The Possible Role of Prescribing Medications, Including Central Nervous System Drugs, in Contributing to Male-Factor Infertility (MFI): Assessment of the Food and Drug Administration (FDA) Pharmacovigilance Database
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mascarenhas, M.N.; Flaxman, S.R.; Boerma, T.; Vanderpoel, S.; Stevens, G.A. National, regional, and global trends in infertility prevalence since 1990: A systematic analysis of 277 health surveys. PLoS Med. 2012, 9, e1001356. [Google Scholar] [CrossRef] [PubMed]
- Practice Committee of the American Society for Reproductive Medicine. Definitions of infertility and recurrent pregnancy loss: A committee opinion. Fertil. Steril. 2020, 113, 533–535. [Google Scholar] [CrossRef] [PubMed]
- Harrison, J.E.; Weber, S.; Jakob, R.; Chute, C.G. ICD-11: An international classification of diseases for the twenty-first century. BMC Med. Inform. Decis. Mak. 2021, 21 (Suppl. S6), 206. [Google Scholar] [CrossRef] [PubMed]
- Levine, H.; Jørgensen, N.; Martino-Andrade, A.; Mendiola, J.; Weksler-Derri, D.; Mindlis, I.; Pinotti, R.; Swan, S.H. Temporal trends in sperm count: A systematic review and meta-regression analysis. Hum. Reprod. Update. 2017, 23, 646–659. [Google Scholar] [CrossRef]
- Pierik, F.H.; Van Ginneken, A.M.; Dohle, G.R.; Vreeburg, J.T.; Weber, R.F. The advantages of standardized evaluation of male infertility. Int. J. Androl. 2000, 23, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Drechsel, K.C.; Pilon, M.C.; Stoutjesdijk, F.; Meivis, S.; Schoonmade, L.J.; Wallace, W.H.B.; van Dulmen-den Broeder, E.; Beishuizen, A.; Kaspers, G.J.; Broer, S.L.; et al. Reproductive ability in survivors of childhood, adolescent, and young adult Hodgkin lymphoma: A review. Hum. Reprod. Update. 2023, 29, 486–517. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A. The Newly Available FAERS Public Dashboard: Implications for Health Care Professionals. Hosp. Pharm. 2019, 54, 75–77. [Google Scholar] [CrossRef] [PubMed]
- Chiappini, S.; Schifano, F. A Decade of Gabapentinoid Misuse: An Analysis of the European Medicines Agency’s ‘Suspected Adverse Drug Reactions’ Database. CNS Drugs 2016, 30, 647–654. [Google Scholar] [CrossRef]
- Capogrosso Sansone, A.; Convertino, I.; Galiulo, M.T.; Salvadori, S.; Pieroni, S.; Knezevic, T.; Mantarro, S.; Marino, A.; Hauben, M.; Blandizzi, C.; et al. Muscular Adverse Drug Reactions Associated with Proton Pump Inhibitors: A Disproportionality Analysis Using the Italian National Network of Pharmacovigilance Database. Drug Saf. 2017, 40, 895–909. [Google Scholar] [CrossRef]
- Maignen, F.; Hauben, M.; Hung, E.; Van Holle, L.; Dogne, J. Assessing the extent and impact of the masking effect of disproportionality analyses on two spontaneous reporting systems databases. Pharmacoepidemiol. Drug Saf. 2014, 23, 195–207. [Google Scholar] [CrossRef]
