Postnatal Migration of Cerebellar Interneurons
Abstract
1. Introduction
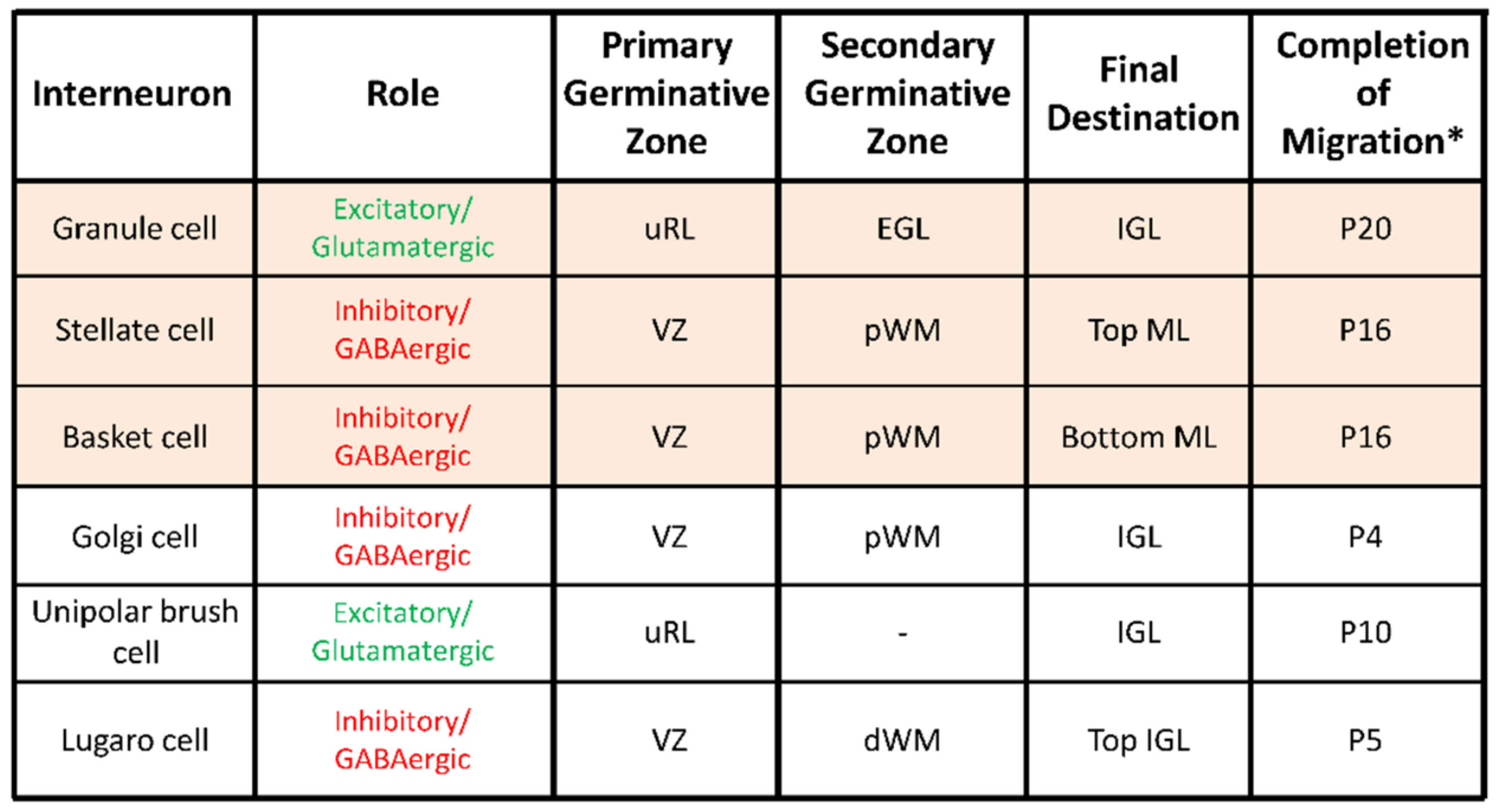
2. Postnatal Cerebellum
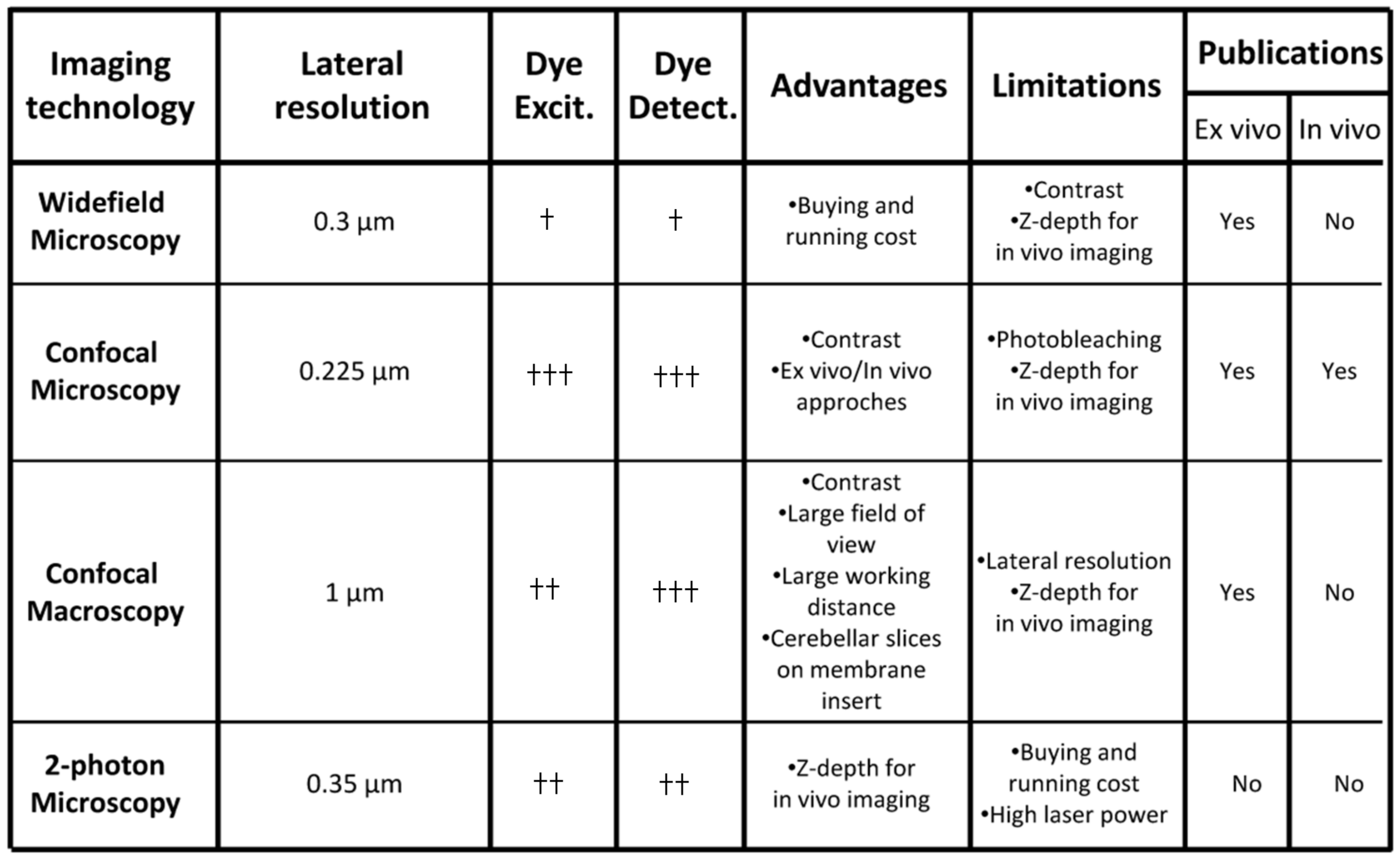
3. Complementary Imaging Approaches for Cell Migration Studies in the Postnatal Cerebellum
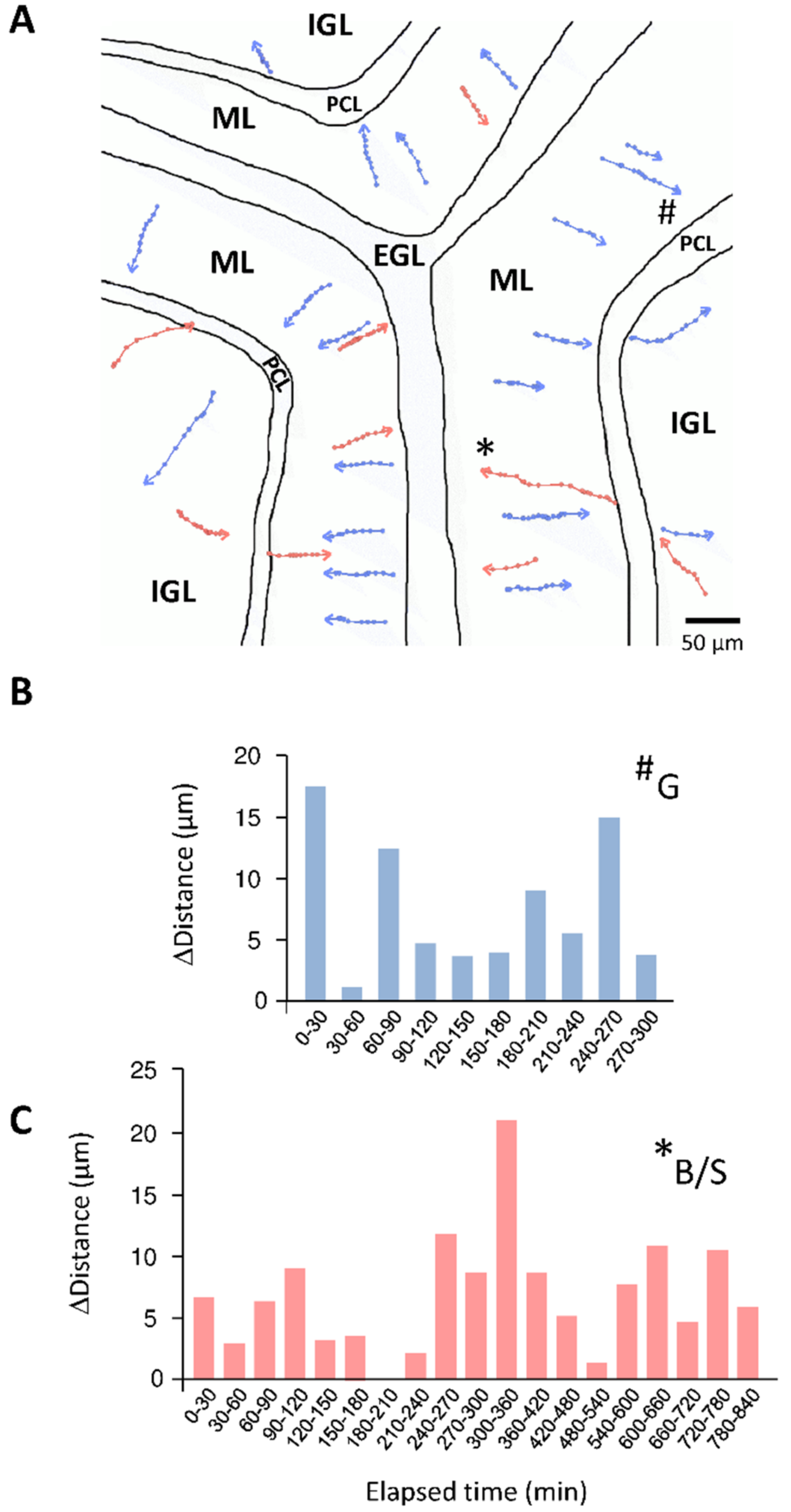
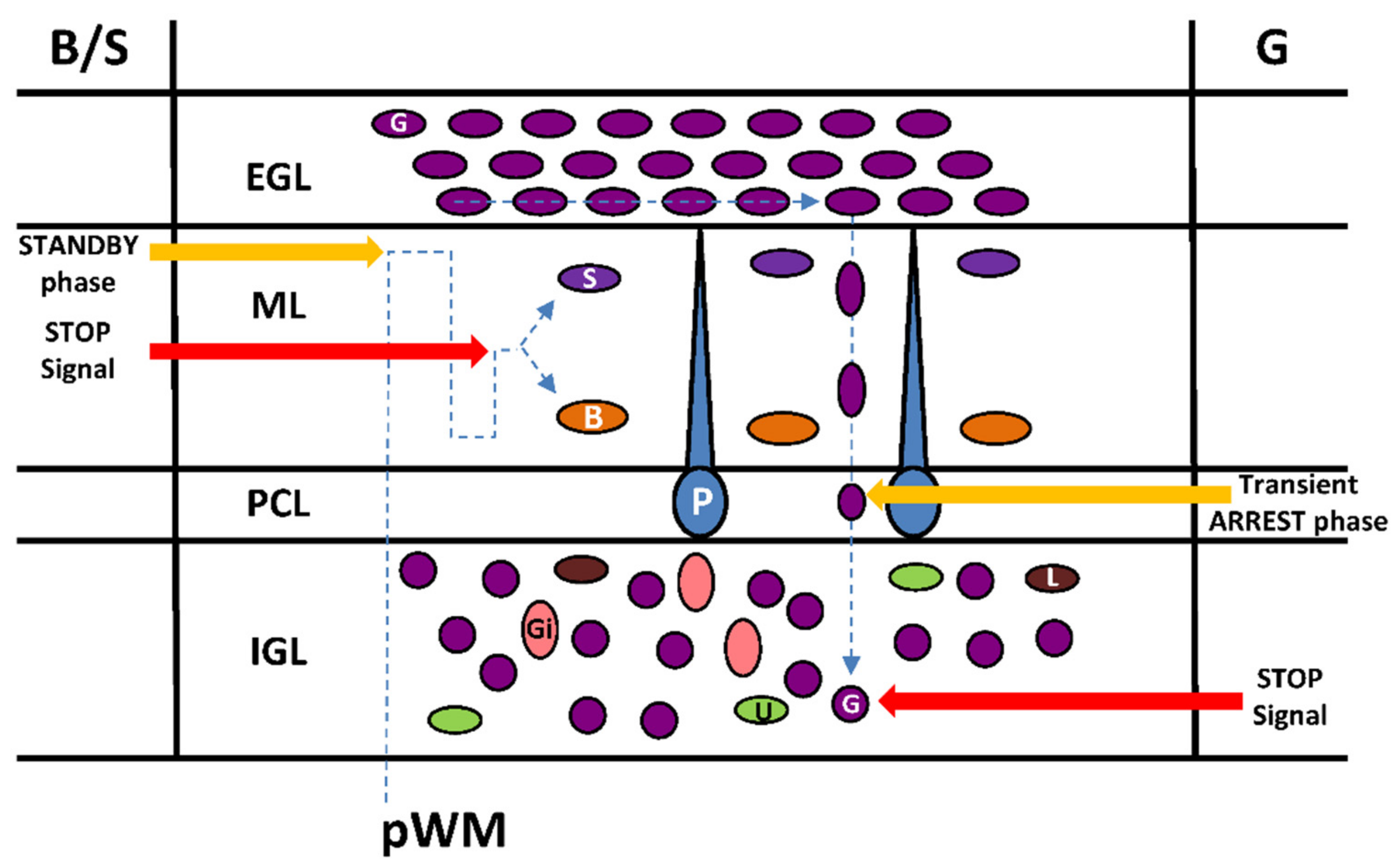
4. Cellular Highway Networks within the First Two Postnatal Weeks of the Cerebellum
5. Transient Arrest Phase, Standby Phase and Final Stop Signal for Cerebellar Interneuron Migration
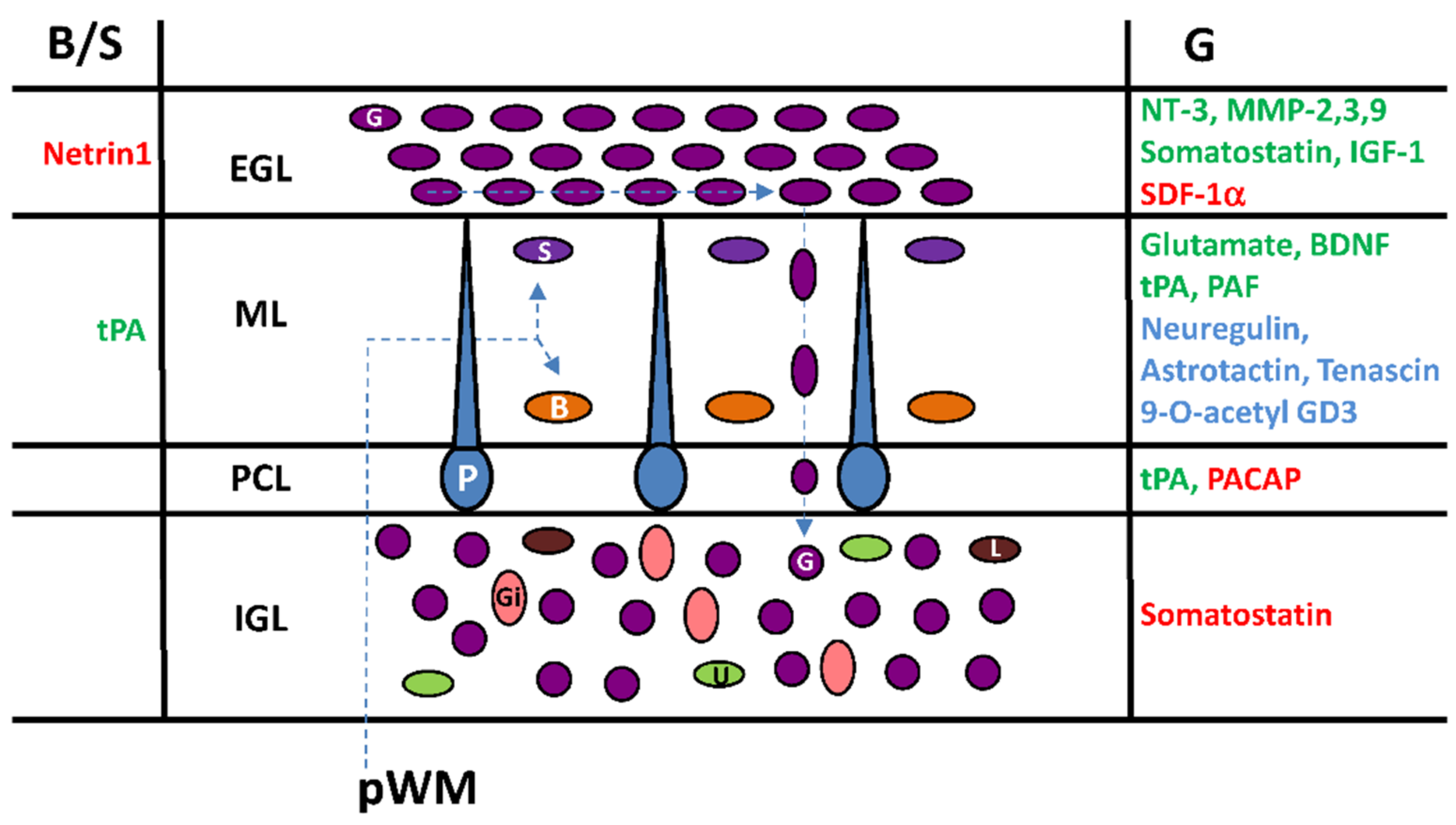
6. Cortical-Layer-Specific Effects of Regulatory Factors during Interneuron Migration
7. Impact of Environmental Conditions, Pollutants, Nutrients, and Drug of Abuse on Interneuron Migration
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ramón y Cajal, S. Cervelet, cerveau moyen, rétine, couche optique, corps strié, écorce cérébrale générale et régionale, grand sympathique. In Histologie du Système Nerveux de L’homme et des Vertébrés, 1st ed.; Azoulay, L., Ed.; Maloine A.: Paris, France, 1911; Volume 2. [Google Scholar]
- Miale, I.L.; Sidman, R.L. An autoradiographic analysis of histogenesis in the mouse cerebellum. Exp. Neurol. 1961, 4, 277–296. [Google Scholar] [CrossRef]
- Rakic, P. Neuron-glia relationship during granule cell migration in developing cerebellar cortex. A Golgi and electonmicroscopic study in Macacus rhesus. J. Comp. Neurol. 1971, 141, 283–312. [Google Scholar] [CrossRef] [PubMed]
- Edmondson, J.C.; Hatten, M.E. Glial-guided granule neuron migration in vitro: A high-resolution time-lapse video microscopic study. J. Neurosci. 1987, 78, 1928–1934. [Google Scholar]
- Sillitoe, R.V.; Joyner, A.L. Morphology, molecular codes, and circuitry produce the three-dimensional complexity of the cerebellum. Annu. Rev. Cell. Dev. Biol. 2007, 23, 549–577. [Google Scholar] [CrossRef] [PubMed]
- Komuro, H.; Yacubova, E. Recent advances in cerebellar granule cell migration. Cell. Mol. Life Sci. 2003, 60, 1084–1098. [Google Scholar] [CrossRef] [PubMed]
- Yuasa, S.; Kawamura, K.; Ono, K.; Yamakuni, T.; Takahashi, Y. Development and migration of Purkinje cells in the mouse cerebellar primordium. Anat. Embryol. 1991, 184, 195–212. [Google Scholar] [CrossRef] [PubMed]
- Maricich, S.M.; Herrup, K. Pax-2 expression defines a subset of GABAergic interneurons and their precursors in the developing murine cerebellum. J. Neurobiol. 1999, 41, 281–294. [Google Scholar] [CrossRef]
- Zhang, L.; Goldman, J.E. Developmental fates and migratory pathways of dividing progenitors in the postnatal rat cerebellum. J. Comp. Neurol. 1996, 370, 536–550. [Google Scholar] [CrossRef]
- Fahrion, J.K.; Komuro, Y.; Ohno, N.; Littner, Y.; Nelson, C.; Kumada, T.; Komuro, H. Cerebellar Patterning. In Comprehensive Developmental Neuroscience: Patterning and Cell Type Specification in the Developing CNS and PNS, 1st ed.; Rubenstein, J., Rakic, P., Eds.; Elsevier Inc.: Oxford, UK, 2013; Volume 1, pp. 211–225. [Google Scholar]
- Komuro, Y.; Kumada, T.; Ohno, N.; Foote, K.D.; Komuro, H. Migration in the Cerebellum. In Cellular Migration and Formation of Neuronal Connections, 1st ed.; Rubenstein, J., Rakic, P., Eds.; Elsevier Inc.: Oxford, UK, 2013; Volume 2, pp. 281–297. [Google Scholar]
- Hsu, Y.H.; Huang, H.Y.; Tsaur, M.L. Contrasting expression of Kv4.3, an A-type K+ channel, in migrating Purkinje cells and other post-migratory cerebellar neurons. Eur. J. Neurosci. 2003, 18, 601–612. [Google Scholar] [CrossRef] [PubMed]
