Neuropathological Applications of Microscopy with Ultraviolet Surface Excitation (MUSE): A Concordance Study of Human Primary and Metastatic Brain Tumors
Abstract
1. Introduction
2. Materials and Methods
2.1. Image Acquisition and Diagnostic Accuracy Comparison
2.2. Whole-Exome Sequencing
2.3. Targeted Sequencing of DNA and RNA Variant Calling
3. Results
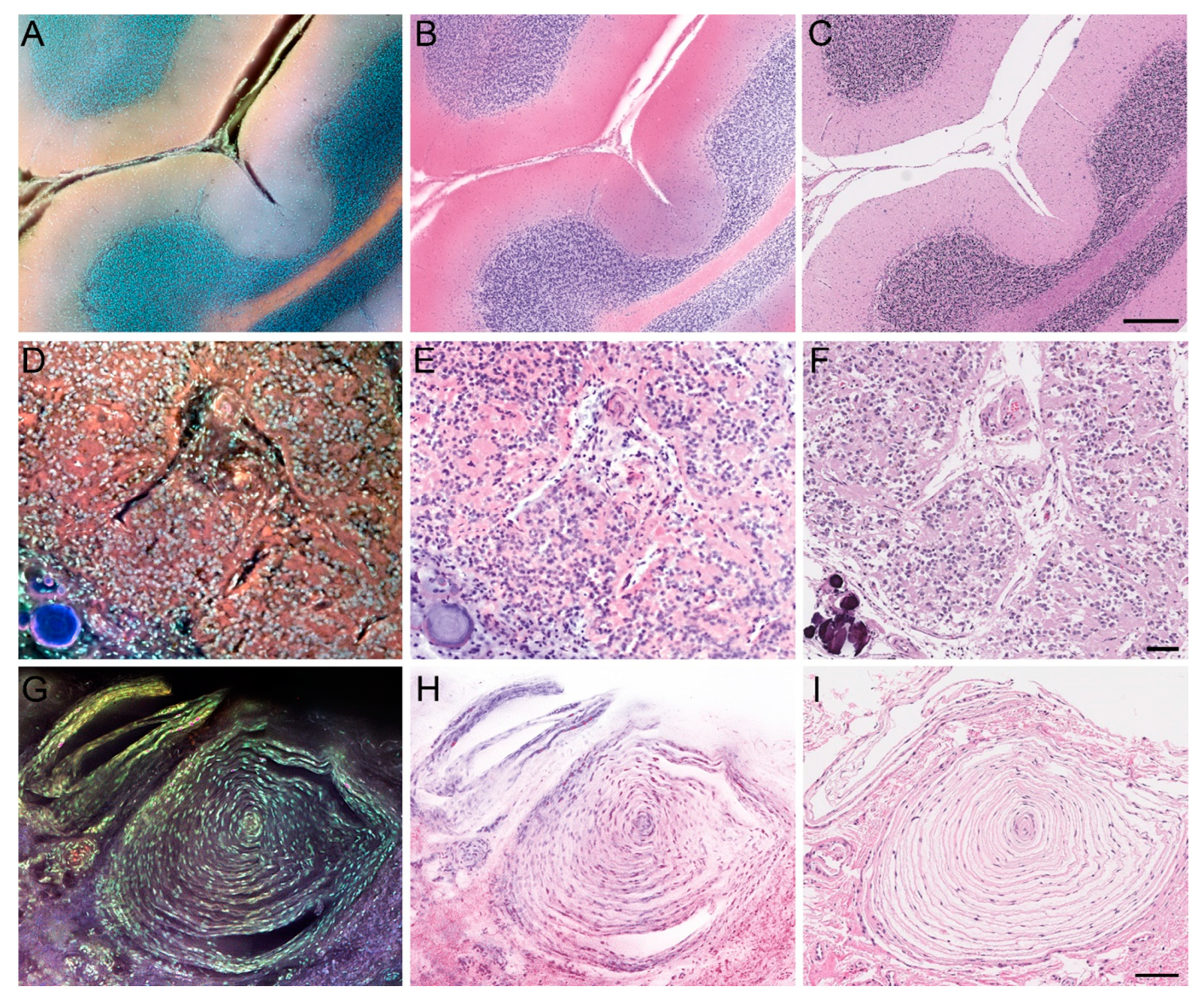
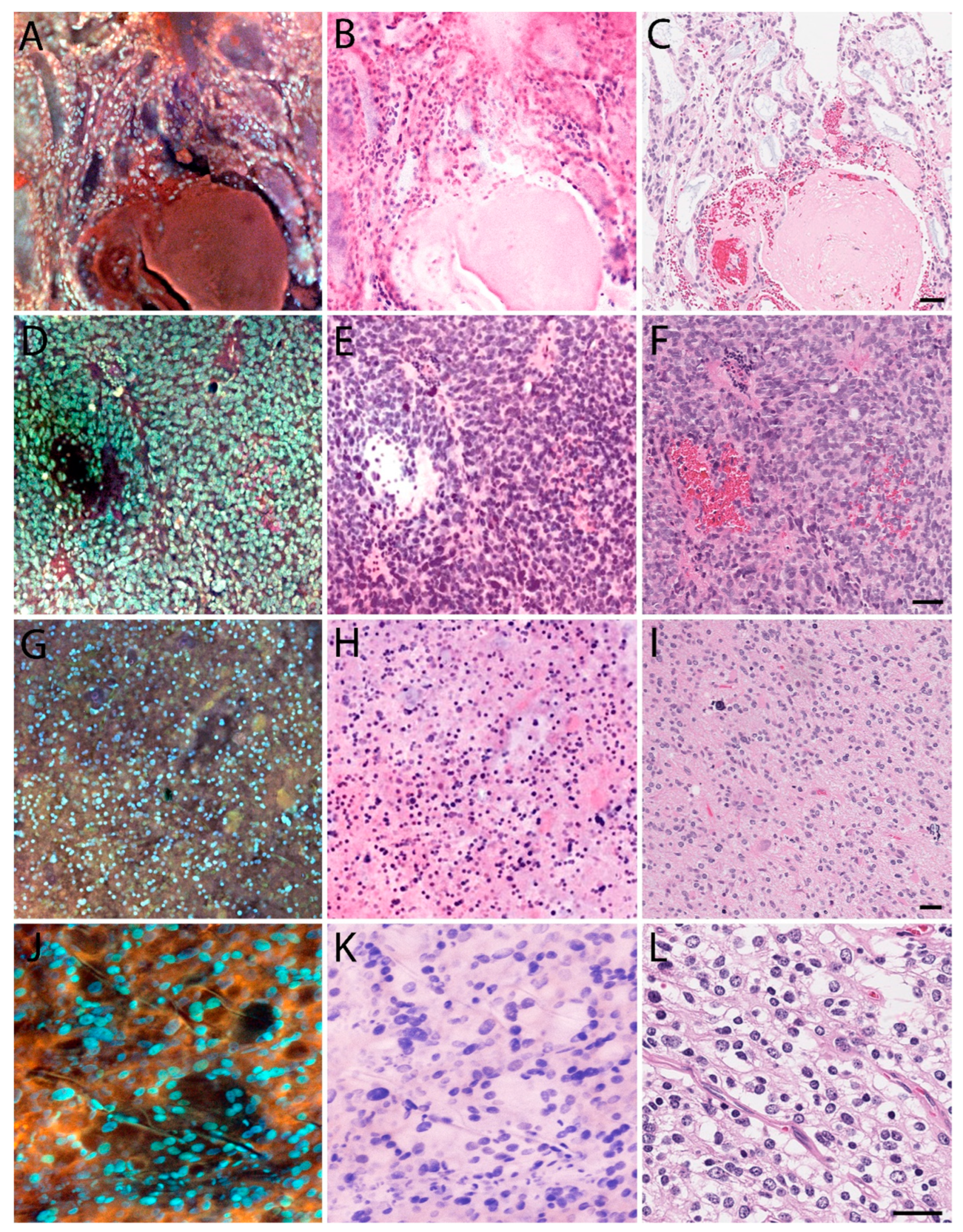
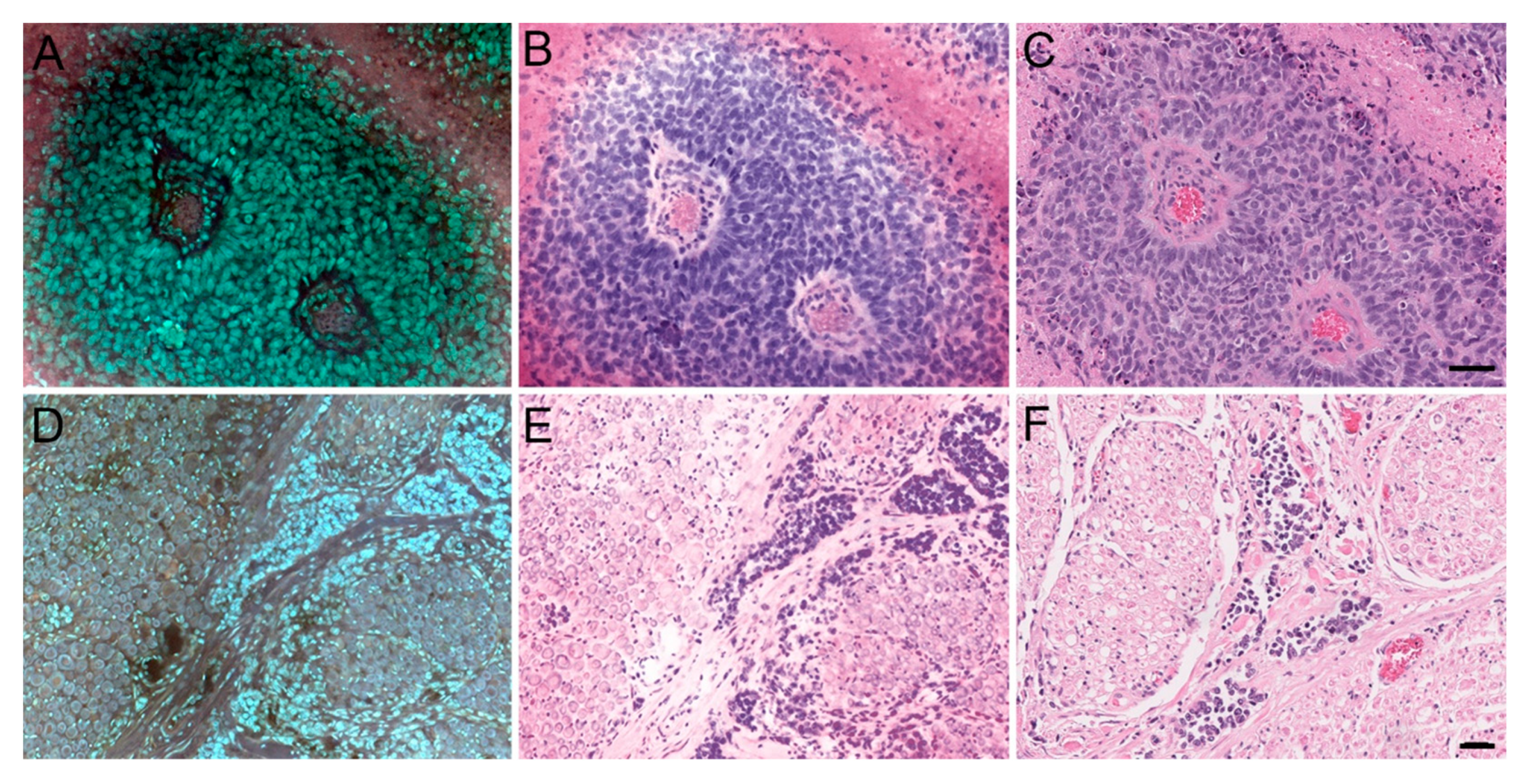
3.1. MUSE Image Quality Compared to Conventional Histology
3.2. Preliminary Validation Studies
| Reviewer 1 | Reviewer 2 | Reviewer 3 | |||||
|---|---|---|---|---|---|---|---|
| Tumor Type | Correct | Incorrect | Correct | Incorrect | Correct | Incorrect | Combined Accuracy |
| Diffuse Astrocytic DA II, AA III, and GBM IV | 10 | 0 | 9 | 1 | 9 | 1 | 93% |
| Oligodendroglial Oligo II, AO III | 4 | 0 | 4 | 0 | 3 | 1 | 92% |
| Meningioma Grades I and II | 6 | 0 | 5 | 1 | 5 | 1 | 89% |
| Metastatic | 3 | 0 | 3 | 0 | 3 | 0 | 100% |
| Normal Anatomic Structures | 4 | 0 | 4 | 0 | 4 | 0 | 100% |
| Combined Accuracy | 100% | 93% | 89% | 94% | |||
| Reviewer 1 | Reviewer 2 | Reviewer 3 | |||||
|---|---|---|---|---|---|---|---|
| Tumor Type | Correct | Incorrect | Correct | Incorrect | Correct | Incorrect | Combined Accuracy |
| Diffuse Astrocytoma Grade II | 5 | 0 | 5 | 0 | 5 | 0 | 100% |
| Anaplastic Astrocytoma Grade III | 6 | 0 | 5 | 1 | 3 | 3 | 78% |
| Glioblastoma Multiforme Grade IV | 10 | 6 | 9 | 7 | 10 | 6 | 60% |
| Oligodendroglioma Grade II | 8 | 0 | 8 | 0 | 7 | 1 | 96% |
| Anaplastic Oligodendroglioma Grade III | 2 | 0 | 0 | 2 | 0 | 2 | 33% |
| Meningioma Grade I | 5 | 0 | 5 | 0 | 5 | 0 | 100% |
