How COVID-19 Affected Sleep Talking Episodes, Sleep and Dreams?
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.1.1. Online Survey
- Participants who self-reported frequent STs, as indicated by the MUPS item related to STs (“How often do you experience the following behaviors? talking during sleep”) ranging from 5 (“Sometimes—one or more times per month) to 7 (“Very frequently—every or nearly every night”);
- Age range: 18–35 years;
- Absence of medical conditions and psychiatric disorders (this was investigated with a specific question in the questionnaire on case history: “Do you have acute or chronic health problems (medical or psychological problems? (If yes, please specify in ‘other’)”);
- Absence of other sleep disorders except ST;
- No drug or alcohol abuse.
2.1.2. Home Monitoring
2.2. Procedure
2.3. Measures
2.3.1. Sleep Diaries
- Sleep onset latency (SOL): the time (in minutes) it takes to fall asleep after turning off the light;
- Number of nocturnal awakenings (NOA);
- Intra-night wakefulness (ISW): subjective duration of wakefulness (in minutes) from falling asleep to the final awakening;
- Total sleep time in minutes (TST): the amount of time spent asleep;
- Total bed time (TBT): the amount of time from lights off to the final awakening;
- Sleep efficiency (SE = TST/TBT × 100).
- Sleep depth (1 = very light sleep, 5 = very deep sleep)
- Sleep quiet (1 = very disturbed sleep, 5 = very quiet sleep)
- Sleep restless (1 = very low-rest sleep, 5 = very high-rest sleep)
2.3.2. Dream Reports
- Emotional intensity—EL: 1 = very low emotional intensity; 6 = very high emotional intensity;
- Vividness—VV: 1 = no image, just thinking about objects; 2 = very vague; 3 = less vague; 4 = moderately clear and vivid; 5 = clear and reasonably vivid; 6 = clear and vivid as normal vision;
- Bizarreness—B: 1 = scenes/thoughts closely related to everyday life, something that belongs to the individual’s life or to current reality, is plausible; 2 = belongs to reality but not to the life of the individual, seems unusual, strange, or illogical; 3 = plot discontinuous, changes in setting, slightly inappropriate roles; 4 = plot highly discontinuous, unlikely elements and particular settings appear, inappropriate roles; 5 = abundance of unlikely elements, settings with imaginary elements, metamorphoses, imaginary characters; 6 = impossible settings, fantastic elements and characters (with unlikely characteristics), illogicality of the plot.
2.3.3. Sleep Talking Episodes
2.4. Statistical Analyses
3. Results
3.1. Preliminary Questionnaire
3.2. Home Monitoring
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jahrami, H.; BaHammam, A.S.; Bragazzi, N.L.; Saif, Z.; Faris, M.; Vitiello, M.V. Sleep Problems during the COVID-19 Pandemic by Population: A Systematic Review and Meta-Analysis. J. Clin. Sleep Med. 2021, 17, 299–313. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, R.P. COVID-19 and Mental Health: A Review of the Existing Literature. Asian J. Psychiatr. 2020, 52, 102066. [Google Scholar] [CrossRef] [PubMed]
- Salfi, F.; Amicucci, G.; Cascioli, J.; Corigliano, D.; Viselli, L.; Tempesta, D.; Ferrara, M. The Impact of Home Confinement Due to COVID-19 Pandemic on Sleep Quality and Insomnia Symptoms among the Italian Population. 2020. Available online: https://www.researchgate.net/profile/Federico-Salfi/publication/344337922_The_impact_of_home_confinement_due_to_COVID-19_pandemic_on_sleep_quality_and_insomnia_symptoms_among_the_Italian_population/links/5f69c99392851c14bc8e03c4/The-impact-of-home-confinement-due-to-COVID-19-pandemic-on-sleep-quality-and-insomnia-symptoms-among-the-Italian-population.pdf (accessed on 15 September 2020).
