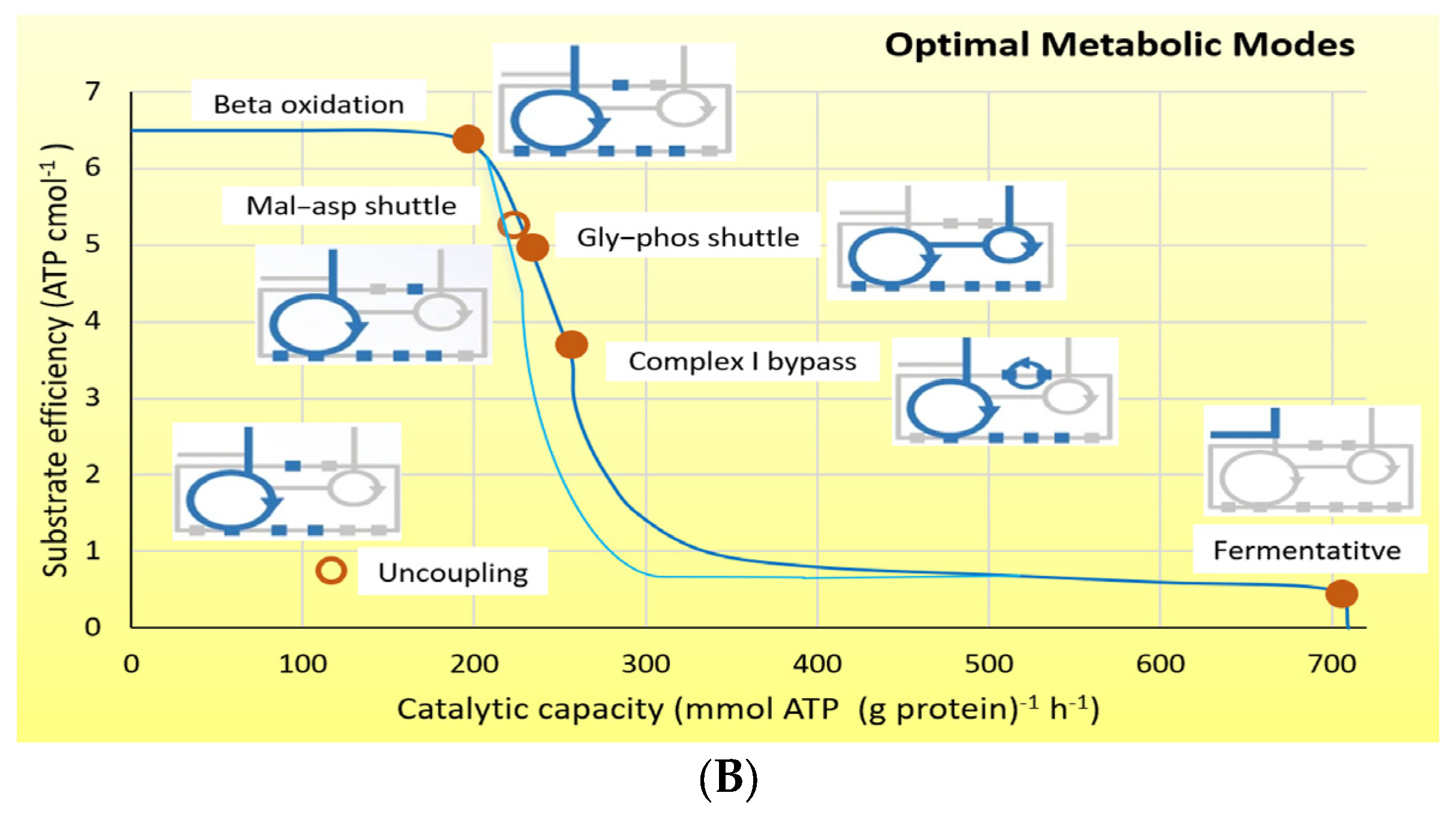
4. Energy Demand Regulates Substrate Preference—Fat Is “Cheap”
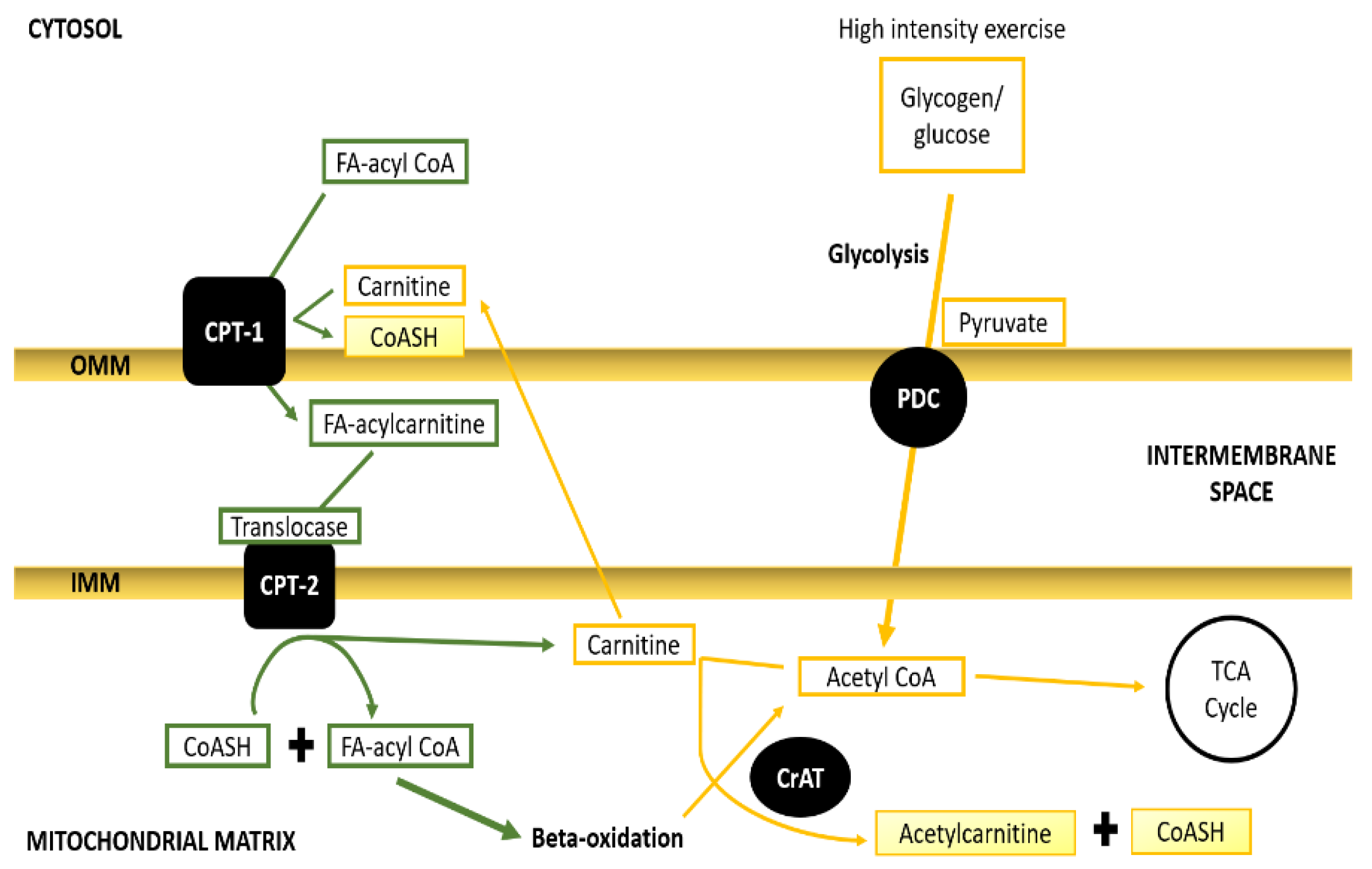
5. Balancing between Fatty Acids and Pyruvate to Meet the Contractile Demand
6. Carnitine Acetyltransferase Mitigates Metabolic Inertia—A Trait of Oxidative Flexibility
7. Lactate Supports the Glycolytic Flux through Oxidation in Mitochondria
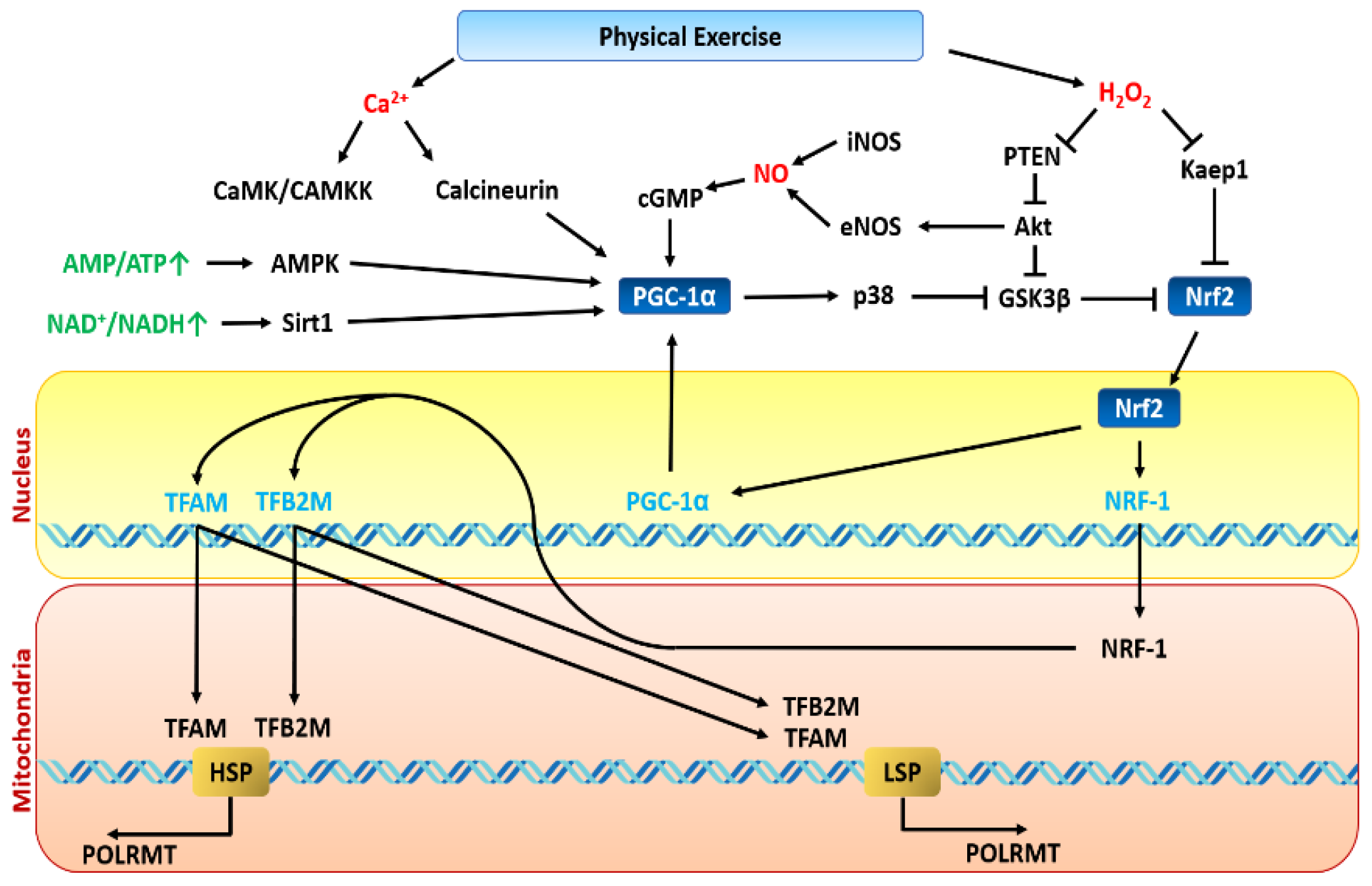
8. Lactate Sets the Stage for PGC-1α Signalling—The Oxidative Booster
9. Reactive Oxygen Species—A Thin Line between Fit and Failed
10. The Higher the Intensity, the Larger the Dyshomeostasis, the Greater the Adaptation?
11. Hypoxia—Friend or Foe?
12. The Balance between Regulated Ageing and Immortal Tumorigenesis—Losing the Oxidative Identity
13. Summary
14. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hill, A.; Long, C.; Lupton, H. Muscular exercise, lactic acid, and the supply and utilisation of oxygen. Parts I–III. Proc. R. Soc. Lond. Ser. B Contain. Pap. A Biol. Character 1924, 96, 438–475. [Google Scholar]
- Taylor, H.; Buskirk, E.; Henschel, A. Maximal Oxygen Intake as an Objective Measure of Cardio-Respiratory Performance. J. Appl. Physiol. 1955, 8, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Astrand, P.; Saltin, B. Maximal oxygen uptake and heart rate in various types of muscular activity. J. Appl. Physiol. 1961, 16, 977–981. [Google Scholar] [PubMed]
- Owles, W. Alterations in the lactic acid content of the blood as a result of light exercise, and associated changes in the co2-combining power of the blood and in the alveolar co2pressure. J. Physiol. 1930, 69, 214–237. [Google Scholar] [CrossRef] [PubMed]
- Wasserman, K.; McIlroy, M. Detecting the threshold of anaerobic metabolism in cardiac patients during exercise. Am. J. Cardiol. 1964, 14, 844–852. [Google Scholar] [CrossRef]
- Sjödin, B.; Jacobs, I. Onset of Blood Lactate Accumulation and Marathon Running Performance. Int. J. Sports Med. 1981, 2, 23–26. [Google Scholar] [CrossRef]
- Billat, V.; Sirvent, P.; Py, G.; Koralsztein, J.; Mercier, J. The Concept of Maximal Lactate Steady State. Sports Med. 2003, 33, 407–426. [Google Scholar] [CrossRef]
- Romijn, J.; Coyle, E.; Sidossis, L.; Gastaldelli, A.; Horowitz, J.; Endert, E.; Wolfe, R. Regulation of endogenous fat and carbohydrate metabolism in relation to exercise intensity and duration. Am. J. Physiol. Endocrinol. Metab. 1993, 265, E380–E391. [Google Scholar] [CrossRef]
- Achten, J.; Jeukendrup, A. Maximal Fat Oxidation during Exercise in Trained Men. Int. J. Sports Med. 2003, 24, 603–608. [Google Scholar] [PubMed]
- Preece, S.; Bramah, C.; Mason, D. The biomechanical characteristics of high-performance endurance running. Eur. J. Sport Sci. 2018, 19, 784–792. [Google Scholar] [CrossRef]
- Barnes, K.; Kilding, A. Running economy: Measurement, norms, and determining factors. Sports Med. Open 2015, 1, 1–15. [Google Scholar] [CrossRef]
- Whipp, B.; Seard, C.; Wasserman, K. Oxygen deficit-oxygen debt relationships and efficiency of anaerobic work. J. Appl. Physiol. 1970, 28, 452–456. [Google Scholar] [CrossRef]
- Wallimann, T.; Wyss, M.; Brdiczka, D.; Nicolay, K.; Eppenberger, H. Intracellular compartmentation, structure and function of creatine kinase isoenzymes in tissues with high and fluctuating energy demands: The ‘phosphocreatine circuit’ for cellular energy homeostasis. Biochem. J. 1992, 281, 21–40. [Google Scholar] [CrossRef]
- Korzeniewski, B.; Zoladz, J. Some factors determining the PCr recovery overshoot in skeletal muscle. Biophys. Chem. 2005, 116, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Brooks, G. Anaerobic threshold. Med. Sci. Sports Exerc. 1985, 17, 22–31. [Google Scholar] [CrossRef]
- Poole, D.; Jones, A. Oxygen Uptake Kinetics. Compr. Physiol. 2012, 2, 933–996. [Google Scholar] [PubMed]
- Huxley, H. Fifty years of muscle and the sliding filament hypothesis. Eur. J. Biochem. 2004, 271, 1403–1415. [Google Scholar] [CrossRef] [PubMed]
- Boron, W.; Boulpaep, E. Medical Physiology, 3rd ed.; Elsevier: Philadelphia, PA, USA, 2017; pp. 228–242. [Google Scholar]
- He, Z.; Bottinelli, R.; Pellegrino, M.; Ferenczi, M.; Reggiani, C. ATP Consumption and Efficiency of Human Single Muscle Fibers with Different Myosin Isoform Composition. Biophys. J. 2000, 79, 945–961. [Google Scholar] [CrossRef]
- Taylor, D.; Styles, P.; Matthews, P.; Arnold, D.; Gadian, D.; Bore, P.; Radda, G. Energetics of human muscle: Exercise-induced ATP depletion. Magn. Reson. Med. 1986, 3, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Brown, R.; Park, C.; Cohn, M.; Chance, B. Energy metabolism of the untrained muscle of elite runners as observed by 31P magnetic resonance spectroscopy: Evidence suggesting a genetic endowment for endurance exercise. Proc. Natl. Acad. Sci. USA 1988, 85, 8780–8784. [Google Scholar] [CrossRef]
- Purdom, T.; Kravitz, L.; Dokladny, K.; Mermier, C. Understanding the factors that effect maximal fat oxidation. J. Int. Soc. Sports Nutr. 2018, 15, 1–10. [Google Scholar] [CrossRef]
- Spriet, L. New Insights into the Interaction of Carbohydrate and Fat Metabolism during Exercise. Sports Med. 2014, 44, 87–96. [Google Scholar] [CrossRef]
- Goodpaster, B.; Sparks, L. Metabolic Flexibility in Health and Disease. Cell Metab. 2017, 25, 1027–1036. [Google Scholar] [CrossRef] [PubMed]
- Randle, P.; Newsholme, E.; Garland, P. Regulation of glucose uptake by muscle. 8. Effects of fatty acids, ketone bodies and pyruvate, and of alloxan-diabetes and starvation, on the uptake and metabolic fate of glucose in rat heart and diaphragm muscles. Biochem. J. 1964, 93, 652–665. [Google Scholar] [CrossRef]
- Brooks, G.; Mercier, J. Balance of carbohydrate and lipid utilization during exercise: The “crossover” concept. J. Appl. Physiol. 1994, 76, 2253–2261. [Google Scholar] [CrossRef]
- Nilsson, A.; Björnson, E.; Flockhart, M.; Larsen, F.; Nielsen, J. Complex I is bypassed during high intensity exercise. Nat. Commun. 2019, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Agüera, M.; Gao, L.; González-Rodríguez, P.; Pintado, C.; Arias-Mayenco, I.; García-Flores, P.; García-Pergañeda, A.; Pascual, A.; Ortega-Sáenz, P.; López-Barneo, J. Oxygen Sensing by Arterial Chemoreceptors Depends on Mitochondrial Complex I Signaling. Cell Metab. 2015, 22, 825–837. [Google Scholar] [CrossRef]
- Arias-Mayenco, I.; González-Rodríguez, P.; Torres-Torrelo, H.; Gao, L.; Fernández-Agüera, M.; Bonilla-Henao, V.; Ortega-Sáenz, P.; López-Barneo, J. Acute O2 Sensing: Role of Coenzyme QH2/Q Ratio and Mitochondrial ROS Compartmentalization. Cell Metab. 2018, 28, 145–158.e4. [Google Scholar] [CrossRef] [PubMed]
- Richardson, R.; Newcomer, S.; Noyszewski, E. Skeletal muscle intracellular Po 2assessed by myoglobin desaturation: Response to graded exercise. J. Appl. Physiol. 2001, 91, 2679–2685. [Google Scholar] [CrossRef] [PubMed]
- Ward, S. Exercise physiology: Exercise hyperpnea. Curr. Opin. Physiol. 2019, 10, 166–172. [Google Scholar] [CrossRef]
- Hureau, T.; Weavil, J.; Thurston, T.; Broxterman, R.; Nelson, A.; Bledsoe, A.; Jessop, J.; Richardson, R.; Wray, D.; Amann, M. Identifying the role of group III/IV muscle afferents in the carotid baroreflex control of mean arterial pressure and heart rate during exercise. J. Physiol. 2018, 596, 1373–1384. [Google Scholar] [CrossRef]
- Hureau, T.; Weavil, J.; Thurston, T.; Wan, H.; Gifford, J.; Jessop, J.; Buys, M.; Richardson, R.; Amann, M. Pharmacological attenuation of group III/IV muscle afferents improves endurance performance when oxygen delivery to locomotor muscles is preserved. J. Appl. Physiol. 2019, 127, 1257–1266. [Google Scholar] [CrossRef]
- Saito, M.; Mano, T.; Iwase, S.; Koga, K.; Abe, H.; Yamazaki, Y. Responses in muscle sympathetic activity to acute hypoxia in humans. J. Appl. Physiol. 1988, 65, 1548–1552. [Google Scholar] [CrossRef]
- Dehvari, N.; Hutchinson, D.; Nevzorova, J.; Dallner, O.; Sato, M.; Kocan, M.; Merlin, J.; Evans, B.; Summers, R.; Bengtsson, T. β2-Adrenoceptors increase translocation of GLUT4 via GPCR kinase sites in the receptor C-terminal tail. Br. J. Pharmacol. 2012, 165, 1442–1456. [Google Scholar] [CrossRef] [PubMed]
- van Loon, L. Use of intramuscular triacylglycerol as a substrate source during exercise in humans. J. Appl. Physiol. 2004, 97, 1170–1187. [Google Scholar] [CrossRef]
- Shaw, C.; Shepherd, S.; Wagenmakers, A.; Hansen, D.; Dendale, P.; van Loon, L. Prolonged exercise training increases intramuscular lipid content and perilipin 2 expression in type I muscle fibers of patients with type 2 diabetes. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E1158–E1165. [Google Scholar] [CrossRef] [PubMed]
- Goodpaster, B.; He, J.; Watkins, S.; Kelley, D. Skeletal Muscle Lipid Content and Insulin Resistance: Evidence for a Paradox in Endurance-Trained Athletes. J. Clin. Endocrinol. Metab. 2001, 86, 5755–5761. [Google Scholar] [CrossRef]
- Daemen, S.; Gemmink, A.; Brouwers, B.; Meex, R.; Huntjens, P.; Schaart, G.; Moonen-Kornips, E.; Jörgensen, J.; Hoeks, J.; Schrauwen, P.; et al. Distinct lipid droplet characteristics and distribution unmask the apparent contradiction of the athlete’s paradox. Mol. Metab. 2018, 17, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Koves, T.; Sparks, L.; Kovalik, J.; Mosedale, M.; Arumugam, R.; DeBalsi, K.; Everingham, K.; Thorne, L.; Phielix, E.; Meex, R.; et al. PPARγ coactivator-1α contributes to exercise-induced regulation of intramuscular lipid droplet programming in mice and humans. J. Lipid Res. 2012, 54, 522–534. [Google Scholar] [CrossRef]
