Neuroprotective and Cognitive Enhancement Potentials of Baicalin: A Review
Abstract
1. Introduction
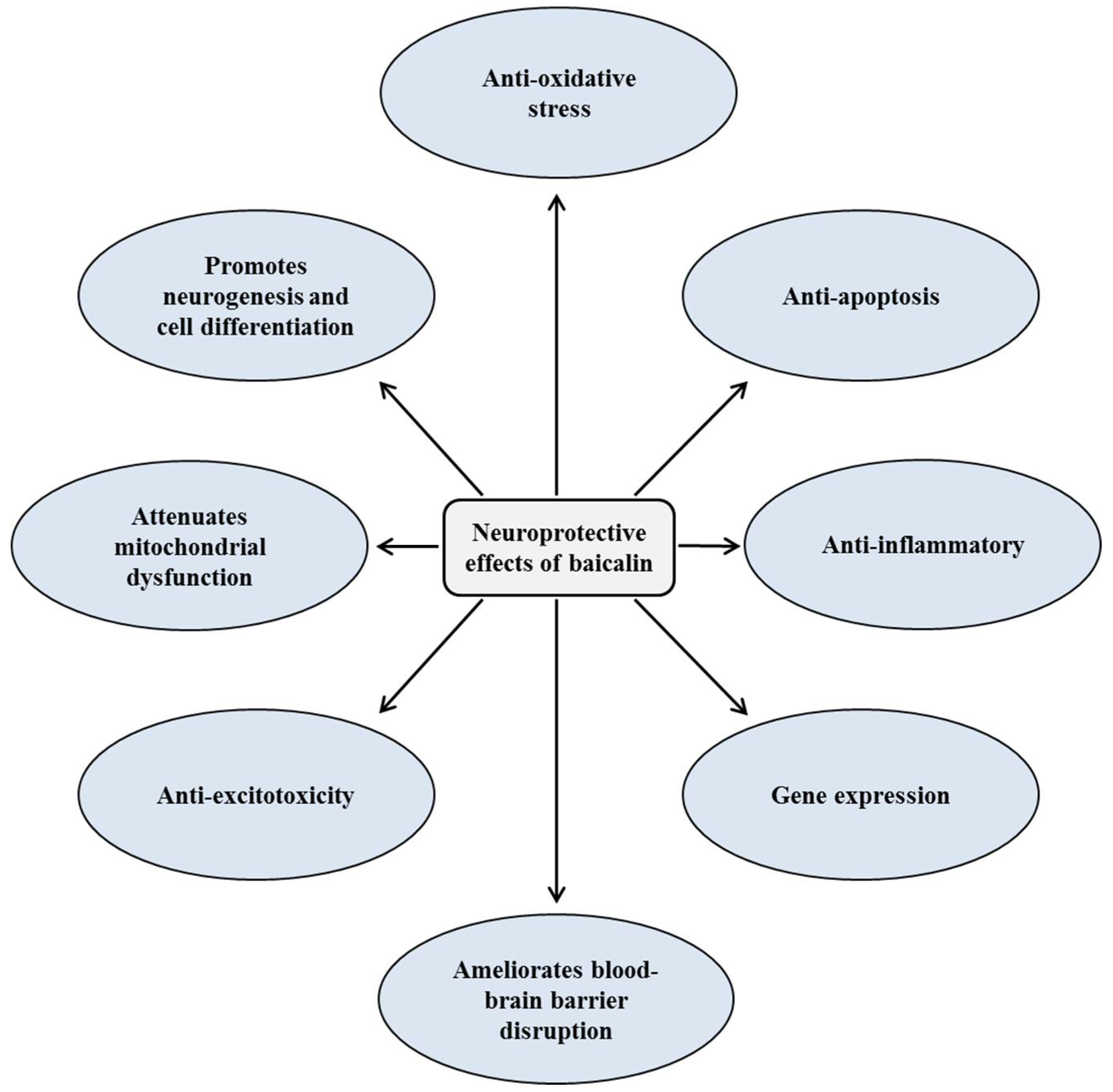
2. Neuroprotective Effects of Baicalin
2.1. Alzheimer’s Disease Model
2.1.1. In Vitro
2.1.2. In Vivo
2.2. Parkinson’s Disease Model
In Vivo
2.3. Ischemia
2.3.1. In Vitro
2.3.2. In Vivo
2.4. Neuroprotective and Cognitive Enhancement Effects
2.4.1. In Vitro
2.4.2. In Vivo
2.5. Epilepsy: In Vivo
2.6. Antidepressant Effect: In Vivo
2.7. Anxiolytic-Like Effect: In Vivo
2.8. Blood–Brain Barrier Penetrating Ability
3. Conclusions and Future Perspectives
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| 11β-HSD2 | 11β-hydroxysteroid dehydrogenase-2 |
| 5-LOX | 5-lipoxygenase (5-LOX) |
| AchE | acetylcholinesterase |
| AD | Alzheimer’s disease |
| Akt | protein kinase B |
| AMPA | α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid |
| APP | amyloid precursor protein |
| APPL2 | adaptor protein, phosphotyrosine interacting with PH domain and leucine zipper 2 |
| Aβ | amyloid-β |
| BBB | blood–brain barrier |
| BCCA | bilateral common carotid artery |
| BCCL | bilateral common carotid artery ligation |
| Bcl-2 | B cell lymphoma 2 family |
| BDNF | brain-derived neurotrophic factor |
| bHLH | basic helix–loop–helix |
| CaMKII | Ca2+/calmodulin-dependent protein kinase II |
| CD11b | cluster of differentiation molecule 11b |
| ChAT | choline acetyltransferase |
| CMS | chronic mild stress |
| CNS | central nervous system |
| CNTF | ciliary neurotrophic factor |
| COX-1/2 | cyclooxygenases-1/2 |
| CREB | cAMP response element-binding protein |
| CXCL3 | chemokine (C–X–C motif) ligand 3 (CXCL-3) |
| Drp-1 | dynamin-related protein 1 |
| DWI | diffusion weighted imaging |
| ERK1/2 | extracellular signal-regulated kinases 1 and 2 |
| GABA(A)R | GABAA receptor |
| GABA | gamma-aminobutyric acid |
| GDNF | glial cell-derived neurotrophic factor |
| GFAP | glial fibrillary acidic protein |
| GR | glucocorticoid receptor |
| GSH-Px | glutathione peroxidase |
| GSH | glutathione |
| GSK-3β | glycogen synthase kinase 3 beta |
| H2O2 | hydrogen peroxide |
| HSP-70 | heat shock protein-70 |
| IL-6 | interleukin 6 |
| IL-β | interleukin beta |
| iNOS | inducible nitric oxide synthase |
| IκB-α | nuclear factor of kappa light polypeptide gene enhancer in B cells inhibitor-alpha |
| JAK2 | Janus kinase 2 |
| JNK | c-Jun N-terminal kinase |
| KCC2 | K–Cl co-transporter 2 |
| LDH | lactate dehydrogenase |
| MAP-2 | microtubule-associated protein |
| MAPK | mitogen-actives protein kinases |
| MCAO | middle cerebral artery occlusion |
| MCL-1 | myeloid cell leukemia-1 |
| MDA | malondialdehyde |
| MFN2 | mitochondrial fission, elevated mitofusin-2 |
| MMP-9 | matrix metallopeptidase 9 |
| MPO | myeloperoxidase |
| MPTP | 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine |
| MRI | magnetic resonance imaging |
| MRTF-A | myocardin-related transcription factor-A |
| MyD88 | myeloid differentiation primary response 88 |
| NF-κB | nuclear factor kappa-light-chain-enhancer of activated B cells |
| NKCC1 | Na–K–Cl cotransporter-1 |
| NLRP3 | nucleotide-binding domain and leucine-rich repeat protein-3 |
| NMDA | N-methyl-d-aspartic acid |
| NMDAR1 | N-methyl-d-aspartic acid receptor-1 |
| NO | nitric oxide |
| NOD2 | nucleotide-binding oligomerization domain protein 2 |
| NOX | nicotinamide adenine dinucleotide 2′-phosphate oxidase |
| Nrf2 | nuclear factor erythroid 2-related factor 2 |
| NSPCs | neural stem/progenitor cells |
| OGD | oxygen/glucose deprivation |
| OGD/RO | oxygen/glucose deprivation/reoxygenation |
| PAR-1 | protease-activated receptor-1 |
| PD | Parkinson’s disease |
| PGE2 | prostaglandin E2 |
| PI3K | phosphatidylinositide 3-kinase |
| POP | prolyl oligopeptidase |
| ROS | reactive oxygen species |
| SCI | spinal cord injury |
| SCs | Schwann cells |
| SGK1 | serum- and glucocorticoid-regulated kinase 1 |
| siRNA | small interfering RNA |
| SOD | superoxide dismutase |
| STAT3 | signal transducer and activator of transcription 3 |
| TH | tyrosine hydroxylase |
| TLR4 | Toll like receptor-4 |
| TNF-α | tumor necrosis factor alpha |
| TRAF6 | tumor necrosis factor receptor-associated factor 6 |
| TRkB | tropomyosin receptor kinase B |
| TTC | 2,3,5-triphenyltetrazolium chloride |
| ZO-1 | zonula occludens-1. |
References
- Zhao, Q.; Chen, X.Y.; Martin, C. Scutellaria baicalensis, the golden herb from the garden of Chinese medicinal plants. Sci. Bull. (Beijing) 2016, 6, 1391–1398. [Google Scholar] [CrossRef] [PubMed]
- Shang, X.; He, X.; He, X.; Li, M.; Zhang, R.; Fan, P.; Zhang, Q.; Jia, Z. The genus Scutellaria an ethnopharmacological and phytochemical review. J. Ethnopharmacol. 2010, 128, 279–313. [Google Scholar] [CrossRef] [PubMed]
- Gaire, B.P.; Moon, S.K.; Kim, H. Scutellaria baicalensis in stroke management: Nature’s blessing in traditional Eastern medicine. Chin. J. Integr. Med. 2014, 20, 712–720. [Google Scholar] [CrossRef] [PubMed]
- Ishimaru, K.; Nishikawa, K.; Omoto, T.; Asai, I.; Yoshihira, K.; Shimomura, K. Two flavone 21-glucosides from Scutellaria baicalensis. Phytochemistry 1995, 40, 279–281. [Google Scholar] [CrossRef]
- Noh, K.; Kang, Y.; Nepal, M.R.; Jeong, K.S.; Oh, D.G.; Kang, M.J.; Lee, S.; Kang, W.; Jeong, H.G.; Jeong, T.C. Role of intestinal microbiota in baicalin-induced drug interaction and its pharmacokinetics. Molecules 2016, 21, 337. [Google Scholar] [CrossRef] [PubMed]
- Zheljazkov, V.D.; Cantrell, C.L.; Ebelhar, M.W.; Coker, C.; Evans, W.B. Quality assessment and yield of baikal skullcap (Scutellaria baicalensis) grown at multiple locations. HortScience 2007, 42, 1183–1187. [Google Scholar]
- Bochoráková, H.; Paulová, H.; Slanina, J.; Musil, P.; Táborská, E. Main flavonoids in the root of Scutellaria baicalensis cultivated in Europe and their comparative antiradical properties. Phytother. Res. 2003, 17, 640–644. [Google Scholar] [CrossRef] [PubMed]
