Increased Voluntary Activation of the Elbow Flexors Following a Single Session of Spinal Manipulation in a Subclinical Neck Pain Population
Abstract
1. Introduction
2. Methods
2.1. Subjects
2.2. Data Recording and Analysis
2.2.1. EMG Recording
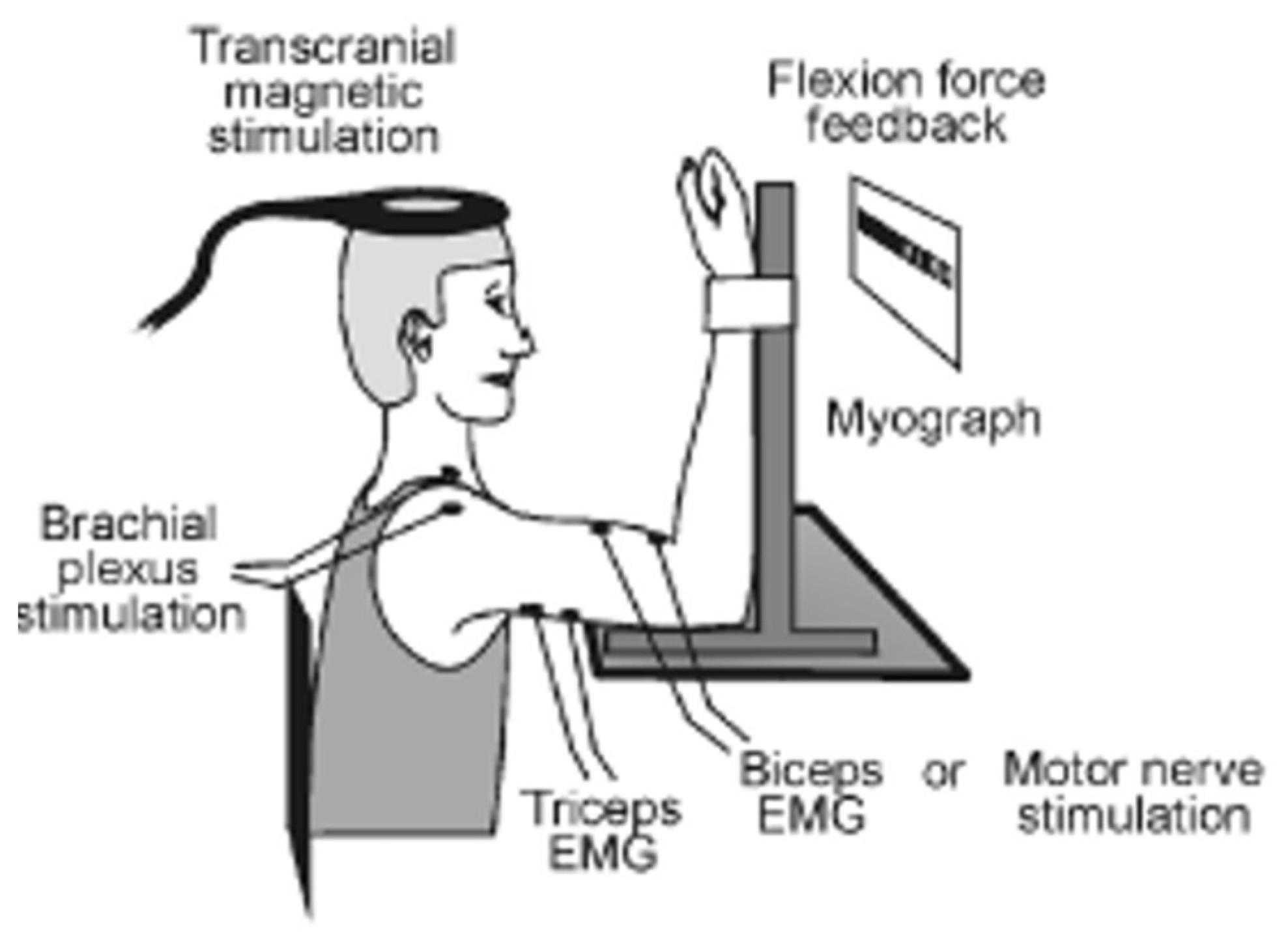
2.2.2. Electrical and Transcranial Magnetic Stimulation
2.2.3. Force Recording and Measurement of Maximal Voluntary Contraction Force
Calculation of Voluntary Activation
2.3. Experimental Protocol
2.4. Interventions
2.4.1. Spinal Manipulation
2.4.2. Control Intervention (Passive Movement)
2.5. Statistical Analysis
3. Results
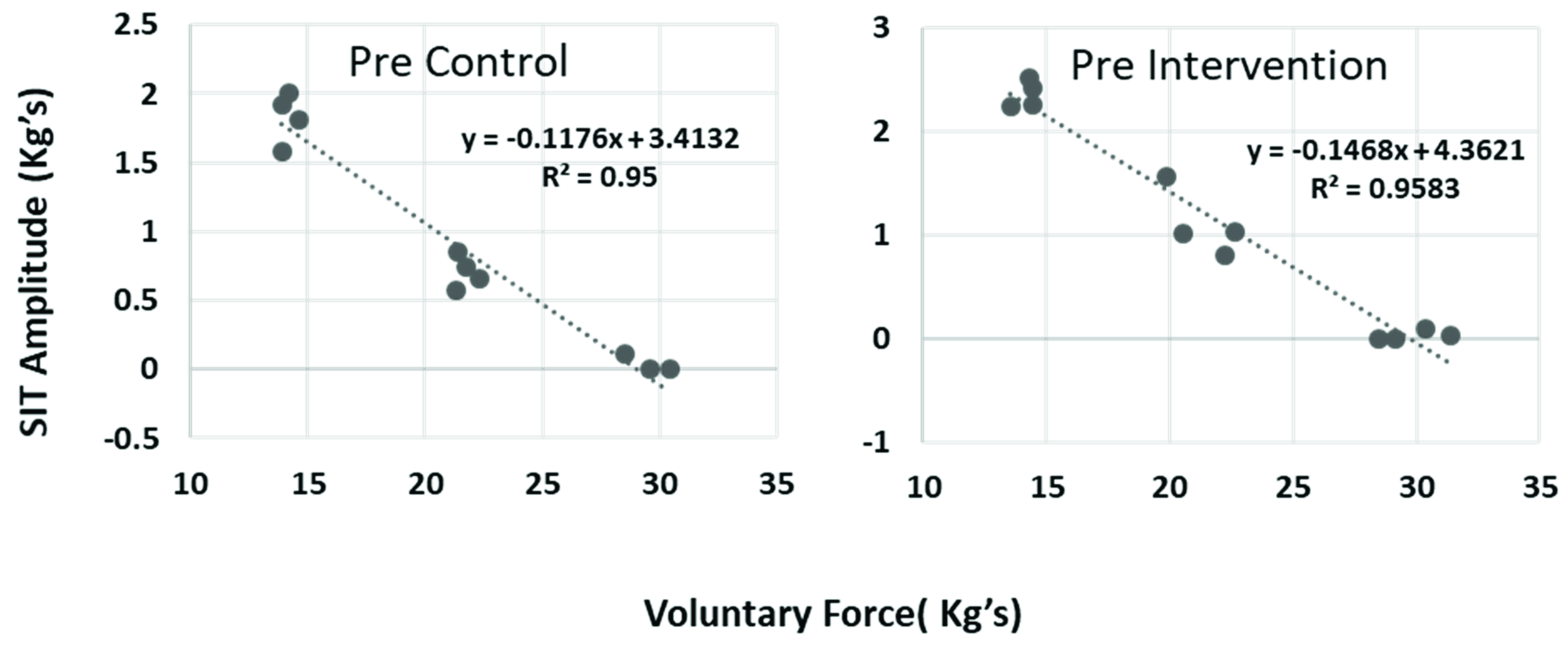
3.1. Baseline Measurements
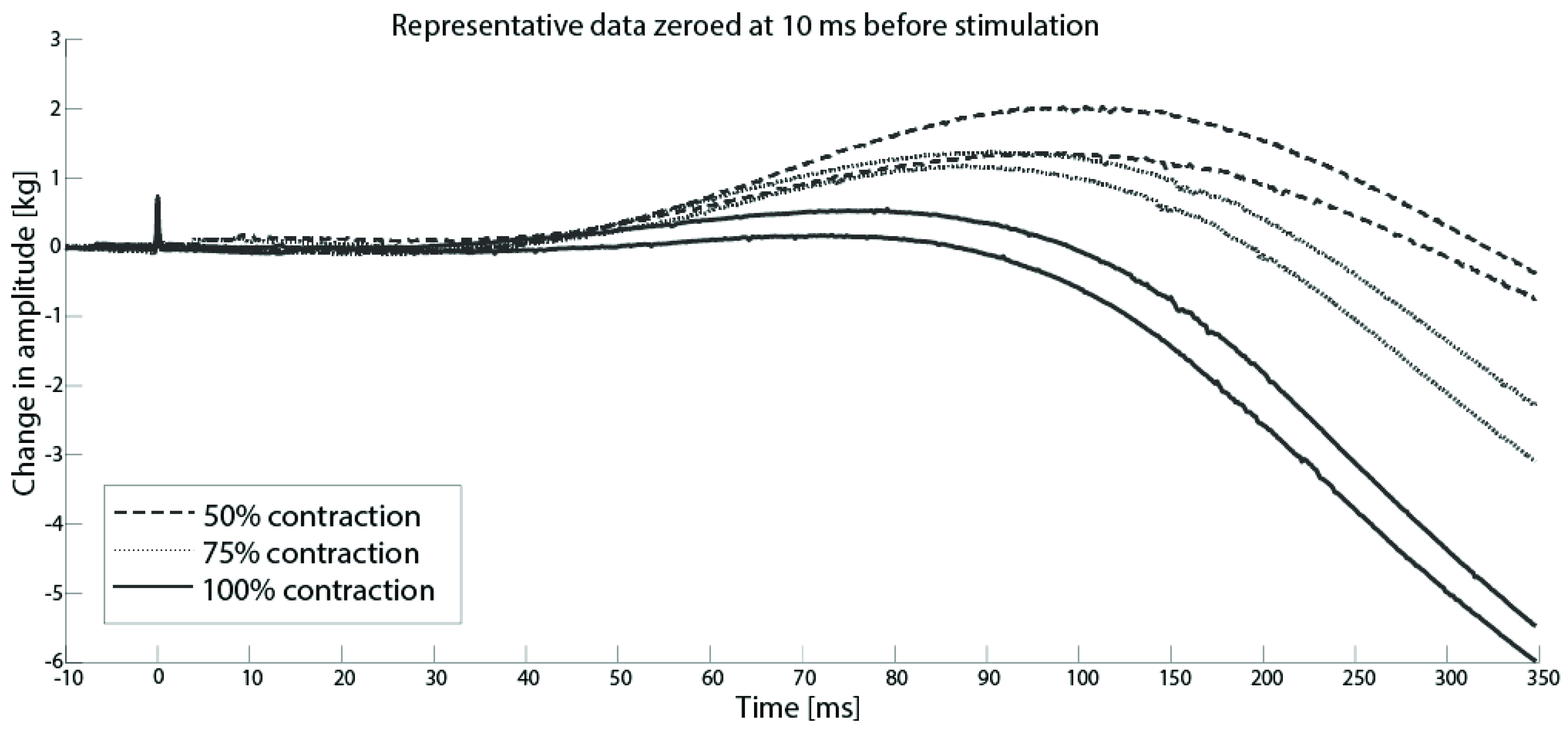
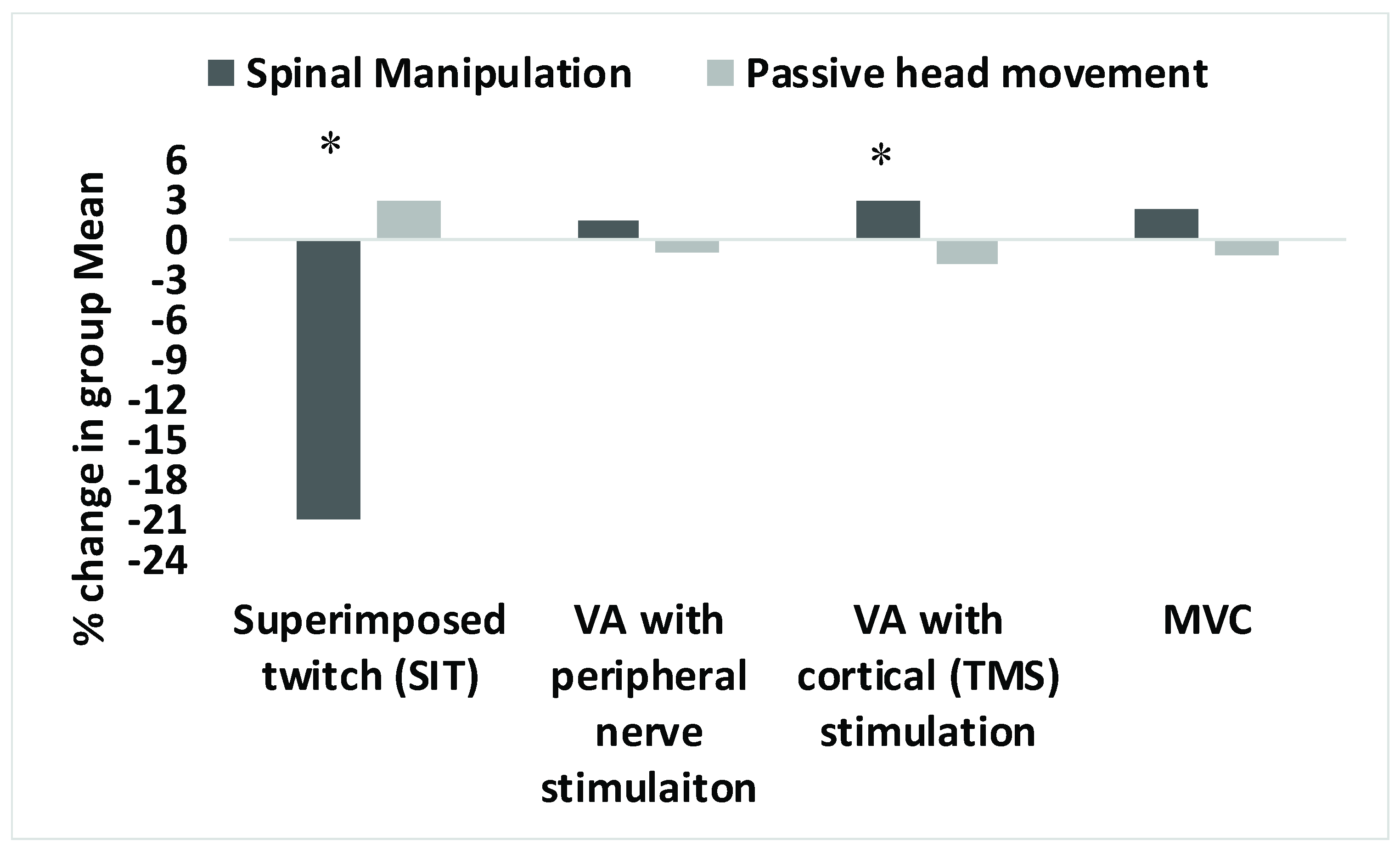
3.2. Superimposed Twitch
3.3. Voluntary Activation
3.4. MVC
4. Discussion
Limitations and Future Directions
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Botelho, M.B.; Andrade, B.B. Effect of Cervical Spine Manipulative Therapy on Judo Athletes’ Grip Strength. J. Manip. Physiol. Ther. 2012, 35, 38–44. [Google Scholar]
- Chilibeck, P.D.; Cornish, S.M.; Schulte, A.; Jantz, N.; Magnus, C.R.; Schwanbeck, S.; Juurlink, B.H. The effect of spinal manipulation on imbalances in leg strength. J. Can. Chiropr. Assoc. 2011, 55, 183–192. [Google Scholar]
- Christiansen, T.L.; Niazi, I.K.; Holt, K.; Nedergaard, R.W.; Duehr, J.; Allen, K.; Marshall, P.; Türker, K.S.; Hartvigsen, J.; Haavik, H. The effects of a single session of spinal manipulation on strength and cortical drive in athletes. Graefe’s Arch. Clin. Exp. Ophthalmol. 2018, 118, 737–749. [Google Scholar]
- Grindstaff, T.L.; Hertel, J.; Beazell, J.R.; Magrum, E.M.; Ingersoll, C.D. Effects of lumbopelvic joint manipulation on quadriceps activation and strength in healthy individuals. Man. Ther. 2009, 14, 415–420. [Google Scholar]
- Hillermann, B.; Gomes, A.N.; Korporaal, C.; Jackson, D. A Pilot Study Comparing the Effects of Spinal Manipulative Therapy with Those of Extra-Spinal Manipulative Therapy on Quadriceps Muscle Strength. J. Manip. Physiol. Ther. 2006, 29, 145–149. [Google Scholar]
