3D Cultivation Techniques for Primary Human Hepatocytes
Abstract
:1. Introduction
2. 2D Cultivation Models for Hepatocytes
| Cultivation | Advantages | Disadvantages | Reference |
|---|---|---|---|
| 2D | |||
| Monolayer | Gold standard for drug metabolism and toxicity | Rapid loss of morphology and cell polarity | [8,9,15] |
| Ideal for testing interindividual and interspecies differences in metabolism | Rapid loss of drug metabolizing capability, decrease of albumin production and cell-cell interaction) | ||
| Maintenance of key functions as carbohydrate metabolism and plasma protein synthesis (24–72 h) | Limited availability | ||
| Co-culture | Improved functionality of all cell types | No real standard established | [8,16,17,18,19] |
| Increased expression of phase I and phase II enzymes | High variability between different laboratories | ||
| Maintenance of cell morphology | |||
| Inducibility of CYPs | |||
| 3D | |||
| Hydrogels, scaffolds scaffold-free | Long-term maintenance of liver-specific functions | Lack of established standards | [8,9,15,20,21,22,23,24,25] |
| Increased sensitivity towards drugs | Not adjusted to high throughput | ||
| Long-term expression of phase I and phase II enzymes | Cell recovery for further analysis is difficult | ||
| Co-culture with liver-drived cell types | Improved expression of phase I and phase II enzymes, Including inducibility by drugs | No real standard established | [17,26,27] |
| Not adjusted to high throughput, high variability regarding cell viability and differentiation | |||
| Maintenance of cell polarity, cell-cell contacts and bile canaliculi | |||
| Mimicks in vitro architecture | |||
| Longer cell viability | |||
| Microfluidic devices | Sustained liver like cell functionality and increased liver specific functions | No standardized system available so far | [8,9,15] |
| Not adjusted to high throughput | |||
| Precisely adjusted flow/drug concentrations | |||
| Enable microscopic examination | |||
| Formation of a sinusoid-like shape (HepaChip®) | |||
| Fast differentiation of the cells after flow induction |
2.1. Heptocyte Monolayer Cultivation
2.2. Co-Cultivation of Hepatocytes
2.3. 3D Cultivation Systems for Hepatocytes
2.3.1. Hydrogels
| 3D System | Source | Cell Type | Observation | Reference |
|---|---|---|---|---|
| Hydrogels | ||||
| Natural | Collagen | Human hepatocytes | Sensitivity to acetaminophen-induced intoxication | [3] |
| Matrigel | Human Hepatocytes | Enhanced formation of bile canalicular networks | [38] | |
| Synthetic | PEG | Functional groups can be attached to macromolecules | [41] | |
| +PGLA | Human hepatocytes | Consistent urea synthesis and increased CYP3A4 activity | [33] | |
| +Heparin | Rat hepatocytes | Stable urea and albumin synthesis for more than 3 weeks | [42] | |
| +RGD | Rat hepatocytes | Spheroid formation, maintenance of urea and albumin production for more than 4 weeks | [43] | |
| PuraMatrix™ | Rat hepatocytes | Higher albumin and urea levels for up to 3 weeks | [44] | |
| Scaffolds | ||||
| Natural | Chitosan | Provision of adhesion for hepatocytes | [46] | |
| +Heparin/alginate | Increased albumin synthesis | [47] | ||
| +GHA | Increased albumin secretion and urea synthesis | [48] | ||
