Conducting Performance-Assisted Resections in the Right Temporo-Insular Cortex: A Real-Time Neuropsychological Testing (RTNT) Protocol
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Inclusion Criteria
2.3. Exclusion Criteria
2.4. RTNT Protocol
2.5. Pre- and Post-Surgery Neuropsychological Assessment
3. Results
3.1. Patients
3.2. RTNT Results
3.3. Intra-RTNT Qualitative Observations
3.4. Neuropsychological Outcome
4. Discussion
Limitations of the Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| RTNT | Real-time neuropsychological testing |
| DES | Direct electrical stimulation |
| TOM | Theory of mind |
| L | Landmark test |
| C | Clock test |
| Ch | Chimera test |
| F | Face recognition |
| I | Emotion processing |
References
- Ojemann, G.; Ojemann, J.; Lettich, E.; Berger, M. Cortical language localization in left, dominant hemisphere. An electrical stimulation mapping investigation in 117 patients. J. Neurosurg. 1989, 71, 316–326. [Google Scholar] [CrossRef] [PubMed]
- Vilasboas, T.; Herbet, G.; Duffau, H. Challenging the Myth of Right Nondominant Hemisphere: Lessons from Corticosubcortical Stimulation Mapping in Awake Surgery and Surgical Implications. World Neurosurg. 2017, 103, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Mamadaliev, D.M.; Saito, R.; Motomura, K.; Ohka, F.; Scalia, G.; Umana, G.E.; Conti, A.; Chaurasia, B. Awake Craniotomy for Gliomas in the Non-Dominant Right Hemisphere: A Comprehensive Review. Cancers 2024, 16, 1161. [Google Scholar] [CrossRef]
- Duffau, H. Awake surgery for nonlanguage mapping. Neurosurgery 2010, 66, 523–529. [Google Scholar] [CrossRef]
- Bartolomeo, P. A parietofrontal network for spatial awareness in the right hemisphere of the human brain. Arch. Neurol. 2006, 63, 1238–1241. [Google Scholar] [CrossRef]
- Lemée, J.M.; Bernard, F.; Ter Minassian, A.; Menei, P. Right Hemisphere Cognitive Functions: From Clinical and Anatomical Bases to Brain Mapping During Awake Craniotomy. Part II: Neuropsychological Tasks and Brain Mapping. World Neurosurg. 2018, 118, 360–367. [Google Scholar] [CrossRef]
- Martín-Monzón, I.; Amores-Carrera, L.; Sabsevitz, D.; Herbet, G. Intraoperative mapping of the right hemisphere: A systematic review of protocols that evaluate cognitive and social cognitive functions. Front. Psychol. 2024, 15, 1415523. [Google Scholar] [CrossRef]
