Adjunct Diagnostic Value of Transcranial Magnetic Stimulation in Mucopolysaccharidosis-Related Cervical Myelopathy: A Pilot Study
Abstract
1. Introduction
1.1. Mucopolysaccharidosis: A Brief Overview
1.2. Cervical Myelopathy in MPS
2. Materials and Methods
2.1. Subjects and Assessment
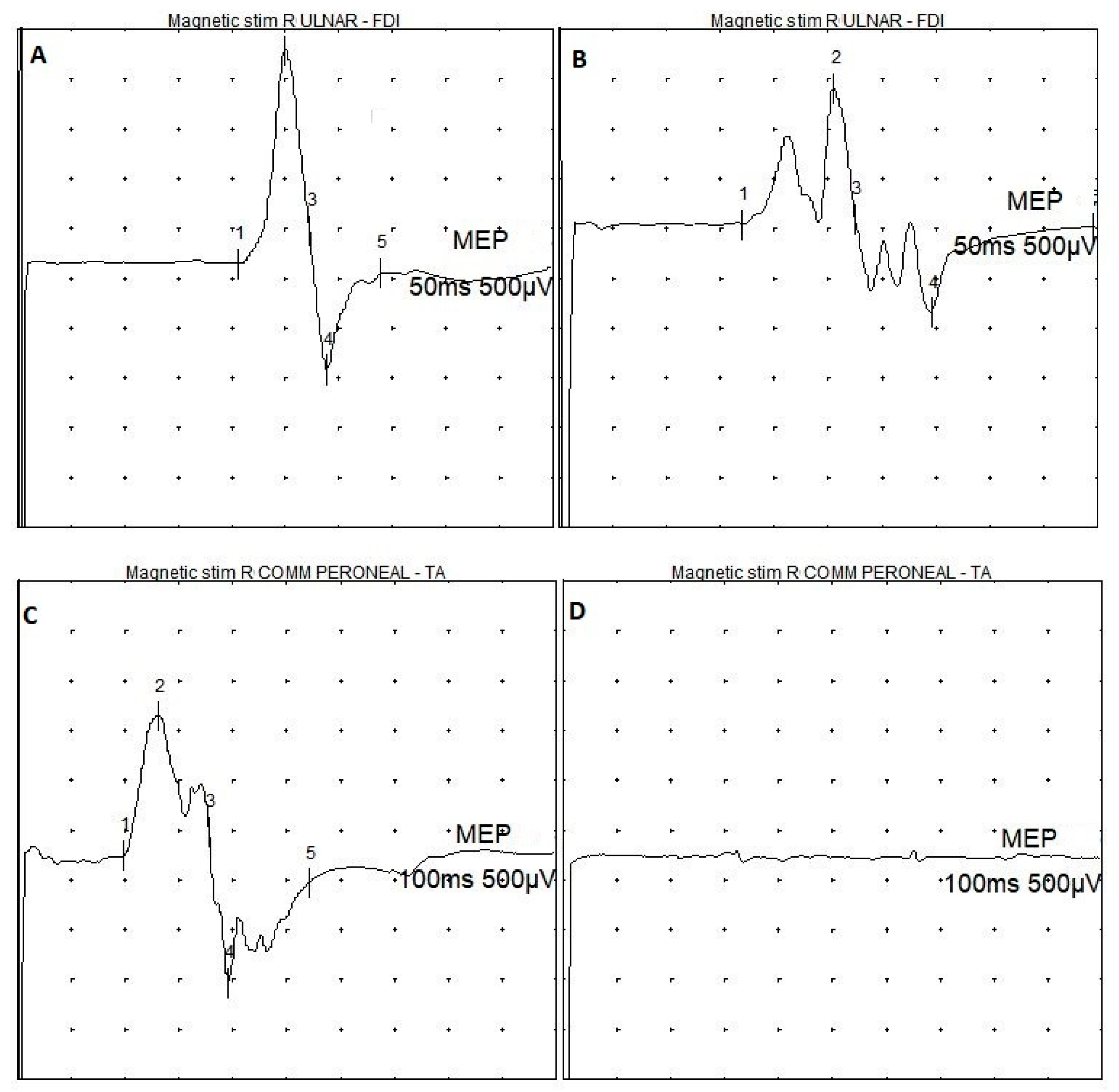
2.2. Transcranial Magnetic Stimulation
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Stapleton, M.; Arunkumar, N.; Kubaski, F.; Mason, R.W.; Tadao, O.; Tomatsu, S. Clinical presentation and diagnosis of mucopolysaccharidoses. Mol. Genet. Metab. 2018, 125, 4–17. [Google Scholar] [CrossRef] [PubMed]
- Barone, R.; Pellico, A.; Pittalà, A.; Gasperini, S. Neurobehavioral phenotypes of neuronopathic mucopolysaccharidoses. Ital. J. Pediatr. 2018, 44, 121. [Google Scholar] [CrossRef] [PubMed]
- Palmucci, S.; Attinà, G.; Lanza, M.L.; Belfiore, G.; Cappello, G.; Foti, P.V.; Milone, P.; Di Bella, D.; Barone, R.; Fiumara, A.; et al. Imaging findings of mucopolysaccharidoses: A pictorial review. Insights Imaging 2013, 4, 443–459. [Google Scholar] [CrossRef] [PubMed]
- Sun, A. Lysosomal storage disease overview. Ann. Transl. Med. 2018, 6, 476. [Google Scholar] [CrossRef] [PubMed]
- Valstar, M.J.; Neijs, S.; Bruggenwirth, H.T.; Olmer, R.; Ruijter, G.J.; Wevers, R.A.; van Diggelen, O.P.; Poorthuis, B.J.; Halley, D.J.; Wijburg, F.A. Mucopolysaccharidosis type IIIA: Clinical spectrum and genotype-phenotype correlations. Ann. Neurol. 2010, 68, 876–887. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, E.G.; Nestrasil, I.; Delaney, K.A.; Rudser, K.; Kovac, V.; Nair, N.; Richard, C.W., 3rd; Haslett, P.; Whitley, C.B. A Prospective Natural History Study of Mucopolysaccharidosis Type IIIA. J. Pediatr. 2016, 170, e1–e4. [Google Scholar] [CrossRef] [PubMed]
- Hendriksz, C.J.; Harmatz, P.; Beck, M.; Jones, S.; Wood, T.; Lachman, R.; Gravance, C.G.; Orii, T.; Tomatsu, S. Review of clinical presentation and diagnosis of mucopolysaccharidosis IVA. Mol. Genet. Metab. 2013, 110, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Montaño, A.M.; Lock-Hock, N.; Steiner, R.D.; Graham, B.H.; Szlago, M.; Greenstein, R.; Pineda, M.; Gonzalez-Meneses, A.; Çoker, M.; Bartholomew, D.; et al. Clinical course of sly syndrome (mucopolysaccharidosis type VII). J. Med. Genet. 2016, 53, 403–418. [Google Scholar] [CrossRef] [PubMed]
- Zielonka, M.; Garbade, S.F.; Kölker, S.; Hoffmann, G.F.; Ries, M. Quantitative clinical characteristics of 53 patients with MPS VII: A cross-sectional analysis. Genet. Med. 2018, 20, 474. [Google Scholar] [CrossRef]
