3D Cell Culture in Alginate Hydrogels
Abstract
:1. Introduction
2. Alginate
2.1. Alginate Structure, Chemistry and Purity
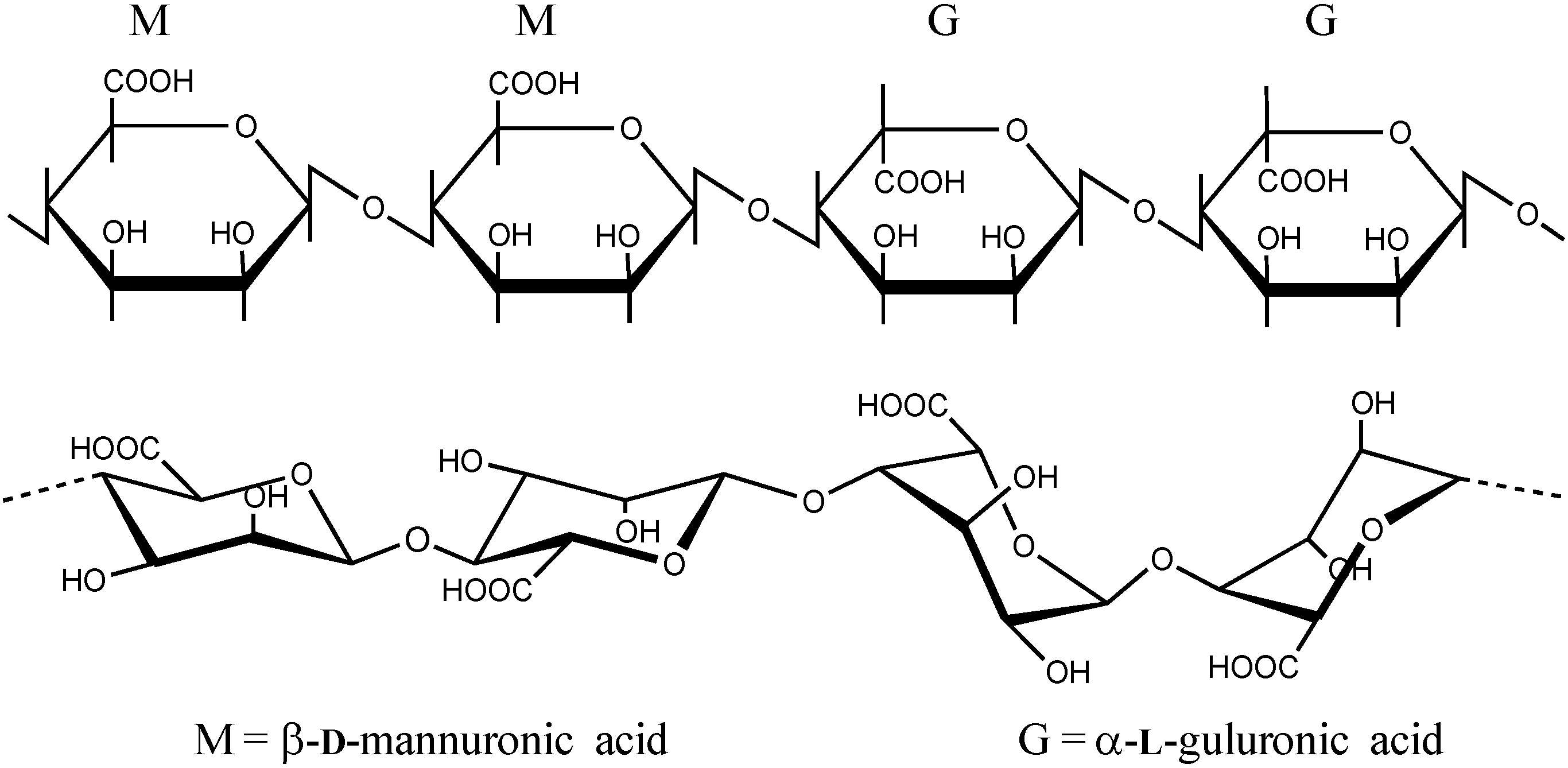
2.2. Alginate Hydrogels
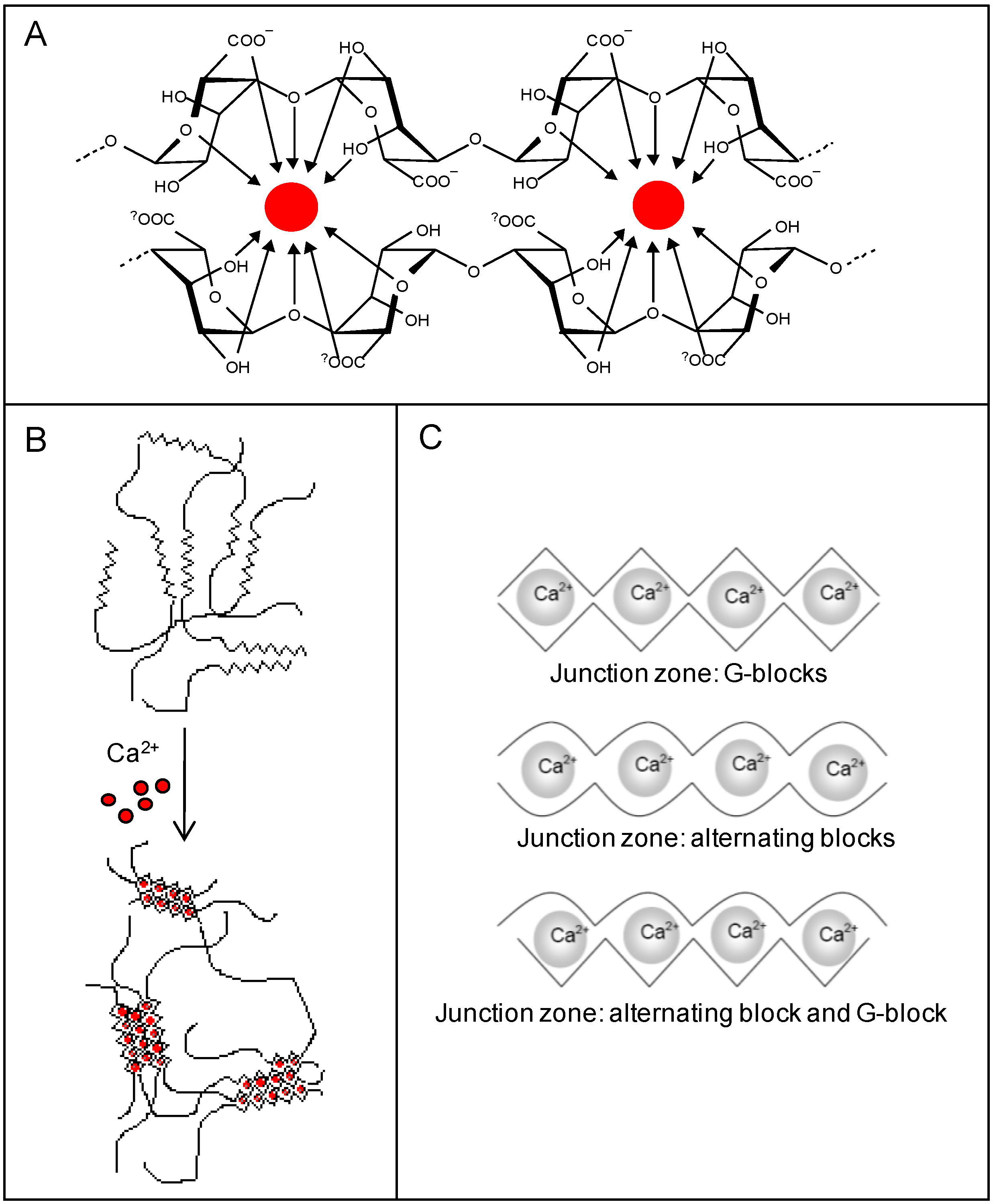
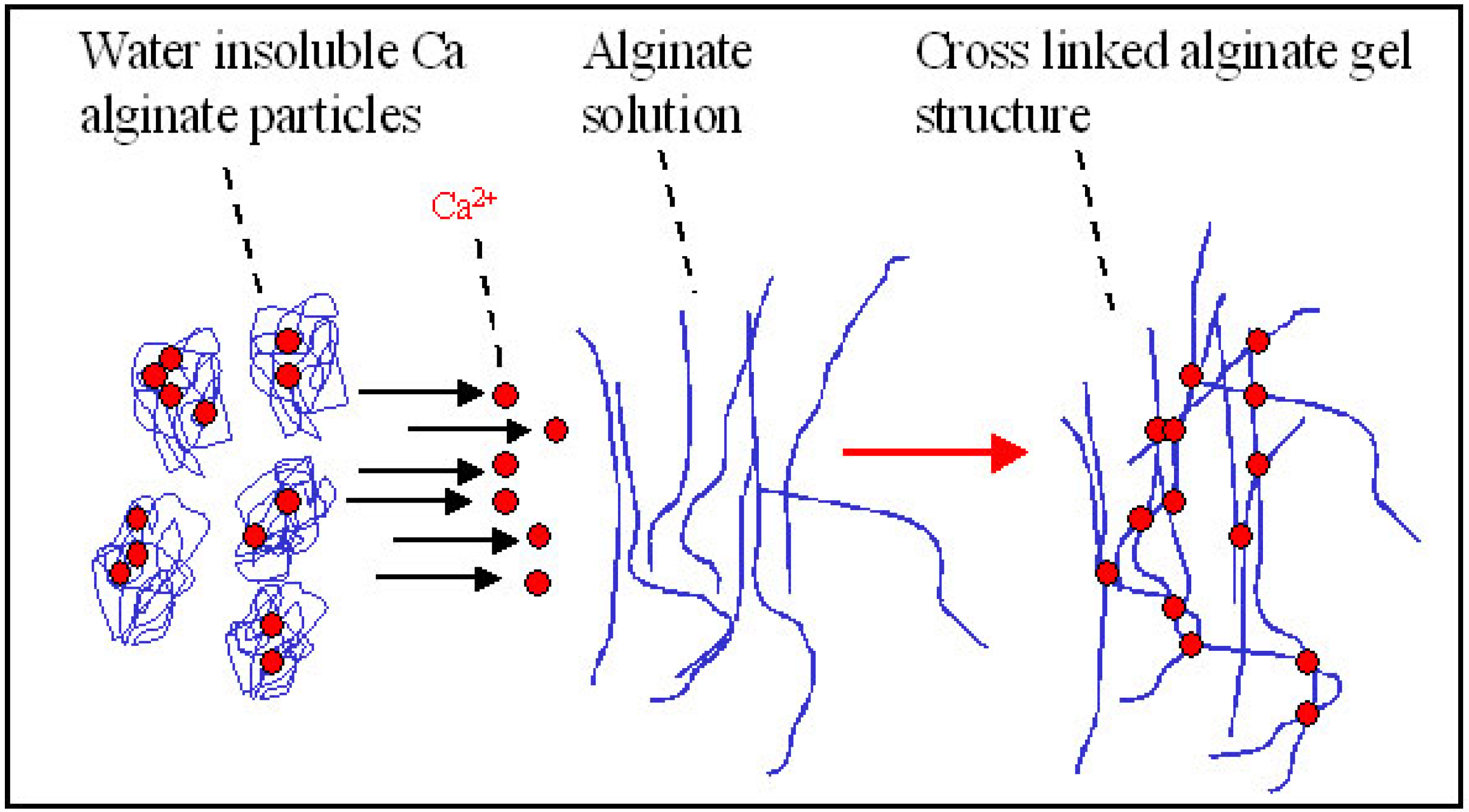
2.2.1. Ionic Gelation
2.2.2. Covalent Gelation
2.3. Alginate Derivatives
2.3.1. Peptide-Coupled Alginates

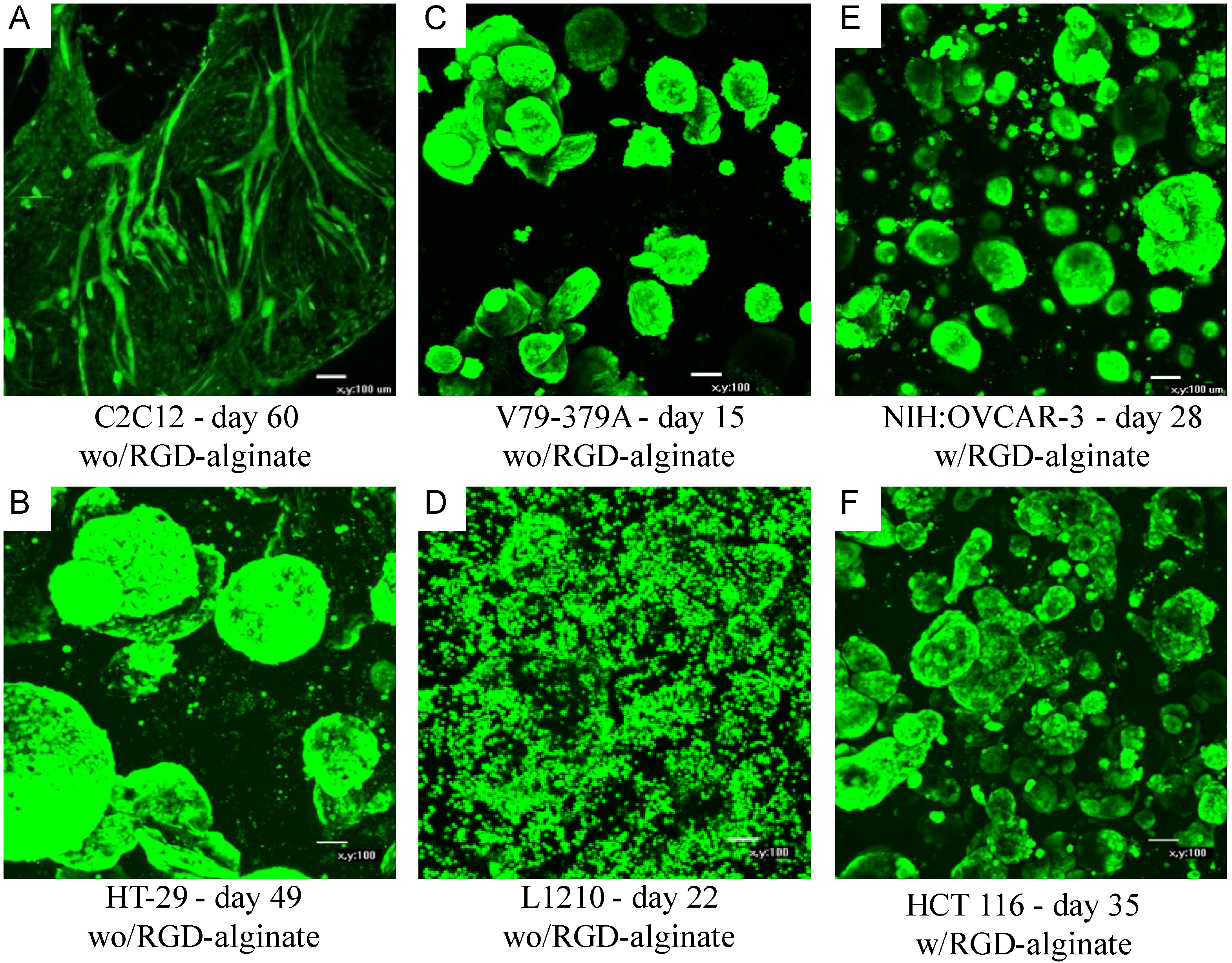
| Cell type | Cell line ID | Source | NOVATACH MVG GRGDSP |
|---|---|---|---|
| Cell lines—Non-tumorigenic origin | |||
| Lung fibroblasts (Chinese hamster) | V79-379A | Former Flow Laboratories | No2 |
| Myoblasts (epithelial, murine) | C2C12 | ATCC CRL-1772 | Yes |
| Fibroblasts (embryonic, murine) | NIH:3T3 | ATCC CRL-1658 | No3 |