- Naranjo, C.A.; Busto, U.; Sellers, E.M.; Sandor, P.; Ruiz, I.; Roberts, E.A.; Janecek, E.; Domecq, C.; Greenblatt, D.J. A method for estimating the probability of adverse drug reactions. Clin. Pharmacol. Ther. 1981, 30, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Kumar, K.d. An assessment of reported adverse drug reactions in a Tertiary Care Hospital in South India: A retrospective cross-sectional study. Int. J. Pharm. Investig. 2017, 7, 193. [Google Scholar] [CrossRef] [PubMed]
- Felicetti, P.; Trotta, F.; Bonetto, C.; Santuccio, C.; Pernus, Y.B.; Burgner, D.; Chandler, R.; Girolomoni, G.; Hadden, R.D.; Kochhar, S.; et al. Spontaneous reports of vasculitis as an adverse event following immunization: A descriptive analysis across three international databases. Vaccine 2016, 34, 6634–6640. [Google Scholar] [CrossRef] [PubMed]
- Csete, J.; Kamarulzaman, A.; Kazatchkine, M.; Altice, F.; Balicki, M.; Buxton, J.; Cepeda, J.; Comfort, M.; Goosby, E.; Goulão, J.; et al. Public health and international drug policy. Lancet 2016, 387, 1427–1480. [Google Scholar] [CrossRef] [PubMed]
- Bihan, K.; Lebrun-Vignes, B.; Funck-Brentano, C.; Salem, J.E. Uses of pharmacovigilance databases: An overview. Therapie 2020, 75, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Hammond, I.W.; Gibbs, T.G.; Seifert, H.A.; Rich, D.S. Database size and power to detect safety signals in pharmacovigilance. Expert. Opin. Drug Saf. 2007, 6, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Postigo, R.; Brosch, S.; Slattery, J.; van Haren, A.; Dogné, J.M.; Kurz, X.; Candore, G.; Domergue, F.; Arlett, P. EudraVigilance Medicines Safety Database: Publicly Accessible Data for Research and Public Health Protection. Drug Saf. 2018, 41, 665–675. [Google Scholar] [CrossRef] [PubMed]
- Schifano, N.; Capogrosso, P.; Boeri, L.; Fallara, G.; Cakir, O.O.; Castiglione, F.; Alnajjar, H.M.; Muneer, A.; Deho’, F.; Schifano, F.; et al. Medications mostly associated with priapism events: Assessment of the 2015–2020 Food and Drug Administration (FDA) pharmacovigilance database entries. Int. J. Impot. Res. 2022. [Google Scholar] [CrossRef]
- Cocuzza, M.; Alvarenga, C.; Pagani, R. The epidemiology and etiology of azoospermia. Clinics 2013, 68 (Suppl. S1), 15–26. [Google Scholar] [CrossRef]
- Sengupta, P.; Roychoudhury, S.; Nath, M.; Dutta, S. Oxidative Stress and Idiopathic Male Infertility. Adv. Exp. Med. Biol. 2022, 1358, 181–204. [Google Scholar]
- Pandruvada, S.; Royfman, R.; Shah, T.A.; Sindhwani, P.; Dupree, J.M.; Schon, S.; Avidor-Reiss, T. Lack of trusted diagnostic tools for undetermined male infertility. J. Assist. Reprod. Genet. 2021, 38, 265–276. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Parekh, N.; Selvam, M.K.P.; Henkel, R.; Shah, R.; Homa, S.T.; Ramasamy, R.; Ko, E.; Tremellen, K.; Esteves, S.; et al. Male Oxidative Stress Infertility (MOSI): Proposed Terminology and Clinical Practice Guidelines for Management of Idiopathic Male Infertility. World J. Mens. Health 2019, 37, 296–312. [Google Scholar] [CrossRef] [PubMed]
