Resident Training in Minimally Invasive Spine Surgery: A Scoping Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Search Strategy
2.2. Inclusion Criteria and Screening Process
2.3. Data Extraction and Synthesis
3. Results
3.1. Study Selection
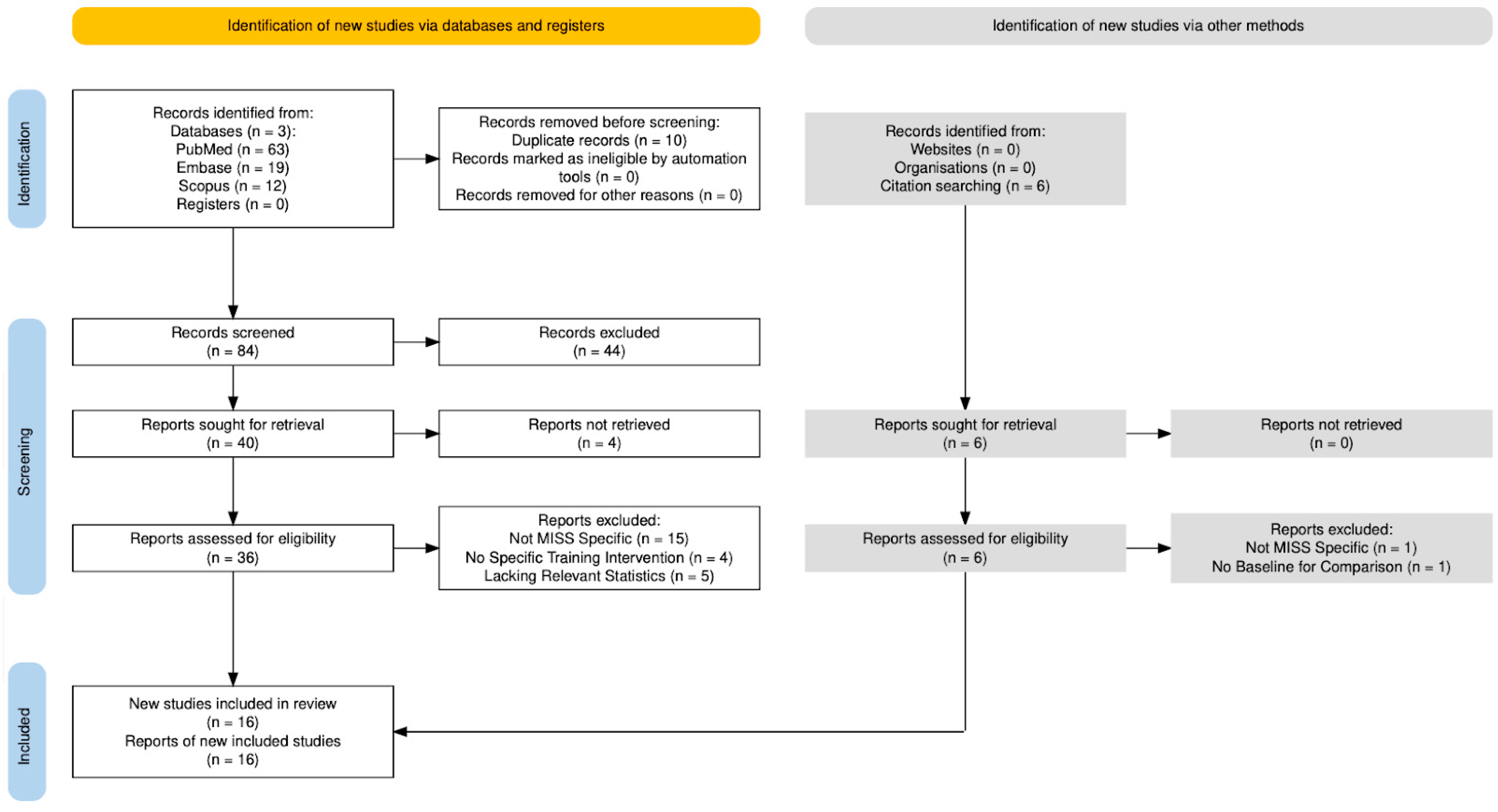
3.2. Study Trends and Key Findings
3.3. Breakdown by Training Modality
3.3.1. Virtual Simulation
3.3.2. Physical Models
3.3.3. Hybrid (Didactic + Simulation)
3.3.4. Mentored Training
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MIS | Minimally Invasive Surgery |
| MISS | Minimally Invasive Spine Surgery |
| TLIF | Transforaminal Lumbar Interbody Fusion |
| LLIF | Lateral Lumbar Interbody Fusion |
| ULBD | Unilateral Laminotomy for Bilateral Decompression |
| PCDF | Posterior Cervical Decompression and Fusion |
| AR | Augmented Reality |
| VR | Virtual Reality |
References
- Soomro, N.A.; Hashimoto, D.A.; Porteous, A.J.; Ridley, C.J.A.; Marsh, W.J.; Ditto, R.; Roy, S. Systematic Review of Learning Curves in Robot-Assisted Surgery. BJS Open 2020, 4, 27–44. [Google Scholar] [CrossRef]
- Müller, P.C.; Kuemmerli, C.; Cizmic, A.; Sinz, S.; Probst, P.; de Santibanes, M.; Shrikhande, S.V.; Tschuor, C.; Loos, M.; Mehrabi, A.; et al. Learning Curves in Open, Laparoscopic, and Robotic Pancreatic Surgery: A Systematic Review and Proposal of a Standardization. Ann. Surg. Open 2022, 3, e111. [Google Scholar] [CrossRef]
- Sharif, S.; Afsar, A. Learning Curve and Minimally Invasive Spine Surgery. World Neurosurg. 2018, 119, 472–478. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Lee, S.B.; Seok, S.O.; Jo, B.W.; Ha, J.W. Perioperative Surgical Complications and Learning Curve Associated with Minimally Invasive Transforaminal Lumbar Interbody Fusion: A Single-Institute Experience. Clin. Orthop. Surg. 2015, 7, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Sclafani, J.A.; Kim, C.W. Complications Associated with the Initial Learning Curve of Minimally Invasive Spine Surgery: A Systematic Review. Clin. Orthop. Relat. Res. 2014, 472, 1711–1717. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Ha, J.W. Comparison of One-Level Posterior Lumbar Interbody Fusion Performed with a Minimally Invasive Approach or a Traditional Open Approach. Available online: https://oce.ovid.com/article/00007632-200703010-00007/PDF (accessed on 25 February 2025).
- Wang, B.; Lü, G.; Patel, A.A.; Ren, P.; Cheng, I. An Evaluation of the Learning Curve for a Complex Surgical Technique: The Full Endoscopic Interlaminar Approach for Lumbar Disc Herniations. Spine J. 2011, 11, 122–130. [Google Scholar] [CrossRef]
- Jin-tao, Q.; Yu, T.; Mei, W.; Xu-dong, T.; Tian-jian, Z.; Guo-hua, S.; Lei, C.; Yue, H.; Zi-tian, W.; Yue, Z. Comparison of MIS vs. Open PLIF/TLIF with Regard to Clinical Improvement, Fusion Rate, and Incidence of Major Complication: A Meta-Analysis. Eur. Spine J. 2015, 24, 1058–1065. [Google Scholar] [CrossRef]