| Meningioma Grade II | 4 | 1 | 0 | 5 | 1 | 4 | 33% |
| Combined Accuracy | 85% | 68% | 66% | 73% | |||
| Reviewer 1 | Reviewer 2 | Reviewer 3 | |||||
|---|---|---|---|---|---|---|---|
| Cellular Feature | Correct | Incorrect | Correct | Incorrect | Correct | Incorrect | Combined Accuracy |
| Cellularity | 36 | 11 | 39 | 8 | 41 | 6 | 82% |
| Atypia | 44 | 3 | 43 | 4 | 43 | 4 | 92% |
| Mitoses | 38 | 9 | 34 | 13 | 33 | 14 | 74% |
| Combined Accuracy | 84% | 82% | 83% | 83% | |||
3.3. Suitability for Downstream Molecular Analysis
4. Discussion
5. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kristensen, B.W.; Priesterbach-Ackley, L.P.; Petersen, J.K.; Wesseling, P. Molecular Pathology of Tumors of the Central Nervous System. Ann. Oncol. 2019, 30, 1265–1278. [Google Scholar] [CrossRef] [PubMed]
- Hardesty, D.A.; Sanborn, M.R.; Parker, W.E.; Storm, P.B. Perioperative seizure incidence and risk factors in 223 pediatric brain tumor patients without prior seizures. J. Neurosurg. Pediatr. 2011, 7, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Giannetti, A.; Alvarenga, A.; de Lima, T.; Pedrosa, H.; Souweidane, M.M. Neuroendoscopic biopsy of brain lesions: Accuracy and complications. J. Neurosurg. 2015, 122, 34–39. [Google Scholar] [CrossRef]
- Fereidouni, F.; Harmany, Z.T.; Tian, M.; Todd, A.; Kintner, J.A.; McPherson, J.D.; Borowski, A.D.; Bishop, J.; Lechpammer, M.; Demos, S.G.; et al. Microscopy with ultraviolet surface excitation for rapid slide-free histology. Nat. Biomed. Eng. 2017, 1, 957–966. [Google Scholar] [CrossRef]
