Major Depressive Disorder Is Associated with Impaired Interoceptive Accuracy: A Systematic Review
Abstract
1. Introduction
2. Methods
2.1. Search Strategy
2.2. Eligibility Criteria
2.3. Study Selection
2.4. Data Extraction
3. Results
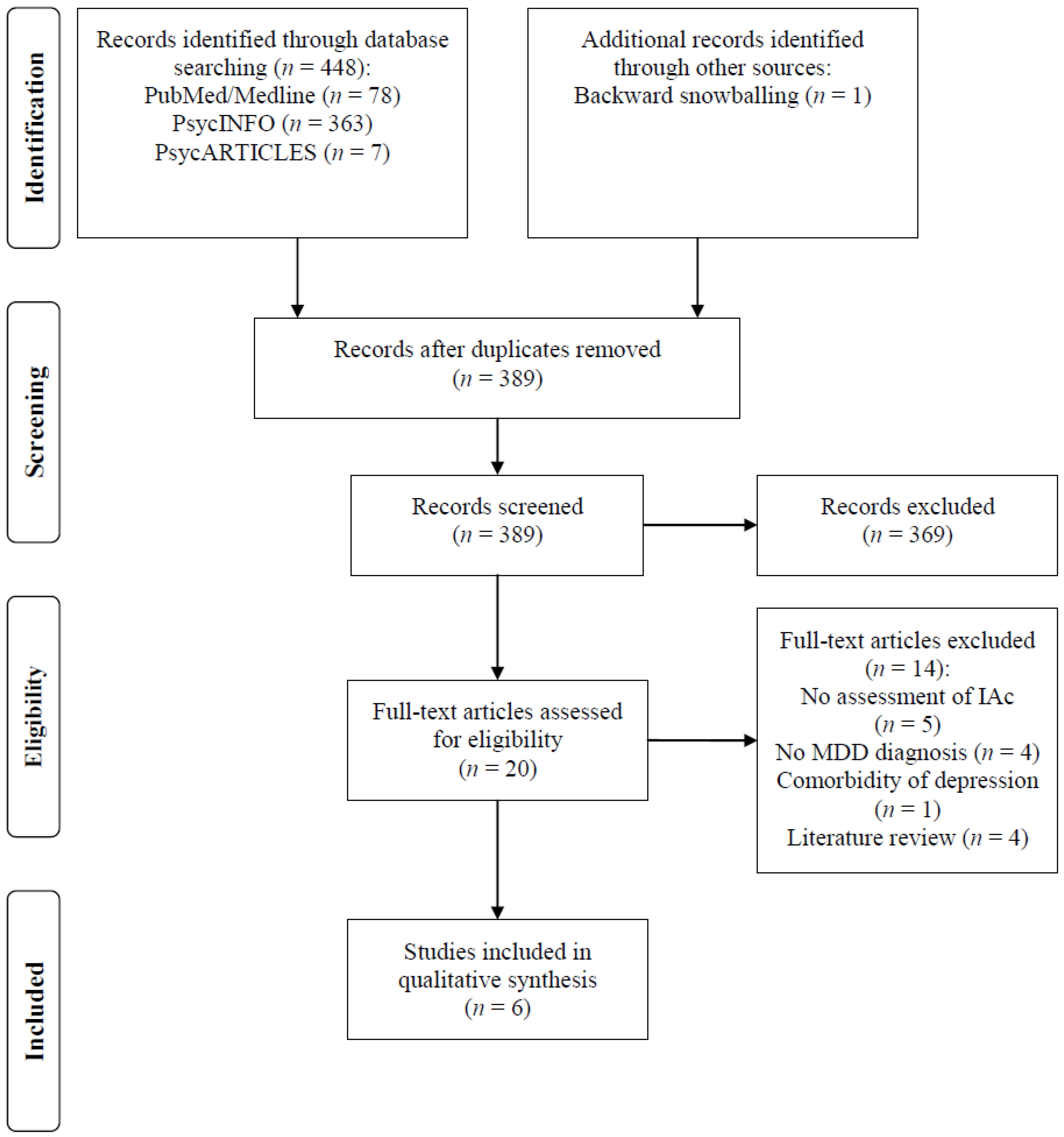
3.1. Process of Study Selection
3.2. Characteristics of Included Studies
3.3. Group Differences on Heartbeat Perception Task Performance
3.4. Association between Interoceptive Accuracy and Depression Severity
3.5. Modern Antidepressants as Possible Confounders
3.6. Associations between Interoceptive Accuracy and MDD-Related Symptoms
4. Discussion
4.1. Preliminary Explanations for the Inverted U-Shaped Relationship
4.2. Interoception and Its Link to Affective and Cognitive Disturbances in MDD
4.3. Limitations
4.4. Clinical Implications
4.5. Future Research Directions
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Arlington, VA, USA, 2013; ISBN 9780890425558. [Google Scholar]
- Grover, S.; Kumar, V.; Chakrabarti, S.; Hollikatti, P.; Singh, P.; Tyagi, S.; Kulhara, P.; Avasthi, A. Prevalence and type of functional somatic complaints in patients with first-episode depression. East Asian Arch. Psychiatry 2012, 22, 146–153. [Google Scholar]
- Vaccarino, A.L.; Sills, T.L.; Evans, K.R.; Kalali, A.H. Prevalence and association of somatic symptoms in patients with Major Depressive Disorder. J. Affect. Disord. 2008, 110, 270–276. [Google Scholar] [CrossRef]
- Henningsen, P.; Zimmermann, T.; Sattel, H. Medically unexplained physical symptoms, anxiety, and depression: A meta-analytic review. Psychosom. Med. 2003, 65, 528–533. [Google Scholar] [CrossRef]
- Chakraborty, K.; Avasthi, A.; Kumar, S.; Grover, S. Psychological and clinical correlates of functional somatic complaints in depression. Int. J. Soc. Psychiatry 2012, 58, 87–95. [Google Scholar] [CrossRef]
- Simon, G.E.; Vonkorff, M.; Piccinelli, M.; Fullerton, C.; Ormel, J. An International Study of the Relation between Somatic Symptoms and Depression. New Engl. J. Med. 1999, 341, 1329–1335. [Google Scholar] [CrossRef]
- Schaefer, M.; Egloff, B.; Witthöft, M. Is interoceptive awareness really altered in somatoform disorders? Testing competing theories with two paradigms of heartbeat perception. J. Psychol. 2012, 121, 719–724. [Google Scholar] [CrossRef]
- Khalsa, S.S.; Lapidus, R.C. Can Interoception Improve the Pragmatic Search for Biomarkers in Psychiatry? Front. Psychol. 2016, 7, 633. [Google Scholar] [CrossRef]
- Niedenthal, P.M. Embodying Emotion. Science 2007, 316, 1002–1005. [Google Scholar] [CrossRef]
- Herbert, B.M.; Pollatos, O. The Body in the Mind: On the Relationship Between Interoception and Embodiment. Top. Cogn. Sci. 2012, 4, 692–704. [Google Scholar] [CrossRef]
- Fuchs, T.; Schlimme, J.E. Embodiment and psychopathology: A phenomenological perspective. Curr. Opin. 2009, 22, 570–575. [Google Scholar] [CrossRef]
- Sherrington, C.S. The Integrative Action of the Nervous System; Yale University Press: New Haven, CT, USA, 1906. [Google Scholar]
- Ceunen, E.; Vlaeyen, J.W.S.; Van Diest, I. On the Origin of Interoception. Front. Psychol. 2016, 7, 577. [Google Scholar] [CrossRef]