- Cao, W.; Fang, Z.; Hou, G.; Han, M.; Xu, X.; Dong, J.; Zheng, J. The Psychological Impact of the COVID-19 Epidemic on College Students in China. Psychiatry Res. 2020, 287, 112934. [Google Scholar] [CrossRef] [PubMed]
- Casagrande, M.; Favieri, F.; Tambelli, R.; Forte, G. The Enemy Who Sealed the World: Effects Quarantine Due to the COVID-19 on Sleep Quality, Anxiety, and Psychological Distress in the Italian Population. Sleep Med. 2020, 75, 12–20. [Google Scholar] [CrossRef]
- Alfonsi, V.; Gorgoni, M.; Scarpelli, S.; Zivi, P.; Sdoia, S.; Mari, E.; Fraschetti, A.; Ferlazzo, F.; Giannini, A.M.; De Gennaro, L. COVID-19 Lockdown and Poor Sleep Quality: Not the Whole Story. J. Sleep Res. 2021, 30, e13368. [Google Scholar] [CrossRef] [PubMed]
- Mazza, C.; Ricci, E.; Colasanti, M.; Ferracuti, S.; Napoli, C.; Roma, P. A Nationwide Survey of Psychological Distress among Italian People during the COVID-19 Pandemic: Immediate Psychological Responses and Associated Factors. Int. J. Environ. Res. Public Health 2020, 17, 3165. [Google Scholar] [CrossRef]
- Moccia, L.; Janiri, D.; Pepe, M.; Dattoli, L.; Molinaro, M.; De Martin, V.; Chieffo, D.; Janiri, L.; Fiorillo, A.; Sani, G.; et al. Affective Temperament, Attachment Style, and the Psychological Impact of the COVID-19 Outbreak: An Early Report on the Italian General Population. Brain. Behav. Immun. 2020, 87, 75–79. [Google Scholar] [CrossRef]
- Salfi, F.; Lauriola, M.; Amicucci, G.; Corigliano, D.; Viselli, L.; Tempesta, D.; Ferrara, M. Gender-Related Time Course of Sleep Disturbances and Psychological Symptoms during the COVID-19 Lockdown: A Longitudinal Study on the Italian Population. Neurobiol. Stress 2020, 13, 100259. [Google Scholar] [CrossRef]
- Ballesio, A.; Zagaria, A.; Musetti, A.; Lenzo, V.; Palagini, L.; Quattropani, M.C.; Vegni, E.; Bonazza, F.; Filosa, M.; Manari, T.; et al. Longitudinal Associations between Stress and Sleep Disturbances during COVID-19. Stress Health 2022, 38, 919–926. [Google Scholar] [CrossRef]
- Holzinger, B.; Mayer, L.; Nierwetberg, F.; Klösch, G. COVID-19 Lockdown—Are Austrians Finally Able to Compensate Their Sleep Debt? Sleep Med. X 2021, 3, 100032. [Google Scholar] [CrossRef]
- Cellini, N.; Canale, N.; Mioni, G.; Costa, S. Changes in Sleep Pattern, Sense of Time and Digital Media Use during COVID-19 Lockdown in Italy. J. Sleep Res. 2020, 29, e13074. [Google Scholar] [CrossRef] [PubMed]
- Kocevska, D.; Blanken, T.F.; Van Someren, E.J.W.; Rösler, L. Sleep Quality during the COVID-19 Pandemic: Not One Size Fits All. Sleep Med. 2020, 76, 86–88. [Google Scholar] [CrossRef] [PubMed]
- Wright, K.P.; Linton, S.K.; Withrow, D.; Casiraghi, L.; Lanza, S.M.; de la Iglesia, H.; Vetter, C.; Depner, C.M. Sleep in University Students Prior to and during COVID-19 Stay-at-Home Orders. Curr. Biol. 2020, 30, R797–R798. [Google Scholar] [CrossRef] [PubMed]
