Exploring the Impact of Flavonoids on Symptoms of Depression: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Methods
2.1. Eligibility Criteria and Search Strategies
2.2. Study Selection and Data Extraction
2.3. Study Quality Assessment
2.4. Statistical Analysis
3. Results
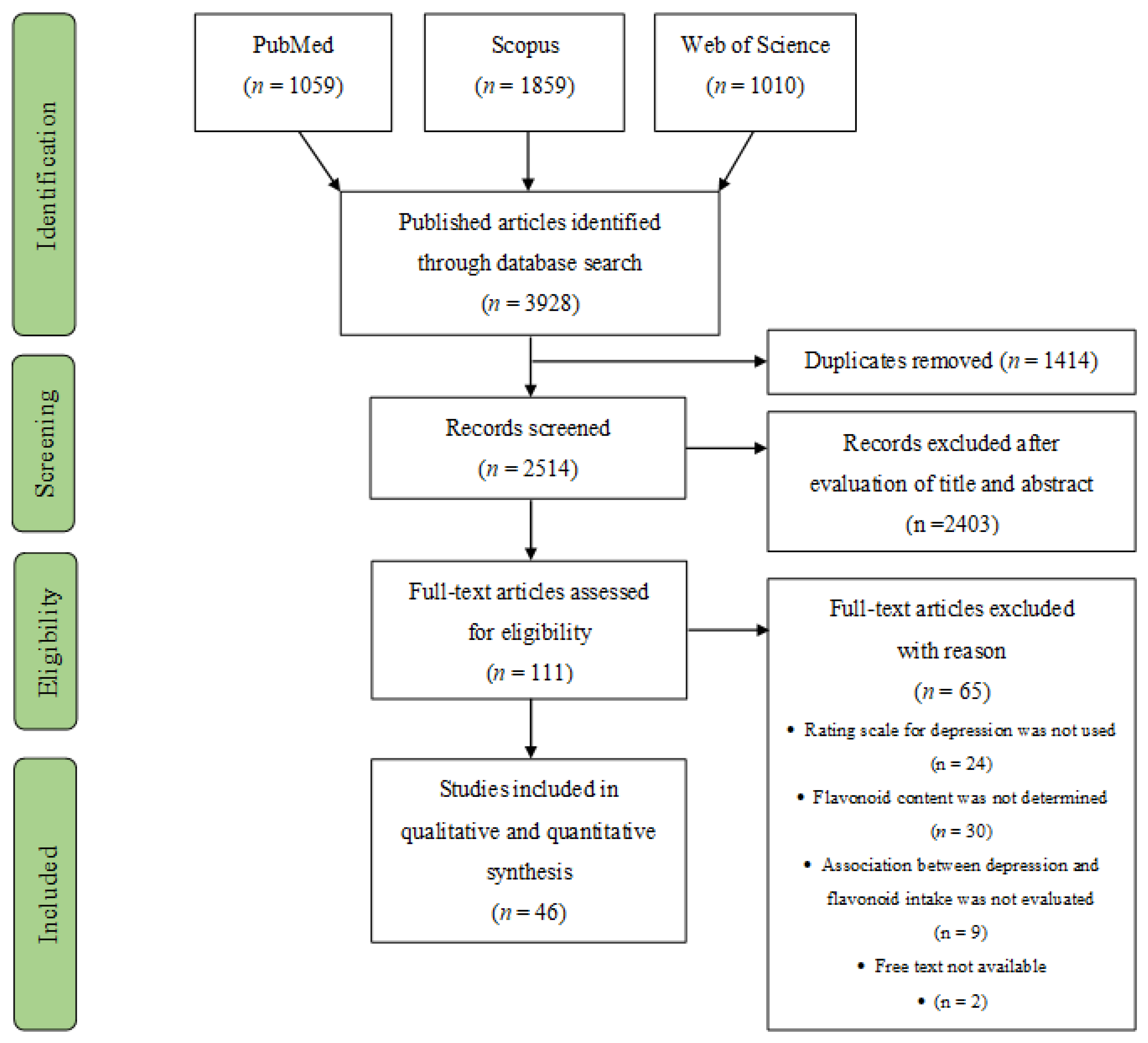
3.1. Study Selection
3.2. Study Characteristics
3.3. Flavonoids
3.4. Depression Scales
3.5. Flavonoids and Depressive Symptoms
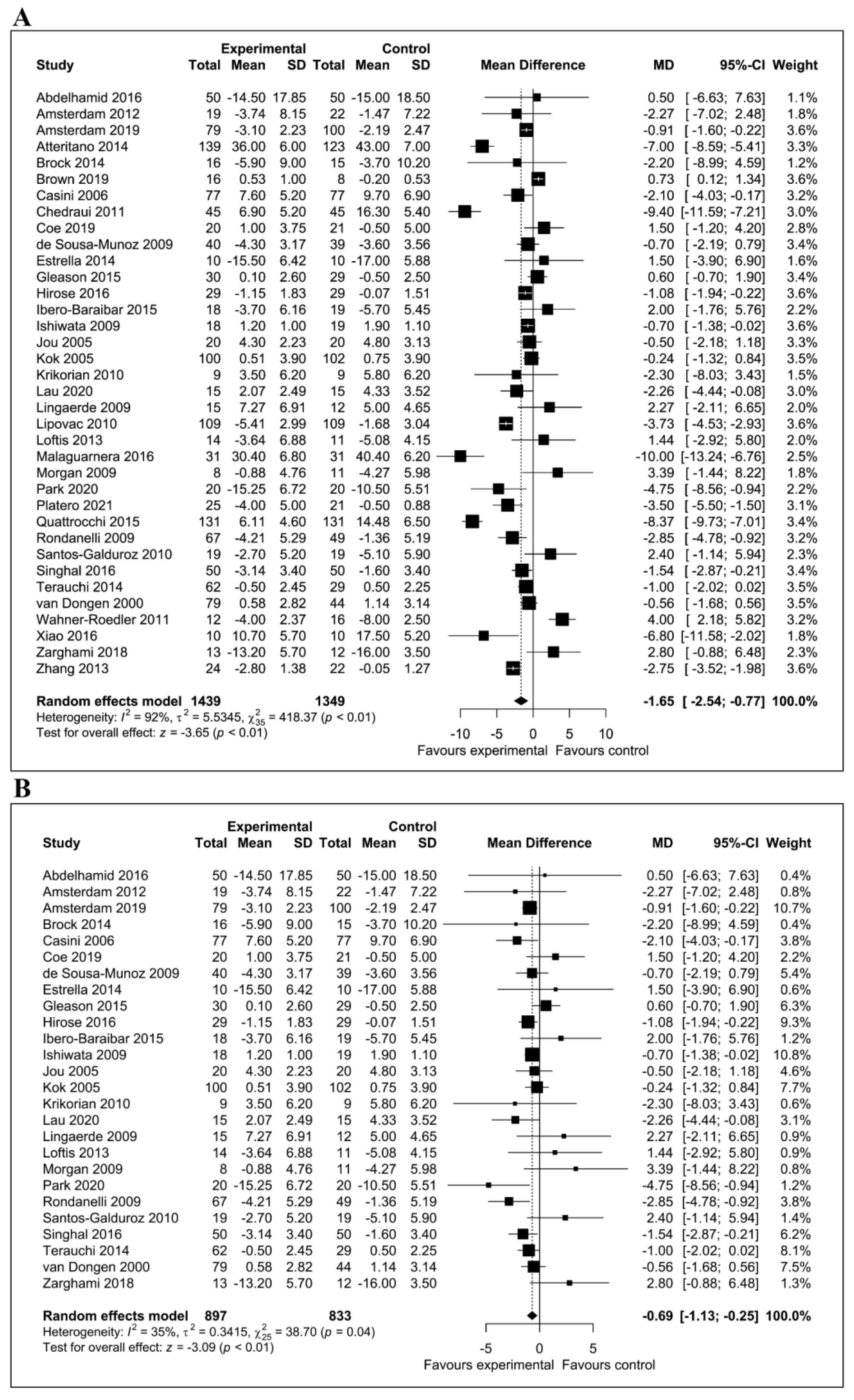
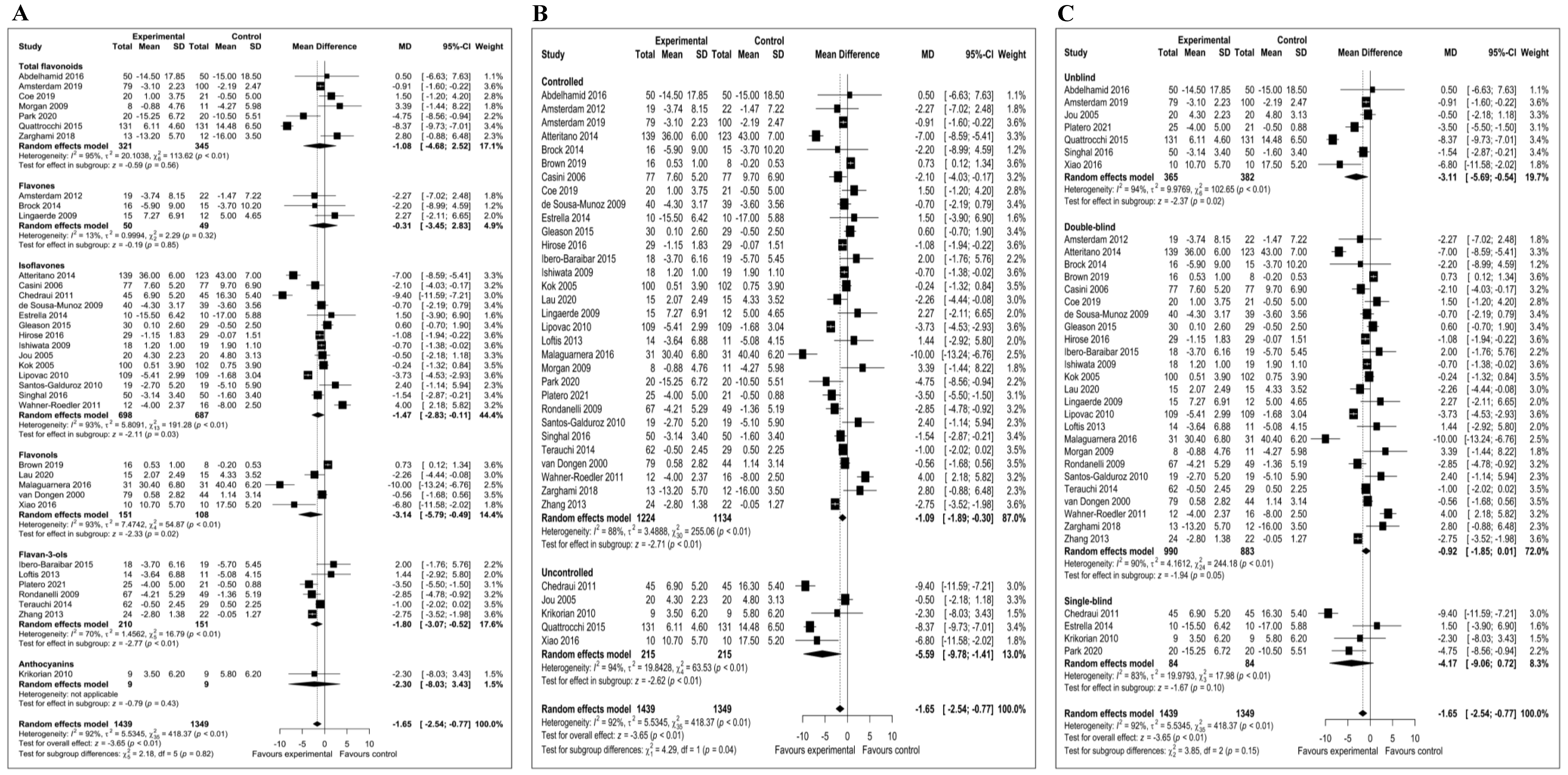
3.6. Meta-Analysis of the Effect of Flavonoids on Depressive Symptoms
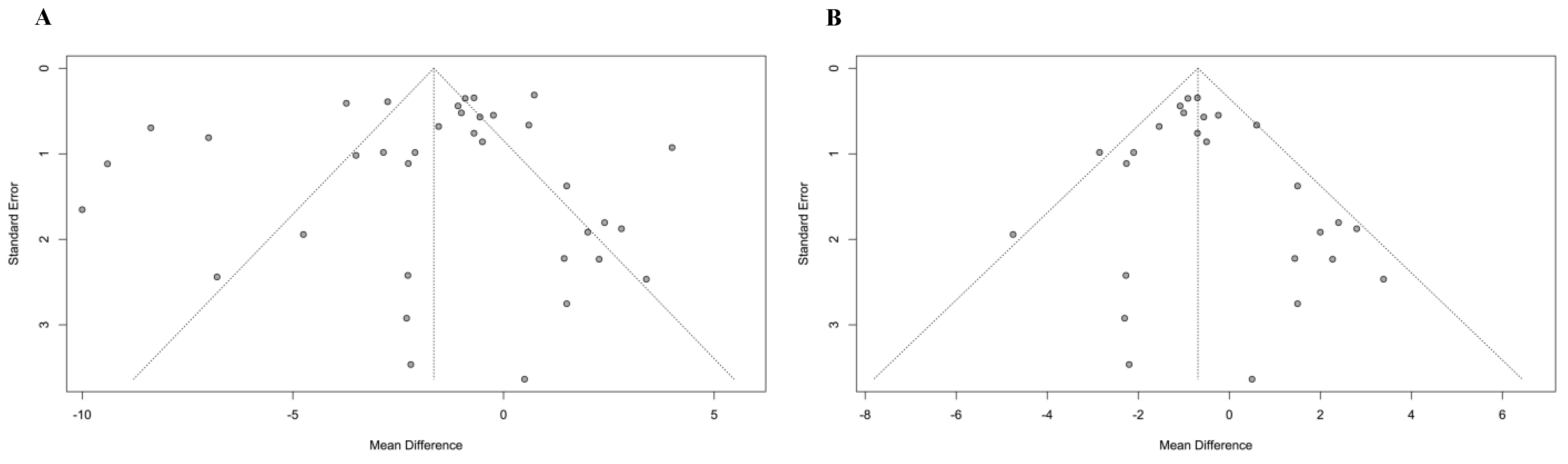
3.7. Publication Bias
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Friedrich, M.J. Depression Is the Leading Cause of Disability Around the World. JAMA 2017, 317, 1517. [Google Scholar] [CrossRef]
- Klein, D.N.; Santiago, N.J. Dysthymia and chronic depression: Introduction, classification, risk factors, and course. J. Clin. Psychol. 2003, 59, 807–816. [Google Scholar] [CrossRef]
- Rotenstein, L.S.; Ramos, M.A.; Torre, M.; Segal, J.B.; Peluso, M.J.; Guille, C.; Sen, S.; Mata, D.A. Prevalence of Depression, Depressive Symptoms, and Suicidal Ideation Among Medical Students: A Systematic Review and Meta-Analysis. JAMA 2016, 316, 2214–2236. [Google Scholar] [CrossRef] [Green Version]
- van den Bosch, M.; Meyer-Lindenberg, A. Environmental Exposures and Depression: Biological Mechanisms and Epidemiological Evidence. Annu. Rev. Public Health 2019, 40, 239–259. [Google Scholar] [CrossRef] [Green Version]
- Bhatt, S.; Nagappa, A.N.; Patil, C.R. Role of oxidative stress in depression. Drug Discov. Today 2020, 25, 1270–1276. [Google Scholar] [CrossRef]
- Liu, T.; Zhong, S.; Liao, X.; Chen, J.; He, T.; Lai, S.; Jia, Y. A Meta-Analysis of Oxidative Stress Markers in Depression. PLoS ONE 2015, 10, e0138904. [Google Scholar] [CrossRef]
- Cipriani, A.; Furukawa, T.A.; Salanti, G.; Chaimani, A.; Atkinson, L.Z.; Ogawa, Y.; Leucht, S.; Ruhe, H.G.; Turner, E.H.; Higgins, J.P.T.; et al. Comparative efficacy and acceptability of 21 antidepressant drugs for the acute treatment of adults with major depressive disorder: A systematic review and network meta-analysis. Lancet 2018, 391, 1357–1366. [Google Scholar] [CrossRef] [Green Version]
- Marx, W.; Moseley, G.; Berk, M.; Jacka, F. Nutritional psychiatry: The present state of the evidence. Proc. Nutr. Soc. 2017, 76, 427–436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marx, W.; Lane, M.; Hockey, M.; Aslam, H.; Berk, M.; Walder, K.; Borsini, A.; Firth, J.; Pariante, C.M.; Berding, K.; et al. Diet and depression: Exploring the biological mechanisms of action. Mol. Psychiatry 2021, 26, 134–150. [Google Scholar] [CrossRef] [PubMed]
- Jacka, F.N.; O’Neil, A.; Opie, R.; Itsiopoulos, C.; Cotton, S.; Mohebbi, M.; Castle, D.; Dash, S.; Mihalopoulos, C.; Chatterton, M.L.; et al. A randomised controlled trial of dietary improvement for adults with major depression (the “SMILES” trial). BMC Med. 2017, 15, 23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Molendijk, M.L.; Fried, E.I.; Van Der Does, W. The SMILES trial: Do undisclosed recruitment practices explain the remarkably large effect? BMC Med. 2018, 16, 243. [Google Scholar] [CrossRef]
- Parletta, N.; Zarnowiecki, D.; Cho, J.; Wilson, A.; Bogomolova, S.; Villani, A.; Itsiopoulos, C.; Niyonsenga, T.; Blunden, S.; Meyer, B.; et al. A Mediterranean-style dietary intervention supplemented with fish oil improves diet quality and mental health in people with depression: A randomized controlled trial (HELFIMED). Nutr. Neurosci. 2019, 22, 474–487. [Google Scholar] [CrossRef] [Green Version]
- Sánchez-Villegas, A.; Martínez-González, M.A.; Estruch, R.; Salas-Salvadó, J.; Corella, D.; Covas, M.I.; Arós, F.; Romaguera, D.; Gómez-Gracia, E.; Lapetra, J.; et al. Mediterranean dietary pattern and depression: The PREDIMED randomized trial. BMC Med. 2013, 11, 208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Firth, J.; Marx, W.; Dash, S.; Carney, R.; Teasdale, S.B.; Solmi, M.; Stubbs, B.; Schuch, F.B.; Carvalho, A.F.; Jacka, F.; et al. The Effects of Dietary Improvement on Symptoms of Depression and Anxiety: A Meta-Analysis of Randomized Controlled Trials. Psychosom. Med. 2019, 81, 265–280. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lv, M.-R.; Wei, Y.-J.; Sun, L.; Zhang, J.-X.; Zhang, H.-G.; Li, B. Dietary patterns and depression risk: A meta-analysis. Psychiatry Res. 2017, 253, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Davinelli, S.; Ali, S.; Solfrizzi, V.; Scapagnini, G.; Corbi, G. Carotenoids and cognitive outcomes: A meta-analysis of randomized intervention trials. Antioxidants 2021, 10, 223. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ali, T.; Kim, T.; Rehman, S.U.; Khan, M.S.; Amin, F.U.; Khan, M.; Ikram, M.; Kim, M.O. Natural Dietary Supplementation of Anthocyanins via PI3K/Akt/Nrf2/HO-1 Pathways Mitigate Oxidative Stress, Neurodegeneration, and Memory Impairment in a Mouse Model of Alzheimer’s Disease. Mol. Neurobiol. 2018, 55, 6076–6093. [Google Scholar] [CrossRef]
