Investigation of Corticomuscular Functional Coupling during Hand Movements Using Vine Copula
Abstract
:1. Introduction
2. Materials and Methods
2.1. Vine Copula
2.2. GARCH Model and Marginal Distribution
2.3. Correlation Measure Based on Copula Function
2.4. Modeling Step of Vine Copula
- Calculate the Kendall correlation coefficients between all variables, compare the sum of absolute values, and choose the largest spanning tree as the structure of the first layer tree;
- Select the optimal pair-copula function of the first layer tree structure by using the Akaike information criterion (AIC) and Bayesian information criterion (BIC), and calculate the conditional marginal distribution function;
- According to step (2), calculate the Kendall correlation coefficients between all conditional variables, and set the generating tree with the maximum sum of the absolute values of all correlation coefficients as the structure of the second layer tree;
- Select the second-level tree structure by using the AIC and BIC, and calculate the conditional marginal distribution function. Repeat steps (3) and (4) until only two nodes and one edge are left;
- The decomposition expression of the joint density function of random variables is expressed by the marginal density function and pair-copula function.
2.5. Granger Causality
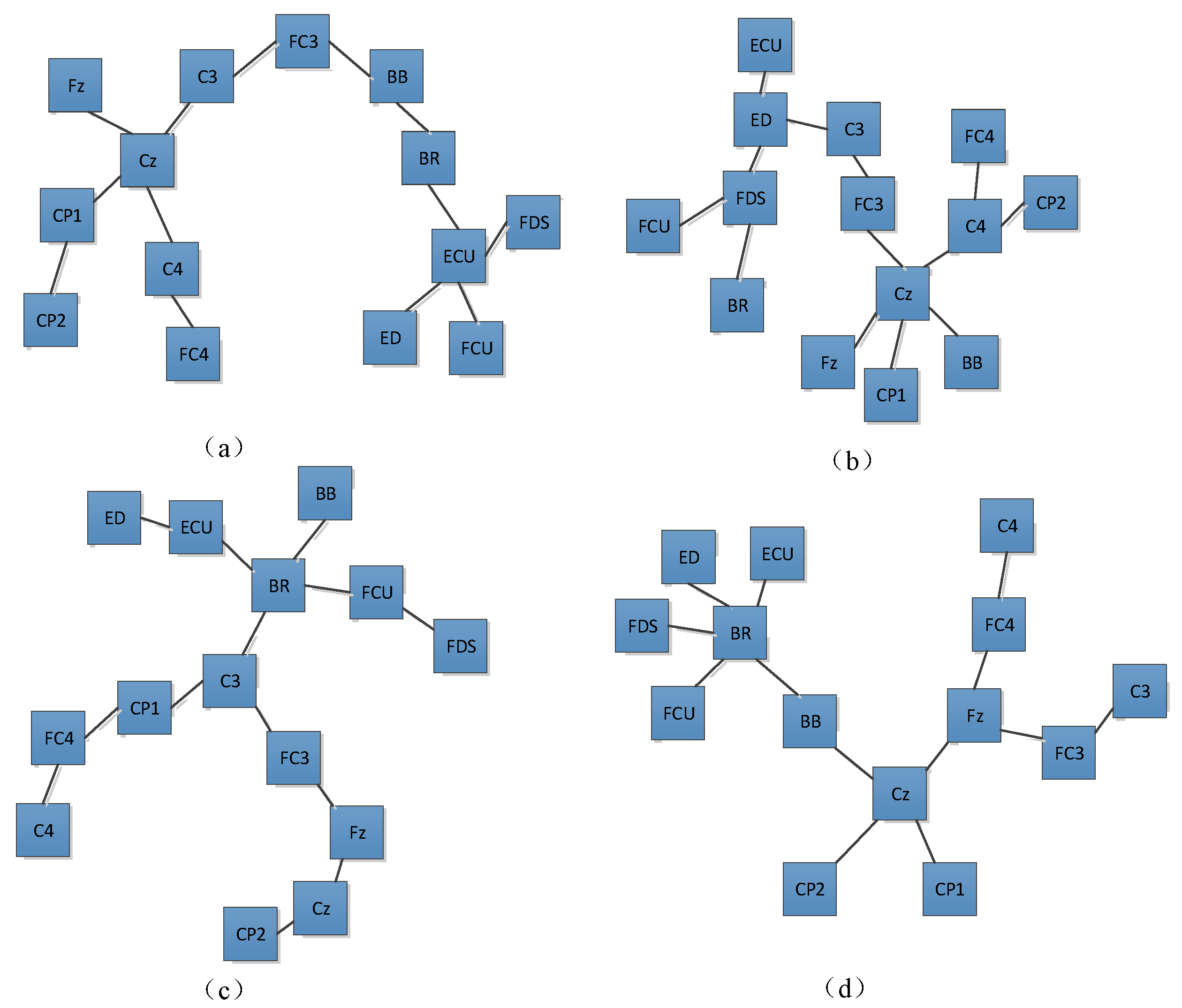
2.6. Computation of Network Characteristics
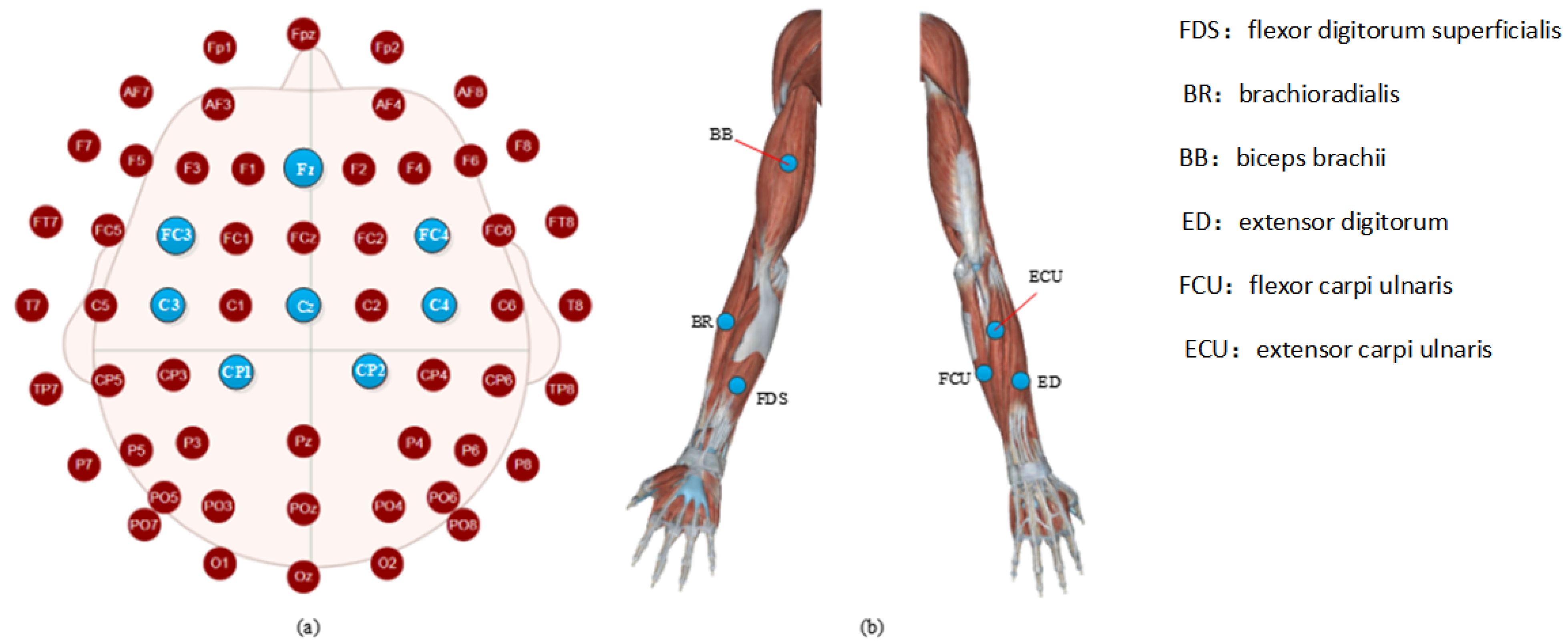