- Galimberti, C.; Madeo, A.; Di Rocco, M.; Fiumara, A. Mucopolysaccharidoses: Early diagnostic signs in infants and children. Ital. J. Pediatr. 2018, 44, 133. [Google Scholar] [CrossRef]
- Giussani, C.; Guida, L.; Canonico, F.; Sganzerla, E.P. Cerebral and occipito-atlanto-axial involvement in mucopolysaccharidosis patients: Clinical, radiological, and neurosurgical features. Ital. J. Pediatr. 2018, 44, 119. [Google Scholar] [CrossRef] [PubMed]
- Illsinger, S.; Lücke, T.; Hartmann, H.; Mengel, E.; Müller-Forell, W.; Donnerstag, F.; Das, A.M. Scheie syndrome: Enzyme replacement therapy does not prevent progression of cervical myelopathy due to spinal cord compression. J. Inherit. Metab. Dis. 2009, 32, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Solanki, G.A.; Martin, K.W.; Theroux, M.C.; Lampe, C.; White, K.K.; Shediac, R.; Lampe, C.G.; Beck, M.; Mackenzie, W.G.; Hendriksz, C.J.; et al. Spinal involvement in mucopolysaccharidosis IVA (Morquio-Brailsford or Morquio A syndrome): Presentation, diagnosis and management. J. Inherit. Metab. Dis. 2013, 36, 339–355. [Google Scholar] [CrossRef] [PubMed]
- Rossini, P.M.; Burke, D.; Chen, R.; Cohen, L.G.; Daskalakis, Z.; Di Iorio, R.; Di Lazzaro, V.; Ferreri, F.; Fitzgerald, P.B.; George, M.S.; et al. Non-invasive electrical and magnetic stimulation of the brain, spinal cord, roots and peripheral nerves: Basic principles and procedures for routine clinical and research application. An updated report from an I.F.C.N. Committee. Clin. Neurophysiol. 2015, 126, 1071–1107. [Google Scholar] [CrossRef] [PubMed]
- Travlos, A.; Pant, B.; Eisen, A. Transcranial magnetic stimulation for detection of preclinical cervical spondylotic myelopathy. Arch. Phys. Med. Rehabil. 1992, 73, 442–446. [Google Scholar] [PubMed]
- Bella, R.; Ferri, R.; Cantone, M.; Pennisi, M.; Lanza, G.; Malaguarnera, G.; Spampinato, C.; Giordano, D.; Raggi, A.; Pennisi, G. Motor cortex excitability in vascular depression. Int. J. Psychophysiol. 2011, 82, 248–253. [Google Scholar] [CrossRef] [PubMed]
- Lanza, G.; Bella, R.; Giuffrida, S.; Cantone, M.; Pennisi, G.; Spampinato, C.; Giordano, D.; Malaguarnera, G.; Raggi, A.; Pennisi, M. Preserved transcallosal inhibition to transcranial magnetic stimulation in nondemented elderly patients with leukoaraiosis. Biomed. Res. Int. 2013, 2013, 351680. [Google Scholar] [CrossRef] [PubMed]
- Bella, R.; Ferri, R.; Lanza, G.; Cantone, M.; Pennisi, M.; Puglisi, V.; Vinciguerra, L.; Spampinato, C.; Mazza, T.; Malaguarnera, G.; et al. TMS follow-up study in patients with vascular cognitive impairment-no dementia. Neurosci. Lett. 2013, 534, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Pennisi, G.; Bella, R.; Lanza, G. Motor cortex plasticity in subcortical ischemic vascular dementia: What can TMS say? Clin. Neurophysiol. 2015, 126, 851–852. [Google Scholar] [CrossRef]
