Four Social Brain Regions, Their Dysfunctions, and Sequelae, Extensively Explain Autism Spectrum Disorder Symptomatology
Abstract
1. Introduction
2. Major Symptoms and Features of ASD
2.1. Sensory Abnormalities
2.2. Motor Abnormalities
2.3. Cognitive Abnormalities
2.4. Emotion Function
2.5. Repetitive Behaviors
2.6. Daily Living Skills Impairments
2.7. Social Impairments
2.8. Summary and Proposed Pathogenic Mechanism
3. Neurocircuitry: The Four Social Brain Regions are Commonly Disordered in ASD
3.1. The Amygdala is Disordered in ASD
3.2. OFC is Disordered in ASD
3.3. TPC is Disordered in ASD
3.4. Insula is Disordered in ASD
3.5. Summary
4. Symptomatology: The Four Social Brain Regions’ Multiple Functions, and Their Direct Contributions to ASD Symptoms and Features
4.1. Amygdala Disruption Likely Underlies Specific ASD Symptoms
4.2. OFC Disruption Likely Underlies Specific ASD Symptoms
4.3. TPC Disruption Likely Underlies Specific ASD Symptoms
4.4. Insula Disruption Likely Underlies Specific ASD Symptoms
5. Sequelae
5.1. Cognitive Abnormalities
5.2. Daily Living Skills Deficits
5.3. Emotional Abnormalities
5.4. Additional Brain Regions Display Likely Secondary Structural and Functional Atypicalities in ASD
5.4.1. Visual cortex
5.4.2. IFG
5.4.3. Caudate Nucleus
5.4.4. Hippocampus
5.5. Heterogeneity
6. Summary, Cautions, and Causation
6.1. Summary
6.2. Cautions
6.3. Causation
7. Testable Predictions and Directions for Future Research
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Quirke, S.; Hagen, A.; Fombonne, E.; Geschwind, D.; Dawson, G. Epidemiology of Pervasive Developmental Disorders. In Autism Spectrum Disorders; Oxford University Press (OUP): New York, NY, USA, 2011; pp. 90–111. [Google Scholar]
- Modabbernia, A.; Velthorst, E.; Reichenberg, A. Environmental risk factors for autism: An evidence-based review of systematic reviews and meta-analyses. Mol. Autism 2017, 8, 13. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, D.A.; Genuis, S.J.; Frye, R.E. Environmental toxicants and autism spectrum disorders: A systematic review. Transl. Psychiatry 2014, 4, e360. [Google Scholar] [CrossRef] [PubMed]
- Weisskopf, M.G.; Kioumourtzoglou, M.-A.; Roberts, A.L. Air Pollution and Autism Spectrum Disorders: Causal or Confounded? Environ. Heal. Rep. 2015, 2, 430–439. [Google Scholar] [CrossRef] [PubMed]
- Baron-Cohen, S.; Scott, F.J.; Allison, C.; Williams, J.; Bolton, P.; Matthews, F.E.; Brayne, C. Prevalence of autism-spectrum conditions: UK school-based population study. Br. J. Psychiatry 2009, 194, 500–509. [Google Scholar] [CrossRef] [PubMed]
- Centres for Disease Control and Prevention. Prevalence and characteristics of autism spectrum disorder among children aged 8 years—Autism and developmental disabilities monitoring network, 11 sites, United States, 2012. MMWR Surveill Summ. 2016, 65, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Strathearn, L.; Liu, B.; O’Brien, M.; Kopelman, T.G.; Zhu, J.; Snetselaar, L.G.; Bao, W. Prevalence and treatment patterns of autism spectrum disorder in the United States, 2016. JAMA Pediatr. 2019, 173, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Schumann, C.M.; Noctor, S.C.; Amaral, D.G. Neuropathology of Autism Spectrum Disorders: Postmortem Studies. In Autism Spectrum Disorders; Oxford University Press (OUP): New York, NY, USA, 2011; pp. 539–565. [Google Scholar]
- Groen, W.B.; Buitelaar, J.K.; Geschwind, D.; Dawson, G. Cognitive and Neural Correlates of Language in Autism. In Autism Spectrum Disorders; Oxford University Press (OUP): New York, NY, USA, 2011; pp. 186–199. [Google Scholar]
- Helen, T.-F.; Lisa, E.; Rhiannon, L.; Geschwind, D.; Dawson, G. Language and Communication in Autism Spectrum Disorders. In Autism Spectrum Disorders; Oxford University Press (OUP): New York, NY, USA, 2011; pp. 172–185. [Google Scholar]
- Dykens, E.M.; Lense, M.; Geschwind, D.; Dawson, G. Intellectual Disabilities and Autism Spectrum Disorder: A Cautionary Note. In Autism Spectrum Disorders; Oxford University Press (OUP): New York, NY, USA, 2011; pp. 263–269. [Google Scholar]
- Howlin, P.; Goode, S.; Hutton, J.; Rutter, M. Savant skills in autism: Psychometric approaches and parental reports. Philos. Trans. Soc. B Boil. Sci. 2009, 364, 1359–1367. [Google Scholar] [CrossRef]
- Kanne, S.M.; Mazurek, M.O. Aggression in children and adolescents with ASD: Prevalence and risk factors. J. Autism Dev. Disord. 2011, 41, 926–937. [Google Scholar] [CrossRef]
- Billstedt, E.; Gillberg, C.; Gillberg, C. Autism after Adolescence: Population-based 13- to 22-year Follow-up Study of 120 Individuals with Autism Diagnosed in Childhood. J. Autism Dev. Disord. 2005, 35, 351–360. [Google Scholar] [CrossRef]
- Bryson, S.E.; Zwaigenbaum, L.; Brian, J.; Roberts, W.; Szatmari, P.; Rombough, V.; McDermott, C. A Prospective Case Series of High-risk Infants who Developed Autism. J. Autism Dev. Disord. 2007, 37, 12–24. [Google Scholar] [CrossRef]
- Duncan, A.W.; Bishop, S.L. Understanding the gap between cognitive abilities and daily living skills in adolescents with autism spectrum disorders with average intelligence. Autism 2015, 19, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Elison, J.T.; Wolff, J.J.; Reznick, J.S.; Botteron, K.N.; Estes, A.M.; Gu, H.; Hazlett, H.C.; Meadows, A.J.; Paterson, S.J.; Zwaigenbaum, L.; et al. Repetitive Behavior in 12-Month-Olds Later Classified With Autism Spectrum Disorder. J. Am. Acad. Child Adolesc. Psychiatry 2014, 53, 1216–1224. [Google Scholar] [CrossRef] [PubMed]
- Estes, A.; Network, I.; Zwaigenbaum, L.; Gu, H.; John, T.S.; Paterson, S.; Elison, J.T.; Hazlett, H.; Botteron, K.; Dager, S.R.; et al. Behavioral, cognitive, and adaptive development in infants with autism spectrum disorder in the first 2 years of life. J. Neurodev. Disord. 2015, 7, 24. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, J.E.; Landa, R.; Bhat, A.; Bauman, M. Head Lag in Infants at Risk for Autism: A Preliminary Study. Am. J. Occup. Ther. 2012, 66, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Gliga, T.; Bedford, R.; Charman, T.; Johnson, M.H.; Baron-Cohen, S.; Bolton, P.; Cheung, C.; Davies, K.; Liew, M.; Fernandes, J.; et al. Enhanced Visual Search in Infancy Predicts Emerging Autism Symptoms. Lesions Head Direction Cell Syst. Increase Hippo. Place Field Repetit. 2015, 25, 1727–1730. [Google Scholar] [CrossRef] [PubMed]
- Green, D.; Charman, T.; Pickles, A.; Chandler, S.; Loucas, T.; Simonoff, E.; Baird, G. Impairment in movement skills of children with autistic spectrum disorders. Dev. Med. Child Neurol. 2009, 51, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Green, J.; Gilchrist, A.; Burton, D.; Cox, A. Social and Psychiatric Functioning in Adolescents with Asperger Syndrome Compared with Conduct Disorder. J. Autism Dev. Disord. 2000, 30, 279–293. [Google Scholar] [CrossRef] [PubMed]
- Howlin, P.; Magiati, I. Autism spectrum disorder: Outcomes in adulthood. Curr. Opin. Psychiatry 2017, 30, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Leonard, H.C.; Bedford, R.; Charman, T.; Elsabbagh, M.; Johnson, M.H.; Hill, E.L. Motor development in children at risk of autism: A follow-up study of infant siblings. Autism 2014, 18, 281–291. [Google Scholar] [CrossRef]
- Riquelme, I.; Hatem, S.M.; Montoya, P. Abnormal Pressure Pain, Touch Sensitivity, Proprioception, and Manual Dexterity in Children with Autism Spectrum Disorders. Neural Plast. 2016, 2016, 1–9. [Google Scholar] [CrossRef]
- Rogers, S.J. What are infant siblings teaching us about autism in infancy? Autism Res. 2009, 2, 125–137. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.L.; Minshew, N.J.; Goldstein, G. Further understanding of complex information processing in verbal adolescents and adults with autism spectrum disorders. Autism 2015, 19, 859–867. [Google Scholar] [CrossRef] [PubMed]
- Cadman, T.; Eklund, H.; Howley, D.; Hayward, H.; Clarke, H.; Findon, J.; Xenitidis, K.; Murphy, D.; Asherson, P.; Glaser, K. Caregiver Burden as People With Autism Spectrum Disorder and Attention-Deficit/Hyperactivity Disorder Transition into Adolescence and Adulthood in the United Kingdom. J. Am. Acad. Child Adolesc. Psychiatry 2012, 51, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Fairthorne, J.; Hammond, G.; Bourke, J.; Jacoby, P.; Leonard, H. Early mortality and primary causes of death in mothers of children with intellectual disability or autism spectrum disorder: A retrospective cohort study. PLoS ONE 2014, 9, e113430. [Google Scholar] [CrossRef] [PubMed]
- Gabriels, R.L.; Geschwind, D.; Dawson, G. Adolescent Transition to Adulthood and Vocational Issues. In Autism Spectrum Disorders; Oxford University Press (OUP): New York, NY, USA, 2011; pp. 1167–1181. [Google Scholar]
- Seltzer, M.M.; Floyd, F.; Greenberg, J.; Orsmond, G.; Bolt, D.; Hartley, S.L.; Barker, E.T. The relative risk and timing of divorce in families of children with an autism spectrum disorder. J. Fam. Psychol. 2010, 24, 449–457. [Google Scholar]
- Moore, A. Letter: Poorest Struggle to Get SEN Diagnosis; The Guardian: London, UK, 2017. [Google Scholar]
- Moreno, S. A Parent’s View of More Able People with Autism. In High-Functioning Individuals with Autism; Springer Nature: Berlin, Germany, 1992; pp. 91–103. [Google Scholar]
- Pickles, A.; Le Couteur, A.; Leadbitter, K.; Salomone, E.; Cole-Fletcher, R.; Tobin, H.; Gammer, I.; Lowry, J.; Vamvakas, G.; Byford, S.; et al. Parent-mediated social communication therapy for young children with autism (PACT): Long-term follow-up of a randomised controlled trial. Lancet 2016, 388, 2501–2509. [Google Scholar] [CrossRef]
- Peters-Scheffer, N.; Didden, R.; Korzilius, H.; Matson, J. Cost comparison of early intensive behavioral intervention and treatment as usual for children with autism spectrum disorder in the Netherlands. Dev. Disabil. 2012, 33, 1763–1772. [Google Scholar] [CrossRef] [PubMed]
- Constantino, J.N.; Gruber, C.P.; Davis, S.; Hayes, S.; Passanante, N.; Przybeck, T. The factor structure of autistic traits. J. Child Psychol. Psychiatry 2004, 45, 719–726. [Google Scholar] [CrossRef]
- Ring, H.; Woodbury-Smith, M.; Watson, P.; Wheelwright, S.; Baron-Cohen, S. Clinical heterogeneity among people with high functioning autism spectrum conditions: Evidence favouring a continuous severity gradient. Behav. Brain Funct. 2008, 4, 11. [Google Scholar] [CrossRef] [PubMed]
- Spiker, D.; Lotspeich, L.J.; DiMiceli, S.; Myers, R.M.; Risch, N. Behavioral phenotypic variation in autism multiplex families: Evidence for a continuous severity gradient. Am. J. Med Genet. 2002, 114, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Bachevalier, J. Medial temporal lobe structures and autism: A review of clinical and experimental findings. Neuropsychologia 1994, 32, 627–648. [Google Scholar] [CrossRef]
- Bachevalier, J.; Loveland, K.A. The orbitofrontal–amygdala circuit and self-regulation of social–emotional behavior in autism. Neurosci. Biobehav. Rev. 2006, 30, 97–117. [Google Scholar] [CrossRef] [PubMed]
- Baron-Cohen, S.; Ring, H.; Bullmore, E.; Wheelwright, S.; Ashwin, C.; Williams, S.; Bullmore, E. The amygdala theory of autism. Neurosci. Biobehav. Rev. 2000, 24, 355–364. [Google Scholar] [CrossRef]
- McPartland, J.C.; Coffman, M.; Pelphrey, K.A. Recent Advances in Understanding the Neural Bases of Autism Spectrum Disorder. Curr. Opin. Pediatr. 2011, 23, 628–632. [Google Scholar] [CrossRef] [PubMed]
- Pelphrey, K.A.; Shultz, S.; Hudac, C.M.; Wyk, B.C.V. Constraining Heterogeneity: The Social Brain and its Development in Autism Spectrum Disorder. J. Child Psychol. Psychiatry 2011, 52, 631–644. [Google Scholar] [CrossRef] [PubMed]
- Schultz, R.T.; Chawarska, K.; Volkmar, F.R. The social brain in autism: Perspectives from neuropsychology and neuroimaging. In Understanding Autism: From Basic Neuroscience to Treatment; Moldin, S.O., Rubenstein, J.L.R., Eds.; Taylor & Francis: Boca Raton, FL, USA, 2006; pp. 323–348. [Google Scholar]
- Nickl-Jockschat, T.; Habel, U.; Michel, T.M.; Manning, J.; Laird, A.R.; Fox, P.T.; Schneider, F.; Eickhoff, S.B. Brain structure anomalies in autism spectrum disorder—A meta-analysis of VBM studies using anatomic likelihood estimation. Hum. Brain Mapp. 2012, 33, 1470–1489. [Google Scholar] [CrossRef] [PubMed]
- Patriquin, M.A.; DeRamus, T.; Libero, L.E.; Laird, A.; Kana, R.K. Neuroanatomical and Neurofunctional Markers of Social Cognition in Autism Spectrum Disorder. Hum. Brain Mapp. 2016, 37, 3957–3978. [Google Scholar] [CrossRef] [PubMed]
- Stanfield, A.C.; McIntosh, A.M.; Spencer, M.D.; Philip, R.; Gaur, S.; Lawrie, S.M. Towards a neuroanatomy of autism: A systematic review and meta-analysis of structural magnetic resonance imaging studies. Eur. Psychiatry 2008, 23, 289–299. [Google Scholar] [CrossRef]
- Via, E.; Radua, J.; Cardoner, N.; Happé, F.; Mataix-Cols, D. Meta-analysis of Gray Matter Abnormalities in Autism Spectrum Disorder. Arch. Psychiatry 2011, 68, 409. [Google Scholar] [CrossRef]
- Wegiel, J.; Flory, M.; Kuchna, I.; Nowicki, K.; Ma, S.Y.; Imaki, H.; Wegiel, J.; Cohen, I.L.; London, E.; Wisniewski, T.; et al. Stereological study of the neuronal number and volume of 38 brain subdivisions of subjects diagnosed with autism reveals significant alterations restricted to the striatum, amygdala and cerebellum. Acta Neuropathol. Commun. 2014, 2, 141. [Google Scholar] [CrossRef]
- Fornito, A.; Zalesky, A.; Breakspear, M. The connectomics of brain disorders. Nat. Rev. Neurosci. 2015, 16, 159–172. [Google Scholar] [CrossRef] [PubMed]
- Duering, M.; Schmidt, R. Remote changes after ischaemic infarcts: A distant target for therapy? Brain 2017, 140, 1818–1820. [Google Scholar] [CrossRef] [PubMed]
- Kuchcinski, G.; Munsch, F.; Lopes, R.; Bigourdan, A.; Su, J.; Sagnier, S.; Renou, P.; Pruvo, J.-P.; Rutt, B.K.; Dousset, V.; et al. Thalamic alterations remote to infarct appear as focal iron accumulation and impact clinical outcome. Brain 2017, 140, 1932–1946. [Google Scholar] [CrossRef] [PubMed]
- Wing, L. Diagnosis, Clinical Description and Prognosis. In Early Childhood Autism: Clinical, Educational and Social Aspects, 2nd ed.; Wing, L., Ed.; Pergamon Press: Oxford, UK, 1976; pp. 15–64. [Google Scholar]
- Insel, T.; Cuthbert, B.; Garvey, M.; Heinssen, R.; Pine, D.S.; Quinn, K.; Sanislow, C.; Wang, P. Research Domain Criteria (RDoC): Toward a New Classification Framework for Research on Mental Disorders. Am. J. Psychiatry 2010, 167, 748–751. [Google Scholar] [CrossRef] [PubMed]
- Insel, T.R.; Cuthbert, B.N. Brain disorders? Precisely. Science 2015, 348, 499–500. [Google Scholar] [CrossRef] [PubMed]
- National Institute of Mental Health. RDoC matrix (version 3) 2017. Available online: https://www.nimh.nih.gov/research-priorities/rdoc/constructs/rdoc-matrix.shtml (accessed on 3 June 2018).