- Raoult, E.; Bénard, M.; Komuro, H.; Lebon, A.; Vivien, D.; Fournier, A.; Vaudry, H.; Vaudry, D.; Galas, L. Cortical-layer-specific effects of PACAP and tPA on interneuron migration during post-natal development of the cerebellum. J. Neurochem. 2014, 130, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Consalez, G.G.; Hawkes, R. The compartmental restriction of cerebellar interneurons. Front. Neural Circuits 2013, 6, 123. [Google Scholar] [CrossRef] [PubMed]
- Bénard, M.; Lebon, A.; Komuro, H.; Vaudry, D.; Galas, L. Ex vivo imaging of postnatal cerebellar granule cell migration using confocal macroscopy. J. Vis. Exp. 2015, e52810. [Google Scholar] [CrossRef] [PubMed]
- Komuro, H.; Rakic, P. Selective role of N-type calcium channels in neuronal migration. Science 1992, 257, 806–809. [Google Scholar] [CrossRef] [PubMed]
- Cameron, D.B.; Galas, L.; Jiang, Y.; Raoult, E.; Vaudry, D.; Komuro, H. Cerebellar cortical-layer-specific control of neuronal migration by pituitary adenylate cyclase-activating polypeptide. Neuroscience 2007, 146, 697–712. [Google Scholar] [CrossRef] [PubMed]
- Cameron, D.B.; Kasai, K.; Jiang, Y.; Hu, T.; Saeki, Y.; Komuro, H. Four distinct phases of basket/stellate cell migration after entering their final destination (the molecular layer) in the developing cerebellum. Dev. Biol. 2009, 332, 309–324. [Google Scholar] [CrossRef] [PubMed]
- Komuro, H.; Rakic, P. Modulation of neuronal migration by NMDA receptors. Science 1993, 260, 95–97. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Komuro, Y.; Fahrion, J.K.; Hu, T.; Ohno, N.; Fenner, K.B.; Wooton, J.; Raoult, E.; Galas, L.; Vaudry, D.; Komuro, H. Light stimuli control neuronal migration by altering of insulin-like growth factor 1 (IGF-1) signaling. Proc. Natl. Acad. Sci. USA 2012, 109, 2630–2635. [Google Scholar] [CrossRef] [PubMed]
- Livet, J.; Weissman, T.A.; Kang, H.; Draft, R.W.; Lu, J.; Bennis, R.A.; Sanes, J.R.; Lichtman, J.W. Transgenic strategies for combinatorial expression of fluorescent proteins in the nervous system. Nature 2007, 450, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Weissman, T.A.; Pan, Y.A. Brainbow: New resources and emerging biological applications for multicolor genetic labeling and analysis. Genetics 2015, 199, 293–306. [Google Scholar] [CrossRef] [PubMed]
- Webb, S.E.; Zanetti-Domingues, L.; Coles, B.C.; Rolfe, D.J.; Wareham, R.J.; Martin-Fernandez, M.L. Multicolour single molecule imaging on cells using a supercontinuum source. Biomed. Opt. Express 2012, 3, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Borlinghaus, R.T.; Birk, H.; Schreiber, F. Detectors for sensitive detection: HyD. In Current Microscopy Contributions to Advances in Science and Technology, 1st ed.; Mendez-Vilas, A., Ed.; Formatex Research Center: Badajoz, Spain, 2012; Volume 2, pp. 818–825. [Google Scholar]
- Fahrion, J.K.; Komuro, Y.; Li, Y.; Ohno, N.; Littner, Y.; Raoult, E.; Galas, L.; Vaudry, D.; Komuro, H. Rescue of neuronal migration deficits in a mouse model of fetal Minamata disease by increasing neuronal Ca2+ spike frequency. Proc. Natl. Acad. Sci. USA 2012, 109, 5057–5062. [Google Scholar] [CrossRef] [PubMed]
- So, P.T.; Dong, C.Y.; Masters, B.R.; Berland, K.M. Two-photon excitation fluorescence microscopy. Annu. Rev. Biomed. Eng. 2000, 2, 399–429. [Google Scholar] [CrossRef] [PubMed]