- Lehman, J.; Barger, P.; Kovacs, A.; Saffitz, J.; Medeiros, D.; Kelly, D. Peroxisome proliferator–activated receptor γ coactivator-1 promotes cardiac mitochondrial biogenesis. J. Clin. Investig. 2000, 106, 847–856. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Puigserver, P.; Andersson, U.; Zhang, C.; Adelmant, G.; Mootha, V.; Troy, A.; Cinti, S.; Lowell, B.; Scarpulla, R.; et al. Mechanisms Controlling Mitochondrial Biogenesis and Respiration through the Thermogenic Coactivator PGC-1. Cell 1999, 98, 115–124. [Google Scholar] [CrossRef]
- Terada, S.; Goto, M.; Kato, M.; Kawanaka, K.; Shimokawa, T.; Tabata, I. Effects of low-intensity prolonged exercise on PGC-1 mRNA expression in rat epitrochlearis muscle. Biochem. Biophys. Res. Commun. 2002, 296, 350–354. [Google Scholar] [CrossRef]
- Pilegaard, H.; Saltin, B.; Neufer, P. Exercise induces transient transcriptional activation of the PGC-1α gene in human skeletal muscle. J. Physiol. 2003, 546, 851–858. [Google Scholar] [CrossRef]
- Maunder, E.; Plews, D.; Kilding, A. Contextualising Maximal Fat Oxidation During Exercise: Determinants and Normative Values. Front. Physiol. 2018, 9, 599. [Google Scholar] [CrossRef]
- Frandsen, J.; Vest, S.; Larsen, S.; Dela, F.; Helge, J. Maximal Fat Oxidation is Related to Performance in an Ironman Triathlon. Int. J. Sports Med. 2017, 38, 975–982. [Google Scholar] [CrossRef]
- Vest, S.; Frandsen, J.; Larsen, S.; Dela, F.; Helge, J. Peak Fat Oxidation is not Independently Related to Ironman Performance in Women. Int. J. Sports Med. 2018, 39, 916–923. [Google Scholar] [CrossRef]
- Knechtle, B.; Muller, G.; Willman, F.; Kotteck, K.; Eser, P.; Knecht, H. Fat Oxidation in Men and Women Endurance Athletes in Running and Cycling. Int. J. Sports Med. 2004, 25, 38–44. [Google Scholar]
- Fletcher, G.; Eves, F.; Glover, E.; Robinson, S.; Vernooij, C.; Thompson, J.; Wallis, G. Dietary intake is independently associated with the maximal capacity for fat oxidation during exercise. Am. J. Clin. Nutr. 2017, 105, 864–872. [Google Scholar] [CrossRef]
- Schreiber, S.; Emter, R.; Hock, M.; Knutti, D.; Cardenas, J.; Podvinec, M.; Oakeley, E.; Kralli, A. The estrogen-related receptor (ERR) functions in PPAR coactivator 1 (PGC-1)-induced mitochondrial biogenesis. Proc. Natl. Acad. Sci. USA 2004, 101, 6472–6477. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, M.; Vongpatanasin, W. Estrogen and hypertension. Curr. Hypertens. Rep. 2006, 8, 368–376. [Google Scholar] [CrossRef]
- Taniyama, Y.; Griendling, K. Reactive Oxygen Species in the Vasculature. Hypertension 2003, 42, 1075–1081. [Google Scholar] [CrossRef] [PubMed]
- Ko, K.; Woo, J.; Bae, J.; Roh, H.; Lee, Y.; Shin, K. Exercise training improves intramuscular triglyceride lipolysis sensitivity in high-fat diet induced obese mice. Lipids Health Dis. 2018, 17, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Scharhag-Rosenberger, F.; Meyer, T.; Walitzek, S.; Kindermann, W. Effects of One Year Aerobic Endurance Training on Resting Metabolic Rate and Exercise Fat Oxidation in Previously Untrained Men and Women. Int. J. Sports Med. 2010, 31, 498–504. [Google Scholar] [CrossRef]
- Bircher, S.; Knechtle, B. Relationship between Fat Oxidation and Lactate Threshold in Athletes and Obese Women and Men. J. Sports Sci. Med. 2004, 3, 174–181. [Google Scholar]
- Lima Silva, A.; Pires, F.; Bertuzzi, R.; Gagliardi, J.; Barros, R.; Hammond, J.; Kiss, M. Relationship between training status and maximal fat oxidation. J. Sports Sci. Med. 2010, 9, 31–35. [Google Scholar] [PubMed]
- Gonzalez, J.; Fuchs, C.; Betts, J.; van Loon, L. Liver glycogen metabolism during and after prolonged endurance-type exercise. Am. J. Physiol. Endocrinol. Metab. 2016, 311, E543–E553. [Google Scholar] [CrossRef]
- Jensen, J.; Rustad, P.; Kolnes, A.; Lai, Y. The Role of Skeletal Muscle Glycogen Breakdown for Regulation of Insulin Sensitivity by Exercise. Front. Physiol. 2011, 2, 1–11. [Google Scholar] [CrossRef]
- Ørtenblad, N.; Westerblad, H.; Nielsen, J. Muscle glycogen stores and fatigue. J. Physiol. 2013, 591, 4405–4413. [Google Scholar] [CrossRef]
- Ørtenblad, N.; Nielsen, J. Muscle glycogen and cell function-Location, location, location. Scand. J. Med. Sci. Sports 2015, 25, 34–40. [Google Scholar] [CrossRef]
- Nielsen, J.; Suetta, C.; Hvid, L.; Schrøder, H.; Aagaard, P.; Ørtenblad, N. Subcellular localization-dependent decrements in skeletal muscle glycogen and mitochondria content following short-term disuse in young and old men. Am. J. Physiol. Endocrinol. Metab. 2010, 299, E1053–E1060. [Google Scholar] [CrossRef]
- Nielsen, J.; Farup, J.; Rahbek, S.; de Paoli, F.; Vissing, K. Enhanced Glycogen Storage of a Subcellular Hot Spot in Human Skeletal Muscle during Early Recovery from Eccentric Contractions. PLoS ONE 2015, 10, e0127808. [Google Scholar] [CrossRef]
- Nielsen, J.; Cheng, A.; Ørtenblad, N.; Westerblad, H. Subcellular distribution of glycogen and decreased tetanic Ca2+ I n fatigued single intact mouse muscle fibres. J. Physiol. 2014, 592, 2003–2012. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, D.; Wada, M. Effects of reduced muscle glycogen on excitation–contraction coupling in rat fast-twitch muscle: A glycogen removal study. J. Muscle Res. Cell Motil. 2019, 40, 353–364. [Google Scholar] [CrossRef] [PubMed]
- Jensen, R.; Nielsen, J.; Ørtenblad, N. Inhibition of glycogenolysis prolongs action potential repriming period and impairs muscle function in rat skeletal muscle. J. Physiol. 2020, 598, 789–803. [Google Scholar] [CrossRef] [PubMed]
- Temesi, J.; Johnson, N.; Raymond, J.; Burdon, C.; O’Connor, H. Carbohydrate Ingestion during Endurance Exercise Improves Performance in Adults. J. Nutr. 2011, 141, 890–897. [Google Scholar] [CrossRef] [PubMed]
- Fogelholm, G.; Tikkanen, H.; Naveri, H.; Naveri, L.; Harkonen, M. Carbohydrate loading in practice: High muscle glycogen concentration is not certain. Br. J. Sports Med. 1991, 25, 41–44. [Google Scholar] [CrossRef]
- Holloway, G.; Lally, J.; Nickerson, J.; Alkhateeb, H.; Snook, L.; Heigenhauser, G.; Calles-Escandon, J.; Glatz, J.; Luiken, J.; Spriet, L.; et al. Fatty acid binding protein facilitates sarcolemmal fatty acid transport but not mitochondrial oxidation in rat and human skeletal muscle. J. Physiol. 2007, 582, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Nickerson, J.; Alkhateeb, H.; Benton, C.; Lally, J.; Nickerson, J.; Han, X.; Wilson, M.; Jain, S.; Snook, L.; Glatz, J.; et al. Greater Transport Efficiencies of the Membrane Fatty Acid Transporters FAT/CD36 and FATP4 Compared with FABPpm and FATP1 and Differential Effects on Fatty Acid Esterification and Oxidation in Rat Skeletal Muscle. J. Biol. Chem. 2009, 284, 16522–16530. [Google Scholar] [CrossRef] [PubMed]
- Watt, M.; van Denderen, B.; Castelli, L.; Bruce, C.; Hoy, A.; Kraegen, E.; Macaulay, L.; Kemp, B. Adipose Triglyceride Lipase Regulation of Skeletal Muscle Lipid Metabolism and Insulin Responsiveness. Mol. Endocrinol. 2008, 22, 1200–1212. [Google Scholar] [CrossRef][Green Version]
- Van Hall, G. The Physiological Regulation of Skeletal Muscle Fatty Acid Supply and Oxidation during Moderate-Intensity Exercise. Sports Med. 2015, 45, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Schenk, S.; Horowitz, J. Coimmunoprecipitation of FAT/CD36 and CPT I in skeletal muscle increases proportionally with fat oxidation after endurance exercise training. Am. J. Physiol. Endocrinol. Metab. 2006, 291, E254–E260. [Google Scholar] [CrossRef]
- Kim, J.; Lim, K. Relationship between FAT/CD36 Protein in Skeletal Muscle and Whole-body Fat Oxidation in Endurance-trained Mice. J. Exerc. Nutr. Biochem. 2016, 20, 48–52. [Google Scholar] [CrossRef]
- Kiens, B.; Roepstorff, C.; Glatz, J.; Bonen, A.; Schjerling, P.; Knudsen, J.; Nielsen, J. Lipid-binding proteins and lipoprotein lipase activity in human skeletal muscle: Influence of physical activity and gender. J. Appl. Physiol. 2004, 97, 1209–1218. [Google Scholar] [CrossRef]
- Watt, M. Triglyceride lipases alter fuel metabolism and mitochondrial gene expression. Appl. Physiol. Nutr. Metab. 2009, 34, 340–347. [Google Scholar] [CrossRef]
- Wojtaszewski, J.; MacDonald, C.; Nielsen, J.; Hellsten, Y.; Hardie, D.; Kemp, B.; Kiens, B.; Richter, E. Regulation of 5′AMP-activated protein kinase activity and substrate utilization in exercising human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2003, 284, E813–E822. [Google Scholar] [CrossRef] [PubMed]
- Moro, C.; Bajpeyi, S.; Smith, S. Determinants of intramyocellular triglyceride turnover: Implications for insulin sensitivity. Am. J. Physiol. Endocrinol. Metab. 2008, 294, E203–E213. [Google Scholar] [CrossRef] [PubMed]
- Watt, M.; Spriet, L. Regulation and role of hormone-sensitive lipase activity in human skeletal muscle. Proc. Nutr. Soc. 2004, 63, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Richter, E.; Ruderman, N. AMPK and the biochemistry of exercise: Implications for human health and disease. Biochem. J. 2009, 418, 261–275. [Google Scholar] [CrossRef]
- O’Neill, H.; Maarbjerg, S.; Crane, J.; Jeppesen, J.; Jorgensen, S.; Schertzer, J.; Shyroka, O.; Kiens, B.; van Denderen, B.; Tarnopolsky, M.; et al. AMP-activated protein kinase (AMPK) B1B2 muscle null mice reveal an essential role for AMPK in maintaining mitochondrial content and glucose uptake during exercise. Proc. Natl. Acad. Sci. USA 2011, 108, 16092–16097. [Google Scholar] [CrossRef]
- Miura, S.; Kai, Y.; Kamei, Y.; Bruce, C.; Kubota, N.; Febbraio, M.; Kadowaki, T.; Ezaki, O. α2-AMPK activity is not essential for an increase in fatty acid oxidation during low-intensity exercise. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E47–E55. [Google Scholar] [CrossRef]
