Automated Detection of Epileptic Seizures in EEG Signals via Micro-Capsule Networks
Abstract
1. Introduction
2. Methods
2.1. EEG Preprocessing and Generation
2.1.1. Data Sets Description
2.1.2. Data Preprocessing
2.1.3. Data Matrix Generation
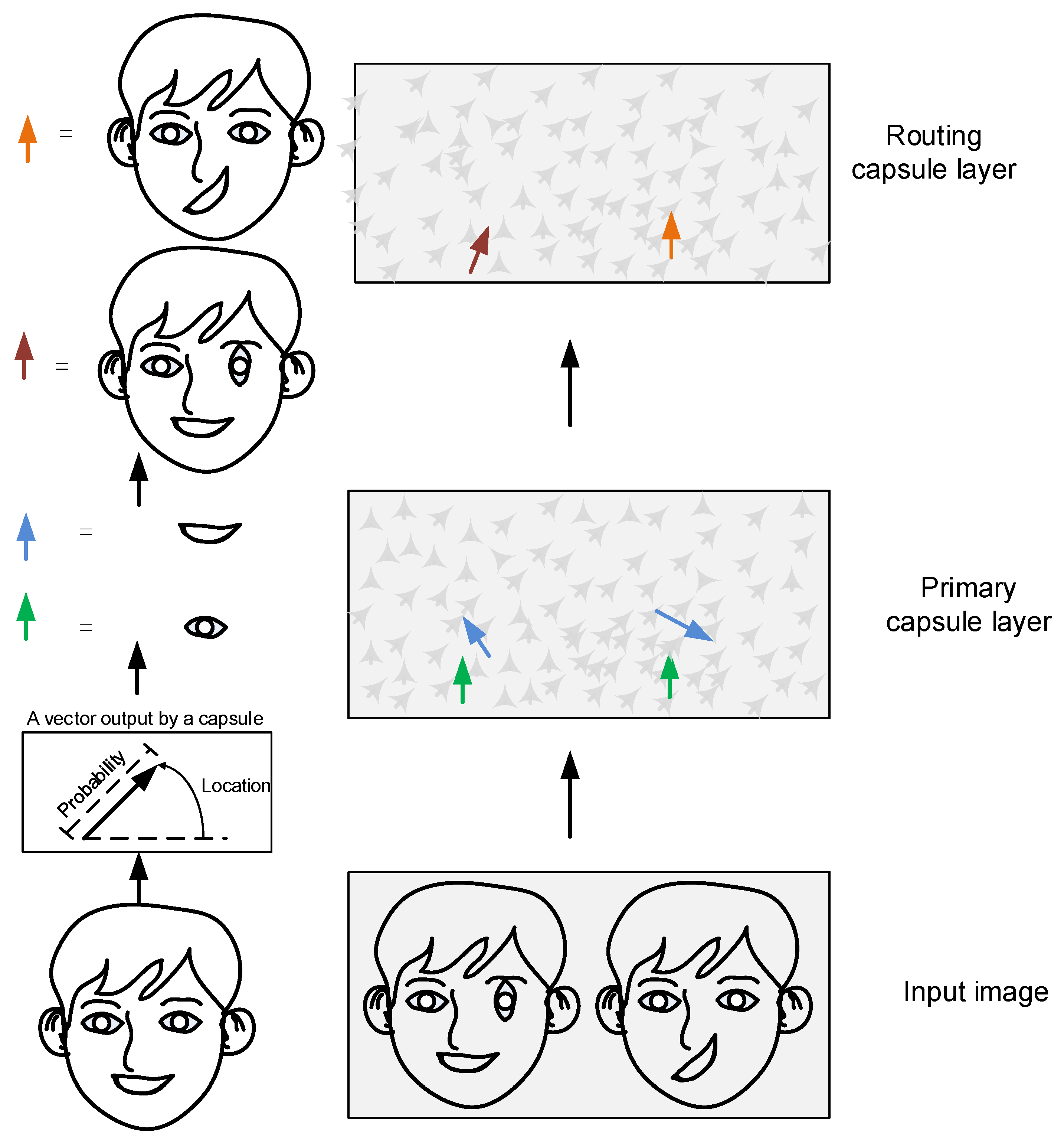
2.2. Construction of the Micro-Capsule Networks
2.2.1. The Micro-Capsule Network Model Establishment
