Implications of Indolethylamine N-Methyltransferase (INMT) in Health and Disease: Biological Functions, Disease Associations, Inhibitors, and Analytical Approaches
Abstract
1. Introduction
1.1. History and Discovery of INMT
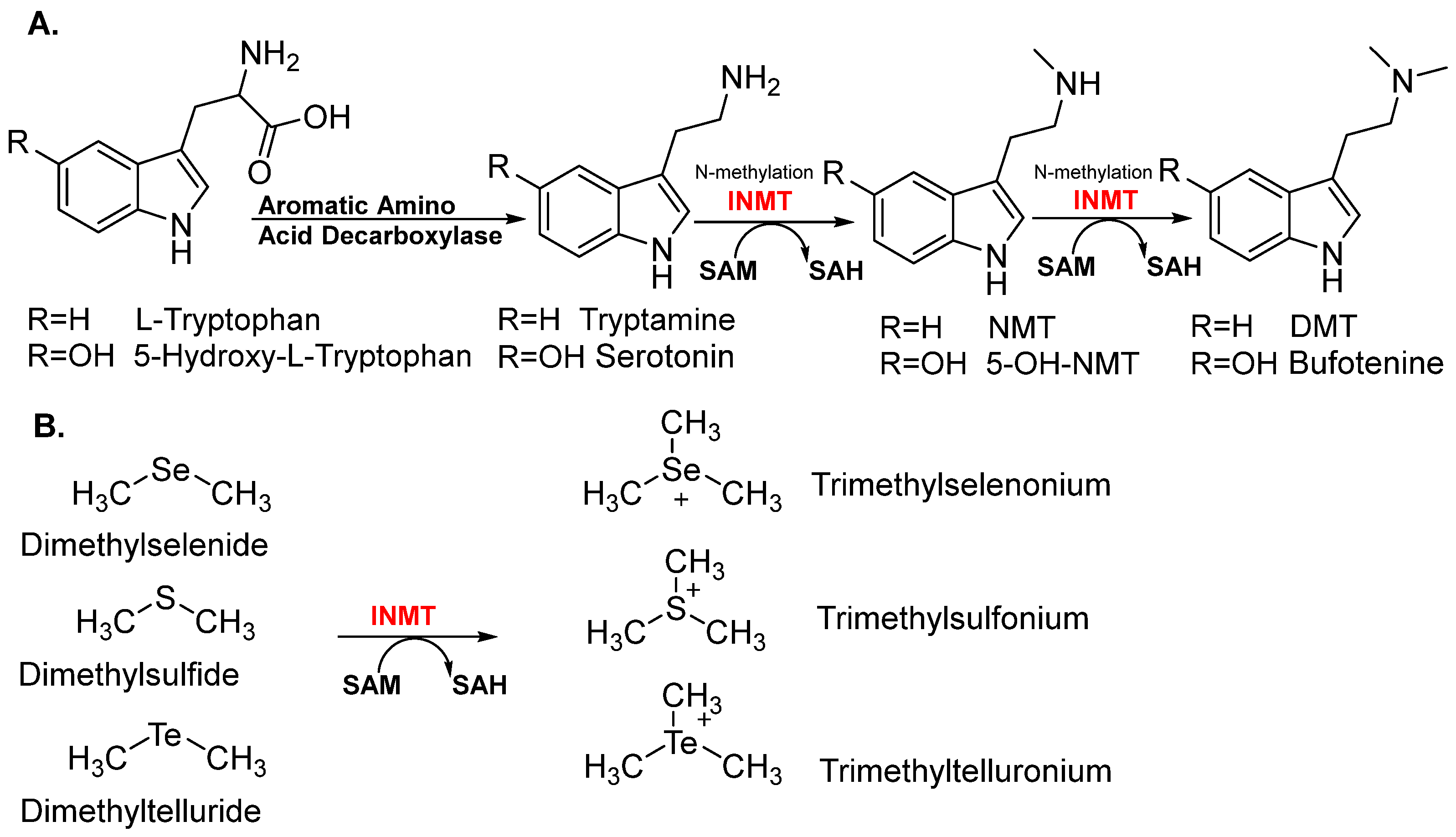
1.2. Substrates and Physiological Relevance
1.3. INMT in Neuropsychiatric and Neurodegenerative Disorders
1.4. INMT in Cancer
1.5. Enzyme Inhibitors and Regulation
1.6. Analytical Approaches for INMT Activity Assessment
2. INMT Characterization and Distribution
2.1. Cloning and Genomic Characterization of INMT
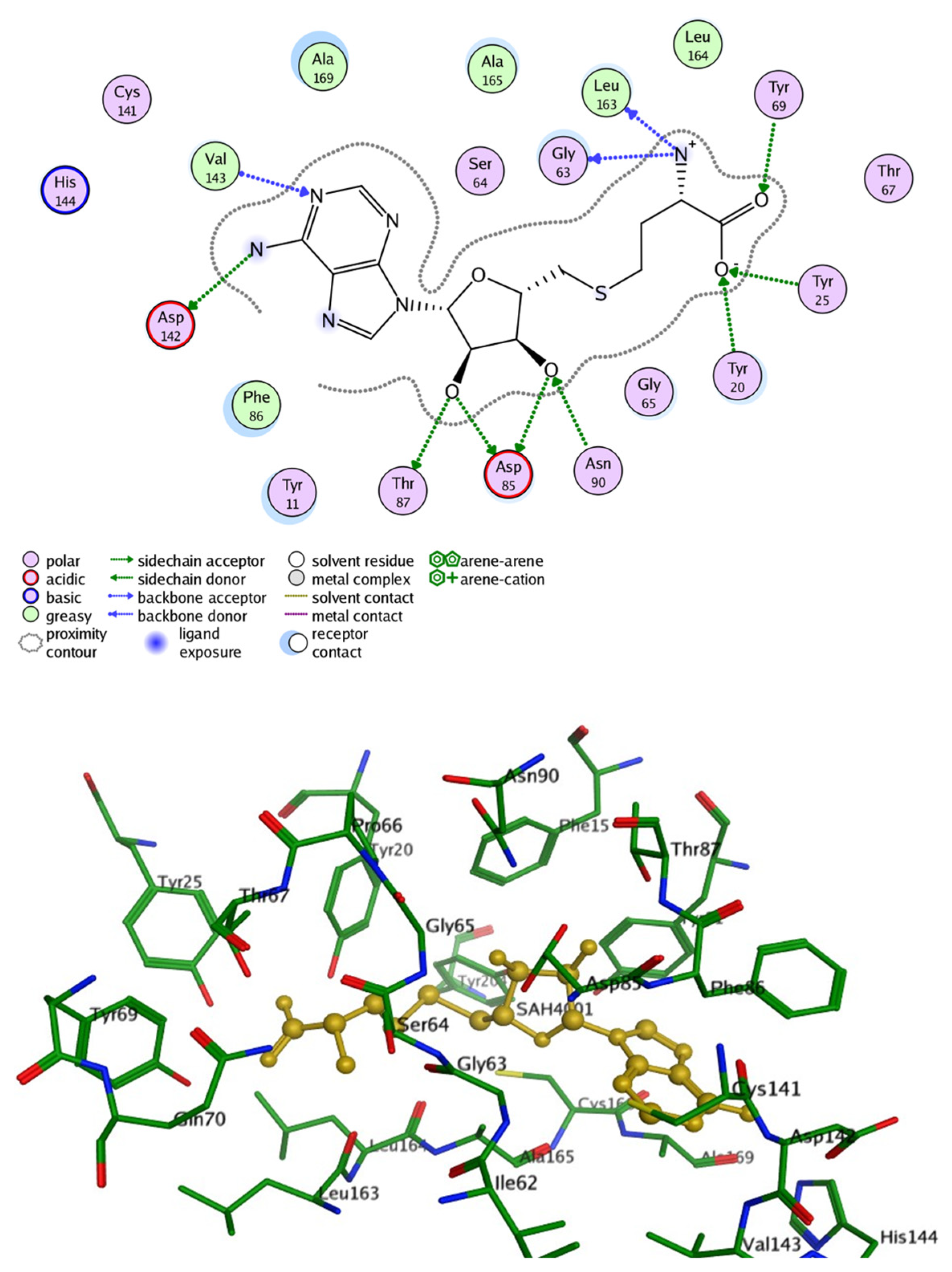
2.2. Protein Structure and Active Site of INMT
2.3. Comparative Structural Features Across Species
2.4. Tissue Distribution and Functional Activity of INMT
3. INMT and Related Disorders
3.1. Schizophrenia
3.2. Alzheimer’s Disease (AD)
3.3. Amyotrophic Lateral Sclerosis (ALS)
3.4. Autism
3.5. Cancer
3.5.1. Castration-Resistant Prostate Cancer (CRPC)
3.5.2. Lung Cancer
3.5.3. Hepatocellular Carcinoma
3.5.4. Other Cancer Types
3.6. Hirschsprung’s Disease (HSCR)
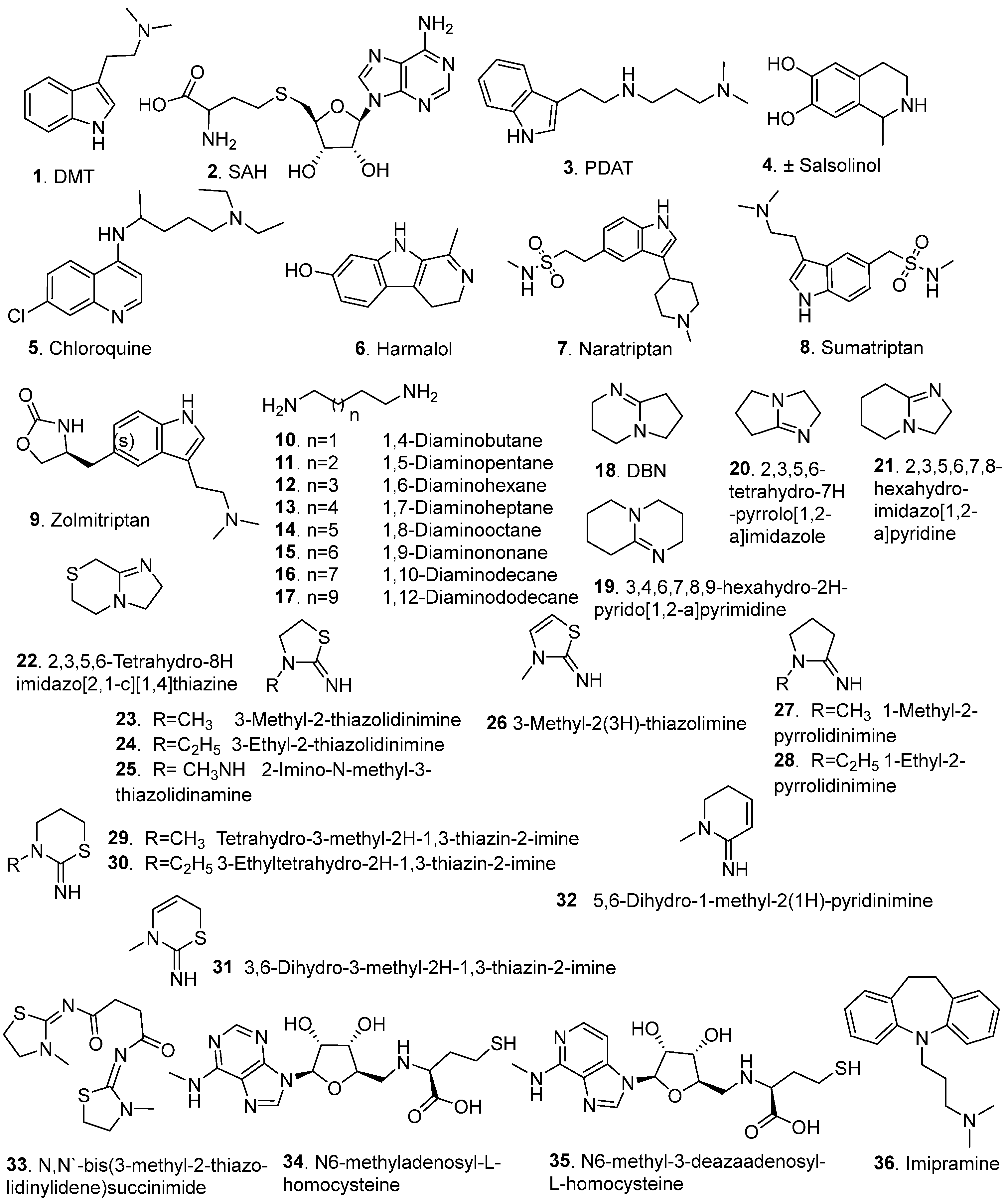
4. INMT Inhibitors
4.1. Endogenous Inhibitors
4.2. Synthetic Inhibitors
5. Measurement of INMT Enzyme Activity by Discontinuous Assays
5.1. Radiometric Assay
5.2. TLC and Spray Reagent Method
5.3. UHPLC-MS/MS Analysis Method
6. Monitoring the INMT Activity Using Real-Time Assays
6.1. Methyltransferase Colorimetric Assay
6.2. Methyltransferase Fluorometric Assay
6.3. Apta Fluor SAH Methyltransferase TR-FRET Assay
6.4. HTRF EPIgeneousTM Methyltransferase Assay
6.5. MTase-Glo™ Assay
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| INMT | Indolethylamine N-methyltransferase |
| DMT | N,N-dimethyltryptamine |
| NMT | N-methyltransferase |
| SAM | S-adenosyl-L-methionine |
| MAO | Monoamine oxidase |
| SAH | S-adenosyl-L-homocysteine |
| NNMT | Nicotinamide N-methyltransferase |
| PNMT | Phenylethanolamine N-methyltransferase |
| CRPC | Castration-resistant prostate cancer |
| NSCLC | Non-small cell lung cancer |
| UCEC | Uterine corpus endometrial carcinoma |
| HSCR | Hirschsprung’s disease |
| ALS | Amyotrophic lateral sclerosis |
References
- Thompson, M.A.; Weinshilboum, R.M. Rabbit Lung Indolethylamine N-Methyltransferase: cDNA And Gene Cloning And Characterization. J. Biol. Chem. 1998, 273, 34502–34510. [Google Scholar] [CrossRef]
- Dean, J.G. Indolethylamine-N-Methyltransferase Polymorphisms: Genetic and Biochemical Approaches for Study of Endogenous N,N,-Dimethyltryptamine. Front. Neurosci. 2018, 12, 232. [Google Scholar] [CrossRef] [PubMed]
- Axelrod, J. Enzymatic Formation of Psychotomimetic Metabolites from Normally Occurring Compounds. Science 1961, 134, 343. [Google Scholar] [CrossRef]
- Barker, S.A.; Monti, J.A.; Christian, S.T. N,N-Dimethyltryptamine: An Endogenous Hallucinogen. Int. Rev. Neurobiol. 1981, 22, 83–110, ISBN 0074-7742. [Google Scholar]
- Thompson, M.A.; Moon, E.; Kim, U.-J.; Xu, J.; Siciliano, M.J.; Weinshilboum, R.M. Human Indolethylamine N-Methyltransferase: CDNA Cloning and Expression, Gene Cloning, and Chromosomal Localization. Genomics 1999, 61, 285–297. [Google Scholar] [CrossRef] [PubMed]
- Wyatt, R.J.; Saavedra, J.M.; Axelrod, J. A Dimethyltryptamine-Forming Enzyme in Human Blood. Am. J. Psychiatry 1973, 130, 754–760. [Google Scholar] [CrossRef] [PubMed]
- Chu, U.B.; Vorperian, S.K.; Satyshur, K.; Eickstaedt, K.; Cozzi, N.V.; Mavlyutov, T.; Hajipour, A.R.; Ruoho, A.E. Noncompetitive Inhibition of Indolethylamine-N-Methyltransferase by N,N-Dimethyltryptamine and N,N-Dimethylaminopropyltryptamine. Biochemistry 2014, 53, 2956–2965. [Google Scholar] [CrossRef]