- Lin, B.; Urayama, S.; Saroufeem, R.M.; Matthews, D.L.; Demos, S.G. Real-time microscopic imaging of esophageal epithelial disease with autofluorescence under ultraviolet excitation. Opt. Express 2009, 17, 12502–12509. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.; Urayama, S.; Saroufeem, R.M.; Matthews, D.L.; Demos, S.G. Establishment of rules for interpreting ultraviolet autofluorescence microscopy images for noninvasive detection of Barrett’s esophagus and dysplasia. J. Biomed. Opt. 2012, 17, 016013. [Google Scholar] [CrossRef] [PubMed]
- Qorbani, A.; Fereidouni, F.; Levenson, R.; Lahoubi, S.Y.; Harmany, Z.T.; Todd, A.; Fung, M.A. Microscopy with ultraviolet surface excitation (MUSE): A novel approach to real-time inexpensive slide-free dermatology. J. Cutan. Pathol. 2018, 45, 498–503. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef] [PubMed]
- DePristo, M.A.; Banks, E.; Poplin, R.; Garimella, K.V.; Maguire, J.R.; Hartl, C.; Philippakis, A.A.; del Angel, G.; Rivas, M.A.; Hanna, M.; et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat. Genet. 2011, 43, 491–498. [Google Scholar] [CrossRef]
- Kim, S.; Scheffler, K.; Halpern, A.L.; Bekritsky, M.A.; Noh, E.; Kallberg, M.; Chen, X.; Kim, Y.; Beyter, D.; Krusche, P.; et al. Strelka2: Fast and accurate calling of germline and somatic variants. Nat. Methods 2018, 15, 591–594. [Google Scholar] [CrossRef] [PubMed]
- Dunn, T.; Berry, G.; Emig-Agius, D.; Jiang, Y.; Lei, S.; Iyer, A.; Udar, N.; Chuang, H.Y.; Hegarty, J.; Dickover, M.; et al. Pisces: An accurate and versatile variant caller for somatic and germline next-generation sequencing data. Bioinformatics 2019, 35, 1579–1581. [Google Scholar] [CrossRef] [PubMed]
- Yates, A.; Akanni, W.; Amode, M.R.; Barrell, D.; Billis, K.; Carvalho-Silva, D.; Cummins, C.; Clapham, P.; Fitzgerald, S.; Gil, L.; et al. Ensembl 2016. Nucleic Acids Res. 2016, 44, D710–D716. [Google Scholar] [CrossRef] [PubMed]