- Allegra Mascaro, A.L.; Sacconi, L.; Pavone, F.S. Laser nanosurgery of cerebellar axons in vivo. J. Vis. Exp. 2014, e51371. [Google Scholar]
- Ahrens, M.B.; Orger, M.B.; Robson, D.N.; Li, J.M.; Keller, P.J. Whole-brain functional imaging at cellular resolution using light-sheet microscopy. Nat. Methods 2013, 10, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Keller, P.J.; Ahrens, M.B. Visualizing whole-brain activity and development at the single-cell level using light-sheet microscopy. Neuron 2015, 85, 462–483. [Google Scholar] [CrossRef] [PubMed]
- Heap, L.A.; Goh, C.C.; Kassahn, K.S.; Scott, E.K. Cerebellar output in zebrafish: An analysis of spatial patterns and topography in eurydendroid cell projections. Front. Neural Circuits 2013, 7, 53. [Google Scholar] [CrossRef] [PubMed]
- Komuro, H.; Rakic, P. Distinct modes of neuronal migration in different domains of developing cerebellar cortex. J. Neurosci. 1998, 18, 1478–1490. [Google Scholar] [PubMed]
- Komuro, H.; Yacubova, E.; Yacubova, E.; Rakic, P. Mode and tempo of tangential cell migration in the cerebellar external granular layer. J. Neurosci. 2001, 21, 527–540. [Google Scholar] [PubMed]
- Milosevic, A.; Goldman, J.E. Potential of progenitors from postnatal cerebellar neuroepithelium and white matter: Lineage specified vs. multipotent fate. Mol. Cell. Neurosci. 2004, 26, 342–353. [Google Scholar] [CrossRef] [PubMed]
- Rakic, P. Kinetics of proliferation and latency between final cell division and onset of differentiation of cerebellar stellate and basket neurons. J. Comp. Neurol. 1973, 147, 523–546. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, H.; Yanagawa, Y.; Obata, K. Development of stellate and basket cells and their apoptosis in mouse cerebellar cortex. Neurosci. Res. 2004, 50, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Leto, K.; Carletti, B.; Williams, I.M.; Magrassi, L.; Rossi, F. Different types of cerebellar GABAergic interneurons originate from a common pool of multipotent progenitor cells. J. Neurosci. 2006, 26, 11682–11694. [Google Scholar] [CrossRef] [PubMed]
- Weisheit, G.; Gliem, M.; Endl, E.; Pfeffer, P.L.; Busslinger, M.; Schilling, K. Postnatal development of the murine cerebellar cortex: Formation and early dispersal of basket, stellate and Golgi neurons. Eur. J. Neurosci. 2006, 24, 466–478. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Yang, Y.; Tang, X.; Zhao, M.; Liang, F.; Xu, P.; Hou, B.; Xing, Y.; Bao, X.; Fan, X. Bergmann glia function in granule cell migration during cerebellum development. Mol. Neurobiol. 2013, 47, 833–844. [Google Scholar] [CrossRef] [PubMed]
- Komuro, H.; Rakic, P. Dynamics of granule cell migration: A confocal microscopic study in acute cerebellar slice preparations. J. Neurosci. 1995, 15, 1110–1120. [Google Scholar] [PubMed]
- Bénard, M.; Schapman, D.; Lebon, A.; Monterroso, B.; Bellenger, M.; Le Foll, F.; Pasquier, J.; Vaudry, H.; Vaudry, D.; Galas, L. Structural and functional analysis of tunneling nanotubes (TnTs) using gCW STED and gconfocal approaches. Biol. Cell 2015, 107, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Chéreau, R.; Tønnesen, J.; Nägerl, U.V. STED microscopy for nanoscale imaging in living brain slices. Methods 2015, 88, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Chéreau, R.; Saraceno, G.E.; Angibaud, J.; Cattaert, D.; Nägerl, U.V. Superresolution imaging reveals activity-dependent plasticity of axon morphology linked to changes in action potential conduction velocity. Proc. Natl. Acad. Sci. USA 2017, 114, 1401–1406. [Google Scholar] [CrossRef] [PubMed]