- Nichols, D.E. Psychedelics. Pharmacol. Rev. 2016, 68, 264–355. [Google Scholar] [CrossRef]
- Carbonaro, T.M.; Gatch, M.B. Neuropharmacology of N,N-Dimethyltryptamine. Brain Res. Bull. 2016, 126, 74–88. [Google Scholar] [CrossRef]
- Strahilevitz, M.; Narasimhachari, N.; Fischer, G.W.; Meltzer, H.Y.; Himwich, H.E. Indolethylamine-N-Methyltransferase Activity in Psychiatric Patients and Controls. Biol. Psychiatry 1975, 10, 287–302. [Google Scholar] [PubMed]
- Osmond, H.; Smythies, J. Schizophrenia: A New Approach. J. Ment. Sci. 1952, 98, 309–315. [Google Scholar] [CrossRef]
- Barbic, D.; Fernandes, J.; Eberdt, C.; Chakraborty, A. N,N-Dimethyltryptamine: DMT-Induced Psychosis. Am. J. Emerg. Med. 2020, 38, 1961.e1–1961.e2. [Google Scholar] [CrossRef]
- Grammenos, D.; Barker, S.A. On the Transmethylation Hypothesis: Stress, N,N-Dimethyltryptamine, and Positive Symptoms of Psychosis. J. Neural. Transm. 2015, 122, 733–739. [Google Scholar] [CrossRef]
- Emanuele, E.; Colombo, R.; Martinelli, V.; Brondino, N.; Marini, M.; Boso, M.; Barale, F.; Politi, P. Elevated Urine Levels of Bufotenine in Patients with Autistic Spectrum Disorders and Schizophrenia. Neuroendocrinol. Lett. 2010, 31, 117–121. [Google Scholar] [PubMed]
- Fontanilla, D.; Johannessen, M.; Hajipour, A.R.; Cozzi, N.V.; Jackson, M.B.; Ruoho, A.E. The Hallucinogen N,N-Dimethyltryptamine (DMT) Is an Endogenous Sigma-1 Receptor Regulator. Science 2009, 323, 934–937. [Google Scholar] [CrossRef]
- Cheng, D.; Lei, Z.-G.; Chu, K.; Lam, O.J.H.; Chiang, C.Y.; Zhang, Z.-J. N, N-Dimethyltryptamine, a Natural Hallucinogen, Ameliorates Alzheimer’s Disease by Restoring Neuronal Sigma-1 Receptor-Mediated Endoplasmic Reticulum-Mitochondria Crosstalk. Alzheimers Res. Ther. 2024, 16, 95. [Google Scholar] [CrossRef] [PubMed]
- Mu, H.; Ye, L.; Wang, B. Detailed Resume of S-Methyltransferases: Categories, Structures, Biological Functions and Research Advancements in Related Pathophysiology and Pharmacotherapy. Biochem. Pharmacol. 2024, 226, 116361. [Google Scholar] [CrossRef]
- Zhou, X.; Zou, B.; Wang, J.; Wu, L.; Tan, Q.; Ji, C. Low Expression of INMT Is Associated with Poor Prognosis but Favorable Immunotherapy Response in Lung Adenocarcinoma. Front. Genet. 2022, 13, 946848. [Google Scholar] [CrossRef]
- Zhong, S.; Jeong, J.-H.; Huang, C.; Chen, X.; Dickinson, S.I.; Dhillon, J.; Yang, L.; Luo, J.-L. Targeting INMT and Interrupting Its Methylation Pathway for the Treatment of Castration Resistant Prostate Cancer. J. Exp. Clin. Cancer Res. 2021, 40, 307. [Google Scholar] [CrossRef]
- Lin, R.-L.; Narasimhachari, N.; Himwich, H.E. Inhibition of Indolethylamine-N-Methyltransferase by S-Adenosylhomocysteine. Biochem. Biophys. Res. Commun. 1973, 54, 751–759. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, J.H.; Bouso, J.C. Significance of Mammalian N, N-Dimethyltryptamine (DMT): A 60-Year-Old Debate. J. Psychopharmacol. 2022, 36, 905–919. [Google Scholar] [CrossRef]
- Rokach, J.; Hamel, P.; Hunter, N.R.; Reader, G.; Rooney, C.S.; Anderson, P.S.; Cragoe, E.J.J.; Mandel, L.R. Cyclic Amidine Inhibitors of Indolamine N-Methyltransferase. J. Med. Chem. 1979, 22, 237–247. [Google Scholar] [CrossRef]
- Porta, R.; Camardella, M.; Esposito, C.; Della Pietra, G. Inhibition of Indolethylamine-N-Methyltransferase by Aliphatic Diamines. Biochem. Biophys. Res. Commun. 1977, 77, 1196–1202. [Google Scholar] [CrossRef]
- Rokach, J.; Girard, Y.; Hamel, P.; Reader, G.; Rooney, C.S.; Mandel, L.R.; Cragoe, E.J., Jr.; Zacchei, A.G. Inhibitors of Indoleethylamine N-Methyltransferase. Derivatives of 3-Methyl-2-Thiazolidinimine. In Vitro, in Vivo, and Metabolic Studies. J. Med. Chem. 1980, 23, 773–780. [Google Scholar] [CrossRef]
