Evaluation of Postnatal Sedation in Full-Term Infants
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Chart Review
2.3. Statistical Analysis
3. Results
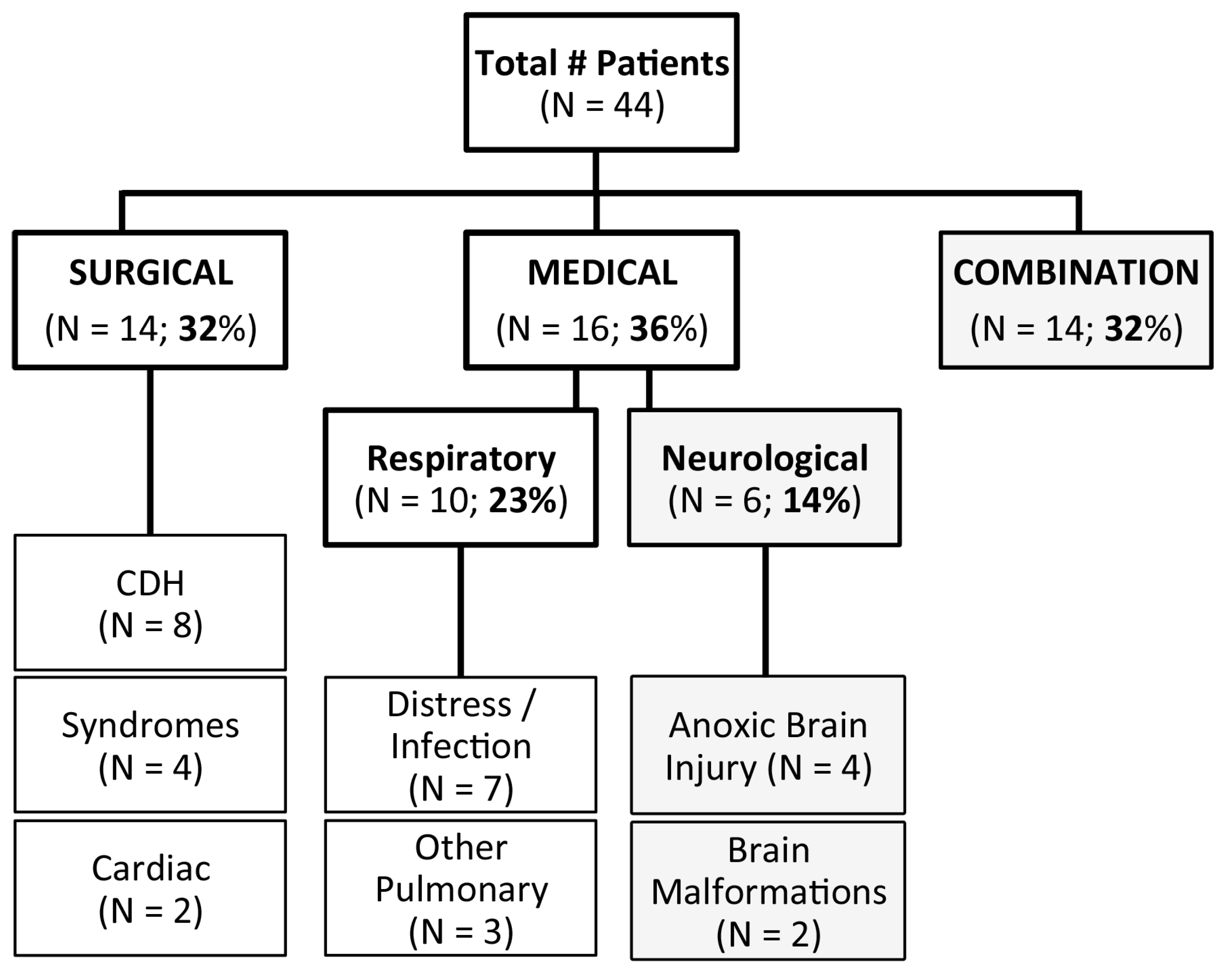
3.1. Patient Diagnoses
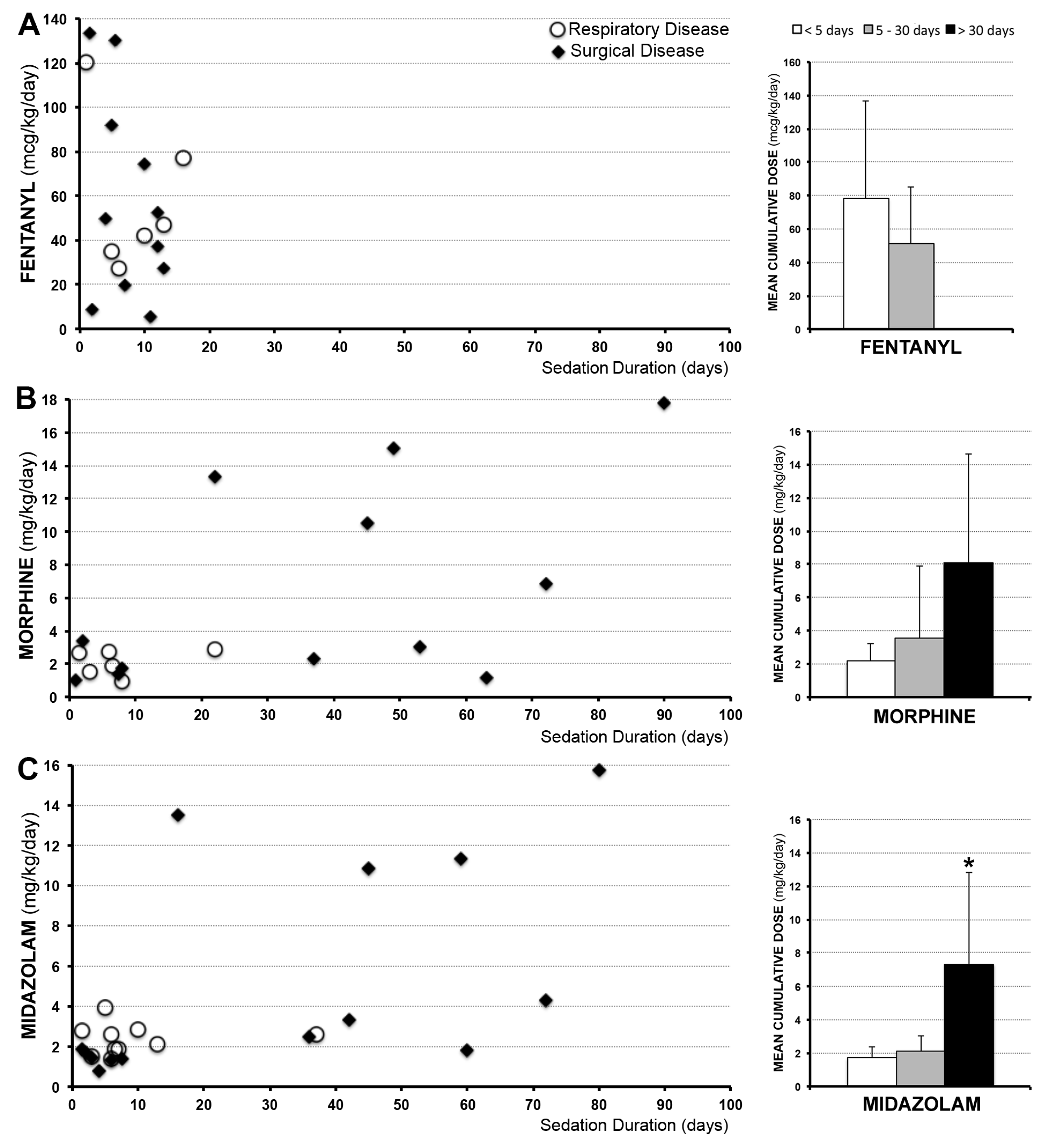
3.2. Sedation Drugs
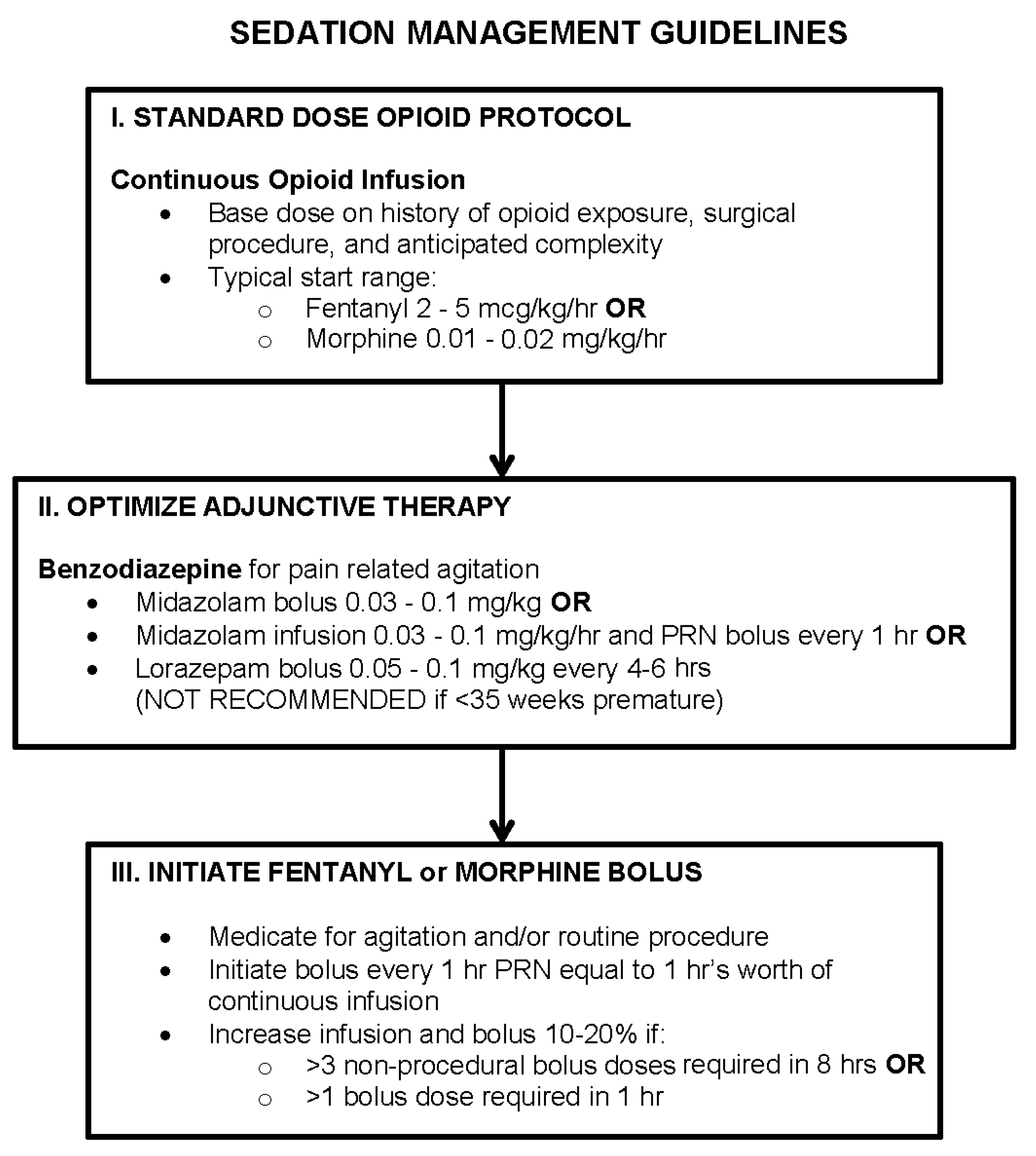
3.3. Sedation Management
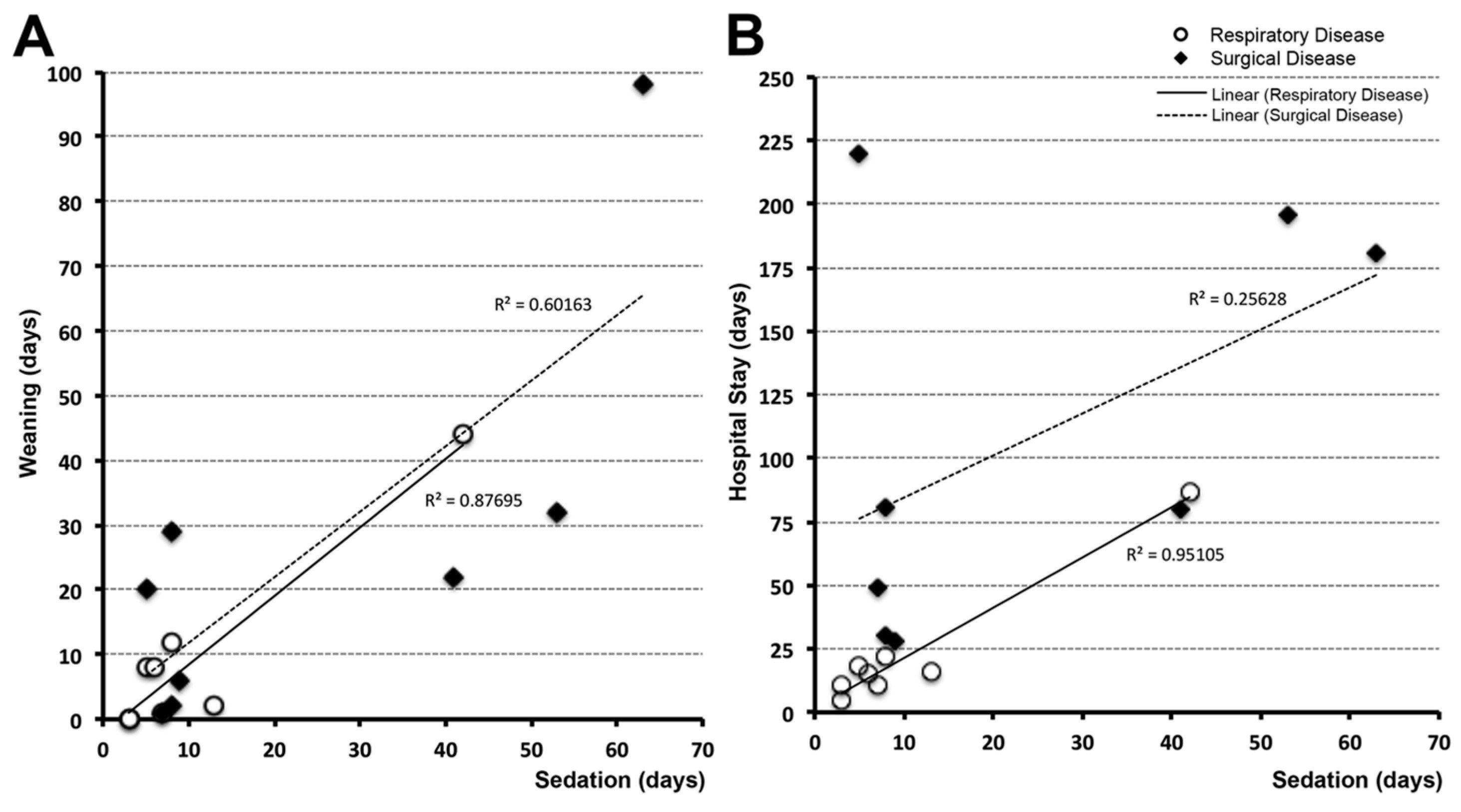
3.4. Time Correlations
4. Discussion
4.1. Limitations of the Retrospective Chart Review
4.1.1. Study Size
4.1.2. Withdrawal Evaluation
4.2. Sedation of Full-Term Infants
4.3. Sequalae of Prolonged Postnatal Opioid and Benzodiazepine Exposure
4.3.1. Opioids
4.3.2. Benzodiazepines
4.3.3. Future Studies
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CDH | congenital diaphragmatic hernia |
| CICU | cardiac intensive care unit |
| i2b2 | Informatics for Integrating Biology and the Bedside |
| MICU | medical intensive care unit |
| NEOPAN | Neurological Outcomes and Preemptive Analgesia in Neonates |
| NICU | neonatal intensive care unit |
| MOTIF | Measuring Opioid Tolerance Induced by Fentanyl (or morphine) |
References
- Dilworth, N.M.; MacKellar, A. Pain relief for the pediatric surgical patient. J. Pediatr. Surg. 1987, 22, 264–266. [Google Scholar] [CrossRef]
- Berde, C.B.; Sethna, N.F. Analgesics for the treatment of pain in children. N. Engl. J. Med. 2002, 347, 1094–1103. [Google Scholar] [CrossRef] [PubMed]
- Anand, K.J.; Arnold, J.H. Opioid tolerance and dependence in infants and children. Crit. Care Med. 1994, 22, 334–342. [Google Scholar] [CrossRef] [PubMed]
- Anand, K.J.; Willson, D.F.; Berger, J.; Harrison, R.; Meert, K.L.; Zimmerman, J.; Carcillo, J.; Newth, C.J.; Prodhan, P.; Dean, J.M.; et al. Tolerance and withdrawal from prolonged opioid use in critically ill children. Pediatrics 2010, 125, e1208–e1225. [Google Scholar] [CrossRef]
- Lewis, T.; Erfe, B.L.; Ezell, T.; Gauda, E. Pharmacoepidemiology of opiate use in the neonatal ICU: Increasing cumulative doses and iatrogenic opiate withdrawal. J. Opioid Manag. 2015, 11, 305–312. [Google Scholar] [CrossRef]
- Arnold, J.H.; Truog, R.D.; Scavone, J.M.; Fenton, T. Changes in the pharmacodynamic response to fentanyl in neonates during continuous infusion. J. Pediatr. 1991, 119, 639–643. [Google Scholar] [CrossRef]