2.2.2. Epilepsy Detection Based on the Micro-Capsule Network Model
2.2.3. Margin Loss of the Micro-Capsule Network Model
2.2.4. Integrated Optimization Algorithm of Micro-Capsule Networks
2.3. Evaluation Metrics
3. Results
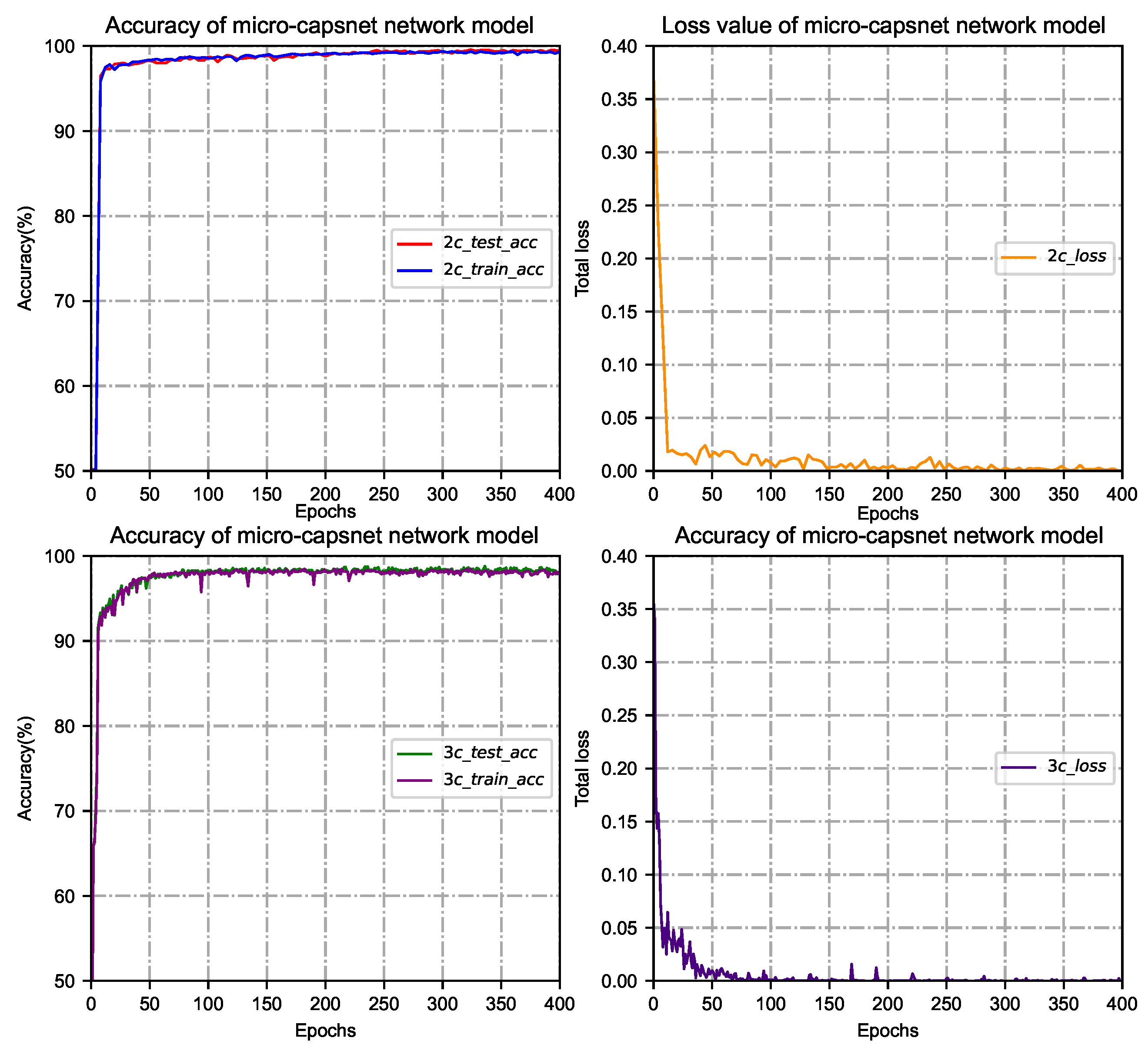
3.1. Effect of Iterations on the Micro-Capsule Networks
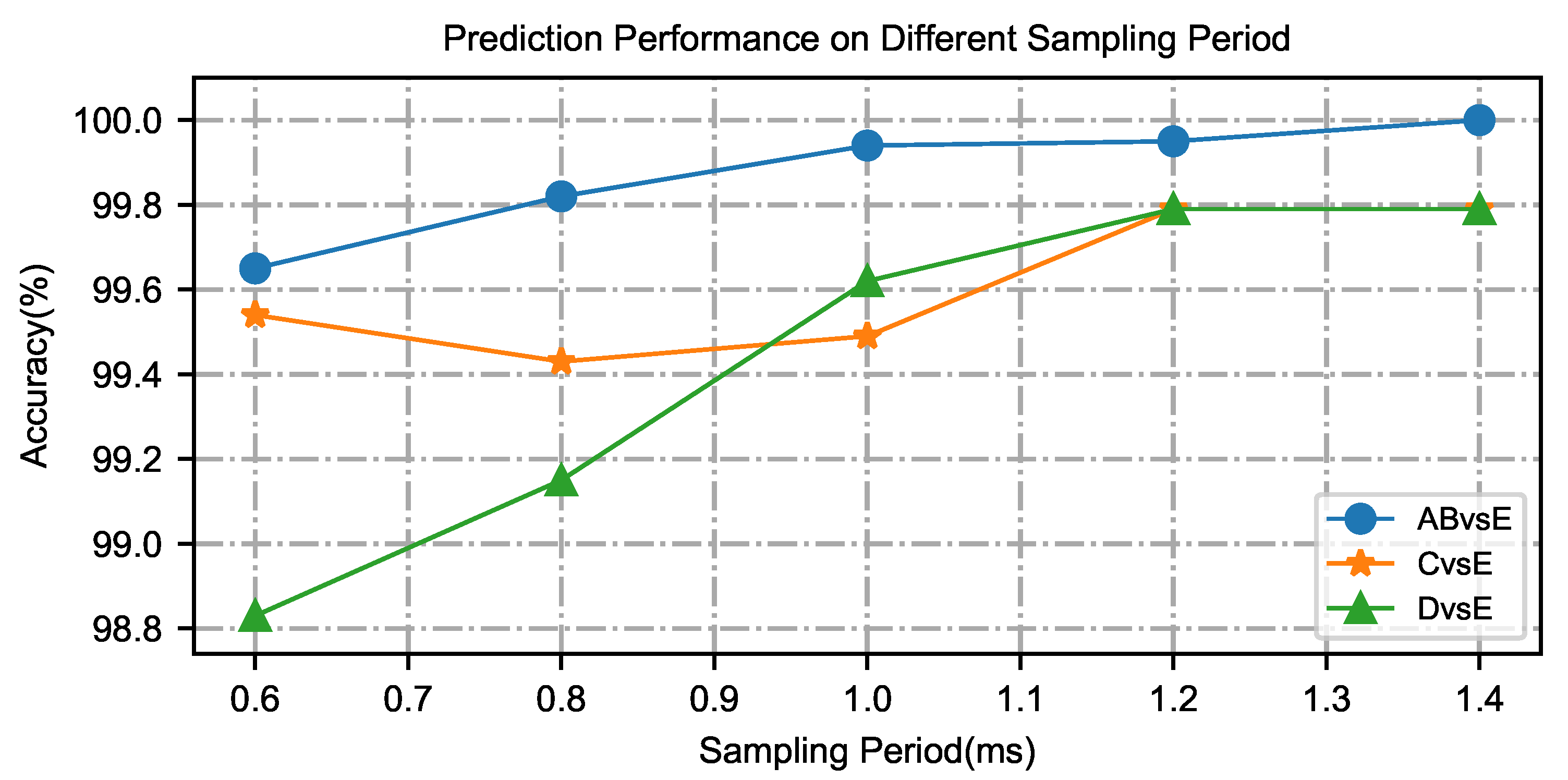
3.2. Influence of Time Interval on the Micro-Capsule Networks
3.3. Performance of the Micro-Capsule Network Model
4. Discussion
4.1. Advantages of the Micro-Capsule Network Model
4.2. Comparisons with Previous Methods
4.3. Limitations and Future Works
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organisation. Epilepsy, Website. 2025. Available online: https://www.who.int/news-room/fact-sheets/detail/epilepsy/ (accessed on 6 February 2025).