3. Experimental Procedure
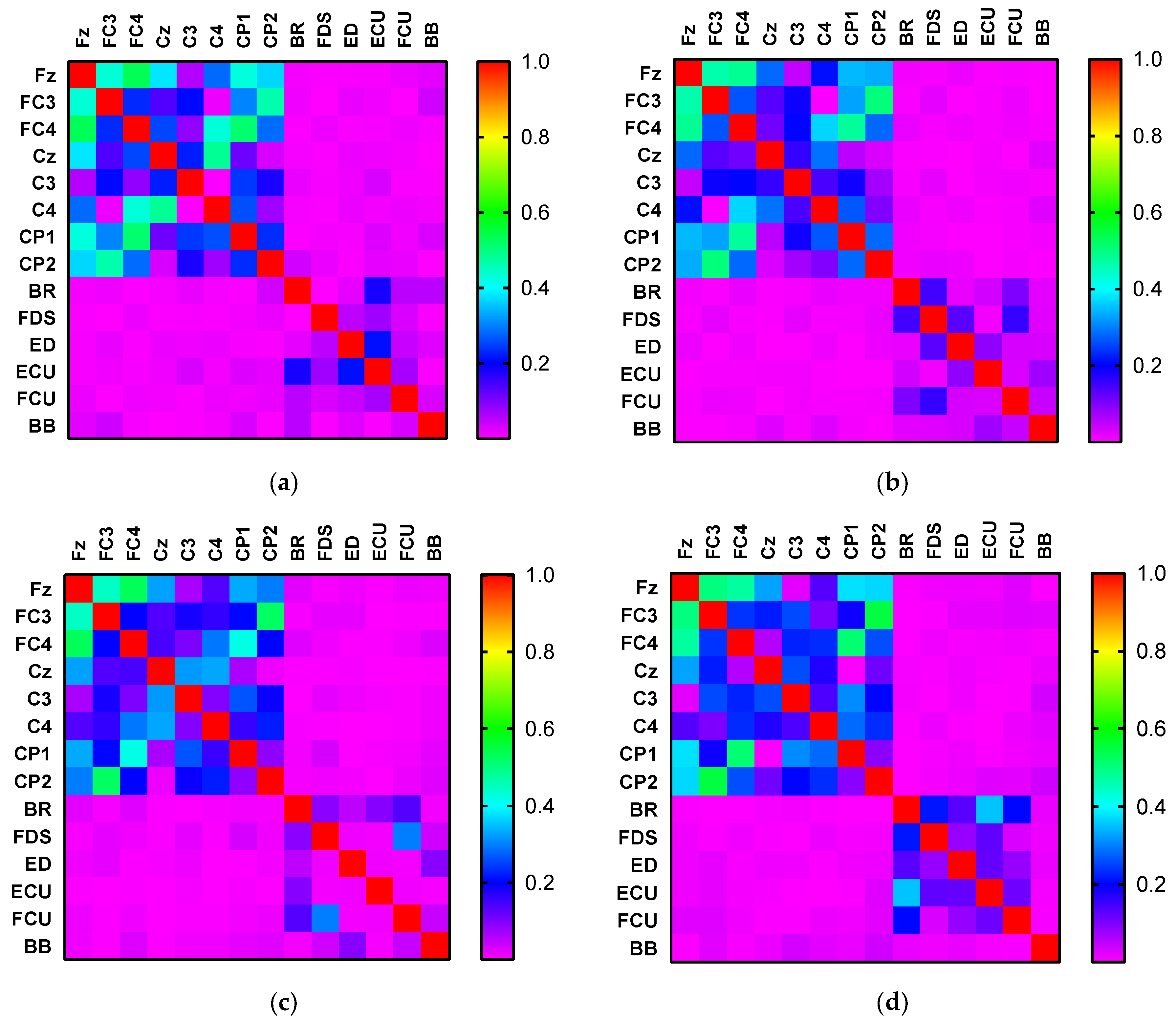
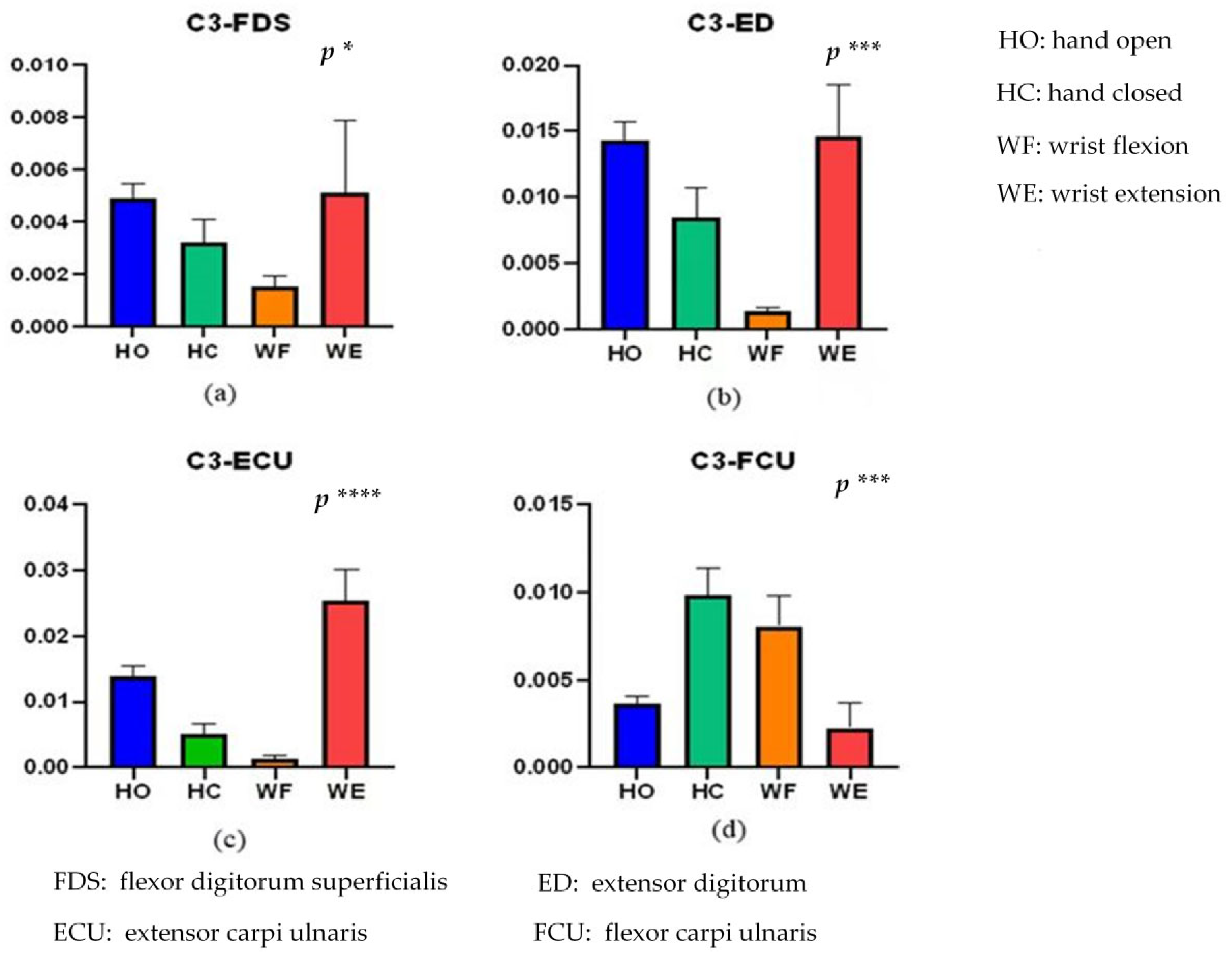
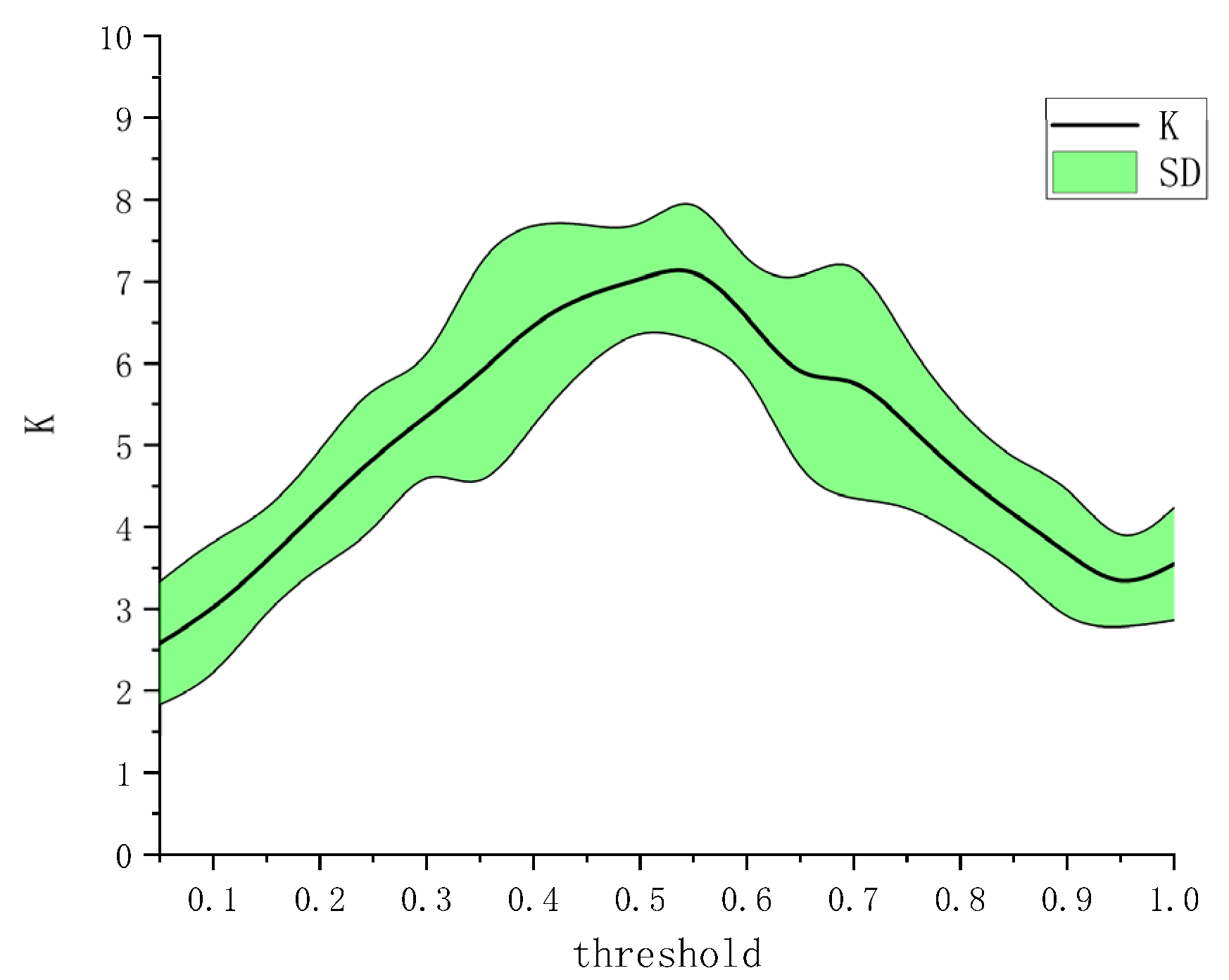
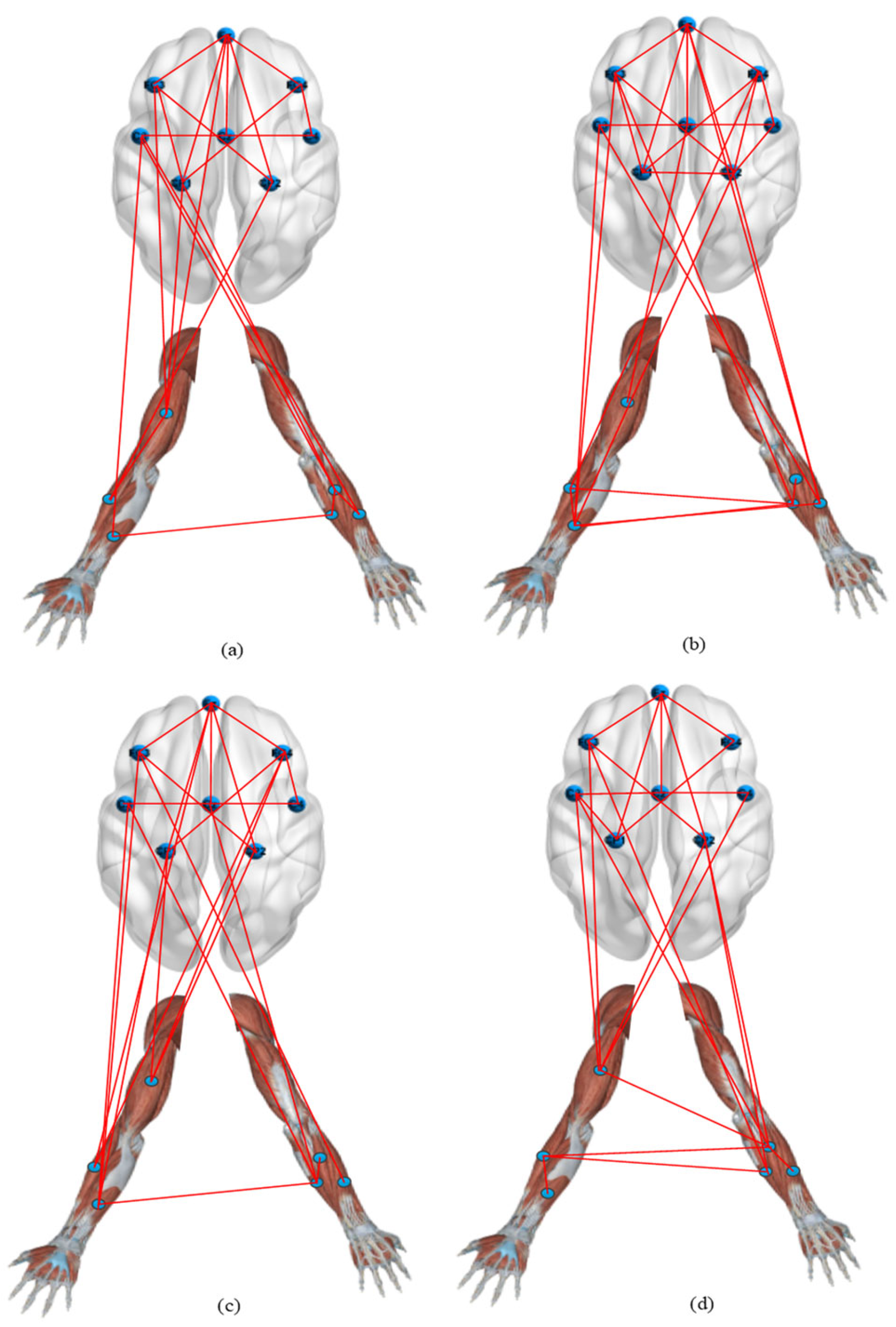
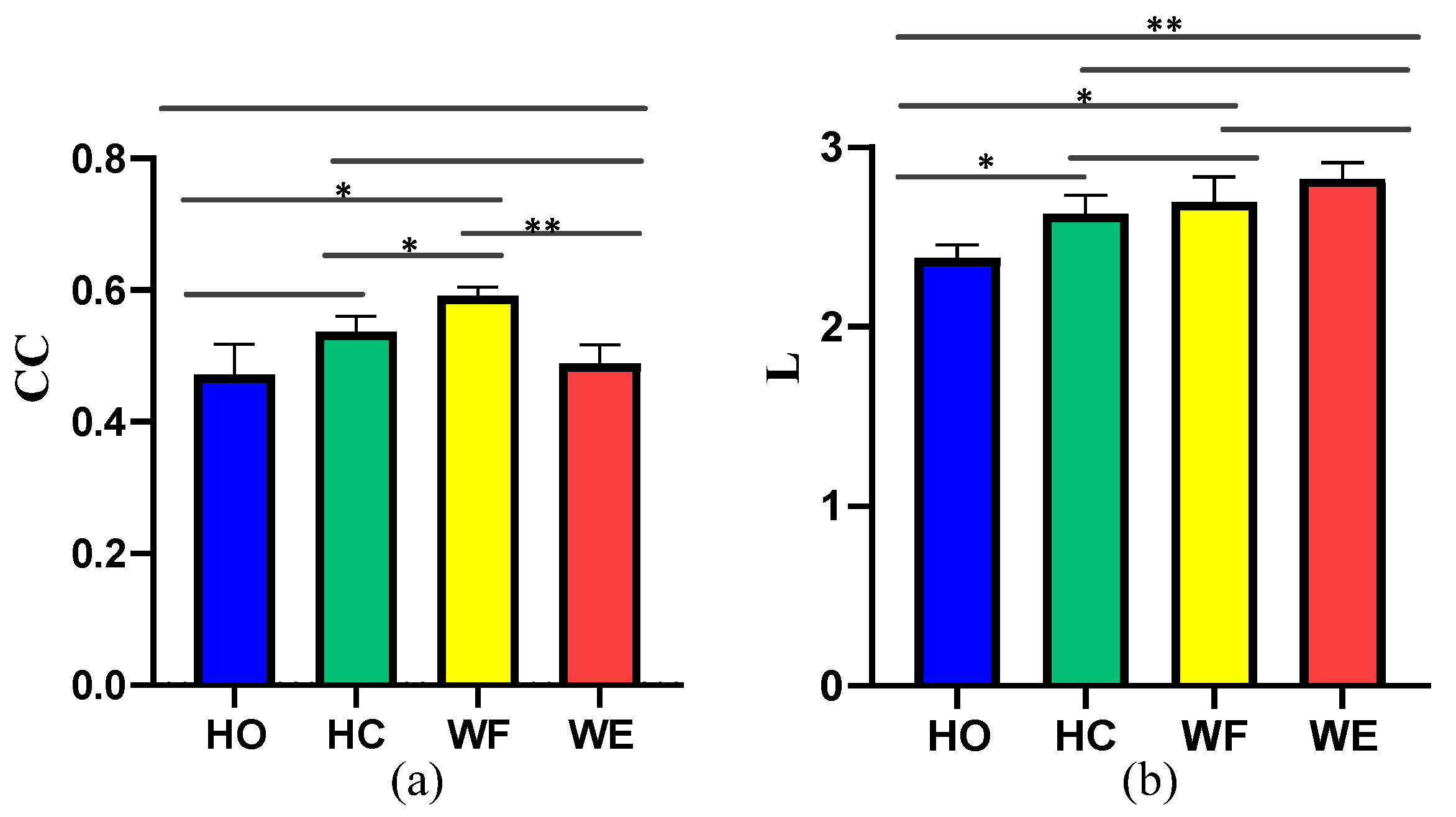
4. Results
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, Z.; Huang, Z.; He, W.; Su, C.-Y. Adaptive impedance control for an upper limb robotic exoskeleton using biological signals. IEEE Trans. Ind. Electron. 2016, 64, 1664–1674. [Google Scholar] [CrossRef]
- Namazi, H.; Ala, T.S. Decoding of simple and compound limb motor imagery movements by fractal analysis of Electroencephalogram (EEG) signal. Fractals 2019, 27, 1950041. [Google Scholar] [CrossRef]
- Qi, J.; Jiang, G.; Li, G.; Sun, Y.; Tao, B. Surface EMG hand gesture recognition system based on PCA and GRNN. Neural Comput. Appl. 2020, 32, 6343–6351. [Google Scholar] [CrossRef]
- van Wijk, B.C.; Beek, P.J.; Daffertshofer, A. Neural synchrony within the motor system: What have we learned so far? Front. Hum. Neurosci. 2012, 6, 252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Vliet, M.; Liljeström, M.; Aro, S.; Salmelin, R.; Kujala, J. Analysis of functional connectivity and oscillatory power using DICS: From raw MEG data to group-level statistics in Python. Front. Neurosci. 2018, 12, 586. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lebedev, M.A.; Nicolelis, M.A. Nicolelis, Brain-machine interfaces: From basic science to neuroprostheses and neurorehabilitation. Physiol. Rev. 2017, 97, 767–837. [Google Scholar] [PubMed]