- Anand, K.J.; Hall, R.W.; Desai, N.; Shephard, B.; Bergqvist, L.L.; Young, T.E.; Boyle, E.M.; Carbajal, R.; Bhutani, V.K.; Moore, M.B.; et al. Effects of morphine analgesia in ventilated preterm neonates: Primary outcomes from the neopain randomised trial. Lancet 2004, 363, 1673–1682. [Google Scholar] [CrossRef]
- Steinhorn, R.; McPherson, C.; Anderson, P.J.; Neil, J.; Doyle, L.W.; Inder, T. Neonatal morphine exposure in very preterm infants-cerebral development and outcomes. J. Pediatr. 2015, 166, 1200–1207. [Google Scholar] [CrossRef] [PubMed]
- Hall, R.W.; Kronsberg, S.S.; Barton, B.A.; Kaiser, J.R.; Anand, K.J.; Group, N.T.I. Morphine, hypotension, and adverse outcomes among preterm neonates: Who’s to blame? Secondary results from the neopain trial. Pediatrics 2005, 115, 1351–1359. [Google Scholar] [CrossRef] [PubMed]
- De Graaf, J.; van Lingen, R.A.; Simons, S.H.; Anand, K.J.; Duivenvoorden, H.J.; Weisglas-Kuperus, N.; Roofthooft, D.W.; Groot Jebbink, L.J.; Veenstra, R.R.; Tibboel, D.; et al. Long-term effects of routine morphine infusion in mechanically ventilated neonates on children’s functioning: Five-year follow-up of a randomized controlled trial. Pain 2011, 152, 1391–1397. [Google Scholar] [CrossRef]
- Ferguson, S.A.; Ward, W.L.; Paule, M.G.; Hall, R.W.; Anand, K.J. A pilot study of preemptive morphine analgesia in preterm neonates: Effects on head circumference, social behavior, and response latencies in early childhood. Neurotoxicol. Teratol. 2012, 34, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, M.B.; West, M.A.; Nathens, A.B.; Harbrecht, B.G.; Moore, F.A.; Bankey, P.E.; Freeman, B.; Johnson, J.L.; McKinley, B.A.; Minei, J.P.; et al. V. Guidelines for sedation and analgesia during mechanical ventilation general overview. J. Trauma 2007, 63, 945–950. [Google Scholar] [CrossRef]
- Bellu, R.; de Waal, K.A.; Zanini, R. Opioids for neonates receiving mechanical ventilation. Cochrane Database Syst. Rev. 2008, CD004212. [Google Scholar] [CrossRef]
- Anand, K.J.; Barton, B.A.; McIntosh, N.; Lagercrantz, H.; Pelausa, E.; Young, T.E.; Vasa, R. Analgesia and sedation in preterm neonates who require ventilatory support: Results from the nopain trial. Neonatal outcome and prolonged analgesia in neonates. Arch. Pediatr. Adolesc. Med. 1999, 153, 331–338. [Google Scholar] [CrossRef]
