Sex Differences in Neuromuscular Fatigability of the Knee Extensors Post-Stroke
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Experimental Setup and Data Collection
2.3. Torque Measurements
2.4. Electromyography Measurements
2.5. Resting Twitch Measurements
2.6. Experimental Protocol
2.7. Data Processing
2.8. Statistical Analysis
3. Results
3.1. Baseline Measurements
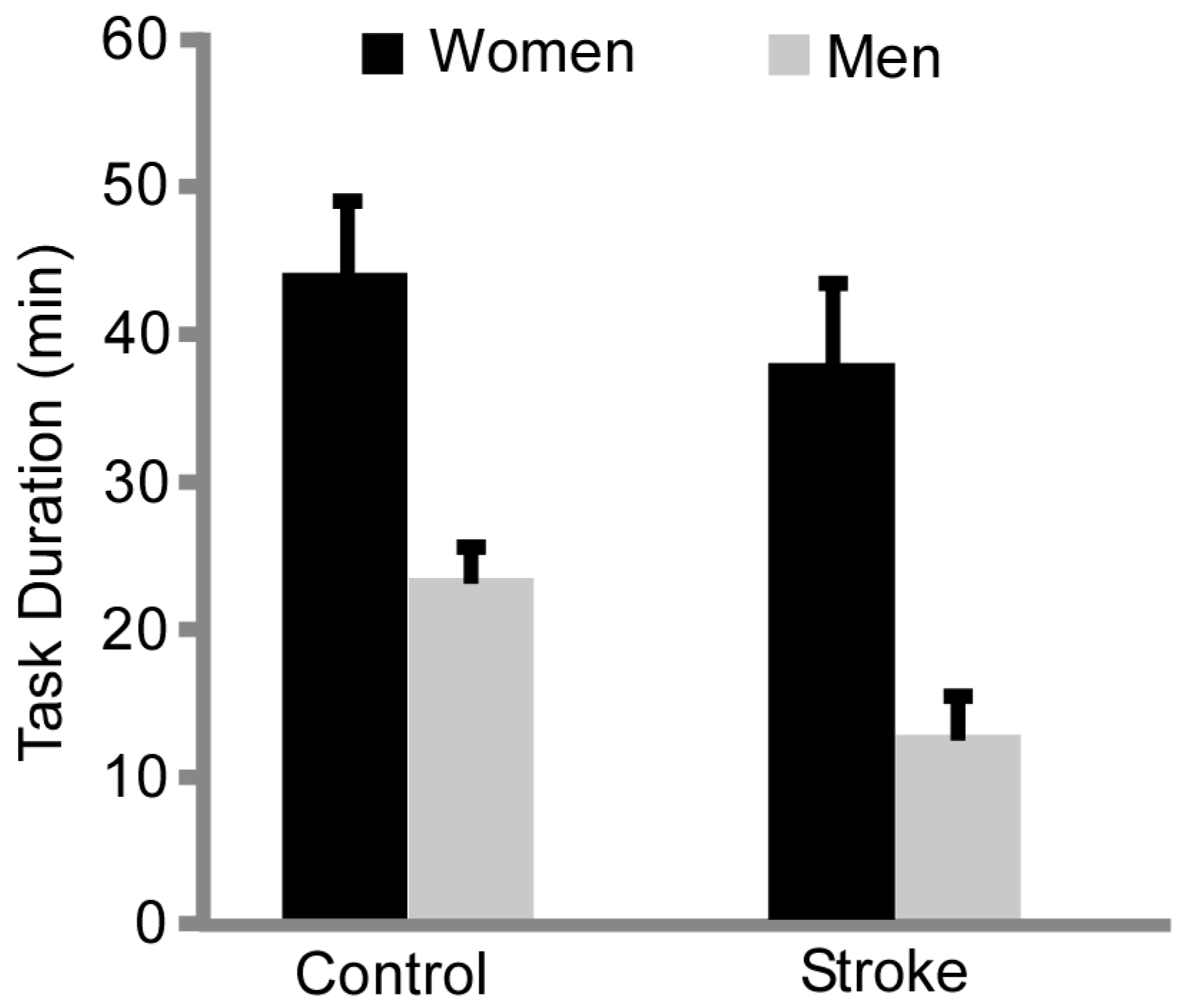
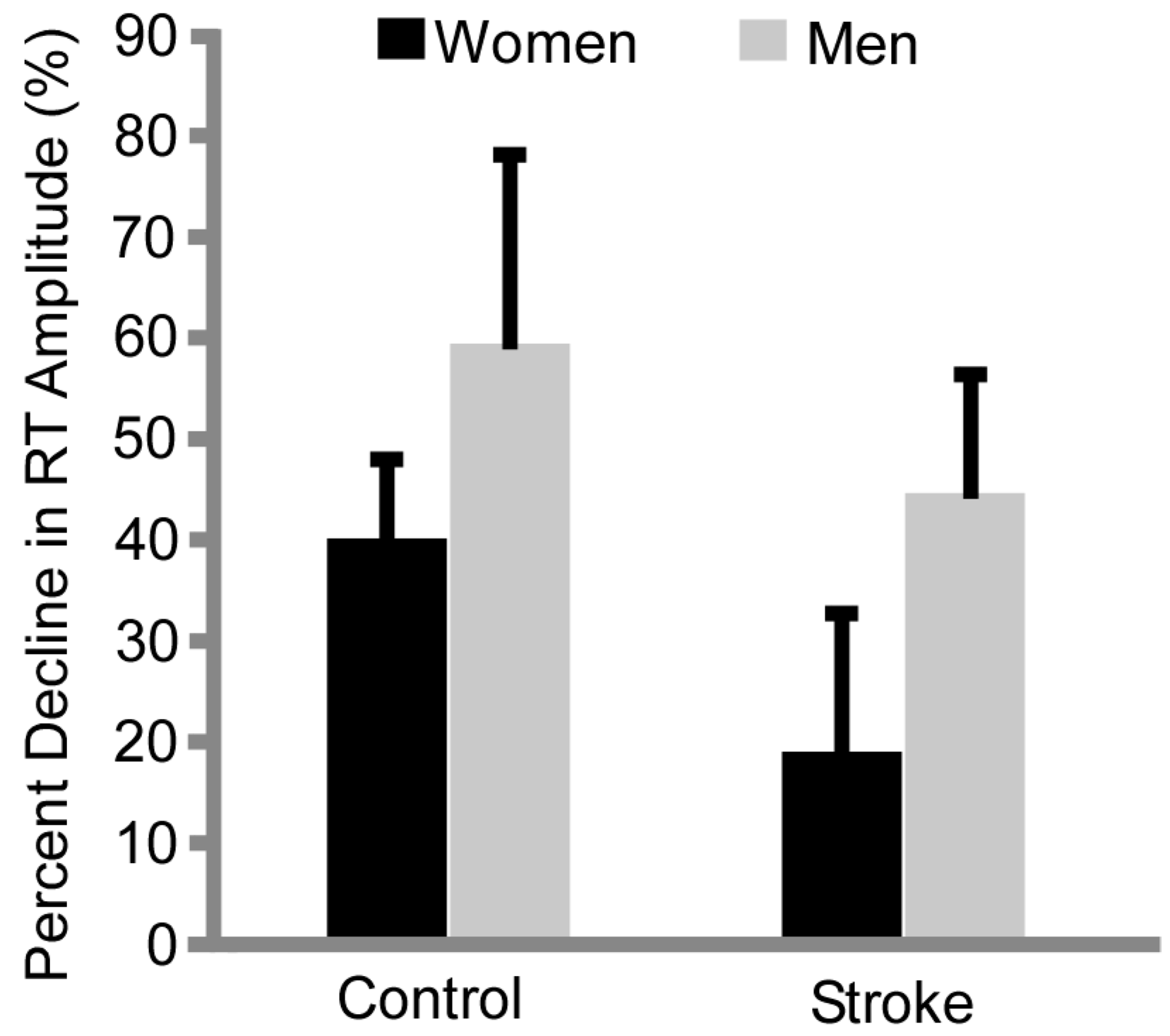
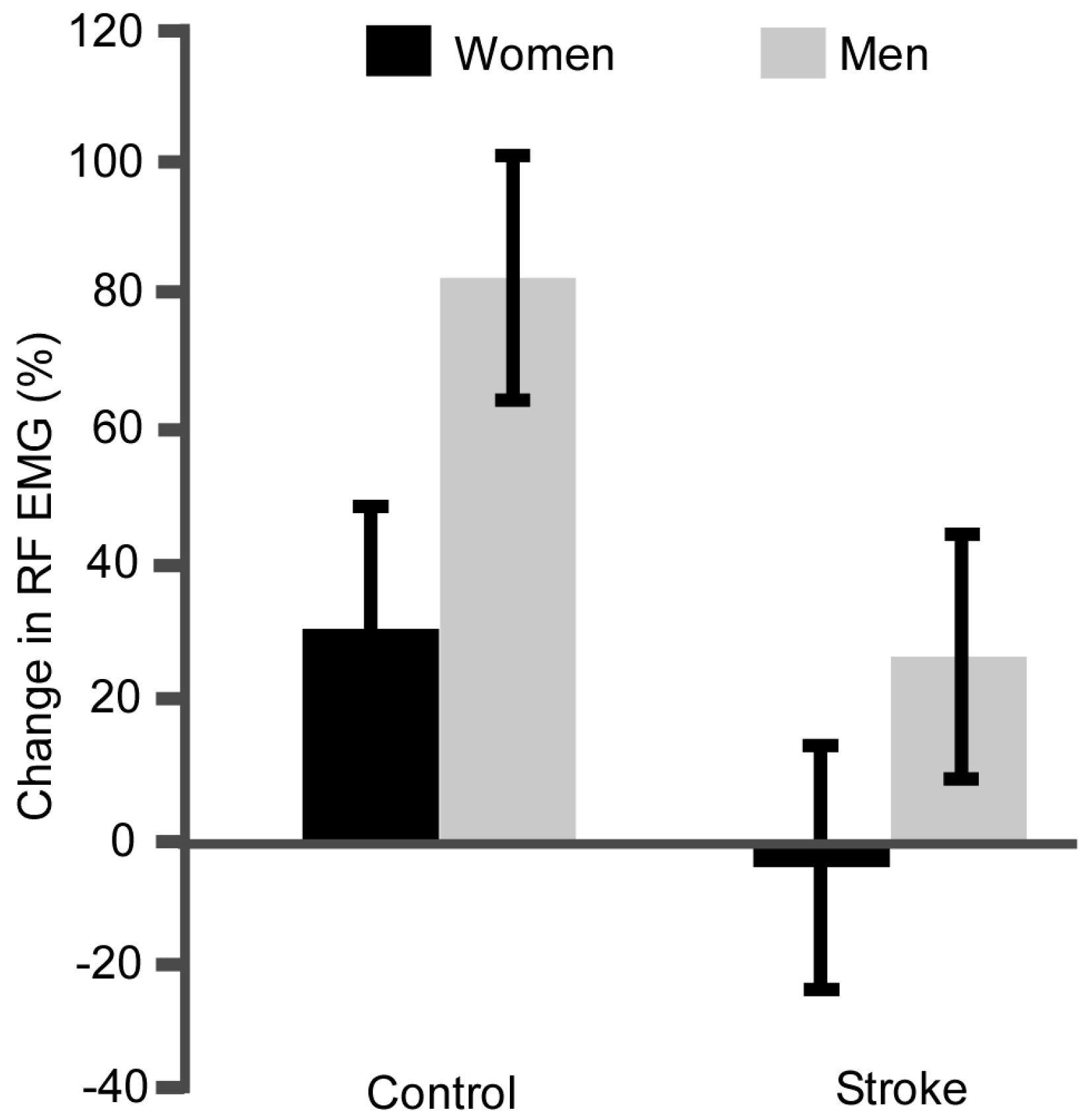
3.2. Fatigability
3.3. Relationship between Neuromuscular Fatigue Metrics and Clinical Measures
4. Discussion
4.1. Sex Differences in Baseline Strength Persist with Stroke
4.2. Sex Differences in Time to Task Failure Maintained with Stroke
4.3. Differences in Resting Twitch Amplitude Provides Insight into Mechanisms of Fatigue
4.4. Neural Strategies of Muscle Activation Differ with Stroke and Sex
4.5. Fatigue Metrics Associated with Neuromuscular Properties Relate to Walking Function
4.6. Next Steps: Perceived Fatigue and Motor Performance Post-Stroke
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lloyd-Jones, D.; Adams, R.; Carnethon, M.; De Simone, G.; Ferguson, T.B.; Flegal, K.; Ford, E.; Furie, K.; Go, A.; Greenlund, K.; et al. Heart disease and stroke statistics—2009 update: A report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation 2009, 119, 480–486. [Google Scholar] [PubMed]
- Nadeau, S.; Arsenault, A.B.; Gravel, D.; Bourbonnais, D. Analysis of the clinical factors determining natural and maximal gait speeds in adults with a stroke. Am. J. Phys. Med. Rehabil. 1999, 78, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Nadeau, S.; Gravel, D.; Arsenault, A.B.; Bourbonnais, D. Plantarflexor weakness as a limiting factor of gait speed in stroke subjects and the compensating role of hip flexors. Clin. Biomech. 1999, 14, 125–135. [Google Scholar] [CrossRef]
- Jonkers, I.; Delp, S.; Patten, C. Capacity to increase walking speed is limited by impaired hip and ankle power generation in lower functioning persons post-stroke. Gait Posture 2009, 29, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Hunter, S.K. The relevance of sex differences in performance fatigability. Med. Sci. Sports Exerc. 2016, 48, 2247–2256. [Google Scholar] [CrossRef] [PubMed]