- Ahn, J.; Iqbal, A.; Manning, B.T.; Leblang, S.; Bohl, D.D.; Mayo, B.C.; Massel, D.H.; Singh, K. Minimally Invasive Lumbar Decompression—The Surgical Learning Curve. Spine J. 2016, 16, 909–916. [Google Scholar] [CrossRef]
- Morgenstern, R.; Morgenstern, C.; Yeung, A.T. The Learning Curve in Foraminal Endoscopic Discectomy: Experience Needed to Achieve a 90% Success Rate. SAS J. 2007, 1, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Nomura, K.; Yoshida, M. Assessment of the Learning Curve for Microendoscopic Decompression Surgery for Lumbar Spinal Canal Stenosis through an Analysis of 480 Cases Involving a Single Surgeon. Glob. Spine J. 2017, 7, 54–58. [Google Scholar] [CrossRef]
- Silva, P.S.; Pereira, P.; Monteiro, P.; Silva, P.A.; Vaz, R. Learning Curve and Complications of Minimally Invasive Transforaminal Lumbar Interbody Fusion. Neurosurg. Focus 2013, 35, E7. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Yeo, W.; Soeharno, H.; Yue, W.M. Learning Curve of a Complex Surgical Technique: Minimally Invasive Transforaminal Lumbar Interbody Fusion (MIS TLIF). J. Spinal Disord. Tech. 2014, 27, E234–E240. [Google Scholar] [CrossRef] [PubMed]
- Flynn, S.C.; Eli, I.M.; Ghogawala, Z.; Yew, A.Y. Minimally Invasive Surgery for Spinal Metastasis: A Review. World Neurosurg. 2022, 159, e32–e39. [Google Scholar] [CrossRef]
- Passias, P.G.; Tretiakov, P.S.; Nunley, P.D.; Wang, M.Y.; Park, P.; Kanter, A.S.; Okonkwo, D.O.; Eastlack, R.K.; Mundis, G.M.; Chou, D.; et al. Incremental Benefits of Circumferential Minimally Invasive Surgery for Increasingly Frail Patients with Adult Spinal Deformity. J. Neurosurg. Spine 2023, 39, 168–174. [Google Scholar] [CrossRef]
- De Win, G.; Van Bruwaene, S.; Kulkarni, J.; Van Calster, B.; Aggarwal, R.; Allen, C.; Lissens, A.; De Ridder, D.; Miserez, M. An Evidence-Based Laparoscopic Simulation Curriculum Shortens the Clinical Learning Curve and Reduces Surgical Adverse Events. Adv. Med. Educ. Pract. 2016, 7, 357–370. [Google Scholar] [CrossRef] [PubMed]
- Walliczek, U.; Förtsch, A.; Dworschak, P.; Teymoortash, A.; Mandapathil, M.; Werner, J.; Güldner, C. Effect of Training Frequency on the Learning Curve on the Da Vinci Skills Simulator. Head Neck 2016, 38, E1762–E1769. [Google Scholar] [CrossRef]
- Walliczek-Dworschak, U.; Mandapathil, M.; Förtsch, A.; Teymoortash, A.; Dworschak, P.; Werner, J.A.; Güldner, C. Structured Training on the Da Vinci Skills Simulator Leads to Improvement in Technical Performance of Robotic Novices. Clin. Otolaryngol. 2017, 42, 71–80. [Google Scholar] [CrossRef]
- Zaki, M.M.; Joshi, R.S.; Joseph, J.R.; Saadeh, Y.S.; Kashlan, O.N.; Godzik, J.; Uribe, J.S.; Park, P. Virtual Reality–Enabled Resident Education of Lateral-Access Spine Surgery. World Neurosurg. 2024, 183, e401–e407. [Google Scholar] [CrossRef]