- Fertig, R.M.; Gamret, A.C.; Darwin, E.; Gaudi, S. Sexual side effects of 5-α-reductase inhibitors finasteride and dutasteride: A comprehensive review. Dermatol. Online J. 2017, 23, 3. [Google Scholar] [CrossRef]
- Azzouni, F.; Godoy, A.; Li, Y.; Mohler, J. The 5 alpha-reductase isozyme family: A review of basic biology and their role in human diseases. Adv. Urol. 2012, 2012, 530121. [Google Scholar] [CrossRef] [PubMed]
- Amory, J.K.; Wang, C.; Swerdloff, R.S.; Anawalt, B.D.; Matsumoto, A.M.; Bremner, W.J.; Walker, S.E.; Haberer, L.J.; Clark, R.V. The effect of 5alpha-reductase inhibition with dutasteride and finasteride on semen parameters and serum hormones in healthy men. J. Clin. Endocrinol. Metab. 2007, 92, 1659–1665. [Google Scholar] [CrossRef] [PubMed]
- Overstreet, J.W.; Fuh, V.L.; Gould, J.; Howards, S.S.; Lieber, M.M.; Hellstrom, W.; Shapiro, S.; Carroll, P.; Corfman, R.S.; Petrou, S.; et al. Chronic treatment with finasteride daily does not affect spermatogenesis or semen production in young men. J. Urol. 1999, 162, 1295–1300. [Google Scholar] [CrossRef] [PubMed]
- Samplaski, M.K.; Lo, K.; Grober, E.; Jarvi, K. Finasteride use in the male infertility population: Effects on semen and hormone parameters. Fertil. Steril. 2013, 100, 1542–1546. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.E.; Binsaleh, S.; Lo, K.C.; Jarvi, K. Propecia-induced spermatogenic failure: A report of two cases. Fertil. Steril. 2008, 90, 849.e17–849.e19. [Google Scholar] [CrossRef]
- Chiba, K.; Yamaguchi, K.; Li, F.; Ando, M.; Fujisawa, M. Finasteride-associated male infertility. Fertil. Steril. 2011, 95, 1786.e9–1786.e11. [Google Scholar] [CrossRef]
- Tu, H.Y.V.; Zini, A. Finasteride-induced secondary infertility associated with sperm DNA damage. Fertil. Steril. 2011, 95, 2125.e13–2125.e14. [Google Scholar] [CrossRef]
- Şalvarci, A.; Istanbulluoğlu, O. Secondary infertility due to use of low-dose finasteride. Int. Urol. Nephrol. 2013, 45, 83–85. [Google Scholar] [CrossRef]
- Schifano, N.; Capogrosso, P.; Boeri, L.; Fallara, G.; Chiappini, S.; Rewhorn, M.; Cakir, O.O.; Harvey, H.; Castiglione, F.; Alnajjar, H.M.; et al. Are finasteride-related penile curvature/Peyronie’s disease Adverse Event Reports worthy of further clinical investigation? Disproportionality analysis based on both the Food and Drug Administration (FDA) and the European Medicines Agency (EMA) pharmacov. Int. J. Impot. Res. 2022, 35, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Garcia, P.V.; Barbieri, M.F.; Perobelli, J.E.; Consonni, S.R.; Mesquita, S.D.F.P.; de Grava Kempinas, W.; Pereira, L.A.V. Morphometric-stereological and functional epididymal alterations and a decrease in fertility in rats treated with finasteride and after a 30-day post-treatment recovery period. Fertil. Steril. 2012, 97, 1444–1451. [Google Scholar] [CrossRef]
