Ictal MEG-EEG Study to Localize the Onset of Generalized Seizures: To See Beyond What Meets the Eye
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. MEG/EEG Data Acquisition Analysis
2.3. MEG/EEG Data Analysis
2.4. Statistical Analysis
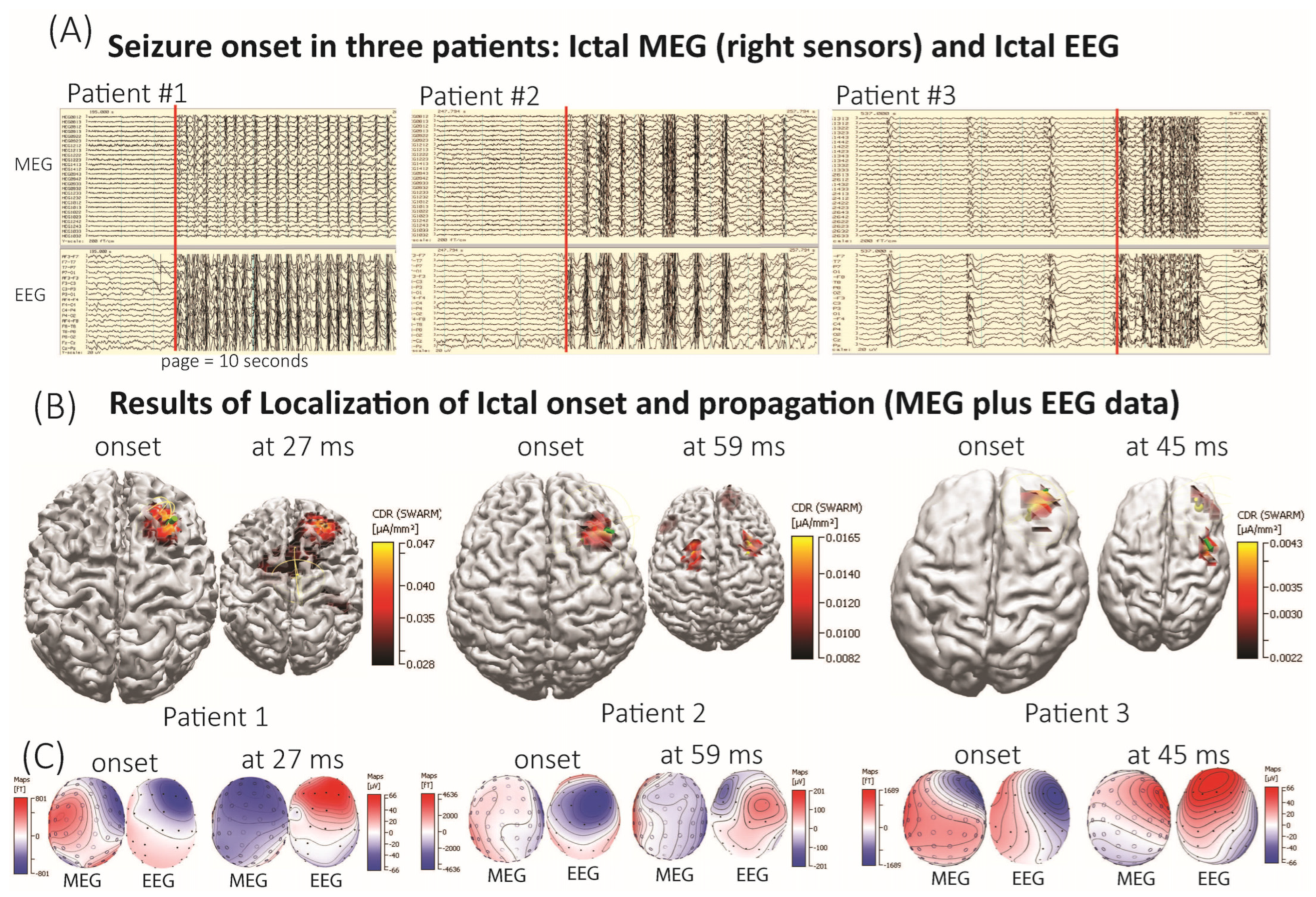
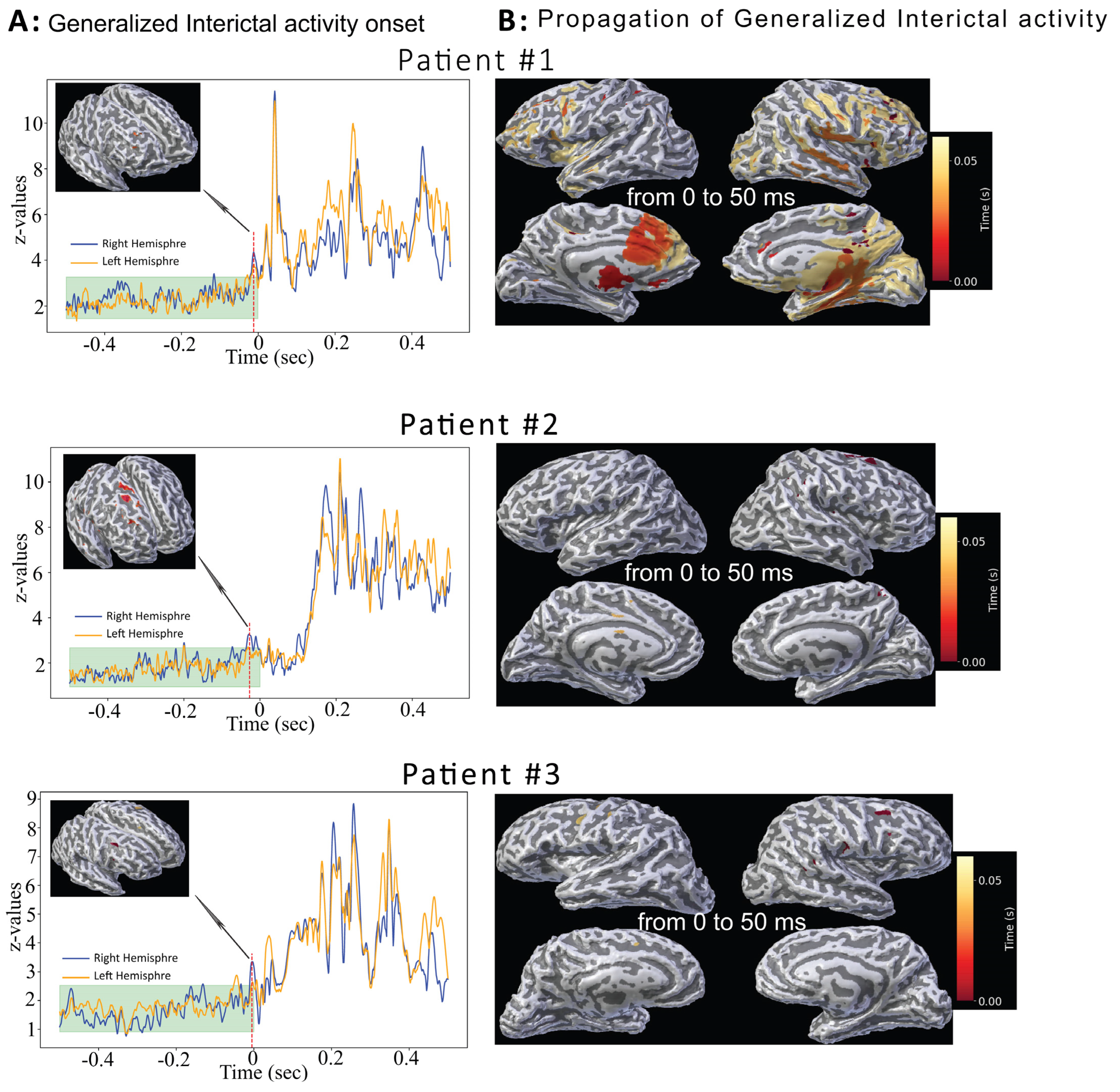
3. Results
3.1. Seizure Onset and Propagation Defined by Z-Values in Normalized Epileptogenic Data to Normal Sleep Data
3.2. Interictal Generalized Discharges
3.3. Comparison Between Ictal and Interictal Source Localization Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jallon, P.; Latour, P. Epidemiology of idiopathic generalized epilepsies. Epilepsia 2005, 46 (Suppl. 9), 10–14. [Google Scholar] [CrossRef]
- Tomson, T.; Nashef, L.; Ryvlin, P. Sudden unexpected death in epilepsy: Current knowledge and future directions. Lancet Neurol. 2008, 7, 1021–1031. [Google Scholar] [CrossRef]
- Harden, C.; Tomson, T.; Gloss, D.; Buchhalter, J.; Cross, J.H.; Donner, E.; French, J.A.; Gil-Nagel, A.; Hesdorffer, D.C.; Smithson, W.H.; et al. Practice guideline summary: Sudden unexpected death in epilepsy incidence rates and risk factors: Report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Neurology 2017, 88, 1674–1680. [Google Scholar] [CrossRef]
- Asadi-Pooya, A.A.; Nikseresht, A.; Yaghoubi, E.; Nei, M. Physical injuries in patients with epilepsy and their associated risk factors. Seizure 2012, 21, 165–168. [Google Scholar] [CrossRef]
- Posner, E. Absence seizures in children. BMJ Clin. Evid. 2008, 2008, 317. [Google Scholar]
- Engel, J., Jr.; International League Against, E. A proposed diagnostic scheme for people with epileptic seizures and with epilepsy: Report of the ILAE Task Force on Classification and Terminology. Epilepsia 2001, 42, 796–803. [Google Scholar] [CrossRef] [PubMed]
- Meeren, H.; van Luijtelaar, G.; Lopes da Silva, F.; Coenen, A. Evolving concepts on the pathophysiology of absence seizures: The cortical focus theory. Arch. Neurol. 2005, 62, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Buzsaki, G. The thalamic clock: Emergent network properties. Neuroscience 1991, 41, 351–364. [Google Scholar] [CrossRef] [PubMed]