- Akbulut, B.B.; Bölük, M.S.; Biçeroğlu, H.; Yurtseven, T. Evaluating the Efficacy of a Cost-Effective, Fully Three-Dimensional-Printed Vertebra Model for Endoscopic Spine Surgery Training for Neurosurgical Residents. Asian Spine J. 2024, 18, 630–638. [Google Scholar] [CrossRef]
- Chitale, R.; Ghobrial, G.M.; Lobel, D.; Harrop, J. Simulated Lumbar Minimally Invasive Surgery Educational Model with Didactic and Technical Components. Neurosurgery 2013, 73, S107–S110. [Google Scholar] [CrossRef]
- Buchanan, I.A.; Min, E.; Pham, M.H.; Donoho, D.A.; Bakhsheshian, J.; Minneti, M.; Zada, G.; Giannotta, S.L.; Hsieh, P.C.; Liu, J.C. Simulation of Dural Repair in Minimally Invasive Spine Surgery with the Use of a Perfusion-Based Cadaveric Model. Oper. Neurosurg. 2019, 17, 616–621. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, F.A.; Hussain, I.; Boadi, B.; Sommer, F.J.; Thomé, C.; Härtl, R. The Use of Augmented Reality as an Educational Tool in Minimally Invasive Transforaminal Lumbar Interbody Fusion. Oper. Neurosurg. 2025, 28, 183–192. [Google Scholar] [CrossRef]
- Rambani, R.; Ward, J.; Viant, W. Desktop-Based Computer-Assisted Orthopedic Training System for Spinal Surgery. J. Surg. Educ. 2014, 71, 805–809. [Google Scholar] [CrossRef] [PubMed]
- Harrop, J.; Rezai, A.R.; Hoh, D.J.; Ghobrial, G.M.; Sharan, A. Neurosurgical Training with a Novel Cervical Spine Simulator: Posterior Foraminotomy and Laminectomy. Neurosurgery 2013, 73 (Suppl. 1), S94–S99. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.B.; Perkins, E.; Harkey, H.L. A Novel Simulation Model for Minimally Invasive Spine Surgery. Oper. Neurosurg. 2009, 65, ons188–ons195. [Google Scholar] [CrossRef]
- Stienen, M.N.; Smoll, N.R.; Hildebrandt, G.; Schaller, K.; Gautschi, O.P. Early Surgical Education of Residents Is Safe for Microscopic Lumbar Disc Surgery. Acta Neurochir. 2014, 156, 1205–1214. [Google Scholar] [CrossRef]
- Ryu, W.H.A.; Mostafa, A.E.; Dharampal, N.; Sharlin, E.; Kopp, G.; Jacobs, W.B.; Hurlbert, R.J.; Chan, S.; Sutherland, G.R. Design-Based Comparison of Spine Surgery Simulators: Optimizing Educational Features of Surgical Simulators. World Neurosurg. 2017, 106, 870–877.e1. [Google Scholar] [CrossRef]
- Luciano, C.J.; Banerjee, P.P.; Sorenson, J.M.; Foley, K.T.; Ansari, S.A.; Rizzi, S.; Germanwala, A.V.; Kranzler, L.; Chittiboina, P.; Roitberg, B.Z. Percutaneous Spinal Fixation Simulation with Virtual Reality and Haptics. Neurosurgery 2013, 72 (Suppl. 1), A89–A96. [Google Scholar] [CrossRef]
- Kirkpatrick, J.S. A Comparison C1-C2 Transarticular Screw Placement after Self-Education and Mentored Education of Orthopaedic Residents. J. Spinal Disord. Tech. 2012, 25, E155–E160. [Google Scholar] [CrossRef]
