The Herbal Formula JI017 Induces ER Stress via Nox4 in Breast Cancer Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. JI017 Extraction
2.2. Reagents
2.3. Cell Cultures
2.4. Cell Viability
2.5. LDH Assay
2.6. Caspase-3 Activity Assays
2.7. Intracellular Ca2+ Assays
2.8. Intracellular ROS Assays
2.9. Establishment of Paclitaxel-Resistant MCF-7 and MDA-MB-231 Cell Lines
2.10. Colony Formation Assay
2.11. Transfection
2.12. Isolation of Total RNA and Protein
2.13. Real-Time PCR and Western Blot Analyses
2.14. Exosome Isolation
2.15. Animals
2.16. Tumor Xenograft Mouse Models
2.17. Statistical Analysis
3. Results
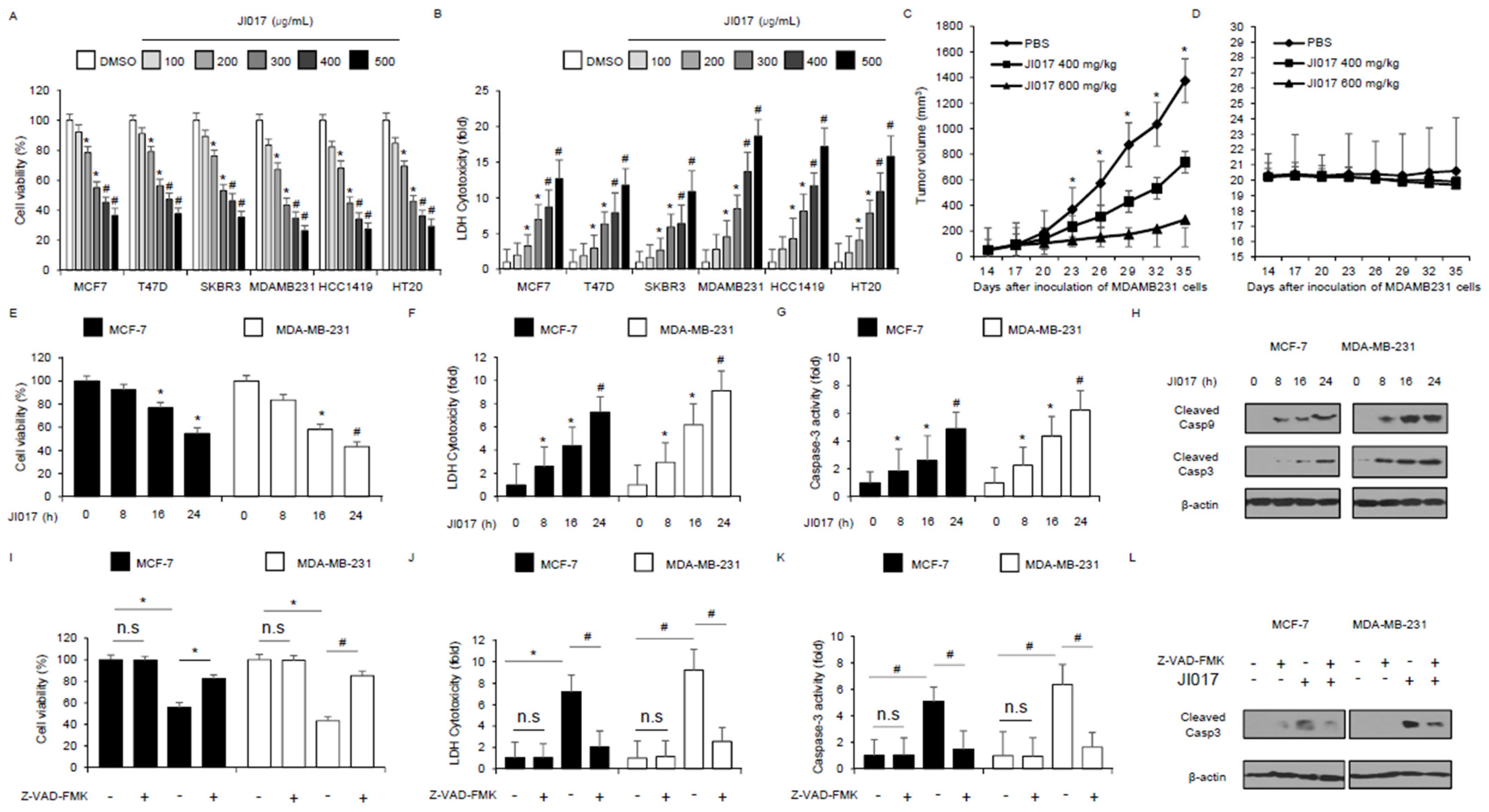
3.1. Anticancer Effects of JI017 In Vitro and In Vivo in Breast Cancer Cells
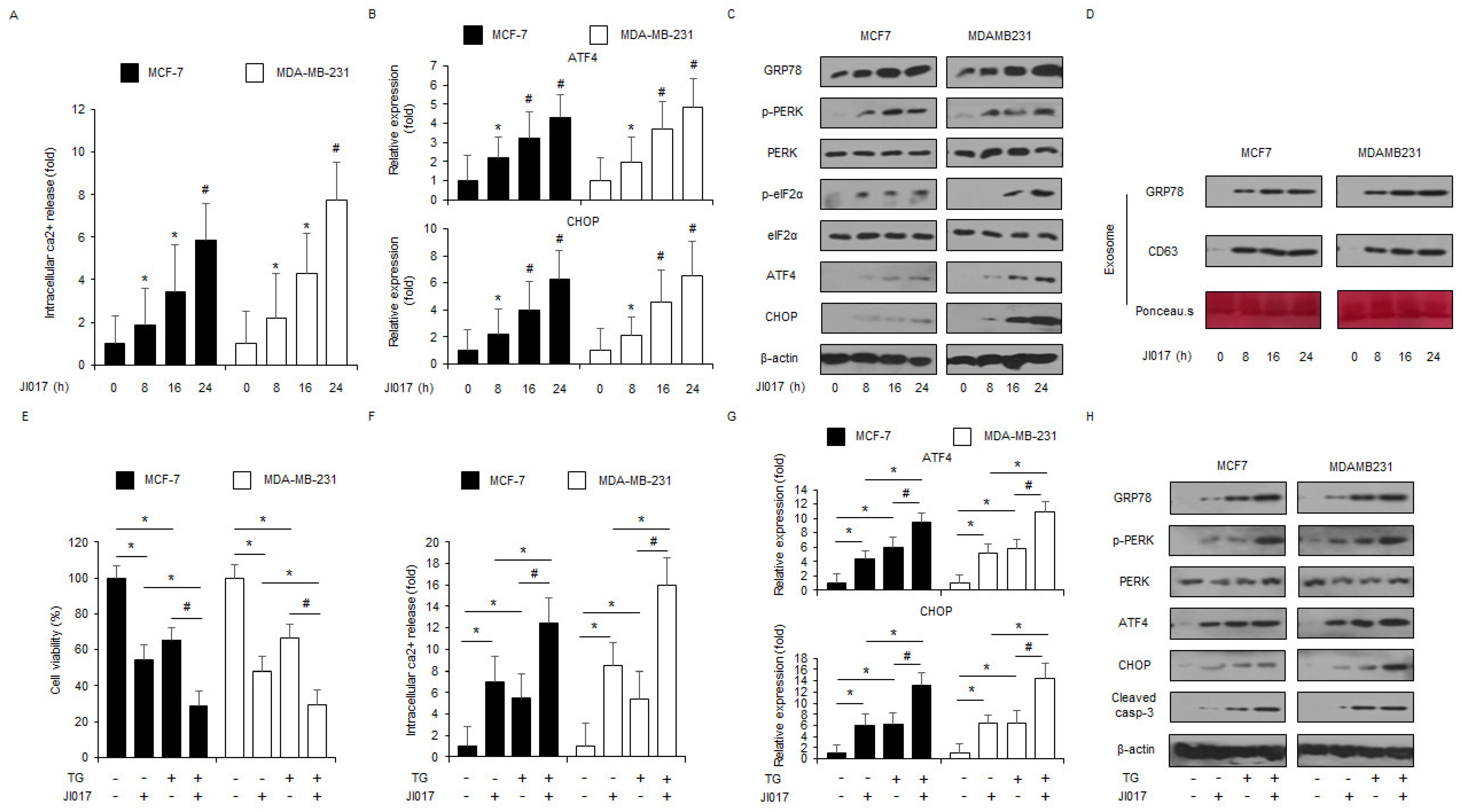
3.2. JI017 Treatment Mediates ER Stress Response in Breast Cancer Cells
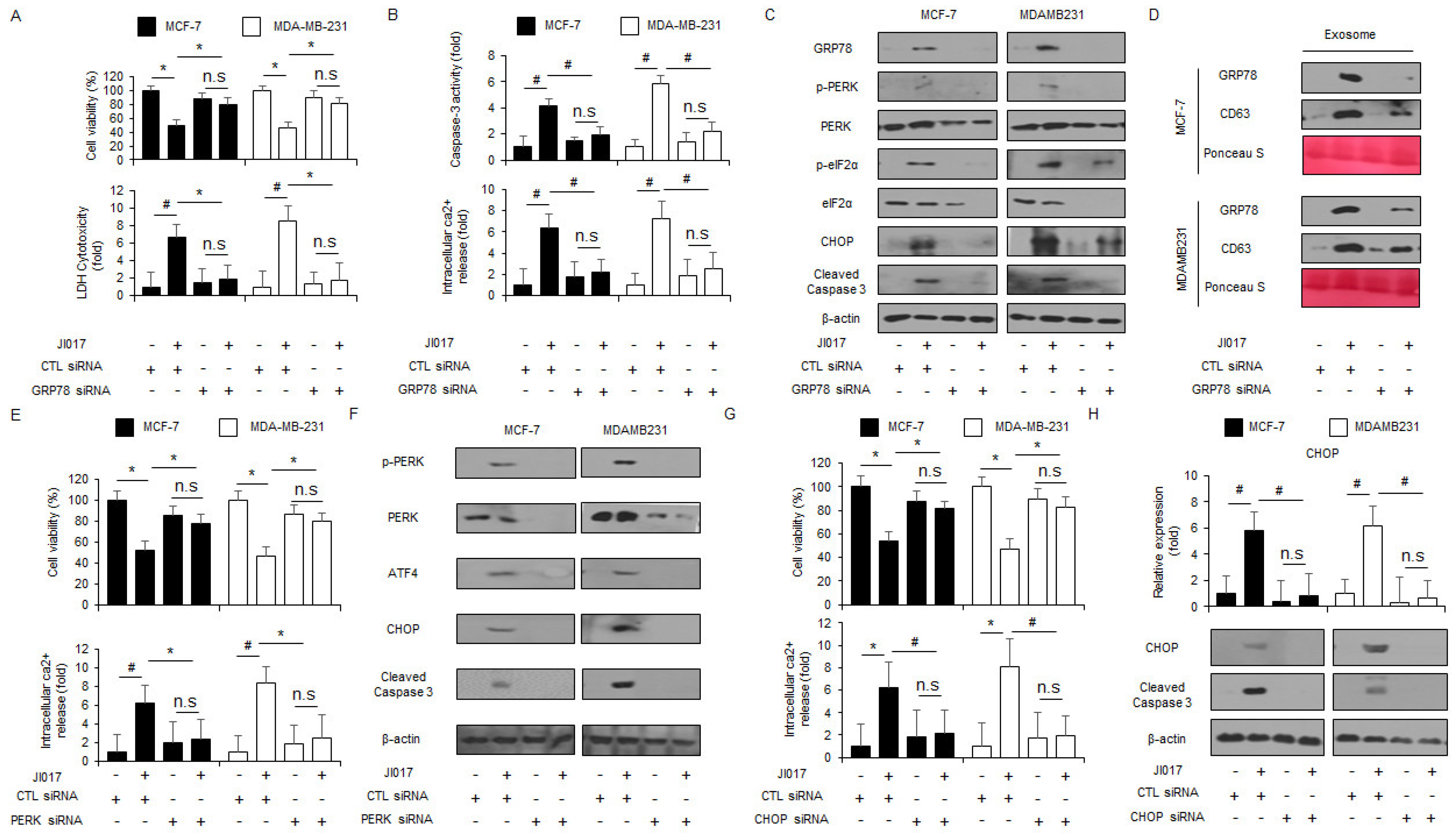
3.3. Targeting ER Stress Suppresses JI017-Mediated Cell Death in Breast Cancer Cells
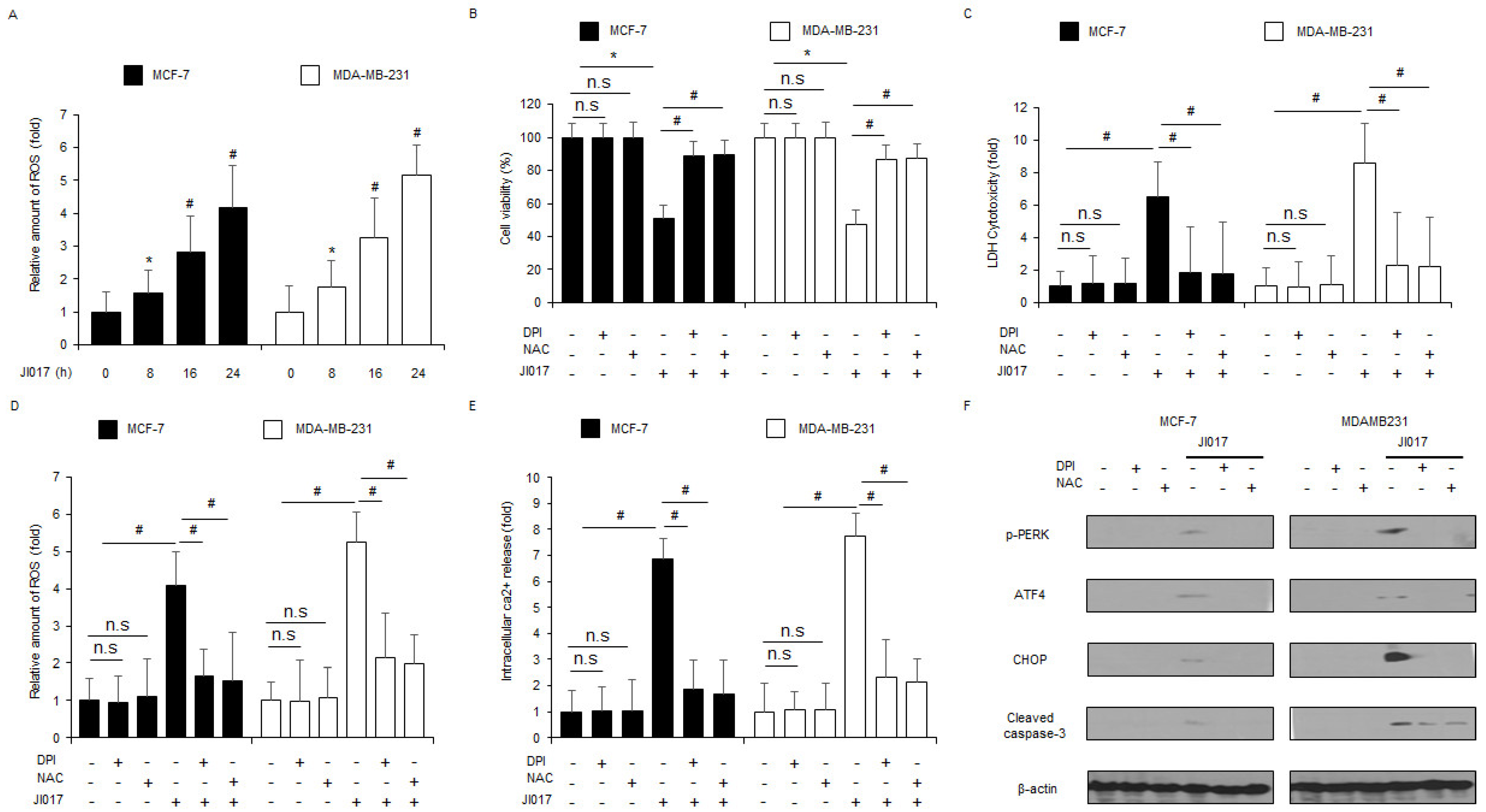
3.4. JI017 Treatment Mediates ER Stress and Cell Death by Releasing ROS in Breast Cancer Cells
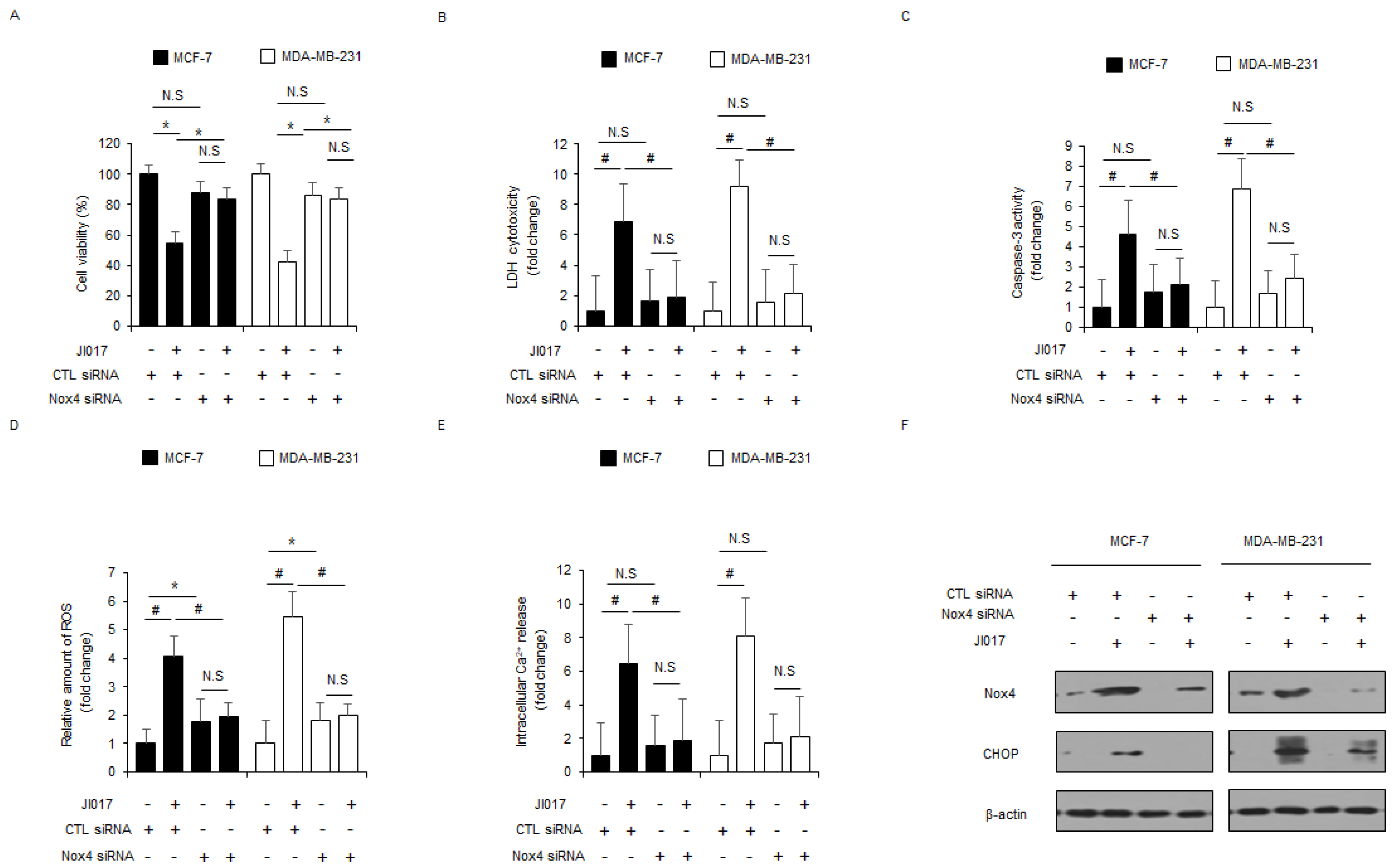
3.5. JI017 Induces ER Stress and Apoptosis via Nox4 and ROS Release in Breast Cancer Cells
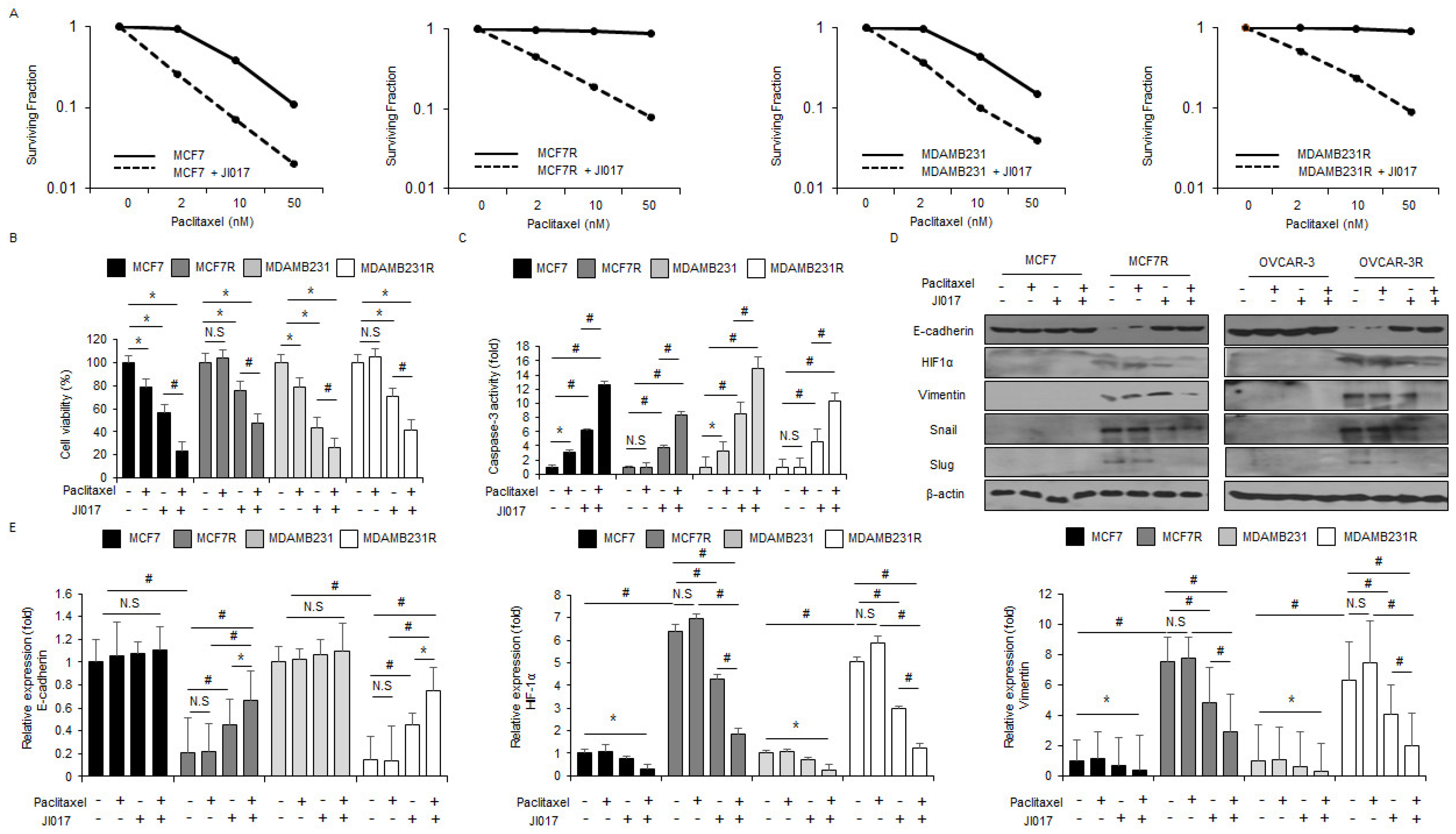
3.6. Paclitaxel in Combination with JI017 Overcome Paclitaxel Resistance by Blocking EMT in Paclitaxel-Resistant Breast Cancer Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Azamjah, N.; Soltan-Zadeh, Y.; Zayeri, F. Global Trend of Breast Cancer Mortality Rate: A 25-Year Study. Asian-Pac. J. Cancer Prev. 2019, 20, 2015–2020. [Google Scholar] [CrossRef]