- Dinda, B.; Dinda, S.; DasSharma, S.; Banik, R.; Chakraborty, A.; Dinda, M. Therapeutic potentials of baicalin and its aglycone, baicalein against inflammatory disorders. Eur. J. Med. Chem. 2017, 131, 68–80. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Huang, X.; Chen, W. The effects of baicalin and baicalein on cerebral ischemia: A review. Aging Dis. 2017, 8, 850–867. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Liu, C. Baicalin ameliorates chronic unpredictable mild stress-induced depressive behavior: Involving the inhibition of NLRP3 inflammasome activation in rat prefrontal cortex. Int. Immunopharmacol. 2017, 48, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Gasiorowski, K.; Lamer-Zarawska, E.; Leszek, J.; Parvathaneni, K.; Yendluri, B.B.; Błach-Olszewska, Z.; Aliev, G. Flavones from root of Scutellaria baicalensis Georgi: Drugs of the future in neurodegeneration? CNS Neurol. Disord. Drug Targets 2011, 10, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Ju, X.N.; Mu, W.N.; Liu, Y.T.; Wang, M.H.; Kong, F.; Sun, C.; Zhou, Q.B. Baicalin protects against thrombin induced cell injury in SH-SY5Y cells. Int. J. Clin. Exp. Pathol. 2015, 8, 14021–14027. [Google Scholar] [PubMed]
- Li, S.; Sun, X.; Xu, L.; Sun, R.; Ma, Z.; Deng, X.; Liu, B.; Fu, Q.; Qu, R.; Ma, S. Baicalin attenuates in vivo and in vitro hyperglycemia-exacerbated ischemia/reperfusion injury by regulating mitochondrial function in a manner dependent on AMPK. Eur. J. Pharmacol. 2017, 815, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.; Wang, C.; Chen, H.; Hu, Y.; Tian, L.; Pan, J.; Geng, M. Aβ-induced microglial cell activation is inhibited by baicalin through the JAK2/STAT3 signaling pathway. Int. J. Neurosci. 2014, 124, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.X.; Wang, F.; Cao, X.L.; Pan, H.Y.; Liu, X.Y.; Hu, X.M.; Sun, Y.Y. Baicalin protects PC-12 cells from oxidative stress induced by hydrogen peroxide via anti-apoptotic effects. Brain Inj. 2014, 28, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Zuo, D.; Lin, Y.; Liu, L.; Wang, C.; Xu, J.; Sun, F.; Li, L.; Li, Z.; Wu, Y. Baicalin attenuates ketamine-induced neurotoxicity in the developing rats: Involvement of PI3K/Akt and CREB/BDNF/Bcl-2 pathways. Neurotox. Res. 2016, 30, 159–172. [Google Scholar] [CrossRef] [PubMed]
- Tejada, S.; Setzer, W.; Daglia, M.; Nabavi, S.F.; Sureda, A.; Braidy, N.; Gortzi, O.; Nabavi, S.M. Neuroprotective effects of ellagitannins: A brief review. Curr. Drug Targets 2016, 7, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, H.L.; Yang, C.M. Role of redox signaling in neuroinflammation and neurodegenerative disease. BioMed Res. Int. 2013, 2013, 484613. [Google Scholar] [CrossRef] [PubMed]
- Sowndhararajan, K.; Deepa, P.; Kim, M.; Park, S.J.; Kim, S. Baicalein as a potent neuroprotective agent: A review. Biomed. Pharmacother. 2017, 95, 1021–1032. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Li, G.; Wang, Y.F.; Fan, Z.K.; Yu, D.S.; Wang, Z.D.; Bi, Y.L. Neuroprotective effect of baicalin on compression spinal cord injury in rats. Brain Res. 2010, 1357, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.X.; Cao, X.L.; Wang, F.; Wang, J.; Ying, T.Z.; Xiao, W.; Zhang, Y.; Xing, H.; Dong, W.; Xu, S.Q.; et al. Baicalin inhibiting cerebral ischemia/hypoxia-induced neuronal apoptosis via MRTF-A-mediated transactivity. Eur. J. Pharmacol. 2015, 767, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Bao-An, C.; Senthilkumar, R.; Rong, F.; Guo, Q.L. Cardioprotective potential of baicalein: A short review of in vitro and in vivo studies. Pharm. Anal. Acta 2014, 5, 280–284. [Google Scholar]
- de Oliveira, M.R.; Nabavi, S.F.; Habtemariam, S.; Erdogan Orhan, I.; Daglia, M.; Nabavi, S.M. The effects of baicalein and baicalin on mitochondrial function and dynamics: A review. Pharmacol. Res. 2015, 100, 296–308. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Snyder, S.A.; Smith, J.N.; Chen, Y.C. Anticancer properties of baicalein: A review. Med. Chem. Res. 2016, 25, 1515–1523. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.R.; Do, C.W.; To, C.H. Potential therapeutic effects of baicalein, baicalin and wogonin in ocular disorders. J. Ocul. Pharmacol. Ther. 2014, 30, 605–614. [Google Scholar] [CrossRef] [PubMed]
- Lai, M.Y.; Hsiu, S.L.; Tsai, S.Y.; Hou, Y.C.; Chao, P.D.L. Comparison of metabolic pharmacokinetics of baicalin and baicalein in rats. J. Pharm. Pharmacol. 2003, 55, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Du, L.; Wang, S.; He, G.; Yang, T.; Li, X. Pharmacokinetic study of baicalein and its major metabolites after IV administration in dogs. Chin. Herb. Med. 2011, 3, 196–201. [Google Scholar]
- Dou, J.; Chen, L.; Xu, G.; Zhang, L.; Zhou, H.; Wang, H.; Su, Z.; Ke, M.; Guo, Q.; Zhou, C. Effects of baicalein on sendai virus in vivo are linked to serum baicalin and its inhibition of hemagglutinin-neuraminidase. Arch. Virol. 2011, 156, 793–801. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.I.; Kim, H.J.; Hwang, K.M.; Jekal, S.J.; Pae, H.O.; Choi, B.M.; Yun, Y.G.; Kwon, T.O.; Chung, H.T.; Kim, Y.C. Hepatoprotective effect of baicalin, a major flavone from Scutellaria radix, on acetaminophen-induced liver injury in mice. Immunopharmacol. Immunotoxicol. 2003, 25, 585–594. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.Y.; Wei, E.Q.; Zhao, Y.M.; Chen, F.X.; Wang, M.L.; Zhang, W.P.; Chen, Z. Protective effects of baicalin on oxygen/glucose deprivation- and NMDA-induced injuries in rat hippocampal slices. J. Pharm. Pharmacol. 2005, 57, 1019–1026. [Google Scholar] [CrossRef] [PubMed]
- Ge, Q.F.; Hu, X.; Ma, Z.Q.; Liu, J.R.; Zhang, W.P.; Chen, Z.; Wei, E.Q. Baicalin attenuates oxygen-glucose deprivation-induced injury via inhibiting NMDA receptor-mediated 5-lipoxygenase activation in rat cortical neurons. Pharmacol. Res. 2007, 55, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.H.; Kang, K.D.; Ji, D.; Fawcett, R.J.; Safa, R.; Kamalden, T.A.; Osborne, N.N. The flavonoid baicalin counteracts ischemic and oxidative insults to retinal cells and lipid peroxidation to brain membranes. Neurochem. Int. 2008, 53, 325–337. [Google Scholar] [CrossRef] [PubMed]