- Amaral, D.G.; Dawson, G.; Geschwind, D.H. (Eds.) Autism Spectrum Disorders; Oxford University Press: New York, NY, USA, 2011. [Google Scholar]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Moldin, S.O.; Rubenstein, J.L.R. (Eds.) Understanding Autism: From Basic Neuroscience to Treatment; Taylor & Francis: Boca Raton, FL, USA, 2006. [Google Scholar]
- Roth, I.; Rezaie, P. (Eds.) Researching the Autism Spectrum: Contemporary Perspectives; Cambridge University Press: Cambridge, UK, 2011. [Google Scholar]
- Jolliffe, T.; Lansdown, R.; Robinson, C. Autism: A personal account. Communication 1992, 26, 12–19. [Google Scholar]
- Grandin, T. A personal perspective of autism. In Handbook of Autism & Pervasive Developmental Disorders, 3rd ed.; Volkmar, F.R., Paul, R., Klin, A., Cohen, D., Eds.; John Wiley & Sons: New York, NY, USA, 2005; pp. 1276–1286. [Google Scholar]
- Grandin, T.; Geschwind, D.; Dawson, G. Top Priorities for Autism/Asperger’s Research: Perspectives from a Person with Autism. In Autism Spectrum Disorders; Oxford University Press (OUP): New York, NY, USA, 2011; pp. 1377–1385. [Google Scholar]
- Grinker, R.R. Unstrange Minds; Icon Books: London, UK, 2007. [Google Scholar]
- Williams, D. Autism: An Inside-Out Approach; Jessica Kingsley Publishers: London, UK, 1996. [Google Scholar]
- Simmons, D.R.; Robertson, A.E.; McKay, L.S.; Toal, E.; McAleer, P.; Pollick, F.E. Vision in autism spectrum disorders. Vis. Res. 2009, 49, 2705–2739. [Google Scholar] [CrossRef] [PubMed]
- Hickey, C.; Chelazzi, L.; Theeuwes, J. Reward-Priming of Location in Visual Search. PLoS ONE 2014, 9, e103372. [Google Scholar] [CrossRef]
- Klin, A.; Jones, W.; Schultz, R.; Volkmar, F. The enactive mind, or from actions to cognition: Lessons from autism. Philos. Trans. Soc. B Boil. Sci. 2003, 358, 345–360. [Google Scholar] [CrossRef]
- Nakano, T.; Tanaka, K.; Endo, Y.; Yamane, Y.; Yamamoto, T.; Nakano, Y.; Ohta, H.; Kato, N.; Kitazawa, S. Atypical gaze patterns in children and adults with autism spectrum disorders dissociated from developmental changes in gaze behaviour. Proc. Soc. B Boil. Sci. 2010, 277, 2935–2943. [Google Scholar] [CrossRef]
- Niu, Y.; Todd, R.M.; Anderson, A.K. Affective Salience Can Reverse the Effects of Stimulus-Driven Salience on Eye Movements in Complex Scenes. Front. Psychol. 2012, 3, 336. [Google Scholar] [CrossRef] [PubMed]
- Towal, R.B.; Mormann, M.; Koch, C. Simultaneous modeling of visual saliency and value computation improves predictions of economic choice. Proc. Acad. Sci. 2013, 110, E3858–E3867. [Google Scholar] [CrossRef] [PubMed]
- Vuilleumier, P. Affective and motivational control of vision. Curr. Opin. Neurol. 2015, 28, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Schomaker, J.; Walper, D.; Wittmann, B.C.; Einhäuser, W. Attention in natural scenes: Affective-motivational factors guide gaze independently of visual salience. Vis. Res. 2017, 133, 161–175. [Google Scholar] [CrossRef] [PubMed]
- Klin, A.; Lin, D.J.; Gorrindo, P.; Ramsay, G.; Jones, W. Two-year-olds with autism orient to non-social contingencies rather than biological motion. Nature 2009, 459, 257–261. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Jiang, M.; Duchesne, X.M.; Laugeson, E.A.; Kennedy, D.P.; Adolphs, R.; Zhao, Q. Atypical visual saliency in autism spectrum disorder quantified through model-based eye tracking. Neuron 2015, 88, 604–616. [Google Scholar] [CrossRef] [PubMed]
- Frazier, T.W.; Strauss, M.; Klingemier, E.W.; Zetzer, E.E.; Hardan, A.Y.; Eng, C.; Youngstrom, E.A. A Meta-Analysis of Gaze Differences to Social and Nonsocial Information between Individuals with and Without Autism. J. Am. Acad. Child Adolesc. Psychiatry 2017, 56, 546–555. [Google Scholar] [CrossRef] [PubMed]
- Klin, A.; Chawarska, K.; Paul, R.; Rubin, E.; Morgan, T.; Wiesner, L.; Volkmar, F. Autism in a 15-Month-Old Child. Am. J. Psychiatry 2004, 161, 1981–1988. [Google Scholar] [CrossRef]
- Plaisted, K.C. Reduced generalization in autism: An alternative to weak central coherence. In The Development of Autism: Perspectives from Theory and Research; Burack, J.A., Charman, T., Yirmiya, N., Zelazo, P.R., Eds.; Lawrence Erlbaum Associates: Mahwah, NJ, USA, 2001; pp. 149–169. [Google Scholar]
- Tavassoli, T.; Miller, L.J.; Schoen, S.A.; Nielsen, D.M.; Baron-Cohen, S. Sensory over-responsivity in adults with autism spectrum conditions. Autism 2014, 18, 428–432. [Google Scholar] [CrossRef]
- Kanner, L. Autistic disturbances of affective contact. Nervous Child 1943, 2, 217–250. [Google Scholar]
- Volkmar, F.R.; Cohen, D.J.; Paul, R. An Evaluation of DSM-III Criteria for Infantile Autism. J. Am. Acad. Child Psychiatry 1986, 25, 190–197. [Google Scholar] [CrossRef]
- Jemel, B.; Mottron, L.; Dawson, M. Impaired Face Processing in Autism: Fact or Artifact? J. Autism Dev. Disord. 2006, 36, 91–106. [Google Scholar] [CrossRef] [PubMed]
- Neil, L.; Cappagli, G.; Karaminis, T.; Jenkins, R.; Pellicano, E. Recognizing the same face in different contexts: Testing within-person face recognition in typical development and in autism. J. Exp. Child Psychol. 2016, 143, 139–153. [Google Scholar] [CrossRef] [PubMed]
- Weigelt, S.; Koldewyn, K.; Kanwisher, N. Face identity recognition in autism spectrum disorders: A review of behavioral studies. Neurosci. Biobehav. Rev. 2012, 36, 1060–1084. [Google Scholar] [CrossRef] [PubMed]
- Grelotti, D.J.; Klin, A.J.; Gauthier, I.; Skudlarski, P.; Cohen, D.J.; Gore, J.C.; Volkmar, F.R.; Schultz, R.T. fMRI activation of the fusiform gyrus and amygdala to cartoon characters but not to faces in a boy with autism. Neuropsychologia 2005, 43, 373–385. [Google Scholar] [CrossRef] [PubMed]
- Hadjikhani, N.; Chabris, C.F.; Joseph, R.M.; Clark, J.; McGrath, L.; Aharon, I.; Feczko, E.; Tager-Flusberg, H.; Harris, G.J. Early visual cortex organization in autism: An fMRI study. NeuroReport 2004, 15, 267–270. [Google Scholar] [CrossRef]
- Morris, J.P.; Pelphrey, K.A.; McCarthy, G. Controlled scanpath variation alters fusiform face activation. Soc. Cogn. Affect Neurosci. 2006, 2, 31–38. [Google Scholar] [CrossRef]
- Nickl-Jockschat, T.; Rottschy, C.; Thommes, J.; Schneider, F.; Laird, A.R.; Fox, P.T.; Eickhoff, S.B. Neural networks related to dysfunctional face processing in autism spectrum disorder. Brain Struct Funct. 2015, 220, 2355–2371. [Google Scholar] [CrossRef]
- Perlman, S.B.; Hudac, C.M.; Pegors, T.; Minshew, N.J.; Pelphrey, K.A. Experimental manipulation of face-evoked activity in the fusiform gyrus of individuals with autism. Soc. Neurosci. 2011, 6, 22–30. [Google Scholar] [CrossRef]
- Pierce, K.; Redcay, E. Fusiform Function in Children with an Autism Spectrum Disorder Is a Matter of “Who”. Boil. Psychiatry 2008, 64, 552–560. [Google Scholar] [CrossRef]
- Samson, F.; Mottron, L.; Soulières, I.; Zeffiro, T.A. Enhanced visual functioning in autism: An ALE meta-analysis. Hum. Brain Mapp. 2012, 33, 1553–1581. [Google Scholar] [CrossRef] [PubMed]
- Iuculano, T.; Rosenberg-Lee, M.; Supekar, K.; Lynch, C.J.; Khouzam, A.; Phillips, J.; Uddin, L.Q.; Menon, V. Brain organization underlying superior mathematical abilities in children with autism. Biol. Psychiatry 2014, 75, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Rosenhall, U.; Nordin, V.; Sandström, M.; Ahlsén, G.; Gillberg, C. Autism and hearing loss. J. Autism Dev. Disord. 1999, 29, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Tas, A.; Yagiz, R.; Tas, M.; Esme, M.; Uzun, C.; Karasalihoglu, A.R. Evaluation of hearing in children with autism by using TEOAE and ABR. Autism 2007, 11, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Tharpe, A.M.; Bess, F.H.; Sladen, D.P.; Schissel, H.; Couch, S.; Schery, T. Auditory Characteristics of Children with Autism. Ear Hear. 2006, 27, 430–441. [Google Scholar] [CrossRef] [PubMed]
- Klin, A. Young autistic children’s listening preferences in regard to speech: A possible characterization of the symptom of social withdrawal. J. Autism Dev. Disord. 1991, 21, 29–42. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.; Iosif, A.-M.; Hill, M.; Young, G.S.; Schwichtenberg, A.J.; Ozonoff, S. Response to Name in Infants Developing Autism Spectrum Disorder: A Prospective Study. J. Pediatr. 2017, 183, 141–146. [Google Scholar] [CrossRef]
- Nadig, A.S.; Ozonoff, S.; Young, G.S.; Rozga, A.; Sigman, M.; Rogers, S.J. A Prospective Study of Response to Name in Infants at Risk for Autism. Arch. Pediatr. Adolesc. Med. 2007, 161, 378–383. [Google Scholar] [CrossRef]
- Čeponienė, R.; Lepistö, T.; Shestakova, A.; Vanhala, R.; Alku, P.; Näätänen, R.; Yaguchi, K. Speech–sound-selective auditory impairment in children with autism: They can perceive but do not attend. Proc. Acad. Sci. 2003, 100, 5567–5572. [Google Scholar] [CrossRef]
- Gervais, H.; Belin, P.; Boddaert, N.; Leboyer, M.; Coez, A.; Sfaello, I.; Barthélémy, C.; Brunelle, F.; Samson, Y.; Zilbovicius, M. Abnormal cortical voice processing in autism. Nat. Neurosci. 2004, 7, 801–802. [Google Scholar] [CrossRef]
- Lai, G.; Pantazatos, S.P.; Schneider, H.; Hirsch, J. Neural systems for speech and song in autism. Brain 2012, 135, 961–975. [Google Scholar] [CrossRef] [PubMed]
- Lepistö, T.; Kujala, T.; Vanhala, R.; Alku, P.; Huotilainen, M.; Näätänen, R. The discrimination of and orienting to speech and non-speech sounds in children with autism. Brain Res. 2005, 1066, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Whitehouse, A.J.; Bishop, D.V. Do children with autism ‘switch off’ to speech sounds? An investigation using event-related potentials. Dev. Sci. 2008, 11, 516–524. [Google Scholar] [CrossRef] [PubMed]
- Elwin, M.; Ek, L.; Schröder, A.; Kjellin, L. Autobiographical Accounts of Sensing in Asperger Syndrome and High-Functioning Autism. Arch. Psychiatr. Nurs. 2012, 26, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Failla, M.D.; Moana-Filho, E.J.; Essick, G.K.; Baranek, G.T.; Rogers, B.P.; Cascio, C.J. Initially intact neural responses to pain in autism are diminished during sustained pain. Autism 2018, 22, 669–683. [Google Scholar] [CrossRef] [PubMed]
- Kientz, M.A.; Dunn, W. A Comparison of the Performance of Children with and without Autism on the Sensory Profile. Am. J. Occup. Ther. 1997, 51, 530–537. [Google Scholar] [CrossRef] [PubMed]
- Charrier, A.; Kermarrec, S.; Canitano, R.; Antoine, C.; Brailly-Tabard, S.; Cohen, D.; Haidar, H.; Trabado, S.; Carlier, M.; Mottron, L.; et al. Relationships Between Self-Injurious Behaviors, Pain Reactivity, and β-Endorphin in Children and Adolescents With Autism. J. Clin. Psychiatry 2018, 79. [Google Scholar] [CrossRef]
- Billstedt, E.; Gillberg, I.C.; Gillberg, C. Autism in adults: Symptom patterns and early childhood predictors. Use of the DISCO in a community sample followed from childhood. J. Child Psychol. Psychiatry 2007, 48, 1102–1110. [Google Scholar] [CrossRef]