- Dewey, W.L. Various factors which affect the rate of development of tolerance and physical dependence to abused drugs. NIDA Res. Monogr. 1984, 54, 39–49. [Google Scholar] [PubMed]
- Hess, D.R. Retrospective studies and chart reviews. Respir. Care 2004, 49, 1171–1174. [Google Scholar] [PubMed]
- Worster, A.; Haines, T. Advanced statistics: Understanding medical record review (MRR) studies. Acad. Emerg. Med. 2004, 11, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Ashton, C.M. Chart review. A need for reappraisal. Eval. Health Prof. 1997, 20, 146–163. [Google Scholar] [CrossRef]
- Vet, N.J.; Kleiber, N.; Ista, E.; de Hoog, M.; de Wildt, S.N. Sedation in critically ill children with respiratory failure. Front. Pediatr. 2016, 4, 89. [Google Scholar] [CrossRef]
- Gearing, R.E.; Mian, I.A.; Barber, J.; Ickowicz, A. A methodology for conducting retrospective chart review research in child and adolescent psychiatry. J. Can. Acad. Child Adolesc. Psychiatry 2006, 15, 126–134. [Google Scholar]
- Jansen, A.C.; van Aalst-Cohen, E.S.; Hutten, B.A.; Buller, H.R.; Kastelein, J.J.; Prins, M.H. Guidelines were developed for data collection from medical records for use in retrospective analyses. J. Clin. Epidemiol. 2005, 58, 269–274. [Google Scholar] [CrossRef]
- Haynes, R.B.; Sackett, D.L.; Guyatt, G.H.; Tugwell, P. Clinical Epidemiology: How to Do Clinical Practice Research, 3rd ed.; Lippincott Williams and Wilkins: Philadelphia, PA, USA, 2006. [Google Scholar]
- Findley, T.W.; Daum, M.C. Research in physical medicine and rehabilitation. III. The chart review or how to use clinical data for exploratory retrospective studies. Am. J. Phys. Med. Rehabil. 1989, 68, 150–157. [Google Scholar] [CrossRef]
- Harrell, F.E., Jr.; Lee, K.L.; Matchar, D.B.; Reichert, T.A. Regression models for prognostic prediction: Advantages, problems, and suggested solutions. Cancer Treat Rep. 1985, 69, 1071–1077. [Google Scholar]
- Raykov, T.; Widaman, K.F. Issues in applied equation modeling research. Struct. Equ. Modeling 1995, 2, 289–318. [Google Scholar] [CrossRef]
- Franck, L.S.; Scoppettuolo, L.A.; Wypij, D.; Curley, M.A. Validity and generalizability of the withdrawal assessment tool-1 (WAT-1) for monitoring iatrogenic withdrawal syndrome in pediatric patients. Pain 2012, 153, 142–148. [Google Scholar] [CrossRef]