- Kuhnen, H.R.; Rybar, M.M.; Onushko, T.; Doyel, R.E.; Hunter, S.K.; Schmit, B.D.; Hyngstrom, A.S. Stroke-related effects on maximal dynamic hip flexor fatigability and functional implications. Muscle Nerve 2015, 51, 446–448. [Google Scholar] [CrossRef] [PubMed]
- Hyngstrom, A.S.; Onushko, T.; Heitz, R.P.; Rutkowski, A.; Hunter, S.K.; Schmit, B.D. Stroke-related changes in neuromuscular fatigue of the hip flexors and functional implications. Am. J. Phys. Med. Rehabil. 2012, 91, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Enoka, R.M.; Duchateau, J. Muscle fatigue: What, why and how it influences muscle function. J. Physiol. 2008, 586, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Rybar, M.; Walker, E.; Kuhnen, H.; Ouellette, D.; Berrios, R.; Hunter, S.K.; Hyngstrom, A.S. The stroke-related effects of hip flexion fatigue on over ground walking. Gait Posture 2014, 39, 1103–1108. [Google Scholar] [CrossRef] [PubMed]
- Pohl, P.S.; Duncan, P.W.; Perera, S.; Liu, W.; Lai, S.M.; Studenski, S.; Long, J. Influence of stroke-related impairments on performance in 6-minute walk test. J. Rehabil. Res. Dev. 2002, 39, 439–444. [Google Scholar] [PubMed]
- Bayat, R.; Barbeau, H.; Lamontagne, A. Speed and temporal-distance adaptations during treadmill and overground walking following stroke. Neurorehabil. Neural Repair. 2005, 19, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Dean, C.M.; Richards, C.L.; Malouin, F. Walking speed over 10 metres overestimates locomotor capacity after stroke. Clin. Rehabil. 2001, 15, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Sibley, K.M.; Tang, A.; Brooks, D.; McIlroy, W.E. Effects of extended effortful activity on spatio-temporal parameters of gait in individuals with stroke. Gait Posture 2008, 27, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Sibley, K.M.; Tang, A.; Patterson, K.K.; Brooks, D.; McIlroy, W.E. Changes in spatiotemporal gait variables over time during a test of functional capacity after stroke. J. Neuroeng. Rehabil. 2009, 6, 27. [Google Scholar] [CrossRef] [PubMed]