| Kidney epithelial cells (Madin Darby, canine) | MDCK | ATCC CCL-34 | Yes |
| Cell lines—Carcinoma/adenocarcinoma origin | |||
| Cervix (human) | NHIK 3025 | Norwegian Radium Hospital | No |
| Ovarian (human) | NIH:OVCAR-3 | ATCC HTB-161 | Yes |
| Colon (myofibroblasts, hTERT-immortalized, human) | CT5.3 | Proprietary | No3 |
| Colorectal (human) | LoVo | ATCC CCL-229 | No3 |
| Lung (human) | A549 | ATCC CCL-185 | No3 |
| Prostate (human) | DU145 | ATCC HTB-81 | No2,3 |
| Breast (human) | MCF7 | ATCC HTB-22 | No3 |
| Pancreas (human) | PANC-1 | ATCC CRL-1469 | No2,3 |
| Leukemia (suspension, murine) | L1210 | ATCC CCL-219 | No3 |
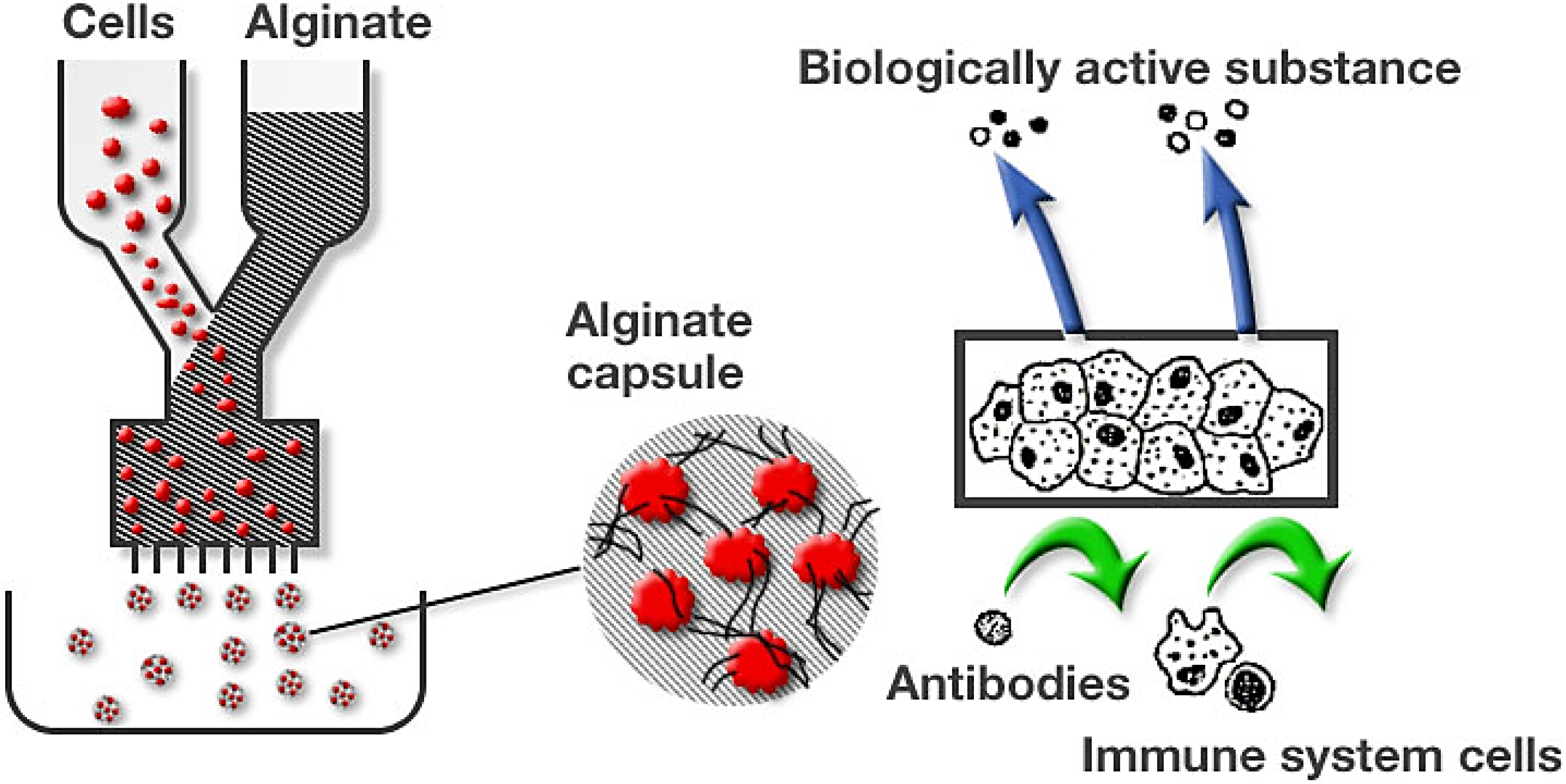