- Sharma, G.N.; Dave, R.; Sanadya, J.; Sharma, P.; Sharma, K.K. Various types and management of breast cancer: An overview. J Adv. Pharm. Technol. Res. 2010, 1, 109–126. [Google Scholar]
- Posner, M.C.; Wolmark, N. Non-invasive breast carcinoma. Breast Cancer Res. Treat. 1992, 21, 155–164. [Google Scholar] [CrossRef]
- Bianchini, G.; Balko, J.M.; Mayer, I.A.; Sanders, M.E.; Gianni, L. Triple-negative breast cancer: Challenges and opportunities of a heterogeneous disease. Nat. Rev. Clin. Oncol. 2016, 13, 674–690. [Google Scholar] [CrossRef] [PubMed]
- Waks, A.G.; Winer, E.P. Breast Cancer Treatment: A Review. JAMA 2019, 321, 288–300. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, H.; Chen, X. Drug resistance and combating drug resistance in cancer. Cancer Drug Resist. 2019, 2, 141–160. [Google Scholar] [CrossRef] [Green Version]
- Kim, B.M.; Hong, Y.K.; Lee, S.H.; Liu, P.D.; Lim, J.H.; Lee, Y.H.; Lee, T.H.; Chang, K.T.; Hong, Y.G. Therapeutic Implications for Overcoming Radiation Resistance in Cancer Therapy. Int. J. Mol. Sci. 2015, 16, 26880–26913. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.; Khor, T.O.; Shu, L.; Su, Z.Y.; Fuentes, F.; Lee, J.H.; Kong, A.N. Plants vs. cancer: A review on natural phytochemicals in preventing and treating cancers and their druggability. Anticancer Agents Med. Chem. 2012, 12, 1281–1305. [Google Scholar] [CrossRef]
- Barbuti, A.M.; Chen, Z.S. Paclitaxel Through the Ages of Anticancer Therapy: Exploring Its Role in Chemoresistance and Radiation Therapy. Cancers 2015, 7, 2360–2371. [Google Scholar] [CrossRef]