- Knorr, S.; Ivanova, T.D.; Doherty, T.J.; Campbell, J.A.; Garland, S.J. The origins of neuromuscular fatigue post-stroke. Exp. Brain Res. 2011, 214, 303–315. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; An, Z.; Zhao, W.; Wang, W.; Gao, C.; Liu, S.; Wang, J.; Wu, J. Sex differences in stroke subtypes, severity, risk factors, and outcomes among elderly patients with acute ischemic stroke. Front. Aging Neurosci. 2015, 7, 174. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; An, Z.; Hong, Y.; Zhou, G.; Liu, B.; Guo, J.; Yang, Y.; Ning, X.; Wang, J. Sex differences in long-term outcomes among acute ischemic stroke patients with diabetes in China. Biol. Sex. Differ. 2015, 6, 29. [Google Scholar] [CrossRef] [PubMed]
- Lindbohm, J.V.; Kaprio, J.; Jousilahti, P.; Salomaa, V.; Korja, M. Sex, smoking, and risk for subarachnoid hemorrhage. Stroke 2016, 47, 1975–1981. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, M.J.; Chin, S.L.; Rangarajan, S.; Xavier, D.; Liu, L.; Zhang, H.; Rao-Melacini, P.; Zhang, X.; Pais, P.; Agapay, S.; et al. Global and regional effects of potentially modifiable risk factors associated with acute stroke in 32 countries (INTERSTROKE): A case-control study. Lancet 2016, 388, 761–765. [Google Scholar] [CrossRef]
- Roth, D.L.; Sheehan, O.C.; Huang, J.; Rhodes, J.D.; Judd, S.E.; Kilgore, M.; Kissela, B.; Bettger, J.P.; Haley, W.E. Medicare claims indicators of healthcare utilization differences after hospitalization for ischemic stroke: Race, gender, and caregiving effects. Int. J. Stroke 2016, 11, 928–934. [Google Scholar] [CrossRef] [PubMed]
- Roquer, J.; Rodriguez-Campello, A.; Jimenez-Conde, J.; Cuadrado-Godia, E.; Giralt-Steinhauer, E.; Vivanco Hidalgo, R.M.; Soriano, C.; Ois, A. Sex-related differences in primary intracerebral hemorrhage. Neurology 2016, 87, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Hunter, S.K. Sex differences in human fatigability: Mechanisms and insight to physiological responses. Acta Physiol. 2014, 210, 768–789. [Google Scholar] [CrossRef] [PubMed]