- Glynos, N.G.; Carter, L.; Lee, S.J.; Kim, Y.; Kennedy, R.T.; Mashour, G.A.; Wang, M.M.; Borjigin, J. Indolethylamine N-Methyltransferase (INMT) Is Not Essential for Endogenous Tryptamine-Dependent Methylation Activity in Rats. Sci. Rep. 2023, 13, 280. [Google Scholar] [CrossRef] [PubMed]
- BellBrookLabs SAH Methyltransferase TR-FRET AssayAptaFluorTM. Available online: https://bellbrooklabs.com/products/assays/aptafluor-sah-methyltransferase-assay-kits/ (accessed on 17 March 2025).
- Revvity HTRF EPIgeneous Methyltransferase Assay Kit. Available online: https://www.revvity.com/product/htrf-methyltransferase-kit-1k-pts-62sahpeb (accessed on 10 March 2025).
- Cayman Chemical Methyltransferase Colorimetric Assay Kit. Available online: https://www.caymanchem.com/product/700140/methyltransferase-colorimetric-assay-kit (accessed on 17 March 2025).
- Cayman Chemical Methyltransferase Fluorometric Assay Kit. Available online: https://www.caymanchem.com/product/700150/methyltransferase-fluorometric-assay-kit (accessed on 17 March 2025).
- Fukumoto, Y.; Kyono, R.; Shibukawa, Y.; Tanaka, Y.; Suzuki, N.; Ogra, Y. Differential Molecular Mechanisms of Substrate Recognition by Selenium Methyltransferases, INMT and TPMT, in Selenium Detoxification and Excretion. J. Biol. Chem. 2024, 300, 105599. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Dong, A.; Zeng, H.; Loppnau, P.; Sundstrom, M.; Arrowsmith, C.H. The Crystal Structure of Human Indolethylamine Nmethyltransferase in Complex with SAH. Protein Data Bank Entry 2A14 2005. [Google Scholar] [CrossRef]
- Zhang, A.-A.; He, Q.-L.; Zhao, Q. Mining and Characterization of Indolethylamine N-Methyltransferases in Amphibian Toad Bufo Gargarizans. Biochemistry 2023, 62, 2371–2381. [Google Scholar] [CrossRef]
- Thompson, M.A.; Weinshilboum, R.M.; El Yazal, J.; Wood, T.C.; Pang, Y.-P. Rabbit Indolethylamine N-Methyltransferase Three-Dimensional Structure Prediction: A Model Approach to Bridge Sequence to Function in Pharmacogenomic Studies. Mol. Model. Annu. 2001, 7, 324–333. [Google Scholar] [CrossRef]
- Mavlyutov, T.A.; Epstein, M.L.; Liu, P.; Verbny, Y.I.; Ziskind-Conhaim, L.; Ruoho, A.E. Development of the Sigma-1 Receptor in C-Terminals of Motoneurons and Colocalization with the N,N′-Dimethyltryptamine Forming Enzyme, Indole-N-Methyl Transferase. Neuroscience 2012, 206, 60–68. [Google Scholar] [CrossRef]
- Barker, S.A. N, N-Dimethyltryptamine (DMT), an Endogenous Hallucinogen: Past, Present, and Future Research to Determine Its Role and Function. Front. Neurosci. 2018, 12, 536. [Google Scholar] [CrossRef]
- Barker, S.A.; Borjigin, J.; Lomnicka, I.; Strassman, R. LC/MS/MS Analysis of the Endogenous Dimethyltryptamine Hallucinogens, Their Precursors, and Major Metabolites in Rat Pineal Gland Microdialysate. Biomed. Chromatogr. 2013, 27, 1690–1700. [Google Scholar] [CrossRef]
- Cozzi, N.V.; Mavlyutov, T.A.; Thompson, M.A.; Ruoho, A.E. Indolethylamine N-Methyltransferase Expression in Primate Nervous Tissue. Soc. Neurosci. 2011, 37, 840. [Google Scholar]
- Axt, A. Autism Viewed as a Consequence of Pineal Gland Malfunction. Farmakoter. w Psychiatr. I Neurol 1998, 98, 112–134. [Google Scholar]
- Kopantzev, E.P.; Monastyrskaya, G.S.; Vinogradova, T.V.; Zinovyeva, M.V.; Kostina, M.B.; Filyukova, O.B.; Tonevitsky, A.G.; Sukhikh, G.T.; Sverdlov, E.D. Differences in Gene Expression Levels between Early and Later Stages of Human Lung Development Are Opposite to Those between Normal Lung Tissue and Non-Small Lung Cell Carcinoma. Lung Cancer 2008, 62, 23–34. [Google Scholar] [CrossRef]