- Yang, Q.; Huang, J.; Wu, Q.; Cai, Y.; Zhu, L.; Lu, X.; Chen, S.; Chen, C.; Wang, Z. Acquisition of epithelial–mesenchymal transition is associated with Skp2 expression in paclitaxel-resistant breast cancer cells. Br. J. Cancer 2014, 110, 1958–1967. [Google Scholar] [CrossRef] [Green Version]
- Seo, H.S.; Ku, J.M.; Lee, H.J.; Woo, J.K.; Cheon, C.H.; Kim, M.A.; Jang, B.H.; Shin, Y.C.; Ko, S.G. SH003 reverses drug resistance by blocking signal transducer and activator of transcription 3 (STAT3) signaling in breast cancer cells. Biosci. Rep. 2017, 37, BSR20170125. [Google Scholar] [CrossRef] [Green Version]
- Zhu, M.L.; Li, J.C.; Wang, L.; Zhong, X.; Zhang, Y.W.; Tan, R.Z.; Wang, H.L.; Fan, J.M.; Wang, L. Decursin inhibits the growth of HeLa cervical cancer cells through PI3K/Akt signaling. J. Asian Nat. Prod. Res. 2021, 23, 584–595. [Google Scholar] [CrossRef]
- Ge, Y.; Yoon, S.H.; Jang, H.; Jeong, J.H.; Lee, Y.M. Decursin promotes HIF-1α proteasomal degradation and immune responses in hypoxic tumour microenvironment. Phytomedicine 2020, 78, 153318. [Google Scholar] [CrossRef]
- Oh, S.T.; Lee, S.; Hua, C.; Koo, B.S.; Pak, S.C.; Kim, D.I.; Jeon, S.; Shin, B.A. Decursin induces apoptosis in glioblastoma cells, but not in glial cells via a mitochondria-related caspase pathway. Korean J. Physiol. Pharmacol. 2019, 23, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Kweon, B.; Han, Y.H.; Kee, J.Y.; Mun, J.G.; Jeon, H.D.; Yoon, D.H.; Choi, B.M.; Hong, S.H. Effect of Angelica gigas Nakai Ethanol Extract and Decursin on Human Pancreatic Cancer Cells. Molecules 2020, 25, 2028. [Google Scholar] [CrossRef]