- Tarragó, T.; Kichik, N.; Claasen, B.; Prades, R.; Teixidó, M.; Giralt, E. Baicalin, a prodrug able to reach the CNS, is a prolyl oligopeptidase inhibitor. Bioorg. Med. Chem. 2008, 16, 7516–7524. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Hu, J.; Ma, L.; Yuan, Z.; Wang, Y.; Wang, X.; Xing, D.; Lei, F.; Du, L. Comprehensive study of baicalin down-regulating NOD2 receptor expression of neurons with oxygen-glucose deprivation in vitro and cerebral ischemia-reperfusion in vivo. Eur. J. Pharmacol. 2010, 649, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Yin, F.; Liu, J.; Ji, X.; Wang, Y.; Zidichouski, J.; Zhang, J. Baicalin prevents the production of hydrogen peroxide and oxidative stress induced by Aβ aggregation in SH-SY5Y cells. Neurosci. Lett. 2011, 492, 76–79. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Wang, J.; Zhang, P.; Li, D.; Zhang, C.; Zhao, H.; Fu, J.; Wang, B.; Liu, J. Baicalin attenuates proinflammatory cytokine production in oxygen-glucose deprived challenged rat microglial cells by inhibiting TLR4 signaling pathway. Int. Immunopharmacol. 2012, 14, 749–757. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhuang, P.; Shen, B.; Zhang, Y.; Shen, J. Baicalin promotes neuronal differentiation of neural stem/progenitor cells through modulating p-stat3 and bHLH family protein expression. Brain Res. 2012, 1429, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Cheng, F.; Lu, Y.; Zhong, X.; Song, W.; Wang, X.; Sun, X.; Qin, J.; Guo, S.; Wang, Q. Baicalin’s therapeutic time window of neuroprotection during transient focal cerebral ischemia and its antioxidative effects in vitro and in vivo. Evid. Based Complement. Alternat. Med. 2013, 2013, 120261. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Lv, P.; Li, J.; Wang, H.; Zhou, T.; Liu, Y.; Lin, W. Baicalin inhibits colistin sulfate-induced apoptosis of PC12 cells. Neural Regen. Res. 2013, 8, 2597–2604. [Google Scholar] [PubMed]
- Xu, M.; Chen, X.; Gu, Y.; Peng, T.; Yang, D.; Chang, R.C.; So, K.F.; Liu, K.; Shen, J. Baicalin can scavenge peroxynitrite and ameliorate endogenous peroxynitrite-mediated neurotoxicity in cerebral ischemia-reperfusion injury. J. Ethnopharmacol. 2013, 150, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Hou, J.; Fu, J.; Li, D.; Zhang, C.; Liu, J. Baicalin protects rat brain microvascular endothelial cells injured by oxygen-glucose deprivation via anti-inflammation. Brain Res. Bull. 2013, 97, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, P.W.; Cui, G.Z.; Zhang, Y.J.; Zhang, M.X.; Guo, H.; Zhang, J.B.; Lu, Z.Q.; Isaiah, A.O.; Lin, Y.X. Baicalin regulates neuronal fate decision in neural stem/progenitor cells and stimulates hippocampal neurogenesis in adult rats. CNS Neurosci. Ther. 2013, 19, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Chen, X.; Xiong, P. Baicalin suppresses iron accumulation after substantia nigra injury: Relationship between iron concentration and transferrin expression. Neural Regen. Res. 2014, 9, 630–636. [Google Scholar] [PubMed]
- Morita, A.; Soga, K.; Nakayama, H.; Ishida, T.; Kawanishi, S.; Sato, E.F. Neuronal differentiation of human iPS cells induced by baicalin via regulation of bHLH gene expression. Biochem. Biophys. Res. Commun. 2015, 465, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Han, X.; Wang, J.; Sun, J. Baicalin may have a therapeutic effect in attention deficit hyperactivity disorder. Med. Hypotheses 2015, 85, 761–764. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Cao, Y.; Yu, J.; Liu, R.; Bai, B.; Qi, H.; Zhang, Q.; Guo, W.; Zhu, H.; Qu, L. Baicalin alleviates ischemia-induced memory impairment by inhibiting the phosphorylation of CaMKII in hippocampus. Brain Res. 2016, 1642, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhao, R.; Yan, W.; Wang, H.; Jia, M.; Zhu, N.; Zhu, Y.; Zhang, Y.; Wang, P.; Lei, H. Compositions, formation mechanism, and neuroprotective effect of compound precipitation from the traditional Chinese prescription Huang-Lian-Jie-Du-Tang. Molecules 2016, 21, 1094. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Li, S.; Zhu, L.; Fang, S.H.; Chen, J.L.; Xu, Q.Q.; Li, H.Y.; Luo, N.C.; Yang, C.; Luo, D.; et al. Effect of baicalin on oxygen-glucose deprivation-induced endothelial cell damage. Neuroreport 2017, 28, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Zuo, W.; Wu, H.; Zhang, K.; Lv, P.; Xu, F.; Jiang, W.; Zheng, L.; Zhao, J. Baicalin promotes the viability of Schwann cells in vitro by regulating neurotrophic factors. Exp. Ther. Med. 2017, 14, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Ding, S.; Zhuge, W.; Hu, J.; Yang, J.; Wang, X.; Wen, F.; Wang, C.; Zhuge, Q. Baicalin reverses the impairment of synaptogenesis induced by dopamine burden via the stimulation of GABAAR-TrkB interaction in minimal hepatic encephalopathy. Psychopharmacology 2018, 235, 1163–1178. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wang, H.; Chen, J.H.; Wang, L.H.; Zhang, H.S.; Fan, Y. Determination of amino acid neurotransmitters in cerebral cortex of rats administered with baicalin prior to cerebral ischemia by capillary electrophoresis-laser-induced fluorescence detection. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2003, 788, 93–101. [Google Scholar] [CrossRef]
- Liao, J.F.; Hung, W.Y.; Chen, C.F. Anxiolytic-like effects of baicalein and baicalin in the Vogel conflict test in mice. Eur. J. Pharmacol. 2003, 464, 141–146. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, Z.; Zhang, X.; Ying, K.; Liu, J.; Wang, Y. Gene expression profile induced by oral administration of baicalin and gardenin after focal brain ischemia in rats. Acta Pharmacol. Sin. 2005, 26, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Wang, F.; Tsang, S.Y.; Ho, K.H.; Zheng, H.; Yuen, C.T.; Chow, C.Y.; Xue, H. Anxiolytic-Like Effect of baicalin and its additivity with other anxiolytics. Planta Med. 2006, 72, 189–192. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Xing, D.; Wang, W.; Wang, R.; Du, L. Kinetic difference of baicalin in rat blood and cerebral nuclei after intravenous administration of Scutellariae Radix extract. J. Ethnopharmacol. 2006, 103, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.P.; Huang, W.T.; Cheng, B.C.; Hsu, C.C.; Lin, M.T. The flavonoid baicalin protects against cerebrovascular dysfunction and brain inflammation in experimental heatstroke. Neuropharmacology 2007, 52, 1024–1033. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhang, N.; Zou, H.Y. Protective effect of baicalin on mouse with Parkinson’s disease induced by MPTP. Zhongguo Zhong Xi Yi Jie He Za Zhi 2007, 27, 1010–1012. [Google Scholar] [PubMed]
- Wang, F.; Xu, Z.; Ren, L.; Tsang, S.Y.; Xue, H. GABA a receptor subtype selectivity underlying selective anxiolytic effect of baicalin. Neuropharmacology 2008, 55, 1231–1237. [Google Scholar] [CrossRef] [PubMed]
- Tu, X.K.; Yang, W.Z.; Shi, S.S.; Wang, C.H.; Chen, C.M. Neuroprotective effect of baicalin in a rat model of permanent focal cerebral ischemia. Neurochem. Res. 2009, 34, 1626–1634. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wu, R.; Li, P.; Liu, F.; Zhang, W.; Zhang, P.; Li, P.; Wang, Y. Baicalin administration is effective in positive regulation of twenty-four ischemia/reperfusion-related proteins identified by a proteomic study. Neurochem. Int. 2009, 54, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Xue, X.; Qu, X.J.; Yang, Y.; Sheng, X.H.; Cheng, F.; Jiang, E.N.; Wang, J.H.; Bu, W.; Liu, Z.P. Baicalin attenuates focal cerebral ischemic reperfusion injury through inhibition of nuclear factor κB p65 activation. Biochem. Biophys. Res. Commun. 2010, 403, 398–404. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Mao, X.; Sun, C.; Zheng, P.; Gao, J.; Wang, X.; Min, D.; Sun, H.; Xie, N.; Cai, J. Baicalin attenuates global cerebral ischemia/reperfusion injury in gerbils via anti-oxidative and anti-apoptotic pathways. Brain Res. Bull. 2011, 85, 396–402. [Google Scholar] [CrossRef] [PubMed]
