Acute Confusional Migraine: Distinct Clinical Entity or Spectrum of Migraine Biology?
Abstract
1. Introduction
2. Materials and Methods
3. Results
Clinical Features
4. Discussion
4.1. Auras
4.2. Premonitory Symptoms
4.3. Headache Phase
4.4. Postdrome Phase
5. Conclusions
Conflicts of Interest
Abbreviations
| ACM | Acute confusional migraine |
| ICHD-3β | International Headache Society’s International Classification of Headache Disoders-3 beta version |
| FHM | Familial hemiplegic migraine |
| EA2 | Episodic ataxia type 2 |
| CADASIL | Cerebral Autosomal-Dominant Arteriopathy with Subcortical Infarcts and Leukoencephalopathy |
| MRA | Magnetic resonance imaging |
| FIRDA | Frontal intermittent rhythmic delta activity |
| CSD | Cortical Spreading Depression |
| EEG | Electroencephalography |
References
- Gascon, G.; Barlow, C. Juvenile migraine, presenting as an acute confusional state. Pediatrics 1970, 45, 628–635. [Google Scholar] [PubMed]
- Ehyai, A.; Fenichel, G.M. The natural history of acute confusional migraine. Arch. Neurol. 1978, 35, 368–369. [Google Scholar] [CrossRef] [PubMed]
- Emery, E.S. Acute confusional state in children with migraine. Pediatrics 1977, 60, 111–114. [Google Scholar]
- Parrino, L.; Pietrini, V.; Spaggiari, M.C.; Terzano, M.G. Acute confusional migraine attacks resolved by sleep: Lack of significant abnormalities in post-ictal polysomnograms. Cephalalgia 1986, 6, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Sacquegna, T.; Cortelli, P.; Baldrati, A.; de Carolis, P.; Tinuper, P.; Lugaresi, E. Impairment of memory and consciousness in migraine: Clinical and EEG study. Funct. Neurol. 1986, 1, 431–436. [Google Scholar] [PubMed]
- Pietrini, V.; D’andrea, G.; Cananzi, A.; Ferro-Milone, F.; Terzano, M.G.; Parrino, L. Acute confusional migraine: Clinical and electroencephalographic aspects. Cephalalgia 1987, 7, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Haan, J.; Ferrari, M.D.; Brouwer, O.F. Acute confusional migraine. Case report and review of literature. Clin. Neurol. Neurosurg. 1988, 90, 275–278. [Google Scholar] [CrossRef]
- Piatella, L.; Tavoni, M.A.; Cardinali, C. Acute Confusional State as a possible manifestation of migraine: A case study. In Juvenile Headache: Etiopathogenesis, Clinical Diagnosis, and Therapy. In Proceedings of the International Juvenile Headache Congress, Rome, Italy, 6–9 March 1991; Excerpta Medica: Amsterdam, The Netherlands; New York, NY, USA; Sole Distributors for the USA and Canada: New York, NY, USA; Elsevier Science Pub. Co.: New York, NY, USA, 1991; pp. 539–542. [Google Scholar]
- D’Cruz, O.F.; Walsh, D.J. Acute confusional migraine: Case series and review of literature. Wis. Med. J. 1992, 91, 130–131. [Google Scholar] [PubMed]