- Keller, T.S.; Colloca, C.J. Mechanical force spinal manipulation increases trunk muscle strength assessed by electromyography: A comparative clinical trial. J. Manip. Physiol. Ther. 2000, 23, 585–595. [Google Scholar]
- Niazi, I.K.; Türker, K.S.; Flavel, S.; Kinget, M.; Duehr, J.; Haavik, H. Changes in H-reflex and V-waves following spinal manipulation. Exp. Brain Res. 2015, 233, 1165–1173. [Google Scholar]
- De Almeida, B.S.N.; Sabatino, J.H.; Giraldo, P.C. Effects of High-Velocity, Low-Amplitude Spinal Manipulation on Strength and the Basal Tonus of Female Pelvic Floor Muscles. J. Manip. Physiol. Ther. 2010, 33, 109–116. [Google Scholar]
- Suter, E.; McMorland, G.; Herzog, W.; Bray, R. Decrease in quadriceps inhibition after sacroiliac joint manipulation in patients with anterior knee pain. J. Manip. Physiol. Ther. 1999, 22, 149–153. [Google Scholar]
- Suter, E.; McMorland, G. Decrease in elbow flexor inhibition after cervical spine manipulation in patients with chronic neck pain. Clin. Biomech. 2002, 17, 541–544. [Google Scholar]
- Suter, E.; McMorland, G.; Herzog, W.; Bray, R. Conservative lower back treatment reduces inhibition in knee-extensor muscles: A randomized controlled trial. J. Manip. Physiol. Ther. 2000, 23, 76–80. [Google Scholar]
- Haavik, H.; Murphy, B. The role of spinal manipulation in addressing disordered sensorimotor integration and altered motor control. J. Electromyogr. Kinesiol. 2012, 22, 768–776. [Google Scholar]
- Taylor, H.H.; Murphy, B. Altered Central Integration of Dual Somatosensory Input after Cervical Spine Manipulation. J. Manip. Physiol. Ther. 2010, 33, 178–188. [Google Scholar]
- Baarbé, J.K.; Holmes, M.W.; Murphy, H.E.; Haavik, H.; Murphy, B.A. Influence of Subclinical Neck Pain on the Ability to Perform a Mental Rotation Task: A 4-Week Longitudinal Study With a Healthy Control Group Comparison. J. Manip. Physiol. Ther. 2016, 39, 23–30. [Google Scholar]
- Haavik, H.; Murphy, B. Subclinical Neck Pain and the Effects of Cervical Manipulation on Elbow Joint Position Sense. J. Manip. Physiol. Ther. 2011, 34, 88–97. [Google Scholar]