- Hunter, S.K.; Enoka, R.M. Sex differences in the fatigability of arm muscles depends on absolute force during isometric contractions. J. Appl. Physiol. 2001, 91, 2686–2694. [Google Scholar] [PubMed]
- Hunter, S.K.; Butler, J.E.; Todd, G.; Gandevia, S.C.; Taylor, J.L. Supraspinal fatigue does not explain the sex difference in muscle fatigue of maximal contractions. J. Appl. Physiol. 2006, 101, 1036–1044. [Google Scholar] [CrossRef] [PubMed]
- Wust, R.C.; Morse, C.I.; de Haan, A.; Jones, D.A.; Degens, H. Sex differences in contractile properties and fatigue resistance of human skeletal muscle. Exp. Physiol. 2008, 93, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Russ, D.W.; Kent-Braun, J.A. Sex differences in human skeletal muscle fatigue are eliminated under ischemic conditions. J. Appl. Physiol. 2003, 94, 2414–2422. [Google Scholar] [CrossRef] [PubMed]
- Martin, P.G.; Gandevia, S.C.; Taylor, J.L. Muscle fatigue changes cutaneous suppression of propriospinal drive to human upper limb muscles. J. Physiol. 2007, 580, 211–223. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.E.; MacDougall, J.D.; Tarnopolsky, M.A.; Sale, D.G. Gender differences in strength and muscle fiber characteristics. Eur. J. Appl. Physiol. Occup. Physiol. 1993, 66, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Keller-Ross, M.L.; Pereira, H.M.; Pruse, J.; Yoon, T.; Schlinder-Delap, B.; Nielson, K.A.; Hunter, S.K. Stressor-induced increase in muscle fatigability of young men and women is predicted by strength but not voluntary activation. J. Appl. Physiol. 2014, 116, 767–778. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, G.; Conrad, M.O.; Qiu, D.; Kamper, D.G. Contributions of voluntary activation deficits to hand weakness after stroke. Top. Stroke Rehabil. 2016, 23, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Kamper, D.G.; Fischer, H.C.; Cruz, E.G.; Rymer, W.Z. Weakness is the primary contributor to finger impairment in chronic stroke. Arch. Phys. Med. Rehabil. 2006, 87, 1262–1269. [Google Scholar] [CrossRef] [PubMed]