- Sheth, R.D.; Riggs, J.E.; Bodensteiner, J.B. Acute confusional migraine: Variant of transient global amnesia. Pediatr. Neurol. 1995, 12, 129–131. [Google Scholar] [CrossRef]
- Ferrera, P.C.; Reicho, P.R. Acute confusional migraine and trauma-triggered migraine. Am. J. Emerg. Med. 1996, 14, 276–278. [Google Scholar] [CrossRef]
- Shaabat, A. Confusional migraine in childhood. Pediatr. Neurol. 1996, 15, 23–25. [Google Scholar] [CrossRef]
- Nezu, A.; Kimura, S.; Ohtsuki, N.; Tanaka, M.; Takebayashi, S. Acute confusional migraine and migrainous infarction in childhood. Brain Dev. 1997, 19, 148–151. [Google Scholar] [CrossRef]
- Neinstein, L.; Milgrom, E. Trauma-triggered migraine and acute confusional migraine. J. Adolesc. Health 2000, 27, 119–224. [Google Scholar] [CrossRef]
- Soriani, S.; Cavaliere, B.; Faggioli, R.; Scarpa, P.; Borgna-Pignatti, C. Confusional migraine precipitated by mild head trauma. Arch. Pediatr. Adolesc. Med. 2000, 154, 90–91. [Google Scholar] [PubMed]
- Al-Twaijri, W.A.; Shevell, M.I. Pediatric migraine equivalents: Occurrence and clinical features in practice. Pediatr. Neurol. 2002, 26, 365–368. [Google Scholar] [CrossRef]
- Bechtel, K. Acute mental status change due to acute confusional migraine. Pediatr. Emerg. Care 2004, 20, 238–241. [Google Scholar] [CrossRef] [PubMed]
- Gascon, G.; Coskun, C.; Brown, W. Acute confusional migraine: Case series and brief review. Int. J. Child Neuropsychiatry 2005, 2, 189–194. [Google Scholar]
- Fujita, M.; Fujiwara, J.; Maki, T.; Shigeta, M.; Shibasaki, K.; Takahashi, N.; Takahashi, M. The efficacy of sodium valproate and a MRA finding in confusional migraine. Brain Dev. 2007, 29, 178–181. [Google Scholar] [CrossRef] [PubMed]
- Sathe, S.; Deperalta, E.; Pastores, G.; Kolodny, E.H. Acute confusional migraine may be a presenting feature of Cadasil. Headache 2009, 49, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Khatri, R.; Hershey, A.D.; Wong, B. Prochlorperazine-Treatment for acute confusional migraine. Headache 2009, 49, 477–480. [Google Scholar] [CrossRef] [PubMed]
- Avraham, S.B.; Har-Gil, M.; Watemberg, N. Acute confusional migraine in an adolescent: Response to intravenous valproate. Pediatrics 2010, 125, e956–e959. [Google Scholar] [CrossRef] [PubMed]
- Gantenbein, A.R.; Riederer, F.; Mathys, J.; Biethahn, S.; Gossrau, G.; Waldvogel, D.; Sándor, P.S. Confusional migraine is an adult as well as a childhood disease. Cephalalgia 2011, 31, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Rota, E.; Morelli, N.; Immovilli, P.; De Mitri, P.; Magnifico, F.; Terlizzi, E.; Mazza, L.; Sala, B.; Biasucci, G.; Guidetti, D. “Possessed”: Acute confusional migraine in an adolescent, prevented by topiramate. Case Rep. Neurol. 2012, 4, 240–243. [Google Scholar] [CrossRef] [PubMed]