- Zhang, J.; Wang, B.; Zhang, C.; Xiao, Y.; Wang, M.Y. An EEG/EMG/EOG-based multimodal human-machine interface to real-time control of a soft robot hand. Front. Neurorobot. 2019, 13, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Y.; Solis-Escalante, T.; Yao, J.; Van Der Helm, F.C.T.; Dewald, J.P.A.; Schouten, A.C.; Van Der Helm, F.C.T. Nonlinear Connectivity in the Human Stretch Reflex Assessed by Cross-Frequency Phase Coupling. Int. J. Neural Syst. 2016, 26, 1650043. [Google Scholar] [CrossRef]
- Larsen, L.H.; Zibrandtsen, I.C.; Wienecke, T.; Kjaer, T.W.; Christensen, M.S.; Nielsen, J.B.; Langberg, H. Corticomuscular coherence in the acute and subacute phase after stroke. Clin. Neurophysiol. 2017, 128, 2217–2226. [Google Scholar] [CrossRef] [Green Version]
- Dal Maso, F.; Longcamp, M.; Cremoux, S.; Amarantini, D. Effect of training status on beta-range corticomuscular coherence in agonist vs. antagonist muscles during isometric knee contractions. Exp. Brain Res. 2017, 235, 3023–3031. [Google Scholar] [CrossRef]
- Babiloni, C.; Vecchio, F.; Bares, M.; Brazdil, M.; Nestrasil, I.; Eusebi, F.; Rossini, P.M.; Rektor, I. Functional coupling between anterior prefrontal cortex (BA10) and hand muscle contraction during intentional and imitative motor acts. NeuroImage 2008, 39, 1314–1323. [Google Scholar] [CrossRef] [PubMed]