- Craig, A.D. How do you feel? Interoception: The sense of the physiological condition of the body. Nat. Rev. Neurosci. 2002, 3, 655–666. [Google Scholar] [CrossRef]
- Khalsa, S.S.; Adolphs, R.; Cameron, O.G.; Critchley, H.D.; Davenport, P.W.; Feinstein, J.S.; Feusner, J.D.; Garfinkel, S.N.; Lane, R.D.; Mehling, W.E.; et al. Interoception and Mental Health: A Roadmap. Biolog. Psychiatry Cog. Neurosci. Neuroimag. 2018, 3, 501–513. [Google Scholar] [CrossRef]
- Craig, A.D. (Bud) How do you feel—Now? The anterior insula and human awareness. Nat. Rev. Neurosci. 2009, 10, 59–70. [Google Scholar] [CrossRef]
- Teh, M.M.; Dunn, J.T.; Choudhary, P.; Samarasinghe, Y.; Macdonald, I.; O’Doherty, M.; Marsden, P.; Reed, L.J.; Amiel, S.A. Evolution and resolution of human brain perfusion responses to the stress of induced hypoglycemia. NeuroImage 2010, 53, 584–592. [Google Scholar] [CrossRef]
- Tsakiris, M.; Critchley, H. Interoception beyond homeostasis: Affect, cognition and mental health. Philos. Trans. Soc. B Boil. Sci. 2016, 371, 20160002. [Google Scholar] [CrossRef]
- Wiens, S. Interoception in emotional experience. Curr. Opin. Neurol. 2005, 18, 442–447. [Google Scholar] [CrossRef]
- Critchley, H.D.; Wiens, S.; Rotshtein, P.; Öhman, A.; Dolan, R.J. Neural systems supporting interoceptive awareness. Nat. Neurosci. 2004, 7, 189–195. [Google Scholar] [CrossRef]
- Paulus, M.P.; Stein, M.B. Interoception in anxiety and depression. Brain Struct. Funct. 2010, 214, 451–463. [Google Scholar] [CrossRef]
- Barrett, L.F.; Simmons, W.K. Interoceptive predictions in the brain. Nat. Rev. Neurosci. 2015, 16, 419–429. [Google Scholar] [CrossRef]
- Seth, A.K.; Friston, K.J. Active interoceptive inference and the emotional brain. Philos. Trans. Soc. B Boil. Sci. 2016, 371, 20160007. [Google Scholar] [CrossRef]
- Quadt, L.; Critchley, H.D.; Garfinkel, S.N. The neurobiology of interoception in health and disease. Ann. N. Y. Acad. Sci. 2018, 1428, 112–128. [Google Scholar] [CrossRef]
- Garfinkel, S.N.; Critchley, H.D. Interoception, emotion and brain: New insights link internal physiology to social behaviour. Commentary on: “Anterior insular cortex mediates bodily sensibility and social anxiety” by Terasawa et al. 2012. Soc. Cogn. Affect. Neurosci. 2013, 8, 231–234. [Google Scholar] [CrossRef]
- Garfinkel, S.N.; Seth, A.K.; Barrett, A.B.; Suzuki, K.; Critchley, H.D. Knowing your own heart: Distinguishing interoceptive accuracy from interoceptive awareness. Boil. Psychol. 2015, 104, 65–74. [Google Scholar] [CrossRef]
- Schandry, R. Heart Beat Perception and Emotional Experience. Psychophysiology 1981, 18, 483–488. [Google Scholar] [CrossRef]
- Whitehead, W.E.; Drescher, V.M.; Heiman, P.; Blackwell, B.; Whitehead, D.W.E. Relation of heart rate control to heartbeat perception. Biofeedback Self Regul. 1977, 2, 371–392. [Google Scholar] [CrossRef]
- Schulz, S.M. Neural correlates of heart-focused interoception: A functional magnetic resonance imaging meta-analysis. Philos. Trans. Soc. B Boil. Sci. 2016, 371, 20160018. [Google Scholar] [CrossRef]