- Salfi, F.; Amicucci, G.; Corigliano, D.; D’Atri, A.; Viselli, L.; Tempesta, D.; Ferrara, M. Changes of Evening Exposure to Electronic Devices during the COVID-19 Lockdown Affect the Time Course of Sleep Disturbances. Sleep 2021, 44, zsab080. [Google Scholar] [CrossRef]
- Salfi, F.; D’Atri, A.; Tempesta, D.; Ferrara, M. Sleeping under the Waves: A Longitudinal Study across the Contagion Peaks of the COVID-19 Pandemic in Italy. J. Sleep Res. 2021, 30, e13313. [Google Scholar] [CrossRef] [PubMed]
- Massar, S.A.A.; Ng, A.S.C.; Soon, C.S.; Ong, J.L.; Chua, X.Y.; Chee, N.I.Y.N.; Lee, T.S.; Chee, M.W.L. Reopening after Lockdown: The Influence of Working-from-Home and Digital Device Use on Sleep, Physical Activity, and Wellbeing Following COVID-19 Lockdown and Reopening. Sleep 2022, 45, zsab250. [Google Scholar] [CrossRef] [PubMed]
- Gorgoni, M.; Scarpelli, S.; Alfonsi, V.; De Gennaro, L. Dreaming during the COVID-19 Pandemic: A Narrative Review. Neurosci. Biobehav. Rev. 2022, 138, 104710. [Google Scholar] [CrossRef] [PubMed]
- Fränkl, E.; Scarpelli, S.; Nadorff, M.R.; Bjorvatn, B.; Bolstad, C.J.; Chan, N.Y.; Chung, F.; Dauvilliers, Y.; Espie, C.A.; Inoue, Y.; et al. How Our Dreams Changed during the COVID-19 Pandemic: Effects and Correlates of Dream Recall Frequency—A Multinational Study on 19,355 Adults. Nat. Sci. Sleep 2021, 13, 1573–1591. [Google Scholar] [CrossRef] [PubMed]
- Schredl, M.; Bulkeley, K. Dreaming and the COVID-19 Pandemic: A Survey in a U.S. Sample. Dreaming 2020, 30, 189–198. [Google Scholar] [CrossRef]
- Scarpelli, S.; Alfonsi, V.; Mangiaruga, A.; Musetti, A.; Quattropani, M.C.; Lenzo, V.; Freda, M.F.; Lemmo, D.; Vegni, E.; Borghi, L.; et al. Pandemic Nightmares: Effects on Dream Activity of the COVID-19 Lockdown in Italy. J. Sleep Res. 2021, 30, e13300. [Google Scholar] [CrossRef]
- Solomonova, E.; Picard-Deland, C.; Rapoport, I.L.; Pennestri, M.H.; Saad, M.; Kendzerska, T.; Veissiere, S.P.L.; Godbout, R.; Edwards, J.D.; Quilty, L.; et al. Stuck in a Lockdown: Dreams, Bad Dreams, Nightmares, and Their Relationship to Stress, Depression and Anxiety during the COVID-19 Pandemic. PLoS ONE 2021, 16, e0259040. [Google Scholar] [CrossRef]
- Scarpelli, S.; Nadorff, M.R.; Bjorvatn, B.; Chung, F.; Dauvilliers, Y.; Espie, C.A.; Inoue, Y.; Matsui, K.; Merikanto, I.; Morin, C.M.; et al. Nightmares in People with COVID-19: Did Coronavirus Infect Our Dreams? Nat. Sci. Sleep 2022, 14, 93–108. [Google Scholar] [CrossRef]
- Gorgoni, M.; Scarpelli, S.; Alfonsi, V.; Annarumma, L.; Cordone, S.; Stravolo, S.; De Gennaro, L. Pandemic Dreams: Quantitative and Qualitative Features of the Oneiric Activity during the Lockdown Due to COVID-19 in Italy. Sleep Med. 2021, 81, 20–32. [Google Scholar] [CrossRef]
- Scarpelli, S.; Alfonsi, V.; Gorgoni, M.; Musetti, A.; Filosa, M.; Quattropani, M.C.; Lenzo, V.; Vegni, E.; Borghi, L.; Margherita, G.; et al. Dreams and Nightmares during the First and Second Wave of the COVID-19 Infection: A Longitudinal Study. Brain Sci. 2021, 11, 1375. [Google Scholar] [CrossRef]