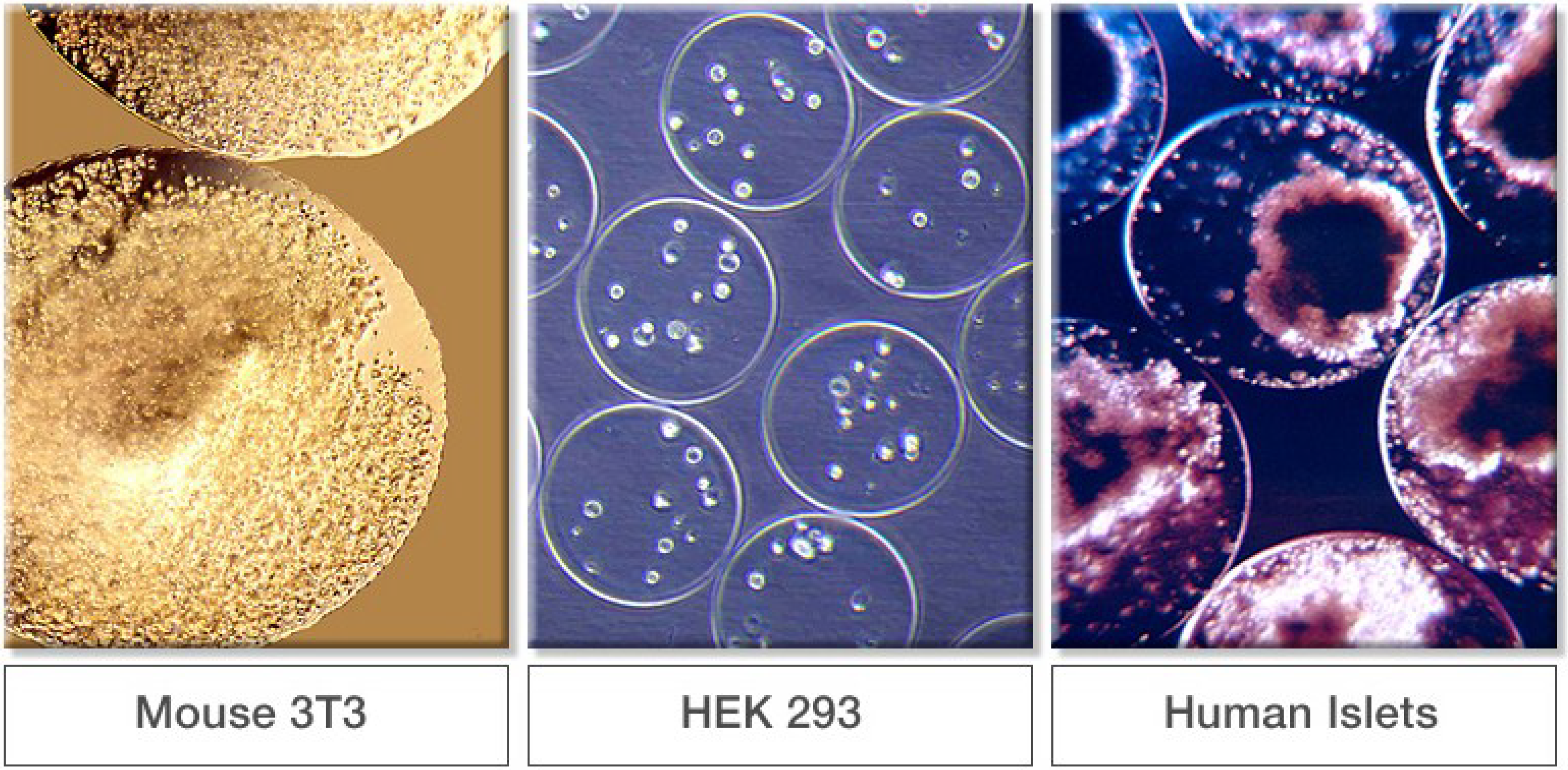
3. 3D Cell Culture
3.1. Beads
- •
- Extrusion through a needle: Beads can be made by dripping an alginate solution from a syringe with appropriate diameter needle directly into a gelling bath. While this method does not require any instrumentation, the size and size distribution of the produced beads are difficult to control.
- •
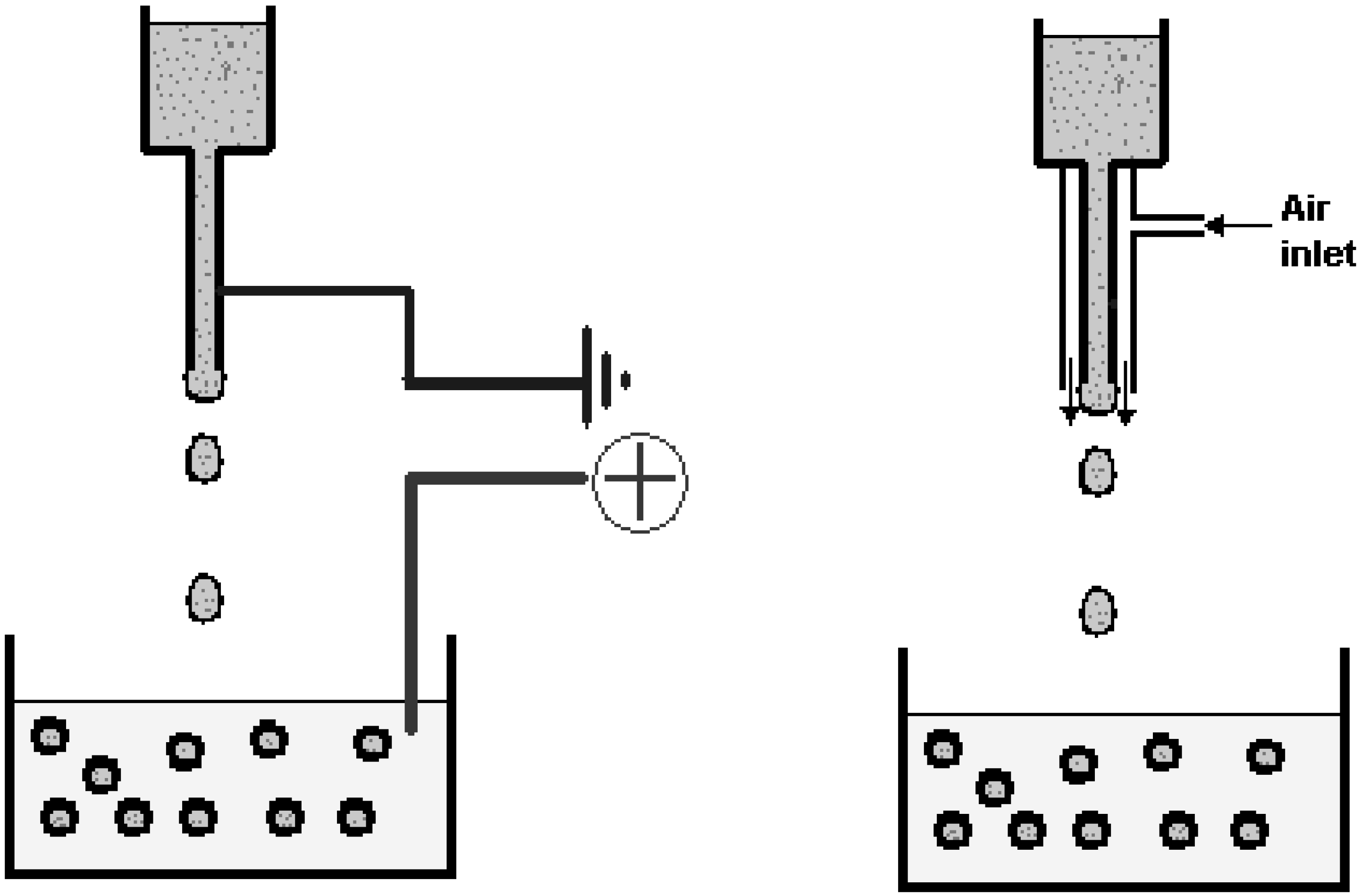
- Coaxial air or liquid flow: The coaxial air jet system is a simple way of generating small beads (down to around 400 µm), although the size distribution will normally be larger as compared to an electrostatic system. In this system, a coaxial air stream is used to pull droplets from a needle tip into a gelling bath (Figure 7).