- Avery, J.A.; Drevets, W.C.; Moseman, S.E.; Bodurka, J.; Barcalow, J.C.; Simmons, W.K. Major depressive disorder is associated with abnormal interoceptive activity and functional connectivity in the insula. Biol. Psychiatry 2014, 76, 258–266. [Google Scholar] [CrossRef]
- Cameron, O.G. Interoception: The Inside Story—A Model for Psychosomatic Processes. Psychosom. Med. 2001, 63, 697–710. [Google Scholar] [CrossRef]
- Duquette, P. Increasing Our Insular World View: Interoception and Psychopathology for Psychotherapists. Front. Neurosci. 2017, 11, 135. [Google Scholar] [CrossRef]
- DeVille, D.C.; Kerr, K.L.; Avery, J.A.; Burrows, K.; Bodurka, J.; Feinstein, J.; Khalsa, S.S.; Paulus, M.P.; Simmons, W.K. The Neural Bases of Interoceptive Encoding and Recall in Healthy Adults and Adults With Depression. Biolog. Psychiatry Cog. Neurosci. Neuroimag. 2018, 3, 546–554. [Google Scholar] [CrossRef]
- Harshaw, C. Interoceptive dysfunction: Toward an integrated framework for understanding somatic and affective disturbance in depression. Psychol. Bull. 2015, 141, 311–363. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. The PRISMA Group Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLOS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Ehlers, A.; Breuer, P. Increased cardiac awareness in panic disorder. J. Psychol. 1992, 101, 371–382. [Google Scholar] [CrossRef]
- Van Der Does, A.W.; van Dyck, R.; Spinhoven, P. Accurate heartbeat perception in panic disorder: Fact and artefact. J. Affect. Disord. 1997, 43, 121–130. [Google Scholar] [CrossRef]
- Mussgay, L.; Klinkenberg, N.; Rüddel, H. Heart Beat Perception in Patients with Depressive, Somatoform, and Personality Disorders. Psychophysiology 1999, 13, 27–36. [Google Scholar] [CrossRef]
- Dunn, B.D.; Dalgleish, T.; Ogilvie, A.D.; Lawrence, A.D. Heartbeat perception in depression. Behav. Res. Ther. 2007, 45, 1921–1930. [Google Scholar] [CrossRef]
- Terhaar, J.; Viola, F.C.; Bär, K.-J.; Debener, S. Heartbeat evoked potentials mirror altered body perception in depressed patients. Clin. Neurophysiol. 2012, 123, 1950–1957. [Google Scholar] [CrossRef]
- Furman, D.J.; Waugh, C.E.; Bhattacharjee, K.; Thompson, R.J.; Gotlib, I.H. Interoceptive Awareness, Positive Affect, and Decision Making in Major Depressive Disorder. J. Affect. Disord. 2013, 151, 780–785. [Google Scholar] [CrossRef]
- Pollatos, O.; Schandry, R.; Traut-Mattausch, E.; Traut-Mattausch, E.; Traut-Mattausch, E. Differential effects of anxiety and depression on interoceptive accuracy. Depression Anxiety 2009, 26, 167–173. [Google Scholar] [CrossRef]
- Brown, C.; Schulberg, H.C.; Madonia, M.J.; Shear, M.K.; Houck, P.R. Treatment outcomes for primary care patients with major depression and lifetime anxiety disorders. Am. J. Psychiatry 1996, 153, 1293–1300. [Google Scholar]
- Fairclough, S.H.; Goodwin, L. The effect of psychological stress and relaxation on interoceptive accuracy: Implications for symptom perception. J. Psychosom. 2007, 62, 289–295. [Google Scholar] [CrossRef]