- Sundar, S.J.; Healy, A.T.; Kshettry, V.R.; Mroz, T.E.; Schlenk, R.; Benzel, E.C. A Pilot Study of the Utility of a Laboratory-Based Spinal Fixation Training Program for Neurosurgical Residents. J. Neurosurg. Spine 2016, 24, 850–856. [Google Scholar] [CrossRef]
- Gardeck, A.M.; Pu, X.; Yang, Q.; Polly, D.W.; Jones, K.E. The Effect of Simulation Training on Resident Proficiency in Thoracolumbar Pedicle Screw Placement Using Computer-Assisted Navigation. J. Neurosurg. Spine 2020, 34, 127–134. [Google Scholar] [CrossRef]
- Melcher, C.; Hussain, I.; Kirnaz, S.; Goldberg, J.L.; Sommer, F.; Navarro-Ramirez, R.; Medary, B.; Härtl, R. Use of a High-Fidelity Training Simulator for Minimally Invasive Lumbar Decompression Increases Working Knowledge and Technical Skills Among Orthopedic and Neurosurgical Trainees. Glob. Spine J. 2023, 13, 2182–2192. [Google Scholar] [CrossRef] [PubMed]
- Alaraj, A.; Charbel, F.T.; Birk, D.; Tobin, M.; Luciano, C.; Banerjee, P.P.; Rizzi, S.; Sorenson, J.; Foley, K.; Slavin, K.; et al. Role of Cranial and Spinal Virtual and Augmented Reality Simulation Using Immersive Touch Modules in Neurosurgical Training. Neurosurgery 2013, 72, A115–A123. [Google Scholar] [CrossRef] [PubMed]
- Suresh, D.; Aydin, A.; James, S.; Ahmed, K.; Dasgupta, P. The Role of Augmented Reality in Surgical Training: A Systematic Review. Surg. Innov. 2023, 30, 366–382. [Google Scholar] [CrossRef] [PubMed]
- Walter, A.J. Surgical Education for the Twenty-First Century: Beyond the Apprentice Model. Obstet. Gynecol. Clin. N. Am. 2006, 33, 233–236. [Google Scholar] [CrossRef]
- Franzese, C.B.; Stringer, S.P. The Evolution of Surgical Training: Perspectives on Educational Models from the Past to the Future. Otolaryngol. Clin. N. Am. 2007, 40, 1227–1235. [Google Scholar] [CrossRef]
- Lallas, C.D.; Davis, J.W. Members of The Society of Urologic Robotic Surgeons Robotic Surgery Training with Commercially Available Simulation Systems in 2011: A Current Review and Practice Pattern Survey from the Society of Urologic Robotic Surgeons. J. Endourol. 2012, 26, 283–293. [Google Scholar] [CrossRef]
- Dunkin, B.; Adrales, G.L.; Apelgren, K.; Mellinger, J.D. Surgical Simulation: A Current Review. Surg. Endosc. 2007, 21, 357–366. [Google Scholar] [CrossRef]
- Ghaednia, H.; Fourman, M.S.; Lans, A.; Detels, K.; Dijkstra, H.; Lloyd, S.; Sweeney, A.; Oosterhoff, J.H.F.; Schwab, J.H. Augmented and Virtual Reality in Spine Surgery, Current Applications and Future Potentials. Spine J. 2021, 21, 1617–1625. [Google Scholar] [CrossRef]
- Ng, D.S.; Yip, B.H.K.; Young, A.L.; Yip, W.W.K.; Lam, N.M.; Li, K.K.; Ko, S.T.; Chan, W.H.; Aryasit, O.; Sikder, S.; et al. Cost-Effectiveness of Virtual Reality and Wet Laboratory Cataract Surgery Simulation. Medicine 2023, 102, e35067. [Google Scholar] [CrossRef]