- •
- Electrostatic potential: An electrostatic potential can be used to pull droplets from a needle tip into a gelling bath. The primary effect on droplet formation by the electrostatic potential is to direct charged molecules to the surface of the droplet to counteract surface tension. Using this type of instrument, beads below 200 µm and with a small size distribution may be generated. The desired bead size is obtained simply by adjusting the voltage (electrostatic potential) of the instrument. The principle for making smaller beads by electrostatic potential bead generators is shown in Figure 7.
- •
- Vibrating capillary jet breakage: A vibrating nozzle generates drops from a pressurized vessel.
- •
- Rotating capillary jet breakage: Bead generation is achieved by cutting a solid jet of fluid coming out of a nozzle by means of a rotating cutting device. The fluid is cut into cylindrical segments that then form beads due to surface tension while falling into a gelling bath.
3.2. Delayed Gelation Systems
3.3. Macroporous Scaffolds
3.4. Alginate as a Bioink and 3D Bioprinting
3.5. Cryopreservation
4. Future
4.1. Drug Discovery
4.1.1. Cancer
4.1.2. Safety and Toxicology
4.2. Tissue Engineering and Regenerative Medicine
4.2.1. Skin
4.2.2. Cartilage
4.2.3. Cardiac
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Lee, J.; Cuddihy, M.J.; Kotov, N.A. Three-Dimensional Cell Culture Matrices: State of the Art. Tissue Eng. 2008, 14, 61–86. [Google Scholar] [CrossRef]
- Gevaert, M. Engineering 3D Tissue Systems to Better Mimic Human Biology. Bridge 2012, 42, 48–55. [Google Scholar]
- Weigelt, B.; Lo, A.T.; Park, C.C.; Gray, J.W.; Bissell, M.J. HER2 Signaling Pathway Activation and Response of Breast Cancer Cells to HER2-Targeting Agents Is Dependent Strongly on the 3D Microenvironment. Breast. Cancer Res. Treat. 2010, 122, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Kenny, P.A.; Lee, G.Y.; Myers, C.A.; Neve, R.M.; Semeiks, J.R.; Spellman, P.T.; Lorenz, K.; Lee, E.H.; Barcellos-Hoff, M.H.; Petersen, O.W.; et al. The Morphologies of Breast Cancer Cell Lines in Three-Dimensional Assays Correlate With Their Profiles of Gene Expression. Mol. Oncol. 2007, 1, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.M.; Mytelka, D.S.; Dunwiddie, C.T.; Persinger, C.C.; Munos, B.H.; Lindborg, S.R.; Schacht, A.L. How to Improve R&D Productivity: The Pharmaceutical Industry's Grand Challenge. Nat. Rev. Drug Discov. 2010, 9, 203–214. [Google Scholar] [PubMed]
- Prestwich, G.D. Evaluating Drug Efficacy and Toxicology in Three Dimensions: Using Synthetic Extracellular Matrices in Drug Discovery. Acc. Chem. Res. 2008, 41, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Russell, W.M. S.; Burch, R.L. The Principles of Humane Experimental Technique; Methuen: London, UK, 1959; pp. 1–238. [Google Scholar]
- Prestwich, G.D. Simplifying the Extracellular Matrix for 3-D Cell Culture and Tissue Engineering: a Pragmatic Approach. J. Cell Biochem. 2007, 101, 1370–1383. [Google Scholar] [CrossRef] [PubMed]
- Drury, J.L.; Mooney, D.J. Hydrogels for Tissue Engineering: Scaffold Design Variables and Applications. Biomaterials 2003, 24, 4337–4351. [Google Scholar] [CrossRef] [PubMed]
- Place, E.S.; George, J.H.; Williams, C.K.; Stevens, M.M. Synthetic Polymer Scaffolds for Tissue Engineering. Chem. Soc. Rev. 2009, 38, 1139–1151. [Google Scholar] [CrossRef] [PubMed]
- Mironi-Harpaz, I.; Wang, D.Y.; Venkatraman, S.; Seliktar, D. Photopolymerization of Cell-Encapsulating Hydrogels: Crosslinking Efficiency Versus Cytotoxicity. Acta Biomater. 2012, 8, 1838–1848. [Google Scholar] [CrossRef] [PubMed]
- Rosso, F.; Giordano, A.; Barbarisi, M.; Barbarisi, A. From Cell-ECM Interactions to Tissue Engineering. J Cell Physiol. 2004, 199, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Lim, F.; Sun, A.M. Microencapsulated Islets As Bioartifical Endocrine Pancreas. Science 1980, 210, 908–910. [Google Scholar] [CrossRef] [PubMed]
- Smidsrød, O.; Skjåk-Bræk, G. Alginate As Immobilization Matrix for Cells. TIBTECH 1990, 8, 71–78. [Google Scholar] [CrossRef]
- Standard Guide for Immobilization of Living Cells or Tissue in Alginate Gels. In Annual Book of ASTM Standards 2011; ASTM International: West Conshohocken, PA, USA, 2011.
- Helgerud, T.; Gåserød, O.; Fjæreide, T.; Andersen, P.O.; Larsen, C.K. Alginates. In Food Stabilizers, Thickeners and Gelling Agents; Imeson, A., Ed.; Wiley-Blackwell: Oxford, UK, 2009. [Google Scholar]
- Andersen, T.; Strand, B.L.; Formo, K.; Alsberg, E.; Christensen, B.E. Alginates As Biomaterials in Tissue Engineering. In Carbohydrate Chemistry; The Royal Society of Chemistry: Cambridge, UK, 2012; Volume 37, Chapter 9. [Google Scholar]
- Andersen, T. Alginate Foams as Biomaterials. Ph.D. Thesis, Norwegian University of Science and Technology, Trondheim, Norway, 2013. [Google Scholar]
- Atkins, E.D.; Mackie, W.; Parker, K.D.; Smolko, E.E. Crystalline Structures of Poly-D-Mannuronic and Poly-L-Guluronic Acid. J. Polymer Sci. B Polymer Lett. 1971, 9, 311–316. [Google Scholar] [CrossRef]
- Atkins, E.D.; Mackie, W.; Smolko, E.E. Crystalline Structures of Alginic Acids. Nature 1970, 225, 626–628. [Google Scholar] [CrossRef] [PubMed]
- Haug, A. Composition and properties of alginates. Ph.D. Thesis, University of Trondheim, Trondheim, Norway, 1964. [Google Scholar]
- Standard Guide for Characterization and Testing of Alginates as Starting Materials Intended for Use in Biomedical and Tissue-Engineered Medical Products Application. In Annual Book of ASTM Standards 2011; ASTM International: West Conshohocken, PA, USA, 2007.