- Zheng, G.; Kong, H. Exploring the correlation between serum α-synuclein and abnormal electroencephalography patterns in children with epilepsy, as well as electroencephalographic discharge index. Int. J. Neurosci. 2024, 135, 779–786. [Google Scholar] [CrossRef]
- Cardeña, E.; Pick, S.; Litwin, R. Differentiating psychogenic nonepileptic from epileptic seizures: A mixed-methods, content analysis study. Epilepsy Behav. 2020, 109, 107121. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, Y.; Zhao, Y.; Zhang, Q.; Han, F. The neurovascular unit dysfunction in the molecular mechanisms of epileptogenesis and targeted therapy. Neurosci. Bull. 2024, 40, 621–634. [Google Scholar] [CrossRef]
- Kanner, A.M.; Bicchi, M.M. Antiseizure Medications for Adults with Epilepsy: A Review. JAMA 2022, 327, 1269–1281. [Google Scholar] [CrossRef] [PubMed]
- Woodbright, M.; Verma, B.; Haidar, A. Autonomous deep feature extraction based method for epileptic EEG brain seizure classification. Neurocomputing 2021, 444, 30–37. [Google Scholar] [CrossRef]
- Dutta, K.K.; Manohar, P.; Indira, K.; Naaz, F.; Lakshminarayan, M.; Rajagopalan, S. Seven Epileptic Seizure Type Classification in Pre-Ictal, Ictal and Inter-Ictal Stages using Machine Learning Techniques. Adv. Mach. Learn. Artif. Intell. 2023, 4, 1–10. [Google Scholar] [CrossRef]
- Wei, X.; Zhou, L.; Chen, Z.; Zhang, L.; Zhou, Y. Automatic seizure detection using three-dimensional CNN based on multi-channel EEG. BMC Med. Inform. Decis. Mak. 2018, 18, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Bagga, S.; Maheshkar, V.; Bhatia, M. Detection of Epileptic Seizures using EEG Signals. In Proceedings of the 2020 International Conference on Artificial Intelligence and Signal Processing (AISP), Amaravati, India, 10–12 January 2020; pp. 1–5. [Google Scholar] [CrossRef]
- Wong, S.; Simmons, A.; Rivera-Villicana, J.; Barnett, S.; Sivathamboo, S.; Perucca, P.; Kwan, P.; Kuhlmann, L.; Vasa, R.; O’Brien, T.J. EEG based automated seizure detection–A survey of medical professionals. Epilepsy Behav. 2023, 149, 109518. [Google Scholar] [CrossRef]
- Shoeibi, A.; Khodatars, M.; Ghassemi, N.; Jafari, M.; Moridian, P.; Alizadehsani, R.; Panahiazar, M.; Khozeimeh, F.; Zare, A.; Hosseini-Nejad, H.; et al. Epileptic seizures detection using deep learning techniques: A review. Int. J. Environ. Res. Public Health 2021, 18, 5780. [Google Scholar] [CrossRef]
- Duan, L.; Lian, Z.; Chen, J.; Qiao, Y.; Miao, J.; Li, M. Classification of epilepsy period based on combination feature extraction methods and spiking swarm intelligent optimization algorithm. Concurr. Comput. Pract. Exp. 2021, 33, e5550. [Google Scholar] [CrossRef]
- Moctezuma, L.A.; Molinas, M. EEG channel-selection method for epileptic-seizure classification based on multi-objective optimization. Front. Neurosci. 2020, 14, 593. [Google Scholar] [CrossRef]
- Omar, A.; Abd El-Hafeez, T. Optimizing epileptic seizure recognition performance with feature scaling and dropout layers. Neural Comput. Appl. 2024, 36, 2835–2852. [Google Scholar] [CrossRef]