- Papanikolaou, I.G.; Haidopoulos, D.; Paschopoulos, M.; Chatzipapas, I.; Loutradis, D.; Vlahos, N.F. Changing the Way We Train Surgeons in the 21th Century: A Narrative Comparative Review Focused on Box Trainers and Virtual Reality Simulators. Eur. J. Obstet. Gynecol. Reprod. Biol. 2019, 235, 13–18. [Google Scholar] [CrossRef]
- Henry, B.; Clark, P.; Sudan, R. Cost and Logistics of Implementing a Tissue-Based American College of Surgeons/Association of Program Directors in Surgery Surgical Skills Curriculum for General Surgery Residents of All Clinical Years. Am. J. Surg. 2014, 207, 201–208. [Google Scholar] [CrossRef]
- Zhao, Z.; Ma, Y.; Mushtaq, A.; Radhakrishnan, V.; Hu, Y.; Ren, H.; Song, W.; Tse, Z.T.H. Engineering Functional and Anthropomorphic Models for Surgical Training in Interventional Radiology: A State-of-the-Art Review. Proc. Inst. Mech. Eng. H 2023, 237, 3–17. [Google Scholar] [CrossRef]
- Abboudi, H.; Khan, M.S.; Aboumarzouk, O.; Guru, K.A.; Challacombe, B.; Dasgupta, P.; Ahmed, K. Current Status of Validation for Robotic Surgery Simulators—A Systematic Review. BJU Int. 2013, 111, 194–205. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Rodrigues Armijo, P.; Krause, C.; Siu, K.-C.; Oleynikov, D. SAGES Robotic Task Force A Comprehensive Review of Robotic Surgery Curriculum and Training for Residents, Fellows, and Postgraduate Surgical Education. Surg. Endosc. 2020, 34, 361–367. [Google Scholar] [CrossRef]
- Herron, D.M.; Marohn, M. The SAGES-MIRA Robotic Surgery Consensus Group A Consensus Document on Robotic Surgery. Surg. Endosc. 2008, 22, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Prasad, D.; Weiss, B.E.; Snyder, L.A.; Arnold, P.M.; Rosenow, J.M. Where are all the neurosurgery robots? J. Robot. Surg. 2025, 19, 267. [Google Scholar] [CrossRef]
- Purnell, S.M.; Bass, B.L.; Benavides, B.; Martinez, S.; McNeil, S.G.; Dickinson, K.J. Template for a Program Tailored ACS/APDS Phase 1 Curriculum: From Needs Assessment to Implementation. Am. J. Surg. 2022, 223, 257–265. [Google Scholar] [CrossRef]
- Ghaderi, I.; Fitzgibbons, S.; Watanabe, Y.; Lachapelle, A.; Paige, J. Surgical Skills Curricula in American College of Surgeons Accredited Education Institutes: An International Survey. Am. J. Surg. 2017, 213, 678–686. [Google Scholar] [CrossRef] [PubMed]
- Niitsu, H.; Hirabayashi, N.; Yoshimitsu, M.; Mimura, T.; Taomoto, J.; Sugiyama, Y.; Murakami, S.; Saeki, S.; Mukaida, H.; Takiyama, W. Using the Objective Structured Assessment of Technical Skills (OSATS) Global Rating Scale to Evaluate the Skills of Surgical Trainees in the Operating Room. Surg. Today 2013, 43, 271–275. [Google Scholar] [CrossRef]
| Study | MISS Technique | Training Type | Primary Outcomes |
|---|---|---|---|
| Zaki et al. (2024) [19], World Neurosurgery | MIS-LLIF | VR lateral spine module | Increased precision scores and decreased radiograph usage for the majority of resident participants. Reduced operation time and increased confidence in performing MIS-LLIF in all participants |