- Gecici, N.N.; Habib, A.; Niranjan, A.; Balzer, J.; Sherry, N.; Zinn, P.O. Optimizing brain mapping: Integrating real-time neuropsychological assessment in awake craniotomy. Neurosurg. Focus. Video 2025, 12, V6. [Google Scholar] [CrossRef]
- Skrap, M.; Marin, D.; Ius, T.; Fabbro, F.; Tomasino, B. Brain mapping: A novel intraoperative neuropsychological approach. J. Neurosurg. 2016, 125, 877–887. [Google Scholar] [CrossRef]
- Tomasino, B.; Guarracino, I.; Ius, T.; Maieron, M.; Skrap, M. Real-Time Neuropsychological Testing Protocol for Left Temporal Brain Tumor Surgery: A Technical Note and Case Report. Front. Hum. Neurosci. 2021, 15, 760569. [Google Scholar] [CrossRef] [PubMed]
- Tomasino, B.; Guarracino, I.; Ius, T.; Budai, R.; Skrap, M. Real-Time Neuropsychological Testing of Sensorimotor Cognition During Awake Surgery in Precentral and Postsomatosensory Areas. World Neurosurg. 2022, 164, e599–e610. [Google Scholar] [CrossRef]
- Tomasino, B.; Guarracino, I.; Ius, T.; Skrap, M. Continuous Real-Time Neuropsychological Testing during Resection Phase in Left and Right Prefrontal Brain Tumors. Curr. Oncol. 2023, 30, 2007–2020. [Google Scholar] [CrossRef]
- Guarracino, I.; Ius, T.; Pauletto, G.; Maieron, M.; Skrap, M.; Tomasino, B. Junior-Real Time neuropsychological testing (j-RTNT) for a young patient undergoing awake craniotomy. Brain Cogn. 2020, 140, 105535. [Google Scholar] [CrossRef]
- Committeri, G.; Pitzalis, S.; Galati, G.; Patria, F.; Pelle, G.; Sabatini, U.; Castriota-Scanderbeg, A.; Piccardi, L.; Guariglia, C.; Pizzamiglio, L. Neural bases of personal and extrapersonal neglect in humans. Brain J. Neurol. 2007, 130 Pt 2, 431–441. [Google Scholar] [CrossRef]
- Rengachary, J.; He, B.J.; Shulman, G.L.; Corbetta, M. A behavioral analysis of spatial neglect and its recovery after stroke. Front. Hum. Neurosci. 2011, 5, 29. [Google Scholar] [CrossRef]
- Wang, X.; Song, Y.; Zhen, Z.; Liu, J. Functional integration of the posterior superior temporal sulcus correlates with facial expression recognition. Brain Mapp. 2016, 37, 1930–1940. [Google Scholar] [CrossRef] [PubMed]