- Gemein, L.A.; Schirrmeister, R.T.; Chrabąszcz, P.; Wilson, D.; Boedecker, J.; Schulze-Bonhage, A.; Hutter, F.; Ball, T. Machine-learning-based diagnostics of EEG pathology. NeuroImage 2020, 220, 117021. [Google Scholar] [CrossRef]
- Kim, K.; Duc, N.T.; Choi, M.; Lee, B. EEG microstate features for schizophrenia classification. PLoS ONE 2021, 16, e0251842. [Google Scholar] [CrossRef]
- Usman, S.M.; Khalid, S.; Aslam, M.H. Epileptic seizures prediction using deep learning techniques. IEEE Access 2020, 8, 39998–40007. [Google Scholar] [CrossRef]
- Bomela, W.; Wang, S.; Chou, C.A.; Li, J.S. Real-time inference and detection of disruptive EEG networks for epileptic seizures. Sci. Rep. 2020, 10, 8653. [Google Scholar] [CrossRef] [PubMed]
- Si, Y. Machine learning applications for electroencephalograph signals in epilepsy: A quick review. Acta Epileptol. 2020, 2, 5. [Google Scholar] [CrossRef]
- Hu, D.; Cao, J.; Lai, X.; Wang, Y.; Wang, S.; Ding, Y. Epileptic state classification by fusing hand-crafted and deep learning EEG features. IEEE Trans. Circuits Syst. II Express Briefs 2020, 68, 1542–1546. [Google Scholar] [CrossRef]
- Yang, Y.; Zhou, M.; Niu, Y.; Li, C.; Cao, R.; Wang, B.; Yan, P.; Ma, Y.; Xiang, J. Epileptic seizure prediction based on permutation entropy. Front. Comput. Neurosci. 2018, 12, 55. [Google Scholar] [CrossRef]
- Jana, R.; Mukherjee, I. Deep learning based efficient epileptic seizure prediction with EEG channel optimization. Biomed. Signal Process. Control 2021, 68, 102767. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, J.; Wang, B.; Dai, J.; Lian, J.; Ke, A.; Zhao, Y.; Zhou, J.; Wang, C. Motion direction prediction through spike timing based on micro Capsnet networks. Sci. China Technol. Sci. 2022, 65, 2763–2775. [Google Scholar] [CrossRef]
- Li, C.; Zhao, Y.; Song, R.; Liu, X.; Qian, R.; Chen, X. Patient-Specific Seizure Prediction From Electroencephalogram Signal via Multichannel Feedback Capsule Network. IEEE Trans. Cogn. Dev. Syst. 2023, 15, 1360–1370. [Google Scholar] [CrossRef]
- Kumari, N.; Anwar, S.; Bhattacharjee, V. Convolutional Neural Network-Based Visually Evoked EEG Classification Model on MindBigData. In Proceedings of Research and Applications in Artificial Intelligence; Springer: Singapore, 2021; pp. 233–241. [Google Scholar] [CrossRef]
- Liu, X.; Xie, Q.; Lv, J.; Huang, H.; Wang, W. P300 event-related potential detection using one-dimensional convolutional capsule networks. Expert Syst. Appl. 2021, 174, 114701. [Google Scholar] [CrossRef]
- Sabour, S.; Frosst, N.; Hinton, G.E. Dynamic routing between capsules. In Proceedings of the 31st Conference on Neural Information Processing Systems (NIPS 2017), Long Beach, CA, USA, 4–9 December 2017. [Google Scholar] [CrossRef]
- Ha, K.W.; Jeong, J.W. Motor imagery EEG classification using capsule networks. Sensors 2019, 19, 2854. [Google Scholar] [CrossRef] [PubMed]
- Toraman, S. Automatic recognition of preictal and interictal EEG signals using 1D-capsule networks. Comput. Electr. Eng. 2021, 91, 107033. [Google Scholar] [CrossRef]