- Scherbakov, N.; Sandek, A.; Doehner, W. Stroke-related sarcopenia: Specific characteristics. J. Am. Med. Dir. Assoc. 2015, 16, 272–276. [Google Scholar] [CrossRef] [PubMed]
- Kent-Braun, J.A.; Fitts, R.H.; Christie, A. Skeletal muscle fatigue. Compr. Physiol. 2012, 2, 997–1044. [Google Scholar] [PubMed]
- Gandevia, S.C. Spinal and supraspinal factors in human muscle fatigue. Physiol. Rev. 2001, 81, 1725–1789. [Google Scholar] [PubMed]
- Carpentier, A.; Duchateau, J.; Hainaut, K. Motor unit behaviour and contractile changes during fatigue in the human first dorsal interosseus. J. Physiol. 2001, 534, 903–912. [Google Scholar] [CrossRef] [PubMed]
- Riley, Z.A.; Maerz, A.H.; Litsey, J.C.; Enoka, R.M. Motor unit recruitment in human biceps brachii during sustained voluntary contractions. J. Physiol. 2008, 586, 2183–2193. [Google Scholar] [CrossRef] [PubMed]
- Hunter, S.K.; Critchlow, A.; Shin, I.S.; Enoka, R.M. Men are more fatigable than strength-matched women when performing intermittent submaximal contractions. J. Appl. Physiol. 2004, 96, 2125–2132. [Google Scholar] [CrossRef] [PubMed]
- Enoka, R.M.; Duchateau, J. Translating fatigue to human performance. Med. Sci. Sports Exerc. 2016, 48, 2228–2238. [Google Scholar] [CrossRef] [PubMed]
- Schepers, V.P.; Visser-Meily, A.M.; Ketelaar, M.; Lindeman, E. Poststroke fatigue: Course and its relation to personal and stroke-related factors. Arch. Phys. Med. Rehabil. 2006, 87, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Van de Port, I.G.; Kwakkel, G.; Schepers, V.P.; Heinemans, C.T.; Lindeman, E. Is fatigue an independent factor associated with activities of daily living, instrumental activities of daily living and health-related quality of life in chronic stroke? Cerebrovasc. Dis. 2007, 23, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Van de Port, I.G.; Kwakkel, G.; Bruin, M.; Lindeman, E. Determinants of depression in chronic stroke: A prospective cohort study. Disabil. Rehabil. 2007, 29, 353–358. [Google Scholar] [CrossRef] [PubMed]