| Alginate | Rat hepatocytes | Cell-cell and cell-extracellular matrix (ECM) interactions, phase I and phase II activity stable for one week, high urea and albumin synthesis | [49] | |
| Synthetic | PVA | Rat hepatocytes | Urea synthesis maintained over 5 days | [50] |
| PLA | Rat hepatocytes | Maintenance of albumin and urea synthesis as well as CYP1A and UGT-activity | [51] | |
| PS | Rat hepatocytes | Higher activity of CYP1A2, CYP2B1, and CYP3A2 | [52] | |
| Human hepatocytes | Higher activity of CYP2B6 and CYP3A4 | [37] | ||
| Scaffold-free | ||||
| Spheroids | Human hepatocytes | Metabolism of lamotrigine and salbutamol | [53] | |
| Nanoculture plate | HepG2 | Enhanced expression of albumin, CYPs and liver-enriched transcription factors (HNF4-α and C/EBPα) | [54] |
2.3.2. Scaffolds
2.3.3. Scaffold-Free Cultivation
3. 3D Microfluidic Cultivation Systems for Primary Hepatocytes
| 3D System | Cell Type | Observation | Reference | Manufacturer |
|---|---|---|---|---|
| Hollow fiber bioreactor | Rat hepatocytes Human hepatocytes | Increased albumin synthesis and diclofenac toxicity higher expression of CYP1A2, CYP3A4/5, CYP2C9, CYP2D6, CYP2B6, transporters, and phase II enzymes | [64,66,67,68] | Unisyn |
| Alginate encapsulated hepatocytes in the bioreactor | Rat hepatocytes | Enhanced biotransformation, CYP inducibility, albumin and urea secretion | [69] | Sartorius Stedim |
| Multichamber modular bioreactor | Human hepatocytes | Up-regulation of CYP1A1, 1A2, 2B6, 2C9, 3A4, UGT, MDR1, and MRP2 | [70] | University of Pisa |
| Quasi-Vivo® | Human hepatocytes | Enables investigation of cross-talk between different cell types | [71] | Kirkstall |
| LiverChip | Human hepatocytes | Maintained mRNA-levels of Phase I/II-enzymes over 7 days, higher or similar CYP-activities after day 4 | [72,73] | CN Bio Innovations Limited |
| HepaChip® | Human hepatocytes | Up-regulation of CYP3A4, CYP2A1, and phase II enzymes | [52] | NMI Reutlingen |
| 3D-KITChip | HepG2, rat hepatocytes | Higher level of differentiation | [40] | KIT |
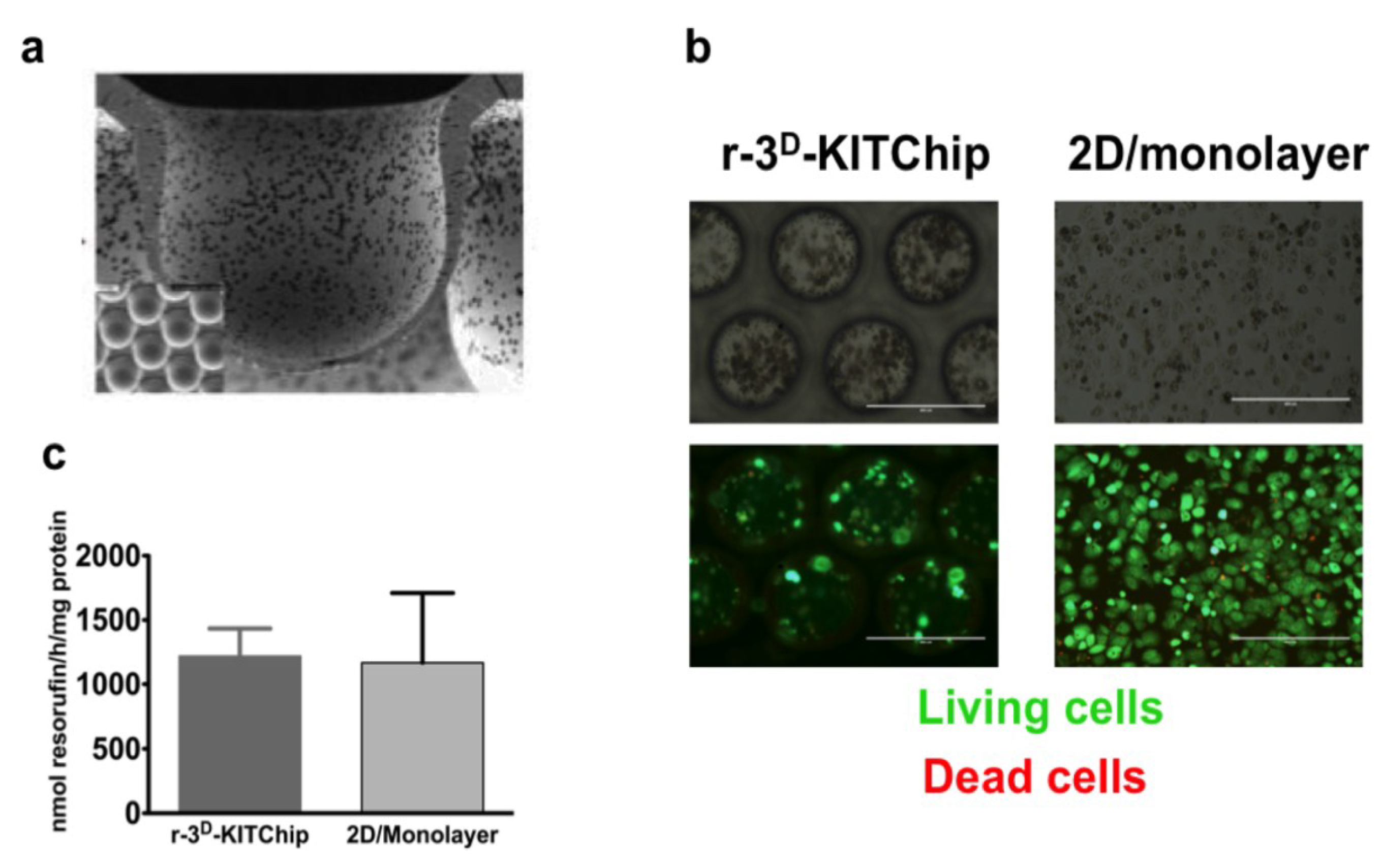
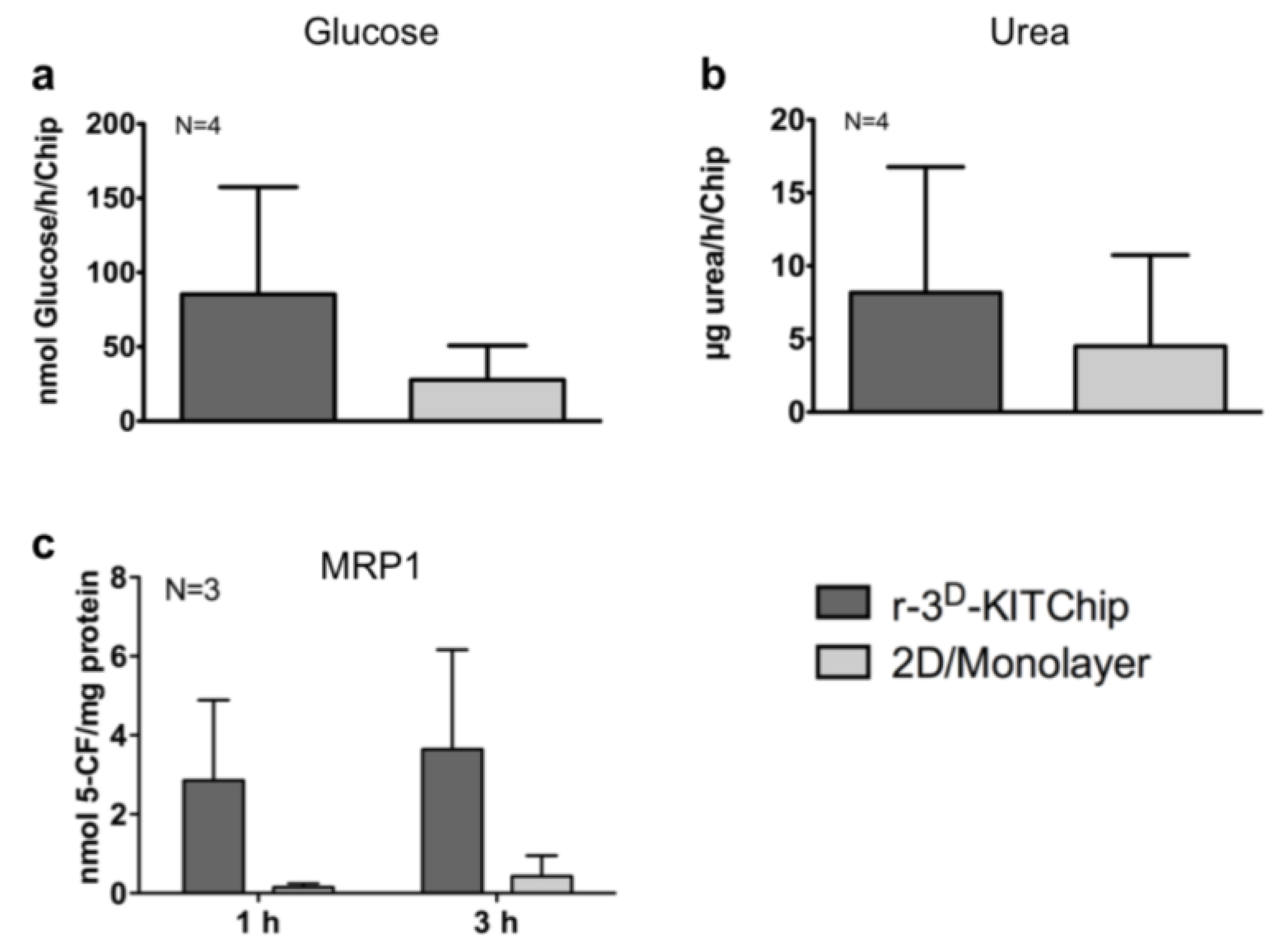

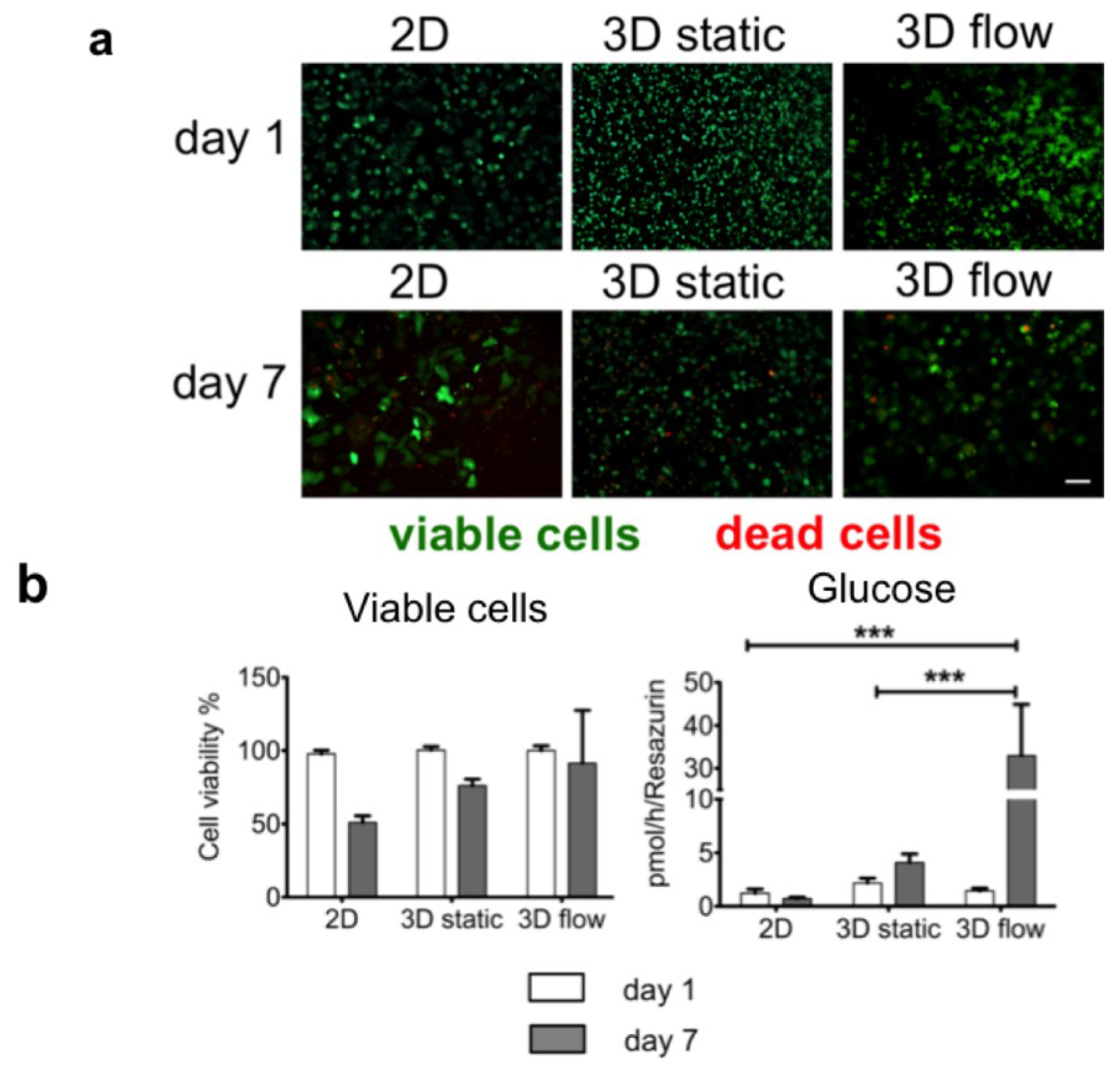
4. Conclusions
Acknowledgments
Author Contributions
Compliance with Ethical Requirements
Conflicts of Interest
References
- Dambach, D.M.; Andrews, B.A.; Moulin, F. New Technologies and Screening Strategies for Hepatotoxicity: Use of in Vitro Models. Toxicol. Pathol. 2004, 33, 17–26. [Google Scholar] [CrossRef]
- De Bruyn, T.; Chatterjee, S.; Fattah, S.; Keemink, J.; Nicolaï, J.; Augustijns, P.; Annaert, P. Sandwich-cultured hepatocytes: Utility for in vitro exploration of hepatobiliary drug disposition and drug-induced hepatotoxicity. Expert Opin. Drug Metab. Toxicol. 2013, 9, 589–616. [Google Scholar]
- Schyschka, L.; Sánchez, J.J.M.; Wang, Z.; Burkhardt, B.; Müller-Vieira, U.; Zeilinger, K.; Bachmann, A.; Nadalin, S.; Damm, G.; Nussler, A.K. Hepatic 3D cultures but not 2D cultures preserve specific transporter activity for acetaminophen-induced hepatotoxicity. Arch. Toxicol. 2013, 87, 1581–1593. [Google Scholar] [CrossRef] [PubMed]
- Decaens, C.; Cassio, D. Spatiotemporal expression of catenins, ZO-1, and occludin during early polarization of hepatic WIF-B9 cells. Am. J. Physiol. Cell Physiol. 2001, 280, C527–C539. [Google Scholar] [PubMed]