- Graven-Nielsen, T.; Lund, H.; Arendt-Nielsen, L.; Danneskiold-Samsøe, B.; Bliddal, H. Inhibition of maximal voluntary contraction force by experimental muscle pain: A centrally mediated mechanism. Muscle Nerve 2002, 26, 708–712. [Google Scholar]
- Lee, H.; Nicholson, L.L.; Adams, R.D. Cervical Range of Motion Associations With Subclinical Neck Pain. Spine 2004, 29, 33–40. [Google Scholar]
- Lee, H.-Y.; Wang, J.-D.; Yao, G.; Wang, S.-F.; Yao, K.-P.G. Association between cervicocephalic kinesthetic sensibility and frequency of subclinical neck pain. Man. Ther. 2008, 13, 419–425. [Google Scholar]
- Daligadu, J.; Haavik, H.; Yielder, P.C.; Baarbé, J.; Murphy, B. Alterations in Cortical and Cerebellar Motor Processing in Subclinical Neck Pain Patients Following Spinal Manipulation. J. Manip. Physiol. Ther. 2013, 36, 527–537. [Google Scholar]
- Baarbé, J.K.; Yielder, P.; Haavik, H.; Holmes, M.W.R.; Murphy, B.A. Subclinical recurrent neck pain and its treatment impacts motor training-induced plasticity of the cerebellum and motor cortex. PLoS ONE 2018, 13, e0193413. [Google Scholar]
- Smith, D.L.; Cox, R.H. Muscular strength and chiropractic: theoretical mechanisms and health implications. J. Vertebr. Subluxation Res. 1999, 2000, 1–13. [Google Scholar]
- Behm, D.G.; St-Pierre, D.M.M.; Pérez, D. Muscle inactivation: Assessment of interpolated twitch technique. J. Appl. Physiol. 1996, 81, 2267–2273. [Google Scholar]
- Shield, A.; Zhou, S.; Shield, D.A. Assessing Voluntary Muscle Activation with the Twitch Interpolation Technique. Sports Med. 2004, 34, 253–267. [Google Scholar]
- Todd, G.; Taylor, J.L.; Gandevia, S.C. Reproducible measurement of voluntary activation of human elbow flexors with motor cortical stimulation. J. Appl. Physiol. 2004, 97, 236–242. [Google Scholar]
- Lee, M.; Gandevia, S.C.; Carroll, T.J. Cortical voluntary activation can be reliably measured in human wrist extensors using transcranial magnetic stimulation. Clin. Neurophysiol. 2008, 119, 1130–1138. [Google Scholar]
