Examining the Impact of Human Face Stimulus on Shape-Contrast Effects during a Brief Presentation
Abstract
1. Introduction
2. Experiment 1: Comparing the Shape-Contrast Effect Evoked by Emoticon Faces and Realistic Faces
2.1. Methods
2.1.1. Participants
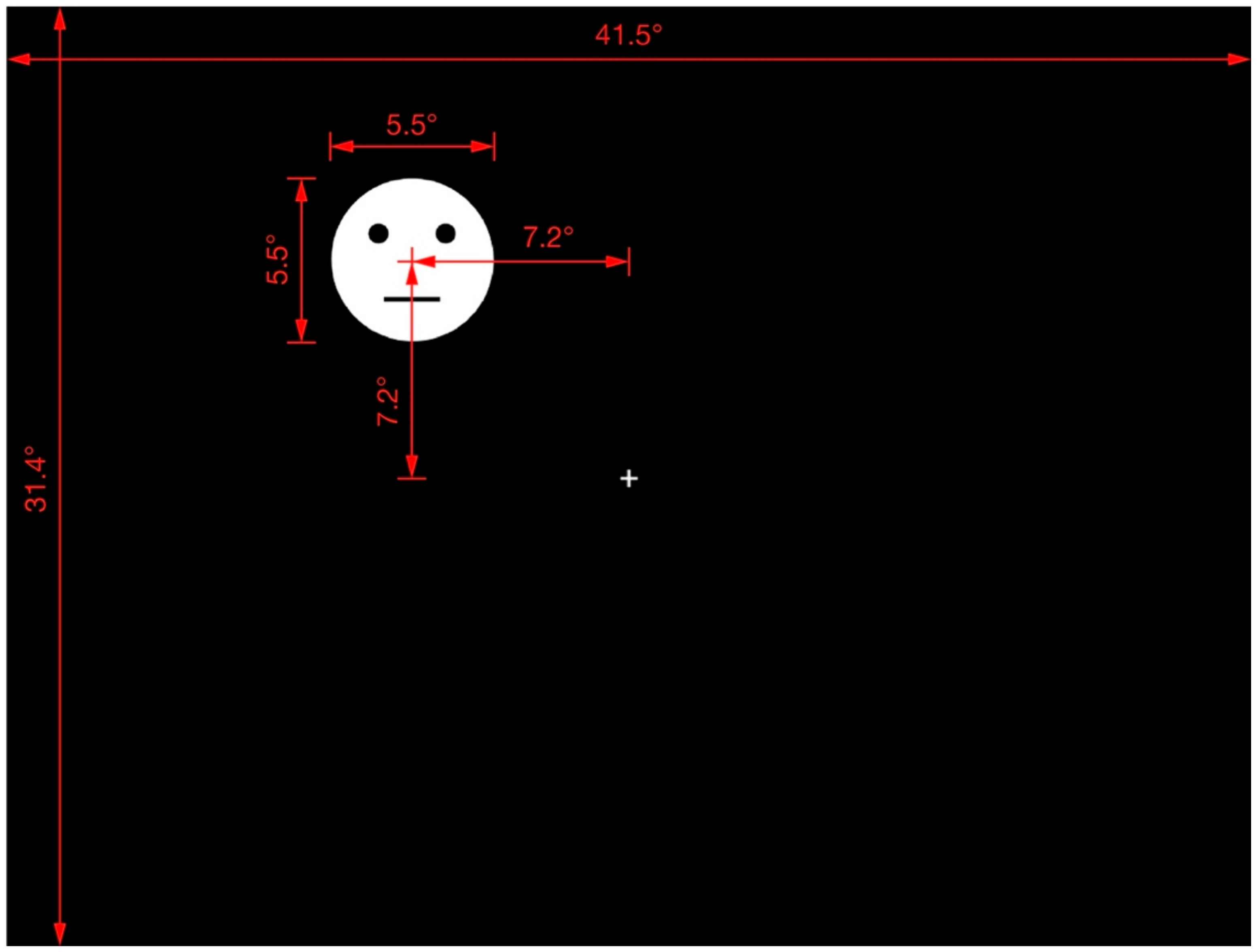
2.1.2. Apparatus and Stimuli
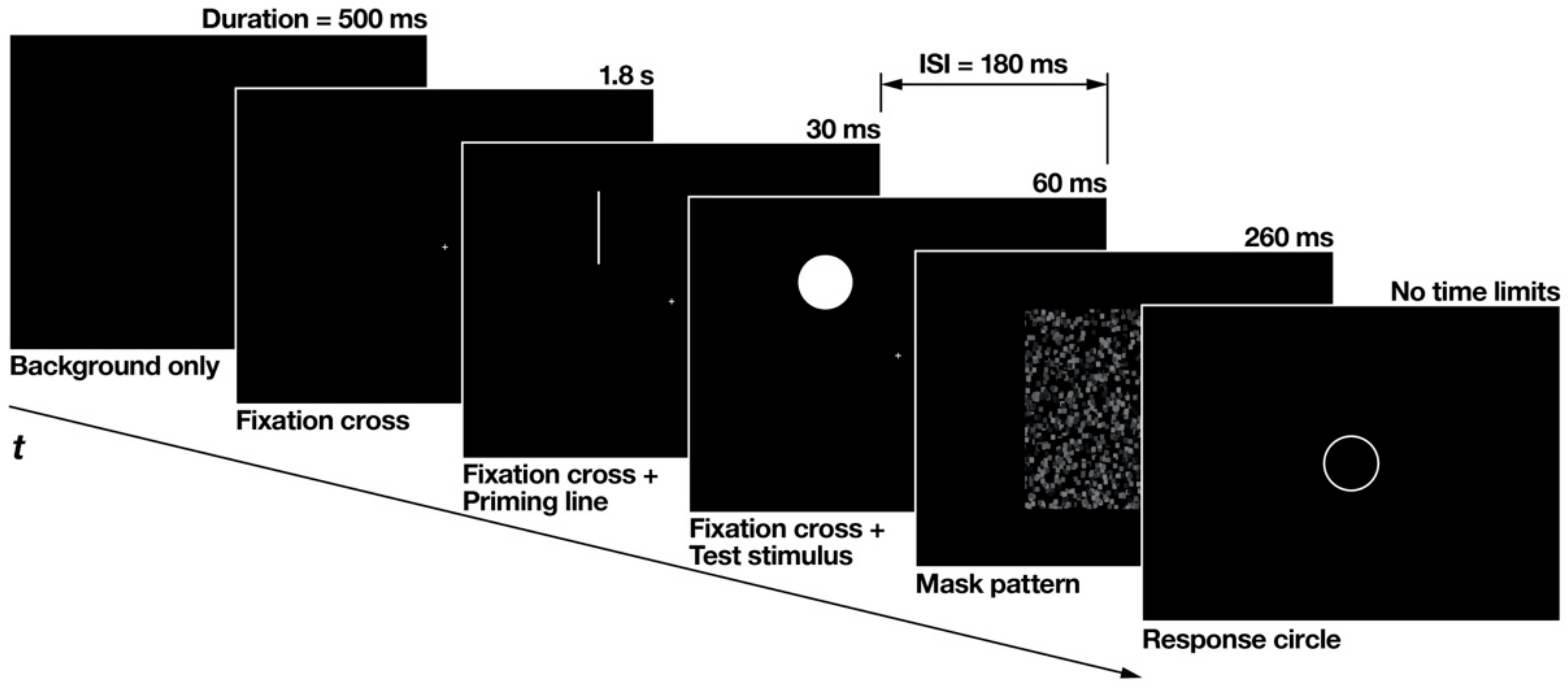
2.1.3. Procedure
2.1.4. Data Analysis
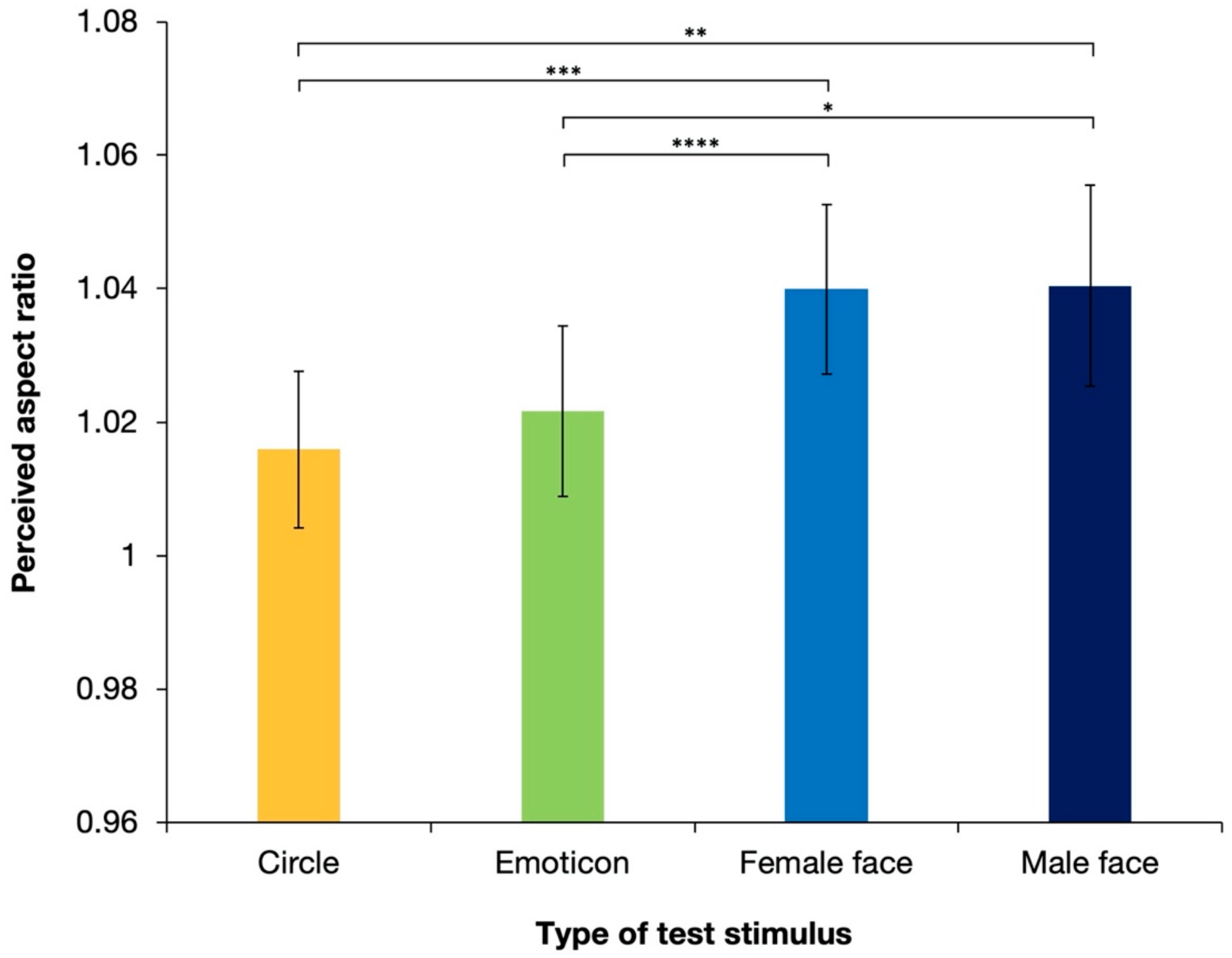
2.2. Results and Discussion
3. Experiment 2: Comparing the Shape-Contrast Effect Evoked by Upright and Inverted Faces
3.1. Methods
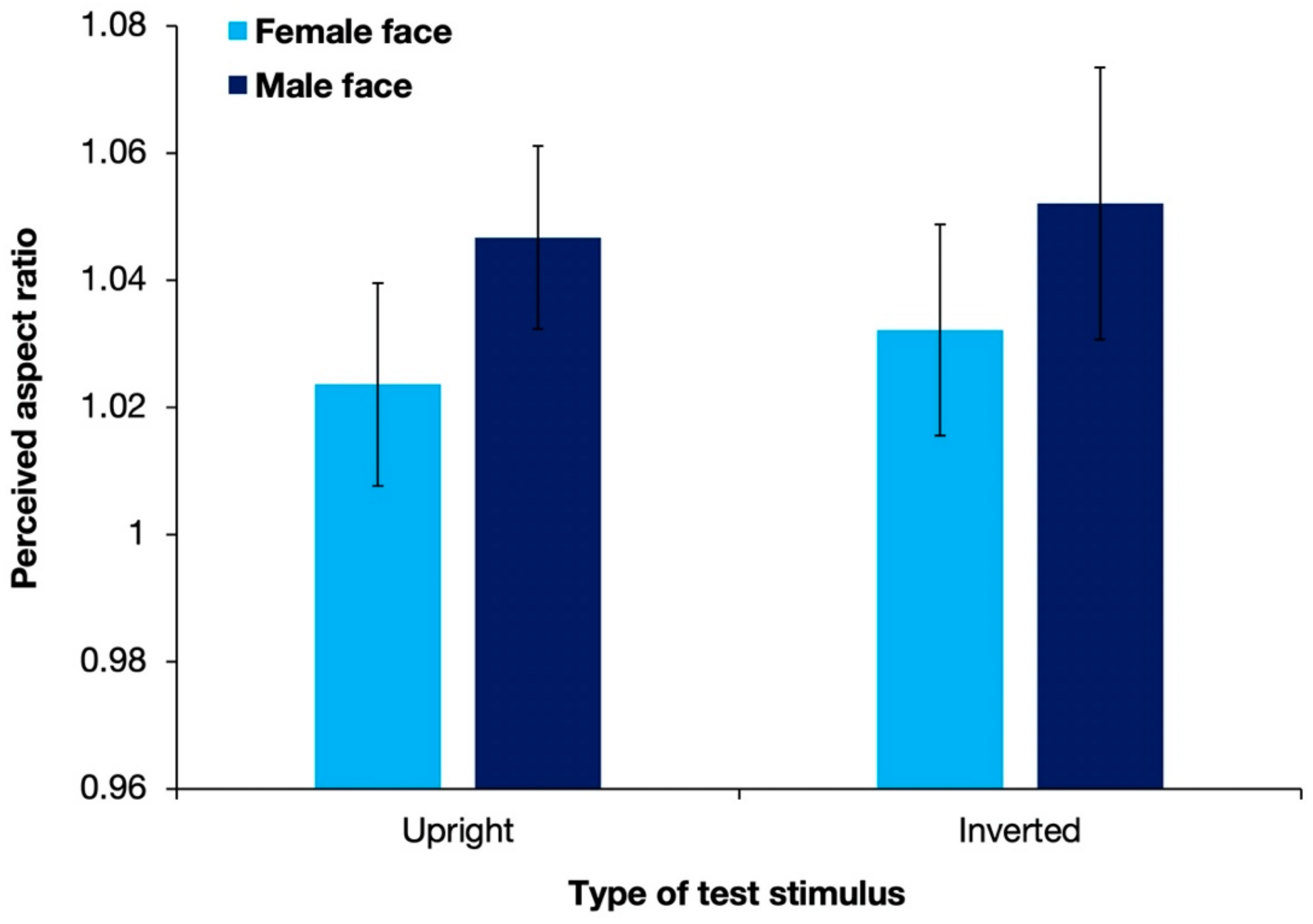
3.2. Results and Discussion
4. General Discussion
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Frisby, J.P. Seeing: Illusion, Brain and Mind; Oxford University Press: Oxford, UK, 1979. [Google Scholar]
- Durgin, F.H.; Proffitt, D.R. Visual learning in the perception of texture: Simple and contingent aftereffects of texture density. Spatial Vis. 1996, 9, 423–474. [Google Scholar] [CrossRef] [PubMed]
- Clifford, C.W.G. Perceptual adaptation: Motion parallels orientation. Trends Cogn. Sci. 2002, 6, 136–143. [Google Scholar] [CrossRef]
- Suzuki, S.; Cavanagh, P. A shape-contrast effect for briefly presented stimuli. J. Exp. Psychol. Hum. Percept. Perform. 1998, 24, 1315–1341. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, S. Attention-dependent brief adaptation to contour orientation: A high-level aftereffect for convexity? Vis. Res. 2001, 41, 3883–3902. [Google Scholar] [CrossRef]