- Tu, X.K.; Yang, W.Z.; Liang, R.S.; Shi, S.S.; Chen, J.P.; Chen, C.M.; Wang, C.H.; Xie, H.S.; Chen, Y.; Ouyang, L.Q. Effect of baicalin on matrix metalloproteinase-9 expression and blood-brain barrier permeability following focal cerebral ischemia in rats. Neurochem. Res. 2011, 36, 2022–2028. [Google Scholar] [CrossRef] [PubMed]
- Tu, X.K.; Yang, W.Z.; Shi, S.S.; Chen, Y.; Wang, C.H.; Chen, C.M.; Chen, Z. Baicalin inhibits TLR2/4 signaling pathway in rat brain following permanent cerebral ischemia. Inflammation 2011, 34, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Cheng, O.; Li, Z.; Han, Y.; Jiang, Q.; Yan, Y.; Cheng, K. Baicalin improved the spatial learning ability of global ischemia/reperfusion rats by reducing hippocampal apoptosis. Brain Res. 2012, 1470, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Gaire, B.P.; Song, J.; Lee, S.H.; Kim, H. Neuroprotective effect of four flavonoids in the root of Scutellaria baicalensis Georgi. Planta Med. 2012, 78, PF71. [Google Scholar] [CrossRef]
- Liu, Y.F.; Gao, F.; Li, X.W.; Jia, R.H.; Meng, X.D.; Zhao, R.; Jing, Y.Y.; Wang, Y.; Jiang, W. The anticonvulsant and neuroprotective effects of baicalin on pilocarpine-induced epileptic model in rats. Neurochem. Res. 2012, 37, 1670–1680. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Zhao, M.; Liu, H.; Wang, L.; Zhang, X. Pharmacokinetic effects of baicalin on cerebral ischemia-reperfusion after iv administration in rats. Chin. Herb. Med. 2012, 4, 53–57. [Google Scholar]
- Xiong, P.; Chen, X.; Guo, C.; Zhang, N.; Ma, B. Baicalin and deferoxamine alleviate iron accumulation in different brain regions of Parkinson’s disease rats. Neural Regen. Res. 2012, 7, 2092–2098. [Google Scholar] [PubMed]
- Zhou, Q.B.; Jia, Q.; Zhang, Y.; Li, L.Y.; Chi, Z.F.; Liu, P. Effects of baicalin on protease-activated receptor-1 expression and brain injury in a rat model of intracerebral hemorrhage. Chin. J. Physiol. 2012, 55, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Chen, L.; Qiu, Y.M.; Li, S.Q.; Xiong, W.H.; Yin, Y.H.; Jia, F.; Jiang, J.Y. Activations of GABAergic signaling, HSP70 and MAPK cascades are involved in baicalin’s neuroprotection against gerbil global ischemia/reperfusion injury. Brain Res. Bull. 2013, 90, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhang, Y.; Yu, Y.; Li, B.; Chen, Y.; Wu, H.; Wang, J.; Li, J.; Xiong, X.; He, Q.; et al. Systemic revealing pharmacological signalling pathway networks in the hippocampus of ischaemia-reperfusion mice treated with baicalin. Evid. Based Complement. Alternat. Med. 2013, 2013, 630723. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Shen, J.D.; Li, J.; Wang, R.; Jiao, S.; Yi, L.T. Chronic treatment with baicalin prevents the chronic mild stress-induced depressive-like behavior: Involving the inhibition of cyclooxygenase-2 in rat brain. Prog. Neuropsychopharmacol. Biol. Psychiatry 2013, 40, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, P.; Wang, Y.; Liu, J.; Zhang, Z.; Cheng, W.; Wang, Y. Ameliorative effects of a combination of baicalin, jasminoidin and cholic acid on ibotenic acid-induced dementia model in rats. PLoS ONE 2013, 8, e56658. [Google Scholar]
- Lee, B.; Sur, B.; Shim, I.; Lee, H.; Hahm, D.H. Baicalin improves chronic corticosterone-induced learning and memory deficits via the enhancement of impaired hippocampal brain-derived neurotrophic factor and cAMP response element-binding protein expression in the rat. J. Nat. Med. 2014, 68, 132–143. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Yin, Z.; Yang, S.; Ma, S. Baicalin reverse AMPA receptor expression and neuron apoptosis in chronic unpredictable mild stress rats. Biochem. Biophys. Res. Commun. 2014, 451, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.B.; Jin, Y.L.; Jia, Q.; Zhang, Y.; Li, L.Y.; Liu, P.; Liu, Y.T. Baicalin attenuates brain edema in a rat model of intracerebral hemorrhage. Inflammation 2014, 37, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Li, X.; Gao, P.; Tu, Y.; Zhao, M.; Li, J.; Zhang, S.; Liang, H. Baicalin attenuates alzheimer-like pathological changes and memory deficits induced by amyloid β1-42 protein. Metab. Brain Dis. 2015, 30, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.; Wang, H.; Zhao, Y.; Sun, D.; Zhai, X. Protective effects of baicalin on Aβ1–42-induced learning and memory deficit, oxidative stress, and apoptosis in rat. Cell Mol. Neurobiol. 2015, 35, 623–632. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Wang, L.L.; Pei, Y.Y.; Shen, J.D.; Li, H.B.; Wang, B.Y.; Bai, M. Baicalin decreases SGK1 expression in the hippocampus and reverses depressive-like behaviors induced by corticosterone. Neuroscience 2015, 311, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Ma, P.; Mao, X.Y.; Li, X.L.; Ma, Y.; Qiao, Y.D.; Liu, Z.Q.; Zhou, H.H.; Cao, Y.G. Baicalin alleviates diabetes-associated cognitive deficits via modulation of mitogen-activated protein kinase signaling, brain-derived neurotrophic factor and apoptosis. Mol. Med. Rep. 2015, 12, 6377–6383. [Google Scholar] [CrossRef] [PubMed]