- Sidhu, S.K.; Bentley, D.J.; Carroll, T.J. Cortical voluntary activation of the human knee extensors can be reliably estimated using transcranial magnetic stimulation. Muscle Nerve 2009, 39, 186–196. [Google Scholar]
- Todd, G.; Taylor, J.L.; Gandevia, S.C. Measurement of voluntary activation of fresh and fatigued human muscles using transcranial magnetic stimulation. J. Physiol. 2003, 551, 661–671. [Google Scholar]
- Gandevia, S.C.; Allen, G.M.; Butler, J.E.; Taylor, J.L. Supraspinal factors in human muscle fatigue: Evidence for suboptimal output from the motor cortex. J. Physiol. 1996, 490, 529–536. [Google Scholar]
- Oldfield, R. The assessment and analysis of handedness: The Edinburgh inventory. Neuropsychologia 1971, 9, 97–113. [Google Scholar]
- Mogk, J.P.; Rogers, L.M.; Murray, W.M.; Perreault, E.J.; Stinear, J.W. Corticomotor excitability of arm muscles modulates according to static position and orientation of the upper limb. Clin. Neurophysiol. 2014, 125, 2046–2054. [Google Scholar]
- Nuzzo, J.L.; Trajano, G.S.; Barry, B.K.; Gandevia, S.C.; Taylor, J.L. Arm posture-dependent changes in corticospinal excitability are largely spinal in origin. J. Neurophysiol. 2016, 115, 2076–2082. [Google Scholar]
- Rothwell, J.; Thompson, P.; Day, B.; Boyd, S.; Marsden, C. Stimulation of the human motor cortex through the scalp. Exp. Physiol. 1991, 76, 159–200. [Google Scholar]
- Rothwell, J.; Rothwell, J. Techniques and mechanisms of action of transcranial stimulation of the human motor cortex. J. Neurosci. Methods 1997, 74, 113–122. [Google Scholar]
- Allen, G.M.; Gandevia, S.C.; McKenzie, D.K. Reliability of measurements of muscle strength and voluntary activation using twitch interpolation. Muscle Nerve 1995, 18, 593–600. [Google Scholar]
- Triano, J.J.; Budgell, B.; Bagnulo, A.; Roffey, B.; Bergmann, T.; Cooperstein, R.; Gleberzon, B.; Good, C.; Perron, J.; Tepe, R. Review of methods used by chiropractors to determine the site for applying manipulation. Chiropr. Man. Ther. 2013, 21, 36. [Google Scholar]