- Suzuki, S. Attentional selection of overlapped shapes: A study using brief shape aftereffects. Vis. Res. 2003, 43, 549–561. [Google Scholar] [CrossRef]
- Suzuki, S. High-level pattern coding revealed by brief shape aftereffects. In Fitting the Mind to the World: Adaptation and After-Effects in High-Level Vision; Clifford, C.W.G., Rhodes, G., Eds.; Oxford University Press: Oxford, UK, 2005; pp. 135–170. [Google Scholar]
- Anderson, N.D.; Habak, C.; Wilkinson, F.; Wilson, H.R. Evaluating shape after-effects with radial frequency patterns. Vis. Res. 2007, 47, 298–308. [Google Scholar] [CrossRef][Green Version]
- Tanaka, K. Inferotemporal cortex and object vision. Annu. Rev. Neurosci. 1996, 19, 109–139. [Google Scholar] [CrossRef] [PubMed]
- Leopold, D.A.; O’Toole, A.J.; Vetter, T.; Blanz, V. Prototype-referenced shape encoding revealed by high-level aftereffects. Nat. Neurosci. 2001, 4, 89–94. [Google Scholar] [CrossRef]
- Nakajima, K.; Minami, T.; Nakauchi, S. Interaction between facial expression and color. Sci. Rep. 2017, 7, 41019. [Google Scholar] [CrossRef]
- Peromaa, T.; Olkkonen, M. Red color facilitates the detection of facial anger—but how much? PLoS ONE 2019, 14, e0215610. [Google Scholar] [CrossRef]