- Chen, L.; Teng, H.; Jia, Z.; Battino, M.; Miron, A.; Yu, Z.; Cao, H.; Xiao, J. Intracellular signaling pathways of inflammation modulated by dietary flavonoids: The most recent evidence. Crit. Rev. Food Sci. Nutr. 2018, 58, 2908–2924. [Google Scholar] [CrossRef]
- Yi, L.-T.; Liu, B.-B.; Li, J.; Luo, L.; Liu, Q.; Geng, D.; Tang, Y.; Xia, Y.; Wu, D. BDNF signaling is necessary for the antidepressant-like effect of naringenin. Prog. Neuropsychopharmacol. Biol. Psychiatry 2014, 48, 135–141. [Google Scholar] [CrossRef]
- Yi, L.-T.; Li, C.-F.; Zhan, X.; Cui, C.-C.; Xiao, F.; Zhou, L.-P.; Xie, Y. Involvement of monoaminergic system in the antidepressant-like effect of the flavonoid naringenin in mice. Prog. Neuropsychopharmacol. Biol. Psychiatry 2010, 34, 1223–1228. [Google Scholar] [CrossRef]
- Khan, H.; Perviz, S.; Sureda, A.; Nabavi, S.M.; Tejada, S. Current standing of plant derived flavonoids as an antidepressant. Food Chem. Toxicol. 2018, 119, 176–188. [Google Scholar] [CrossRef]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; Group, P.-P. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [Green Version]
- Higgins, J.P.T.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [Green Version]
- Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 2010, 25, 603–605. [Google Scholar] [CrossRef] [Green Version]
- Higgins, J.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions. Res. Syn. Meth. 2011, 2, 126–130. [Google Scholar]
- Begg, C.B.; Mazumdar, M. Operating Characteristics of a Rank Correlation Test for Publication Bias. Biometrics 1994, 50, 1088. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Smith, G.D.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. Br. Med. J. 1997, 315, 629–634. [Google Scholar] [CrossRef] [Green Version]
- Abdelhamid, Z.S.A.; Serry, Z.M.; Elnahas, N.M.G.; Ammar, N.M. Serum serotonin response to aerobic exercise verus phoenix. Int. J. PharmTech Res. 2016, 9, 108–114. [Google Scholar]
- Amsterdam, J.D.; Shults, J.; Soeller, I.; Mao, J.J.; Rockwell, K.; Newberg, A.B. Chamomile (Matricaria recutita) may provide antidepressant activity in anxious, depressed humans: An exploratory study. Altern. Ther. Health Med. 2012, 18, 44–49. [Google Scholar] [PubMed]
- Estrella, R.E.N.; Landa, A.I.; Lafuente, J.V.; Gargiulo, P.A. Effects of antidepressants and soybean association in depressive menopausal women. Acta Pol. Pharm. 2014, 71, 323–327. [Google Scholar]
- Gleason, C.E.; Fischer, B.L.; Dowling, N.M.; Setchell, K.D.R.; Atwood, C.S.; Carlsson, C.M.; Asthana, S. Cognitive Effects of Soy Isoflavones in Patients with Alzheimer’s Disease. J. Alzheimer’s Dis. JAD 2015, 47, 1009–1019. [Google Scholar] [CrossRef] [Green Version]
- Hirose, A.; Terauchi, M.; Akiyoshi, M.; Owa, Y.; Kato, K.; Kubota, T. Low-dose isoflavone aglycone alleviates psychological symptoms of menopause in Japanese women: A randomized, double-blind, placebo-controlled study. Arch. Gynecol. Obstet. 2016, 293, 609–615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ibero-Baraibar, I.; Perez-Cornago, A.; Ramirez, M.J.; Martínez, J.A.; Zulet, M.A. An Increase in Plasma Homovanillic Acid with Cocoa Extract Consumption Is Associated with the Alleviation of Depressive Symptoms in Overweight or Obese Adults on an Energy Restricted Diet in a Randomized Controlled Trial. J. Nutr. 2015, 146, 897S–904S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ishiwata, N.; Melby, M.K.; Mizuno, S.; Watanabe, S. New equol supplement for relieving menopausal symptoms: Randomized, placebo-controlled trial of Japanese women. Menopause 2009, 16, 141–148. [Google Scholar] [CrossRef]
- Jou, H.J.; Ling, P.Y.; Wu, S.C. Comparison of 70 mg and 35 mg isoflavone soya supplement for menopause symptoms. Int. J. Gynecol. Obstet. 2005, 90, 159–160. [Google Scholar] [CrossRef] [PubMed]
- Kok, L.; Kreijkamp-Kaspers, S.; Grobbee, D.E.; Lampe, J.W.; van der Schouw, Y.T. A randomized, placebo-controlled trial on the effects of soy protein containing isoflavones on quality of life in postmenopausal women. Menopause 2005, 12, 56–62. [Google Scholar] [CrossRef] [Green Version]
- Krikorian, R.; Shidler, M.D.; Nash, T.A.; Kalt, W.; Vinqvist-Tymchuk, M.R.; Shukitt-Hale, B.; Joseph, J.A. Blueberry supplementation improves memory in older adults. J. Agric. Food Chem. 2010, 58, 3996–4000. [Google Scholar] [CrossRef] [Green Version]
- Lau, H.; Shahar, S.; Mohamad, M.; Rajab, N.F.; Yahya, H.M.; Din, N.C.; Hamid, H.A. The effects of six months Persicaria minor extract supplement among older adults with mild cognitive impairment: A double-blinded, randomized, and placebo-controlled trial. BMC Complement. Med. Ther. 2020, 20, 315. [Google Scholar] [CrossRef]
- Lingaerde, O.; Føreland, A.R.; Magnusson, A. Can winter depression be prevented by Ginkgo biloba extract? A placebo-controlled trial. Acta Psychiatr. Scand. 1999, 100, 62–66. [Google Scholar] [CrossRef]
- Amsterdam, J.D.; Li, Q.S.; Xie, S.X.; Mao, J.J. Putative Antidepressant Effect of Chamomile (Matricaria chamomilla L.) Oral Extract in Subjects with Comorbid Generalized Anxiety Disorder and Depression. J. Altern. Complement. Med. 2020, 26, 813–819. [Google Scholar] [CrossRef]