- Bella, R.; Cantone, M.; Lanza, G.; Ferri, R.; Vinciguerra, L.; Puglisi, V.; Pennisi, M.; Ricceri, R.; Di Lazzaro, V.; Pennisi, G. Cholinergic circuitry functioning in patients with vascular cognitive impairment--no dementia. Brain Stimul. 2016, 9, 225–233. [Google Scholar] [CrossRef]
- Lanza, G.; Bramanti, P.; Cantone, M.; Pennisi, M.; Pennisi, G.; Bella, R. Vascular Cognitive Impairment through the Looking Glass of Transcranial Magnetic Stimulation. Behav. Neurol. 2017, 2017, 1421326. [Google Scholar] [CrossRef] [PubMed]
- Cantone, M.; Bramanti, A.; Lanza, G.; Pennisi, M.; Bramanti, P.; Pennisi, G.; Bella, R. Cortical Plasticity in Depression. ASN Neuro 2017, 9, 1759091417711512. [Google Scholar] [CrossRef] [PubMed]
- Boor, R.; Fricke, G.; Brühl, K.; Spranger, J. Abnormal subcortical somatosensory evoked potentials indicate high cervical myelopathy in achondroplasia. Eur. J. Pediatr. 1999, 158, 662–667. [Google Scholar] [CrossRef] [PubMed]
- Boor, R.; Miebach, E.; Brühl, K.; Beck, M. Abnormal somatosensory evoked potentials indicate compressive cervical myelopathy in mucopolysaccharidoses. Neuropediatrics 2000, 31, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Müller-Forell, W.; Oberman, B.; Boor, R. Subcortical somatosensory evoked potentials after median nerve and posterior tibial nerve stimulation in high cervical cord compression of achondroplasia. Brain Dev. 2008, 30, 499–503. [Google Scholar] [CrossRef] [PubMed]
- Mut, M.; Cila, A.; Varli, K.; Akalan, N. Multilevel myelopathy in Maroteaux-Lamy syndrome and review of the literature. Clin. Neurol. Neurosurg. 2005, 107, 230–235. [Google Scholar] [CrossRef]
- Kandil, A.I.; Pettit, C.S.; Berry, L.N.; Busso, V.O.; Careskey, M.; Chesnut, E.; Buck, D.W.; Leslie, N.D.; Tamai, J.; McAuliffe, J.J.; et al. Tertiary Pediatric Academic Institution’s Experience With Intraoperative Neuromonitoring for Nonspinal Surgery in Children With Mucopolysaccharidosis, Based on a Novel Evidence-Based Care Algorithm. Anesth. Analg. 2019. [Google Scholar] [CrossRef]
- Wraith, J.E. Mucopolysaccharidoses and mucolipidoses. Handb. Clin. Neurol. 2013, 113, 1723–1729. [Google Scholar] [CrossRef]
- Amassian, V.E.; Cracco, R.Q.; Maccabee, P.J. Focal stimulation of human cerebral cortex with the magnetic coil: A comparison with electrical stimulation. Electroencephalogr. Clin. Neurophysiol. 1989, 74, 401–416. [Google Scholar] [CrossRef]