- Chen, S.T.; Lee, T.Y.; Tsai, T.H.; Huang, Y.C.; Lin, Y.C.; Lin, C.P.; Shieh, H.R.; Hsu, M.L.; Chi, C.W.; Lee, M.C.; et al. The Traditional Chinese Medicine DangguiBuxue Tang Sensitizes Colorectal Cancer Cells to Chemoradiotherapy. Oncotarget 2017, 8, 88563–88574. [Google Scholar] [CrossRef] [Green Version]
- Shukla, Y.; Singh, M. Cancer preventive properties of ginger: A brief review. Food Chem. Toxicol. 2007, 45, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Jeyakumar, S.M.; Nalini, N.; Menon, V.P. Antioxidant activity of ginger (Zingiber officinale) in rats fed a high fat diet. Med. Sci. Res. 1999, 27, 341–344. [Google Scholar]
- Huang, S.; DeGuzman, A.; Bucana, C.D.; Fidler, I.J. Nuclear factor-kappaB activity correlates with growth, angiogenesis, and metastasis of human melanoma cells in nude mice. Clin. Cancer Res. 2000, 6, 2573–2581. [Google Scholar]
- Lee, H.S.; Seo, E.Y.; Kang, N.E.; Kim, W.K. [6]-Gingerol inhibits metastasis of MDA-MB-231 human breast cancer cells. J. Nutr. Biochem. 2008, 19, 313–319. [Google Scholar] [CrossRef]
- Nipin, S.P.; Kang, D.Y.; Lee, J.M.; Bae, S.W.; Jang, K.J. Potential antitumor effect of 6-gingerolin p53-dependent mitochondrial apoptosis and inhibition of tumor sphere formation in breast cancer cells. Int. J. Mol. Sci. 2021, 22, 4660. [Google Scholar]
- Akimoto, M.; Iizuka, M.; Kanematsu, R.; Yoshida, M.; Takenaga, K. Anticancer Effect of Ginger Extract against Pancreatic Cancer Cells Mainly through Reactive Oxygen Species-Mediated Autotic Cell Death. PLoS ONE 2015, 10, e0126605. [Google Scholar] [CrossRef] [PubMed]