- Longnecker, M.P.; Taylor, P.R.; Levander, O.A.; Howe, M.; Veillon, C.; McAdam, P.A.; Patterson, K.Y.; Holden, J.M.; Stampfer, M.J.; Morris, J.S. Selenium in Diet, Blood, and Toenails in Relation to Human Health in a Seleniferous Area. Am. J. Clin. Nutr. 1991, 53, 1288–1294. [Google Scholar] [CrossRef] [PubMed]
- Ľupták, M.; Michaličková, D.; Fišar, Z.; Kitzlerová, E.; Hroudová, J. Novel Approaches in Schizophrenia-from Risk Factors and Hypotheses to Novel Drug Targets. World J. Psychiatry 2021, 11, 277–296. [Google Scholar] [CrossRef] [PubMed]
- Insel, T.R. Rethinking Schizophrenia. Nature 2010, 468, 187–193. [Google Scholar] [CrossRef]
- Fišar, Z. Biological Hypotheses, Risk Factors, and Biomarkers of Schizophrenia. Prog. Neuro Psychopharmacol. Biol. Psychiatry 2023, 120, 110626. [Google Scholar] [CrossRef] [PubMed]
- Biedermann, F.; Fleischhacker, W.W. Psychotic Disorders in DSM-5 and ICD-11. CNS Spectr. 2016, 21, 349–354. [Google Scholar] [CrossRef]
- Howes, O.; McCutcheon, R.; Stone, J. Glutamate and Dopamine in Schizophrenia: An Update for the 21st Century. J. Psychopharmacol. 2015, 29, 97–115. [Google Scholar] [CrossRef]
- Murray, R.M.; Oon, M.C.H.; Rodnight, R.; Birley, J.L.T.; Smith, A. Increased Excretion of Dimethyltryptamine and Certain Features of Psychosis: A Possible Association. Arch. Gen. Psychiatry 1979, 36, 644–649. [Google Scholar] [CrossRef]
- Uebelhack, R.; Franke, L.; Seidel, K. Methylated and unmethylated indolamine in the cisternal fluid in acute endogenous psychoses. Biomed. Biochim. Acta 1983, 42, 1343–1346. [Google Scholar] [PubMed]
- Lataster, T.; Valmaggia, L.; Lardinois, M.; van Os, J.; Myin-Germeys, I. Increased Stress Reactivity: A Mechanism Specifically Associated with the Positive Symptoms of Psychotic Disorder. Psychol. Med. 2013, 43, 1389–1400. [Google Scholar] [CrossRef] [PubMed]
- Myin-Germeys, I.; Delespaul, P.H.; Van Os, J. Behavioural Sensitization to Daily Life Stress in Psychosis. Psychol. Med. 2005, 35, 733–741. [Google Scholar] [CrossRef] [PubMed]
- Harrison, R.E.W.; Christian, S.T. Individual Housing Stress Elevates Brain and Adrenal Tryptamine Content B-Neurobiology of the Trace Amines: Analytical, Physiological, Pharmacological, Behavioral, and Clinical Aspects; Boulton, A.A., Baker, G.B., Dewhurst, W.G., Sandler, M., Eds.; Humana Press: Totowa, NJ, USA, 1984; pp. 249–255. ISBN 978-1-4612-5312-9. [Google Scholar]
- Curzon, G.; Joseph, M.H.; Knott, P.J. EFFECTS OF IMMOBILIZATION AND FOOD DEPRIVATION ON RAT BRAIN TRYPTOPHAN METABOLISM. J. Neurochem. 1972, 19, 1967–1974. [Google Scholar] [CrossRef]
- Cozzi, N.V.; Gopalakrishnan, A.; Anderson, L.L.; Feih, J.T.; Shulgin, A.T.; Daley, P.F.; Ruoho, A.E. Dimethyltryptamine and Other Hallucinogenic Tryptamines Exhibit Substrate Behavior at the Serotonin Uptake Transporter and the Vesicle Monoamine Transporter. J. Neural Transm. 2009, 116, 1591–1599. [Google Scholar] [CrossRef] [PubMed]
- Govitrapong, P.; Mukda, S.; Turakitwanakan, W.; Dumrongphol, H.; Chindaduangratn, C.; Sanvarinda, Y. Platelet Serotonin Transporter in Schizophrenic Patients with and without Neuroleptic Treatment. Neurochem. Int. 2002, 41, 209–216. [Google Scholar] [CrossRef]
- Zucker, M.; Valevski, A.; Weizman, A.; Rehavi, M. Increased Platelet Vesicular Monoamine Transporter Density in Adult Schizophrenia Patients. Eur. Neuropsychopharmacol. 2002, 12, 343–347. [Google Scholar] [CrossRef]
- Tillinger, A.; Sollas, A.; Serova, L.I.; Kvetnansky, R.; Sabban, E.L. Vesicular Monoamine Transporters (VMATs) in Adrenal Chromaffin Cells: Stress-Triggered Induction of VMAT2 and Expression in Epinephrine Synthesizing Cells. Cell. Mol. Neurobiol. 2010, 30, 1459–1465. [Google Scholar] [CrossRef] [PubMed]