- Pacheva, I.; Ivanov, I. Acute confusional migraine: Is it a distinct form of migraine? Int. J. Clin. Pract. 2013, 67, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Pacheva, I.H.; Ivanov, I.S. Migraine variants-Occurrence in pediatric neurology practice. Clin. Neurol. Neurosurg. 2013, 115, 1775–1783. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.; Sahu, R.; Jaiswal, A.; Kumar, N. Acute confusional migraine: A variant not to be missed. BMJ Case Rep. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.E.; Shin, J.H.; Kim, Y.H.; Eom, T.H.; Kim, S.H.; Kim, J.M. Source localization of intermittent rhythmic delta activity in a patient with acute confusional migraine: Cross-spectral analysis using standardized low-resolution brain electromagnetic tomography (sLORETA). Neurol. Sci. 2016, 37, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Hida, A.; Arai, N.; Takeuchi, S. Low-dose intravenous propofol as a possible therapeutic option for acute confusional migraine. Am. J. Emerg. Med. 2017, 35, 195.e5–195.e6. [Google Scholar] [CrossRef] [PubMed]
- Olesen, J. The International Classification of Headache Disorders, 3rd edition. Cephalagia 2013, 33, 629–808. [Google Scholar] [CrossRef]
- Fisher, C.M. Late-life migraine accompaniments as a cause of unexplained transient ischemic attacks. Can. J. Neurol. Sci. 1980, 7, 9–17. [Google Scholar] [PubMed]
- Gowers, W.R. The Border-Land of Epilepsy: Faints, Vagal Attacks, Vertigo, Migraine, Sleep Symptoms and Their Treatment; Churchill: London, UK, 1907. [Google Scholar]
- Mendez, M.F.; Kremen, S.A.; Daroff, R.B.; Fenichel, G.M.; Jankovic, J.; Mazziotta, J.C.; Delirium. Bradley’s Neurology in Clinical Practice, Volume 1: Principles of Diagnosis and Management, 6th ed.; Daroff, R.B., Fenichel, G.M., Jankovic, J., Mazziotta, J.C., Eds.; Saunders: Philadelphia, PA, USA, 2012; pp. 26–36. ISBN 9996085309, 978-1437704341. [Google Scholar]
- American Psychiatric Association. Neurocognitive Disorders. In Diagnostic and Statistical Manual of Mental Disorders; American Psychiatric Association: Arlington, VA, USA, 2013; ISBN 978–0890425558. [Google Scholar]
- Gil-Gouveia, R.; Oliveira, A.G.; Martins, I.P. Assessment of cognitive dysfunction during migraine attacks: A systematic review. J. Neurol. 2015, 262, 654–665. [Google Scholar] [CrossRef] [PubMed]
- Merwick, A.; Fernandez, D.; McNamara, B.; Harrington, H. Acute encephalopathy in familial hemiplegic migraine with ATP1A2 mutation. BMJ Case Rep. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Spranger, M.; Spranger, S.; Schwab, S.; Benninger, C.; Dichgans, M. Familial hemiplegic migraine with cerebellar ataxia and paroxysmal psychosis. Eur. Neurol. 1999, 41, 150–152. [Google Scholar] [CrossRef] [PubMed]