- Hamza, A.A.; Heeba, G.H.; Hamza, S.; Abdalla, A.; Amin, A. Standardized extract of ginger ameliorates liver cancer by reducing proliferation and inducing apoptosis through inhibition oxidative stress/inflammation pathway. Biomed. Pharmacother. 2021, 134, 111102. [Google Scholar] [CrossRef]
- Nedungadi, D.; Binoy, A.; Vinod, V.; Vanuopadath, M.; Nair, S.S.; Nair, B.G.; Mishra, N. Ginger extract activates caspase independent paraptosis in cancer cells via ER stress, mitochondrial dysfunction, AIF translocation and DNA damage. Nutr. Cancer 2021, 73, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Nemoto, Y.; Satoh, K.; Toriizuka, K.; Hirai, Y.; Tobe, T.; Sakagami, H.; Nakashima, H.; Ida, Y. Cytotoxic and radical scavenging activity of blended herbal extracts. In Vivo 2002, 16, 327–332. [Google Scholar] [PubMed]
- Aumeeruddy, M.Z.; Mahomoodally, M.F. Combating breast cancer using combination therapy with 3 phytochemicals: Piperine, sulforaphane, and thymoquinone. Cancer 2019, 125, 1600–1611. [Google Scholar] [CrossRef]
- Tang, L.; Wei, F.; Wu, Y.F.; He, Y.; Shi, L.; Xiong, F.; Gong, Z.J.; Guo, C.; Li, X.; Deng, H.; et al. Role of metabolism in cancer cell radioresistance and radiosensitization methods. J. Exp. Clin. Cancer Res. 2018, 37, 87. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Tang, Y.; Xiong, F.; He, Y.; Wei, F.; Zhang, S.; Guo, C.; Xiang, B.; Zhou, M.; Xie, N.; et al. LncRNAs regulate cancer metastasis via binding to functional proteins. Oncotarget 2017, 9, 1426–1443. [Google Scholar] [CrossRef]
- Quennet, V.; Yaromina, A.; Zips, D.; Rosner, A.; Walenta, S.; Baumann, M.; Mueller-Klieser, W. Tumor lactate content predicts for response to fractionated irradiation of human squamous cell carcinomas in nude mice. Radiother. Oncol. 2006, 81, 130–135. [Google Scholar] [CrossRef]