- Dang, H.; Li, K.; Yu, Y.; Zhang, Y.; Liu, J.; Wang, P.; Li, B.; Wang, H.; Li, H.; Wang, Z.; et al. Variation of pathways and network profiles reveals the differential pharmacological mechanisms of each effective component to treat middle cerebral artery ischemia-reperfusion mice. Exp. Biol. Med. 2016, 241, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Liao, Z.J.; Liang, R.S.; Shi, S.S.; Wang, C.H.; Yang, W.Z. Effect of baicalin on hippocampal damage in kainic acid-induced epileptic mice. Exp. Ther. Med. 2016, 12, 1405–1411. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.Y.; Yin, Z.J.; Yang, S.J.; Ma, S.P.; Qu, R. Baicalin reverses depressive-like behaviours and regulates apoptotic signalling induced by olfactory bulbectomy. Phytother. Res. 2016, 30, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Bitto, A.; Giuliani, D.; Pallio, G.; Irrera, N.; Vandini, E.; Canalini, F.; Zaffe, D.; Ottani, A.; Minutoli, L.; Rinaldi, M.; et al. Effects of COX1-2/5-LOX blockade in Alzheimer transgenic 3xTg-AD mice. Inflamm. Res. 2017, 66, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Guan, B.; Chen, X.; Chen, X.; Li, C.; Qiu, J.; Yang, D.; Liu, K.J.; Qi, S.; Shen, J. Baicalin attenuates blood-brain barrier disruption and hemorrhagic transformation and improves neurological outcome in ischemic stroke rats with delayed t-PA treatment: Involvement of ONOO--MMP-9 pathway. Transl. Stroke Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Liu, J.; Wang, P.; Zhang, Y.; Li, B.; Yu, Y.; Dang, H.; Li, H.; Zhang, X.; Wang, Z. Poly-dimensional network comparative analysis reveals the pure pharmacological mechanism of baicalin in the targeted network of mouse cerebral ischemia. Brain Res. 2017, 1666, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Fu, Y.; Zhang, S.; Ding, H.; Chen, J. Baicalin attenuates subarachnoid hemorrhagic brain injury by modulating blood-brain barrier disruption, inflammation, and oxidative damage in mice. Oxid. Med. Cell Longev. 2017, 2017, 1401790. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.Q.; Li, Y.L.; Ao, Z.B.; Wen, Z.L.; Chen, Q.W.; Huang, Z.G.; Xiao, B.; Yan, X.H. Baicalin protects neonatal rat brains against hypoxic-ischemic injury by upregulating glutamate transporter 1 via the phosphoinositide 3-kinase/protein kinase B signaling pathway. Neural Regen. Res. 2017, 12, 1625–1631. [Google Scholar] [PubMed]
- Gao, C.; Du, Q.; Li, W.; Deng, R.; Wang, Q.; Xu, A.; Shen, J. Baicalin modulates APPL2/glucocorticoid receptor signaling cascade, promotes neurogenesis, and attenuates emotional and olfactory dysfunctions in chronic corticosterone-induced depression. Mol. Neurobiol. 2018. [CrossRef] [PubMed]
- Zhao, J.; Lu, S.; Yu, H.; Duan, S.; Zhao, J. Baicalin and ginsenoside Rb1 promote the proliferation and differentiation of neural stem cells in Alzheimer’s disease model rats. Brain Res. 2018, 1678, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Ma, S.; Qu, R.; Kang, D.; Liu, Y. Antidepressant effect of baicalin extracted from the root of Scutellaria baicalensis in mice and rats. Pharm. Biol. 2006, 44, 503–510. [Google Scholar] [CrossRef]
- Zhang, K.; Pan, X.; Wang, F.; Ma, J.; Su, G.; Dong, Y.; Yang, J.; Wu, C. Baicalin promotes hippocampal neurogenesis via SGK1- and FKBP5-mediated glucocorticoid receptor phosphorylation in a neuroendocrine mouse model of anxiety/depression. Sci. Rep. 2016, 6, 30951. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.M.; Liu, H.Y.; Xie, L.; Liu, X.D. Effect of baicalin and berberine on transport of nimodipine on primary-cultured, rat brain microvascular endothelial cells. Acta Pharmacol. Sin. 2007, 28, 573–578. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Zhang, L.; He, Q.; Liu, X.; Okeke, C.I.; Tong, L.; Guo, L.; Yang, H.; Zhang, Q.; Zhao, H.; et al. Effect of Baicalin-loaded PEGylated cationic solid lipid nanoparticles modified by OX26 antibody on regulating the levels of baicalin and amino acids during cerebral ischemia-reperfusion in rats. Int. J. Pharm. 2015, 489, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Zhao, H.; Shu, L.; Zhang, Y.; Okeke, C.; Zhang, L.; Li, J.; Li, N. Preparation and evaluation of Baicalin-loaded cationic solid lipid nanoparticles conjugated with OX26 for improved delivery across the BBB. Drug Dev. Ind. Pharm. 2015, 41, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Je, Y.J.; Yang, M.; Jiang, X.H.; Ma, J.H. Pharmacokinetics of baicalin-phospholipid complex in rat plasma and brain tissues after intranasal and intravenous administration. Pharmazie 2011, 66, 374–377. [Google Scholar] [PubMed]
- Zhang, Z.J.; Li, P.; Wang, Z.; Li, P.T.; Zhang, W.S.; Sun, Z.H.; Zhang, X.J.; Wang, Y.Y. A comparative study on the individual and combined effects of baicalin and jasminoidin on focal cerebral ischemia-reperfusion injury. Brain Res. 2006, 1123, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.F.; Li, Z.; Xin, W.F.; Wang, Y.Y.; Zhang, W.S. Pharmacokinetics and brain distribution differences of baicalin in rat underlying the effect of Panax notoginsenosides after intravenous administration. Chin. J. Nat. Med. 2014, 12, 632–640. [Google Scholar] [CrossRef]
- Fong, S.Y.K.; Li, C.; Ho, Y.C.; Li, R.; Wang, Q.; Wong, Y.C.; Xue, H.; Zuo, Z. Brain uptake of bioactive flavones in Scutellariae Radix and its relationship to anxiolytic effect in mice. Mol. Pharm. 2017, 14, 2908–2916. [Google Scholar] [CrossRef] [PubMed]
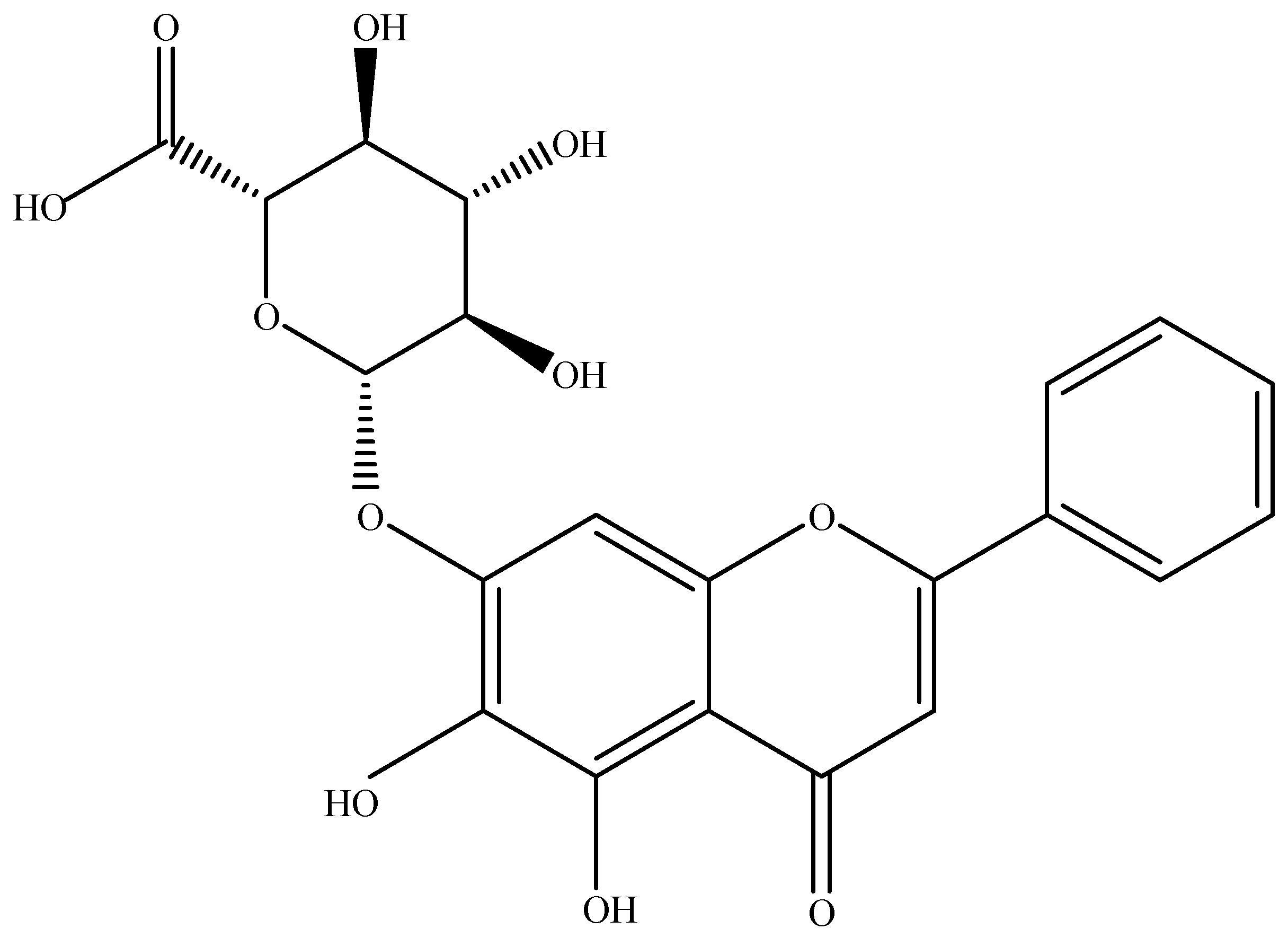
| S. No. | Model | Mechanism | Dose | Reference |
|---|---|---|---|---|
| 1 | Oxygen/glucose deprivation (OGD)- and N-methyl-d-aspartate (NMDA)-induced injuries in rat hippocampal slices | Inhibited viability reduction and acute neuron swelling. Inhibited the increased membrane portion of PKC(α). | 0.1, 1, and 10 μM/L | [30] |
| 2 | OGD-induced ischemic-like injury in rat cortical neurons | Attenuated Injuries. Inhibited 5-LOX translocation to the nuclear envelope translocation. Inhibited the production of cysteinyl leukotrienes. Decreased intracellular calcium level. | 0.2, 1, and 5 µM | [31] |
| 3 | Light-, H2O2-, and serum deprivation-induced toxicity in RGC-5 cells | Attenuated the negative insult of light, H2O2, and serum withdrawal to RGC-5 cells. | 0.1, 0.5, 1, 5, and 10 μM | [32] |
| 4 | Prolyl oligopeptidase (POP) inhibition assay | Inhibited POP in a dose-dependent manner. | 20, 50, 100, and 150 µM | [33] |
| 5 | OGD-induced toxicity in BC2, PC12 and primary neuron cells | Downregulated the expression of NOD2 and TNFα. | 10 μg/mL | [34] |
| 6 | Aβ-induced toxicity in SH-SY5Y cells | Inhibited Aβ1-42 aggregation. Protected oxidative injuries by decreasing H2O2 production. | 2.5, 5, and 10 μM | [35] |
| 7 | OGD-induced toxicity in rat microglial cells | Suppressed the release of TNF-α, IL-1β, IL-6, and IL-8. Downregulated TLR4 mRNA expression. Reduced TRAF6 levels. Downregulated the phosphorylation of IκB-α, c-jun, ERK1/2, JNK, p38 and inhibited the transfer of MyD88 from cytoplasm to membrane. | 10, 20, and 40 μg/mL | [36] |
| 8 | Neural stem/progenitor cells (NSPCs) from the cortex of embryonic E15–16 rats | Increased the percentages of MAP-2 positive-staining cells and decreased GFAP staining cells. Downregulated the expression of p-stat3 and Hes1. Upregulated the expressions of NeuroD1 and Mash1. | 2 and 20 μM | [37] |
| 9 | H2O2-induced toxicity in primary rat cortical neurons | Attenuated neuronal injury and improved superoxide dismutase (SOD) activity. | 10, 20, 40, 80, and 200 µM | [38] |