- Godoy, P.; Hengstler, J.G.; Ilkavets, I.; Meyer, C.; Bachmann, A.; Mueller, A.; Tuschl, G.; Mueller, S.O.; Dooley, S. Doole Extracellular matrix modulates sensitivity of hepatocytes to fibroblastoid dedifferentiation and transforming growth factor induced apoptosis. Hepatology 2009, 49, 2031–2043. [Google Scholar] [CrossRef] [PubMed]
- Acosta, D.; Anuforo, D.; McMillin, R.; Soine, W.H.; Smith, R.V. Comparison of cytochrome P-450 levels in adult rat liver, postnatal rat liver, and primary cultures of postnatal rat hepatocytes. Life Sci. 1979, 1413–1418. [Google Scholar] [CrossRef]
- Enat, R.; Jefferson, D.M.; Ruiz-Opazo, N.; Gatmaitan, Z.; Leinwand, L.A.; Reid, L.M. Hepatocyte proliferation in vitro: Its dependence on the use of serum-free hormonally defined medium and substrata of extracellular matrix. Proc. Natl. Acad. Sci. USA 1984, 81, 1411–1415. [Google Scholar] [CrossRef] [PubMed]
- Godoy, P.; Hewitt, N.J.; Albrecht, U.; Andersen, M.E.; Ansari, N.; Bhattacharya, S.; Bode, J.G.; Bolleyn, J.; Borner, C.; Boettger, J.; et al. Recent advances in 2D and 3D in vitro systems using primary hepatocytes, alternative hepatocyte sources and non-parenchymal liver cells and their use in investigating mechanismsof hepatotoxicity, cell signaling and ADME. Arch. Toxicol. 2013, 87, 1315–1530. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Lechón, M.J.; Tolosa, L.; Conde, I.; Donato, M.T. Competency of different cell models to predict human hepatotoxic drugs. Expert Opin. Drug Metab. Toxicol. 2014, 10, 1553–1568. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Bisgin, H.; Tong, L.; Hong, H.; Fang, H.; Borlak, J.; Tong, W. Toward predictive models for drug-induced liver injury in humans: Are we there yet? Biomark. Med. 2014, 8, 201–213. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Lechon, M.J.; Lahoz, A.; Gombau, L.; Castell, J.V.; Donato, M.T. Evaluation of Potential Hepatotoxicity Induced by Drugs. Curr. Pharm. Des. 2010, 16, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Björnsson, E.S.; Bergmann, O.M.; Björnsson, H.K.; Kvaran, R.B.; Olafsson, S. Incidence, Presentation, and Outcomes in Patients with Drug-Induced Liver Injury in the General Population of Iceland. YGAST 2013, 144, 1419–1425. [Google Scholar]
- Lewis, D.F.; Ioannides, C.; Parke, D.V. Cytochromes P450 and species differences in xenobiotic metabolism and activation of carcinogen. Environ. Health Perspect. 1998, 106, 633–641. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, P.J.; Chan, K.; Silber, P.M. Human and animal hepatocytes in vitro with extrapolation in vivo. Chem. Biol. Interact. 2004, 150, 97–114. [Google Scholar] [CrossRef] [PubMed]
- Soldatow, V.Y.; Griffith, L.G.; Rusyn, I.; LeCluyse, E.L. In vitro models for liver toxicity testing. Toxicol. Res. 2013, 2, 23–39. [Google Scholar] [CrossRef]
- Pfeiffer, E.; Kegel, V.; Zeilinger, K.; Hengstler, J.G.; Nussler, A.K.; Seehofer, D.; Damm, G. Isolation, characterization, and cultivation of human hepatocytes and non-parenchymal liver cells. Exp. Biol. Med. 2014. [Google Scholar] [CrossRef]
- Guguen-Guillouzo, C.; Guillouzo, A. General Review on In Vitro Hepatocyte Models and Their Applications. In Hepatocytes; Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2010; Volume 640, pp. 1–40. [Google Scholar]