- Duerden, E.G.; Taylor, M.J.; Lee, M.; McGrath, P.A.; Davis, K.D.; Roberts, S.W. Decreased Sensitivity to Thermal Stimuli in Adolescents With Autism Spectrum Disorder: Relation to Symptomatology and Cognitive Ability. J. Pain 2015, 16, 463–471. [Google Scholar] [CrossRef]
- Mikkelsen, M.; Wodka, E.L.; Mostofsky, S.H.; Puts, N.A.J. Autism spectrum disorder in the scope of tactile processing. Dev. Cogn. Neurosci. 2018, 29, 140–150. [Google Scholar] [CrossRef]
- Dalrymple, N.J.; Ruble, L.A. Toilet training and behaviors of people with autism: Parent views. J. Autism Dev. Disord. 1992, 22, 265–275. [Google Scholar] [CrossRef] [PubMed]
- Fiene, L.; Brownlow, C. Investigating interoception and body awareness in adults with and without autism spectrum disorder. Autism Res. 2015, 8, 709–716. [Google Scholar] [CrossRef] [PubMed]
- Reber, A.S.; Allen, R.; Reber, E.S. Penguin Dictionary of Psychology; Penguin: London, UK, 2009. [Google Scholar]
- Teitelbaum, P.; Teitelbaum, O.; Nye, J.; Fryman, J.; Maurer, R.G. Movement analysis in infancy may be useful for early diagnosis of autism. Proc. Acad. Sci. 1998, 95, 13982–13987. [Google Scholar] [CrossRef] [PubMed]
- Teitelbaum, O.; Benton, T.; Shah, P.K.; Prince, A.; Kelly, J.L.; Teitelbaum, P. Eshkol–Wachman movement notation in diagnosis: The early detection of Asperger’s syndrome. Proc. Acad. Sci. 2004, 101, 11909–11914. [Google Scholar] [CrossRef] [PubMed]
- Weimer, A.K.; Schatz, A.M.; Lincoln, A.; Ballantyne, A.O.; Trauner, D.A. “Motor” impairment in Asperger syndrome: Evidence for a deficit in proprioception. J. Dev. Behav. Pediatr. 2001, 22, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, C.T.; Mostofsky, S.H.; Bastian, A.J. No proprioceptive deficits in autism despite movement-related sensory and execution impairments. J. Autism Dev. Disord. 2011, 41, 1352–1361. [Google Scholar] [CrossRef] [PubMed]
- Germani, T.; Zwaigenbaum, L.; Sacrey, L.-A.R.; Bryson, S.E. Reaching and grasping in autism spectrum disorder: A review of recent literature. Front. Neurol. 2014, 5, 6. [Google Scholar]
- Doumas, M.; McKenna, R.; Murphy, B. Postural control deficits in autism spectrum disorder: The role of sensory integration. J. Autism Dev. Disord. 2016, 46, 853–861. [Google Scholar] [CrossRef]
- Molloy, C.A.; Dietrich, K.N.; Bhattacharya, A. Postural Stability in Children with Autism Spectrum Disorder. J. Autism Dev. Disord. 2003, 33, 643–652. [Google Scholar] [CrossRef]
- Shetreat-Klein, M.; Shinnar, S.; Rapin, I. Abnormalities of joint mobility and gait in children with autism spectrum disorders. Brain Dev. 2014, 36, 91–96. [Google Scholar] [CrossRef]
- Linkenauger, S.A.; Lerner, M.D.; Ramenzoni, V.C.; Proffitt, D.R. A Perceptual-Motor Deficit Predicts Social and Communicative Impairments in Individuals With Autism Spectrum Disorders. Autism Res. 2012, 5, 352–362. [Google Scholar] [CrossRef]
- Pokorny, J.J.; Hatt, N.V.; Colombi, C.; Vivanti, G.; Rogers, S.J.; Rivera, S.M. The Action Observation System when Observing Hand Actions in Autism and Typical Development. Autism Res. 2015, 8, 284–296. [Google Scholar] [CrossRef]
- Velikonja, T.; Fett, A.-K.; Velthorst, E. Patterns of non-social and social cognitive functioning in adults with autism spectrum disorder. JAMA Psychiatry 2019, 76, 135–151. [Google Scholar] [CrossRef] [PubMed]
- Heider, F.; Simmel, M. An Experimental Study of Apparent Behavior. Am. J. Psychol. 1944, 57, 243. [Google Scholar] [CrossRef]
- Abell, F.; Happé, F.; Frith, U. Do triangles play tricks? Attribution of mental states to animated shapes in normal and abnormal development. Cogn. Dev. 2000, 15, 1–16. [Google Scholar] [CrossRef]
- Klin, A. Attributing Social Meaning to Ambiguous Visual Stimuli in Higher-functioning Autism and Asperger Syndrome: The Social Attribution Task. J. Child Psychol. Psychiatry 2000, 41, 831–846. [Google Scholar] [CrossRef] [PubMed]
- Klin, A.; Jones, W. Attributing social and physical meaning to ambiguous visual displays in individuals with higher-functioning autism spectrum disorders. Brain Cogn. 2006, 61, 40–53. [Google Scholar] [CrossRef] [PubMed]
- Salter, G.; Seigal, A.; Claxton, M.; Lawrence, K.; Skuse, D. Can autistic children read the mind of an animated triangle? Autism 2008, 12, 349–371. [Google Scholar] [CrossRef]
- Grandin, T. How does visual thinking work in the mind of a person with autism? A personal account. Philos. Trans. Soc. B Boil. Sci. 2009, 364, 1437–1442. [Google Scholar] [CrossRef]
- Jones, E.J.H.; Webb, S.J.; Estes, A.; Dawson, G. Rule Learning in Autism: The Role of Reward Type and Social Context. Dev. Neuropsychol. 2013, 38, 58–77. [Google Scholar] [CrossRef]
- Klinger, L.G.; Dawson, G. Prototype formation in autism. Dev. Psychopathol. 2001, 13, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Kuschner, E.S.; Bennetto, L.; Yost, K. Patterns of nonverbal cognitive functioning in young children with autism spectrum disorders. J. Autism Dev. Disord. 2007, 37, 795–807. [Google Scholar] [CrossRef] [PubMed]
- Minshew, N.J.; Meyer, J.; Goldstein, G. Abstract reasoning in autism: A disassociation between concept formation and concept identification. Neuropsychology 2002, 16, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Ropar, D.; Peebles, D. Sorting preference in children with autism: The dominance of concrete features. J. Autism Dev. Disord. 2007, 37, 270–280. [Google Scholar] [CrossRef]
- Anthony, L.G.; Kenworthy, L.; Yerys, B.E.; Jankowski, K.F.; James, J.D.; Harms, M.B.; Martin, A.; Wallace, G.L. Interests in high-functioning autism are more intense, interfering, and idiosyncratic, but not more circumscribed, than those in neurotypical development. Dev. Psychopathol. 2013, 25, 643–652. [Google Scholar] [CrossRef] [PubMed]
- Baron-Cohen, S.; Wheelwright, S. ‘Obsessions’ in children with autism or Asperger syndrome. Br. J. Psychiatry 1999, 175, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Klin, A.; Danovitch, J.H.; Merz, A.B.; Volkmar, F.R. Circumscribed Interests in Higher Functioning Individuals with Autism Spectrum Disorders: An Exploratory Study. Pr. Pers. Sev. Disabil. 2007, 32, 89–100. [Google Scholar] [CrossRef]
- Treffert, D.A. Savant syndrome: Realities, myths and misconceptions. J. Autism Dev. Disord. 2014, 44, 564–571. [Google Scholar] [CrossRef]
- Brown, L.M.T.-; Lam, K.S.L.; Holtzclaw, T.N.; Dichter, G.S.; Bodfish, J.W. Phenomenology and measurement of circumscribed interests in autism spectrum disorders. Autism 2011, 15, 437–456. [Google Scholar] [CrossRef]
- Pierce, K.; Conant, D.; Hazin, R.; Stoner, R.; Desmond, J. Preference for geometric patterns early in life as a risk factor for autism. Arch. Gen. Psychiatry 2011, 68, 101–109. [Google Scholar] [CrossRef]
- Pierce, K.; Marinero, S.; Hazin, R.; McKenna, B.; Barnes, C.C.; Malige, A. Eye tracking reveals abnormal visual preference for geometric images as an early biomarker of an autism spectrum disorder subtype associated with increased symptom severity. Biol. Psychiatry 2016, 79, 657–666. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.R.G.; Happé, F.; Pickles, A.; Marsden, A.J.S.; Tregay, J.; Baird, G.; Simonoff, E.; Charman, T. ‘Everyday memory’ impairments in autism spectrum disorders. J. Autism Dev. Disord. 2011, 41, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Chevallier, C.; Grèzes, J.; Molesworth, C.; Berthoz, S.; Happé, F. Brief report: Selective social anhedonia in high functioning autism. J. Autism Dev. Disord. 2012, 42, 1504–1509. [Google Scholar] [CrossRef] [PubMed]
- Stokes, M.; Newton, N.; Kaur, A. Stalking, and Social and Romantic Functioning Among Adolescents and Adults with Autism Spectrum Disorder. J. Autism Dev. Disord. 2007, 37, 1969–1986. [Google Scholar] [CrossRef] [PubMed]
- Mendelson, J.L.; Gates, J.A.; Lerner, M.D. Friendship in school-age boys with autism spectrum disorders: A meta-analytic summary and developmental, process-based model. Psychol. Bull. 2016, 142, 601–622. [Google Scholar] [CrossRef]
- Bauminger, N.; Shulman, C.; Agam, G. Peer Interaction and Loneliness in High-Functioning Children with Autism. J. Autism Dev. Disord. 2003, 33, 489–507. [Google Scholar] [CrossRef] [PubMed]
- Whitehouse, A.J.; Durkin, K.; Jaquet, E.; Ziatas, K. Friendship, loneliness and depression in adolescents with Asperger’s Syndrome. J. Adolesc. 2009, 32, 309–322. [Google Scholar] [CrossRef] [PubMed]
- Bishop-Fitzpatrick, L.; Minshew, N.J.; Mazefsky, C.A.; Eack, S.M. Perception of life as stressful, not biological response to stress, is associated with greater social disability in adults with autism spectrum disorder. J. Autism Dev. Disord. 2017, 47, 1–16. [Google Scholar] [CrossRef]
- Hirvikoski, T.; Blomqvist, M. High self-perceived stress and poor coping in intellectually able adults with autism spectrum disorder. Autism 2015, 19, 752–757. [Google Scholar] [CrossRef]
- Ogawa, S.; Lee, Y.-A.; Yamaguchi, Y.; Shibata, Y.; Goto, Y. Associations of acute and chronic stress hormones with cognitive functions in autism spectrum disorder. Neuroscience 2017, 343, 229–239. [Google Scholar] [CrossRef]
- Bradley, E.A.; Summers, J.A.; Wood, H.L.; Bryson, S.E. Comparing Rates of Psychiatric and Behavior Disorders in Adolescents and Young Adults with Severe Intellectual Disability with and without Autism. J. Autism Dev. Disord. 2004, 34, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Joshi, G.; Wozniak, J.; Petty, C.; Martelon, M.K.; Fried, R.; Bolfek, A.; Kotte, A.; Stevens, J.; Furtak, S.L.; Bourgeois, M.; et al. Psychiatric comorbidity and functioning in a clinically referred population of adults with autism spectrum disorders: A comparative study. J. Autism Dev. Disord. 2013, 43, 1314–1325. [Google Scholar] [CrossRef] [PubMed]
- Lugnegård, T.; Hallerbäck, M.U.; Gillberg, C. Psychiatric comorbidity in young adults with a clinical diagnosis of Asperger syndrome. Res. Dev. Disabil. 2011, 32, 1910–1917. [Google Scholar] [CrossRef] [PubMed]
- Muris, P.; Steerneman, P.; Merckelbach, H.; Holdrinet, I.; Meesters, C. Comorbid Anxiety Symptoms in Children with Pervasive Developmental Disorders. J. Anxiety Disord. 1998, 12, 387–393. [Google Scholar] [CrossRef]
- McConnell, B.; Szatmari, P.; Geschwind, D.; Dawson, G. Anxiety and Mood Disorders in Individuals with Autism Spectrum Disorder. In Autism Spectrum Disorders; Oxford University Press (OUP): New York, NY, USA, 2011; pp. 330–338. [Google Scholar]
- Bodfish, J.W.; Geschwind, D.; Dawson, G. Repetitive Behaviors in Individuals with Autism Spectrum Disorders. In Autism Spectrum Disorders; Oxford University Press (OUP): New York, NY, USA, 2011; pp. 200–212. [Google Scholar]