- Stübner, S.; Grohmann, R.; Greil, W.; Zhang, X.; Müller-Oerlinghausen, B.; Bleich, S.; Rüther, E.; Möller, H.-J.; Engel, R.; Falkai, P.; et al. Suicidal Ideation and Suicidal Behavior as Rare Adverse Events of Antidepressant Medication: Current Report from the AMSP Multicenter Drug Safety Surveillance Project. Int. J. Neuropsychopharmacol. 2018, 21, 814–821. [Google Scholar] [CrossRef]
- Beach, S.R.; Kostis, W.J.; Celano, C.M.; Januzzi, J.L.; Ruskin, J.N.; Noseworthy, P.A.; Huffman, J.C. Meta-Analysis of Selective Serotonin Reuptake Inhibitor–Associated QTc Prolongation. J. Clin. Psychiatry 2014, 75, 441–449. [Google Scholar] [CrossRef]
- Rottenberg, J.; Gross, J.J.; Gotlib, I.H. Emotion Context Insensitivity in Major Depressive Disorder. J. Psychol. 2005, 114, 627–639. [Google Scholar] [CrossRef]
- James, W. What is an emotion? Mind 1884, 9, 188–205. [Google Scholar] [CrossRef]
- Schachter, S.; Singer, J. Cognitive, social, and physiological determinants of emotional state. Psychol. Rev. 1962, 69, 379–399. [Google Scholar] [CrossRef]
- Damasio, A.R. Descartes’ Error. Emotion, Rationality and the Human Brain; G.P. Putnam’s Sons: New York, NY, USA, 1994. [Google Scholar]
- Damasio, A.R. The Feeling of What Happens. Body and Emotion in the Making of Consciousness; Harcourt Brace & Company: New York, NY, USA, 1999; ISBN 978-0156010757. [Google Scholar]
- Ainley, V.; Tsakiris, M. Body Conscious? Interoceptive Awareness, Measured by Heartbeat Perception, Is Negatively Correlated with Self-Objectification. PloS ONE 2013, 8, e55568. [Google Scholar] [CrossRef]
- Forrest, L.N.; Smith, A.R.; White, R.D.; Joiner, T.E. (Dis)connected: An examination of interoception in individuals with suicidality. J. Abnorm. Psychol. 2015, 124, 754–763. [Google Scholar] [CrossRef]
- Rogers, M.L.; Hagan, C.R.; Joiner, T.E. Examination of interoception along the suicidality continuum. J. Clin. Psychol. 2018, 74, 1004–1016. [Google Scholar] [CrossRef]
- Craig, A.D. A new view of pain as a homeostatic emotion. Trends Neurosci. 2003, 26, 303–307. [Google Scholar] [CrossRef]
- Dunn, B.D.; Galton, H.C.; Morgan, R.; Evans, D.; Oliver, C.; Meyer, M.; Cusack, R.; Lawrence, A.D.; Dalgleish, T. Listening to your heart. How interoception shapes emotion experience and intuitive decision making. Psychol. Sci. 2010, 21, 1835–1844. [Google Scholar] [CrossRef]
- Weitkunat, R.; Schandry, R.; Sparrer, B.; Beck, K. The trainability of heart perception. Z. Exp. Angew. Psychol. 1987, 34, 340–350. [Google Scholar]
- Murphy, J.; Brewer, R.; Hobson, H.; Catmur, C.; Bird, G. Is alexithymia characterised by impaired interoception? Further evidence, the importance of control variables, and the problems with the Heartbeat Counting Task. Boil. Psychol. 2018, 136, 189–197. [Google Scholar] [CrossRef]
- Herbert, B.M.; Muth, E.R.; Pollatos, O.; Herbert, C. Interoception across Modalities: On the Relationship between Cardiac Awareness and the Sensitivity for Gastric Functions. PLoS ONE 2012, 7, e36646. [Google Scholar] [CrossRef]
- Whitehead, W.E.; Drescher, V.M. Perception of Gastric Contractions and Self-Control of Gastric Motility. Psychophysiology 1980, 17, 552–558. [Google Scholar] [CrossRef]