- Lipovac, M.; Chedraui, P.; Gruenhut, C.; Gocan, A.; Stammler, M.; Imhof, M. Improvement of postmenopausal depressive and anxiety symptoms after treatment with isoflavones derived from red clover extracts. Maturitas 2010, 65, 258–261. [Google Scholar] [CrossRef]
- Loftis, J.M.; Wilhelm, C.J.; Huckans, M. Effect of epigallocatechin gallate supplementation in schizophrenia and bipolar disorder: An 8-week, randomized, double-blind, placebo-controlled study. Ther. Adv. Psychopharmacol. 2013, 3, 21–27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malaguarnera, G.; Bertino, G.; Chisari, G.; Motta, M.; Vecchio, M.; Vacante, M.; Caraci, F.; Greco, C.; Drago, F.; Nunnari, G.; et al. Silybin supplementation during HCV therapy with pegylated interferon-α plus ribavirin reduces depression and anxiety and increases work ability. BMC Psychiatry 2016, 16, 398. [Google Scholar] [CrossRef] [Green Version]
- Morgan, S.L.; Baggott, J.E.; Moreland, L.; Desmond, R.; Kendrach, A.C. The safety of flavocoxid, a medical food, in the dietary management of knee osteoarthritis. J. Med. Food 2009, 12, 1143–1148. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Choi, J.; Lee, H.J. Flavonoid-rich orange juice intake and altered gut microbiome in young adults with depressive symptom: A randomized controlled study. Nutrients 2020, 12, 1815. [Google Scholar] [CrossRef] [PubMed]
- Platero, J.L.; Cuerda-Ballester, M.; Sancho-Cantus, D.; Benlloch, M.; Ceron, J.J.; Rubio, C.P.; García-Pardo, M.P.; López-Rodríguez, M.M.; de la Rubia Ortí, J.E. The impact of epigallocatechin gallate and coconut oil treatment on cortisol activity and depression in multiple sclerosis patients. Life 2021, 11, 353. [Google Scholar] [CrossRef] [PubMed]
- Quattrocchi, T.; Micali, E.; Gentile, A.; La Ferrera, E.G.; Barbaro, L.; Ciarcià, S.; Corrado, F.; Di Costa, M.; Fazio, R.; Licenziato, R.; et al. Effects of a phyto complex on well-being of climacteric women. J. Obstet. Gynaecol. Res. 2015, 41, 1093–1098. [Google Scholar] [CrossRef]
- Rondanelli, M.; Opizzi, A.; Solerte, S.B.; Trotti, R.; Klersy, C.; Cazzola, R. Administration of a dietary supplement (N-oleyl-phosphatidylethanolamine and epigallocatechin-3-gallate formula) enhances compliance with diet in healthy overweight subjects: A randomized controlled trial. Br. J. Nutr. 2009, 101, 457–464. [Google Scholar] [CrossRef] [Green Version]
- Santos-Galduróz, R.F.; Galduróz, J.C.F.; Facco, R.L.; Hachul, H.; Tufik, S. Effects of isoflavone on the learning and memory of women in menopause: A double-blind placebo-controlled study. Braz. J. Med. Biol. Res. 2010, 43, 1123–1126. [Google Scholar] [CrossRef] [Green Version]
- Singhal, S.; Shullai, W. Comparative study of gabapentin and isoflavone in menopausal vasomotor symptoms. J. MidLife Health 2016, 7, 132–139. [Google Scholar] [CrossRef]
- Atteritano, M.; Mazzaferro, S.; Bitto, A.; Cannata, M.L.; D’Anna, R.; Squadrito, F.; Macrì, I.; Frisina, A.; Frisina, N.; Bagnato, G. Genistein effects on quality of life and depression symptoms in osteopenic postmenopausal women: A 2-year randomized, double-blind, controlled study. Osteoporos. Int. J. Establ. Result Coop. Eur. Found. Osteoporos. Natl. Osteoporos. Found. USA 2014, 25, 1123–1129. [Google Scholar] [CrossRef] [PubMed]
- Terauchi, M.; Horiguchi, N.; Kajiyama, A.; Akiyoshi, M.; Owa, Y.; Kato, K.; Kubota, T. Effects of grape seed proanthocyanidin extract on menopausal symptoms, body composition, and cardiovascular parameters in middle-aged women: A randomized, double-blind, placebo-controlled pilot study. Menopause 2014, 21, 990–996. [Google Scholar] [CrossRef] [PubMed]
- van Dongen, M.C.J.M.; van Rossum, E.; Kessels, A.G.H.; Sielhorst, H.J.G.; Knipschild, P.G. The efficacy of ginkgo for elderly people with dementia and age-associated memory impairment: New results of a randomized clinical trial. J. Am. Geriatr. Soc. 2000, 48, 1183–1194. [Google Scholar] [CrossRef] [PubMed]
- Wahner-Roedler, D.L.; Thompson, J.M.; Luedtke, C.A.; King, S.M.; Cha, S.S.; Elkin, P.L.; Bruce, B.K.; Townsend, C.O.; Bergeson, J.R.; Eickhoff, A.L.; et al. Dietary soy supplement on fibromyalgia symptoms: A randomized, double-blind, placebo-controlled, early phase trial. Evid. Based Complement. Altern. Med. 2011, 2011, 350697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiao, H.; Wignall, N.; Brown, E.S. An open-label pilot study of icariin for co-morbid bipolar and alcohol use disorder. Am. J. Drug Alcohol Abuse 2016, 42, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Zarghami, M.; Chabra, A.; Azadbakht, M.; Khalilian, A.; Asghar Hoseini, A. Antidepressant Effect of Asperugo procumbens L. in Comparison with Fluoxetine: A Randomized Double Blind Clinical Trial. Res. J. Pharmacogn. 2018, 5, 15–20. [Google Scholar] [CrossRef]
- Zhang, Q.; Yang, H.; Wang, J.; Li, A.; Zhang, W.; Cui, X.; Wang, K. Effect of green tea on reward learning in healthy individuals: A randomized, double-blind, placebo-controlled pilot study. Nutr. J. 2013, 12, 84. [Google Scholar] [CrossRef] [Green Version]
- Brock, C.; Whitehouse, J.; Tewfik, I.; Towell, T. American Skullcap (Scutellaria lateriflora): A randomised, double-blind placebo-controlled crossover study of its effects on mood in healthy volunteers. Phyther. Res. PTR 2014, 28, 692–698. [Google Scholar] [CrossRef]