- Wang, S.; Wang, G.; Pei, G.; Yan, T. An EEG-based approach for Parkinson’s disease diagnosis using capsule network. In Proceedings of the 2022 7th International Conference on Intelligent Computing and Signal Processing (ICSP), Xi’an, China, 15–17 April 2022; pp. 1641–1645. [Google Scholar] [CrossRef]
- Dan, J.; Pale, U.; Amirshahi, A.; Cappelletti, W.; Ingolfsson, T.M.; Wang, X.; Cossettini, A.; Bernini, A.; Benini, L.; Beniczky, S.; et al. SzCORE: Seizure Community Open-Source Research Evaluation framework for the validation of electroencephalography-based automated seizure detection algorithms. Epilepsia, 2024; Online ahead of print. [Google Scholar] [CrossRef]
- Andrzejak, R.G.; Lehnertz, K.; Mormann, F.; Rieke, C.; David, P.; Elger, C.E. Indications of nonlinear deterministic and finite-dimensional structures in time series of brain electrical activity: Dependence on recording region and brain state. Phys. Rev. E 2001, 64, 061907. [Google Scholar] [CrossRef]
- Emami, A.; Kunii, N.; Matsuo, T.; Shinozaki, T.; Kawai, K.; Takahashi, H. Seizure detection by convolutional neural network-based analysis of scalp electroencephalography plot images. NeuroImage Clin. 2019, 22, 101684. [Google Scholar] [CrossRef]
- Mazzia, V.; Salvetti, F.; Chiaberge, M. Efficient-capsnet: Capsule network with self-attention routing. Sci. Rep. 2021, 11, 14634. [Google Scholar] [CrossRef]
- Li, C.; Wang, B.; Zhang, S.; Liu, Y.; Song, R.; Cheng, J.; Chen, X. Emotion recognition from EEG based on multi-task learning with capsule network and attention mechanism. Comput. Biol. Med. 2022, 143, 105303. [Google Scholar] [CrossRef]
- Zhao, W.; Zhao, W.; Wang, W.; Jiang, X.; Zhang, X.; Peng, Y.; Zhang, B.; Zhang, G. A Novel Deep Neural Network for Robust Detection of Seizures Using EEG Signals. Comput. Math. Methods Med. 2020, 2020, 9689821. [Google Scholar] [CrossRef]
- Sharmila, A.; Geethanjali, P. DWT based detection of epileptic seizure from EEG signals using naive Bayes and k-NN classifiers. IEEE Access 2016, 4, 7716–7727. [Google Scholar] [CrossRef]
- Abbasi, M.U.; Rashad, A.; Basalamah, A.; Tariq, M. Detection of epilepsy seizures in neo-natal EEG using LSTM architecture. IEEE Access 2019, 7, 179074–179085. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Pachori, R.B.; Upadhyay, A.; Acharya, U.R. Tunable-Q wavelet transform based multiscale entropy measure for automated classification of epileptic EEG signals. Appl. Sci. 2017, 7, 385. [Google Scholar] [CrossRef]
- Zhang, T.; Chen, W.; Li, M. Fuzzy distribution entropy and its application in automated seizure detection technique. Biomed. Signal Process. Control 2018, 39, 360–377. [Google Scholar] [CrossRef]
- Behara, D.S.T.; Kumar, A.; Swami, P.; Panigrahi, B.K.; Gandhi, T.K. Detection of epileptic seizure patterns in EEG through fragmented feature extraction. In Proceedings of the 2016 3rd International Conference on Computing for Sustainable Global Development (INDIACom), New Delhi, India, 16–18 March 2016; pp. 2539–2542. Available online: https://ieeexplore.ieee.org/document/7724719 (accessed on 20 June 2023).