- Tang, Y.; He, Y.; Zhang, P.; Wang, J.; Fan, C.; Yang, L.; Xiong, F.; Zhang, S.; Gong, Z.; Nie, S.; et al. LncRNAs regulate the cytoskeleton and related Rho/ROCK signaling in cancer metastasis. Mol. Cancer 2018, 17, 77. [Google Scholar] [CrossRef] [Green Version]
- Tabas, I.; Ron, D. Interating the mechanisms of apoptosis induced by endoplasmic reticulum stress. Nat. Cell Biol. 2011, 13, 183–190. [Google Scholar] [CrossRef]
- Bettigole, S.E.; Glimcher, L.H. Endoplasmic reticulum stress in immunity. Annu. Rev. Immunol. 2015, 33, 107–138. [Google Scholar] [CrossRef] [PubMed]
- Yamamori, T.; Meike, S.; Nagane, M.; Yasui, H.; Inanami, O. ER stress suppresses DNA double-strand break repair and sensitizes tumor cells to ionizing radiation by stimulating proteasomal degradation of Rad51. FEBS Lett. 2013, 587, 3348–3353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bahar, E.; Kim, H.S.; Yoon, H.N. ER stress-mediated signaling: Action potential and Ca2+ as key players. Int. J. Mol. Sci. 2016, 17, 1558. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Li, H.Z.; Ren, G.S. Epithelial-mesenchymal transition and drug resistance in breast cancer (Review). Int. J. Oncol. 2015, 47, 840–848. [Google Scholar] [CrossRef] [Green Version]
- Kim, C.M.; Kim, B.L. Anti-cancer natural products and their bioactive compounds inducing ER stress-mediated apoptosis: A review. Nutrients 2018, 10, 1021. [Google Scholar] [CrossRef] [Green Version]