- Traish, A.M. Post-finasteride syndrome: A surmountable challenge for clinicians. Fertil. Steril. 2020, 113, 21–50. [Google Scholar] [CrossRef] [PubMed]
- Neha, R.; Subeesh, V.; Beulah, E.; Gouri, N.; Maheswari, E. Existence of Notoriety Bias in FDA Adverse Event Reporting System Database and Its Impact on Signal Strength. Hosp. Pharm. 2021, 56, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Mulhall, J.P.; Trost, L.W.; Brannigan, R.E.; Kurtz, E.G.; Redmon, J.B.; Chiles, K.A.; Lightner, D.J.; Miner, M.M.; Murad, M.H.; Nelson, C.J.; et al. Evaluation and Management of Testosterone Deficiency: AUA Guideline. J. Urol. 2018, 200, 423–432. [Google Scholar] [CrossRef]
- Bhasin, S.; Brito, J.P.; Cunningham, G.R.; Hayes, F.J.; Hodis, H.N.; Matsumoto, A.M.; Snyder, P.J.; Swerdloff, R.S.; Wu, F.C.; Yialamas, M.A. Testosterone Therapy in Men With Hypogonadism: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2018, 103, 1715–1744. [Google Scholar] [CrossRef]
- Amory, J.K.; Bremner, W. Endocrine regulation of testicular function in men: Implications for contraceptive development. Mol. Cell Endocrinol. 2001, 182, 175–179. [Google Scholar] [CrossRef]
- Contraceptive efficacy of testosterone-induced azoospermia and oligozoospermia in normal men. Fertil. Steril. 1996, 65, 821–829. [CrossRef]
- Esposito, M.; Salerno, M.; Calvano, G.; Agliozzo, R.; Ficarra, V.; Sessa, F.; Favilla, V.; Cimino, S.; Pomara, C. Impact of anabolic androgenic steroids on male sexual and reproductive function: A systematic review. Panminerva Med. 2023, 65, 43–50. [Google Scholar] [CrossRef]
- Christou, M.A.; Christou, P.A.; Markozannes, G.; Tsatsoulis, A.; Mastorakos, G.; Tigas, S. Effects of Anabolic Androgenic Steroids on the Reproductive System of Athletes and Recreational Users: A Systematic Review and Meta-Analysis. Sports Med. 2017, 47, 1869–1883. [Google Scholar] [CrossRef] [PubMed]
- Schifano, N.; Chiappini, S.; Mosca, A.; Miuli, A.; Santovito, M.C.; Pettorruso, M.; Capogrosso, P.; Dehò, F.; Martinotti, G.; Schifano, F. Recreational Drug Misuse and Its Potential Contribution to Male Fertility Levels’ Decline: A Narrative Review. Brain Sci. 2022, 12, 1582. [Google Scholar] [CrossRef] [PubMed]
- Ghodke-Puranik, Y.; Thorn, C.F.; Lamba, J.K.; Leeder, J.S.; Song, W.; Birnbaum, A.K.; Altman, R.B.; Klein, T.E. Valproic acid pathway: Pharmacokinetics and pharmacodynamics. Pharmacogenet Genom. 2013, 23, 236–241. [Google Scholar] [CrossRef] [PubMed]
- Mishra, M.K.; Kukal, S.; Paul, P.R.; Bora, S.; Singh, A.; Kukreti, S.; Saso, L.; Muthusamy, K.; Hasija, Y.; Kukreti, R. Insights into Structural Modifications of Valproic Acid and Their Pharmacological Profile. Molecules 2021, 27, 104. [Google Scholar] [CrossRef] [PubMed]