- Türk, Ö.; Özerdem, M.S. Epilepsy detection by using scalogram based convolutional neural network from EEG signals. Brain Sci. 2019, 9, 115. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.C.; Chen, C.J.; Chiang, C.T.; Wu, H.C.; Yang, R.C.; Ouyang, C.S. Classification Preictal and Interictal Stages via Integrating Interchannel and Time-Domain Analysis of EEG Features. Clin. EEG Neurosci. 2017, 48.2, 139–145. [Google Scholar] [CrossRef]
- Ma, M.; Cheng, Y.; Wei, X.; Chen, Z.; Zhou, Y. Research on epileptic EEG recognition based on improved residual networks of 1-D CNN and indRNN. BMC Med. Inform. Decis. Mak. 2021, 21, 100. [Google Scholar] [CrossRef] [PubMed]
- Billeci, L.; Tonacci, A.; Varanini, M.; Detti, P.; Vatti, G. Epileptic seizures prediction based on the combination of EEG and ECG for the application in a wearable device. In Proceedings of the 2019 IEEE 23rd International Symposium on Consumer Technologies (ISCT), Ancona, Italy, 19–21 June 2019. [Google Scholar] [CrossRef]
- Pachori, R.B.; Patidar, S. Epileptic seizure classification in EEG signals using second-order difference plot of intrinsic mode functions. Comput. Methods Programs Biomed 2014, 113, 494–502. [Google Scholar] [CrossRef]
- Wong, S.; Simmons, A.; Rivera-Villicana, J.; Barnett, S.; Sivathamboo, S.; Perucca, P.; Ge, Z.; Kwan, P.; Kuhlmann, L.; Vasa, R.; et al. EEG datasets for seizure detection and prediction—A review. Epilepsia Open 2023, 8, 252–267. [Google Scholar] [CrossRef]
- Chen, W.; Wang, Y.; Ren, Y.; Jiang, H.; Du, G.; Zhang, J.; Li, J. An automated detection of epileptic seizures EEG using CNN classifier based on feature fusion with high accuracy. BMC Med. Inform. Decis. Mak. 2023, 23, 96. [Google Scholar] [CrossRef] [PubMed]
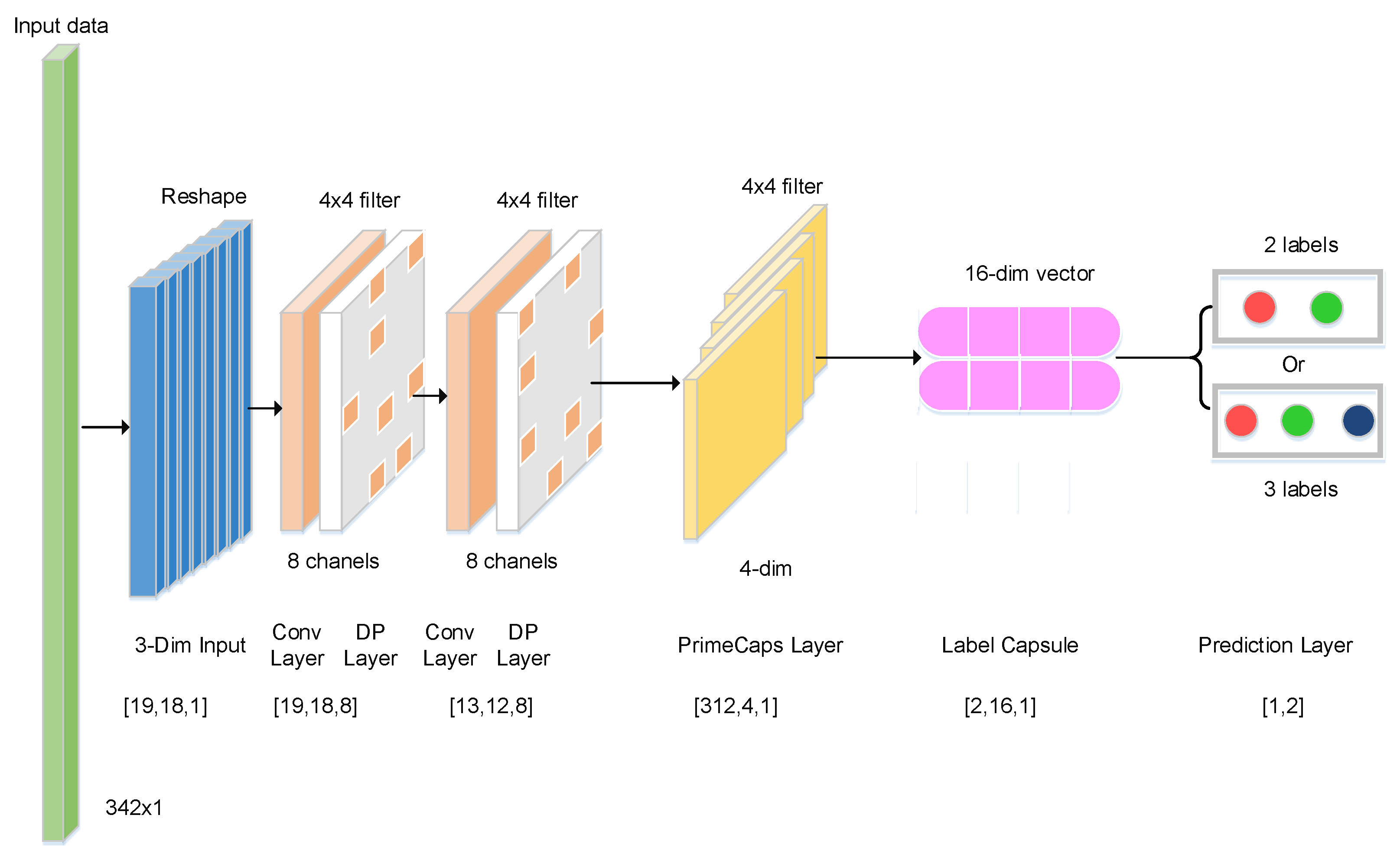

| Name | Input Shape | Output Shape | Convolution Kernel |
|---|---|---|---|
| Input | (1, 342) | (19, 18, 1) | - |
| Con-1 | (19, 18, 1) | (19, 18, 8) | (4, 4) |