- Richler, J.; Huerta, M.; Bishop, S.L.; Lord, C. Developmental trajectories of restricted and repetitive behaviors and interests in children with autism spectrum disorders. Dev. Psychopathol. 2010, 22, 55–69. [Google Scholar] [CrossRef]
- Lam, K.S.; Bodfish, J.W.; Piven, J. Evidence for three subtypes of repetitive behavior in autism that differ in familiality and association with other symptoms. J. Child Psychol. Psychiatry 2008, 49, 1193–1200. [Google Scholar] [CrossRef]
- Richler, J.; Bishop, S.L.; Kleinke, J.R.; Lord, C. Restricted and repetitive behaviors in young children with autism spectrum disorders. J. Autism Dev. Disord. 2007, 37, 73–85. [Google Scholar] [CrossRef]
- Goldman, S.; Wang, C.; Salgado, M.W.; Greene, P.E.; Kim, M.; Rapin, I. Motor stereotypies in children with autism and other developmental disorders. Dev. Med. Child. Neurol. 2008, 51, 30–38. [Google Scholar] [CrossRef]
- Wolff, J.J.; Botteron, K.N.; Dager, S.R.; Elison, J.T.; Estes, A.M.; Gu, H.; Hazlett, H.C.; Pandey, J.; Paterson, S.J.; Schultz, R.T.; et al. Longitudinal Patterns of Repetitive Behavior in Toddlers with Autism. J. Child Psychol. Psychiatry 2014, 55, 945–953. [Google Scholar] [CrossRef]
- Esbensen, A.J.; Bishop, S.L.; Seltzer, M.M.; Greenberg, J.S.; Taylor, J.L. Comparisons Between Individuals With Autism Spectrum Disorders and Individuals With Down Syndrome in Adulthood. Am. J. Intellect. Dev. Disabil. 2010, 115, 277–290. [Google Scholar] [CrossRef]
- Klin, A.; Saulnier, C.A.; Sparrow, S.S.; Cicchetti, D.V.; Volkmar, F.R.; Lord, C. Social and communication abilities and disabilities in higher functioning individuals with autism spectrum disorders: The Vineland and the ADOS. J. Autism Dev. Disord. 2007, 37, 748–759. [Google Scholar] [CrossRef] [PubMed]
- Kuschner, E.S.; Eisenberg, I.W.; Orionzi, B.; Simmons, W.K.; Kenworthy, L.; Martin, A.; Wallace, G.L. A preliminary study of self-reported food selectivity in adolescents and young adults with autism spectrum disorder. Res. Autism Spectr. Disord. 2015, 15, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Sharp, W.G.; Berry, R.C.; McCracken, C.; Nuhu, N.N.; Marvel, E.; Saulnier, C.A.; Klin, A.; Jones, W.; Jaquess, D.L. Feeding Problems and Nutrient Intake in Children with Autism Spectrum Disorders: A Meta-analysis and Comprehensive Review of the Literature. J. Autism Dev. Disord. 2013, 43, 2159–2173. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; American Psychiatric Association: Washington DC, USA, 1994. [Google Scholar]
- Dubbelink, L.M.E.O.; Geurts, H.M. Planning Skills in Autism Spectrum Disorder Across the Lifespan: A Meta-analysis and Meta-regression. J. Autism. Dev. Disord. 2017, 47, 1148–1165. [Google Scholar] [CrossRef] [PubMed]
- Seltzer, M.M.; Greenberg, J.S.; Orsmond, G.I.; Esbensen, A.; Hong, J.; Taylor, J.L.; Smith, L.; Geschwind, D.; Dawson, G. Adolescents and Adults with Autism Spectrum Disorders. In Autism Spectrum Disorders; Oxford University Press (OUP): New York, NY, USA, 2011; pp. 241–252. [Google Scholar]
- Bauman, M.L.; Geschwind, D.; Dawson, G. Autism Spectrum Disorders: Identification and Implications of Associated Medical Conditions. In Autism Spectrum Disorders; Oxford University Press (OUP): New York, NY, USA, 2011; pp. 1215–1224. [Google Scholar]
- Buie, T.; Geschwind, D.; Dawson, G. Gastrointestinal Problems in Individuals with Autism Spectrum Disorders. In Autism Spectrum Disorders; Oxford University Press (OUP): New York, NY, USA, 2011; pp. 420–430. [Google Scholar]
- Gillberg, C.; Neville, B. Autism and epilepsy. In Researching the Autism Spectrum: Contemporary Perspectives; Roth, I., Rezaie, P., Eds.; Cambridge University Press: Cambridge, UK, 2011; pp. 176–189. [Google Scholar]
- Porter, A.E.; Glaze, D.G. Sleep problems. In Autism Spectrum Disorders; Amaral, D.G., Dawson, G., Geschwind, D.H., Eds.; Oxford University Press: New York, NY, USA, 2011; pp. 431–444. [Google Scholar]
- Sacco, R.; Gabriele, S.; Persico, A.M. Head circumference and brain size in autism spectrum disorder: A systematic review and meta-analysis. Psychiatry Res. Neuroimaging 2015, 234, 239–251. [Google Scholar] [CrossRef] [PubMed]
- Howlin, P.; Goode, S.; Hutton, J.; Rutter, M. Adult outcome for children with autism. J. Child Psychol. Psychiatry 2004, 45, 212–229. [Google Scholar] [CrossRef]
- Grandin, T. An inside view of autism. In High-Functioning Individuals with Autism; Schopler, E., Mesibov, G.B., Eds.; Plenum Press: New York, NY, USA, 1992; pp. 105–126. [Google Scholar]
- Baron-Cohen, S. Hey! It was just a joke! Understanding propositions and propositional attitudes by normally developing children and children with autism. J. Psychiatry Relat. Sci. 1997, 34, 174–178. [Google Scholar]
- McCann, J.; Peppé, S.; Gibbon, F.E.; O’Hare, A.; Rutherford, M.; Cleland, J. Prosody and its relationship to language in school-aged children with high-functioning autism. Int. J. Commun. Disord. 2007, 42, 682–702. [Google Scholar] [CrossRef]
- Paul, R.; Augustyn, A.; Klin, A.; Volkmar, F.R. Perception and Production of Prosody by Speakers with Autism Spectrum Disorders. J. Autism Dev. Disord. 2005, 35, 205–220. [Google Scholar] [CrossRef]
- Rosenblau, G.; Kliemann, D.; Dziobek, I.; Heekeren, H.R. Emotional prosody processing in autism spectrum disorder. Soc. Cogn. Affect Neurosci. 2017, 12, 224–239. [Google Scholar] [CrossRef]
- Rapin, I. Atypical sensory/perceptual responsiveness. In Autism: A Neurological Disorder of Early Brain Development; Tuchman, R., Rapin, I., Eds.; Mac Keith Press: London, UK, 2006; pp. 202–230. [Google Scholar]
- Freese, J.L.; Amaral, D.G. Neuroanatomy of the primate amygdala. In The Human Amygdala; Whalen, P.J., Phelps, E.A., Eds.; Guilford Press: New York, NY, USA, 2009; pp. 3–42. [Google Scholar]
- Schumann, C.M.; Amaral, D.G. Stereological Analysis of Amygdala Neuron Number in Autism. J. Neurosci. 2006, 26, 7674–7679. [Google Scholar] [CrossRef] [PubMed]
- Von dem Hagen, E.A.H.; Stoyanova, R.S.; Baron-Cohen, S.; Calder, A.J. Reduced functional connectivity within and between ‘social’ resting state networks in autism spectrum conditions. Soc. Cogn. Affect Neurosci. 2013, 8, 694–701. [Google Scholar] [CrossRef] [PubMed]
- LaBar, K.S.; Warren, L.H. Methodological approaches to studying the human amygdala. In The Human Amygdala; Whalen, P.J., Phelps, E.A., Eds.; Guilford Press: New York, NY, USA, 2009; pp. 155–176. [Google Scholar]
- Rutishauser, U.; Tudusciuc, O.; Wang, S.; Mamelak, A.N.; Ross, I.B.; Adolphs, R. Single-neuron correlates of atypical face processing in autism. Neuron 2013, 80, 887–899. [Google Scholar] [CrossRef] [PubMed]
- Sturm, V.; Fricke, O.; Bührle, C.P.; Lenartz, D.; Maarouf, M.; Treuer, H.; Mai, J.K.; Lehmkuhl, G.; Buehrle, C.P. DBS in the basolateral amygdala improves symptoms of autism and related self-injurious behavior: A case report and hypothesis on the pathogenesis of the disorder. Front. Hum. Neurosci. 2013, 6, 341. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.E.; Lyoo, I.K.; Estes, A.M.; Renshaw, P.F.; Shaw, D.W.; Friedman, S.D.; Kim, D.J.; Yoon, S.J.; Hwang, J.; Dager, S.R. Laterobasal Amygdalar Enlargement in 6- to 7-Year-Old Children With Autism Spectrum Disorder. Arch. Psychiatry 2010, 67, 1187–1197. [Google Scholar] [CrossRef] [PubMed]
- Nordahl, C.W.; Scholz, R.; Yang, X.; Buonocore, M.H.; Simon, T.; Rogers, S.; Amaral, D.G. Increased Rate of Amygdala Growth in Children Aged 2 to 4 Years With Autism Spectrum Disorders. Arch. Psychiatry 2012, 69, 53–61. [Google Scholar] [CrossRef]
- Sato, W.; Kochiyama, T.; Uono, S.; Yoshimura, S.; Kubota, Y.; Sawada, R.; Sakihama, M.; Toichi, M. Reduced gray matter volume in the social brain network in adults with autism spectrum disorder. Front. Hum. Neurosci. 2017, 11, 395. [Google Scholar] [CrossRef] [PubMed]
- Schumann, C.M.; Amaral, D.G. The human amygdala in autism. In The Human Amygdala; Whalen, P.J., Phelps, E.A., Eds.; Guilford Press: New York, NY, USA, 2009; pp. 362–381. [Google Scholar]
- Munson, J.; Dawson, G.; Abbott, R.; Faja, S.; Webb, S.J.; Friedman, S.D.; Shaw, D.; Artru, A.; Dager, S.R. Amygdalar Volume and Behavioral Development in Autism. Arch. Psychiatry 2006, 63, 686–693. [Google Scholar] [CrossRef]
- Nacewicz, B.M.; Dalton, K.M.; Johnstone, T.; Long, M.T.; McAuliff, E.M.; Oakes, T.R.; Alexander, A.L.; Davidson, R.J. Amygdala Volume and Nonverbal Social Impairment in Adolescent and Adult Males With Autism. Arch. Psychiatry 2006, 63, 1417–1428. [Google Scholar] [CrossRef]
- Price, J.L. Definition of the Orbital Cortex in Relation to Specific Connections with Limbic and Visceral Structures and Other Cortical Regions. Ann. N. Y. Acad. Sci. 2007, 1121, 54–71. [Google Scholar] [CrossRef]
- Cruz-Rizzolo, R.J.; Cavada, C.; Compañy, T.; Tejedor, J.; Reinoso-Suárez, F. The Anatomical Connections of the Macaque Monkey Orbitofrontal Cortex. A Review. Cereb. Cortex 2000, 10, 220–242. [Google Scholar]
- Tamminga, C.A. The human brain. Am. J. Psychiatry 2004, 161, 1169. [Google Scholar] [CrossRef] [PubMed]
- Clark, L.; Bechara, A.; Damasio, H.; Aitken, M.R.F.; Sahakian, B.J.; Robbins, T.W. Differential effects of insular and ventromedial prefrontal cortex lesions on risky decision-making. Brain 2008, 131, 1311–1322. [Google Scholar] [CrossRef] [PubMed]
- Kringelbach, M.L. The functional neuroanatomy of the human orbitofrontal cortex: Evidence from neuroimaging and neuropsychology. Prog. Neurobiol. 2004, 72, 341–372. [Google Scholar] [CrossRef] [PubMed]
- Rudebeck, P.H.; Murray, E.A. Balkanizing the primate orbitofrontal cortex: Distinct subregions for comparing and contrasting values. Ann. N. Y. Acad. Sci. 2011, 1239, 1–13. [Google Scholar] [CrossRef]
- Kaiser, M.D.; Hudac, C.M.; Shultz, S.; Lee, S.M.; Cheung, C.; Berken, A.M.; Deen, B.; Pitskel, N.B.; Sugrue, D.R.; Voos, A.C.; et al. Neural signatures of autism. Proc. Acad. Sci. 2010, 107, 21223–21228. [Google Scholar] [CrossRef]
- Boes, A.D.; Mehta, S.; Rudrauf, D.; van der Plas, E.; Grabowski, T.; Adolphs, R.; Nopoulo, P. Changes in cortical morphology resulting from long-term amygdala damage. Soc. Cogn. Affect Neurosci. 2012, 7, 588–595. [Google Scholar] [CrossRef]
- Herringshaw, A.J.; Ammons, C.J.; DeRamus, T.P.; Kana, R.K. Hemispheric differences in language processing in autism spectrum disorders: A meta-analysis of neuroimaging studies. Autism Res. 2016, 9, 1046–1057. [Google Scholar] [CrossRef]
- Muñoz-Yunta, J.; Ortíz, T.; Palau-Baduell, M.; Martín-Muñoz, L.; Salvadó-Salvadó, B.; Valls-Santasusana, A.; Perich-Alsina, J.; Cristobal, I.; Fernández, A.; Maestu, F.; et al. Magnetoencephalographic pattern of epileptiform activity in children with early-onset autism spectrum disorders. Clin. Neurophysiol. 2008, 119, 626–634. [Google Scholar] [CrossRef]