- Jeppesen, J.; Kiens, B. Regulation and limitations to fatty acid oxidation during exercise. J. Physiol. 2012, 590, 1059–1068. [Google Scholar] [CrossRef]
- Ngo, K.; Denis, C.; Saafi, M.; Feasson, L.; Verney, J. Endurance but not resistance training increases intra-myocellular lipid content and β-hydroxyacyl coenzyme A dehydrogenase activity in active elderly men. Acta Physiol. 2012, 205, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Winder, W.; Arogyasami, J.; Barton, R.; Elayan, I.; Vehrs, P. Muscle malonyl-CoA decreases during exercise. J. Appl. Physiol. 1989, 67, 2230–2233. [Google Scholar] [CrossRef] [PubMed]
- Odland, L.; Heigenhauser, G.; Lopaschuk, G.; Spriet, L. Human skeletal muscle malonyl-CoA at rest and during prolonged submaximal exercise. Am. J. Physiol. Endocrinol. Metab. 1996, 270, E541–E544. [Google Scholar] [CrossRef] [PubMed]
- Odland, L.; Howlett, R.; Heigenhauser, G.; Hultman, E.; Spriet, L. Skeletal muscle malonyl-CoA content at the onset of exercise at varying power outputs in humans. Am. J. Physiol. Endocrinol. Metab. 1998, 274, E1080–E1085. [Google Scholar] [CrossRef] [PubMed]
- Roepstorff, C.; Halberg, N.; Hillig, T.; Saha, A.; Ruderman, N.; Wojtaszewski, J.; Richter, E.; Kiens, B. Malonyl-CoA and carnitine in regulation of fat oxidation in human skeletal muscle during exercise. Am. J. Physiol. Endocrinol. Metab. 2005, 288, E133–E142. [Google Scholar] [CrossRef] [PubMed]
- Starritt, E.; Howlett, R.; Heigenhauser, G.; Spriet, L. Sensitivity of CPT I to malonyl-CoA in trained and untrained human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2000, 278, E462–E468. [Google Scholar] [CrossRef] [PubMed]
- Bezaire, V.; Bruce, C.; Heigenhauser, G.; Tandon, N.; Glatz, J.; Luiken, J.; Bonen, A.; Spriet, L. Identification of fatty acid translocase on human skeletal muscle mitochondrial membranes: Essential role in fatty acid oxidation. Am. J. Physiol. Endocrinol. Metab. 2006, 290, E509–E515. [Google Scholar] [CrossRef] [PubMed]
- Stephens, F.; Constantin-Teodosiu, D.; Greenhaff, P. New insights concerning the role of carnitine in the regulation of fuel metabolism in skeletal muscle. J. Physiol. 2007, 581, 431–444. [Google Scholar] [CrossRef]
- Arenas, J.; Ricoy, J.; Encinas, A.; Pola, P.; D’Iddio, S.; Zeviani, M.; DiDonato, S.; Corsi, M. Carnitine in muscle, serum, and urine of nonprofessional athletes: Effects of physical exercise, training, and L-carnitine administration. Muscle Nerve 1991, 14, 598–604. [Google Scholar] [CrossRef]
- Wächter, S.; Vogt, M.; Kreis, R.; Boesch, C.; Bigler, P.; Hoppeler, H.; Krähenbühl, S. Long-term administration of l-carnitine to humans: Effect on skeletal muscle carnitine content and physical performance. Clin. Chim. Acta 2002, 318, 51–61. [Google Scholar] [CrossRef]
- Broad, E.; Maughan, R.; Galloway, S. Carbohydrate, Protein, and Fat Metabolism during Exercise after Oral Carnitine Supplementation in Humans. Int. J. Sport Nutr. Exerc. Metab. 2008, 18, 567–584. [Google Scholar] [CrossRef] [PubMed]
- Broad, E.; Maughan, R.; Galloway, S. Effects of Exercise Intensity and Altered Substrate Availability on Cardiovascular and Metabolic Responses to Exercise after Oral Carnitine Supplementation in Athletes. Int. J. Sport Nutr. Exerc. Metab. 2011, 21, 385–397. [Google Scholar] [CrossRef][Green Version]
- Wall, B.; Stephens, F.; Constantin-Teodosiu, D.; Marimuthu, K.; Macdonald, I.; Greenhaff, P. Chronic oral ingestion of l-carnitine and carbohydrate increases muscle carnitine content and alters muscle fuel metabolism during exercise in humans. J. Physiol. 2011, 589, 963–973. [Google Scholar] [CrossRef] [PubMed]
- Noland, R.; Koves, T.; Seiler, S.; Lum, H.; Lust, R.; Ilkayeva, O.; Stevens, R.; Hegardt, F.; Muoio, D. Carnitine Insufficiency Caused by Aging and Overnutrition Compromises Mitochondrial Performance and Metabolic Control. J. Biol. Chem. 2009, 284, 22840–22852. [Google Scholar] [CrossRef] [PubMed]
- Seiler, S.; Martin, O.; Noland, R.; Slentz, D.; DeBalsi, K.; Ilkayeva, O.; An, J.; Newgard, C.; Koves, T.; Muoio, D. Obesity and lipid stress inhibit carnitine acetyltransferase activity. J. Lipid Res. 2014, 55, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Pieklik, J.; Guynn, R. Equilibrium constants of the reactions of choline acetyltransferase, carnitine acetyltransferase, and acetylcholinesterase under physiological conditions. J. Biol. Chem. 1975, 250, 4445–4450. [Google Scholar] [CrossRef]
- Sugden, M.; Holness, M. Recent advances in mechanisms regulating glucose oxidation at the level of the pyruvate dehydrogenase complex by PDKs. Am. J. Physiol. Endocrinol. Metab. 2003, 284, E855–E862. [Google Scholar] [CrossRef]
- Sahlin, K. Muscle carnitine metabolism during incremental dynamic exercise in humans. Acta Physiol. Scand. 1990, 138, 259–262. [Google Scholar] [CrossRef]
- Muoio, D.; Noland, R.; Kovalik, J.; Seiler, S.; Davies, M.; DeBalsi, K.; Ilkayeva, O.; Stevens, R.; Kheterpal, I.; Zhang, J.; et al. Muscle-Specific Deletion of Carnitine Acetyltransferase Compromises Glucose Tolerance and Metabolic Flexibility. Cell Metab. 2012, 15, 764–777. [Google Scholar] [CrossRef]
- Seiler, S.; Koves, T.; Gooding, J.; Wong, K.; Stevens, R.; Ilkayeva, O.; Wittmann, A.; DeBalsi, K.; Davies, M.; Lindeboom, L.; et al. Carnitine Acetyltransferase Mitigates Metabolic Inertia and Muscle Fatigue during Exercise. Cell Metab. 2015, 22, 65–76. [Google Scholar] [CrossRef]
- Fathizadeh, H.; Milajerdi, A.; Reiner, Ž.; Asemi, Z.; Kolahdooz, F. The effects of L-carnitine supplementation on glycemic control: A systematic review and meta-analysis of randomized controlled trials. Excli J. 2019, 18, 631–643. [Google Scholar]
- Sidhu, S.; Weavil, J.; Thurston, T.; Rosenberger, D.; Jessop, J.; Wang, E.; Richardson, R.; McNeil, C.; Amann, M. Fatigue-related group III/IV muscle afferent feedback facilitates intracortical inhibition during locomotor exercise. J. Physiol. 2018, 596, 4789–4801. [Google Scholar] [CrossRef] [PubMed]
- Sidhu, S.; Weavil, J.; Mangum, T.; Jessop, J.; Richardson, R.; Morgan, D.; Amann, M. Group III/IV locomotor muscle afferents alter motor cortical and corticospinal excitability and promote central fatigue during cycling exercise. Clin. Neurophysiol. 2017, 128, 44–55. [Google Scholar] [CrossRef] [PubMed]
- Newgard, C.; An, J.; Bain, J.; Muehlbauer, M.; Stevens, R.; Lien, L.; Haqq, A.; Shah, S.; Arlotto, M.; Slentz, C.; et al. A Branched-Chain Amino Acid-Related Metabolic Signature that Differentiates Obese and Lean Humans and Contributes to Insulin Resistance. Cell Metab. 2009, 9, 565–566. [Google Scholar] [CrossRef]
- Magkos, F.; Bradley, D.; Schweitzer, G.; Finck, B.; Eagon, J.; Ilkayeva, O.; Newgard, C.; Klein, S. Effect of Roux-en-Y Gastric Bypass and Laparoscopic Adjustable Gastric Banding on Branched-Chain Amino Acid Metabolism. Diabetes 2013, 62, 2757–2761. [Google Scholar] [CrossRef]
- Oku, H.; Kaneda, T. Biosynthesis of branched-chain fatty acids in Bacillus subtilis. A decarboxylase is essential for branched-chain fatty acid synthetase. J. Biol. Chem. 1988, 263, 18386–18396. [Google Scholar] [CrossRef]
- Lackey, D.; Lynch, C.; Olson, K.; Mostaedi, R.; Ali, M.; Smith, W.; Karpe, F.; Humphreys, S.; Bedinger, D.; Dunn, T.; et al. Regulation of adipose branched-chain amino acid catabolism enzyme expression and cross-adipose amino acid flux in human obesity. Am. J. Physiol. Endocrinol. Metab. 2013, 304, E1175–E1187. [Google Scholar] [CrossRef]
- Su, X.; Magkos, F.; Zhou, D.; Eagon, J.; Fabbrini, E.; Okunade, A.; Klein, S. Adipose tissue monomethyl branched-chain fatty acids and insulin sensitivity: Effects of obesity and weight loss. Obesity 2014, 23, 329–334. [Google Scholar] [CrossRef]
- Wallace, M.; Green, C.; Roberts, L.; Lee, Y.; McCarville, J.; Sanchez-Gurmaches, J.; Meurs, N.; Gengatharan, J.; Hover, J.; Phillips, S.; et al. Enzyme promiscuity drives branched-chain fatty acid synthesis in adipose tissues. Nat. Chem. Biol. 2018, 14, 1021–1031. [Google Scholar] [CrossRef]
- Davies, M.; Kjalarsdottir, L.; Thompson, J.; Dubois, L.; Stevens, R.; Ilkayeva, O.; Brosnan, M.; Rolph, T.; Grimsrud, P.; Muoio, D. The Acetyl Group Buffering Action of Carnitine Acetyltransferase Offsets Macronutrient-Induced Lysine Acetylation of Mitochondrial Proteins. Cell Rep. 2016, 14, 243–254. [Google Scholar] [CrossRef]
- Hirschey, M.; Shimazu, T.; Jing, E.; Grueter, C.; Collins, A.; Aouizerat, B.; Stančáková, A.; Goetzman, E.; Lam, M.; Schwer, B.; et al. SIRT3 Deficiency and Mitochondrial Protein Hyperacetylation Accelerate the Development of the Metabolic Syndrome. Mol. Cell 2011, 44, 177–190. [Google Scholar] [CrossRef] [PubMed]
- Morales, J.; Castillo, E.; Silva-Platas, C.; Torre-Amione, G.; Youker, K.; García-Rivas, G. Mitochondrial Hyperacetylation Contributes with Ventricular Dysfunction as Consequence of SIRT3 Deficiency in Obesity and Metabolic Syndrome. J. Card. Fail. 2017, 23, S39. [Google Scholar] [CrossRef][Green Version]
- Essner, R.; Smith, A.; Jamnik, A.; Ryba, A.; Trutner, Z.; Carter, M. AgRP Neurons Can Increase Food Intake during Conditions of Appetite Suppression and Inhibit Anorexigenic Parabrachial Neurons. J. Neurosci. 2017, 37, 8678–8687. [Google Scholar] [CrossRef] [PubMed]
- Reichenbach, A.; Stark, R.; Mequinion, M.; Lockie, S.; Lemus, M.; Mynatt, R.; Luquet, S.; Andrews, Z. Carnitine acetyltransferase (Crat) in hunger-sensing AgRP neurons permits adaptation to calorie restriction. FASEB J. 2018, 32, 6923–6933. [Google Scholar] [CrossRef] [PubMed]
- Brooks, G. Lactate: Glycolytic end product and oxidative substrate during sustained exercise in mammals—the “lactate shuttle”. In Circulation, Respiration, and Metabolism; Gilles, R., Ed.; Springer: Berlin/Heidelberg, Germany, 1985; pp. 208–218. [Google Scholar]