- Fan, C.; Yang, Y.; Liu, Y.; Jiang, S.; Di, S.; Hu, W.; Ma, Z.; Li, T.; Zhu, Y.; Xin, Z.; et al. Icariin displays anticancer activity against human esophageal cancer cells via regulating endoplasmic reticulum stress-mediated apoptotic signaling. Sci. Rep. 2016, 6, 21145. [Google Scholar] [CrossRef] [Green Version]
- Xipell, E.; Aragón, T.; Martínez-Velez, N.; Vera, B.; Idoate, M.A.; Martínez-Irujo, J.J.; Garzón, A.G.; Gonzalez-Huarriz, M.; Acanda, A.M.; Jones, C.; et al. Endoplasmic reticulum stress-inducing drugs sensitize glioma cells to temozolomide through downregulation of MGMT, MPG, and Rad51. Neuro-Oncology 2016, 18, 1109–1119. [Google Scholar] [CrossRef] [Green Version]
- Sehgal, P.; Szalai, P.; Olesen, C.; Praetorius, H.A.; Nissen, P.; Christensen, S.B.; Engedal, N.; Møller, J.V. Inhibition of the sarco/endoplasmic reticulum (ER) Ca2+-ATPase by thapsigargin analogs induces cell death via ER Ca2+ depletion and the unfolded protein response. J. Biol. Chem. 2017, 292, 19656–19673. [Google Scholar] [CrossRef] [Green Version]
- Yang, L.; Wang, P.; Wang, H.; Li, Q.; Teng, H.; Liu, Z.; Yang, W.; Hou, L.; Zou, X. Fucoidan derived from Undaria pinnatifida induces apoptosis in human hepatocellular carcinoma SMMC-7721 cells via the ROS-mediated mitochondrial pathway. Mar. Drugs 2013, 11, 1961–1976. [Google Scholar] [CrossRef] [Green Version]
- Choi, A.Y.; Choi, J.H.; Hwang, K.Y.; Jeong, Y.J.; Choe, W.; Yoon, K.S.; Ha, J.; Kim, S.S.; Youn, J.H.; Yeo, E.J.; et al. Licochalcone A induces apoptosis through endoplasmic reticulum stress via a phospholipase Cγ1-, Ca(2+)-, and reactive oxygen species-dependent pathway in HepG2 human hepatocellular carcinoma cells. Apoptosis 2014, 19, 682–697. [Google Scholar] [CrossRef]