- Gastaut, H. The Physiopathogenesis of the Epilepsies; Charles C Thomas Publisher: Springfield, IL, USA, 1969; Volume xxi, 316p. [Google Scholar]
- Luders, H.; Lesser, R.P.; Dinner, D.S.; Morris, H.H., 3rd. Generalized epilepsies: A review. Cleve. Clin. Q. 1984, 51, 205–226. [Google Scholar] [CrossRef]
- Niedermeyer, E. The Generalized Epilepsies: A Clinical Electroencephalographic Study; Charles C Thomas Publisher: Springfield, IL, USA, 1972; Volume xii, 247p. [Google Scholar]
- Penfield, W.; Jasper, H.H. Epilepsy and the Functional Anatomy of the Human Brain, 1st ed.; Little: Boston, MA, USA, 1954; 896p. [Google Scholar]
- Vlachou, M.; Ryvlin, P.; Armand Larsen, S.; Beniczky, S. Focal electroclinical features in generalized tonic-clonic seizures: Decision flowchart for a diagnostic challenge. Epilepsia 2024, 65, 725–738. [Google Scholar] [CrossRef]
- Meeren, H.K.; Pijn, J.P.; Van Luijtelaar, E.L.; Coenen, A.M.; Lopes da Silva, F.H. Cortical focus drives widespread corticothalamic networks during spontaneous absence seizures in rats. J. Neurosci. 2002, 22, 1480–1495. [Google Scholar] [CrossRef] [PubMed]
- Ferrie, C.D. Idiopathic generalized epilepsies imitating focal epilepsies. Epilepsia 2005, 46 (Suppl. 9), 91–95. [Google Scholar] [CrossRef] [PubMed]
- Gloor, P. Generalized cortico-reticular epilepsies. Some considerations on the pathophysiology of generalized bilaterally synchronous spike and wave discharge. Epilepsia 1968, 9, 249–263. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.R.; Cohen, J.; Mayman, C.I.; Scholl, M.L.; Hendrix, D.E. Relationship of the magnetoencephalogram to abnormal activity in the electroencephalogram. J. Neurol. 1977, 217, 79–93. [Google Scholar] [CrossRef]
- Aung, T.; Tenney, J.R.; Bagic, A.I. Contributions of Magnetoencephalography to Understanding Mechanisms of Generalized Epilepsies: Blurring the Boundary Between Focal and Generalized Epilepsies? Front. Neurol. 2022, 13, 831546. [Google Scholar] [CrossRef]
- Zhou, D.J.; Gumenyuk, V.; Taraschenko, O.; Grobelny, B.T.; Stufflebeam, S.M.; Peled, N. Visualization of the Spatiotemporal Propagation of Interictal Spikes in Temporal Lobe Epilepsy: A MEG Pilot Study. Brain Topogr. 2024, 37, 116–125. [Google Scholar] [CrossRef]