- Wright, S.D.; Ramos, R.A.; Tobias, P.S.; Ulevitch, R.J.; Mathison, J.C. CD14, a receptor for complexes of lipopolysaccharide (LPS) and LPS binding protein. Science 1990, 249, 1431–1433. [Google Scholar]
- Smidsrød, O.; Haug, A. Dependence Upon Uronic Acid Composition of Some Ion-Exchange Properties of Alginates. Acta Chem. Scand. 1968, 22, 1989–1997. [Google Scholar] [CrossRef]
- Haug, A.; Smidsrød, O. Selectivity of Some Anionic Polymers for Divalent Metal Ions. Acta Chem. Scand. 1970, 24, 843–854. [Google Scholar] [CrossRef]
- Smidsrød, O. Molecular Basis for Some Physical Properties of Alginates in the Gel State. Faraday Discussions of the Chemical Society 1974, 57, 263–274. [Google Scholar] [CrossRef]
- Donati, I.; Holtan, S.; Mørch, Y.A.; Borgogna, M.; Dentini, M.; Skjåk-Bræk, G. New Hypothesis on the Role of Alternating Sequences in Calcium-Alginate Gels. Biomacromolecules 2005, 6, 1031–1040. [Google Scholar] [CrossRef] [PubMed]
- Haug, A.; Smidsrod, O. The Effect of Divalent Metals on the Properties of Alginate Solutions 2. Comparison of Different Metal Ions. Acta Chem. Scand. 1965, 19, 341–351. [Google Scholar] [CrossRef]
- Gombotz, W.R.; Wee, S.F. Protein Release From Alginate Matrices. Adv. Drug Delivery Rev. 1998, 31, 267–285. [Google Scholar] [CrossRef]
- Schmidt, J.J.; Rowley, J.; Kong, H.J. Hydrogels Used for Cell-Based Drug Delivery. J. Biomed. Mater. Res. 2008, 87A, 1113–1122. [Google Scholar] [CrossRef]
- Martinsen, A.; Storrø, I.; Skjåk-Bræk, G. Alginate As Immobilization Material: III. Diffusional Properties. Biotechnol. Bioeng. 1992, 39, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Martinsen, A.; Skjåk-Bræk, G.; Smidsrød, O. Alginate As Immobilization Material: I. Correlation Between Chemical and Physical Properties of Alginate Gel Beads. Biotechnol. Bioeng. 1989, 33, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Donati, I.; Paoletti, S. Material Properties of Alginates. In Alginates: Biology and Applications; Rehm, B.H.A., Ed.; Springer: Berlin/Heidelberg, Germany, 2009; Volume 13, pp. 1–53. [Google Scholar]
- Kuo, C.K.; Ma, P.X. Ionically Crosslinked Alginate Hydrogels As Scaffolds for Tissue Engineering: Part 1. Structure, Gelation Rate and Mechanical Properties. Biomaterials 2001, 22, 511–521. [Google Scholar] [CrossRef] [PubMed]
- Landa, N.; Miller, L.; Feinberg, M.S.; Holbova, R.; Shachar, M.; Cohen, S.; Leor, J. Effect of Injectable Alginate Implant on Cardiac Remodeling and Function After Recent and Old Infarcts in Rat. Circulation 2008, 117, 1388–1396. [Google Scholar] [CrossRef] [PubMed]
- Draget, K.I.; Østgaard, K.; Smidsrød, O. Homogeneous Alginate Gels: a Technical Approach. Carbohydrate Polymers 1991, 14, 159–178. [Google Scholar] [CrossRef]
- Moe, S.T.; Draget, K.I.; Skjåk-Bræk, G.; Smidsrød, O. Alginates. In Food Polysaccharides; Stephen, A., Ed.; Marcel Dekker: New York, NY, USA, 1998. [Google Scholar]
- Draget, K.I.; Østgaard, K.; Smidsrød, O. Alginate-Based Solid Media for Plant Tissue Culture. Appl. Microbiol. Biotechnol. 1989, 31, 79–83. [Google Scholar]
- Alsberg, E.; Jeon, O. Photocrosslinked biodegradable hydrogel. U.S. Patent 8,273,373, 25 September 2012. [Google Scholar]
- Lee, K.Y.; Bouhadir, K.H.; Mooney, D.J. Controlled Degradation of Hydrogels Using Multi-Functional Cross-Linking Molecules. Biomaterials 2004, 25, 2461–2466. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and Biomedical Applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef] [PubMed]
- Rouillard, A.D.; Berglund, C.M.; Lee, J.Y.; Polacheck, W.J.; Tsui, Y.; Bonassar, L.J.; Kirby, B.J. Methods for Photocrosslinking Alginate Hydrogel Scaffolds With High Cell Viability. Tissue Eng. C Meth. 2011, 17, 173–179. [Google Scholar] [CrossRef]
- Jeon, O.; Powell, C.; Ahmed, S.M.; Alsberg, E. Biodegradable, Photocrosslinked Alginate Hydrogels With Independently Tailorable Physical Properties and Cell Adhesivity. Tissue Eng. A 2010, 16, 2915–2925. [Google Scholar] [CrossRef]
- Jeon, O.; Bouhadir, K.H.; Mansour, J.M.; Alsberg, E. Photocrosslinked Alginate Hydrogels With Tunable Biodegradation Rates and Mechanical Properties. Biomaterials 2009, 30, 2724–2734. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, K.B.; Bidarra, S.J.; Oliveira, M.J.; Granja, P.L.; Barrias, C.C. Molecularly Designed Alginate Hydrogels Susceptible to Local Proteolysis As Three-Dimensional Cellular Microenvironments. Acta Biomater. 2011, 7, 1674–1682. [Google Scholar] [CrossRef] [PubMed]
- Bouhadir, K.H.; Lee, K.Y.; Alsberg, E.; Damm, K.L.; Anderson, K.W.; Mooney, D.J. Degradation of partially oxidized alginate and its potential application for tissue engineering. Biotechnol. Prog. 2001, 17, 945–950. [Google Scholar] [CrossRef] [PubMed]
- Jeon, O.; Alt, D.S.; Ahmed, S.M.; Alsberg, E. The effect of oxidation on the degradation of photocrosslinkable alginate hydrogels. Biomaterials 2012, 33, 3503–3514. [Google Scholar] [CrossRef] [PubMed]
- Kristiansen, K.A.; Tomren, H.B.; Christensen, B.E. Periodate oxidized alginates: Depolymerization kinetics. Carbohydr. Polymer. 2011, 86, 1595–1601. [Google Scholar] [CrossRef]
- Yang, J.-S.; Xie, Y.-J.; He, W. Research Progress on Chemical Modification of Alginate: A Review. Carbohydrate Polymers 2011, 84, 33–39. [Google Scholar] [CrossRef]
- Pawar, S.N.; Edgar, K.J. Alginate Derivatization: A Review of Chemsitry, Properties and Applications. Biomaterials 2012, 33, 3279–3305. [Google Scholar] [CrossRef] [PubMed]
- West, J.L. Biofunctional polymers. In Encyclopedia of Biomaterials and Biomedical Engineering; Bowlin, G.L., Ed.; Marcel Dekker, Inc.: New York, NY, USA, 2004; Volume 1, pp. 89–95. [Google Scholar]
- Vacharathit, V.; Silva, E.A.; Mooney, D.J. Viability and Functionality of Cells Delivered From Peptide Conjugated Scaffolds. Biomaterials 2011, 32, 3721–3728. [Google Scholar] [CrossRef] [PubMed]
- Hsiong, S.X.; Huebsch, N.; Fischbach, C.; Kong, H.J.; Mooney, D.J. Integrin-Adhesion Ligand Bond Formation of Preosteoblasts and Stem Cells in Three-Dimensional RGD Presenting Matrices. Biomacromolecules 2008, 9, 1843–1851. [Google Scholar] [CrossRef] [PubMed]
- Sapir, Y.; Kryukov, O.; Cohen, S. Integration of Multiple Cell-Matrix Interactions into Alginate Scaffolds for Promoting Cardiac Tissue Regeneration. Biomaterials 2011, 32, 1838–1847. [Google Scholar] [CrossRef] [PubMed]
- Melvik, J.E.; Dornish, M. Alginate As a Carrier for Cell Immobilisation. In Fundamentals of Cell Immobilisation Biotechnology; Nedovic, V., Willaert, R., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2004. [Google Scholar]
- Rowley, J.A.; Madlambayan, G.; Mooney, D.J. Alginate Hydrogels As Synthetic Extracellular Matrix Materials. Biomaterials 1999, 20, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Alsberg, E.; Anderson, K.W.; Albeiruti, A.; Rowley, J.A.; Mooney, D.J. Engineering Growing Tissues. Proc. Natl. Acad. Sci. USA 2002, 99, 12025–12030. [Google Scholar] [CrossRef] [PubMed]
- Cell types cultured in NovaMatrix-3D. 2014. Available online: http://novamatrix.biz/Portals/Novamatrix3D/Content/Docs/NovaMatrix-3D_Cell%20types.pdf, NovaMatrix (accessed on 30 January 2015).
- Rowley, J.A.; Mooney, D.J. Alginate Type and RGD Density Control Myoblast Phenotype. J. Biomed. Mater Res. 2002, 60, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Example studies with NovaMatrix-3D. 2014. Available online: http://novamatrix.biz/3d/Documentation/Examples.aspx, NovaMatrix (accessed on 30 January 2015).