| Akbulut et al. (2024) [20], Asian Spine Journal | MIS Lumbar Discectomy | 3D-printed MIS spinal model | Mean operative time decreased from 21 min 18 s to 6 min 45 s after fourth practice with model (p < 0.0001) |
| Chitale et al. (2013) [21], Neurosurgery | Pedicle Screw Insertion | Didactic curriculum + MIS simulation model | Mean written test score improved from 78% to 100% after a 2 h didactic curriculum; Improvements in technical score for CT and fluoroscopic navigation also improved, although this was not statistically significant |
| Buchanan et al. (2019) [22], Operative Neurosurgery | MIS Dural Repair | Perfusion-based cadaveric model | Mean dural closure time improved from 12 min 7 s to 7 min 4 s (p = 0.02) |
| Schmidt et al. (2025) [23], Operative Neurosurgery | MIS-TLIF | High-Fidelity Lumbar Spine Simulation model ± Augmented Reality | AR supplementation resulted in significantly decreased mental demand (p = 0.003) and significantly less difficulty in maintaining performance levels during the procedure (p = 0.019) |
| Rambani et al. (2014) [24], Journal of Surgical Education | Pedicle Screw Insertion | Computer Simulation | Significant improvements in operative time, fixation accuracy, and reduction in fluoroscopy exposures (p < 0.05) |
| Harrop et al. (2013) [25], Neurosurgery | Navigation | Didactic curriculum + PCDF Simulation Model | Didactic scores improved in 78% of participants (p = 0.005); technical scores increased from a mean of 14.1 to 22.4 (p = 0.02). |
| Walker et al. (2009) [26], Neurosurgery | Pedicle Screw Insertion/MISS Laminectomy | Surgical simulator with animal model | Improvements in self-reported junior and senior resident confidence in MISS laminectomy and pedicle screw insertion, respectively |
| Stienen et al. (2014) [27], Acta Neurochirurgica | Microscopic Lumbar Disk Herniation repair | Resident involvement in surgery | No significant differences in intraoperative blood loss, surgery duration, complication rates, post-surgical pain reduction, or quality of life outcomes between teaching and non-teaching cases. |
| Ryu et al. (2017) [28], World Neurosurgery | Pedicle Screw Insertion | Computer-based simulator; synthetic model | Computer-based simulators successfully incorporate procedural guidance and real-time feedback, while synthetic models provide more realistic haptic feedback and allow for utilization of real surgical tools |
| Luciano et al. (2013) [29], Neurosurgery | Percutaneous Needle Placement | Mixed Augmented Reality + Haptic Feedback Simulator | Performance accuracy significantly improved between first and second attempts (p = 0.04) |