- Guzzardi, M.A.; Vozzi, F.; Ahluwalia, A.D. Study of the Crosstalk between Hepatocytes and Endothelial Cells Using a Novel Multicompartmental Bioreactor: A Comparison between Connected Cultures and Cocultures. Tissue Eng. Part A 2009, 15, 3635–3645. [Google Scholar] [CrossRef] [PubMed]
- Khetani, S.R.; Bhatia, S.N. Microscale culture of human liver cells for drug development. Nat. Biotechnol. 2008, 26, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Lau, T.T.; Priscilyn Lee, L.Q.; Leong, W.; Wang, D.A. Formation of model hepatocellular aggregates in a hydrogel scaffold using degradable genipin crosslinked gelatin microspheres as cell carriers. Biomed. Mater. 2012, 7, 1–8. [Google Scholar] [CrossRef]
- Dash, A.; Inman, W.; Hoffmaster, K.; Sevidal, S.; Kelly, J.; Obach, R.S.; Tannenbaum, S.R. Liver tissue engineering in the evaluation of drug safety. Expert Opin. Drug Metab. Toxicol. 2009, 5, 1159–1174. [Google Scholar] [CrossRef] [PubMed]
- Schmelzer, E.; Triolo, F.; Turner, M.; Thompson, R.L.; Zeillinger, K.; Reid, L.M.; Gridelli, B.; Gerlach, J.G. Three-Dimensional Perfusion Bioreactor Culture Supports Differentiation of Human Fetal Liver Cells. Tissue Eng. Part A 2010, 16, 2007–2016. [Google Scholar] [CrossRef] [PubMed]
- Mueller, D.; Tascher, G.; Mueller-Viera, U.; Nuessler, A.K.; Zeillinger, K.; Heinzle, E.; Noor, F. In-depth physiological characterization of primary human hepatocytes in a 3D hollow- fiber bioreactor. J. Tissue Eng. Regen. Med. 2011, 5, e207–e218. [Google Scholar] [CrossRef] [PubMed]
- Legendre, A.; Baudoin, R.; Alberto, G.; Paullier, P.; Naudot, M.; Bricks, T.; Brocheton, J.; Jaques, S.; Cotton, J.; Leclerc, E. Metabolic Characterization of Primary Rat Hepatocytes Cultivated in Parallel Microfluidic Biochips. Eur. J. Pharm. Sci. 2013, 102, 3264–3276. [Google Scholar] [CrossRef]
- Hegde, M.; Jindal, R.; Bhushan, A.; Bale, S.S.; McCarty, W.J.; Golberg, I.; Usta, O.B.; Yarmush, M.L. Dynamic interplay of flow and collagen stabilizes primary hepatocytes culture in a microfluidic platform. Lab Chip 2014, 14, 2033–2039. [Google Scholar] [CrossRef] [PubMed]
- Inamori, M.; Eng, M.; Mizumoto, H.; Kajiwara, T. An Approach for Formation of Vascularized Liver Tissue by Endothelial Cell–Covered Hepatocyte Spheroid Integration. Tissue Eng. Part A 2009, 15, 2029–2037. [Google Scholar] [CrossRef] [PubMed]
- No, D.Y.; Lee, S.A.; Choi, Y.Y.; Park, D.Y.; Jang, J.Y.; Kim, D.S.; Lee, H.L. Functional 3D Human Primary Hepatocyte Spheroids Made by Co-Culturing Hepatocytes from Partial Hepatectomy Specimens and Human Adipose-Derived Stem Cells. PLoS One 2012, 7, 1–8. [Google Scholar] [CrossRef]
- Knobeloch, D.; Ehnert, S.; Schyschka, L.; Büchler, P.; Schoenberg, M.; Kleeff, J.; Thasler, W.E.; Nussler, N.C.; Godoy, P.; Hengstler, J.; et al. Human Hepatocytes: Isolation, Culture, and Quality Procedures. In Hepatocytes; Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2011; Volume 806, pp. 99–120. [Google Scholar]
- Hewitt, N.J.; Lechón, M.J.G.; Houston, J.B.; Hallifax, D.; Brown, H.S.; Maurel, P.; Kenna, J.G.; Gustavsson, L.; Lohmann, C.; Skonberg, C.; et al. Primary hepatocytes: Current understanding of the regulation of metabolic enzymes and transporter proteins, and pharmaceutical practice for the use of hepatocytes in metabolism, enzyme induction, transporter, clearance, and hepatotoxicity studies. Drug Metab. Rev. 2007, 39, 159–234. [Google Scholar] [CrossRef] [PubMed]