- Hessell, B.W.; Herzog, W.; Conway, P.J.; McEwen, M.C. Experimental measurement of the force exerted during spinal manipulation using the Thompson technique. J. Manip. Physiol. Ther. 1990, 13, 448–453. [Google Scholar]
- Lelic, D.; Niazi, I.K.; Holt, K.; Jochumsen, M.; Dremstrup, K.; Yielder, P.; Murphy, B.; Drewes, A.M.; Haavik, H.; Drewes, A.; et al. Manipulation of Dysfunctional Spinal Joints Affects Sensorimotor Integration in the Prefrontal Cortex: A Brain Source Localization Study. Neural Plast. 2016, 2016, 1–9. [Google Scholar]
- Pickar, J.G.; Wheeler, J.D. Response of muscle proprioceptors to spinal manipulative-like loads in the anesthetized cat. J. Manip. Physiol. Ther. 2001, 24, 2–11. [Google Scholar]
- Reed, W.R.; Pickar, J.G. Paraspinal muscle spindle response to intervertebral fixation and segmental thrust level during spinal manipulation in an animal model. Spine 2015, 40, E752–E759. [Google Scholar]
- Taylor, H.H.; Murphy, B. Altered Sensorimotor Integration With Cervical Spine Manipulation. J. Manip. Physiol. Ther. 2008, 31, 115–126. [Google Scholar]
- Haavik, H.; Niazi, I.K.; Jochumsen, M.; Uginčius, P.; Sebik, O.; Yılmaz, G.; Navid, M.S.; Ozyurt, M.G.; Türker, K.S.; Yilmaz, G. Chiropractic spinal manipulation alters TMS induced I-wave excitability and shortens the cortical silent period. J. Electromyogr. Kinesiol. 2018, 42, 24–35. [Google Scholar]
- Wall, J.; Xu, J.; Wang, X. Human brain plasticity: An emerging view of the multiple substrates and mechanisms that cause cortical changes and related sensory dysfunctions after injuries of sensory inputs from the body. Brain Res. Rev. 2002, 39, 181–215. [Google Scholar]
- Falla, D.; Cescon, C.; Lindstroem, R.; Barbero, M. Muscle Pain Induces a Shift of the Spatial Distribution of Upper Trapezius Muscle Activity during a Repetitive Task: A Mechanism for Perpetuation of Pain with Repetitive Activity? Clin. J. Pain 2017, 33, 1006–1013. [Google Scholar]
- Sterling, M.; Jull, G.; Vicenzino, B.; Kenardy, J.; Darnell, R. Development of motor system dysfunction following whiplash injury. Pain 2003, 103, 65–73. [Google Scholar]