- Doyle, A.; Hucklebridge, F.; Evans, P.; Clow, A. Urinary Output of Endogenous Monoamine Oxidase Inhibitory Activity Is Related to Everyday Stress. Life Sci. 1996, 58, 1723–1730. [Google Scholar] [CrossRef]
- Riga, M.S.; Soria, G.; Tudela, R.; Artigas, F.; Celada, P. The Natural Hallucinogen 5-MeO-DMT, Component of Ayahuasca, Disrupts Cortical Function in Rats: Reversal by Antipsychotic Drugs. Int. J. Neuropsychopharmacol. 2014, 17, 1269–1282. [Google Scholar] [CrossRef]
- Uhlhaas, P.J.; Singer, W. Neural Synchrony in Brain Disorders: Relevance for Cognitive Dysfunctions and Pathophysiology. Neuron 2006, 52, 155–168. [Google Scholar] [CrossRef]
- Barker, S.A.; McIlhenny, E.H.; Strassman, R. A Critical Review of Reports of Endogenous Psychedelic N, N-Dimethyltryptamines in Humans: 1955–2010. Drug Test. Anal. 2012, 4, 617–635. [Google Scholar] [CrossRef] [PubMed]
- Carlsson, A. The Current Status of the Dopamine Hypothesis of Schizophrenia. Neuropsychopharmacology 1988, 1, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Davis, K.L.; Kahn, R.S.; Ko, G.; Davidson, M. Dopamine in Schizophrenia: A Review and Reconceptualization. Am. J. Psychiatry 1991, 148, 1474–1486. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.L.E.E. Some Neurochemical Effects Of N,N-Dimethyltryptamine And Their Possible Relation To Acute Schizophrenia. Ph.D. Thesis, University of Nevada, Reno, NV, USA, 1975. [Google Scholar]
- Smith, T.L. Increased Synthesis of Striatal Dopamine by N,N-Dimethyltryptamine. Life Sci. 1977, 21, 1597–1601. [Google Scholar] [CrossRef]
- Waldmeier, P.C.; Maître, L. Neurochemical Investigations of the Interaction of N,N-Dimethyltryptamine with the Dopaminergic System in Rat Brain. Psychopharmacology 1977, 52, 137–144. [Google Scholar] [CrossRef]
- Glynos, N.G.; Huels, E.R.; Nelson, A.; Kim, Y.; Kennedy, R.T.; Mashour, G.A.; Pal, D. Neurochemical and Neurophysiological Effects of Intravenous Administration of N,N-Dimethyltryptamine in Rats. bioRxiv Prepr. Serv. Biol. 2025, 2024, 19.589047. [Google Scholar] [CrossRef]
- Van den Buuse, M. Modeling the Positive Symptoms of Schizophrenia in Genetically Modified Mice: Pharmacology and Methodology Aspects. Schizophr. Bull. 2010, 36, 246–270. [Google Scholar] [CrossRef]
- Jenner, P.; Marsden, C.D.; Thanki, C.M. Behavioural Changes Induced by N,N-Dimethyltryptamine in Rodents [Proceedings]. Br. J. Pharmacol. 1978, 63, 380. [Google Scholar] [CrossRef]
- Su, T.-P.; Hayashi, T.; Vaupel, D.B. When the Endogenous Hallucinogenic Trace Amine N,N-Dimethyltryptamine Meets the Sigma-1 Receptor. Sci. Signal. 2009, 2, pe12. [Google Scholar] [CrossRef] [PubMed]
- Athar, T.; Al Balushi, K.; Khan, S.A. Recent Advances on Drug Development and Emerging Therapeutic Agents for Alzheimer’s Disease. Mol. Biol. Rep. 2021, 48, 5629–5645. [Google Scholar] [CrossRef]
- Dhakal, S.; Kushairi, N.; Phan, C.W.; Adhikari, B.; Sabaratnam, V.; Macreadie, I. Dietary Polyphenols: A Multifactorial Strategy to Target Alzheimer’s Disease. Int. J. Mol. Sci. 2019, 20, 5090. [Google Scholar] [CrossRef]
- Eysert, F.; Kinoshita, P.F.; Mary, A.; Vaillant-Beuchot, L.; Checler, F.; Chami, M. Molecular Dysfunctions of Mitochondria-Associated Membranes (MAMs) in Alzheimer’s Disease. Int. J. Mol. Sci. 2020, 21, 9521. [Google Scholar] [CrossRef] [PubMed]
- Hedskog, L.; Pinho, C.M.; Filadi, R.; Rönnbäck, A.; Hertwig, L.; Wiehager, B.; Larssen, P.; Gellhaar, S.; Sandebring, A.; Westerlund, M.; et al. Modulation of the Endoplasmic Reticulum-Mitochondria Interface in Alzheimer’s Disease and Related Models. Proc. Natl. Acad. Sci. USA 2013, 110, 7916–7921. [Google Scholar] [CrossRef] [PubMed]