- Allegra Mascaro, A.L.; Silvestri, L.; Sacconi, L.; Pavone, F.S. Towards a comprehensive understanding of brain machinery by correlative microscopy. J. Biomed. Opt. 2015, 20, 61105. [Google Scholar] [CrossRef] [PubMed]
- Noctor, S.C.; Martínez-Cerdeño, V.; Ivic, L.; Kriegstein, A.R. Cortical neurons arise in symmetric and asymmetric division zones and migrate through specific phases. Nat. Neurosci. 2004, 7, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Yacubova, E.; Komuro, H. Stage-specific control of neuronal migration by somatostatin. Nature 2002, 415, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Alcántara, S.; Ruiz, M.; De Castro, F.; Soriano, E.; Sotelo, C. Netrin 1 acts as an attractive or as a repulsive cue for distinct migrating neurons during the development of the cerebellar system. Development 2000, 127, 1359–1372. [Google Scholar] [PubMed]
- Nishimoto, M.; Furuta, A.; Aoki, S.; Kudo, Y.; Miyakawa, H.; Wada, K. PACAP/PAC1 autocrine system promotes proliferation and astrogenesis in neural progenitor cells. Glia 2007, 55, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Vaillant, C.; Meissirel, C.; Mutin, M.; Belin, M.F.; Lund, L.R.; Thomasset, N. MMP-9 deficiency affects axonal outgrowth, migration, and apoptosis in the developing cerebellum. Mol. Cell. Neurosci. 2003, 24, 395–408. [Google Scholar] [CrossRef]
- Ayoub, A.E.; Cai, T.Q.; Kaplan, R.A.; Luo, J. Developmental expression of matrix metalloproteinases 2 and 9 and their potential role in the histogenesis of the cerebellar cortex. J. Comp. Neurol. 2005, 481, 403–415. [Google Scholar] [CrossRef] [PubMed]
- Van Hove, I.; Verslegers, M.; Buyens, T.; Delorme, N.; Lemmens, K.; Stroobants, S.; Gantois, I.; D’Hooge, R.; Moons, L. An aberrant cerebellar development in mice lacking matrix metalloproteinase-3. Mol. Neurobiol. 2012, 45, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Borghesani, P.R.; Peyrin, J.M.; Klein, R.; Rubin, J.; Carter, A.R.; Schwartz, P.M.; Luster, A.; Corfas, G.; Segal, R.A. BDNF stimulates migration of cerebellar granule cells. Development 2002, 129, 1435–1442. [Google Scholar] [PubMed]
- Ma, Q.; Jones, D.; Borghesani, P.R.; Segal, R.A.; Nagasawa, T.; Kishimoto, T.; Bronson, R.T.; Springer, T.A. Impaired B-lymphopoiesis, myelopoiesis, and derailed cerebellar neuron migration in CXCR4- and SDF-1-deficient mice. Proc. Natl. Acad. Sci. USA 1998, 95, 9448–9453. [Google Scholar] [CrossRef] [PubMed]
- Seeds, N.W.; Basham, M.E.; Haffke, S.P. Neuronal migration is retarded in mice lacking the tissue plasminogen activator gene. Proc. Natl. Acad. Sci. USA 1999, 96, 14118–14123. [Google Scholar] [CrossRef] [PubMed]
- Adams, N.C.; Tomoda, T.; Cooper, M.; Dietz, G.; Hatten, M.E. Mice that lack astrotactin have slowed neuronal migration. Development 2002, 129, 965–972. [Google Scholar] [PubMed]
- Doughty, M.L.; Lohof, A.; Campana, A.; Delhaye-Bouchaud, N.; Mariani, J. Neurotrophin-3 promotes cerebellar granule cell exit from the EGL. Eur. J. Neurosci. 1998, 10, 3007–3011. [Google Scholar] [CrossRef] [PubMed]
- Husmann, K.; Faissner, A.; Schachner, M. Tenascin promotes cerebellar granule cell migration and neurite outgrowth by different domains in the fibronectin type III repeats. J. Cell. Biol. 1992, 116, 1475–1486. [Google Scholar] [CrossRef] [PubMed]