- Curley, M.A.; Wypij, D.; Watson, R.S.; Grant, M.J.; Asaro, L.A.; Cheifetz, I.M.; Dodson, B.L.; Franck, L.S.; Gedeit, R.G.; Angus, D.C.; et al. Protocolized sedation vs usual care in pediatric patients mechanically ventilated for acute respiratory failure: A randomized clinical trial. JAMA 2015, 313, 379–389. [Google Scholar] [CrossRef]
- Arnold, J.H.; Truog, R.D.; Orav, E.J.; Scavone, J.M.; Hershenson, M.B. Tolerance and dependence in neonates sedated with fentanyl during extracorporeal membrane oxygenation. Anesthesiology 1990, 73, 1136–1140. [Google Scholar] [CrossRef]
- Kudchadkar, S.R.; Yaster, M.; Punjabi, N.M. Sedation, sleep promotion, and delirium screening practices in the care of mechanically ventilated children: A wake-up call for the pediatric critical care community. Crit. Care Med. 2014, 42, 1592–1600. [Google Scholar] [CrossRef]
- Best, K.M.; Asaro, L.A.; Franck, L.S.; Wypij, D.; Curley, M.A.; Randomized Evaluation of Sedation Titration for Respiratory Failure Baseline Study Investigators. Patterns of sedation weaning in critically ill children recovering from acute respiratory failure. Pediatr. Crit. Care Med. 2016, 17, 19–29. [Google Scholar] [CrossRef]
- Anand, K.J.; Clark, A.E.; Willson, D.F.; Berger, J.; Meert, K.L.; Zimmerman, J.J.; Harrison, R.; Carcillo, J.A.; Newth, C.J.; Bisping, S.; et al. Opioid analgesia in mechanically ventilated children: Results from the multicenter measuring opioid tolerance induced by fentanyl study. Pediatr. Crit. Care Med. 2013, 14, 27–36. [Google Scholar] [CrossRef]
- Jenkins, I.A.; Playfor, S.D.; Bevan, C.; Davies, G.; Wolf, A.R. Current United Kingdom sedation practice in pediatric intensive care. Paediatr. Anaesth. 2007, 17, 675–683. [Google Scholar] [CrossRef]
- Twite, M.D.; Rashid, A.; Zuk, J.; Friesen, R.H. Sedation, analgesia, and neuromuscular blockade in the pediatric intensive care unit: Survey of fellowship training programs. Pediatr. Crit. Care Med. 2004, 5, 521–532. [Google Scholar] [CrossRef]
- Walhovd, K.B.; Moe, V.; Slinning, K.; Due-Tonnessen, P.; Bjornerud, A.; Dale, A.M.; van der Kouwe, A.; Quinn, B.T.; Kosofsky, B.; Greve, D.; et al. Volumetric cerebral characteristics of children exposed to opiates and other substances in utero. Neuroimage 2007, 36, 1331–1344. [Google Scholar] [CrossRef] [PubMed]