- Ehnert, S.; Lukoschek, T.; Bachmann, A.; Martínez Sánchez, J.J.; Damm, G.; Nussler, N.C.; Pscherer, S.; Stöckle, U.; Dooley, S.; Mueller, S.; et al. The right choice of antihypertensives protects primary human hepatocytes from ethanol- and recombinant human TGF-β1-induced cellular damage. Hepat. Med. 2013, 5, 31–41. [Google Scholar] [PubMed]
- Buxboim, A.; Ivanovska, I.L.; Discher, D.E. Matrix elasticity, cytoskeletal forces and physics of the nucleus: How deeply do cells “feel” outside and in? J. Cell. Sci. 2010, 123, 297–308. [Google Scholar] [CrossRef] [PubMed]
- Asthana, A.; Kisaalita, W.S. Biophysical microenvironment and 3D culture physiological relevance. Drug Discov. Today 2013, 18, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.; Cho, N.C.; Xiong, A.; Glenn, J.S.; Frank, C.W. Hydrophobic nanoparticles improve permeability of cell-encapsulating poly(ethylene glycol) hydrogelswhile maintaining patternability. Proc. Natl. Acad. Sci. USA 2010, 107, 120709–120714. [Google Scholar]
- Burkhardt, B.; Martinez-Sanchez, J.J.; Bachmann, A.; Ladurner, R.; Nussler, A.K. Long-term culture of primary hepatocytes: New matrices and microfluidic devices. Hepatol. Int. 2013, 8, 14–22. [Google Scholar] [CrossRef]
- Berthiaume, F.; Moghe, P.V.; Toner, M.; Yarmush, M.L. Effect of extracellular matrix topology on cell structure, function, and physiological responsiveness: Hepatocytes cultured in a sandwich configuration. FASEB 2004, 10, 1471–1484. [Google Scholar]
- Messner, S.; Agarkova, L.; Moritz, W.; Kelm, J.M. Multi-cell type human liver microtissues for hepatotoxicity testing. Arch. Toxicol. 2012, 87, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Burkard, A.; Dähn, C.; Heinz, S.; Zutavern, A.; Sonntag-Buck, V.; Maltman, D. Generation of proliferating human hepatocytes using upcyte® technology: Characterisation and applications in induction and cytotoxicity assays. Xenobiotica 2012, 42, 939–956. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, G.A.; Jolley, S.L.; Gilbert, D.; Coon, J.D.; Barros, S.; LeCluyse, E.L. Regulation of cell morphology and cytochrome P450 expressionin human hepatocytes by extracellular matrix and cell-cell interactions. Cell Tissue Res. 2012, 306, 85–99. [Google Scholar]
- Gottwald, E.; Giselbrecht, S.; Augspurger, C.; Lahni, B.; Dambrowski, N.; Truckenmueller, R.; Poitter, V.; Giezelt, T.; Wendt, O.; Pfleging, W.; et al. A chip-based platform for the in vitro generation of tissues in three-dimensional organization. Lab Chip 2007, 7, 777–785. [Google Scholar] [CrossRef] [PubMed]
- Altmann, B.; Giselbrecht, S.; Weibezahn, K.F.; Welle, A.; Gottwald, E. The three-dimensional cultivation of the carcinoma cell line HepG2 in a perfused chip system leads to a more differentiated phenotype of the cells compared to monolayer culture. Biomed. Mater. 2008, 3, 034120–034110. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Marchan, R.E. Design properties of hydrogel tissue-engineering scaffolds. Expert Rev. Med. Devices 2011, 8, 607–626. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Lee, J.Y.; Johnes, C.N.; Revzinc, A.; Tae, G. Heparin-Based Hydrogel as a Matrix for Encapsulation and Cultivation of Primary Hepatocytes. Biomaterials 2010, 31, 3596–3603. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.C.; Anseth, K.S. PEG Hydrogels for the Controlled Release of Biomolecules in Regenerative Medicine. Pharm. Res. 2009, 26, 631–643. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Nagrath, D.; Chen, P.C.; Berthiaume, F.; Yarmush, M.L. Three-Dimensional Primary Hepatocyte Culture in Synthetic Self-Assembling Peptide Hydrogel. Tissue Eng. Part A 2008, 14, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Genove, E.; Schmitmeier, S.; Sala, A.; Borros, S.; Bader, A.; Griffith, L.G.; Semino, C.E. Functionalized self-assembling peptide hydrogel enhancemaintenance of hepatocyte activity in vitro. J. Cell Mol. Med. 2009, 13, 3387–3397. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Pan, J.; Zhang, L.; Guo, X.; Yu, Y. Culture of primary rat hepatocytes within porous chitosan scaffolds. J. Biomed. Mater. Res. 2003, 67, 938–943. [Google Scholar] [CrossRef]
- Seo, S.J.; Choi, Y.J.; Akaike, T.; Higuchi, A.; Cho, C.S. Alginate/Galactosylated Chitosan/Heparin Scaffold As a New Synthetic Extracellular Matrix for Hepatocytes. Tissue Eng. 2006, 12, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Shang, Y.; Yuan, Y. Preparation and characterization of chitosan/galactosylated hyaluronic acid scaffolds for primary hepatocytes culture. J. Mater. Sci. 2009, 21, 319–327. [Google Scholar]
- Dvir-Ginzberg, M.; Gamlieli-Bonshtein, I.; Agbaria, R.; Cohen, S. Liver Tissue Engineering within Alginate Scaffolds: Effects of Cell-Seeding Density on Hepatocyte Viability, Morphology, and Function. Tissue Eng. 2004, 9, 757–766. [Google Scholar] [CrossRef]
- Saavedra, Y.G.L.; Mateescu, M.A.; Averill-Bates, D.A.; Denizeau, F. Polyvinylalcohol three-dimensional matrices for improved long-term dynamic culture of hepatocytes. J. Biomed. Mater. Res. A 2003, 66, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Feng, Z.Q.; Leach, M.K.; Wu, J.; Jiang, Q. Nanoporous fibers of type-I collagen coated poly(L-lactic acid) for enhancing primary hepatocyte growth and function. J. Mater.Chem. B 2012, 1, 339–346. [Google Scholar] [CrossRef]
- Schutte, M.; Fox, B.; Baradez, M.O.; Devonshire, A.; Minguez, J.; Bokhari, M.; Przyborski, S.; Marshall, D. Rat primary hepatocytes show enhanced performance and sensitivity to acetaminophen during three-dimensional culture on a polystyrene scaffold designed for routine use. Assay Drug Dev. Technol. 2011, 9, 475–486. [Google Scholar] [CrossRef] [PubMed]
- Ohkura, T.; Ohta, K.; Nagao, T.; Kusumoto, K.; Koeda, A.; Ueda, T.; Jomura, T.; Ikeya, T.; Ozeki, E.; Kazuki, W.; et al. Evaluation of Human Hepatocytes Cultured by Three-dimensional Spheroid Systems for Drug Metabolism. Drug Metab. Pharmacokinet. 2014, 29, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Kato, N.; Aizawa, K.; Mizutani, R.; Yamauchi, J.; Tanoue, A. Expression of albumin and cytochrome P450 enzymes in HepG2 cells cultured with a nanotechnology-based culture plate with microfabricated scaffold. J. Toxicol. Sci. 2011, 36, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Moghe, P.V.; Ciger, R.N.; Toner, M.; Yarmush, M.L. Cell–Cell Interactions Are Essential for Maintenance of Hepatocyte Function in Collagen Gel But Not on Matrigel. Biotechnol. Bioeng. 1997, 56, 706–711. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; WAng, Y.; Xu, D.; Tang, Y.; Feng, M. Chitosan/gelatin composite microcarrier for hepatocyte culture. Biotechnol. Lett. 2004, 26, 879–883. [Google Scholar] [CrossRef] [PubMed]