- Feely, M.P.; O’Hare, J.; Veale, D.; Callaghan, N. Episodes of acute confusion or psychosis in familial hemiplegic migraine. Acta Neurol. Scand. 1982, 65, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Tan, R.Y.Y.; Markus, H.S. CADASIL: Migraine, encephalopathy, stroke and their inter-relationships. PLoS ONE 2016, 11, e0157613. [Google Scholar] [CrossRef] [PubMed]
- Cleves, C.; Parikh, S.; Rothner, A.D.; Tepper, S.J. Link between confusional migraine, hemiplegic migraine and episodic ataxia type 2: Hypothesis, family genealogy, gene typing and classification. Cephalalgia 2010, 30, 740–743. [Google Scholar] [CrossRef] [PubMed]
- Lapkin, M.L.; French, J.H.; Golden, G.S.; Rowan, A.J. The electroencephalogram in childhood basilar artery migraine. Neurology 1977, 27, 580–583. [Google Scholar] [CrossRef] [PubMed]
- Ganji, S. Basilar artery migraine: EEG and evoked potential patterns during acute stage. Headache 1986, 26, 220–223. [Google Scholar] [CrossRef] [PubMed]
- Frequin, S.T.; Linssen, W.H.; Pasman, J.W.; Hommes, O.R.; Merx, H.L. Recurrent prolonged coma due to basilar artery migraine. A case report. Headache 1991, 31, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, Y.; Nakajima, S.; Nishioka, R.; Nakamura, H. Basilar artery migraine with transient MRI and EEG abnormalities. Rinsho Shinkeigaku 1993, 33, 61–67. [Google Scholar] [PubMed]
- Ganji, S.; Hellman, S.; Stagg, S.; Furlow, J. Episodic coma due to acute basilar artery migraine: Correlation of EEG and brainstem auditory evoked potential patterns. Clin. Electroencephalogr. 1993, 24, 44–48. [Google Scholar] [CrossRef] [PubMed]
- Muellbacher, W.; Mamoli, B. Prolonged impaired consciousness in basilar artery migraine. Headache 1994, 34, 282–285. [Google Scholar] [CrossRef] [PubMed]
- La Spina, I.; Vignati, A.; Porazzi, D. Basilar artery migraine: Transcranial doppler EEG and SPECT from the aura phase to the end. Headache 1997, 37, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Ramelli, G.P.; Sturzenegger, M.; Donati, F.; Karbowski, K. EEG findings during basilar migraine attacks in children. Electroencephalogr. Clin. Neurophysiol. 1998, 107, 374–378. [Google Scholar] [CrossRef]
- Pisani, F.; Fusco, C. Ictal and interictal EEG findings in children with migraine. J. Headache Pain. 2004, 5, 23–29. [Google Scholar] [CrossRef]
- Hooshmand, H. The clinical significance of frontal intermittent rhythmic delta activity (FIRDA). Clin. Electroencephalogr. 1983, 14, 135–137. [Google Scholar] [CrossRef] [PubMed]
- Accolla, E.A.; Kaplan, P.W.; Maeder-Ingvar, M.; Jukopila, S.; Rossetti, A.O. Clinical correlates of frontal intermittent rhythmic delta activity (FIRDA). Clin. Neurophysiol. 2011, 122, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Desai, J.D.; Toczek, M.T.; Mitchell, W.G. Frontal intermittent rhythmic delta activity (FIRDA): Is there a clinical significance in children and adolescents? Eur. J. Paediatr. Neurol. 2012, 16, 138–141. [Google Scholar] [CrossRef] [PubMed]