- Do, H.; Dobrovic, A. Sequence artifacts in DNA from formalin-fixed tissues: Causes and strategies for minimization. Clin. Chem. 2015, 61, 64–71. [Google Scholar] [CrossRef] [PubMed]


| DNA ng/μL (Total Yield, ng) [260/280 Ratio] | RNA ng/μL (Total Yield, ng) [260/280 Ratio] | |||
|---|---|---|---|---|
| Sample | MUSE-Treated | Fresh Frozen | MUSE-Treated | Fresh Frozen |
| 39N | 28.7 (2009) [1.85] | 16 (1120) [1.91] | 49.2 (1968) [1.68] | 113.5 (4540) [1.86] |
| 53 | 113 (7910) [1.87] | 85.3 (5971) [1.87] | 195.4 (7816) [2.02] | 250.5 (10,020) [1.97] |
| 57T | 46.5 (3255) [1.85] | 30.8 (2156) [1.78] | 75.5 (3020) [1.92] | 101 (4040) [1.98] |
| Sample 39N | Sample 53 | |||||
|---|---|---|---|---|---|---|
| Metric | Pass Metric | MUSE-Treated | Fresh Frozen | MUSE-Treated | Fresh Frozen | |
| DNA | Mapped reads | ≥5,000,000 | 12,334,171 | 14,797,491 | 11,613,269 | 9,929,415 |
| Mean read length (bp) | ≥75 | 115 | 114 | 112 | 112 | |
| Uniformity (%) | ≥90 | 95.9 | 96.4 | 97.5 | 97.7 | |
| Mean target coverage | ≥800 | 3703 | 4389 | 3426 | 2922 | |
| On-target coverage (%) | ≥85 | 96.0 | 95.9 | 96.1 | 96.2 | |
| C > T transitions (%) | <8.0 | 5.7 | 5.8 | 5.8 | 5.8 | |
| RNA | Mapped reads | ≥500,000 | 969,724 | 764,965 | 1,302,634 | 1,207,136 |
| Mean read length (bp) | ≥60 | 108 | 93 | 111 | 109 | |
| Sample 53 | ||
|---|---|---|
| Metric | MUSE-Treated | Fresh Frozen |
| Mapped reads | 942,438,994 | 1,016,643,517 |
| On-target reads | 457,723,211 | 479,992,813 |
| On-target duplicate reads | 215,608,334 | 207,313,493 |