- Witham, C.L.; Riddle, C.N.; Baker, M.R.; Baker, S.N. Contributions of descending and ascending pathways to corticomuscular coherence in humans. J. Physiol. 2011, 589, 3789–3800. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lóopez-Larraz, E.; Birbaumer, N.; Ramos-Murguialday, A. Ramos-Murguialday, A hybrid EEG-EMG BMI improves the detection of movement intention in cortical stroke patients with complete hand paralysis. In Proceedings of the 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Honolulu, HI, USA, 18–21 July 2018; IEEE: Piscataway, NJ, USA; pp. 2000–2003. [Google Scholar]
- Antelis, J.M.; Montesano, L.; Ramos-Murguialday, A.; Birbaumer, N.; Minguez, J. Decoding upper limb movement attempt from EEG measurements of the contralesional motor cortex in chronic stroke patients. IEEE Trans. Biomed. Eng. 2016, 64, 99–111. [Google Scholar] [CrossRef] [PubMed]
- Edwards, L.; King, E.M.; Buetefisch, C.M.; Borich, M.R. Putting the “Sensory” Into Sensorimotor Control: The Role of Sensorimotor Integration in Goal-Directed Hand Movements After Stroke. Front. Integr. Neurosci. 2019, 13, 16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chowdhury, A.; Raza, H.; Dutta, A.; Prasad, G. EEG-EMG based hybrid brain computer interface for triggering hand exoskeleton for neuro-rehabilitation. In Proceedings of the Advances in Robotics, New Delhi, India, 28 June–2 July 2017; pp. 1–6. [Google Scholar]