- Genova, H.M.; Rajagopalan, V.; Chiaravalloti, N.; Binder, A.; Deluca, J.; Lengenfelder, J. Facial affect recognition linked to damage in specific white matter tracts in traumatic brain injury. Soc. Neurosci. 2015, 10, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Philippi, C.L.; Mehta, S.; Grabowski, T.; Adolphs, R.; Rudrauf, D. Damage to association fiber tracts impairs recognition of the facial expression of emotion. J. Neurosci. 2009, 29, 15089–15099. [Google Scholar] [CrossRef]
- Lang, P.J.; Bradley, M.M.; Cuthbert, B.N. International affective picture system (IAPS): Technical manual and affective ratings. NIMH Cent. Study Emot. Atten. 1997, 1, 3. [Google Scholar]
- Humphreys, G.W.; Riddoch, J.M. Birmingham Object Recognition Battery; Psychology Press: East Sussex, UK, 1993. [Google Scholar]
- Bizzozero, I.; Ferrari, F.; Pozzoli, S.; Saetti, M.C.; Spinnler, H. Who is who: Italian norms for visual recognition and identification of celebrities. Neurol. Sci. 2005, 26, 95–107. [Google Scholar] [CrossRef]
- Prior, M.; Marchi, S.; Sartori, G. Social Cognition and Behavior. A Tool for Assessment Cognizione Sociale e Comportamento. Uno Strumento Per la Misurazione; Upsel Domenighini Editore: Padova, Italy, 2003. [Google Scholar]
- Antonietti, A.; Bartolomeo, P.; Colombi, A.; Incorpora, C.; Oliveri, S. Batteria Immaginazione e Percezione (BIP) per la valutazione della cognizione visuo-spaziale-Italian Translation and Adaptation of the Corresponding Battery Devised by P. Bartolomeo, A. C. Bachoud-Levi and S. Chokron at the INSERM, Paris; ISU-Università Cattolica del Sacro Cuore: Milan, Italy, 2008. [Google Scholar]
- Capitani, E.; Neppi-Mòdona, M.; Bisiach, E. Verbal-response and manual-response versions of the Milner Landmark task: Normative data. Cortex 2000, 36, 593–600. [Google Scholar] [CrossRef] [PubMed]
- Basso, A.; Capitani, E.; Laiacona, M. Raven’s coloured progressive matrices: Normative values on 305 adult normal controls. Funct. Neurol. 1987, 2, 189–194. [Google Scholar] [PubMed]
- Giovagnoli, A.R.; Del Pesce, M.; Mascheroni, S.; Simoncelli, M.; Laiacona, M.; Capitani, E. Trail making test: Normative values from 287 normal adult controls. Ital. J. Neurol. Sci. 1996, 17, 305–309. [Google Scholar] [CrossRef] [PubMed]