- Wang, M.; Kaufman, R.J. The impact of the endoplasmic reticulum protein-folding environment on cancer development. Nat. Rev. Cancer 2014, 14, 581–597. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Siegel, D.; Newsome, J.; Chilloux, A.; Moody, C.J.; Ross, D. Antitumor indolequinones induced apoptosis in human pancreatic cancer cells via inhibition of thioredoxin reductase and activation of redox signaling. Mol. Pharmacol. 2012, 81, 401–410. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weyemi, U.; Caillou, B.; Talbot, M.; Ameziane-El-Hassani, R.; Lacroix, L.; Lagent-Chevallier, O.; Al Ghuzlan, A.; Roos, D.; Bidart, J.M.; Virion, A. Intracellular expression of reactive oxygen species-generating NADPH oxidase NOX4 in normal and cancer thyroid tissues. Endocr. Relat. Cancer 2010, 17, 27–37. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, T.W.; Ko, S.-G. The Herbal Formula JI017 Induces ER Stress via Nox4 in Breast Cancer Cells. Antioxidants 2021, 10, 1881. https://doi.org/10.3390/antiox10121881
Kim TW, Ko S-G. The Herbal Formula JI017 Induces ER Stress via Nox4 in Breast Cancer Cells. Antioxidants. 2021; 10(12):1881. https://doi.org/10.3390/antiox10121881
Chicago/Turabian StyleKim, Tae Woo, and Seong-Gyu Ko. 2021. "The Herbal Formula JI017 Induces ER Stress via Nox4 in Breast Cancer Cells" Antioxidants 10, no. 12: 1881. https://doi.org/10.3390/antiox10121881
APA StyleKim, T. W., & Ko, S.-G. (2021). The Herbal Formula JI017 Induces ER Stress via Nox4 in Breast Cancer Cells. Antioxidants, 10(12), 1881. https://doi.org/10.3390/antiox10121881