- Scarpelli, S.; Alfonsi, V.; Camaioni, M.; Gorgoni, M.; Albano, A.; Musetti, A.; Quattropani, M.C.; Plazzi, G.; De Gennaro, L.; Franceschini, C. Longitudinal Findings on the Oneiric Activity Changes Across the Pandemic. Nat. Sci. Sleep 2023, 15, 435–447. [Google Scholar] [CrossRef]
- Remedios, A.; Marin-Dragu, S.; Routledge, F.; Hamm, S.; Iyer, R.S.; Orr, M.; Meier, S.; Michael, S. Nightmare Frequency and Nightmare Distress during the COVID-19 Pandemic. J. Clin. Sleep Med. 2023, 19, 163–169. [Google Scholar] [CrossRef]
- Pesonen, A.K.; Lipsanen, J.; Halonen, R.; Elovainio, M.; Sandman, N.; Mäkelä, J.M.; Antila, M.; Béchard, D.; Ollila, H.M.; Kuula, L. Pandemic Dreams: Network Analysis of Dream Content During the COVID-19 Lockdown. Front. Psychol. 2020, 11, 573961. [Google Scholar] [CrossRef]
- Montplasir, J.; Zadra, A.; Nielsen, T.; Petit, D. Basic Science, Technical Considerations and Clinical Aspects. In Sleep Disorders Medicine, 4th ed.; Springer: Berlin/Heidelberg, Germany, 2017; pp. 1–1269. [Google Scholar] [CrossRef]
- Liu, Y.; Partinen, E.; Chan, N.Y.; Dauvilliers, Y.; Inoue, Y.; De Gennaro, L.; Plazzi, G.; Bolstad, C.J.; Nadorff, M.R.; Merikanto, I.; et al. Dream-Enactment Behaviours during the COVID-19 Pandemic: An International COVID-19 Sleep Study. J. Sleep Res. 2023, 32, e13613. [Google Scholar] [CrossRef]
- Koulack, D.; Goodenough, D.R. Dream Recall and Dream Recall Failure: An Arousal-Retrieval Model. Psychol. Bull. 1976, 83, 975–984. [Google Scholar] [CrossRef]
- van Wyk, M.; Solms, M.; Lipinska, G. Increased Awakenings from Non-Rapid Eye Movement Sleep Explain Differences in Dream Recall Frequency in Healthy Individuals. Front. Hum. Neurosci. 2019, 13, 370. [Google Scholar] [CrossRef]
- Alfonsi, V.; D’Atri, A.; Scarpelli, S.; Mangiaruga, A.; De Gennaro, L. Sleep Talking: A Viable Access to Mental Processes during Sleep. Sleep Med. Rev. 2019, 44, 12–22. [Google Scholar] [CrossRef]
- Mangiaruga, A.; D’Atri, A.; Scarpelli, S.; Alfonsi, V.; Camaioni, M.; Annarumma, L.; Gorgoni, M.; Pazzaglia, M.; De Gennaro, L. Sleep Talking versus Sleep Moaning: Electrophysiological Patterns Preceding Linguistic Vocalizations during Sleep. Sleep 2022, 45, zsab284. [Google Scholar] [CrossRef]
- Camaioni, M.; Scarpelli, S.; Alfonsi, V.; Gorgoni, M.; De Bartolo, M.; Calzolari, R.; De Gennaro, L. The Influence of Sleep Talking on Nocturnal Sleep and Sleep-Dependent Cognitive Processes. J. Clin. Med. 2022, 11, 6489. [Google Scholar] [CrossRef]
- Hublin, C.; Kaprio, J.; Partinen, M.; Koskenvuo, M. Parasomnias: Co-Occurrence and Genetics. Psychiatr. Genet. 2001, 11, 65–70. [Google Scholar] [CrossRef]
- Klackenberg, G. Incidence of Parasomnias in Children in a General Population. In Sleep and Its Disorders in Children; Guilleminault, C., Ed.; Raven Press: New York, NY, USA, 1987. [Google Scholar]
- Curcio, G.; Tempesta, D.; Scarlata, S.; Marzano, C.; Moroni, F.; Rossini, P.M.; Ferrara, M.; De Gennaro, L. Validity of the Italian Version of the Pittsburgh Sleep Quality Index (PSQI). Neurol. Sci. 2013, 34, 511–519. [Google Scholar] [CrossRef]