- Youssofzadeh, V.; Zanotto, D.; Wong-Lin, K.; Agrawal, S.K.; Prasad, G. Directed functional connectivity in fronto-centroparietal circuit correlates with motor adaptation in gait training. IEEE Trans. Neural Syst. Rehabil. Eng. 2016, 24, 1265–1275. [Google Scholar] [CrossRef] [PubMed]
- Conway, B.A.; Halliday, D.M.; Shahani, U.; Maas, P.; Weir, A.I.; Rosenberg, J.R.; Farmer, S.F. Common frequency components identified from correlations between magnetic recordings of cortical activity and the electromyogram in man. J. Physiol. 1995, 483, P35. [Google Scholar]
- de Vries, I.E.J.; Daffertshofer, A.; Stegeman, D.F.; Boonstra, T.W. Functional connectivity in the neuromuscular system underlying bimanual coordination. J. Neurophysiol. 2016, 116, 2576–2585. [Google Scholar] [CrossRef] [Green Version]
- Hu, G.; Yang, W.; Chen, X.; Qi, W.; Li, X.; Du, Y.; Xie, P. Estimation of time-varying coherence amongst synergistic muscles during wrist movements. Front. Neurosci. 2018, 12, 537. [Google Scholar] [CrossRef]
- Fallani, F.D.V.; Pichiorri, F.; Morone, G.; Molinari, M.; Babiloni, F.; Cincotti, F.; Mattia, D. Multiscale topological properties of functional brain networks during motor imagery after stroke. Neuroimage 2013, 83, 438–449. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.; Sheng, Y.; Zeng, J.; Liu, H. Corticomuscular coherence for upper arm flexor and extensor muscles during isometric exercise and cyclically isokinetic movement. Front. Neurosci. 2019, 13, 522. [Google Scholar] [CrossRef]