- Brown, E.S.; Bice, C.; Putnam, W.C.; Leff, R.; Kulikova, A.; Nakamura, A.; Ivleva, E.I.; Van Enkevort, E.; Holmes, T.; Miingi, N. Human safety and pharmacokinetics study of orally administered icariin: Randomized, double-blind, placebo-controlled trial. Nat. Prod. Commun. 2019, 14. [Google Scholar] [CrossRef]
- Casini, M.L.; Marelli, G.; Papaleo, E.; Ferrari, A.; D’Ambrosio, F.; Unfer, V. Psychological assessment of the effects of treatment with phytoestrogens on postmenopausal women: A randomized, double-blind, crossover, placebo-controlled study. Fertil. Steril. 2006, 85, 972–978. [Google Scholar] [CrossRef]
- Chedraui, P.; San Miguel, G.; Schwager, G. The effect of soy-derived isoflavones over hot flushes, menopausal symptoms and mood in climacteric women with increased body mass index. Gynecol. Endocrinol. Off. J. Int. Soc. Gynecol. Endocrinol. 2011, 27, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Coe, S.; Cossington, J.; Collett, J.; Soundy, A.; Izadi, H.; Ovington, M.; Durkin, L.; Kirsten, M.; Clegg, M.; Cavey, A.; et al. A randomised double-blind placebo-controlled feasibility trial of flavonoid-rich cocoa for fatigue in people with relapsing and remitting multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 2019, 90, 507–513. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Sousa-Muñoz, R.L.; Filizola, R.G. Efficacy of soy isoflavones for depressive symptoms of the climacteric syndrome. Maturitas 2009, 63, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Hakim, B.N.; Suzana, S.; Haron, H. Association between isoflavones consumption and cognitive function and comorbidities among older adults residing in the State of Johor, Malaysia. Malays. J. Nutr. 2016, 22, 29–39. [Google Scholar]
- Chang, S.-C.; Cassidy, A.; Willett, W.C.; Rimm, E.B.; O’Reilly, E.J.; Okereke, O.I. Dietary flavonoid intake and risk of incident depression in midlife and older women. Am. J. Clin. Nutr. 2016, 104, 704–714. [Google Scholar] [CrossRef] [Green Version]
- Godos, J.; Castellano, S.; Ray, S.; Grosso, G.; Galvano, F. Dietary polyphenol intake and depression: Results from the mediterranean healthy eating, lifestyle and aging (MEAL) study. Molecules 2018, 23, 999. [Google Scholar] [CrossRef] [Green Version]
- Miyake, Y.; Tanaka, K.; Okubo, H.; Sasaki, S.; Furukawa, S.; Arakawa, M. Soy isoflavone intake and prevalence of depressive symptoms during pregnancy in Japan: Baseline data from the Kyushu Okinawa Maternal and Child Health Study. Eur. J. Nutr. 2018, 57, 441–450. [Google Scholar] [CrossRef]
- Miyake, Y.; Tanaka, K.; Okubo, H.; Sasaki, S.; Furukawa, S.; Arakawa, M. Dietary patterns and depressive symptoms during pregnancy in Japan: Baseline data from the Kyushu Okinawa Maternal and Child Health Study. J. Affect. Disord. 2018, 225, 552–558. [Google Scholar] [CrossRef]
- Nagata, C.; Shimizu, H.; Takami, R.; Hayashi, M.; Takeda, N.; Yasuda, K. Hot flushes and other menopausal symptoms in relation to soy product intake in Japanese women. Climacteric J. Int. Menopause Soc. 1999, 2, 6–12. [Google Scholar] [CrossRef]
- Richard, A.; Rohrmann, S.; Mohler-Kuo, M.; Rodgers, S.; Moffat, R.; Güth, U.; Eichholzer, M. Urinary phytoestrogens and depression in perimenopausal US women: NHANES 2005–2008. J. Affect. Disord. 2014, 156, 200–205. [Google Scholar] [CrossRef] [PubMed]
- Rosli, H.; Shahar, S.; Din, N.C.; Haron, H.; Rajab, N.F. Prevalence of Poor Mental Health and Cognitive Status among Middle-Aged Adults and Its Predictors in Relation to Polyphenols Intake. Malays. J. Med. Sci. MJMS 2019, 26, 72–89. [Google Scholar] [CrossRef] [PubMed]
- Woo, J.; Lynn, H.; Lau, W.Y.; Leung, J.; Lau, E.; Wong, S.Y.S.; Kwok, T. Nutrient intake and psychological health in an elderly Chinese population. Int. J. Geriatr. Psychiatry 2006, 21, 1036–1043. [Google Scholar] [CrossRef]
- Cui, Y.; Huang, C.; Momma, H.; Niu, K.; Nagatomi, R. Daily dietary isoflavone intake in relation to lowered risk of depressive symptoms among men. J. Affect. Disord. 2020, 261, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Krishnamoorthy, Y.; Rajaa, S.; Rehman, T. Diagnostic accuracy of various forms of geriatric depression scale for screening of depression among older adults: Systematic review and meta-analysis. Arch. Gerontol. Geriatr. 2020, 87, 104002. [Google Scholar] [CrossRef] [PubMed]
- Lewinsohn, P.M.; Seeley, J.R.; Roberts, R.E.; Allen, N.B. Center for epidemiologic studies depression scale (CES-D) as a screening instrument for depression among community-residing older adults. Psychol. Aging 1997, 12, 277–287. [Google Scholar] [CrossRef]
- Richter, P.; Werner, J.; Heerlein, A.; Kraus, A.; Sauer, H. On the validity of the Beck Depression Inventory. A review. Psychopathology 1998, 31, 160–168. [Google Scholar] [CrossRef]
- Dunlop, B.W.; Parikh, S.V.; Rothschild, A.J.; Thase, M.E.; Debattista, C.; Conway, C.R.; Forester, B.P.; Mondimore, F.M.; Shelton, R.C.; Macaluso, M.; et al. Comparing sensitivity to change using the 6-item versus the 17-item Hamilton depression rating scale in the GUIDED randomized controlled trial. BMC Psychiatry 2019, 19, 420. [Google Scholar] [CrossRef] [Green Version]