- Fulda, S.; Hornyak, M.; Müller, K.; Cerny, L.; Beitinger, P.A.; Wetter, T.C. Development and Validation of the Munich Parasomnia Screening (MUPS): A Questionnaire for Parasomnias and Nocturnal Behaviors. Somnologie 2008, 12, 56–65. [Google Scholar] [CrossRef]
- De Gennaro, L.; Ferrara, M.; Cristiani, R.; Curcio, G.; Martiradonna, V.; Bertini, M. Alexithymia and Dream Recall upon Spontaneous Morning Awakening. Psychosom. Med. 2003, 65, 301–306. [Google Scholar] [CrossRef]
- Casagrande, M.; Cortini, P. Spoken and Written Dream Communication: Differences and Methodological Aspects. Conscious. Cogn. 2008, 17, 145–158. [Google Scholar] [CrossRef]
- De Gennaro, L.; Cipolli, C.; Cherubini, A.; Assogna, F.; Cacciari, C.; Marzano, C.; Curcio, G.; Ferrara, M.; Caltagirone, C.; Spalletta, G. Amygdala and Hippocampus Volumetry and Diffusivity in Relation to Dreaming. Hum. Brain Mapp. 2010, 32, 1458–1470. [Google Scholar] [CrossRef]
- Lehmann, S.; Skogen, J.C.; Haug, E.; Mæland, S.; Fadnes, L.T.; Sandal, G.M.; Hysing, M.; Bjørknes, R. Perceived Consequences and Worries among Youth in Norway during the COVID-19 Pandemic Lockdown. Scand. J. Public Health 2021, 49, 755–765. [Google Scholar] [CrossRef]
- Ohayon, M.M.; Morselli, P.L.; Guilleminault, C. Prevalence of Nightmares and Their Relationship to Psychopathology and Daytime Functioning in Insomnia Subjects. Sleep 1997, 20, 340–348. [Google Scholar] [CrossRef]
- Berlin, R.M.; Litovitz, G.L.; Diaz, M.A.; Ahmed, S.W. Sleep Disorders on a Psychiatric Consultation Service. Am. J. Psychiatry 1984, 141, 582–584. [Google Scholar] [CrossRef]
- Cernovsky, Z.Z.; Paitich, D.; Crawford, G. MMPI and Nightmare Reports in Women Addicted to Alcohol and Other Drugs. Percept. Mot. Skills 1986, 62, 717–718. [Google Scholar] [CrossRef]
- Munezawa, T.; Kaneita, Y.; Osaki, Y.; Kanda, H.; Ohtsu, T.; Suzuki, H.; Minowa, M.; Suzuki, K.; Higuchi, S.; Mori, J.; et al. Nightmare and Sleep Paralysis among Japanese Adolescents: A Nationwide Representative Survey. Sleep Med. 2011, 12, 56–64. [Google Scholar] [CrossRef]
- Tanskanen, A.; Tuomilehto, J.; Viinamäki, H.; Vartiainen, E.; Lehtonen, J.; Puska, P. Nightmares as Predictors of Suicide. Sleep 2001, 24, 845–848. [Google Scholar] [CrossRef][Green Version]
- Kales, A.; Soldatos, C.R.; Caldwell, A.B.; Charney, D.S.; Kales, J.D.; Markel, D.; Cadieux, R. Nightmares: Clinical Characteristics and Personality Patterns. Am. J. Psychiatry 1980, 137, 1197–1201. [Google Scholar] [CrossRef]
- Kramer, M.; Schoen, L.S.; Kinney, L. Psychological and Behavioral Features of Disturbed Dreamers. Psychiatr. J. Univ. Ott. 1984, 3, 102–106. [Google Scholar]
- Berquier, A.; Ashton, R. Characteristics of the Frequent Nightmare Sufferer. J. Abnorm. Psychol. 1992, 101, 246–250. [Google Scholar] [CrossRef]
- Husni, M.; Cernovsky, Z.Z.; Koye, N.; Haggarty, J. Nightmares of Refugees from Kurdistan. J. Nerv. Ment. Dis. 2001, 189, 557–559. [Google Scholar] [CrossRef]