- Alexeeva, N.; Broton, J.G.; Calancie, B. Latency of changes in spinal motoneuron excitability evoked by transcranial magnetic brain stimulation in spinal cord injured individuals. Electroencephalogr. Clin. Neurophysiol. 1998, 109, 297–303. [Google Scholar] [CrossRef]
- Garry, M.I.; Kamen, G.; Nordstrom, M.A. Hemispheric differences in the relationship between corticomotor excitability changes following a fine-motor task and motor learning. J. Neurophysiol. 2004, 91, 1570–1578. [Google Scholar] [CrossRef]
- Cantone, M.; Lanza, G.; Vinciguerra, L.; Puglisi, V.; Ricceri, R.; Fisicaro, F.; Vagli, C.; Bella, R.; Ferri, R.; Pennisi, G.; et al. Age, Height, and Sex on Motor Evoked Potentials: Translational Data From a Large Italian Cohort in a Clinical Environment. Front. Hum. Neurosci. 2019, 13, 185. [Google Scholar] [CrossRef]
- Charrow, J.; Alden, T.D.; Breathnach, C.A.; Frawley, G.P.; Hendriksz, C.J.; Link, B.; Mackenzie, W.G.; Manara, R.; Offiah, A.C.; Solano, M.L.; et al. Diagnostic evaluation, monitoring, and perioperative management of spinal cord compression in patients with Morquio syndrome. Mol. Genet. Metab. 2015, 114, 11–18. [Google Scholar] [CrossRef]
- Solanki, G.A.; Sun, P.P.; Martin, K.W.; Hendriksz, C.J.; Lampe, C.; Guffon, N.; Hung, A.; Sisic, Z.; Shediac, R.; Harmatz, P.R. CSP Study Group.Cervical cord compression in mucopolysaccharidosis VI (MPS VI): Findings from the MPS VI Clinical Surveillance Program (CSP). Mol. Genet. Metab. 2016, 118, 310–318. [Google Scholar] [CrossRef]
- Ransford, A.O.; Crockard, H.A.; Stevens, J.M.; Modaghegh, S. Occipito-atlanto-axial fusion in Morquio-Brailsford syndrome. A ten-year experience. J. Bone Joint Surg. Br. 1996, 78, 307–313. [Google Scholar] [CrossRef]
- Baldo, G.; Mayer, F.Q.; Martinelli, B.; Dilda, A.; Meyer, F.; Ponder, K.P.; Giugliani, R.; Matte, U. Evidence of a progressive motor dysfunction in Mucopolysaccharidosis type I mice. Behav. Brain Res. 2012, 233, 169–175. [Google Scholar] [CrossRef]
- Pennisi, G.; Lanza, G.; Giuffrida, S.; Vinciguerra, L.; Puglisi, V.; Cantone, M.; Pennisi, M.; D’Agate, C.C.; Naso, P.; Aprile, G.; et al. Excitability of the motor cortex in de novo patients with celiac disease. PLoS ONE 2014, 9, e102790. [Google Scholar] [CrossRef]
- Bella, R.; Lanza, G.; Cantone, M.; Giuffrida, S.; Puglisi, V.; Vinciguerra, L.; Pennisi, M.; Ricceri, R.; D’Agate, C.C.; Malaguarnera, G.; et al. Effect of a Gluten-Free Diet on Cortical Excitability in Adults with Celiac Disease. PLoS ONE 2015, 10, e0129218. [Google Scholar] [CrossRef]