- Thomas-Odenthal, F.; Molero, P.; van der Does, W.; Molendijk, M. Impact of review method on the conclusions of clinical reviews: A systematic review on dietary interventions in depression as a case in point. PLoS ONE 2020, 15, e0238131. [Google Scholar] [CrossRef]
- Molendijk, M.; Molero, P.; Ortuño Sánchez-Pedreño, F.; Van der Does, W.; Angel Martínez-González, M. Diet quality and depression risk: A systematic review and dose-response meta-analysis of prospective studies. J. Affect. Disord. 2018, 226, 346–354. [Google Scholar] [CrossRef] [Green Version]
- Kimball, S.M.; Mirhosseini, N.; Rucklidge, J. Database analysis of depression and anxiety in a community sample—response to a micronutrient intervention. Nutrients 2018, 10, 152. [Google Scholar] [CrossRef] [Green Version]
- Edman, J.S.; Morrow, F.D.; Marby, D.W.; Mirages, S.; Perrone, G.; Kayne, H.L.; Cole, J.O. B Complex Vitamin Patterns in Geriatric and Young Adult Inpatients with Major Depression. J. Am. Geriatr. Soc. 1991, 39, 252–257. [Google Scholar] [CrossRef]
- Eby, G.A.; Eby, K.L. Rapid recovery from major depression using magnesium treatment. Med. Hypotheses 2006, 67, 362–370. [Google Scholar] [CrossRef]
- Deacon, G.; Kettle, C.; Hayes, D.; Dennis, C.; Tucci, J. Omega 3 polyunsaturated fatty acids and the treatment of depression. Crit. Rev. Food Sci. Nutr. 2017, 57, 212–223. [Google Scholar] [CrossRef]
- Spedding, S. Vitamin D and depression: A systematic review and meta-analysis comparing studies with and without biological flaws. Nutrients 2014, 6, 1501–1518. [Google Scholar] [CrossRef]
- Li, Z.; Wang, W.; Xin, X.; Song, X.; Zhang, D. Association of total zinc, iron, copper and selenium intakes with depression in the US adults. J. Affect. Disord. 2018, 228, 68–74. [Google Scholar] [CrossRef]
- Guan, L.P.; Liu, B.Y. Antidepressant-like effects and mechanisms of flavonoids and related analogues. Eur. J. Med. Chem. 2016, 121, 47–57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brown, D. Do Food Frequency Questionnaires Have Too Many Limitations? J. Am. Diet. Assoc. 2006, 106, 1541–1542. [Google Scholar] [CrossRef] [PubMed]
- Rendeiro, C.; Rhodes, J.S.; Spencer, J.P.E. The mechanisms of action of flavonoids in the brain: Direct versus indirect effects. Neurochem. Int. 2015, 89, 126–139. [Google Scholar] [CrossRef] [PubMed]
- Maes, M.; Fišar, Z.; Medina, M.; Scapagnini, G.; Nowak, G.; Berk, M. New drug targets in depression: Inflammatory, cell-mediated immune, oxidative and nitrosative stress, mitochondrial, antioxidant, and neuroprogressive pathways. And new drug candidates—Nrf2 activators and GSK-3 inhibitors. Inflammopharmacology 2012, 20, 127–150. [Google Scholar] [CrossRef] [PubMed]
- Felger, J.C.; Lotrich, F.E. Inflammatory cytokines in depression: Neurobiological mechanisms and therapeutic implications. Neuroscience 2013, 246, 199–229. [Google Scholar] [CrossRef] [Green Version]
- Scapagnini, G.; Davinelli, S.; Drago, F.; De Lorenzo, A.; Oriani, G. Antioxidants as antidepressants: Fact or fiction? CNS Drugs 2012, 26, 477–490. [Google Scholar] [CrossRef]
- Davinelli, S.; Maes, M.; Corbi, G.; Zarrelli, A.; Willcox, D.C.; Scapagnini, G. Dietary phytochemicals and neuro-inflammaging: From mechanistic insights to translational challenges. Immun. Ageing 2016, 13, 16. [Google Scholar] [CrossRef] [Green Version]
- Williams, C.M.; El Mohsen, M.A.; Vauzour, D.; Rendeiro, C.; Butler, L.T.; Ellis, J.A.; Whiteman, M.; Spencer, J.P.E. Blueberry-induced changes in spatial working memory correlate with changes in hippocampal CREB phosphorylation and brain-derived neurotrophic factor (BDNF) levels. Free Radic. Biol. Med. 2008, 45, 295–305. [Google Scholar] [CrossRef]
- Spencer, J.P.E.; Rice-Evans, C.; Williams, R.J. Modulation of pro-survival Akt/protein kinase B and ERK1/2 signaling cascades by quercetin and its in vivo metabolites underlie their action on neuronal viability. J. Biol. Chem. 2003, 278, 34783–34793. [Google Scholar] [CrossRef] [Green Version]
- Spencer, J.P.E. Flavonoids and brain health: Multiple effects underpinned by common mechanisms. Genes Nutr. 2009, 4, 243–250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pei, R.; Liu, X.; Bolling, B. Flavonoids and gut health. Curr. Opin. Biotechnol. 2020, 61, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Rémésy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2005, 81, 230S–242S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Williamson, G.; Manach, C. Bioavailability and bioefficacy of polyphenols in humans. II. Review of 93 intervention studies. Am. J. Clin. Nutr. 2005, 81, 243S–255S. [Google Scholar] [CrossRef]
- Messina, M.; Gleason, C. Evaluation of the potential antidepressant effects of soybean isoflavones. Menopause 2016, 23, 1348–1360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davinelli, S.; Scapagnini, G.; Marzatico, F.; Nobile, V.; Ferrara, N.; Corbi, G. Influence of equol and resveratrol supplementation on health-related quality of life in menopausal women: A randomized, placebo-controlled study. Maturitas 2017, 96, 77–83. [Google Scholar] [CrossRef] [PubMed]
| Study (Author, Year, Country, Ref.) | Study Design | Subjects | Condition | Intervention | Depression Scale | Results |
|---|---|---|---|---|---|---|