- Takeuchi, T.; Fukuda, K.; Sasaki, Y.; Inugami, M.; Murphy, T.I. Factors Related to the Occurrence of Isolated Sleep Paralysis Elicited during a Multi-Phasic Sleep-Wake Schedule. Sleep 2002, 25, 89–96. [Google Scholar] [CrossRef]
- Hinton, D.E.; Pich, V.; Chhean, D.; Pollack, M.H.; McNally, R.J. Sleep Paralysis among Cambodian Refugees: Association with PTSD Diagnosis and Severity. Depress. Anxiety 2005, 22, 47–51. [Google Scholar] [CrossRef]
- Ohayon, M.M.; Shapiro, C.M. Sleep Disturbances and Psychiatric Disorders Associated with Posttraumatic Stress Disorder in the General Population. Compr. Psychiatry 2000, 7, 628–637. [Google Scholar] [CrossRef]
- Yeung, A.; Xu, Y.; Chang, D.F. Prevalence and Illness Beliefs of Sleep Paralysis among Chinese Psychiatric Patients in China and the United States. Transcult. Psychiatry 2005, 42, 135–145. [Google Scholar] [CrossRef]
- Castelnovo, A.; Lopez, R.; Proserpio, P.; Nobili, L.; Dauvilliers, Y. NREM Sleep Parasomnias as Disorders of Sleep-State Dissociation. Nat. Rev. Neurol. 2018, 14, 470–481. [Google Scholar] [CrossRef]
- Morin, C.M.; Bjorvatn, B.; Chung, F.; Holzinger, B.; Partinen, M.; Penzel, T.; Ivers, H.; Wing, Y.K.; Chan, N.Y.; Merikanto, I.; et al. Insomnia, Anxiety, and Depression during the COVID-19 Pandemic: An International Collaborative Study. Sleep Med. 2021, 87, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Salari, N.; Hosseinian-Far, A.; Jalali, R.; Vaisi-Raygani, A.; Rasoulpoor, S.; Mohammadi, M.; Rasoulpoor, S.; Khaledi-Paveh, B. Prevalence of Stress, Anxiety, Depression among the General Population during the COVID-19 Pandemic: A Systematic Review and Meta-Analysis. Global. Health 2020, 16, 57. [Google Scholar] [CrossRef] [PubMed]
- Barone, D.A.; Henchcliffe, C. Rapid Eye Movement Sleep Behavior Disorder and the Link to Alpha-Synucleinopathies. Clin. Neurophysiol. 2018, 129, 1551–1564. [Google Scholar] [CrossRef]
- Scarpelli, S.; Alfonsi, V.; Gorgoni, M.; De Gennaro, L. What about Dreams? State of the Art and Open Questions. J. Sleep Res. 2022, 31, e13609. [Google Scholar] [CrossRef] [PubMed]
- Scarpelli, S.; Bartolacci, C.; D’Atri, A.; Gorgoni, M.; De Gennaro, L. The Functional Role of Dreaming in Emotional Processes. Front. Psychol. 2019, 10, 459. [Google Scholar] [CrossRef]
- Rechtschaffen, A.; Goodenough, D.R.; Shapiro, A. Patterns of Sleep Talking. Arch. Gen. Psychiatry 1962, 7, 418–426. [Google Scholar] [CrossRef]
- Castelnovo, A.; Loddo, G.; Provini, F.; Miano, S.; Manconi, M. Mental Activity during Episodes of Sleepwalking, Night Terrors or Confusional Arousals: Differences between Children and Adults. Nat. Sci. Sleep 2021, 13, 829–840. [Google Scholar] [CrossRef] [PubMed]
- Fasiello, E.; Scarpelli, S.; Gorgoni, M.; Alfonsi, V.; Galbiati, A.; De Gennaro, L. A Systematic Review of Dreams and Nightmares Recall in Patients with Rapid Eye Movement Sleep Behaviour Disorder. J. Sleep Res. 2023, 32, e13768. [Google Scholar] [CrossRef] [PubMed]