- Michaelson, P.; Michaelson, M.; Jaric, S.; Latash, L.M.; Sjölander, P.; Djupsjöbacka, M. VERTICAL POSTURE AND HEAD STABILITY IN PATIENTS WITH CHRONIC NECK PAIN. J. Rehabilitation Med. 2003, 35, 229–235. [Google Scholar]
- Haavik-Taylor, H.; Murphy, B. Cervical spine manipulation alters sensorimotor integration: A somatosensory evoked potential study. Clin. Neurophysiol. 2007, 118, 391–402. [Google Scholar]
- Goldman-Rakic, P.S. Motor control function of the prefrontal cortex. Ciba Found. Symp. 1987, 132, 187–200. [Google Scholar]
- Paulus, I.; Brumagne, S. Altered interpretation of neck proprioceptive signals in persons with subclinical recurrent neck pain. J. Rehabilitation Med. 2008, 40, 426–432. [Google Scholar]
- Zabihhosseinian, M.; Holmes, M.W.; Howarth, S.; Ferguson, B.; Murphy, B. Neck muscle fatigue differentially alters scapular and humeral kinematics during humeral elevation in subclinical neck pain participants versus healthy controls. J. Electromyogr. Kinesiol. 2017, 33, 73–82. [Google Scholar]
- Hodges, P.W.; James, G.; Blomster, L.; Hall, L.; Schmid, A.B.; Shu, C.; Little, C.; Melrose, J. Can Proinflammatory Cytokine Gene Expression Explain Multifidus Muscle Fiber Changes After an Intervertebral Disc Lesion? Spine 2014, 39, 1010–1017. [Google Scholar]
- Brown, S.H.M.; Gregory, D.E.; Carr, J.A.; Ward, S.R.; Masuda, K.; Lieber, R.L. ISSLS prize winner: Adaptations to the multifidus muscle in response to experimentally induced intervertebral disc degeneration. Spine 2011, 36, 1728–1736. [Google Scholar]
- Hodges, P.W.; James, G.; Blomster, L.; Hall, L.; Schmid, A.; Shu, C.; Little, C.; Melrose, J. Multifidus Muscle Changes after Back Injury Are Characterized by Structural Remodeling of Muscle, Adipose and Connective Tissue, but Not Muscle Atrophy: Molecular and Morphological Evidence. Spine 2015, 40, 1057–1071. [Google Scholar]
- James, G.; Blomster, L.; Hall, L.; Schmid, A.B.; Shu, C.C.; Little, C.B.; Melrose, J.; Hodges, P.W. Mesenchymal stem cell treatment of intervertebral disc lesion prevents fatty infiltration and fibrosis of the multifidus muscle, but not cytokine and muscle fiber changes. Spine 2016, 41, 1208–1217. [Google Scholar]