| Kirkpatrick (2012) [30], Journal of Spinal Disorders & Techniques | Pedicle Screw Insertion | Mentored surgical training on models | Mentored residents showed significantly greater improvements in performance scores (p = 0.0068). Subsequent screw placement error rate was significantly lower in the mentored group than non-mentored controls (p = 0.004) |
| Sundar et al. (2016) [31], Journal of Neurosurgery: Spine | Pedicle Screw Insertion | Cadaver or Sawbones model + surgical navigation training session | Significant reduction in overall surgical error (p = 0.04) compared to controls. Fewer errors in thoracic (p = 0.02) and lumbar (p = 0.04) regions, with more optimal screw placement in the cervical, thoracic, and lumbar regions (p = 0.02, p = 0.04, p = 0.04, respectively) |
| Gardeck et al. (2020) [32], Journal of Neurosurgery: Spine | Pedicle Screw Insertion | Lecture + Synthetic spine model w/3D computer-assisted navigation | Regardless of previous experience, all residents showed significant improvement on subjective measures for navigated screw placement (p < 0.001). Nearly all residents showed improvements on objective measures for navigated screw placement (p < 0.001) and reduced their screw placement time from session 1 to session 2 (p = 0.006) |
| Melcher et al. (2023) [33], Global Spine Journal | MIS-ULBD | High-Fidelity Simulator | By the third practice, the average procedural time decreased by 31.7 min, while skipped steps and surgical errors significantly declined. Surgical proficiency improved, particularly in efficiency, smoothness, and instrument handling. Knowledge gap decreased by 30% (p = 0.001), with the greatest gains among junior residents. |
| Alaraj et al. (2013) [34], Neurosurgery | Pedicle Screw Insertion | Multi-modal augmented reality simulator | Less fluoroscopy necessary to achieve accurate pedicle screw placement |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oblich, M.C.; Lyman, J.G.; Jain, R.; Prasad, D.; Romanos, S.; Dahdaleh, N.; El Tecle, N.E.; Ahuja, C.S. Resident Training in Minimally Invasive Spine Surgery: A Scoping Review. Brain Sci. 2025, 15, 936. https://doi.org/10.3390/brainsci15090936
Oblich MC, Lyman JG, Jain R, Prasad D, Romanos S, Dahdaleh N, El Tecle NE, Ahuja CS. Resident Training in Minimally Invasive Spine Surgery: A Scoping Review. Brain Sciences. 2025; 15(9):936. https://doi.org/10.3390/brainsci15090936
Chicago/Turabian StyleOblich, Michael C., James G. Lyman, Rishi Jain, Dillan Prasad, Sharbel Romanos, Nader Dahdaleh, Najib E. El Tecle, and Christopher S. Ahuja. 2025. "Resident Training in Minimally Invasive Spine Surgery: A Scoping Review" Brain Sciences 15, no. 9: 936. https://doi.org/10.3390/brainsci15090936
APA StyleOblich, M. C., Lyman, J. G., Jain, R., Prasad, D., Romanos, S., Dahdaleh, N., El Tecle, N. E., & Ahuja, C. S. (2025). Resident Training in Minimally Invasive Spine Surgery: A Scoping Review. Brain Sciences, 15(9), 936. https://doi.org/10.3390/brainsci15090936