- Uguccioni, G.; Golmard, J.-L.; de Fontreaux, A.N.; Leu-Semenescu, S.; Brion, A.; Arnulf, I. Fight or Flight? Dream Content during Sleepwalking/Sleep Terrors vs. Rapid Eye Movement Sleep Behavior Disorder. Sleep Med. 2013, 14, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Antrobus, J. Dreaming: Cognitive Processes During Cortical Activation and High Afferent Thresholds. Psychol. Rev. 1991, 98, 96–121. [Google Scholar] [CrossRef] [PubMed]
- Casagrande, M.; Violani, C.; Lucidi, F.; Buttinelli, E.; Bertini, M. Variations in Sleep Mentation as a Function of Time of Night. Int. J. Neurosci. 1996, 85, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Foulkes, D. Nonrapid Eye Movement Mentation. Exp. Neurol. 1967, 19, 28–38. [Google Scholar] [CrossRef] [PubMed]
- Rechtschaffen, A.; Verdone, P.; Wheaton, J. Reports of mental activity during sleep. Can. Psychiatr. Assoc. J. 1963, 8, 409–414. [Google Scholar] [CrossRef] [PubMed]
- MacNeilage, P.F. Motor Control of Serial Ordering of Speech. Psychol. Rev. 1970, 77, 182–196. [Google Scholar] [CrossRef]
- Arkin, A.M.; Toth, F.; Baker, J.; Hastey, J.M. The Frequency of Sleep Talking in the Laboratory among Chronic Sleep Talkers and Good Dream Recallers. J. Nerv. Ment. Dis. 1970, 151, 369–374. [Google Scholar] [CrossRef]

| Preliminary Questionnaires | ||||||||
|---|---|---|---|---|---|---|---|---|
| MUPS | Pandemic (%) | Pre-Pandemic (%) | Fisher’s Exact | p | ||||
| I | II | III | I | II | III | |||
| Hypnic jerks | 11.10 | 33.30 | 55.60 | 11.10 | 22.20 | 66.70 | 0.95 | 0.73 |
| Rhythmic foot movements | 59.30 | 18.50 | 22.20 | 40.70 | 14.80 | 44.40 | 3.02 | 0.28 |
| Rhythmic movement disorders | 92.60 | 7.40 | 0.00 | 81.50 | 11.10 | 7.40 | 2.08 | 0.50 |
| Exploding head syndrome | 74.10 | 14.80 | 11.10 | 85.20 | 7.40 | 7.40 | 1.13 | 0.62 |
| Hypnagogic hallucinations | 63.00 | 25.90 | 11.10 | 77.80 | 14.80 | 7.40 | 1.48 | 0.56 |
| Periodic leg movements | 81.50 | 11.10 | 7.40 | 70.40 | 18.50 | 11.10 | 0.99 | 0.65 |
| Nocturnal leg cramps | 55.60 | 37.00 | 7.40 | 59.30 | 29.60 | 11.10 | 0.54 | 0.85 |
| Sleep-related bruxism | 63.00 | 7.40 | 29.60 | 66.70 | 3.70 | 29.60 | 0.48 | 1.00 |
| Abnormal swallowing | 81.50 | 7.40 | 11.10 | 85.20 | 11.10 | 3.70 | 1.22 | 0.75 |
| Groaning | 48.10 | 14.80 | 37.00 | 55.60 | 18.50 | 25.90 | 0.83 | 0.76 |
| Sleep enuresis | 96.30 | 3.70 | 0.00 | 96.30 | 3.70 | 0.00 | 1.00 | |
| Nightmares | 3.70 | 37.00 | 59.30 | 3.70 | 48.10 | 48,10 | 0.95 | 0.79 |
| Sleep terrors | 70.40 | 14.80 | 14.80 | 51.90 | 29.60 | 18.50 | 2.19 | 0.35 |
| Nocturnal eating | 100.00 | 0.00 | 0.00 | 88.90 | 7.40 | 3.70 | 2.74 | 0.24 |
| Sleep-related eating | 92.60 | 7.40 | 0.00 | 96.30 | 3.70 | 0.00 | 1.00 | |
| Confusing arousals | 59.30 | 25.90 | 14.80 | 59.30 | 22.20 | 18.50 | 0.27 | 1.00 |
| Sleep paralysis | 85.20 | 7.40 | 7.40 | 88.90 | 7.40 | 3.70 | 0.56 | 1.00 |
| Sleepwalking | 88.90 | 11.10 | 0.00 | 77.80 | 14.80 | 7.40 | 2.02 | 0.39 |
| Violent behavior | 77.80 | 18.50 | 3.70 | 85.20 | 11.10 | 3.70 | 0.84 | 0.85 |