- Røste, L.S.; Taubøll, E.; Haugen, T.B.; Bjørnenak, T.; Saetre, E.R.; Gjerstad, L. Alterations in semen parameters in men with epilepsy treated with valproate or carbamazepine monotherapy. Eur. J. Neurol. 2003, 10, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Chen, L.; Wu, D.; Yu, L.; Sun, H.; Zhu, Q. A Comparative Study of the Effects of Valproate and Oxcarbazepine on Sexual Function, Sperm Quality, and Sex Hormones in Males with Epilepsy. Biomed. Res. Int. 2021, 2021, 6624101. [Google Scholar] [CrossRef] [PubMed]
- Tallon, E.; O’Donovan, L.; Delanty, N. Reversible male infertility with valproate use: A review of the literature. Epilepsy Behav. Rep. 2021, 16, 100446. [Google Scholar] [CrossRef]
- Soliman, G.A.; Abd, A. Effects of antiepileptic drugs carbamazepine and sodium valproate on fertility of male rats. Dtsch. Tierarztl. Wochenschr. 1999, 106, 110–113. [Google Scholar]
- Rättyä, J.; Pakarinen, A.J.; Knip, M.; Repo-Outakoski, M.; Myllylä, V.V.; Isojärvi, J.I. Early hormonal changes during valproate or carbamazepine treatment: A 3-month study. Neurology 2001, 57, 440–444. [Google Scholar] [CrossRef]
- Kühn-Velten, W.N.; Herzog, A.G.; Müller, M.R. Acute effects of anticonvulsant drugs on gonadotropin-stimulated and precursor-supported androgen production in the rat testis. Eur. J. Pharmacol. 1990, 181, 151–155. [Google Scholar] [CrossRef]
- Coulter, D.L. Carnitine, valproate, and toxicity. J. Child. Neurol. 1991, 6, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Hamed, S.A.; Moussa, E.M.; Tohamy, A.M.; Mohamed, K.O.; Mohamad, M.E.; Sherif, T.M.; Abdellah, M.M. Seminal fluid analysis and testicular volume in adults with epilepsy receiving valproate. J. Clin. Neurosci. 2015, 22, 508–512. [Google Scholar] [CrossRef] [PubMed]
- Jeulin, C.; Lewin, L.M. Role of free L-carnitine and acetyl-L-carnitine in post-gonadal maturation of mammalian spermatozoa. Hum. Reprod. Update 1996, 2, 87–102. [Google Scholar] [CrossRef]
- Nolten, W.E.; Sholiton, L.J.; Srivastava, L.S.; Knowles, H.C.J.; Werk, E.E.J. The effects of diethylstilbestrol and medroxyprogesterone acetate on kinetics and production of testosterone and dihydrotestosterone in patients with prostatic carcinoma. J. Clin. Endocrinol. Metab. 1976, 43, 1226–1233. [Google Scholar] [CrossRef] [PubMed]
- Gomella, L.G. Effective testosterone suppression for prostate cancer: Is there a best castration therapy? Rev. Urol. 2009, 11, 52–60. [Google Scholar] [PubMed]
- Glaze, G.M. Diethylstilbestrol exposure in utero: Review of literature. J. Am. Osteopath. Assoc. 1984, 83, 435–438. [Google Scholar] [CrossRef] [PubMed]
- Dameshek, W.; Weisfuse, L.; Stein, T. Nitrogen mustard therapy in Hodgkin’s disease; analysis of 50 consecutive cases. Blood. 1949, 4, 338–379. [Google Scholar] [CrossRef]