- Webster, M.A.; MacLeod, D.I.A. Visual adaptation and face perception. Philos. Trans. R. Soc. 2011, 366, 1702–1725. [Google Scholar] [CrossRef] [PubMed]
- Müller, K.-M.; Wilke, M.; Leopold, D.A. Visual adaptation to convexity in macaque area V4. Neuroscience 2009, 161, 655–662. [Google Scholar] [CrossRef][Green Version]
- Rhodes, G.; Jeffery, L.; Watson, T.L.; Clifford, C.W.; Nakayama, K. Fitting the mind to the world: Face adaptation and attractiveness aftereffects. Psychol. Sci. 2003, 14, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Afraza, S.-R.; Cavanagh, P. Retinotopy of the face aftereffect. Vis. Res. 2008, 48, 42–54. [Google Scholar] [CrossRef] [PubMed]
- Vakli, P.; Németh, K.; Zimmer, M.; Schweinberger, S.; Kovács, G. Face distortion aftereffects evoked by featureless first-order stimulus configurations. Front. Psychol. 2012, 3, 566. [Google Scholar] [CrossRef] [PubMed]
- Walton, B.; Hills, P. Face distortion aftereffects in personally familiar, famous, and unfamiliar faces. Front. Psychol. 2012, 3, 258. [Google Scholar] [CrossRef]
- Webster, M.A.; Kaping, D.; Mizokami, Y.; Duhamel, P. Adaptation to natural facial categories. Nature 2004, 428, 557–561. [Google Scholar] [CrossRef]
- Webster, M.A.; MacLin, O.H. Figural aftereffects in the perception of faces. Psychon. Bull. Rev. 1999, 6, 647–653. [Google Scholar] [CrossRef]
- Tsao, D.Y.; Livingstone, M.S. Mechanisms of face perception. Annu. Rev. Neurosci. 2008, 31, 411–437. [Google Scholar] [CrossRef]