- Haas, D.C.; Lourie, H. Trauma-triggered migraine: An explanation for common neurological attacks after mild head injury. Review of the literature. J. Neurosurg. 1988, 68, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Karsan, N.; Prabhakar, P.; Goadsby, P.J. Characterising the premonitory stage of migraine in children: A clinic-based study of 100 patients in a specialist headache service. J. Headache Pain 2016, 17, 94. [Google Scholar] [CrossRef] [PubMed]
- Giffin, N.J.; Ruggiero, L.; Lipton, R.B.; Silberstein, S.D.; Tvedskov, J.F.; Olesen, J.; Altman, J.; Goadsby, P.J.; Macrae, A. Premonitory symptoms in migraine—An electronic diary study. Neurology 2003, 60, 935–940. [Google Scholar] [CrossRef] [PubMed]
- Kelman, L. The premonitory symptoms (Prodrome): A tertiary care study of 893 migraineurs. Headache 2004, 44, 865–872. [Google Scholar] [CrossRef] [PubMed]
- Kelman, L. The postdrome of the acute migraine attack. Cephalalgia 2006, 26, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Santangelo, G.; Russo, A.; Trojano, L.; Falco, F.; Marcuccio, L.; Siciliano, M.; Conte, F.; Garramone, F.; Tessitore, A.; Tedeschi, G. Cognitive dysfunctions and psychological symptoms in migraine without aura: A cross-sectional study. J. Headache Pain 2016, 17, 76. [Google Scholar] [CrossRef] [PubMed]
- Ayata, C.; Jin, H.; Kudo, C.; Dalkara, T.; Moskowitz, M.A. Suppression of cortical spreading depression in migraine prophylaxis. Ann. Neurol. 2006, 59, 652–661. [Google Scholar] [CrossRef] [PubMed]
- Andreou, A.P.; Shields, K.G.; Goadsby, P.J. GABA and valproate modulate trigeminovascular nociceptive transmission in the thalamus. Neurobiol. Dis. 2010, 37, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Andreou, A.P.; Goadsby, P.J. Topiramate in the treatment of migraine: A kainate (glutamate) receptor antagonist within the trigeminothalamic pathway. Cephalalgia 2011, 31, 1343–1358. [Google Scholar] [CrossRef] [PubMed]
- Schipper, S.; Riederer, F.; Sándor, P.S.; Gantenbein, A.R. Acute confusional migraine: Our knowledge to date. Expert Rev. Neurother. 2012, 12, 307–314. [Google Scholar] [CrossRef] [PubMed]


| Case Report (Reference Number) | No. of Patients (n) | Gender Ratio (M:F) | Age Range (Years) | Mean Age (Mean ± SD) | Clinical Presentation α | Headache Onset Pre/Post-Confusion | Duration of Confusion (h) | Treatment | Recurrence | Outcomes |
|---|---|---|---|---|---|---|---|---|---|---|
| Gascon G. and Barlow C. [1] | 4 | 3:1 | 8–16 | 12.3 ± 3.5 | Disorientation (100%), Agitation (100%), Speech (100%), somatosensory (25%) and memory disturbances (50%) | Pre-confusion (4) | 4–24 h | Ergotamine and Phenobarbital | None | Two patients had multiple episodes of headache afterwards |