- Brooks, G. The Science and Translation of Lactate Shuttle Theory. Cell Metab. 2018, 27, 757–785. [Google Scholar] [CrossRef]
- Brooks, G.; Brown, M.; Butz, C.; Sicurello, J.; Dubouchaud, H. Cardiac and skeletal muscle mitochondria have a monocarboxylate transporter MCT1. J. Appl. Physiol. 1999, 87, 1713–1718. [Google Scholar] [CrossRef]
- McClelland, G.; Khanna, S.; González, G.; Eric Butz, C.; Brooks, G. Peroxisomal membrane monocarboxylate transporters: Evidence for a redox shuttle system? Biochem. Biophys. Res. Commun. 2003, 304, 130–135. [Google Scholar] [CrossRef]
- Hashimoto, T.; Masuda, S.; Taguchi, S.; Brooks, G. Immunohistochemical analysis of MCT1, MCT2 and MCT4 expression in rat plantaris muscle. J. Physiol. 2005, 567, 121–129. [Google Scholar] [CrossRef]
- Hashimoto, T.; Hussien, R.; Brooks, G. Colocalization of MCT1, CD147, and LDH in mitochondrial inner membrane of L6 muscle cells: Evidence of a mitochondrial lactate oxidation complex. Am. J. Physiol. Endocrinol. Metab. 2006, 290, E1237–E1244. [Google Scholar] [CrossRef]
- Stanley, W.; Gertz, E.; Wisneski, J.; Neese, R.; Morris, D.; Brooks, G. Lactate extraction during net lactate release in legs of humans during exercise. J. Appl. Physiol. 1986, 60, 1116–1120. [Google Scholar] [CrossRef] [PubMed]
- Henneman, E. Relation between Size of Neurons and Their Susceptibility to Discharge. Science 1957, 126, 1345–1347. [Google Scholar] [CrossRef] [PubMed]
- Leberer, E.; Pette, D. Lactate dehydrogenase isozymes in type I, IIA and IIB fibres of rabbit skeletal muscles. Histochemistry 1984, 80, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Rogatzki, M.; Ferguson, B.; Goodwin, M.; Gladden, L. Lactate is always the end product of glycolysis. Front. Neurosci. 2015, 9, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Allen, D.; Lamb, G.; Westerblad, H. Skeletal Muscle Fatigue: Cellular Mechanisms. Physiol. Rev. 2008, 88, 287–332. [Google Scholar] [CrossRef] [PubMed]
- Marcinek, D.; Kushmerick, M.; Conley, K. Lactic acidosis in vivo: Testing the link between lactate generation and H+ accumulation in ischemic mouse muscle. J. Appl. Physiol. 2010, 108, 1479–1486. [Google Scholar] [CrossRef]
- Azevedo, J.; Tietz, E.; Two-Feathers, T.; Paull, J.; Chapman, K. Lactate, Fructose and Glucose Oxidation Profiles in Sports Drinks and the Effect on Exercise Performance. PLoS ONE 2007, 2, e927. [Google Scholar] [CrossRef]
- Nalos, M.; Leverve, X.; Huang, S.; Weisbrodt, L.; Parkin, R.; Seppelt, I.; Ting, I.; Mclean, A. Half-molar sodium lactate infusion improves cardiac performance in acute heart failure: A pilot randomised controlled clinical trial. Crit. Care 2014, 18, R48. [Google Scholar] [CrossRef]
- De Paoli, F.; Overgaard, K.; Pedersen, T.; Nielsen, O. Additive protective effects of the addition of lactic acid and adrenaline on excitability and force in isolated rat skeletal muscle depressed by elevated extracellular K+. J. Physiol. 2007, 581, 829–839. [Google Scholar] [CrossRef]
- Young, A.; Oldford, C.; Mailloux, R. Lactate dehydrogenase supports lactate oxidation in mitochondria isolated from different mouse tissues. Redox Biol. 2020, 28, 101339. [Google Scholar] [CrossRef]
- Passarella, S.; Paventi, G.; Pizzuto, R. The mitochondrial L-lactate dehydrogenase affair. Front. Neurosci. 2014, 8, 1–4. [Google Scholar] [CrossRef]
- Fulghum, K.; Rood, B.; Shang, V.; McNally, L.; Zheng, Y.; Hill, B. Mitochondria-localized lactate dehydrogenase is not a biologically significant contributor to bioenergetic function in striated muscle. Free Radic. Biol. Med. 2018, 128, S83. [Google Scholar] [CrossRef]
- Sahlin, K.; Fernström, M.; Svensson, M.; Tonkonogi, M. No evidence of an intracellular lactate shuttle in rat skeletal muscle. J. Physiol. 2002, 541, 569–574. [Google Scholar] [CrossRef]
- Chicco, A.; Le, C.; Gnaiger, E.; Dreyer, H.; Muyskens, J.; D’Alessandro, A.; Nemkov, T.; Hocker, A.; Prenni, J.; Wolfe, L.; et al. Adaptive remodeling of skeletal muscle energy metabolism in high-altitude hypoxia: Lessons from AltitudeOmics. J. Biol. Chem. 2018, 293, 6659–6671. [Google Scholar] [CrossRef] [PubMed]
- San-Millán, I.; Brooks, G. Assessment of Metabolic Flexibility by Means of Measuring Blood Lactate, Fat, and Carbohydrate Oxidation Responses to Exercise in Professional Endurance Athletes and Less-Fit Individuals. Sports Med. 2017, 48, 467–479. [Google Scholar] [CrossRef] [PubMed]
- Bergman, B.; Butterfield, G.; Wolfel, E.; Lopaschuk, G.; Casazza, G.; Horning, M.; Brooks, G. Muscle net glucose uptake and glucose kinetics after endurance training in men. Am. J. Physiol. Endocrinol. Metab. 1999, 277, E81–E92. [Google Scholar] [CrossRef] [PubMed]
- Bergman, B.; Wolfel, E.; Butterfield, G.; Lopaschuk, G.; Casazza, G.; Horning, M.; Brooks, G. Active muscle and whole body lactate kinetics after endurance training in men. J. Appl. Physiol. 1999, 87, 1684–1696. [Google Scholar] [CrossRef] [PubMed]
- Summermatter, S.; Santos, G.; Perez-Schindler, J.; Handschin, C. Skeletal muscle PGC-1a controls whole-body lactate homeostasis through estrogen-related receptor -dependent activation of LDH B and repression of LDH A. Proc. Natl. Acad. Sci. USA 2013, 110, 8738–8743. [Google Scholar] [CrossRef]
- Lund, J.; Aas, V.; Tingstad, R.; Van Hees, A.; Nikolić, N. Utilization of lactic acid in human myotubes and interplay with glucose and fatty acid metabolism. Sci. Rep. 2018, 8, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Wehrlin, J.; Hallén, J. Linear decrease in VO2max and performance with increasing altitude in endurance athletes. Eur. J. Appl. Physiol. 2005, 96, 404–412. [Google Scholar] [CrossRef]
- Hochachka, P.; Beatty, C.; Burelle, Y.; Trump, M.; McKenzie, D.; Matheson, G. The Lactate Paradox in Human High-Altitude Physiological Performance. Physiology 2002, 17, 122–126. [Google Scholar] [CrossRef][Green Version]
- Levett, D.; Radford, E.; Menassa, D.; Graber, E.; Morash, A.; Hoppeler, H.; Clarke, K.; Martin, D.; Ferguson-Smith, A.; Montgomery, H.; et al. Acclimatization of skeletal muscle mitochondria to high-altitude hypoxia during an ascent of Everest. FASEB J. 2011, 26, 1431–1441. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Wu, H.; Tarr, P.; Zhang, C.; Wu, Z.; Boss, O.; Michael, L.; Puigserver, P.; Isotani, E.; Olson, E.; et al. Transcriptional co-activator PGC-1α drives the formation of slow-twitch muscle fibres. Nature 2002, 418, 797–801. [Google Scholar] [CrossRef] [PubMed]
- Wende, A.; Schaeffer, P.; Parker, G.; Zechner, C.; Han, D.; Chen, M.; Hancock, C.; Lehman, J.; Huss, J.; McClain, D.; et al. A Role for the Transcriptional Coactivator PGC-1α in Muscle Refueling. J. Biol. Chem. 2007, 282, 36642–36651. [Google Scholar] [CrossRef] [PubMed]
- Calvo, J.; Daniels, T.; Wang, X.; Paul, A.; Lin, J.; Spiegelman, B.; Stevenson, S.; Rangwala, S. Muscle-specific expression of PPARγ coactivator-1α improves exercise performance and increases peak oxygen uptake. J. Appl. Physiol. 2008, 104, 1304–1312. [Google Scholar] [CrossRef] [PubMed]
- Vega, R.; Huss, J.; Kelly, D. The Coactivator PGC-1 Cooperates with Peroxisome Proliferator-Activated Receptor alpha in Transcriptional Control of Nuclear Genes Encoding Mitochondrial Fatty Acid Oxidation Enzymes. Mol. Cell. Biol. 2000, 20, 1868–1876. [Google Scholar] [CrossRef] [PubMed]
- Michael, L.; Wu, Z.; Cheatham, R.; Puigserver, P.; Adelmant, G.; Lehman, J.; Kelly, D.; Spiegelman, B. Restoration of insulin-sensitive glucose transporter (GLUT4) gene expression in muscle cells by the transcriptional coactivator PGC-1. Proc. Natl. Acad. Sci. USA 2001, 98, 3820–3825. [Google Scholar] [CrossRef]
- Constantin-Teodosiu, D.; Baker, D.; Constantin, D.; Greenhaff, P. PPARδ agonism inhibits skeletal muscle PDC activity, mitochondrial ATP production and force generation during prolonged contraction. J. Physiol. 2009, 587, 231–239. [Google Scholar] [CrossRef]
- Varga, T.; Czimmerer, Z.; Nagy, L. PPARs are a unique set of fatty acid regulated transcription factors controlling both lipid metabolism and inflammation. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2011, 1812, 1007–1022. [Google Scholar] [CrossRef]
- Zajac, A.; Poprzecki, S.; Maszczyk, A.; Czuba, M.; Michalczyk, M.; Zydek, G. The Effects of a Ketogenic Diet on Exercise Metabolism and Physical Performance in Off-Road Cyclists. Nutrients 2014, 6, 2493–2508. [Google Scholar] [CrossRef]
- Cox, P.; Kirk, T.; Ashmore, T.; Willerton, K.; Evans, R.; Smith, A.; Murray, A.; Stubbs, B.; West, J.; McLure, S.; et al. Nutritional Ketosis Alters Fuel Preference and Thereby Endurance Performance in Athletes. Cell Metab. 2016, 24, 256–268. [Google Scholar] [CrossRef]
- Dalvi, P.; Chalmers, J.; Luo, V.; Han, D.; Wellhauser, L.; Liu, Y.; Tran, D.; Castel, J.; Luquet, S.; Wheeler, M.; et al. High fat induces acute and chronic inflammation in the hypothalamus: Effect of high-fat diet, palmitate and TNF-α on appetite-regulating NPY neurons. Int. J. Obes. 2016, 41, 149–158. [Google Scholar] [CrossRef]
- Morselli, E.; Fuente-Martin, E.; Finan, B.; Kim, M.; Frank, A.; Garcia-Caceres, C.; Navas, C.; Gordillo, R.; Neinast, M.; Kalainayakan, S.; et al. Hypothalamic PGC-1α Protects Against High-Fat Diet Exposure by Regulating ERα. Cell Rep. 2014, 9, 633–645. [Google Scholar] [CrossRef]
- Crunkhorn, S.; Dearie, F.; Mantzoros, C.; Gami, H.; da Silva, W.; Espinoza, D.; Faucette, R.; Barry, K.; Bianco, A.; Patti, M. Peroxisome Proliferator Activator Receptor γ Coactivator-1 Expression Is Reduced in Obesity. J. Biol. Chem. 2007, 282, 15439–15450. [Google Scholar] [CrossRef]