- Tamilia, E.; AlHilani, M.; Tanaka, N.; Tsuboyama, M.; Peters, J.M.; Grant, P.E.; Madsen, J.R.; Stufflebeam, S.M.; Pearl, P.L.; Papadelis, C. Assessing the localization accuracy and clinical utility of electric and magnetic source imaging in children with epilepsy. Clin. Neurophysiol. 2019, 130, 491–504. [Google Scholar] [CrossRef]
- Wagner, M.; Fuchs, M.; Kastner, J. Evaluation of sLORETA in the presence of noise and multiple sources. Brain Topogr. 2004, 16, 277–280. [Google Scholar] [CrossRef]
- Laohathai, C.; Ebersole, J.S.; Mosher, J.C.; Bagic, A.I.; Sumida, A.; Von Allmen, G.; Funke, M.E. Practical Fundamentals of Clinical MEG Interpretation in Epilepsy. Front. Neurol. 2021, 12, 722986. [Google Scholar] [CrossRef]
- Corona, L.; Rijal, S.; Tanritanir, O.; Shahdadian, S.; Keator, C.G.; Tran, L.; Malik, S.I.; Bosemani, M.; Hansen, D.; Shahani, D.; et al. Electromagnetic Source Imaging in Presurgical Evaluation of Children with Drug-Resistant Epilepsy. J. Vis. Exp. 2024, 211, 1–38. [Google Scholar] [CrossRef]
- Ebersole, J.S.; Ebersole, S.M. Combining MEG and EEG source modeling in epilepsy evaluations. J. Clin. Neurophysiol. 2010, 27, 360–371. [Google Scholar] [CrossRef] [PubMed]
- Taulu, S.; Simola, J. Spatiotemporal signal space separation method for rejecting nearby interference in MEG measurements. Phys. Med. Biol. 2006, 51, 1759–1768. [Google Scholar] [CrossRef] [PubMed]
- Felsenstein, O.; Peled, N.; Hahn, E.; Rockhill, A.P.; Frank, D.; Libster, A.M.; Nossenson, Y.; Folsom, L.; Gholipour, T.; Macadams, K.; et al. Multi-modal neuroimaging analysis and visualization tool (MMVT). arXiv 2019, arXiv:1912.10079. [Google Scholar]
- Plummer, C.; Wagner, M.; Fuchs, M.; Vogrin, S.; Litewka, L.; Farish, S.; Bailey, C.; Harvey, A.S.; Cook, M.J. Clinical utility of distributed source modelling of interictal scalp EEG in focal epilepsy. Clin. Neurophysiol. 2010, 121, 1726–1739. [Google Scholar] [CrossRef]
- Tomlinson, S.B.; Bermudez, C.; Conley, C.; Brown, M.W.; Porter, B.E.; Marsh, E.D. Spatiotemporal Mapping of Interictal Spike Propagation: A Novel Methodology Applied to Pediatric Intracranial EEG Recordings. Front. Neurol. 2016, 7, 229. [Google Scholar] [CrossRef]