| Mean target coverage | 939X | 989X |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lechpammer, M.; Todd, A.; Tang, V.; Morningstar, T.; Borowsky, A.; Shahlaie, K.; Kintner, J.A.; McPherson, J.D.; Bishop, J.W.; Fereidouni, F.; et al. Neuropathological Applications of Microscopy with Ultraviolet Surface Excitation (MUSE): A Concordance Study of Human Primary and Metastatic Brain Tumors. Brain Sci. 2024, 14, 108. https://doi.org/10.3390/brainsci14010108
Lechpammer M, Todd A, Tang V, Morningstar T, Borowsky A, Shahlaie K, Kintner JA, McPherson JD, Bishop JW, Fereidouni F, et al. Neuropathological Applications of Microscopy with Ultraviolet Surface Excitation (MUSE): A Concordance Study of Human Primary and Metastatic Brain Tumors. Brain Sciences. 2024; 14(1):108. https://doi.org/10.3390/brainsci14010108
Chicago/Turabian StyleLechpammer, Mirna, Austin Todd, Vivian Tang, Taryn Morningstar, Alexander Borowsky, Kiarash Shahlaie, John A. Kintner, John D. McPherson, John W. Bishop, Farzad Fereidouni, and et al. 2024. "Neuropathological Applications of Microscopy with Ultraviolet Surface Excitation (MUSE): A Concordance Study of Human Primary and Metastatic Brain Tumors" Brain Sciences 14, no. 1: 108. https://doi.org/10.3390/brainsci14010108
APA StyleLechpammer, M., Todd, A., Tang, V., Morningstar, T., Borowsky, A., Shahlaie, K., Kintner, J. A., McPherson, J. D., Bishop, J. W., Fereidouni, F., Harmany, Z. T., Coley, N., Zagzag, D., Wong, J. W. H., Tao, J., Hesson, L. B., Burnett, L., & Levenson, R. (2024). Neuropathological Applications of Microscopy with Ultraviolet Surface Excitation (MUSE): A Concordance Study of Human Primary and Metastatic Brain Tumors. Brain Sciences, 14(1), 108. https://doi.org/10.3390/brainsci14010108