- Pennisi, M.; Lanza, G.; Cantone, M.; Ricceri, R.; Ferri, R.; D’Agate, C.C.; Pennisi, G.; Di Lazzaro, V.; Bella, R. Cortical involvement in celiac disease before and after long-term gluten-free diet: A Transcranial Magnetic Stimulation study. PLoS ONE 2017, 12, e0177560. [Google Scholar] [CrossRef]
- Sasso, V.; Bisicchia, E.; Latini, L.; Ghiglieri, V.; Cacace, F.; Carola, V.; Molinari, M.; Viscomi, M.T. Repetitive transcranial magnetic stimulation reduces remote apoptotic cell death and inflammation after focal brain injury. J. Neuroinflamm. 2016, 13, 150. [Google Scholar] [CrossRef]
- Bordet, R.; Ihl, R.; Korczyn, A.D.; Lanza, G.; Jansa, J.; Hoerr, R.; Guekht, A. Towards the concept of disease-modifier in post-stroke or vascular cognitive impairment: A consensus report. BMC Med. 2017, 15, 107. [Google Scholar] [CrossRef]
- Lanza, G.; Cantone, M.; Aricò, D.; Lanuzza, B.; Cosentino, F.I.I.; Paci, D.; Papotto, M.; Pennisi, M.; Bella, R.; Pennisi, G.; et al. Clinical and electrophysiological impact of repetitive low-frequency transcranial magnetic stimulation on the sensory-motor network in patients with restless legs syndrome. Ther. Adv. Neurol. Disord. 2018, 11, 1756286418759973. [Google Scholar] [CrossRef]
- Lanza, G.; Lanuzza, B.; Aricò, D.; Cantone, M.; Cosentino, F.I.I.; Bella, R.; Pennisi, G.; Ferri, R.; Pennisi, M. Impaired short-term plasticity in restless legs syndrome: A pilot rTMS study. Sleep Med. 2018, 46, 1–4. [Google Scholar] [CrossRef]
- Medina-Fernández, F.J.; Escribano, B.M.; Padilla-Del-Campo, C.; Drucker-Colín, R.; Pascual-Leone, A.; Túnez, I. Transcranial magnetic stimulation as an antioxidant. Free Radic. Res. 2018, 52, 381–389. [Google Scholar] [CrossRef]
| Patient | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
|---|---|---|---|---|---|---|---|---|
| MPS type | IVA | IVA | IVA | IVA | IVA | IVA | VI | VI |
| Sex/age (years) | F/14 | M/15 | F/16 | M/13 | F/20 | F/40 | F/13 | F/14 |
| ERT (age, years) | - | - | - | - | - | - | + (9) | + (10) |
| Height (cm) | 98 | 100 | 102 | 110 | 150 | 113 | 120 | 110 |
| Spinal cord surgery (age, years) | + (5) | + (4) | + (8) | + (10) | - | - | + (10) | + (11) |
| Diffuse brisk tendon reflex | + | - | + | + | - | - | + | + |
| Limbs paresis/weakness | + | - | + | - | - | - | + | + |
| Walking assistance | + | - | + | - | - | - | - | + |
| MRI cervical cord compression | + | + | + | + | - | - | + | + |
| MRI cervical myelopathy | + | - | + | - | - | - | - | + |