- Ebersole, J.S.; Hawes-Ebersole, S. Clinical application of dipole models in the localization of epileptiform activity. J. Clin. Neurophysiol. 2007, 24, 120–129. [Google Scholar] [CrossRef]
- Barkley, G.L.; Baumgartner, C. MEG and EEG in epilepsy. J. Clin. Neurophysiol. 2003, 20, 163–178. [Google Scholar] [CrossRef]
- Goldenholz, D.M.; Ahlfors, S.P.; Hamalainen, M.S.; Sharon, D.; Ishitobi, M.; Vaina, L.M.; Stufflebeam, S.M. Mapping the signal-to-noise-ratios of cortical sources in magnetoencephalography and electroencephalography. Hum. Brain Mapp. 2009, 30, 1077–1086. [Google Scholar] [CrossRef]
- Pitetzis, D.; Frantzidis, C.; Psoma, E.; Ketseridou, S.N.; Deretzi, G.; Kalogera-Fountzila, A.; Bamidis, P.D.; Spilioti, M. The Pre-Interictal Network State in Idiopathic Generalized Epilepsies. Brain Sci. 2023, 13, 1671. [Google Scholar] [CrossRef]
- Tenney, J.R.; Glauser, T.A. The current state of absence epilepsy: Can we have your attention? Epilepsy Curr. 2013, 13, 135–140. [Google Scholar] [CrossRef]
- Mitsuhashi, T.; Sonoda, M.; Iwaki, H.; Sakakura, K.; Asano, E. Detection of absence seizures using a glasses-type eye tracker. Clin. Neurophysiol. 2021, 132, 720–722. [Google Scholar] [CrossRef]


| Pt | Age (Years)/Sex | Seizure Types | Medications | Imaging Results | Surface EEG Results | Clinical MEG Results |
| 1 | 16/F | Type 1: Absence Type 2: GTC | Diamox, Clobazam, Diazepam, Lorazepam | MRI: Normal PET: Decreased signal in the right frontal and left temporal | Occasional generalized spike-wave discharges with right frontal onset | Right frontal for ictal and interictal discharges |
| 2 | 13/M | GTC | Depakote, Lamotrigine and Clobazam | MRI: Stable pineal region cyst PET: Normal | Generalized spike-and-wave discharges with slight right frontal predominance | Right frontal for ictal and interictal discharges |
| 3 | 33/M | Bilateral clonic | Divalproex, Felbamate, Perampanel, Rufinamide | MRI: Normal PET: Left parietotemporal hypermetabolism | Generalized seizure pattern with right predominance | Right frontal for ictal Right fronto-centro-parietal for interictal discharges |