- Santiago, M.F.; Berredo-Pinho, M.; Costa, M.R.; Gandra, M.; Cavalcante, L.A.; Mendez-Otero, R. Expression and function of ganglioside 9-O-acetyl GD3 in postmitotic granule cell development. Mol. Cell. Neurosci. 2001, 17, 488–499. [Google Scholar] [CrossRef] [PubMed]
- Bix, G.J.; Clark, G.D. Platelet-activating factor receptor stimulation disrupts neuronal migration in vitro. J. Neurosci. 1998, 18, 307–318. [Google Scholar] [PubMed]
- Rio, C.; Rieff, H.I.; Qi, P.; Khurana, T.S.; Corfas, G. Neuregulin and erbB receptors play a critical role in neuronal migration. Neuron 1997, 19, 39–50. [Google Scholar] [CrossRef]
- Patel, Y.C. Somatostatin and its receptor family. Front. Neuroendocrinol. 1999, 20, 157–198. [Google Scholar] [CrossRef] [PubMed]
- Tostivint, H.; Ocampo Daza, D.; Bergqvist, C.A.; Quan, F.B.; Bougerol, M.; Lihrmann, I.; Larhammar, D. Molecular evolution of GPCRs: Somatostatin/urotensin II receptors. J. Mol. Endocrinol. 2014, 52, T61–T86. [Google Scholar] [CrossRef] [PubMed]
- Thoss, V.S.; Duc, D.; Hoyer, D. Somatostatin receptors in the developing rat brain. Eur. J. Pharmacol. 1996, 297, 145–155. [Google Scholar] [CrossRef]
- Miyata, A.; Arimura, A.; Dahl, R.R.; Minamino, N.; Uehara, A.; Jiang, L.; Culler, M.D.; Coy, D.H. Isolation of a novel 38 residue-hypothalamic polypeptide which stimulates adenylate cyclase in pituitary cells. Biochem. Biophys. Res. Commun. 1989, 164, 567–574. [Google Scholar] [CrossRef]
- Miyata, A.; Jiang, L.; Dahl, R.D.; Kitada, C.; Kubo, K.; Fujino, M.; Minamino, N.; Arimura, A. Isolation of a neuropeptide corresponding to the N-terminal 27 residues of the pituitary adenylate cyclase activating polypeptide with 38 residues (PACAP38). Biochem. Biophys. Res. Commun. 1990, 170, 643–648. [Google Scholar] [CrossRef]
- Vaudry, D.; Gonzalez, B.J.; Basille, M.; Yon, L.; Fournier, A.; Vaudry, H. Pituitary adenylate cyclase-activating polypeptide and its receptors: From structure to functions. Pharmacol. Rev. 2000, 52, 269–324. [Google Scholar] [PubMed]
- Nielsen, H.S.; Hannibal, J.; Fahrenkrug, J. Expression of pituitary adenylate cyclase activating polypeptide (PACAP) in the postnatal and adult rat cerebellar cortex. NeuroReport 1998, 9, 2639–2642. [Google Scholar] [CrossRef] [PubMed]
- Hannibal, J. Pituitary adenylate cyclase-activating peptide in the rat central nervous system: An immunohistochemical and in situ hybridization study. J. Comp. Neurol. 2002, 453, 389–417. [Google Scholar] [CrossRef] [PubMed]
- Basille, M.; Gonzalez, B.J.; Fournier, A.; Vaudry, H. Ontogeny of pituitary adenylate cyclase-activating polypeptide (PACAP) receptors in the rat cerebellum: A quantitative autoradiographic study. Brain Res. Dev. Brain Res. 1994, 82, 81–89. [Google Scholar] [CrossRef]
- Basille, M.; Vaudry, D.; Coulouarn, Y.; Jegou, S.; Lihrmann, I.; Fournier, A.; Vaudry, H.; Gonzalez, B. Comparative distribution of pituitary adenylate cyclase-activating polypeptide (PACAP) binding sites and PACAP receptor mRNAs in the rat brain during development. J. Comp. Neurol. 2000, 425, 495–509. [Google Scholar] [CrossRef]
- Osborne, J.G.; Kindy, M.S.; Spruce, B.A.; Hauser, K.F. Ontogeny of proenkephalin mRNA and enkephalin peptide expression in the cerebellar cortex of the rat: Spatial and temporal patterns of expression follow maturational gradients in the external granular layer and in Purkinje cells. Brain Res. Dev. Brain Res. 1993, 76, 1–12. [Google Scholar] [CrossRef]