- Koh, J.; Johnson, M.; Dasari, S.; LeBrasseur, N.; Vuckovic, I.; Henderson, G.; Cooper, S.; Manjunatha, S.; Ruegsegger, G.; Shulman, G.; et al. TFAM Enhances Fat Oxidation and Attenuates High Fat Diet Induced Insulin Resistance in Skeletal Muscle. Diabetes 2019, 68, 1552–1564. [Google Scholar] [CrossRef] [PubMed]
- Cai, T.; Ren, N.; Jin, L.; Cheng, K.; Kash, S.; Chen, R.; Wright, S.; Taggart, A.; Waters, M. Role of GPR81 in lactate-mediated reduction of adipose lipolysis. Biochem. Biophys. Res. Commun. 2008, 377, 987–991. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Wu, J.; Zhu, J.; Kuei, C.; Yu, J.; Shelton, J.; Sutton, S.; Li, X.; Yun, S.; Mirzadegan, T.; et al. Lactate Inhibits Lipolysis in Fat Cells through Activation of an Orphan G-protein-coupled Receptor, GPR81. J. Biol. Chem. 2008, 284, 2811–2822. [Google Scholar] [CrossRef]
- Chang, A.; Ortega, F.; Riegler, J.; Madison, D.; Krasnow, M. Oxygen regulation of breathing through an olfactory receptor activated by lactate. Nature 2015, 527, 240–244. [Google Scholar] [CrossRef]
- Morland, C.; Andersson, K.; Haugen, Ø.; Hadzic, A.; Kleppa, L.; Gille, A.; Rinholm, J.; Palibrk, V.; Diget, E.; Kennedy, L.; et al. Exercise induces cerebral VEGF and angiogenesis via the lactate receptor HCAR1. Nat. Commun. 2017, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Nalbandian, M.; Radak, Z.; Takeda, M. N-acetyl-L-cysteine Prevents Lactate-Mediated PGC1-alpha Expression in C2C12 Myotubes. Biology 2019, 8, 44. [Google Scholar] [CrossRef] [PubMed]
- Powers, S.; Jackson, M. Exercise-Induced Oxidative Stress: Cellular Mechanisms and Impact on Muscle Force Production. Physiol. Rev. 2008, 88, 1243–1276. [Google Scholar] [CrossRef]
- Steinbacher, P.; Eckl, P. Impact of Oxidative Stress on Exercising Skeletal Muscle. Biomolecules 2015, 5, 356–377. [Google Scholar] [CrossRef] [PubMed]
- Sakellariou, G.; Jackson, M.; Vasilaki, A. Redefining the major contributors to superoxide production in contracting skeletal muscle. The role of NAD(P)H oxidases. Free Radic. Res. 2013, 48, 12–29. [Google Scholar] [CrossRef] [PubMed]
- Peake, J.; Suzuki, K.; Wilson, G.; Hordern, M.; Nosaka, K.; Mackinnon, L.; Coombes, J. Exercise-Induced Muscle Damage, Plasma Cytokines, and Markers of Neutrophil Activation. Med. Sci. Sports Exerc. 2005, 37, 737–745. [Google Scholar] [CrossRef] [PubMed]
- Duarte, J.; Appell, H.; Carvalho, F.; Bastos, M.; Soares, J. Endothelium-Derived Oxidative Stress May Contribute to Exercise-Induced Muscle Damage. Int. J. Sports Med. 1993, 14, 440–443. [Google Scholar] [CrossRef] [PubMed]
- Fulle, S.; Protasi, F.; Di Tano, G.; Pietrangelo, T.; Beltramin, A.; Boncompagni, S.; Vecchiet, L.; Fanò, G. The contribution of reactive oxygen species to sarcopenia and muscle ageing. Exp. Gerontol. 2004, 39, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Burns, D.; Ali, I.; Rieux, C.; Healy, J.; Jasionek, G.; O’Halloran, K. Tempol Supplementation Restores Diaphragm Force and Metabolic Enzyme Activities in mdx Mice. Antioxidants 2017, 6, 101. [Google Scholar] [CrossRef] [PubMed]
- Lewis, P.; O’Halloran, K. Diaphragm Muscle Adaptation to Sustained Hypoxia: Lessons from Animal Models with Relevance to High Altitude and Chronic Respiratory Diseases. Front. Physiol. 2016, 7, 1–11. [Google Scholar] [CrossRef]
- Reid, M.; Khawli, F.; Moody, M. Reactive oxygen in skeletal muscle. III. Contractility of unfatigued muscle. J. Appl. Physiol. 1993, 75, 1081–1087. [Google Scholar] [CrossRef]
- Debold, E. Potential molecular mechanisms underlying muscle fatigue mediated by reactive oxygen and nitrogen species. Front. Physiol. 2015, 6, 1–7. [Google Scholar] [CrossRef]
- Ristow, M.; Zarse, K.; Oberbach, A.; Kloting, N.; Birringer, M.; Kiehntopf, M.; Stumvoll, M.; Kahn, C.; Bluher, M. Antioxidants prevent health-promoting effects of physical exercise in humans. Proc. Natl. Acad. Sci. USA 2009, 106, 8665–8670. [Google Scholar] [CrossRef]
- Tanskanen, M.; Atalay, M.; Uusitalo, A. Altered oxidative stress in overtrained athletes. J. Sports Sci. 2010, 28, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Palazzetti, S.; Richard, M.; Favier, A.; Margaritis, I. Overloaded Training Increases Exercise-Induced Oxidative Stress and Damage. Can. J. Appl. Physiol. 2003, 28, 588–604. [Google Scholar] [CrossRef] [PubMed]
- 176 Gumucio, J.; Mendias, C. Atrogin-1, MuRF-1, and sarcopenia. Endocrine 2012, 43, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Brault, J.; Gygi, S.; Glass, D.; Valenzuela, D.; Gartner, C.; Latres, E.; Goldberg, A. During muscle atrophy, thick, but not thin, filament components are degraded by MuRF1-dependent ubiquitylation. J. Cell Biol. 2009, 185, 1083–1095. [Google Scholar] [CrossRef] [PubMed]
- St-Pierre, J.; Lin, J.; Krauss, S.; Tarr, P.; Yang, R.; Newgard, C.; Spiegelman, B. Bioenergetic Analysis of Peroxisome Proliferator-activated Receptor γ Coactivators 1α and 1β (PGC-1α and PGC-1β) in Muscle Cells. J. Biol. Chem. 2003, 278, 26597–26603. [Google Scholar] [CrossRef]
- St-Pierre, J.; Drori, S.; Uldry, M.; Silvaggi, J.; Rhee, J.; Jäger, S.; Handschin, C.; Zheng, K.; Lin, J.; Yang, W.; et al. Suppression of Reactive Oxygen Species and Neurodegeneration by the PGC-1 Transcriptional Coactivators. Cell 2006, 127, 397–408. [Google Scholar] [CrossRef]
- Valle, I.; Alvarezbarrientos, A.; Arza, E.; Lamas, S.; Monsalve, M. PGC-1α regulates the mitochondrial antioxidant defense system in vascular endothelial cells. Cardiovasc. Res. 2005, 66, 562–573. [Google Scholar] [CrossRef]
- Brown, M.; McClean, C.; Davison, G.; Brown, J.; Murphy, M. The acute effects of walking exercise intensity on systemic cytokines and oxidative stress. Eur. J. Appl. Physiol. 2018, 118, 2111–2120. [Google Scholar] [CrossRef]
- Larsen, E.; Poulsen, H.; Michaelsen, C.; Kjær, L.; Lyngbæk, M.; Andersen, E.; Petersen-Bønding, C.; Lemoine, C.; Gillum, M.; Jørgensen, N.; et al. Differential time responses in inflammatory and oxidative stress markers after a marathon: An observational study. J. Sports Sci. 2020, 38, 2080–2091. [Google Scholar] [CrossRef]
- Withee, E.; Tippens, K.; Dehen, R.; Tibbitts, D.; Hanes, D.; Zwickey, H. Effects of Methylsulfonylmethane (MSM) on exercise-induced oxidative stress, muscle damage, and pain following a half-marathon: A double-blind, randomized, placebo-controlled trial. J. Int. Soc. Sports Nutr. 2017, 14, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Jamurtas, A.; Fatouros, I.; Deli, C.; Georgakouli, K.; Poulios, A.; Draganidis, D.; Papanikolaou, K.; Tsimeas, P.; Chatzinikolaou, A.; Avloniti, A.; et al. The Effects of Acute Low-Volume HIIT and Aerobic Exercise on Leukocyte Count and Redox Status. J. Sports Sci. Med. 2018, 17, 501–508. [Google Scholar]
- Holloway, T.; Bloemberg, D.; da Silva, M.; Simpson, J.; Quadrilatero, J.; Spriet, L. High Intensity Interval and Endurance Training Have Opposing Effects on Markers of Heart Failure and Cardiac Remodeling in Hypertensive Rats. PLoS ONE 2015, 10, e0121138. [Google Scholar] [CrossRef] [PubMed]
- Holloway, T.; Bloemberg, D.; da Silva, M.; Quadrilatero, J.; Spriet, L. High-intensity interval and endurance training are associated with divergent skeletal muscle adaptations in a rodent model of hypertension. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 308, R927–R934. [Google Scholar] [CrossRef] [PubMed]
- Sharma, B.; Patil, M.; Satyanarayana, A. Negative Regulators of Brown Adipose Tissue (BAT)-Mediated Thermogenesis. J. Cell. Physiol. 2014, 229, 1901–1907. [Google Scholar] [CrossRef]
- Miwa, S.; Brand, M. Mitochondrial matrix reactive oxygen species production is very sensitive to mild uncoupling. Biochem. Soc. Trans. 2003, 31, 1300–1301. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.; Brown, K.; Hirschey, M.; Verdin, E.; Chen, D. Calorie Restriction Reduces Oxidative Stress by SIRT3-Mediated SOD2 Activation. Cell Metab. 2010, 12, 662–667. [Google Scholar] [CrossRef]
- Ahn, B.; Kim, H.; Song, S.; Lee, I.; Liu, J.; Vassilopoulos, A.; Deng, C.; Finkel, T. A role for the mitochondrial deacetylase Sirt3 in regulating energy homeostasis. Proc. Natl. Acad. Sci. USA 2008, 105, 14447–14452. [Google Scholar] [CrossRef]
- Finley, L.; Haas, W.; Desquiret-Dumas, V.; Wallace, D.; Procaccio, V.; Gygi, S.; Haigis, M. Succinate Dehydrogenase Is a Direct Target of Sirtuin 3 Deacetylase Activity. PLoS ONE 2011, 6, e23295. [Google Scholar] [CrossRef]
- Jia, Y.; Wei, W.; Ma, B.; Xu, Y.; Liu, W.; Wang, Y.; Lv, K.; Tang, H.; Wei, D.; Xia, Z. Activation of p38 MAPK by Reactive Oxygen Species Is Essential in a Rat Model of Stress-Induced Gastric Mucosal Injury. J. Immunol. 2007, 179, 7808–7819. [Google Scholar] [CrossRef]
- Wang, Y.; Huang, X.; Cang, H.; Gao, F.; Yamamoto, T.; Osaki, T.; Yi, J. The endogenous reactive oxygen species promote NF-κB activation by targeting on activation of NF-κB-inducing kinase in oral squamous carcinoma cells. Free Radic. Res. 2007, 41, 963–971. [Google Scholar] [CrossRef]
- Wagner, S.; Ruff, H.; Weber, S.; Bellmann, S.; Sowa, T.; Schulte, T.; Anderson, M.; Grandi, E.; Bers, D.; Backs, J.; et al. Reactive Oxygen Species–Activated Ca/Calmodulin Kinase IIδ Is Required for Late I(Na) Augmentation Leading to Cellular Na and Ca Overload. Circ. Res. 2011, 108, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; New, L.; Kravchenko, V.; Kato, Y.; Gram, H.; di Padova, F.; Olson, E.; Ulevitch, R.; Han, J. Regulation of the MEF2 Family of Transcription Factors by p38. Mol. Cell. Biol. 1999, 19, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Handschin, C.; Rhee, J.; Lin, J.; Tarr, P.; Spiegelman, B. An autoregulatory loop controls peroxisome proliferator-activated receptor coactivator 1 expression in muscle. Proc. Natl. Acad. Sci. USA 2003, 100, 7111–7116. [Google Scholar] [CrossRef] [PubMed]