- Lo, S.-K. The nonverbal communication functions of emoticons in computer-mediated communication. CyberPsychology Behav. 2008, 11, 595–597. [Google Scholar] [CrossRef]
- Tossell, C.C.; Kortum, P.; Shepard, C.; Barg-Walkow, L.H.; Rahmati, A.; Zhong, L. A longitudinal study of emoticon use in text messaging from smartphones. Comput. Hum. Behav. 2012, 28, 659–663. [Google Scholar] [CrossRef]
- Takahashi, T.; Oishi, T.; Shimada, M. Is smiling? cross-cultural study on recognition of emoticon’s emotion. J. Cross-Cult. Psychol. 2017, 48, 1578–1586. [Google Scholar] [CrossRef]
- Butler, A.; Oruc, I.; Fox, C.J.; Barton, J.J. Factors contributing to the adaptation aftereffects of facial expression. Brain Res. 2008, 1191, 116–126. [Google Scholar] [CrossRef]
- Xu, H.; Liu, P.; Dayan, P.; Qian, N. Multi-level visual adaptation: Dissociating curvature and facial-expression aftereffects produced by the same adapting stimuli. Vis. Res. 2012, 72, 42–53. [Google Scholar] [CrossRef] [PubMed]
- Peirce, J.W.; Gray, J.R.; Simpson, S.; MacAskill, M.; Höchenberger, R.; Sogo, H.; Kastman, E.; Lindeløv, J.K. Psychopy2: Experiments in behavior made easy. Behav. Res. Methods 2019, 51, 195. [Google Scholar] [CrossRef] [PubMed]
- Fujimura, T.; Umemura, H. Development and validation of a facial expression database based on the dimensional and categorical model of emotions. Cogn. Emot. 2018, 32, 1663–1670. [Google Scholar] [CrossRef]