- Lee, K.Y.; Alsberg, E.; Hsiong, S.; Comisar, W.; Linderman, J.; Ziff, R.; Mooney, D. Nanoscale Adhesion Ligand Organization Regulates Osteoblast Proliferation and Differentiation. Nano Lett. 2004, 4, 1501–1506. [Google Scholar] [CrossRef] [PubMed]
- Fischbach, C.; Kong, H.J.; Hsiong, S.X.; Evangelista, M.B.; Yuen, W.; Mooney, D.J. Cancer cell angiogenic capability is regulated by 3-D culture and integrin engagement. Proc. Natl. Acad. Sci. USA 2009, 106, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Hsiong, S.X.; Boontheekul, T.; Huebsch, N.; Mooney, D.J. Cyclic Arginine-Glycine-Aspartate Peptides Enhance Three-Dimensional Stem Cell Osteogenic Differentiation. Tissue Eng. A 2009, 2, 263–272. [Google Scholar] [CrossRef]
- Perlin, L.; MacNeil, S.; Rimmer, S. Production and Performance of Biomaterials Containing RGD Peptides. Soft Matter 2008, 4, 2331–2349. [Google Scholar] [CrossRef]
- Dhoot, N.O.; Tobias, C.A.; Fischer, I.; Wheatley, M.A. Peptide-modified Alginate Surfaces as a Growth Permissive Substrate for Neurite Outgrowth. J. Biomed. Mater. Res. A 2004, 71, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Justice, B.A.; Badr, N.A.; Felder, R.A. 3D Cell Culture Opens New Dimensions in Cell-Based Assays. Drug Discovery Today 2009, 14, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Buxboim, A.; Ivanovska, I.L.; Discher, D.E. Matrix Elasticity, Cytoskeletal Forces and Physics of the Nucleus: How Deeply Do Cells 'Feel' Outside and in? J. Cell Sci. 2010, 123, 297–308. [Google Scholar] [CrossRef] [PubMed]
- Reilly, G.C.; Engler, A.J. Intrinsic Extracellular Matrix Properties Regulate Stem Cell Differentiation. J. Biomech. 2010, 43, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Huebsch, N.; Arany, P.R.; Mao, A.S.; Shvartsman, D.; Ali, O.A.; Bencherif, S.A.; Rivera-Feliciano, J.; Mooney, D.J. Harnessing Traction-Mediated Manipulation of the Cell/Matrix Interface to Control Stem-Cell Fate. Nat. Mater. 2010, 9, 518–526. [Google Scholar] [CrossRef]
- Yamamoto, M.; James, D.; Li, H.; Butler, J.; Rafii, S.; Rabbany, S. Generation of Stable Co-Cultures of Vascular Cells in a Honeycom Alginate Scaffold. Tissue Eng. Part A 2010, 16, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Sapir, Y.; Cohen, S.; Friedman, G.; Polyak, B. The Promotion of In Vitro Vessel-like Organization of Endothelial Cells in Magnetically Responsive Alginate Scaffolds. Biomaterials 2012, 33, 4100–4109. [Google Scholar] [CrossRef] [PubMed]
- Suárez-González, D.; Barnhart, K.; Saito, E.; Vanderby, R.; Hollister, S.J.; Murphy, W.L. Controlled nucleation of hydroxyapatite on alginate scaffolds for stem cell-based bone tissue engineering. J. Biomed. Mater. Res. A 2010, 95, 222–234. [Google Scholar] [CrossRef] [PubMed]
- Westhrin, M.; Xie, M.; Olderøy, M.Ø.; Sikorski, P.; Strand, B.L.; Standal, T. (2015) Osteogenic Differentiation of Human Mesenchymal Stem Cells in Mineralized Alginate Matrices. PLoS One 2015, 10, e0120374. [Google Scholar] [CrossRef] [PubMed]
- Discher, D.E.; Mooney, D.J.; Zandstra, P.W. Growth Factors, Matrices, and Forces Combine and Control Stem Cells. Science 2009, 324, 1673–1677. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Arha, M.; Choudhary, S.; Ashton, R.S.; Bhatia, S.R.; Schaffer, D.V.; Kane, R.S. The Influence of Hydrogel Modulus on the Proliferation and Differentiation of Encapsulated Neural Stem Cells. Biomaterials 2009, 30, 4695–4699. [Google Scholar] [CrossRef] [PubMed]
- Engler, A.J.; Sen, S.; Sweeney, H.L.; Discher, D.E. Matrix Elasticity Directs Stem Cell Lineage Specification. Cell 2006, 126, 677–689. [Google Scholar] [CrossRef] [PubMed]
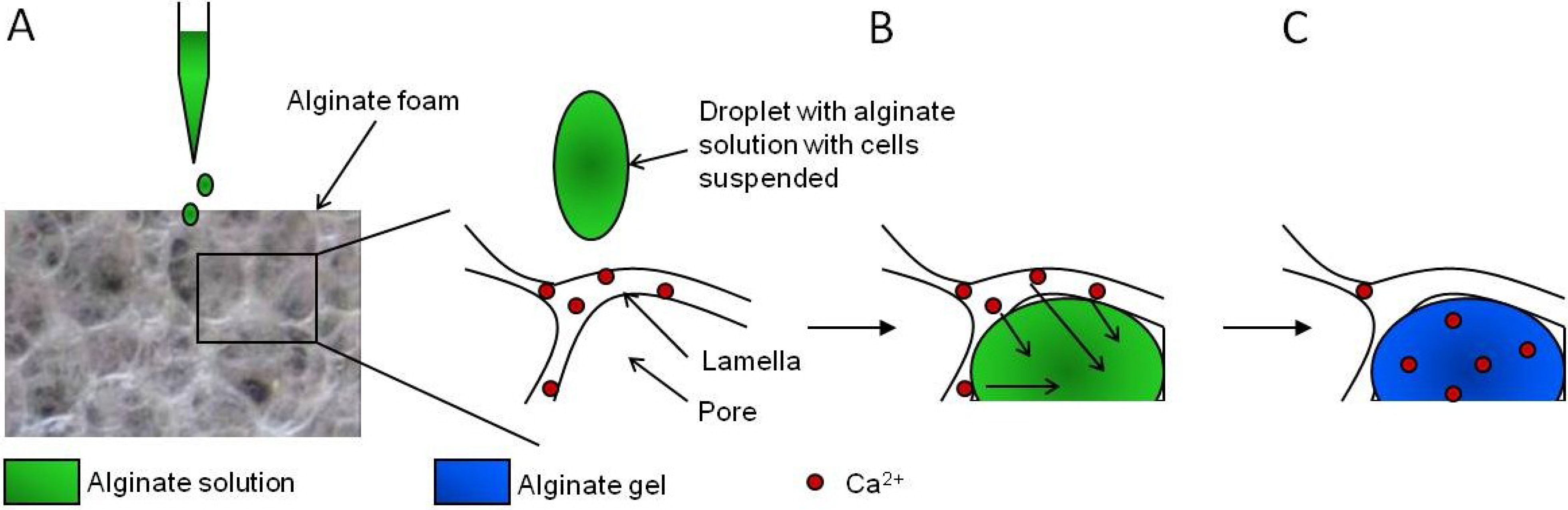
- Andersen, T.; Markussen, C.; Dornish, M.; Heier-Baardson, H.; Melvik, J.E.; Alsberg, E.; Christensen, B.E. In Situ Gelation for Cell Immobilization and Culture in Alginate Foam Scaffolds. Tissue Eng. A 2013, 20, 600–610. [Google Scholar]
- Skjåk-Bræk, G.; Espevik, T. Application of Alginate Gels in Biotechnology and Biomedicine. Carbohydr. Eur. 1996, 14, 19–25. [Google Scholar]
- Dulieu, C.; Poncelet, D.; Neufeld, R.J. Encapsulation and Immobilization Techniques. In Cell Encapsulation Technology and Therapeutics; Kühtreiber, W.M., Lanza, R.P., Chick, W.L., Eds.; Birkhäuser: Boston, MA, USA, 1999. [Google Scholar]
- Bead generators. 2014. Available online: http://www.novamatrix.biz/Products/BeadGenerators.aspx, NovaMatrix (accessed online 6 February 2015).