| Abdelhamid et al., 2016; Egypt [29] | Randomized clinical trial Duration: 24 weeks | 100 subjects (60–70 y) (n = 50 women; n = 50 men) | Mild depression | Flavonoid-rich extract from date palm fruit (rutin 11.2 mg/100 mg) | GDS | Significant effect compared with baseline (p = 0.0001) |
| Amsterdam et al., 2012; US [30] | Randomized double-blind placebo-controlled trial Duration: 8 weeks | 41 subjects (mean age 42.9 y) (n = 26 women; n = 15 men) | Anxiety with or without depression | Chamomile capsules (220 mg/d; 1.2% apigenin) | HAM-D | Significant effect (treatment vs. placebo p < 0.05) |
| Amsterdam et al., 2019; US [41] | Randomized open-label trial Duration: 8 weeks | 179 subjects (mean age 45.7 y) (n = 119 women; n = 60 men) | Anxiety with or without depression | Chamomile capsules (1500 mg/d; 18 mg of flavonoids) | HRSD | Significant effect in subjects with depression (p < 0.02) |
| Atteritano et al., 2014; Italy [52] | Randomized double-blind controlled trial Duration: 2 years | 262 women (49–67 y) | Osteopenia and Postmenopause | Genistein tablets (54 mg/d) | ZSDS | Significant effect (treatment vs. placebo p < 0.01) |
| Brock et al., 2014; UK [59] | Randomized double-blind placebo-controlled crossover trial Duration: 2 weeks | 31 subjects (mean age 34 y) (n = 25 women; n = 6 men) | Anxious and non-anxious population | Scutellaria lateriflora capsules (350 mg/d; 11.7 mg/g baicalein; 7.7 mg/g wogonin) | POMS | No significant effect |
| Brown et al., 2019; US [60] | Randomized double-blind placebo-controlled trial Duration: 5 days | 24 subjects (18–50 y) (men and women) | Healthy subjects | Icariin capsules (from 100 to 1.680 mg/d) | QIDS-C | Significant effect (treatment vs. placebo p = 0.02) |
| Casini et al., 2006; Italy [61] | Randomized double-blind crossover placebo-controlled study Duration: 6 months | 77 women (49–50 y) | Postmenopause | Phytoestrogens tablets (soy isoflavones 60 mg/d; 45% genisytein 45% daidzein 10% glycitein) | BDI and POMS | Significant effect in BDI and POMS scores (treatment vs. placebo BDI, p < 0.01; POMS, p < 0.001) |
| Chedraui et al., 2011; Ecuador [62] | Single blinded pilot clinical trial Duration: 3 months | 45 women (age 40–59 y) | Menopausal symptoms | Soy isoflavone capsules (100 mg/d; 40% isoflavones) | HAM-D | Significant effect compared with baseline (p < 0.05) |
| Coe et al., 2019; UK [63] | Randomized, double-blind placebo- controlled trial Duration: 6 weeks | 41 subjects (mean age 44 y) (n = 30 women; n = 10 men) | Relapsing and remitting multiple sclerosis | Flavonoid-rich cocoa drink (intervention group 200 mg/day) | HADS | No significant effect |
| de Sousa-Muñoz, and Filizola, 2009; Brazil [64] | Placebo-controlled double-blind randomized trial Duration:16 weeks | 79 women (mean age 53.3 y) | Depressive symptoms in climacteric syndrome | Soy isoflavone capsules (120 mg/d; 60 mg isoflavones; 20 mg daidzein; 14 mg genistein) | CES-D | Significant effect compared with baseline (p = 0.001) |
| Estrella et al., 2014; Chile [31] | Randomized clinical trial Duration: 3 months | 20 women (45–55 y) | Depression in menopause | Soybean concentrate (100 mg/d; 50 mg isoflavones) | HAM-D and ZSDS | Significant effect in depression scores compared with baseline (p < 0.001) |
| Gleason et al., 2015; US [32] | Randomized double-blind parallel group clinical trial Duration:6 months | 59 subjects (>60 y) (men and women) | Alzheimer’s disease | Soy isoflavone capsules (100 mg/d; 85% daidzein, and genistein) | GDS and POMS | No significant effect |
| Hirose et al., 2016; Japan [33] | Randomized double-blind placebo-controlled trial Duration 8 weeks | 58 women (40–60 y) | Menopause | Soy isoflavone tablets (12.5 mg/d; 25 mg/d; genistein 51.8%, daidzein 43.3%, glycitein 4.9%) | HADS | Significant effect in compared with placebo (p = 0.033) |
| Ibero-Baraibar et al., 2015; Spain [34] | Randomized double-blind placebo-controlled parallel trial Duration: 4 weeks | 37 subjects (mean age 57 y) (n = 25 women; n = 22 men) | Overweight or obese adults | Cocoa extract (1.4 g/d; 414 mg flavan-3-ols; 153 mg epicatechin; 15 mg catechin; 246 mg procyanidins), | BDI | Significant effect compared with baseline (p < 0.01) |
| Ishiwata et al., 2009; Japan [35] | Randomized double-blind placebo-controlled trial Duration: 12 weeks | 37 women (mean age 46.6 y) | Menopause | Equol capsules (10 or 30 mg/d) | POMS | Significant effect compared with placebo (p < 0.05) |
| Jou et al., 2005; Taiwan [36] | Single-centre prospective randomized trial Duration: 6 weeks | 20 women (age N/A) | Menopause | Soy isoflavone capsules (35 or 70 mg/d isoflavones) | GCS | Significant effect in 35 mg groups compared with baseline (p < 0.05) |
| Kok et al., 2005; Netherlands [37] | Randomized double-blind placebo-controlled trial Duration: 12 months | 202 women (60–75 y) | Postmenopause | Soy powder (25.6 g/d; 52 mg/g genistein; 41 mg/g daidzein; 6 mg/g glycitein) | GDS | No significant effect |
| Krikorian et al., 2010; Canada [38] | Single-blind placebo-controlled trial Duration:12 weeks | 9 subjects (mean age 76.2) (men and women) | Memory decline | Blueberry juice (three doses; 444 mL/d, 0.42 g anthocyanins; 532 mL/d, 0.51 g anthocyanins; 621 mL/d, 0.59 g anthocyanins) | GDS | No significant effect |