- Monaco, M.; Costa, A.; Caltagirone, C.; Carlesimo, G.A. Erratum to: Forward and backward span for verbal and visuo-spatial data: Standardization and normative data from an Italian adult population. Neurol. Sci. 2015, 36, 345–347. [Google Scholar] [CrossRef]
- Wechsler, D. Wechsler Adult Intelligence Scale—Third Edition (WAIS-III); Psychological Corporation: San Antonio, TX, USA, 1997. [Google Scholar]
- Caffarra, P.; Vezzadini, G.; Dieci, F.; Zonato, F.; Venneri, A. Rey-Osterrieth complex figure: Normative values in an Italian population sample. Neurol. Sci. 2002, 22, 443–447. [Google Scholar] [CrossRef]
- Bizzozero, I.; Lucchelli, F.; Pozzoli, S.; Saetti, M.C.; Spinnler, H. “What do you know about Ho Chi Minh?” Italian norms of proper name comprehension. Neurol. Sci. 2007, 28, 16–30. [Google Scholar] [CrossRef]
- Spinnler, M.; Tognoni, G. Standardizzazione e taratura italiana di test neuropsicologici. Ital. J. Neurol. Sci. 1987, 6, 1–120. [Google Scholar]
- Wilson Barbara, A.; Cockburn, J.; Halligan, P.W.; Spinazzola, L.; Pagliari, C.; Beschin, N. BIT: Behavioural Inattention Test. Giunti O.S. Organizzazioni Speciali: Firenze, Italy, 2010. [Google Scholar]
- Ratcliff, G. Spatial thought, mental rotation and the right cerebral hemisphere. Neuropsychologia 1979, 17, 49–54. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef]
- Tomaselli, A.; Luca, A.; Ferini, G.; Umana, G.E.; Chaurasia, B.; Scalia, G. Cognitive Profiles and Determinants of Eligibility for Awake Surgery in Non-Dominant Hemisphere Gliomas: A Narrative Review. Brain Behav. 2025, 15, e70604. [Google Scholar] [CrossRef]
- Raffa, G.; Quattropani, M.C.; Marzano, G.; Curcio, A.; Rizzo, V.; Sebestyén, G.; Germanò, A. Mapping and preserving the visuospatial network by repetitive nTMS and DTI tractography in patients with right parietal lobe tumors. Front. Oncol. 2021, 11, 677172. [Google Scholar] [CrossRef]
- Noll, K.R.; Ziu, M.; Weinberg, J.S.; Wefel, J.S. Neurocognitive functioning in patients with glioma of the left and right temporal lobes. J. Neuro-Oncol. 2016, 128, 323–331. [Google Scholar] [CrossRef]
- Young, J.S.; Morshed, R.A.; Andrews, J.P.; Cha, S.; Berger, M.S. Prosopagnosia following nonlanguage dominant inferior temporal lobe low-grade glioma resection in which the inferior longitudinal fasciculus was disrupted preoperatively: Illustrative case. J. Neurosurg. Case Lessons 2021, 2, CASE21277. [Google Scholar] [CrossRef] [PubMed]