- Nieuwenhuys, R. The insular cortex: A review. In Evolution of the Primate Brain: From Neuron to Behavior; Hofman, M.A., Falk, D., Eds.; Progress in Brain Research; Elsevier: Amsterdam, The Netherlands, 2012; Volume 195, pp. 123–163. [Google Scholar]
- Fanselow, M.S.; Wassum, K.M. The origins and organization of vertebrate Pavlovian conditioning. Cold Spring Harb. Perspect. Biol. 2016, 8, a021717. [Google Scholar] [CrossRef]
- LeDoux, J.E. Coming to terms with fear. Proc. Acad. Sci. 2014, 111, 2871–2878. [Google Scholar] [CrossRef] [PubMed]
- McGaugh, J.L. Making lasting memories: Remembering the significant. Proc. Acad. Sci. 2013, 110, 10402–10407. [Google Scholar] [CrossRef] [PubMed]
- McIntyre, C.K.; McGaugh, J.L.; Williams, C.L. Interacting brain systems modulate memory consolidation. Neurosci. Biobehav. Rev. 2012, 36, 1750–1762. [Google Scholar] [CrossRef] [PubMed]
- Shin, L.M.; Liberzon, I. The neurocircuitry of fear, stress, and anxiety disorders. Neuropsychopharmacology 2010, 35, 169–191. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A.K.; Phelps, E.A. Is the Human Amygdala Critical for the Subjective Experience of Emotion? Evidence of Intact Dispositional Affect in Patients with Amygdala Lesions. J. Cogn. Neurosci. 2002, 14, 709–720. [Google Scholar] [CrossRef] [PubMed]
- Feinstein, J.S.; Buzza, C.; Hurlemann, R.; Follmer, R.L.; Dahdaleh, N.S.; Coryell, W.H.; Welsh, M.J.; Tranel, D.; Wemmie, J.A. Fear and panic in humans with bilateral amygdala damage. Nat. Neurosci. 2013, 16, 270–272. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, R. Disorders of facial recognition, social behaviour and affect after combined bilateral amygdalotomy and subcaudate tractotomy—A clinical and experimental study. Psychol. Med. 1986, 16, 439–450. [Google Scholar] [CrossRef]
- Khalsa, S.S.; Feinstein, J.S.; Li, W.; Feusner, J.D.; Adolphs, R.; Hurlemann, R. Panic Anxiety in Humans with Bilateral Amygdala Lesions: Pharmacological Induction via Cardiorespiratory Interoceptive Pathways. J. Neurosci. 2016, 36, 3559–3566. [Google Scholar] [CrossRef]
- Koenigs, M.; Huey, E.D.; Raymont, V.; Cheon, B.; Solomon, J.; Wassermann, E.M.; Grafman, J. Focal brain damage protects against post-traumatic stress disorder in combat veterans. Nat. Neurosci. 2008, 11, 232–237. [Google Scholar] [CrossRef]
- Shi, T.-Y.; Feng, S.-F.; Wei, M.-X.; Huang, Y.; Liu, G.; Wu, H.-T.; Zhang, Y.-X.; Zhou, W.-X. Kainate receptor mediated presynaptic LTP in agranular insular cortex contributes to fear and anxiety in mice. Neuropharmacology 2018, 128, 388–400. [Google Scholar] [CrossRef]
- Hernádi, I.; Grabenhorst, F.; Schultz, W. Planning activity for internally generated reward goals in monkey amygdala neurons. Nat. Neurosci. 2015, 18, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Amaral, D.G.; Price, J.L. Amygdalo-cortical projections in the monkey (Macaca fascicularis). J. Comp. Neurol. 1984, 230, 465–496. [Google Scholar] [CrossRef] [PubMed]
- Geschwind, N. Disconnexion Syndromes in Animals and Man. Brain 1965, 88, 237. [Google Scholar] [CrossRef] [PubMed]
- Weston, C.S.E. Amygdala Represents Diverse Forms of Intangible Knowledge, That Illuminate Social Processing and Major Clinical Disorders. Front. Hum. Neurosci. 2018, 12, 336. [Google Scholar] [CrossRef] [PubMed]
- Sander, D.; Grafman, J.; Zalla, T. The Human Amygdala: An Evolved System for Relevance Detection. Rev. Neurosci. 2003, 14, 303–316. [Google Scholar] [CrossRef] [PubMed]
- Whalen, P.J.; Phelps, E.A. (Eds.) The Human Amygdala; Guilford Press: New York, NY, USA, 2009. [Google Scholar]
- Andino, S.L.G.; Menendez, R.G.D.P.; De Peralta, R.G. Coding of saliency by ensemble bursting in the amygdala of primates. Front. Behav. Neurosci. 2012, 6, 38. [Google Scholar]
- Gerbella, M.; Baccarini, M.; Borra, E.; Rozzi, S.; Luppino, G. Amygdalar connections of the macaque areas 45A and 45B. Brain Struct. Funct. 2014, 219, 831–842. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; DiNicola, L.; Heymann, P.; Hampson, M.; Chawarska, K. Impaired Value Learning for Faces in Preschoolers With Autism Spectrum Disorder. J. Am. Acad. Child Adolesc. Psychiatry 2018, 57, 33–40. [Google Scholar] [CrossRef]
- Freese, J.L.; Amaral, D.G. The organization of projections from the amygdala to visual cortical areas TE and V1 in the macaque monkey. J. Comp. Neurol. 2005, 486, 295–317. [Google Scholar] [CrossRef]
- Freese, J.L.; Amaral, D.G. The Synaptic Organization of Projections from the Amygdala to Visual Cortical Areas TE and V1 in the Macaque Monkey. J. Comp. Neurol. 2006, 496, 655–667. [Google Scholar] [CrossRef]
- Sato, W.; Kochiyama, T.; Uono, S.; Matsuda, K.; Usui, K.; Usui, N.; Inoue, Y.; Toichi, M. Bidirectional electric communication between the inferior occipital gyrus and the amygdala during face processing. Hum. Brain Mapp. 2017, 38, 4511–4524. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.D.; Lori, N.F.; Akbudak, E.; Sorar, E.; Gultepe, E.; Shimony, J.S.; McKinstry, R.C.; Conturo, T.E. MRI diffusion tensor tracking of a new amygdalo-fusiform and hippocampo-fusiform pathway system in humans. J. Magn. Reson. Imaging 2009, 29, 1248–1261. [Google Scholar] [CrossRef] [PubMed]
- Conturo, T.E.; Williams, D.L.; Smith, C.D.; Gultepe, E.; Akbudak, E.; Minshew, N.J. Neuronal fiber pathway abnormalities in autism: An initial MRI diffusion tensor tracking study of hippocampo-fusiform and amygdalo-fusiform pathways. J. Int. Neuropsychol. Soc. 2008, 14, 933–946. [Google Scholar] [CrossRef] [PubMed]
- Kleinhans, N.M.; Richards, T.; Sterling, L.; Stegbauer, K.C.; Mahurin, R.; Johnson, L.C.; Greenson, J.; Dawson, G.; Aylward, E. Abnormal functional connectivity in autism spectrum disorders during face processing. Brain 2008, 131, 1000–1012. [Google Scholar] [CrossRef] [PubMed]
- Hadj-Bouziane, F.; Liu, N.; Bell, A.H.; Gothard, K.M.; Luh, W.-M.; Tootell, R.B.H.; Murray, E.A.; Ungerleider, L.G. Amygdala lesions disrupt modulation of functional MRI activity evoked by facial expression in the monkey inferior temporal cortex. Proc. Acad. Sci. 2012, 109, E3640–E3648. [Google Scholar] [CrossRef] [PubMed]
- Yukie, M. Connections between the amygdala and auditory cortical areas in the macaque monkey. Neurosci. Res. 2002, 42, 219–229. [Google Scholar] [CrossRef]
- Elison, J.T.; Wolff, J.J.; Heimer, D.C.; Paterson, S.J.; Gu, H.; Hazlett, H.C.; Styner, M.; Gerig, G.; Piven, J. The IBIS Network. Frontolimbic neural circuitry at 6 months predicts individual differences in joint attention at 9 months. Dev. Sci. 2013, 16, 186–197. [Google Scholar] [CrossRef]
- Rapin, I. Introduction. Autism turns 65: A neurologist’s bird’s eye view. In Autism Spectrum Disorders; Amaral, D.G., Dawson, G., Geschwind, D.H., Eds.; Oxford University Press: New York, NY, USA, 2011; pp. 3–14. [Google Scholar]
- Zangemeister, L.; Grabenhorst, F.; Schultz, W. Neural Basis for Economic Saving Strategies in Human Amygdala-Prefrontal Reward Circuits. Curr. Boil. 2016, 26, 3004–3013. [Google Scholar] [CrossRef]
- Anderson, S.W.; Barrash, J.; Bechara, A.; Tranel, D. Impairments of emotion and real-world complex behavior following childhood- or adult-onset damage to ventromedial prefrontal cortex. J. Int. Neuropsychol. Soc. 2006, 12, 224–235. [Google Scholar] [CrossRef]
- Damasio, A.R. Descartes’ Error: Emotion, Reason & the Human Brain; Picador: London, UK, 1994. [Google Scholar]
- Dimitrov, M.; Phipps, M.; Zahn, T.P.; Grafman, J. A thoroughly modern gage. Neurocase 1999, 5, 345–354. [Google Scholar] [CrossRef]
- Kavaliers, M.; Colwell, D.D.; Choleris, E.; Ossenkopp, K.-P. Learning to cope with biting flies: Rapid NMDA-mediated acquisition of conditioned analgesia. Behav. Neurosci. 1999, 113, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Anderson, S.W.; Bechara, A.; Damasio, H.; Tranel, D.; Damasio, A.R. Impairment of social and moral behavior related to early damage in human prefrontal cortex. Nat. Neurosci. 1999, 2, 1032–1037. [Google Scholar] [CrossRef] [PubMed]
- Boes, A.D.; Grafft, A.H.; Joshi, C.; Chuang, N.A.; Nopoulos, P.; Anderson, S.W. Behavioral effects of congenital ventromedial prefrontal cortex malformation. BMC Neurol. 2011, 11, 151. [Google Scholar] [CrossRef] [PubMed]
- Koenigs, M.; Sutterer, M.; Tranel, D.; Taber-Thomas, B.C.; Asp, E.W.; Anderson, S.W. Arrested development: Early prefrontal lesions impair the maturation of moral judgement. Brain 2014, 137, 1254–1261. [Google Scholar]
- Young, L.; Koenigs, M. Investigating emotion in moral cognition: A review of evidence from functional neuroimaging and neuropsychology. Br. Med. 2007, 84, 69–79. [Google Scholar] [CrossRef] [PubMed]
- McNamee, D.; Rangel, A.; O’Doherty, J.P. Category-dependent and category-independent goal-value codes in human ventromedial prefrontal cortex. Nat. Neurosci. 2013, 16, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Piech, R.M.; Lewis, J.; Parkinson, C.H.; Owen, A.M.; Roberts, A.C.; Downing, P.E.; Parkinson, J.A. Neural Correlates of Appetite and Hunger-Related Evaluative Judgments. PLoS ONE 2009, 4, e6581. [Google Scholar] [CrossRef] [PubMed]
- Price, J.L. Connections of orbital cortex. In The Orbitofrontal Cortex; Zald, D.H., Rauch, S.L., Eds.; Oxford University Press: New York, NY, USA, 2006; pp. 39–55. [Google Scholar]
- Rolls, E.T. Taste, olfactory, and food texture processing in the brain, and the control of food intake. Physiol. Behav. 2005, 85, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Reber, J.; Feinstein, J.S.; O’Doherty, J.P.; Liljeholm, M.; Adolphs, R.; Tranel, D. Selective impairment of goal-directed decision-making following lesions to the human ventromedial prefrontal cortex. Brain 2017, 140, 1743–1756. [Google Scholar] [CrossRef]
- Kim, H.; Adolphs, R.; O’Doherty, J.P.; Shimojo, S. Temporal isolation of neural processes underlying face preference decisions. Proc. Acad. Sci. 2007, 104, 18253–18258. [Google Scholar] [CrossRef]
- Dougherty, D.D.; Rauch, S.L.; Deckersbach, T.; Marci, C.; Loh, R.; Shin, L.M.; Alpert, N.M.; Fischman, A.J.; Fava, M. Ventromedial Prefrontal Cortex and Amygdala Dysfunction During an AngerInduction Positron Emission Tomography Study in Patients With Major DepressiveDisorder With Anger Attacks. Arch. Psychiatry 2004, 61, 795–804. [Google Scholar] [CrossRef] [PubMed]
- Fabiansson, E.C.; Denson, T.F.; Moulds, M.L.; Grisham, J.R.; Schira, M.M. Don’t look back in anger: Neural correlates of reappraisal, analytical rumination, and angry rumination during recall of an anger-inducing autobiographical memory. NeuroImage 2012, 59, 2974–2981. [Google Scholar] [CrossRef] [PubMed]
- Grafman, J.; Schwab, K.; Warden, D.; Pridgen, A.; Brown, H.R.; Salazar, A.M. Frontal lobe injuries, violence, and aggression: A report of the Vietnam Head Injury Study. Neurology 1996, 46, 1231. [Google Scholar] [CrossRef]