- Farah, M.J.; Wilson, K.D.; Drain, M.; Tanaka, J.N. What is “special” about face perception? Psychol. Rev. 1998, 105, 482–498. [Google Scholar] [CrossRef]
- Khuu, S.K.; Phu, J.; Khambiye, S. Apparent motion distorts the shape of a stimulus briefly presented along the motion path. J. Vis. 2010, 10, 1–15. [Google Scholar] [CrossRef]
- Palumbo, R.; Laeng, B.; Tommasi, L. Gender-specific aftereffects following adaptation to silhouettes of human bodies. Vis. Cogn. 2013, 21, 1–12. [Google Scholar] [CrossRef]
- Greene, M.R.; Oliva, A. High-level aftereffects to global scene properties. J. Exp. Psychol. Hum. Percept. Perform. 2010, 36, 1430–1442. [Google Scholar] [CrossRef]
- Palumbo, R.; D’Ascenzo, S.; Quercia, A.; Tommasi, L. Adaptation to complex pictures: Exposure to emotional valence induces assimilative aftereffects. Front. Psychol. 2017, 8, 54. [Google Scholar] [CrossRef] [PubMed]
- Daelli, V.; van Rijsbergen, N.J.; Treves, A. How recent experience affects the perception of ambiguous objects. Brain Res. 2010, 1322, 81–91. [Google Scholar] [CrossRef]
- Daelli, V. High-level adaptation aftereffects for novel objects: The role of pre-existing representations. Neuropsychologia 2011, 49, 1923–1927. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, R.; D’Ascenzo, S.; Tommasi, L. Cross-category adaptation: Exposure to faces produces gender aftereffects in body perception. Psychol. Res. 2015, 79, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Habtegiorgis, S.W.; Rifai, K.; Lappe, M.; Wahl, S. Adaptation to skew distortions of natural scenes and retinal specificity of its aftereffects. Front. Psychol. 2017, 8, 1158. [Google Scholar] [CrossRef] [PubMed]
- Dennett, H.W.; Edwards, M.; McKone, E. Global face distortion aftereffects tap face-specific and shape-generic processes. J. Vis. 2012, 12, 11. [Google Scholar] [CrossRef]
- Storrs, K.R.; Arnold, D.H. Shape aftereffects reflect shape constancy operations: Appearance matters. J. Exp. Psychol. Hum. Percept. Perform. 2013, 39, 616–622. [Google Scholar] [CrossRef]
- Ono, F.; Watanabe, K. Attention can retrospectively distort visual space. Psychol. Sci. 2011, 22, 472–477. [Google Scholar] [CrossRef]
- Ono, F.; Watanabe, K. Shape-assimilation effect: Retrospective distortion of visual shapes. Atten. Percept. Psychophys. 2014, 76, 5–10. [Google Scholar] [CrossRef]
- Qian, K.; Mitsudo, H. Eggs illusion: Local shape deformation generated by a grid pattern. J. Vis. 2016, 16, 27. [Google Scholar] [CrossRef]
- Qian, K.; Mitsudo, H. Role of orientation processing in the eggs illusion. Perception 2019, 48, 1197–1213. [Google Scholar] [CrossRef] [PubMed]
- Qian, K.; Mitsudo, H. Spatial dynamics of the eggs illusion: Visual field anisotropy and peripheral vision. Vision Res. 2020, 177, 12–19. [Google Scholar] [CrossRef] [PubMed]