- Ansell, S.M. Hodgkin lymphoma: MOPP chemotherapy to PD-1 blockade and beyond. Am. J. Hematol. 2016, 91, 109–112. [Google Scholar] [CrossRef]
- Talpur, R.; Venkatarajan, S.; Duvic, M. Mechlorethamine gel for the topical treatment of stage IA and IB mycosis fungoides-type cutaneous T-cell lymphoma. Expert. Rev. Clin. Pharmacol. 2014, 7, 591–597. [Google Scholar] [CrossRef]
- Strawn, J.R.; Geracioti, L.; Rajdev, N.; Clemenza, K.; Levine, A. Pharmacotherapy for generalized anxiety disorder in adult and pediatric patients: An evidence-based treatment review. Expert. Opin. Pharmacother. 2018, 19, 1057–1070. [Google Scholar] [CrossRef]
- Gul, M.; Bocu, K.; Serefoglu, E.C. Current and emerging treatment options for premature ejaculation. Nat. Rev. Urol. 2022, 19, 659–680. [Google Scholar] [CrossRef] [PubMed]
- Gollenberg, A.L.; Liu, F.; Brazil, C.; Drobnis, E.Z.; Guzick, D.; Overstreet, J.W.; Redmon, J.B.; Sparks, A.; Wang, C.; Swan, S.H. Semen quality in fertile men in relation to psychosocial stress. Fertil. Steril. 2010, 93, 1104–1111. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.S.; Sharma, V.L.; Tiwari, P.; Singh, D.; Maikhuri, J.P.; Gupta, G.; Singh, M.M. The spermicidal and antitrichomonas activities of SSRI antidepressants. Bioorg Med. Chem. Lett. 2006, 16, 2509–2512. [Google Scholar] [CrossRef] [PubMed]
- Tanrikut, C.; Schlegel, P.N. Antidepressant-associated changes in semen parameters. Urology 2007, 69, 185.e5–185.e7. [Google Scholar] [CrossRef] [PubMed]
- Elnazer, H.Y.; Baldwin, D.S. Treatment with citalopram, but not with agomelatine, adversely affects sperm parameters: A case report and translational review. Acta Neuropsychiatr. 2014, 26, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Safarinejad, M.R. Sperm DNA damage and semen quality impairment after treatment with selective serotonin reuptake inhibitors detected using semen analysis and sperm chromatin structure assay. J. Urol. 2008, 180, 2124–2128. [Google Scholar] [CrossRef] [PubMed]
- Tanrikut, C.; Feldman, A.S.; Altemus, M.; Paduch, D.A.; Schlegel, P.N. Adverse effect of paroxetine on sperm. Fertil. Steril. 2010, 94, 1021–1026. [Google Scholar] [CrossRef]
- Pham, M.N.; Siebert, A.L.; Faw, C.A.; Dubin, J.M.; Hudnall, M.T.; Lai, J.D.; Wren, J.M.; Bennett, N.E.; Brannigan, R.E.; Halpern, J.A. Selective Serotonin Reuptake Inhibitor (SSRI) Use is Not Associated With Impaired Semen Parameters. Urology 2022, 164, 140–144. [Google Scholar] [CrossRef]
- Göçmez, S.S.; Utkan, T.; Ulak, G.; Gacar, N.; Erden, F. Effects of long-term treatment with fluoxetine and venlafaxine on rat isolated vas deferens. Auton. Autacoid Pharmacol. 2010, 30, 197–202. [Google Scholar] [CrossRef]