- Alaranta, H.; Tallroth, K.; Soukka, A.; Heliövaara, M. Fat content of lumbar extensor muscles and low back disability: A radiographic and clinical comparison. J. Spinal Disord. 1993, 6, 137–140. [Google Scholar]
- Kjaer, P.; Bendix, T.; Sorensen, J.S.; Korsholm, L.; Leboeuf-Yde, C. Are MRI-defined fat infiltrations in the multifidus muscles associated with low back pain? BMC Med. 2007, 5, 2. [Google Scholar] [CrossRef]
- Sun, D.; Liu, P.; Cheng, J.; Ma, Z.; Liu, J.; Qin, T. Correlation between intervertebral disc degeneration, paraspinal muscle atrophy, and lumbar facet joints degeneration in patients with lumbar disc herniation. BMC Musculoskelet. Disord. 2017, 18, 167. [Google Scholar]
- Fortin, M.; Lazáry, À.; Varga, P.P.; McCall, I.; Battié, M.C. Paraspinal muscle asymmetry and fat infiltration in patients with symptomatic disc herniation. Eur. Spine J. 2016, 25, 1452–1459. [Google Scholar]
- Zhao, W.P.; Kawaguchi, Y.; Matsui, H.; Kanamori, M.; Kimura, T. Histochemistry and morphology of the multifidus muscle in lumbar disc herniation: Comparative study between diseased and normal sides. Spine 2000, 25, 2191–2199. [Google Scholar]
- Meier, M.L.; Vrana, A.; Schweinhardt, P. Low Back Pain: The Potential Contribution of Supraspinal Motor Control and Proprioception. Neuroscientist 2018. [Google Scholar] [CrossRef]
- Manzoni, D. The cerebellum and sensorimotor coupling: Looking at the problem from the perspective of vestibular reflexes. Cerebellum 2007, 6, 24–37. [Google Scholar]
- Moulton, E.A.; Schmahmann, J.D.; Becerra, L.; Borsook, D. The cerebellum and pain: Passive integrator or active participator? Brain Res. Rev. 2010, 65, 14–27. [Google Scholar]
- Andrew, D.; Yielder, P.; Haavik, H.; Murphy, B. The effects of subclinical neck pain on sensorimotor integration following a complex motor pursuit task. Exp. Brain Res. 2017, 236, 1–11. [Google Scholar]
- Taylor, H.H.; Murphy, B. Transient modulation of intracortical inhibition following spinal manipulation. Chiropr. J. Aust. 2007, 37, 106–116. [Google Scholar]