- Lamm, C.; Singer, T. The role of anterior insular cortex in social emotions. Brain Struct. Funct. 2010, 214, 579–591. [Google Scholar] [CrossRef]
- Murphy, J.; Brewer, R.; Catmur, C.; Bird, G. Interoception and psychopathology: A developmental neuroscience perspective. Dev. Cogn. Neurosci. 2017, 23, 45–56. [Google Scholar] [CrossRef]
- Hatfield, T.R.; Brown, R.F.; Giummarra, M.J.; Lenggenhager, B. Autism spectrum disorder and interoception: Abnormalities in global integration? Autism 2019, 23, 212–222. [Google Scholar] [CrossRef]
- Jenkinson, P.M.; Taylor, L.; Laws, K.R. Self-reported interoceptive deficits in eating disorders: A meta-analysis of studies using the eating disorder inventory. J. Psychosom. 2018, 110, 38–45. [Google Scholar] [CrossRef]
- Klabunde, M.; Acheson, D.T.; Boutelle, K.N.; Matthews, S.C.; Kaye, W.H. Interoceptive sensitivity deficits in women recovered from bulimia nervosa. Eat. Behav. 2013, 14, 488–492. [Google Scholar] [CrossRef]
- Strawn, J.R.; Cotton, S.; Luberto, C.M.; Patino, L.R.; Stahl, L.A.; Weber, W.A.; Eliassen, J.C.; Sears, R.; DelBello, M.P. Neural Function Before and After Mindfulness-Based Cognitive Therapy in Anxious Adolescents at Risk for Developing Bipolar Disorder. J. Child Adolesc. Psychopharmacol. 2016, 26, 372–379. [Google Scholar] [CrossRef]
- Piet, J.; Hougaard, E. The effect of mindfulness-based cognitive therapy for prevention of relapse in recurrent major depressive disorder: A systematic review and meta-analysis. Clin. Psychol. 2011, 31, 1032–1040. [Google Scholar] [CrossRef]
- Janssen, C.W.; Lowry, C.A.; Mehl, M.R.; Allen, J.J.B.; Kelly, K.L.; Gartner, D.E.; Medrano, A.; Begay, T.K.; Rentscher, K.; White, J.J.; et al. Whole-Body Hyperthermia for the Treatment of Major Depressive Disorder: A Randomized Clinical Trial. JAMA Psychiatry 2016, 73, 789–795. [Google Scholar] [CrossRef]
- Eggart, M.; Queri, S.; Müller-Oerlinghausen, B. Are the antidepressive effects of massage therapy mediated by restoration of impaired interoceptive functioning? A novel hypothetical mechanism. Med. Hypotheses 2019, 128, 28–32. [Google Scholar] [CrossRef]
- Khalsa, S.; Rudrauf, D.; Sandesara, C.; Olshansky, B.; Tranel, D. Bolus isoproterenol infusions provide a reliable method for assessing interoceptive awareness. Int. J. Psychophysiol. 2009, 72, 34–45. [Google Scholar] [CrossRef]
- Ewing, D.L.; Manassei, M.; Van Praag, C.G.; Philippides, A.O.; Critchley, H.D.; Garfinkel, S.N. Sleep and the heart: Interoceptive differences linked to poor experiential sleep quality in anxiety and depression. Boil. Psychol. 2017, 127, 163–172. [Google Scholar] [CrossRef]
- Thönes, S.; Oberfeld, D. Time perception in depression: A meta-analysis. J. Affect. Disord. 2015, 175, 359–372. [Google Scholar] [CrossRef]
- Coull, J.T. fMRI studies of temporal attention: Allocating attention within, or towards, time. Cogn. Brain 2004, 21, 216–226. [Google Scholar] [CrossRef]
- Craig, A. (Bud) Emotional moments across time: A possible neural basis for time perception in the anterior insula. Philos. Trans. Soc. B Boil. Sci. 2009, 364, 1933–1942. [Google Scholar] [CrossRef]