- Cargnelutti, E.; Ius, T.; Skrap, M.; Tomasino, B. What do we know about pre- and postoperative plasticity in patients with glioma? A review of neuroimaging and intraoperative mapping studies. Neuroimage Clin. 2020, 28, 102435. [Google Scholar] [CrossRef] [PubMed]
- Doherty, C.; Nowacki, A.S.; Pat McAndrews, M.; McDonald, C.R.; Reyes, A.; Kim, M.S.; Hamberger, M.; Najm, I.; Bingaman, W.; Jehi, L.; et al. Predicting mood decline following temporal lobe epilepsy surgery in adults. Epilepsia 2021, 62, 450–459. [Google Scholar] [CrossRef] [PubMed]
- Van Sweden, B. Sleep and the temporal lobe. Acta Neurol. Belg. 1996, 96, 19–30. [Google Scholar]
- Ayoub, L.J.; Barnett, A.; Leboucher, A.; Golosky, M.; McAndrews, M.P.; Seminowicz, D.A.; Moayedi, M. The medial temporal lobe in nociception: A meta-analytic and functional connectivity study. Pain 2019, 160, 1245–1260. [Google Scholar] [CrossRef]
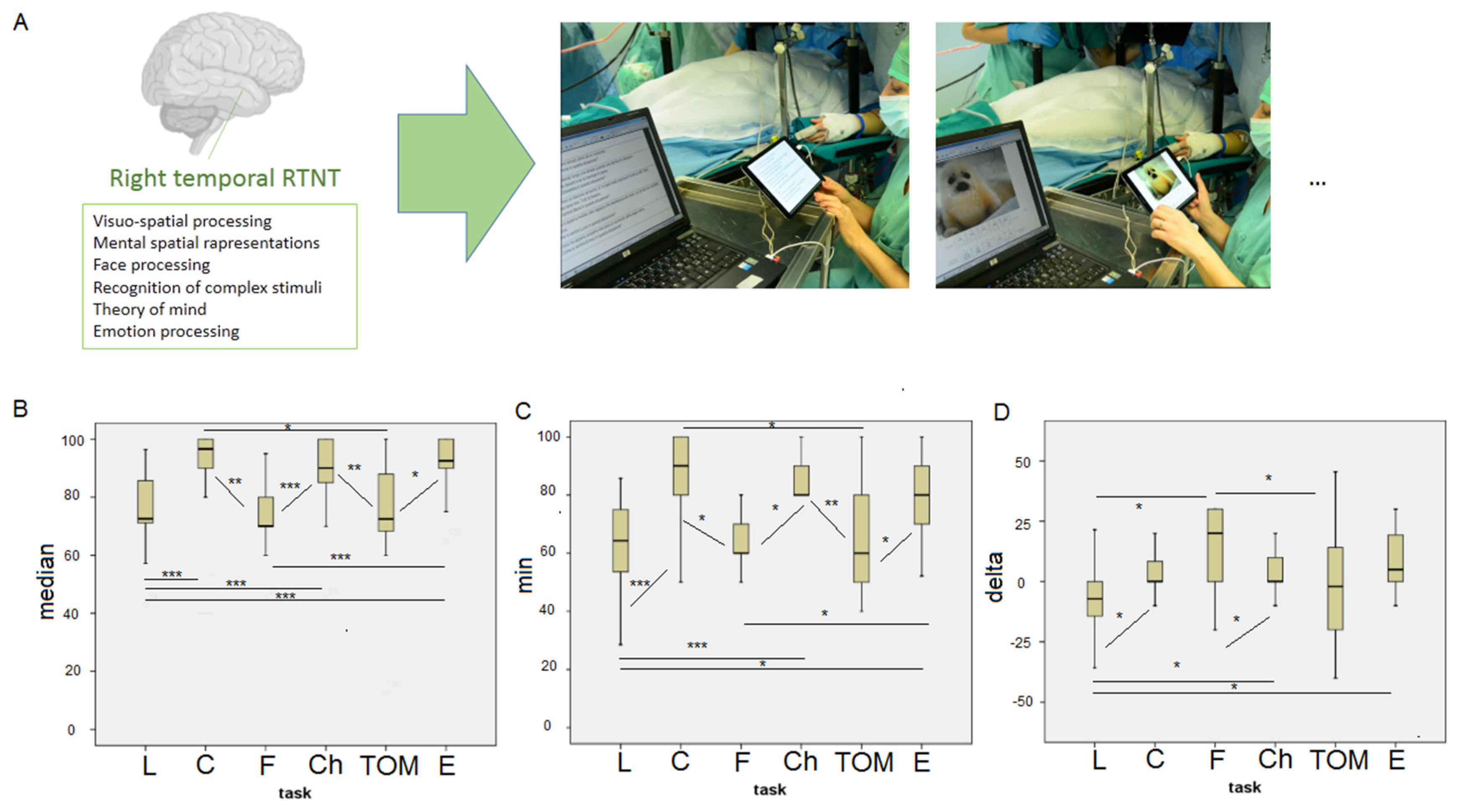
| RTNT Test-Run | Test | Assessed Ability |
| International Affective Picture System (E) [19] | The patient is presented with positive, negative, or neutral images. The patient using the SAM Manikin provides a score on the Likert scale for pleasure and arousal. | Explicit measures of emotion-related processing |
| Real object or chimera (Ch) [20] | Black and white images are presented corresponding to real figures (living and non-living) or chimeras (figures formed by the fusion of 2 living or 2 non-living). The patient says whether the image is real or a chimera. | Visual–perceptual and semantic skills |
| Face Recognition (guess of familiarity) and identification (F) [21] | Black and white photos of famous and unknown people are presented. The subject says whether the person is famous or not and if they are, the patient says their name. | Semantic memory (Familiarity Recognition Units, Personal Identity Nodes) |
| Theory of Mind (TOM) [22] | The subject is presented with very short scenes with a protagonist. The subject says what emotion the protagonist will feel in the specific situation described. | Ability to put oneself in the shoes of others and understand their mental states |