- Haavik, H.; Niazi, I.K.; Jochumsen, M.; Sherwin, D.; Flavel, S.; Türker, K.S. Impact of spinal manipulation on cortical drive to upper and lower limb muscles. Brain Sci. 2017, 7, 2. [Google Scholar]
- Shibasaki, H.; Hallett, M. What is the Bereitschaftspotential? Clin. Neurophysiol. 2006, 117, 2341–2356. [Google Scholar]
| (Spinal Manipulation) | (Passive Movements) | (Spinal Manipulation) | (Passive Movements) | (Spinal Manipulation) | (Passive Movements) |
|---|---|---|---|---|---|
| MVC (Kg’s) | MVC (Kg’s) | VA Cortical (TMS) | VA Cortical (TMS) | VA Peripheral Nerve Stimulation) | VA Peripheral Nerve Stimulation) |
| Pre 20.37 (SD = 7.36) | 20.14 (SD 7.39) | 88.80 (SD 8.17) | 91.36 (SD 7.39) | 89.5 (SD 13.22) | 94.74 (SD 5.68) |
| Post 20.84 (SD = 7.345) | 19.89 (SD 7.41) | 91.47 (SD 7.16) | 89.70 (SD 12.26) | 90.77 (SD 9.28) | 93.83 (SD 7.50) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kingett, M.; Holt, K.; Niazi, I.K.; Nedergaard, R.W.; Lee, M.; Haavik, H. Increased Voluntary Activation of the Elbow Flexors Following a Single Session of Spinal Manipulation in a Subclinical Neck Pain Population. Brain Sci. 2019, 9, 136. https://doi.org/10.3390/brainsci9060136
Kingett M, Holt K, Niazi IK, Nedergaard RW, Lee M, Haavik H. Increased Voluntary Activation of the Elbow Flexors Following a Single Session of Spinal Manipulation in a Subclinical Neck Pain Population. Brain Sciences. 2019; 9(6):136. https://doi.org/10.3390/brainsci9060136
Chicago/Turabian StyleKingett, Mat, Kelly Holt, Imran Khan Niazi, Rasmus Wiberg Nedergaard, Michael Lee, and Heidi Haavik. 2019. "Increased Voluntary Activation of the Elbow Flexors Following a Single Session of Spinal Manipulation in a Subclinical Neck Pain Population" Brain Sciences 9, no. 6: 136. https://doi.org/10.3390/brainsci9060136
APA StyleKingett, M., Holt, K., Niazi, I. K., Nedergaard, R. W., Lee, M., & Haavik, H. (2019). Increased Voluntary Activation of the Elbow Flexors Following a Single Session of Spinal Manipulation in a Subclinical Neck Pain Population. Brain Sciences, 9(6), 136. https://doi.org/10.3390/brainsci9060136