- Ryan, M.C.; Loiacono, R.E.; Gundlach, A.L. Galanin messenger RNA during postnatal development of the rat brain: Expression patterns in Purkinje cells differentiate anterior and posterior lobes of cerebellum. Neuroscience 1997, 78, 1113–1127. [Google Scholar] [CrossRef]
- García-Rocha, M.; Avila, J.; Armas-Portela, R. Tissue-type plasminogen activator (tPA) is the main plasminogen activator associated with isolated rat nerve growth cones. Neurosci. Lett. 1994, 180, 123–126. [Google Scholar] [CrossRef]
- Yepes, M.; Lawrence, D.A. Tissue-type plasminogen activator and neuroserpin: A well-balanced act in the nervous system. Trends Cardiovasc. Med. 2004, 14, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Friedman, G.C.; Seeds, N.W. Tissue plasminogen activator mRNA expression in granule neurons coincides with their migration in the developing cerebellum. J. Comp. Neurol. 1995, 360, 658–670. [Google Scholar] [CrossRef] [PubMed]
- Ware, J.H.; DiBenedetto, A.J.; Pittman, R.N. Localization of tissue plasminogen activator mRNA in the developing rat cerebellum and effects of inhibiting tissue plasminogen activator on granule cell migration. J. Neurobiol. 1995, 28, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Krystosek, A.; Seeds, N.W. Plasminogen activator release at the neuronal growth cone. Science 1981, 213, 1532–1534. [Google Scholar] [CrossRef] [PubMed]
- Krystosek, A.; Seeds, N.W. Plasminogen activator secretion by granule neurons in cultures of developing cerebellum. Proc. Natl. Acad. Sci. USA 1981, 78, 7810–7814. [Google Scholar] [CrossRef] [PubMed]
- Seeds, N.W.; Siconolfi, L.B.; Haffke, S.P. Neuronal extracellular proteases facilitate cell migration, axonal growth, and pathfinding. Cell Tissue Res. 1997, 290, 367–370. [Google Scholar] [CrossRef] [PubMed]
- Basham, M.E.; Seeds, N.W. Plasminogen expression in the neonatal and adult mouse brain. J. Neurochem. 2001, 77, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Allin, M.P. Novel insights from quantitative imaging of the developing cerebellum. Semin. Fetal Neonatal Med. 2016, 21, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Bakkum, B.W.; Benevento, L.A.; Cohen, R.S. Effects of light/dark- and dark-rearing on synaptic morphology in the superior colliculus and visual cortex of the postnatal and adult rat. J. Neurosci. Res. 1991, 28, 65–80. [Google Scholar] [CrossRef] [PubMed]
- Brooks, E.; Waters, E.; Farrington, L.; Canal, M.M. Differential hypothalamic tyrosine hydroxylase distribution and activation by light in adult mice reared under different light conditions during the suckling period. Brain Struct. Funct. 2011, 216, 357–370. [Google Scholar] [CrossRef] [PubMed]
- Dulcis, D.; Spitzer, N.C. Illumination controls differentiation of dopamine neurons regulating behaviour. Nature 2008, 456, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Harada, M. Neuropsychiatric disturbances due to organic mercury poisoning during the prenatal period. Seishin Shinkeigaku Zasshi 1964, 66, 429–468. [Google Scholar] [PubMed]
- Snyder, R.D. Congenital mercury poisoning. N. Engl. J. Med. 1971, 284, 1014–1016. [Google Scholar] [CrossRef] [PubMed]
- Eto, K. Pathology of Minamata disease. Toxicol. Pathol. 1997, 25, 614–623. [Google Scholar] [CrossRef] [PubMed]
- Barkur, R.R.; Bairy, L.K. Histological study on hippocampus, amygdala and cerebellum following low lead exposure during prenatal and postnatal brain development in rats. Toxicol. Ind. Health 2016, 32, 1052–1063. [Google Scholar] [CrossRef] [PubMed]