- Di Lernia, D.; Serino, S.; Pezzulo, G.; Pedroli, E.; Cipresso, P.; Riva, G. Feel the Time. Time Perception as a Function of Interoceptive Processing. Front. Hum. Neurosci. 2018, 12, 74. [Google Scholar] [CrossRef]
| Study | Diagnostic Criteria | Participants and Comparison Groups (N, % Female, Age: M ± SD) | Exclusion Criteria | Heartbeat Perception Task (Number of Trials) | Main Findings (IAc Scores Resp. Error Rates in % Reported) |
|---|---|---|---|---|---|
| Ehlers and Breuer (1992) [36] | DSM-III-R | MDD or dysthymia (combined N = 16, 47% female, age: 36.80 ± 7.30 years) vs. panic disorder (N = 13, 38% female, age: 41.00 ± 6.80 years) vs. generalized anxiety disorder (N = 15, 44% female, age: 40.50 ± 9.50 years) | Pregnancy, endocrine or cardiovascular disorder, cardiac medication | HMTT (3 trials) | Depressed subjects exhibited significantly higher error for heartbeat perception (43.00%) than subjects with panic (23.00%) or generalized anxiety disorder (21.00%). Group differences remained significant after controlling for medication status. |
| van der Does et al. (1997) [37] | DSM-IV | MDD or dysthymia (combined N = 16, 81% female, age: 40.40 ± 13.00 years) vs. panic disorder (N = 23, 65% female, age: 41.10 ± 10.60 years) vs. healthy controls (N = 21, 62% female, age: 44.50 ± 14.10 years) | Lifetime history of psychosis or substance abuse, comorbidity of panic disorder | HMTT (3 trials) | No significant IAc difference between individuals with mood disorder (42.70%), panic disorder (30.30%), and healthy controls (37.10%). However, a significant lower proportion of accurate heartbeat perceivers was found in the mood disorder group (0.00%) as compared with subjects with panic disorder (30.40%), but no significant differences with regard to healthy controls (9.50%). |
| Mussgay et al. (1999) [38] | ICD-9 | Neurotic depression (N = 141, 87% female, age: 41.10 years ± NA) vs. depressive psychogenic reactions (N = 106, 77% female, age: 42.80 years ± NA) vs. functional disorder (N = 43, 79% female, age: 38.90 years ± NA) vs. functional cardiac disorder (N = 48, 58% female, age: 45.10 years ± NA) vs. personality disorder (N = 26, 69% female, age: 35.40 years ± NA) vs. panic disorder (N = 53, 58% female, age: 41.30 years ± NA) vs. healthy controls (N = 48, 60% female, age: 35.80 years ± NA) | No exclusion criteria (observational study that recruited all inpatients in the sequence of their admission) | HMTT (3 trials) | The groups significantly differed regarding performance in HMTT. There was a post-hoc trend (p = 0.06) towards lower IAc in reactive depression (0.45) as compared with healthy controls (0.56). No IAc score difference between medicated and unmedicated subjects (IAc scores not reported), except for panic disorder: medicated (0.30) vs. unmedicated (0.57). |