- Walhovd, K.B.; Moe, V.; Slinning, K.; Siqveland, T.; Fjell, A.M.; Bjornebekk, A.; Smith, L. Effects of prenatal opiate exposure on brain development—A call for attention. Nat. Rev. Neurosci. 2009, 10, 390. [Google Scholar] [CrossRef] [PubMed]
- Roze, J.C.; Denizot, S.; Carbajal, R.; Ancel, P.Y.; Kaminski, M.; Arnaud, C.; Truffert, P.; Marret, S.; Matis, J.; Thiriez, G.; et al. Prolonged sedation and/or analgesia and 5-year neurodevelopment outcome in very preterm infants: Results from the epipage cohort. Arch. Pediatr. Adolesc. Med. 2008, 162, 728–733. [Google Scholar] [CrossRef] [PubMed]
- De Graaf, J.; van Lingen, R.A.; Valkenburg, A.J.; Weisglas-Kuperus, N.; Groot Jebbink, L.; Wijnberg-Williams, B.; Anand, K.J.; Tibboel, D.; van Dijk, M. Does neonatal morphine use affect neuropsychological outcomes at 8 to 9 years of age? Pain 2013, 154, 449–458. [Google Scholar] [CrossRef]
- McGlone, L.; Mactier, H.; Weaver, L.T. Drug misuse in pregnancy: Losing sight of the baby? Arch. Dis. Child 2009, 94, 708–712. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.H.; Sweitzer, S.M. Neonatal morphine enhances nociception and decreases analgesia in young rats. Brain Res. 2008, 1199, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Ferenczi, S.; Nunez, C.; Pinter-Kubler, B.; Foldes, A.; Martin, F.; Markus, V.L.; Milanes, M.V.; Kovacs, K.J. Changes in metabolic-related variables during chronic morphine treatment. Neurochem. Int. 2010, 57, 323–330. [Google Scholar] [CrossRef]
- Anderson, E.M.; Neubert, J.K.; Caudle, R.M. Long-term changes in reward-seeking following morphine withdrawal are associated with altered n-methyl-d-aspartate receptor 1 splice variants in the amygdala. Neuroscience 2012, 223, 45–55. [Google Scholar] [CrossRef]
- Hintz, S.R.; Barnes, P.D.; Bulas, D.; Slovis, T.L.; Finer, N.N.; Wrage, L.A.; Das, A.; Tyson, J.E.; Stevenson, D.K.; Carlo, W.A.; et al. Neuroimaging and neurodevelopmental outcome in extremely preterm infants. Pediatrics 2015, 135, e32–e42. [Google Scholar] [CrossRef]
- Kiechl-Kohlendorfer, U.; Ralser, E.; Pupp Peglow, U.; Reiter, G.; Trawoger, R. Adverse neurodevelopmental outcome in preterm infants: Risk factor profiles for different gestational ages. Acta Paediatr. 2009, 98, 792–796. [Google Scholar] [CrossRef]
- Duerden, E.G.; Guo, T.; Dodbiba, L.; Chakravarty, M.M.; Chau, V.; Poskitt, K.J.; Synnes, A.; Grunau, R.E.; Miller, S.P. Midazolam dose correlates with abnormal hippocampal growth and neurodevelopmental outcome in preterm infants. Ann. Neurol. 2016, 79, 548–559. [Google Scholar] [CrossRef] [PubMed]