| Subject | Sex | Age (Years) | Time since Stroke (Months) | Lower Extremity Fugl–Meyer | Self-Selected Walking Speed (m/s) | Berg Balance Test |
|---|---|---|---|---|---|---|
| S2 | women | 66 | 93 | 17 | 0.28 | 38 |
| S3 | women | 62 | 270 | 30 | 1.22 | 47 |
| S5 | women | 57 | 235 | 14 | 0.40 | 46 |
| S6 | women | 76 | 51 | 30 | 1.09 | 48 |
| S9 | women | 79 | 52 | 27 | 0.66 | 49 |
| S13 | women | 62 | 191 | 12 | 0.84 | 46 |
| S16 | women | 64 | 110 | 32 | 0.80 | 54 |
| S17 | women | 80 | 94 | 21 | 0.60 | 26 |
| Average (±SD) | women | 68.25 ± 9 | 137.00 ± 84 | 22.88 ± 7.90 | 0.74 ± 0.3 | 44.25 ± 9 |
| S1 | men | 55 | 118 | 29 | 0.96 | 45 |
| S4 | men | 48 | 439 | 23 | 1.35 | 55 |
| S7 | men | 67 | 17 | 11 | 0.14 | 24 |
| S8 | men | 48 | 140 | 21 | 0.34 | 43 |
| S10 | men | 55 | 65 | 22 | 0.83 | 42 |
| S11 | men | 67 | 113 | 19 | 0.67 | 48 |
| S12 | men | 64 | 65 | 27 | 1.24 | 49 |
| S14 | men | 47 | 49 | 28 | 1.04 | 52 |
| S15 | men | 59 | 110 | 24 | 1.10 | 49 |
| S18 | men | 62 | 90 | 21 | 0.90 | 39 |
| Average (±SD) | men | 57 ± 8 | 120.6 ± 117 | 22.5 ± 5 | 0.86 ± 0.4 | 44.6 ± 8 |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kirking, M.; Berrios Barillas, R.; Nelson, P.A.; Hunter, S.K.; Hyngstrom, A. Sex Differences in Neuromuscular Fatigability of the Knee Extensors Post-Stroke. Brain Sci. 2017, 7, 8. https://doi.org/10.3390/brainsci7010008
Kirking M, Berrios Barillas R, Nelson PA, Hunter SK, Hyngstrom A. Sex Differences in Neuromuscular Fatigability of the Knee Extensors Post-Stroke. Brain Sciences. 2017; 7(1):8. https://doi.org/10.3390/brainsci7010008
Chicago/Turabian StyleKirking, Meghan, Reivian Berrios Barillas, Philip Andrew Nelson, Sandra Kay Hunter, and Allison Hyngstrom. 2017. "Sex Differences in Neuromuscular Fatigability of the Knee Extensors Post-Stroke" Brain Sciences 7, no. 1: 8. https://doi.org/10.3390/brainsci7010008
APA StyleKirking, M., Berrios Barillas, R., Nelson, P. A., Hunter, S. K., & Hyngstrom, A. (2017). Sex Differences in Neuromuscular Fatigability of the Knee Extensors Post-Stroke. Brain Sciences, 7(1), 8. https://doi.org/10.3390/brainsci7010008