- Sameshima, K.; Baccala, L.A. Methods in Brain Connectivity Inference Through Multivariate Time Series Analysis; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Marinazzo, D.; Liao, W.; Chen, H.; Stramaglia, S. Nonlinear connectivity by Granger causality. NeuroImage 2011, 58, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Shen, W.; Ting, C.-M.; Cramer, S.C.; Srinivasan, R.; Ombao, H. Estimating Brain Connectivity Using Copula Gaussian Graphical Models. In Proceedings of the International Symposium on Biomedical Imaging, Venice, Italy, 8–11 April 2019; pp. 108–112. [Google Scholar]
- Sklar, M. Fonctions de repartition an dimensions et leurs marges. Publ. Inst. Statist. Univ. Paris 1959, 8, 229–231. [Google Scholar]
- Dauwels, J.; Yu, H.; Wang, X.; Vialatte, F.; Latchoumane, C.-F.V.; Jeong, J.; Cichocki, A. Inferring brain networks through graphical models with hidden variables. In Machine Learning and Interpretation in Neuroimaging; Springer: Berlin/Heidelberg, Germany, 2012; pp. 194–201. [Google Scholar]
- Aas, K.; Czado, C.; Frigessi, A.; Bakken, H. Pair-copula constructions of multiple dependence. Insur. Math. Econ. 2009, 44, 182–198. [Google Scholar] [CrossRef] [Green Version]
- Bedford, T.; Daneshkhah, A.; Wilson, K.J. Approximate uncertainty modeling in risk analysis with vine copulas. Risk Anal. 2016, 36, 792–815. [Google Scholar] [CrossRef] [Green Version]
- Schepsmeier, U. A goodness-of-fit test for regular vine copula models. Econom. Rev. 2019, 38, 25–46. [Google Scholar] [CrossRef] [Green Version]