| Lau et al., 2020; Malaysia [39] | Multi-center randomized double-blind placebo-controlled trial Duration: 6 months | 30 subjects (mean age 66.4 y) (n = 23 women; n = 7 men) | Mild cognitive impairment | Persicaria minor extract (500 mg/d; 0.45% quercetin-3-glucuronide; 0.15% quercitrin) | POMS | No significant effect |
| Lingaerde et al., 1999; Norway [40] | Randomized double-blind placebo-controlled trial Duration: 10 weeks | 27 subjects (26–58 y) (n = 21 women; n = 6 men) | Seasonal affective disorder | Ginkgo biloba tablets (120–160 mg/d; 24 mg flavones) | MADRS | No significant effect |
| Lipovac et al., 2010; Austria [42] | Randomized double-blind placebo-controlled trial Duration: 3 months | 109 women (mean age 53.5 y) | Postmenopausal symptoms | Isoflavone capsules (80 mg/d; extract in form of | HADS and ZSDS | Significant effect in HADS and ZSDS scores compared |
| Loftis et al., 2013; US [43] | Randomized double-blind placebo-controlled trial Duration: 8 weeks | 25 subjects (≥18 years) (men and women) | Schizophrenia and bipolar disorder | biochanin A, formononetin, genistein and daidzein) EGCG capsules (N/A mg/d; capsules enriched with 150 mg of theaflavin) | HAM-D | with control (p < 0.001) No significant effect |
| Malaguarnera et al.; 2016; Italy [44] | Randomized double-blind placebo-controlled trial Duration: 1 year | 62 subjects (mean age 46 y) (n = 26 women; n = 36 men) | Hepatitis C virus infection | Silybin pills (active group: 1.5 mg/kg/week Peg–IFN plus RBV + silybin 94 mg + vitamin E 30 mg + phospholipids 194 mg) | BDI | Significant effect in intervention group compared with placebo (p < 0.05) |
| Morgan et al., 2009; US [45] | Randomized double-blind placebo-controlled trial Duration: 12 weeks | 19 subjects (40–75 y) (men and women) | Moderate osteoarthritis | Scutellaria baicalensis and Acacia catechu pills (500 mg/d; N/A mg/g of flavonoids) | BDI | No significant effect |
| Park et al., 2020; South Korea [46] | Randomized single-blind controlled trial Duration: 8 weeks | 40 subjects (mean age 21.8 y) (n = 24 women; n = 16 men) | Depressed and non-depressed population | Flavonoid-rich orange juice (380 mL/d; 600 mg flavonoids) | CES-D | Significant effect compared with baseline (p < 0.0001) |
| Platero et al., 2021; Spain [47] | Randomized placebo-controlled trial Duration: 4 months | 46 subjects (mean age 47.1 y) (n = 32 women; n = 14 men) | Multiple sclerosis | EGCG capsules (800 mg/d) with coconut oil (60 mL/d) | BDI | Significant effect compared with baseline (p = 0.007) |
| Quattrocchi et al., 2015; Italy [48] | Intervention trial Duration: 6 months | 131 women (42–67 y) | Menopausal symptoms | Phyto complex (200 mg/d; 20% isoflavones, 1.5% vitexin) | BDI | Significant effect compared with baseline (p < 0.05) |
| Rondanelli et al., 2009; Italy [49] | Randomized double-blind placebo-controlled parallel trial Duration: 2 months | 116 subjects (18–50 y) (n = 89 women; n = 27 men) | Overweight | EGCG capsules (50 mg/d) | BDI | Significant effect compared with placebo (p < 0·005) |
| Santos- Galduróz et al., 2010; Brazil [50] | Randomized double-blind placebo-controlled trial Duration: 4 months | 38 women (50–65 y) | Menopause | Isoflavone tablets (80 mg/d; 60.8 mg genistein; 16 mg daidzein; 3.2 mg glycitein) | GDS | No significant effect |
| Singhal et al., 2016; India [51] | Comparative clinical Trial Duration: 3 months weeks | 100 women (46–55 y) | Menopausal Vasomotor symptoms | Isoflavone tablets (60 mg/d) | ZSDS | Significant effect compared with control (p = 0.02) |
| Terauchi et al., 2014; Japan [53] | Randomized double-blind placebo-controlled pilot trial Duration: 8 weeks | 91 women (40–60 y) | Menopausal symptoms | Grape seed Proanthocyanidin tablets (100 or 200 mg/d; 85% proanthocyanidins; 15% flavan-3-ols) | HADS | No significant effect |
| van Dongen et al., 2000; Netherlands [54] | Randomized double-blind placebo-controlled parallel group multicenter trial Duration: 24 weeks | 123 subjects (mean age 82.8) (men and women) | Age-associated Memory impairment | Ginko biloba tablets (160 or 240 mg/d; 24% ginkgo flavonols) | GDS | No significant effect |
| Wahner-Roedler et al., 2011; US [55] | Randomized double-blind placebo-controlled early phase trial. Duration: 6 weeks | 28 women (mean age 53.9 y) | Fibromyalgia symptoms | Soy shake (N/A mL/d; 160 mg soy isoflavones) | CES-D | No significant effect |
| Xiao et al., 2016; US [56] | Open-label Trial Duration: 8 weeks | 10 subjects (18–70 y) (men and women) | Alcohol abuse and bipolar disorder | Icariin capsules (300 mg/d) | HAM-D | Significant effect compared with baseline (p = 0.01) |
| Zarghami et al., 2018; Iran [57] | Randomized double-blind clinical trial Duration: 6 weeks | 25 subjects (18–70 y) (n = 19 women; n = 6 men) | Mild depression | Asperugo procumbens capsules (1.2 g/d; 6 mg total flavonoids) | HRSD | Significant lower effect compared with antidepressant control (p = 0.03) |