- Dhakal, S.; Macreadie, I. Potential Contributions of Trace Amines in Alzheimer’s Disease and Therapeutic Prospects. Neural Regen. Res. 2021, 16, 1394–1396. [Google Scholar] [CrossRef]
- Ilieva, H.; Polymenidou, M.; Cleveland, D.W. Non-Cell Autonomous Toxicity in Neurodegenerative Disorders: ALS and Beyond. J. Cell Biol. 2009, 187, 761–772. [Google Scholar] [CrossRef]
- Mancuso, R.; Navarro, X. Sigma-1 Receptor in Motoneuron Disease. Adv. Exp. Med. Biol. 2017, 964, 235–254. [Google Scholar] [CrossRef]
- Mavlyutov, T.A.; Baker, E.M.; Losenegger, T.M.; Kim, J.R.; Torres, B.; Epstein, M.L.; Ruoho, A.E. The Sigma-1 Receptor–A Therapeutic Target for the Treatment of ALS? BT-Sigma Receptors: Their Role in Disease and as Therapeutic Targets; Smith, S.B., Su, T.-P., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 255–265. ISBN 978-3-319-50174-1. [Google Scholar]
- Mavlyutov, T.A.; Epstein, M.L.; Verbny, Y.I.; Huerta, M.S.; Zaitoun, I.; Ziskind-Conhaim, L.; Ruoho, A.E. Lack of Sigma-1 Receptor Exacerbates ALS Progression in Mice. Neuroscience 2013, 240, 129–134. [Google Scholar] [CrossRef]
- Ono, Y.; Tanaka, H.; Takata, M.; Nagahara, Y.; Noda, Y.; Tsuruma, K.; Shimazawa, M.; Hozumi, I.; Hara, H. SA4503, a Sigma-1 Receptor Agonist, Suppresses Motor Neuron Damage in in Vitro and in Vivo Amyotrophic Lateral Sclerosis Models. Neurosci. Lett. 2014, 559, 174–178. [Google Scholar] [CrossRef]
- Shomrat, T.; Nesher, N. Updated View on the Relation of the Pineal Gland to Autism Spectrum Disorders. Front. Endocrinol. 2019, 10, 37. [Google Scholar] [CrossRef] [PubMed]
- Geoffray, M.-M.; Nicolas, A.; Speranza, M.; Georgieff, N. Are Circadian Rhythms New Pathways to Understand Autism Spectrum Disorder? J. Physiol. 2016, 110, 434–438. [Google Scholar] [CrossRef]
- Cohen, S.; Conduit, R.; Lockley, S.W.; Rajaratnam, S.M.W.; Cornish, K.M. The Relationship between Sleep and Behavior in Autism Spectrum Disorder (ASD): A Review. J. Neurodev. Disord. 2014, 6, 44. [Google Scholar] [CrossRef]
- Rossignol, D.A.; Frye, R.E. Melatonin in Autism Spectrum Disorders: A Systematic Review and Meta-Analysis. Dev. Med. Child Neurol. 2011, 53, 783–792. [Google Scholar] [CrossRef]
- Ly, C.; Greb, A.C.; Cameron, L.P.; Wong, J.M.; Barragan, E.V.; Wilson, P.C.; Burbach, K.F.; Soltanzadeh Zarandi, S.; Sood, A.; Paddy, M.R.; et al. Psychedelics Promote Structural and Functional Neural Plasticity. Cell Rep. 2018, 23, 3170–3182. [Google Scholar] [CrossRef]
- Phillips, M.; Pozzo-Miller, L. Dendritic Spine Dysgenesis in Autism Related Disorders. Neurosci. Lett. 2015, 601, 30–40. [Google Scholar] [CrossRef]
- Karantanos, T.; Corn, P.G.; Thompson, T.C. Prostate Cancer Progression after Androgen Deprivation Therapy: Mechanisms of Castrate Resistance and Novel Therapeutic Approaches. Oncogene 2013, 32, 5501–5511. [Google Scholar] [CrossRef]
- Amaral, T.M.S.; Macedo, D.; Fernandes, I.; Costa, L. Castration-Resistant Prostate Cancer: Mechanisms, Targets, and Treatment. Prostate Cancer 2012, 2012, 327253. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Wang, L.; Cheng, Z.; Pei, Z.; Zhang, Z.; Li, Z.; Zhang, X.; Yan, D.; Xia, Q.; Feng, Y.; et al. Molecular Changes of Lung Malignancy in HIV Infection. Sci. Rep. 2018, 8, 13128. [Google Scholar] [CrossRef] [PubMed]
- López-Torres, C.D.; Torres-Mena, J.E.; Castro-Gil, M.P.; Villa-Treviño, S.; Arellanes-Robledo, J.; del Pozo-Yauner, L.; Pérez-Carreón, J.I. Downregulation of Indolethylamine N-Methyltransferase Is an Early Event in the Rat Hepatocarcinogenesis and Is Associated with Poor Prognosis in Hepatocellular Carcinoma Patients. J. Gene Med. 2022, 24, e3439. [Google Scholar] [CrossRef]