- Krithikaivasan, B.; Zeng, Y.; Deka, K.; Medhi, D. ARCH-based traffic forecasting and dynamic bandwidth provisioning for periodically measured nonstationary traffic. IEEE/ACM Trans. Netw. 2007, 15, 683–696. [Google Scholar] [CrossRef]
- Kim, S. Forecasting internet traffic by using seasonal GARCH models. J. Commun. Netw. 2011, 13, 621–624. [Google Scholar] [CrossRef]
- Cormen, T.H.; Leiserson, C.E.; Rivest, R.L. Introduction to Algorithms (3); MIT Press: Cambridge, MA, USA, 2009. [Google Scholar]
- Xi, X. Construction and analysis of cortical–muscular functional network based on EEG-EMG coherence using wavelet coherence. Neurocomputing 2021, 438, 248–258. [Google Scholar] [CrossRef]
- Muthuraman, M.; Galka, A.; Deuschl, G.; Heute, U.; Raethjen, J. Dynamical correlation of non-stationary signals in time domain—A comparative study. Biomed. Signal Process. Control 2010, 5, 205–213. [Google Scholar] [CrossRef]
- Zhao, J.; Zhou, W.; Liu, K.; Cai, D. Application of SVM and Wavelet Analysis in EEG Classif ication. J. Biomed. Eng. 2011, 28, 277–279. [Google Scholar]
- Clemens, B.; Puskás, S.; Besenyei, M.; Spisák, T.; Opposits, G.; Hollódy, K.; Fogarasi, A.; Fekete, I.; Emri, M. Neurophysiology of juvenile myoclonic epilepsy: EEG-based network and graph analysis of the interictal and immediate preictal states. Epilepsy Res. 2013, 106, 357–369. [Google Scholar] [CrossRef] [PubMed]