- Jones, B.E. Basic Mechanisms of Sleep-Wake States. In Principles and Practice of Sleep Medicine; Elsevier BV: Amsterdam, The Netherlands, 2005; pp. 136–153. [Google Scholar]
- Mander, B.A.; Rao, V.; Lu, B.; Saletin, J.M.; Lindquist, J.R.; Ancoli-Israel, S.; Jagust, W.; Walker, M.P. Prefrontal atrophy, disrupted NREM slow waves and impaired hippocampal-dependent memory in aging. Nat. Neurosci. 2013, 16, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Nofzinger, E.A.; Maquet, P. What Brain Imaging Reveals about Sleep Generation and Maintenance. In Principles and Practice of Sleep Medicine; Elsevier BV: Amsterdam, The Netherlands, 2011; pp. 201–214. [Google Scholar]
- Shan, Z.Y.; Kwiatek, R.; Burnet, R.; Del Fante, P.; Staines, D.R.; Marshall-Gradisnik, S.M.; Barnden, L.R. Medial prefrontal cortex deficits correlate with unrefreshing sleep in patients with chronic fatigue syndrome. NMR Biomed. 2017, 30, e3757. [Google Scholar] [CrossRef]
- Atomi, T.; Noriuchi, M.; Oba, K.; Atomi, Y.; Kikuchi, Y. Self-Recognition of One’s Own Fall Recruits the Genuine Bodily Crisis-Related Brain Activity. PLoS ONE 2014, 9, 115303. [Google Scholar] [CrossRef] [PubMed]
- Kaas, J.H. Somatosensory system. In The Human Nervous System, 2nd ed.; Paxinos, G., Mai, J.K., Eds.; Elsevier Academic Press: London, UK, 2004; pp. 1059–1092. [Google Scholar]
- Rozzi, S.; Calzavara, R.; Belmalih, A.; Borra, E.; Gregoriou, G.G.; Matelli, M.; Luppino, G. Cortical connections of the inferior parietal cortical convexity of the macaque monkey. Cereb. Cortex 2006, 16, 1389–1417. [Google Scholar] [CrossRef]
- Sereno, M.I.; Huang, R.-S. Multisensory maps in parietal cortex. Curr. Opin. Neurobiol. 2014, 24, 39–46. [Google Scholar] [CrossRef]
- Mosconi, M.W.; Takarae, Y.; Sweeney, J.A.; Geschwind, D.; Dawson, G. Motor Functioning and Dyspraxia in Autism Spectrum Disorders. In Autism Spectrum Disorders; Oxford University Press (OUP): New York, NY, USA, 2011; pp. 355–380. [Google Scholar]
- Igelström, K.M.; Webb, T.W.; Kelly, Y.T.; Graziano, M.S.A. Topographical Organization of Attentional, Social, and Memory Processes in the Human Temporoparietal Cortex123. eNeuro 2016, 3, 1–12. [Google Scholar] [CrossRef]
- Carter, R.M.; Huettel, S.A. A Nexus Model of the Temporal-Parietal Junction. Trends Cogn. Sci. 2013, 17, 328–336. [Google Scholar] [CrossRef]
- Geng, J.J.; Vossel, S. Re-evaluating the role of TPJ in attentional control: Contextual updating? Neurosci. Biobehav. Rev. 2013, 37, 2608–2620. [Google Scholar] [CrossRef] [PubMed]
- Bzdok, D.; Schilbach, L.; Vogeley, K.; Schneider, K.; Laird, A.R.; Langner, R.; Eickhoff, S.B. Parsing the neural correlates of moral cognition: ALE meta-analysis on morality, theory of mind, and empathy. Anat. Embryol. 2012, 217, 783–796. [Google Scholar] [CrossRef] [PubMed]
- Eres, R.; Louis, W.R.; Molenberghs, P. Common and distinct neural networks involved in fMRI studies investigating morality: An ALE meta-analysis. Soc. Neurosci. 2018, 13, 384–398. [Google Scholar] [CrossRef] [PubMed]
- Blanke, O. Multisensory brain mechanisms of bodily self-consciousness. Nat. Rev. Neurosci. 2012, 13, 556–571. [Google Scholar] [CrossRef] [PubMed]
- Dieguez, S.; Annoni, J.-M. Asomatognosia: Disorders of the bodily self. In The Behavioral and Cognitive Neurology of Stroke; Cambridge University Press (CUP): Cambridge, UK, 2013; pp. 170–192. [Google Scholar]
- Halligan, P.W.; Marshall, J.C.; Wade, D.T. Unilateral Somatoparaphrenia After Right Hemisphere Stroke: A Case Description. Cortex 1995, 31, 173–182. [Google Scholar] [CrossRef]
- Hécaen, H.; Penfield, W.; Bertrand, C.; Malmo, R. The Syndrome of Apractognosia Due to Lesions of the Minor Cerebral Hemisphere. Arch. Neurol. Psychiatry 1956, 75, 400. [Google Scholar] [CrossRef]
- Heydrich, L.; Blanke, O. Distinct illusory own-body perceptions caused by damage to posterior insula and extrastriate cortex. Brain 2013, 136, 790–803. [Google Scholar] [CrossRef] [PubMed]
- Ionta, S.; Heydrich, L.; Lenggenhager, B.; Mouthon, M.; Fornari, E.; Chapuis, D.; Gassert, R.; Blanke, O. Multisensory Mechanisms in Temporo-Parietal Cortex Support Self-Location and First-Person Perspective. Neuron 2011, 70, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Ionta, S.; Martuzzi, R.; Salomon, R.; Blanke, O. The brain network reflecting bodily self-consciousness: A functional connectivity study. Soc. Cogn. Affect. Neurosci. 2014, 9, 1904–1913. [Google Scholar] [CrossRef]
- Salvato, G.; Gandola, M.; Veronelli, L.; Agostoni, E.C.; Sberna, M.; Corbo, M.; Bottini, G. The spatial side of somatoparaphrenia: A case study. Neurocase 2016, 22, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Evans, N.; Blanke, O. Shared electrophysiology mechanisms of body ownership and motor imagery. NeuroImage 2013, 64, 216–228. [Google Scholar] [CrossRef]
- McDonald, I. Musical alexia with recovery: A personal account. Brain 2006, 129, 2554–2561. [Google Scholar] [CrossRef] [PubMed]
- Ethofer, T.; Bretscher, J.; Gschwind, M.; Kreifelts, B.; Wildgruber, D.; Vuilleumier, P. Emotional voice areas: Anatomic location, functional properties, and structural connections revealed by combined fMRI/DTI. Cereb. Cortex 2012, 22, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Gerbella, M.; Belmalih, A.; Borra, E.; Rozzi, S.; Luppino, G. Cortical connections of the macaque caudal ventrolateral prefrontal areas 45A and 45B. Cereb. Cortex 2010, 20, 141–168. [Google Scholar] [CrossRef] [PubMed]
- Humphries, C.; Love, T.; Swinney, D.; Hickok, G. Response of anterior temporal cortex to syntactic and prosodic manipulations during sentence processing. Hum. Brain Mapp. 2005, 26, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Jahanshahi, M.; Jones, C.R.G.; Zijlmans, J.; Katzenschlager, R.; Lee, L.; Quinn, N.; Frith, C.D.; Lees, A.J. Dopaminergic modulation of striato-frontal connectivity during motor timing in Parkinson’s disease. Brain 2010, 133, 727–745. [Google Scholar] [CrossRef] [PubMed]
- Jezzini, A.; Rozzi, S.; Borra, E.; Gallese, V.; Caruana, F.; Gerbella, M. A shared neural network for emotional expression and perception: An anatomical study in the macaque monkey. Front. Behav. Neurosci. 2015, 9, 1099. [Google Scholar] [CrossRef]
- Kristensen, L.B.; Wang, L.; Petersson, K.M.; Hagoort, P. The interface between language and attention: Prosodic focus marking recruits a general attention network in spoken language comprehension. Cereb. Cortex 2013, 23, 1836–1848. [Google Scholar] [CrossRef]
- McFarland, H.R.; Fortin, D. Amusia due to Right Temporoparietal Infarct. Arch. Neurol. 1982, 39, 725–727. [Google Scholar] [CrossRef]
- Terao, Y.; Mizuno, T.; Shindoh, M.; Sakurai, Y.; Ugawa, Y.; Kobayashi, S.; Nagai, C.; Furubayashi, T.; Arai, N.; Okabe, S.; et al. Vocal amusia in a professional tango singer due to a right superior temporal cortex infarction. Neuropsychologia 2006, 44, 479–488. [Google Scholar] [CrossRef]
- Forkel, S.J.; De Schotten, M.T.; Dell’Acqua, F.; Kalra, L.; Murphy, D.G.M.; Williams, S.C.R.; Catani, M.; Dell’Acqua, F. Anatomical predictors of aphasia recovery: A tractography study of bilateral perisylvian language networks. Brain 2014, 137, 2027–2039. [Google Scholar] [CrossRef] [PubMed]
- Lacroix, A.N.; Diaz, A.F.; Rogalsky, C. The relationship between the neural computations for speech and music perception is context-dependent: An activation likelihood estimate study. Front. Psychol. 2015, 6, 1138. [Google Scholar] [CrossRef] [PubMed]
- Rogalsky, C.; Rong, F.; Saberi, K.; Hickok, G. Functional anatomy of language and music perception: Temporal and structural factors investigated using functional magnetic resonance imaging. J. Neurosci. 2011, 31, 3843–3852. [Google Scholar] [CrossRef] [PubMed]
- Ueno, T.; Saito, S.; Rogers, T.T.; Ralph, M.A.L. Lichtheim 2: Synthesizing Aphasia and the Neural Basis of Language in a Neurocomputational Model of the Dual Dorsal-Ventral Language Pathways. Neuron 2011, 72, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Xing, S.; Lacey, E.H.; Skipper-Kallal, L.M.; Jiang, X.; Harris-Love, M.L.; Zeng, J.; Turkeltaub, P.E. Right hemisphere grey matter structure and language outcomes in chronic left hemisphere stroke. Brain 2016, 139, 227–241. [Google Scholar] [CrossRef] [PubMed]
- Afif, A.; Minotti, L.; Kahane, P.; Hoffmann, D. Anatomofunctional organization of the insular cortex: A study using intracerebral electrical stimulation in epileptic patients. Epilepsia 2010, 51, 2305–2315. [Google Scholar] [CrossRef] [PubMed]
- Bamiou, D.-E.; Musiek, F.E.; Luxon, L.M. The insula (Island of Reil) and its role in auditory processing. Brain Rev. 2003, 42, 143–154. [Google Scholar] [CrossRef]
- Mazzola, L.; Isnard, J.; Mauguière, F. Somatosensory and pain responses to stimulation of the second somatosensory area (SII) in humans. A comparison with SI and insular responses. Cereb. Cortex 2006, 16, 960–968. [Google Scholar] [CrossRef]
- Cereda, C.; Ghika, J.; Maeder, P.; Bogousslavsky, J. Strokes restricted to the insular cortex. Neurology 2002, 59, 1950–1955. [Google Scholar] [CrossRef]
- Khalsa, S.S.; Rudrauf, D.; Feinstein, J.S.; Tranel, D. The pathways of interoceptive awareness. Nat. Neurosci. 2009, 12, 1494–1496. [Google Scholar] [CrossRef]
- Egan, G.F.; Johnson, J.; Farrell, M.; McAllen, R.; Zamarripa, F.; McKinley, M.J.; Lancaster, J.; Denton, D.; Fox, P.T. Cortical, thalamic, and hypothalamic responses to cooling and warming the skin in awake humans: A positron-emission tomography study. Proc. Acad. Sci. 2005, 102, 5262–5267. [Google Scholar] [CrossRef] [PubMed]
- Diwadkar, V.A.; Murphy, E.R.; Freedman, R.R. Temporal sequence of brain activations during naturally occurring thermoregulatory events. Cereb. Cortex 2014, 24, 3006–3013. [Google Scholar] [CrossRef] [PubMed]
- Weston, C. Another major function of the anterior cingulate cortex: The representation of requirements. Neurosci. Biobehav. Rev. 2012, 36, 90–110. [Google Scholar] [CrossRef] [PubMed]
- Kapur, N. Paradoxical functional facilitation in brain-behaviour research. Brain 1996, 119, 1775–1790. [Google Scholar] [CrossRef] [PubMed]
- Midorikawa, A.; Kawamura, M. The emergence of artistic ability following traumatic brain injury. Neurocase 2015, 21, 90–94. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Miller, B.L.; Cummings, J.; Mishkin, F.; Boone, K.; Prince, F.; Ponton, M.; Cotman, C. Emergence of artistic talent in frontotemporal dementia. Neurology 1998, 51, 978–982. [Google Scholar] [CrossRef] [PubMed]
- Rauschecker, J.P.; Tian, B.; Korte, M.; Egert, U. Crossmodal changes in the somatosensory vibrissa/barrel system of visually deprived animals. Proc. Acad. Sci. 1992, 89, 5063–5067. [Google Scholar] [CrossRef]