- Georgieff, M.K. Nutrition and the developing brain: Nutrient priorities and measurement. Am. J. Clin. Nutr. 2007, 85, 614S–620S. [Google Scholar] [PubMed]
- Thompson, B.L.; Levitt, P.; Stanwood, G.D. Prenatal exposure to drugs: Effects on brain development and implications for policy and education. Nat. Rev. Neurosci. 2009, 10, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Sidman, R.L.; Rakic, P. Neuronal migration, with special reference to developing human brain: A review. Brain Res. 1973, 62, 1–35. [Google Scholar] [CrossRef]
- Johnson, J.; Wu, V.; Donovan, M.; Majumdar, S.; Rentería, R.C.; Porco, T.; Van Gelder, R.N.; Copenhagen, D.R. Melanopsin-dependent light avoidance in neonatal mice. Proc. Natl. Acad. Sci. USA 2010, 107, 17374–17378. [Google Scholar] [CrossRef] [PubMed]
- Amin-Zaki, L.; Elhassani, S.; Majeed, M.A.; Clarkson, T.W.; Doherty, R.A.; Greenwood, M.R. Studies of infants postnatally exposed to methylmercury. J. Pediatr. 1974, 85, 81–84. [Google Scholar] [CrossRef]
- Matsumoto, H.; Koya, G.; Takeuchi, T. Fetal Minamata disease. A neuropathological study of two cases of intrauterine intoxication by a methyl mercury compound. J. Neuropathol. Exp. Neurol. 1965, 24, 563–574. [Google Scholar] [CrossRef] [PubMed]
- Eto, K.; Oyanagi, S.; Itai, Y.; Tokunaga, H.; Takizawa, Y.; Suda, I. A fetal type of Minamata disease. An autopsy case report with special reference to the nervous system. Mol. Chem. Neuropathol. 1992, 16, 171–186. [Google Scholar] [CrossRef] [PubMed]
- Cace, I.B.; Milardovic, A.; Prpic, I.; Krajina, R.; Petrovic, O.; Vukelic, P.; Spiric, Z.; Horvat, M.; Mazej, D.; Snoj, J. Relationship between the prenatal exposure to low-level of mercury and the size of a newborn’s cerebellum. Med. Hypotheses 2011, 76, 514–516. [Google Scholar] [CrossRef] [PubMed]
- Levitsky, D.A.; Strupp, B.J. Malnutrition and the brain: Changing concepts, changing concerns. J. Nutr. 1995, 125, 2212S–2220S. [Google Scholar] [PubMed]
- Golub, M.S.; Takeuchi, P.T.; Keen, C.L.; Gershwin, M.E.; Hendrickx, A.G.; Lonnerdal, B. Modulation of behavioral performance of prepubertal monkeys by moderate dietary zinc deprivation. Am. J. Clin. Nutr. 1994, 60, 238–243. [Google Scholar] [PubMed]
- Penland, J.G.; Prohaska, J.R. Abnormal motor function persists following recovery from perinatal copper deficiency in rats. J. Nutr. 2004, 134, 1984–1988. [Google Scholar] [PubMed]
- McNall, A.D.; Etherton, T.D.; Fosmire, G.J. The impaired growth induced by zinc deficiency in rats is associated with decreased expression of the hepatic insulin-like growth factor I and growth hormone receptor genes. J. Nutr. 1995, 125, 874–879. [Google Scholar] [PubMed]
- Keunen, K.; van Elburg, R.M.; van Bel, F.; Benders, M.J. Impact of nutrition on brain development and its neuroprotective implications following preterm birth. Pediatr. Res. 2015, 77, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.L.; Smith, D.W. Recognition of the fetal alcohol syndrome in early infancy. Lancet 1973, 302, 999–1001. [Google Scholar] [CrossRef]
- Little, R.E.; Anderson, K.W.; Ervin, C.H.; Worthington-Roberts, B.; Clarren, S.K. Maternal alcohol use during breast-feeding and infant mental and motor development at one year. N. Engl. J. Med. 1989, 321, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Chiriboga, C.A. Fetal alcohol and drug effects. Neurologist 2003, 9, 267–279. [Google Scholar] [CrossRef] [PubMed]