- Xi, X. Emotion-movement relationship: A study using functional brain network and cortico-muscular coupling. J. Neurosci. Methods 2021, 362, 109320. [Google Scholar] [CrossRef] [PubMed]
- Bezruchko, B.P.; Ponomarenko, V.I.; Prokhorov, M.D.; Smirnov, D.A.; Tass, P.A. Modeling nonlinear oscillatory systems and diagnostics of coupling between them using chaotic time series analysis: Applications in neurophysiology. Phys. Uspekhi 2008, 51, 304–310. [Google Scholar] [CrossRef]
- Granger, C.W.J. Testing For Causality: A Personal Viewpoint. J. Econ. Dyn. Control 1980, 2, 329–352. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ye, F.; Ding, J.; Chen, K.; Xi, X. Investigation of Corticomuscular Functional Coupling during Hand Movements Using Vine Copula. Brain Sci. 2022, 12, 754. https://doi.org/10.3390/brainsci12060754
Ye F, Ding J, Chen K, Xi X. Investigation of Corticomuscular Functional Coupling during Hand Movements Using Vine Copula. Brain Sciences. 2022; 12(6):754. https://doi.org/10.3390/brainsci12060754
Chicago/Turabian StyleYe, Fei, JinSuo Ding, Kai Chen, and Xugang Xi. 2022. "Investigation of Corticomuscular Functional Coupling during Hand Movements Using Vine Copula" Brain Sciences 12, no. 6: 754. https://doi.org/10.3390/brainsci12060754
APA StyleYe, F., Ding, J., Chen, K., & Xi, X. (2022). Investigation of Corticomuscular Functional Coupling during Hand Movements Using Vine Copula. Brain Sciences, 12(6), 754. https://doi.org/10.3390/brainsci12060754