- Schott, G.D. Pictures as a neurological tool: Lessons from enhanced and emergent artistry in brain disease. Brain 2012, 135, 1947–1963. [Google Scholar] [CrossRef]
- Thomas-Antérion, C.; Dionet, E.; Borg, C.; Extier, C.; Faillenot, I.; Peyron, R.; Créac’h, C. De novo artistic activity following insular–SII ischemia. Pain 2010, 150, 121–127. [Google Scholar]
- Lagarde, J.; Valabrègue, R.; Corvol, J.-C.; Garcin, B.; Volle, E.; Le Ber, I.; Vidailhet, M.; Dubois, B.; Levy, R. Why do patients with neurodegenerative frontal syndrome fail to answer: ‘In what way are an orange and a banana alike?’. Brain 2015, 138, 456–471. [Google Scholar] [CrossRef]
- Martin, A. GRAPES-Grounding representations in action, perception, and emotion systems: How object properties and categories are represented in the human brain. Psychon. Bull. Rev. 2016, 23, 979–990. [Google Scholar] [CrossRef] [PubMed]
- Pellicano, E.; Dinsmore, A.; Charman, T. What should autism research focus upon? Community views and priorities from the United Kingdom. Autism 2014, 18, 756–770. [Google Scholar] [CrossRef] [PubMed]
- Maïano, C.; Normand, C.L.; Salvas, M.-C.; Moullec, G.; Aimé, A. Prevalence of school bullying among youth with autism spectrum disorders: A systematic review and meta-analysis. Autism Res. 2016, 9, 601–615. [Google Scholar] [CrossRef] [PubMed]
- Paul, A.; Gallot, C.; Lelouche, C.; Bouvard, M.P.; Amestoy, A. Victimisation in a French population of children and youths with autism spectrum disorder: A case control study. Child Adolesc. Psychiatry Heal. 2018, 12, 48. [Google Scholar] [CrossRef] [PubMed]
- Mazurek, M.O. Loneliness, friendship, and well-being in adults with autism spectrum disorders. Autism 2014, 18, 223–232. [Google Scholar] [CrossRef]
- Halls, G.; Cooper, P.J.; Creswell, C. Social communication deficits: Specific associations with Social Anxiety Disorder. J. Affect. Disord. 2015, 172, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Pickard, H.; Rijsdijk, F.; Happé, F.; Mandy, W. Are Social and Communication Difficulties a Risk Factor for the Development of Social Anxiety? J. Am. Acad. Child Adolesc. Psychiatry 2017, 56, 344–351.e3. [Google Scholar] [CrossRef]
- Singham, T.; Viding, E.; Schoeler, T.; Arseneault, L.; Ronald, A.; Cecil, C.M.; McCrory, E.; Rijsdijk, F.; Pingault, J.-B. Concurrent and Longitudinal Contribution of Exposure to Bullying in Childhood to Mental Health. JAMA Psychiatry 2017, 74, 1112–1119. [Google Scholar] [CrossRef]
- Storch, E.A.; Larson, M.J.; Ehrenreich-May, J.; Arnold, E.B.; Jones, A.M.; Renno, P.; Fujii, C.; Lewin, A.B.; Mutch, P.J.; Murphy, T.K.; et al. Peer Victimization in Youth with Autism Spectrum Disorders and Co-occurring Anxiety: Relations with Psychopathology and Loneliness. J. Dev. Phys. Disabil. 2012, 24, 575–590. [Google Scholar] [CrossRef]
- Adams, R.E.; Fredstrom, B.K.; Duncan, A.W.; Holleb, L.J.; Bishop, S.L. Using self- and parent-reports to test the association between peer victimization and internalizing symptoms in verbally fluent adolescents with ASD. J. Autism Dev. Disord. 2014, 44, 861–872. [Google Scholar] [CrossRef]
- Bromberger, J.T.; Kravitz, H.M.; Matthews, K.; Youk, A.; Brown, C.; Feng, W. Predictors of first lifetime episodes of major depression in midlife women. Psychol. Med. 2009, 39, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Cole, D.A.; Peeke, L.G.; Martin, J.M.; Truglio, R.; Seroczynski, A.D. A longitudinal look at the relation between depression and anxiety in children and adolescents. J. Consult. Clin. Psychol. 1998, 66, 451–460. [Google Scholar] [CrossRef]
- Goodwin, R.D. Anxiety disorders and the onset of depression among adults in the community. Psychol. Med. 2002, 32, 1121–1124. [Google Scholar] [CrossRef] [PubMed]
- Gotham, K.; Bishop, S.L.; Brunwasser, S.; Lord, C. Rumination and Perceived Impairment Associated with Depressive Symptoms in a Verbal Adolescent-Adult ASD Sample. Autism Res 2014, 7, 381–391. [Google Scholar] [CrossRef] [PubMed]
- Hedley, D.; Young, R. Social comparison processes and depressive symptoms in children and adolescents with Asperger syndrome. Autism 2006, 10, 139–153. [Google Scholar] [CrossRef]
- Rice, F.; Sellers, R.; Hammerton, G.; Eyre, O.; Bevan-Jones, R.; Collishaw, S.; Thapar, A.K.; Harold, G.T. Antecedents of New-Onset Major Depressive Disorder in Children and Adolescents at High Familial Risk. JAMA Psychiatry 2017, 74, 153. [Google Scholar] [CrossRef]
- Sterling, L.; Dawson, G.; Estes, A.; Greenson, J. Characteristics associated with presence of depressive symptoms in adults with autism spectrum disorder. J. Autism Dev. Disord. 2008, 38, 1011–1018. [Google Scholar] [CrossRef]
- Gottlieb, D.H.; Capitanio, J.P.; McCowan, B. Risk factors for stereotypic behavior and self-biting in rhesus macaques (Macaca mulatta): Animal’s history, current environment, and personality. Am. J. Primatol. 2013, 75, 995–1008. [Google Scholar] [CrossRef]
- Novak, M.A. Self-injurious behavior in rhesus monkeys: New insights into its etiology, physiology, and treatment. Am. J. Primatol. 2003, 59, 3–19. [Google Scholar] [CrossRef]
- Novak, M.A.; Meyer, J.S.; Lutz, C.; Tiefenbacher, S. Deprived environments: Developmental insights from primatology. In Stereotypic Animal Behaviour: Fundamentals & Applications to Welfare, 2nd ed.; Mason, G., Rushen, J., Eds.; CAB International: Wallingford, UK, 2006; pp. 153–189. [Google Scholar]
- Rushen, J.; Mason, G. A decade-or-more’s progress in understanding stereotypic behaviour. In Stereotypic Animal Behaviour: Fundamentals & Applications to Welfare, 2nd ed.; Mason, G., Rushen, J., Eds.; CAB International: Wallingford, UK, 2006; pp. 1–18. [Google Scholar]
- Muehlmann, A.; Edington, G.; Mihalik, A.; Buchwald, Z.; Koppuzha, D.; Korah, M.; Lewis, M. Further characterization of repetitive behavior in C58 mice: Developmental trajectory and effects of environmental enrichment. Behav. Brain 2012, 235, 143–149. [Google Scholar] [CrossRef]
- Cullen, K.R.; Westlund, M.K.; Lariviere, L.L.; Klimes-Dougan, B. An Adolescent With Nonsuicidal Self-Injury: A Case and Discussion of Neurobiological Research on Emotion Regulation. Am. J. Psychiatry 2013, 170, 828–831. [Google Scholar] [CrossRef] [PubMed]
- Gratz, K.L.; Chapman, A.L.; Dixon-Gordon, K.L.; Tull, M.T. Exploring the association of deliberate self-harm with emotional relief using a novel implicit association test. Personal Disord. 2016, 7, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Klonsky, E.D. The functions of deliberate self-injury: A review of the evidence. Clin. Psychol. 2007, 27, 226–239. [Google Scholar] [CrossRef] [PubMed]
- Klonsky, E.D. The functions of self-injury in young adults who cut themselves: Clarifying the evidence for affect regulation. Psychiatry Res. 2009, 166, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Klonsky, E.D.; Victor, S.E.; Saffer, B.Y. Nonsuicidal Self-Injury: What We Know, and What We Need to Know. Can. J. Psychiatry 2014, 59, 565–568. [Google Scholar] [CrossRef] [PubMed]
- Nock, M.K. Self-Injury. Annu. Rev. Clin. Psychol. 2010, 6, 339–363. [Google Scholar] [CrossRef] [PubMed]
- Nock, M.K.; Prinstein, M.J.; Sterba, S.K. Revealing the form and function of self-injurious thoughts and behaviors: A real-time ecological assessment study among adolescents and young adults. J. Psychol. 2009, 118, 816–827. [Google Scholar] [CrossRef]
- Victor, S.E.; Klonsky, E.D. Daily emotion in non-suicidal self-injury. J. Clin. Psychol. 2014, 70, 364–375. [Google Scholar] [CrossRef]
- Seltzer, M.M.; Taylor, J.L.; Baker, J.K.; Smith, L.E.; Greenberg, J.S. Change in maternal criticism and behavior problems in adolescents and adults with autism across a 7-year period. J. Psychol. 2011, 120, 465–475. [Google Scholar]
- Garcia-Villamisar, D.; Rojahn, J. Cormorbid psychopathology and stress mediate the relationship between autistic traits and repetitive behaviours in adults with autism. J. Intellect. Disabil. Res. 2015, 59, 116–124. [Google Scholar] [CrossRef]
- Smith, L.E.; Greenberg, J.S.; Seltzer, M.M.; Hong, J. Symptoms and Behavior Problems of Adolescents and Adults with Autism: Effects of Mother–Child Relationship Quality, Warmth, and Praise. Am. J. Retard. 2008, 113, 387–402. [Google Scholar] [CrossRef] [PubMed]
- Courchesne, E.; Mouton, P.R.; Calhoun, M.E.; Semendeferi, K.; Ahrens-Barbeau, C.; Hallet, M.J.; Barnes, C.C.; Pierce, K. Neuron Number and Size in Prefrontal Cortex of Children With Autism. JAMA 2011, 306, 2001. [Google Scholar] [CrossRef] [PubMed]
- Grayson, D.; Bliss-Moreau, E.; Bennett, J.; Lavenex, P.; Amaral, D. Neural Reorganization Due to Neonatal Amygdala Lesions in the Rhesus Monkey: Changes in Morphology and Network Structure. Cereb. Cortex 2017, 27, 3240–3253. [Google Scholar] [CrossRef] [PubMed]
- Vuilleumier, P.; Richardson, M.P.; Armony, J.L.; Driver, J.; Dolan, R.J. Distant influences of amygdala lesion on visual cortical activation during emotional face processing. Nat. Neurosci. 2004, 7, 1271–1278. [Google Scholar] [CrossRef] [PubMed]
- Bickart, K.C.; Wright, C.I.; Dautoff, R.J.; Dickerson, B.C.; Barrett, L.F. Amygdala volume and social network size in humans. Nat. Neurosci. 2011, 14, 163–164. [Google Scholar] [CrossRef] [PubMed]
- Sallet, J.; Mars, R.B.; Noonan, M.P.; Andersson, J.L.; O’Reilly, J.X.; Jbabdi, S.; Croxson, P.L.; Jenkinson, M.; Miller, K.L.; Rushworth, M.F.S. Social Network Size Affects Neural Circuits in Macaques. Science 2011, 334, 697–700. [Google Scholar] [CrossRef]
- Gerbella, M.; Borra, E.; Rozzi, S.; Luppino, G. Connections of the macaque Granular Frontal Opercular (GrFO) area: A possible neural substrate for the contribution of limbic inputs for controlling hand and face/mouth actions. Brain Struct. Funct. 2016, 221, 59–78. [Google Scholar] [CrossRef]
- Brass, M.; Derrfuss, J.; Forstmann, B.; Cramon, D.; Von Cramon, D.Y. The role of the inferior frontal junction area in cognitive control. Trends Cogn. Sci. 2005, 9, 314–316. [Google Scholar] [CrossRef]
- Patel, S.; Oishi, K.; Wright, A.; Sutherland-Foggio, H.; Saxena, S.; Sheppard, S.M.; Hillis, A.E. Right Hemisphere Regions Critical for Expression of Emotion Through Prosody. Front. Neurol. 2018, 9, 224. [Google Scholar] [CrossRef]
- Tyler, L.K.; Marslen-Wilson, W.D.; Randall, B.; Wright, P.; Devereux, B.J.; Zhuang, J.; Papoutsi, M.; Stamatakis, E.A. Left inferior frontal cortex and syntax: Function, structure and behaviour in patients with left hemisphere damage. Brain 2011, 134, 415–431. [Google Scholar] [CrossRef]
- Tyler, L.K.; Wright, P.; Randall, B.; Stamatakis, E.A.; Marslen-Wilson, W.D.; Marslen-Wilson, W.D.; Marslen-Wilson, W. Reorganization of syntactic processing following left-hemisphere brain damage: Does right-hemisphere activity preserve function? Brain 2010, 133, 3396–3408. [Google Scholar] [CrossRef] [PubMed]