- Kesim, M.; Kadioglu, M.; Yaris, E.; Kalyoncu, N.I.; Ozyavuz, R.; Ulku, C. Paroxetine alters KCl- or ATP-induced contractile responses of isolated vas deferens in rats chronically treated with ethanol. Pharmacol. Res. 2004, 49, 51–57. [Google Scholar] [CrossRef]
- Tektemur, A.; Etem Önalan, E.; Kaya Tektemur, N.; Güngör, İ.H.; Türk, G.; Kuloğlu, T. Verapamil-induced ion channel and miRNA expression changes in rat testis and/or spermatozoa may be associated with male infertility. Andrologia 2020, 52, e13778. [Google Scholar] [CrossRef] [PubMed]
- Almeida, S.A.; Teófilo, J.M.; Anselmo Franci, J.A.; Brentegani, L.G.; Lamano-Carvalho, T.L. Antireproductive effect of the calcium channel blocker amlodipine in male rats. Exp. Toxicol. Pathol. 2000, 52, 353–356. [Google Scholar] [PubMed]
- Morton, B.E.; Sagadraca, R.; Fraser, C. Sperm motility within the mammalian epididymis: Species variation and correlation with free calcium levels in epididymal plasma. Fertil. Steril. 1978, 29, 695–698. [Google Scholar] [CrossRef] [PubMed]
- Morakinyo, A.; Iranloye, B.; Adegoke, O. Calcium antagonists modulate oxidative stress and acrosomal reaction in rat spermatozoa. Arch. Med. Sci. 2011, 7, 613–618. [Google Scholar] [CrossRef] [PubMed]
- Omolaoye, T.S.; Halabi, M.O.; Mubarak, M.; Cyril, A.C.; Duvuru, R.; Radhakrishnan, R.; Du Plessis, S.S. Statins and Male Fertility: Is There a Cause for Concern? Toxics 2022, 10, 627. [Google Scholar] [CrossRef] [PubMed]
- Frenoux, J.M.; Vernet, P.; Volle, D.H.; Britan, A.; Saez, F.; Kocer, A.; Henry-Berger, J.; Mangelsdorf, D.J.; Lobaccaro, J.A.; Drevet, J.R. Nuclear oxysterol receptors, LXRs, are involved in the maintenance of mouse caput epididymidis structure and functions. J. Mol. Endocrinol. 2004, 33, 361–375. [Google Scholar] [CrossRef] [PubMed]
- Schifano, N.; Chiappini, S.; Castiglione, F.; Salonia, A.; Schifano, F. Is medicinal ketamine associated with urinary dysfunction issues? Assessment of both the European Medicines Agency (EMA) and the UK Yellow Card Scheme pharmacovigilance database-related reports. LUTS Low. Urin. Tract. Symptoms 2021, 13, 230–237. [Google Scholar] [CrossRef]
- Pradhan, P.; Lavallée, M.; Akinola, S.; Escobar Gimenes, F.R.; Bérard, A.; Méthot, J.; Piché, M.E.; Gonella, J.M.; Cloutier, L.; Leclerc, J. Causality assessment of adverse drug reaction: A narrative review to find the most exhaustive and easy-to-use tool in post-authorization settings. J. Appl. Biomed. 2023, 21, 59–66. [Google Scholar] [CrossRef]


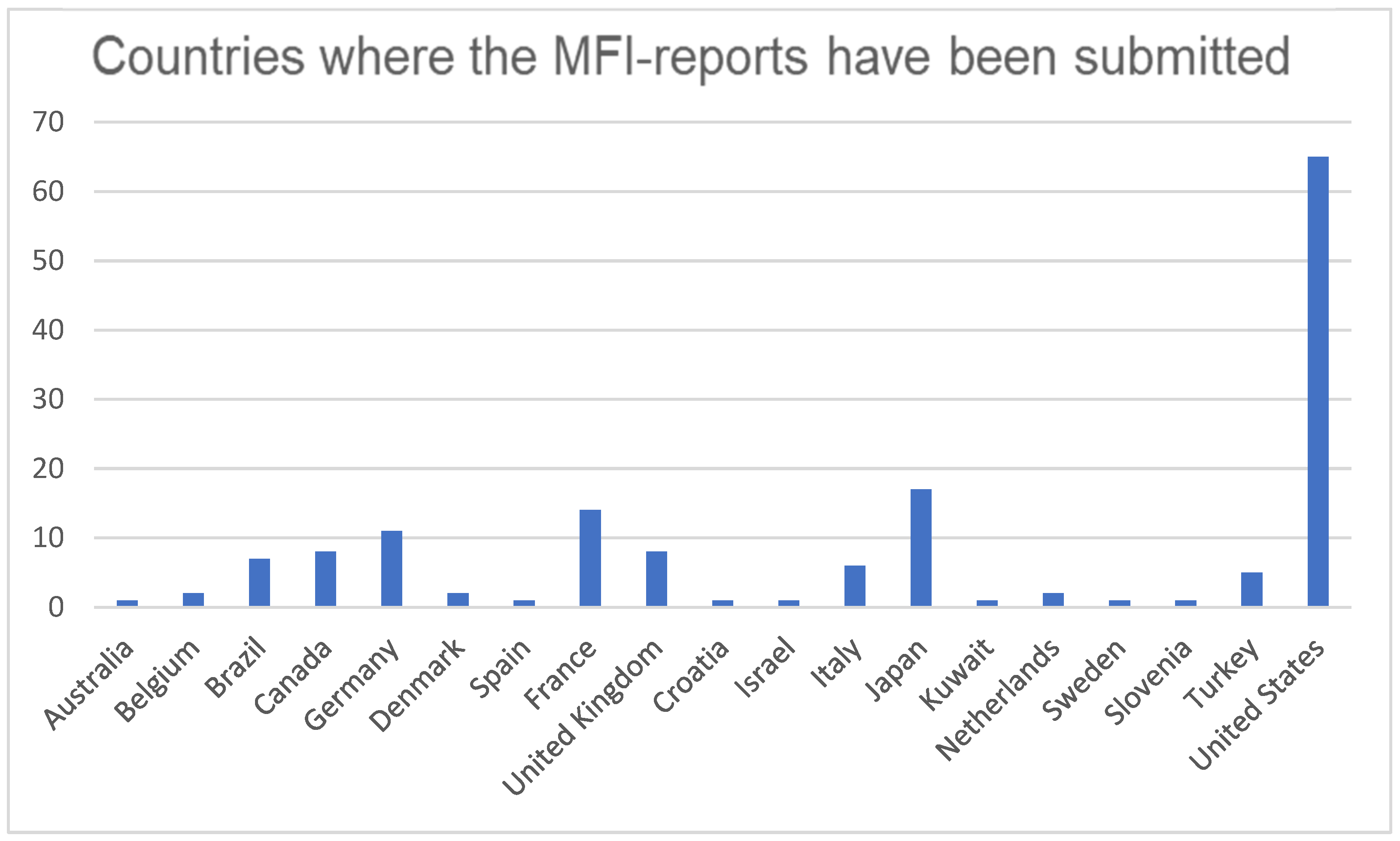
| Pharmacological Class | Drug | Number of Individual Cases Associated with MFI in the Database (n) | Percentage of the Individual Cases of MFI When Compared to All the Compounds in the Database (%) | PRR (CI95%) * |
|---|---|---|---|---|
| 5 alpha-reductase inhibitors | Finasteride | 86 | 21.08% | 16.04 (12.67–20.3) |
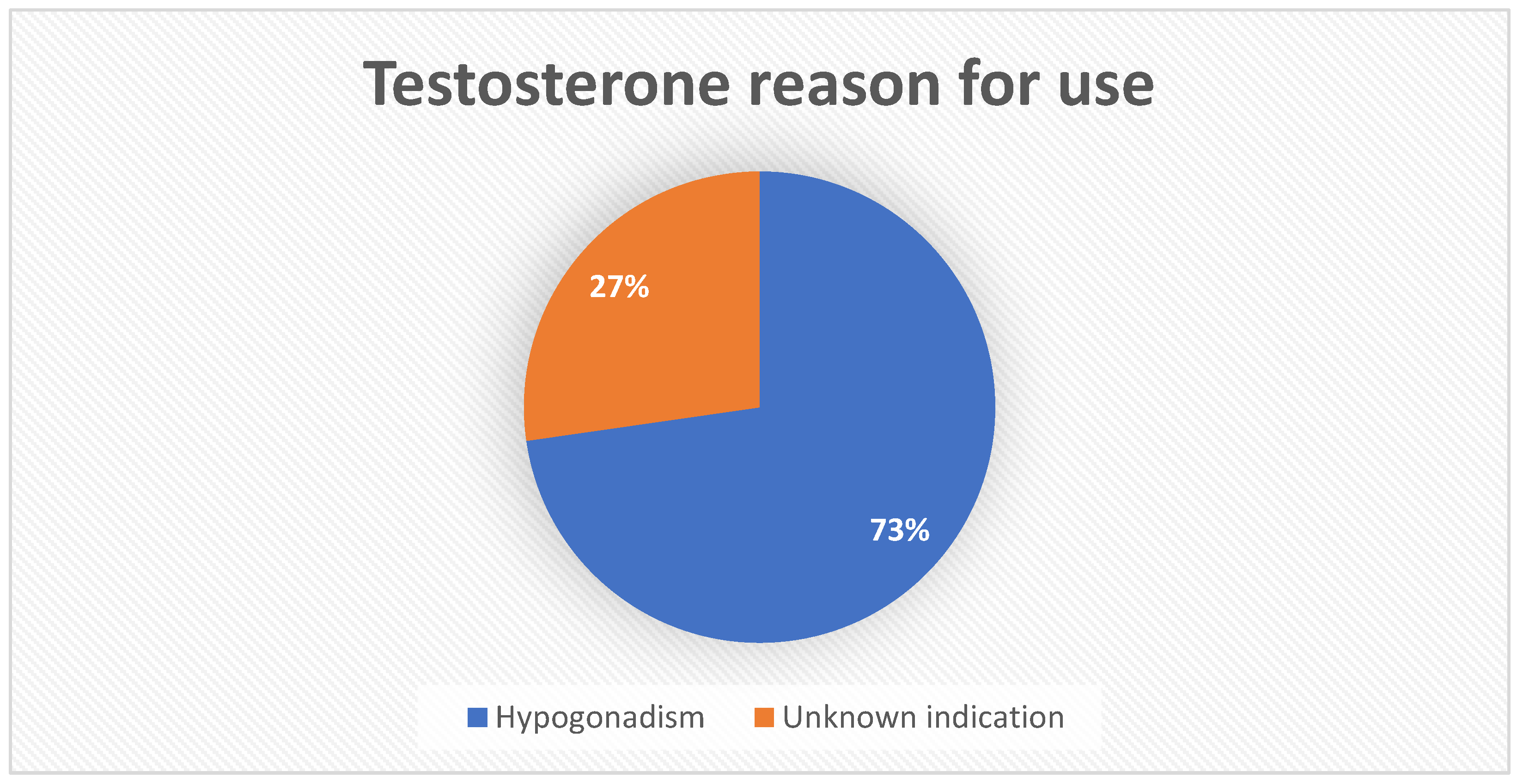
| Steroid hormones | Testosterone | 33 | 8.09% | 3.03 (2.12–4.32) |