| Zhang et al., 2013; China [58] | Randomized double-blind placebo-controlled pilot trial Duration: 5 weeks | 46 subjects (mean age: 25.7 y) (n = 23 women; n = 23 men) | Reward function in healthy subject | Green tea (400 mg/d; EGCG 45.6%; epigallocatechin 16.7%; epicatechin-3-gallate 11.4%; epicatechin 6.8%) | MADRS And HRSD | Significant effect compared with control (MADRS p < 0.01; HRSD p < 0.001) |
| Study (Author, Year, Country, Ref.) | Study Design | Subjects and Sample Size | Main Variable | Exposure Measure | Depression Scale | Results |
|---|---|---|---|---|---|---|
| Chang et al., 2016; US [66] | Longitudinal cohort study Duration: 1976–2001 | 82,648 women who participated in the NHS (mean age of 67 y at baseline) and NHSII (mean age of 47 y at baseline) | Dietary Flavonoid intake | FFQ (total flavonoid intake ranged from 127.6 to 779.4 mg/d) | CES-D and GDS | Participants in the highest flavonoid consumption group had a 7–10% reduction in depression risk compared with the lowest intake group (p-trend = 0.0004–0.08). Flavones and proanthocyanidins showed the strongest associations |
| Cui et al., 2020; Japan [74] | Cross-sectional study Duration: 2008–2011 | 1335 men (19–83 y) | Dietary Isoflavone intake | DHQ (isoflavone intake ranged from ≤10.61 to ≥25.79 mg/d) | SDS | A high level of dietary isoflavone intake was associated with a lower prevalence of depressive symptoms in all the adjusted models (p-trend = 0.002–0.029) |
| Godos et al., 2018; Italy [67] | Cross-sectional study Duration: 2014–2015 | 1572 participants who participated in the MHELAS (18–92 y) (n = 912 women; n = 660 men) | Dietary Polyphenol intake | FFQ (total flavonoid intake ranged from 157.0 to 543.7 mg/d) | CES-D | Higher dietary intake of flavanones and anthocyanins was inversely associated with depressive symptoms (p-trend = 0.001) |
| Hakim et al., 2016; Malaysia [65] | Longitudinal cohort study Duration: 18 months | 400 elderly participants (≥60 y) (n = 231 women; n = 169 men) | Dietary Isoflavone intake | DHQ (mean intake: isoflavones 19.1 mg/d, daidzein 11.7 mg/d, genistein 7.6 mg/d) | GDS | No association between isoflavone intake and depression |
| Miyake et al., 2018; Japan [68] | Cross-sectional study | 1745 pregnant women (mean age 31 y) who participated in the KOMCHS (an ongoing prospective prebirth cohort study) | Soy Isoflavone intake | DHQ (isoflavone intake ranged from 10.5 to 49.4 mg/d) | CES-D | Isoflavone intake was associated with a lower prevalence of depressive symptoms during pregnancy in all the quartiles (p-trend = 0.002) |
| Miyake et al., 2018; Japan [69] | Cross-sectional study | 1744 pregnant women (mean age 31.2 y) who participated in the KOMCHS | Dietary patterns | DHQ (maximum isoflavone intake: Healthy pattern 43.5 mg/d Japanese pattern 32.7 mg/d Western pattern 31.2 mg/d) | CES-D | The healthy and Japanese patterns were inversely associated with depressive symptoms during pregnancy (Healthy pattern p-trend< 0.0001; Japanese pattern p-trend = 0.008) |
| Nagata et al., 1999; Japan [70] | Cross-sectional study Duration: September 1996– August 1997 | 284 menopausal women (mean age 47.1 y) | Soy product intake | FFQ (total isoflavone intake from soy product: 38.6 mg/d; isoflavone intake from fermented soy product 12.6 mg/d) | CES-D | No association between soy products and depression |
| Richard et al., 2014; Switzerland [71] | Cross-sectional study Duration: 2005–2008 | 193 perimenopausal women who participated in the NHANES (mean age 49 y) | Urinary phyto- estrogens | HPLC-APPI-MS/MS (mean value depressed vs. non-depressed women: isoflavones 128.3 and 97.4 μg/g; daidzein 57.4 and 48.3 μg/g; genistein 27.6 and 21.6 μg/g) | PHQ-9 | No association between urinary isoflavone concentrations and depression |
| Rosli et al., 2019; Malaysia [72] | Cross-sectional study | 349 participants with mild mental health problem (45–59 y) (n = 228 women; n = 121 men) | Dietary Polyphenols intake | FFQ (total flavonoid intake 265 mg/d) | GHQ-28 | No association between flavonoid intake and depression |
| Woo et al., 2006; Hong Kong [73] | Cross-sectional study | 3999 participants (≥65 y) (women and men) | Nutrient intake | FFQ (total isoflavone intake ranged from 4 to ≥19 mg/d) | GDS | No association between isoflavone intake and depression |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ali, S.; Corbi, G.; Maes, M.; Scapagnini, G.; Davinelli, S. Exploring the Impact of Flavonoids on Symptoms of Depression: A Systematic Review and Meta-Analysis. Antioxidants 2021, 10, 1644. https://doi.org/10.3390/antiox10111644
Ali S, Corbi G, Maes M, Scapagnini G, Davinelli S. Exploring the Impact of Flavonoids on Symptoms of Depression: A Systematic Review and Meta-Analysis. Antioxidants. 2021; 10(11):1644. https://doi.org/10.3390/antiox10111644
Chicago/Turabian StyleAli, Sawan, Graziamaria Corbi, Michael Maes, Giovanni Scapagnini, and Sergio Davinelli. 2021. "Exploring the Impact of Flavonoids on Symptoms of Depression: A Systematic Review and Meta-Analysis" Antioxidants 10, no. 11: 1644. https://doi.org/10.3390/antiox10111644
APA StyleAli, S., Corbi, G., Maes, M., Scapagnini, G., & Davinelli, S. (2021). Exploring the Impact of Flavonoids on Symptoms of Depression: A Systematic Review and Meta-Analysis. Antioxidants, 10(11), 1644. https://doi.org/10.3390/antiox10111644