- Garner, J.P. Perseveration and stereotypy–Systems-level insights from clinical psychology. In Stereotypic Animal Behaviour: Fundamentals & Applications to Welfare, 2nd ed.; Mason, G., Rushen, J., Eds.; CAB International: Wallingford, UK, 2006; pp. 121–152. [Google Scholar]
- Guehl, D.; Benazzouz, A.; Aouizerate, B.; Cuny, E.; Rotge, J.-Y.; Rougier, A.; Tignol, J.; Bioulac, B.; Burbaud, P. Neuronal Correlates of Obsessions in the Caudate Nucleus. Boil. Psychiatry 2008, 63, 557–562. [Google Scholar] [CrossRef] [PubMed]
- Whitehouse, C.M.; Lewis, M.H. Repetitive Behavior in Neurodevelopmental Disorders: Clinical and Translational Findings. Behav. Anal. 2015, 38, 163–178. [Google Scholar] [CrossRef] [PubMed]
- Hollander, E.; Anagnostou, E.; Chaplin, W.; Esposito, K.; Haznedar, M.M.; LiCalzi, E.; Wasserman, S.; Soorya, L.; Buchsbaum, M. Striatal Volume on Magnetic Resonance Imaging and Repetitive Behaviors in Autism. Boil. Psychiatry 2005, 58, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Langen, M.; Schnack, H.G.; Nederveen, H.; Bos, D.; Lahuis, B.E.; De Jonge, M.V.; Van Engeland, H.; Durston, S. Changes in the Developmental Trajectories of Striatum in Autism. Boil. Psychiatry. 2009, 66, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Langen, M.; Bos, D.; Noordermeer, S.D.; Nederveen, H.; Van Engeland, H.; Durston, S. Changes in the Development of Striatum Are Involved in Repetitive Behavior in Autism. Boil. Psychiatry. 2014, 76, 405–411. [Google Scholar] [CrossRef]
- Wolff, J.J.; Hazlett, H.C.; Lightbody, A.A.; Reiss, A.L.; Piven, J. Repetitive and self-injurious behaviors: Associations with caudate volume in autism and fragile X syndrome. J. Neurodev. Disord. 2013, 5, 12. [Google Scholar] [CrossRef] [PubMed]
- Calderoni, S.; Bellani, M.; Hardan, A.Y.; Muratori, F.; Brambilla, P. Basal ganglia and restricted and repetitive behaviours in Autism Spectrum Disorders: Current status and future perspectives. Epidemiol. Psychiatr. Sci. 2014, 23, 235–238. [Google Scholar] [CrossRef] [PubMed]
- Frodl, T.; O’Keane, V. How does the brain deal with cumulative stress? A review with focus on developmental stress, HPA axis function and hippocampal structure in humans. Neurobiol. Dis. 2013, 52, 24–37. [Google Scholar] [CrossRef]
- Kim, E.J.; Pellman, B.; Kim, J.J. Stress effects on the hippocampus: A critical review. Learn. Mem. 2015, 22, 411–416. [Google Scholar] [CrossRef]
- Bernasconi, N.; Caramanos, Z.; Antel, S.B.; Arnold, D.L.; Andermann, F. Mesial temporal damage in temporal lobe epilepsy: A volumetric MRI study of the hippocampus, amygdala and parahippocampal region. Brain 2003, 126, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Cendes, F. Progressive hippocampal and extrahippocampal atrophy in drug resistant epilepsy: Review. Curr. Opin. Neurol. 2005, 18, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Cormack, F.; Gadian, D.G.; Vargha-Khadem, F.; Cross, J.H.; Connelly, A.; Baldeweg, T. Extra-hippocampal grey matter density abnormalities in paediatric mesial temporal sclerosis. NeuroImage 2005, 27, 635–643. [Google Scholar] [CrossRef] [PubMed]
- Brandt, T.; Schautzer, F.; Hamilton, D.A.; Brüning, R.; Markowitsch, H.J.; Kalla, R.; Darlington, C.; Smith, P.; Strupp, M. Vestibular loss causes hippocampal atrophy and impaired spatial memory in humans. Brain 2005, 128, 2732–2741. [Google Scholar] [CrossRef] [PubMed]
- Lawrie, S.M. Parsing Heterogeneity. JAMA Psychiatry 2017, 74, 1089. [Google Scholar] [CrossRef] [PubMed]
- Kadohisa, M.; Verhagen, J.; Rolls, E.; Rolls, E. The primate amygdala: Neuronal representations of the viscosity, fat texture, temperature, grittiness and taste of foods. Neuroscience 2005, 132, 33–48. [Google Scholar] [CrossRef] [PubMed]
- Vogt, B.A. Regions and subregions of the cingulate cortex. In Cingulate Neurobiology & Disease; Vogt, B.A., Ed.; Oxford University Press: New York, NY, USA, 2009; pp. 3–30. [Google Scholar]
- Young, M.P.; Scanneil, J.W.; Burns, G.A.P.; Blakemore, C. Analysis of Connectivity: Neural Systems in the Cerebral Cortex. Rev. Neurosci. 1994, 5, 227–250. [Google Scholar] [CrossRef] [PubMed]
- Ziemann, A.E.; Allen, J.E.; Dahdaleh, N.S.; Drebot, I.I.; Coryell, M.W.; Wunsch, A.M.; Lynch, C.M.; Faraci, F.M.; Howard, M.A.; Welsh, M.J.; et al. The Amygdala Is a Chemosensor that Detects Carbon Dioxide and Acidosis to Elicit Fear Behavior. Cell 2009, 139, 1012–1021. [Google Scholar] [CrossRef] [PubMed]
- Warner, C.E.; Kwan, W.C.; Wright, D.; Johnston, L.A.; Egan, G.F.; Bourne, J.A. Preservation of Vision by the Pulvinar following Early-Life Primary Visual Cortex Lesions. Curr. Boil. 2015, 25, 424–434. [Google Scholar] [CrossRef] [PubMed]
- Paul, L.K.; Corsello, C.; Tranel, D.; Adolphs, R. Does bilateral damage to the human amygdala produce autistic symptoms? J. Neurodev. Disord. 2010, 2, 165–173. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Heberlein, A.S.; Adolphs, R. Impaired spontaneous anthropomorphizing despite intact perception and social knowledge. Proc. Acad. Sci. 2004, 101, 7487–7491. [Google Scholar] [CrossRef] [PubMed]
- Fatemi, S.H.; Aldinger, K.A.; Ashwood, P.; Bauman, M.L.; Blaha, C.D.; Blatt, G.J.; Chauhan, A.; Chauhan, V.; Dager, S.R.; Dickson, P.E.; et al. Consensus Paper: Pathological Role of the Cerebellum in Autism. Cerebellum 2012, 11, 777–807. [Google Scholar] [CrossRef] [PubMed]
- Whitney, E.R.; Kemper, T.L.; Bauman, M.L.; Rosene, D.L.; Blatt, G.J. Cerebellar Purkinje Cells are Reduced in a Subpopulation of Autistic Brains: A Stereological Experiment Using Calbindin-D28k. Cerebellum 2008, 7, 406–416. [Google Scholar] [CrossRef] [PubMed]
- Corkin, S.; Amaral, D.G.; Gonzalez, R.G.; Johnson, K.A.; Hyman, B.T. HM’s Medial Temporal Lobe Lesion: Findings from Magnetic Resonance Imaging. J. Neurosci. 1997, 17, 3964–3979. [Google Scholar] [CrossRef] [PubMed]
- Kansal, K.; Yang, Z.; Fishman, A.M.; Sair, H.I.; Ying, S.H.; Jedynak, B.M.; Prince, J.L.; Onyike, C.U. Structural cerebellar correlates of cognitive and motor dysfunctions in cerebellar degeneration. Brain 2017, 140, 707–720. [Google Scholar] [CrossRef] [PubMed]
- Hegarty, J.P.; Gu, M.; Spielman, D.M.; Cleveland, S.C.; Hallmayer, J.F.; Lazzeroni, L.C.; Raman, M.M.; Frazier, T.W.; Phillips, J.M.; Reiss, A.L.; et al. A proton MR spectroscopy study of the thalamus in twins with autism spectrum disorder. Prog. Neuropsychopharmacol Biol. Psychiatry 2018, 81, 153–160. [Google Scholar] [CrossRef]
- Sandin, S.; Lichtenstein, P.; Kuja-Halkola, R.; Larsson, H.; Hultman, C.M.; Reichenberg, A. THE FAMILIAL RISK OF AUTISM. JAMA 2014, 311, 1770–1777. [Google Scholar] [CrossRef] [PubMed]
- Amaral, D.G. Examining the causes of autism. Cerebrum 2017, 1, 1–12. [Google Scholar]
- Carvill, S. Sensory impairments, intellectual disability and psychiatry. J. Intellect. Disabil. 2001, 45, 467–483. [Google Scholar] [CrossRef]
- Gainotti, G.; Guido, G. The Role of Body-Related and Environmental Sources of Knowledge in the Construction of Different Conceptual Categories. Front. Psychol. 2012, 3, 430. [Google Scholar] [CrossRef]
- Taylor, K.I.; Moss, H.E.; Tyler, L.K. The conceptual structure account: A cognitive model of semantic memory and its neural instantiation. In Neural Basis of Semantic Memory; Hart, J., Kraut, M.A., Eds.; Cambridge University Press: Cambridge, UK, 2007; pp. 265–301. [Google Scholar]
- Arana, F.S.; Parkinson, J.A.; Hinton, E.; Holland, A.J.; Owen, A.M.; Roberts, A.C. Dissociable Contributions of the Human Amygdala and Orbitofrontal Cortex to Incentive Motivation and Goal Selection. J. Neurosci. 2003, 23, 9632–9638. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.L. The Neuropsychiatric Inventory: Assessing psychopathology in dementia patients. Neurology 1997, 48, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Owens, J.A.; Dalzell, V. Use of the ‘BEARS’ sleep screening tool in a pediatric residents’ continuity clinic: A pilot study. Sleep Med. 2005, 6, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Maenner, M.J.; Smith, L.E.; Hong, J.; Makuch, R.; Greenberg, J.S.; Mailick, M.R. Evaluation of an activities of daily living scale for adolescents and adults with developmental disabilities. Disabil. Health J. 2013, 6, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Reitan, R.M.; Wolfson, D. The Halstead-Reitan Neuropsychological Test Battery: Theory & Clinical Interpretation; Neuropsychology Press: Tucson, AZ, USA, 1985. [Google Scholar]
- Reitan, R.M.; Wolfson, D. The Halstead-Reitan Neuropsychological Test Battery: Theory & Clinical Interpretation, 2nd ed.; Neuropsychology Press: Tucson, AZ, USA, 1993. [Google Scholar]
- Lam, K.S.L.; Aman, M.G. The Repetitive Behavior Scale-Revised: Independent validation in individuals with autism spectrum disorders. J. Autism Dev. Disord. 2007, 37, 855–866. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, M.S.; Velicer, W.F.; Intille, S.S. Telemetric monitoring in the behavior sciences. Behav. Res. Methods 2008, 40, 328–341. [Google Scholar] [CrossRef]
- Rutter, M. Changing concepts and findings on autism. J. Autism Dev. Disord. 2013, 43, 1749–1757. [Google Scholar] [CrossRef]
- Frazier, T.W.; Klingemier, E.W.; Beukemann, M.; Speer, L.; Markowitz, L.; Parikh, S.; Wexberg, S.; Giuliano, K.; Schulte, E.; Delahunty, C.; et al. Development of an Objective Autism Risk Index Using Remote Eye Tracking. J. Am. Acad. Child Adolesc. Psychiatry 2016, 55, 301–309. [Google Scholar] [CrossRef]
- Cameron, O.G. Interoception: The Inside Story—A Model for Psychosomatic Processes. Psychosom. Med. 2001, 63, 697–710. [Google Scholar] [CrossRef]
- Wolff, J.J.; Boyd, B.A.; Elison, J.T. A quantitative measure of restricted and repetitive behaviors for early childhood. J. Neurodev. Disord. 2016, 8, 572. [Google Scholar] [CrossRef]
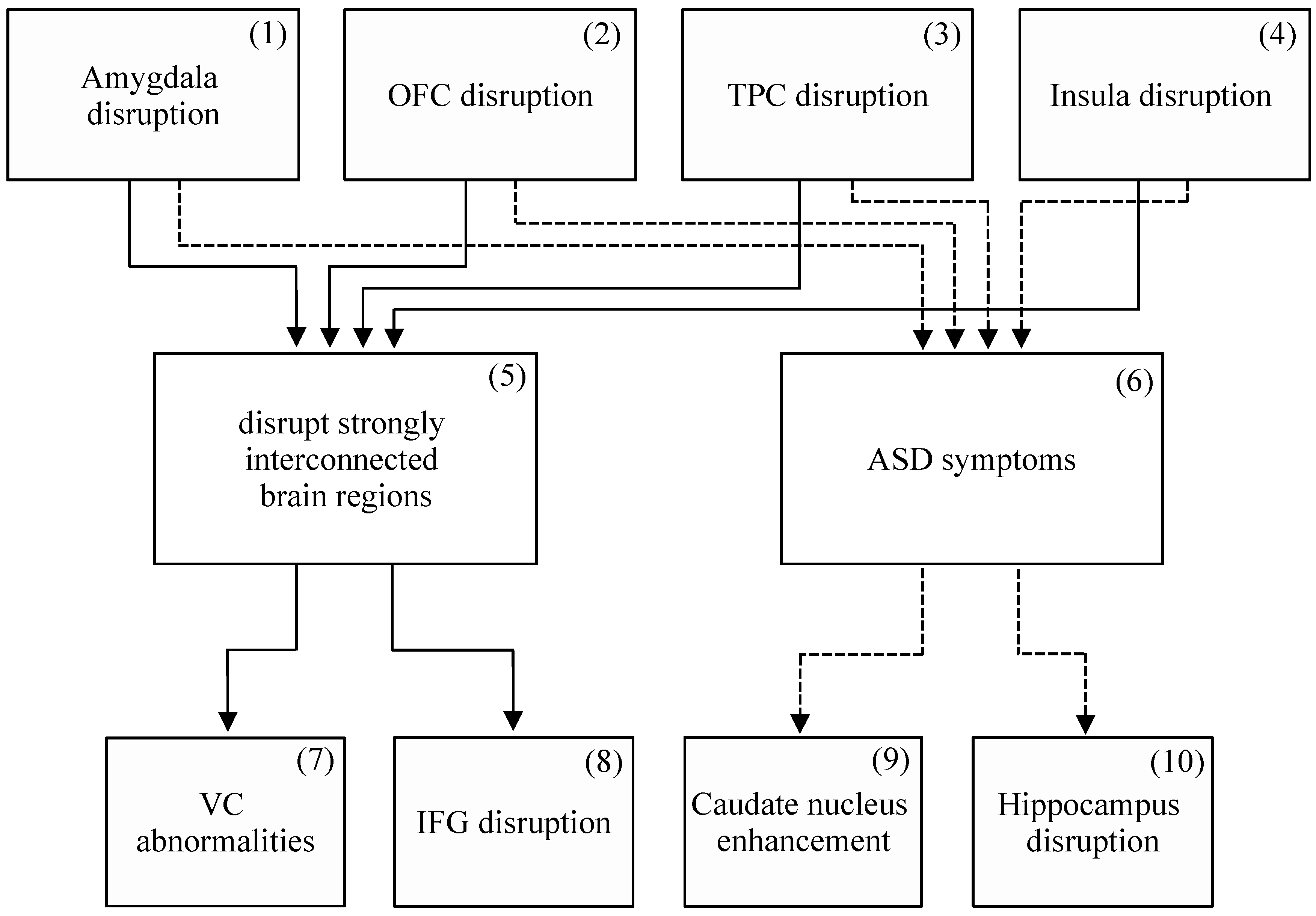
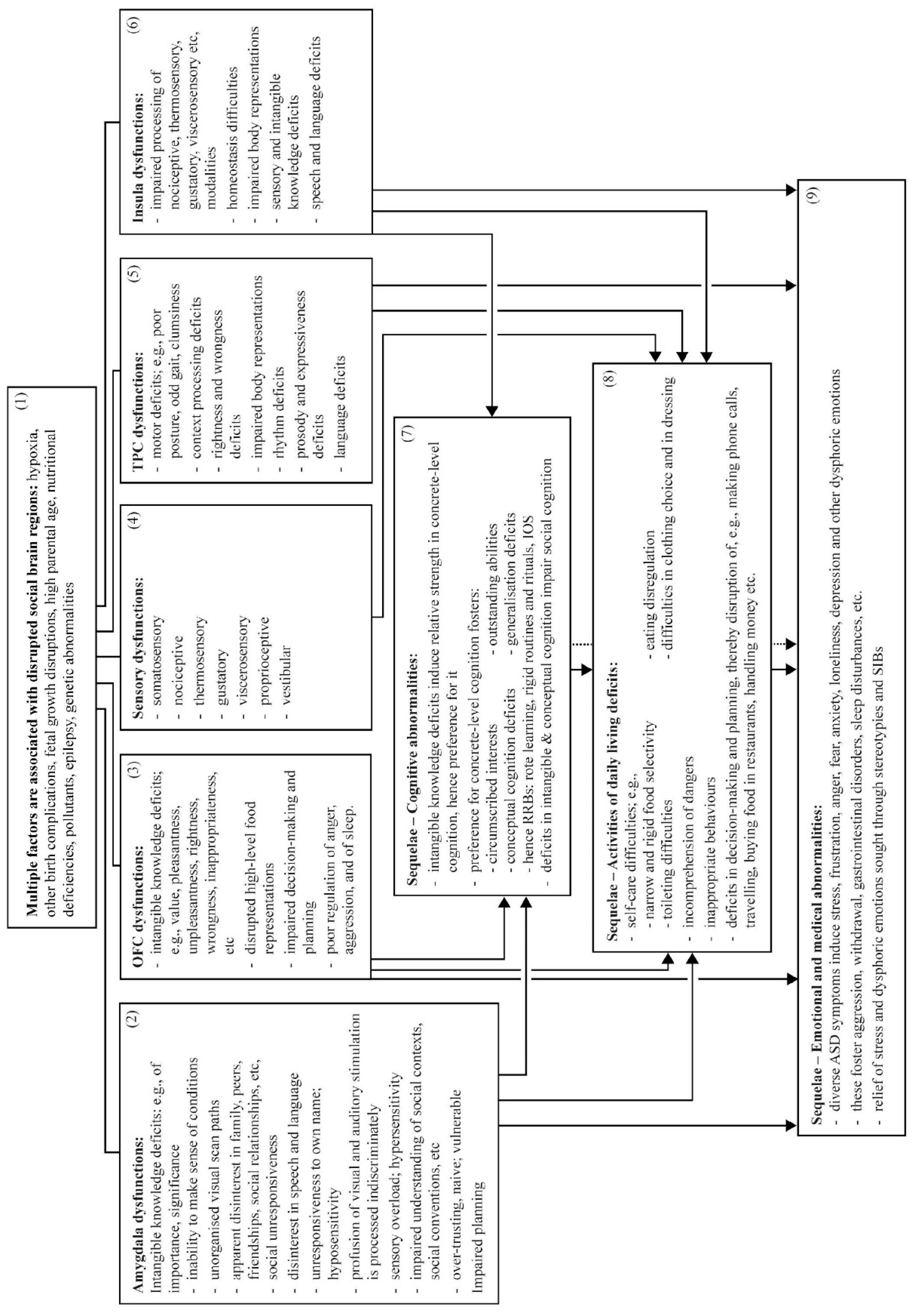
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weston, C.S.E. Four Social Brain Regions, Their Dysfunctions, and Sequelae, Extensively Explain Autism Spectrum Disorder Symptomatology. Brain Sci. 2019, 9, 130. https://doi.org/10.3390/brainsci9060130
Weston CSE. Four Social Brain Regions, Their Dysfunctions, and Sequelae, Extensively Explain Autism Spectrum Disorder Symptomatology. Brain Sciences. 2019; 9(6):130. https://doi.org/10.3390/brainsci9060130
Chicago/Turabian StyleWeston, Charles S. E. 2019. "Four Social Brain Regions, Their Dysfunctions, and Sequelae, Extensively Explain Autism Spectrum Disorder Symptomatology" Brain Sciences 9, no. 6: 130. https://doi.org/10.3390/brainsci9060130
APA StyleWeston, C. S. E. (2019). Four Social Brain Regions, Their Dysfunctions, and Sequelae, Extensively Explain Autism Spectrum Disorder Symptomatology. Brain Sciences, 9(6), 130. https://doi.org/10.3390/brainsci9060130