- Boss, O.; Bachman, E.; Vidal-Puig, A.; Zhang, C.; Peroni, O.; Lowell, B. Role of the β3-Adrenergic Receptor and/or a Putative β4-Adrenergic Receptor on the Expression of Uncoupling Proteins and Peroxisome Proliferator-Activated Receptor-γ Coactivator-1. Biochem. Biophys. Res. Commun. 1999, 261, 870–876. [Google Scholar] [CrossRef] [PubMed]
- Akimoto, T.; Pohnert, S.; Li, P.; Zhang, M.; Gumbs, C.; Rosenberg, P.; Williams, R.; Yan, Z. Exercise Stimulates Pgc-1α Transcription in Skeletal Muscle through Activation of the p38 MAPK Pathway. J. Biol. Chem. 2005, 280, 19587–19593. [Google Scholar] [CrossRef]
- Puigserver, P.; Rhee, J.; Lin, J.; Wu, Z.; Yoon, J.; Zhang, C.; Krauss, S.; Mootha, V.; Lowell, B.; Spiegelman, B. Cytokine Stimulation of Energy Expenditure through p38 MAP Kinase Activation of PPARγ Coactivator-1. Mol. Cell 2001, 8, 971–982. [Google Scholar] [CrossRef]
- Deng, Y.; Zhang, Q.; Luo, H.; Chen, X.; Han, Q.; Wang, F.; Huang, P.; Lai, W.; Guan, X.; Pan, X.; et al. Sustained elevation of NF-κB activity sensitizes offspring of maternal inflammation to hypertension via impairing PGC-1α recovery. Sci. Rep. 2016, 6, 1–14. [Google Scholar]
- Álvarez-Guardia, D.; Palomer, X.; Coll, T.; Davidson, M.; Chan, T.; Feldman, A.; Laguna, J.; Vázquez-Carrera, M. The p65 subunit of NF-κB binds to PGC-1α, linking inflammation and metabolic disturbances in cardiac cells. Cardiovasc. Res. 2010, 87, 449–458. [Google Scholar] [CrossRef]
- Eisele, P.; Salatino, S.; Sobek, J.; Hottiger, M.; Handschin, C. The Peroxisome Proliferator-activated Receptor γ Coactivator 1α/β (PGC-1) Coactivators Repress the Transcriptional Activity of NF-κB in Skeletal Muscle Cells. J. Biol. Chem. 2013, 288, 2246–2260. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Jeoung, N.; Burgess, S.; Rosaaen-Stowe, K.; Inagaki, T.; Latif, S.; Shelton, J.; McAnally, J.; Bassel-Duby, R.; Harris, R.; et al. Overexpression of pyruvate dehydrogenase kinase 4 in heart perturbs metabolism and exacerbates calcineurin-induced cardiomyopathy. Am. J. Physiol. Heart Circ. Physiol. 2008, 294, H936–H943. [Google Scholar] [CrossRef]
- Wilkins, B.; Molkentin, J. Calcineurin and cardiac hypertrophy: Where have we been? Where are we going? J. Physiol. 2002, 541, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, N.; Gill, J.; Brink, M.; Handschin, C. Moderate Modulation of Cardiac PGC-1α Expression Partially Affects Age-Associated Transcriptional Remodeling of the Heart. Front. Physiol. 2018, 9, 1–13. [Google Scholar] [CrossRef]
- Berridge, M.; Bootman, M.; Roderick, H. Calcium signalling: Dynamics, homeostasis and remodelling. Nat. Rev. Mol. Cell Biol. 2003, 4, 517–529. [Google Scholar] [CrossRef]
- Russ, D.; Krause, J.; Wills, A.; Arreguin, R. “SR stress” in mixed hindlimb muscles of aging male rats. Biogerontology 2012, 13, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Gill, J.; Delezie, J.; Santos, G.; McGuirk, S.; Schnyder, S.; Frank, S.; Rausch, M.; St-Pierre, J.; Handschin, C. Peroxisome proliferator-activated receptor γ coactivator 1α regulates mitochondrial calcium homeostasis, sarcoplasmic reticulum stress, and cell death to mitigate skeletal muscle aging. Aging Cell 2019, 18, e12993. [Google Scholar] [CrossRef] [PubMed]
- Wright, D.; Han, D.; Garcia-Roves, P.; Geiger, P.; Jones, T.; Holloszy, J. Exercise-induced Mitochondrial Biogenesis Begins before the Increase in Muscle PGC-1α Expression. J. Biol. Chem. 2006, 282, 194–199. [Google Scholar] [CrossRef]
- Little, J.; Safdar, A.; Cermak, N.; Tarnopolsky, M.; Gibala, M. Acute endurance exercise increases the nuclear abundance of PGC-1α in trained human skeletal muscle. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 298, R912–R917. [Google Scholar] [CrossRef] [PubMed]
- Carling, D.; Thornton, C.; Woods, A.; Sanders, M. AMP-activated protein kinase: New regulation, new roles? Biochem. J. 2012, 445, 11–27. [Google Scholar] [CrossRef] [PubMed]
- Cantó, C.; Gerhart-Hines, Z.; Feige, J.; Lagouge, M.; Noriega, L.; Milne, J.; Elliott, P.; Puigserver, P.; Auwerx, J. AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature 2009, 458, 1056–1060. [Google Scholar] [CrossRef]
- Gurd, B.; Yoshida, Y.; McFarlan, J.; Holloway, G.; Moyes, C.; Heigenhauser, G.; Spriet, L.; Bonen, A. Nuclear SIRT1 activity, but not protein content, regulates mitochondrial biogenesis in rat and human skeletal muscle. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 301, R67–R75. [Google Scholar] [CrossRef]
- Jager, S.; Handschin, C.; St.-Pierre, J.; Spiegelman, B. AMP-activated protein kinase (AMPK) action in skeletal muscle via direct phosphorylation of PGC-1. Proc. Natl. Acad. Sci. USA 2007, 104, 12017–12022. [Google Scholar] [CrossRef]
- Wojtaszewski, J.; Nielsen, P.; Hansen, B.; Richter, E.; Kiens, B. Isoform-specific and exercise intensity-dependent activation of 5′-AMP-activated protein kinase in human skeletal muscle. J. Physiol. 2000, 528, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Wojtaszewski, J.; Mourtzakis, M.; Hillig, T.; Saltin, B.; Pilegaard, H. Dissociation of AMPK activity and ACCβ phosphorylation in human muscle during prolonged exercise. Biochem. Biophys. Res. Commun. 2002, 298, 309–316. [Google Scholar] [CrossRef]
- Chen, Z.; Stephens, T.; Murthy, S.; Canny, B.; Hargreaves, M.; Witters, L.; Kemp, B.; McConell, G. Effect of Exercise Intensity on Skeletal Muscle AMPK Signaling in Humans. Diabetes 2003, 52, 2205–2212. [Google Scholar] [CrossRef]
- Brenman, J.; Chao, D.; Xia, H.; Aldape, K.; Bredt, D. Nitric oxide synthase complexed with dystrophin and absent from skeletal muscle sarcolemma in Duchenne muscular dystrophy. Cell 1995, 82, 743–752. [Google Scholar] [CrossRef]
- Percival, J. nNOS regulation of skeletal muscle fatigue and exercise performance. Biophys. Rev. 2011, 3, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Lee, K.; Minhas, K.; Gonzalez, D.; Raju, S.; Tejani, A.; Li, D.; Berkowitz, D.; Hare, J. Neuronal nitric oxide synthase negatively regulates xanthine oxidoreductase inhibition of cardiac excitation-contraction coupling. Proc. Natl. Acad. Sci. USA 2004, 101, 15944–15948. [Google Scholar] [CrossRef]
- Balon, T.; Nadler, J.; Jasman, A. Evidence that nitric oxide increases glucose transport in skeletal muscle. J. Appl. Physiol. 1997, 82, 359–363. [Google Scholar] [CrossRef] [PubMed]
- Kellogg, D.; McCammon, K.; Hinchee-Rodriguez, K.; Adamo, M.; Roman, L. Neuronal nitric oxide synthase mediates insulin- and oxidative stress-induced glucose uptake in skeletal muscle myotubes. Free Radic. Biol. Med. 2017, 110, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, C.; Sánchez, G.; Barrientos, G.; Aracena-Parks, P. A Transverse Tubule NADPH Oxidase Activity Stimulates Calcium Release from Isolated Triads via Ryanodine Receptor Type 1S-Glutathionylation. J. Biol. Chem. 2006, 281, 26473–26482. [Google Scholar] [CrossRef] [PubMed]
- Adhihetty, P.; Ljubicic, V.; Menzies, K.; Hood, D. Differential susceptibility of subsarcolemmal and intermyofibrillar mitochondria to apoptotic stimuli. Am. J. Physiol. Cell Physiol. 2005, 289, C994–C1001. [Google Scholar] [CrossRef]
- Rastogi, R.; Geng, X.; Li, F.; Ding, Y. NOX Activation by Subunit Interaction and Underlying Mechanisms in Disease. Front. Cell. Neurosci. 2017, 10, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Loureiro, A.; Rêgo-Monteiro, I.; Louzada, R.; Ortenzi, V.; Aguiar, A.; Abreu, E.; Cavalcanti-de-Albuquerque, J.; Hecht, F.; Oliveira, A.; Ceccatto, V.; et al. Differential Expression of NADPH Oxidases Depends on Skeletal Muscle Fiber Type in Rats. Oxidative Med. Cell. Longev. 2016, 2016, 1–10. [Google Scholar] [CrossRef]
- Chen, K.; Pittman, R.; Popel, A. Nitric Oxide in the Vasculature: Where Does It Come from and Where Does It Go? A Quantitative Perspective. Antioxid. Redox Signal. 2008, 10, 1185–1198. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Cahill-Smith, S.; Geng, L.; Du, J.; Brooks, G.; Li, J. Aging-associated metabolic disorder induces Nox2 activation and oxidative damage of endothelial function. Free Radic. Biol. Med. 2017, 108, 940–951. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.; Heng, B.; He, W.; Shi, L.; Lai, C.; Xiao, L.; Ren, H.; Mo, S.; Su, Z. Chronic reactive oxygen species exposure inhibits glucose uptake and causes insulin resistance in C2C12 myotubes. Biochem. Biophys. Res. Commun. 2016, 478, 798–803. [Google Scholar] [CrossRef]
- Semenza, G. HIF-1: Mediator of physiological and pathophysiological responses to hypoxia. J. Appl. Physiol. 2000, 88, 1474–1480. [Google Scholar] [CrossRef]
- Kaijser, L.; Sundberg, C.; Eiken, O.; Nygren, A.; Esbjornsson, M.; Sylven, C.; Jansson, E. Muscle oxidative capacity and work performance after training under local leg ischemia. J. Appl. Physiol. 1990, 69, 785–787. [Google Scholar] [CrossRef]
- Desplanches, D.; Hoppeler, H.; Linossier, M.; Denis, C.; Claassen, H.; Dormois, D.; Lacour, J.; Geyssant, A. Effects of training in normoxia and normobaric hypoxia on human muscle ultrastructure. Pflug. Arch. Eur. J. Physiol. 1993, 425, 263–267. [Google Scholar] [CrossRef]
- Chi, W.; Gan, X.; Xiao, W.; Wang, W.; He, S. Different evolutionary patterns of hypoxia-inducible factor α (HIF-α) isoforms in the basal branches of Actinopterygii and Sarcopterygii. Febs Open Bio 2013, 3, 479–483. [Google Scholar] [CrossRef]
- Stroka, D.; Burkhardt, T.; Desbaillets, I.; Wenger, R.; Neil, D.; Bauer, C.; Gassman, M.; Candinas, D. HIF-1 is expressed in normoxic tissue and displays an organ-specific regulation under systemic hypoxia. FASEB J. 2001, 15, 2445–2453. [Google Scholar] [CrossRef]