- Andropoulos, D.B.; Easley, R.B.; Brady, K.; McKenzie, E.D.; Heinle, J.S.; Dickerson, H.A.; Shekerdemian, L.; Meador, M.; Eisenman, C.; Hunter, J.V.; et al. Changing expectations for neurological outcomes after the neonatal arterial switch operation. Ann. Thorac. Surg. 2012, 94, 1250–1255. [Google Scholar] [CrossRef] [PubMed]
- Andropoulos, D.B.; Easley, R.B.; Brady, K.; McKenzie, E.D.; Heinle, J.S.; Dickerson, H.A.; Shekerdemian, L.S.; Meador, M.; Eisenman, C.; Hunter, J.V.; et al. Neurodevelopmental outcomes after regional cerebral perfusion with neuromonitoring for neonatal aortic arch reconstruction. Ann. Thorac. Surg. 2013, 95, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Garcia Guerra, G.; Robertson, C.M.; Alton, G.Y.; Joffe, A.R.; Cave, D.A.; Yasmin, F.; Dinu, I.A.; Creighton, D.E.; Ross, D.B.; Rebeyka, I.M.; et al. Neurotoxicity of sedative and analgesia drugs in young infants with congenital heart disease: 4-year follow-up. Paediatr. Anaesth. 2014, 24, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Guerra, G.G.; Robertson, C.M.; Alton, G.Y.; Joffe, A.R.; Cave, D.A.; Dinu, I.A.; Creighton, D.E.; Ross, D.B.; Rebeyka, I.M.; Western Canadian Complex Pediatric Therapies Follow-up Group. Neurodevelopmental outcome following exposure to sedative and analgesic drugs for complex cardiac surgery in infancy. Paediatr. Anaesth. 2011, 21, 932–941. [Google Scholar] [CrossRef] [PubMed]
- Mongerson, C.R.L.; Wilcox, S.L.; Goins, S.M.; Pier, D.B.; Zurakowski, D.; Jennings, R.W.; Bajic, D. Infant brain structural mri analysis in the context of thoracic noncardiac surgery and critical care. Front. Pediatr. Neurol. 2019, Accepted. [Google Scholar]
- Stolwijk, L.J.; Keunen, K.; de Vries, L.S.; Groenendaal, F.; van der Zee, D.C.; van Herwaarden, M.Y.A.; Lemmers, P.M.A.; Benders, M. Neonatal surgery for noncardiac congenital anomalies: Neonates at risk of brain injury. J. Pediatr. 2017, 182, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Stolwijk, L.J.; Lemmers, P.M.; Harmsen, M.; Groenendaal, F.; de Vries, L.S.; van der Zee, D.C.; Benders, M.J.; van Herwaarden-Lindeboom, M.Y. Neurodevelopmental outcomes after neonatal surgery for major noncardiac anomalies. Pediatrics 2016, 137, e20151728. [Google Scholar] [CrossRef]