| Clock test: Imaginative (C) subtest [23] | The patient is orally told a time, e.g., 14:30, mentally pictures the clock, and says whether the hands are positioned both on the right half of the dial, both on the left half, or are one on the right and one on the left. | Spatial cognition in an imaginative dimension |
| Landmark test (L) [24] | Segments are presented. Each segment is made up of a red and a black portion. The subject says whether the longer portion is the red or the black one. | Highlights the presence of spatial neglect |
| Title 1 | Title 2 |
|---|---|
| Number of patients | 24 |
| Sex | |
| Male | 16 |
| Female | 8 |
| Age (years) | |
| Median (years and range) | 44 (66–23) |
| Tumor side | |
| Right | 24 |
| Handedness | |
| Right | 24/24 |
| Education | |
| Median (years and range) | 13 (20–8) |
| Molecular class ^ | |
| High-grade glioma | 11 |
| Oligodendrogliomas, IDH1/2 mutant 1p/19q codeleted | 4 |
| Astrocytomas, IDH1/2 mutant | 6 |
| Astrocytomas, IDH1/2 wild type | 1 |
| Cavernoma | 2 |
| Preoperative tumoral volume (cm3) | |
| Median (range) | 42 (15–118) |
| EOR | |
| Median (range) | 99 (34–100) |
| Intraoperative protocol | |
| Awake surgery | 24/24 |
| 95% CI Lower | 95% CI Upper | Median | Min (Baseline: 1st RTNT Run) | Max | |
|---|---|---|---|---|---|
| Median RTNT scores (χ2 (5) = 41.02, p < 0.001) | |||||
| L | 63.56 | 80.57 | 71.42 | 42.85 (78.57) | 89.28 |
| C | 92.65 | 100 | 100 | 85 (93.3) | 100 |
| F | 63.69 | 79.94 | 70 | 45 (70) | 90 |
| Ch | 87.91 | 98.44 | 95 | 80 (90) | 100 |
| TOM | 68.7 | 87.82 | 75 | 60 (80) | 100 |
| E | 88.59 | 98.81 | 95 | 75.71 (90) | 100 |
| Minimum RTNT score (χ2 (5) = 31.23, p < 0.001) | |||||
| L | 42.24 | 67.73 | 57.14 | 28.57 | 78.57 |
| C | 85.94 | 97.68 | 90 | 80 | 100 |
| F | 49.86 | 69.53 | 60 | 30 | 80 |
| Ch | 79.1 | 88.16 | 80 | 80 | 100 |
| TOM | 54.62 | 78.1 | 60 | 40 | 100 |
| E | 72.77 | 93.07 | 80 | 52.14 | 100 |
| Delta: last RTNT run—first RTNT run (χ2 (5) = 15.71, p < 0.01) | |||||
| L | −19.17 | 0.99 | −7.1 | −35.72 | 14.29 |
| C | −3.34 | 8.80 | 0 | −10 | 20 |
| F | −1.85 | 23.06 | 20 | −20 | 30 |
| Ch | −1.61 | 7.07 | 0 | −10 | 10 |
| TOM | −25.82 | 3.6 | −20 | −40 | 20 |
| E | −0.26 | 16.36 | 0 | −10 | 30 |
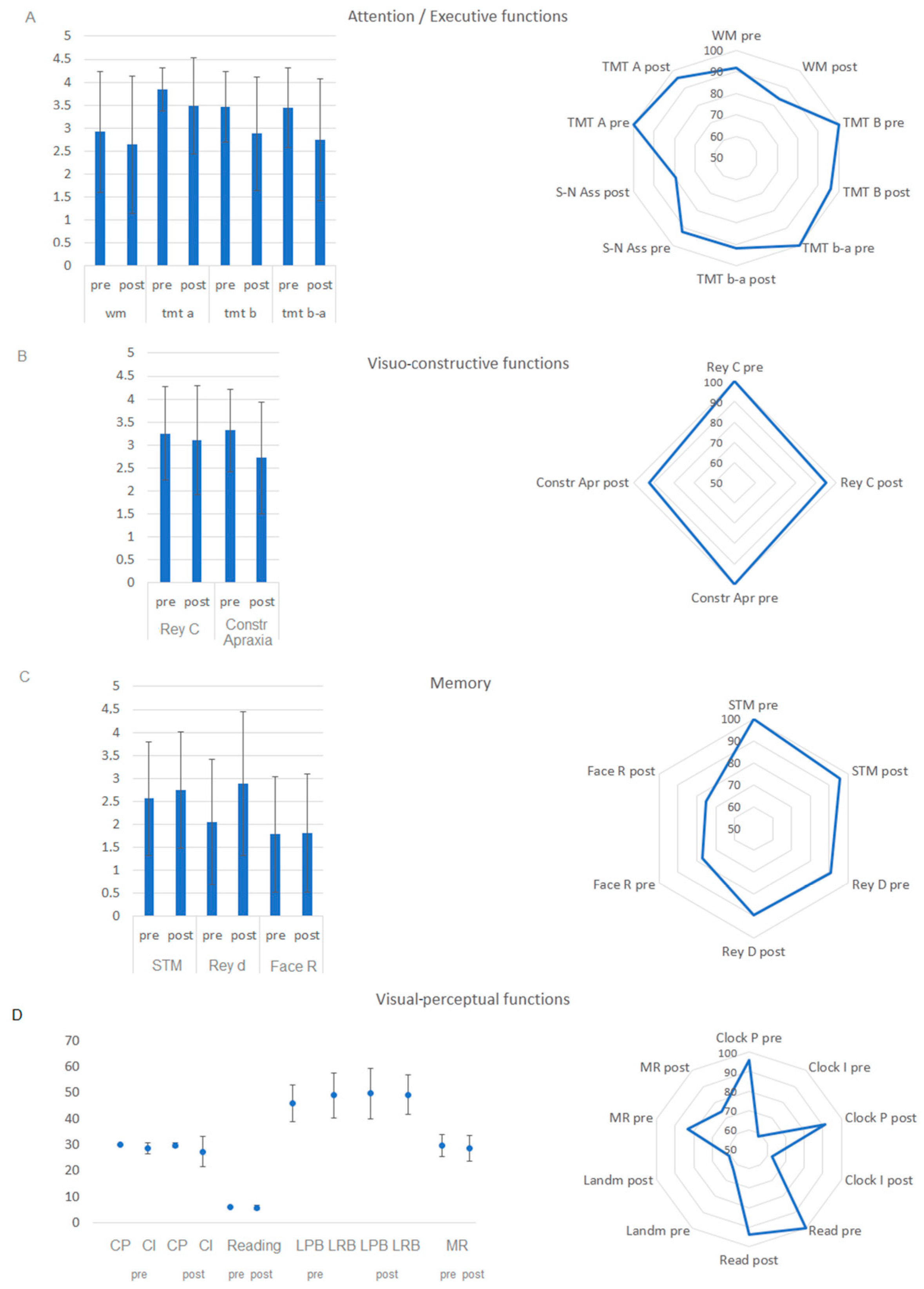