- Mikos, A.G.; Sarakinos, G.; Leite, S.M.; Vacanti, J.P.; Langer, R. Laminated three-dimensional biodegradable foams for use in tissueengineering. Biomaterials 2002, 14, 323–330. [Google Scholar] [CrossRef]
- reliver. Available online: http://www.reliver.eu/news and dissemination/ (accessed on 11 November 2014).
- reliver. Available online: http://www.reliver.eu (accessed on 11 November 2014).
- Kermanizadeh, A.; Løhr, M.; Roursgaard, M.; Messner, S.; Gunness, P.; Kelm, J.M.; Møller, P.; Stone, V.; Loft, S. Hepatic toxicology following single and multiple exposure of engineered nanomaterials utilising a novel primary human 3D liver microtissue model. Part. Fibre Toxicol. 2014, 11, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Tilles, A.W.; Baskaran, H.; Roy, P.; Yarmus, M.L.; Toner, M. Effects of oxygenation and flow on the viability and function of rat hepatocytes cocultured in a microchannel flat-plate bioreactor. Biotechnol. Bioeng. 2015, 73, 379–389. [Google Scholar] [CrossRef]
- Powers, M.J.; Janigian, D.M.; Wack, K.E.; Baker, C.S.; Beer, S.; Griffith, L.G. Functional Behavior of Primary Rat Liver Cells in a Three-Dimensional Perfused Microarray Bioreactor. Tissue Eng. 2002, 8, 499–513. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y.; Yamato, M.; Okano, T.; Kitamori, T.; Sato, K. Evaluation of effects of shear stress on hepatocytes by a microchip-based system. Meas. Sci. Technol. 2006, 17, 3167–3170. [Google Scholar] [CrossRef]
- Nussler, A.K.; Wang, A.; Neuhaus, P.; Fischer, J.; Yuan, J.; Liu, L.; Zeilinger, K.; Gerlach, J.; Arnold, P.J.; Albrecht, W. The suitability of hepatocyte culture models to study various aspects of drug metabolism. ALTEX 2001, 18, 91–101. [Google Scholar] [PubMed]
- Hoffmann, S.A.; Muller-Viera, U.; Biemel, K.; Knobeloch, D.; Heydel, S.; Luebberstedt, M.; Nuessler, A.K.; Andersson, T.B.; Gerlach, J.C.; Katrin, Z. Analysis of drug metabolism activities in a miniaturized liver cell bioreactor for use in pharmacological studies. Biotechnol. Bioeng. 2012, 109, 3172–3181. [Google Scholar] [CrossRef] [PubMed]
- Müller-Vieira, U.; Biemel, K.M.; Darnell, M.; Hoffmann, S.A.; Knöspel, F.; Wönne, E.C.; Knobeloch, D.; Nüssler, A.K.; Gerlach, J.C.; Andersson, T.B.; et al. Serum-free culture of primary human hepatocytes in a miniaturized hollow-fibre membrane bioreactor for pharmacological in vitro studies. J. Tissue Eng. Regen. Med. 2012. [Google Scholar] [CrossRef]
- Mazzei, D.; Guazzardi, S. A low shear stress modular bioreactor for connected cell culture under high flow rates. Bioctechnol. Bioeng. 2010, 106, 127–137. [Google Scholar]
- Unger, K.J.; Kuehlein, G.; Schroers, A.; Gerlach, J.G.; Rossaint, R. Adsorption of xenobiotics to plastic tubing incorporated into dynamic in vitro systems used in pharmacological research * limits and progress. Biomaterials 2001, 22, 2031–2037. [Google Scholar] [CrossRef] [PubMed]