| 4 | 13/F | Type 1: Absence Type 2: GTC | Keppra, Lamotringine | MRI: Lesion left temporal white matter PET: Hypometabolism R fronto-parietal | Absence seizure recorded maximum frontocentral | Defused bilateral frontal |
| 5 | 12/M | GTC | Keppra | MRI: Normal PET: Normal | Generalized interictal activity with no lateralizing features | Right frontal |
| 6 | 14/M | GTC | Depakote, Lamotringine | MRI: Normal PET: N/A | Generalized seizures and interictal activity with no lateralizing features | No cluster found |
| 7 | 28/M | Bilateral convulsive seizures | Vimpat, Lamictal XR, Xcopri, Nayzilam | MRI: Normal PET: Decrease signal in left mesial temporal lobe | Focal and bilateral tonic clonic possible left frontal | Right frontal |
| Pt # | CDR: Dipole Moving and SWARM | Brain Structure (Talairach Atlas) |
| 1 | Ictal—Right frontal Interictals—Right frontal | R-Superior Frontal gyrus |
| 2 | Ictal—Right frontal Interictals—Right frontal | R-Middle Frontal gyrus |
| 3 | Ictal—Right frontal Interictals—Right frontal | R-Middle Frontal gyrus |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gumenyuk, V.; Korzyukov, O.; Peled, N.; Landazuri, P.; Taraschenko, O.; Parker, S.M.; Frank, D.; Pavuluri, S. Ictal MEG-EEG Study to Localize the Onset of Generalized Seizures: To See Beyond What Meets the Eye. Brain Sci. 2025, 15, 938. https://doi.org/10.3390/brainsci15090938
Gumenyuk V, Korzyukov O, Peled N, Landazuri P, Taraschenko O, Parker SM, Frank D, Pavuluri S. Ictal MEG-EEG Study to Localize the Onset of Generalized Seizures: To See Beyond What Meets the Eye. Brain Sciences. 2025; 15(9):938. https://doi.org/10.3390/brainsci15090938
Chicago/Turabian StyleGumenyuk, Valentina, Oleg Korzyukov, Noam Peled, Patrick Landazuri, Olga Taraschenko, Sheridan M. Parker, Darya Frank, and Spriha Pavuluri. 2025. "Ictal MEG-EEG Study to Localize the Onset of Generalized Seizures: To See Beyond What Meets the Eye" Brain Sciences 15, no. 9: 938. https://doi.org/10.3390/brainsci15090938
APA StyleGumenyuk, V., Korzyukov, O., Peled, N., Landazuri, P., Taraschenko, O., Parker, S. M., Frank, D., & Pavuluri, S. (2025). Ictal MEG-EEG Study to Localize the Onset of Generalized Seizures: To See Beyond What Meets the Eye. Brain Sciences, 15(9), 938. https://doi.org/10.3390/brainsci15090938