- Soon-Shiong, P.; Feldman, E.; Nelson, R.; Heintz, R.; Yao, Q.; Yao, Z.; Zheng, T.; Merideth, N.; Skjåk-Bræk, G.; Espevik, T.; Smidsrød, O.; Sandford, P. Long-Term Reversal of Diabetes by the Injection of Immunoprotected Islets. Proc. Natl. Acad. Sci. USA 1993, 90, 5843–5847. [Google Scholar] [CrossRef] [PubMed]
- Calafiore, R.; Basta, G.; Luca, G.; Lemmi, A.; Montanucci, M.P.; Calabrese, G.; Racanicchi, L.; Mancuso, F.; Brunetti, P. Microencapsulated Pancreatic Islet Allografts into Nonimmunosuppressed Patients With Type 1 Diabetes. Diabetes Care 2006, 29, 137–138. [Google Scholar] [CrossRef] [PubMed]
- Tuch, B.E.; Keogh, G.W.; Williams, L.J.; Wu, W.; Foster, J.L.; Vaithilingam, V.; Philips, R. Safety and Viability of Microencapsulated Human Islets Transplanted into Diabetic Humans. Diabetes Care 2009, 32, 1887–1889. [Google Scholar] [CrossRef] [PubMed]
- Bjerkvig, R.; Read, T.-A.; Vajkoczy, P.; Aebischer, P.; Pralong, W.; Platt, S.; Melvik, J.E.; Hagen, A.; Dornish, M. Cell Therapy Using Encapsulated Cells Producing Endostatin. Acta Neurochir. Suppl. 2003, 88, 137–141. [Google Scholar] [PubMed]
- Read, T.-A.; Sørensen, D.R.; Mahesparan, R.; Enger, P.Ø.; Timpl, R.; Olsen, B.R.; Hjelstuen, M.; Haraldseth, O.; Bjerkvig, R. Local Endostatin Treatment of Gliomas Administered by Microcapsulated Producer Cells. Nat. Biotechnol. 2001, 19, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Read, T.-A.; Farhadi, M.; Holtan, S.; Olsen, B.R.; Huzthy, P.; Bjerkvig, R.; Vajkoczy, P. Intravital Microscopy reveals novel anti-vascular and tumour effects ef endostatin delivered locally by alginate encapsulated cells. Cancer Res. 2001, 61, 6830–6837. [Google Scholar] [PubMed]
- Moyer, H.R.; Kinney, R.C.; Singh, K.A.; Williams, J.K.; Schwartz, Z.; Boyan, B.D. Alginate microencapsulation technology for the percutaneous delivery of adipose-derived stem cells. Ann. Plast. Surg. 2010, 65, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Khosravizadeh, Z.; Razavi, S.; Bahramian, H.; Kazemi, M. The beneficial effect of encapsulated human adipose-derived stem cells in alginate hydrogel on neural differentiation. J. Biomed. Mater. Res. B Appl. Biomater. 2014, 102, 749–755. [Google Scholar] [CrossRef] [PubMed]
- Olderøy, M.Ø.; Lilledahl, M.B.; Sandvold, M.; Melvik, J.E.; Reinholt, F.; Sikorski, P.; Brinchmann, J.E. Biochemical and structural characterization of neocartilage formed by mesenchymal stem cells in alginate hydrogels. PLoS One 2014, 9, e91662. [Google Scholar] [CrossRef] [PubMed]
- Bidarra, S.J.; Barrias, C.C.; Barbosa, M.A.; Soares, R.; Granja, P.L. Immobilization of human mesenchymal stem cells within RGD-grafted alginate microspheres and assessment of their angiogenic potential. Biomacromolecules 2010, 11, 1956–1964. [Google Scholar] [CrossRef] [PubMed]
- Bittencourt, R.A. C.; Pereira, H.R.; Felisbino, S.L.; Ferreira, R.R.; Guilherme, G.R. B.; Moroz, A.; Deffune, E. Chondrocyte cultures in tridimensional scaffold: alginate hydrogel. Acta Ortop. Bras. 2009, 17, 242–246. [Google Scholar] [CrossRef]
- Lin, Y.-J.; Yen, C.-N.; Hu, Y.-C.; Liao, C.-J.; Chu, I.-M. Chondrocytes culture in three-dimensional porous alginate scaffolds enhanced cell proliferation, matrix synthesis and gene expression. J. Biomed. Mater. Res. 2008, 88A, 23–33. [Google Scholar]
- NTCELL® for Parkinson's disease. Available online: http://www.lctglobal.com/products/ntcell/about-parkinsons-disease, Living Cell Technologies (accessed on 20 March 2015).
- DIABECELL® for type 1 diabetes. Available online: http://www.lctglobal.com/products/diabecell/about-type-1-diabetes, Living Cell Technologies (accessed on 20 March 2015).
- Herlofsen, S.R.; Küchler, A.M.; Melvik, J.E.; Brinchmann, J.E. Chondrogenic Differentiation of Human Bone Marrow-Derived Mesenchymal Stem Cells in Self-Gelling Alginate Discs Reveals Novel Chondrogenic Signature Gene Clusters. Tissue Eng. A 2011, 17, 1003–1013. [Google Scholar] [CrossRef]
- Jang, J.; Seol, Y.J.; Kim, H.J.; Kundu, J.; Kim, S.W.; Cho, D.W. Effects of alginate hydrogel cross-linking density on mechanical and biological behaviors for tissue engineering. J. Mech. Behav. Biomed. Mater 2014, 37, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Alsberg, E.; Anderson, K.W.; Albeiruti, A.; Franceschi, R.T.; Mooney, D.J. Cell-Interactive Alginate Hydrogels for Bone Tissue Engineering. J. Dent. Res. 2001, 80, 2025–2029. [Google Scholar] [CrossRef] [PubMed]
- Atala, A.; Kim, W.; Paige, K.T.; Vacanti, C.A.; Retik, A.B. Endoscopic Treatment of Vesicoureteral Reflux With a Chondrocyte-Alginate Suspension. J. Urol. 1994, 152, 641–643. [Google Scholar] [PubMed]
- Principle of self-gelling alginate. 2011. Available online: http://novamatrix.biz/Portals/novamatrix/Content/Docs/Technology/Injectable%20alginate%20self%20gelling%20technology-060911.pdf, NovaMatrix (accessed on 30 January 2015).
- Hwang, C.M.; Sant, S.; Masaeli, M.; Kachouie, N.N.; Zamanian, B.; Lee, S.H.; Khademhosseini, A. Fabrication of Three-Dimensional Porous Cell-Laden Hydrogel for Tissue Engineering. Biofabrication 2010, 2, 035003. [Google Scholar] [CrossRef] [PubMed]
- Thornton, A.J.; Alsberg, E.; Hill, E.E.; Mooney, D.J. Shape Retaining Injectable Hydrogels for Minimally Invasive Bulking. J. Urol. 2004, 172, 763–768. [Google Scholar] [CrossRef] [PubMed]
- Henderson, T.M. A.; Ladewig, K.; Haylock, D.N.; McLean, K.M.; O'Connor, A.J. Cryogels for Biomedical Applications. J. Mater. Chem. 2013, 1, 2682–2695. [Google Scholar] [CrossRef]
- Dvir-Ginzberg, M.; Elkayam, T.; Cohen, S. Induced Differentiation and Maturation of Newborn Liver Cells into Functional Hepatic Tissue in Macroporous Alginate Scaffolds. FASEB J. 2008, 22, 1440–1449. [Google Scholar] [CrossRef] [PubMed]
- Zmora, S.; Glicklis, R.; Cohen, S. Tailoring the Pore Architecture in 3-D Alginate Scaffolds by Controlling the Freezing Regime During Fabrication. Biomaterials 2002, 23, 4087–4094. [Google Scholar] [CrossRef] [PubMed]
- Soletti, L.; Nieponice, A.; Guan, J.; Stankus, J.J.; Wagner, W.R.; Vorp, D.A. A Seeding Device for Tissue Engineered Tubular Structures. Biomaterials 2006, 27, 4863–4870. [Google Scholar] [CrossRef] [PubMed]
- Papadimitropoulos, A.; Riboldi, S.A.; Tonnarelli, B.; Piccinini, E.; Woodruff, M.A.; Hutmacher, D.W.; Martin, I. A Collagen Network Phase Improves Cell Seeding of Open-Pore Structure Scaffolds Under Perfusion. J. Tissue Eng. Regen. Med. 2013, 7, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Dar, A.; Shachar, M.; Leor, J.; Cohen, S. Optimization of Cardiac Cell Seeding and Distribution in 3D Porous Alginate Scaffolds. Biotechnol. Bioeng. 2002, 80, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Solchaga, L.A.; Tognana, E.; Penick, K.; Baskaran, H.; Goldberg, V.M.; Caplan, A.I.; Welter, J.F. A Rapid Seeding Technique for the Assembly of Large Cell/Scaffold Composite Constructs. Tissue Eng. 2006, 12, 1851–1863. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Michaud, H.; Bhowmick, S. Controlled Vacuum Seeding As a Means of Generating Uniform Cellular Distribution in Electrospun Polycaprolactone (PCL) Scaffolds. J. Biomech. Eng. 2009, 131, 074521. [Google Scholar] [CrossRef] [PubMed]
- Godugu, C.; Patel, A.R.; Desai, U.; Andey, T.; Sams, A.; Singh, M. AlgiMatrix Based 3D Cell Culture System As an in-Vitro Tumor Model for Anticancer Studies. PLoS One 2013, 8, e53708. [Google Scholar] [CrossRef] [PubMed]
- Murphy, S.V.; Atala, A. 3D Bioprinting of Tissues and Organs. Nat. Biotechnol. 2014, 32, 773–785. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Zhang, Y.; Martin, J.A.; Ozbolat, I.T. Evaluation of Cell Viability and Functionality in Vessel-Like Bioprintable Cell-Laden Tubular Channels. J Biomech. Eng. 2013, 135, 91011. [Google Scholar] [CrossRef] [PubMed]
- Lee, V.; Singh, G.; Trasatti, J.P.; Bjornsson, C.; Xu, X.; Tran, T.N.; Yoo, S.S.; Dai, G.; Karande, P. Design and Fabrication of Human Skin by Three-Dimensional Bioprinting. Tissue Eng. C Meth. 2014, 20, 473–484. [Google Scholar] [CrossRef]
- Cui, X.; Breitenkamp, K.; Finn, M.G.; Lotz, M.; D'Lima, D.D. Direct Human Cartilage Repair Using Three-Dimensional Bioprinting Technology. Tissue Eng. A 2012, 18, 1304–1312. [Google Scholar] [CrossRef]
- Tasoglu, S.; Demirci, U. Bioprinting for Stem Cell Research. Trends Biotechnol. 2013, 31, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Public Workshop—Additive Manufacturing of Medical Devices: An Interactive Discussion on the Technical Considerations of 3D Printing, October 8–9, 2014. Available online: http://www.fda.gov/MedicalDevices/NewsEvents/WorkshopsConferences/ucm397324.htm, U.S. Food and Drug Administration (accessed on 20 March 2015).