| Con-2 | (19, 18, 8) | (13, 12, 8) | (4, 4) |
| Primary Capsule | (13, 12, 8) | (312, 4, 1) | (4, 4), (4, 4), (2, 4, 16) |
| Label Capsule | (312, 4, 1) | (2, 16, 1) | - |
| Output | (2, 16, 1) | (1, 2) | - |
| Data Category | Acc | Spec | Sen |
|---|---|---|---|
| AB vs. E | |||
| C vs. E | |||
| D vs. E | |||
| A vs. C vs. E |
| Data Category | Methods | Auther | Acc (%) | Our Acc (%) |
|---|---|---|---|---|
| AB vs. E | 1D - CNN | Zhao et al. [36] | ||
| DWT + KNN | Sharmila et al. [37] | 99.16% | ||
| LSTM | Abbasi et al. [38] | 99.17% | ||
| C vs. E | 1D - CNN | Zhao et al. [36] | ||
| TQWT + SVM | Bhattacharyya et al. [39] | 99.50% | ||
| WPD + KNN | Zhang et al. [40] | 99.36% | ||
| D vs. E | 1D - CNN | Zhao et al. [36] | ||
| CNN + SVM | Bhattacharyya et al. [39] | 98.00% | ||
| WPD + KNN | Zhang et al. [40] | 98.36% | ||
| A vs. C vs. E | 1D - CNN | Zhao et al. [36] | ||
| LSSVM | Behara et al. [41] | 97.19% | ||
| CWT + CNN | Türk and Ömer [42] | 97.00% | ||
| LSTM | Abbasi et al. [38] | 97.00% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, B.; Zhou, J.; Zhang, H.; Zhou, J.; Wang, C. Automated Detection of Epileptic Seizures in EEG Signals via Micro-Capsule Networks. Brain Sci. 2025, 15, 842. https://doi.org/10.3390/brainsci15080842
Wang B, Zhou J, Zhang H, Zhou J, Wang C. Automated Detection of Epileptic Seizures in EEG Signals via Micro-Capsule Networks. Brain Sciences. 2025; 15(8):842. https://doi.org/10.3390/brainsci15080842
Chicago/Turabian StyleWang, Baozeng, Jiayue Zhou, Hualiang Zhang, Jin Zhou, and Changyong Wang. 2025. "Automated Detection of Epileptic Seizures in EEG Signals via Micro-Capsule Networks" Brain Sciences 15, no. 8: 842. https://doi.org/10.3390/brainsci15080842
APA StyleWang, B., Zhou, J., Zhang, H., Zhou, J., & Wang, C. (2025). Automated Detection of Epileptic Seizures in EEG Signals via Micro-Capsule Networks. Brain Sciences, 15(8), 842. https://doi.org/10.3390/brainsci15080842