| Emery III et al. [3] | 4 | 3:1 | 5–14 | 10 ± 4.2 | Confusion (100%), agitation (100%), visual (50%), somatosensory (50%) speech (25%), and memory disturbances (75%) | Pre-confusion (4) | 1.5–9 h | NA | Two patients reported similar episodes in the past | Three patients had intermittent episodes of headache |
| Ehayi A. and Fenichel G. M. [2] | 5 | 3:2 | 9–14 | 11 ± 2 | Confusion and disorientation (100%), Agitation (100%), visual (80%), somatosensory (20%), speech (40%), motor (20%) and memory disturbances (80%) | Pre- (4), post-confusion (1) | 0.5–24 h | Ergotamine, methysergide | Over brief period of time all patients had recurrent ACM episodes | Migraine |
| Parrino L. et al. [4] | 2 | 2:0 | 14–15 | 14.5 ± 0.7 | Confusion and disorientation (100%), agitation (100%), Photophobia (50%), visual (100%), somatosensory (50%), speech (100%), and memory disturbances (100%) | Pre-confusion (2) | 24 h | NA | One patient reported similar episode in the past | None |
| Sacquegna T. et al. 1 [5] | 1 | 0:1 | 17 | 17 ± 0 | Confusion and disorientation (100%), visual (100%), somatosensory (100%), and memory disturbances (100%) | Pre-confusion (1) | 2 h | NA | Several episodes marked by less confusion | NA |
| Pietrini V. et al. [6] | 12 | 6:6 | 8–60 | 19.4 ± 13.4 | Confusion (100%), agitation (100%), visual (42%), somatosensory (42%), speech (25%), and motor symptoms (17%) | Pre-confusion (10) | 1–12 h | NA | NA | NA |
| Haan J. et al. [7] | 1 | 0:1 | 13 | 13 ± 0 | Confusion and disorientation (100%), agitation (100%), memory disturbance (100%) | Pre-confusion (1) | 12 h | NA | One similar episode in the past | NA |
| Piatella L. et al. [8] | 5 | 4:1 | 10–16 | 12.6 ± 2.3 | Confusion and disorientation (%100), agitation (20%), speech (80%), somatosensory disturbances (20%) | Pre-confusion (4) 2 | 15 min–24 h | NA | NA | Three patients developed migraine |
| D’Cruz O. and Walsh D. J. [9] | 3 | 0:3 | 11 | 11 ± 0 | Confusion and disorientation (100%), speech (67%), visual (67%), memory disturbances (100%) | Pre-confusion (3) | 6 h | NA | NA | NA |
| Sheth R. D. et al. [10] | 6 | 1:5 | 7.5–17 | 11.8 ± 3.5 | Confusion and disorientation (100%), agitation (100%), photophobia (50%), visual (50%), memory disturbances (100%) | NA 2 | 1–12 h | Propranolol | Two patients had recurrent ACM episodes | NA |
| Ferrera P. and Reicho P [11] | 2 | 1:1 | 6–9 | 7.5 ± 2.1 | Confusion (100%), agitation (50%), visual (50%), somatosensory (100%), speech (50%), and motor disturbances (50%) | Pre-confusion (1) 2 | NA | Sodium Valproate | Both patients had episodes of confusion in past | One patient had 2 episodes of headache |
| Shaabat A. et al. [12] | 13 | 11:2 | 6–15 | 10.8 ± 2.9 | Confusion (100%), agitation (62%) | Pre-confusion (13) | 1.5–24 h | NA | Four patients had recurrent ACM episodes | NA |