- Lemoine, P.; Harousseau, H.; Borteyru, J.P.; Menuet, J.C. Children of alcoholic parents-observed anomalies: Discussion of 127 cases. Ther. Drug Monit. 2003, 25, 132–136. [Google Scholar] [CrossRef] [PubMed]
- Sokol, R.J.; Delaney-Black, V.; Nordstrom, B. Fetal alcohol spectrum disorder. JAMA 2003, 290, 2996–2999. [Google Scholar] [CrossRef] [PubMed]
- Manzardo, A.M.; Penick, E.C.; Knop, J.; Nickel, E.J.; Hall, S.; Jensen, P.; Gabrielli, W.F., Jr. Developmental differences in childhood motor coordination predict adult alcohol dependence: Proposed role for the cerebellum in alcoholism. Alcohol. Clin. Exp. Res. 2005, 29, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Kornguth, S.E.; Rutledge, J.J.; Sunderland, E.; Siegel, F.; Carlson, I.; Smollens, J.; Juhl, U.; Young, B. Impeded cerebellar development and reduced serum thyroxine levels associated with fetal alcohol intoxication. Brain Res. 1979, 177, 347–360. [Google Scholar] [CrossRef]
- Sakata-Haga, H.; Sawada, K.; Hisano, S.; Fukui, Y. Abnormalities of cerebellar foliation in rats prenatally exposed to ethanol. Acta. Neuropathol. 2001, 102, 36–40. [Google Scholar] [PubMed]
- Dikranian, K.; Qin, Y.Q.; Labruyere, J.; Nemmers, B.; Olney, J.W. Ethanol-induced neuroapoptosis in the developing rodent cerebellum and related brain stem structures. Brain Res. Dev. Brain Res. 2005, 155, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Kumada, T.; Lakshmana, M.K.; Komuro, H. Reversal of neuronal migration in a mouse model of fetal alcohol syndrome by controlling second-messenger signalings. J. Neurosci. 2006, 26, 742–756. [Google Scholar] [CrossRef] [PubMed]
- Kumada, T.; Komuro, Y.; Li, Y.; Hu, T.; Wang, Z.; Littner, Y.; Komuro, H. Inhibition of cerebellar granule cell turning by alcohol. Neuroscience 2010, 170, 1328–1344. [Google Scholar] [CrossRef] [PubMed]
- Bihannic, L.; Ayrault, O. Insights into cerebellar development and medulloblastoma. Bull. Cancer 2016, 103, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Stoodley, C.J.; Limperopoulos, C. Structure-function relationships in the developing cerebellum: Evidence from early-life cerebellar injury and neurodevelopmental disorders. Semin. Fetal Neonatal Med. 2016, 21, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Wiethoff, S.; Arber, C.; Li, A.; Wray, S.; Houlden, H.; Patani, R. Using human induced pluripotent stem cells to model cerebellar disease: Hope and hype. J. Neurogenet. 2015, 29, 95–102. [Google Scholar] [CrossRef] [PubMed]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galas, L.; Bénard, M.; Lebon, A.; Komuro, Y.; Schapman, D.; Vaudry, H.; Vaudry, D.; Komuro, H. Postnatal Migration of Cerebellar Interneurons. Brain Sci. 2017, 7, 62. https://doi.org/10.3390/brainsci7060062
Galas L, Bénard M, Lebon A, Komuro Y, Schapman D, Vaudry H, Vaudry D, Komuro H. Postnatal Migration of Cerebellar Interneurons. Brain Sciences. 2017; 7(6):62. https://doi.org/10.3390/brainsci7060062
Chicago/Turabian StyleGalas, Ludovic, Magalie Bénard, Alexis Lebon, Yutaro Komuro, Damien Schapman, Hubert Vaudry, David Vaudry, and Hitoshi Komuro. 2017. "Postnatal Migration of Cerebellar Interneurons" Brain Sciences 7, no. 6: 62. https://doi.org/10.3390/brainsci7060062
APA StyleGalas, L., Bénard, M., Lebon, A., Komuro, Y., Schapman, D., Vaudry, H., Vaudry, D., & Komuro, H. (2017). Postnatal Migration of Cerebellar Interneurons. Brain Sciences, 7(6), 62. https://doi.org/10.3390/brainsci7060062