| 10 | Colistin sulfate-induced toxicity in PC12 cells | Changed the cell morphology and increased the cell viability. Decreased caspase-3 activity, tate dehydrogenase level, and free radical content. | 25, 50, and 100 μg/mL | [39] |
| 11 | Peroxynitrite-mediated toxicity in SH-SY5Y cells | Protected the neuronal cell damage. | 5, 20, and 50 μM | [40] |
| 12 | OGD-induced injury in brain microvascular endothelial cells | Increased cell viability, decreased the rate of LDH leakage, and the levels of TNF-α, IL-1β, and IL-6. Downregulated the phosphorylation of MAPK signaling pathway, such as p-MRK1/2, p-ERK and p-p38. Suppressed the phosphorylation of NF-кB signaling pathway, such as p-IKKα, p-IKKβ, and p-IκBα. Inhibited nuclear transcriptional activity triggered via NF-κB p65 and p-IκBα. | 10 and 100 μM | [41] |
| 13 | NSPCs from rats | Transient exposure of NSPCs to baicalin during proliferation activated Mash1. | 7.5, 15, and 30 µM | [42] |
| 14 | C6 glioma cells | Downregulated iron concentration by positively regulating divalent metal transporter 1 expression and negatively regulating ferroportin 1 expression. Decreased iron accumulation in the substantia nigra. | 100 μg/mL | [43] |
| 15 | Aβ42-induced toxicity in BV2 microglial cells | Reduced the expression of CD11b, decreased chemotactic ability, and inhibited the secretion of IL-6, TNF-α, and NO. Suppressed the phosphorylation of JAK2 and STAT3. | 50 and 100 μM | [14] |
| 16 | H2O2-induced toxicity in PC12 cells | Reduced the viability loss and apoptotic rate. Increased SOD, GSH-Px activities, and decreased MDA level. Increased the expression of survivin, Bcl-2, and p-STAT3, and decreased caspase-3 expression. | 1, 2, and 5 µM | [15] |
| 17 | Thrombin-induced toxicity in SH-SY5Y cells | Reduced cell death by inhibiting NF-κB activation and suppressing PAR-1 expression. Reduced caspase-3 expression. | 5, 10, and 20 μM | [12] |
| 18 | Neuronal differentiation of human iPS cells | Promoted neuronal differentiation and inhibited glial differentiation. Reduced Hes1 protein levels and upregulated Ascl1 gene expression. | 10 mmol/L | [44] |
| 19 | H2O2-induced toxicity in primary rat cortical neurons | Inhibited neuronal apoptosis by enhancing the transcription and expression of MCL-1 and BCL-2. Increased MRTF-A level. The anti-apoptosis effect of baicalin was inhibited by small interfering RNA of MRTF-A. Enhanced the transactivity of MCL-1 and BCL-2 promoter. LY294002 (PI3K inhibitor) and PD98059 (ERK1/2 inhibitor) reduced baicalin-induced MRTF-A expression and transactivity and expression of MCL-1 and BCL-2. | 0.7, 1.4, and 2.8 µM | [21] |
| 20 | OGD/reoxygenation (RO)-induced toxicity in SH-SY5Y cells | Attenuated apoptotic cell death. Decreased caspase-3 expression. Downregulated of NF-κB and NMDAR1. | 1, 5, and 25 μmol/L | [45] |
| 21 | OGD-induced toxicity in hippocampal neurons and SH-SY5Y cells | Suppressed the phosphorylation level of CaMKII. | 1 µM | [46] |
| 22 | Ketamine-induced toxicity in primary rat cortical neuron–glia mixed cultures | Alleviated cell viability decrease, morphological change, and caspase-3 expression increase. | 20, 50, and 100 µM | [16] |
| 23 | Cobalt chloride-induced toxicity in PC12 cells | Baicalin–berberine complex showed protective effects | 15 µg/mL | [47] |
| 24 | OGD/reperfusion-induced toxicity in PC12 cells | Inhibited Drp-1 expression, decreased mitochondrial fission, promoted MFN2 generation, increased Drp-1 Ser637 phosphorylation, and elevated mitochondrial membrane potential. Suppressed cell apoptosis and enhanced mitophagy. | 0.1, 1, 10, and 20 µM | [13] |
| 25 | OGD-induced toxicity in microvascular endothelial cells from mouse brain | Inhibited cell death, reduced cell membrane damage, and maintained the integrity of the nucleus. Decreased the necroptosis ratio. Regulated the expression of RIP-1 and RIP-3 in bEnd.3 cells. Inhibited the production of ROS and malondialdehyde. Increased the activity of SOD. | 100 and 200 μM | [48] |
| 26 | Schwann cells (SCs) of RSC96 | Promoted the viability of RSC96 SCs and gene expression of GDNF, BDNF, and CNTF. | 5, 10, and 20 μM | [49] |
| 27 | Dopamine-induced minimal hepatic encephalopathy in primary hippocampal neurons | Blocked dopamine-induced reduction of GABAAR levels. Improved the interaction of GABAAR with TrkB. Prevents dopamine-induced impairment of synaptogenesis. | 1, 2.5, 5, 10, and 30 μM | [50] |
| S. No. | Model | Mechanism | Dose | References |
|---|---|---|---|---|
| 1 | Middle cerebral artery occlusion (MCAO)-induced focal cerebral ischemia in rats | Attenuated the elevations of Glu and Asp. | 300 mg/kg, intra-sublingually | [51] |
| 2 | Anxiolytic-like effects in mice | Increased the number of shocks as measured in Vogel lick-shock conflict paradigm. Anxiolytic-like effect baicalin was antagonized by a benzodiazepine receptor antagonist, flumazenil (2 mg/kg, ip). | 20 mg/kg, ip | [52] |
| 3 | MCAO-induced cerebral ischemia in rats | Reduced the infarction areas. Increased the gene expression of RpL19 and Csnk2. | 40 mg/kg, po | [53] |
| 4 | Anxiolytic-like effect in mice | Increased entries into and time spent in open arms. Improved the performance in the hole-board and horizontal wire tests. | 3.75, 7.5, 15, and 30 mg/kg, po | [54] |
| 5 | Focal cerebral ischemia–reperfusion injury in rats | Ameliorated the results of TTC and histological examination. Baicalin/jasminoidin combination ameliorated DWI of MRI and behavior examination results. Promoted the expression of BDNF and inhibited the expression of caspase-3. | Baicalin—15 mg/kg, or a combination of baicalin (15 mg/kg) and jasminoidin (15 mg/kg), iv | [55] |
| 6 | Heat stress on cerebrovascular and metabolic functions in rats | Improved survival during heatstroke. Reduced the hyperthermia, intracranial hypertension, and increased levels of NO metabolite, glutamate, glycerol, lactate/pyruvate ratio, and dihydroxybenzoic acid in the hypothalamus. Suppressed the levels of IL-1β and TNF-α in the serum and hypothalamus. | 10, 20, and 40 mg/kg, iv | [56] |
| 7 | MPTP-induced toxicity in mice | Decreased score in the hanging and swimming tests. Prevented the loss of TH-positive neurons and the decrease of dopamine content. Increased the content of GSH in the brain. | 100 mg/kg, po | [57] |
| 8 | Picrotoxin-induced seizure in mice | Improved behavioral performances as measured in step-through passive avoidance and rotarod tests. Showed preference for alpha2- and alpha3-containing GABA(A) subtypes. | 3.3, 10, and 30 mg/kg, po | [58] |