- Ivan, M.; Kondo, K.; Yang, H.; Kim, W.; Valiando, J.; Ohh, M.; Salic, A.; Asara, J.; Lane, W.; Kaelin, W. HIFalpha Targeted for VHL-Mediated Destruction by Proline Hydroxylation: Implications for O2 Sensing. Science 2001, 292, 464–468. [Google Scholar] [CrossRef]
- Lundby, C.; Gassmann, M.; Pilegaard, H. Regular endurance training reduces the exercise induced HIF-1α and HIF-2α mRNA expression in human skeletal muscle in normoxic conditions. Eur. J. Appl. Physiol. 2005, 96, 363–369. [Google Scholar] [CrossRef]
- Capitanio, D.; Fania, C.; Torretta, E.; Viganò, A.; Moriggi, M.; Bravatà, V.; Caretti, A.; Levett, D.; Grocott, M.; Samaja, M.; et al. TCA cycle rewiring fosters metabolic adaptation to oxygen restriction in skeletal muscle from rodents and humans. Sci. Rep. 2017, 7, 9723. [Google Scholar] [CrossRef]
- Hashimoto, T.; Shibasaki, F. Hypoxia-Inducible Factor as an Angiogenic Master Switch. Front. Pediatrics 2015, 3, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Mason, S.; Howlett, R.; Kim, M.; Olfert, I.; Hogan, M.; McNulty, W.; Hickey, R.; Wagner, P.; Kahn, C.; Giordano, F.; et al. Loss of Skeletal Muscle HIF-1α Results in Altered Exercise Endurance. PLoS Biol. 2004, 2, e288. [Google Scholar] [CrossRef] [PubMed]
- Mason, S.; Rundqvist, H.; Papandreou, I.; Duh, R.; McNulty, W.; Howlett, R.; Olfert, I.; Sundberg, C.; Denko, N.; Poellinger, L.; et al. HIF-1α in endurance training: Suppression of oxidative metabolism. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 293, R2059–R2069. [Google Scholar] [CrossRef] [PubMed]
- Lindholm, M.; Fischer, H.; Poellinger, L.; Johnson, R.; Gustafsson, T.; Sundberg, C.; Rundqvist, H. Negative regulation of HIF in skeletal muscle of elite endurance athletes: A tentative mechanism promoting oxidative metabolism. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2014, 307, R248–R255. [Google Scholar] [CrossRef]
- LaGory, E.; Wu, C.; Taniguchi, C.; Ding, C.; Chi, J.; von Eyben, R.; Scott, D.; Richardson, A.; Giaccia, A. Suppression of PGC-1α Is Critical for Reprogramming Oxidative Metabolism in Renal Cell Carcinoma. Cell Rep. 2015, 12, 116–127. [Google Scholar] [CrossRef]
- Zhang, H.; Gao, P.; Fukuda, R.; Kumar, G.; Krishnamachary, B.; Zeller, K.; Dang, C.; Semenza, G. HIF-1 Inhibits Mitochondrial Biogenesis and Cellular Respiration in VHL-Deficient Renal Cell Carcinoma by Repression of C-MYC Activity. Cancer Cell 2007, 11, 407–420. [Google Scholar] [CrossRef]
- Gomes, A.; Price, N.; Ling, A.; Moslehi, J.; Montgomery, M.; Rajman, L.; White, J.; Teodoro, J.; Wrann, C.; Hubbard, B.; et al. Declining NAD+ Induces a Pseudohypoxic State Disrupting Nuclear-Mitochondrial Communication during Aging. Cell 2013, 155, 1624–1638. [Google Scholar] [CrossRef]
- Sowter, H.; Ratclife, P.; Watson, P.; Greenberg, A.; Harris, A. HIF-1-dependent regulation of hypoxic induction of the cell death factors BNIP3 and NIX in human tumors. Cancer Res. 2001, 61, 6669–6673. [Google Scholar] [PubMed]
- Namas, R.; Metukuri, M.; Dhupar, R.; Velosa, C.; Jefferson, B.; Myer, E.; Constantine, G.; Billiar, T.; Vodovotz, Y.; Zamora, R. Hypoxia-Induced Overexpression of BNIP3 is Not Dependent on Hypoxia-Inducible Factor 1α in Mouse Hepatocytes. Shock 2011, 36, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, A.; Sumi, C.; Tanaka, H.; Kusunoki, M.; Iwai, T.; Nishi, K.; Matsuo, Y.; Harada, H.; Takenaga, K.; Bono, H.; et al. HIF-1-mediated suppression of mitochondria electron transport chain function confers resistance to lidocaine-induced cell death. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhou, Y.; Li, L.; Li, S.; Long, D.; Chen, X.; Zhang, J.; Feng, L.; Li, Y. HIF-1α protects against oxidative stress by directly targeting mitochondria. Redox Biol. 2019, 25, 1–15. [Google Scholar] [CrossRef]
- Yuan, G.; Khan, S.; Luo, W.; Nanduri, J.; Semenza, G.; Prabhakar, N. Hypoxia-inducible factor 1 mediates increased expression of NADPH oxidase-2 in response to intermittent hypoxia. J. Cell. Physiol. 2011, 226, 2925–2933. [Google Scholar] [CrossRef]
- Henríquez-Olguin, C.; Knudsen, J.; Raun, S.; Li, Z.; Dalbram, E.; Treebak, J.; Sylow, L.; Holmdahl, R.; Richter, E.; Jaimovich, E.; et al. Cytosolic ROS production by NADPH oxidase 2 regulates muscle glucose uptake during exercise. Nat. Commun. 2019, 10, 1–11. [Google Scholar] [CrossRef]
- Nanduri, J.; Vaddi, D.; Khan, S.; Wang, N.; Makarenko, V.; Semenza, G.; Prabhakar, N. HIF-1α Activation by Intermittent Hypoxia Requires NADPH Oxidase Stimulation by Xanthine Oxidase. PLoS ONE 2015, 10, e0119762. [Google Scholar]
- Nanduri, J.; Vaddi, D.; Khan, S.; Wang, N.; Makerenko, V.; Prabhakar, N. Xanthine Oxidase Mediates Hypoxia-Inducible Factor-2α Degradation by Intermittent Hypoxia. PLoS ONE 2013, 8, e75838. [Google Scholar] [CrossRef]
- Rasbach, K.; Gupta, R.; Ruas, J.; Wu, J.; Naseri, E.; Estall, J.; Spiegelman, B. PGC-1 regulates a HIF2-dependent switch in skeletal muscle fiber types. Proc. Natl. Acad. Sci. USA 2010, 107, 21866–21871. [Google Scholar] [CrossRef]
- Block, K.; Gorin, Y.; Hoover, P.; Williams, P.; Chelmicki, T.; Clark, R.; Yoneda, T.; Abboud, H. NAD(P)H Oxidases Regulate HIF-2α Protein Expression. J. Biol. Chem. 2007, 282, 8019–8026. [Google Scholar] [CrossRef]
- Abe, T.; Kitaoka, Y.; Kikuchi, D.; Takeda, K.; Numata, O.; Takemasa, T. High-intensity interval training-induced metabolic adaptation coupled with an increase in Hif-1α and glycolytic protein expression. J. Appl. Physiol. 2015, 119, 1297–1302. [Google Scholar] [CrossRef] [PubMed]
- Cięszczyk, P.; Eider, J.; Arczewska, A.; Ostanek, M.; Leońska-Duniec, A.; Sawczyn, S.; Ficek, K.; Jascaniene, N.; Kotarska, K.; Sygit, K. The HIF1A Gene Pro582Ser in Polish Power-Orientated Athletes. Biol. Sport 2011, 28, 111–114. [Google Scholar] [CrossRef]
- Vyas, S.; Zaganjor, E.; Haigis, M. Mitochondria and Cancer. Cell 2016, 166, 555–566. [Google Scholar] [CrossRef] [PubMed]
- Golpich, M.; Amini, E.; Mohamed, Z.; Azman Ali, R.; Mohamed Ibrahim, N.; Ahmadiani, A. Mitochondrial Dysfunction and Biogenesis in Neurodegenerative diseases: Pathogenesis and Treatment. CNS Neurosci. Ther. 2016, 23, 5–22. [Google Scholar] [CrossRef]
- Jang, J.; Blum, A.; Liu, J.; Finkel, T. The role of mitochondria in aging. J. Clin. Investig. 2018, 128, 3662–3670. [Google Scholar] [CrossRef]
- Baldelli, S.; Aquilano, K.; Ciriolo, M. Punctum on two different transcription factors regulated by PGC-1α: Nuclear factor erythroid-derived 2-like 2 and nuclear respiratory factor 2. Biochim. Biophys. Acta (BBA) Gen. Subj. 2013, 1830, 4137–4146. [Google Scholar] [CrossRef]
- Rowe, G.; El-Khoury, R.; Patten, I.; Rustin, P.; Arany, Z. PGC-1α is Dispensable for Exercise-Induced Mitochondrial Biogenesis in Skeletal Muscle. PLoS ONE 2012, 7, e41817. [Google Scholar] [CrossRef]
- Virbasius, J.; Scarpulla, R. Activation of the human mitochondrial transcription factor A gene by nuclear respiratory factors: A potential regulatory link between nuclear and mitochondrial gene expression in organelle biogenesis. Proc. Natl. Acad. Sci. USA 1994, 91, 1309–1313. [Google Scholar] [CrossRef]
- Ohtsuji, M.; Katsuoka, F.; Kobayashi, A.; Aburatani, H.; Hayes, J.; Yamamoto, M. Nrf1 and Nrf2 Play Distinct Roles in Activation of Antioxidant Response Element-dependent Genes. J. Biol. Chem. 2008, 283, 33554–33562. [Google Scholar] [CrossRef]
- Ruttkay-Nedecky, B.; Nejdl, L.; Gumulec, J.; Zitka, O.; Masarik, M.; Eckschlager, T.; Stiborova, M.; Adam, V.; Kizek, R. The Role of Metallothionein in Oxidative Stress. Int. J. Mol. Sci. 2013, 14, 6044–6066. [Google Scholar] [CrossRef]
- Satoh, J.; Kawana, N.; Yamamoto, Y. Pathway Analysis of ChIP-Seq-Based NRF1 Target Genes Suggests a Logical Hypothesis of their Involvement in the Pathogenesis of Neurodegenerative Diseases. Gene Regul. Syst. Biol. 2013, 7, GRSB.S13204. [Google Scholar] [CrossRef] [PubMed]
- Piantadosi, C.; Carraway, M.; Babiker, A.; Suliman, H. Heme Oxygenase-1 Regulates Cardiac Mitochondrial Biogenesis via Nrf2-Mediated Transcriptional Control of Nuclear Respiratory Factor-1. Circ. Res. 2008, 103, 1232–1240. [Google Scholar] [CrossRef] [PubMed]
- Whitman, S.; Long, M.; Wondrak, G.; Zheng, H.; Zhang, D. Nrf2 modulates contractile and metabolic properties of skeletal muscle in streptozotocin-induced diabetic atrophy. Exp. Cell Res. 2013, 319, 2673–2683. [Google Scholar] [CrossRef]
- Aquilano, K.; Baldelli, S.; Pagliei, B.; Cannata, S.; Rotilio, G.; Ciriolo, M. p53 Orchestrates the PGC-1α-Mediated Antioxidant Response Upon Mild Redox and Metabolic Imbalance. Antioxid. Redox Signal. 2013, 18, 386–399. [Google Scholar] [CrossRef]
- Zhang, D.; Hannink, M. Distinct Cysteine Residues in Keap1 Are Required for Keap1-Dependent Ubiquitination of Nrf2 and for Stabilization of Nrf2 by Chemopreventive Agents and Oxidative Stress. Mol. Cell. Biol. 2003, 23, 8137–8151. [Google Scholar] [CrossRef]
- Kansanen, E.; Kuosmanen, S.; Leinonen, H.; Levonen, A. The Keap1-Nrf2 pathway: Mechanisms of activation and dysregulation in cancer. Redox Biol. 2013, 1, 45–49. [Google Scholar] [CrossRef]
- Dinkova-Kostova, A.; Abramov, A. The emerging role of Nrf2 in mitochondrial function. Free Radic. Biol. Med. 2015, 88, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.; Barger, J.; Edwards, M.; Braun, K.; O’Connor, C.; Prolla, T.; Weindruch, R. Dynamic regulation of PGC-1α localization and turnover implicates mitochondrial adaptation in calorie restriction and the stress response. Aging Cell 2008, 7, 101–111. [Google Scholar] [CrossRef]