| Comparison | Mean Difference | SE | df | t | p | ptukey | |
|---|---|---|---|---|---|---|---|
| Circle | - Emoticon | −0.864 | 1.03 | 48 | −0.841 | 0.405 | 0.835 |
| - Female face | −3.599 | 1.03 | 48 | −3.504 | 0.001 | 0.005 | |
| - Male face | −3.680 | 1.03 | 48 | −3.583 | <0.001 | 0.004 | |
| Emoticon | - Female face | −2.736 | 1.03 | 48 | −2.663 | 0.011 | 0.050 |
| - Male face | −2.817 | 1.03 | 48 | −2.742 | 0.009 | 0.041 | |
| Female face | - Male face | −0.081 | 1.03 | 48 | −0.080 | 0.937 | 1.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qian, K. Examining the Impact of Human Face Stimulus on Shape-Contrast Effects during a Brief Presentation. Brain Sci. 2022, 12, 914. https://doi.org/10.3390/brainsci12070914
Qian K. Examining the Impact of Human Face Stimulus on Shape-Contrast Effects during a Brief Presentation. Brain Sciences. 2022; 12(7):914. https://doi.org/10.3390/brainsci12070914
Chicago/Turabian StyleQian, Kun. 2022. "Examining the Impact of Human Face Stimulus on Shape-Contrast Effects during a Brief Presentation" Brain Sciences 12, no. 7: 914. https://doi.org/10.3390/brainsci12070914
APA StyleQian, K. (2022). Examining the Impact of Human Face Stimulus on Shape-Contrast Effects during a Brief Presentation. Brain Sciences, 12(7), 914. https://doi.org/10.3390/brainsci12070914