| 9 | Ischemic insult to retina of one eye of a rat | Regulated the localization of Thy-1 and ChAT, and the content of various proteins and mRNAs. | 12.5 mg/kg, ip | [32] |
| 10 | MCAO-induced focal cerebral ischemia in rats | Reduced neurological deficit scores and cerebral infarct volume. Decreased the enzymatic activity of MPO and the expression of iNOS and COX-2. Inhibited neuronal apoptosis and the expression of cleaved caspase-3 protein. | 10, 30, and 100 mg/kg, ip | [59] |
| 11 | MCAO-induced focal cerebral ischemia in mice | Performed well in regulating proteins in energy metabolism. | 20 mg/kg, i.v | [60] |
| 12 | Spinal cord injury (SCI) in rat | Decreased the water content of spinal cord tissue, the permeability of blood–spinal cord barrier, oxidant stress. Downregulated the expression of TNF-α, NF-κB, Bax, Bcl-2, and caspase-3. Improved the recovery of limb function. | 10, 30, and 100 mg/kg, ip | [20] |
| 13 | Bilateral common carotid artery ligation (BCCL)-induced cerebral ischemia in rats | Downregulated the expression of NOD2 and TNFα in protein levels. | 10, and 50 mg/kg, ip | [34] |
| 14 | MCAO-induced focal cerebral ischemia in rats | Decreased neurological deficit scores and reduced the volume of infarction. Decreased the level of NF-κB p65. | 50, 100, and 200 mg/kg, ip | [61] |
| 15 | Occlusion of common carotid arteries-induced ischemia in gerbils | Attenuated neuronal cell damage. Reduced the level of MDA. Elevated SOD, GSH, and GSH-PX activities. Promoted the expression of BDNF and inhibited the expression of caspase-3. | 50, 100, and 200 mg/kg, ip | [62] |
| 16 | MCAO-induced focal cerebral ischemia in rats | Reduced the neuronal damage, brain edema, and blood–brain barrier (BBB) permeability. Downregulated the expression of MMP-9 protein and mRNA. Upregulated the expression of occludin. | 100 mg/kg, ip | [63] |
| 17 | MCAO-induced cerebral ischemia in rats | Reduced cerebral infarct area and infarct volume. Decreased the expression of TLR2/4, NF-κB, iNOS, and COX-2. Attenuated TNF-α and IL-1β levels. | 100 mg/kg, ip | [64] |
| 18 | Global ischemia/reperfusion injury in rats | Improved learning and memory. Decreased hippocampal apoptosis and reduced the level of COX-2 expression. | 100 mg/kg, po | [65] |
| 19 | 4-vessel occlusion-induced global ischemic model in rat | Inhibited the hippocampal neuronal cell death. | 10 mg/kg, po | [66] |
| 20 | Pilocarpine-induced epileptic model in rats | Delayed the onset of the first limbic seizures and status epilepticus. Reduced the mortality rate, and attenuated the changes of lipid peroxidation, nitrite content, and reduced glutathione levels. Attenuated the neuronal cell loss, apoptosis, and degeneration. | 100 mg/kg, ip | [67] |
| 21 | BCCL-induced cerebral ischemia-reperfusion in rats | Prolonged the terminal half-life of baicalin | 90 mg/kg, iv | [68] |
| 22 | Rotenone-induced Parkinson’s disease in rats | Inhibited iron accumulation in different brain regions. | 78 mg/kg, po | [69] |
| 23 | Collagenase VII-induced ICH in rats | Attenuated brain edema and inhibited cell apoptosis. Suppressed the expression of PAR-1. | 25, 50, or 100 mg/kg, ip | [70] |
| 24 | MCAO-induced cerebral ischemia in rats | Improved neurological function and decreased brain infarction. Reduced cell apoptosis and inhibited the production of ROS and MDA. Interfered with SOD and NOX oxidase activities. | 15 mg/kg, iv | [38] |
| 25 | Bilateral common carotid arteries (BCCA)-induced global ischemia/reperfusion injury in gerbils | Facilitated neurological function and suppressed neuronal damage. Increased GABA(A)R α1, GABA(A)R γ2 and KCC2. Decreased NKCC1 level. Upregulated the protein expressions of HSP70 and p-ERK, and diminished the expression of p-JNK and p-p38. | 200 mg/kg, ip | [71] |
| 26 | MCAO-induced cerebral ischemia in mice | Out of the 10 most significant molecular functions, 7 were common to baicalin and controls, and only 3 occurred in baicalin group. | 5 mg/kg, iv | [72] |
| 27 | Chronic unpredictable mild stress (CMS)-induced depressive-like behavior in rats | Prevented the abnormalities induced by CMS. Decreased COX-2 activity and expression, and reduced the level of PGE2. | 10, 20, and 40 mg/kg, po | [73] |
| 28 | MCAO-induced cerebral ischemia injury in rats | Inhibited the formation of 3-nitrotyrosine, reduced infarct size, and attenuated apoptotic cell death. | 10, 25, and 50 mg/kg, iv | [40] |
| 29 | Ibotenic acid-induced dementia in rats | A combination of three chemicals attenuated abnormalities in cognition, brain functional images, and brain histological morphology. Influenced the expression levels of 19 genes in the forebrain. | 3 mL/kg, po (1.25 mg/mL baicalin, 6.25 mg/mL jasminoidin, and 1.75 mg/mL cholic acid) | [74] |
| 30 | BCCA-induced transient cerebral ischemia in rats | Increased the number of newly generated cells and promoted new neuron production. Improved cognitive impairment in Morris water maze test. | 50 mg/kg, ip | [42] |
| 31 | Chronic corticosterone-induced learning and memory deficits in rats | Improved memory impairment in the passive avoidance test and reduced the escape latency in the Morris water maze test. Upregulated the expression of BDNF and CREB. | 20, 50, and 100 mg/kg, ip | [75] |
| 32 | AD transgenic mice (APPswe, PSEN1de9) | Inhibited microglial cell activation by regulating the JAK2/STAT3 signaling pathway. | 100 mg/kg, po | [14] |
| 33 | Rats induced with CMS | Increased AMPA receptor expression and decreased neuron apoptosis. | 20, and 40 mg/kg, po | [76] |
| 34 | Collagenase VII-induced ICH in rats | Reduced brain edema, inhibited NF-κB activation, and suppressed MMP-9 expression. Reduced IL-1β and IL-6 production, and BBB permeability. | 25, 50, and 100 mg/kg, ip | [77] |
| 35 | Aβ-induced AD in mice | Ameliorated memory impairment in the Morris water maze and probe tests. Attenuated glial cell activations, and increase of TNF-α and IL-6 expressions. | 30, 50, and 100 mg/kg, po | [78] |
| 36 | Aβ1–42-induced learning and memory deficits in rats | Improved learning and memory deficits. Attenuated the hippocampus injury caused by Aβ. Increased SOD, catalase, and GSH-px activities and upregulated their gene expression. Increased mitochondrial membrane potential, and decreased Bax/Bcl-2 ratio, cytochrome c release, and caspase-9/-3 activation. Activated Nrf2 signaling. | 50, 100, and 200 mg/kg, i.p | [79] |