| Disease Category | ||
|---|---|---|
| Characteristics | Surgical (N = 14) | Respiratory (N = 10) |
| Male Gender—n (%) | 11 (79%) | 5 (50%) |
| Gestational age at birth (wks) ± SD | 39.1 ± 1.6 | 39.3 ± 1.1 |
| Multiple births— n (%) | 0 | 1 (10%) |
| Birth by C/S— n (%) | 6 (43%) | 2 (20%) |
| # | Sedation/Intubation (days) | LOS | Location | Drugs | Average Dose (unit/kg/day) | Other Sedatives | Weaning |
|---|---|---|---|---|---|---|---|
| Respiratory Distress/Infection | |||||||
| 1 | 3 | 4 | MSICU | Morphine | 2.67 mg | None | No |
| Midazolam | 2.80 mg | ||||||
| 2 | 3 | 10 | MSICU | Morphine | 1.56 mg | None | No |
| Midazolam | 1.52 mg | ||||||
| 3 | 5 | 17 | NICU | Fentanyl | 35.21 mcg | Lorazepam | Yes |
| Midazolam | 3.96 mg | ||||||
| 4 | 7 | 8 | MSICU | Morphine | 1.92 mg | Lorazepam | Yes |
| Midazolam | 1.88 mg | ||||||
| 5 | 7 | 10 | MSICU | Morphine | 2.72 mg | Lorazepam | Yes |
| Midazolam | 2.63 mg | ||||||
| 6 | 13 | 15 | NICU | Fentanyl | 46.86 mcg | Lorazepam | Yes |
| Midazolam | 2.11 mg | ||||||
| 7 | 14 | 14 | MSICU | Fentanyl | 27.20 mcg | Clonidine, dexmedetomidine (infusion), lorazepam | Yes |
| Midazolam | 1.37 mg | ||||||
| Other Pulmonary Disease | |||||||
| 8 | 9 | 21 | MSICU | Fentanyl | 120.14 mcg | Lorazepam | Yes |
| Morphine | 0.95 mg | ||||||
| Midazolam | 1.88 mg | ||||||
| 9 | 10 | 10 | NICU | Fentanyl | 42.01 mcg | None | Yes |
| Midazolam | 2.82 mg | ||||||
| 10 | 37 | 86 | MSICU | Fentanyl | 77.20 mcg | Clonidine (patch) | Yes |
| Morphine | 2.91 mg | ||||||
| Midazolam | 2.60 mg | ||||||
| # | Sedation/Intubation (days) | LOS | Location | Drugs | Average Dose (unit/kg/day) | Other Sedatives | Weaning |
|---|---|---|---|---|---|---|---|
| Syndromes | |||||||
| 1 | 4 | 170 | MSICU | Fentanyl | 27.24 mcg | None | No |
| Midazolam | 1.47 mg | ||||||
| 2 | 5 | 219 | MSICU | Fentanyl | 91.96 mcg | Oxycodone (liquid) | Yes |
| Morphine | 1.03 mg | ||||||
| Midazolam | 0.81 mg | ||||||
| 3 | 7 | 48 | NICU | Fentanyl | 19.71 mcg | None | Yes |
| Midazolam | 1.70 mg | ||||||
| 4 | 8 | 80 | NICU | Morphine | 1.38 mg | None | Yes |
| Midazolam | 1.41 mg | ||||||
| Cardiac | |||||||
| 5 | 8 | 164 | CICU | Fentanyl | 133.25 mcg | Dexmedetomidine (infusion), lorazepam (liquid and bolus), methadone (liquid and bolus) | Yes |