- Hayes, J.; Chowdhry, S.; Dinkova-Kostova, A.; Sutherland, C. Dual regulation of transcription factor Nrf2 by Keap1 and by the combined actions of β-TrCP and GSK-3. Biochem. Soc. Trans. 2015, 43, 611–620. [Google Scholar] [CrossRef] [PubMed]
- Kitagishi, Y.; Matsuda, S. Redox regulation of tumor suppressor PTEN in cancer and aging. Int. J. Mol. Med. 2013, 31, 511–515. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Kim, H.; Park, J.; Kim, I.; Bae, E.; Ma, S.; Kim, S. PGC-1α attenuates hydrogen peroxide-induced apoptotic cell death by upregulating Nrf-2 via GSK3β inactivation mediated by activated p38 in HK-2 Cells. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Gureev, A.; Shaforostova, E.; Popov, V. Regulation of Mitochondrial Biogenesis as a Way for Active Longevity: Interaction between the Nrf2 and PGC-1α Signaling Pathways. Front. Genet. 2019, 10, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hota, K.; Hota, S.; Chaurasia, O.; Singh, S. Acetyl-L-carnitine-mediated neuroprotection during hypoxia is attributed to ERK1/2-Nrf2-regulated mitochondrial biosynthesis. Hippocampus 2011, 22, 723–736. [Google Scholar] [CrossRef] [PubMed]
- Faubert, B.; Vincent, E.; Griss, T.; Samborska, B.; Izreig, S.; Svensson, R.; Mamer, O.; Avizonis, D.; Shackelford, D.; Shaw, R.; et al. Loss of the tumor suppressor LKB1 promotes metabolic reprogramming of cancer cells via HIF-1. Proc. Natl. Acad. Sci. USA 2014, 111, 2554–2559. [Google Scholar] [CrossRef]
- Taylor, E.; Lamb, J.; Hurst, R.; Chesser, D.; Ellingson, W.; Greenwood, L.; Porter, B.; Herway, S.; Winder, W. Endurance training increases skeletal muscle LKB1 and PGC-1α protein abundance: Effects of time and intensity. Am. J. Physiol. Endocrinol. Metab. 2005, 289, E960–E968. [Google Scholar] [CrossRef]
- Cerda-Kohler, H.; Henríquez-Olguín, C.; Casas, M.; Jensen, T.; Llanos, P.; Jaimovich, E. Lactate administration activates the ERK1/2, mTORC1, and AMPK pathways differentially according to skeletal muscle type in mouse. Physiol. Rep. 2018, 6, e13800. [Google Scholar] [CrossRef]
- Merry, T.; Ristow, M. Nuclear factor erythroid-derived 2-like 2 (NFE2L2, Nrf2) mediates exercise-induced mitochondrial biogenesis and the anti-oxidant response in mice. J. Physiol. 2016, 594, 5195–5207. [Google Scholar] [CrossRef]
- Żołądź, J.; Koziel, A.; Broniarek, I.; Woyda-Ploszczyca, A.; Ogrodna, K.; Majerczak, J.; Celichowski, J.; Szkutnik, Z.; Jarmuszkiewicz, W. Effect of temperature on fatty acid metabolism in skeletal muscle mitochondria of untrained and endurance-trained rats. PLoS ONE 2017, 12, e0189456. [Google Scholar] [CrossRef]
- Aguiar, A.; Duzzioni, M.; Remor, A.; Tristão, F.; Matheus, F.; Raisman-Vozari, R.; Latini, A.; Prediger, R. Moderate-Intensity Physical Exercise Protects Against Experimental 6-Hydroxydopamine-Induced Hemiparkinsonism Through Nrf2-Antioxidant Response Element Pathway. Neurochem. Res. 2015, 41, 64–72. [Google Scholar] [CrossRef]
- Gkotinakou, I.; Befani, C.; Simos, G.; Liakos, P. ERK1/2 phosphorylates HIF-2α and regulates its activity by controlling its CRM1-dependent nuclear shuttling. J. Cell Sci. 2019, 132, jcs225698. [Google Scholar] [CrossRef] [PubMed]
- Jin, W.; Wu, J.; Wang, H.; Kong, J.; Ni, H.; Liang, W. Erythropoietin Administration Modulates Pulmonary Nrf2 Signaling Pathway After Traumatic Brain Injury in Mice. J. Trauma Inj. Infect. Crit. Care 2011, 71, 680–686. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Zhao, J.; Chen, M.; Wang, H.; Yao, Q.; Fan, J.; Zhang, M. The Anti-Aging Effect of Erythropoietin via the ERK/Nrf2-ARE Pathway in Aging Rats. J. Mol. Neurosci. 2017, 61, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Zhang, X.; Yu, Y.; Bian, X.; Dong, S. Cardioprotective effect of erythropoietin on sepsis-induced myocardial injury in rats. World J. Emerg. Med. 2013, 4, 215. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, L.; Jia, Y.; Rogers, H.; Suzuki, N.; Gassmann, M.; Wang, Q.; McPherron, A.; Kopp, J.; Yamamoto, M.; Noguchi, C. Erythropoietin contributes to slow oxidative muscle fiber specification via PGC-1α and AMPK activation. Int. J. Biochem. Cell Biol. 2013, 45, 1155–1164. [Google Scholar] [CrossRef]
- Harman, D. Origin and evolution of the free radical theory of aging: A brief personal history, 1954–2009. Biogerontology 2009, 10, 773–781. [Google Scholar] [CrossRef]
- Webb, A.; Brunet, A. FOXO transcription factors: Key regulators of cellular quality control. Trends Biochem. Sci. 2014, 39, 159–169. [Google Scholar] [CrossRef]
- Sanchez, A.; Bernardi, H.; Py, G.; Candau, R. Autophagy is essential to support skeletal muscle plasticity in response to endurance exercise. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2014, 307, R956–R969. [Google Scholar] [CrossRef]
- Martin-Rincon, M.; Morales-Alamo, D.; Calbet, J. Exercise-mediated modulation of autophagy in skeletal muscle. Scand. J. Med. Sci. Sports 2017, 28, 772–781. [Google Scholar] [CrossRef]
- Drake, J.; Wilson, R.; Yan, Z. Molecular mechanisms for mitochondrial adaptation to exercise training in skeletal muscle. FASEB J. 2015, 30, 13–22. [Google Scholar] [CrossRef]
- Fang, E.; Waltz, T.; Kassahun, H.; Lu, Q.; Kerr, J.; Morevati, M.; Fivenson, E.; Wollman, B.; Marosi, K.; Wilson, M.; et al. Tomatidine enhances lifespan and healthspan in C. elegans through mitophagy induction via the SKN-1/Nrf2 pathway. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef]
- Song, Z.; Moore, D.; Hodson, N.; Ward, C.; Dent, J.; O’Leary, M.; Shaw, A.; Hamilton, D.; Sarkar, S.; Gangloff, Y.; et al. Resistance exercise initiates mechanistic target of rapamycin (mTOR) translocation and protein complex co-localisation in human skeletal muscle. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Bonuccelli, G.; Tsirigos, A.; Whitaker-Menezes, D.; Pavlides, S.; Pestell, R.; Chiavarina, B.; Frank, P.; Flomenberg, N.; Howell, A.; Martinez-Outschoorn, U.; et al. Ketones and lactate “fuel” tumor growth and metastasis. Cell Cycle 2010, 9, 3506–3514. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Outschoorn, U.; Lin, Z.; Trimmer, C.; Flomenberg, N.; Wang, C.; Pavlides, S.; Pestell, R.; Howell, A.; Sotgia, F.; Lisanti, M. Cancer cells metabolically “fertilize” the tumor microenvironment with hydrogen peroxide, driving the Warburg effect. Cell Cycle 2011, 10, 2504–2520. [Google Scholar] [CrossRef]
- Fu, Y.; Liu, S.; Yin, S.; Niu, W.; Xiong, W.; Tan, M.; Li, G.; Zhou, M. The reverse Warburg effect is likely to be an Achilles’ heel of cancer that can be exploited for cancer therapy. Oncotarget 2017, 8, 57813–57825. [Google Scholar] [CrossRef]
- Tauffenberger, A.; Fiumelli, H.; Almustafa, S.; Magistretti, P. Lactate and pyruvate promote oxidative stress resistance through hormetic ROS signaling. Cell Death Dis. 2019, 10, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Lacher, S.; Levings, D.; Freeman, S.; Slattery, M. Identification of a functional antioxidant response element at the HIF1A locus. Redox Biol. 2018, 19, 401–411. [Google Scholar] [CrossRef] [PubMed]
- Archibald, J. Endosymbiosis and Eukaryotic Cell Evolution. Curr. Biol. 2015, 25, R911–R921. [Google Scholar] [CrossRef]
- Laker, R.; Lillard, T.; Okutsu, M.; Zhang, M.; Hoehn, K.; Connelly, J.; Yan, Z. Exercise Prevents Maternal High-Fat Diet–Induced Hypermethylation of thePgc-1α Gene and Age-Dependent Metabolic Dysfunction in the Offspring. Diabetes 2014, 63, 1605–1611. [Google Scholar] [CrossRef] [PubMed]
- Longo, M.; Refuerzo, J.; Mann, L.; Leon, M.; Moussa, H.; Sibai, B.; Blackwell, S. Adverse Effect of High-Fat Diet on Metabolic Programming in Offspring Born to a Murine Model of Maternal Hypertension. Am. J. Hypertens. 2016, 29, 1366–1373. [Google Scholar] [CrossRef] [PubMed]
- Stanford, K.; Rasmussen, M.; Baer, L.; Lehnig, A.; Rowland, L.; White, J.; So, K.; De Sousa-Coelho, A.; Hirshman, M.; Patti, M.; et al. Paternal Exercise Improves Glucose Metabolism in Adult Offspring. Diabetes 2018, 67, 2530–2540. [Google Scholar] [CrossRef] [PubMed]
- Engineer, A.; Saiyin, T.; Greco, E.; Feng, Q. Say NO to ROS: Their Roles in Embryonic Heart Development and Pathogenesis of Congenital Heart Defects in Maternal Diabetes. Antioxidants 2019, 8, 436. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Alves-Wagner, A.; Stanford, K.; Prince, N.; So, K.; Mul, J.; Dirice, E.; Hirshman, M.; Kulkarni, R.; Goodyear, L. Maternal and paternal exercise regulate offspring metabolic health and beta cell phenotype. Bmj Open Diabetes Res. Care 2020, 8, e000890. [Google Scholar] [CrossRef] [PubMed]
- Sies, H. Oxidative eustress: On constant alert for redox homeostasis. Redox Biol. 2021, 41, 101867. [Google Scholar] [CrossRef]
- Silva, L.; Tromm, C.; Doyenart, R.; Thirupathi, A.; Silveira, P.; Pinho, R. Effects of different frequencies of physical training on electron transport chain and oxidative damage in healthy mice. Mot. Rev. Educ. Física 2018, 24. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kolodziej, F.; O’Halloran, K.D. Re-Evaluating the Oxidative Phenotype: Can Endurance Exercise Save the Western World? Antioxidants 2021, 10, 609. https://doi.org/10.3390/antiox10040609
Kolodziej F, O’Halloran KD. Re-Evaluating the Oxidative Phenotype: Can Endurance Exercise Save the Western World? Antioxidants. 2021; 10(4):609. https://doi.org/10.3390/antiox10040609
Chicago/Turabian StyleKolodziej, Filip, and Ken D. O’Halloran. 2021. "Re-Evaluating the Oxidative Phenotype: Can Endurance Exercise Save the Western World?" Antioxidants 10, no. 4: 609. https://doi.org/10.3390/antiox10040609
APA StyleKolodziej, F., & O’Halloran, K. D. (2021). Re-Evaluating the Oxidative Phenotype: Can Endurance Exercise Save the Western World? Antioxidants, 10(4), 609. https://doi.org/10.3390/antiox10040609