- Sun, H.; Long, J.; Zuo, B.; Li, Y.; Song, Y.; Yu, M.; Xun, Z.; Wang, Y.; Wang, X.; Sang, X.; et al. Development and Validation of a Selenium Metabolism Regulators Associated Prognostic Model for Hepatocellular Carcinoma. BMC Cancer 2023, 23, 451. [Google Scholar] [CrossRef] [PubMed]
- Jianfeng, W.; Yutao, W.; Jianbin, B. Indolethylamine-N-Methyltransferase Inhibits Proliferation and Promotes Apoptosis of Human Prostate Cancer Cells: A Mechanistic Exploration. Front. Cell Dev. Biol. 2022, 10, 805402. [Google Scholar] [CrossRef]
- Parisi, M.A. Hirschsprung Disease Overview–Retired Chapter, For Historical Reference Only; Adam, M.P., Feldman, J., Mirzaa, G.M., Pagon, R.A., Wallace, S.E., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
- Kim, J.Y.; Seo, J.-M.; Kim, D.-Y.; Oh, J.-T.; Park, K.-W.; Kim, H.-Y.; Jung, K.; Park, B.L.; Kim, J.-H.; Shin, H.D. Potential Association of INMT Nonsynonymous Variant (His46Pro) with Hirschsprung’s Disease. Neonatology 2015, 108, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Mandel, L.R. Inhibition of Indoleamine-N-Methyltransferase by 2,3,4,6,7,8-Hexahydropyrrolo[1,2-a]Pyrimidine. Biochem. Pharmacol. 1976, 25, 2251–2256. [Google Scholar] [CrossRef]
- Borchardt, R.T.; Huber, J.A.; Wu, Y.S. Potential Inhibitors of S-Adenosylmethionine-Dependent Methyltransferases. 4. Further Modifications of the Amino Acid and Base Portions of S-Adenosyl-L-Homocysteine. J. Med. Chem. 1976, 19, 1094–1099. [Google Scholar] [CrossRef] [PubMed]
- Gomes, U.C.R.; Shanley, B.C. An endogenous inhibitor of indoleamine-N-methyltransferase in cerebrospinal fluid. Life Sci. 1978, 23, 697–704. [Google Scholar] [CrossRef]
- Hsiao, K.; Zegzouti, H.; Goueli, S.A. Methyltransferase-Glo: A Universal, Bioluminescent and Homogenous Assay for Monitoring All Classes of Methyltransferases. Epigenomics 2016, 8, 321–339. [Google Scholar] [CrossRef]
- Dong, G.; Yasgar, A.; Peterson, D.L.; Zakharov, A.; Talley, D.; Cheng, K.C.-C.; Jadhav, A.; Simeonov, A.; Huang, R. Optimization of High-Throughput Methyltransferase Assays for the Discovery of Small Molecule Inhibitors. ACS Comb. Sci. 2020, 22, 422–432. [Google Scholar] [CrossRef]




| Method | Assay | Description | Reference |
|---|---|---|---|
| M1 | Radiometric (in vitro) | 14C-SAM is used as the methyl donor and tryptamine as the substrate. Formation of 14C-methylated tryptamine (14C-NMT) is quantified by liquid scintillation counting. | [1] |
| M2 | Fluorometric | N-methylation of N-methylserotonin (NMS) to bufotenine. The resulting bufotenine was derivatized with O-phthalaldehyde (OPT). The fluorescent bufotenine-OPT adduct was then quantified using fluorescence detection. | [20] |
| M3 | Radiometric (in vivo) | Intravenous administration of radiolabeled 14C-NMT. After incubation, tissues and plasma were collected, and the radiolabeled product 14C-DMT was isolated, quantified by liquid scintillation (LS) counting, and confirmed using reverse isotope dilution analysis. | [93] |
| Compound ID | Scaffold/Class | Inhibitory Potency | Inhibition Type | Selectivity | Reference |
|---|---|---|---|---|---|
| DMT (1) | Tryptamine derivative | IC50 = 67 μM | Competitive/Non-competitive | Selective | [1,7] |
| SAH (2) | SAM analog | Ki = 10 µM | Competitive | Selective | [20] |
| PDAT (3) | Tryptamine derivative | Ki = 84 μM | Non-competitive | Selective | [7] |
| Salsolinol (4) | Isoquinoline | 97% inhibition at 0.1 mM | Competitive | N/A | [1] |
| Chloroquine (5) | Quinoline | 98% inhibition at 0.1 mM | Competitive | N/A | [1] |
| Harmalol (6) | β-Carboline | 89% inhibition at 1 mM | Competitive | N/A | [1] |
| Naratriptan (7) | Triptan | IC50 = 147 μM | Competitive | Non-selective | [1] |