| Test | Level of Performance | Number of Within the Normal Range Performances |
|---|---|---|
| Short-term memory | Z = −691 p = 0.490 | Z = −1 p = 0.317 |
| Working memory | Z = −1.312 p = 0.190 | Z = −1 p = 0.317 |
| Rey Copy | Z = 0.00 p = 1 | Z = −1 p = 0.317 |
| Rey Delayed Recall | Z = −1.448 p = 0.148 | Z = 0 p = 1000 |
| Clock Perceptive | Z = −0.816 p = 0.414 | Z = −0.577 p = 0.564 |
| Clock Imagery | Z = −0.834 p = 0.404 | Z = −0.302 p = 0.763 |
| Constructional praxis | Z = −2.712 p = 0.007 | Z = −1.414 p = 0.157 |
| Reading | Z = −1.00 p = 0.317 | Z = −1.00 p = 0.317 |
| Landmark Perceptual Bias | Z = −0.426 p = 0.670 | Z = −1.00 p = 0.317 |
| Landmark Response Bias | Z = −0.877 p= 0.381 | |
| Digit symbol association | Z = −2.613 p = 0.009 | Z = −1.342 p = 0.180 |
| Trail Making Test A | Z = −1.461 p = 0.144 | Z = −1 p = 0.317 |
| Trail Making Test B | Z = −1.586 p = 0.113 | Z = −1 p = 0.317 |
| Trail Making Test-B-A | Z = −1.5361 p = 0.124 | Z = −1.414 p = 0.157 |
| Face recognition | Z = −1.612 p = 0.107 | Z = −1 p = 0.317 |
| Little Man | Z = −1.944 p = 0.052 | Z = −1.732 p = 0.083 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomasino, B.; Guarracino, I.; Ius, T.; Skrap, M. Conducting Performance-Assisted Resections in the Right Temporo-Insular Cortex: A Real-Time Neuropsychological Testing (RTNT) Protocol. Brain Sci. 2025, 15, 949. https://doi.org/10.3390/brainsci15090949
Tomasino B, Guarracino I, Ius T, Skrap M. Conducting Performance-Assisted Resections in the Right Temporo-Insular Cortex: A Real-Time Neuropsychological Testing (RTNT) Protocol. Brain Sciences. 2025; 15(9):949. https://doi.org/10.3390/brainsci15090949
Chicago/Turabian StyleTomasino, Barbara, Ilaria Guarracino, Tamara Ius, and Miran Skrap. 2025. "Conducting Performance-Assisted Resections in the Right Temporo-Insular Cortex: A Real-Time Neuropsychological Testing (RTNT) Protocol" Brain Sciences 15, no. 9: 949. https://doi.org/10.3390/brainsci15090949
APA StyleTomasino, B., Guarracino, I., Ius, T., & Skrap, M. (2025). Conducting Performance-Assisted Resections in the Right Temporo-Insular Cortex: A Real-Time Neuropsychological Testing (RTNT) Protocol. Brain Sciences, 15(9), 949. https://doi.org/10.3390/brainsci15090949