| Anticonvulsants | Valproic Acid | 32 | 7.42% | 1.72 (1.20–2.47) |
| Carbamazepine | 17 | 4.17% | 1.07 (0.66–1.74) | |
| SSRIs | Sertraline | 29 | 7.11% | 0.94 (0.64–1.37) |
| Paroxetine | 17 | 4.17% | 0.92 (0.56–1.5) | |
| Fluoxetine | 15 | 3.68% | 0.51 (0.30–0.85) | |
| Direct Vasodilators | Minoxidil | 24 | 5.88% | 0.95 (0.62–1.43) |
| Nonsteroidal estrogens | Diethylstilbestrol | 20 | 4.90% | 14.3 (9.13–22.37) |
| Calcium-channel blockers | Amlodipine Besylate | 19 | 4.66% | 0.69 (0.43–1.09) |
| Verapamil | 14 | 3.43% | 1.83 (1.07–3.12) | |
| Nifedipine | 12 | 2.94% | 1.85 (1.04–3.28) | |
| Diltiazem Hydrochloride | 11 | 2.70% | 1.49 (0.82–2.72) | |
| Skin and Mucous Membrane Agents | Isotretinoin | 19 | 4.66% | 0.84 (0.53–1.33) |
| Alkylating agents | Mechlorethamine | 17 | 4.17% | 58.71 (36.30–94.94) |
| Histamine H2-Antagonists | Ranitidine | 16 | 3.92% | 0.10 (0.06–0.16) |
| Antineoplastic Agents | Vincristine | 13 | 3.19% | 1.12 (0.65–1.95) |
| Glucocorticoids | Prednisone | 13 | 3.19% | 0.28 (0.16–0.48) |
| Statins | Lovastatin | 13 | 3.19% | 2.51 (1.44–4.36) |
| Immunosuppressive Agents | Mycophenolate Mofetil | 11 | 2.70% | 0.47 (0.26–0.86) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baldini, S.; Khattak, A.; Capogrosso, P.; Antonini, G.; Dehò, F.; Schifano, F.; Schifano, N. The Possible Role of Prescribing Medications, Including Central Nervous System Drugs, in Contributing to Male-Factor Infertility (MFI): Assessment of the Food and Drug Administration (FDA) Pharmacovigilance Database. Brain Sci. 2023, 13, 1652. https://doi.org/10.3390/brainsci13121652
Baldini S, Khattak A, Capogrosso P, Antonini G, Dehò F, Schifano F, Schifano N. The Possible Role of Prescribing Medications, Including Central Nervous System Drugs, in Contributing to Male-Factor Infertility (MFI): Assessment of the Food and Drug Administration (FDA) Pharmacovigilance Database. Brain Sciences. 2023; 13(12):1652. https://doi.org/10.3390/brainsci13121652
Chicago/Turabian StyleBaldini, Sara, Ahmed Khattak, Paolo Capogrosso, Gabriele Antonini, Federico Dehò, Fabrizio Schifano, and Nicolò Schifano. 2023. "The Possible Role of Prescribing Medications, Including Central Nervous System Drugs, in Contributing to Male-Factor Infertility (MFI): Assessment of the Food and Drug Administration (FDA) Pharmacovigilance Database" Brain Sciences 13, no. 12: 1652. https://doi.org/10.3390/brainsci13121652
APA StyleBaldini, S., Khattak, A., Capogrosso, P., Antonini, G., Dehò, F., Schifano, F., & Schifano, N. (2023). The Possible Role of Prescribing Medications, Including Central Nervous System Drugs, in Contributing to Male-Factor Infertility (MFI): Assessment of the Food and Drug Administration (FDA) Pharmacovigilance Database. Brain Sciences, 13(12), 1652. https://doi.org/10.3390/brainsci13121652