| Sumatriptan (8) | Triptan | IC50 = 370 μM | Competitive | Non-selective | [1] |
| Zolmitriptan (9) | Triptan | IC50 = 483 μM | Competitive | Non-selective | [1] |
| 1,7-diaminoheptane (13) | Aliphatic diamine | 84.9% inhibition at 10 mM | Competitive | Non-selective | [23] |
| 1,8-diaminooctane (14) | Aliphatic diamine | 94.3% inhibition at 10 mM | Competitive | Non-selective | [23] |
| DBN (18) | Bicyclic amidine | IC50 = 20 μM | Non-competitive | Selective | [93] |
| 3,4,6,7,8,9-hexahydro-2H-pyrido [1,2-a]pyrimidine (19) | Bicyclic amidine | IC50 = 0.7 μM | Non-competitive | Selective | [22] |
| 2,3,5,6-tetrahydro-7H-pyrrolo [1,2-a]imidazole (20) | Bicyclic amidine | IC50 = 0.04 μM | Non-competitive | Selective | [22] |
| 2,3,5,6,7,8-hexahydroimidazo [1,2-a]pyridine (21) | Bicyclic amidine | IC50 = 0.17 μM | Non-competitive | Selective | [22] |
| 2,3,5,6-Tetrahydro-8H-imidazo [2,1-c][1,4]thiazine (22) | Bicyclic amidine | IC50 = 0.5 μM | Non-competitive | Selective | [22] |
| 3-methylthiazolidin-2-imine (23) | Monocyclic amidine | IC50 = 0.8 μM | Non-competitive | Selective | [22] |
| 3-ethylthiazolidin-2-imine (24) | Monocyclic amidine | IC50 = 0.2 μM | Non-competitive | Selective | [22] |
| 2-imino-N-methylthiazolidin-3-amine (25) | Monocyclic amidine | IC50 = 1.5 μM | Non-competitive | Selective | [22] |
| 3-methylthiazol-2(3H)-imine (26) | Monocyclic amidine | IC50 = 2 μM | Non-competitive | Selective | [22] |
| 1-methylpyrrolidin-2-imine (27) | Monocyclic amidine | IC50 = 1.5 μM | Non-competitive | Selective | [22] |
| 1-ethylpyrrolidin-2-imine (28) | Monocyclic amidine | IC50 = 1.6 μM | Non-competitive | Selective | [22] |
| 3-methyl-1,3-thiazinan-2-imine (29) | Monocyclic amidine | IC50 = 2 μM | Non-competitive | Selective | [22] |
| 3-ethyl-1,3-thiazinan-2-imine (30) | Monocyclic amidine | IC50 = 1.8 μM | Non-competitive | Selective | [22] |
| 3-methyl-3,6-dihydro-2H-1,3-thiazin-2-imine (31) | Monocyclic amidine | IC50 = 2 μM | Non-competitive | Selective | [22] |
| 1-methyl-5,6-dihydropyridin-2(1H)-imine (32) | Monocyclic amidine | IC50 = 0.9 μM | Non-competitive | Selective | [22] |
| N6-methyl-3-deazaadenosyl-L-homocysteine (34) | Homocysteine derivatives | N/A | Competitive | Selective | [94] |
| N6-methyladenosyl-L-homocysteine (35) | Homocysteine derivatives | IC50 = 17.4 μM | Competitive | Selective | [94] |
| Imipramine (36) | Tricyclic antidepressant | IC50 = 166 μM | Competitive | N/A | [1] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abouheif, S.; Awad, A.; McCurdy, C.R. Implications of Indolethylamine N-Methyltransferase (INMT) in Health and Disease: Biological Functions, Disease Associations, Inhibitors, and Analytical Approaches. Brain Sci. 2025, 15, 935. https://doi.org/10.3390/brainsci15090935
Abouheif S, Awad A, McCurdy CR. Implications of Indolethylamine N-Methyltransferase (INMT) in Health and Disease: Biological Functions, Disease Associations, Inhibitors, and Analytical Approaches. Brain Sciences. 2025; 15(9):935. https://doi.org/10.3390/brainsci15090935
Chicago/Turabian StyleAbouheif, Seif, Ahmed Awad, and Christopher R. McCurdy. 2025. "Implications of Indolethylamine N-Methyltransferase (INMT) in Health and Disease: Biological Functions, Disease Associations, Inhibitors, and Analytical Approaches" Brain Sciences 15, no. 9: 935. https://doi.org/10.3390/brainsci15090935
APA StyleAbouheif, S., Awad, A., & McCurdy, C. R. (2025). Implications of Indolethylamine N-Methyltransferase (INMT) in Health and Disease: Biological Functions, Disease Associations, Inhibitors, and Analytical Approaches. Brain Sciences, 15(9), 935. https://doi.org/10.3390/brainsci15090935