| Nezu A. et al. [13] | 2 | 1:1 | 7–12 | 9.5 ± 3.5 | Confusion (100%), Photophobia (50%), visual (50%), somatosensory (50%), speech (50%), motor (50%) and memory disturbances (100%) | Post-confusion (2) | 6–12 h | Dihydergot | NA | NA |
| Neinstein L. and Milgrom E. [14] | 1 | 1:0 | 14 | 14 ± 0 | Confusion (100%), anisocoria (100%), and ataxic gait (100%) | Pre-confusion (1) | NA | High-dose Oxygen and Sumatriptan | One similar episode | NA |
| Soriani S. et al. [15] | 11 | 8:3 | 6–14 | 9 ± 3 | Confusion (100%), agitation (45%), somnolence (55%), visual (27%), speech disturbances (9%) | Six patients had headache 3 | 1–12 h | NA | NA | Four patients developed migraine with aura & one w/o aura |
| Al-Twaijri W. and Shevell M. [16] | 5 | 2:3 | 6.5–15 | 10.9 | Confused, agitated and memory disturbances 4 | NA 2 | NA | NA | NA | NA |
| Bechtel K. et al. [17] | 2 | 1:1 | 11–14 | 12.5 ± 2.1 | Confusion (100%), speech (100%), visual (50%), somatosensory (50%) and memory disturbances (50%) | Pre- (1), post confusion (1) | Several hours | Acetaminophen | None | One patient had several episodes of headache |
| Gascon G. G. et al. [18] | 13 | 6:7 | 6–16 | 12.3 ± 3.8 | Confusion (69%), speech (46%), somatosensory (7.7%) and memory disturbances (8%) | Pre- (7), post confusion (4) 3 | NA | NA | Two patients had recurrent episodes of ACM | None |
| Fujita M. et al. [19] | 1 | 0:1 | 10 | 10 ± 0 | Confusion and disorientation (100%) visual disturbances (100%) | Pre-confusion (1) | 5–10 h | Sodium Valproate | Recurrent ACM episodes | Attacks were controlled after increasing the dose of sodium valproate |
| Sathe S. et al. [20] | 7 | 5:2 | 42–58 | 51.9 ± 7.3 | Confusion (100%), agitation (100%), visual (100%), somatosensory (57%), speech (57%), motor (14%) and memory disturbances (100%) | NA 2 | NA | NA | Recurrent ACM episodes | CADASIL |
| Khatri et al. [21] | 2 | 1:1 | 11–16 | 13.5 ± 3.5 | Confusion (100%), speech (50%) and memory disturbances (50%) | Pre-confusion (2) | 0.5–72 h | Prochlorperazine | Recurrent ACM episodes | Prochlorperazine was effective in acute management |
| Avraham S. B. et al. [22] | 1 | 1:0 | 12 | 12 ± 0 | Confusion (100%), speech (100%), visual (100%) and somatosensory symptoms (100%) | Pre- (3), during (5), post-confusion (1) | NA | Sodium Valproate | None | None |
| Gantebein A. et al. [23] | 10 | 6:4 | 16–62 | 30.5 ± 14.7 | Confusion (100%), agitation (20%), photophobia (10%), visual (40%), somatosensory (10%), motor (10%) speech (40%), and memory disturbances (60%) | Pre- (4), post-confusion (3) 2 | 1–6 h | NA | Seven patients had recurrent confusional episodes | NA |