| Dunn et al. (2007) [39] | DSM-IV | MDD in-/outpatients (N = 18, 72% female, age: 47.10 ± 9.90 years) vs. moderately depressed community volunteers (N = 18, 72% female, age: 40.10 ± 15.60 years) vs. healthy controls (N = 18, 78% female, age: 44.80 ± 13.00 years) | Comorbidity of panic disorder, no history of brain injury, psychosis, learning disability or substance abuse | HMTT (6 trials) | The moderately depressed community sample exhibited lower heartbeat perception accuracy (39.60%) than healthy controls (29.00%). No significant difference in IAc between subjects with MDD (25.31%) and healthy controls. Significant negative correlation between IAc and depression severity in community volunteers, r = −0.53, p = 0.03. No significant correlation between IAc and depression severity in subjects with MDD. A curvilinear inverted U-shaped relationship was found between depression severity and IAc (error rate) across all participants (R2 = 0.13). Higher IAc in medicated subjects (mainly SSRI) than in unmedicated individuals after collapsing the three samples (adjusted error rates not reported). |
| Terhaar et al. (2012) [40] | DSM-IV | MDD (N = 16, 81% female, age: 21.75 ± 12.40 years) vs. healthy matched controls (N = 16, 75% female, age: 39.81 ± 17.60 years) | Comorbidity of panic disorder | HMTT (3 trials) | Participants with MDD exhibited significantly lower IAc (0.67) than healthy controls (0.81, large effect, Cohen’s d = 0.85). No significant IAc difference between medicated and unmedicated subjects (IAc scores not reported). |
| Furman et al. (2013) [41] | DSM-IV-TR | MDD without anxiety disorder (N = 25, 100% female, age: 38.20 ± 11.90 years) vs. healthy controls (N = 36, 100% female, age: 36.00 ± 12.50 years) | Psychosis, substance abuse or impaired mental status, cardiovascular symptoms | HMTT (3 trials) | IAc was significantly lower in subjects with MDD (0.55) than in healthy controls (0.65). Depression severity and IAc were uncorrelated in MDD as well as in healthy controls. IAc was positively correlated with positive affectivity in MDD. Low IAc was a predictor of indecisiveness, i.e., difficulties in decision making were more severe for inaccurate heartbeat perceivers (0.47) diagnosed with MDD than in subjects without decision-making difficulties (0.67). No significant IAc difference between individuals under psychoactive medication and unmedicated participants (no IAc scores reported). |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eggart, M.; Lange, A.; Binser, M.J.; Queri, S.; Müller-Oerlinghausen, B. Major Depressive Disorder Is Associated with Impaired Interoceptive Accuracy: A Systematic Review. Brain Sci. 2019, 9, 131. https://doi.org/10.3390/brainsci9060131
Eggart M, Lange A, Binser MJ, Queri S, Müller-Oerlinghausen B. Major Depressive Disorder Is Associated with Impaired Interoceptive Accuracy: A Systematic Review. Brain Sciences. 2019; 9(6):131. https://doi.org/10.3390/brainsci9060131
Chicago/Turabian StyleEggart, Michael, Andreas Lange, Martin J. Binser, Silvia Queri, and Bruno Müller-Oerlinghausen. 2019. "Major Depressive Disorder Is Associated with Impaired Interoceptive Accuracy: A Systematic Review" Brain Sciences 9, no. 6: 131. https://doi.org/10.3390/brainsci9060131
APA StyleEggart, M., Lange, A., Binser, M. J., Queri, S., & Müller-Oerlinghausen, B. (2019). Major Depressive Disorder Is Associated with Impaired Interoceptive Accuracy: A Systematic Review. Brain Sciences, 9(6), 131. https://doi.org/10.3390/brainsci9060131