| N | First Dorsal Interosseous Muscle | Tibialis Anterior Muscle | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MEPs Amp (mV) | ID | Poly-Phasic Shape | MEPs Latency (ms) | ID | CMCT (ms) | ID | MEPs Amp (mV) | ID | MEPs Latency (ms) | ID | CMCT (ms) | ID | ||||||||
| R | L | R | L | R | L | R | L | R | L | R | L | |||||||||
| >2.8 | >2.8 | <4.0 | - | - | <22.5 | <22.5 | <1.5 | <7.6 | <7.6 | <1.5 | >1.9 | >1.9 | <4.0 | <31.2 | <31.2 | <4.1 | <17.2 | <17.2 | <3.0 | |
| 1 | 0.2 | 0.1 | 0.1 | + | + | 19.2 | 20.0 | 0.8 | nr | 10.1 | / | 0.3 | nr | / | 29.3 | nr | / | 21.2 | nr | / |
| 2 | 2.6 | 2.2 | 0.4 | - | + | 16.5 | 17.1 | 0.6 | 8.1 | 8.1 | 0.0 | 3.2 | 1.7 | 1.5 | 19.6 | 21.5 | 1.9 | 12.3 | 14.0 | 1.7 |
| 3 | 0.3 | 0.2 | 0.1 | + | + | 19.4 | 20.6 | 1.2 | nr | nr | / | nr | nr | / | nr | nr | / | nr | nr | / |
| 4 | 0.8 | 0.6 | 0.2 | + | + | 17.3 | 17.2 | 0.1 | 9.6 | 9.4 | 0.2 | 2.9 | 2.5 | 0.4 | 18.5 | 19.8 | 1.3 | 12.5 | 12.6 | 0.1 |
| 5 | 8.0 | 7.0 | 1.0 | - | - | 17.4 | 16.2 | 1.2 | 5.4 | 6.0 | 0.6 | 7.0 | 6.0 | 1.0 | 20.0 | 19.5 | 0.5 | 11.6 | 11.6 | 0.0 |
| 6 | 3.8 | 3.5 | 0.3 | - | - | 18.3 | 18.5 | 0.2 | 6.1 | 6.4 | 0.3 | 3.0 | 4.0 | 1.0 | 24.9 | 25.2 | 0.3 | 14.7 | 14.3 | 0.4 |
| 7 | nr | nr | / | / | / | nr | nr | / | nr | nr | / | nr | nr | / | nr | nr | / | nr | nr | / |
| 8 | nr | nr | / | / | / | nr | nr | / | nr | nr | / | nr | nr | / | nr | nr | / | nr | nr | / |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cantone, M.; Lanza, G.; Le Pira, A.; Barone, R.; Pennisi, G.; Bella, R.; Pennisi, M.; Fiumara, A. Adjunct Diagnostic Value of Transcranial Magnetic Stimulation in Mucopolysaccharidosis-Related Cervical Myelopathy: A Pilot Study. Brain Sci. 2019, 9, 200. https://doi.org/10.3390/brainsci9080200
Cantone M, Lanza G, Le Pira A, Barone R, Pennisi G, Bella R, Pennisi M, Fiumara A. Adjunct Diagnostic Value of Transcranial Magnetic Stimulation in Mucopolysaccharidosis-Related Cervical Myelopathy: A Pilot Study. Brain Sciences. 2019; 9(8):200. https://doi.org/10.3390/brainsci9080200
Chicago/Turabian StyleCantone, Mariagiovanna, Giuseppe Lanza, Alice Le Pira, Rita Barone, Giovanni Pennisi, Rita Bella, Manuela Pennisi, and Agata Fiumara. 2019. "Adjunct Diagnostic Value of Transcranial Magnetic Stimulation in Mucopolysaccharidosis-Related Cervical Myelopathy: A Pilot Study" Brain Sciences 9, no. 8: 200. https://doi.org/10.3390/brainsci9080200
APA StyleCantone, M., Lanza, G., Le Pira, A., Barone, R., Pennisi, G., Bella, R., Pennisi, M., & Fiumara, A. (2019). Adjunct Diagnostic Value of Transcranial Magnetic Stimulation in Mucopolysaccharidosis-Related Cervical Myelopathy: A Pilot Study. Brain Sciences, 9(8), 200. https://doi.org/10.3390/brainsci9080200