- Khalil, S.; Sun, W. Bioprinting Endothelial Cells With Alginate for 3D Tissue Constructs. J. Biomech. Eng. 2009, 131, 111002. [Google Scholar] [CrossRef] [PubMed]
- Neufurth, M.; Wang, X.; Schroder, H.C.; Feng, Q.; Diehl-Seifert, B.; Ziebart, T.; Steffen, R.; Wang, S.; Muller, W.E. Engineering a Morphogenetically Active Hydrogel for Bioprinting of Bioartificial Tissue Derived From Human Osteoblast-Like SaOS-2 Cells. Biomaterials 2014, 35, 8810–8819. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Tolba, E.; Schroder, H.C.; Neufurth, M.; Feng, Q.; Diehl-Seifert, B.; Muller, W.E. Effect of Bioglass on Growth and Biomineralization of SaOS-2 Cells in Hydrogel After 3D Cell Bioprinting. PLoS One 2014, 9, e112497. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Yao, R.; Ouyang, L.; Ding, H.; Zhang, T.; Zhang, K.; Cheng, S.; Sun, W. Three-Dimensional Printing of Hela Cells for Cervical Tumor Model in Vitro. Biofabrication 2014, 6, 035001. [Google Scholar] [CrossRef] [PubMed]
- Jeon, O.; Alt, D.S.; Ahmed, S.M.; Alsberg, E. The Effect of Oxidation on the Degradation of Photocrosslinkable Alginate Hydrogels. Biomaterials 2012, 33, 3503–3514. [Google Scholar] [CrossRef] [PubMed]
- Jia, J.; Richards, D.J.; Pollard, S.; Tan, Y.; Rodriguez, J.; Visconti, R.P.; Trusk, T.C.; Yost, M.J.; Yao, H.; Markwald, R.R.; Mei, Y. Engineering Alginate As Bioink for Bioprinting. Acta Biomater. 2014, 10, 4323–4331. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Zhang, M.; Huang, Y.; Ogale, A.; Fu, J.; Markwald, R.R. Study of Droplet Formation Process During Drop-on-Demand Inkjetting of Living Cell-Laden Bioink. Langmuir 2014, 30, 9130–9138. [Google Scholar] [CrossRef] [PubMed]
- Gasperini, L.; Maniglio, D.; Motta, A.; Migliaresi, C. An Electrohydrodynamic Bioprinter for Alginate Hydrogels Containing Living Cells. Tissue Eng. C. Meth. 2015, 21, 123–132. [Google Scholar] [CrossRef]
- Karlsson, J.O.; Toner, M. Long-Term Storage of Tissues by Cryopreservation: Critical Issues. Biomaterials 1996, 17, 243–256. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, H.; Ehrhart, F.; Zimmermann, D.; Müller, K.; Katsen-Globa, A.; Behringer, M.; Feilen, P.J.; Gessner, P.; Zimmermann, G.; Shirley, S.G.; et al. Hydrogel-Based Encapsulation of Biological, Functional Tissue: Fundamentals, Technologies and Applications. Appl. Phys. A 2007, 89, 909–922. [Google Scholar] [CrossRef]
- Zhang, W.; He, X. Microencapsulating and Banking Living Cells for Cell-Based Medicine. J. Healthc. Eng. 2011, 2, 427–446. [Google Scholar] [CrossRef] [PubMed]
- Abramson, S. Methods for cryopreserving and encapsulating cells. Anthrogenesis Corp. PCT/US2011/067716[WO 2012/092420], 29 December 2010. [Google Scholar]
- Mukherjee, N.; Chen, Z.; Sambanis, A.; Song, Y. Effects of Cryopreservation on Cell Viability and Insulin Secretion in a Model Tissue-Engineered Pancreatic Substitute (TEPS). Cell Transplant. 2005, 14, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Kupchik, H.Z.; Collins, E.A.; O'Brien, M.J.; McCaffrey, R.P. Chemotherapy Screening Assay Using 3-Dimensional Cell Culture. Cancer Lett. 1990, 51, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Burdett, E.; Kasper, F.K.; Mikos, A.G.; Ludwig, J.A. Engineering Tumors: A Tissue Engineering Perspective in Cancer Biology. Tissue Eng. B Rev. 2010, 16, 351–359. [Google Scholar] [CrossRef]
- Lan, S.-F.; Safiejko-Mroczka, B.; Starly, B. Long-Term Cultivation of HepG2 Liver Cells Encapsulated in Alginate Hydrogels: A Study of Cell Viability, Morphology and Drug Metabolism. Toxicol. Vitro 2010, 24, 1314–1323. [Google Scholar] [CrossRef]
- Tran, N.M.; Dufresne, M.; Duverlie, G.; Castelain, S.; Defarge, C.; Paullier, P.; Legallais, C. An Appropriate Selection of a 3D Alginate Culture Model for Hepatic Huh-7 Cell Line Encapsulation Intended for Viral Studies. Tissue Eng. A 2013, 19, 103–113. [Google Scholar] [CrossRef]
- Miranda, J.P.; Rodrigues, A.; Tostöes, R.M.; Leite, S.; Zimmermann, H.; Carrondo, M.J. T.; Alves, P.M. Extending Hepatocyte Functionality for Drug-Testing Applications Using High-Viscosity Alginate–Encapsulated Three-Dimensional Cultures in Bioreactors. Tissue Eng. C. Meth. 2010, 16, 1223–1232. [Google Scholar] [CrossRef]
- Oh, J.W.; Hsi, T.C.; Guerrero-Juarez, C.F.; Ramos, R.; Plikus, M.V. Organotypic Skin Culture. J. Invest. Dermatol. 2013, 133, e14. [Google Scholar] [CrossRef] [PubMed]
- Canton, I.; Cole, D.M.; Kemp, E.H.; Watson, P.F.; Chunthapong, J.; Ryan, A.J.; MacNeil, S.; Haycock, J.W. Development of a 3D Human in Vitro Skin Co-Culture Model for Detecting Irritants in Real-Time. Biotechnol. Bioeng. 2010, 106, 794–803. [Google Scholar] [CrossRef] [PubMed]
- Olderøy, M.Ø.; Lilledahl, M.B.; Sandvold, M.; Melvik, J.E.; Reinholt, F.; Sikorski, P.; Brinchmann, J.E. Biochemical and Structural Characterization of Neocartilage Formed by Mesenchymal Stem Cells in Alginate Hydrogels. PLoS One 2014, 9, e91662. [Google Scholar] [CrossRef] [PubMed]
- Ceccaldi, C.; Fullana, S.G.; Alfarano, C.; Lairez, O.; Calise, D.; Cussac, D.; Parini, A.; Sallerin, B. Alginate Scaffolds for Mesenchymal Stem Cell Cardiac Therapy: Influence of Alginate Composition. Cell Transplant. 2012, 21, 1969–1984. [Google Scholar] [CrossRef] [PubMed]
- Levit, R.D.; Landazuri, N.; Phelps, E.A.; Brown, M.E.; Garcia, A.J.; Davis, M.E.; Joseph, G.; Long, R.; Safley, S.A.; Suever, J.D.; et al. Cellular Encapsulation Enhances Cardiac Repair. J. Am. Heart Assoc. 2013, 2, e000367. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andersen, T.; Auk-Emblem, P.; Dornish, M. 3D Cell Culture in Alginate Hydrogels. Microarrays 2015, 4, 133-161. https://doi.org/10.3390/microarrays4020133
Andersen T, Auk-Emblem P, Dornish M. 3D Cell Culture in Alginate Hydrogels. Microarrays. 2015; 4(2):133-161. https://doi.org/10.3390/microarrays4020133
Chicago/Turabian StyleAndersen, Therese, Pia Auk-Emblem, and Michael Dornish. 2015. "3D Cell Culture in Alginate Hydrogels" Microarrays 4, no. 2: 133-161. https://doi.org/10.3390/microarrays4020133
APA StyleAndersen, T., Auk-Emblem, P., & Dornish, M. (2015). 3D Cell Culture in Alginate Hydrogels. Microarrays, 4(2), 133-161. https://doi.org/10.3390/microarrays4020133