- Miranda, J.P.; Rodrigues, A.; Tostoes, R.M.; Leite, S.; Zimmerman, H.; Carrondo, M.J.T.; Alves, P.M. Extending Hepatocyte Functionality for Drug-Testing Applications Using High-Viscosity Alginate–Encapsulated Three-Dimensional Cultures in Bioreactors. Tissue Eng. Part C 2010, 16, 1223–1232. [Google Scholar] [CrossRef]
- Vinci, B.; Duret, C.; Klieber, S.; Gerbal-Chaloin, S.; Sa-Cunha, A.; Laporte, Z.; Suc, B.; Maurel, P.; Ahluwalia, A.; Daujat-Chavanieu, M. Modular bioreactor for primary human hepatocyte culture: Medium flow stimulates expression and activity of detoxification genes. Biotechnol. J. 2011, 6, 554–564. [Google Scholar] [CrossRef] [PubMed]
- Sbrana, T.; Ahuluwalia, A. Engineering Quasi-Vivo® in Vitro Organ Models. In Advances in Experimental Medicine and Biology; Advances in Experimental Medicine and Biology; Springer US: New York, NY, USA, 2012; Volume 745, pp. 138–153. [Google Scholar]
- Sivaraman, A.; Leach, J.K.; Townsend, S.; Iida, T.; Hogan, B.J.; Stolz, D.B.; Fry, R.; Samson, L.D.; Tannenbaum, S.R.; Griffith, L.G. A microscale in vitro physiological model of the liver: Predictive screens for drug metabolism and enzyme induction. Curr. Drug Metab. 2005, 6, 569–591. [Google Scholar] [CrossRef] [PubMed]
- Vivares, A.; Salle-Lefort, S.; Arabeyre-Fabre, C.; Ngo, R.; Penarier, G.; Bremond, M.; Moliner, P.; Gallas, J.F.; Fabre, G.; Klieber, S. Morphological behaviour and metabolic capacity of cryopreserved human primary hepatocytes cultivated in a perfused multiwell device. Xenobiotica 2015, 45, 29–44. [Google Scholar] [CrossRef] [PubMed]
- Schuette, J.; Hagmeyer, B.; Holzner, F.; Kubon, M.; Werner, S.; Freudigmann, C.; Benz, K.; Boettger, J.; Gebhardt, R.; Becker, H.; et al. “Artificial micro organs”—A microfluidic device for dielectrophoretic assembly of liver sinusoids. Biomed. Microdevices 2011, 13, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Insphero. Available online: http://www.insphero.com/company/eurostars-project-hts-dili/ (accessed on 11 November 2014).
- Merck Millipore. Available online: https://www.emdmillipore.com/US/en/20140312_12470?bd=1 (accesses online 11 November 2014).
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bachmann, A.; Moll, M.; Gottwald, E.; Nies, C.; Zantl, R.; Wagner, H.; Burkhardt, B.; Sánchez, J.J.M.; Ladurner, R.; Thasler, W.; et al. 3D Cultivation Techniques for Primary Human Hepatocytes. Microarrays 2015, 4, 64-83. https://doi.org/10.3390/microarrays4010064
Bachmann A, Moll M, Gottwald E, Nies C, Zantl R, Wagner H, Burkhardt B, Sánchez JJM, Ladurner R, Thasler W, et al. 3D Cultivation Techniques for Primary Human Hepatocytes. Microarrays. 2015; 4(1):64-83. https://doi.org/10.3390/microarrays4010064
Chicago/Turabian StyleBachmann, Anastasia, Matthias Moll, Eric Gottwald, Cordula Nies, Roman Zantl, Helga Wagner, Britta Burkhardt, Juan J. Martínez Sánchez, Ruth Ladurner, Wolfgang Thasler, and et al. 2015. "3D Cultivation Techniques for Primary Human Hepatocytes" Microarrays 4, no. 1: 64-83. https://doi.org/10.3390/microarrays4010064
APA StyleBachmann, A., Moll, M., Gottwald, E., Nies, C., Zantl, R., Wagner, H., Burkhardt, B., Sánchez, J. J. M., Ladurner, R., Thasler, W., Damm, G., & Nussler, A. K. (2015). 3D Cultivation Techniques for Primary Human Hepatocytes. Microarrays, 4(1), 64-83. https://doi.org/10.3390/microarrays4010064