| Rota E. et al [24] | 1 | 0:1 | 12 | 12 ± 0 | Confusion (100%), agitation (100%) | Not specified 2 | 4 h | Topiramate prophylaxis | Previous attack characterized by less agitation | No further episodes of confusion and headache after topiramate |
| Pacheva I. and Ivanov I. [25,26] | 3 | 1:2 | 12–14 | 12.7 ± 1.2 | Confusion (100%), agitation (67%), visual (33%), somatosensory (33%), motor (33%), speech (100%) and memory disturbances (67%) | Pre-confusion (3) | 8–10 h | Diazepam and phenobarbital | None | Two patients had 1–2 episodes of migraine without aura per month |
| Verma R. et al. [27] | 1 | 0:1 | 29 | 29 ± 0 | Confusion (100%), Agitation (100%), and memory disturbances (100%) | Pre-confusion (1) | NA | Sodium Valproate | None | NA |
| Kim D. et al. [28] | 1 | 0:1 | 9 | 9 ± 0 | Confusion (100%), agitation (100%), speech (100%) and memory disturbance (100%) | NA 2 | 2 h | Propranolol & Flunarizine | Similar episode in the past | Migraine |
| Sato K. et al [29] | 1 | 1:0 | 24 | 24 ± 0 | Confusion (100%), agitation (100%), visual (100%) and speech disturbance (100%) | Pre-confusion (1) | NA | Propofol | None | NA |
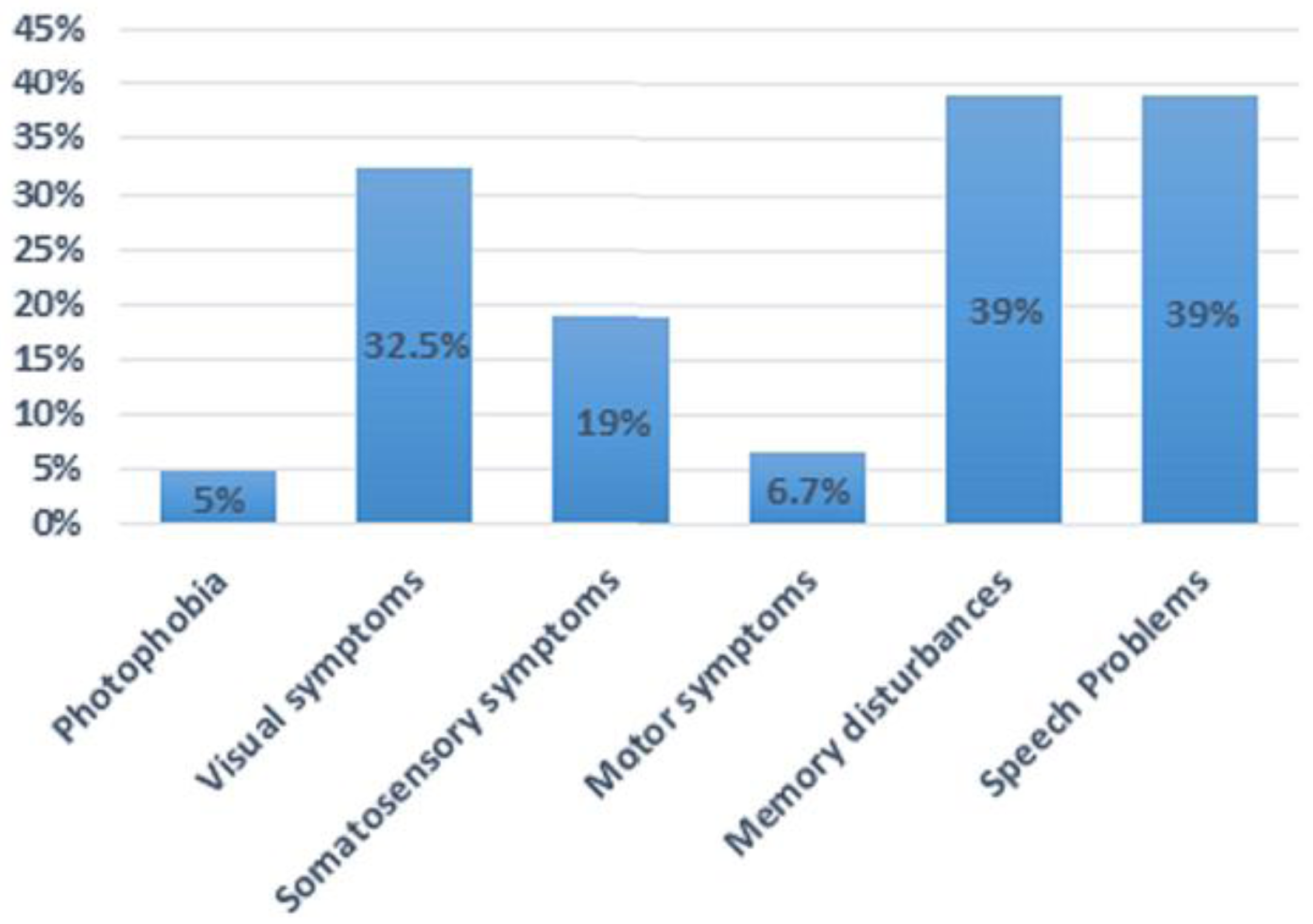
| Total (n = 120) | 68:52 | 5–62 years | Confusion and disorientation (100%), agitation (53%), photophobia (5%), visual (33%), somatosensory (19%), motor (6.7%), speech (39%), memory disturbances (39%) | Pre- (69), post-confusion (11) | 15 min–72 h | ---------- | ---------- | ----------- |
| (A) At least one attack, fulfilling criteria B to G, not attributed to other medical disorder and/or drug intoxication: |
| (B) At least one of the following: |
|
| (C) At least one of the following: |
|
| (D) Complete resolution within 24 h or after sleep with partial or complete amnesia of event |
| (E) Normal neurological or no persistent neurologic deficit examination following the attack |
| (F) At least one of the following: |
|
| (G) Not attributed to another disorder |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farooqi, A.M.; Padilla, J.M.; Monteith, T.S. Acute Confusional Migraine: Distinct Clinical Entity or Spectrum of Migraine Biology? Brain Sci. 2018, 8, 29. https://doi.org/10.3390/brainsci8020029
Farooqi AM, Padilla JM, Monteith TS. Acute Confusional Migraine: Distinct Clinical Entity or Spectrum of Migraine Biology? Brain Sciences. 2018; 8(2):29. https://doi.org/10.3390/brainsci8020029
Chicago/Turabian StyleFarooqi, Ashar M., Jennifer M. Padilla, and Teshamae S. Monteith. 2018. "Acute Confusional Migraine: Distinct Clinical Entity or Spectrum of Migraine Biology?" Brain Sciences 8, no. 2: 29. https://doi.org/10.3390/brainsci8020029
APA StyleFarooqi, A. M., Padilla, J. M., & Monteith, T. S. (2018). Acute Confusional Migraine: Distinct Clinical Entity or Spectrum of Migraine Biology? Brain Sciences, 8(2), 29. https://doi.org/10.3390/brainsci8020029