| 37 | Corticosterone-induced depressive-like behaviors in mice | Increased sucrose preference and decreased duration of immobility. Downregulated the mRNA and protein expression of glucocorticoid receptor and BDNF. Upregulated the serum- and SGK1 in the hippocampus. Increased the expression of 11β-HSD2 protein in the hippocampus. | 10, and 20 mg/kg, po | [80] |
| 38 | Streptozotocin-induced diabetes-associated cognitive deficits in rats | Improved memory performances and neuronal survival. Increased ChAT, p-ERK, BDNF, and Bcl-2. Downregulated the levels of hippocampal AChE, p-JNK, p-p38, caspase-3, Bax, and plasma glucose. | 50, 100, and 200 mg/kg, ip | [81] |
| 39 | MCAO-induced ischemia/reperfusion in rats | Inhibited neuronal apoptosis and enhanced transcription and expression of MCL-1 and BCL-2. Increased myocardin-related transcription factor-A (MRTF-A) level in ischemic hemisphere. | 50, 100, and 200 mg/kg, po | [21] |
| 40 | MCAO-induced reperfusion in mice | Targeted pathways associated with development, neurophysiological processes, and cytoskeleton remodeling. | 20 mg/kg, iv | [82] |
| 41 | Kainic acid-induced epileptic mice | Attenuated neuronal damage and apoptosis in the hippocampus. Decreased the expression of miR-497 and cleaved caspase-3 protein. Upregulated the expression of Bcl-2 protein. | 100 mg/kg, ip | [83] |
| 42 | Global cerebral ischemia in gerbils | Improved learning and memory dysfunction by downregulating the phosphorylation level of CaMKII. | 100 mg/kg, ip | [46] |
| 43 | Ketamine-induced toxicity in rats | Alleviated morphological change and apoptosis. Downregulated caspase-3 activity and caspase-3 mRNA expression. Inhibited p-Akt and p-GSK-3β decrease, and relieved p-CREB and BDNF expression decrease. Increased Bcl-2/Bax and decreased caspase-3 expression. | 25, 50, and 100 mg/kg, ip | [16] |
| 44 | Olfactory bulbectomy-induced depression in rats | Increased the performance in depression-like behavioral tests. Decreased oxidative stress, synaptophysin expression, and hippocampal apoptosis. Modulated the levels MDA, SOD, and GSH-Px. Prevented apoptotic protease-activating factor-1 expression. Suppressed caspase-mediated apoptosis signaling cascades. | 20, and 40 mg/kg, po | [84] |
| 45 | In triple-transgenic (3xTg-AD) mice | Flavocoxid (a mixture of purified baicalin and catechin) improved learning and memory function. Decreased eicosanoid production and reduced the phosphorylation level of APP-p-Thr668, p-Thr181 and p-ERK, and the activation of the NLRP3 inflammasome. | 20 mg/kg, ip | [85] |
| 46 | MCAO-induced cerebral ischemia in rats | Reduced the mortality rates, ameliorated the tissue plasminogen activator-mediated BBB disruption and hemorrhagic transformation. Scavenged peroxynitrite and inhibited MMP-9 expression. | 50, 100, and 150 mg/kg, femoral vein | [86] |
| 47 | Hyperglycemia-exacerbated MCAO-induced ischemia/reperfusion in rats | Reduced blood glucose, relieved neurological deficit, and decreased infarct volume. | 100 mg/kg | [13] |
| 48 | CMS-induced rats | Reversed the changes of depressive-like behavior. Decreased the activation of NLRP3 inflammasome and IL-1β and IL-6 levels. | 20 and 40 mg/kg | [10] |
| 49 | MCAO-induced cerebral ischemia in mice | Reduced the ischemic infarct volume. BA resulted in targeting of pathways related to development, G-protein signaling, apoptosis, signal transduction, and immunity. | 5 mg/mL, iv | [87] |
| 50 | Subarachnoid hemorrhagic brain injury in mice via filament perforation | Restored the level of tight junction proteins such as occludin, claudin-5, ZO-1, and collagen IV. Inhibited the production of IL-1β, IL-6, and CXCL-3. Attenuated the induction of NOS-2 and NOX-2. | 100 mg/kg, ip | [88] |
| 51 | Left common carotid artery ligation followed by hypoxia in rats | Reduced cerebral infarct volume and neuronal loss. Inhibited apoptosis, and upregulated the expression of p-Akt and glutamate transporter 1. | 120 mg/kg, ip | [89] |
| 52 | Dopamine-induced minimal hepatic encephalopathy in rats | Reversed the inactivation of the GABA(A)Rβ/TrkB signaling pathway. Prevented the impairment of synaptogenesis and improved the memory performance. | 20, 50, and 100 mg/kg, ip | [50] |
| 53 | Chronic corticosterone-induced depression in mice | Inhibited APPL2-mediated GR hyperactivity and promoted adult neurogenesis. Released depressive and anxiety symptoms and enhanced olfactory functions. | 3.35 and 6.7 mg/kg, po | [90] |
| 54 | Aβ1-40-induced Alzheimer’s disease in rats | The percentages of astrocytes and neurons were increased. Enhanced the expressions of Nestin and nucleotide sugar epimerase. | 10 mg/kg, ip | [91] |
| 55 | CMS-induced depressant-like effect in mice and rats | Reduced immobility time in tail suspension test and the forced swimming test in mice. Decreased immobility time in forced swimming test in rats. Showed a significant recovery in sucrose intake. Inhibited monoamine oxidase A and B activity in a dose-dependent manner in rats. | Mice—10, 20, 40, 60, and 80 mg/kg, po Rats—6.25, 12.5, 25, 50, and 100 mg/k, po | [92] |
| 56 | Chronic corticosterone-induced anxiety/depression in mice | Alleviated several anxiety/depression-like behaviors. Increased Ki-67- and DCX-positive cells. Normalized the chronic corticosterone-induced decrease in GR protein levels, the increase in GR nuclear translocation, and the increase in GR phosphorylation at Ser203 and Ser211. Further, regulated the level of FK506-binding protein 51 and phosphorylated serum- and glucocorticoid-inducible kinase 1 at Ser422 and Thr256. | 40, 80, and 160 mg/kg, po | [93] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sowndhararajan, K.; Deepa, P.; Kim, M.; Park, S.J.; Kim, S. Neuroprotective and Cognitive Enhancement Potentials of Baicalin: A Review. Brain Sci. 2018, 8, 104. https://doi.org/10.3390/brainsci8060104
Sowndhararajan K, Deepa P, Kim M, Park SJ, Kim S. Neuroprotective and Cognitive Enhancement Potentials of Baicalin: A Review. Brain Sciences. 2018; 8(6):104. https://doi.org/10.3390/brainsci8060104
Chicago/Turabian StyleSowndhararajan, Kandhasamy, Ponnuvel Deepa, Minju Kim, Se Jin Park, and Songmun Kim. 2018. "Neuroprotective and Cognitive Enhancement Potentials of Baicalin: A Review" Brain Sciences 8, no. 6: 104. https://doi.org/10.3390/brainsci8060104
APA StyleSowndhararajan, K., Deepa, P., Kim, M., Park, S. J., & Kim, S. (2018). Neuroprotective and Cognitive Enhancement Potentials of Baicalin: A Review. Brain Sciences, 8(6), 104. https://doi.org/10.3390/brainsci8060104