| Morphine | 1.73 mg | ||||||
| Midazolam | 1.31 mg | ||||||
| 6 | 9 | 27 | CICU | Fentanyl | 50.00 mcg | None | Yes |
| Morphine | 3.40 mg | ||||||
| Midazolam | 1.90 mg | ||||||
| Congenital Diaphragmatic Hernia | |||||||
| 7 | 27 | 115 | MICU | Fentanyl | 130.12 mcg | Dexmedetomidine (infusion), ketamine, lorazepam, methadone | Yes |
| Morphine | 13.29 mg | ||||||
| Midazolam | 13.53 mg | ||||||
| 8 | 41 | 105 | MSICU | Morphine | 2.29 mg | Clonidine (patch and liquid), lorazepam (liquid and bolus), methadone (liquid) | Yes |
| Midazolam | 2.50 mg | ||||||
| 9* | 45 | 45 | MSICU | Fentanyl | 37.15 mcg | Dexmedetomidine (infusion), lorazepam | Yes |
| Morphine | 10.51 mg | ||||||
| Midazolam | 10.84 mg | ||||||
| 10 | 53 | 53 | MSICU | Morphine | 3.05 mg | Clonidine (patch), dexmedetomidine (infusion), ketamine, lorazepam (liquid and bolus) | Yes |
| Midazolam | 3.33 mg | ||||||
| 11 | 63 | 148 | MSICU | Fentanyl | 74.54 mcg | Clonidine (patch and liquid), dexmedetomidine (infusion), ketamine, lorazepam (liquid) | Yes |
| Morphine | 15.06 mg | ||||||
| Midazolam | 11.32 mg | ||||||
| 12 | 64 | 91 | MSICU | Fentanyl | 8.53 mcg | Clonidine (patch), lorazepam (liquid) | Yes |
| Morphine | 1.16 mg | ||||||
| Midazolam | 1.83 mg | ||||||
| 13 * | 70 | 71 | MSICU | Fentanyl | 52.44 mcg | None | Yes |
| Morphine | 6.82 mg | ||||||
| Midazolam | 4.30 mg | ||||||
| 14 | 90 | 248 | MSICU | Fentanyl | 5.40 mcg | Dexmedetomidine (infusion), lorazepam, methadone | Yes |
| Morphine | 17.77 mg | ||||||
| Midazolam | 15.75 mg | ||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Solodiuk, J.C.; Jennings, R.W.; Bajic, D. Evaluation of Postnatal Sedation in Full-Term Infants. Brain Sci. 2019, 9, 114. https://doi.org/10.3390/brainsci9050114
Solodiuk JC, Jennings RW, Bajic D. Evaluation of Postnatal Sedation in Full-Term Infants. Brain Sciences. 2019; 9(5):114. https://doi.org/10.3390/brainsci9050114
Chicago/Turabian StyleSolodiuk, Jean Carmela, Russell William Jennings, and Dusica Bajic. 2019. "Evaluation of Postnatal Sedation in Full-Term Infants" Brain Sciences 9, no. 5: 114. https://doi.org/10.3390/brainsci9050114
APA StyleSolodiuk, J. C., Jennings, R. W., & Bajic, D. (2019). Evaluation of Postnatal Sedation in Full-Term Infants. Brain Sciences, 9(5), 114. https://doi.org/10.3390/brainsci9050114