| RBD | 55.60 | 33.30 | 11.10 | 51.90 | 33.30 | 14.80 | 0.27 | 1.00 |
| Others | 88.90 | 7.40 | 3.70 | 66.70 | 18.50 | 14.80 | 3.71 | 0.18 |
| PSQI | Pandemic | Pre-Pandemic | t | p | ||||
| Global Score | 5.85 (0.55) | 6.04 (0.52) | −0.25 | 0.80 | ||||
| Variables | Pandemic Mean (sd) | Pre-Pandemic Mean (sd) | |
|---|---|---|---|
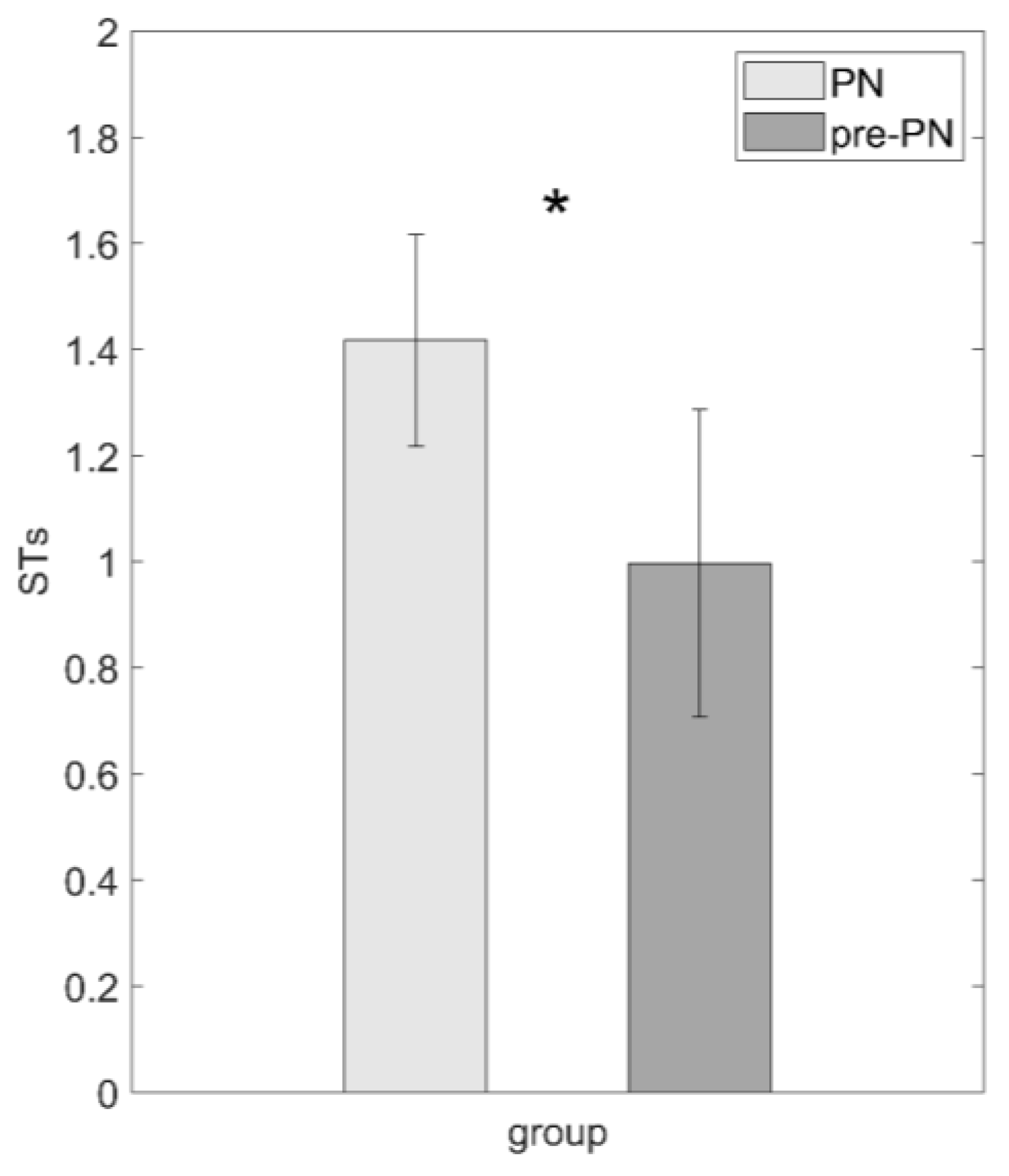
| STs * | 1.417 (1.06) | 0.996 (1.51) | |
| Sleep variables | SOL * | 14.087 (10.22) | 10.102 (4.09) |
| NOA * | 1.218 (0.79) | 1.593 (1.37) | |
| ISW * | 4.320 (3.23) | 6.709 (7.74) | |
| TBT | 493.330 (49.14) | 503.367 (76.60) | |
| TST | 441.661 (36.90) | 457.102 (64.07) | |
| SE (%) | 90.040 (4.38) | 90.930 (5.19) | |
| Sleep depth | 3.787 (0.58) | 3.73 (0.58) | |
| Sleep quiet | 3.371 (0.63) | 3.312 (0.72) | |
| Sleep restless | 3.453 (0.59) | 3.279 (0.56) | |
| Dream variables | DR frequency * | 0.701 (0.52) | 0.599 (0.69) |
| TWC * | 73.059 (64.14) | 124.819 (88.89) | |
| EL * | 2.429(1.12) | 2.767 (0.88) | |
| B | 1.903 (0.92) | 2.750 (0.86) | |
| VV | 2.972 (0.82) | 3.582 (0.90) | |
| Dependent Variables | Predictors | Standardized β Coefficients | t | p |
|---|---|---|---|---|
| TWC | ||||
| Adjusted R2 = 0.087 | Group | −0.289 | −1.869 | 0.068 |
| F = 2.488 | STs | −0.166 | −1.082 | 0.285 |
| p = 0.073 | ISW | 0.019 | 0.128 | 0.899 |
| EL | ||||
| Adjusted R2 = 0.129 | Group | −0.090 | −0.599 | 0.553 |
| F = 3.327 | STs | −0.393 | −2.626 | 0.012 * |
| p = 0.028 | ISW | 0.030 | 0.208 | 0.836 |
| B | ||||
| Adjusted R2 = 0.163 | Group | −0.395 | −2.673 | 0.011 * |
| F = 4.057 | STs | −0.018 | −0.124 | 0.902 |
| p = 0.012 | ISW | 0.171 | 1.217 | 0.230 |
| VV | ||||
| Adjusted R2 = 0.056 | Group | −0.336 | −2.138 | 0.038 |
| F = 1.928 | STs | −0.006 | −0.037 | 0.971 |
| p = 0.139 | ISW | 0.015 | 0.097 | 0.923 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Camaioni, M.; Scarpelli, S.; Alfonsi, V.; Gorgoni, M.; Calzolari, R.; De Bartolo, M.; Mangiaruga, A.; Couyoumdjian, A.; De Gennaro, L. How COVID-19 Affected Sleep Talking Episodes, Sleep and Dreams? Brain Sci. 2024, 14, 486. https://doi.org/10.3390/brainsci14050486
Camaioni M, Scarpelli S, Alfonsi V, Gorgoni M, Calzolari R, De Bartolo M, Mangiaruga A, Couyoumdjian A, De Gennaro L. How COVID-19 Affected Sleep Talking Episodes, Sleep and Dreams? Brain Sciences. 2024; 14(5):486. https://doi.org/10.3390/brainsci14050486
Chicago/Turabian StyleCamaioni, Milena, Serena Scarpelli, Valentina Alfonsi, Maurizio Gorgoni, Rossana Calzolari, Mina De Bartolo, Anastasia Mangiaruga, Alessandro Couyoumdjian, and Luigi De Gennaro. 2024. "How COVID-19 Affected Sleep Talking Episodes, Sleep and Dreams?" Brain Sciences 14, no. 5: 486. https://doi.org/10.3390/brainsci14050486
APA StyleCamaioni, M., Scarpelli, S., Alfonsi, V., Gorgoni, M., Calzolari, R., De Bartolo, M., Mangiaruga, A., Couyoumdjian, A., & De Gennaro, L. (2024). How COVID-19 Affected Sleep Talking Episodes, Sleep and Dreams? Brain Sciences, 14(5), 486. https://doi.org/10.3390/brainsci14050486










